Abstract
Background:
Hepatic artery infusion (HAI) is a liver-directed therapy that delivers high-dose chemotherapy to the liver through the hepatic arterial system for colorectal liver metastases and intrahepatic cholangiocarcinoma. Utilization of HAI is rapidly expanding worldwide.
Objective and Methods:
This review describes the conduct of HAI pump implantation, with focus on common technical pitfalls and their associated solutions. Perioperative identification and management of common postoperative complications is also described.
Results:
HAI therapy is most commonly performed with the surgical implantation of a subcutaneous pump, and placement of its catheter into the hepatic arterial system for inline flow of pump chemotherapy directly to the liver. Intraoperative challenges and abnormal hepatic perfusion can arise due to aberrant anatomy, vascular disease, technical or patient factors. However, solutions to prevent or overcome technical pitfalls are present for the majority of cases. Postoperative HAI-specific complications arise in 22% to 28% of patients in the form of pump pocket (8%–18%), catheter (10%–26%), vascular (5%–10%), or biliary (2%–8%) complications. The majority of patients can be rescued from these complications with early identification and aggressive intervention to continue to deliver safe and effective HAI therapy.
Conclusions:
This HAI toolkit provides the HAI team a reference to manage commonly encountered HAI-specific perioperative obstacles and complications. Overcoming these challenges is critical to ensure safe and effective pump implantation and delivery of HAI therapy, and key to successful implementation of new programs and expansion of HAI to patients who may benefit from such a highly specialized treatment strategy.
Keywords: colorectal liver metastases, hepatic artery infusion pump
Hepatic artery infusion (HAI) is a liver-directed treatment strategy to deliver high-dose chemotherapy directly to the liver by way of the hepatic arterial system. In the modern era, HAI is most commonly performed by an implantable subcutaneous pump and intra-arterial catheter, which is the focus of this review. HAI is generally considered in three clinical scenarios: unresectable colorectal liver metastases (CRLM), resected CRLM as adjuvant therapy, and in the setting of liver-confined advanced intrahepatic cholangiocarcinoma.1–6 HAI therapy takes advantage of inherent liver physiology: primary and secondary hepatic malignancies derive vascularity disproportionately from the hepatic arteries, whereas normal parenchyma receives dual blood supply from the arterial and portal venous circulations.7–14 Furthermore, the cornerstone of HAI chemotherapy is floxuridine (FUDR), a pro-drug of 5-fluorouracil, which has the ideal pharmacokinetic and pharmacodynamic properties of high first-pass hepatic metabolism and a short half-life.15–17 These factors allow for physiologic and pharmacologic isolation of the liver and the delivery of high concentrations of FUDR to hepatic tumors while sparing normal liver parenchyma and limiting systemic exposure and therefore systemic toxicity. Due to the complex and specialized nature of this therapy and unique complication profile, HAI was predominantly adopted at only a few specialized centers over the past 4 decades.
Along with advances in systemic chemotherapy, most commonly administered concurrently with HAI in the modern era, recent single and multi-institutional studies have demonstrated that HAI can be safely implemented at other tertiary referral hospitals in the United States and internationally.4,18–22 The overall objective of HAI is to treat and control liver disease burden, increase the number of patients eligible for definitive resection, or provide liver-specific adjuvant therapy to decrease hepatic recurrence. Although few centers historically utilized HAI routinely, > 15 new HAI programs have formed in the past 5 years in the United States and Europe. Given the rapid expansion of HAI programs and need for ongoing analysis of patient selection and clinical outcomes, a consortium was established in 2020, which now consists of 50 international centers actively using HAI or in the process of implementation. The HAI Consortium Research Network (HCRN) will serve as a resource for new HAI programs, allow for retrospective evaluation of HAI therapy and outcomes, and importantly provide a collaborative effort to design and execute contemporary prospective, multicenter trials.
The present body of HAI literature primarily originates from surgeons and medical oncologists that have trained at Memorial Sloan Kettering Cancer Center, where the majority of research and practice of this therapy has oringinated.1–3,5,11,23,24 Many of the surgical pearls and chemotherapeutic management recommendations have been experiential or communicated provider to provider within a limited network. Program implementation and development have been described previously and are not the focus of this review.4,18–21 However, as more surgeons and trainees encounter HAI and this tool is expanded to new programs nationally and internationally, a reference for operative decision-making and management of HAI-specific complications is needed to ensure the safety and continued effectiveness of this therapy.
The goal of this review is to create an HAI toolkit to address frequently encountered intraoperative challenges, guide perioperative management, and help identify and salvage early and late complications of HAI device implantation and therapy. This toolkit aims to provide a relevant and critical reference for practitioners treating patients with HAI to improve outcomes.
SURGICAL PLANNING AND PUMP IMPLANTATION
Preoperative Imaging
HAI chemotherapy must be delivered to the liver via a single inflow vessel to ensure perfusion of the entire liver without flow to adjacent organs. In addition, there must be a conduit by which the catheter can deliver chemotherapy without obstructing hepatic arterial inflow. Computed tomography angiography (CTA) is performed preoperatively to assess the relevant vascular anatomy, pathology and anomalies. Specifically and most commonly, replaced or accessory hepatic arteries are identified preoperatively to guide surgical planning. In cases of renal impairment or severe contrast allergy, MR angiography can also be considered. Catheter-based angiography is now rarely utilized due to the invasiveness of the diagnostic modality, but it enables diagnostic and therapeutic evaluation in the appropriate clinical setting (eg, celiac stenosis).
Operative Technique
HAI pump placement is traditionally performed as an open procedure as previously described.12 Minimally invasive techniques, including robot-assisted implantation, have more recently been adopted and are likely to become standard as experience grows.25–28 Regardless of the surgical approach, key steps to the procedure are the same and include exploration to confirm there is no extrahepatic disease which would preclude HAI pump placement. After abdominal entry, the falciform ligament and ligamentum teres are divided to prevent perfusion of the abdominal wall via terminal branches from the hepatic artery. Next, the right gastric artery is divided and the hepatic artery lymph node is resected to gain access to the common hepatic artery and identify the gastroduodenal artery (GDA). The GDA is isolated circumferentially and all lymphovascular tissue, including that between the proper hepatic artery, bile duct and portal vein, is cleared for at least 1 to 2 cm in each direction from the origin of the GDA to avoid common sources of extra-hepatic perfusion (EHP).29 This includes dissection proximally on the common hepatic artery, and distally along the proper, right and/or left hepatic arteries. The GDA is dissected distally to the superior border of the pancreas, thereby maximizing GDA length and disrupting collaterals that could result in EHP to the duodenum and pancreatic head. Proximal arterial branches on the GDA, down to and often including the supraduodenal branch, are ligated with nonabsorbale ties with careful attention not to narrow the GDA. A partial Kocher maneuver of the first and second portions of the duodenum, including disruption of the lateral portocaval nodal chain and lymphatics, may further mitigate EHP, and facilitates evaluation of the dorsal aspect of the duodenal sweep and pancreatic head when assessing for EHP intraoperatively. The gastrohepatic ligament can also be divided up to the left hepatic vein to reduce the risk of extrahepatic or competitive perfusion from an accessory or replaced left hepatic artery. When the gallbladder is present, a cholecystectomy is performed to avoid chemical cholecystitis. Papaverine is then applied to the GDA to optimize vasodilation while the pocket is fashioned.
At this point, a limited transverse incision that will accommodate the device is created overlying the planned subcutaneous pump pocket, typically in the left hemiabdomen. Left abdominal placement limits artifact from the pump upon the liver on future imaging studies, and is clear of a future right-sided Kocher incision in patients whom future hepatectomy is possible. However, laterality of pump placement is not a major consideration, and the device can be adjusted based on prior surgical incisions, other concurrently implanted devices, the presence or requirement for an ostomy, or complication necessitating pump relocation as noted below. Incision for the pocket should be placed such that the pump avoids bony prominences, typically midway between the costal margin and the anterior superior iliac spine. The incision should also overlie the upper third of the device to enable reservoir access while avoiding having to traverse the incision with the needle (access inferior to the incision), and the device should fit snugly when inserted to prevent seroma formation (Fig. 1A). The catheter is passed intraperitoneally so the pump can be secured flush on the fascia with the majority of the catheter protected behind the device. In this orientation, the reservoir port will lie just inferior to the pump pocket incision and the catheter access port, if using a dual-access device, is positioned laterally to allow easy access for pump perfusion studies.
FIGURE 1.

Hepatic artery infusion (HAI) pump placement in normal anatomy and common variants. A, HAI pump placement with conventional arterial anatomy. B, Replaced left hepatic artery (originating from the left gastric) in situ and following HAI pump placement (C). D, Replaced right hepatic artery (originating from the SMA) in situ and following HAI pump placement after ligation of the aberrant artery lateral to the common bile duct (E). F, Intraoperative photos of uncommon arterial variant: common hepatic artery (long white arrow) branches into the right (asterisk) and left (triangle) hepatic arteries and the GDA (short white arrow) emanates from the right hepatic artery. An accessory right hepatic artery (looped) is also present. G, Following ligation and division of the accessory right hepatic artery and native left hepatic artery (clipped), the GDA (short white arrow) is cannulated which facilitated single vessel in-line flow to the liver via the right hepatic artery (asterisk).
Once the pump is placed within the pocket, the catheter is positioned for insertion in the GDA. After ligation of the GDA distally, proximal control is obtained at the origin of the GDA with a vascular clamp (alternatively may apply separate clamps proximally and distally on the common and proper hepatic arteries). A transverse arteriotomy is made as far distal on the GDA as can be accommodated with care not to disrupt the intima to avoid arterial dissection. The beaded tip catheter, already prepared with a bevel immediately distal to the terminal bead, is inserted with the assistance of a vein pick such that the tip of the catheter terminates at the junction of the GDA and hepatic artery and at least 1 bead is completely within the vessel. It should be noted that the catheter tip should neither terminate within the hepatic artery lumen, as it may impede flow or result in thrombosis, nor should it terminate too far from the GDA ostium, which could result in pooling of chemotherapy and resultant arteritis. Once the catheter is in place and secured with the first 2–0 non-absorbable tie proximal to the terminal bead, the GDA is unclamped and the pump is flushed with heparinized saline (100U/mL) through the bolus port (Fondaparinux can be used in cases of heparin allergy). Additional nonabsorbable ties are placed as space allows, for added security, during which time the catheter may be flushed following each tie to ensure patency and to identify any suture which may occlude the catheter in real time. Next, 2 to 3 mL of undiluted blue dye (most commonly, but alternative agents can be used as described below) is slowly flushed to confirm bilobar hepatic perfusion and to rule out EHP. The following text will describe potential pitfalls and proposed solutions for common intraoperative challenges.
ABERRANT OR UNFAVORABLE ANATOMY
Intraoperative challenges can arise due to aberrant anatomy, small native vessel caliber, or vascular disease. Challenges can also arise due to patient body habitus or prior surgical procedures precluding pump placement in the left abdomen. Each of these scenarios requires careful consideration; however, they can be overcome in the majority of cases.
Aberrant Arterial Anatomy
Several variant celiac and superior mesenteric vascular patterns are documented.12,30–32 In the seminal work, Daly et al characterized the relative frequency of many of these variants and described the implications to catheter placement in the GDA for each.12 The most common arterial variants involve a replaced/accessory left hepatic artery arising from the celiac or left gastric arteries (15%–20%) or a replaced/accessory right hepatic artery arising from the superior mesenteric artery (SMA, 3%–8%) (Fig. 1B–E). As long as it of suitable caliber and length and has inline flow to the liver, the GDA should be utilized for catheter canalization because catheter placement in vessels other than the GDA are associated with higher complication and EHP rates.33 Replaced or accessory arteries, when present, are routinely ligated to remove competitive intrahepatic flow that otherwise results in preferential flow of chemotherapy to only 1 hemiliver. Perfusion following ligation of a replaced or accessory hepatic artery relies on intraparenchymal and hilar cross-perfusion between left and right hepatic arterial systems and hepatic ischemia is extremely rare. It is prudent to consider test clamping the aberrant vessel before ligation to ensure cross-perfusion and lack of ischemia, the latter being a rare event. Nevertheless, the consequence of ligating a replaced vessel must be considered, for example, the potential ramifications of relying on that lobe as the future liver remnant.
Other common variants include variable takeoff of the left and right hepatic artery in relation to the GDA, including GDA emanating from the right hepatic artery (Fig. 1F–G), the GDA branching off the left hepatic artery, trifurcation of these three vessels from the common hepatic, or even lack of communication of the GDA to either hepatic artery.32 Each of these variants may require ligation of one of the native hepatic arteries, such that all remaining hepatic arterial flow is in line with the GDA (and HAI delivery) to ensure equal bilobar perfusion.
Unsuitable Target Vessel
The most common catheter (Intera silicone arterial beaded tip catheter, Intera Oncology Inc, Wellesley, MA, previously Codman) used for HAI has a 0.6 mm inner diameter and 2.3 mm outer diameter, and thus requires a conduit vessel of at least 2 to 3 mm. In some instances, a diminutive GDA is encountered which cannot accommodate the catheter tip. Alternatively, intraoperative pitfalls may include vasospasm, insufficient GDA length, or GDA/hepatic artery dissection, each of which presents a unique challenge in management. Vasospasm often results in cases of reoperation or otherwise difficult dissection and may preclude passage of the arterial catheter due to diminutive size. When encountering a GDA of small size, it must be determined whether the vessel is simply in spasm or whether it is truly diminutive and therefore unsuitable for cannulation. It is common practice to apply a smooth muscle relaxant such as papaverine directly to the vessel to facilitate successful passage of the catheter routinely. However, if the vessel is still too small, alternate cannulation sites have to be entertained (see below).
In cases wherein the GDA length is short (<1cm), insertion of the catheter beyond the first bead may not be possible. If the catheter cannot be inserted enough to accommodate the first bead without preventing in line flow from the common hepatic artery (catheter tip sits within the lumen of the common hepatic artery), the vessel should not be used.
Arterial Dissection or Injury
Although meticulous catheter insertion, including visualization of the arterial wall following arteriotomy and careful manipulation with the vein pick decreases the incidence, arterial injury, including dissection, may occur during cannulation. Intraoperatively, arterial dissection may be identified by non-perfusion, incomplete hepatic perfusion (described below) or blue dye accumulation within the wall of the hepatic artery. Alternatively, delayed or preferential perfusion to the right or left hemiliver may be present when blue dye is flushed due to propagation of the false lumen into the hepatic circulation. If dissection is suspected intraoperatively, the catheter can be removed and the GDA flashed to confirm blood return from the true lumen and allow for inspection for an intimal flap. If the vessel returns blood and no flap is seen, a second attempt at cannulation can be attempted and the proper position confirmed with blue dye, fluorescein, or indocyanine green (ICG) infusion. Catheter placement should only continue after confirmation that the tip is inserted in the true lumen and there is no ongoing dissection. In some instances, dissection may spontaneously resolve and recanalize with time, so it is acceptable to leave the catheter in place and evaluate postoperatively with flow scans. However, if target artery cannulation is not successful due to dissection, consideration should be given to attempt cannulation of an alternative conduit vessel (see below) or to abort the procedure. Despite these heroic measures, in the case of arterial dissection, the pump may not be salvageable.
Alternate Cannulation Strategies
In the circumstances described above when the GDA is not suitable for cannulation, alternate strategies for cannulation can be undertaken. Assuming otherwise standard hepatic arterial anatomy, one option is retrograde cannulation with ligation of the left or, less commonly, right hepatic artery, the choice of which may be dictated by the future liver remnant, which also takes advantage of the intrahepatic bilobar collaterals. This method best recapitulates the standard approach of GDA cannulation; however, it requires significant hilar dissection to isolate an adequate segment of left (or right) hepatic artery, which may be diminutive itself. Additionally, there is decreased ability to stabilize the catheter and as a result left or right hepatic artery cannulation may be at higher risk to kink or dislodge. Use of a venous conduit to the common hepatic artery (saphenous or right gonadal vein) to accommodate the HAI catheter has also been described and is the preferred bailout strategy for several authors.34,35 Another alternative is to cannulate the splenic artery proximally and direct in line flow to the common hepatic artery at its origin from the celiac axis. The primary issue with this method is the rich collateral flow from this proximal infusion point. Splenic artery cannulation thus requires ligation of the GDA and commonly, the left and right gastric arteries to prevent EHP. Ligation of celiac axis collaterals leaves the stomach at higher risk of ischemia given the reliance of collateral back-flow through the gastroepiploic and to a lesser extent the short gastric arteries.
Occasionally, as a result of celiac stenosis, the majority of arterial flow to the liver is retrograde from SMA collaterals via the GDA, which may be indicated by a large caliber GDA and collaterals throughout the head of the pancreas. If celiac stenosis is suspected based on preoperative CT angiography, interventional angiography or lysis of a pathologic arcuate ligament can be attempted to correct the stenosis versus planned alternate artery cannulation. Intraoperatively, the surgeon should routinely test-clamp the GDA before ligation and catheter insertion to ensure pulsatile flow in the hepatic arteries. If hepatic arterial perfusion relies upon retrograde flow through the GDA (and thus the proper hepatic arterial pulse diminishes when the GDA is clamped), it cannot be cannulated as there will not be adequate inline flow. Instead, the catheter may be inserted into the common hepatic artery after ligation proximally. Despite technical feasibility, there is an increased risk of perioperative and pump related complications when anatomic variants require cannulation of a vessel other than the GDA.33 In these cases, it is critical to understand the goals of HAI therapy for the particular patient to best assess the risks and beneflts of alternative access before pump placement.
Unfavorable Patient Characteristics
Critical steps to pump pocket creation and device implantation include establishing a subcutaneous pocket that will accommodate the pump with a snug fit, anchoring the device to the anterior abdominal wall fascia to prevent rotation, revolution, or migration of the pump, unimpeded transfascial catheter insertion, and ensuring the tissue overlying the pump permits palpation of pump access and reservoir ports for ongoing use. Several patient characteristics, including obesity, previous surgical incisions or mesh, or the presence of an ostomy preceding or concurrent with pump placement, may complicate pump insertion. However, each can be overcome with minimal change in technique.
Obesity is a common comorbidity, even among patients with advanced cancers. In many obese patients, the subcutaneous space is too thick to successfully place the device without risk of pump displacement/flipping and difficulty accessing the reservoir. In this case, placement may be performed in an area with less subcutaneous tissue such as the anterior chest wall, in which the catheter is tunneled for insertion into the peritoneum through the upper abdomen. Additionally, with the increased anterior-posterior distance of the abdominal wall in the obese patient, the pump runs the particular risk of seroma formation or flipping within the pocket if not properly anchored. To prevent each of these complications which would leave the pump inoperable, polypropylene mesh fixation can be used to both anchor and harness the pump. Finally, if pump placement into a deep subcutaneous space is necessary and future device access is anticipated to be challenging, fat debridement to include Scarpa’s fascia can be performed. Such debridement lessens the distance between the skin and device to ease pump access, however, may increase the risk of seroma formation.
INTRAOPERATIVE INCOMPLETE PERFUSION, NONPERFUSION, AND EXTRAHEPATIC PERFUSION
Following catheter placement, bilobar hepatic perfusion, and the absence of EHP, is confirmed directly with intra-operative injection of blue dye, most commonly (Fig. 2A). Alternative agents include ICG or fluorescein, which are visualized with fluorescent filters or a Woods lamp, respectively (Fig. 2B). The surgeon must be prepared with strategies to troubleshoot each of the various issues with perfusion to ensure optimal pump function before leaving the operating room.
FIGURE 2.

Introperative and postoperative perfusion studies. A, Intraoperative blue dye and (B) indocyanine green (ICG) injection confirming normal bilobar perfusion. C, Incomplete perfusion with blue dye only perfusing left hemiliver due to competitive flow from accessory right hepatic artery (yellow vessel loop). Note demarcation along Cantlie’s line. Bilobar flow via proper hepatic artery in-line from HAI pump was achieved after ligation of accessory vessel (not shown). D, Extrahepatic perfusion (EHP) to duodenal bulb (black arrow), which is resolved following further dissection along GDA and periportal lymphatics, confirmed with ICG (E). F, Planar images from a normal postoperative perfusion study of the anterior (left) and posterior (right) abdomen with perfusion confined to the liver. Note heterogeneous perfusion in the liver reflects known liver metastases. G, In contrast, EHP is identified with Technetium-99 uptake in duodenal bulb (white arrows). H, This was further confirmed on axial SPECT/CT to reflect EHP to the pancreatic uncinate and duodenum (white arrows). I, Confirmation of EHP by celiac arteriography demonstrates an anomalous origin of the posterior superior pancreaticoduodenal artery (white arrows) from an accessory segment 6 artery that arises from the distal proper hepatic artery as the source of nontarget perfusion. J, After coil embolization of the pancreaticoduodenal and accessory segment 6 artery (white arrow), there is resolution of nontarget artery opacification on angiography. Subsequent scintigraphy confirmed absence of radiotracer uptake in the pancreatic head and duodenum (inset). Note, HAI catheter remains in GDA and was functional following embolization.
Incomplete Perfusion
Incomplete perfusion appears as lobar or segmental perfusion and typically indicates competing flow through the hepatic circulation (Fig. 2C). This occurs most commonly from ongoing perfusion from aberrant vascular anatomy, typically via replaced or accessory hepatic arteries that were not identified preoperatively, or from aberrant insertions of the GDA. To avoid competitive perfusion, we recommend high-quality pre-operative CT angiography, which will identify nearly all physiologically significant arterial anomalies. Intraoperatively, the authors recommend complete dissection of the gastrohepatic ligament to divide accessory or replaced left hepatic perfusion, and lateral portal dissection to identify missed accessory or replaced right hepatic perfusion. Incomplete perfusion due to competitive flow can thus be resolved with ligation of these aberrant vessels. If extensive dissection is performed, but bilateral perfusion cannot be achieved, arterial occlusion due to advancement of the catheter into the left or right hepatic artery, arterial dissection, ligation or injury should be investigated and treated as described above. If complete hepatic perfusion cannot be achieved in the operating room, and persists on postoperative nuclear medicine evaluation (interlobar perfusion may become more robust over time), then postoperative angiography should be performed for further evaluation of potential sites of competitive perfusion.
Nonperfusion
Nonperfusion indicates that no dye is visible in the liver parenchyma following injection through the pump bolus port. Most commonly, nonperfusion may occur due to over-diluted dye or insufficient concentration of dye in the hepatic circulation caused by high flow rate through the hepatic artery. It is our practice to utilize undiluted blue dye to avoid this type of event and to improve visualization. If color change is still not observed and no occlusion is present, alternate fluoroscopic dye selection should be attempted as ICG may have higher sensitivity than blue dye. Additionally, the surgeon should inspect the pump pocket to ensure that dye is not leaking out of the catheter system or that the pocket was not inadvertently injected due to accidental withdrawal of the needle from the septum.
Contrarily, nonperfusion may be caused by proximal occlusion or arterial dissection. First, one must confirm there is still a pulse in the proper hepatic artery, the absence of which may indicate dissection (see above) or thrombosis. If there is a strong pulse, ensure that the catheter flushes easily. It may be necessary to use a smaller volume, 3cc or 5cc, syringe to flush given the occasional high resistance in the catheter, though this should be done with care to avoid disruption of the catheter from the pump or arterial lumen. If it is still difficult to flush, the ties securing the catheter to the cannulated vessel should be checked to ensure they are not occlusive, and that one of these ties is not proximal to the first catheter bead, inadvertently ligating the GDA. This can also be avoided during placement by heparinized saline injection after each tie to confirm each is not too tight. In the rare instance that nonperfusion persists, postoperative pump angiography can be utilized to determine whether the catheter is patent and adequate hepatic flow via the true arterial lumen is retained. Transarterial hepatic angiography or CTA may also be utilized to identify hepatic artery dissection, thrombosis, or other less common etiologies for nonperfusion.
Extrahepatic Perfusion
Finally, EHP appears as blue dye uptake in a nontarget location, most commonly in the stomach, duodenal sweep, or pancreas, but it can appear in any abdominal organ (Fig. 2D).29 EHP most commonly results from incomplete dissection of small unnamed periportal vessels or lymphatics (often visible containing blue dye). When present and visible, further dissection should be carried out to divide the culprit vessel and/or lymphovascular tissue supplying the distribution of EHP observed, whereas balancing the risk of EHP with the risk of further dis-section, including duodenal injury or biliary injury or devascularization. Once more extensive dissection is completed, alternate dye (eg, ICG) can be utilized to confirm isolated hepatic perfusion if staining from the blue dye persists (Fig. 2E). Postoperative nuclear medicine confirmation of bilobar hepatic perfusion without EHP (as well as lung shunt evaluation depending on institutional protocols) should be performed in all patients, regardless of the findings on the intraoperative evaluation.
POSTOPERATIVE PROCEDURES AND HAI INITIATION
After successful HAI pump placement, a nuclear medicine perfusion study using planar scintigraphy, or more commonly, single-photon emission computed tomography (SPECT)/CT, is performed before initiation of pump therapy in all patients to confirm isolated hepatic perfusion. Some institutions also evaluate for pulmonary shunting, as protocols are extrapolated from radioembolization planning. Pump SPECT/CT represents an opportunity to identify EHP with higher sensitivity than pre-operative or intraoperative modalities.36,37 SPECT/CT may be obtained before discharge from the index hospitalization or as an outpatient, depending on institutional practices, work-flow, and patient factors (eg, pump pocket swelling), but must be completed before initiation of FUDR. The study is conducted in the Nuclear Medicine suite under sterile conditions as previously described.38 In a patient with favorable body habitus, pump access can be accomplished by direct palpation. In obese patients or those with thicker abdominal wall, palpation of the device may be difficult, necessitating access under fluoroscopy in the interventional radiology suite. This has become standard practice at several of the author’s institutions for all or in selected patients (eg, BMI > 30). Once the access/bolus port is cannulated, the port is flushed with heparinized saline to ensure patency followed by 5 mCi Technitium-99 macroaggregated albumin for acquisition of SPECT/CT images (Fig. 2F). In the event there is difficulty flushing the catheter despite successful cannulation, a smaller 3cc or 5cc syringe may again be used.
The postoperative, pre-treatment SPECT/CT is primarily used to rule out EHP and determine the lung shunt fraction, both of which may dictate need for additional procedures before pump chemotherapy, impact timing of HAI initiation, and/or require pump chemotherapy dose modifications. Normal perfusion studies show radiotracer uptake only in the liver. It is not uncommon to see moderate pertechnetate pooling at the catheter tip or diffuse, mild uptake in the gastric mucosa (which may coincide with symmetric renal parenchyma and/or splenic uptake) due to variable time delay between Technitium-99 injection and image acquisition. However, focal radiotracer uptake, most commonly in the stomach, duodenum, or pancreas, indicates EHP and should be confirmed with pump and/or trans-arterial angiography, followed by embolization of extrahepatic feeding vessels to avoid nontarget organ toxicity to pump chemotherapy (Fig. 2G–J).29 Effective endovascular embolization of off target vessels can be difficult to remedy, however, as the aberrant vessels are often small, thus posing a challenge to selective embolization. In cases where EHP cannot be disrupted despite best efforts, repeat SPECT/CT may be obtained to determine if the EHP from small, unnamed vessles resolved spontaneously. Infrequently, it may otherwise be necessary to return to the operating room to surgically ligate the associated vessel after weighing the risks of delaying therapy. Pump therapy can continue with extreme caution, typically initiated at a reduced dose, with close attention to potential side effects including gastritis, pancreatitis, or gastrointestinal bleeding. In contrast, incomplete perfusion on SPECT/CT is uncommon as this study has higher sensitivity than intraoperative blue dye or ICG assessment.36’37 Unless there is associated EHP, or significant lung shunting as described below, therapy should proceed at the normal dose.
The lung shunt fraction, if determined (institution dependent), is a surrogate for the amount of chemotherapy entering systemic circulation.39 A shunt fraction threshold below 10% is used to safely dose pump chemotherapy without dose reduction. Values between 10% to 20% do not preclude patients from pump therapy, however, FUDR dose reductions of 25% to 50% are typically employed for initial doses to ensure tolerance of therapy. Patients with a lung shunt fraction exceeding 20% should be considered for initiation of pump chemotherapy on an individual basis given the concern for chemotherapy induced pneumonitis and systemic toxicity.16’38
HAI-ASSOCIATED COMPLICATIONS
Complications related to external pump placement and pump therapy were initially reported in up to 35% to 80%, but have improved in the modern era and with subcutaneous pump implantation.40’41 At high-volume centers, HAI pump placement is performed with low mortality (<1%) and acceptable morbidity. In addition, 2 recent reports demonstrated that the morbidity, which can prolong recovery and prevent initiation of therapy, is primarily driven by synchronous procedures (eg, minor/major hepatectomy, colorectal resection), not by the HAI pump placement.22’42 HAI-related complications can be classified as either pump/pocket-related, catheter-related, vascular, or biliary, and may occur early in the postoperative course or following initiation of pump therapy. More recent series from high-volume centers report pump specific complications in 22% to 28% of patients, however up to 45% are salvageable with early and aggressive intervention.20’33’43 Therefore, it is critical for any practitioner treating patients with HAI to be able to rapidly recognize, evaluate, and treat these complications.
Pump and Pocket Complications
HAI therapy relies on the subcutaneous implantation of the pump, mostly commonly within the anterior abdominal wall. Many pitfalls are present which can lead to difficult access, contamination, and nonfunction. The postoperative SPECT/CT and subsequent initiation of therapy requires adequate recovery from pump placement. Pump pocket complications, including seroma, hematoma, wound infection or abscess occur in 8% to 18% of patients following pump placement in contemporary series (Fig. 3A–F).4,18–20,33 These are typically diagnosed on physical examination, including findings of erythema (Fig. 3A), tenderness, fullness, or fluctuance around the pump site, or drainage from the pump pocket incision, all of which can occur early in the postoperative course or in a delayed fashion. Ultrasound or CT imaging can aid in diagnosis if physical examination findings are inconclusive (Fig. 3B). When a small seroma in the absence of any signs of infection is encountered, the patient may be managed expectantly. If significant fluid is encountered, there may be undue pressure on the pocket incision or act as a nidus for infection. In this scenario, the pocket should be aspirated completely under sterile conditions, either at the bedside or under ultrasound guidance, to evacuate the space, with careful attention not to injure the pump catheter, which should be positioned mostly posteriorly to the pump. Cultures of the aspirate should be sent to direct therapy if there is a concern for associated infection. Aspiration can be done repeatedly if the contents are sterile, but reaccumulate, and do not preclude regular filling of the pump with either saline or FUDR. A low threshold for antibiotics covering skin flora and broad coverage of gram negative and gram positive organisms should be started if erythema or warmth is found on examination, or if systemic signs of infection (including leukocytosis) are present. Pump salvage is achieved when the pocket remains dry following aspiration and local infection subsides. Pocket hematoma, if small and nonexpanding, can also be managed expectantly with compression using an abdominal binder. However, if the pocket is expanding rapidly, or there is obvious pressure or tension on the overlying skin or incision, reexploration should be performed to achieve hemostasis and evacuate the hematoma, an otherwise nidus for infection. If there are clear signs of pocket infection, relocation of a new device, either through laparoscopic or open approaches, may be considered, which requires disconnecting the catheter intraabdominally and splicing the catheter to the new pump using an extension or repair kit. Relocation can also be performed completely extraperitoneally by tunneling the catheter to the newly chosen pump site (Fig. 3C), but this runs the risk of ongoing infection and should be utilized sparingly. Alternatively, some pumps may be salvaged after pocket exploration, washout, irrigation with antimicrobial solution (ie, betadine) and a course of intravenous antibiotics; however, this maneuver typically only provides rescue for 1 to 2 months of HAI. If salvage is unachievable, pump removal or re-siting may be necessary.
FIGURE 3.

Pump-and pocket-related complications. A, Erythema and blistering over pump pocket which was warm on physical exam with (B) corresponding CT findings of pump pocket seroma, which resolved after aspiration of serous fluid and empiric antibiotics. C, Coronal maximum intensity projection following pump relocation to right abdomen using catheter extension tunneled through the subcutaneous tissue (white arrow). D, Pump erosion presenting at blister (left) and full thickness ulcer with exposed device (right). E, Fluoroscopy shows evidence of a flipped pump: note the orientation of the access port and catheter connector (black arrow) is superior-lateral rather than inferior indicating the opening faces inward and accounts for the inability to access the pump. In addition, the catheter has disconnected from the intraabdominal portion and only a short length remains attached to the pump (white arrow), whereas the intraabdominal portion terminates inferiorly (out of view). F, Catheter migration through pump pocket incision following pump removal. This was managed by ligation and trimming of catheter at the level of the fascia and closure of the anterior rectus sheath defect.
Additionally, when the pocket is not created snugly, the device is loosely secured to the fascia, and/or after prolonged usage of the pump, migration may arise within the pocket. Erosion, ulceration, blistering or displacement of the pump on physical examination should lead to concern for pump migration (Fig. 3D). In the case of pump erosion or ulceration through the dermis, one should consider the pump contaminated and it should be removed from the original pocket and re-sited. If ulceration is noted before pump exposure and/or the patient still requires pump therapy, the pump may be re-sited under sterile operative conditions as described above. The pump is at higher risk for infectious complication at this point and should be removed if recurrent ulcer, infection, or abscess is encountered.
Further, the device can rotate or flip within the pocket pump (Fig. 3E), which is most commonly confirmed on plain films or fluoroscopically after failed attempts to cannulate the reservoir or bolus ports. In the case of rotated or flipped pump, where the integrity of the overlying dermis is not in question, re-siting in the same pocket, with or without the use of a mesh, may be possible. Alternatively, as a temporizing measure, the pump can be manually flipped at the bedside by an experienced clinician, although this should be avoided repeatedly as it may lead to catheter kinking or dislodgement. If these maneuvers are successful, definitive re-siting (with or without anchoring mesh), either in the established pocket or relocation to a new pocket (eg, chest wall) may be necessary.
Complications related to malfunction or nonfunction of the device are also seen, albeit in rare instances (<5%).33 Unfortunately, any interruption of infusion through the pump catheter may lead to thrombosis/occlusion resulting in pump nonfunction. However, if the catheter flushes, salvage can be attempted. A malfunctioned pump may be removed and replaced in the subcutaneous pocket and therapy re-attempted/re-initiated. If therapy is still not possible following device replacement, therapy will likely need to be aborted.
Catheter-Related Complications
Complications assocated with the catheter were commonly encountered in early series involving pump placement via Seldinger technique via peripheral vessels.40 The incidence of catheter-related complications has decreased significantly with advancement in implantable devices, however, the insertion of a foreign material in the abdominal cavity and vascular system is fraught with challenges. Catheter related complications can be summarized as occlusive or migratory in nature and occur in up to 10% to 26% of patients.33
Occlusive complications including catheter thrombosis or mechanical occlusion should be considered if difficulty flushing the catheter at the time of SPECT/CT or unexpectedly high residual reservoir volume at the time of pump refill is encountered. Occlusion due to any cause can be ruled out by fluoroscopic pump injection to confirm the proper orientation of the pump and catheter and free flow of contrast into the hepatic arteries. If occlusion is due to kinking of the catheter or malpostioning of the pump is found, the pump should be repositioned or re-sited as described above such that the catheter is freed of its obstruction. If catheter or arterial thrombosis is suspected, CT angiography in combination with pump angiography discern between the two etiologies of occlusion. If thrombosis is confined to the catheter itself, treatment depends on the timing of this complication. Early in the postoperative course, gentle heparin flush can be attempted and may clear the tube of freshly formed clot. Late thrombosis, however, may be due to extensive fibrin sheath that forms within the catheter lumen, for which injection of 1 mg tissue plasminogen activator may be considered in the interventional radiology suite. If thrombosis extends into the hepatic artery, catheter directed therapy should not be attempted, as this can result in pseudoaneurysm formation and catastrophic bleeding. Rather, the patient should undergo systemic anticoagulation with concurrent heparinized saline in the pump until clot resolution. Unfortunately, given the small caliber of the pump catheter, prolonged pause of forward flow typically results in unrecoverable catheter thrombosis.
Catheter migration or dislodgement can also occur in 5% to 18% of patients and have variable severity depending on the timing and location in which migration occurs.11,33 Distally, catheter dislodgement from the GDA may occur gradually over a prolonged period, most commonly, or can occur acutely in conjunction with pseudoaneurysm (see vascular complications). When the catheter migrates gradually, bleeding is uncommon as the GDA slowly thromboses and/or a well-developed tract is formed and likely fills with thrombus or fibrosis. As clinically significant bleeding is uncommon and the catheter is free to flow into the peritoneal cavity, patients are often asymptomatic, and catheter migration and pseudoaneurysm are incidentally detected on surveillance or restaging CT (Fig. 4A–C). Imaging findings include separation of the catheter tip from the GDA, with or without perivascular fluid collection near the catheter tip or pseudoaneurysm, which may be related to extraluminal infusion of pump therapy or contained hemorrhage, respectively. When identified, pump therapy should be permanently dis-continued. In the absence of symptoms, no further intervention is required, however pump removal may be considered.
FIGURE 4.

Catheter-related complications. (A–C) Catheter disloged from GDA. A, Axial CT of a patient with fluid incidentally noted adjacent to GDA (white arrow) and pump catheter tubing that dislodged from the GDA insertion. B, 3D-rendering of same patient shows the catheter tip (black arrow) paralleling the course of the common hepatic artery (white arrow), and is no longer sited within the vessel. Optimal positioning of the catheter will mimic the position of the GDA and is commonly more perpen-dicular to the common hepatic artery. C, Contrast injection through the pump demonstrates opacification from the pump catheter tip (black arrow) into an amorphous collection (white arrows) that was confirmed to be extraluminal on cone-beam CT scan (not shown). D, Coronal maximum intensity projection in a patient with an HAI pump in the left abdomen. The catheter is correctly positioned distally at the hepatic hilum (white arrow), whereas the tubing is inappropriately disconnected proximally, residing freely in the pelvis (long black arrow). The tubing disconnected close to the pump; a short segment of tubing is seen exiting the pump proximal to the tubing fracture (short black arrow). E, CT image of a catheter which appears intraluminal (white arrow) at the gastrojejunostomy of a patient with a prior gastric bypass. F, Intraluminal catheter erosion (white arrow) was confirmed on upper endoscopy adjacent to an Axios stent placed for access to the gastric remnant, which likely created friction leading to catheter erosion. Jejunal ulcer healed following catheter removal and laparoscopic repair.
Proximal dislodgement can also occur, particularly in patients who have implanted pumps that require splicing of two catheters together, as is the case with the Medtronic device, or in scenarios where the catheter needs to be repaired or pump relocated. The catheters may become separated at the level of the metal connector, resulting in a free floating intraperitoneal catheter, or sometimes within the pocket (especially in the setting of a flipped pump, see Fig. 3E), with the distal end still within the arterial insertion site. Diagnosis is again typically incidental on surveillance or restaging scans (Fig. 4D). Fortunately, due to the small internal caliber of the catheter, this rarely results in intra-peritoneal hemorrhage as the catheter often thromboses, and surgeons and radiologists alike should assess for this phenomenon each time imaging is obtained. Although this is typically an unsalvageable complication, simple techniques may decrease its incidence. These include placement of multiple permanent sutures securing each catheter on either side of the metal connector when splicing is necessary, and/or maintaining a long loop of catheter in the pump pocket to keep the spliced portion above the fascia thereby preventing friction to the spliced segment as the catheter traverses the abdominal wall fascia and musculature. When identified, removal of the pump should be considered.
Finally, in rare circumstances, the intra-abdominal portion of the catheter can also erode into abdominal viscera, either into the duodenum near the catheter insertion site, or at a remote site (Figure 4E–F). This may be detected incidentally on upper endoscopy, or patients may present with abdominal pain, gastrointestinal bleeding, systemic infection, or even free perforation. Axial imaging, often obtained to evaluate the presenting symptoms, can facilitate diagnosis of catheter erosion. Pump therapy should be stopped immediately if chemotherapy is infusing, and antibiotics promptly initiated. Full thickness erosion into abdominal viscera can be confirmed by endoscopy and catheter removal and bowel repair/resection should be considered.
Vascular Complications
Complications related to cannulation of the GDA or alternate arteries can occur intraoperatively, as described above, or early or late in the postoperative course. Vascular complications occur in less than 10% of HAI patients and include arterial dissection, thrombosis, or pseudoaneurysm. If not identified at the time of pump placement, arterial injury in the form of dis-section or thrombosis, though most commonly asymptomatic, may present with symptoms or signs of hepatic ischemia or injury, including RUQ pain, transaminitis, hyperbilirubinemia, or alkaline phosphatemia. CT angiography is typically the first imaging modality obtained to evaluate for these complications (Fig. 5A), and if nondiagnostic, catheterbased arteriography should be considered (Fig. 5B and C). Dissection does impede delivery of pump chemotherapy, and attempts to fenestrate the dissection flap such that the true and false lumens communicate can be attempted, but are rarely successful. As a result, this should only be performed after a complete risk assessment, as such heroic salvage attempt may result in complete occlusion or thrombosis of the hepatic artery.
FIGURE 5.

Vascular complications. (A–C) Arterial dissection. A, Axial CT angiography of GDA dissection of the common hepatic artery which has propagated beyond the bifurcation of the right and left hepatic arteries. B, In the same patient, contrast injection through the pump demonstrates opacification from the pump catheter tip (black arrow) into the false lumen of an extensive dissection (white arrow) that extends from the common hepatic artery into the right hepatic artery, with opacification of right hepatic artery branches (black asterisk) via distal right hepatic artery fenestrations into the true lumen, but does not communicate with the left hepatic artery (nonopacified). C, Celiac arteriogram confirms a narrow common hepatic artery (true lumen, black arrow) with filling only of the left hepatic artery (white arrow). (D–F) Pseudoaneurysm. D, Abdominal CT shows dense layered fluid (white arrows) surrounding the catheter insertion into the GDA, concerning for pseudoaneurysm. E, Celiac arteriogram of the same patient demonstrates active filling of a pseudoaneurysm (white arrow) originating at the site of the pump catheter tip (black arrow). F, Rescue was achieved by deployment of a self-expanding covered stent graft positioned to traverse the gastroduodenal artery origin (margins denoted by bracket), to prevent active filling of the pseudoaneurysm. Note the residual contrast within the now thrombosed pseudoaneurysm (white arrow).
Pseudoaneurysm, often related to erosion of the catheter through the GDA, occurs in fewer than 5% of patients, is often asymptomatic and is incidentally identified on re-staging scans, or patients may present with acute abdominal pain, acute anemia, hypotension, and in rare circumstances, shock. Given the potential for serious morbidity associated with an unidentified and untreated pseudoaneurysm, a low threshold of suspicion should be maintained until this complication is ruled out. Physical examination is critical, as is blood work, including CBC and metabolic panels. In a hemodynamically stable patient, CT angiography can be obtained, which may show pseudoaneurysm, fluid near the catheter and GDA, often with a layering hematocrit level or perhaps active hemorrhage (Fig. 5D). Alternatively, in hemodynamically unstable patients, interventional radiology should be urgently consulted for diagnostic and therapeutic angiography (Fig. 5E). Intervention directed at the vascular injury, including coil embolization, but most commonly placement of a covered metal stent in the common hepatic artery crossing the origin of the GDA is required (Fig. 5F). With this maneuver, patient salvage is prioritized at the expense of a functional HAI pump, which is sacrificed.
Biliary Complications
Although HAI with FUDR relies upon high first-pass metabolism in the liver, delivery through the hepatic arterial system places the biliary tree at risk for toxicity, the most feared being irreversible biliary sclerosis (Fig. 6A–G). Biliary toxicity most commonly is detected on routine protocolized bloodwork during each HAI cycle. Elevation of the transaminases, bilirubin, and alkaline phosphatase are early indicators of biliary toxicity and occurs in approximately 26% of patients.16,33 Close attention to liver function tests, which drive the need for dose reductions or holds and/or elongated dosing intervals, combined with concurrent delivery of dexamethasone with FUDR, and timely termination of HAI, minimizes the risk of biliary sclerosis requiring intervention, but the incidence remains 2% to 8% in the modern era and is highest when HAI is used adjuvantly following hepatic resection.2,21,44 Bevacizumab, which previously was used in conjunction with FUDR therapy, is no longer utilized due to increased incidence of biliary toxicity with concomitant use.2
FIGURE 6.
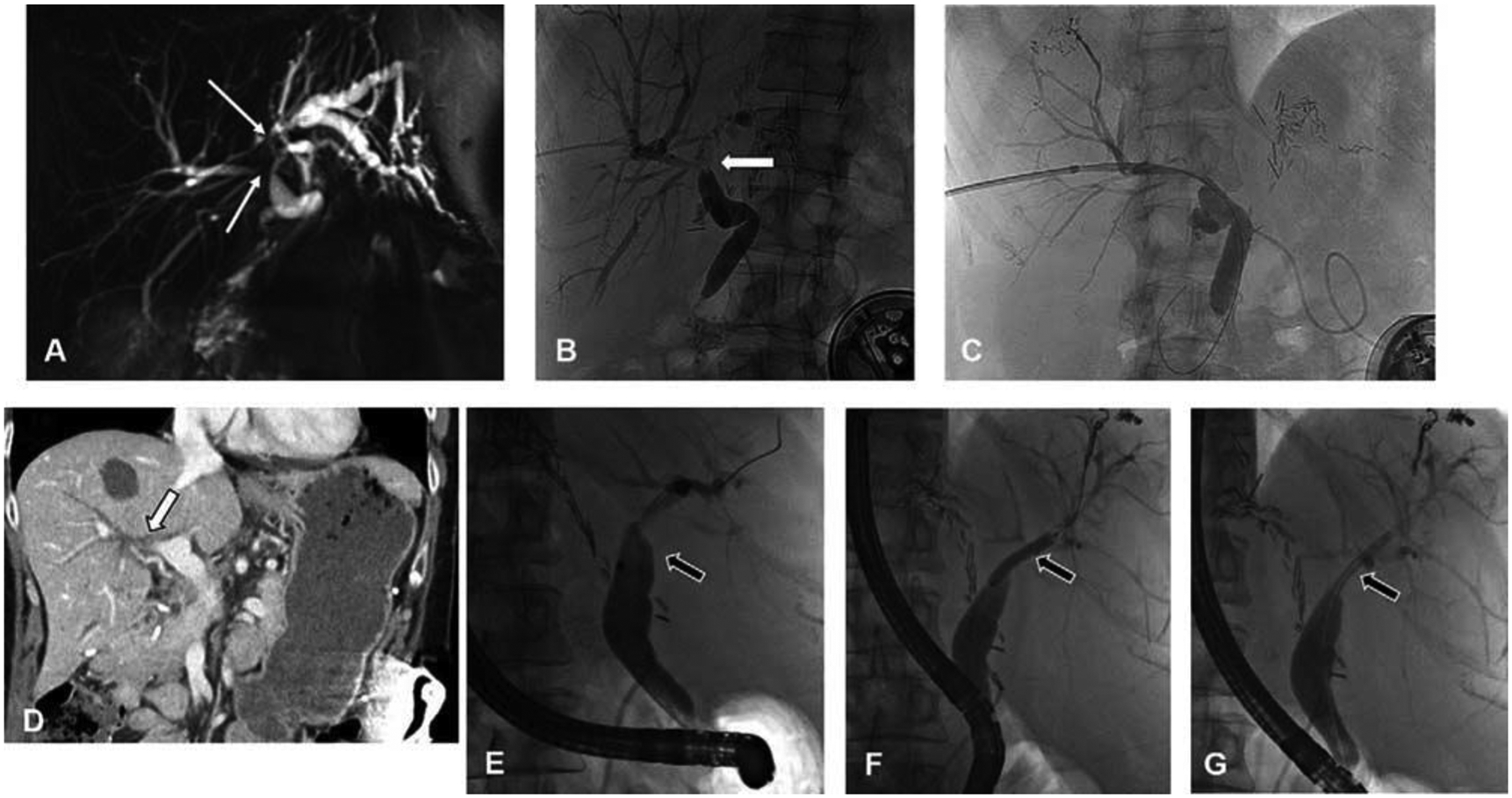
Biliary complications. A, Short segment right hepatic duct stricture and significant dilation of left hepatic ductal system on MRCP. B, Transhepatic cholangiogram demonstrates a central stricture involving the origins of both the right and left hepatic ducts (white arrow) with otherwise normal common bile duct and peripheral right ducts. A long segment stricture of the left hepatic duct is also present. C, The central stricture was managed with percutaneous biliary drainage. D, Long segment right hepatic duct stricture identified as biliary dilatation on CT (white arrow) obtained for hyperbilirubinemia. E, Stricture at origin of right hepatic duct (black arrow) was confirmed by endoscopic retrograde cholangiography and managed by (F) balloon dilation (black arrow) and (G) deployment of an indwelling plastic biliary stent (black arrow). Note, A–C from patient 1 and D–G from patient 2.
Although some patients may present with abdominal pain or cholangitis, most patients develop asymptomatic but persistent elevation in liver function tests despite the above maneuvers, and should be imaged with magnetic resonance cholangiopancreatography (MRCP). Cross-sectional imaging typically shows unifocal or multifocal common bile duct and/or hepatic duct strictures with intrahepatic ductal dilatation (Fig. 6A and D). Biliary sclerosis usually requires termination of HAI/FUDR, and in 2% to 4% of patients treated with HAI for unresectable disease and 5% to 6% of patients treated with adjuvant HAI, biliary stenting is required.2,3,44,45 Biliary decompression by dilation or stenting mitigates additional risk of cholangitis or hepatic failure and can be accomplished endoscopically or percutaneously (Fig. 6B and C, E–G). In very rare instances, biliary toxicity can progress to hepatic failure, a rare phenomenon with modern HAI.
PUMP REMOVAL
Although many patients will live with the HAI pump until they succumb to their disease, there are several indications for pump removal. Pumps may be removed after any complication that necessitates termination of HAI therapy, including but not limited to pump pocket infection, device malfunction, catheter dislodgement, pseudoaneurysm, and biliary sclerosis. In patients without complications, it is our practice to maintain pumps as functional for indefinite future use. The indication for pump therapy should also be considered. For either unresectable disease or adjuvant HAI following resection, pump chemotherapy can be reinitiated in the event of hepatic or liver dominant recurrence or progression, and therefore the pump should remain indefinitely. Alternatively, when used as adjuvant therapy, most progression will occur in the first 2 to 3 years following therapy24,45; accordingly, pump removal may be considered after achieving long-term hepatic progression-free survival or based on patient preference. However, it should be noted that once the pump is removed, implantation of a new pump and resumption of HAI is challenging, and requires cannulation of a secondary artery (ie, left or right hepatic artery).
Pump removal is performed as an outpatient procedure. The subcutaneous pocket is incised to expose the pump and free it from the fascial sutures. The catheter is tied off with permanent suture to prevent back bleeding through the GDA and ligated just above the level of the fascia. The catheter is then pushed below the fascia or secured to the fascia with a stitch to prevent migration through the incision (Fig. 3F). The capsule that forms around the pump may also be resected or fulgurated with cautery before closure to decrease the risk of seroma.
SUMMARY
Hepatic artery infusion offers a liver directed therapeutic approach to metastatic and primary hepatic tumors. A growing number of centers have implemented HAI programs, and recently the HAI Consortium Research Network was established to share experiences, standardize practice, evaluate HAI outcomes in a multicenter fashion, and conduct clinical trials. This HAI toolkit for commonly encountered intraoperative findings during surgical implantation and nuances of management of a variety of complications specific to HAI is a resource for new and veteran centers alike. With close follow-up, attention to detail, awareness of signs and symptoms specific to HAI complications, and aggressive work-up when potential problems arise, 95% of pumps are functional for HAI initiation and approximately 50% of complications that subsequently arise during therapy can be salvaged. The technical ability to implant HAI pumps in patients, including those with aberrant arterial anatomy, and overcoming clinical challenges and rescuing patients from HAI specific complications, when they arise, is key to successful implementation of new programs and expansion of HAI to patients who may benefit from such aggressive treatment strategy.
Footnotes
The authors report no conflict of interests.
REFERENCES
- 1.Kemeny NE, Niedzwiecki D, Hollis DR, et al. Hepatic arterial infusion versus systemic therapy for hepatic metastases from colorectal cancer: a randomized trial of efficacy, quality of life, and molecular markers (CALGB 9481). J Clin Oncol. 2006;24:1395–1403. [DOI] [PubMed] [Google Scholar]
- 2.D’Angelica MI, Correa-Gallego C, Paty PB, et al. Phase II trial of hepatic artery infusional and systemic chemotherapy for patients with unresectable hepatic metastases from colorectal cancer: conversion to resection and longterm outcomes. Ann Surg. 2015;261:353–360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pak LM, Kemeny NE, Capanu M, et al. Prospective phase II trial of combination hepatic artery infusion and systemic chemotherapy for unresectable colorectal liver metastases: long term results and curative potential. J Surg Oncol. 2018;117:634–643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dhir M, Jones HL, Shuai Y, et al. Hepatic arterial infusion in combination with modern systemic chemotherapy is associated with improved survival compared with modern systemic chemotherapy alone in patients with isolated unresectable colorectal liver metastases: a case-control study. Ann Surg Oncol. 2017;24:150–158. [DOI] [PubMed] [Google Scholar]
- 5.Jarnagin WR, Schwartz LH, Gultekin DH, et al. Regional chemotherapy for unresectable primary liver cancer: results of a phase II clinical trial and assessment of DCE-MRI as a biomarker of survival. Ann Oncol. 2009;20:1589–1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Anteby R, Kemeny NE, Kingham TP, et al. Getting chemotherapy directly to the liver: the historical evolution of hepatic artery chemotherapy. J Am Coll Surg. 2021;232:332–338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kemeny N, Cohen A, Bertino JR, et al. Continuous intrahepatic infusion of floxuridine and leucovorin through an implantable pump for the treatment of hepatic metastases from colorectal carcinoma. Cancer. 1990;65:2446–2450. [DOI] [PubMed] [Google Scholar]
- 8.Kemeny N, Cohen A, Seiter K, et al. Randomized trial of hepatic arterial floxuridine, mitomycin, and carmustine versus floxuridine alone in previously treated patients with liver metastases from colorectal cancer. J Clin Oncol. 1993;11:330–335. [DOI] [PubMed] [Google Scholar]
- 9.Kemeny N, Conti JA, Cohen A, et al. Phase II study of hepatic arterial floxuridine, leucovorin, and dexamethasone for unresectable liver metastases from colorectal carcinoma. J Clin Oncol. 1994;12:2288–2295. [DOI] [PubMed] [Google Scholar]
- 10.Kemeny N, Conti JA, Sigurdson E, et al. A pilot study of hepatic artery floxuridine combined with systemic 5-fluorouracil and leucovorin. A potential adjuvant program after resection of colorectal hepatic metastases. Cancer. 1993;71:1964–1971. [DOI] [PubMed] [Google Scholar]
- 11.Kemeny N, Huang Y, Cohen AM, et al. Hepatic arterial infusion of chemotherapy after resection of hepatic metastases from colorectal cancer. N Engl J Med. 1999;341:2039–2048. [DOI] [PubMed] [Google Scholar]
- 12.Daly JM, Kemeny N, Oderman P, et al. Long-term hepatic arterial infusion chemotherapy. Anatomic considerations, operative technique, and treatment morbidity. Arch Surg. 1984;119:936–941. [DOI] [PubMed] [Google Scholar]
- 13.Ackerman NB The blood supply of experimental liver metastases. IV. Changes in vascularity with increasing tumor growth. Surgery. 1974;75:589–596. [PubMed] [Google Scholar]
- 14.Breedis C, Young G. The blood supply of neoplasms in the liver. Am J Pathol. 1954;30:969–977. [PMC free article] [PubMed] [Google Scholar]
- 15.Dizon DS, Schwartz J, Kemeny N. Regional chemotherapy: a focus on hepatic artery infusion for colorectal cancer liver metastases. Surg Oncol Clin N Am. 2008;17:759–771. viii. [DOI] [PubMed] [Google Scholar]
- 16.Power DG, Kemeny NE The role of floxuridine in metastatic liver disease. Mol Cancer Ther. 2009;8:1015–1025. [DOI] [PubMed] [Google Scholar]
- 17.Sigurdson ER, Ridge JA, Kemeny N, et al. Tumor and liver drug uptake following hepatic artery and portal vein infusion. J Clin Oncol. 1987;5:1836–1840. [DOI] [PubMed] [Google Scholar]
- 18.Buisman FE, Grunhagen DJ, Homs MYV, et al. Adjuvant hepatic arterial infusion pump chemotherapy after resection of colorectal liver metastases: results of a safety and feasibility study in The Netherlands. Ann Surg Oncol. 2019;26:4599–4607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chakedis J, Beal EW, Sun S, et al. Implementation and early outcomes for a surgeon-directed hepatic arterial infusion pump program for colorectal liver metastases. J Surg Oncol. 2018;118:1065–1073. [DOI] [PubMed] [Google Scholar]
- 20.Creasy JM, Napier KJ, Reed SA, et al. Implementation of a hepatic artery infusion program: initial patient selection and perioperative outcomes of concurrent hepatic artery infusion and systemic chemotherapy for colorectal liver metastases. Ann Surg Oncol. 2020;27:5086–5095. [DOI] [PubMed] [Google Scholar]
- 21.Muaddi H, D’Angelica M, Wiseman JT, et al. Safety and feasibility of initiating a hepatic artery infusion pump chemotherapy program for unresectable colorectal liver metastases: amulticenter, retrospective cohort study. J Surg Oncol. 2021;123:252–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Brajcich BC, Bentrem DJ, Yang AD, et al. Short-term risk of performing concurrent procedures with hepatic artery infusion pump placement. Ann Surg Oncol. 2020;27:5098–5106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cercek A, Boerner T, Tan BR, et al. Assessment of hepatic arterial infusion of floxuridine in combination with systemic gemcitabine and oxaliplatin in patients with unresectable intrahepatic cholangiocarcinoma: a phase 2 clinical trial. JAMA Oncol. 2020;6:60–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Datta J, Narayan RR, Kemeny NE, et al. Role of hepatic artery infusion chemotherapy in treatment of initially unresectable colorectal liver metastases: a review. JAMA Surg. 2019;154:768–776. [DOI] [PubMed] [Google Scholar]
- 25.Dhir M, Magge D, Novak S, et al. Robotic-assisted placement of an hepatic artery infusion pump and catheter for regional chemotherapy of the liver. Ann Surg Oncol. 2016;23(suppl 5):755–756. [DOI] [PubMed] [Google Scholar]
- 26.Dhir M, Zenati MS, Padussis JC, et al. Robotic assisted placement of hepatic artery infusion pump is a safe and feasible approach. J Surg Oncol. 2016;114:342–347. [DOI] [PubMed] [Google Scholar]
- 27.Qadan M, D’Angelica MI, Kemeny NE, et al. Robotic hepatic arterial infusion pump placement. HPB (Oxford). 2017;19:429–435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Urbach DR, Hansen PD Laparoscopic placement of a continuous hepatic artery infusion pump. Semin Laparosc Surg. 2000;7:140–147. [PubMed] [Google Scholar]
- 29.Perez DR, Kemeny NE, Brown KT, et al. Angiographic identification of extrahepatic perfusion after hepatic arterial pump placement: implications for surgical prevention. HPB (Oxford). 2014;16:744–748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Catalano OA, Singh AH, Uppot RN, et al. Vascular and biliary variants in the liver: implications for liver surgery. Radiographics. 2008;28:359–378. [DOI] [PubMed] [Google Scholar]
- 31.Hiatt JR, Gabbay J, Busuttil RW Surgical anatomy of the hepatic arteries in 1000 cases. Ann Surg. 1994;220:50–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Song SY, Chung JW, Yin YH, et al. Celiac axis and common hepatic artery variations in 5002 patients: systematic analysis with spiral CT and DSA. Radiology. 2010;255:278–288. [DOI] [PubMed] [Google Scholar]
- 33.Allen PJ, Nissan A, Picon AI, et al. Technical complications and durability of hepatic artery infusion pumps for unresectable colorectal liver metastases: an institutional experience of 544 consecutive cases. JAm Coll Surg. 2005;201:57–65. [DOI] [PubMed] [Google Scholar]
- 34.Yezhelyev M, Osgood M, Egnatashvili V, et al. Saphenous vein graft conduits for insertion of hepatic arterial infusion pumps in patients with abnormal hepatic arterial anatomy. J Surg Oncol. 2008;97:85–89. [DOI] [PubMed] [Google Scholar]
- 35.Aust J Saphenous vein graft as a conduit for hepatic artery catheters in anomalies of the hepatic arterial system. Surgery. 1986;100:581–583. [PubMed] [Google Scholar]
- 36.Callahan MK, Kemeny NE Implanted hepatic arterial infusion pumps. Cancer J. 2010;16:142–149. [DOI] [PubMed] [Google Scholar]
- 37.Sofocleous CT, Schubert J, Kemeny N, et al. Arterial embolization for salvage of hepatic artery infusion pumps. J Vasc Interv Radiol. 2006;17:801–806. [DOI] [PubMed] [Google Scholar]
- 38.Napier KJ, Lidsky ME, James OG, et al. Hepatic arterial infusion pumps: what the radiologist needs to know. Radiographics. 2021;41:895–908. [DOI] [PubMed] [Google Scholar]
- 39.Uliel L, Royal HD, Darcy MD, et al. From the angio suite to the gammacamera: vascular mapping and 99mTc-MAA hepatic perfusion imaging before liver radioembolization—a comprehensive pictorial review. J Nucl Med. 2012;53:1736–1747. [DOI] [PubMed] [Google Scholar]
- 40.Oberfield RA, McCaffrey JA, Polio J, et al. Prolonged and continuous percutaneous intra-arterial hepatic infusion chemotherapy in advanced metastatic liver adenocarcinoma from colorectal primary. Cancer. 1979;44:414–423. [DOI] [PubMed] [Google Scholar]
- 41.Reed ML, Vaitkevicius VK, Al-Sarraf M, et al. The practicality of chronic hepatic artery infusion therapy of primary and metastatic hepatic malignancies: ten-year results of 124 patients in a prospective protocol. Cancer. 1981;47:402–409. [DOI] [PubMed] [Google Scholar]
- 42.Turley M, Moore C, Creasy JM, et al. Resection of the primary tumor combined with hepatic artery infusion pump placement is a safe approach for unresectable colorectal liver metastases. J Gastrointest Surg. 2021. In Press. [DOI] [PubMed] [Google Scholar]
- 43.Curley SA, Chase JL, Roh MS, et al. Technical considerations and complications associated with the placement of 180 implantable hepatic arterial infusion devices. Surgery. 1993;114:928–935. [PubMed] [Google Scholar]
- 44.Ito K, Ito H, Kemeny NE, et al. Biliary sclerosis after hepatic arterial infusion pump chemotherapy for patients with colorectal cancer liver metastasis: incidence, clinical features, and risk factors. Ann Surg Oncol. 2012;19:1609–1617. [DOI] [PubMed] [Google Scholar]
- 45.Kemeny N, Capanu M, D’Angelica M, et al. Phase I trial of adjuvant hepatic arterial infusion (HAI) with floxuridine (FUDR) and dexamethasone plus systemic oxaliplatin, 5-fluorouracil and leucovorin in patients with resected liver metastases from colorectal cancer. Ann Oncol. 2009;20:1236–1241. [DOI] [PMC free article] [PubMed] [Google Scholar]


