Abstract
A novel core–shell nanomaterial, ZnO@SiO2, based on rice husk for antibiotic and bacteria removal, was successfully fabricated. The ZnO@SiO2 nanoparticles were characterized by X-ray diffraction (XRD), energy-dispersive X-ray spectroscopy (EDX), Fourier transform infrared (FTIR) spectroscopy, transmission electron microscopy (TEM), photoluminescence spectroscopy (PL), Brunauer–Emmett–Teller (BET) method, diffuse reflectance ultraviolet–vis (DR-UV–vis) spectroscopy, X-ray photoelectron spectroscopy (XPS), Raman spectroscopy, and ζ-potential measurements. β-Lactam antibiotic amoxicillin (AMX) was removed using ZnO@SiO2 nanoparticles with an efficiency greater than 90%, while Escherichia coli removal was higher than 91%. The optimum effective conditions for AMX removal using ZnO@SiO2, including solution pH, adsorption time, and ZnO@SiO2 dosage, were 8, 90 min, and 25 mg/mL, respectively. The maximum adsorption capacity reached 52.1 mg/g, much higher than those for other adsorbents. Adsorption isotherms of AMX on ZnO@SiO2 were more in accordance with the Freundlich model than the Langmuir model. The electrostatic attraction between negative species of AMX and the positively charged ZnO@SiO2 surface induced adsorption, while the removal of E. coli was governed by both electrostatic and hydrophobic interactions. Our study demonstrates that ZnO@SiO2 based on rice husk is a useful core–shell nanomaterial for antibiotic and bacteria removal from water.
1. Introduction
Core–shell nanomaterials have recently attracted intense interest due to the advantages of their surface and internal properties. Metal oxide nanomaterials are widely used for different applications in both chemical engineering and bioresource technology.1 Among various semiconductor metal oxides, ZnO is a well-known material because of its high performance for industrial and pharmaceutical applications.2 ZnO is also an effective material in photocatalysis and adsorption.3 Another advantage of ZnO is its antibacterial efficacy.4,5 Previously, nanostructured ZnO with high photocatalytic performance was synthesized and applied for the degradation of emerging pollutants.3,4,6,7 ZnO nanoparticles with high charge density were also used for the adsorptive removal of organic pollutants.8,9
Nevertheless, ZnO has some limitations, such as low photocatalytic activity under visible illumination due to its high band-gap energy.10 Also, ZnO is not a very stable adsorbent at low pH, although it has a high point of zero charge.11 To improve interfacial characteristics and stability of ZnO in acidic media, recent work has used silica to create composite, coated, or core–shell materials known as ZnO@SiO2.3,12,13
ZnO@SiO2 was synthesized by different techniques such as chemical precipitation,12,14 sol–gel formation,13 and sonication combined with dispersion.15 Among them, sol–gel is the simplest method to synthesize ZnO@SiO2 without using surfactants or organic solvents. Since nanosilica is easily fabricated from rice husk, a common agricultural byproduct,16−19 ZnO@SiO2 based on rice husk is evidently a low-cost adsorbent.
Antibiotics are serious organic pollutants because their residue can provide selection pressure for antibiotic resistance genes in bacteria.20−24 Among various techniques for antibiotic remediation, nanomaterial and nanocrystal adsorption and photocatalysis have high performance that can be used for the removal of both antibiotics and bacteria.25−30 Antibacterial activity of a ZnO hybrid indicated the highest biodegradation rate after 80 days.31 Amoxicillin (AMX) belongs to the β-lactam-containing penicillin family that is widely used to treat various bacterial infections. However, excessive use has led to bacterial strains becoming highly resistant to AMX.32,33 For an antibacterial study, various strains of Escherichia coli are often used as test bacteria.34−36 To our best knowledge, the removal of AMX and E. coli using ZnO@SiO2 based on rice husk has not been previously investigated.
This is the first study on the fabrication of a new core–shell adsorbent ZnO@SiO2 based on rice husk for the removal of AMX and E. coli. ZnO@SiO2 nanomaterials were thoroughly characterized by various physicochemical methods and then applied as a novel adsorbent to investigate AMX and E. coli removal under different experimental conditions. Our study demonstrates that core–shell nanomaterial ZnO@SiO2 is an excellent material for removing both AMX and E. coli.
2. Materials and Methods
2.1. Materials
Nanosilica was fabricated based on rice husk by a simple acid digestion and thermal process.16 Amoxicillin trihydrate (AMX), with purity >98% for high-performance liquid chromatography (HPLC), was delivered from AK Scientific (USA). Zinc acetate dihydrate (Zn(CH3COO)2·2H2O) (purity ≥99.0%) was purchased from Scharlau, Spain. Glass electrode pH measurements were conducted with a pH meter (HI 2215, Hanna). Other chemicals were of analytical grade (p.A, Merck). Ultrapure water was produced by Thermo Scientific Barnstead MicroPure with a resistivity of 18.2 MΩ·cm.
2.2. Fabrication and Characterization
Nanosilica rice husk was easily fabricated by following the procedure in our previous paper,16 while ZnO@SiO2 was synthesized by the sol–gel method with minor modification.13 To synthesize ZnO@SiO2 core–shell nanomaterials, 5.4880 g of Zn(CH3COO)2 was dissolved with stirring in 100 mL of anhydrous ethanol at 80 °C. Overall, 0.6000 g of nanosilica rice husk was added to the solution and heated at 80 °C for 1 h. A NaOH solution containing 2 g of a NaOH solid and 50 mL of anhydrous ethanol was added with 100 mL of n-hexane at room temperature to the above mixture. A white gel was obtained after 24 h of refrigeration, from which a solid was obtained by centrifugation and drying at 100 °C. ZnO@SiO2 was then prepared by calcination at 400 °C over 3 h. Finally, ZnO@SiO2 was cooled to room temperature before keeping the material in a polyethylene container. Figure 1 shows photos of ZnO@SiO2 before and after calcination.
Figure 1.
Photos of synthesized ZnO@SiO2 based on rice husk before calcination (A) and after calcination (B). (Photograph courtesy of Tien-Duc Pham. Copyright 2022).
The phase structure of synthesized ZnO@SiO2 was confirmed by X-ray diffraction (Bruker D8 Advance Diffractometer). Elemental composition was determined by energy-dispersive X-ray spectroscopy, energy-dispersive X-ray spectroscopy (EDX) (Oxford Instrument). Active vibration groups were determined by Fourier transform infrared (FTIR) spectroscopy using an Affinity-1S spectrophotometer (Shimadzu, Japan) from 400 to 4000 cm–1 with a resolution of 4 cm–1. The specific surface area of ZnO@SiO2 was determined by the Brunauer–Emmett–Teller (BET) method using a surface area analyzer (NOVA touch 4LX). Particle size distribution and morphology of ZnO@SiO2 were determined by transmission electron microscopy (TEM) using a JEM-2100, Jeol, Japan. Diffuse reflectance ultraviolet–vis (DR-UV–vis) spectra of the material were measured at RT using a Cary 5000 spectrometer (Varian) equipped with a diffuse reflectance accessory (praying mantis, Harrick). The spectra were collected in the range of 200–800 nm using BaSO4 as a reference material and converted into the Kubelka–Munk function F(R). Surface elemental composition was determined by X-ray photoelectron spectroscopy (XPS) using a Thermo Electron Escalab 250 using a monochromated Al Kα radiation (1486.6 eV) and a photoelectron take-off angle of 90°. Survey and high-resolution spectra energy were 100 and 20 eV, respectively. A Thermo Electron software Avantage was used for the curve fitting of the XPS spectra. Before deconvolution, all peaks were corrected by Shirley’s background. Raman spectra were recorded with a Horiba Jobin Yvon LabRam micro-spectrometer iHR 550 using a 473 nm laser. A diffraction grating of 1800 mm–1 and an entrance slit in the spectrometer of 10 mm was used. Spectra were acquired at different points of the sample to check for sample homogeneity, using a laser power on the samples of 0.1 to 10 mW with a power density of 2.8 × 105 W·cm–2. Data analysis was performed by LabSpec 6, Jobin Yvon Horiba, built-in software.
Total organic carbon was determined by an organic carbon analyzer (TOC-VCSN, Shimadzu, Japan). Materials before and after AMX adsorption were evaluated by TOC measurements.
After AMX adsorption, TOC concentrations decreased compared with the initial solution (not shown in detail). It implies that a high amount of AMX occurred on the ZnO@SiO2 surface.
The surface charge behavior of ZnO@SiO2 nanoparticles using a background electrolyte of 1 mM KCl was measured by electrophoretic mobility using Smoluchowski’s equation37 with a Zetasizer Nano ZS (Malvern).
2.3. Adsorption Experiments
All batch adsorption experiments were conducted using 15 mL Falcon tubes at a room temperature of 25 ± 2 °C. Optimization studies for AMX removal used 10 mg/L antibiotics with various values of pH, contact time, and adsorbent dosage. The AMX and ZnO@SiO2 nanoparticles were mixed by an orbital shaker OS-350D (Digisystem laboratory, Taiwan). Then, the supernatants were separated by centrifuging at 12,000 rpm at 10 °C for 15 min using a refrigerated centrifuge (MR23i, JOUAN, France). After that, AMX concentrations were determined using an ultraviolet–visible (UV–vis) spectrophotometer (UV-1650 PC, Shimadzu, Japan) at a wavelength of 228 nm.19
The removal efficiency of AMX (% R) was calculated by eq 1
| 1 |
where Ci (mg/L) and Ce (mg/L) denote the concentrations of AMX initially and at equilibria, respectively.
The AMX adsorption capacity Γ (mg/g) on ZnO@SiO2 using adsorbent dosage m (g/L) was determined by eq 2
| 2 |
All adsorption experiments were carried out at least twice.
2.4. Adsorption Isotherms
The adsorption isotherms of AMX on ZnO@SiO2 were fitted by Langmuir and Freundlich models.
The linear Langmuir’s equation is shown in eq 3(38)
| 3 |
where KL (L/g) is the Langmuir constant and qmax (mg/g) and qe (mg/g) are the maximum adsorption capacity and the equilibrium adsorption amount of AMX ZnO@SiO2, respectively.
The linear Freundlich isotherm is given in eq 4(39)
| 4 |
where KF (mgn–1Ln/g) is the Freundlich constant and 1/nF is the adsorption intensity.
2.5. Antibacterial Study
The potential for the antibacterial activity of ZnO@SiO2 against E. coli was studied in sterile media under fixed experimental conditions according to a published method.34 ZnO@SiO2 was added to the bacterial strain and incubated. The growth of E. coli was estimated with and without an adsorbent by counting the number of colony-forming units (CFU). The minimum inhibitory concentration (MIC) was used to evaluate adsorbent antibacterial activity. E. coli was dispersed in 0.9% NaCl at 37 °C. The broth medium containing E. coli was centrifuged at 6000 rpm (4 °C) for 10 min. Different volumes of 50 mg/mL ZnO@SiO2 were taken and agitated at 300 rpm at 37 °C to find the optimum dosage. The number of E. coli was counted using the spread plate technique.34 After a contact time of 30 min, an accurate amount of bacteria culture (1 mL) was transferred from the flask and appropriately diluted. Subsequently, a drop of the diluted suspension was put onto agar plates and incubated at 37 °C for 16 h. The CFU was then calculated with the appropriate dilution factor and indicated as CFU mL–1.
3. Results and Discussion
3.1. Characterizations of Synthesized ZnO@SiO2 Based on Rice Husk
ZnO@SiO2 based on rice husk was characterized by X-ray diffraction (XRD), FTIR, scanning electron microscopy (SEM), EDX, BET, XPS, and ζ-potential measurements.
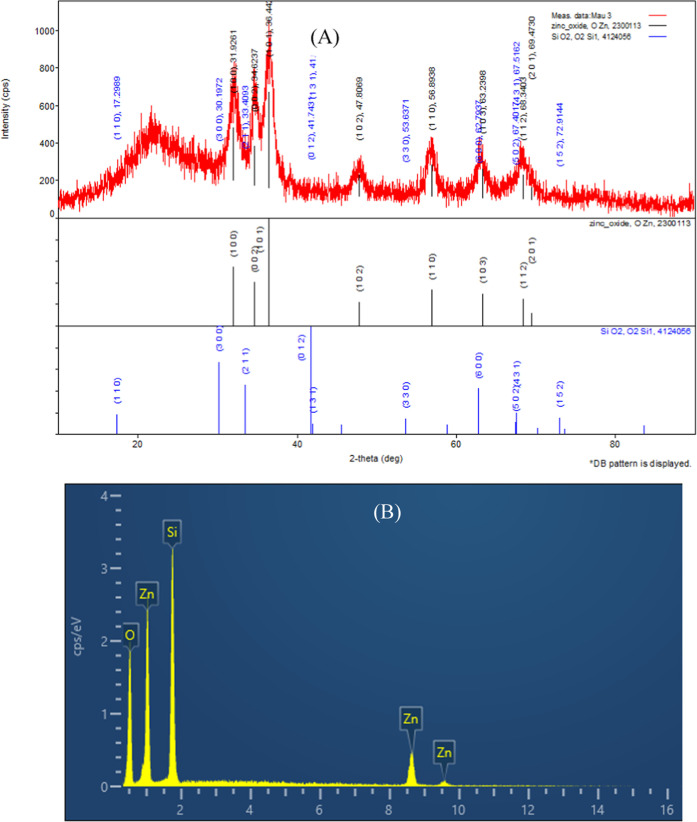
Figure 2A shows that a large broad peak near 22° indicated amorphous SiO2,19 while the peaks appearing at 31.9, 34.6, 47.8, 56.9, 62.2, and 68.3 were assigned to the ZnO crystal phase. All peaks for ZnO were in good agreement with standard patterns (JCPDS 36-1451) without the impurities.13
Figure 2.
XRD pattern (A) and the EDX spectrum (B) of synthesized ZnO@SiO2.
The EDX spectrum in Figure 2B shows that the ZnO@SiO2 material contained Zn, O, and Si with weight percentages of 34.3, 27.5, and 38.2%, respectively, indicating that a significant amount of ZnO was present when using nanosilica rice husk.
Figure 3A reveals the sharp peaks at 797 and 463 cm–1, which are assigned to the asymmetric and bending vibrations of Si–O–Si,18 which are similar to SiO2 rice husk. A large boarding peak at 3470 cm–1 shows the −OH group. The FTIR spectra in Figure 3 also indicate that a very strong symmetric vibration shifted to a longer wavenumber, to 1104 cm–1, while ZnO nanomaterials do not contain the wavenumber of 1104 cm–1. It indicates that ZnO was coated on the SiO2 shell.14
Figure 3.
FTIR spectra (A), TEM image (B), and photoluminescence spectra (C) of the synthesized ZnO@SiO2 material.
The TEM image of ZnO@SiO2 with a resolution of 100 nm shown in Figure 3B indicates that the materials are spherical particles with an average particle size of 30 ± 5 nm. This implies that synthesized ZnO@SiO2 is a nanomaterial.
The photoluminescence (PL) spectra in Figure 3C show that the emission peaks at 445 nm with an excitation wavelength of 350 nm appeared because electrons (radical anions) and holes (radical cations) directly accumulated at the interfaces of ZnO@SiO2. The carriers occurred at the material interface due to the average energy barriers for both hole injection and electron injection.
The adsorption and desorption isotherms of N2 on ZnO@SiO2 nanomaterials are indicated in Figure 4A. Based on BET calculations, the specific surface area of ZnO@SiO2 was found to be 29.6 m2/g, slightly smaller than the material synthesized by the precipitation method.12 In addition, ZnO@SiO2 is nonporous, with a pore volume of 0.162 cc/g as, calculated by the BET method.40
Figure 4.
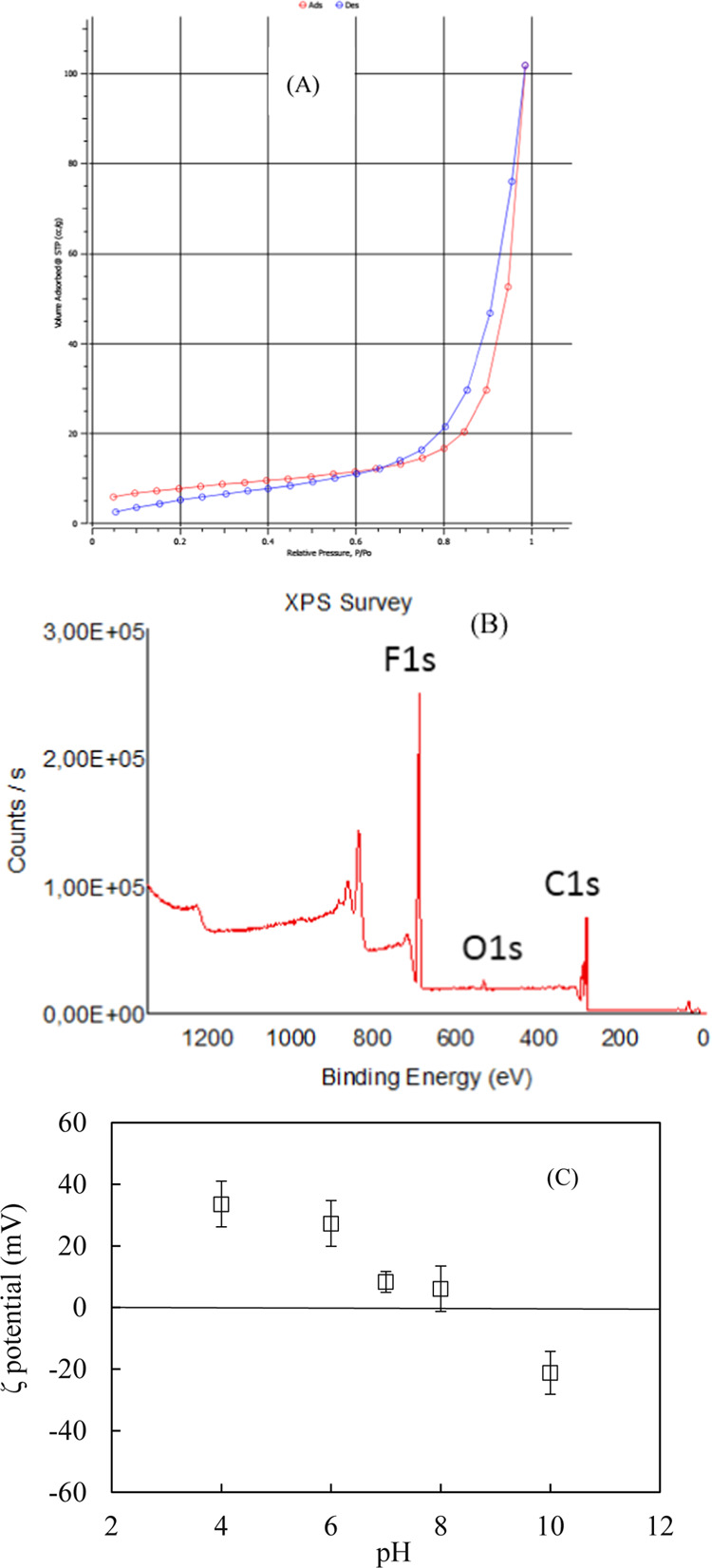
Adsorption–desorption of N2 on ZnO@SiO2 nanoparticles (A). The X-ray photoelectron spectra of ZnO@SiO2 nanoparticles (B). The ζ-potential of ZnO@SiO2 nanoparticles as a function of pH at 1 mM KCl (C).
The XPS analysis (survey spectrum) in Figure 4B indicates the presence of F, O, and C atoms since there is fluoride in the materials due to the F auger peak masking. The XPS analysis also shows no substantial elements in the structure of the ZnO@SiO2 material.
The ZnO@SiO2 surface charge as a function of pH in 1 mM KCl is shown in Figure 4C. The isoelectric point (IEP) of ZnO@SiO2 was about 8.7. The IEP of ZnO@SiO2, in this case, agreed well with previously published reports.1 A very high IEP is helpful for the removal of negative antibiotic AMX species and Gram-negative bacteria E. coli.
3.2. Adsorptive Removal of AMX Using Core–Shell ZnO@SiO2 Nanoparticles
Three effective parameters, including solution pH, contact time, and ZnO@SiO2 dosage, were investigated to find optimum parameters for AMX removal using core–shell ZnO@SiO2 nanoparticles.
Figure 5A shows that AMX removal using ZnO@SiO2 increased with increasing pH from 4.0 to 8.0, followed by trend reversal in the pH range of 8.0–11.0. At pH >7.5, AMX has a negative charge due to pH > pKa2.27 Since the IEP of ZnO@SiO2 is 8.7, a positive adsorbent surface charge is evident at pH <8.7. In other words, AMX adsorption on ZnO@SiO2 is influenced by the electrostatic interaction between the negative AMX charge and positive ZnO@SiO2 surface charge. It should be noted that AMX removal, in this case, is much higher than with SiO2 rice husk alone,19 indicating that the role of ZnO as a shell is of great importance. Since the highest AMX removal was achieved at pH 8.0, we chose pH 8.0 for further studies.
Figure 5.
Effect of pH (A), contact time (B), and adsorbent dosage (C) on AMX adsorption (removal) using ZnO@SiO2 (Ci, AMX 10 mg/L). Error bars show the measurement range of two replicates.
Contact time influences the equilibration process that significantly affects AMX removal. AMX removal using 10 mg/mL ZnO@SiO2 was carried out at pH 8 with 1 mM KCl while contact time ranged from 5 to 180 min. Figure 5B indicates that AMX removal improved dramatically when contact time increased from 0 to 90 min. A further increase in contact time did not induce significant additional AMX removal. This indicates that adsorption reached equilibrium at 90 min. The contact time in our case is faster than AMX adsorption on bentonite, which equilibrates in 400 min.41 Thus, we kept contact time of 90 min for AMX adsorption using ZnO@SiO2.
The optimum dosage of ZnO@SiO2 is another important factor influenced by net material charge density as well as the specific surface area. For AMX adsorption, the total charge density is more important because almost all antibiotics are charged molecules. As can be seen in Figure 5C, AMX removal using ZnO@SiO2 grew sharply when increasing the adsorbent from 1 to 25 mg/mL. Although the adsorbent dosage for antibiotic removal using modified nanosilica rice husk was lower in our case,16−18 adsorption capacity for AMX was much higher than using rice husk without nanosilica as well as the ZnO composite. Therefore, the optimum ZnO@SiO2 dosage for AMX removal was 25 mg/mL.
3.3. Adsorption Isotherms of AMX on ZnO@SiO2
Adsorption isotherms of AMX on ZnO@SiO2 were determined at different KCl concentrations to evaluate the effect of ionic strength. The AMX concentrations were in the range of 2.5–1000 mg/L. The linear fits and parameters for AMX adsorption isotherms on ZnO@SiO2 as fitted to Langmuir and Freundlich models are shown in Table 1.
Table 1. Parameters for Amoxicillin (AMX) Adsorption Isotherms on ZnO@SiO2 at Different Ionic Strengths Fitted by Langmuir and Freundlich Models.
| KCl concentration
(mM) |
|||
|---|---|---|---|
| model | parameters | 0.1 | 10 |
| langmuir | qmax(mg/g) | 52.1 | 34.9 |
| KL(L/g) | 0.0020 | 0.0019 | |
| R2 | 0.6453 | 0.6854 | |
| freundlich | KF(mgn–1Ln/g) | 0.413 | 0.437 |
| nF | 1.10 | 1.24 | |
| R2 | 0.9967 | 0.9763 | |
As can be seen in Table 1, the Freundlich model fits the data well, while the Langmuir model poorly correlates with our data. As nF is greater than 1, AMX can apparently form multilayers on the ZnO@SiO2 surface.
Table 1 also indicates that maximum adsorption capacity decreased by a factor of 4.5 when increasing the KCl concentration from 0.1 to 10 mM. This trend is due to a decrease in electrostatic interaction between anionic AMX and the positively charged ZnO@SiO2 surface. A maximum adsorption capacity of 52 mg/g is much higher than common adsorbents such as activated carbon,42 demonstrating that ZnO@SiO2 is a useful material for AMX removal from an aqueous solution. The maximum adsorption capacity of AMX using ZnO-NPs is only 37 mg/g, which is lower than ZnO@SiO2 when using the same conditions. It implies that ZnO@SiO2 is more effective for AMX removal than ZnO-NPs.
3.4. Adsorption Mechanism of AMX on ZnO@SiO2
In this section, the adsorption mechanism of AMX on ZnO@SiO2 is proposed based on the change in binding energy, TOC measurements, Raman spectroscopy, and the change in the surface charge monitored by ζ-potential measurement and the AMX adsorption isotherms on ZnO@SiO2. After AMX adsorption, TOC concentrations decreased compared with the initial solution (not shown in detail). It implies that a high amount of AMX occurred on the ZnO@SiO2 surface.
We also compare with standard TiO2 nanoparticles to emphasize the advantages of ZnO@SiO2.
The DR-UV–vis spectra of different nanomaterials shown in Figure 6 have strong absorption spectra from 200 to 400 nm. The intensity of ZnO@SiO2 is higher than TiO2 due to the difference in photo energies. The band-gap energies (Egs) of ZnO@SiO2 and TiO2 were found to be 3.15 and 3.06 eV, respectively. The differences in Eg are due to strong sp–d exchange interaction between the band electrons of ZnO and TiO2.43 Interestingly, after AMX adsorption, the characteristics of ZnO@SiO2 changed insignificantly, indicating that adsorption is dominant. Raman spectra of ZnO@SiO2 could not be obtained, so the photocatalyst is rather low compared with the TiO2 material.
Figure 6.
DR-UV–vis spectra of TiO2 and ZnO@SiO2 before and after AMX adsorption.
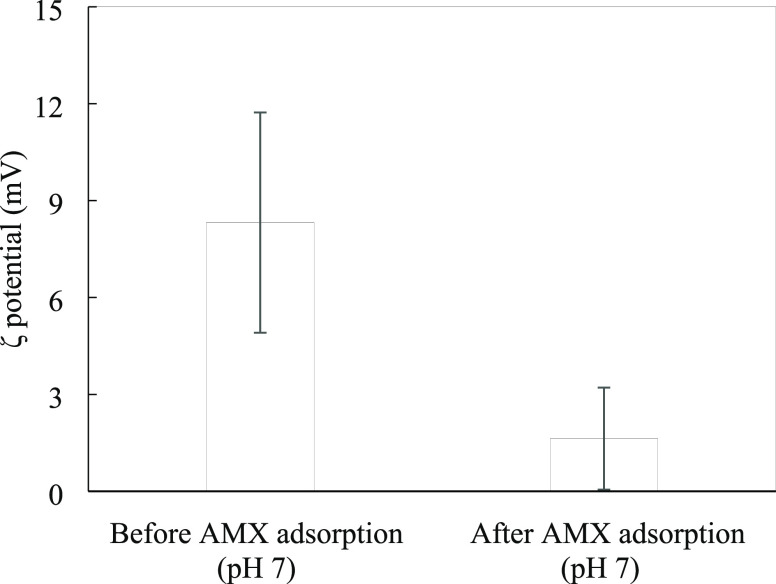
The charging behavior of various adsorbents during antibiotic adsorption has been evaluated by ζ-potential measurements,17−21,28 but the change in the surface charge of ZnO@SiO2 before and after AMX adsorption has not previously been evaluated. Figure 7 shows that the ζ-potential of ZnO@SiO2 using 1 mM KCl at pH 7 was positive at 8.32 mV. However, after adsorption with the anionic form of AMX (at pH 8.0 > pKa2), a significant decrease was observed as the ζ-potential was reduced to 1.63 mV. This result agreed well with the pH influence on AMX removal (Section 3.2). The surface charge change was in good agreement with adsorption isotherms and DR-UV–vis results. Thus, we confirm that the electrostatic attraction between the negative AMX species and the positively charged ZnO@SiO2 surface mainly governs adsorption.
Figure 7.
ζ-potential of ZnO@SiO2 nanoparticles before and after AMX adsorption at 1 mM KCl (pH 7.0).
3.5. Stability and Comparison of ZnO@SiO2 and Other Adsorbents for AMX Removal
Section 3.3 indicates that the maximum adsorption capacity reached 52 mg/g, while the removal efficiency of AMX using ZnO@SiO2 was about 90%. This adsorption process has previously been investigated. Table 2 shows that the adsorption capacity of AMX using ZnO@SiO2 is the highest compared with other adsorbents. These results again show that ZnO@SiO2 is an excellent adsorbent for AMX removal from an aqueous solution.
Table 2. Adsorption Capacity and the Removal Efficiency of Amoxicillin (AMX) Using ZnO@SiO2 Based on Rice Husk and Other Absorbentsa.
| adsorbent | adsorption capacity (mg/g) | removal efficiency (%) | refs |
|---|---|---|---|
| activated carbon | 7.4 | 50 | (42) |
| nanosilica with surface modification by a polyelectrolyte | 7.5 | 92 | (44) |
| chitosan beads | 8.7 | NF | (25) |
| bentonite | 18.8 | 88 | (41) |
| ZnO@SiO2-based rice husk | 52.1 | 90 | this study |
NF: no information.
To demonstrate the stability of ZnO@SiO2, the reuse potential of the material was conducted. Figure 8 shows that the AMX removal was slightly decreased, but the removal was still higher than 74%. It implies that ZnO@SiO2 is a reusable material with high performance.
Figure 8.
Reuse potential of ZnO@SiO2 nanoparticles after three regeneration.
3.6. Removal of E. coli Bacteria Using ZnO@SiO2
To demonstrate bacterial removal efficiency, E. coli growth was compared among 0.9% NaCl, ZnO, and a medium containing ZnO@SiO2.
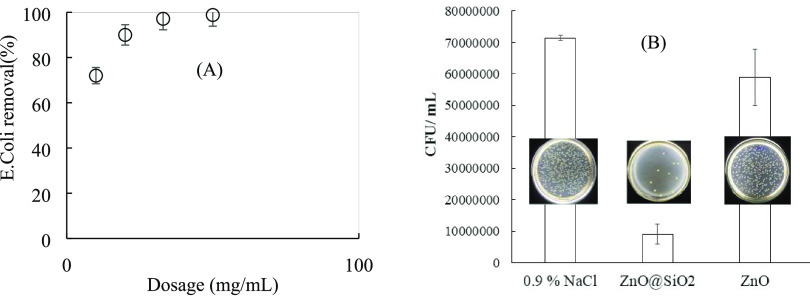
Figure 9A shows the effect of the ZnO@SiO2 dosage on the removal of E. coli. It implies that E. coli removal increased with increasing ZnO@SiO2 concentration. The number of E. coli colonies is reduced by greater than 90% when the ZnO@SiO2 dosage is higher than 20 mg/mL. Figure 9B shows that the antibacterial activity of ZnO@SiO2 nanoparticles was much higher than ZnO and 0.9% NaCl. ZnO@SiO2 nanoparticles reduced the number of E. coli colonies by about 91%, while only 20% E. coli colonies were decreased using ZnO. As can be seen in Figure 9B, the agar plate using ZnO@SiO2 had a much lower MIC than that using ZnO or 0.9% NaCl. The removal efficiency by preventing the growth of E. coli could be controlled by electrostatic and nonelectrostatic interactions.45 Nonelectrostatic, hydrophobic interactions may increase the growth of E. coli cells.46 The positive charge of ZnO@SiO2 at neutral pH had a strong interaction with the negatively charged E. coli cytoplasmic membrane that increased the removal efficiency. It again implies the important role of producing nanoshell ZnO on the surface of the nanocore SiO2 to form a high-performance core–shell nanomaterial, ZnO@SiO2, based on rice husk for antibacterial activity.
Figure 9.
Test results of antibacterial activities for E. coli. The effect of the dosage (A) using ZnO@SiO2 nanoparticles. The comparative antibacterial activities using ZnO and ZnO@SiO2 (B). (Photograph courtesy of Thi-Thuy-Trang Truong. Copyright 2022).
4. Conclusions
We studied the adsorptive removal of antibiotic AMX and bacteria E. coli using synthesized core–shell ZnO@SiO2 nanoparticles based on rice husk. The ZnO@SiO2 nanoparticles were examined by XRD, EDX, FTIR, TEM, BET, XPS, DR-UV–vis, and ζ-potential measurements. Optimum conditions for AMX removal using ZnO@SiO2 were found to be pH 8, an adsorption time of 90 min, and an adsorbent dosage of 25 mg/mL. Under optimum conditions, the removal of antibiotic amoxicillin (AMX) using ZnO@SiO2 nanoparticles was about 90%, and a very high adsorption capacity of 52.1 mg/g was achieved. The Freundlich isotherm better fits AMX behavior on ZnO@SiO2 than does the Langmuir model. E. coli growth suppression was greater than 91%, higher than other adsorbents. Adsorption of AMX on ZnO@SiO2 is governed by electrostatics, whereas the adsorptive removal of E. coli is controlled by both electrostatic and hydrophobic interactions.
Acknowledgments
The authors would like to thank Dr. Thanh Huyen Vuong at Leibniz-Institut für Katalyse e.V. (LIKAT Rostock) for DR-UV–vis measurements. They are grateful to Prof. Alexander Scheeline, University of Illinois at Urbana-Champaign, U.S.A., for critical reading to improve this manuscript’s English.
Glossary
Abbreviations
- XRD
X-ray diffraction
- EDS
energy-dispersive X-ray spectroscopy
- FTIR
Fourier transform infrared
- UV–vis
ultraviolet–visible
- TEM
transmission electron microscopy
- BET
Brunauer–Emmett–Teller
- AMX
amoxicillin
- E. coli
Escherichia coli
- DR-UV–vis
diffuse reflectance UV–vis
- PL
photoluminescence spectroscopy
- XPS
X-ray photoelectron spectroscopy
Author Contributions
Conceptualization was given by T.-D.P.; methodology was contributed by T.-D.P.; software was given by T.-T.-T.T. and V.-P.B.; validation was performed by T.-D.P. and T.-T.-T.T.; formal analysis was done by T.-T.-T.T., H.-L.N., L.-B.-L.H., V.-P.B., T.-T.-M.T., and T.-D.D.; investigation was performed by T.-D.P. and T.-T.-T.T.; resources were contributed by T.-T.-T.T., H.-L.N., L.-B.-L.H., V.-P.B., and T.-T.-M.T.; data curation was done by T.-T.-T.T., H.-L.N., L.-B.-L.H., V.-P.B., T.-T.-My.T., T.-D.D., and T.-D.L.; writing—original draft preparation was done by T.-D.P. and T.-T.-T.T.; writing—review and editing was done by T.-D.P.; visualization was performed by T.-D.P., T.-T.-T.T., T.-D.D., and T.-D.L.; supervision was done by T.-D.P.; and project administration was done by T.-D.P.. All authors reviewed and approved the manuscript.
The authors declare no competing financial interest.
References
- Kosmulski M. The pH dependent surface charging and points of zero charge. VIII. Update. Adv. Colloid Interface Sci. 2020, 275, 102064 10.1016/j.cis.2019.102064. [DOI] [PubMed] [Google Scholar]
- Truong T. T.; Pham T. T.; Truong T. T. T.; Pham T. D. Synthesis, characterization of novel ZnO/CuO nanoparticles, and the applications in photocatalytic performance for rhodamine B dye degradation. Environ. Sci. Pollut. Res. 2022, 29, 22576–22588. 10.1007/s11356-021-17106-0. [DOI] [PubMed] [Google Scholar]
- Saravanan R.; Gupta V. K.; Narayanan V.; Stephen A. Comparative study on photocatalytic activity of ZnO prepared by different methods. J. Mol. Liq. 2013, 181, 133–141. 10.1016/j.molliq.2013.02.023. [DOI] [Google Scholar]
- Wang X.; Yang F.; Zhao J.; Xu Y.; Mao D.; Zhu X.; Luo Y.; Alvarez P. J. J. Bacterial exposure to ZnO nanoparticles facilitates horizontal transfer of antibiotic resistance genes. NanoImpact 2018, 10, 61–67. 10.1016/j.impact.2017.11.006. [DOI] [Google Scholar]
- La D. D.; Nguyen-Tri P.; Le K. H.; Nguyen P. T. M.; Nguyen M. D.-B.; Vo A. T. K.; Nguyen M. T. H.; Chang S. W.; Tran L. D.; Chung W. J.; Nguyen D. D. Effects of antibacterial ZnO nanoparticles on the performance of a chitosan/gum arabic edible coating for post-harvest banana preservation. Prog. Org. Coat. 2021, 151, 106057 10.1016/j.porgcoat.2020.106057. [DOI] [Google Scholar]
- Chen Y.-r.; Guo X.-p.; Feng J.-n.; Lu D.-p.; Niu Z.-s.; Tou F.-y.; Hou L.-j.; Liu M.; Yang Y. Impact of ZnO nanoparticles on the antibiotic resistance genes (ARGs) in estuarine water: ARG variations and their association with the microbial community. Environ. Sci.: Nano 2019, 6, 2405–2419. 10.1039/C9EN00338J. [DOI] [Google Scholar]
- Saravanan R.; Karthikeyan S.; Gupta V. K.; Sekaran G.; Narayanan V.; Stephen A. Enhanced photocatalytic activity of ZnO/CuO nanocomposite for the degradation of textile dye on visible light illumination. Mater. Sci. Eng., C 2013, 33, 91–98. 10.1016/j.msec.2012.08.011. [DOI] [PubMed] [Google Scholar]
- Chaudhary S.; Kaur Y.; Umar A.; Chaudhary G. R. Ionic liquid and surfactant functionalized ZnO nanoadsorbent for Recyclable Proficient Adsorption of toxic dyes from waste water. J. Mol. Liq. 2016, 224, 1294–1304. 10.1016/j.molliq.2016.10.116. [DOI] [Google Scholar]
- Shengcong L.; Hanning X.; Yuping L. Adsorption of polyelectrolyte on the surface of ZnO nanoparticles and the stability of colloidal dispersions. Chin. Sci. Bull. 2005, 50, 1570–1575. 10.1360/982004-575. [DOI] [Google Scholar]
- Lekota M. W.; Dimpe K. M.; Nomngongo P. N. MgO-ZnO/carbon nanofiber nanocomposite as an adsorbent for ultrasound-assisted dispersive solid-phase microextraction of carbamazepine from wastewater prior to high-performance liquid chromatographic detection. J. Anal. Sci. Technol. 2019, 10, 25 10.1186/s40543-019-0185-1. [DOI] [Google Scholar]
- Degen A.; Kosec M. Influence of pH and Ionic Impurities on the Adsorption of Poly(acrylic) Dispersant onto a Zinc Oxide Surface. J. Am. Ceram. Soc. 2003, 86, 2001–2010. 10.1111/j.1151-2916.2003.tb03600.x. [DOI] [Google Scholar]
- Justine M.; Prabu H. J.; Johnson I.; Raj D. M. A.; Sundaram S. J.; Kaviyarasu K. Synthesis and characterizations studies of ZnO and ZnO-SiO2 nanocomposite for biodiesel applications. Mater. Today: Proc. 2021, 36, 440–446. 10.1016/j.matpr.2020.05.034. [DOI] [Google Scholar]
- Xu H.; Yu T.; Liu Y. Characteristics and Performance of Nanozinc Oxide/Mesoporous Silica Gel Photocatalytic Composite Prepared by a Sol-Gel Method. J. Nanomater. 2015, 2015, 1–5. 10.1155/2015/154865. [DOI] [Google Scholar]
- Xia H.-L.; Tang F.-Q. Surface Synthesis of Zinc Oxide Nanoparticles on Silica Spheres: Preparation and Characterization. J. Phys. Chem. B 2003, 107, 9175–9178. 10.1021/jp0261511. [DOI] [Google Scholar]
- Zhang J.; Yan X.; Hu M.; Hu X.; Zhou M. Adsorption of Congo red from aqueous solution using ZnO-modified SiO2 nanospheres with rough surfaces. J. Mol. Liq. 2018, 249, 772–778. 10.1016/j.molliq.2017.11.109. [DOI] [Google Scholar]
- Pham T. D.; Bui T. T.; Truong T. T. T.; Hoang T. H.; Le T. S.; Duong V. D.; Yamaguchi A.; Kobayashi M.; Adachi Y. Adsorption characteristics of beta-lactam cefixime onto nanosilica fabricated from rice HUSK with surface modification by polyelectrolyte. J. Mol. Liq. 2020, 298, 111981 10.1016/j.molliq.2019.111981. [DOI] [Google Scholar]
- Pham T. D.; Vu T. N.; Nguyen H. L.; Le P. H. P.; Hoang T. S. Adsorptive Removal of Antibiotic Ciprofloxacin from Aqueous Solution Using Protein-Modified Nanosilica. Polymers 2020, 12, 57 10.3390/polym12010057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Truong T. T. T.; Vu T. N.; Dinh T. D.; Pham T. T.; Nguyen T. A. H.; Nguyen M. H.; Nguyen T. D.; Yusa S.-i.; Pham T. D. Adsorptive removal of cefixime using a novel adsorbent based on synthesized polycation coated nanosilica rice husk. Prog. Org. Coat. 2021, 158, 106361 10.1016/j.porgcoat.2021.106361. [DOI] [Google Scholar]
- Pham T.-D.; Le T.-M.-A.; Pham T.-M.-Q.; Dang V.-H.; Vu K.-L.; Tran T.-K.; Hoang T.-H. Synthesis and Characterization of Novel Hybridized CeO2@SiO2 Nanoparticles Based on Rice Husk and Their Application in Antibiotic Removal. Langmuir 2021, 37, 2963–2973. 10.1021/acs.langmuir.0c03632. [DOI] [PubMed] [Google Scholar]
- Pham T. D.; Tran T. T.; Le V. A.; Pham T. T.; Dao T. H.; Le T. S. Adsorption characteristics of molecular oxytetracycline onto alumina particles: The role of surface modification with an anionic surfactant. J. Mol. Liq. 2019, 287, 110900 10.1016/j.molliq.2019.110900. [DOI] [Google Scholar]
- Nguyen N. T.; Dao T. H.; Truong T. T.; Nguyen T. M. T.; Pham T. D. Adsorption characteristic of ciprofloxacin antibiotic onto synthesized alpha alumina nanoparticles with surface modification by polyanion. J. Mol. Liq. 2020, 309, 113150 10.1016/j.molliq.2020.113150. [DOI] [Google Scholar]
- Tran N. H.; Chen H.; Reinhard M.; Mao F.; Gin K. Y.-H. Occurrence and removal of multiple classes of antibiotics and antimicrobial agents in biological wastewater treatment processes. Water Res. 2016, 104, 461–472. 10.1016/j.watres.2016.08.040. [DOI] [PubMed] [Google Scholar]
- Tran N. H.; Hoang L.; Nghiem L. D.; Nguyen N. M. H.; Ngo H. H.; Guo W.; Trinh Q. T.; Mai N. H.; Chen H.; Nguyen D. D.; Ta T. T.; Gin K. Y.-H. Occurrence and risk assessment of multiple classes of antibiotics in urban canals and lakes in Hanoi, Vietnam. Sci. Total Environ. 2019, 692, 157–174. 10.1016/j.scitotenv.2019.07.092. [DOI] [PubMed] [Google Scholar]
- Tran N. H.; Reinhard M.; Khan E.; Chen H.; Nguyen V. T.; Li Y.; Goh S. G.; Nguyen Q. B.; Saeidi N.; Gin K. Y.-H. Emerging contaminants in wastewater, stormwater runoff, and surface water: Application as chemical markers for diffuse sources. Sci. Total Environ. 2019, 676, 252–267. 10.1016/j.scitotenv.2019.04.160. [DOI] [PubMed] [Google Scholar]
- Adriano W. S.; Veredas V.; Santana C. C.; Gonçalves L. R. B. Adsorption of amoxicillin on chitosan beads: Kinetics, equilibrium and validation of finite bath models. Biochem. Eng. J. 2005, 27, 132–137. 10.1016/j.bej.2005.08.010. [DOI] [Google Scholar]
- Saucier C.; Karthickeyan P.; Ranjithkumar V.; Lima E. C.; dos Reis G. S.; de Brum I. A. S. Efficient removal of amoxicillin and paracetamol from aqueous solutions using magnetic activated carbon. Environ. Sci. Pollut. Res. 2017, 24, 5918–5932. 10.1007/s11356-016-8304-7. [DOI] [PubMed] [Google Scholar]
- Zha S. x.; Zhou Y.; Jin X.; Chen Z. The removal of amoxicillin from wastewater using organobentonite. J. Environ. Manage. 2013, 129, 569–576. 10.1016/j.jenvman.2013.08.032. [DOI] [PubMed] [Google Scholar]
- Dao T.-H.; Vu T.-Q.-M.; Nguyen N.-T.; Pham T.-T.; Nguyen T.-L. Yusa, S.-i.; Pham, T.-D., Adsorption Characteristics of Synthesized Polyelectrolytes onto Alumina Nanoparticles and their Application in Antibiotic Removal. Langmuir 2020, 36, 13001–13011. 10.1021/acs.langmuir.0c02352. [DOI] [PubMed] [Google Scholar]
- Dao T. H.; Tran T. T.; Nguyen V. R.; Pham T. N. M.; Vu C. M.; Pham T. D. Removal of antibiotic from aqueous solution using synthesized TiO2 nanoparticles: characteristics and mechanisms. Environ. Earth Sci. 2018, 77, 359 10.1007/s12665-018-7550-z. [DOI] [Google Scholar]
- Chen L.; Yu H.; Deutschman C.; Yang T.; Tam K. C. Novel design of Fe-Cu alloy coated cellulose nanocrystals with strong antibacterial ability and efficient Pb2+ removal. Carbohydr. Polym. 2020, 234, 115889 10.1016/j.carbpol.2020.115889. [DOI] [PubMed] [Google Scholar]
- Lu G.; Zhu J.; Yu H.; Jin M.; Abdalkarim S. Y. H.; Wei Y. Degradation mechanism of green biopolyester nanocomposites with various cellulose nanocrystal based nanohybrids. Cellulose 2021, 28, 7735–7748. 10.1007/s10570-021-04031-1. [DOI] [Google Scholar]
- Chandrasekaran A.; Patra C.; Narayanasamy S.; Subbiah S. Adsorptive removal of Ciprofloxacin and Amoxicillin from single and binary aqueous systems using acid-activated carbon from Prosopis juliflora. Environ. Res. 2020, 188, 109825 10.1016/j.envres.2020.109825. [DOI] [PubMed] [Google Scholar]
- Le T.-H.; Ng C.; Tran N. H.; Chen H.; Gin K. Y.-H. Removal of antibiotic residues, antibiotic resistant bacteria and antibiotic resistance genes in municipal wastewater by membrane bioreactor systems. Water Res. 2018, 145, 498–508. 10.1016/j.watres.2018.08.060. [DOI] [PubMed] [Google Scholar]
- Mpatani F. M.; Aryee A. A.; Han R.; Kani A. N.; Li Z.; Qu L. Green fabrication of a novel cetylpyridinium-bagasse adsorbent for sequestration of micropollutant 2,4-D herbicide in aqueous system and its antibacterial properties against S. aureus and E. coli. J. Environ. Chem. Eng. 2021, 9, 106714 10.1016/j.jece.2021.106714. [DOI] [Google Scholar]
- Pisharody L.; Suresh S.; Mukherji S. Evaluation of adsorbents and eluents for application in virus concentration and adsorption-desorption isotherms for coliphages. Chem. Eng. J. Adv. 2021, 403, 126267 10.1016/j.cej.2020.126267. [DOI] [Google Scholar]
- Nguyen T. V.; Do T. V.; Ha M. H.; Le H. K.; Le T. T.; Nguyen T. N. L.; Dam X. T.; Lu L. T.; Tran D. L.; Vu Q. T.; Dinh D. A.; Dang T. C.; Nguyen -Tri P. Crosslinking process, mechanical and antibacterial properties of UV-curable acrylate/Fe3O4-Ag nanocomposite coating. Prog. Org. Coat. 2020, 139, 105325 10.1016/j.porgcoat.2019.105325. [DOI] [Google Scholar]
- Delgado A. V.; González-Caballero F.; Hunter R. J.; Koopal L. K.; Lyklema J. Measurement and interpretation of electrokinetic phenomena. J. Colloid Interface Sci. 2007, 309, 194–224. 10.1016/j.jcis.2006.12.075. [DOI] [PubMed] [Google Scholar]
- Langmuir I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. 10.1021/ja02242a004. [DOI] [Google Scholar]
- Freundlich H. M. F. Über die adsorption in Lösungen. Z. Phys. Chem. 1906, 57, 385–490. [Google Scholar]
- Barrett E. P.; Joyner L. G.; Halenda P. P. The Determination of Pore Volume and Area Distributions in Porous Substances. I. Computations from Nitrogen Isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. 10.1021/ja01145a126. [DOI] [Google Scholar]
- Putra E. K.; Pranowo R.; Sunarso J.; Indraswati N.; Ismadji S. Performance of activated carbon and bentonite for adsorption of amoxicillin from wastewater: Mechanisms, isotherms and kinetics. Water Res. 2009, 43, 2419–2430. 10.1016/j.watres.2009.02.039. [DOI] [PubMed] [Google Scholar]
- Pouretedal H. R.; Sadegh N. Effective removal of Amoxicillin, Cephalexin, Tetracycline and Penicillin G from aqueous solutions using activated carbon nanoparticles prepared from vine wood. J. Water Process Eng. 2014, 1, 64–73. 10.1016/j.jwpe.2014.03.006. [DOI] [Google Scholar]
- Truong T. T.; Pham T. T.; Truong T. T. T.; Pham T. D. Synthesis, characterization of novel ZnO/CuO nanoparticles, and the applications in photocatalytic performance for rhodamine B dye degradation. Environ. Sci. Pollut. Res. 2022, 29, 22576–22588. 10.1007/s11356-021-17106-0. [DOI] [PubMed] [Google Scholar]
- Pham T. D.; Bui T. T.; Nguyen V. T.; Bui T. K. V.; Tran T. T.; Phan Q. C.; Pham T. D.; Hoang T. H. Adsorption of Polyelectrolyte onto Nanosilica Synthesized from Rice Husk: Characteristics, Mechanisms, and Application for Antibiotic Removal. Polymers 2018, 10, 220 10.3390/polym10020220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shafi Q. I.; Ihsan H.; Hao Y.; Wu X.; Ullah N.; Younas M.; He B.; Rezakazemi M. Multi-ionic electrolytes and E. coli removal from wastewater using chitosan-based in-situ mediated thin film composite nanofiltration membrane. J. Environ. Manage. 2021, 294, 112996 10.1016/j.jenvman.2021.112996. [DOI] [PubMed] [Google Scholar]
- Li Z.; Ma J.; Ruan J.; Zhuang X. Using Positively Charged Magnetic Nanoparticles to Capture Bacteria at Ultralow Concentration. Nanoscale Res. Lett. 2019, 14, 195 10.1186/s11671-019-3005-z. [DOI] [PMC free article] [PubMed] [Google Scholar]