Abstract
The use of antibiotics in farm animals is one of the main reasons for the development of resistant bacterial strains (e.g., zoonotic pathogens). Therefore, save alternatives are needed. Here, we examined how post-hatch application (day one to seven of life) of the probiotic Enterococcus faecium AL41 (EF) affects the development and tissue properties of the broiler pectoralis major muscle (PM). Expression of regulators, namely IGF-1, PAX7, and MYF5, was also investigated. At day 1 (n = 6), and days 5, 8, and 12 (n = 10), muscle samples were taken from control and EF supplemented chicks. From day 5 on, myonuclei number per fiber was elevated in EF chicks. Improved capillarization (from day 8), larger myofibers, increased body and PM weights (day 12) were found in the EF group. Part of our findings is explainable by higher intramuscular expression of IGF-1 and lower MYF5 expression in EF birds. In both groups IGF-1 expression decreases with age, thereby increasing the cellular myogenic potential. However, a strong increase in PAX7 expression and more PAX7-positive nuclei were found in EF chicks at day 12. We conclude that EF supplementation improves PM growth and health due to positive effects on bioavailability and fusion capacity of SATC progeny and better tissue perfusion.
Keywords: poultry, probiotics, Enterococcus faecium, muscle tissue, IGF-1, myogenic regulatory factors, PAX7, antibiotics replacement
1. Introduction
Over the last few decades, the consumption of chicken meat has increased at the same time as consumer demands regarding its quality and safety have been increasing. [1,2].
To meet the growing demand for chicken meat, traditional forms of husbandry have over time been replaced by scaled industrial production systems in which the animals live mostly or completely indoors during their life span. Associated negative effects relate in particular to the intestinal microbiome [3]. Chicks hatched in incubators show a delayed colonization of the gut with commensal bacteria [4,5] and, later on, density and complexity of the population is reduced [6]. Consequently, a lower resistance to pathogens, disturbed nutrient absorption, higher mortality rates, and production costs (mainly due to a reduced feed conversion rate) have been increasingly observed [7,8].
For a long time, attempts were made to solve these problems by regular application of low, sub-therapeutic amounts of antibiotics to animals [9]. These antimicrobial growth promoters (AGPs) reduced the occurrence of gastrointestinal infections and promoted feed efficiency, growth performance, and, thereby, meat mass [7,10,11].
However, the intensive use of antibiotics at sub-therapeutic doses has contributed to development of resistant bacterial strains in commensals and zoonotic pathogens [12,13]. Thus, to prevent negative effects on human health, AGP’s have been banned in the European Union since 2006. However, the use of therapeutic antibiotics for farm animals increased significantly thereafter [3,14].
Consequently, there is growing global interest in effective and save AGP replacements enabling human and animal health, food security, and efficiency of production. Various alternatives have been proposed and tested, among them the application of protective microbiota [15,16] such as probiotics (live microbial feed supplements).
Per definition, probiotics are “products that contain sufficient values of viable bacteria that can improve the intestinal microbial balance of the host resulting in detectable beneficial health effects” [16,17]. Various bacteria such as Lactobacillus spp., Bacillus spp., Bifidobacterium spp., Streptococcus spp., and Enterococcus spp. are known to have probiotic properties when added to poultry diets [5,7,8,10,18,19,20]. In addition, beneficial effects on poultry growth performance and on feed utilization efficiency have been reported for various probiotics [5,7,8,20,21] but results show a higher variability than with AGPs [10,22].
The probiotic species Enterococcus faecium (EF) suppress colonization and proliferation of enteric bacteria among them Salmonella Enteritidis (SE) [15,19,23,24,25] and Listeria innocua HPB13 [16]. In addition, EF strains are able to produce antimicrobial substances, so-called bacteriocins [16,19,25]. However, in contrast to antibiotics, the killing spectrum of EF bacteriocins is relative narrow and their toxicity is only directed against close relatives of the producing strain [25,26]. Early post-hatch administration of EF to chicken reduced cecal colonization with pathogenic SE, stimulate innate and acquired immune responses [16,27,28], stabilized the protective function of the intestinal epithelial barrier, and promotes small intestine development [20,24,25,29]. Thus, at least in part, a higher absorptive capacity for nutrients, specifically for glucose, will contribute to the positive effects of EF strains on growth performance [20,29].
However, economically, the growth and quality of the skeletal muscle is most significant for broiler meat production [30]. Modern broiler strains are selected for fast muscle growth and, in particular, an accelerated growth of the pectoralis muscle (PM) [31].
The number of muscle fibers is already established at hatch [32]. Therefore, muscle hypertrophy occurring by the accretion of protein and new myonuclei to existing myofibers is responsible for post-hatch muscle growth [33]. New myonuclei for this process originate from so-called satellite cells (SATC) [34,35,36]. The majority of SATC is quiescent in adult animals, and can be found adjacent to myofibers in a niche underneath their basement membrane [33]. In contrast, SATC show a high proliferative capacity in growing broiler muscles reaching a peak activity already between days two and four after hatching [37,38]. After differentiation, most SATC fuse to existing myofibers to enlarge their diameter [30,34,39] whereas some SATC self-renew to replenish their own population [40,41]. A group of myogenic genes regulates SATC bioavailability and their molecular and functional properties. During quiescence, paired box protein 7 (PAX7) and the determination gene myogenic factor 5 (MYF5) are expressed [41,42,43]. In activated, proliferating cells (myoblasts) determination protein 1 (MYOD) is induced [44,45]. Downregulation of PAX7 and induction of myogenin (MYOG) characterize the process of early differentiation (myocytes) whereas sarcomeric myosin heavy chain (MYHC) is expressed in terminally differentiated cells [44,45].
However, the molecular and functional phenotype of SATC can also be affected by environmental stimuli (e.g., nutrients or growth factors) [46,47]. Short-term reductions in SATC mitotic activity during the early post-hatch period (e.g., due to feed deprivation or high ambient temperature) decrease muscle size at maturity, showing its critical role for maintaining SATC activity and subsequent muscle growth [33,38,48]. In our previous study [49], early post-hatch SE infection induced dramatic negative effects in muscle tissue, among them permanently reduced myofiber cross sectional area (FCSA) and disturbed capillarization. However, preventive application of the EF strain AL41 completely restored blood supply pointing to more direct effects of EF AL41 on muscle development [49]. To our knowledge, there is no experimental work on the effect of oral probiotics on processes of early postnatal muscle development. However, we think that such information is prerequisite for commercial application of probiotics.
Therefore, in addition to morphological and biochemical properties of muscle tissue, the current study examines if and how selected regulatory systems of muscle development will be affected by a seven-day post-hatch EF AL41 treatment. Our results show clear positive effects of probiotic application on daily weight gain, body and PM weight accompanied by improved hypertrophic myofiber growth and capillarization. In addition, our study provides new findings showing that the intramuscular IGF-1 system and main regulators of SATC functionality, namely the myogenic regulatory factors PAX7 and MYF5, are modulated by EF treatment giving a possible explanation for positive effects of the probiotic on growth performance.
2. Materials and Methods
2.1. Animals
The chicks were handled and killed according to the Slovak state regulations. The experiment was approved by the Ethics Committee of the University of Veterinary Medicine and Pharmacy, Košice, Slovakia, and by the Committee for Animal Welfare of the Ministry of Agriculture of the Slovak Republic (permission number 1184-3/2020-220).
Eighty one-day-old COBB 500 male chicks were randomly divided into two groups, a control group (Con) and a treatment group, respectively. Birds of the treatment group were inoculated with the probiotic strain Enterococcus faecium (E. faecium) AL41 (CCM 8558) as described by Zitnan et al. [49]. The probiotic strain E. faecium AL41 (EF) was grown as described by Letnická et al. [50]. From the first to the seventh day of the experiment, a suspension with 109 colony-forming units of EF in 0.2 mL PBS was applied to every chicken of the treatment group per os. To simulate the same stress to chicken of the control group, an equal volume of PBS only was applied with a Pasteur pipette. The chicks were placed in hardwood pens with wood shavings and fed a standard BR-1 compound feed. The diet composition has been published in [51]. Access to water was provided ad libitum. Housing conditions were chosen according to Cobb-Vantress Inc. guidelines [52]. Prior to the start of the experiment, faecal control samples were taken from the chickens for microbiological examination.
For the present experiments, six birds per group were slaughtered at the beginning of the experiment (day one of life). Subsequently, 10 birds per group were slaughtered at days 5, 8, and 12 of life. Body weights were recorded at slaughter and pectoralis muscle (PM), liver, stomach, and intestine were weighed and collected. Muscle tissue was cut into pieces that were separately frozen in liquid nitrogen for biochemical and histological analyses. Samples from four randomly selected animals per group were immediately placed in RNA Later solution (Qiagen, Manchester, UK) for gene expression analyses and stored at −70 °C until further processing.
2.2. Biochemical Analyses
Frozen muscle samples were prepared as described by Zitnan et al. [49] for determination of DNA, RNA, and protein content, as well as enzyme activities. All muscle homogenates were diluted (1:20), then centrifuged at 14,000× g (15 min, 4 °C), and the resulting supernatants were kept on ice. All enzyme activity assays and the determination of the protein content were conducted the same day. The content of DNA and RNA in diluted supernatants (1:2 and 1:50, respectively) were measured using Hoechst 33258 and SYBR Green II, against respective standards (all Sigma-Aldrich, Steinheim, Germany) in an Flx-800-I microplate fluorescence reader (Bio-Tek Instruments, Bad Friedrichshall, Germany) as described earlier [49]. The protein content was analysed in 1:50 diluted supernatants according to Peterson [53]. Enzyme activities of creatine kinase (CK, EC 2.7.3.2), isocitrate dehydrogenase (ICDH, EC 1.1.1.42), and lactate dehydrogenase (LDH, EC 1.1.1.28) were determined as described by Lösel et al. [54]. Supernatants from 1-day-old chicken were diluted 1:100 and all others 1:200 for determination of CK activity using a commercial kit (CK-NAC-Hit kit, IFCC method, BIOMED Labordiagnostik GmbH, Oberschleißheim, Germany) adapted for microplates. Activities of ICDH and LDH were analysed as described by Zitnan et al. [49] in undiluted or diluted (1:5 at d 1, 1:15 at d 5, 1:30 at d 8 and 12) supernatants, respectively. A Spectramax Plus384 spectrophotometer/plate reader (Molecular Devices, Sunnyvale, CA, USA) was used for quantification.
2.3. Histomorphological Analysis
PM samples were cut 10 µm thick at −20 °C using a cryostat microtome Leica CM3050 S (Leica, Wetzlar, Germany). Sections were stained and analysed as described by Zitnan et al. [49] using standard protocols for hematoxylin/eosin (HE) [55] and for capillaries with eosin and alkaline phosphatase (EAP) [56]. At least three randomly selected images per sample were taken using an Olympus BX43 microscope (Olympus, Hamburg, Germany) equipped with a UC30 colour camera and Cell^D imaging software (OSIS, Münster, Germany). Muscle fiber und nuclei number were counted in HE stained slides using the interactive measurement function of Cell^D. The muscle fiber size, capillary size and density (number per mm2) were determined in EAP-stained slides with a self-made macro program as described by [49]. The ratio between counted numbers of myonuclei and muscle fibers was calculated as nuclei/fiber. The size of muscle fibers was an estimated muscle fiber cross sectional area (FCSA) determined by dividing the analyzed area by the number of muscle fibers. The total area analysed per sample was about 25,900 µm2 at d 1, 36,970 µm2 at d 5, 60,263 µm² at d 8, and 94,567 µm2 at d 12.
2.4. Gene Expression Analysis
Gene expression of IGF1, PAX7, MYF5, and GAPDH (glyceraldehyde-3-phosphate dehydrogenase) was determined. Based on confirmed expression stability using the Bestkeeper program, GAPDH was selected as the reference gene. About 20 mg muscle tissue was used for RNA purification and transcription as described by Karaffová et al. [57].
Amplification and detection of target products were performed using the CFX 96 RT system (Bio-Rad, Hercules, CA, USA) and Maxima SYBR Green qPCR Master Mix (Thermo Scientific, Waltham, MA, USA). Subsequent RT-qPCR to detect relative mRNA abundance was performed for 36 cycles under the following conditions: initial denaturation at 95 °C for 2 min, subsequent denaturation at 95 °C for 15 s, annealing (as listed in Table 1) and extension step 2 min at 72 °C. A melting curve from 50 °C to 95 °C with readings at every 0.5 °C was recorded for each individual RT-qPCR plate. Analysis was performed after every run to ensure a single amplified product for each reaction. All reactions for qPCR were carried out in duplicate. The primer sequences, annealing temperatures and times for each primer used for RT-qPCR are listed in Table 1. All primer sets allowed cDNA amplification efficiencies between 94% and 100%. It could be confirmed that the efficiency of amplification for each target gene (including GAPDH) was essentially 100% in the exponential phase of the reaction, where the quantification cycle (Cq) was calculated. The Cq values of the studied genes were normalised to an average Cq value of the reference gene (ΔCq), and the relative expression of each gene was calculated as 2−ΔCq. The relative gene expression in muscle of control chicken at day one was set to 1 to calculate fold changes for the values at the other ages and in the EF group.
Table 1.
List of primers used for the chicken mRNA quantification.
| Primer | Sequence 5′–3′ | Annealing Temperature/Time | References |
|---|---|---|---|
| IGF1 Fw | GAGCTGGTTGATGCTCTTCAGTT | 60 °C/1 min | Xiao et al., 2017 [58] |
| IGF1 Rev | CCAGCCTCCTCAGGTCACAACT | ||
| MYF5 Fw | CAGAGACTCCCCAAAGTGGAGAT | 60 °C/1 min | |
| MYF5 Rev | GTCCCGGCAGGTGATAGTAGTTC | ||
| PAX7 Fw | AGGCTGACTTCTCCATCTCTCCT | 60 °C/1 min | Adhikari et al., 2019 [59] |
| PAX7 Rev | TGTAACTGGTGGTGCTGTAGGTG | ||
| GAPDH Fw | CCTGCATCTGCCCATTT | 59 °C/30 s | De Boever et al., 2008 [60] |
| GAPDH Rev | GGCACGCCATCACTATC |
2.5. Immunohistochemistry and Image Analysis
Muscle tissue of each two animals per age and treatment group were randomly selected in addition to all samples of 12-day old chickens for immunohistochemistry and quantification of PAX7 positive cells. Tissue was cut 10 µm thick with a cryostat microtome Leica CM3050 S (Leica, Wetzlar, Germany). Fixation with 4% paraformaldehyde in PBS, was followed by washing three times for 10 min with PBS including Tween 20 (PBST) and a blocking step with 10% normal goat serum (NGS) and 10% BSA in PBS for 30 min. Incubation over night with the primary antibody against PAX7 (Developmental Studies Hybridoma Bank, Iowa, USA; 1:50 in PBS with 1% BSA) was followed by three times wash with PBST and incubation with the secondary antibody Alexa Fluor 594 goat anti-mouse IgG H & L (1:500, Thermo Fischer Scientific, Waltham, MA, USA) for 1.5 h at room temperature. Slides were washed three times with PBS for 5 min and covered with Roti Fluor Care DAPI (Roth, Karlsruhe, Germany) to counterstain all nuclei. A negative control, omitting the primary antibody, was processed to detect unspecific binding of the secondary antibody. No unspecific staining was observed. Immunofluorescence of PAX7 and DAPI-stained nuclei was detected using a Nikon Microphot SA fluorescence microscope (Nikon, Duesseldorf, Germany) equipped with a CC-12 color camera (OSIS, Münster, Germany) and Cell^F image analysis software (OSIS). A macro program was developed to count the total number of nuclei (stained blue) and the number of PAX7-positive nuclei (stained red) as follows. The blue image of total nuclei was opened, a shading correction was performed, the blue channel extracted, followed by a mean filter. A region of interest (ROI) was defined. An ultimate erosion followed by two dilatation steps enabled separation of closely located nuclei. Nuclei were then detected by a threshold operation and counted, after manual correction/deletion of false detected particles, such as artifacts. Afterwards, the red image of PAX7-positive nuclei was opened, the contrast was enhanced, the red channel extracted and the nuclei detected by threshold operation. False detected particles were deleted interactively and the results were saved. Ten images per animal were analyzed with a total area of about 2.37 mm2. For each animal, the number of nuclei per mm², the number of PAX7 nuclei per mm2, the percentage of PAX7 nuclei, the number of nuclei per muscle fiber and the number of PAX7 nuclei per muscle fiber were calculated.
2.6. Statistical Analysis
Statistical analysis was performed using the ANOVA model of SAS statistical software (Version 9.4, SAS Inst., Cary, NC, USA). Data was analyzed with the MIXED procedure with fixed factors treatment (Con and EF), age (1, 5, 8, and 12 d), and their interaction. All data are presented as least square means (LSM) and standard error (SE). As a post-hoc test, the Tukey-Kramer correction was used. The SLICE statement of the MIXED procedure was applied to enable the partitioned analysis of the effect of the treatment within the same age. A p-value of <0.05 was considered statistically significant and 0.05 < p < 0.1 a trend.
3. Results
3.1. Weight Development
In both experimental groups, slaughter weight as well as weights of the PM and inner organs increased from day 1 to day 12 of life (p < 0.001, Table 2). The fast growth during the first days of life was reflected by an increasing daily gain from day five to day eight of age (p < 0.001, Table 2). The growth slowed down thereafter, as indicated by a lower daily gain for the 12 day group. Over all ages, the daily gain was significantly higher in EF compared to Con chicks (19.8 vs. 18.0 g/d, p > 0.001). Control and EF-supplemented chicks developed similarely up to the eighth day of life. However, at day 12 of age, EF chicks had a higher slaughter (p = 0.015) and PM (p = 0.009) weight than Con chicks (Table 2). The livers of Con chicks were heavier at the 5th and 12th days of age (p < 0.001 and p = 0.038, respectively) whereas the stomach weight was greater in EF chicks at day 5 of age (p = 0.009). Regarding the intestinal weights no group differences (p > 0.05) were found.
Table 2.
Daily weight gain, body weight, muscle weight and organ development of control and E. faecium AL41 (EF)-treated chicks between day 1 and 12 of life.
| Control | EF | p-Value | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Day of Age | 1 (n = 6) |
5 (n = 10) |
8 (n = 10) |
12 (n = 10) |
1 (n = 6) |
5 (n = 10) |
8 (n = 10) |
12 (n = 10) |
SE d 1 | SE d 5–12 | Age | Group | Age × Group |
| Daily gain, g | 14.0 c | 22.8 a | 17.3 b | 14.9 c | 24.1 a | 18.4 b | 0.39 | <0.001 | <0.001 | 0.919 | |||
| Weights, g | |||||||||||||
| Slaughter weight | 43.2 d | 112.7 c | 224.4 b | 250.5 a,B | 43.8 d | 116.8 c | 233.1 b | 261.4 a,A | 4.0 | 3.1 | <0.001 | 0.012 | 0.456 |
| Pectoralis muscle | 1.74 d | 8.26 c | 35.66 b | 58.54 a,B | 1.64 d | 8.19 c | 37.21 b | 62.63 a,A | 1.39 | 1.07 | <0.001 | 0.100 | 0.212 |
| Liver | 2.36 c | 7.47 b,A | 11.32 a | 12.46 a,A | 1.69 c | 4.50 b,B | 10.67 a | 11.21 a,B | 0.54 | 0.42 | <0.001 | <0.001 | 0.030 |
| Stomach | 5.01 b | 4.77 b,B | 12.34 a | 11.31 a | 4.32 b | 6.53 b,A | 11.14 a | 11.64 a | 0.60 | 0.46 | <0.001 | 0.901 | 0.014 |
| Intestine | 5.62 c | 9.88 b | 21.61 a | 24.46 a | 4.90 c | 9.28 b | 22.13 a | 22.39 a | 1.04 | 0.80 | <0.001 | 0.246 | 0.464 |
a–d Different lower case letters indicate significant differences among ages (p < 0.05). A,B Different upper case letters indicate significant differences between groups (p < 0.05).
3.2. DNA, RNA, Protein and Muscle Enzymes
Table 3 displays changes of DNA, RNA, and protein amounts in PM between day 1 and 12 of age in control and EF-treated chicks. In both groups, total DNA, RNA, and protein increased (p > 0.05). The DNA/protein and RNA/protein ratios were highest at d 5 in both groups (p < 0.05).
Table 3.
Changes of DNA, RNA, and protein contents and concentrations as well as enzyme activities in pectoralis muscle of control and E. faecium AL41 (EF) supplemented chicks during day 1 to 12 of age.
| Control | EF | p-Value | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Day of Age | 1 (n = 6) |
5 (n = 10) |
8 (n = 10) |
12 (n = 10) |
1 (n = 6) |
5 (n = 10) |
8 (n = 10) |
12 (n = 10) |
SE d 1 | SE d 5–12 | Age | Group | Age × Group |
| Total DNA, mg | 0.48 d | 3.03 c | 7.72 b | 11.38 a | 0.37 d | 2.95 c | 7.58 b | 12.36 a | 0.34 | 0.27 | <0.001 | 0.435 | 0.123 |
| Total RNA, mg | 2.54 d | 20.42 c | 48.56 b | 59.78 a,B | 2.16 d | 19.80 c | 44.34 b | 67.04 a,A | 1.96 | 1.52 | <0.001 | 0.660 | 0.003 |
| Total protein, mg | 80 c | 501 c | 2551 b | 4620 a | 78 c | 482 c | 2753 b | 4961 a | 131 | 102 | <0.001 | 0.098 | 0.276 |
| DNA/RNA | 0.188 a,A | 0.148 b | 0.159 b,B | 0.191 a | 0.170 b,B | 0.149 c | 0.171 b,A | 0.184 a | 0.003 | 0.003 | <0.001 | 0.138 | <0.001 |
| RNA/DNA | 5.32 c,B | 6.76 a | 6.28 b,A | 5.25 c | 5.93 b,A | 6.74 a | 5.83 bc,B | 5.44 c | 0.11 | 0.09 | <0.001 | 0.249 | <0.001 |
| DNA, µg/g | 273 b,A | 366 a | 217 c | 195 d | 226 b,B | 360 a | 204 c | 197 c | 5 | 4 | <0.001 | <0.001 | <0.001 |
| RNA, µg/g | 1453 b | 2474 a | 1362 b,A | 1024 c | 1340 b | 2423 a | 1192 bc,B | 1071 c | 44 | 34 | <0.001 | 0.008 | 0.020 |
| Protein, mg/g | 45.9 d | 60.7 c | 71.6 b | 79.0 a | 47.3 c | 58.8 c | 74.0 b | 79.1 a | 1.6 | 1.2 | <0.001 | 0.588 | 0.327 |
| DNA/protein, µg/mg | 5.98 a,A | 6.04 a | 3.03 b | 2.46 c | 4.79 b,B | 6.13 a | 2.76 c | 2.50 c | 0.12 | 0.09 | <0.001 | <0.001 | <0.001 |
| RNA/protein, µg/mg | 31.8 b | 40.8 a | 19.1 c,A | 12.9 d | 28.4 b | 41.3 a | 16.1 c,B | 13.6 c | 0.8 | 0.6 | <0.001 | 0.009 | 0.004 |
| Protein/RNA, mg/µg | 0.032 c | 0.025 c | 0.053 b,B | 0.077 a | 0.036 c | 0.024 d | 0.063 b,A | 0.074 a | 0.002 | 0.001 | <0.001 | 0.019 | <0.001 |
| Total CK, IU | 1591 c | 13,347 c | 65,827 b | 116,724 a | 1612c | 12,557 c | 67,609 b | 126,644 a | 3553 | 2752 | <0.001 | 0.198 | 0.218 |
| CK, IU/g | 919 c | 1622 b | 1845 ab | 1994 a | 968 c | 1540 b | 1822 a | 2026 a | 70 | 54 | <0.001 | 0.882 | 0.668 |
| CK/protein, IU/mg | 20.2 b | 26.8 a | 25.7a | 25.2a | 20.4b | 26.3a | 24.6 a | 25.7 a | 1.0 | 0.8 | <0.001 | 0.703 | 0.751 |
| ICDH, IU/g | 3.12 a | 2.28 b | 1.41 c | 1.25 c | 2.96 a | 2.31 b | 1.39 c | 1.13 c | 0.09 | 0.07 | <0.001 | 0.218 | 0.533 |
| LDH, IU/g | 43 d | 330 c | 751 b | 929 a | 54 d | 315 c | 765 b | 892 a | 29 | 22 | <0.001 | 0.694 | 0.658 |
| ICDH/protein, IU/mg | 0.068 a | 0.038 b | 0.020 c | 0.016 c | 0.062 a | 0.039 b | 0.019 c | 0.014 c | 0.001 | 0.001 | <0.001 | 0.070 | 0.040 |
| LDH/protein, IU/mg | 0.93 d | 5.42 c | 10.5 b | 11.74 a | 1.14 c | 5.36 b | 10.34 a | 11.28 a | 0.32 | 0.25 | <0.001 | 0.536 | 0.695 |
| LDH/ICDH | 14 d | 146 c | 540 b | 748 a | 19 d | 138 c | 554 b | 796 a | 29 | 23 | <0.001 | 0.394 | 0.660 |
a–d Different lower case letters indicate significant differences among ages (p < 0.05). A,B Different upper case letters indicate significant differences between groups (p < 0.05).
Activity of CK and LDH increased with age (p < 0.05), whereas the ICDH activity decreased (p < 0.05) in both groups. No group differences in the activity of investigated muscle enzymes were observed. The increase of LDH/ICDH ratio with age was also similar in both groups.
3.3. Muscle Structure
Next, myonuclei density, fiber cross sectional area (FCSA), myonuclei per fiber and capillarization of PM were determined in control and EF treated chicks. The results are summarized in Figure 1 and Table 4.
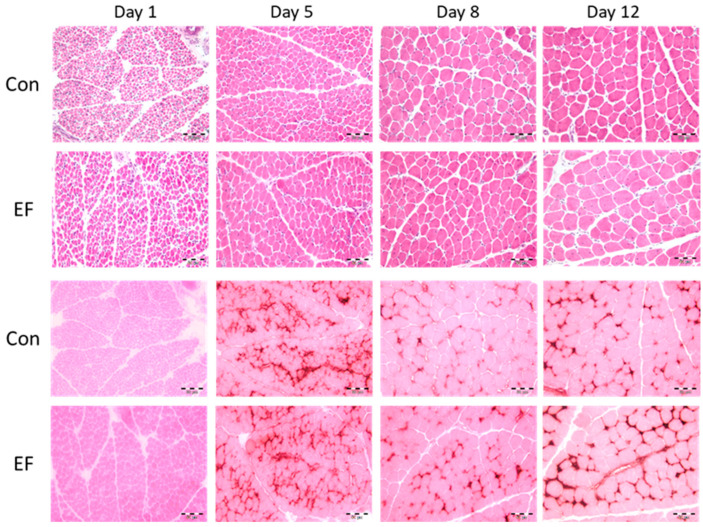
Figure 1.
Muscle structure of control (Con) and E. faecium AL41 (EF) supplemented chicks. Representative pectoralis muscle histological sections of chicks at 1, 5, 8, and 12 d of age, stained with hematoxylin/eosin (HE, upper panel) or eosin/alkaline phosphatase (EAP, lower panel) as used for image analysis. Scale bars represent 50 µm.
Table 4.
Muscle structure of pectoralis muscle in untreated control and E. faecium AL41 (EF) supplemented chicks between 5 and 12 d of age.
| Control | EF | p-Value | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Day of Age | 1 (n = 6) |
5 (n = 10) |
8 (n = 10) |
12 (n = 10) |
1 (n = 6) |
5 (n = 9) |
8 (n = 10) |
12 (n = 10) |
SE d 1 | SE d 5–12 |
Age | Group | Age × Group |
| Nuclei/mm2 | 7.622 c,A | 2.689 b,B | 1.648 a | 1.198 a | 6.515 c,B | 3.356 b,A | 1.803 a | 1.142 a | 257 | 199 | <0.001 | 0.580 | 0.004 |
| FCSA, µm2 | 54.5 d | 227.7 c | 577.5 b | 816.1 a,B | 67.9 d | 220.6 c | 586.4 b | 1107.8 a,A | 48.9 | 37.9 | <0.001 | 0.011 | <0.001 |
| Nuclei/fiber | 0.40 d | 0.57 c,B | 0.92 b,B | 0.95 a,B | 0.43 c | 0.71 b,A | 1.04 a,A | 1.23 a,A | 0.05 | 0.04 | <0.001 | <0.001 | 0.021 |
| CCSA, µm2 | 3.59 c | 10.02 b | 13.31 a,B | 4.77 c | 11.78 b | 22.44 a,A | 0.88 | <0.001 | <0.001 | <0.001 | |||
| Capillary area, % | 2.29 | 2.21 B | 1.92 B | 2.41 | 3.01 A | 2.89 A | 0.22 | 0.461 | 0.001 | 0.141 | |||
| Capillaries/mm2 | 6496 a,A | 2162 b | 1476 b | 5246 a,B | 2616 b | 1324 c | 222 | <0.001 | 0.091 | 0.002 | |||
| Capillaries/fiber | 1.50 A | 1.30 | 1.29 | 1.01 B | 1.39 | 1.45 | 0.13 | 0.656 | 0.453 | 0.033 | |||
a–d Different lower case letters indicate significant differences among ages (p < 0.05). A,B Different upper case letters indicate significant differences between groups (p < 0.05). FCSA, fiber cross sectional area; CCSA, capillary cross sectional area.
In both groups, the FCSA increased with age (p < 0.001). However, at day 12 of age larger muscle fibers were found in EF chicks (p < 0.001). As expected, the number of nuclei per mm2 decreased (p < 0.001) from day 1 to day 12 post-hatch in Con and EF chicks. At day 1, myonuclei density was higher in the Con group whereas it was lower in Con than EF chicks at day 5. In addition, the number of myonuclei per muscle fiber was higher in EF group chicks at days 5, 8, and 12 (p < 0.05).
Due to the immaturity of the muscle tissue on day one of life, the EAP staining did not work. Therefore, it was impossible to investigate the capillarization at this age. Control chicks had more capillaries per mm2 and a higher number of capillaries per myofiber at day five of age. At 12 d of age, capillaries in the PM of EF supplemented chicks had a larger CCSA and thus, the area covered by capillaries was larger than in the control group (p < 0.05).
3.4. Gene Expression
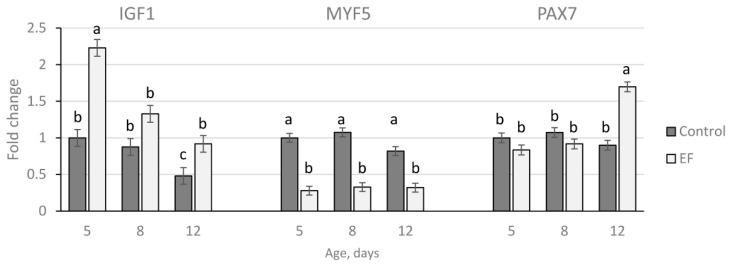
Gene expression, calculated as fold changes to Con at day five of age, is presented in Figure 2. A reduction of IGF1 mRNA was observed from the 5th to the 12th day of life in both groups (p < 0.05). The relative expression for IGF1 was higher in the EF group compared to the control at day 5 and day 12 (p < 0.001). In contrast, MYF5 expression was relatively constant over age in both groups (p > 0.1). The mRNA abundance was markedly down-regulated in the EF group (p < 0.001) at days 5, 8, and 12 of age. The transcription factor PAX7 was up-regulated in the EF group (p < 0.001) at day 12.
Figure 2.
Gene expression of IGF1, MYF5, and PAX7 in pectoralis muscles of untreated control and E. faecium AL41 (EF) supplemented chicks. Results at each time point are fold changes of expression of control at five days of age. a–c different letters indicate significant differences among groups and time points at p < 0.05.
3.5. PAX7 Protein Abundance in Pectoralis Muscle
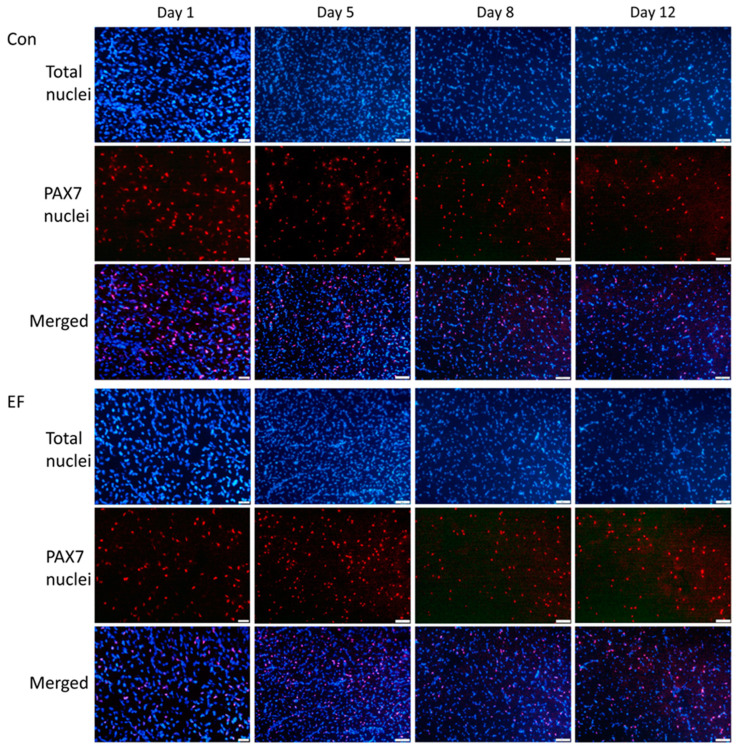
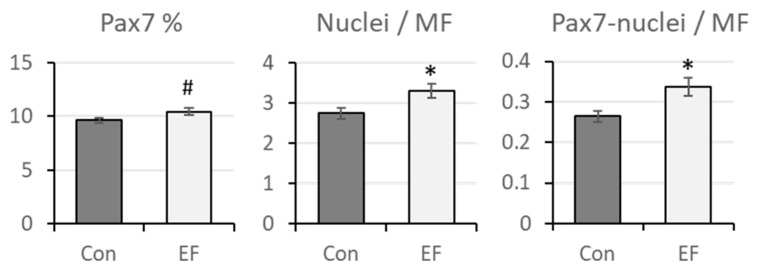
Increased gene expression of PAX7 in EF chicks at 12 d of age was further elucidated on protein level. Immunohistochemistry was applied to detect PAX7-positive nuclei in muscle sections as shown in Figure 3. As expected, PAX7 staining was observed in nuclei, verified by co-localization with DAPI nuclear staining, and PAX7-positive nuclei were observed close to muscle fibers. The proportion of PAX7-positive nuclei and the number of Pax7-positive nuclei per myofiber were quantified in muscle sections of chicks at 12 d of age solely (Figure 4), since the difference in gene expression between Con and EF chicks was observed only in this age (Figure 2). There was a trend for a greater percentage of PAX7-positive nuclei in EF chicks (p = 0.09). The numbers of nuclei per muscle fiber and PAX7-positive nuclei per muscle fiber were both higher in EF chicks (p = 0.021 and 0.013, respectively).
Figure 3.
Immunefluorescence of PAX7 in pectoralis muscle cross sections of untreated control (Con) and E. faecium AL41 (EF) supplemented chicks. Total nuclei were counterstained with DAPI (blue). Merged images indicate nuclear localization of PAX7. Scale bars represent 20 µm at day 1 and 50 µm at other ages.
Figure 4.
Quantification of total and PAX-7 positive nuclei in pectoralis muscle of untreated control (Con) and E. faecium AL41 (EF) supplemented chicks at 12 d of age. The percentage of Pax7-positive nuclei, the total number of nuclei per myofiber and the number of Pax7-positive nuclei per myofiber are given. * indicate significant differences between the groups at p < 0.05 and # indicates a trend at p = 0.09.
4. Discussion
Whereas probiotics regularly show positive health effects, its growth promoting effects are often inconsistent [22]. As PM growth is a main component of performance in broiler chickens, we aimed at getting information on the possible direct effects of the poultry-specific probiotic E. feacium AL41 concerning its development and tissue properties. SATC and their progeny are essential for post-hatch muscle development. Therefore, in addition to biochemical properties, we also examined myogenic factors (Pax7 and MYF5) and IGFI known as important regulators of SATC bioavailability and functionality [41,44,47].
As expected, body and PM weight increased strongly over the experimental period and the contribution of PM to BW increased from about 4 to 24% within twelve days. Accelerated growth of the breast muscle is a characteristic of poultry strains selected for meat production [31]. In our study, this is reflected by a fast increase of total amounts of DNA, RNA, protein and CK in muscle samples from both groups. In addition, the overall RNA/DNA ratio amounts to about 5.9 reflecting the high overall transcriptional activity of cells in broiler PM or in other words, its high capacity to accumulate ribosomes [61]. For example, The RNA/DNA ratio of liver cells, another cell type showing high rates of protein synthesis, amounts to 4.7, whereas those of thymocytes amounts to 0.3 [61]. However, daily weight gain (19.8 g/d) and relative increases in PM weight (day 1 to 8: 21.7; day 8 to 12: 0.68) were higher in EF compared with Con (18.0 g/d, day 1 to 8: 19.5; day 8 to 12: 0.64) chicks. Thus, at the end of the study period, body weight and PM weight are higher in EF-treated animals. Exclusively in muscle samples from EF-treated chicks, we found a higher amount of total RNA at day 12 of age, again indicating a higher anabolic potential after probiotic supplementation. As 85% of total RNA is ribosomal, higher levels represent an elevation in the machinery necessary for mRNA translation into protein [62].
On the one hand, the age-dependent increase in total CK activity of PM indicates a higher proportion of myofiber proteins [63]. However, muscular CK is predominantly expressed in fetal and adult fast fibers [64,65]. Thus, the observed increase of CK could reflect processes of muscle maturation including the conversion of slow to fast myofibers, the predominant fiber type in the adult PM [66]. In accord, ICDH activity, a marker of oxidative muscle metabolism decreased whereas LDH activity, a measure of glycolytic muscle metabolism, increased with age. The resulting increase of the LDH/ICDH ratio, confirms an age-dependent shift to the more glycolytic type of muscle metabolism typically for fast twitch PM [67,68].
Interestingly, maximum values of RNA/DNA ratio as well as DNA and RNA concentration per gram of muscle were found at day five in control and EF-supplemented chicks whereas protein content and CK activity, have increased continuously over the experimental period. This could reflect that SATC/myoblast proliferative activity and the number of SATC per gram muscle is highest at hatching and reaches its peak between days 2 and 4 of life [37,39,41] whereas myogenic processes (differentiation, protein accretion) increase further on. This idea is also supported by lower DNA/protein and RNA/protein ratios at day 8 and 12 compared with day 1 and 5 of age. Interestingly, data from Halevy et al. [39,46] showed that GH-R-mediated inhibition of differentiation by chicken growth hormone is responsible for the high proliferation rate of SATC directly post-hatch.
Typically, SATC mitotic activity declines rapidly after hatching [41]. For example, mitotic activity at day seven after hatch was reduced to one-third compared to that seen at day one [37]. Indeed, in both groups, the number of total nuclei per mm2 decreased significantly from day five on.
Nevertheless, in our study, myonuclei number per myofiber and FCSA increased over the complete experimental period in both groups. However, compared with controls, an elevated number of myonuclei per fiber was observed in EF chicks from day five on. One explanation for these results comes from our data showing a higher IGF1 mRNA expression in muscle samples from EF-supplemented compared with Con chicken. As shown for other species [47], IGF-I exerts positive effects on multiplication, growth and differentiation of SATC, myoblasts, and myotubes originating from chicken [69,70,71]. Our data indicate, that due to higher IGF1 expression in PM of EF-treated chicks more SATC escape terminal differentiation and proliferate for a longer time, thereby increasing the SATC pool size and the number of myonuclei per fiber over those of control animals. In both groups, an age depending decrease of intramuscular IGF1 mRNA expression was observed which step by step will increase the myogenic potential [47]. Induction of the myogenic regulatory factor MYOG is an essential component of IGF-I-dependent initiation of terminal differentiation [72,73]. MYOG expression is needed for the generation of fusion competent myoblasts that can supply new DNA to growing fibers [74], but its expression has not been investigated in our study. Nevertheless, at day 12 of life, the FCSA was larger in the EF group probably reflecting an increased fusion potential of MYOG-expressing cells [35].
Instead of the differentiation factor MYOG [45,74], we investigated the mRNA expression of the determination gene MYF5. MYF5 primes SATC for myogenic commitment thereby preventing them from adopting other than the myogenic fate [45,74]. Although MYF5 is expressed (mRNA and protein) in quiescent SATC/reserve cells and SATC-derived myoblasts it regulates myogenic process independently of PAX7 [43,74,75]. In the present study, we found in both groups a low variability in the MYF5 mRNA expression level over time. However, the expression level was generally higher in controls pointing to a higher proportion of cells primed for differentiation in the total SATC population of this group [42]. The lower level of MYF5 expression in the EF group was unexpected and, at first sight, it does not fit with data showing that PAX7 directly regulates the expression of MYF5 mRNA and protein [76]. Gayraud-Morel et al. [43] investigated the commitment and self-renewal potential of SATC with one compromised MYF5 allele (MYF5 heterozygous cells) and a 50% reduction of MYF5 levels compared to the wild type SATC. Quiescent MYF5 heterozygous cells express normal PAX7 levels but had a more committed status as reflected by expression of MYOD, MYOG and the adult structural protein Troponin-T. Interestingly, transplantation in regenerating muscle reveals a higher self-renewing capacity in MYF5 heterozygous compared with normal SATC. It seems thus possible that low MYF5 expressing SATC from the EF group show similar properties, which is in accord with our observation of more PAX7+ cells. Angiopoietin and its receptor Tie2 have been shown to downregulate MYF5 in SATC [77]. Both are expressed by SATC [43,77] but can also be provided by perivascular or interstitial cell types. Interestingly, compared with controls, PAX7 mRNA levels are upregulated in muscle samples from EF-treated chicks at day 12 post-hatch. PAX7 has essential functions in early postnatal/post-hatch development of SATC [41,42]. During this period, PAX7 is required for the expansion and survival of muscle progenitor cells, and for maintenance of their myogenic potential but it also regulates the transition from cycling muscle progenitor cells to the adult, quiescent SATC status [40,78]. From our mRNA expression data, we cannot exclude that higher PAX7 levels only promote the latter process. Therefore, we also quantified the number of PAX7 positive cells present in pectoralis muscle cross sections of 12-day-old control and EF-treated chicks. The proportion of PAX7+ to total nuclei is in accord with data from Halevy et al. [41] and amounts to about 10%, with a trend for higher levels in the EF group. More importantly, EF-treated chicks had more myonuclei per fiber and more of them were PAX7+ compared with controls. Halevy et al. [41] have shown that the majority of PAX7+ cells are in the pre-myogenin state during this developmental stage. However, most cells co-express PAX7 and MYOD (Pax7+/MyoD+) and the ratio between them is critical for the fate determination of myoblasts [79]. MYOD is found in myoblasts and drives them into the next steps of differentiation. A higher expression of PAX7 has been shown to delay or prevent the expression of MYOG thereby favoring proliferation (and survival) of SATC over terminal differentiation [45,79], which is in agreement with our results for 12-day-old EF-treated chicks.
The early post-hatch period is not only critical for myofiber growth but also for tissue vascularization, which is rudimentary at hatching. To ensure normal muscle development, both processes must develop in parallel [80] and are regulated by common factors [81,82,83]. By secreting proangiogenic growth factors such as the master driver VEGF (vascular endothelial growth factor), HGF, FGF as well as HIF, SATC play an important role in the initiation of the angiogenic program. Therefore, it was not surprising that in our previous study [49], disturbed SATC function induced by early post-hatch SE infection led to significantly reduced myofiber growth and strong negative effects on capillarization. Interestingly, application of EF could not prevent negative effects of SE infection on FCSA growth, but completely restored normal capillarization [49]. Here, we observed positive effects of EF treatment on both, hypertrophic myofiber growth and capillarization that manifests at day 8 and/or day 12 of age. Capillary supply decreased with age in Con while an increase was found in EF chicks. As was found by others [84], capillary density decreases with age in both groups. However, the area covered by capillaries (day 8 and day 12) and the CCSA (day 12) were higher in EF chicks compared with controls. Myofiber diameters increased with age in both groups but at day 12 EF-treated chicks had lager fibers. Large myofiber diameters are speculated to lead to metabolic stress in fast growing broilers muscles such as PM as they are associated with longer diffusion distances for O2, nutrients and metabolites such as lactate. Larger capillaries as in EF treated chicks will provide an increased exchange surface. Possibly, this will help to reduce or to overcome negative consequences of fast myofiber growth such as hypoxia, increased oxidative stress and reduced intramuscular pH values. These factors are thought to increase the risk of myopathies particularly in animals with very high weight gain during the first two weeks after hatch [85,86].
5. Conclusions
The probiotic EF AL41 when applied to chicks for seven days after hatching seems to have positive effects on myofiber growth but also improves the capillarization needed for supplying adequate amounts of O2, nutrients, endocrine, and growth factors, as well as the removal of metabolic waste. Thus, it could be a good option to achieve reliable positive effects on broiler health and performance without increasing their susceptibility to myopathies.
Acknowledgments
We would like to thank Andrea Lauková (Institute of Animal Physiology, Slovak Academy of Science, Košice, Slovakia) for providing the probiotic bacterial strain Enterococcus faecium AL41. In addition the authors thank Angela Steinborn and Marie Jugert-Lund for excellent technical assistance.
Author Contributions
Conceptualization, M.R. and R.Z.; methodology, E.A., M.R. and V.K.; validation, E.A. and M.R.; formal analysis, E.A., R.Z. and V.K.; investigation, E.A., M.Č., R.Z. and V.K.; resources, E.A., M.L.J., M.R. and V.R.; data curation, E.A. and M.R.; writing—original draft preparation, E.A., M.R. and R.Z.; writing—review and editing, V.K., V.R. and M.L.J.; project administration, M.R. and R.Z.; funding acquisition, M.R., R.Z. and V.K. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Ethics Committee of the University of Veterinary Medicine and Pharmacy, Košice, Slovakia, and by the Committee for Animal Welfare of the Ministry of Agriculture of the Slovak Republic (permission number 1184-3/2020-220).
Informed Consent Statement
Not applicable.
Data Availability Statement
The raw data were generated at the FBN Dummerstorf (Germany), the Research Institute for Animal Production, Nitra and the University of Veterinary Medicine and Pharmacy, Košice (Slovakia). Derived data supporting the findings of this study are available from the corresponding author on request.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This work was supported by the Slovak Research and Development Agency (APVV-21-0129) and by the Alexander von Humboldt Foundation (R.Z. and M.R.).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Marangoni F., Corsello G., Criselli C., Ferrara N., Ghiselli A., Luccin L., Poli A. Role of poultry meat in a balanced diet aimed at maintaining health and wellbeing; an Italien consensus document. Food Nutr. Res. 2015;59:27606. doi: 10.3402/fnr.v59.27606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Scanes C.G. The global importance of poultry. Poult. Sci. 2007;86:1057–1058. doi: 10.1093/ps/86.6.1057. [DOI] [PubMed] [Google Scholar]
- 3.Wyszyńska A.K., Godlewska R. Lactic Acid Bacteria—A Promising Tool for Controlling Chicken Campylobacter Infection. Front. Microbiol. 2021;12:703441. doi: 10.3389/fmicb.2021.70344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Crhanova M., Hradecka H., Faldynova M., Matulova M., Havlickova H., Sisak F., Rychlik I. Immune response of chicken gut to natural colonization by gut microflora and to Salmonella enterica serovar Enteritidis infection. Infect. Immun. 2011;79:2755–2763. doi: 10.1128/IAI.01375-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Biloni A., Quintana C.F., Menconi A., Kallapura G., Latorre J., Pixley C., Layton S., Dalmagro M., Hernandez-Velasco X., Wolfenden A., et al. Evaluation of effects of EarlyBird associated with FloraMax-B11 on Salmonella Enteritidis, intestinal morphology, and performance of broiler chickens. Poult. Sci. 2013;92:2337–2346. doi: 10.3382/ps.2013-03279. [DOI] [PubMed] [Google Scholar]
- 6.Pan D., Yu Z. Intestinal microbiome of poultry and its interaction with host and diet. Gut Microbes. 2014;5:108–119. doi: 10.4161/gmic.26945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Abdel Hamid F.M., El-Gohary F.A., Risha E.F. Incorporation efficacy comparison of probiotic and antibiotic on growth performance, some immunological and biochemical parameters in Salmonella Enteritidis challenged chicks. Life Sci. J. 2013;10:3550–3558. [Google Scholar]
- 8.Dina M.W., El-Hamd S., Ahmed Hams M. Effect of probiotic on Salmonella Enteritidis infection on broiler chickens. Egypt. J. Chem. Environ. Health. 2016;2:298–314. [Google Scholar]
- 9.Mehdi Y., Létourneau-Montminy M.-P., Gaucher Y.C., Suresh G., Rouissi T., Kaur Brar S., Cote C., Ramirez A.A. Use of antibiotics in broiler production: Global impacts and alternatives. Anim. Nutr. 2018;4:170–178. doi: 10.1016/j.aninu.2018.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iheukwumere I.H., Uneze B.C., Ejike C.E. Efficacy of some selected antimicrobial substances in prevention of enteric bacterial infection in broiler chicks. J. Biol. Agric. Healthc. 2017;7:58–66. [Google Scholar]
- 11.Miles R.D., Butcher G.D., Henry P.R., Littell R.C. Effect of antibiotic growth promotors on broiler performance, intestinal growth parameters, and quantitative morphology. Poult. Sci. 2006;85:476–485. doi: 10.1093/ps/85.3.476. [DOI] [PubMed] [Google Scholar]
- 12.Kilonzo-Nthenge A., Nahashon S.N., Chen F., Adefope N. Prevalence and antimicrobial resistance of pathogenic bacteria in chicken and guinea fowl. Poult. Sci. 2008;87:1841–1848. doi: 10.3382/ps.2007-00156. [DOI] [PubMed] [Google Scholar]
- 13.Roth N., Käsbohrer A., Mayrhofer S., Zitz U., Hofacre C., Domig K.J. The application of antibiotics in broiler production and the resulting antibiotic resistance in Escherichia coli: A global overview. Poult. Sci. 2019;98:1791–1804. doi: 10.3382/ps/pey539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maron D.F., Smith T.J., Nachmann K.E. Restrictions on antimicrobial use in food animal production: An international regulatory and economic survey. Glob. Health. 2013;9:48. doi: 10.1186/1744-8603-9-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Téllez G., Lauková A., Latorre J.D., Hernandez-Velasco X., Hargis B.M., Callaway T. Food-producing animals and their health in relation to human health. Microb. Ecol. Health Dis. 2015;26:25876. doi: 10.3402/mehd.v26.25876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zommiti M., Cambronel M., Maillot O., Barreau M., Sebei K., Feuilloley M., Ferchichi M., Connil N. Evaluation of probiotic properties and safety of Enterococcus faecium isolated from artisanal Tunisian meat “Dried Ossban”. Front. Microbiol. 2018;9:1685. doi: 10.3389/fmicb.2018.01685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schrezenmeir J., de Vrese M. Probiotics, prebiotics, and synbiotics—Approaching a definition. Am. J. Clin. Nutr. 2001;73:361S–364S. doi: 10.1093/ajcn/73.2.361s. [DOI] [PubMed] [Google Scholar]
- 18.Van der Wielen P.W.J.J., Lipman L.J.A., van Knapen F., Biesterveld S. Competitive exclusion of salmonella enterica serovar Enteritidis by lactobacillus crispatus and clostridium lactifermentans in a sequencing fed-batch culture. Appl. Environ. Microbiol. 2002;68:555–559. doi: 10.1128/AEM.68.2.555-559.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Audisio M., Oliver G., Apella M.C. Antagonistic effect of Enterococcus faecium J96 against human and poultry pathogenic Salmonella spp. J. Food Protect. 1999;62:751–755. doi: 10.4315/0362-028X-62.7.751. [DOI] [PubMed] [Google Scholar]
- 20.Awad W.A., Ghareeb K., Abdel-Raheen S., Böhm J. Effects of dietary inclusion of probiotic and symbiotic on growth performance, organ weights, and intestinal histomorphology of broiler chickens. Poult. Sci. 2009;88:49–55. doi: 10.3382/ps.2008-00244. [DOI] [PubMed] [Google Scholar]
- 21.De Lima Almeida Paz I.C., de Lima Almeida I.G., de La Vega L.T., Milbrandt E.L., Rodrigues Borges M., Coelho Chaves G.H., dos Ouros C.C., da Silva M.I.L., Caldara F.R., Filho R.L. Productivity and well-being of broiler chickens supplemented with probiotic. J. Appl. Poult. Res. 2019;28:930–942. doi: 10.3382/japr/pfz054. [DOI] [Google Scholar]
- 22.Niewold T.A. The nonantibiotic anti-inflammatory effect of antimicrobial growth promotors, the real mode of action? A hypothesis. Poult. Sci. 2007;86:605–609. doi: 10.1093/ps/86.4.605. [DOI] [PubMed] [Google Scholar]
- 23.Levkut M., Pistl J., Lauková A., Revajová V., Herich R., Ševčíková Z., Strompfová V., Szabóová R., Kokinčáková T. Antimicrobial activity of Enterococcus faecium EF55 against Salmonella Enteritidis in chicks. Acta Vet. Hung. 2009;57:13–24. doi: 10.1556/avet.57.2009.1.2. [DOI] [PubMed] [Google Scholar]
- 24.Herich R., Kokinčáková T., Lauková A., Levkutová M. Effect of preventive application of Enterococcus faecium EF55 on intestinal mucosa during salmonellosis in chickens. Czech J. Anim. Sci. 2010;55:42–47. doi: 10.17221/19/2009-CJAS. [DOI] [Google Scholar]
- 25.Lauková A., Chrastinová L., Pogány Simonová M., Strompfová V., Plachá I., Čobanová K., Formelová Z., Chrenková M., Ondruška L. Enterococcus faecium AL 41: Its enterocin M and their beneficial use in rabbits husbandry. Probiotics Antimicrob. 2012;4:243–249. doi: 10.1007/s12602-012-9118-7. [DOI] [PubMed] [Google Scholar]
- 26.Lauková A., Mareková M., Štyriak I. Inhibitory effect of different enterocins against fecal bacterial isolates. Berl. Muench. Tierärztl. Wochenschr. 2003;116:37–40. [PubMed] [Google Scholar]
- 27.Levkut M., Revajová V., Lauková A., Ševčíková Z., Spiŝáková V., Faixová Z., Levkutová M., Strompfová V., Pistl J., Levkut M. Leucocytic responses and intestinal mucin dynamics of broilers protected with Enterococcus faecium EF55 and challenged with Salmonella Enteritidis. Res. Vet. Sci. 2012;93:195–201. doi: 10.1016/j.rvsc.2011.06.021. [DOI] [PubMed] [Google Scholar]
- 28.Karaffová V., Bobíková K., Husáková E., Levkut M., Herich R., Ravajová V., levkutová M., Levkut M. Interaction of TGF-ß4 and IL-17 with IgA secretion in the intestine of chickens fed with E. faecium AL41 and challenged with S. Enteritidis. Res. Vet. Sci. 2015;100:75–79. doi: 10.1016/j.rvsc.2015.04.005. [DOI] [PubMed] [Google Scholar]
- 29.Samli E.S., Senkoylu N., Koc F., Kanter M., Agma A. Effects of Enterococcus faecium and dried whey on broiler performance, gut histomorphology and intestinal microbiota. Arch. Anim. Nutr. 2007;61:42–49. doi: 10.1080/17450390601106655. [DOI] [PubMed] [Google Scholar]
- 30.Berri C., Godet E., Hattab N.H., Duclos M.J. Growth and differentiation of the chicken Pectoralis major muscle: Effect of genotype and early nutrition. Arch. Anim. Breed. 2006;49:31–32. [Google Scholar]
- 31.Tixier-Boichard M. From the jungle fowl to highly performing chickens: Are we reaching limits? World’s Poult. Sci. J. 2020;76:2–17. doi: 10.1080/00439339.2020.1729676. [DOI] [Google Scholar]
- 32.Smith J.H. Relation of body size to muscle cell size and number in the chicken. Poult. Sci. 1963;42:283–290. doi: 10.3382/ps.0420283. [DOI] [Google Scholar]
- 33.Mozdziak P.E., Schultz E., Cassens R.G. Myonuclear accretion is a major determinant of avian skeletal muscle growth. Am. J. Physiol. 1997;272:C565–C571. doi: 10.1152/ajpcell.1997.272.2.C565. [DOI] [PubMed] [Google Scholar]
- 34.Duclos M.J., Molette C., Guernec A., Remignon H., Berri C. Cellular aspects of breast muscle development in chickens with high and low growth rate. Arch. Anim. Breed. 2006;49:147–151. [Google Scholar]
- 35.Jacquemin V., Butler-Browne G.S., Furling D., Mouly V. IL-13 mediates the recruitment of reserve cells for fusion during IGF-1-induced hypertrophy of human myotubes. J. Cell Sci. 2007;120:670–681. doi: 10.1242/jcs.03371. [DOI] [PubMed] [Google Scholar]
- 36.Geiger A.E., Daughtry M.R., Gow C.M., Siegel P.B., Shi H., Gerrard D.E. Long-term selection of chickens for body weight alters muscle satellite cell behaviors. Poult. Sci. 2018;97:2557–2567. doi: 10.3382/ps/pey050. [DOI] [PubMed] [Google Scholar]
- 37.Moore D.T., Ferket P.R., Mozdziak P.E. Muscle development in the late embryonic and early post-hatch poult. Int. J. Poult. Sci. 2005;4:138–142. doi: 10.3923/ijps.2005.138.142. [DOI] [Google Scholar]
- 38.Velleman S.G., Coy C.S., Emmerson D.A. Effect of the timing of posthatch feed restrictions on broiler breast muscle development and muscle transcriptional regulatory factor gene expression. Poult. Sci. 2014;93:1484–1494. doi: 10.3382/ps.2013-03813. [DOI] [PubMed] [Google Scholar]
- 39.Halevy O., Gayra A., Barak M., Uni Z., Sklan D. Early posthatch starvation decreases satellite cell proliferation and skeletal muscle growth in chicks. J. Nutr. 2000;130:858–864. doi: 10.1093/jn/130.4.858. [DOI] [PubMed] [Google Scholar]
- 40.Olguin H.C., Olwin B.B. Pax-7 up-regulation inhibits myogenesis and cell cycle progression in satellite cells: A potential mechanism for self-renewal. Dev. Biol. 2004;275:375–388. doi: 10.1016/j.ydbio.2004.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Halevy O., Piestun Y., Allouh M.Z., Rosser B.W.C., Rinkevich Y., Reshef R., Rozenboim I., Wleklinski-Lee M., Yablonka-Reuveni Z. The pattern of Pax7 expression during myogenesis in the posthatch chicken establishes a model for satellite cell differentiation and renewal. Dev. Dyn. 2004;231:489–502. doi: 10.1002/dvdy.20151. [DOI] [PubMed] [Google Scholar]
- 42.Seale P., Rudnicki M.A. A new look at the origin, function, and “stem-cell” status of muscle satellite cells. Dev. Biol. 2000;218:115–124. doi: 10.1006/dbio.1999.9565. [DOI] [PubMed] [Google Scholar]
- 43.Gayraud-Morel B., Chrétien F., Jory A., Sambasivan R., Negroni E., Flamant P., Soubigou G., Coppé J.-Y., Di Santo J., Cumano A., et al. MYF5 haploinsufficiency reveals distinct cell fate potentials for adult skeletal muscle stem cells. J. Cell Sci. 2012;125:1738–1749. doi: 10.1242/jcs.128678. [DOI] [PubMed] [Google Scholar]
- 44.Yablonka-Reuveni Z., Paterson B.M. MyoD and myogenin expression patterns in cultures of fetal and adult chicken myoblasts. J. Histochem. Cytochem. 2001;49:455–462. doi: 10.1177/002215540104900405. [DOI] [PubMed] [Google Scholar]
- 45.Zammit P.S., Relaix F., Nagata Y., Pérez Ruiz A., Collins C.A., Partridge T.A., Beauchamp J.R. Pax7 and myogenic progression in skeletal muscle satellite cells. J. Cell Sci. 2006;119:1824–1832. doi: 10.1242/jcs.02908. [DOI] [PubMed] [Google Scholar]
- 46.Halevy O., Hodik V., Mett A. The effects of growth hormone on avian skeletal muscle satellite cell proliferation and differentiation. Gen. Comp. Endocrinol. 1996;101:43–52. doi: 10.1006/gcen.1996.0006. [DOI] [PubMed] [Google Scholar]
- 47.Florini J.R., Ewton D.Z., Coolican S.A. Growth hormone and the insulin-like growth factor system in myogenesis. Endocr. Rev. 1996;17:481–517. doi: 10.1210/edrv-17-5-481. [DOI] [PubMed] [Google Scholar]
- 48.Clark D.L., Walter K.G., Velleman S.G. Incubation temperature and time of hatch impact broiler muscle growth and morphology. Poult. Sci. 2017;96:4085–4095. doi: 10.3382/ps/pex202. [DOI] [PubMed] [Google Scholar]
- 49.Zitnan R., Albrecht E., Kalbe C., Miersch C., Revajova V., Levkut Jr. M., Röntgen M. Muscle characteristics in chicks challenged with Salmonella Enteritidis and the effect of preventive application of the probiotic Enterococcus faecium. Poult. Sci. 2019;98:2014–2025. doi: 10.3382/ps/pey561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Letnická A., Karaffová V., Levkut M., Revajová V., Herich R. Influence of oral application of Enterococcus faecium AL41 on TGF-β4 and IL-17 expression and immunocompetent cell distribution in chickens challenged with Campylobacter jejuni. Acta Vet. Hung. 2017;65:317–326. doi: 10.1556/004.2017.031. [DOI] [PubMed] [Google Scholar]
- 51.Karaffová V., Tóthová C., Szabóová R., Revajová V., Lauková A., Ševcíková Z., Herich R., Žitnan R., Levkut M., Levkut M., et al. The effect of Enterococcus faecium AL41 on the acute phase proteins and selected mucosal immune molecules in broiler chickens. Life. 2022;12:598. doi: 10.3390/life12040598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cobb-Vantress Inc. Cobb500 Slow Feather Breeder Management Supplement. Cobb-Vantress; Siloam Springs, AR, USA: 2020. [Google Scholar]
- 53.Peterson G.L. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal. Biochem. 1977;83:346–356. doi: 10.1016/0003-2697(77)90043-4. [DOI] [PubMed] [Google Scholar]
- 54.Lösel D., Franke A., Kalbe C. Comparison of different skeletal muscles from growing domestic pigs and wild boars. Arch. Anim. Breed. 2013;56:766–777. doi: 10.7482/0003-9438-56-076. [DOI] [Google Scholar]
- 55.Romeis B. Mikroskopische Technik. Urban & Schwarzenberg; Munich, Germany: 1989. [Google Scholar]
- 56.Spannhof L. Einführung in die Praxis der Histochemie. VEB Gustav-Fischer-Verlag; Jena, Germany: 1967. [Google Scholar]
- 57.Karaffová V., Bobíková K., Levkut M., Revajová V., Ševčíková Z., Levkut M. The influence of Farmatan® and Flimabend® on the mucosal immunity of broiler chicken. Poult. Sci. 2019;98:1161–1166. doi: 10.3382/ps/pey517. [DOI] [PubMed] [Google Scholar]
- 58.Xiao Y., Wu C., Li K., Gui G., Zhang G., Yang H. Association of growth rate with hormone levels and myogenic gene expression profile in broilers. J. Anim. Sci. Biotechnol. 2017;8:43. doi: 10.1186/s40104-017-0170-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Adhikari R., Chen C., Waters E., West F.D., Kim W.K. Isolation and differentiation of mesenchymal stem cells from broiler chicken compact bones. Front. Physiol. 2019;9:1892. doi: 10.3389/fphys.2018.01892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.De Boever S., Vangestel C., De Backer P., Croubels S., Sys S.U. Identification and validation of housekeeping genes as internal control for gene expression in an intravenous LPS inflammation model in chickens. Vet. Immunol. Immunopathol. 2008;122:312–317. doi: 10.1016/j.vetimm.2007.12.002. [DOI] [PubMed] [Google Scholar]
- 61.Schmidt E.E., Schibler U. Cell size regulation, a mechanism that controls cellular RNA accumulation: Consequences on regulation of the ubiquitous transcription factors Oct1 and NF-Y, and liver-enriched transcription factor DBP. J. Cell Biol. 1995;128:467–483. doi: 10.1083/jcb.128.4.467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Haddad F., Baldwin K.M., Tesch P.A. Pretranslational markers of contractile protein expression in human skeletal muscle: Effect of limb unloading plus resistance exercise. J. Appl. Physiol. 2005;98:46–52. doi: 10.1152/japplphysiol.00553.2004. [DOI] [PubMed] [Google Scholar]
- 63.Rehfeldt C., Renne U., Sawitzky M., Binder G., Hoeflich A. Increased fat mass, decreased myofiber size, and a shift to glycolytic muscle metabolism in adolescent male transgenic mice overexpressing IGFBP-2. Am. J. Physiol. Endocrinol. Metabol. 2010;299:E287–E298. doi: 10.1152/ajpendo.00492.2009. [DOI] [PubMed] [Google Scholar]
- 64.Biressi S., Molinaro M., Cossa G. Cellular heterogeneity during vertebrate skeletal muscle development. Dev. Biol. 2007;308:281–293. doi: 10.1016/j.ydbio.2007.06.006. [DOI] [PubMed] [Google Scholar]
- 65.Bottinelli R., Reggiani C. Human skeletal muscle fibres, molecular and functional diversity. Prog. Biophys. Mol. Biol. 2000;73:195–262. doi: 10.1016/S0079-6107(00)00006-7. [DOI] [PubMed] [Google Scholar]
- 66.Verdiglione R., Cassandro M. Characterization of muscle fiber type in the pectoralis major muscle of slow-growing local and commercial chicken strains. Poult. Sci. 2013;92:2433–2437. doi: 10.3382/ps.2013-03013. [DOI] [PubMed] [Google Scholar]
- 67.Kocamis H., McFarland D.C., Killefer J. Temporal expression of growth factor genes during myogenesis of satellite cells derived from the biceps femoris and pectoralis major muscles of the chicken. J. Cell. Physiol. 2001;186:146–152. doi: 10.1002/1097-4652(200101)186:1<146::AID-JCP1014>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 68.Doherty M.K., McLean L., Hayter J.R., Pratt J.M., Robertson D.H., El-Shafei A., Gaskell S.J., Beynon R.J. The proteome of chicken skeletal muscle: Changes in soluble protein expression during growth in a layer strain. Proteomics. 2004;4:2082–2093. doi: 10.1002/pmic.200300716. [DOI] [PubMed] [Google Scholar]
- 69.Schmid C., Steiner T., Froesch E.R. Preferential enhancement of myoblast differentiation by insulin-like growth factors (IGF I and IGF II) in primary cultures of chicken embryonic cells. FEBS Lett. 1983;16:117–121. doi: 10.1016/0014-5793(83)80742-X. [DOI] [PubMed] [Google Scholar]
- 70.Duclos M.J., Wilkie R.S., Goddard C. Stimulation of DNA synthesis in chicken muscle satellite cells by insulin and insulin-like growth factors: Evidence for exclusive mediation by a type-I insulin-like growth factor receptor. J. Endocrinol. 1991;128:35–42. doi: 10.1677/joe.0.1280035. [DOI] [PubMed] [Google Scholar]
- 71.Vandenburgh H.H., Karlisch P., Shansky J., Feldstein R. Insulin and IGF-I induce pronounced hypertrophy of skeletal myofibers in tissue culture. Am. J. Physiol. Cell Physiol. 1991;29:C475–C484. doi: 10.1152/ajpcell.1991.260.3.C475. [DOI] [PubMed] [Google Scholar]
- 72.Florini J.R., Ewton D.Z., Roof S.L. Insulin-like growth factor-I stimulates terminal myogenic differentiation by induction of myogenin gene expression. Mol. Endocrinol. 1991;5:718–724. doi: 10.1210/mend-5-5-718. [DOI] [PubMed] [Google Scholar]
- 73.Musaro A., Rosenthal N. Maturation of the myogenic program is induced by postmitotic expression of insulin-like growth factor I. Mol. Cell. Biol. 1999;19:3115–3124. doi: 10.1128/MCB.19.4.3115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zammit P.S. Function of the myogenic regulatory factors MYF5, MyoD, Myogenin and MRF4 in skeletal muscle, satellite cells and regenerative myogenesis. Semin. Cell. Dev. Biol. 2017;72:19–32. doi: 10.1016/j.semcdb.207.11.011. [DOI] [PubMed] [Google Scholar]
- 75.Beauchamp J.R., Heslop L., Yu D.S.W., Tajbakhsh S., Kelly R.G., Wernig A., Buckingham M.E., Partridge T.A., Zammit P.S. Expression of CD34 and of MYF5 defines the majority pf quiescent adult skeletal muscle satellite cells. J. Cell Biol. 2000;151:1221–1233. doi: 10.1083/jcb.151.6.1221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.McKinnell I.W., Ishibashi J., Le Grand F., Punch V.G.J., Addicks G.C., Greenblatt J.F., Dilworth F.J., Rudnicki M.A. Pac7 activates myogenic genes by recruitment of a histone methyltransferase complex. Nat. Cell Biol. 2008;10:77–84. doi: 10.1038/ncb1671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Abu Khalil R., Le Grand F., Pallafacchina G., Valable S., Authier F.-J., Rudnicki M.A., Gheradi R.K., Germain S., Chretien F., Sotiropoulos A., et al. Autocrine and paracrine angiopoietin 1/Tie1-2 signaling promotes muscle satellite cell self-renewal. Cell Stem Cell. 2009;5:298–309. doi: 10.1016/j.stem.2009.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lepper C., Conway S.J., Fan C.M. Adult satellite cells and embryonic muscle progenitors have distinct genetic requirements. Nature. 2009;460:627–631. doi: 10.1038/nature08209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Olguin H.C., Yang Z., Tapscott S.J., Stephen J., Olwin B.B. Reciprocal inhibition between Pax7 and muscle regulatory factors modulates myogenic cell fate determination. J. Cell Biol. 2007;177:769–779. doi: 10.1083/jcb.200608122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hoving-Bolink A.H., Kranen R.W., Klont R.E., Gerritsen C.L.M., de Greef K.H. Fibre area and capillary supply in broiler breast muscle in relation to productivity and ascites. Meat Sci. 2000;66:397–402. doi: 10.1016/S0309-1740(00)00071-1. [DOI] [PubMed] [Google Scholar]
- 81.Coultas L., Chawengsaksophak K., Rossant J. Endothelial cells and VEGF in vascular development. Nature. 2005;438:937–945. doi: 10.1038/nature04479. [DOI] [PubMed] [Google Scholar]
- 82.Christov C., Chrétien F., Abou-Khalil R., Bassez G., Vallet G., Authier F.-J., Bassaglia Y., Shinin V., Tajbakhsh S., Chazaud B., et al. Muscle satellite cells and endothelial cells: Close neighbors and privileged partners. Mol. Biol. Cell. 2007;18:1397–1409. doi: 10.1091/mbc.e06-08-0693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bryan B.A., Walshe T.E., Mitchell D.C., Havumaki J.S., Saint-Geniez M., Maharaj A.S., Maldonado A.E., D’Amore P.A. Coordinated vascular endothelial growth factor expression and signaling during skeletal myogenic differentiation. Mol. Biol. Cell. 2008;19:994–1006. doi: 10.1091/mbc.e07-09-0856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Radaelli G., Piccirilo A., Birolo M., Bertotto D., Gratta F., Ballarina C., Vascellari M., Xiccato G., Trocino A. Effect of age on the occurrence of muscle fiber degeneration associated with myopathies in broiler chickens submitted to feed restriction. Poult. Sci. 2017;96:309–319. doi: 10.3382/ps/pew270. [DOI] [PubMed] [Google Scholar]
- 85.Papah M.B., Brannick E.M., Schmidt C.J., Abasht B. Evidence and role of phlebitis and lipid infiltration in the onset and pathogenesis of wooden breast disease in modern broiler checkens. Avian Pathol. 2017;46:623–643. doi: 10.1080/03079457.2017.1339346. [DOI] [PubMed] [Google Scholar]
- 86.Sihvo H.-K., Airas N., Lindén J., Puolanne E. Pectoral vessel density and early ultrastructural changes in broiler chicken wooden breast myopathy. J. Comp. Path. 2018;161:1–10. doi: 10.1016/j.jcpa.2018.04.002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The raw data were generated at the FBN Dummerstorf (Germany), the Research Institute for Animal Production, Nitra and the University of Veterinary Medicine and Pharmacy, Košice (Slovakia). Derived data supporting the findings of this study are available from the corresponding author on request.