Abstract
Background:
Pregnancy is associated with physiological changes to meet the metabolic demands of the growing fetus. To understand adverse pregnancy outcomes it is important to establish vascular changes throughout pregnancy. We examined longitudinal changes in vascular measurements from prepregnancy through postpartum.
Materials and Methods:
Seventy women planning to conceive in Ulaanbaatar, Mongolia enrolled in this prospective study. Within 6 months, 44 (63%) had conceived; of which 36 (82%) delivered. Ten (28%) developed complex pregnancies and were analyzed separately. Vascular measures included central systolic blood pressure (cSBP), central diastolic blood pressure (cDBP), augmentation index corrected for heart rate of 75 beats/minute (AIx-75), and pulse wave velocity (PWV). Measurements were performed at prepregnancy, second trimester (22–24 weeks), third trimester (34–36 weeks), and 2 months postpartum. Missing values (n = 0–6 per time period) were replaced by multiple imputation. A repeated measures analysis of variance analyzed changes within individual women over the four time points adjusted for age, parity, and body mass index.
Results:
We observed significant reductions from prepregnancy to second trimester for mean Alx-75 (17.1%–12.6%; p = 0.006) and cSBP (91.7–86.8 mmHg; p = 0.03) but not for cDBP or PWV. Both mean AIx-75 and cSBP increased in third trimester. In the postpartum, cSBP returned to prepregnancy levels, whereas AIx-75 exceeded prepregnancy levels, although this fell short of significance (p = 0.09). Similar vascular patterns were observed in women with complex pregnancies for AIx-75; however, PWV increased from the second trimester and remained higher through postpartum, although not significant.
Conclusion:
In Mongolian women, we observed a decrease in AIx-75 and cSBP from prepregnancy through second trimester, which resolved postpartum. These results provide an understanding of changes across pregnancies in an Asian country. Future studies should assess vascular changes across pregnancies to determine if it can predict conditions such as pre-eclampsia.
Keywords: vascular health, pregnancy, prepregnancy, Mongolia
Introduction
Pregnancy-induced vascular changes develop early in pregnancy to meet the metabolic demands of the mother and growing fetus. To provide optimal support to the growing fetal-placental circulation, a variety of changes occur in the cardiovascular system, including an up to 40% increase in heart rate, cardiac output, and uterine artery blood flow.1–3 Peripheral vasodilatation leads to a 25%–30% decrease in systemic vascular resistance with a reduction in blood pressure until approximately week 18 of gestation, after which it increases toward term delivery.4 These changes and adaptations have been observed as early as 6 weeks of gestation; however, they are more profound after 12 weeks of gestation when the uteroplacental circulation begins.5
Vascular changes can be measured using noninvasive arterial tonometry to assess carotid-femoral pulse wave velocity (PWV), a measure of arterial stiffness and central blood pressure and augmentation index (AIx), a measure of arterial wave reflections. Both are predictive of future cardiovascular disease events6,7 and mortality.8 Studies in healthy pregnancies have found lower central systolic blood pressure (cSBP), central diastolic blood pressure (cDBP), and AIx during pregnancy compared with levels observed in nonpregnant women.9 Few studies have documented the trajectories of arterial measurements across healthy pregnancies,9–14 and those that have report a decrease in levels from the first to the second trimester. However, only one study has investigated the trajectory of arterial measurements from prepregnancy through pregnancy and into the postpartum period and this was collected in healthy British women.15
There remain several gaps of scientific evidence related to arterial changes during pregnancy. First, there is limited knowledge regarding changes from prepregnancy to pregnancy to postpartum across other ethnic diverse populations. Second, to understand conditions such as pre-eclampsia and preterm delivery,16–20 it is important to establish norms for arterial measurements throughout pregnancy. The vascular endothelium plays a crucial role in arterial tone and stiffness.
Third, early diagnosis of pre-eclampsia is important during pregnancy, but early detection is limited and predictive models are weak; noninvasive assessment of vascular measurements may play a role in improving risk prediction model for pre-eclampsia. Therefore, studying in populations such as Mongolia would provide further important insight into maternal cardiovascular adaptation and maladaptation in pregnancy. The purpose of our study was to examine longitudinal vascular changes from prepregnancy through the postpartum period in urban Mongolian women, using noninvasive arterial tonometry.
Materials and Methods
Study population
Mongolian women aged 18–40 years who were planning to conceive were eligible for this prospective study. To increase the likelihood of recruiting women who would become pregnant, we restricted enrollment to women who had not been trying to conceive for >4 months. Radio, newspaper, television advertisements, and posters in marriage registry offices were used to recruit women in Ulaanbaatar, the capital city of Mongolia. Interested women underwent a telephone screen to determine eligibility. Eligible women were scheduled for their first study appointment at either the Health Sciences University of Mongolia or the National Center for Maternal and Child Health (NCMCH) in Ulaanbaatar, Mongolia. Seven hundred forty-seven women responded to the study advertisements, of which 81 were eligible.
The primary reasons for ineligibility were as follows: current pregnancy or trying to become pregnant for >4 months (n = 439, 65.9%), lack of interest (n = 157, 23.6%), and not currently trying to get pregnant (n = 70, 10.5%). The first 70 eligible women were invited to join the study, and all 70 agreed to enroll. Women were given prenatal vitamins (Twin lab Prenatal Formula, Boca Raton, FL, USA) for their participation from enrollment through delivery and the postpartum period. Information was abstracted from pregnancy charts, including the physician's diagnosis of pre-eclampsia and gestational hypertension. The Institutional Review Boards at NCMCH, Cedars-Sinai Medical Center, the Harvard T.H. Chan School of Public Health, and the Ethical Review Board at the Ministry of Health, Mongolia all approved the study.
Sample size calculation
The study was designed as a feasibility study to evaluate the logistics of forming a prepregnancy cohort with tonometry throughout pregnancy. One of its goals was to document the proportion who would become pregnant in a 6-month time frame. The original sample was calculated to be a sample size of 60. However, when fewer women became pregnant than expected, the sample size was increased to 70. We reasoned that, assuming a 50% pregnancy rate and a 15% spontaneous abortion rate, enrolling 70 women attempting to conceive would yield 30 completed pregnancies.
Study protocol and visits
Written informed consent was obtained from participants at the baseline study visit (before conception). Urine pregnancy tests were performed to rule out current pregnancy. Women then completed a questionnaire that included a history of all prior pregnancies and any related complications. Height and weight were measured, and a cardiovascular assessment was conducted, including peripheral SBP and DBP measurement through sphygmomanometer and vascular tests through tonometry (described in vascular measurements section). At the end of the baseline visit, women were asked to contact the study staff if they became pregnant. After the baseline study visit, participants were contacted monthly by study staff to inquire about pregnancy status. All enrolled participants were followed for 6 months and assessed for pregnancy.
Women who reported that they were pregnant underwent three study visits after the baseline visit; these visits occurred during the second trimester (22–24 weeks), third trimester (34–36 weeks), and at 2 months postpartum (two visit occurred between 2 and 3 months; five occurred between 5 and 6 months). Vascular measurements were performed starting in second trimester due to effort and budgeting for this international stud, and taking into account that systemic vascular changes are more profound after week 12 of gestation.21 The gestational age was based on the first pregnancy ultrasound. At each visit, blood pressure, weight, and vascular function were assessed.
During the second trimester visit, we also collected information on pregnancy complications that had arisen during the pregnancy, such as high blood pressure and pre-eclampsia. Pre-eclampsia was physician-diagnosed and based on new onset of hypertension (persistent SBP ≥140 mmHg and DBP >90 mmHg) plus either proteinuria (>0.3 g/24 h) or other systemic symptoms.22,23 Preterm birth referred to a delivery that occurred <37 weeks of gestation, where 32 to <37 defined as moderate preterm, 28 to <32 very preterm, and <28 extremely preterm.24 At the postpartum visit, information on late pregnancy complications and pregnancy outcomes (including gestation length and birthweight) were additionally obtained.
Vascular measurements
Before noninvasive vascular testing, all study participants refrained from consuming caffeine for 24 hours and were instructed to fast for 8 hours. At each study visit, trained study staff measured peripheral SBP and DBP with a mercury sphygmomanometer and an appropriate-sized cuff. Two measurements separated by at least 5 minutes were taken from the right arm of subjects after being seated for at least 5 minutes; these measurements were then averaged for use in the analysis. Vascular testing was performed using noninvasive arterial tonometry (SphygmoCor, Atcor Medical, Sydney, Australia), as previously described.25–30
This device records the pulse waveform using tonometry applied to the radial artery. The arterial waveform is recorded and corrected for brachial pressure. This test produces a central pulse pressure waveform from the ascending aorta. The central pulse pressure is the vector sum of the forward vascular pressure from the heart and backward reflection pressure of the peripheral vascular system. The ability of a vessel to distend and increase volume with increasing pressure is quantified through a PWA, which determines the pressure waveform produced by arterial tonometry.
The peak and t trough of the pulse pressure waveform represent the cSBP and cDBP. PWA indices measure vascular compliance and include the augmentation pressure (AP) and AIx. AP is the additional pressure reflected from peripheral waves back toward the heart that is added to the central systolic peak pressure, whereas AIx is a ratio of the AP to the central pulse pressure.31 The AIx adjusted to a heart rate of 75 beats per minute (AIx-75) is expressed as a percent and is calculated as the difference between the magnitude of the forward and backward aortic pulse waves divided by the central pulse pressure.30,32,33 PWV measures the velocity of the pressure wave from the carotid to the femoral arteries with higher PWV indicates greater arterial stiffness.
Vascular tests were screened for quality by an expert reader (C.S.) for all measurements at a dedicated noninvasive vascular core laboratory (Cedars-Sinai Medical Center, Los Angeles, CA, USA). The mean of the first two measurements for each outcome was used in the analysis.
Statistical methods
To evaluate changes in vascular testing from prepregnancy to postpartum separately for each outcome of interest (cSBP and cDBP, peripheral SBP and DBP, AIx-75, and PWV), a repeated measures analysis of variance (repeated ANOVA) was used. Missing data were replaced using multiple imputation; no data were missing at baseline; three women (8%) were missing outcomes in the second trimester, six (17%) in the third trimester, and three (8%) postpartum. Specifically, 25 imputed data sets were created from regression (imputation) models, including the following variables considered to be important predictors of the missing outcomes: prepregnancy age, body mass index (BMI), history of hypertension (yes/no), and parity (yes/no).
The normality of dependent variables was evaluated graphically by examining the linearity of data in a quantile-quantile plot (Q-Q plot); all values were normally distributed, and log transformation did not improve normality. Unadjusted means were calculated at each time point, and a global F-test of the null hypothesis that the means were equal across time points was obtained from an unadjusted repeated measures ANOVA. We then adjusted each repeated measure ANOVA for prepregnancy age, parity (yes/no), and BMI. To depict the covariate-adjusted results graphically, these models were used to predict mean cSBP and cDBP, AIx-75, and PWV at ages 25, 30, and 35 years for all parous women using a mean BMI of 22 kg/m2.
Associations of age and parity with vascular function were also examined. Formal F-tests from the adjusted repeated measures ANOVA tested mean changes in outcomes: prepregnancy versus the second trimester; prepregnancy versus postpartum; and a global test comparing all four time points. A multivariate linear regression model was used to determine the effect of maternal age on vascular outcomes during pregnancy. In the regression model, Alx-75, cSBP, cDBP, and PWV during pregnancy were taken as the dependent variables. The main independent variable was maternal age, and other control variables were parity and BMI. Statistical analyses were performed using SAS (version 9.4; SAS Institute, Inc., Cary, NC, USA) statistical software.
Results
Study participants
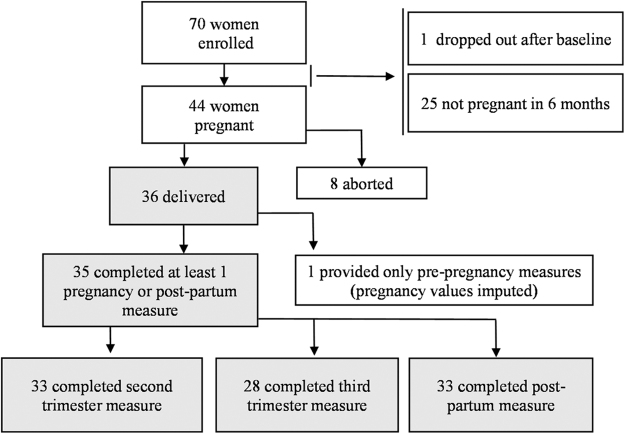
Of the 70 enrolled women, 44 (64%) became pregnant within 6 months, 8 (18%) subsequently aborted, whereas the remaining 36 women (82% of those who became pregnant) were followed through postpartum and were included in our analyses (Fig. 1). There are no differences in the baseline measurements between the women who did and did not conceive within 6 months. Ten women developed complex pregnancies: 2.7% (n = 1) developed pre-eclampsia, 16.7% (n = 6) had gestational hypertension, and 8.3% (n = 3) delivered late preterm between 35.2 and 35.4 weeks of gestational age, of which one delivered a baby with low birth weight (2400 g). Table 1 depicts the characteristics of the study participants.
FIG. 1.
Participant flowchart. Participants in the gray shaded boxes are included in the current analysis.
Table 1.
Characteristics of Study Participants
| All (n = 36) | Nulliparous (n = 10) | Parous (n = 26) | Noncomplex (n = 26) | Complex (n = 10) | |
|---|---|---|---|---|---|
| Agea | 30.3 (5.7) | 27.2 (5.1) | 31.4 (5.6) | 30.7 (5.7) | 29.9 (4.5) |
| Prepregnancy BMIa | 22.0 (2.9) | 22.1 (2.7) | 22.0 (3.1) | 22.1 (3.0) | 21.9 (2.1) |
| Ever smoker(%) | 14 | 10 | 15 | 17 | 10 |
| Parental history of hypertension(%) | 67 | 70 | 65 | 58 | 76 |
| Gestational length (weeks)a | 39.6 (3.6) | 38.6 (2.2) | 40.6 (2.6) | 40.0 (1.9) | 37.2 (1.8) |
| Birthweight (g)a | 3462 (590) | 3461 (718) | 3463 (551) | 3577 (621) | 3209 (444) |
Mean (standard deviation).
BMI, body mass index.
Vascular testing from prepregnancy to postpartum
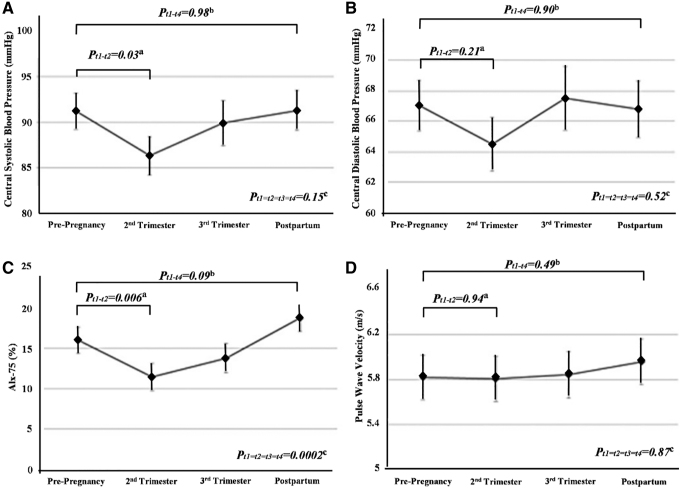
Unadjusted means for vascular measurements over time are presented in Table 2 and predicted means from models adjusted for age, parity, and BMI are shown in Figure 2. Mean prepregnancy SBP was 101.8 (12.2) and DBP was 66.1 (7.6), consistent with a recent study among Mongolian women of reproductive age.34 Figure 2 depicts predicted values for a 30-year-old parous woman of average BMI (22 kg/m2). There was a statistically significant 4.92 mm Hg (95% confidence interval [CI]: −9.37 to −0.48) drop in the adjusted mean cSBP from prepregnancy to the second trimester (Fig. 2A, p = 0.03); however, this result must be interpreted with caution given that the global test of mean differences across the time periods was not significant (p = 0.15).
Table 2.
Unadjusted Cardiovascular Measures from Prepregnancy to Pregnancy to Postpartum (n = 36)
| Measurement | Prepregnancy |
Second trimester |
Third trimester |
Postpartum |
p
a
|
|---|---|---|---|---|---|
| Mean (SD) | |||||
| All pregnancies: n = 36 | |||||
| AIx-75 (%) | 17.1 (9.9) | 12.6 (10.4) | 14.8 (10.3) | 19.8 (10.4) | 0.0002 |
| Peripheral SBP (mmHg) | 101.8 (12.2) | 99.3 (9.9) | 103.1 (13.6) | 100.4 (13.5) | 0.60 |
| Peripheral DBP (mmHg) | 66.1 (7.6) | 63.6 (8.3) | 66.1 (10.3) | 65.9 (9.1) | 0.57 |
| Central SBP (mmHg) | 91.7 (10.6) | 86.8 (9.0) | 90.3 (13.0) | 91.7 (13.8) | 0.15 |
| Central DBP (mmHg) | 67.3 (7.7) | 64.8 (7.9) | 67.8 (11.2) | 67.0 (9.9) | 0.52 |
| PWV (m/s) | 6.01 (1.18) | 5.99 (1.09) | 6.03 (1.16) | 6.15 (1.09) | 0.87 |
| Noncomplex pregnancy: n = 26 | |||||
| AIx-75 (%) | 16.2 (9.7) | 13.0 (10.1) | 15.6 (10.1) | 20.8 (10.8) | 0.0005 |
| Peripheral SBP (mmHg) | 101.3 (10.5) | 98.6 (10.1) | 95.5 (15.6) | 99.4 (11.0) | 0.88 |
| Peripheral DBP (mmHg) | 65.6 (7.0) | 63.2 (7.1) | 62.6 (9.0) | 65.7 (8.5) | 0.48 |
| Central SBP (mmHg) | 90.9 (9.3) | 86.6 (9.0) | 86.8 (12.6) | 91.1 (12.4) | 0.26 |
| Central DBP (mmHg) | 66.8 (7.1) | 64.6 (7.4) | 64.8 (8.7) | 66.4 (8.6) | 0.73 |
| PWV (m/s) | 6.0 (1.3) | 5.9 (1.1) | 6.0 (1.1) | 6.2 (1.0) | 0.78 |
| Complex pregnancy: n = 10 | |||||
| AIx-75 (%) | 20.3 (11.6) | 11.2 (12.1) | 14.9 (11.1) | 18.8 (9.3) | 0.005 |
| Peripheral SBP (mmHg) | 101.5 (17.3) | 101.4 (9.9) | 110.2 (13.8) | 103.3 (21.0) | 0.28 |
| Peripheral DBP (mmHg) | 64.3 (7.3) | 65.8 (8.8) | 73.6 (12.7) | 66.3 (13.1) | 0.23 |
| Central SBP (mmHg) | 92.1 (15.1) | 88.6 (9.9) | 97.7 (13.0) | 93.7 (19.6) | 0.583 |
| Central DBP (mmHg) | 65.7 (7.6) | 67.1 (9.0) | 75.2 (12.7) | 67.4 (13.3) | 0.65 |
| PWV (m/s) | 6.09 (0.95) | 6.34 (1.11) | 6.47 (1.27) | 6.49 (1.14) | 0.26 |
Significant differences are marked in bold.
p-Value is from unadjusted repeated measures analysis of variance testing the null hypothesis that the means at each time point are all equal (prepregnancy = second trimester = third trimester = postpartum).
AIx-75, augmentation index adjusted to a heart rate of 75 beats per minute; DBP, diastolic blood pressure; PWV, pulse wave velocity; SBP, systolic blood pressure.
FIG. 2.
Vascular changes from prepregnancy to postpartum. Each data point is predicted from a linear mixed model at for parous women at mean BMI (22 kg/m2). (A) cSBP; (B) cDBP; (C) AIx-75%; (D) PWV. aValue is from a linear mixed model adjusting for age, parity, and BMI comparing the second trimester mean cSBP (A), cDBP (B), AIx-75 (C), and PWV (D) to the prepregnancy mean cSBP (A), cDBP (B), AIx-75 (C), and PWV (D). bp-Value is from a linear mixed model adjusting for age, parity, and BMI comparing the postpartum mean cSBP (A), cDBP (B), AIx-75 (C), and PWV (D) to the prepregnancy mean cSBP (A), cDBP (B), AIx-75 (C), and PWV (D). cGlobal test p-value from a linear mixed model adjusting for age, parity, and BMI testing whether the means of cSBP (A), cDBP (B), AIx-75 (C), and PWV (D) are different for at least one time point. Each data point is predicted from a linear mixed model at for parous women at mean BMI (22 kg/m2). AIx-75, augmentation index adjusted to a heart rate of 75 beats per minute; BMI, body mass index; cDBP, central diastolic blood pressure; cSBP, central systolic blood pressure; PWV, pulse wave velocity.
cDBP also appeared to drop in the second trimester; however, this change in the adjusted mean was not statistically significant (Fig. 2B). Figure 2C shows predicted means of Alx-75 across time periods. We observed statistically significant differences in the adjusted means across the time periods (p < 0.0002), which was primarily driven by the significant 4.5% (95% CI: −7.7 to −1.3) drop in AIx-75 from prepregnancy to the second trimester. AIx-75 levels were qualitatively higher after pregnancy than before, but this was not statistically significant (p = 0.09). In contrast, mean PWV did not change from prepregnancy, through pregnancy, and into the postpartum period (Fig. 2D).
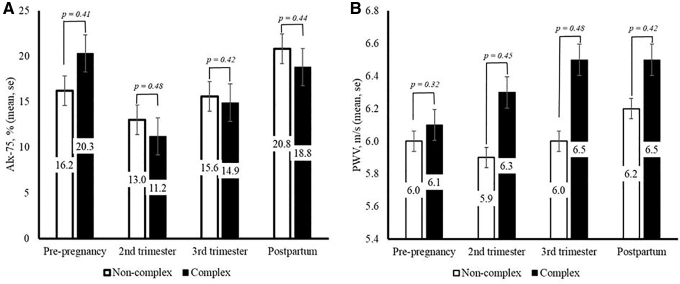
Similar patterns were observed when noncomplex and complex pregnancies were examined separately (Table 2). There were no statistically significant differences between means of complex and noncomplex pregnancies at each time point (Fig. 3). In contrast to the noncomplex pregnancies, in the complex pregnancies, PWV appeared to increase from the second trimester and remain high through postpartum (Fig. 3B); however, these differences were not statistically significant, perhaps due to the small size and heterogeneity of the complex pregnancies.
FIG. 3.
Vascular changes from pre-pregnancy to postpartum by pregnancy complications. (A) AIx-75; (B) pulse wave velocity.
Effect of age on vascular measurements
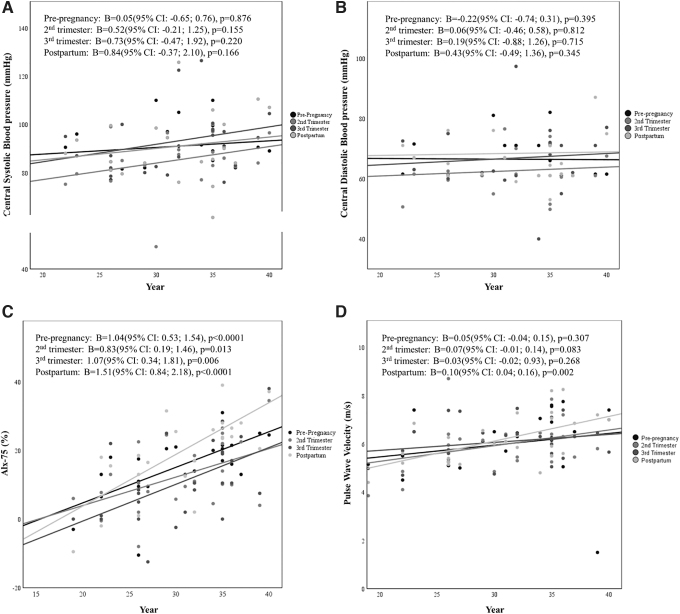
The results of the multiple linear regression analysis found that in noncomplex pregnancies, for every 1-year increase in maternal age, AIx-75 increases by 1.04%, 0.83%, 1.07%, and 1.51% during prepregnancy, second trimester, third trimester, and postpartum, respectively (all p < 0.05). In contrast, PWV was not significantly associated with age during pregnancy, but was significantly higher postpartum in older women (p = 0.002; Fig. 4).
FIG. 4.
Linear regression between age and vascular change during pre-pregnancy, pregnancy, and postpartum in noncomplex pregnancies. (A) cSBP; (B) cDBP; (C) AIx-75; (D) PWV. se, standard error.
Discussion
Our prospective study reports changes in maternal vascular function from prepregnancy to pregnancy to postpartum in a group of Mongolian women recruited from the general population. Central blood pressures and AIx-75 values were lowest in the second trimester, although the drop in cDBP was not statistically significant. AIx-75 appeared to increase in the third trimester through the postpartum period; the other parameters also appeared to increase after the second trimester nadir, but these changes were not statistically significant over the four time points.
Patterns observed in this study are similar to those reported by the only other longitudinal study examining vascular measures from prepregnancy to the postpartum. In a cohort of 54 healthy British pregnant women, Mahendru and colleagues reported drops in central blood pressure and AIx in early pregnancy as compared with prepregnancy.15 In the second trimester, central blood pressure and AIx reached their lowest points, before rebounding to early pregnancy levels by the third trimester and postpartum, demonstrating a U-shaped trajectory. Similar to our results, the drop in blood pressure seen during pregnancy should not account for the change in AIx as pulse pressure remains the same over each trimester.
The unadjusted PWV decreased significantly from preconception levels in the second trimester, remained lower in the third trimester, but increased back to baseline levels in the postpartum period.12,15 This is also consistent with other cross-sectional studies' findings that early pregnancy is a time of lower PWV, perhaps coinciding with placentation, observed across multiple studies in the context of increased cardiac output (from 20% to 50%), a drop in mean arterial pressure associated with vasodilatation,1,35,36 and hormonal variations associated with implantation.37,38 In contrast, the higher postpartum arterial wave reflection observed in our study is not consistent with the previously reported tendency of blood pressure to become lower with increasing parity.39
Pregnancy complications such as pre-eclampsia are thought to be related to vascular abnormalities that persist after delivery. Women who experience pre-eclampsia are at a higher risk for future cardiovascular disease.23,40–47 However, the vascular etiology of pre-eclampsia remains incompletely understood. It is unclear whether women who develop pre-eclampsia enter pregnancy with vascular abnormalities or if they develop vascular abnormalities during the pregnancy. It is important to understand and establish vascular changes during a healthy pregnancy to understand pregnancies characterized by adverse outcomes related to vascular dysfunction,33,40–44,48 such as pre-eclampsia16,33,40,42,45,46 and preterm delivery.17,41,44,48
Vascular stiffness and arterial wave reflection were measured in a small group of women that developed complex pregnancies at delivery, including late preterm delivery, gestational hypertension, and pre-eclampsia in our study. Although our statistical power was limited to contrast the complex and noncomplex pregnancies, there appeared to be an interesting pattern in which arterial stiffness increased and remained high postpartum in complex pregnancies. As other studies have found, in normal pregnancies arterial stiffness typically falls as vasodilation occurs; therefore, our nonsignificant observation of rising PWV in complex pregnancies should be further explored in future studies. Foo et al. recently examined total peripheral resistance and found that women who develop pre-eclampsia or fetal growth restriction have a hemodynamic profile characterized by low cardiac output, high vascular resistance, and higher prepregnancy blood pressure.
Although studies report that advanced maternal age increases the risk of vascular dysfunction49 and pregnancy complications,50–52 there remain limited data regarding the impact of aging on the maternal vascular system. Aging is associated with increased vascular stiffness and increased wave reflection in the general population.53–55 However, studies have found that age-related changes in AIx are more marked in younger individuals, whereas aortic PWV increases significantly in individuals after the age 50 years.56 We observed a similar pattern in prepregnancy, where only AIx consistently increased with age among these women of reproductive age. This longitudinal study on preconception through postpartum adds more information to existing evidence suggesting that, despite cardiovascular adaptations occurring in pregnancy, vascular changes due to aging observed prepregnancy are evident throughout pregnancy and into the postpartum.
The primary limitation of this study was the modest sample size relative to the variability of some of the outcome measures. However, although there was some movement of participants in and out of our medical center, which prevented the collection of every vascular measurement at every time point, the overall rate of follow-up was high. Moreover, we were able to impute the relatively small amount (3%) of missing data to retain all participants who delivered in our analysis, increasing the power of the analysis and producing more generalizable results.57,58
Furthermore, due to the scope of the study, we could not perform echocardiography and report cardiac output and vascular resistance as others have. Additional strengths of this study included its use of validated vascular testing methods and protocols within the same environment, reducing measurement-related variability.28,30 Furthermore, all pregnancies were confirmed by urine test. To our knowledge, this is the second published study to report maternal arterial function from preconception through pregnancy and postpartum.
Conclusion
We report longitudinal cardiovascular parameters from prepregnancy, across pregnancy, and through the postpartum period in Mongolia, extending the current evidence of the female vascular changes that occur during pregnancy. In women who developed complex pregnancies we observed that arterial stiffness in complex pregnancies increased throughout pregnancy and remained elevated postpartum, although not significant due to limited sample size. Future studies should focus on whether similar changes occur in pregnancies that subsequently result in pre-eclampsia or preterm birth and to what extent arterial function throughout pregnancy can predict such pregnancy-related complications.
Authors' Contributions
Conception and design, data collection, acquisition, interpretation, writing, and editing by D.E. Conception, interpretation, writing, revision, and editing by G.D. Literature review, interpretation, and analysis by L.J.T. Design, interpretation, first draft, acquisition, analysis, revision, and editing by J.W.R.-E. Literature review, data cleaning, and initial analysis by J.J.S. Conception and design, revision, and editing by S.E., M.B., and C.N.B.M. Literature review, data collection, and data entry by G.G. and B.B. Interpretation and analysis by G.F. and E.N. Conception and design, acquisition, interpretation, data validity, writing, editing by C.L.S. All authors approved the final article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported the National Heart, Lung and Blood Institutes (Grant No. K23HL127262), the UNESCO-L'Oreal Fellowship for Young Women in Life Sciences, the Society for Women's Health Research and the Edythe L. Broad and the Constance Austin Women's Heart Research Fellowships, Cedars-Sinai Medical Center, Los Angeles, California, the Barbra Streisand Women's Cardiovascular Research and Education Program, Cedars-Sinai Medical Center, Los Angeles, Washington, DC, USA, The Linda Joy Pollin Women's Heart Health Program, and the Erika Glazer Women's Heart Health Project, Cedars-Sinai Medical Center, Los Angeles, CA, USA.
References
- 1. Duvekot JJ, Cheriex EC, Pieters FA, Menheere PP, Peeters LH. Early pregnancy changes in hemodynamics and volume homeostasis are consecutive adjustments triggered by a primary fall in systemic vascular tone. Am J Obstet Gynecol 1993;169:1382–1392. [DOI] [PubMed] [Google Scholar]
- 2. Lees MM, Taylor SH, Scott DB, Kerr MG. A study of cardiac output at rest throughout pregnancy. J Obstet Gynaecol Br Commonw 1967;74:319–328. [DOI] [PubMed] [Google Scholar]
- 3. Clark SL, Cotton DB, Lee W, et al. Central hemodynamic assessment of normal term pregnancy. Am J Obstet Gynecol 1989;161(6 Pt 1):1439–1442. [DOI] [PubMed] [Google Scholar]
- 4. Macdonald-Wallis C, Lawlor DA, Fraser A, May M, Nelson SM, Tilling K. Blood pressure change in normotensive, gestational hypertensive, preeclamptic, and essential hypertensive pregnancies. Hypertens 2012;59:1241–1248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Mahendru AA, Everett TR, Wilkinson IB, Lees CC, McEniery CM. Maternal cardiovascular changes from pre-pregnancy to very early pregnancy. J Hypertens 2012;30:2168–2172. [DOI] [PubMed] [Google Scholar]
- 6. Vlachopoulos C, Aznaouridis K, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with arterial stiffness: A systematic review and meta-analysis. J Am Coll Cardiol 2010;55:1318–1327. [DOI] [PubMed] [Google Scholar]
- 7. Ben-Shlomo Y, Spears M, Boustred C, et al. Aortic pulse wave velocity improves cardiovascular event prediction: An individual participant meta-analysis of prospective observational data from 17,635 subjects. J Am Coll Cardiol 2014;63:636–646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Wang K-L, Cheng H-M, Sung S-H, et al. Wave reflection and arterial stiffness in the prediction of 15-year all-cause and cardiovascular mortalities. Hypertension 2010;55:799–805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Macedo ML, Luminoso D, Savvidou MD, McEniery CM, Nicolaides KH. Maternal wave reflections and arterial stiffness in normal pregnancy as assessed by applanation tonometry. Hypertens 2008;51:1047–1051. [DOI] [PubMed] [Google Scholar]
- 10. Fujime M, Tomimatsu T, Okaue Y, et al. Central aortic blood pressure and augmentation index during normal pregnancy. Hypertens Res 2012;35:633–638. [DOI] [PubMed] [Google Scholar]
- 11. Osman MW, Nath M, Khalil A, Webb DR, Robinson TG, Mousa HA. Longitudinal study to assess changes in arterial stiffness and cardiac output parameters among low-risk pregnant women. Pregnancy Hypertens 2017;10:256–261. [DOI] [PubMed] [Google Scholar]
- 12. Gomez YH, Hudda Z, Mahdi N, et al. Pulse pressure amplification and arterial stiffness in low-risk, uncomplicated pregnancies. Angiology 2016;67:375–383. [DOI] [PubMed] [Google Scholar]
- 13. Mersich B, RigO J, LEnArd Z, Studinger P, Visontai Z, Kollai M. Carotid artery stiffening does not explain baroreflex impairment in pre-eclampsia. Clin Sci (Lond) 2004;107:407–413. [DOI] [PubMed] [Google Scholar]
- 14. Khalil A, Jauniaux E, Cooper D, Harrington K. Pulse wave analysis in normal pregnancy: A prospective longitudinal study. PLoS One 2009;4:e6134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Mahendru AA, Everett TR, Wilkinson IB, Lees CC, McEniery CM. A longitudinal study of maternal cardiovascular function from preconception to the postpartum period. J Hypertens 2014;32:849–856. [DOI] [PubMed] [Google Scholar]
- 16. Kirollos S, Skilton M, Patel S, Arnott C. A systematic review of vascular structure and function in pre-eclampsia: Non-invasive assessment and mechanistic links. Front Cardiovasc Med 2019;6:166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Minissian MB, Kilpatrick S, Shufelt CL, et al. Vascular function and serum lipids in women with spontaneous preterm delivery and term controls. J Womens Health (Larchmt) 2019;28:1522–1528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Irgens HU, Reisaeter L, Irgens LM, Lie RT. Long term mortality of mothers and fathers after pre-eclampsia: Population based cohort study. BMJ 2001;323:1213–1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Robb AO, Mills NL, Din JN, et al. Influence of the menstrual cycle, pregnancy, and preeclampsia on arterial stiffness. Hypertens 2009;53:952–958. [DOI] [PubMed] [Google Scholar]
- 20. Catov JM, Newman AB, Roberts JM, et al. Preterm delivery and later maternal cardiovascular disease risk. Epidemiology 2007;18:733–739. [DOI] [PubMed] [Google Scholar]
- 21. Mustafa R, Ahmed S, Gupta A, Venuto RC. A comprehensive review of hypertension in pregnancy. J Pregnancy 2012;2012:105918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Emery SP. Hypertensive disorders of pregnancy: Overdiagnosis is appropriate. Cleve Clin J Med 2005;72:345–352. [DOI] [PubMed] [Google Scholar]
- 23. Marchand NE, Davaasambuu G, McElrath TF, Davaasambuu E, Baatar T, Troisi R. Prevalence of pregnancy hypertensive disorders in Mongolia. Pregnancy Hypertens 2016;6:413–417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Robinson JN, Norwitze E (2021) Preterm Birth: Risk factors, interventions for risk reduction and maternal prognosis. In Barss V (Ed). Up to Date. Retrieved on February 01, 2022. [Google Scholar]
- 25. Smith SA, Morris JM, Gallery EDM. Methods of assessment of the arterial pulse wave in normal human pregnancy. Am J Obstet Gynecol 2004;190:472–476. [DOI] [PubMed] [Google Scholar]
- 26. Karamanoglu M, O'Rourke MF, Avolio AP, Kelly RP. An analysis of the relationship between central aortic and peripheral upper limb pressure waves in man. Eur Heart J 1993;14:160–167. [DOI] [PubMed] [Google Scholar]
- 27. Pauca AL, O'Rourke MF, Kon ND. Prospective evaluation of a method for estimating ascending aortic pressure from the radial artery pressure waveform. Hypertens 2001;38:932–937. [DOI] [PubMed] [Google Scholar]
- 28. Wilkinson IB, Fuchs SA, Jansen IM, et al. Reproducibility of pulse wave velocity and augmentation index measured by pulse wave analysis. J Hypertens 1998;16(12 Pt 2):2079–2084. [DOI] [PubMed] [Google Scholar]
- 29. Butlin M, Qasem A. Large artery stiffness assessment using SphygmoCor technology. Pulse (Basel, Switzerland) 2017;4:180–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Wilkinson IB, MacCallum H, Flint L, Cockcroft JR, Newby DE, Webb DJ. The influence of heart rate on augmentation index and central arterial pressure in humans. J Physiol 2000;525 Pt 1(Pt 1):263–270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Vlachopoulos C, O'Rourke M, Nichols WW. McDonald's blood flow in arteries: Theoretical, experimental and clinical principles. Hodder Arnold, London, UK. 2011. [Google Scholar]
- 32. Wilkinson IB, Mohammad NH, Tyrrell S, et al. Heart rate dependency of pulse pressure amplification and arterial stiffness. Am J Hypertens 2002;15(1 Pt 1):24–30. [DOI] [PubMed] [Google Scholar]
- 33. Kaihura C, Savvidou MD, Anderson JM, McEniery CM, Nicolaides KH. Maternal arterial stiffness in pregnancies affected by preeclampsia. Am J Physiol Heart Circ Physiol 2009;297:H759–H764. [DOI] [PubMed] [Google Scholar]
- 34. Potts H, Baatarsuren U, Myanganbayar M, et al. Hypertension prevalence and control in Ulaanbaatar, Mongolia. J Clin Hypertens (Greenwich) 2020;22:103–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Turi V, Dragan S, Iurciuc M, et al. Arterial function in healthy pregnant women vs. non-pregnant women: A 10-year study. Diagnostics (Basel, Switzerland) 2020;10:374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Carlin A, Alfirevic Z. Physiological changes of pregnancy and monitoring. Best Pract Res Clin Obstet Gynaecol 2008;22:801–823. [DOI] [PubMed] [Google Scholar]
- 37. Chapman AB, Abraham WT, Zamudio S, et al. Temporal relationships between hormonal and hemodynamic changes in early human pregnancy. Kidney Int 1998;54:2056–2063. [DOI] [PubMed] [Google Scholar]
- 38. Conrad KP. Emerging role of relaxin in the maternal adaptations to normal pregnancy: Implications for preeclampsia. Semin Nephrol 2011;31:15–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Haug EB, Horn J, Markovitz AR, et al. The impact of parity on life course blood pressure trajectories: The HUNT study in Norway. Eur J Epidemiol 2018;33:751–761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Stuart JJ, Tanz LJ, Missmer SA, et al. Hypertensive disorders of pregnancy and maternal cardiovascular disease risk factor development: An observational cohort study. Ann Intern Med 2018;169:224–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Wu P, Gulati M, Kwok CS, et al. Preterm delivery and future risk of maternal cardiovascular disease: A systematic review and meta-analysis. J Am Heart Assoc 2018;7:e007809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Wu P, Haththotuwa R, Kwok CS, et al. Preeclampsia and future cardiovascular health: A systematic review and meta-analysis. Circ Cardiovasc Qual Outcomes 2017;10:e003497. [DOI] [PubMed] [Google Scholar]
- 43. Yinon Y, Kingdom JCP, Odutayo A, et al. Vascular dysfunction in women with a history of preeclampsia and intrauterine growth restriction: Insights into future vascular risk. Circulation 2010;122:1846–1853. [DOI] [PubMed] [Google Scholar]
- 44. Tanz LJ, Stuart JJ, Williams PL, et al. Preterm delivery and maternal cardiovascular disease risk factors: The nurses' health study II. J Womens Health (Larchmt) 2019;28:677–685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Stuart JJ, Tanz LJ, Cook NR, et al. Hypertensive disorders of pregnancy and 10-year cardiovascular risk prediction. J Am Coll Cardiol 2018;72:1252–1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Enkhmaa D, Wall D, Mehta PK, et al. Preeclampsia and vascular function: A window to future cardiovascular disease risk. J Womens Health (Larchmt) 2016;25:284–291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Elvan-Taşpinar A, Bots ML, Franx A, Bruinse HW, Engelbert RHH. Stiffness of the arterial wall, joints and skin in women with a history of pre-eclampsia. J Hypertens 2005;23:147–151. [DOI] [PubMed] [Google Scholar]
- 48. Tanz LJ, Stuart JJ, Williams PL, et al. Preterm delivery and maternal cardiovascular disease in young and middle-aged adult women. Circulation 2017;135:578–589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Morton JS, Care AS, Kirschenman R, Cooke C-L, Davidge ST. Advanced maternal age worsens postpartum vascular function. Front Physiol 2017;8:465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Luo J, Fan C, Luo M, Fang J, Zhou S, Zhang F. Pregnancy complications among nulliparous and multiparous women with advanced maternal age: A community-based prospective cohort study in China. BMC Pregnancy Childbirth 2020;20:581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Shan D, Qiu P-Y, Wu Y-X, et al. Pregnancy outcomes in women of advanced maternal age: A retrospective cohort study from China. Sci Rep 2018;8:12239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Sauer MV. Reproduction at an advanced maternal age and maternal health. Fertil Steril 2015;103:1136–1143. [DOI] [PubMed] [Google Scholar]
- 53. Csipo T, Lipecz A, Fulop GA, et al. Age-related decline in peripheral vascular health predicts cognitive impairment. GeroScience 2019;41:125–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Camici GG, Savarese G, Akhmedov A, Lüscher TF. Molecular mechanism of endothelial and vascular aging: Implications for cardiovascular disease. Eur Heart J 2015;36:3392–3403. [DOI] [PubMed] [Google Scholar]
- 55. Lakatta EG. So! What's aging? Is cardiovascular aging a disease? J Mol Cell Cardiol 2015;83:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. McEniery CM, Yasmin, Hall IR, Qasem A, Wilkinson IB, Cockcroft JR. Normal vascular aging: Differential effects on wave reflection and aortic pulse wave velocity: The Anglo-Cardiff Collaborative Trial (ACCT). J Am Coll Cardiol 2005;46:1753–1760. [DOI] [PubMed] [Google Scholar]
- 57. Clark TG, Altman DG. Developing a prognostic model in the presence of missing data: An ovarian cancer case study. J Clin Epidemiol 2003;56:28–37. [DOI] [PubMed] [Google Scholar]
- 58. Royston P. Multiple imputation of missing values. Stata J 2004;4:227–241. [Google Scholar]