Abstract
Peripheral arterial disease (PAD) has a worldwide prevalence and is a significant cause of cardiovascular morbidity and mortality. Due to its high prevalence and higher rates of ischemic cardiovascular and lower-extremity events, its treatment is essential. Increased levels of oxidative stress cause disease. This review aimed to evaluate different studies of antioxidant treatments for PAD patients. A systematic search for relevant studies was performed on the PubMed, SCOPUS, and ScienceDirect databases, and 18 studies fulfilled the inclusion criteria. In total, 16.6% of the studies used natural antioxidants, and 83.3% used synthetic antioxidants. The reviewed studies show that natural antioxidants were completely effective in treating PAD, and synthetic antioxidants showed effective results in only 53% of the studies. A less-than-optimal pro-oxidant–antioxidant balance does not improve the symptoms of PAD. In conclusion, antioxidants in their natural forms are more effective for PAD patients, and ensuring the optimal pro-oxidant–antioxidant balance is an effective method for managing treatment with antioxidants.
Keywords: peripheral arterial disease, oxidative stress, antioxidant treatment, natural antioxidants, synthetic antioxidants, pro-oxidant–antioxidant balance, intermittent claudication, walking distance
1. Introduction
Peripheral arterial disease (PAD) is a significant cause of cardiovascular morbidity and mortality. An estimated 230 million people are affected worldwide [1]. Despite the high prevalence, PAD remains underrated, underrecognized, and undertreated [2]. Higher rates of ischemic cardiovascular and lower-extremity events, and increased prescription medication, combined with outpatient and inpatient care, can result in an increased burden on healthcare services and add to health expenses [3]. Furthermore, significant amputations can have socio-economic and psychological consequences.
PAD prevalence increases with age and affects 12–14% of the general population. Two main complications in elderly patients with PAD, and also considered to be among the leading causes of morbidity and mortality, are coronary artery disease (CAD) and cerebrovascular disease (CVD) [4]. PAD is recognized to have a greater-than-20% predictive value for coronary heart disease (CHD), mainly manifesting as a significant coronary event in the next ten years. It has been shown in patients over 50 years of age that there is a coexisting incidence of CAD in 68% of patients, while 42% have a chance of having a stroke [4,5].
Although no specific treatment has been developed for the definitive improvement of PAD, supportive therapeutic protocols are routinely used to reduce symptoms in patients with PAD. The treatment provided to a patient with PAD depends on the clinical history and the underlying disease that triggered the PAD [6]. To date, many mechanisms have been proposed for the causes of PAD. One of the most relevant causes is high levels of oxidative stress. Elevated levels of oxidants have been reported in patients with PAD, although there has been some evidence of antioxidant dysfunction in patients.
Furthermore, the balance between oxidative stress and antioxidants can be impaired in PAD [7]. High levels of oxidative stress can lead to endothelial damage, stimulate the formation of atherosclerotic plaques by LDL oxidation, and increase the levels of free radicals [8]. Therefore, some studies on PAD have considered antioxidant therapies in patients with PAD [9]. Different results have been shown for the effect of antioxidants, which can be discussed. This systematic review aimed to collect information about the impact of antioxidants in treating PAD.
2. Materials and Methods
2.1. Literature Search
In this systematic review, recommendations stated in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were followed. The systematic review protocol was registered on PROSPERO (registration ID 345836). A comprehensive search of the PubMed, SCOPUS, and ScienceDirect databases was conducted from 20 September 2020 to 20 December 2021. It was found that the most important sources of antioxidants are (1) in natural and dietary forms and (2) in the form of synthetic and medicinal supplements. Diets containing vitamins E, A, and C, beta-carotene, lycopene, and selenium are the most important sources of natural antioxidants. Additionally, masoprocol, pramipexole, allopurinol, pentoxifylline, melatonin, dimethyl sulfoxide, and acetylcysteine are among the synthetic and medicinal forms. Additionally, a search was performed using relevant keywords or title headings, including ‘peripheral arterial disease’, ‘antioxidant therapy’, ‘supplementation’, ‘therapeutic effect’, and ‘complications’. Full details of the search strategy can be found in Table 1. Finally, reference lists of related review articles were manually searched.
Table 1.
Full details of the search strategy terms.
| Terms 1 | Search Strategy Terms |
|---|---|
| Term 1 | “Antioxidants” OR “Antioxidant treatments” “Antioxidant therapy” |
| Term 2 | “Antioxidants drugs” OR “vitamin c” OR “Ascorbic acid” OR “Vitamin A” OR “Vitamin E” OR “Lipoic acid” OR “Masoprocol” OR “Pramipexole” OR “Nitric Oxide” OR “Allopurinol” OR “Pentoxifylline” OR “Melatonin” OR “Dimethyl sulfoxide” OR “Probucol” OR “Resveratrol” OR “3-hydroxyanthranilic acid” OR Acetylcysteine OR Nicaraven OR Lodoxamide OR Mequinol OR “Hydroquinone” OR “Selenic acid” OR “Selenium” OR “Lycopene” OR “Tocopherol” OR “Rebamipide” OR “Allicin” OR “Anisodamine” OR “Bucillamine” OR “Carvedilol” OR “Pentoxifylline” |
| Term 3 | Consumption OR “Dietary intake” OR Supplement OR Supplementation OR “Nutritional supplement” |
| Term 4 | “Peripheral arterial disease” OR “Peripheral artery disease” OR “PAD” OR “Peripheral vascular disease” OR “PVD”, “Atherosclerosis” OR “Coronary artery disease” OR “CAD” |
1 Term 1, 2, 3, and 4 were joined with ‘AND’.
2.2. Study Selection
The inclusion and exclusion criteria established for the review are indicated in Table 2. For studies to be included in the review, all the studies had to meet all the inclusion criteria. Two authors, SK and HS, separately screened the articles based on the inclusion criteria. Thus, complete reports were received for all studies that were unclear or appeared to meet the inclusion criteria. Additionally, the conflicts that occurred were resolved through analysis until an agreement was reached. When agreement could not be reached, a third reviewer (BF) participated.
Table 2.
Inclusion and exclusion criteria.
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Studies that are randomized controlled trials | Studies including participants at risk of PAD, including otherwise healthy smokers and hypertensive or diabetic patients |
| Studies with a sample size ≤ 10 participants | Studies that administer mixed nutrient supplementation where no group receives any specific antioxidant supplement alone |
| Studies including participants who either are healthy or have established PAD | Studies incorporating dual treatments such as exercise and supplementation |
| Studies that orally administer a single antioxidant intervention through supplementation or dietary or drug interventions | studies on animal models |
3. Data Extraction and Quality Assessment
The reviewers independently extracted and recorded data, including the author and year of publication, population characteristics such as the sample size, antioxidant intervention (supplementation/dietary and medicinal forms), duration of follow-up, status after intervention, and quality control score as determined by the two independent reviewers.
The studies were assessed using a study design and sampling method appropriate for the research, an adequate sample size considering the prevalence of PAD, approved criteria used for the evaluation of the results, unbiased analysis of the outcomes, an adequate response rate, reporting of the statistics with confidence intervals, and detailed descriptions of the study subjects. Finally, the quality assessment showed that the selected studies could be approved in terms of all the mentioned criteria.
4. Results
4.1. Search Results
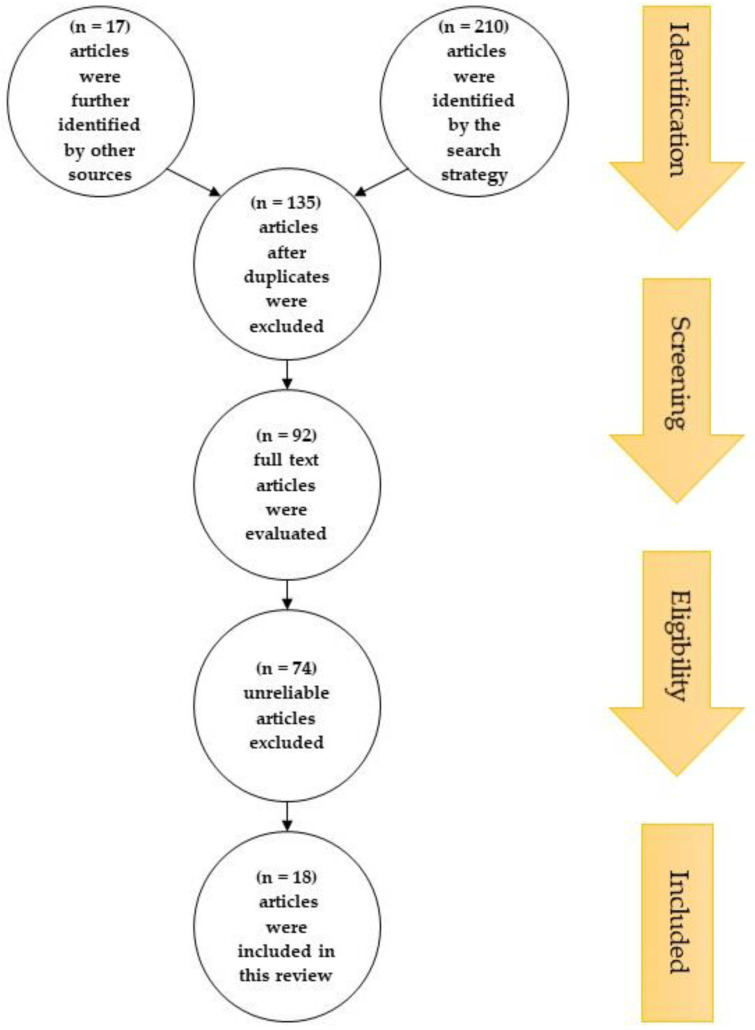
The process of choosing relevant studies is summarized in Figure 1. The search strategy identified 210 articles; additionally, 17 articles were further identified by searching through the reference lists of relevant reviews. After duplicates were excluded, 135 articles were selected by title and abstract for their eligibility. From these, 92 articles were evaluated based on their full texts, with 74 articles excluded due to unreliable study designs, patient populations, interventions, outcome measures, sample sizes smaller than 10, or no full text being available. Consequently, 18 articles were included in this review.
Figure 1.
PRISMA flow diagram showing the study selection and identification. There were 210 articles; additionally, 17 articles were further identified by searching through the reference lists of relevant review. After duplicates were excluded, 135 articles were selected by title and abstract for their eligibility. From these, 92 articles were evaluated by their full texts, with 74 articles excluded due to unreliable data. Consequently, 18 articles were included in this review.
4.2. Study Characteristics
Of these 18 studies, 3 (16.6%) used natural antioxidants in the form of fruits and vegetables [10,11,12]. Fifteen (83.3%) studies used synthetic forms of antioxidants in dietary supplements and medications [13,14,15,16,17,18,19,20,21,22,23,24,25,26,27]. Of the 15 studies with the synthetic forms of antioxidants, 8 (53.3%) studies used dietary supplements [13,14,16,17,18,20,21,22] and 7 (46.6%) studies used medications [15,19,23,24,25,26,27].
Vitamins were the commonest antioxidants used, and six (33.3%) studies used vitamins as antioxidant therapy. Four (22.2%) studies used cocoa and flavonoid compounds [10,11,12,13,14,19,20,21,22,23].
Overall, no relationship between the dose and duration of antioxidant treatment was reported [10,23]. In 11 (61.1%) studies, the results for antioxidant therapy were reported to show it to be significantly effective [10,11,12,17,20,22,23,24,25,26,27], and in 7 (38.8%) studies, no significant differences from the placebo group were observed [13,14,15,16,18,19,21]. The results are summarized in Table 3.
Table 3.
The summarized results of 18 articles that fulfilled the inclusion criteria.
| Study | Year | Sample Size (N) | Age (y) | Gender | Intervention | Form of Intervention (Natural or Synthetic) |
Dose/Day | Duration | Control | Status after Intervention | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Dalgad et al. [10] | 2009 | 48 | Not mentioned | Not mentioned | Orange and blackcurrant juice (vitamin C); orange and blackcurrant juice + vitamin E | Natural: orange and blackcurrant juice | 210 mg | 4 weeks | Sugar-containing reference beverage (0 mg vit c) | There were significant effects in orange and blackcurrant juice-treated patients but not in combination with vitamin E |
| 2 | Klipstein-Grobusch et al. [11] | 2001 | 574 | 55–94 | 204 men and 370 women | Dietary β-carotene, vitamin C, and vitamin E | Natural: 70 food items in 13 food groups | Was not considered due to insufficient accuracy in the recording of the dose and duration | 1 year | There were no controls in this study | In women, vitamin C intake was significantly inversely associated with the risk of PAD and lower ankle–arm systolic blood pressure Index (AAI); in men, inverse associations of PAD and AAI with vitamin E were observed; no association of αtocopherol and β-carotene with intermittent claudication |
| 3 | Woessner et al. [12] | 2018 | 24 | 40–80 | Not mentioned | Beetroot juice | Natural | 70 mL | 12 weeks | Placebo beverage | There was a significant improvement in walking distance |
| 4 | Kleijnen et al. [13] | 1998 | 154 | 50–60 | Men | Vitamin E | Synthetic: dietary supplement | 300–900 mg | 8–10 months | Placebo | There were no significant effects |
| 5 | Catalano et al. [14] | 2007 | 210 | 60–70 | Men | Oral antioxidant vitamins (vitamin E, vitamin C, and beta-carotene) | Synthetic: dietary supplement | 600 mg vitamin E, 250 mg vitamin C, and 20 mg beta-carotene | >20 months | Placebo | There were no significant effects |
| 6 | McDermott [15] | 2017 | 66 | 65 | 45 men and 65 women | Resveratrol | Synthetic medicinal supplement | Group 1: 500 mg (N = 22) Group 2: 125 mg (N = 22) |
6 months | Placebo | There were no significant effects |
| 7 | Jepson et al. [16] | 2013 | 78 | 40–70 | Men and women | Allium sativum (garlic) | Synthetic: dietary supplement | 800 mg | 12 weeks | Placebo | There were no significant effects |
| 8 | Horsch et al. [17] | 2004 | 619 | Not mentioned | Not mentioned | Ginkgo biloba (ginkgo) | Synthetic: dietary supplement | 120–160 mg | 6–24 weeks | Placebo | There was a significant improvement in pain-free walking |
| 9 | Sommerfield et al. [18] | 2007 | 425 | Not mentioned | Not mentioned | Omega-3 fatty acids | Synthetic: dietary supplement | 45 mg to 3 g | 4 months to 2 years | Placebo | There were no significant effects |
| 10 | Curtis et al. [19] | 2013 | 93 | Not mentioned | Not mentioned | Combined isoflavone and flavan-3-ols | Synthetic combination of isoflavone and flavan-3-ols | 100 mg isoflavone; 850 mg flavan-3-ols | 1 year | Placebo | There were no significant effects |
| 11 | Loffredo et al. [20] | 2014 | 20 | 60–70 | 14 men and 6 women | Cocoa | Synthetic dark chocolate (>85% cocoa) | 40 g | 2 h after chocolate ingestion | Milk chocolate (≤35% cocoa) | There was a significant improvement in maximal walking distance serum NOx and decreased serum isoprostanes |
| 12 | Hammer et al. [21] | 2015 | 21 | 60–70 | 17 men and 4 women | Cocoa | Synthetic dark chocolate | 50 g | 2 h after chocolate ingestion | Placebo | There was no significant effect |
| 13 | McDermott et al. [22] | 2020 | 44 | 70–80 | Men | Cocoa | Synthetic cocoa beverage | 15 g | 6 months | Placebo beverage | There was a significant improvement in walking distance |
| 14 | Belch et al. [23] | 2008 | 320 | 40 or more | Men and women | Antioxidant capsule contained α-tocopherol, ascorbic acid, pyridoxine hydrochloride, zinc sulfate, nicotinamide, lecithin, and sodium selenite | Synthetic antioxidant capsule combination | α-tocopherol 200 mg, ascorbic acid 100 mg, pyridoxine hydrochloride 25 mg, zinc sulfate 10 mg, nicotinamide 10 mg, lecithin 9.4 mg, and sodium selenite 0.8 m |
1–5 years | Placebo | There was a significant decrease in deaths from coronary heart disease |
| 15 | Loffredo et al. [24] | 2006 | 40 | 40–80 | Not mentioned | Propionyl -L-carnitine | Synthetic intravenous propionyl-L-carnitine | 6 mg | 7 days | Placebo | There was a significant improvement in maximum walking distance |
| 16 | Loffredo et al. [25] | 2007 | 25 | 40–80 | Not mentioned | Propionyl-L-carnitine | Synthetic intravenous propionyl-L-carnitine | 6 mg | 7 days | Placebo | There was a significant improvement in the oxidative stress marker and flow-mediated dilation |
| 17 | Singh JA et al. [26] | 2018 | 3167 | 75–85 | Men and women | Allopurinol | Synthetic | Not enough evidence to determine | 5 years | There were no controls in this study | There was a significantly lower risk of PAD in the longer allopurinol users. |
| 18 | Poggesi et al. [27] | 1985 | 10 | 32–50 | 8 men and 2 women | Pentoxifylline | Synthetic pentoxifylline ampules; pentoxifylline tablets | 100 mg pentoxifylline ampules; 400 mg pentoxifylline tablets | 1–20 days | Placebo | There was a significant improvement in arterial blood flow and antithrombotic effect |
5. Discussion
Oxidative stress plays a vital role in the development of PAD, but not in isolation, as other risk factors including hypertension, diabetes, smoking, hypercholesterolemia, obesity, and physical inactivity also contribute to the development of symptoms of PAD [7,28]. However, it has been shown that increased levels of oxidative stress can be an important mechanism in the pathophysiology of these risk factors, leading to increased levels of oxidized LDL (ox-LDL), increased thrombus formation, endothelial dysfunction, and the development of atherosclerotic plaques [7,8]. Therefore, it can be deduced that antioxidants may be effective in PAD treatment [29,30].
The current study was designed as a systematic review investigating the effect of antioxidant therapy in PAD patients. Two forms of antioxidants were studied—natural antioxidants (in the form of fruits and vegetables) and synthetic antioxidants (such as dietary supplements or medications).
The reviewed studies show that natural antioxidants have been thoroughly effective in treating PAD (100%), but fewer studies have used this method [10,11,12]. This beneficial effect is not consistently seen with synthetic antioxidants, and only half (53%) of the studies have shown effective results [17,20,22,23,24,25,26,27]. Based on these data, it could be assumed that antioxidants in their natural form may be more effective than synthetic forms in treating PAD. In addition, fruits and vegetables are rich sources of minerals along with antioxidants. On the other hand, the presence of minerals such as sodium, potassium, selenium, magnesium, zinc, copper, and calcium together with antioxidants and an optimal balance with them has been shown to increase the effect of antioxidants [31].
Furthermore, in 38.8% of the reviewed studies, there was no significant effect on the improvement in patients using antioxidants [13,14,15,16,18,19,21]. Furthermore, based on the reviewed studies, these also showed that the dose and duration of antioxidant therapy did not significantly affect its effectiveness.
Overall, studies on the effect of antioxidants causing improvements in PAD patients show some contradictory outcomes. On the contrary, this contradiction regarding the antioxidant effects for PAD treatment may be due to the oxidative effect of antioxidants. It has been shown that, if the level of antioxidants in the body exceeds the required level, they produce oxidative effects, and excess levels of antioxidants can lead to increased levels of oxidative stress [32]. Therefore, although the therapeutic perspective on antioxidant supplements has been optimistic, they should not be administered indiscriminately and/or excessively. The intrinsic antioxidant system in cells can prevent damage from the effects of oxidants. Therefore, checking the pro-oxidant–antioxidant balance can be an effective method for managing antioxidant treatment. The measurement of oxidant and antioxidant capacity has been shown to help in understanding the balance between them. Therefore, prescribing antioxidants may be more effective in patients after considering the pro-oxidant–antioxidant balance/ratio [32,33].
Finally, due to the multifactorial nature of PAD, using antioxidants as a definitive treatment alone may not be effective [6]. Therefore, using additional therapies according to the patient’s underlying disease (anticoagulants, antiplatelet agents, antihypertensives, antidiabetic agents, and statins) along with the appropriate amount of antioxidants seems to be more effective [6,34]. Due to the positive feedback between the initiator (cause) of pathogenesis and its consequences, there is an interaction between the oxidative stress level and thrombosis, leading to the elevation of both [35]. This is also true about the interaction between inflammation and thrombosis [36]. Therefore, interactions at multiple critical points need to be identified and addressed. Although oxidative stress is the hub of this network, we need to consider other strategic issues that can trigger a feedback loop and break the cycle started by the initiator compound. As a result, combination therapy can be more comprehensive and effective [6,35,36]. Additionally, potassium and calcium in some fruits and vegetables, such as oranges, can regulate blood pressure. Based on this, it can be assumed that natural antioxidants in fruits and vegetables can be more efficacious [31]. The review also shows a lack of further similar studies on the effectiveness of antioxidants in populations at risk of developing PAD (such as older adults and people with unhealthy lifestyles) in preventing PAD.
6. Conclusions
In conclusion, though there is optimism regarding using antioxidants in treating PAD, the outcomes do not support the optimism. Using antioxidants in the natural forms and vitamins in the form of fruits and vegetables is likely to be more effective for PAD patients.
Finally, two essential factors must be considered to be more effective in treating PAD. First, evaluating the optimal pro-oxidant–antioxidant balance can be an effective method for managing treatment with antioxidants. In other words, prescribing antioxidants at the appropriate dose makes antioxidants more effective and strongly suggests that custom therapies need to be devised. Secondly, using the optimal amount of antioxidants for treating the underlying disease in PAD patients, together with other agents such as anticoagulants, antiplatelet agents, antihypertensives, hypoglycemic agents, antidiabetic agents, statins, and other cholesterol-balancing drugs, is a strong recommendation.
Author Contributions
S.K., data analysis, writing—original draft preparation, and visualization; H.S., data analysis; M.P., supervision and review; B.F., conceptualization, data analysis, and writing—review and editing; A.S., conceptualization, supervision, and review. All the authors have read and agreed to the published version of the manuscript. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
We used PubMed and Web of Science to screen articles for this narrative review. We do not report any data.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research received no external funding.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Olinic D.-M., Spinu M., Olinic M., Homorodean C., Tataru D.-A., Liew A., Schernthaner G., Stanek A., Fowkes G., Catalano M. Epidemiology of peripheral artery disease in Europe: VAS Educational Paper. Int. Angiol. 2018;37:327–334. doi: 10.23736/S0392-9590.18.03996-2. [DOI] [PubMed] [Google Scholar]
- 2.Frank U., Nikol S., Belch J.J., Boc V., Brodmann M., Carpentier P.H., Chraim A., Canning C., Dimakakos E., Gottsäter A., et al. ESVM Guideline on peripheral arterial disease. Vasa. 2019;48:1–79. doi: 10.1024/0301-1526/a000834. [DOI] [PubMed] [Google Scholar]
- 3.Scully R.E., Arnaoutakis D.J., Smith A.D., Semel M., Nguyen L.L. Estimated annual health care expenditures in individuals with peripheral arterial disease. J. Vasc. Surg. 2018;67:558–567. doi: 10.1016/j.jvs.2017.06.102. [DOI] [PubMed] [Google Scholar]
- 4.Aday A.W., Matsushita K. Epidemiology of Peripheral Artery Disease and Polyvascular Disease. Circ. Res. 2021;128:1818–1832. doi: 10.1161/CIRCRESAHA.121.318535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ness J., Aronow W.S. Prevalence of Coexistence of Coronary Artery Disease, Ischemic Stroke, and Peripheral Arterial Disease in Older Persons, Mean Age 80 Years, in an Academic Hospital-Based Geriatrics Practice. J. Am. Geriatr. Soc. 1999;47:1255–1256. doi: 10.1111/j.1532-5415.1999.tb05208.x. [DOI] [PubMed] [Google Scholar]
- 6.Bevan G.H., Solaru K.T.W. Evidence-Based Medical Management of Peripheral Artery Disease. Arterioscler. Thromb. Vasc. Biol. 2020;40:541–553. doi: 10.1161/ATVBAHA.119.312142. [DOI] [PubMed] [Google Scholar]
- 7.Signorelli S.S., Scuto S., Marino E., Xourafa A., Gaudio A. Oxidative Stress in Peripheral Arterial Disease (PAD) Mechanism and Biomarkers. Antioxidants. 2019;8:367. doi: 10.3390/antiox8090367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stanek A., Fazeli B., Bartuś S., Sutkowska E. The Role of Endothelium in Physiological and Pathological States: New Data. BioMed Res. Int. 2018;2018:1098039. doi: 10.1155/2018/1098039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Koutakis P., Ismaeel A., Farmer P., Purcell S., Smith R.S., Eidson J.L., Bohannon W.T. Oxidative stress and antioxidant treatment in patients with peripheral artery disease. Physiol. Rep. 2018;6:e13650. doi: 10.14814/phy2.13650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dalgård C., Nielsen F., Morrow J.D., Enghusen-Poulsen H., Jonung T., Hørder M., De Maat M.P.M. Supplementation with orange and blackcurrant juice, but not vitamin E, improves inflammatory markers in patients with peripheral arterial disease. Br. J. Nutr. 2008;101:263–269. doi: 10.1017/S0007114508995660. [DOI] [PubMed] [Google Scholar]
- 11.Klipstein-Grobusch K., Breeijen J.H.D., Grobbee D.E., Boeing H., Hofman A., Witteman J.C.M. Dietary Antioxidants and Peripheral Arterial Disease: The Rotterdam Study. Am. J. Epidemiol. 2001;154:145–149. doi: 10.1093/aje/154.2.145. [DOI] [PubMed] [Google Scholar]
- 12.Woessner M., Van Bruggen M.D., Pieper C.F., Sloane R., Kraus W.E., Gow A.J., Allen J.D. Beet the Best? Circ. Res. 2018;123:654–659. doi: 10.1161/CIRCRESAHA.118.313131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kleijnen J., Mackerras D. Vitamin E for intermittent claudication. Cochrane Database Syst. Rev. 1998;2000:CD000987. doi: 10.1002/14651858.CD000987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Critical Leg Ischaemia Prevention Study (CLIPS) Group. Catalano M., Born G., Peto R. Prevention of serious vascular events by aspirin amongst patients with peripheral arterial disease: Randomized, double-blind trial. J. Intern. Med. 2007;261:276–284. doi: 10.1111/j.1365-2796.2006.01763.x. [DOI] [PubMed] [Google Scholar]
- 15.McDermott M.M., Leeuwenburgh C., Guralnik J.M., Tian L., Sufit R., Zhao L., Criqui M.H., Kibbe M.R., Stein J.S., Lloyd-Jones D., et al. Effect of Resveratrol on Walking Performance in Older People with Peripheral Artery Disease: The RESTORE Randomized Clinical Trial. JAMA Cardiol. 2017;2:902–907. doi: 10.1001/jamacardio.2017.0538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jepson R.G., Kleijnen J., Leng G.C. Garlic for peripheral arterial occlusive disease. Cochrane Database Syst. Rev. 2013;2013:CD000095. doi: 10.1002/14651858.CD000095.pub2. [DOI] [PubMed] [Google Scholar]
- 17.Horsch S., Walther C. Ginkgo biloba special extract EGb 761 in the treatment of peripheral arterial occlusive disease (PAOD)—A review based on randomized, controlled studies. Int. J. Clin. Pharmacol. Ther. 2004;42:63–72. doi: 10.5414/CPP42063. [DOI] [PubMed] [Google Scholar]
- 18.Campbell A., Price J., Hiatt W.R. Omega-3 fatty acids for intermittent claudication. Cochrane Database Syst. Rev. 2013;7:CD003833. doi: 10.1002/14651858.CD003833.pub4. [DOI] [PubMed] [Google Scholar]
- 19.Curtis P., Potter J., Kroon P., Wilson P., Dhatariya K., Sampson M., Cassidy A. Vascular function and atherosclerosis progression after 1 y of flavonoid intake in statin-treated postmenopausal women with type 2 diabetes: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2013;97:936–942. doi: 10.3945/ajcn.112.043745. [DOI] [PubMed] [Google Scholar]
- 20.Loffredo L., Perri L., Catasca E., Pignatelli P., Brancorsini M., Nocella C., De Falco E., Bartimoccia S., Frati G., Carnevale R., et al. Dark Chocolate Acutely Improves Walking Autonomy in Patients with Peripheral Artery Disease. J. Am. Heart Assoc. 2014;3:e001072. doi: 10.1161/JAHA.114.001072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hammer A., Koppensteiner R., Steiner S., Niessner A., Goliasch G., Gschwandtner M., Hoke M. Dark chocolate and vascular function in patients with peripheral artery disease: A randomized, controlled cross-over trial. Clin. Hemorheol. Microcirc. 2015;59:145–153. doi: 10.3233/CH-141817. [DOI] [PubMed] [Google Scholar]
- 22.McDermott M.M., Criqui M.H., Domanchuk K., Ferrucci L., Guralnik J.M., Kibbe M.R., Kosmac K., Kramer C.M., Leeuwenburgh C., Li L., et al. Cocoa to Improve Walking Performance in Older People with Peripheral Artery Disease: The COCOA-PAD Pilot Randomized Clinical Trial. Circ. Res. 2020;126:589–599. doi: 10.1161/CIRCRESAHA.119.315600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Belch J., MacCuish A., Campbell I., Cobbe S., Taylor R., Prescott R., Lee R., Bancroft J., MacEwan S., Shepherd J., et al. The prevention of progression of arterial disease and diabetes (POPADAD) trial: Factorial randomized placebo-controlled trial of aspirin and antioxidants in patients with diabetes and asymptomatic peripheral arterial disease. BMJ. 2008;337:a1840. doi: 10.1136/bmj.a1840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Loffredo L., Pignatelli P., Cangemi R., Andreozzi P., Panico M.A., Meloni V., Violi F. Imbalance between nitric oxide generation and oxidative stress in patients with peripheral arterial disease: Effect of an antioxidant treatment. J. Vasc. Surg. 2006;44:525–530. doi: 10.1016/j.jvs.2006.05.023. [DOI] [PubMed] [Google Scholar]
- 25.Loffredo L., Marcoccia A., Pignatelli P., Andreozzi P., Borgia M.C., Cangemi R., Chiarotti F., Violi F. Oxidative-stress-mediated arterial dysfunction in patients with peripheral arterial disease. Eur. Heart J. 2007;5:608–612. doi: 10.1016/S1567-5688(07)71408-3. [DOI] [PubMed] [Google Scholar]
- 26.Singh J.A., Cleveland J. Allopurinol and the risk of incident peripheral arterial disease in the elderly: A US Medicare claims data study. Rheumatology. 2018;57:451–461. doi: 10.1093/rheumatology/kex232. [DOI] [PubMed] [Google Scholar]
- 27.Poggesi L., Scarti L., Boddi M., Masotti G., Serneri G.G.N. Pentoxifylline Treatment in Patients with Occlusive Peripheral Arterial Disease. Circulatory Changes and Effects on Prostaglandin Synthesis. Angiology. 1985;36:628–637. doi: 10.1177/000331978503600907. [DOI] [PubMed] [Google Scholar]
- 28.Majid K.A., Lohana P., Anvekar P., Mustafa S.H., Kumar R., Adnan L.N.U., Bhimani P., Ali S.R., Lnu A., Shah S.H.A. Risk Factors of Peripheral Vascular Disease in Diabetes Mellitus in Abbottabad, Pakistan: A Cross-Sectional Study. Cureus. 2021;13:e17556. doi: 10.7759/cureus.17556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Poznyak A.V., Nikiforov N.G., Markin A.M., Kashirskikh D.A., Myasoedova V.A., Gerasimova E.V., Orekhov A.N. Overview of OxLDL and Its Impact on Cardiovascular Health: Focus on Atherosclerosis. Front. Pharmacol. 2021;11:2248. doi: 10.3389/fphar.2020.613780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wen C., Ying Y., Yu F., Zhou J. Research Progress of Oxidative Stress and MicroRNAs in the Prevention of Catheter-Related Thrombus Under Resistance Exercise. Clin. Appl. Thromb. 2020;26:1076029620931931. doi: 10.1177/1076029620931931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chiu H.-F., Venkatakrishnan K., Golovinskaia O., Wang C.-K. Impact of Micronutrients on Hypertension: Evidence from Clinical Trials with a Special Focus on Meta-Analysis. Nutrients. 2021;13:588. doi: 10.3390/nu13020588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Alamdari D.H., Paletas K., Pegiou T., Sarigianni M., Befani C., Koliakos G. A novel assay for the evaluation of the prooxidant–antioxidant balance, before and after antioxidant vitamin administration in type II diabetes patients. Clin. Biochem. 2007;40:248–254. doi: 10.1016/j.clinbiochem.2006.10.017. [DOI] [PubMed] [Google Scholar]
- 33.Alamdari D.H., Ghayour-Mobarhan M., Tavallaie S., Parizadeh M.R., Moohebati M., Ghafoori F., Kazemi-Bajestani S.M.R., Paletas K., Pegiou T., Koliakos G. Prooxidant–antioxidant balance as a new risk factor in patients with angiographically defined coronary artery disease. Clin. Biochem. 2008;41:375–380. doi: 10.1016/j.clinbiochem.2007.12.008. [DOI] [PubMed] [Google Scholar]
- 34.Jansen-Chaparro S., López-Carmona M.D., Cobos-Palacios L., Sanz-Cánovas J., Bernal-López M.R., Gómez-Huelgas R. Statins and Peripheral Arterial Disease: A Narrative Review. Front. Cardiovasc. Med. 2021;8:777016. doi: 10.3389/fcvm.2021.777016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang Q., Zennadi R. Oxidative Stress and Thrombosis during Aging: The Roles of Oxidative Stress in RBCs in Venous Thrombosis. Int. J. Mol. Sci. 2020;21:4259. doi: 10.3390/ijms21124259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stark K., Massberg S. Interplay between inflammation and thrombosis in cardiovascular pathology. Nat. Rev. Cardiol. 2021;18:666–682. doi: 10.1038/s41569-021-00552-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
We used PubMed and Web of Science to screen articles for this narrative review. We do not report any data.