Abstract
Mycotoxicosis are a common problem in livestock, where a group of six major mycotoxins represents a high risk for animal health and production profits. Mycotoxin binders (MTB) can reduce the mycotoxin burden in the gastrointestinal tract of the animal. Mycotoxin binders are classified in inorganic, as clays and activated carbon (AC), and organic, as yeast cell wall (YCW) and micro-ionized fibers. The adsorption of mycotoxins into MTB is due to: 1) chemical interactions where the cation exchange capacity involves different types of bounds like ion-dipole, Van der Walls forces, or hydrogen bonds; and 2) to physical characteristics of MTB like pore size, or mycotoxin structure and shape. The adsorption capacity of MTB is determined using different in vitro tests that mimic the gastrointestinal tract of the animals. A literature search was conducted to identify in vitro research where the efficacy of adsorption of MTB was determined. The search was based on 8 MTB [AC, bentonite, clinoptilolite, hydrated sodium calcium aluminosilicate (HSCAS), montmorillonite (MMT), sepiolite, YCW and zeolite] and 6 mycotoxins [aflatoxin (AF), deoxynivalenol (DON), fumonisin (FUM), ochratoxin (OTA), T-2 toxin and zearalenone (ZEA)]. Sixty-eight papers with 1842 data were selected and analyzed with the PROC MIXED of SAS. The response variable was the percentage mycotoxins adsorption by MTB, and the model included the fixed effects of MTB, mycotoxins, incubation media, pH and their interactions, and the random effect of the study. Differences were considered significant when P < 0.05 and with tendency when 0.05 < P < 0.10. The mycotoxins adsorption capacity was 83% ± 1.0 for AC, 76% ± 3.1 for MMT, 62% ± 1.0 for bentonite, 55% ± 1.9 for HSCAS, 52% ± 9.1 for sepiolite, 52% ± 4.3 for clinoptilolite and 44% ± 0.4 for YCW. For mycotoxins, the adsorption of AF was 76% ± 0.6, for FUM was 50% ± 1.8, for OTA was 42% ± 1.0, for ZEA was 48% ± 1.1, for DON was 35% ± 1.6, and for T-2 was 27% ± 2.8. The pH affected the adsorption capacity of YCW with higher adsorption at low pH, and the adsorption of OTA and ZEA, where OTA adsorption tended to be lower at intermediate pH, and adsorption of ZEA tended to be higher at the two-steps pH. The potential adsorption of some essential nutrients, including amino acids and vitamins, should also be considered. Results should be used as a guide in the selection of the appropriate mycotoxin binder based on the predominant mycotoxin in feeds.
Keywords: efficacy, interaction, mycotoxin, mycotoxin binder, review
Mycotoxin binders are an effective strategy to sequester mycotoxins ingested by animals. This review summarizes the classification, mechanism of action of adsorption, and possible interactions with other nutrients due to their unselective mechanism of action. Published in vitro data on the capacity of mycotoxin binders to adsorbed mycotoxins indicate a large variation in their efficacy among mycotoxins and mycotoxin binders.
Introduction
Mycotoxicoses are the consequence of consuming feeds contaminated with mycotoxins, causing acute, chronic, or subclinical effects. More than 400 mycotoxins have been identified and six are classified as highly toxic and frequent in animal feeds: aflatoxin (AF), ochratoxin A (OTA), fumonisin (FUM), deoxynivalenol (DON), zearalenone (ZEA) and T-2 toxin (T-2) (Krska et al., 2016; Arroyo-Manzanares et al., 2019). Three major species of molds are responsible for the production of these mycotoxins: Fusarium, Aspergillus, and Penicillium (Yiannikouris and Jouany, 2002), each one producing different types of mycotoxins. A recent report indicated that more than 88% of feed samples analyzed worldwide in 2019 contained more than one mycotoxin (Gruber-Dorninger et al., 2019). Mold growth is dependent on climate conditions where temperature, humidity, and drought influence the type of mold and, as a consequence, the type of mycotoxins produced (Schatzmayr and Streit, 2013; Moretti et al., 2019). For example, Gruber-Dorninger et al. (2019) reported that the first contaminant of corn samples was FUM (80%), followed by DON (67%) and ZEA (44%). Mycotoxicosis may reduce feed intake, nutrient digestion, and absorption and metabolism, resulting in a negative impact on milk yield, average daily gain, growth efficiency, and(or) fertility, among others (Jouany et al., 2009; Bryden, 2012; Papatsiros et al., 2021; Xu et al., 2022).
Mycotoxin binders (MTB) are an effective strategy to sequester mycotoxins into their matrix and avoid their absorption in the gastrointestinal (GI) tract of animals (Di Gregorio et al., 2014; Čolović et al., 2019). Mycotoxin binders are classified as Generally Recognized as Safe by the U.S. Food and Drug Administration (code of federal regulations, 21 CFR 582.2729). This regulation recognizes that MTB produce no harm to physiological functions of animals. The mycotoxin-MTB complex passes through the GI tract of animals and is eliminated in feces (Gimeno and Martins, 2007). The European Food Safety Authority (EFSA, 2011) requires that the efficacy of MTB must be tested by one in vitro and two in vivo tests where mycotoxins are supplemented at the minimal mycotoxin toxic level for each species before being authorized (European Commission EC, 2006). These tests must prove the capacity of the MTB to adsorb mycotoxins through a wide pH range to guarantee the stability of the mycotoxin-MTB complex throughout the GI tract. These tests should also prove high affinity and rapidity to adsorb mycotoxins at a low MTB inclusion rate in diets (1–2 kg/t) to allow a high degree of adsorption before the mycotoxin is absorbed into the bloodstream. These properties depend on the physical and chemical properties of MTB and mycotoxins as pH, polarity, pores dimension, and shape. Furthermore, EFSA (2010) required also that MTB do not adsorb essential nutrients like AA, vitamins, and minerals as was demonstrated in several studies (Barrientos-Velázquez et al., 2016; Kihal et al., 2020).
The objectives of this paper are to review the main characteristics, properties, and mechanisms of action of MTB, to evaluate the efficacy of MTB to adsorb mycotoxins and to determine to what extent they can absorb some essential nutrients.
Classification of Mycotoxin Binders
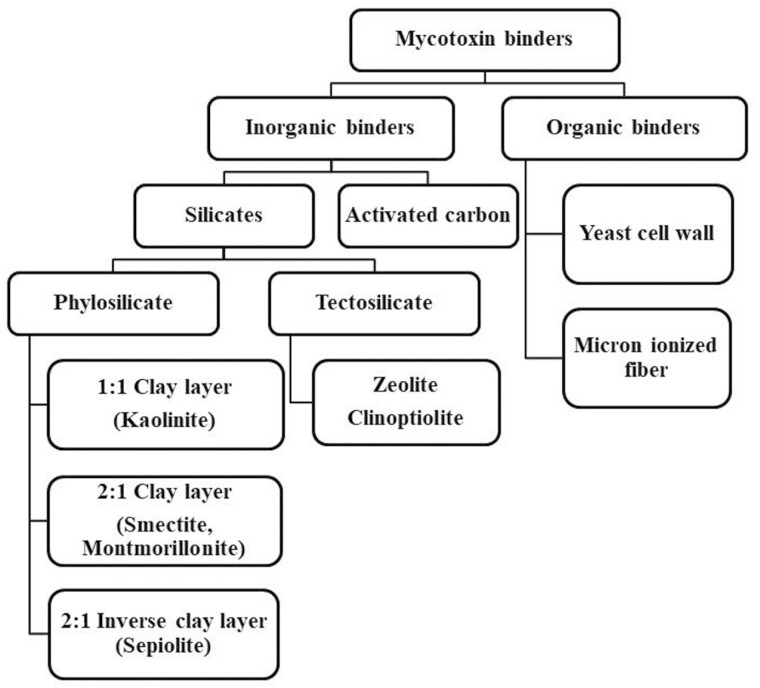
Mycotoxin binders are classified by their nature into two major groups: 1) inorganic binders constituted by silicate minerals and activated carbon (AC) binders, and 2) organic binders constituted by yeast cell wall (YCW) or micro-ionized fiber extracted from different plant materials (Figure 1).
Figure 1.
A diagram representing the classification of different mycotoxin binders by their source, nature and structural composition.
Inorganic binders
There is no consensus on the classification of clay binders that is acceptable to different disciplines such as agriculture, environment, or construction applications (Bergaya and Lagaly, 2013). Therefore, we report a classification of inorganic binders based on their properties to bind mycotoxins as proposed by Grim (1962) and updated by Murray (2007).
Silicate binders
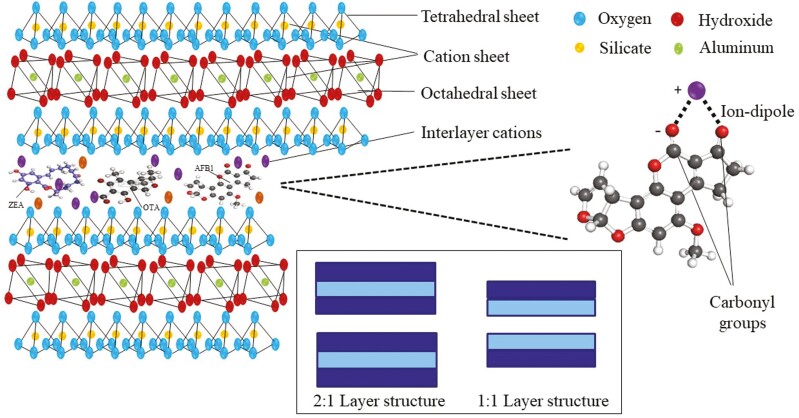
Silicates are the most abundant elements found on earth crust (Kandel, 2018). Silicate is a mineral combining silicon dioxide (SiO24−) with a tetrahedral structure, where the silicon ion is in the center and surrounded by four oxygen atoms. The interaction of the positive silicon charges and negative oxygen charges results in an unbalanced structure. This allows the free oxygen charges to be bound to other silicon ions forming a chain of tetrahedral structures in different combinations, resulting in chains, sheets, rings, and three-dimensional structures. The tetrahedral sheet is the basis of silicate binders where different subgroups of silicate are formed in combination with other mineral ions in bi or three-dimensional structures. The two main subclasses of silicates are phyllosilicate (sheets of silicate) or tectosilicate (framework silicate, Figure 2).
Figure 2.
Molecular structure of octahedral and tetrahedral sheets of tectosilicate binders, and an illustration of the contribution of ions to the adsorption mechanism of mycotoxins.
Phyllosilicate binders
Phyllosilicates are bidimensional laminar or tubular structures characterized by the interaction of the oxygen ions of the tetrahedral silicate sheet with the hydroxyl ions of a second sheet formed by aluminum or magnesium ions located in the center of 6 hydroxyl ions (Al/MgOH)6 to give an octahedral sheet (Figure 2, Di Gregorio et al, 2014). The six coordinating hydroxy ions have a potential of six negative charges. To compensate this charge difference, two Al3+ ions or three Mg2+ ions are added to the structure. Then, the octahedral sheets are named trioctahedral or dioctahedral sheets, respectively (Schoonheydt and Johnston, 2011). The structure of phyllosilicate minerals is formed by piling up tetrahedral and octahedral sheets in different combinations (Murray, 2007). There are two main combinations: 1) A 1:1 tetrahedral and octahedral sheets, represented by the clay group kaolinite-serpentite, and 2) A 2:1 tetrahedral to octahedral sheet in the middle (sandwich structure) like smectite (Figure 2). In some cases, silicate Si4+, and Al3+ ions on the tetrahedral and octahedral sheet can be substituted by Mg2+, Fe2+, or Li+ ions. These substitutions lead to negatively charged layers that need to be balanced with exchangeable cations (Na+, K+, Ca+2, …). These cations can be interchangeable and provide the clay with the swelling and ions exchange capacity responsible for binding mycotoxins.
Many clays are effective in binding mycotoxins, including smectite, montmorillonite (MMT), bentonite, and sepiolite. The smectite is one of the largest classes of phyllosilicate groups that contains MMT known for its high adsorption capacity. Bentonite is another clay composed of 80% MMT and reproduces most of its properties (Grim and Güven, 1978). Sodium MMT is referred to as sodium bentonite and calcium MMT is referred to as calcium bentonite. These different types of MMT come from exchangeable cations that substitute the Al3+ or Mg2+ in the octahedral sheet giving the molecule high cations exchange capacity, electrical conductivity, and water absorption capacity. For instance, sodium MMT has an exchange capacity between 80 and 130 meq/100 g and a surface area between 150 and 200 m2/g. The interlayer space varies with the exchangeable cation and the degree of inter-laminar hydration. A complete dehydration generates a small space between sheets (0.95–1.0 nm). In contact with water, the clay swallows, and expands the interlayer space to tens of nanometers, allowing to increase the adsorption capacity of bentonites (Sánchez et al., 2012). In contrast, calcium MMT has a lower exchange capacity (between 40 and 70 meq/100 g) and a smaller interlayer space (Murray, 2007), which would result in lower adsorption capacity.
Tectosilicate binders
The tectosilicates group is a crystalline aluminosilicate mineral with zeolite as the main constituent. It is formed by assembling multiple tetrahedral structures by the union of the apical oxygen atom in a three-dimensional way. This arrangement generates different pores of the same dimension occupied by exchangeable cations and water molecules, giving a ring, or a cage-like structure. The pores formed by the tridimensional structure are the basis of the adsorbing capacity where the in-site potassium and calcium cations interact with mycotoxins that can get inside the pores depending on their size (Nadziakiewicza et al., 2019; Samantray et al., 2022). There are nearly 50 different types of zeolites with different physical and chemical properties. The classification of the different types of zeolites is based on the crystal structure and chemical composition, cations, pore size, and strength of the structure. Clinoptilolite is the most commonly used zeolite type due to its strength properties and high resistance at low pH and high temperatures. Clinoptilolite is known as a molecular sieve for its pores that represent 50% of the molecular structure with an approximate size of 3–8 Angstrom (Å). The application of heat treatment or enrichment with different cations (K+, Na+, or Ca2+) may increase its porosity and adsorption capacity (Eseceli et al., 2017).
Activated carbon
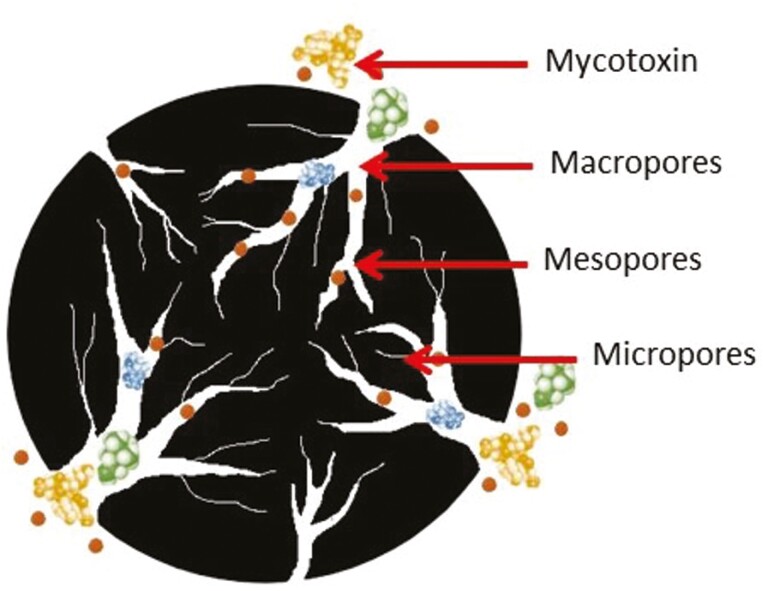
Activated carbon is a non-soluble powder formed by the carbonization of almost any organic compound that contains carbon (wood, bamboo or coal) by a pyrolysis heating process at temperatures up to 2000 °C (Galvano et al., 1996). The resulting powder requires an activation process necessary to acquire a higher adsorption capacity. Chemical and physical processes allow the development of a large number of highly porous structures (Figure 3). The chemical treatment consists on the impregnation of AC with different chemicals such as potassium hydroxide, phosphoric acid, or zinc chloride followed by a temperature exposure of 250–600 °C. The chemical treatment results in impure and ineffective AC with a low number of pores and produces chemical residues harmful to the environment (Danish and Ahmed, 2018). The physical treatment consists on an oxidation process where carbon passes through a heating chamber at 600–900 °C with an oxygen or carbon dioxide thrust with proper conditions of pressure, temperature, and time (Ma et al., 2021). The treatment results in a highly microporous carbon that increases the surface area of the AC (500–3000 m2/g, Ramos and Hernández, 1996; Galvano et al., 2001). Therefore, the efficiency of AC is related to the number of microporous available for adsorption of mycotoxins. Galvano et al. (1997) compared the efficacy of different AC sources (olive residues, peach stone, and almond shells) to adsorb FUM in vitro. Results showed that the adsorption capacity ranged from 100% in olive residues to 35% in peach stone, and was correlated to the availability of pores among AC sources.
Figure 3.
Structure of macro and micropores of activated carbon for the adsoprtion of mycotoxins and other nutrients.
Organic binders
Yeast cell wall
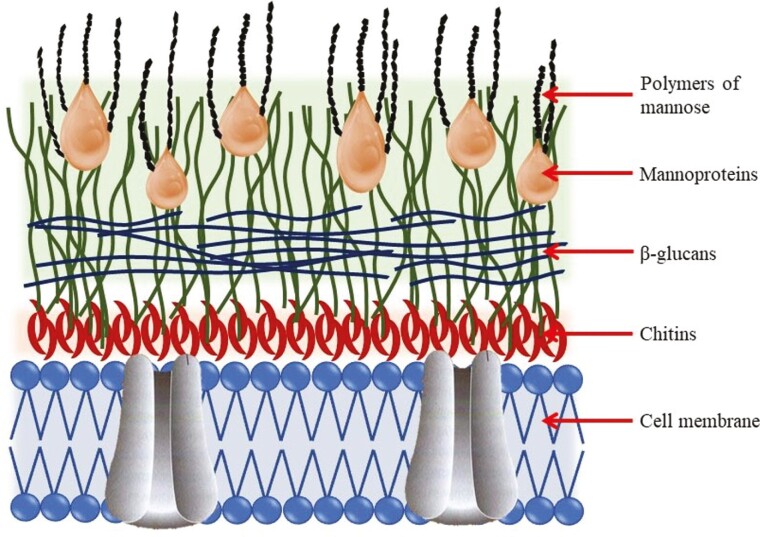
The YCW fraction of yeast represents 15–30% of the dry weight of yeast cells and is considered responsible for mycotoxin adsorption. Cell walls are organized in two sheets: the inner sheet provides stiffness and determines the morphology of the yeast and it is composed of β-(1,3)-d-glucans helix chains organized in a complex 3D structure, and β-(1,6)-d-glucans linear side chains, representing 50–60% of the dry weight of cell walls. The β-d-glucans are firmly attached to the cytoplasm membrane by chitins that provide the cell wall with their insolubility and plasticity. When the chitin proportion is higher in the cell wall it may decrease its flexibility and reduce the affinity to bind mycotoxins (Jouany et al., 2005). The outer layer of the cell wall is constituted by glucomannans and mannoproteins (40%, Figure 4, Kogan and Kocher, 2007) that determine the superficial properties of the cell wall. The adsorption capacity of yeast increases as the proportion of β-d-glucans present in the yeast strain increases (Yiannikouris et al., 2004).
Figure 4.
The composition of different yeast cell wall sheets and their components (adapted from Talavera et al., 2013).
Micro-ionized fiber
Micro-ionized fibers have emerged as a new MTB that has the ability to bind different mycotoxins. Many biomaterials have been identified with a mycotoxin binding potential in several in vitro and in vivo studies as grape pomace, grape stem, olive pomace, alfalfa hay, and wheat straw, with a binding capacity ranging from 27 to 90% depending on the binder and the mycotoxin (Avantaggiato et al., 2014; Čolović et al., 2019; Fernandes et al., 2019). The adsorption mechanism of micro-ionized fibers is similar to that of silicates or AC binders, where physico-chemical interactions with lignin, cellulose, and polyphenol groups with mycotoxins are involved (Greco et al., 2018; Nava-Ramírez et al., 2021). The main limitation of micro-ionized fibers to be used as MTB is that they should be fed at a high inclusion rate (20 kg/t) to be effective in vivo, which may not be adequate in monogastric animals (Čolović et al., 2019). In contrast, the higher fiber content in ruminant diets may help reduce the toxicity of mycotoxins in these animals.
Adsorption Mechanism of Different Binders
Mycotoxin binder properties
The adsorption mechanism of silicates is directly related to the physico-chemical properties of the binder and indirectly to mycotoxin properties. Cation exchange capacity and total net charges of the surface determine the capacity of silicate binders to adsorb mycotoxins. By definition, the cation exchange capacity is the capacity of the binder to exchange cations present on the surface with other molecules like mycotoxins. However, this exchange capacity is highly dependent on the pH of the binder, which varies among mine sources. In fact, each binder has its own pH, named pH at point zero charges, where the surface of the binder has equal positive and negative charges. For instance, MMT extracted from a Greece mine had a pH of 9.4 while another extracted from a Bosnia mine had a pH of 7.7 (Ismadji et al., 2015). If the pH of the medium is lower than the pH of the binder, hydrogen ions are bound to the binder that loses its charge. This situation is similar to the gastric environment where the low pH reduces the ionization capacity of cations and the adsorption of those MTB will be lower (De Mil et al., 2015). However, if the pH of the medium is higher than the pH of the binder, the binder will release hydrogen ions andexpose negative charges, increasing the capacity to attract cations (Na+, Ca2+, K+, Al2+) within the interlayer space of sheets and in the edges of the clay responsible for the interaction with the carbonyl oxygen group of many mycotoxins. The adsorption capacity increases as the cation exchange capacity of the binder increases (Diaz et al., 2004; Ismadji et al., 2015). The interaction of cations (positive charge) and the carbonyl group of mycotoxins (negative charge) is due to weak ion-dipole or Van der Walls interactions. When water is present in the interlayer space of the MTB, hydrogen molecules from H2O interact with oxygen molecules of the carbonyl groups of mycotoxins and make a complex hydrogen-carbonyl oxygen-cation bonds.
In addition to the external cation binding, the capacity of adsorption is also related to the interlayer space of MTB, which is dependent on the size of the interlayer space and the size of mycotoxins. The interlayer space is a determining factor and is highly correlated to the adsorption capacity of MTB (De Mil et al., 2015). The interlayer space varies among MTB. Mortland and Lawless (1983) reported that differences in the interlayer space between sodium and calcium bentonites affect the adsorption capacity of AF and suggested that the higher interlayer space of sodium bentonite allowed higher adsorption compared with calcium bentonite. For instance, zeolite has a lower adsorption capacity of AF because its interlayer space (4–7 Å) is much smaller than the size of AF (10–12 Å). In contrast, bentonite has a higher adsorption capacity of AF due to its larger interlayer space (15–20 Å) that allows the AF to get inside the interlayer space (Vekiru et al., 2014).
New silicate binders can be developed by the structural modification of the original silicates. Chemical processes allow the addition of organic molecules in the sheets of silicate and mineral binders to increase the positive charge of layers. Jaynes and Zartman (2011) reported that MMT treated with choline and carnitine increased by four the adsorption of AF compared with the untreated clays and suggested that lysine, methionine, or phenylalanine treatment may also increase the adsorption capacity of clays. The modification of zeolite mineral with octadecyl-dimethyl benzyl ammonium can also increase the surface hydrophobicity and enhanced the adsorption capacity of ZEA (Dacović et al., 2005). Tomasevic et al. (2003) also reported that the adsorption capacity of zeolite treated with organic compounds increased the adsorption capacity of ZEA from 5 to 94%.
The adsorption mechanism of AC depends on different factors as pore size and surface area (Goto et al., 2015). Differently to clay minerals, AC is not a polar molecule, and provides AC the capacity to bind non-polar mycotoxins rather than polar mycotoxins (Bueno et al., 2004). The binding mechanism of AC is by hydrophobic interactions and pi-bonds. The activation process of AC increases the surface oxygen complexes in the surface of AC like carboxyl or phenol groups, which increases the polarity and the hydrophilic properties of AC. Therefore, the AC acquires the capacity to also adsorb polar compounds such as AF and FUM (Moreno-Castilla et al., 2003). The pore size and their distribution within the AC are also important to determine the efficacy of adsorption. Pore dimensions are categorized in three types: micropores (<2 nm), mesopores (2–50 nm) and macropores (>50 nm). Therefore, the diffusion of mycotoxins into the AC can be slowed if the pore size is inadequate to the size of mycotoxins that limit the accessibility to the inner surface of the AC.
The adsorption mechanism of YCW is mainly related to the interaction of β-(1,3)-d-glucans with mycotoxins. The two bonds involved in this interaction include the Van der Walls bonds between the aromatic cycle of mycotoxins and β-d-glucopyranose ring of the YCW, and the hydrogen bonds between hydroxyl, ketone, and lactone groups of mycotoxins and the hydroxyl group of glucose units of β-d-glucans in YCW (Jouany et al., 2005). The geometrical structure also plays an important role in the binding mechanism of YCW, where the match between the three-dimensional structure of the mycotoxin and the β-d-glucans helix improves the strength of the complex (Yiannikouris et al., 2004).
Mycotoxin properties
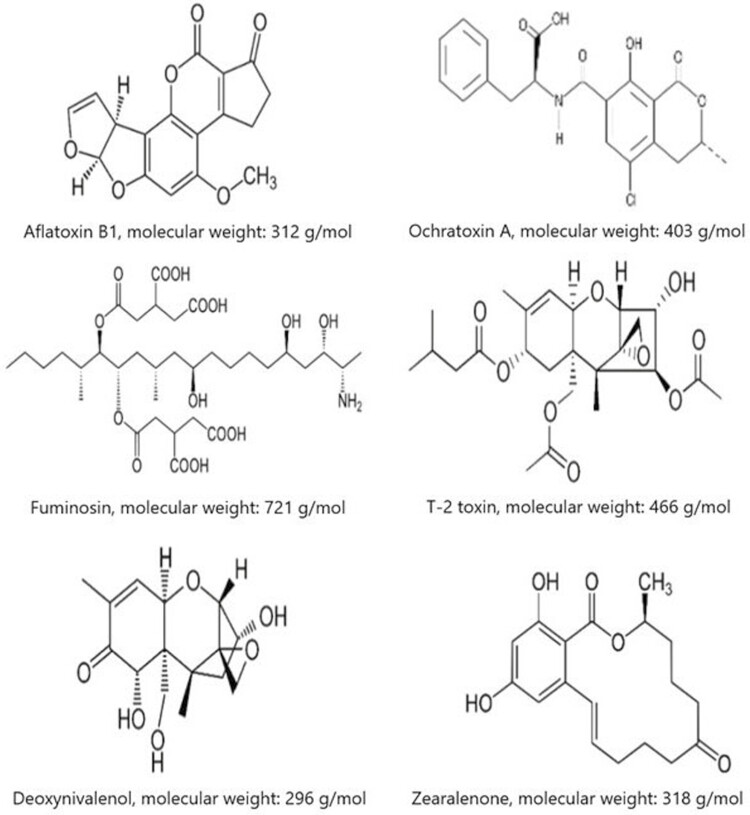
Physico-chemical characteristics of mycotoxins also affect the adsorption capacity of MTB (Galvano et al., 1997). Mycotoxins can be classified by their polarity, solubility, and chemical structure (Figure 5). The polarity of mycotoxins reflects the charge arrangement within the molecule that can be classified as polar or non-polar molecules. For example, AF and FUM are the highest polar mycotoxins, ZEA is non-polar, and DON, T-2 and OTA have an intermediate polarity. Solubility of mycotoxins in the medium is important for their adsorption. Most mycotoxins are soluble in different organic solvents such as methanol, acetonitrile, or acetone. However, their solubility in water depends on their polarity, being the more polar mycotoxins the more soluble. The chemical structure, size, and shape of the molecule are another important characteristic of mycotoxins that affect their adsorption. For instance, AF is a flat shape and small molecule that can easily get into the interlayer space of binders and be adsorbed. In contrast, the large branched structure of FUM makes the entrance of the mycotoxin into the interlayer space of MTB difficult, which reduces its adsorption (Galvano et al., 1996).
Figure 5.
Chemical structure of the major mycotoxins and their molecular weight.
Methods to Determine the Adsorption Capacity of Mycotoxin Binders
In vitro tests are commonly used as a screening method to determine the capacity of MTB to adsorb mycotoxins. However, there are several in vitro methods described in the literature (Galvano et al., 1996; Lemke et al., 2001; Gallo and Masoero, 2010).
Single in vitro test
The single concentration test consists on the use of in vitro models that mimic the GI tract to test the interaction between MTB and mycotoxins in an artificial incubation medium. The method consists on a single concentration of mycotoxin incubated with a defined concentration of MTB. Results of this test are expressed as percentage of mycotoxin adsorption. The simplest model consists of using distilled water as an incubation medium where substrates are incubated in one-step at pH = 7 and ambient temperature for 24 h (Lemke et al., 2001) or incubated for 2 h at 39 °C (Gallo and Maseoro, 2010). To better simulate the differences of pH in the GI tract, a two-steps method was proposed by Dawson et al. (2001). In this model, substrates are incubated first in a citrate buffer at pH 3.0 and then in a phosphate buffer at pH 6.0 at 39 °C for 2 h each. Lemke et al. (2001) modified the method by adding enzymes to simulate the gastric and intestinal digestion environments. This two-steps method consists on the preparation of two distinct buffers containing pepsin enzyme, citric acid, malic acid, acetic acid, and lactic acid adjusted to pH 3.0 to simulate the gastric environment and incubated for 2 h at 38 °C. Then, the pH of the first medium is increased to 7.0 with sodium bicarbonate and mixed with a second buffer containing pancreatin and bile salts to simulate the intestinal environment for 2 additional hours. Gallo and Masoero (2010) reported that deionized water does not simulate properly the adsorption capacity of MTB when they compared the adsorption of AF by clinoptilolite using water or a buffer that simulates the GI digestion model, with an adsorption of 48 vs. 97%, respectively. Authors suggested that the GI model was more appropriate because it simulated closer the physiological conditions. However, no method has been validated. The pH is an important factor for the adsorption of mycotoxins and the determination of MTB adsorption capacity. Avantaggiato et al. (2005) reported that the adsorption capacity of zeolite at pH 3 was higher than at pH 8.0 for FUM (59 vs. 6%) and ZEA (54 vs. 17%). Similarly for YCW, Dawson et al. (2001) reported that the optimal pH of AF adsorption was 4.0. In contrast, the AC adsorption of OTA and ZEA was not affected by pH (Rotter et al., 1989; Bueno et al., 2004). Gallo and Masoero (2010) later proposed to use sterilized ruminal contents followed by the two-steps method of Lemke et al. (2001) to simulate cattle conditions.
This in vitro method is subjected to variation due to experimental conditions. One factor that has not been defined is the effect of the mycotoxin to MTB ratio on the adsorption capacity. A revision of the methodology of different in vitro studies showed a high variability in the MTB to mycotoxin ratios among different studies, ranging from 1:0.2 to 1:12 mg MTB/µg mycotoxin for YCW:DON ratio, and 1:0.00007 to 1:200 mg MTB/µg mycotoxin for bentonite:AF ratio (Table 1). The adsorption capacity of MTB is dose-dependent because the adsorption mechanism is limited by the available sites in the binder. A small dose of mycotoxin with a high concentration of MTB will result in a higher adsorption capacity. In contrast, a high concentration of mycotoxin will saturate the adsorption sites of the MTB resulting in lower adsorption. Bueno et al. (2004) and Avantaggiato et al. (2005) confirmed that the adsorption of ZEA by bentonite and AC was higher with increasing doses of MTB. Alternatively, increasing concentrations of ZEA with the same amount of YCW reduced the adsorption rate of the toxin (Joannis-Cassan et al., 2011). Therefore, there is an urgent need to define the adequate MTB to mycotoxin ratio for a fair evaluation of the MTB adsorption capacity.
Table 1.
Ranges of ratios of mycotoxin binder to mycotoxins doses used in in vitro tests to determine the mycotoxin adsorption capacity of different mycotoxin binders
| Binders1 | Mycotoxin2 | |||||
|---|---|---|---|---|---|---|
| AFB1 mg:µg | DON mg:µg | FUM mg:µg | OTA mg:µg | T-2 mg:µg | ZEA mg:µg | |
| AC | 1:0.008–1:461 | 1:0.2–1:90 | 1:0.92–1:25 | 1:0.025–1:125 | 1:0.1 | 1:0.05–1:20 |
| Bentonite | 1:0.00007–1:200 | 1:0.2–1:12 | 1:2–1:20 | 1:0.002–1:12.5 | 1:0.1–1:0.2 | 1:0.1–1:20 |
| Clinoptilolite | 1:0.02–1:40 | 1:1.2 | . | 1:2 | . | 1:0.05–1:12 |
| HSCAS | 1:0.002–1:600 | 1:0.4–1:12 | 1:2–1:20 | 1:0.025–1:10 | 1:0.1 | 1:0.001–1:20 |
| MMT | 1:0.0002–1:20 | 1:0.5–1:12 | 1:2.5 | 1:0.025 | 1:0.1 | 1:0.05–1:1 |
| Sepiolite | 1:1.6–1:10 | 1:2–1:12 | 1:2 | 1:10 | . | 1:0.05 |
| YCW | 1:0.001–1:461 | 1:0.2–1:12 | 1:2–1:20 | 1:0.001–1:10 | 1:0.1 | 1:0.001–1:20 |
| Zeolite | 1:0.002–1:20 | 1:0.2–1:10 | 1:0.2–1:20 | 1:0.016–1:5 | 1:0.2 | 1:0.05–1:20 |
1AC, activated carbon; HSCAS, hydrated sodium calcium aluminosilicate; MMT, montmorillonite; YCW, yeast cell wall.
2AFB1, aflatoxin B1; DON, deoxynivalenol; FUM, Fumonisin; OTA, ochratoxin; T-2, T-2 toxin; ZEA, zearalenone.
Adsorption isotherm test
The adsorption isotherm test consists on the evaluation of the adsorption capacity of MTB by the assessment of the amount of mycotoxin adsorbed per unit of weight of MTB. The model is based on the incubation of increasing doses of a mycotoxin with a constant concentration of a MTB in a phosphate medium at a fixed temperature and pH for 1 h in a continuous shaking water-bath (Lau et al., 2016). The free mycotoxin concentration left at each concentration gradient after the incubation is analyzed to fit the isotherm equations model (Kinniburgh, 1986). The application of the adsorption isotherm test to the adsorption of mycotoxins by MTB was described by Grant and Phillips (1998). However, there are also some shortcomings. The method assumes that the adsorption mechanism is specific for mycotoxins and no other molecules can be adsorbed or compete with the adsorption sites on MTB, and it is limited to a fixed pH, which fails to simulate the GI tract environment.
Dynamic gastrointestinal models
The TNO GI model was designed to mimic the conditions of the stomach and small intestine lumen continuously, and was validated as a good system to simulate the GI tract (Minekus et al., 1995). The model simulates the stomach, duodenum, jejunum, and ileum at the same time, where the different compartments are connected by peristaltic pumps that ensure the chyme transfer at the required passage rate. The system is adapted to reproduce many physiological conditions of the GI tract as meal transit, peristaltic movements, pH, gastric, and intestinal secretions, absorption of digested products, and water in each segment, and the removal of undigested compounds. Avantaggiato et al. (2003, 2004, 2005, 2007) evaluated the capacity of MTB to reduce the absorption of mycotoxins using this model. Results showed that the intestinal absorption of DON (51%) and nivalenol (21%) occurred mainly in the jejunum and ileum, and the addition of 2% of AC decreased the intestinal absorption of DON by 21% and that of nivalenol by 45% in comparison to the control (Avantaggiato et al., 2004).
Efficacy of Mycotoxin Binder to Adsorb Mycotoxins in In Vitro Studies
Materials and methods
Inclusion criteria and data extraction
A comprehensive literature search was conducted using PubMed, Google Scholar, and Science Direct engines to identify experiments reporting the capacity of MTB to adsorb mycotoxins in vitro. The research used as keywords the six mycotoxins mentioned previously, eight MTB (bentonite; zeolite; clinoptilolite; sepiolite; MMT; hydrated sodium calcium aluminosilicate (HSCAS); AC and YCW), adsorption capacity and in vitro. After the initial search, a total of 97 papers were identified. Papers were retained if, 1) the MTB was described; 2) the single, well described MTB was tested; and 3) the incubation medium of the experiment and the method used for the determination of the adsorption capacity was described. From the initial search, 29 papers were excluded for the following reasons: 23 papers did not describe the incubation medium used; 3 papers did not report data of adsorption; 2 papers did not report the MTB tested in the study; and 1 paper was not used for animal purposes. The summary of the procedure used to select papers is shown in the Supplementary Figure S1. The final analysis included 68 papers with 1843 data for the adsorption capacity of different MTB and mycotoxins. Data were extracted from text, tables, or figures. Adsorption values in figures were extracted using an extraction data program (Origin-Lab 2019, OriginLab Corp, Massachusetts, USA). The collected data included the main predictor variables, the mycotoxin, and the MTB, the doses used, the MTB:mycotoxin ratio, the pH of the medium, the time and temperature of incubation when available, and the type of the incubation medium used. In general, the selected papers used the simple concentration model for the incubation procedure carried on with one- or two-step methods with different incubation media. For the one-step method, buffers were water, water with methanol or water with hydrochloric acid. For the two-step method, buffers were used to mimic the GI tract pH using phosphate buffer at high pH (pH > 7.0), or low (pH < 4), acetate buffer for intermediate pH (pH 4–6), or citrate buffer for low pH (pH < 4), and data were obtained at each pH point separately. Other media were also identified to simulate the GI digestion using digestive enzymes (pepsin, bile salts, and pancreatin), rumen fluid, or gastric juice.
Statistical analysis
The response variable of percentage adsorption was analyzed using the PROC MIXED of SAS (version 9.4; SAS Institute Inc., Cary, NC). The mixed model was: Yijkl = µ + Si + MTBj + MTxk + Ml + pHm + MTBj * MTxk + MTBj * MTxk * Ml * pHm+ Sijk + eijklm, where Yijkl: is the dependent variable; µ: overall adsorption capacity mean; Si: the random effect of the ith study; MTBj: the fixed effect of the jth MTB; MTxk: the fixed effect of the kth mycotoxin; Ml: the fixed effect of the method; pHm: the fixed effect of the pH; MTBj * MTxk: the MTB by mycotoxin interaction; MTBj * MTxk * Ml * pHm: the method by MTB by mycotoxin by pH interaction; Sijk: the random interaction between the ith study, the jth level of MTB and the kth level of MTx; and eijklm: the residual error. The adsorption capacity results are presented as least squares of means. When a significant effect was detected, differences among means were tested using the Tukey’s multiple comparison test. Differences at a level of P < 0.05 were declared significant, and trends were considered at 0.05 < P ≤ 0.10.
Results and Discussion
Effect of incubation media on the MTB adsorption capacity
In vitro studies are used to evaluate the adsorption capacity of MTB, but results may be affected by the incubation conditions that can affect the interaction between MTB and mycotoxins. The two-step method represented 69% of the overall data. Media of water and simulated GI tract represented 10% of the data each, gastric juice media 6%, hydrochloric acid:water 3%, and methanol:water 2%. Analysis of the effect of the media on the adsorption capacity showed that gastric juice was the only method that differed from the other two-step methods (P < 0.05) and, because results were affected by this method and represented only 6% of data, they were removed from the dataset.
The overall adsorption capacity of different mycotoxin binders
Table 2 shows the MTB binding capacity results for each mycotoxin. Within MTB, YCW, and bentonite had the highest number of observations with 36 and 29% of total data, respectively, and clinoptilolite and sepiolite had the lowest number of observations with 2 and 1% of data, respectively. The adsorption capacity was the highest for AC (average of 81%) and was not different among mycotoxins (ranged from 53% with T-2 toxin to 93% with AF). The other MTB had lower adsorption compared with AC but were similar among them, ranging from 32% for zeolite to 48% for HSCAS. The average adsorption of HSCAS (48%) was the highest for AF and ZEA, and the lowest for DON. The average adsorption of MMT (48%) and bentonite (45%) was the highest for AF and the lowest for the other mycotoxins. The average adsorption of sepiolite (46%) and YCW (34%) was similar among mycotoxins ranging from 13 and 20% for DON to 95 and 49% for AF, respectively. No adsorption data were reported for sepiolite for FUM, OTA, and T-2. The average adsorption of clinoptilolite (32%) was the highest for AF and the lowest for ZEA, with no reported data for DON, FUM, and OTA. The adsorption of zeolite (32%) was the highest for AF and the lowest for DON. The average adsorption of bentonite (45%) was the highest for AF and the lowest in the other mycotoxins.
Table 2.
The adsorption efficacy of different mycotoxin binders against most common mycotoxins measured with in vitro methods (average adsorption ± SEM; number of samples in parenthesis)
| Binders1 | Mycotoxin | Average | |||||
|---|---|---|---|---|---|---|---|
| Aflatoxin B1 | Deoxynivalenol | Fumonisin | Ochratoxin | T-2 toxin | Zearalenone | ||
| AC | 93a ± 0.8 (n = 56) |
69a ± 0.8 (n = 59) |
83a ± 1.7 (n = 35) |
88a ± 1.8 (n = 28) |
53 ± 7.9 (n = 5) |
93a ± 1.6 (n = 23) |
81a ± 0.4 (n =2 06) |
| Bentonite | 86a,x ± 0.3 (n = 295) |
18b,y ± 1.4 (n = 25) |
32b,y ± 4.2 (n = 8) |
30b,y ± 0.6 (n = 136) |
22y ± 6.9 (n = 4) |
29b,y ± 1.1 (n = 39) |
45b ± 0.2 (n = 507) |
| Clip | 75ab,x ± 1.5 (n = 26) |
. | . | . | 29xy ± 16.0 (n = 2) |
14b,y ± 2.8 (n = 13) |
32b ± 1.2 (n = 41) |
| HSCAS | 83a,x ± 0.8 (n = 50) |
11b,y ± 1.6 (n = 26) |
52ab,xy ± 2.8 (n = 15) |
43ab,xy ± 5.1 (n = 5) |
32xy ± 12.6 (n = 2) |
52b,x ± 1.4 (n = 29) |
48b ± 0.5 (n = 127) |
| MMT | 88a,x ± 1.0 (n = 51) |
9b,y ± 6.3 (n = 4) |
42ab,y ± 12.7 (n = 2) |
26ab,y ±11.9 (n = 2) |
24y ± 13.1 (n = 2) |
47b,y ± 1.7 (n = 33) |
48b ± 0.8 (n = 94) |
| Sepiolite | 95ab ± 8.3 (n = 4) |
13ab ± 12.6 (n = 2) |
. | . | . | 39ab ± 11.3 (n = 3) |
46b ± 3.9 (n = 9) |
| YCW | 49b ± 0.4 (n = 165) |
20b ± 1.2 (n = 35) |
30b ± 2.5 (n = 18) |
43b ± 0.4 (n = 196) |
28 ± 3.8 (n = 9) |
48b ± 0.4 (n = 213) |
34b ± 0.2 (n = 636) |
| Zeolite | 61ab,x ± 1.5 (n = 22) |
10b,y ± 2.9 (n = 11) |
26b,x ± 2.3 (n = 27) |
44ab,x ± 1.3 (n = 45) |
5x ± 13.5 (n = 2) |
33b,x ± 2.1 (n = 19) |
32b ± 0.5 (n = 126) |
| Average | 77x ± 0.1 (n = 669) |
23z ± 0.5 (n = 162) |
45yz ± 1.0 (n = 105) |
47y ± 0.3 (n = 412) |
31yz ± 2.3 (n = 26) |
50y ± 0.3 (n = 372) |
(n = 1,746) |
1AC, activated carbon; Clip, clinoptilolite; HSCAS, hydrated sodium calcium aluminosilicate; MMT, montmorillonite; YCW, yeast cell wall.
a, b, cDifferent superscripts in the same column indicate a significant effect between binders (P < 0.05).
x, y, zDifferent superscripts in the same row indicate a significant effect between mycotoxins (P < 0.05).
The highest adsorption capacity of AC may be related to its adsorption mechanism discussed previously. The sizes of the pores in AC are measured in nanometers and are larger than the interlayer space of clay minerals measured in angstrom. Thus, mycotoxins with complex chemical structures can get easily into AC pores and not in the interlayer space of clays. Additionally, the activation of AC improves the binding capacity of polar and non-polar mycotoxins which makes it less selective and adsorbs different types of mycotoxins. Clay adsorbents were less effective than AC, with similar adsorption capacity among different types of clays. The adsorption mechanism of clays is based on their cation exchange capacity that includes different weak ionic interactions. Variations among mycotoxins may be associated with the cation exchange capacity and the interlayer space, which varies among and within clays depending on their sources (Nuryono et al., 2012; De Mil et al., 2015). The average adsorption capacity of YCW was similar to clay minerals, although the adsorption mechanism is different and based on the matching of structures of β-glucans and mycotoxins (Jouany, 2007; Yiannikouris et al. 2013).
When analyzing results from the mycotoxin adsorption point of view, AF, OTA and ZEA had the highest number of observations with 38, 24, and 21% of total data, respectively, and DON, FUM, and T-2 had the lowest observations with 9, 6 and 1% of total data, respectively. The average adsorption was the highest for AF (77%) among all mycotoxins, being the highest in AC (93%), bentonite (86%) and MMT (88%), and the lowest in YCW (49%). Two main characteristics allow a high adsorption of AF: the small and flat structure that favors its entrance into the interlayer space and pores of clays and AC, and the high polarity that facilitates the ionic interactions with MTB. In contrast, these properties do not facilitate the adsorption of AF by YCW. In addition to AF, ZEA (50%) and OTA (47%) had a similar average adsorption. For ZEA, adsorption was the highest in AC (93%) and the lowest in the other MTB (average of 38%), except for sepiolite that was not different (39%) probably due to the small number of treatments reported (n = 3). Similarly, OTA adsorption was the highest in AC (88%), and the lowest in YCW (43%) and bentonite (22%). Joannis-Cassan et al. (2011) reported that the high potential of YCW to adsorb OTA is due to the high correlation of mannoprotein in YCW that represents the key factor in the adsorption of OTA. In contrast, for other mycotoxins, β-glucans are the main adsorbing factor. Fumonisin and T-2 were the mycotoxins with fewer observations, 6 and 1%, respectively. For FUM (average of 45%), the adsorption capacity was the highest in AC (83%), and the lowest in bentonite (32%), YCW (30%), and zeolite (26%). For T-2 (average of 31%) the adsorption was not different among MTB (ranged from 5.3% with zeolite to 53% with AC). There is limited research available on the T-2 binding capacity, likely due to the fact that it is a controlled biological substance difficult to conduct research with. Carson and Smith (1983) and Bratich et al. (1990) reported that T-2 adsorption is MTB dose-dependent and suggested that MTB dose must be 10 times higher than the usual dose used for the AF binding. Deoxynivalenol had the lowest average adsorption (23%) with the highest adsorption with AC (69%) and the lowest with YCW (20%), bentonite (18%), HSCAS (11%), zeolite (10%) and MMT (9%), but the adsorption of sepiolite (13%) was not different due to the low number of observations (n = 2). The low adsorption of DON could be due to its hydrophobicity, attributed to the aromatic cycles that limit its binding to MTB with hydrophilic characteristics.
Although AF has been the most prevalent mycotoxin in feeds, AF occurrence has changed due to drastic control strategies and climate change that affect the type of mold proliferation in certain regions (Moretti et al., 2019). Several studies reported the prevalence of different types of mycotoxins worldwide (Streit et al., 2013; Eloska et al., 2019; Gruber-Dorninger et al., 2019). Results of sample analysis were coherent among studies. Streit et al. (2013) reported data on mycotoxins prevalence between 2004 and 2011, and showed that DON (64%) and FUM (63%) were the first contaminants of feeds. However, AF occurrence was only important in South East of Asia that increased from 33% in 2004 to 70% in 2011. The authors attributed the high incidence of AF in this region to the hot climate. Later, Gruber-Dorninger et al. (2019) reported data on mycotoxin occurrence between 2008 and 2017, and results also showed a high incidence of DON (64%) and FUM (60%), and lower occurrence of AF (23%). Eskola et al. (2019) compared the prevalence of mycotoxins from datasets of EFSA that were obtained after information access request (EFSA Ref. 17238686; PAD 2017 017), and of Biomin (Kovalsky et al., 2016), and results showed similar incidences between the two datasets with the highest incidence for ZEA (80%) and DON (60%). Surprisingly, the collected data was much lower for T-2 (n = 26) and FUM (n = 105) than for AF (n = 669). These results report the importance given to AF in detriment to other mycotoxins that also have high prevalence in crops. It is likely that the occurrence of mycotoxins and the changes in the prevalence of specific mycotoxins will increase in the future due to the impact of climate change, where higher average temperature across the world are expected (Yu et al., 2022)
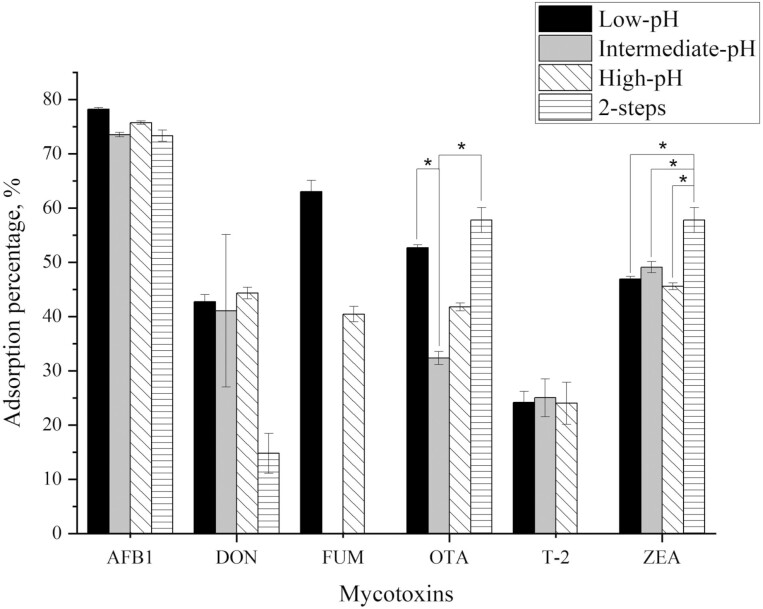
Effect of the pH on the adsorption capacity of mycotoxin binders
The pH values of the incubation media were grouped in four ranges: low (pH from 1 to 4, 42% of data); intermediate (from 5 to 6, 17% of data); high (from 7 to 9, 33% of data) and the two-step methods from low to high pH recording only the final adsorption values of the incubation procedure.
The analysis of data showed that pH affected the adsorption of OTA and ZEA (Figure 6, P < 0.08). The adsorption of OTA was the highest with the two-steps pH (58%), and the lowest with the low (53%) and intermediate (32%) pH. Similarly, the adsorption of ZEA was the highest with the two-steps pH (58%), and the lowest with the low (47%), intermediate (49%) and high (45%) pH. Results showed that the adsorption of OTA and ZEA was more efficient when the two-steps pH method was used. Faucet-Marquis et al. (2014) reported that alkaline pH resulted in the desorption of mycotoxins from adsorption sites of MTB. It is reasonable to think that pH affects more the adsorption of polar molecules (Thieu and Pettersson, 2008). However, FUM and AF are the highest polar mycotoxins and their adsorption was not affected by pH. In contrast, ZEA is the lowest polar mycotoxins and was affected by pH. In fact, other factors could influence the adsorption of mycotoxins as molecular size, structural shape, or solubility.
Figure 6.
Effect of pH (low, intermediate, high, and two-steps) on the adsorption percentage of different mycotoxins. (Bars represent standard error, *P < 0.08).
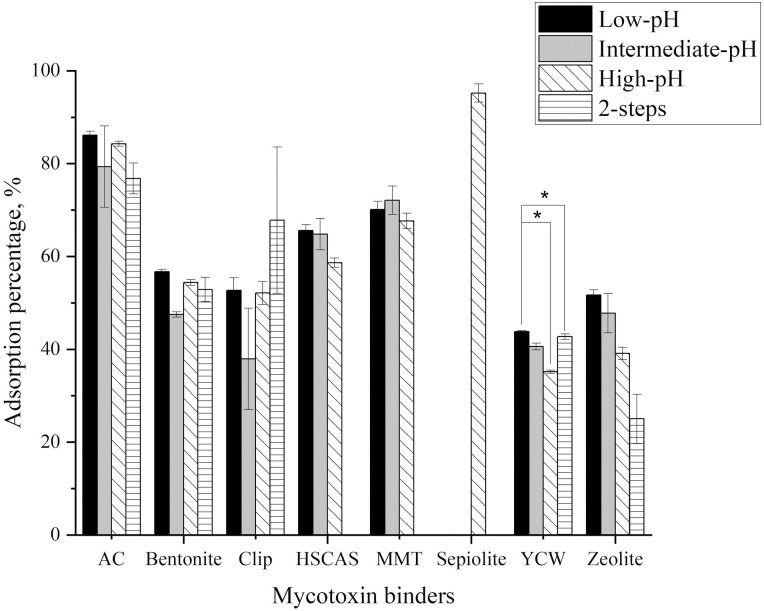
Data analysis also showed an effect of pH on the adsorption capacity of MTB (Figure 7). Among different MTB, the adsorption capacity of YCW was affected by pH (P < 0.05), and was higher with the low pH (43%) and lower with the high pH (35%). Results are consistent with Faucet-Marquis et al. (2014) that reported that the adsorption capacity of YCW was higher at low or neutral pH where the stability of β-glucans, responsible of YCW adsorption capacity, was improved. The pH of the media did not affect the other inorganic MTB.
Figure 7.
Effect of pH (low, intermediate, high, and two-steps) on the adsorption percentage of different mycotoxin binders. (Bars represent standard error, *P < 0.05).
Effect of the dose ratio MTB:mycotoxin on the adsorption capacity of mycotoxins
The mechanism of adsorption is a saturable process (Bueno et al., 2004; Avantaggiato et al., 2005). Therefore, the ratio MTB to mycotoxin may have a relevant effect on absorption results. Using the data selected for the analysis of the effectiveness of MTB, the MTB to mycotoxin ratios resulted in a wide range of ratios independently of the type of mycotoxin or MTB (1:0.00007–1:600 mg/µg, Table 1). Table 1 illustrates the different ranges used for each mycotoxin. The AF is the mycotoxin with the widest range of ratios (from 1:0.00007 to 1:600 mg/µg), while the range was narrower for DON (from 1:0.2 to 1:90 mg/µg) and FUM (from 1:0.2 to 1:25 mg/µg). However, in all cases, the range was very wide and, to the best of our knowledge, there are no established recommendations. This may justify the large variability in results observed in literature for the adsorption of mycotoxins and MTB. Therefore, it is important to set up guidelines for the in in vitro tests where the ratio MTB to mycotoxin is close to physiological conditions.
The Capacity of Mycotoxin Binders to Adsorb Nutrients
The non-selective mechanism of adsorption of MTB to adsorb mycotoxins allows a possible interaction with other essential nutrients. Organic compounds like fatty acids, amines, AA, vitamins, and aromatic compounds with similar molecular structure, molecular size, or surface charges to mycotoxins may also be adsorbed by MTB, and have negative effects on animal health (Vekiru et al., 2007; Barrientos-Velázquez et al., 2016; Kihal et al., 2020, 2021). The EFSA (2010) established guidelines for the assessment of feed additives that reduce mycotoxin feed contamination, requiring that MTB do not affect the apparent digestibility of crude protein and the bioavailability of vitamins B1, B6, A, and E when supplemented to animal diets. In fact, the EFSA (2011) warned against the use of bentonites at doses higher than 0.5% of diets because of its potential to reduce nutrient availability in the GI tract of animals. The MTB capacity to adsorb nutrients has been studied using in vitro models. Kihal et al. (2020, 2021) studied the interaction of six different MTB with AA and vitamins in an in vitro simulated GI model. Authors reported a range of adsorption from 27 to 37% for AA, 25–58% for water-soluble vitamins, and 10–29% for fat-soluble vitamins (Table 3). Barrientos-Velázquez et al. (2016) and Vekiru et al. (2007) also studied the capacity of bentonite and AC to adsorb vitamins B1, B8, and B12 in an in vitro simulated GI model. Vekiru et al. (2007) reported that AC adsorbed a large proportion of vitamin B8 (78%) and B12 (99%), while bentonite had lower adsorption of vitamin B12 (47%). Barrientos-Velázquez et al. (2016) reported that bentonite adsorbed 34% of vitamin B1 and the adsorption of AF was reduced by 34%, indicating a direct competition of other nutrients for the adsorption sites. Mortland et al. (1983) reported that smectite has the capacity to adsorb vitamin B2 (50%). Bentonite and MMT have been also reported to adsorb protein in an in vitro simulated GI model (Ralla et al., 2010; Barrientos-Velázquez et al., 2016). The capacity of MTB to adsorb minerals was also investigated in vitro by Tomasevic-Canovic et al. (2001) that reported a high capacity of bentonite to adsorb copper (56%) and cobalt (73%), but the adsorption of zinc (12%) and manganese (12%) was relatively low. In contrast, vitamins A, D, B3, B5, and B8, and AA tryptophan and phenylalanine were not adsorbed by bentonite and zeolite (Tomasevic-Canovic et al., 2001; Vekiru et al., 2007; Kihal et al., 2020). This difference in the adsorption capacity among nutrients is most likely related to the shape, size, and charges of the different micronutrients.
Table 3.
The capacity of 6 mycotoxin binders to adsorb amino acids and water-soluble and fat-soluble vitamins in vitro (percentage adsorption) (adapted from Kihal et al. 2020, 2021).
| Substrate | AA1 | WSV2 | FSV3 |
|---|---|---|---|
| Bentonite | 45ab | 49b | 25a |
| Clinoptilolite | 51a | 27cd | 19ab |
| Sepiolite | 40bc | 33c | 13b |
| Montmorillonite | 47a | 56a | 25a |
| Active carbon | 36c | 18e | 14b |
| Yeast cell wall | 48 a | 22de | 25a |
| SEM | 5.9 | 6.9 | 5.9 |
| Average | 45 | 34 | 20 |
1AA, amino acids: lysine, methionine, and threonine.
2WSV, water-soluble vitamins: B1, B2, B3, and B6.
3FSV, fat-soluble vitamins: D and E.
a, b, c, d, eDifferent superscripts in the same column indicate a significant effect between binders (P < 0.05).
Vitamin availability was also studied in vivo. Briggs and Fox (1956) supplemented chick diets with 2–3% of bentonite and reported a vitamin A deficiency. The Zinc content was also decreased in chick bones after HSCAS was supplemented at 0.5–1% of the diet (Chung et al., 1990). In contrast, Afriyie-Gyawu (2004) and Pimpukdee et al. (2004) reported that the inclusion of 0.5% bentonite did not affect liver vitamin A concentration. Similarly, HSCAS did not affect the availability of vitamin A, vitamin B2, and manganese in chicks at 0.5–1% inclusion in the diet (Chung et al., 1990). Sulzberger et al. (2016) and Kihal et al. (2022) reported that the supplementation of 1.2 and 2% of MMT in the diet of dairy cows, respectively, did not affect the plasma concentration of vitamins A, D, E, B1, and B6. Maki et al. (2016) supplemented HSCAS to dairy cows at 1.2% of the diet DM and reported no effects on the bioavailability of vitamins A and B2 in milk. Table 4 summarizes the available literature on the interaction of MTB with nutrients.
Table 4.
Summary of studies that determine the capacity of different mycotoxin binders to adsorb nutrients
| Binders1 | Nutrient interaction effects | Observation | References |
|---|---|---|---|
| Bentonite | High adsorption of vitamins E, B1, B2, and B6 and amino acids: lysine, methionine and threonine | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| Low adsorption of vitamins A, D, and B3 | |||
| High adsorption of vitamin B1 and pepsin No adsorption of vitamins D, and E |
In vitro gastric fluid simulation | Barrientos-Velázquez et al. (2016) | |
| High adsorption of vitamin B12 and B8 No adsorption of vitamin B5 |
In vitro gastric fluid simulation and real gastric fluid | Vekiru et al. (2007) | |
| High adsorption of vitamin B6 Adsorption of Zn and Co No adsorption of Cu and Mn |
In vitro in aqueous solution | Tomasevic-Canovic et al. (2000) | |
| Adsorption of vitamin B2 | In vitro in aqueous solution | Mortland and Lawless (1983) | |
| No adsorption of vitamin A | In vivo in chicks | Pimpukdee et al. (2004) | |
| No adsorption of vitamin A | In vivo in chicks | Afriyie-Gyawu (2004) | |
| MMT1 | High adsorption of vitamins E, B1, B2, and B6 and amino acids: lysine, methionine, and threonine | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| Low adsorption of vitamins A, D and B3 | |||
| Adsorption of vitamin B1 | In vitro gastric fluid simulation | Ghanshyam et al. (2009) | |
| Adsorption of protein, urea, and antibiotics | In vitro in agar culture | Pinck (1962) | |
| No adsorption of vitamins A, D, E, B1, and B6 | In vivo in dairy cows | Kihal et al. (2022) | |
| Ca MMT | No adsorption of vitamins A and B1 | In vivo in dairy cows | Maki et al. (2016) |
| AC2 | High adsorption of vitamins E, B1, B2 and B6 and amino acids: lysine, methionine and threonine | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| Low adsorption of vitamins A, D and B3 | |||
| Adsorption of vitamins B8 and B12 | In vitro simulation of gastric fluid and real gastric fluid | Vekiru et al. (2007) | |
| Clinoptilolite | High adsorption of vitamins E, B1, B2, and B6 and amino acids: lysine, methionine and threonine | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| No adsorption of vitamins A, D, and B3 | |||
| No adsorption of amino acids: tryptophan; Phenilanaline and vitamins: A, D, and E | In vitro in aqueous solution | Tomasevic-Canovic et al. (2000) | |
| HSCAS3 | No adsorption of vitamins A, B1, and minerals Zn, Mn | In vivo in chicks | Chung et al. (1990) |
| Sepiolite | High adsorption of vitamins E, B1, B2, and B6 and amino acids: lysine, methionine, and threonine | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| Low adsorption of vitamins A, D, and B3 | |||
| Zeolite | High adsorption of vitamins E, B1, B2, and B6 and amino acids: lysine, methionine, and threonine | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| Low adsorption of vitamins A, D, and B3 |
1MMT, montmorillonite.
2AC, activated carbon.
3HSCAS, hydrated sodium calcium aluminosilicate.
Conclusions
The presence of mycotoxins in feeds is a relevant problem in the animal feed industry. The presence of mycotoxins in raw materials is affected by many factors and their prevalence may change in favor of some mycotoxins over others. The adsorption of mycotoxins by MTB in in vitro tests is variable, with the highest adsorption capacity for AC and the lowest adsorption for clay adsorbents and YCW. For mycotoxins, the adsorption of AF was the highest and that of DON the lowest. The pH of the in vitro media affects the adsorption capacity of YCW, with the highest adsorption at low pH. For mycotoxins, pH affected the adsorption of OTA and ZEA. In general, when MTB are used at recommended doses are effective in reducing the bioavailability of mycotoxins. Yet, it is difficult to select the appropriate adsorbent for each mycotoxin. The in vitro tests that are widely used to assess the adsorption capacity of MTB have many limitations that have been demonstrated in this review (incubation medium type, pH conditions, appropriate MTB to mycotoxin ratio and nutrient interactions) that result in high variation among studies. In vitro tests need to be standardized to have an objective evaluation of the capacity of MTB to adsorb mycotoxins.
Supplementary Data
Supplementary data are available at Journal of Animal Science online.
Figure S1. Summary of revised studies included or excluded from the analysis.
Acknowledgments
Project funded by ACCIO (Departament d’Innovació, Universitats i Empresa, Generalitat de Catalunya), project INNOTEC ACE014/20/000068.
Glossary
Abbreviations
- AC
activated carbon
- AF
aflatoxin
- DON
deoxynivalenol
- FUM
fumonisin
- GI
gastrointestinal
- MMT
montmorillonite
- MTB
mycotoxin binder
- OTA
ochratoxin
- T-2
T-2 toxin
- YCW
yeast cell wall
- ZEA
zearalenone
Contributor Information
Abdelhacib Kihal, Animal Nutrition and Welfare Service, Departament de Ciència Animal i dels Aliments, Universitat Autònoma de Barcelona, 08193 Bellaterra, Spain.
María Rodríguez-Prado, Animal Nutrition and Welfare Service, Departament de Ciència Animal i dels Aliments, Universitat Autònoma de Barcelona, 08193 Bellaterra, Spain.
Sergio Calsamiglia, Animal Nutrition and Welfare Service, Departament de Ciència Animal i dels Aliments, Universitat Autònoma de Barcelona, 08193 Bellaterra, Spain.
Conflict of interest statement
The authors declare no conflicts of interest.
REFERENCES
- Afriyie-Gyawu, E. 2004. Safety and efficacy of Novasil clay as a dietary supplement to prevent aflatoxicosis [Ph.D. Diss]. Texas: Texas A&M University. [Google Scholar]
- Arroyo-Manzanares, N., Rodriguez-Estevez V., Arenas-Fernández P., García-Campaña A. M., and Gamiz-Gracia L.. . 2019. Occurrence of mycotoxins in swine feeding from Spain. Toxins. 2019:342. doi: 10.3390/toxins11060342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avantaggiato, G., Greco D., Damascelli A., Solfrizzo M., and Visconti A.. . 2014. Assessment of multi-mycotoxin adsorption efficacy of grape pomace. J. Agric. Food Chem. 62:497–507. doi: 10.1021/jf404179h. [DOI] [PubMed] [Google Scholar]
- Avantaggiato, G., Havenaar R., and Visconti A.. . 2003. Assessing the zearalenone-binding activity of adsorbent materials during passage through a dynamic in vitro gastrointestinal model. Food Chem. Toxicol. 41:1283–1290. doi: 10.1016/s0278-6915(03)00113-3. [DOI] [PubMed] [Google Scholar]
- Avantaggiato, G., Havenaar R., and Visconti A.. . 2004. Evaluation of the intestinal absorption of deoxynivalenol and nivalenol by an in vitro gastrointestinal model, and the binding efficacy of activated carbon and other adsorbent materials. Food Chem. Toxicol. 42:817–824. doi: 10.1016/j.fct.2004.01.004. [DOI] [PubMed] [Google Scholar]
- Avantaggiato, G., Havenaar R., and Visconti A.. . 2007. Assessment of the multi-mycotoxin-binding efficacy of a carbon/aluminosilicate-based product in an in vitro gastrointestinal model. J. Agric. Food Chem. 55:4810–4819. doi: 10.1021/jf0702803. [DOI] [PubMed] [Google Scholar]
- Avantaggiato, G., Solfrizzo M., and Visconti A.. . 2005. Recent advances on the use of adsorbent materials for detoxification of fusarium mycotoxins. Food Addit. Contam. 22:379–388. doi: 10.1080/02652030500058312. [DOI] [PubMed] [Google Scholar]
- Barrientos-Velázquez, A. L., Arteaga S., Dixon J. B., and Deng Y.. . 2016. The effect of pH, pepsin, exchange cation, and vitamins on aflatoxin adsorption on smectite in simulated gastric fluids. Appl. Clay Sci. 120:17–23. doi: 10.1016/j.clay.2015.11.014. [DOI] [Google Scholar]
- Bergaya, F., and Lagaly G.. . 2013. Handbook of clay science. Vol 5, 2nd ed.Amsterdam. Netherland: Elsevier. [Google Scholar]
- Bratich, P. M., Buck W. B., and Haschek W. M.. . 1990. Prevention of T-2 toxin-induced morphologic effects in the rat by highly activated charcoal. Arch. Toxicol. 64:251–253. doi: 10.1007/BF02010733. [DOI] [PubMed] [Google Scholar]
- Briggs, G. M., and Fox M. R. S.. . 1956. Vitamin a deficiency in chicks produced by adding high levels of bentonite to synthetic diet. Poult. Sci. 35:570–576. doi: 10.3382/ps.0350570. [DOI] [Google Scholar]
- Bryden, W. L. 2012. Mycotoxin contamination of the feed supply chain: implications for animal productivity and feed security. Anim. Feed Sci. Technol. 173:134–158. doi: 10.1016/j.anifeedsci.2011.12.014. [DOI] [Google Scholar]
- Bueno, D. J., Di Marco L., Oliver G., and Bardon A.. . 2004. In vitro binding of zearalenone to different adsorbents. J. Food Prot. 68:613–615. doi: 10.4315/0362-028x-68.3.613. [DOI] [PubMed] [Google Scholar]
- Carson, M. S., and Smith T. K.. . 1983. Role of bentonite in prevention of T-2 toxicosis in rats. J. Anim. Sci. 57:1498–1506. doi: 10.2527/jas1983.5761498x. [DOI] [PubMed] [Google Scholar]
- Chung, T. K., Erdman J. W., and Baker D. H.. . 1990. Hydrated sodium calcium aluminosilicate: effects on zinc, manganese, vitamin A and riboflavin utilization. Poult. Sci. 69:1364–1370. doi: 10.3382/ps.0691364. [DOI] [PubMed] [Google Scholar]
- Čolović, R., Puvaca N., Cheli F., Avantaggiato G., Greco D., Duragic O., and Pinotti L.. . 2019. Decontamination of mycotoxin-contaminated feedstuffs and compound feed. Toxins. 11:617. doi: 10.3390/toxins11110617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danish, M., and Ahmed T.. . 2018. A review on utilization of wood biomass as a sustainable precursor for activated carbon production and application. Renew. Sust. Energ. Rev. 87:1–21. doi: 10.1016/j.rser.2018.02.003. [DOI] [Google Scholar]
- Dawson, K. A., Evans J., and Kudupoje M.. . 2001. Understanding the adsorption characteristics of yeast cell wall preparations associated with mycotoxin binding. In: Lyons T. P., and Jacques K. A, editors. Science and technology in the feed industry. Nottingham, UK: Nottingham University Press; p. 169–181. [Google Scholar]
- De Mil, T., Devreese M., Baere S., Van Ranst E., Eexkhout M., Backer P., and Croubels S.. . 2015. Characterization of 27 mycotoxin binders and the relation with in vitro zearalenone adsorption at a single concentration. Toxins. 7:21–33. doi: 10.3390/toxins7010021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaz, D. E., Hagler W. M., Blackwelder J. T. Jr, Eve J. A., Hopkins B. A., Anderson K. L., Jones F. T., and Whitlow L. W.. . 2004. Aflatoxin binders II: reduction of aflatoxin M1 in milk by sequestering agents of cows consuming aflatoxin in feed. Mycopathology. 157:233–241. doi: 10.1023/B:MYCO.0000020587.93872.59. [DOI] [PubMed] [Google Scholar]
- Di Gregorio, M. C., Neef D. V., Jager A. V., Corassin C. H., Carao A. C. P., Albuquereque R., Azevedo A. C., and Oliveira C. A. F.. . 2014. Mineral adsorbents for prevention of mycotoxins in animal feeds. Toxin Rev. 33:125–135. doi: 10.3109/15569543.2014.905604. [DOI] [Google Scholar]
- Eseceli, H., Ozcan M. A., Demir E., and Bilal T.. . 2017. Increasing the adsorption of clinoptilolite as toxin binder by heat treatment. Bulg. J. Vet. Med. 20:18–26. [Google Scholar]
- Eskola, M., Kos G., Elliott C. T., Hajšlová J., Mayar S., and Krska R.. . 2019. Worldwide contamination of food-crops with mycotoxins: validity of the widely cited ‘FAO estimate’ of 25%. Crit. Rev. Food Sci. Nutr. 60:2773–2789. doi: 10.1080/10408398.2019.1658570. [DOI] [PubMed] [Google Scholar]
- European Commission (EC). 2006. EC regulation No 401/2006. Laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Off. J. Eur. Comm. L. 70:12–34. [Google Scholar]
- European Food Safety Authority (EFSA). 2010. Statement on the establishment of guidelines for the assessment of additives from the functional group substances for reduction of the contamination of feed by mycotoxins. EFSA J. 8:1963. doi: 10.2903/j.efsa.2010.1459. [DOI] [Google Scholar]
- European Food Safety Authority (EFSA). 2011. Opinion of the scientific panel on additives and products or substances used in animal feed (FEEDAP panel) on the safety and efficacy of bentonite (dioctahedral montmorillonite) as feed additive for all species. EFSA J. 9:2007 doi: 10.2903/j.efsa.2014.3694. [DOI] [Google Scholar]
- Faucet-Marquis, V., Joannis-Cassan C., Hadjeba-Medjdoub K., Ballet N., and Pfohl-Leszkowicz A.. . 2014. Development of an in vitro method for the prediction of mycotoxin binding on yeast-based products: case of aflatoxin B1, zearalenone and ochratoxin A. Appl. Microbiol. Biotechnol. 98:7583–7596. doi: 10.1007/s00253-014-5917-y. [DOI] [PubMed] [Google Scholar]
- Fernandes, J. M., Calado T., Guimaraes A., Rodrigues M. A. M., and Abrunhosa L.. . 2019. In vitro adsorption of aflatoxin B1, ochratoxin A, and zearalenone by micronized grape stems and olive pomace in buffer solutions. J. Mycotoxin Res 35:243–252. doi: 10.1007/s12550-019-00349-9. [DOI] [PubMed] [Google Scholar]
- Gallo, A., and Masoero F.. . 2010. In vitro models to evaluate the capacity of different sequestering agents to adsorb aflatoxins. Ital. J. Anim. Sci. 9:e21. doi: 10.4081/ijas.2010.e21. [DOI] [Google Scholar]
- Galvano, F., Pietri A., Bertuzzi T., Bognanno M., Chies L., Angelis A., and Galvano M.. . 1997. Activated carbons: in vitro affinity for fumonisin B1 and relation of adsorption ability to physicochemical parameters. J. Food Prot. 60:985–991. doi: 10.4315/0362-028X-60.8.985. [DOI] [PubMed] [Google Scholar]
- Galvano, F., Pietri A., Fallico B., Bertuzzi T., Scire S., Galvano M., and Maggiore R.. . 1996. Activated carbons: in vitro affinity for aflatoxin B1 and relation of adsorption ability to physicochemical parameters. J. Food Prot. 59:545–550. doi: 10.4315/0362-028X-59.5.545. [DOI] [PubMed] [Google Scholar]
- Galvano, F., Piva A., Ritieni A., and Galvano G.. . 2001. Dietary strategies to counteract the effect of mycotoxins: a review. J. Food Prot. 64:120–131. doi: 10.4315/0362-028x-64.1.120. [DOI] [PubMed] [Google Scholar]
- Ghanshyam, V., Joshi G. V., Patel H. A., Kevadiya B. D., and Bajaj H. C.. . 2009. Montmorillonite intercalated with vitamin B1 as drug carrier. Appl. Clay Sci. 45:248–253. doi: 10.1016/j.clay.2009.06.001. [DOI] [Google Scholar]
- Gimeno, A., and Martins M. L.. . 2007. Micotoxinas y micotoxicosis en animales y humanos. 1st ed.Douglas road, Miami, FL. USA: Special nutrients; p. 128. [Google Scholar]
- Goto, T., Amano Y., Machida M., and Imazeki F.. . 2015. Effect of polarity of activated carbon surface, solvent and adsorbate on adsorption of aromatic compounds from liquid phase. Chem. Pharm. Bull. 63:726–730. doi: 10.1248/cpb.c15-00039. [DOI] [PubMed] [Google Scholar]
- Grant, P. G., and Phillips T.. . 1998. Isothermal adsorption of aflatoxin B1 on HSCAS clay. J. Agric. Food Chem. 46:599–605. doi: 10.1021/jf970604v. [DOI] [PubMed] [Google Scholar]
- Greco, D., Ascanio V., Sanovito E., Logrieco A. F., and Avantaggiato G.. . 2018. Comparative efficacy of agricultural by-products in sequestering mycotoxins. J. Sci. Food Agric. 99:1623–1634. doi: 10.1002/jsfa.9343. [DOI] [PubMed] [Google Scholar]
- Grim, R. E. 1962. Applied clay mineralogy. New York: McGraw Hill. [DOI] [PubMed] [Google Scholar]
- Grim, R. E., and Güven N.. . 1978. Bentonites, geology, mineralogy, properties and uses, development in sedimentology. Vol. 24. Amsterdam: Elsevier. [Google Scholar]
- Gruber-Dorninger, C., Jenkins T., and Schatzmayr G.. . 2019. Global mycotoxin occurrence in feed: a ten-year survey. Toxins. 11:375. doi: 10.3390/toxins11070375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ismadji, S., Soetaredjo F. E., and Ayucitra A.. . 2015. Clay materials for environmental remediation. Springer briefs in molecular science. Cham: Springer. doi: 10.1007/978-3-319-16712-1 [DOI] [Google Scholar]
- Jaynes, W. F., and Zartman R. E.. . 2011. Aflatoxin toxicity reduction in feed by enhanced binding to surface-modified clay additives. Toxins. 3:551–565. doi: 10.3390/toxins3060551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joannis-Cassan, C., Tozlovanu M., Hadjeba-medjdoub K., Ballet N., and Pfohl-leszkowicz A.. . 2011. Binding of zearalenone, aflatoxin b1, and ochratoxin a by yeast-based products: a method for quantification of adsorption performance. J. Food Prot. 74:1175–1185. doi: 10.4315/0362-028X.JFP-11-023. [DOI] [PubMed] [Google Scholar]
- Jouany, J. P. 2007. Methods for preventing, decontaminating and minimizing the toxicity of mycotoxins in feeds. Anim. Feed Sci. Technol. 137:342–362. doi: 10.1016/j.anifeedsci.2007.06.009. [DOI] [Google Scholar]
- Jouany, J. P., Yiannikouris A., and Bertin G.. . 2005. How yeast cell wall components can alleviate mycotoxicosis in animal production and improve the safety of edible animal products. J. Anim. Sci. 14:171–190. doi: 10.22358/jafs/70361/2005. [DOI] [Google Scholar]
- Jouany, J. P., Yiannikouris A., and Bertin G.. . 2009. Risk assessment of mycotoxins in ruminants and ruminant products. Opt. Mediterr. 85:205–224. [Google Scholar]
- Kandel, R. 2018. Potential for using aluminosilicates for removal of heavy metals and mycotoxins from feed and water [Master Diss]. Ås, Norway: Norwegian University of Life Science. [Google Scholar]
- Kihal, A., Marquès C., Rodríguez-Prado M. E., Cunilleras E. J., and Calsamiglia S.. . 2022. Effect of diet supplementation with the mycotoxin binder montmorillonite on the bioavailability of vitamins in dairy cows. Toxins. 14:26. doi: 10.3390/toxins14010026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kihal, A., Rodríguez-Prado M. E., Cristofol C., and Calsamiglia S.. . 2021. Short communication: quantification of the effect of mycotoxin binders on the bioavailability of fat-soluble vitamins in vitro. Animals. 11:2251. doi: 10.3390/ani11082251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kihal, A., Rodriguez-Prado M. E., Godoy C., Cristofol C., and Calsamiglia S.. . 2020. In vitro assessment of the capacity of certain mycotoxin binders to adsorb some amino acids and water-soluble vitamins. J. Dairy Sci. 103:3125–3132. doi: 10.3168/jds.2019-17561. [DOI] [PubMed] [Google Scholar]
- Kinniburgh, D. G. 1986. General purpose adsorption isotherms. Environ. Sci. Technol. 20:895–904. doi: 10.1021/es00151a008. [DOI] [PubMed] [Google Scholar]
- Kogan, G., and Kocher A.. . 2007. Role of yeast cell wall polysaccharides in pig nutrition and health protection. Livest. Sci. 109:161–165. doi: 10.1016/j.livsci.2007.01.134. [DOI] [Google Scholar]
- Kovalsky, P., Kos G., Nährer K., Schwab C., Jenkins T., Schatzmayr G., Sulyok M., and Krska R.. . 2016. Co-occurrence of regulated, masked and emerging mycotoxins and secondary metabolites in finished feed and maize—an extensive survey. Toxins. 8:363. doi: 10.3390/toxins8120363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krska, R., de Nijs M., McNerney O., Pichler M., Gilbert J., Edwards S., Suman M., Magan N., Rossi V., van der Fels-Klerx H. J., . et al. 2016. Safe food and feed through an integrated toolbox for mycotoxin management: the MyToolBox approach. World Mycotox. J. 9:487–495. doi: 10.3920/WMJ2016.2136. [DOI] [Google Scholar]
- Lau, L. C., Nor N. M., Teong K., and Mohamed A.. . 2016. Hydrogen sulphide removal using CeO2/NaOH/PSAC: effect of process conditions and regeneration study. J. Environ. Chem. Eng. 4:3479–3483. doi: 10.1016/j.jece.2016.07.025. [DOI] [Google Scholar]
- Lemke, S. L., Ottinger S. E., Mayura K., Ake C. L., Pimpukdee K., Wang N., and Phillips T. D.. . 2001. Development of a multi-tiered approach to the in vitro pre-screening of clay-based enterosorbents. Anim. Feed Sci. Technol. 93:17–29. doi: 10.1016/S0377-8401(01)00272-3. [DOI] [Google Scholar]
- Ma, Y., Liang H., Yin J., Yao D., Xia Y., Zuo K., and Zeng Y.. . 2021. Air activation of charcoal monoliths for capacitive energy storage. RSC Adv. 11:15118–15130. doi: 10.1039/d1ra02192c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maki, C. R., Monteiro A. P. A., Elmore S. E., Tao S., Bernard J. K., Harvey R. B., Romoser A. A., and Phillips T. D.. . 2016. Calcium montmorillonite clay in dairy feed reduces aflatoxin concentrations in milk without interfering with milk quality, composition or yield. Anim. Feed Sci. Technol. 2141:130–135. doi: 10.1096/fj.07-8720com. [DOI] [Google Scholar]
- Minekus, M., Marteau P., Havenaar R., and Huis in’t Veld J. H. J.. . 1995. A multicompartmental dynamic computer-controlled model simulating the stomach and small intestine. ATLA. 23:197–209. doi: 10.1177/026119299502300205. [DOI] [Google Scholar]
- Moreno-Castilla, C., Pérez-Cadenas A. F., Maldonado-Hodar F. J., Carrasco-Marin F., and Fierro J. L. G.. . 2003. Influence of carbon–oxygen surface complexes on the surface acidity of tungsten oxide catalysts supported on activated carbons. Carbon. 41:1157–1167. doi: 10.1016/S0008-6223(03)00023-X. [DOI] [Google Scholar]
- Moretti, A., Pascale M., and Logrieco A. F.. . 2019. Mycotoxin risks under a climate change scenario in Europe. Trends Food Sci. Technol. 84:38–40. doi: 10.1016/j.tifs.2018.03.008. [DOI] [Google Scholar]
- Mortland, M. M., and Lawless J. G.. . 1983. Smectite interaction with riboflavin. Clays Clay Miner. 31:435–439. doi: 10.1346/CCMN.1983.0310604. [DOI] [PubMed] [Google Scholar]
- Murray, H. H. 2007. Developments in clay science. Occurrences, processing and applications of kaolins, bentonites, palygorskitesepiolite, and common clays; vol. 2. UK: Elsevier Science & Technology; p. 1–180. [Google Scholar]
- Nadziakiewicza, M., Kehoe S., and Micek P.. . 2019. Physico-chemical properties of clay minerals and their use as a health promoting feed additive. Animals. 9:714. doi: 10.3390/ani9100714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nava-Ramírez, M. J., Salazar M., Sordo M., López-Coello C., Téllez-Isaías G., Méndez-Albores A., and Vázquez-Durán A.. . 2021. Ability of low contents of biosorbents to bind the food carcinogen aflatoxin B1 in vitro. Food Chem. 345:128863. doi: 10.1016/j.foodchem.2020.128863. [DOI] [PubMed] [Google Scholar]
- Nuryono, N., Agus A., Wedhastri S., Maryudhani Y. M. S., Pranowo, Yunianto D., and Razzazi-Fazeli E.. . 2012. Adsorption of aflatoxin B1 in corn on natural zeolite and bentonite. Indo. J. Chem. 12:28. doi: 10.22146/ijc.21343. [DOI] [Google Scholar]
- Papatsiros, V. G., Stylianaki I., Tsekouras N., Papakonstantinou G., Gómez-Nicolau N. S., Letsios M., and Papaioannou N.. . 2021. Exposure biomarkers and histopathological analysis in pig liver after exposure to mycotoxins under field conditions: special report on Ffmonisin B1. Foodb. Path. Dis. 18:315–321. doi: 10.1089/fpd.2020.2867. [DOI] [PubMed] [Google Scholar]
- Pimpukdee, K., Kubena L. F., Bailey C. A., Huebner H. J., Afriyie-Gyawu E., and Phillips T. D.. . 2004. Aflatoxin-induced toxicity and depletion of hepatic vitamin a in young broiler chicks: protection of chicks in the presence of low levels of NovaSil PLUS in the diet. Poult. Sci. 83:737–744. doi: 10.1093/ps/83.5.737. [DOI] [PubMed] [Google Scholar]
- Pinck, L. A. 1962. Adsorption of proteins, enzymes and antibiotics by montmorillonite; vol. 9. In: Swineford, A., editor. Clays and Clay Minerals: Proceedings of 9th National Conference on Clays and Clay Minerals. New York, NY: Pergamon Press; p. 520–529. doi: 10.1346/CCMN.1960.0090138 [DOI] [Google Scholar]
- Ralla, K., Sohling U., Riechers D., Kasper C., Ruf F., and Scheper T.. . 2010. Adsorption and separation of proteins by a smectite clay mineral. Bioprocess. Biosyst. Eng. 33:847–861. doi: 10.1007/s00449-010-0408-8. [DOI] [PubMed] [Google Scholar]
- Ramos, A. J., and Hernandez E.. . 1996. Prevention of aflatoxicosis in farm animals by means of hydrated sodium calcium aluminosilicate addition to feedstuffs: a review. Anim. Feed Sci. Technol. 65:197–206. doi: 10.1016/S0377-8401(96)01084-X. [DOI] [Google Scholar]
- Rotter, R. G., Frohlich A. A., and Marquardt R. R.. . 1989. Influence of dietary charcoal on ochratoxin a toxicity in leghorn chicks. Can. J. Vet. Res. 53:449–453. [PMC free article] [PubMed] [Google Scholar]
- Samantray, J., Anand A., Dash B., Ghosh M. K., and Behera A. K.. . 2022. Silicate minerals potential source of potash—a review. Miner. Eng. 179:107463. doi: 10.1016/j.mineng.2022.107463. [DOI] [Google Scholar]
- Schatzmayr, G., and Streit E.. . 2013. Global occurrence of mycotoxins in the food and feed chain: facts and figures. World Mycotoxin J. 6:213–222. doi: 10.3920/WMJ2013.1572. [DOI] [Google Scholar]
- Schoonheydt, R. A., and Johnston C. T.. . 2011. The surface properties of clay minerals. EMU Notes Mineral. 11:337–373. doi: 10.1180/EMU-notes.11.10. [DOI] [Google Scholar]
- Streit, E., Naehrer K., Rodrigues I., and Schatzmayr G.. . 2013. Mycotoxin occurrence in feed and feed raw materials worldwide: long-term analysis with special focus on Europe and Asia. J. Sci. Food Agric. 93:2892–2899. doi: 10.1002/jsfa.6225. [DOI] [PubMed] [Google Scholar]
- Sulzberger, S. A., Melnichenko S., and Cardoso F. C.. . 2016. Effects of clay after an aflatoxin challenge on aflatoxin clearance, milk production, and metabolism of Holstein cows. J. Dairy Sci. 100:1856–1869. doi: 10.3168/jds.2016-11612. [DOI] [PubMed] [Google Scholar]
- Talavera, N., Navarro M., Sifontes Á. B., Díaz Y., Villalobos H., Niño-Vega G., Boada-Sucre A. A., and González I.. . 2013. Green synthesis of nanosized vanadium pentoxide using Saccharomyces cerevisiae as bio template. In: Pandalai, S. G., editor. Recent research developments in materials science. Kerala, India: Research Signpost; p. 89–102. [Google Scholar]
- Thieu, N. Q., and Pettersson H.. . 2008. In vitro evaluation of the capacity of zeolite and bentonite to adsorb aflatoxin B1 in simulated gastrointestinal fluids. Mycotox. Res. 24:124–129. doi: 10.1007/BF03032338. [DOI] [PubMed] [Google Scholar]
- Tomasevic-Canovic, M., Dakovic A., and Markovic V.. . 2001. The effect of exchangeable cations in clinoptilolite and montmorillonite on the adsorption of aflatoxin B1. J. Serb. Chem. Soc. 66:555–561. doi: 10.2298/jsc0108555t. [DOI] [Google Scholar]
- Tomasevic-Canovic, M., Dakovic A., Markovic V., Radosavljevic-Mihajlovic A., and Vukicevic J.. . 2000. Adsorption effects of mineral adsorbents; part III: adsorption behaviour in the presence of vitamin B6 and microelements. Acta Vet. 50:23–30. [Google Scholar]
- Tomasevic-Canovic, M., Dakovic A., Rottinfhaus G., Matijasevic S., and Duricic M.. . 2003. Surfactant modified zeolites new efficient adsorbents for mycotoxins. Micropor. Mesopor. Mat. 61:173–180. [Google Scholar]
- Vekiru, E., Fruhauf S., Rodrigues I., Ottner F., Krska R., Schatzmayr G., Ledoux D. R., G. E.Rottinghaus., and Bermudez A. J.. . 2014. In vitro binding assessment and in vivo efficacy of several adsorbents against aflatoxin B1. World Mycotoxin J. 8:477–488. doi: 10.3920/WMJ2014.1800. [DOI] [Google Scholar]
- Vekiru, E., Fruhauf S., Sahin M., Ottner F., Schatzmayr G., and Krska R.. . 2007. Investigation of various adsorbents for their ability to bind aflatoxin B1. Mycotoxin Res. 23:27–33. doi: 10.1007/BF02946021. [DOI] [PubMed] [Google Scholar]
- Xu, R., Kiarie E. G., Yiannikouris A., Sun L., and Niel A.. . 2022. Nutritional impact of mycotoxins in food animal production and strategies for mitigation. J. Anim. Sci. Biotechnol. 13:69. doi: 10.1186/s40104-022-00714-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yiannikouris, A., and Jouany J. P.. . 2002. Mycotoxins in feeds and their fate in animals: a review. Anim. Res. 51:81–99. doi: 10.1051/animres:2002012. [DOI] [Google Scholar]
- Yiannikouris, A., François J., Poughon L., Dussap C. G., Bertin G., Jeminet G., and Jouany J. P.. . 2004. Adsorption of zearalenone by β-d-Glucans in the Saccharomyces cerevisiae cell wall. J. Food Prot. 67:1195–1200. doi: 10.4315/0362-028x-67.6.1195. [DOI] [PubMed] [Google Scholar]
- Yiannikouris, A., Kettunen H., Apajalahti J., Pennala E., and Moran C. A.. . 2013. Comparison of the sequestering properties of yeast cell wall extract and hydrated sodium calcium aluminosilicate in three in vitro models accounting for the animal physiological bioavailability of zearalenone. Food Addit. Contam. 30:1641–1650. doi: 10.1080/19440049.2013.809625. [DOI] [PubMed] [Google Scholar]
- Yu, J., Hennessy D. A., Tack J., and Wu F.. . 2022. Climate change will increase aflatoxin presence in US Corn. Environ. Res. Lett. 17:054017. doi: 10.1088/1748-9326/ac6435. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.