Abstract
Recently, we reported that uric acid and salicylic acid are photosensitizers of the reaction of nucleosides with UV light via radical formation and energy transfer, respectively. In the present study, effects of 45 biologically relevant compounds on nucleoside reactions photosensitized by uric acid and salicylic acid were examined. When a mixed solution of 2'-deoxycytidine, 2'-deoxyguanosine, thymidine, and 2'-deoxyadenosine with uric acid was irradiated with UV light of a wavelength longer than 300 nm, all the nucleosides decreased. The addition of antioxidants suppressed the consumption of nucleosides. When the UV reaction of nucleosides was conducted with salicylic acid, thymidine decreased almost exclusively. Several antioxidants such as ascorbates, thiols, catecholamines, trans-2-hexen-1-ol, penicillin G, and NaHSO3 enhanced the consumption of thymidine, although the other antioxidants suppressed it. The results suggest that antioxidants may be beneficial to protect against DNA damage by photosensitization via radical formation, but that several of them may be detrimental as they facilitate DNA damage by photosensitization via energy transfer.
Keywords: uric acid, salicylic acid, nucleoside, photosensitizer, antioxidant
Introduction
UV radiation from the sun is the most harmful factor for human skin, causing skin cancer.(1,2) UV light of a wavelength shorter than 300 nm is absorbed by DNA directly to generate photoproducts.(3,4) However, since the atmosphere absorbs this almost completely, the strength of UV light at wavelengths shorter than 300 nm in sunlight is very low on the ground surface.(1) This suggests that the mechanism of skin cancer in humans caused by sunlight involves substances promoting photosensitization, photosensitizers. Photosensitization includes many different prosesses such as radical formation including electron transfer and hydrogen atom abstraction, and energy transfer including direct excitation of substrates and singet oxygen formation.(4,5) We recently reported that uric acid is a photosensitizer of the reaction of nucleosides, 2'-deoxycytidine (dC), 2'-deoxyguanosine (dG), thymidine (dT), and 2'-deoxyadenosine (dA), with UV light at wavelengths longer than 300 nm.(6) These reactions were inhibited by the addition of radical scavengers, ethanol and sodium azide. For the reaction of dC, N4,5-cyclic amide-2'-deoxycytidine was formed by cycloaddition of an amide group from uric acid. When a 15N-labeled uric acid, having two 14N and two 15N atoms in the molecule, was used, N4,5-cyclic amide-2'-deoxycytidine containing both 14N and 15N atoms was generated. For both the UV reactions of dG and dA acetylated on their ribose moiety, several products are generated.(7) All the identified products of the acetylated dG and dA had already been reported in the reaction with reactive free radicals and oxidants.(5,8,9) These results suggest that unidentified radicals derived from uric acid with a delocalized unpaired electron are generated and they react with nucleosides, generating the products. We also reported that salicylic acid is a photosensitizer of the reaction of nucleosides with UV light at wavelengths longer than 300 nm.(10) The photosensitization reaction by salicylic acid occurred for dT almost exclusively. The major products of dT are isomers of cyclobutane thymidine dimers. Energy transfer may be involved in the reaction mechanism for the thymidine dimer formation reaction by salicylic acid, since similar results have been reported for other energy transfer photosensitizers such as acetophenone and p-aminobenzoic acid.(4,11) When the photosensitization reaction of dT by salicylic acid was conducted in the presence of ascorbic acid, two pairs of diastereomers of ascorbic acid adducts of dT and 5,6-dihydrothymidine were produced.(12) dT consumption was markedly increased in an ascorbic acid dose-dependent manner. These results suggest that photosensitization reactions are strongly affected by co-existing compounds. However, there is limited information on this. In the present study, effects of 45 biologically relevant compounds on reactions photosensitized by uric acid and salicylic acid were examined.
Materials and Methods
Materials
The chemicals dC, dG, dT, dA, uric acid, (±)-α-tocopherol phosphate disodium (α-tocopherol phosphate), (±)-6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid (Trolox), l-glutathione reduced (glutathione), l-glutathione oxidized (glutathione disulfide), N-acetyl-l-cysteine (N-acetylcysteine), l-cysteine (cysteine), l-methionine (methionine), glycine, l-lysine monohydrochloride (lysine), l-tyrosine (tyrosine), (−)-epinephrine (adrenaline), sodium fluoride (NaF), sodium chloride (NaCl), sodium bromide (NaBr), sodium iodide (NaI), sodium nitrite (NaNO2), and sodium nitrate (NaNO3) were purchased from Sigma-Aldrich (St. Louis, MO). Sodium salicylate (salicylic acid), l-ascorbic acid sodium salt (ascorbic acid), l-ascorbic acid 2-phosphate trisodium salt (ascorbic acid 2-phosphate), taurine, l-tryptophan (tryptophan), l-DOPA, dopamine hydrochloride (dopamine), l-noradrenaline (noradrenaline), d-(+)-glucose (glucose), d-(−)-fructose (fructose), sucrose, pyruvic acid sodium salt (pyruvic acid), lactic acid, fumaric acid disodium salt (fumaric acid), maleic acid disodium salt (maleic acid), trans-p-coumaric acid, trans-2-hexenal, hexanoic acid, donepezil hydrochloride (donepezil), penicillin G potassium salt (penicillin G), urea, sodium hydrogensulfite (NaHSO3), sodium sulfate (Na2SO4), and sodium hydrogen carbonate (NaHCO3) were obtained from Nacalai Tesque (Kyoto, Japan). The chemicals 2-glyceryl ascorbate, trans-2-hexen-1-ol, and hexanal were from Tokyo Chemical Industry (Tokyo, Japan). Allantoin was obtained from Kanto Chemical (Tokyo, Japan). Metformin hydrochloride (metformin) was obtained from LKT Labs (St. Paul, MN). Other chemicals were obtained from Sigma-Aldrich, Nacalai Tesque, and Kanto Chemical. Water was purified with a Millipore Milli-Q deionizer (Billerica, MA).
Irradiation conditions
UV light from a high-pressure mercury lamp (250 W, SP9-250UB, Ushio, Tokyo, Japan) with an optical filter through a light guide was used to directly irradiate the surface of a solution (1 ml) in a glass vial (12 mm i.d.) at 37°C. A longpass optical filter with a cut-on of 300 nm (LU0300, Asahi Spectra, Tokyo, Japan) was used. The intensity of radiation on the surface of the sample solution was measured by a photometer (UIT-150, Ushio) equipped with the sensor UVD-S254 or UVD-S365.
Spectrophotometer and HPLC conditions
Transmittance and absorption spectra were obtained by a MultiSpec-1500 UV/Vis spectrometer (Shimadzu, Kyoto, Japan). The UV/Vis spectra were measured at room temperature in the wavelength range of 200–800 nm. An HPLC system including an SPD-M10Avp UV/Vis photodiode-array detector (Shimadzu) with an Inertsil ODS-3 octadecylsilane column of 4.6 × 250 mm and particle size of 5 μm (GL Sciences, Tokyo, Japan) was used. The eluent was 20 mM ammonium acetate (pH 7.0) containing methanol. The methanol concentration was increased from 0 to 50% over 15 min, and maintained at 50% from 15 to 30 min. The column temperature was 40°C and flow rate was 1 ml/min. The reversed phase (RP)-HPLC chromatogram was detected at 200–500 nm.
Results and Discussion
Irradiated UV light
UV light from an Hg lamp through a 300-nm longpass filter was used for the present UV irradiation reaction. Figure 1 shows the transmittance spectrum of the longpass filter for wavelengths ranging from 200 to 400 nm. The value of percentage transmittance was smaller than 1% at wavelengths from 200 to 290 nm. It increased from 290 to 310 nm. At wavelengths longer than 310 nm, it was around 90%. The intensities of the irradiated UV light on the surface of the sample solution through the filter were 0 mW/cm2 for 254 nm and 244 mW/cm2 for 365 nm.
Fig. 1.
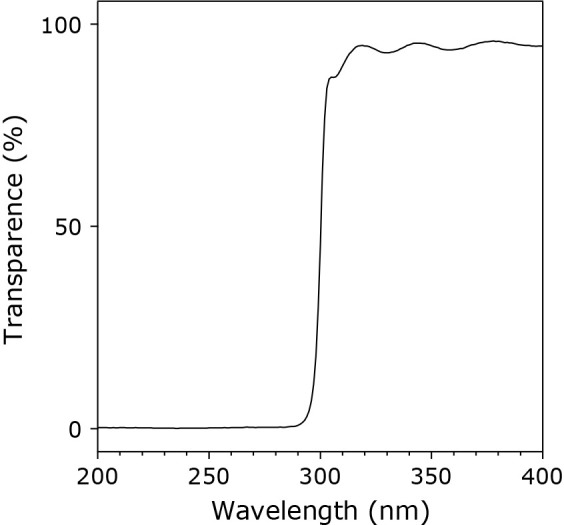
A UV transmittance spectrum of the longpass filter for wavelengths ranging from 200 to 400 nm. The percentage transmittance (%) was measured by a spectrophotometer at room temperature.
UV/Vis absorption spectra of additives
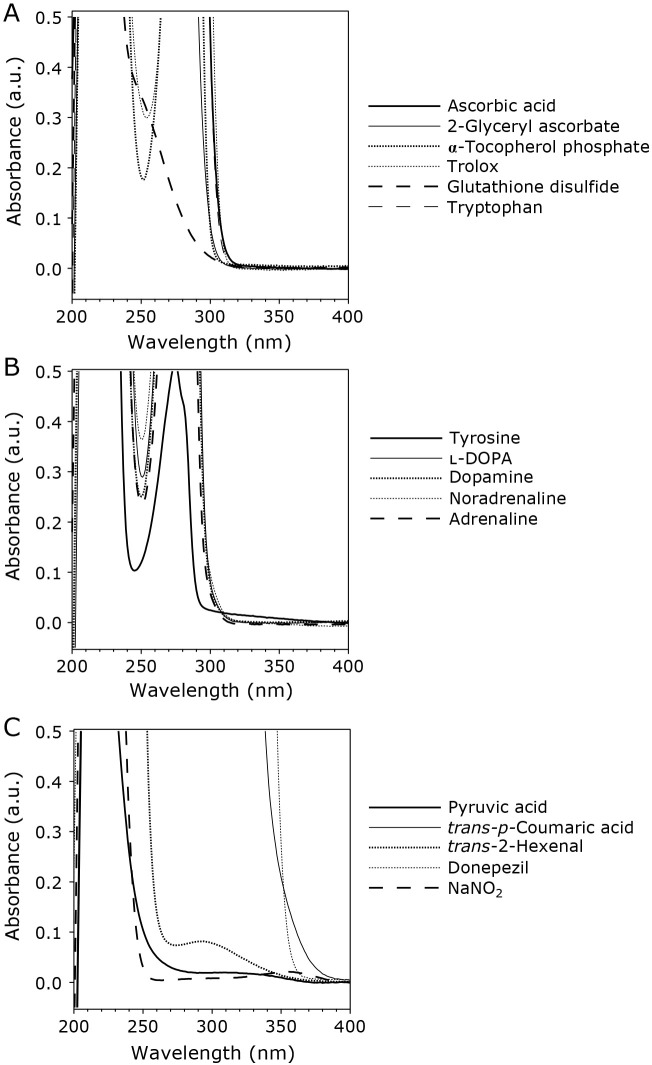
Forty-five biologically relevant compounds were used as additives. UV/Vis absorption spectra of 1 mM additives in 100 mM potassium phosphate buffer of pH 7.4 at room temperature were measured by a UV/Vis spectrometer at wavelengths ranging from 200 to 800 nm. Spectra of 16 out of the 45 compounds showed absorbance larger than 0.01 absorbance unit (a.u.) at wavelengths above 300 nm. Figure 2 shows UV spectra of the 16 compounds at wavelengths ranging from 200 to 400 nm. Among these compounds, trans-p-coumaric acid and donepezil showed strong absorption up to 350 nm.
Fig. 2.
UV absorption spectra from 200 to 400 nm of the 16 compounds used as additives, which showed an absorbance value larger than 0.01 (a.u.) at wavelengths ranging from 300–800 nm. UV spectra were measured by a spectrophotometer for 1 mM additives in 100 mM potassium phosphate buffer of pH 7.4 at room temperature.
UV irradiation without photosensitizers
At first, to clarify the effects of the 45 additives themselves on nucleosides irradiated by UV light, UV reactions were investigated in the absence of the photosensitizers, uric acid and salicylic acid. A mixed solution of dC, dG, dT, and dA (100 μM each) with 5 mM additives in 100 mM potassium phosphate buffer at pH 7.4 and 37°C was irradiated with UV light from an Hg lamp through a 300-nm longpass filter for 20 min. The reaction mixture was analyzed by RP-HPLC. The nucleoside concentration was determined from the HPLC peak area at 260 nm. Table 1 shows the concentrations of nucleosides in the UV-irradiated solutions. Without additives, no nucleoside reacted. Among the 45 additives, those that clearly induced a decrease of nucleoside concentrations were α-tocopherol phosphate, Trolox, tryptophan, l-DOPA, dopamine, noradrenaline, adrenaline, pyruvic acid, donepezil, NaNO2, and NaNO3. α-Tocopherol phosphate decreased the nucleosides, especially dT. α-Tocopherol phosphate is a naturally occurring water-soluble α-tocopherol analog detected in biological tissues and fluids.(13) α-Tocopherol phosphate is used as an ingredient of cosmetics due to its more favorable hydrophilicity and stability than α-tocopherol.(14) Recently, we reported that α-tocopherol phosphate induces the UV reaction of nucleosides, especially dT, generating 5,6-dihydrothymidine as the major product.(15) Trolox slightly decreased dC, dG, and dT. Trolox is another water-soluble α-tocopherol analog, substituting a hydrocarbon chain for a carboxy group.(16) Reportedly, Trolox induces the reaction of nucleosides by UV irradiation, although 5,6-dihydrothymidine is not generated.(15) Tryptophan reduced the dT concentration. It has been reported that tryptophan reacts with thymine to generate photoadducts by UV irradiation.(17) l-DOPA, dopamine, noradrenaline, and adrenaline mainly induced the consumption of dT. The color of all solutions of catecholamines changed from clear to brown-black or dark-red by UV irradiation. These catecholamines can be polymerized by light in the presence of oxygen, resulting in a certain type of melanin.(18–20) Although melanin absorbs UV light and protects the skin against DNA damage induced by UV irradiation, it also acts as a photosensitizer, generating reactive oxygen species including superoxide anion radicals and singlet oxygen, and also hydroxyl radicals in the presence of metal-ions.(21) Pyruvic acid markedly decreased the dG concentration. It has been reported that singlet oxygen is generated by pyruvic acid with UV irradiation.(22) Singlet oxygen can react with dG preferentially, generating several products including 8-oxo-7,8-dihydro-2'-deoxyguanosine and imidazolone and oxazolone derivatives.(23–25) Donepezil mainly decreased dG, and also dT and dC with lower efficiencies. Donepezil is an acetylcholinesterase inhibitor used for Alzheimer’s disease.(26) We could not find any report on the photosensitization activity of donepezil. NaNO2 reduced the concentrations of all nucleosides. Reportedly, UV irradiation at around 350 nm of the nitrite ion (NO2−) generates both the hydroxyl radical (•OH) and nitric oxide (•NO), inducing base release from nucleosides by •OH attack of their deoxyribose moiety and its subsequent decomposition.(27,28) NaNO3 reduced all the nucleosides with efficiency lower than that of NaNO2. Reportedly, UV irradiation of the nitrate ion (NO3−) generates both •OH and nitric dioxide (•NO2).(28)
Table 1.
Effects of additives on the reaction of nucleosides with UVa)
| Additives | Absorptionb)
(300–800 nm) |
dC (μM) | dG (μM) | dT (μM) | dA (μM) |
|---|---|---|---|---|---|
| None | − | 99.7 ± 0.2 | 99.7 ± 0.4 | 98.8 ± 0.1 | 99.8 ± 0.3 |
| Ascorbic acid | + | 98.2 ± 0.2 | 98.3 ± 0.7 | 94.0 ± 1.5 | 98.4 ± 0.5 |
| Ascorbic acid 2-phosphate | − | 100.0 ± 1.4 | 98.1 ± 0.4 | 96.9 ± 1.2 | 98.6 ± 1.5 |
| 2-Glyceryl ascorbate | + | 98.1 ± 0.2 | 97.9 ± 0.1 | 96.0 ± 0.1 | 97.9 ± 0.2 |
| α-Tocopherol phosphate | + | 86.0 ± 0.7 | 92.9 ± 1.0 | 37.6 ± 2.4 | 90.5 ± 0.6 |
| Trolox | + | 92.7 ± 0.5 | 95.4 ± 0.2 | 91.2 ± 2.2 | 98.6 ± 1.0 |
| Glutathione | − | 100.0 ± 0.6 | 100.7 ± 0.6 | 99.0 ± 0.4 | 100.6 ± 0.5 |
| Glutathione disulfide | + | 100.0 ± 0.2 | 99.4 ± 0.2 | 98.3 ± 0.4 | 100.2 ± 0.2 |
| N-Acetylcysteine | − | 100.3 ± 0.1 | 100.6 ± 0.1 | 99.2 ± 0.2 | 100.8 ± 0.1 |
| Cysteine | − | 100.9 ± 0.4 | 101.3 ± 0.3 | 99.3 ± 0.3 | 101.8 ± 0.6 |
| Methionine | − | 100.6 ± 0.4 | 101.0 ± 0.5 | 98.6 ± 1.1 | 100.6 ± 0.5 |
| Taurine | − | 100.4 ± 0.1 | 100.0 ± 0.2 | 98.7 ± 0.2 | 100.4 ± 0.2 |
| Glycine | − | 100.0 ± 0.1 | 100.0 ± 0.2 | 98.9 ± 0.4 | 100.3 ± 0.1 |
| Lysine | − | 101.7 ± 1.0 | 99.0 ± 1.0 | 98.2 ± 1.0 | 99.7 ± 1.1 |
| Tryptophan | + | 95.0 ± 0.5 | 98.9 ± 0.8 | 74.4 ± 2.6 | 94.9 ± 0.5 |
| Tyrosine | + | 97.3 ± 0.4 | 97.5 ± 0.4 | 94.8 ± 0.7 | 97.3 ± 0.4 |
| l-DOPA | + | 92.8 ± 2.4 | 95.4 ± 1.8 | 56.2 ± 1.3 | 91.7 ± 1.5 |
| Dopamine | + | 93.2 ± 0.8 | 97.7 ± 0.7 | 81.5 ± 2.5 | 97.9 ± 0.7 |
| Noradrenaline | + | 96.6 ± 2.4 | 95.9 ± 0.9 | 79.8 ± 2.6 | 95.6 ± 0.6 |
| Adrenaline | + | 93.7 ± 0.6 | 95.5 ± 1.3 | 81.0 ± 0.9 | 95.0 ± 1.6 |
| Glucose | − | 100.6 ± 1.1 | 100.7 ± 1.2 | 99.5 ± 1.2 | 100.8 ± 1.3 |
| Fructose | − | 99.6 ± 0.4 | 98.6 ± 1.4 | 98.6 ± 0.4 | 100.4 ± 0.0 |
| Sucrose | − | 100.2 ± 0.9 | 99.8 ± 0.8 | 98.5 ± 0.8 | 100.1 ± 0.7 |
| Pyruvic acid | + | 96.1 ± 1.6 | 14.3 ± 1.6 | 91.3 ± 1.9 | 97.2 ± 1.4 |
| Lactic acid | − | 99.6 ± 0.7 | 99.6 ± 0.6 | 99.9 ± 0.9 | 99.7 ± 0.9 |
| Fumaric acid | − | 99.1 ± 0.6 | 99.5 ± 0.2 | 98.1 ± 0.5 | 99.5 ± 0.4 |
| Maleic acid | − | 99.5 ± 1.2 | 99.0 ± 1.1 | 97.8 ± 1.2 | 99.4 ± 1.2 |
| trans-p-Coumaric acid | + | 99.9 ± 0.7 | 98.9 ± 0.7 | 98.6 ± 0.8 | 99.5 ± 0.5 |
| trans-2-Hexenal | + | 99.7 ± 0.2 | 97.9 ± 0.1 | 99.3 ± 0.3 | 100.5 ± 0.2 |
| trans-2-Hexen-1-ol | − | 100.1 ± 0.7 | 100.0 ± 0.6 | 98.5 ± 0.8 | 100.1 ± 0.8 |
| Hexanoic acid | − | 100.0 ± 0.4 | 99.8 ± 0.8 | 98.9 ± 0.5 | 100.4 ± 0.2 |
| Hexanal | − | 100.4 ± 1.0 | 99.2 ± 1.4 | 99.6 ± 1.0 | 100.2 ± 1.2 |
| Donepezil | + | 94.8 ± 1.0 | 72.0 ± 1.5 | 90.0 ± 0.9 | 101.8 ± 0.4 |
| Penicillin G | − | 98.7 ± 1.1 | 98.5 ± 1.3 | 98.2 ± 1.4 | 98.7 ± 1.1 |
| Metformin | − | 100.7 ± 1.0 | 101.1 ± 1.0 | 99.5 ± 0.9 | 101.3 ± 0.9 |
| Allantoin | − | 98.5 ± 0.2 | 98.6 ± 0.3 | 97.0 ± 0.4 | 98.7 ± 0.2 |
| Urea | − | 100.0 ± 0.5 | 99.1 ± 0.4 | 97.8 ± 0.8 | 99.4 ± 0.7 |
| NaF | − | 98.5 ± 0.5 | 98.3 ± 0.9 | 97.9 ± 0.6 | 98.6 ± 0.5 |
| NaCl | − | 99.1 ± 1.6 | 100.2 ± 0.1 | 98.3 ± 0.8 | 100.4 ± 0.2 |
| NaBr | − | 98.9 ± 0.3 | 98.3 ± 0.8 | 98.2 ± 0.2 | 99.0 ± 0.4 |
| NaI | − | 99.4 ± 0.3 | 99.8 ± 0.4 | 98.4 ± 0.4 | 99.5 ± 0.5 |
| NaNO2 | + | 64.0 ± 1.4 | 75.6 ± 0.2 | 63.1 ± 1.4 | 80.9 ± 0.3 |
| NaNO3 | − | 92.2 ± 0.6 | 93.4 ± 0.7 | 91.7 ± 0.6 | 97.3 ± 0.7 |
| NaHSO3 | − | 96.2 ± 0.9 | 97.2 ± 1.1 | 93.2 ± 1.0 | 100.3 ± 0.8 |
| Na2SO4 | − | 99.3 ± 0.7 | 99.8 ± 0.6 | 98.5 ± 0.6 | 99.9 ± 0.8 |
| NaHCO3 | − | 100.7 ± 0.3 | 100.9 ± 0.3 | 99.6 ± 0.4 | 101.3 ± 0.3 |
a)A mixed solution of 100 μM dC, dG, dT, and dA in 100 mM potassium phosphate buffer at pH 7.4 in the presence of 5 mM additives was irradiated with UV light through a 300-nm longpass filter at a temperature of 37°C for 20 min. b)“+” means that 1 mM additive in 100 mM potassium phosphate buffer at pH 7.4 shows an absorbance larger than 0.01 a.u. at wavelengths from 300 to 800 nm.
UV irradiation with uric acid
The experiments to examine the effects of the 45 additives on nucleosides irradiated by UV light were conducted in the presence of uric acid. A solution of dC, dG, dT, dA (100 μM each), and 400 μM uric acid with 5 mM additives in 100 mM potassium phosphate buffer at pH 7.4 and 37°C was irradiated with UV light for 20 min. The nucleoside and uric acid concentrations were determined from the RP-HPLC peak area at 260 or 300 nm. Table 2 shows the concentrations of nucleosides and uric acid in the UV-irradiated solutions. Without additives, all nucleosides were consumed in the order of dC>dT>dG>dA with the disappearance of almost all uric acid. Among the 45 compounds, additives that strongly suppressed the consumption of nucleosides were ascorbic acid and its derivatives, Trolox, thiols and its derivatives, tryptophan, tyrosine, catecholamines, trans-p-coumaric acid, trans-2-hexenal, trans-2-hexen-1-ol, donepezil, penicillin G, metformin, NaBr, NaI, and NaHSO3. Ascorbic acid is an effective antioxidant and a radical scavenger.(29,30) Various ascorbic acid derivatives are synthesized as ingredients of cosmetics for antioxidant protection against UV-induced photodamage.(31–33) Trolox acts as a multiple free radical scavenger.(34) Thiols are important antioxidants in cells and body fluids.(35) In addition to thiols, glutathione disulfide, a disulfide, and methionine, a thioether, also reduce oxidative DNA damage.(36,37) Although tryptophan is not a good antioxidant, its oxidative metabolites scavenge peroxyl radicals with high efficiency.(38) Tyrosine, a nomophenolic compound, shows antioxidant and antiradical activities.(39) Catecholamines, diphenolic compounds, are scavenger of radicals.(40,41) trans-p-Coumaric acid, a monophenolic acid, is ubiquitously distributed in fruits and vegetables and shows antioxidant and radical scavenging activities.(42) trans-2-Hexenal, an α,β-unsaturated aldehyde, and trans-2-hexen-1-ol, an α,β-unsaturated alcohol, are major volatile compounds in green leaves and olive oil.(43) Although trans-2-hexenal is mutagenic due to generating a cyclic adduct with dG, it acts as a hydroxyl radical scavenger.(44,45) It has been reported that cis-2-hexen-1-ol, a cis isomer of trans-2-hexen-1-ol, shows radical scavenging activity greater than that of trans-2-hexenal.(46) Donepezil can act as an antioxidant and a radical scavenger.(47) Penicillin G, an antibiotic, can act as a scavenger of radicals, since it has a cyclic thioether moiety, which is acceptable for free radicals.(48,49) Metformin, an antihyperglycemic drug for type 2 diabetes, shows radical scavenging effects.(50) The bromide ion (Br−) and iodide ion (I−) show scavenging effects for •OH.(51) The bisulfite ion (HSO3−), a reducing reagent, is widely added to tap water and food because of its antioxidant, antibacterial, and bleaching effects.(52) All of the above compounds, which strongly suppress the reaction, can act as good antioxidants or radical scavengers, and they would scavenge uric acid radicals effectively. Compounds that moderately suppressed the UV reaction were taurine, lysine, glucose, fructose, sucrose, pyruvic acid, lactic acid, fumaric acid, maleic acid, hexanoic acid, hexanal, NaNO2, and Na2SO4. These compounds may react with radicals formed from uric acid with relatively lower efficiencies. Although pyruvic acid induced decrease of dG concentration almost exclusively in the absence of uric acid probably due to generation of 1O2 (Table 1),(22) the consumption of dG was reduced in the presence of uric acid (Table 2). Uric acid may scavenge 1O2 generated by pyruvic acid, since uric acid is an excellent scavenger of 1O2.(53) Compounds showing no or little effects were α-tocopherol phosphate, glycine, allantoin, urea, NaF, NaCl, NaNO3, and NaHCO3. Although α-tocopherol phosphate induced decrease of dT concentration almost exclusively in the absence of uric acid probably due to hydrogenation of dT (Table 1),(15) the consumption of dT was reduced in the presence of uric acid (Table 2). Uric acid may suppress the hydrogenation of dT by α-tocopherol phosphate. Among the 45 compounds tested, no additive accelerated the reaction of nucleosides with UV light in the presence of uric acid.
Table 2.
Effects of additives on the reaction of nucleosides with UV in the presence of uric acida)
| Additives | dC (μM) | dG (μM) | dT (μM) | dA (μM) | Uric acid (μM) |
|---|---|---|---|---|---|
| None | 36.5 ± 1.4 | 70.8 ± 1.5 | 49.8 ± 1.2 | 81.1 ± 0.5 | 13 ± 7 |
| Ascorbic acid | 97.9 ± 0.3 | 100.1 ± 0.2 | 94.5 ± 0.7 | 99.3 ± 0.2 | 391 ± 8 |
| Ascorbic acid 2-phosphate | 83.4 ± 2.7 | 97.8 ± 1.0 | 88.6 ± 2.2 | 97.9 ± 1.2 | 114 ± 31 |
| 2-Glyceryl ascorbate | 88.2 ± 1.4 | 98.7 ± 1.8 | 93.5 ± 1.4 | 99.5 ± 2.0 | ND |
| α-Tocopherol phosphate | 50.4 ± 1.8 | 85.0 ± 1.8 | 57.1 ± 1.6 | 86.0 ± 1.8 | 33 ± 4 |
| Trolox | 96.0 ± 1.8 | 101.6 ± 1.3 | 95.1 ± 0.8 | 103.9 ± 1.2 | 268 ± 15 |
| Glutathione | 97.1 ± 0.6 | 99.6 ± 0.6 | 98.4 ± 0.3 | 99.3 ± 0.6 | 375 ± 6 |
| Glutathione disulfide | 97.0 ± 0.7 | 100.8 ± 0.9 | 96.4 ± 0.9 | 101.3 ± 1.3 | 144 ± 6 |
| N-Acetylcysteine | 100.3 ± 0.4 | 101.0 ± 0.0 | 100.1 ± 0.7 | 101.5 ± 0.5 | 387 ± 5 |
| Cysteine | 98.9 ± 0.2 | 100.8 ± 0.1 | 99.7 ± 0.7 | 100.9 ± 0.3 | 385 ± 4 |
| Methionine | 95.8 ± 1.2 | 98.2 ± 0.6 | 97.9 ± 0.5 | 98.2 ± 0.5 | 359 ± 22 |
| Taurine | 75.0 ± 3.2 | 88.2 ± 3.2 | 81.0 ± 3.6 | 91.6 ± 1.2 | 234 ± 17 |
| Glycine | 37.1 ± 0.3 | 74.0 ± 0.2 | 51.2 ± 0.0 | 83.5 ± 0.0 | 15 ± 0 |
| Lysine | 72.3 ± 3.3 | 90.9 ± 1.1 | 78.3 ± 2.9 | 93.0 ± 0.4 | 46 ± 24 |
| Tryptophan | 97.8 ± 1.5 | 97.8 ± 1.3 | 89.3 ± 2.7 | 88.3 ± 2.2 | 45 ± 2 |
| Tyrosine | 84.4 ± 1.9 | 98.3 ± 2.4 | 88.9 ± 1.5 | 98.7 ± 3.2 | 13 ± 4 |
| l-DOPA | 100.5 ± 0.4 | 100.8 ± 0.2 | 87.9 ± 7.4 | 101.1 ± 0.1 | 380 ± 4 |
| Dopamine | 89.2 ± 0.6 | 101.3 ± 0.5 | 96.2 ± 0.8 | 101.7 ± 1.7 | 425 ± 69 |
| Noradrenaline | 98.6 ± 2.0 | 100.8 ± 2.6 | 98.0 ± 2.5 | 101.4 ± 2.2 | ND |
| Adrenaline | 97.0 ± 0.4 | 97.2 ± 0.4 | 89.9 ± 0.4 | 97.2 ± 0.1 | ND |
| Glucose | 75.4 ± 0.8 | 90.5 ± 2.1 | 82.5 ± 0.8 | 97.0 ± 0.5 | 163 ± 18 |
| Fructose | 79.5 ± 2.4 | 91.0 ± 2.1 | 85.1 ± 2.0 | 97.3 ± 1.7 | 217 ± 7 |
| Sucrose | 71.7 ± 2.2 | 90.6 ± 2.2 | 81.7 ± 3.3 | 98.3 ± 2.2 | 54 ± 10 |
| Pyruvic acid | 70.7 ± 1.4 | 79.0 ± 0.6 | 78.1 ± 1.4 | 89.7 ± 0.8 | 117 ± 16 |
| Lactic acid | 65.4 ± 1.9 | 90.2 ± 0.8 | 77.5 ± 1.7 | 91.7 ± 1.7 | 54 ± 13 |
| Fumaric acid | 78.7 ± 5.3 | 94.0 ± 5.7 | 90.3 ± 5.9 | 99.3 ± 6.3 | 62 ± 24 |
| Maleic acid | 75.7 ± 2.5 | 87.2 ± 1.6 | 83.4 ± 3.7 | 93.2 ± 2.4 | 178 ± 21 |
| trans-p-Coumaric acid | 95.4 ± 0.6 | 96.5 ± 0.3 | 98.4 ± 0.6 | 96.7 ± 0.6 | 349 ± 19 |
| trans-2-Hexenal | 90.0 ± 0.4 | 92.0 ± 1.7 | 95.5 ± 0.7 | 95.4 ± 3.8 | 114 ± 49 |
| trans-2-Hexen-1-ol | 98.7 ± 0.7 | 101.0 ± 0.4 | 100.5 ± 0.8 | 101.3 ± 0.8 | 371 ± 15 |
| Hexanoic acid | 76.7 ± 1.0 | 94.7 ± 2.1 | 84.5 ± 2.3 | 97.0 ± 1.6 | 74 ± 11 |
| Hexanal | 79.3 ± 1.5 | 87.9 ± 2.2 | 83.5 ± 1.4 | 96.3 ± 1.1 | 56 ± 5 |
| Domepezil | 96.8 ± 4.3 | 87.1 ± 4.2 | 98.2 ± 4.4 | 98.6 ± 4.3 | 26 ± 3 |
| Penicillin G | 93.9 ± 1.6 | 95.6 ± 1.5 | 96.0 ± 1.3 | 95.2 ± 2.2 | 196 ± 11 |
| Metformin | 84.2 ± 0.5 | 87.5 ± 0.1 | 86.9 ± 0.5 | 92.9 ± 0.4 | 255 ± 10 |
| Allantoin | 45.9 ± 1.7 | 78.2 ± 3.2 | 58.9 ± 1.6 | 82.8 ± 4.4 | 14 ± 7 |
| Urea | 43.3 ± 1.5 | 73.9 ± 4.1 | 55.0 ± 2.6 | 82.8 ± 2.0 | 16 ± 4 |
| NaF | 37.9 ± 2.5 | 73.3 ± 3.0 | 52.5 ± 3.6 | 82.2 ± 2.8 | 22 ± 14 |
| NaCl | 57.9 ± 3.0 | 85.1 ± 2.0 | 67.9 ± 3.0 | 87.6 ± 1.2 | 36 ± 8 |
| NaBr | 84.2 ± 1.1 | 94.2 ± 2.4 | 90.0 ± 1.2 | 96.2 ± 1.6 | 126 ± 18 |
| NaI | 96.2 ± 0.2 | 98.9 ± 0.3 | 98.5 ± 0.2 | 98.7 ± 0.2 | 374 ± 20 |
| NaNO2 | 61.8 ± 5.0 | 78.7 ± 3.3 | 63.8 ± 6.6 | 81.6 ± 3.1 | 113 ± 3 |
| NaNO3 | 48.6 ± 2.8 | 75.4 ± 4.3 | 62.1 ± 2.0 | 83.6 ± 2.4 | 85 ± 45 |
| NaHSO3 | 88.1 ± 7.3 | 97.2 ± 5.9 | 86.5 ± 6.4 | 97.7 ± 5.7 | 237 ± 29 |
| Na2SO4 | 72.7 ± 5.3 | 88.5 ± 2.0 | 77.8 ± 4.1 | 91.7 ± 1.8 | 189 ± 47 |
| NaHCO3 | 48.4 ± 2.4 | 73.2 ± 3.4 | 55.3 ± 2.7 | 83.8 ± 0.9 | 16 ± 5 |
a)A mixed solution of 100 μM dC, dG, dT, and dA in 100 mM potassium phosphate buffer at pH 7.4 in the presence of 400 μM uric acid and 5 mM additives was irradiated with UV light through a 300-nm longpass filter at a temperature of 37°C for 20 min. ND denotes not determined due to overlapping peaks on the PR-HPLC chromatogram.
UV irradiation with salicylic acid
The experiments to examine the effects of 45 additives on the nucleosides irradiated by UV light were conducted in the presence of salicylic acid. A solution of dC, dG, dT, dA (100 μM each), and 1 mM salicylic acid with 5 mM additives in 100 mM potassium phosphate buffer at pH 7.4 and 37°C was irradiated with UV light for 20 min. The nucleoside and salicylic acid concentrations were determined from the RP-HPLC peak area at 260 nm. Table 3 shows the concentrations of nucleosides and salicylic acid in the UV-irradiated solutions. Without additives, almost exclusive consumption of dT was observed. The consumption of dT was markedly increased by additions of ascorbic acid and its derivatives, thiols and its derivatives, catecholamines except for adrenaline, trans-2-hexen-1-ol, penicillin G, and NaHSO3. All the additives strongly enhancing the reaction were antioxidants. Among these compounds, the effects of additions of ascorbic acid and glutathione on the UV reaction of dT or thymine have been reported. Ascorbic acid accelerates the reaction of dT with UV in a reaction photosensitized by salicylic acid, generating products of two adducts of ascorbic acid, 5-(2-deoxy-2-l-ascorbyl)-5,6-dihydrothymidine and 5-(2-l-ascorbyl)-5,6-dihydrothymidine, and an adduct of two hydrogen atoms, 5,6-dihydrothymidine.(12) Glutathione reacts with thymine to generate several adducts including 5-S-glutathione-5,6-dihydrothymine and 5-S,S-glutathione-5,6-dihydrothymine by both direct UV excitation and photosensitization.(54) On the other hand, Trolox, tryptophan, pyruvic acid, trans-p-coumaric acid, trans-2-hexenal, donepezil, and NaI strongly suppressed the dT reaction. Among them, NaI showed no absorption at wavelengths from 270 to 800 nm (<0.01 a.u. at 1 mM, data not shown). I− did not suppress the consumption of salicylic acid (Table 3). It has been reported that I− efficiently deactivates excited states of pterin and lumazine caused by UV light with rate constants close to the diffusion-controlled limit.(55) In the present system, I− would quench excited states of dT but not those of salicylic acid. Pyruvic acid and trans-2-hexenal showed only weak absorption at above 300 nm (Fig. 2C), and they did not suppress the consumption of salicylic acid. Trolox and tryptophan showed only weak absorption at above 300 nm (Fig. 2A), but they partially suppressed consumption of salicylic acid. trans-p-Coumaric acid and donepezil showed strong absorption at wavelengths shorter than 350 nm (Fig. 2C). Since donepezil did not suppress the consumption of salicylic acid, it may quench the excited states of dT. trans-p-Coumaric acid showed the strongest suppression of nucleoside consumption by UV irradiation among the 45 compounds examined in the present study (Table 3). In addition, no consumption of salicylic acid was observed. trans-p-Coumaric acid would quench the excited state of salicylic acid or block excitation of salicylic acid by absorbing UV light. The other compounds not mentioned above showed little or no effect.
Table 3.
Effects of additives on the reaction of nucleosides with UV in the presence of salicylic acida)
| Additives | dC (μM) | dG (μM) | dT (μM) | dA (μM) | Salicylic acid (μM) |
|---|---|---|---|---|---|
| None | 97.0 ± 1.2 | 95.4 ± 1.1 | 59.1 ± 3.9 | 101.4 ± 1.2 | 719 ± 30 |
| Ascorbic acid | 101.7 ± 0.1 | 93.4 ± 1.0 | 2.4 ± 0.5 | 89.0 ± 1.6 | 828 ± 3 |
| Ascorbic acid 2-phosphate | 94.9 ± 0.4 | 95.3 ± 0.3 | 5.4 ± 4.3 | 98.2 ± 0.2 | 834 ± 16 |
| 2-Glyceryl ascorbate | 93.5 ± 0.7 | 97.6 ±3.1 | 22.7 ± 7.8 | 99.1 ± 2.1 | 857 ± 39 |
| α-Tocopherol phosphate | 96.9 ± 0.3 | 97.2 ± 0.8 | 31.8 ± 3.0 | 99.8 ± 0.4 | 820 ± 21 |
| Trolox | 92.8 ± 0.9 | 93.2 ± 0.9 | 93.0 ± 1.2 | 102.0 ± 1.5 | 870 ± 21 |
| Glutathione | 95.2 ± 3.5 | 100.8 ± 3.5 | 0.2 ± 0.0 | 97.6 ± 1.5 | 712 ± 20 |
| Glutathione disulfide | 101.2 ± 2.8 | 99.4 ± 3.0 | 22.6 ± 2.1 | 100.3 ± 2.7 | 846 ± 6 |
| N-Acetylcysteine | 97.0 ± 0.6 | 101.9 ± 0.1 | 13.5 ± 1.8 | 98.3 ± 0.4 | 761 ± 4 |
| Cysteine | 99.0 ± 0.8 | 100.7 ± 0.5 | 2.7 ± 0.9 | 96.9 ± 0.8 | 831 ± 3 |
| Methionine | 97.9 ± 5.5 | 98.3 ± 5.5 | 10.3 ± 5.3 | 103.4 ± 1.0 | 767 ± 24 |
| Taurine | 98.3 ± 3.2 | 95.0 ± 2.9 | 66.5 ± 5.6 | 100.5 ± 3.1 | 713 ± 18 |
| Glycine | 99.1 ± 1.3 | 95.9 ± 1.8 | 59.4 ± 2.9 | 100.9 ± 2.7 | 734 ± 12 |
| Lysine | 98.7 ± 0.5 | 94.3 ± 0.6 | 64.9 ± 5.7 | 100.3 ± 0.8 | 744 ± 22 |
| Tryptophan | 88.9 ± 1.9 | 100.7 ± 0.5 | 84.5 ± 2.8 | 94.4 ± 0.1 | 913 ± 13 |
| Tyrosine | 96.7 ± 0.4 | 94.4 ± 0.7 | 36.1 ± 4.5 | 97.3 ± 0.5 | 712 ± 32 |
| l-DOPA | 97.7 ± 0.6 | 100.1 ± 0.4 | 3.4 ± 0.8 | 100.0 ± 0.6 | 864 ± 11 |
| Dopamine | 98.2 ± 2.8 | 99.9 ± 3.0 | 5.0 ± 0.4 | 98.8 ± 2.9 | 858 ± 24 |
| Noradrenaline | 100.3 ± 2.4 | 99.7 ± 2.7 | 11.7 ± 2.0 | 100.8 ± 2.4 | 860 ± 8 |
| Adrenaline | 100.4 ± 0.6 | 99.4 ± 0.2 | 43.9 ± 4.7 | 99.6 ± 0.2 | 935 ± 8 |
| Glucose | 98.5 ± 0.6 | 95.7 ± 0.8 | 66.9 ± 2.2 | 100.8 ± 0.7 | 727 ± 2 |
| Fructose | 98.5 ± 1.0 | 95.6 ± 1.0 | 65.3 ± 5.0 | 101.5 ± 0.5 | 720 ± 17 |
| Sucrose | 90.9 ± 0.2 | 87.2 ± 4.0 | 62.5 ± 3.3 | 94.4 ± 1.0 | 736 ± 76 |
| Pyruvic acid | 96.7 ± 0.2 | 97.1 ± 1.3 | 94.1 ± 0.3 | 98.4 ± 0.2 | 648 ± 27 |
| Lactic acid | 91.9 ± 1.8 | 92.3 ± 0.3 | 58.3 ± 1.5 | 97.4 ± 1.7 | 745 ± 21 |
| Fumaric acid | 96.0 ± 2.4 | 95.0 ± 1.7 | 54.2 ± 1.0 | 98.5 ± 2.7 | 805 ± 25 |
| Maleic acid | 93.4 ± 2.0 | 92.0 ± 1.9 | 40.7 ± 2.0 | 97.2 ± 2.0 | 707 ± 69 |
| trans-p-Coumaric acid | 98.6 ± 0.3 | 97.7 ± 0.2 | 98.8 ± 0.2 | 98.2 ± 0.2 | 989 ± 5 |
| trans-2-Hexenal | 97.2 ± 0.3 | 94.5 ± 0.7 | 91.1 ± 0.5 | 99.7 ± 0.5 | 637 ± 19 |
| trans-2-Hexen-1-ol | 100.2 ± 4.5 | 94.1 ± 3.1 | 6.4 ± 2.4 | 100.2 ± 3.6 | 715 ± 31 |
| Hexanoic acid | 93.5 ± 2.3 | 91.5 ± 2.0 | 56.3 ± 4.6 | 97.4 ± 2.4 | 737 ± 51 |
| Hexanal | 99.3 ± 0.5 | 90.8 ± 0.4 | 30.7 ± 3.8 | 96.8 ± 0.4 | 675 ± 18 |
| Donepezil | 97.2 ± 2.3 | 86.0 ± 2.6 | 95.7 ± 1.8 | 98.8 ± 1.7 | 521 ± 20 |
| Penicillin G | 99.3 ± 2.0 | 99.7 ± 2.4 | 11.6 ± 0.9 | 101.0 ± 2.3 | 892 ± 9 |
| Metformin | 102.6 ± 0.2 | 93.1 ± 0.6 | 65.2 ± 4.4 | 99.2 ± 0.4 | 717 ± 7 |
| Allantoin | 98.2 ± 3.9 | 92.6 ± 1.5 | 70.5 ± 2.8 | 100.4 ± 4.0 | 808 ± 19 |
| Urea | 99.0 ± 1.7 | 92.8 ± 1.4 | 65.0 ± 7.2 | 98.0 ± 0.7 | 744 ± 11 |
| NaF | 94.7 ± 1.1 | 94.3 ± 1.8 | 67.2 ± 2.4 | 100.7 ± 0.8 | 723 ± 20 |
| NaCl | 92.4 ± 1.9 | 90.8 ± 2.0 | 60.1 ± 0.1 | 96.4 ± 1.9 | 726 ± 84 |
| NaBr | 97.4 ± 2.3 | 91.6 ± 3.8 | 54.6 ± 3.8 | 98.4 ± 3.9 | 722 ± 55 |
| NaI | 98.6 ± 0.4 | 95.1 ± 0.5 | 91.9 ± 2.4 | 100.4 ± 0.1 | 725 ± 19 |
| NaNO2 | 72.9 ± 0.7 | 82.5 ± 0.6 | 79.5 ± 0.5 | 85.2 ± 0.6 | 467 ± 32 |
| NaNO3 | 88.5 ± 1.7 | 87.1 ± 1.8 | 61.9 ± 0.5 | 92.6 ± 1.8 | 646 ± 25 |
| NaHSO3 | 80.0 ± 3.0 | 93.6 ± 0.4 | 4.0 ± 0.5 | 98.3 ± 0.5 | 747 ± 7 |
| Na2SO4 | 92.7 ± 2.9 | 91.3 ± 2.8 | 57.3 ± 5.3 | 97.1 ± 2.9 | 679 ± 42 |
| NaHCO3 | 93.7 ± 2.0 | 89.8 ± 1.9 | 56.4 ± 3.5 | 97.6 ± 2.4 | 712 ± 20 |
a)A mixed solution of 100 μM dC, dG, dT, and dA in 100 mM potassium phosphate buffer at pH 7.4 in the presence of 1 mM salicylic acid and 5 mM additives was irradiated with UV light through a 300-nm longpass filter at a temperature of 37°C for 20 min.
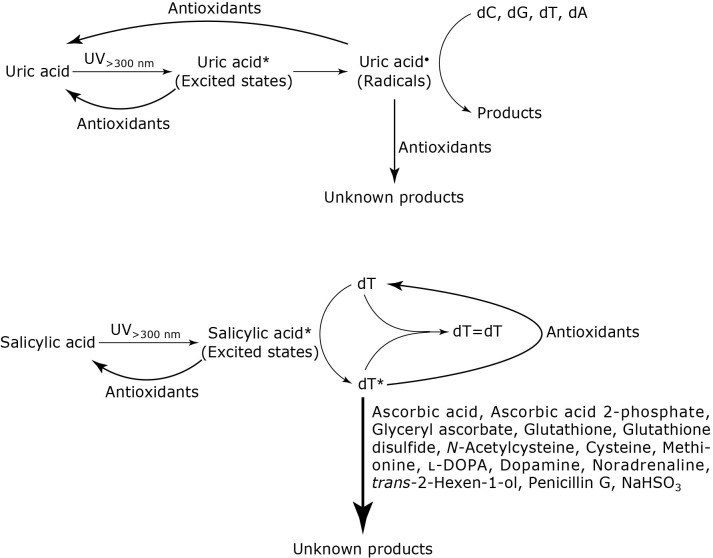
Possible scheme for effects of antioxidants
In the present results, the nucleoside reaction photosensitized by uric acid was suppressed by antioxidants with radical scavenging ability. A possible scheme for inhibition of the reaction is shown in Fig. 3, upper part. Uric acid is activated by UV light at wavelengths longer than 300 nm to form radicals (Uric acid•) through excited states (Uric acid*). Uric acid• reacts with nucleosides to generate products.(6,7) Some antioxidants quench Uric acid* and return it to a ground state. Other antioxidants react with Uric acid•, returning it to uric acid or generating unknown products. On the other hand, regarding the dT reaction photosensitized by salicylic acid, some antioxidants facilitated the reaction but other antioxidants suppressed it. A possible scheme for the reaction is shown in Fig. 3, lower part. Salicylic acid is excited by UV light at wavelengths longer than 300 nm. Energy of excited salicylic acid (Salicylic acid*) is transferred to dT, resulting in excited dT (dT*). Although the lifetime of dT* in H2O is short and it returns to a ground state spontaneously,(56) a small amount of dT* exists continuously during UV irradiation. When dT* encounters ground-state dT, thymidine dimers (dT=dT) are formed. Several antioxidants such as ascorbates, thiols, catecholamines, trans-2-hexen-1-ol, penicillin G, and NaHSO3 react with dT*. When dT* reacts with the antioxidants more efficiently than with dT at the reaction concentrations, the consumption of dT is increased by the addition of antioxidants.
Fig. 3.
Schemes for possible roles of the antioxidants in the reaction of nucleosides with UV irradiation in the presence of uric acid and salicylic acid.
Conclusion
The present study showed that although the reaction of nucleosides with UV light in the presence of uric acid was suppressed by antioxidants, the reaction of dT in the presence of salicylic acid was enhanced by several antioxidants. This suggests that antioxidants would suppress UV DNA damage via radical formation, but several of them may enhance UV DNA damage caused via energy transfer on thymine sites. Various antioxidants are used as ingredients of cosmetics to suppress cell damage by UV light. We should pay attention to the genotoxicity of antioxidants in terms of DNA damage caused by sunlight.
Conflict of Interest
No potential conflicts of interest were disclosed.
References
- 1.IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Vol. 55 “Solar and ultraviolet radiation”. Lyon: IARC Press, 1992; 50–58. [PMC free article] [PubMed] [Google Scholar]
- 2.IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Vol. 100D “Solar and ultraviolet radiation”. Lyon: IARC Press, 2012; 35–101. [Google Scholar]
- 3.Hicks MR, Kowałski J, Rodger A. LD spectroscopy of natural and synthetic biomaterials. Chem Soc Rev 2010; 39: 3380–3393. [DOI] [PubMed] [Google Scholar]
- 4.Cadet J, Vigny P. The photochemistry of nucleic acids. In: Morrison H., ed. Bioorganic Photochemistry: Photochemistry and the Nucleic Acids. Vol. 1, Chap. 1. New York: Wiley, 1990; 1–272. [Google Scholar]
- 5.von Sonntag C. Free-Radical-Induced DNA Damage and Its Repair: A Chemical Perspective. Berlin: Springer-Verlag, 2006. [Google Scholar]
- 6.Suzuki T, Ozawa-Tamura A, Takeuchi M, Sasabe Y. Uric acid as a photosensitizer in the reaction of deoxyribonucleosides with UV light of wavelength longer than 300 nm: identification of products from 2'-deoxycytidine. Chem Pharm Bull (Tokyo) 2021; 69: 1067–1074. [DOI] [PubMed] [Google Scholar]
- 7.Suzuki T, Takeuchi M, Ozawa-Tamura A. Reactions of 3',5'-di-O-acetyl-2'-deoxyguansoine and 3',5'-di-O-acetyl-2'-deoxyadenosine to UV light in the presence of uric acid. Genes Environ 2022; 44: 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Suzuki T, Nakamura A, Inukai M. Reaction of 3',5'-di-O-acetyl-2'-deoxyguansoine with hypobromous acid. Bioorg Med Chem 2013; 21: 3674–3679. [DOI] [PubMed] [Google Scholar]
- 9.Chattopadhyaya R, Goswami B. Oxidative damage to DNA constituents by iron-mediated Fenton reactions: the deoxyadenosine family. J Biomol Struct Dyn 2012; 30: 394–406. [DOI] [PubMed] [Google Scholar]
- 10.Suzuki T, Ota H, Namba Y, Fujino T. Salicylic acid as a photosensitizer for thymidine dimerization induced by UV. Chem Pharm Bull (Tokyo) 2019; 67: 130–134. [DOI] [PubMed] [Google Scholar]
- 11.Aliwell SR, Martincigh BS, Salter LF. p-Aminobenzoic acid-photosensitized dimerization of thymine—I. In DNA-related model systems. J Photochem Photobiol A 1993; 71: 147–153. [Google Scholar]
- 12.Suzuki T, Kishida Y. Reaction of thymidine and ascorbic acid induced by UV in the presence of salicylic acid. Bioorg Med Chem 2019; 27: 115046. [DOI] [PubMed] [Google Scholar]
- 13.Zingg JM. Water-soluble vitamin E-tocopheryl phosphate. Adv Food Nutr Res 2018; 83: 311–363. [DOI] [PubMed] [Google Scholar]
- 14.Fiume MM, Bergfeld WF, Belsito DV, et al. Safety assessment of tocopherols and tocotrienols as used in cosmetics. Int J Toxicol 2018; 37 (2_suppl): 61S–94S. [DOI] [PubMed] [Google Scholar]
- 15.Suzuki T, Ono C. α-Tocopherol phosphate as a photosensitizer in the reaction of nucleosides with UV light: formation of 5,6-dihydrothymidine. Genes Environ 2022; 44: 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Davies MJ, Forni LG, Willson RL. Vitamin E analogue Trolox C. E.s.r. and pulse-radiolysis studies of free-radical reactions. Biochem J 1988; 255: 513–522. [PMC free article] [PubMed] [Google Scholar]
- 17.Saito I, Sugiyama H, Matsuura T, Fukuyama K, Katsube Y. Photochemical reaction between tryptophan and thymine. Tetrahedron Lett 1984; 25: 3243–3246. [Google Scholar]
- 18.Kollias N, Sayre RM, Zeise L, Chedekel MR. Photoprotection by melanin. J Photochem Photobiol B 1991; 9: 135–160. [DOI] [PubMed] [Google Scholar]
- 19.Hill HZ. The function of melanin or six blind people examine an elephant. Bioessays 1992; 14: 49–56. [DOI] [PubMed] [Google Scholar]
- 20.Du X, Li L, Li J, et al. UV-triggered dopamine polymerization: control of polymerization, surface coating, and photopatterning. Adv Mater 2014; 26: 8029–8033. [DOI] [PubMed] [Google Scholar]
- 21.Korytowski W, Pilas B, Sarna T, Kalyanaraman B. Photoinduced generation of hydrogen peroxide and hydroxyl radicals in melanins. Photochem Photobiol 1987; 45: 185–190. [DOI] [PubMed] [Google Scholar]
- 22.Eugene AJ, Guzman MI. Production of singlet oxygen (1O2) during the photochemistry of aqueous pyruvic acid: the effects of pH and photon flux under steady-state O2(aq) concentration. Environ Sci Technol 2019; 53: 12425–12432. [DOI] [PubMed] [Google Scholar]
- 23.Cadet J, Teoule R. Comparative study of oxidation of nucleic acid components by hydroxyl radicals, singlet oxygen and superoxide anion radicals. Photochem Photobiol 1978; 28: 661–667. [DOI] [PubMed] [Google Scholar]
- 24.Suzuki T, Friesen MD, Ohshima H. Formation of a diimino-imidazole nucleoside from 2'-deoxyguanosine by singlet oxygen generated by methylene blue photooxidation. Bioorg Med Chem 2003; 11: 2157–2162. [DOI] [PubMed] [Google Scholar]
- 25.Martinez GR, Medeiros MHG, Ravanat JL, Cadet J, Di Mascio P. [18O]-labeled singlet oxygen as a tool for mechanistic studies of 8-oxo-7,8-dihydroguanine oxidative damage: detection of spiroiminodihydantoin, imidazolone and oxazolone derivatives. Biol Chem 2002; 383: 607–617. [DOI] [PubMed] [Google Scholar]
- 26.Brewster JT 2nd, Dell'Acqua S, Thach DQ, Sessler JL. Classics in chemical neuroscience: donepezil. ACS Chem Neurosci 2019; 10: 155–167. [DOI] [PubMed] [Google Scholar]
- 27.Bilski P, Chignell CF, Szychlinski J, Borkowski A, Oleksy E, Reszka K. Photooxidation of organic and inorganic substrates during UV photolysis of nitrite anion in aqueous solution. J Am Chem Soc 1992; 114: 549–556. [Google Scholar]
- 28.Suzuki T, Inukai M. Effects of nitrite and nitrate on DNA damage induced by ultraviolet light. Chem Res Toxicol 2006; 19: 457–462. [DOI] [PubMed] [Google Scholar]
- 29.Niki E. Action of ascorbic acid as a scavenger of active and stable oxygen radicals. Am J Clin Nutr 1991; 54 (6 Suppl): 1119S–1124S. [DOI] [PubMed] [Google Scholar]
- 30.Wang K, Jiang H, Li W, Qiang M, Dong T, Li H. Role of vitamin C in skin diseases. Front Physiol 2018; 9: 819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stamford NP. Stability, transdermal penetration, and cutaneous effects of ascorbic acid and its derivatives. J Cosmet Dermatol 2012; 11: 310–317. [DOI] [PubMed] [Google Scholar]
- 32.Takebayashi J, Tai A, Gohda E, Yamamoto I. Characterization of the radical-scavenging reaction of 2-O-substituted ascorbic acid derivatives, AA-2G, AA-2P, and AA-2S: a kinetic and stoichiometric study. Biol Pharm Bull 2006; 29: 766–771. [DOI] [PubMed] [Google Scholar]
- 33.Gérard V, Ay E, Graff B, et al. Ascorbic acid derivatives as potential substitutes for ascorbic acid to reduce color degradation of drinks containing ascorbic acid and anthocyanins from natural extracts. J Agric Food Chem 2019; 67: 12061–12071. [DOI] [PubMed] [Google Scholar]
- 34.Kamogawa E, Sueishi Y. A multiple free-radical scavenging (MULTIS) study on the antioxidant capacity of a neuroprotective drug, edaravone as compared with uric acid, glutathione, and trolox. Bioorg Med Chem Lett 2014; 24: 1376–1379. [DOI] [PubMed] [Google Scholar]
- 35.Ulrich K, Jakob U. The role of thiols in antioxidant systems. Free Radic Biol Med 2019; 140: 14–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Eteshola EOU, Haupt DA, Koos SI, Siemer LA, Morris DL. The role of metal ion binding in the antioxidant mechanisms of reduced and oxidized glutathione in metal-mediated oxidative DNA damage. Metallomics 2020; 12: 79–91. [DOI] [PubMed] [Google Scholar]
- 37.Stadtman ER. Cyclic oxidation and reduction of methionine residues of proteins in antioxidant defense and cellular regulation. Arch Biochem Biophys 2004; 423: 2–5. [DOI] [PubMed] [Google Scholar]
- 38.Christen S, Peterhans E, Stocker R. Antioxidant activities of some tryptophan metabolites: possible implication for inflammatory diseases. Proc Natl Acad Sci USA 1990; 87: 2506–2510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gülçin I. Comparison of in vitro antioxidant and antiradical activities of l-tyrosine and l-Dopa. Amino Acids. 2007;32:431–438. doi: 10.1007/s00726-006-0379-x. [DOI] [PubMed] [Google Scholar]
- 40.Kawashima T, Ohkubo K, Fukuzumi S. Radical scavenging reactivity of catecholamine neurotransmitters and the inhibition effect for DNA cleavage. J Phys Chem B 2010; 114: 675–680. [DOI] [PubMed] [Google Scholar]
- 41.Jodko-Piórecka K, Litwinienko G. Antioxidant activity of dopamine and l-DOPA in lipid micelles and their cooperation with an analogue of α-tocopherol. Free Radic Biol Med. 2015;83:1–11. doi: 10.1016/j.freeradbiomed.2015.02.006. [DOI] [PubMed] [Google Scholar]
- 42.Pei K, Ou J, Huang J, Ou S. p-Coumaric acid and its conjugates: dietary sources, pharmacokinetic properties and biological activities. J Sci Food Agric 2016; 96: 2952–2962. [DOI] [PubMed] [Google Scholar]
- 43.Sonda A, Akram Z, Boutheina G, Guido F, Mohamed B. Effect of addition of olive leaves before fruits extraction process to some monovarietal Tunisian extra-virgin olive oils using chemometric analysis. J Agric Food Chem 2014; 62: 251–263. [DOI] [PubMed] [Google Scholar]
- 44.Eder E, Hoffman C, Sporer S, Scheckenbach S. Biomonitoring studies and susceptibility markers for acrolein congeners and allylic and benzyl compounds. Environ Health Perspect 1993; 99: 245–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jiménez E, Lanza B, Martínez E, Albaladejo J. Daytime tropospheric loss of hexanal and trans-2-hexenal: OH kinetics and UV photolysis. Atmos Chem Phys 2007; 7: 1565–1574. [Google Scholar]
- 46.Sarang K, Otto T, Rudzinski K, et al. Reaction kinetics of green leaf volatiles with sulfate, hydroxyl, and nitrate radicals in tropospheric aqueous phase. Environ Sci Technol 2021; 55: 13666–13676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Munishamappa V, Seethalakshmi, Vijayakumar AE, Rajathilagam T. Evaluation of the antioxidant activity of donepezil - in vitro study. Natl J Physiol Pharm Pharmacol 2019; 9: 108–110. [Google Scholar]
- 48.Berczyński P, Kładna A, Kruk I, Aboul-Enein HY. Radical-scavenging activity of penicillin G, ampicillin, oxacillin, and dicloxacillin. Luminescence 2017; 32: 434–442. [DOI] [PubMed] [Google Scholar]
- 49.Szabó L, Tóth T, Rácz G, Takács E, Wojnárovits L. Drugs with susceptible sites for free radical induced oxidative transformations: the case of a penicillin. Free Radic Res 2016; 50: 26–38. [DOI] [PubMed] [Google Scholar]
- 50.Khouri H, Collin F, Bonnefont-Rousselot D, Legrand A, Jore D, Gardès-Albert M. Radical-induced oxidation of metformin. Eur J Biochem 2004; 271: 4745–4772. [DOI] [PubMed] [Google Scholar]
- 51.Suzuki T, Yamada K, Inukai M. Effects of chloride, bromide, and iodide upon decomposition of nucleosides induced by ultrasound in neutral solution. Nucleosides Nucleotides Nucleic Acids 2010; 29: 606–615. [DOI] [PubMed] [Google Scholar]
- 52.Yuan G, Zhou L, Yang Q, Ding H, Tan L, Peng L. Rational development of a new reaction-based ratiometric fluorescent probe with a large stokes shift for selective detection of bisulfite in tap water, real food samples, onion tissues, and zebrafish. J Agric Food Chem 2021; 69: 4894–4902. [DOI] [PubMed] [Google Scholar]
- 53.Ames BN, Cathcart R, Schwiers E, Hochstein P. Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesis. Proc Natl Acad Sci USA 1981; 78: 6858–6862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Varghese AJ. Photochemical addition of glutathione to uracil and thymine. Photochem Photobiol 1974; 20: 339–343. [DOI] [PubMed] [Google Scholar]
- 55.Denofrio MP, Ogiby PR, Oliveros E, Thomas AH, Lorente C. Selective quenching of triplet excited states of pteridines. Photochem Photobiol Sci 2014; 13: 1058–1065. [DOI] [PubMed] [Google Scholar]
- 56.Gustavsson T, Sarkar N, Lazzarotto E, Markovitsi D, Improta R. Singlet excited state dynamics of uracil and thymine derivatives: a femtosecond fluorescence upconversion study in acetonitrile. Chem Phys Lett 2006; 429: 551–557. [Google Scholar]