Abstract
We present a case of a 75-year-old man who developed an acute left atrial appendage thrombus immediately following mitral valve transcatheter edge to edge repair despite adequate intraprocedural anticoagulation. The patient was managed with enoxaparin to warfarin bridging with no obvious thromboembolic events on follow-up. Attention to anticoagulation is important to reduce thromboembolic risk during mitral valve transcatheter edge to edge repair. (Level of Difficulty: Intermediate.)
Key Words: anticoagulation, left atrial appendage thrombus, mitral regurgitation, transcatheter mitral valve edge to edge repair
Abbreviations and Acronyms: ACT, activated clotting time; INR, international normalized ratio; LAA, left atrial appendage; NYHA, New York Heart Association; TEE, transesophageal echocardiogram; TEER, transcatheter edge to edge repair
Central Illustration
History of Presentation
We present the case of a 75-year-old man with severe symptomatic secondary mitral valve regurgitation who presented to the cardiac catheterization laboratory for an elective mitral valve transcatheter edge to edge repair (TEER). At the time of presentation, the patient reported symptoms of shortness of breath with minimal activity, but he had no shortness of breath at rest, suggestive of New York Heart Association (NYHA) functional class IIIa, American College of Cardiology Foundation/American Heart Association Stage C heart failure. He was on appropriate guideline-directed medical therapy for chronic severe ischemic cardiomyopathy with carvedilol, losartan, empagliflozin, torsemide, and spironolactone. His clinical examination revealed borderline jugular venous distension and an apical holosystolic murmur. He had normal respiratory effort at rest and normal breath sounds. He had no peripheral edema. He had a heart rate of 88 beats/min, his blood pressure was 96/70 mm Hg, and his oxygen saturation was 98% on room air.
Learning Objectives
-
•
To recognize the formation or an acute LAA thrombus immediately following mitral valve TEER as a rare complication of this procedure.
-
•
To learn about clinical risk factors associated with acute LAA thrombus formation in patients undergoing mitral valve TEER, such as a history of atrial fibrillation, low LAA emptying velocity, chronic heart failure and elevated CHA2DS2-VASc score.
-
•
To understand the importance of comprehensive post-procedural TEE to identify this complication following mitral valve TEER.
-
•
To learn about appropriate post-procedural anticoagulation strategy in patients who have this complication.
Past Medical History and Differential Diagnosis
The patient had a past medical history of severe ischemic cardiomyopathy (left ventricular ejection fraction of 20%-25% by echocardiography), known coronary artery disease (with percutaneous coronary interventions to left anterior descending and right coronary arteries in 2011) and atrioventricular node ablation with cardiac resynchronization therapy for permanent atrial fibrillation (CHA2DS2-VASc score-5, HAS-BLED score 2). The patient had eccentric, posteriorly directed severe mitral regurgitation due to mal-coaptation of leaflets. The mechanism of the mitral regurgitation was thought to be a combination of annular dilatation and restricted posterior mitral valve leaflet motion due to severe ischemic cardiomyopathy. The etiology of the mitral regurgitation was consistent with secondary mitral regurgitation. The patient’s prior workup included myocardial perfusion imaging, which showed that he had a large anterior and inferior fixed defect without any evidence of reversible myocardial ischemia. His had normal blood cell counts and renal function. His Society of Thoracic Surgeons mortality risk was estimated to be 6% for a surgical mitral valve replacement and 3.6% for a surgical mitral valve repair.
Management
After review by a multidisciplinary valve team, he was thought to be a suitable candidate for mitral valve transcatheter edge to edge repair due to elevated surgical risk, poor functional status, suitable valve morphology and presence of severe symptomatic secondary (Stage D) mitral regurgitation despite optimal goal-directed medical therapy and cardiac resynchronization therapy. The current 2020 American College of Cardiology/American Heart Association guidelines for management of patients with valvular heart disease have a Class 2a recommendation for transcatheter mitral valve repair in this setting. Before mitral valve TEER, he was on aspirin and warfarin because of a history of coronary artery disease and atrial fibrillation. Warfarin was held for the procedure and his international normalized ratio (INR) was 1.2 on the day of procedure. His other cardiac medications were listed previously.
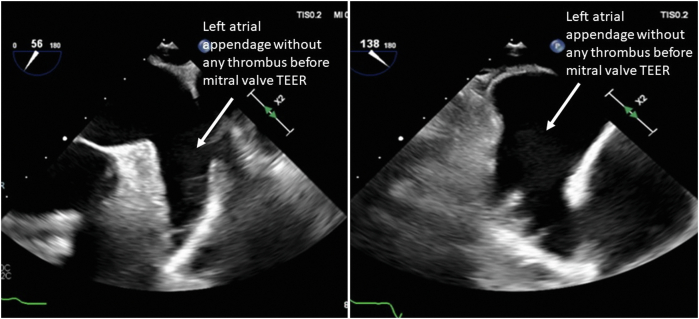
The patient had no prior history of left atrial appendage (LAA) or other intracardiac thrombus and no prior thromboembolic events. The left atrium was known to be severely enlarged with an end-systolic atrial volume index of 77 mL/m2. Pre-TEER transesophageal echocardiogram (TEE) showed no evidence of LAA thrombus (Figure 1). LAA emptying velocity by spectral Doppler was reduced (20 cm/s). At the time of the transseptal puncture, the patient received prompt systemic anticoagulation with heparin. He was given a total of 13,000 units of intravenous heparin, targeting an activated clotting time (ACT) >250 seconds (reference range 74-137 seconds) during the procedure. ACT was 235 seconds after initial bolus of heparin; subsequent readings at the time of mitral valve TEER deployment were 274, 257, and 246 seconds, respectively. A G4 NTW MitraClip (Abbott) was successfully deployed with immediate reduction in mitral regurgitation from severe to mild. Mean mitral valve gradient was 2 mm Hg pre-TEER and increased to 3 mm Hg post clip deployment. Spontaneous echo-contrast was noted in the left atrium and the LAA immediately after mitral valve TEER, suggestive of stasis secondary to the reduction in mitral regurgitation (Figure 2). A few minutes later, the spontaneous echo-contrast continued to thicken, leading to the formation a well-defined thrombus in the LAA (Figure 3). Videos 1, 2, and 3 show spontaneous echo-contrast in the LAA with relatively rapid formation of a well-defined thrombus immediately after clip deployment.
Figure 1.
Preprocedural Transesophageal Echocardiogram Showing No Evidence of Left Atrial Appendage Thrombus
Arrows point to the left atrial appendage. (Left) Left atrial appendage at 56°; (right) left atrial appendage at 138°, showing absence of a thrombus in 2 orthogonal planes. TEER = transcatheter edge to edge repair
Figure 2.
Spontaneous Echo-Contrast in the Left Atrium and LAA Immediately After Mitral Valve TEER
LAA = left atrial appendage; TEER = transcatheter edge to edge repair
Figure 3.
Well-Formed Thrombus in the LAA Within Minutes of Mitral Valve TEER
Abbreviations as in Figure 2.
At the end of the case, the venous sheath was removed, and the access site closed with a purse-string suture. Typically, protamine is administered to reverse ACT before sheath removal; however, in this case, it was not given because of the formation of the acute LAA thrombus and the risk of thrombus propagation. Therapeutic dose enoxaparin (1 mg/kg) was given immediately after the procedure and patient was started on warfarin the same day. He subsequently underwent enoxaparin to warfarin bridging with a target INR of >2. There were no immediate vascular, bleeding, or other complications. He did well clinically and was discharged home without any complications. He had an uneventful recovery. Aspirin and warfarin were continued. He had no post-procedural complications, including no stroke or major bleeding at 45-day follow-up.
Follow-Up
The patient has been doing well following his procedure. He was recently seen in clinic for his 1-year follow-up visit after his mitral valve TEER. He reported improved exercise tolerance and he is now short of breath with moderate activity (NYHA functional class II), which is improved compared with when he initially presented for his mitral valve TEER. He remains on warfarin, and he did not have any thromboembolic complications during this follow-up period. His transthoracic echocardiogram at 1 year follow-up shows only mild mitral regurgitation with a mean mitral gradient of 3 mm Hg. There was no obvious intracardiac thrombus. His left ventricular ejection fraction has improved marginally to 30%.
Ethics/institutional review board approval was not needed, as this is not a requirement for case reports at our institution.
Discussion
Percutaneous valve interventions have significantly expanded the treatment options for severe valve disease, particularly in high-risk patients. Mitral valve TEER is an increasingly used procedure for the management of severe mitral regurgitation in patients with high surgical risk. Several clinical trials have shown that mitral valve TEER is a safe alternative compared with surgical mitral valve repair or replacement for treatment of severe mitral regurgitation in selected patients.1,2 Patients with mitral valve disease have an increased risk of developing atrial fibrillation. The presence of atrial fibrillation is common in patients undergoing mitral valve TEER and does not affect acute procedural success or in-hospital mortality.3 However, presence of atrial fibrillation in these patients is associated with worse clinical outcomes at 1 year, such as increased rate of death or heart failure.3 Patients undergoing mitral valve TEER who have atrial fibrillation are also more likely to have additional comorbidities and more advanced valve disease.4 Patients with atrial fibrillation also have an elevated risk of LAA thrombus formation. Intraprocedural TEE plays a crucial role in the success of mitral valve TEER by guiding transseptal puncture, device insertion, adequacy of valve treatment, and identification of acute complications, such as formation of pericardial effusion or acute intracardiac thrombus.5 Overall, the incidence of stroke in patients undergoing mitral valve TEER remains low, ranging from 0.7% to 2.6% at 30 days.6 However, there are several factors related to the procedure that present a risk for intracardiac thrombus formation intraprocedurally, including need for transseptal puncture, exposure of the mitral valve TEER sheath/delivery system to the blood stream, and the clips themselves being foreign bodies that can develop a thrombus over time. Hence, therapeutic anticoagulation during the procedure and dual antiplatelet therapy for 6 months afterward are the standard practice, although there are limited studies guiding this practice. Patients with atrial fibrillation undergoing mitral valve TEER, as well as patients with other indications for anticoagulation, have been typically managed with continuation of their oral anticoagulation and a single antiplatelet agent.
There are handful of case reports of acute thrombus formation during the mitral valve TEER procedure in the left and right atria: at the site of transseptal puncture, as well as on the clip delivery system/guide catheter and on the mitral valve TEER device itself.7,8 Acute thrombus formation in the LAA immediately after mitral valve TEER is rare. The authors identified only 1 prior case report of acute LAA thrombus formation immediately after mitral valve TEER, which was reported from Germany in 2018.9 In that reported case, the patient had severe left ventricular impairment (left ventricular ejection fraction 20%) and history of atrial fibrillation, similar to the current case. Also, both patients had low LAA emptying velocities. In the previously reported case, the authors performed a thrombo-elastogram on blood sampled from the left atrium directly, showing gain of thrombogenicity. There were no instances of overt clinical thromboembolism in both cases. Both patients were managed with warfarin and bridging with low molecular weight heparin. In both these cases, thrombus formation was preceded by the development of spontaneous echo-contrast. Thus, blood stasis likely plays an important role. We speculate that the abrupt reduction in mitral regurgitation and increase in the mitral valve mean gradient immediately after successful mitral valve TEER, coupled with elevated left ventricular filling pressures in the setting of severe left ventricular dysfunction, as well as low LAA emptying velocity in chronic atrial fibrillation together created conditions that favored blood stasis in the LAA, leading to initial sludge formation that quickly progressed to organized left atrial thrombus in this patient. Ohno et al10 reported development of spontaneous echo-contrast in the left atrium as well as suspicion for evolving left atrial/LAA thrombus after deployment of a second mitral clip in a patient with severe eccentric mitral regurgitation, chronic atrial fibrillation, and persistent NYHA functional class III heart failure. The spontaneous echo-contrast promptly resolved after the second mitral clip was removed.10 In another case reported from Japan in 2017, LAA thrombus was noted subacutely after mitral valve TEER on follow-up transesophageal echocardiogram 1 week after mitral valve TEER.11 This patient also had reduced left ventricular ejection fraction and a history of atrial fibrillation. The patient was on warfarin already and maintained at a higher INR target of 2.5 to 3.0 after the procedure with resolution of thrombus after 5 weeks. No systemic embolization was noted at 1 year in that reported case. In a recent study, patients were noted to have reduced incidence of stroke (0.2%) with oral anticoagulation with warfarin for 30 days after mitral valve TEER compared with dual antiplatelet therapy.12 This indicates that selective use of oral anticoagulation, at least for a short period of time (4-6 weeks) after mitral valve TEER, rather than dual antiplatelet therapy, should be considered for patients with risk factors for left atrial thrombus formation such as a history of atrial fibrillation, heart failure, low appendage velocity, and elevated CHA2DS2-VASc score.
Conclusions
Acute LAA thrombus formation immediately after mitral valve TEER is a rare complication. The common clinical risk factors in 3 such reported cases of acute/subacute LAA thrombus formation immediately after mitral valve TEER, including our case report, were a history of atrial fibrillation, low LAA emptying velocity, severely impaired left ventricular systolic dysfunction (or heart failure with preserved ejection fraction), and elevated CHA2DS2-VASc score. All these factors increase the likelihood of stasis in the LAA, particularly immediately after mitral valve TEER. Post-procedural TEE is crucial in identifying this complication of mitral valve TEER. With appropriate post-procedural anticoagulation, the outcomes in the cases reported so far have been good without any episodes of systemic thromboembolism. Thus, in patients with the previously mentioned risk factors undergoing mitral valve TEER, perioperative use of systemic anticoagulation should be considered to prevent acute/subacute LAA appendage thrombus formation and address the associated thromboembolic risk.
Funding Support and Author Disclosures
The authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Footnotes
The authors attest they are in compliance with human studies committees and animal welfare regulations of the authors’ institutions and Food and Drug Administration guidelines, including patient consent where appropriate. For more information, visit the Author Center.
Appendix
For supplemental videos, please see the online version of this paper.
Appendix
Spontaneous echo-contrast and sludge in left atrial appendage immediately following mitral valve transcatheter edge to edge repair.
Progressive formation of a left atrial appendage thrombus along with persistent spontaneous echo-contrast, immediately following mitral valve transcatheter edge to edge repair.
Acute formation of a well-defined thrombus in the left atrial appendage immediately following mitral valve transcatheter edge to edge repair.
References
- 1.Feldman T., Kar S., Rinaldi M., et al. Percutaneous mitral repair with the MitraClip system: safety and midterm durability in the initial EVEREST (Endovascular Valve Edge-to-Edge REpair Study) cohort. J Am Coll Cardiol. 2009;54(8):686–694. doi: 10.1016/j.jacc.2009.03.077. [DOI] [PubMed] [Google Scholar]
- 2.Whitlow P.L., Feldman T., Pedersen W.R., et al. Acute and 12-month results with catheter-based mitral valve leaflet repair: the EVEREST II (Endovascular Valve Edge-to-Edge Repair) High Risk Study. J Am Coll Cardiol. 2012;59(2):130–139. doi: 10.1016/j.jacc.2011.08.067. [DOI] [PubMed] [Google Scholar]
- 3.Arora S., Vemulapalli S., Stebbins A., et al. The prevalence and impact of atrial fibrillation on 1-year outcomes in patients undergoing transcatheter mitral valve repair: results from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry. J Am Coll Cardiol Intv. 2019;12(6):569–578. doi: 10.1016/j.jcin.2018.12.012. [DOI] [PubMed] [Google Scholar]
- 4.Herrmann H.C., Gertz Z.M., Silvestry F.E., et al. Effects of atrial fibrillation on treatment of mitral regurgitation in the EVEREST II (Endovascular Valve Edge-to-Edge Repair Study) randomized trial. J Am Coll Cardiol. 2012;59(14):1312–1319. doi: 10.1016/j.jacc.2011.12.023. [DOI] [PubMed] [Google Scholar]
- 5.Silvestry F.E., Rodriguez L.L., Herrmann H.C., et al. Echocardiographic guidance and assessment of percutaneous repair for mitral regurgitation with the Evalve MitraClip: lessons learned from EVEREST I. J Am Soc Echocardiogr. 2007;20(10):1131–1140. doi: 10.1016/j.echo.2007.02.003. [DOI] [PubMed] [Google Scholar]
- 6.Nusca A., Bressi E., Colaiori I., Miglionico M., Di Sciascio G. Antiplatelet therapy in valvular and structural heart disease interventions. Cardiovasc Diagn Ther. 2018;8(5):678–693. doi: 10.21037/cdt.2018.06.08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bilge M., Saatci Yasar A., Ali S., Alemdar R. Left atrial spontaneous echo contrast and thrombus formation at septal puncture during percutaneous mitral valve repair with the MitraClip system of severe mitral regurgitation: a report of two cases. Anadolu Kardiyol Derg. 2014;14(6):549–550. doi: 10.5152/akd.2014.5355. [DOI] [PubMed] [Google Scholar]
- 8.Huntgeburth M., Muller-Ehmsen J., Brase C., Baldus S., Rudolph V. Thrombus formation at the MitraClip system during percutaneous mitral valve repair. J Am Coll Cardiol Intv. 2014;7(9):e111–e112. doi: 10.1016/j.jcin.2014.03.010. [DOI] [PubMed] [Google Scholar]
- 9.Glatthaar A., Seizer P., Patzelt J., Langer H., Schreieck J., Gawaz M. Formation of a left atrial thrombus during percutaneous mitral valve edge-to-edge repair induced by acute reduction of mitral regurgitation. J Cardiol Cases. 2018;17(1):33–35. doi: 10.1016/j.jccase.2017.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ohno Y., Attizzani G.F., Capodanno D., et al. Acute left atrial spontaneous echocardiographic contrast and suspicious thrombus formation following mitral regurgitation reduction with the MitraClip system. J Am Coll Cardiol Intv. 2014;7(11):1322–1323. doi: 10.1016/j.jcin.2014.04.027. [DOI] [PubMed] [Google Scholar]
- 11.Amaki M., Kataoka Y., Kanzaki H., et al. Subacute left atrial thrombus formation after percutaneous edge-to-edge mitral valve repair for functional mitral regurgitation. Circ J. 2018;82(6):1712–1713. doi: 10.1253/circj.CJ-17-0781. [DOI] [PubMed] [Google Scholar]
- 12.Geis N., Raake P., Kiriakou C., et al. Temporary oral anticoagulation after MitraClip—a strategy to lower the incidence of post-procedural stroke? Acta Cardiol. 2020;75(1):61–67. doi: 10.1080/00015385.2018.1550886. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Spontaneous echo-contrast and sludge in left atrial appendage immediately following mitral valve transcatheter edge to edge repair.
Progressive formation of a left atrial appendage thrombus along with persistent spontaneous echo-contrast, immediately following mitral valve transcatheter edge to edge repair.
Acute formation of a well-defined thrombus in the left atrial appendage immediately following mitral valve transcatheter edge to edge repair.