Abstract
Small molecular nucleic acid drugs produce antiviral effects by activating pattern recognition receptors (PRRs). In this study, a small molecular nucleotide containing 5′triphosphoric acid (5′PPP) and possessing a double-stranded structure was designed and named nCoV-L. nCoV-L was found to specifically activate RIG-I, induce interferon responses, and inhibit duplication of four RNA viruses (Human enterovirus 71, Human poliovirus 1, Human coxsackievirus B5 and Influenza A virus) in cells. In vivo, nCoV-L quickly induced interferon responses and protected BALB/c suckling mice from a lethal dose of the enterovirus 71. Additionally, prophylactic administration of nCoV-L was found to reduce mouse death and relieve morbidity symptoms in a K18-hACE2 mouse lethal model of SARS-CoV-2. In summary, these findings indicate that nCoV-L activates RIG-I and quickly induces effective antiviral signals. Thus, it has potential as a broad-spectrum antiviral drug.
Keywords: RIG-I, antiviral, RNA agonist, SARS-CoV-2
1. Introduction
Innate immunity is the first line to defend against viral infection. Innate immunity recognises pathogen-associated molecular patterns (PAMPs) through pattern recognition receptors (PRRs) to induce immune responses [1,2]. In mammals, the viral RNA recognition function of innate immunity begins with RIG-I-like receptors (RLRs), Toll-like receptors (TLRs), and NOD-like receptors (NLRs) [3,4]. Among them, RIG-I is a PRR in the RLRs family. Located in the cytoplasm, RIG-I recognises short double-stranded RNA with 5′PPP [5,6,7]. Conformational change of RIG-I is induced after recognising the viral double-stranded RNA [8]. Activated RIG-I interacts with mitochondrial antiviral signal protein (MAVS) to form a protein complex [9,10]. The MAVS complex then recruits serine protein kinase (IKKε/TBK1) to induce phosphorylation of interferon regulatory factor 3 and 7 (p-IRF 3 and p-IRF 7) and activate nuclear factor kappa-B (NF-κB). p-IRF3 and p-IRF7 are transferred to the cell nucleus and induce the expression of type I interferon (IFN-I) [11,12]. IFN-I induces expression of interferon-stimulated genes (ISGs) through the Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway, directly inhibiting transcription of viral genes, degrading viral RNA, and inhibiting translation and modification of viral proteins [13,14,15]. NF-κB could mediate the expression of several proinflammatory cytokines, such as TNF-α and IL-6, which contributes to the antiviral effects [16,17].
Compared to antiviral therapy with recombinant IFN-I injection, RIG-I agonists can induce robust endogenous IFN-I responses and multiple inflammatory cytokines, maximising the likelihood of functional downstream responses [18]. Moreover, recombinant interferon, which has a different molecular structure to human endogenous interferon, may induce interferon antibodies in vivo, thus influencing the treatment outcomes [19,20,21]. Chiang, et al. [22] designed a 99 nt fragment of 5′ppp RNA named M8, which activated RIG-I and significantly inhibited multiple influenza viruses and chikungunya virus. Therefore, RIG-I agonists have significant research, development, and application prospects.
The RIG-I receptor can recognise double-stranded RNA sequences with 5′PPP [23,24,25]. Thus, in this study, a sequence with a stem-loop structure in the 5′ untranslated regions (UTR) of SARS-CoV-2 was selected and reconstructed to obtain a 100 nt fragment with 5′PPP. A hairpin was confirmed, owing to the reverse complementary pairing intramolecularly, and this small molecular nucleotide was named nCoV-L. Subsequently, the protective effects of nCoV-L against infection with several RNA viruses, including Human enterovirus 71 C2-2 strain (EV-71/C2-2), Human coxsackievirus B5 JS417 strain (CV-B5/JS417), Human poliovirus 1 Sabin strain (PV-1/Sabin), Influenza A virus H9N2 (IAV/H9N2) and SARS-CoV-2 Delta strain (SARS-CoV-2/Delta), were analysed through in vitro and in vivo experiments.
2. Materials and Methods
2.1. Biosafety and Ethics Approval
Experiments with live SARS-CoV-2 were performed in a biosafety level 3 (ABSL3) facility at the Wuhan Institute of Biological Products Co., Ltd. (Wuhan, China). All research staff were trained and qualified in the experimental procedures. Animal procedures were approved by the Ethics Committee on Laboratory Animals of the Wuhan Institute of Biological Products Co., Ltd. The number of ethical permissions for this experiment was WIBP-AII442021005.
Other experiments with live viruses were performed in a biosafety level 2 (ABSL2) facility at the National Institutes for Food and Drug Control (Beijing, China). Animal research protocols were approved by the Institutional Animal Care and Use Committee at the National Institutes for Food and Drug Control, China. The ethical permission number for this experiment was 2022-B020.
2.2. Cell Culture
A549 (CRM-CCL-185), RD (CCL-81), HEp-2 (CCL-23), Vero (CCL-81), MDCK (CCL-34) and C2C12 (CRL-1772) cells were purchased from ATCC. The HEK293 cell lines with ISG54-luciferase reporter gene (hkl-null) were purchased from InvivoGene (San Diego, CA, USA). TLR3, TLR7, TLR8, MDA5 and RIG-I in the HEK293 cell lines with ISG54-luciferase reporter gene were knocked out with the CRISPR/Cas9 system. The backbone plasmids lenti-sgRNA and lenti-cas9-zeocin were obtained from Genescript Co., Ltd. (Nanjing, China). sgRNAs were designed and subcloned into a Cas9 backbone. All the sgRNAs used are described in Table 1. ISG54-luciferase reporter HEK293 cells were first infected with lenti-cas9-zeocin and then selected using zeocin. The stable sub-lines were then infected with lenti-sgRNA to specifically knock out the target genes. The CRISPR knockout cell lines were sanger sequenced to confirm frame shift (Table A1).
Table 1.
sgRNA sequences.
| Gene | sgRNA |
|---|---|
| TLR3 | CAACTTTCTTGGGACTAAAG |
| TLR7 | TTCAGCATGTGCCCCCAAGA |
| TLR8 | TTAGTGGGAGAAATAGCCTC |
| MDA5 | GCTCAGGCCTTACCAAATGG |
| RIG-I | AATTCCCACAAGGACAAAAG |
C2C12 cells transfected by lentivirus expressing CXADR gene (c2c12-CAR) were cultivated in DMEM (Gibco, Carlsbad, CA, USA), supplemented with 10% fetal bovine serum (FBS) (Gibco, Carlsbad, CA, USA) and 100 U/mL penicillin-streptomycin (Gibco, Carlsbad, Carlsbad, CA, USA) in a humidified incubator in the presence of 5% CO2. RD, HEp-2, MDCK and Vero cells were cultivated in MEM (Gibco, Carlsbad, CA, USA). The wild type (WT), RIG-I −/−, MDA5 −/−, TLR3 −/−, TLR7 −/−, and TLR8 −/− HEK293 cell lines with ISG54-luciferase reporter gene and A549 cells were cultivated in DMEM (Gibco, Carlsbad, CA, USA).
2.3. Viruses
EV-71/C2-2, CV-B5/JS417 (GenBank accession no. KY303900) and IAV/H9N2 (GenBank accession no. FJ499463-FJ499470) were preserved in the Hepatitis and Enterovirus Laboratory. PV-1/Sabin (GenBank accession no. V01150.1) was donated by the Influenza and Respiratory Virus Laboratory of NIFDC. SARS-CoV-2/Delta (GenBank accession no. OK091006.1) was preserved in a biosafety level 3 (ABSL3) facility at the Wuhan Institute of Biological Products Co., Ltd.
EV-71/C2-2 was used to infect RD cells at a multiplicity of infection (MOI) of 0.1. The virus was allowed to adsorb for 1 h at 35 °C in serum-free MEM. Serum-free MEM was used to wash the monolayer and then replaced with 2% FBS MEM. After seven days of infection, the medium was harvested and cleared by centrifugation (12,000 rpm for 20 min) at 4 °C. Viral titers were determined using cytopathic effect-based end-point titrations.
CV-B5/JS417 and SARS-CoV-2/Delta were propagated in Vero cells. PV-1/Sabin was propagated in HEp-2 cells. IAV/H9N2 were propagated in MDCK cells.
2.4. Mice
Seven-day-old BALB/c mice were purchased from the Laboratory Animal Resource Centre, National Institute for Food and Drug Control, and were housed in a specific pathogen-free (SPF) animal facility.
Six-week-old SPF K18-hACE2 transgenic mice were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China).
2.5. In Vitro Transcription and Gel Analysis
The sequence of nCoV-L was as follows:
5′-GGUUUAAUACCUAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAA
UCCCUUUUUUUUUUUUUUUUUUUUUUUUUUUUUUUUUUUUAGGUAUUAAA
CC-3′ (100 nt).
The target sequence was constructed in the pUC19 plasmid vector and amplified. The amplified plasmid was digested with the endonuclease BsmBI-v2 (New England Biolabs, Ipswich, MA, USA). Then, the target sequence was collected and purified. nCoV-L and M8 were synthesised with a T7 RiboMaxTM Express Large Scale RNA Production System (Promega, Madison, WI, USA) for 3 h. RNA transcripts were DNase digested for 20 min at 37 °C and then purified according to the manufacturer’s instructions. RNA transcripts were analysed on 2% agarose gel with NorthernMax GlyGel Prep/Running buffer (Invitrogen, Carlsbad, CA, USA) for 1 h. The secondary structure of nCoV-L was predicted using the RNAfold Webserver (http://rna.tbi.univie.ac.at/cgibin/RNAWebSuite/RNAfold.cgi) (accessed on 7 June 2021).
2.6. Transfections
Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA) was used for all in vitro and in vivo transfections. nCoV-L and Lipofectamine 3000 reagent (1:1.5 ratio) were mixed in Opti-MEM (Gibco, Carlsbad, CA, USA) and incubated for 10 min. The volume of the mixture for in vitro transfection was 50 μL, and for in vivo transfection was 100 μL.
2.7. Luciferase Assays
For the luciferase assays, the TLR3 −/−, TLR7 −/−, TLR8 −/−, MDA5 −/−, RIG-I −/−, and WT HEK293 cell lines with ISG54-luciferase reporter gene were transfected with 50 ng nCoV-L. The activity of the reporter gene was measured by a Dual-luciferase Reporter Assay (Promega, Madison, WI, USA), according to the manufacturer’s instructions. Relative luciferase activity was measured.
2.8. Protein Extraction and Western Blot
5′ppp RNA-transfected cells were collected by centrifugation at 12,000 rpm for 1 min. The cell precipitation was lysed in RIPA buffer (Sigma-Aldrich, Milwaukee, WI, USA) and cleared by centrifugation at 12,000 rpm for 15 min at 4 °C. The cleared lysis was subjected to SDS-PAGE. Proteins were electrophoretically transferred to nitrocellulose membranes and then subjected to immunoblotting analysis using the indicated antibodies. Anti-RIG-I, anti-MDA5, anti-IRF3, anti-pIRF3 Ser 396, anti-ISG56 and anti-β-actin antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA). Horseradish peroxidase (HRP)-conjugated goat anti-rabbit antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA). Images were visualised by using an Amersham Imager 680.
2.9. RNA Extraction
Total RNA in cells or tissues was isolated using a Kingfisher Flex Automatic Nucleic Acid Extractor (Thermo Fisher Scientific, Waltham Mass, MA, USA) with Pre-packaged Nucleic Acid Extraction and Purification Kits (Thermo Fisher Scientific, Waltham, MA, USA), according to the manufacturer’s instructions.
2.10. Real-Time Quantitative Polymerase Chain Reaction (RT-qPCR)
Extracted RNA was subjected to RT-qPCR with a PrimeScript One Step RT-PCR Kit (Takara, Kyoto, JPN), according to the manufacturer’s instructions. PCR primers were designed with PrimerBank and purchased from Sangon Biotech Company (Shanghai, China). All the primers used are described in Table 2. RT-qPCR was performed on an ABI7500 (Applied Biosystems, Arlington, VA, USA). All data are presented as relative quantification with gadph as the internal control.
Table 2.
Primer sequences used for RT-qPCR.
| Gene | Direction | Primer Sequence (5′-3′) |
|---|---|---|
| EV71/C2-2 | Forward | TAG TTT CTT CAG CAG GGC GG |
| Reverse | ACC ATT GGT AAG CAC TCG CA | |
| CV-B5/JS417 | Forward | GGT GTC CGT GTT TCC TTT TAT TCC TAC |
| Reverse | CAA GTA GAT AAT AGC TCT GTT TGT CAC CG | |
| PV-1/Sabin | Forward | TGA TCA CAA CCC GAC CAA GG |
| Reverse | TAA TCC ACT CCA GGG CCG TA | |
| IAV/H9N2 | Forward | CCG GAA TTT CTG GAG AGG CG |
| Reverse | TAC ACA AGC AGG CAA GCA GG | |
| IFN-β | Forward | GCT TGG ATT CCT ACA AAG AAG CA |
| Reverse | ATA GAT GGT CAA TGC GGC GTC |
2.11. In Vitro Virus Infection
For in vitro virus infection experiments, 2 × 105 cells were infected with virus strains in a small volume of serum-free medium for 1 h at 37 °C. The medium was replaced with a complete medium 24 h prior to analysis.
2.12. In Vivo Virus Infection
Seven-day-old BALB/c mice were challenged intraperitoneally with 1.26 × 106 TCID50 EV71/C2-2 in 100 μL of MEM.
Six-week-old K18-hACE2 transgenic mice were challenged intranasally with 200 TCID50 SARS-CoV-2 Delta in 50 μL of DMEM.
2.13. Histopathological Examination
The harvested tissues were fixed in 4% paraformaldehyde, embedded in paraffin, sectioned, and stained with haematoxylin and eosin (H&E) for histopathological analysis. An experienced and qualified pathologist confirmed the results.
2.14. ELISA
The concentration of IFN-β in mouse serum was determined using ELISA with Legend Max Mouse IFN-β ELISA Kits (BioLegend, San Diego, CA, USA), according to the manufacturer’s instructions.
2.15. Statistical Analyses
Data are presented as the mean ± standard derivation (SD). Statistical analysis was performed using GraphPad Prism software 8.0. Unpaired, two-tailed Student’s t-tests were performed to determine the statistical significance of differences. Differences were considered statistically significant when p was less than 0.05.
3. Results
3.1. nCoV-L Induce Interferon Response via the RIG-I Receptor
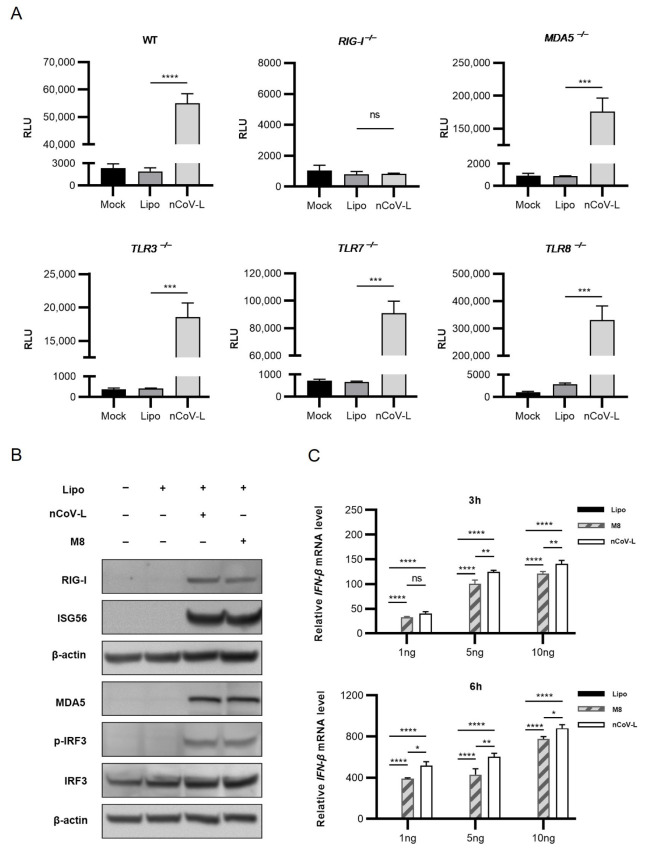
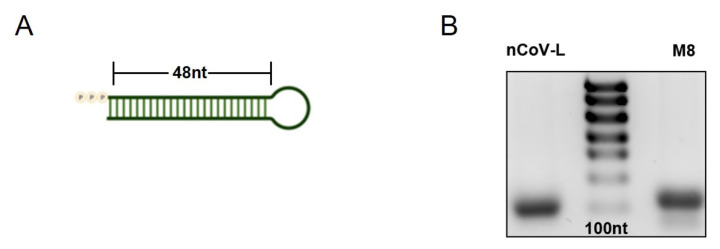
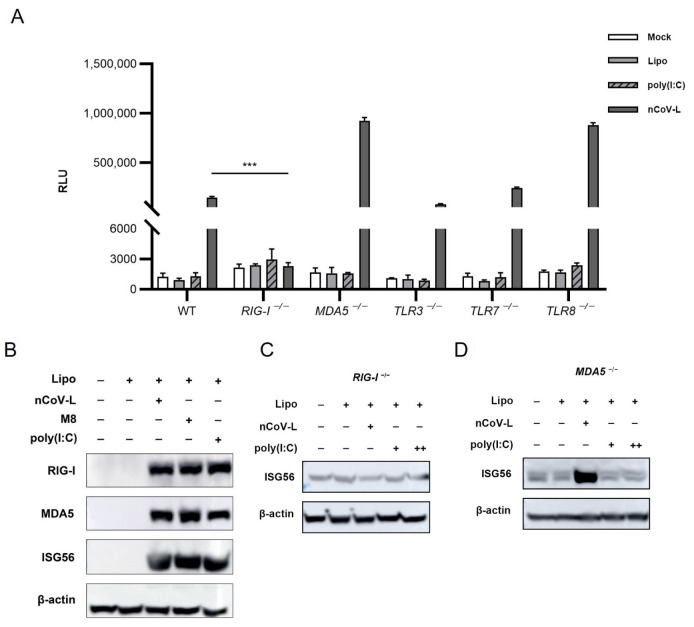
The predicted structure of nCoV-L is shown in Figure A1A in Appendix A, demonstrating that double-stranded stem-loop structures can be formed through complementary pairing. The 5′ppp RNA products were acquired through in vitro transcription and purification. A single stripe was identified through RNA electrophoresis detection (Figure A1B). The TLR3 −/−, TLR7 −/−, TLR8 −/−, MDA5 −/−, RIG-I −/−, and WT HEK293 cell lines with ISG54-luciferase reporter gene were transfected with 50 ng nCoV-L. The expression levels of reporter gene were analysed 24 h after different treatments (Figure 1A and Figure A2A). nCoV-L activated the expression of the reporter gene in TLR3 −/−, TLR7 −/−, TLR8 −/−, MDA5 −/−, and WT cell lines, but not in RIG-I −/− cells, consistent with the induction of a downstream factor ISG56 (Figure A2C,D), suggesting that nCoV-L can specifically activate the RIG-I receptor. When using poly(I:C) as control, no induction of reporter gene or ISG56 was detected (Figure A2A,C,D), which may be due to the insensitive of knockout cell lines to poly(I:C) compared to nCoV-L.
Figure 1.
nCoV-L induces an interferon response by activating the RIG-I receptor. (A) 1 × 104 wild-type (WT), RIG-I −/−, MDA5 −/−, TLR3 −/−, TLR7 −/−, TLR8 −/−, and HEK293 cells expressing ISG54-luciferase reporter gene were cultivated in 96-well plate for 12 h, and then transfected with 50 ng nCoV-L using Lipofectamine 3000 (short for Lipo) or treated with equal dosage of Lipo as the negative control. Relative luciferase activity was tested after 24 h. N = 3, RLU indicates relative light unit, bar indicates SD. (B) 2 × 106 A549 cells were transfected with 1 ng/mL nCoV-L or M8 using Lipo. Cell lysis was prepared and subjected to Western blot analysis 24 h after transfection. (C) 2 × 106 A549 cells were transfected with 1 ng/mL,5 ng/mL or 10 ng/mL nCoV-L or M8 using Lipo. Cells were harvested 3 h or 6 h after transfection and total RNA was extracted. Relative IFN-β gene expression levels were determined by RT-qPCR. N = 3, bar indicates SD. ns, p > 0.05, *, p < 0.05, **, p < 0.01, ***, p < 0.005, ****, p < 0.001.
Western blot analysis was conducted to detect the expression and activation of proteins involved in the RIG-I signalling pathway. Increased phosphorylation levels of IRF3 (Ser396) and upregulated ISG56 were detected after transfection of 1 ng/mL nCoV-L or M8 in A549 cells (Figure 1B and Figure A2B), indicating the activation of RIG-I signalling. RIG-I, MDA5, and IRF3 were also upregulated (Figure 1B and Figure A2B), which was dependent on the positive feedback regulation [26,27]. The IFN-β expression levels at different times post-transfection in A549 cells revealed that IFN-β could be effectively induced by nCoV-L, which was higher than that of M8 (p < 0.05) under our laboratory condition (Figure 1C). These findings indicated that the double-stranded molecular nCoV-L, with 5′ PPP and a stem-loop structure, induced the expression of interferon and downstream ISGs to activate innate antiviral immunity by specifically activating the RIG-I receptor and mediating IRF3 phosphorylation.
3.2. In Vitro Antiviral Effects of nCoV-L against RNA Viruses EV-71, CV-B5, PV-1 and IAV
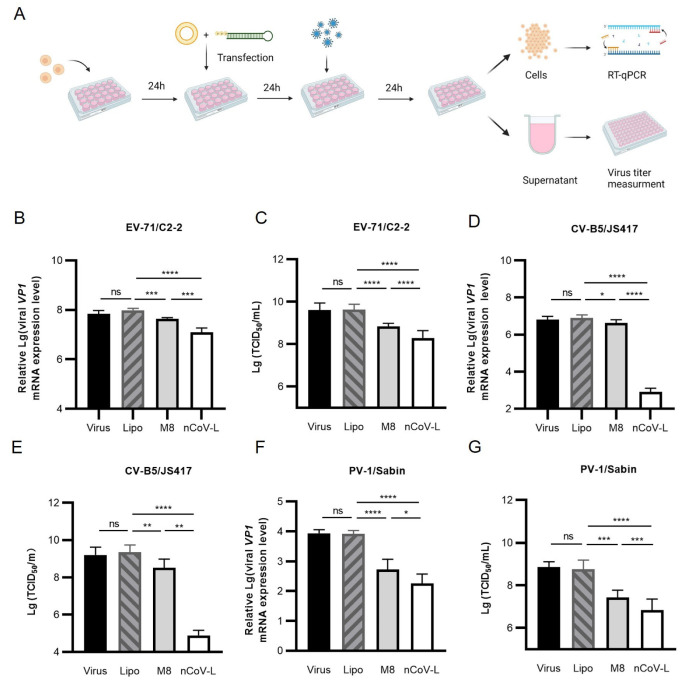
Next, the in vitro inhibition effects of nCoV-L against four RNA viruses (EV-71/C2-2, CV-B5/JS417, PV-1/Sabin, and IAV/H9N2) were analysed. As shown in Figure 2A, the cells were collected after treatment, and RNA was extracted to measure the viral load. The virus titers in the culture supernatants were also evaluated. The results demonstrated that, compared with the liposome negative control, 30 ng/mL nCoV-L inhibited duplication of EV-71/C2-2 in RD cells, decreasing the viral load by approximately 7.8-fold (p < 0.0001). Accordingly, the viral titer in the culture supernatant was decreased 22-fold (p < 0.0001). Under our experimental conditions, nCoV-L decreased the viral load in cells and the virus titer in the supernatants by 3.5-fold and 3.6-fold compared to the equivalent dosage of M8 (p < 0.01). The intracellular viral loads and virus titers of CV-B5/JS417 and PV-1/Sabin in the supernatants were assessed using the same method. nCoV-L significantly inhibited the replication of CV-B5/JS417 and PV-1/Sabin (p < 0.001), markedly better than M8 at the same dosage (p < 0.05). As shown in Figure A3, 1 ng/mL nCoV-L was also proven to inhibit intracellular IAV/H9N2 virus infection and was more effective than the same dose of M8 (p < 0.001). In summary, nCoV-L significantly inhibited the EV-71/C2-2, CV-B5/JS417, PV-1/Sabin, and IAV/H9N2 viral strains.
Figure 2.
The in vitro antiviral effect of nCoV-L against three different RNA viruses. (A) Experimental scheme. 2 × 105 cells were transfected with nCoV-L or M8 for 24 h and then infected with virus (0.1 MOI) for 24 h. Cells were harvested, and total RNA was extracted. Viral loads were measured by RT-qPCR. Viral titers in cell culture supernatants were also determined. RD cells were transfected with 30 ng/mL nCoV-L or M8, and then infected with EV-71/C2-2. Viral loads (B) and viral titers (C) are shown. c2c12-CAR cells were transfected with 10 ng/mL nCoV-L or M8, and then infected with CV-B5/JS417. Viral loads (D) and viral titers (E) are shown. HEp-2 cells were transfected with 10 ng/mL nCoV-L or M8, and then infected with PV-1/Sabin. Viral loads (F) and Viral titers (G) are shown. N = 3, bar indicates SD. ns, p > 0.05, *, p < 0.05, **, p < 0.01, ***, p < 0.005, ****, p < 0.001.
3.3. nCoV-L Protected Suckling Mice from Lethal EV71 Virus Infection
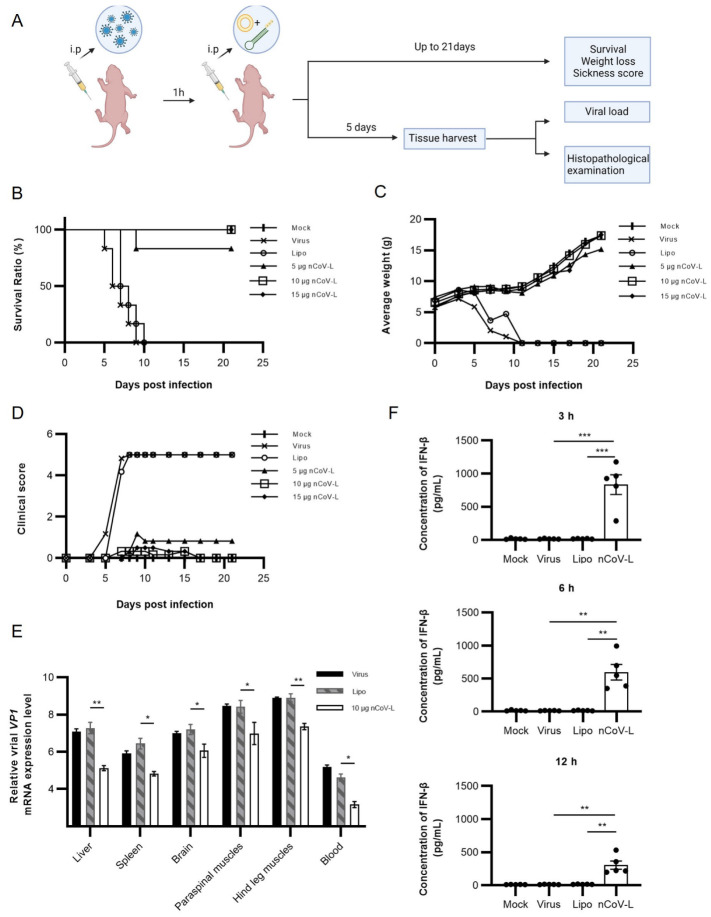
The stable lethal dosage of EV-71/C2-2 was determined to be 1.26 × 106 TCID50 in a seven-day BALB/c suckling mouse model (Figure A4A) so that the in vivo antiviral activity of nCoV-L could be quantitatively assessed. As the experimental procedure shows in Figure 3A, the lethal dosage of EV-71/C2-2 was injected intraperitoneally. One hour later, 5, 10, or 15 μg nCoV-L was intraperitoneally injected. Five days post-infection (DPI), mice from the PBS-treated group and the negative liposome control group developed obvious morbidity symptoms, such as convulsions and paralysis. All mice in these two groups died at 9 DPI (Figure 3B). Mice treated with 5 μg nCoV-L exhibited muscular paralysis of the rear legs at 5 DPI. One mouse died at 8 DPI. Mice treated with 10 or 15 μg nCoV-L developed mild symptoms, such as slow action at 5 DPI, which disappeared gradually within one week. No death occurred during the observation period. Therefore, treatment of 10 μg nCoV-L 1 h after viral infection prevented mouse death from lethal dosage infection of EV-71/C2-2.
Figure 3.
The in vivo antiviral effect of nCoV-L against EV71/C2-2. (A) Experimental scheme. 7-day-old BALB/c suckling mice (N = 6) were challenged intraperitoneally with 1.26 × 106 TCID50 EV-71/C2-2. Mice were injected intraperitoneally with PBS, Lipo, 5 μg, 10 μg, or 15 μg nCoV-L complexed with Lipo 1 h later. Survival (B), average weight (C), and sickness (D) were monitored or scored every two days up to 21 DPI. In a separate cohort (N = 3, 10 μg nCoV-L), blood, liver, spleen, brain tissues, paraspinal, and hind leg muscle were collected for virological and immunological analysis at 5 DPI. (E) Viral loads in the above tissues were measured by RT-qPCR. Suckling mice were injected intraperitoneally with PBS, 1.26 × 106 TCID50 EV-71/C2-2, Lipo or 10 μg nCoV-L complexed with Lipo. Blood was collected 3, 6, 12 h after injection (N = 5). (F) Concentration of IFN-β in serum were measured by ELISA. N = 5, Bar indicates SD. *, p < 0.05, **, p < 0.01, ***, p < 0.005.
Moreover, as shown in Figure 3A, the viral loads in different tissues of mice, including brains, livers, spleens, paraspinal muscles, hind leg muscles, and blood from each group, were evaluated at 5 DPI (Figure 3E). Viral loads in those tissues of mice from the 10 μg nCoV-L treated group were significantly lower (about 100-fold) than that in the control group (p < 0.05). EV71/C2-2 showed prominent tropism in muscle, and viral loads in the paraspinal muscles and hindquarters were significantly higher than that in the other tissues (p < 0.05). To directly assess the treatment effects of nCoV-L on immunopathology, histological analyses on HE-stained paraspinal and hind leg muscle sections from mice in each group were conducted. Muscular fibre necrosis and lymphocyte infiltration were observed in the paraspinal and hind leg muscles of mice treated with liposome. Mild muscular fibre necrosis, lymphocyte infiltration, and muscular fibres repair were observed in the nCoV-L-treated group (Figure A4D,E). The pathological experimental results inferred that nCoV-L had relatively good in vivo antiviral activity. Elisa assays were performed to analyse the IFN-β levels in mice treated with 10 μg nCoV-L for 3, 6, and 12 h. Compared with mice treated with liposome, the IFN-β level was significantly higher when treated with nCoV-L (p < 0.01) (Figure 3F). The highest IFN-β level was observed 3 h after treatment, then declined over time. In summary, nCoV-L effectively induced an IFN-I response and activated antiviral immunity in vivo.
3.4. Prophylactic Administration of nCoV-L Protects Mice from Lethal SARS-CoV-2 Virus Infection
The antiviral effect of nCoV-L against SARS-CoV-2 was also examined in this study. The lethal model constructed by McCray, et al. [28] was used. Briefly, K18-hACE2 mice were intranasally infected with 200 TCID50 of SARS-CoV-2/Delta. nCoV-L was injected intraperitoneally 6 h before or 0.5 h after infection (Figure 4A). The survival ratio and weight loss of the mice were shown in Figure 4B–D. Mice in the non-treated negative control group developed back arching and trembling symptoms at 4 DPI, and all mice died at 5 DPI (5/5). Only one mouse died when treated prophylactically with 30 μg nCoV-L (1/5) (Figure 4B), while the rest survived well. The prophylactic administration of nCoV-L not only protected mice from the challenge of a lethal dosage of SARS-CoV-2/Delta and relieved morbidity symptoms effectively but also inhibited weight loss in mice to some extent (Figure 4C). Administration of nCoV-L 0.5 h after virus infection protected mice from death (three out of five) but did not inhibit weight loss (Figure 4D). All surviving mice were killed by euthanasia at 5 DPI, when all mice in the control group died, to analyse the pathological state of lung tissue. Typical symptoms induced by SARS-CoV-2 infection, such as diffuse capillary congestion in the pulmonary vein and alveolar wall, necrosis and shedding of alveolar epithelial cells, and increased inflammatory cells in blood vessels, were found in lung tissues of mice without treatment [29,30]. Whereas prophylactic administration of 30 μg nCoV-L can protect mice from death, the surviving mice showed similar symptoms but with evidently less severe lesions (Figure 4E).
Figure 4.
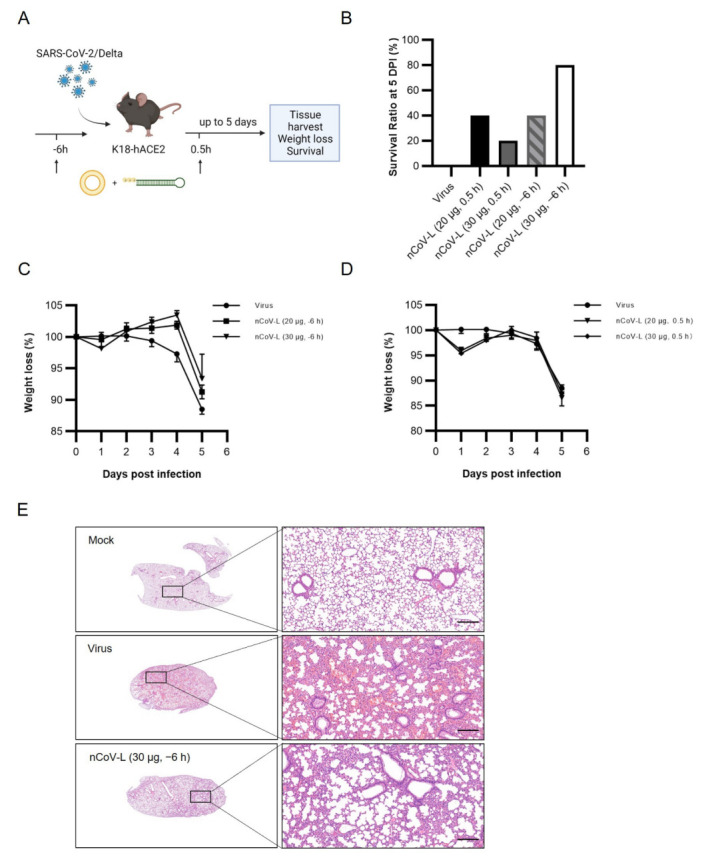
The in vivo antiviral effect of nCoV-L against SARS-CoV-2. (A) Experimental scheme. K18-hACE2 mice were intranasally infected with 200 TCID50 SARS-CoV-2/Delta. 20 μg or 30 μg nCoV-L complexed with Lipo were intraperitoneally injected 6 h before or 0.5 h after infection. Survival and weight loss were monitored daily up to 5 DPI when mice infected with virus were all dead. (B) Survival ratio of each group at 5 DPI are shown. (C) Weight loss of mice treated with nCoV-L or Lipo 6 h before infection. (D) Weight loss of mice treated with nCoV-L or Lipo 0.5 h after infection. Bar indicates SEM. (E) HE staining of lung from mock, PBS treated or 30 μg nCoV-L prophylactically treated mice at 5 DPI are shown. Scale bar represents 200 μm.
4. Discussion
Selection and reconstruction of sequences with hairpin structures from virus 5′ or 3′ UTRs are one of the primary research and development directions for developing RIG-I agonists [31]. A trefoil-type 5′PPP RNA fragment CV-B3 CL from the Human coxsackievirus B3 (CV-B3) virus inhibited the duplication of Dengue virus and Vesicular stomatitis Alagoas virus in vitro [32]. Chiang et al. [22] reported that M8, a fragment of 5′PPP RNA reconstructed from the Vesicular stomatitis Alagoas virus 5′UTR, had potent antiviral effects against multiple influenza viruses and Chikungunya virus in vivo and in vitro [33]. In this study, a fragment of 5′PPP RNA, nCoV-L, reconstructed from the SARS-CoV-2 5′UTR was designed and found to significantly inhibit infection of EV-71/C2-2, CV-B5/JS417, PV-1/Sabin and IAV/H9N2 in vitro (Figure 2 and Figure A3). In vivo, therapeutic administration of nCoV-L protected suckling mice from a lethal dosage of EV-71/C2-2 infection and mitigated morbidity symptoms (Figure 3). Prophylactic administration of nCoV-L also protected mice from a lethal dosage of SARS-CoV-2/Delta infection. The results demonstrate that nCoV-L has broad-spectrum antiviral effects. Recently, the role of activating the RLR/MAVS signalling pathways during DNA virus infection has been identified [34]. However, few studies have examined the effect of RIG-I agonists against DNA viruses, which will be investigated in further research.
This study examined the in vivo antiviral effects of nCoV-L against the SARS-CoV-2/Delta using the K18-hACE2 mouse lethal model. Prophylactic administration of nCoV-L generated a protective outcome to some extent. Pathologic changes were still observed in the lungs of the mice treated with nCoV-L; however, the symptoms were milder than those observed in the control group, indicating that the virus was not totally eliminated. This might be attributed to the immune escape of SARS-CoV-2/Delta, which encodes several proteins that inhibit the IFN-mediated signalling pathway [35,36]. The SARS-CoV-2 nucleocapsid protein (N) and membrane protein (M) were reported to play a role in suppressing host innate immunity by targeting the upstream event of RNA sensing [37,38]. SARS-CoV-2 non-structural protein 3 (NSP3) inhibits IFN-I production by cleaving the ubiquitin-like protein, ISG15, decreasing IRF3 phosphorylation, or directly cleaving IRF3 [39]. NSP1 can inhibit the translation of host mRNA, thus decreasing interferon and ISGs production [19]. Currently, the Omicron strain has overwhelmed the Delta strain to be the primary strain worldwide. Many known SARS-CoV-2 proteins inhibit the interferon response (NSP3, NSP6, NSP14, N, and M) have mutated in the Omicron strain. As a result, the Omicron strain provides weaker inhibition upon IFN signalling than Delta [39,40]. Thus, nCoV-L may provide a better antiviral effect against Omicron infection than Delta.
The experimental results also evaluated the effects of administration timing of nCoV-L on the antiviral outcomes in vivo. The results indicate that the in vivo protective effect of nCoV-L against EV-71/C2-2 virus is attenuated with delayed administration (Figure A4B,C). Consistent with the protective effect of SLR14, a similar result was observed when investigating the in vivo antiviral effect of nCoV-L against the SARS-CoV-2/Delta [41]. The therapeutic administration of nCoV-L could protect mice from the lethal dosage challenge of SARS-CoV-2/Delta and prolong survival time after viral infection. Prophylactic administration achieved a better protective effect, further reduced mouse mortality, and protected mice from weight loss after infection. It can be speculated that prophylactic administration of nCoV-L can induce a systematic interferon response to block the viral infection, thus improving the protective outcomes [18]. Hence, an appropriate immune strategy should be adopted to achieve a better protective effect.
This study proves that nCoV-L can activate the RIG-I receptor and induce interferon responses. Additionally, prophylactic administration of nCoV-L or administration in the early stage of infection protect mice from lethal viral infection. In conclusion, nCoV-L was found to generate a broad-spectrum antiviral effect by inducing an innate antiviral immune response, serving as a feasible antiviral strategy with great potential for future drug development prospects.
Acknowledgments
We thank the biosafety level 3 (ABSL3) facility of the Wuhan Institute of Biological Products Co., Ltd. for supplying the experimental platform. Figure 2A and Figure 3A, and Figure 4A were created with BioRender.com (accessed on 25 July 2022).
Appendix A
Table A1.
Sequences of knockout cell lines.
| Gene | Wild-Type Sequence | Post-Editing Sequence | Mutation Type |
|---|---|---|---|
| TLR3 | CAACTTTCTTGGGACTA----AAGTGGACAAATCTCAC | CAACTTTCTTGGGACTAATTTAAGTGGACAAATCTCAC | frame shift |
| TLR7 | AACTCTCTTCAGCATGTGCCCCCAAGATGGTTT | AACTCTCTT-------------CAAGATGGTTT | frame shift |
| TLR8 | ATTTAGTGGGAGAAATAGC--CTCTGGGGCATTTTT | ATTTAGTGGGAGAAATAGCGCCTCTGGGGCATTTTT | frame shift |
| RIG-I | AAAAAATTCCCACAAGGACAAAAGGGGAAAG | AAAAAATTCCCACAAGGACAA--GGGGAAAG | frame shift |
| MDA5 | CCCCGGAGCCAGAACTCCAGCTCAGGCCTTACCAA | CCCCGGAGCCAGAACTCC----CAGGCCTTACCAA | frame shift |
Figure A1.
Features of nCoV-L. (A) Schematic diagram of secondary structure of nCoV-L was predicted using the RNAfold Webserver. The loop is 4 nt and the stem is 96 nt. (B) In vitro transcribed nCoV-L and M8 were purified, and then run on a TAE gel with denaturing buffer.
Figure A2.
nCoV-L specifically activates the RIG-I receptor. (A) 1 × 104 wild-type (WT), RIG-I −/−, MDA5 −/−, TLR3 −/−, TLR7 −/−, TLR8 −/− HEK293 cells expressing ISG54-luciferase reporter gene were cultivated in 96-well plate for 12 h, and then transfected with 50 ng nCoV-L or poly(I:C) using Lipo, or treated with equal dosage of Lipo as the negative control. Relative luciferase activity was tested after 24 h. N = 3, RLU indicates relative light unit, bar indicates SD. ***, p < 0.005. (B) 2 × 106 A549 cells were transfected with 1 ng/mL nCoV-L, M8 or 100 ng/mL poly(I:C) using Lipo, cell lysis was prepared and subjected to Western blot analysis 24 h after transfection. 2 × 106 RIG-I −/− (C) or MDA5 −/− (D) HEK293 cells expressing ISG54-luciferase reporter gene were transfected with 1 μg/mL nCoV-L, 1 μg/mL poly(I:C) (+) or 2 μg/mL poly(I:C) (++) using Lipo. Cell lysis was prepared and subjected to Western blot analysis 24 h after transfection.
Figure A3.
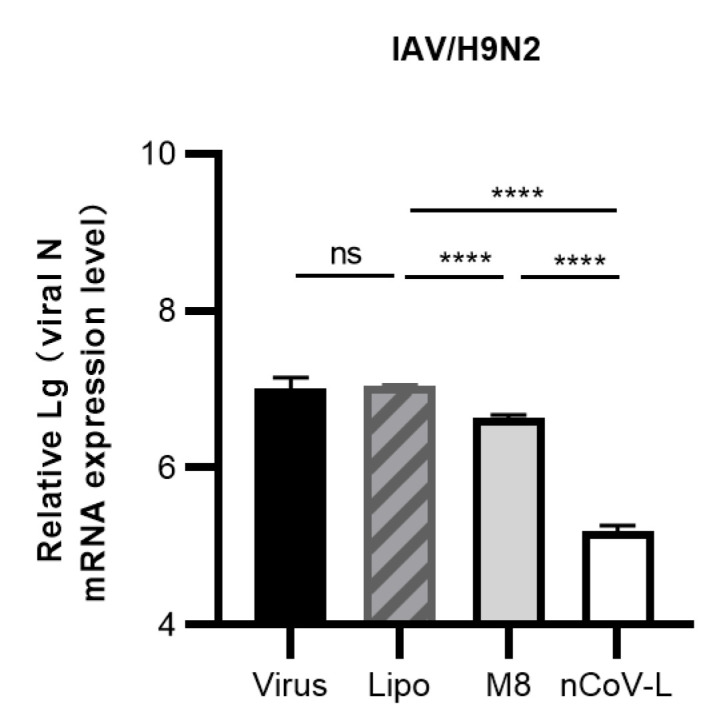
The in vitro antiviral effect of nCoV-L against IAV/H9N2. 2 × 105 A549 cells were transfected with 1 ng/mL nCoV-L or M8 for 24 h and then infected with IAV/H9N2 (1 MOI) for 24 h. Cells were harvested, and total RNA was extracted. Viral loads were measured by RT-qPCR. Viral loads are shown. N = 3, bar indicates SD. ns, p > 0.05, ****, p < 0.005.
Figure A4.
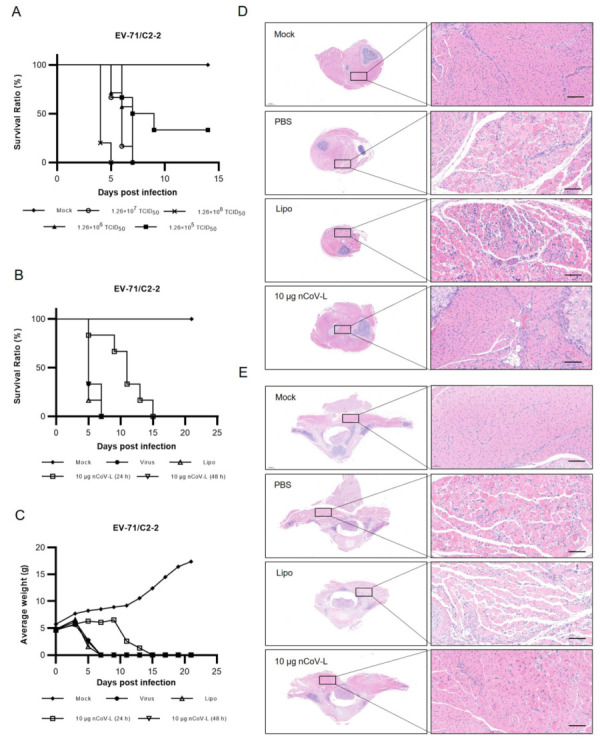
The therapeutic effect of nCoV-L against EV-71/C2-2 in vivo. (A) Seven-day-old BALB/c suckling mice (N = 6) were infected with 1.26 × 105, 1.26 × 106, 1.26 × 107 or 1.26 × 108 TCID50 EV-71/C2-2 intraperitoneally. (B,C) Suckling mice (N = 6) were challenged intraperitoneally with 1.26 × 106 TCID50 EV-71/C2-2. 10 μg nCoV-L complexed with Lipo were intraperitoneally injected 24 h, 48 h after infection. Survival ratio and average weight were monitored every two days up to 21 DPI. Suckling mice (N = 3) were challenged intraperitoneally with 1.26 × 106 TCID50 EV-71/C2-2, then injected intraperitoneally with 10 μg nCoV-L complexed with Lipo 1 h later. (D,E) HE staining of hind leg and paraspinal muscle from Mock, PBS, Lipo or 10 μg nCoV-L treated mice at 5 DPI are shown. Scale bar represents 100 μm.
Author Contributions
Conceptualisation, Z.L., X.L. and F.G.; methodology, Z.S., Q.W. and L.B.; formal analysis, Z.S., Q.W., L.B., C.A., B.C., Q.H., Y.B., J.L., L.S., D.L. and J.Z.; writing—original draft preparation, Z.S., Q.W. and L.B.; writing—review and editing, Z.L.; supervision, Q.M. and X.W.; funding acquisition, Z.L. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Experiments with live SARS-CoV-2 were performed in a biosafety level 3 (ABSL3) facility at the Wuhan Institute of Biological Products Co., Ltd. All research staff were trained and qualified in the experimental procedures. Animal procedures were approved by the Ethics Committee on Laboratory Animals of the Wuhan Institute of Biological Products Co., Ltd. (Approval number: WIBP-AII442021005). Other experiments with live viruses were performed in a biosafety level 2 (ABSL2) facility at the National Institutes for Food and Drug Control. Animal research protocols were approved by the Institutional Animal Care and Use Committee at the National Institutes for Food and Drug Control, China (Approval number: 2022-B020).
Informed Consent Statement
Not applicable.
Data Availability Statement
All data, generated or analysed, and materials during this study are included in this published article.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research was funded by CAMS Innovation Fund for Medical Sciences [grant numbers 2021-I2M-5-005].
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kanneganti T.-D. Intracellular innate immune receptors: Life inside the cell. Immunol. Rev. 2020;297:5–12. doi: 10.1111/imr.12912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barbalat R., Ewald S.E., Mouchess M.L., Barton G.M. Nucleic acid recognition by the innate immune system. Annu. Rev. Immunol. 2011;29:185–214. doi: 10.1146/annurev-immunol-031210-101340. [DOI] [PubMed] [Google Scholar]
- 3.Beutler B., Eidenschenk C., Crozat K., Imler J.L., Takeuchi O., Hoffmann J.A., Akira S. Genetic analysis of resistance to viral infection. Nat. Rev. Immunol. 2007;7:753–766. doi: 10.1038/nri2174. [DOI] [PubMed] [Google Scholar]
- 4.Schlee M., Hartmann G. The chase for the RIG-I ligand--recent advances. Mol. Ther. 2010;18:1254–1262. doi: 10.1038/mt.2010.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Goubau D., Schlee M., Deddouche S., Pruijssers A.J., Zillinger T., Goldeck M., Schuberth C., Van der Veen A.G., Fujimura T., Rehwinkel J., et al. Antiviral immunity via RIG-I-mediated recognition of RNA bearing 5′-diphosphates. Nature. 2014;514:372–375. doi: 10.1038/nature13590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Okamoto M., Kouwaki T., Fukushima Y., Oshiumi H. Regulation of RIG-I Activation by K63-Linked Polyubiquitination. Front. Immunol. 2017;8:1942. doi: 10.3389/fimmu.2017.01942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fujita T. A nonself RNA pattern: Tri-p to panhandle. Immunity. 2009;31:4–5. doi: 10.1016/j.immuni.2009.06.014. [DOI] [PubMed] [Google Scholar]
- 8.Bamming D., Horvath C.M. Regulation of signal transduction by enzymatically inactive antiviral RNA helicase proteins MDA5, RIG-I, and LGP2. J. Biol. Chem. 2009;284:9700–9712. doi: 10.1074/jbc.M807365200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kawai T., Takahashi K., Sato S., Coban C., Kumar H., Kato H., Ishii K.J., Takeuchi O., Akira S. IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nat. Immunol. 2005;6:981–988. doi: 10.1038/ni1243. [DOI] [PubMed] [Google Scholar]
- 10.Seth R.B., Sun L., Ea C.K., Chen Z.J. Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-kappaB and IRF 3. Cell. 2005;122:669–682. doi: 10.1016/j.cell.2005.08.012. [DOI] [PubMed] [Google Scholar]
- 11.Kato H., Takeuchi O., Akira S. Cell type specific involvement of RIG-I in antiviral responses. Nihon Rinsho. 2006;64:1244–1247. doi: 10.1016/j.immuni.2005.04.010. [DOI] [PubMed] [Google Scholar]
- 12.Onomoto K., Onoguchi K., Yoneyama M. Regulation of RIG-I-like receptor-mediated signaling: Interaction between host and viral factors. Cell. Mol. Immunol. 2021;18:539–555. doi: 10.1038/s41423-020-00602-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.González-Navajas J.M., Lee J., David M., Raz E. Immunomodulatory functions of type I interferons. Nat. Rev. Immunol. 2012;12:125–135. doi: 10.1038/nri3133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ivashkiv L.B., Donlin L.T. Regulation of type I interferon responses. Nat. Rev. Immunol. 2014;14:36–49. doi: 10.1038/nri3581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lin F.C., Young H.A. Interferons: Success in anti-viral immunotherapy. Cytokine Growth Factor Rev. 2014;25:369–376. doi: 10.1016/j.cytogfr.2014.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Goubau D., Deddouche S., Reis e Sousa C. Cytosolic sensing of viruses. Immunity. 2013;38:855–869. doi: 10.1016/j.immuni.2013.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rathinam V.A., Fitzgerald K.A. Inflammasomes and anti-viral immunity. J. Clin. Immunol. 2010;30:632–637. doi: 10.1007/s10875-010-9431-4. [DOI] [PubMed] [Google Scholar]
- 18.Linehan M.M., Dickey T.H., Molinari E.S., Fitzgerald M.E., Potapova O., Iwasaki A., Pyle A.M. A minimal RNA ligand for potent RIG-I activation in living mice. Sci. Adv. 2018;4:e1701854. doi: 10.1126/sciadv.1701854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bastard P., Rosen L.B., Zhang Q., Michailidis E., Hoffmann H.H., Zhang Y., Dorgham K., Philippot Q., Rosain J., Béziat V., et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. 2020;370:eabd4585. doi: 10.1126/science.abd4585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Giovannoni G., Munschauer F.E., 3rd, Deisenhammer F. Neutralising antibodies to interferon beta during the treatment of multiple sclerosis. J. Neurol. Neurosurg. Psychiatry. 2002;73:465–469. doi: 10.1136/jnnp.73.5.465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Matsuda F., Torii Y., Enomoto H., Kuga C., Aizawa N., Iwata Y., Saito M., Imanishi H., Shimomura S., Nakamura H., et al. Anti-interferon-α neutralizing antibody is associated with nonresponse to pegylated interferon-α plus ribavirin in chronic hepatitis C. J. Viral Hepat. 2012;19:694–703. doi: 10.1111/j.1365-2893.2012.01598.x. [DOI] [PubMed] [Google Scholar]
- 22.Chiang C., Beljanski V., Yin K., Olagnier D., Ben Yebdri F., Steel C., Goulet M.L., DeFilippis V.R., Streblow D.N., Haddad E.K., et al. Sequence-Specific Modifications Enhance the Broad-Spectrum Antiviral Response Activated by RIG-I Agonists. J. Virol. 2015;89:8011–8025. doi: 10.1128/JVI.00845-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hornung V., Ellegast J., Kim S., Brzózka K., Jung A., Kato H., Poeck H., Akira S., Conzelmann K.K., Schlee M., et al. 5′-Triphosphate RNA is the ligand for RIG-I. Science. 2006;314:994–997. doi: 10.1126/science.1132505. [DOI] [PubMed] [Google Scholar]
- 24.Kato H., Takeuchi O., Sato S., Yoneyama M., Yamamoto M., Matsui K., Uematsu S., Jung A., Kawai T., Ishii K.J., et al. Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature. 2006;441:101–105. doi: 10.1038/nature04734. [DOI] [PubMed] [Google Scholar]
- 25.Pichlmair A., Schulz O., Tan C.P., Näslund T.I., Liljeström P., Weber F., Reis e Sousa C. RIG-I-mediated antiviral responses to single-stranded RNA bearing 5′-phosphates. Science. 2006;314:997–1001. doi: 10.1126/science.1132998. [DOI] [PubMed] [Google Scholar]
- 26.Jiang M., Zhang S., Yang Z., Lin H., Zhu J., Liu L., Wang W., Liu S., Liu W., Ma Y., et al. Self-Recognition of an Inducible Host lncRNA by RIG-I Feedback Restricts Innate Immune Response. Cell. 2018;173:906–919.e913. doi: 10.1016/j.cell.2018.03.064. [DOI] [PubMed] [Google Scholar]
- 27.Brisse M., Ly H. Comparative Structure and Function Analysis of the RIG-I-Like Receptors: RIG-I and MDA5. Front. Immunol. 2019;10:1586. doi: 10.3389/fimmu.2019.01586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McCray P.B., Jr., Pewe L., Wohlford-Lenane C., Hickey M., Manzel L., Shi L., Netland J., Jia H.P., Halabi C., Sigmund C.D., et al. Lethal infection of K18-hACE2 mice infected with severe acute respiratory syndrome coronavirus. J. Virol. 2007;81:813–821. doi: 10.1128/JVI.02012-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zheng J., Wong L.R., Li K., Verma A.K., Ortiz M.E., Wohlford-Lenane C., Leidinger M.R., Knudson C.M., Meyerholz D.K., McCray P.B., Jr., et al. COVID-19 treatments and pathogenesis including anosmia in K18-hACE2 mice. Nature. 2021;589:603–607. doi: 10.1038/s41586-020-2943-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Winkler E.S., Bailey A.L., Kafai N.M., Nair S., McCune B.T., Yu J., Fox J.M., Chen R.E., Earnest J.T., Keeler S.P., et al. SARS-CoV-2 infection of human ACE2-transgenic mice causes severe lung inflammation and impaired function. Nat. Immunol. 2020;21:1327–1335. doi: 10.1038/s41590-020-0778-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schlee M., Roth A., Hornung V., Hagmann C.A., Wimmenauer V., Barchet W., Coch C., Janke M., Mihailovic A., Wardle G., et al. Recognition of 5′ triphosphate by RIG-I helicase requires short blunt double-stranded RNA as contained in panhandle of negative-strand virus. Immunity. 2009;31:25–34. doi: 10.1016/j.immuni.2009.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Feng Q., Langereis M.A., Olagnier D., Chiang C., van de Winkel R., van Essen P., Zoll J., Hiscott J., van Kuppeveld F.J. Coxsackievirus cloverleaf RNA containing a 5′ triphosphate triggers an antiviral response via RIG-I activation. PLoS ONE. 2014;9:e95927. doi: 10.1371/journal.pone.0095927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Goulet M.L., Olagnier D., Xu Z., Paz S., Belgnaoui S.M., Lafferty E.I., Janelle V., Arguello M., Paquet M., Ghneim K., et al. Systems analysis of a RIG-I agonist inducing broad spectrum inhibition of virus infectivity. PLoS Pathog. 2013;9:e1003298. doi: 10.1371/annotation/8fa70b21-32e7-4ed3-b397-ab776b5bbf30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jia J., Fu J., Tang H. Activation and Evasion of RLR Signaling by DNA Virus Infection. Front. Microbiol. 2021;12:804511. doi: 10.3389/fmicb.2021.804511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Konno Y., Kimura I., Uriu K., Fukushi M., Irie T., Koyanagi Y., Sauter D., Gifford R.J., Nakagawa S., Sato K. SARS-CoV-2 ORF3b Is a Potent Interferon Antagonist Whose Activity Is Increased by a Naturally Occurring Elongation Variant. Cell Rep. 2020;32:108185. doi: 10.1016/j.celrep.2020.108185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Setaro A.C., Gaglia M.M. All hands on deck: SARS-CoV-2 proteins that block early anti-viral interferon responses. Curr. Res. Virol. Sci. 2021;2:100015. doi: 10.1016/j.crviro.2021.100015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lu X., Pan J., Tao J., Guo D. SARS-CoV nucleocapsid protein antagonizes IFN-β response by targeting initial step of IFN-β induction pathway, and its C-terminal region is critical for the antagonism. Virus Genes. 2011;42:37–45. doi: 10.1007/s11262-010-0544-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yamada T., Sato S., Sotoyama Y., Orba Y., Sawa H., Yamauchi H., Sasaki M., Takaoka A. RIG-I triggers a signaling-abortive anti-SARS-CoV-2 defense in human lung cells. Nat. Immunol. 2021;22:820–828. doi: 10.1038/s41590-021-00942-0. [DOI] [PubMed] [Google Scholar]
- 39.Xu D., Biswal M., Neal A., Hai R. Review Devil’s tools: SARS-CoV-2 antagonists against innate immunity. Curr. Res. Virol. Sci. 2021;2:100013. doi: 10.1016/j.crviro.2021.100013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bojkova D., Widera M., Ciesek S., Wass M.N., Michaelis M., Cinatl J., Jr. Reduced interferon antagonism but similar drug sensitivity in Omicron variant compared to Delta variant of SARS-CoV-2 isolates. Cell Res. 2022;32:319–321. doi: 10.1038/s41422-022-00619-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mao T., Israelow B., Lucas C., Vogels C.B.F., Gomez-Calvo M.L., Fedorova O., Breban M.I., Menasche B.L., Dong H., Linehan M., et al. A stem-loop RNA RIG-I agonist protects against acute and chronic SARS-CoV-2 infection in mice. J. Exp. Med. 2022;219:e20211818. doi: 10.1084/jem.20211818. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data, generated or analysed, and materials during this study are included in this published article.