Abstract
Background
Obstructive sleep apnea (OSA), nocturnal hypoxemia and excessive daytime sleepiness (EDS) are common comorbidities in people with cystic fibrosis (pwCF). Most of the data showing this originates from children and adolescents. The aim of this study was to collect data on sleep parameters, EDS and pulmonary function from a large cohort of adult pwCF.
Methods
Full overnight polysomnography (PSG) was performed. EDS was determined using the Epworth Sleepiness Scale (ESS). Demographic and clinical data (body mass index [BMI], pulmonary function, capillary blood gases) were collected.
Results
A total of 52 adult pwCF were included (mean age 30.7 ± 8.0 years, mean percent predicted forced expiratory volume in 1 s [ppFEV1] of 52.1 ± 14.8). Overall AHI was in the normal range (4.5 ± 4.0/h); 21/52 pwCF (40%) had an apnea-hypopnea index > 5/h. Nocturnal hypoxemia was found in 25% of participants and this was associated with ppFEV1 (p = 0.014), awake oxygen saturation (SpO2; p = 0.021) and awake partial pressure of oxygen (pO2; p = 0.003); there were no significant differences in age, lung function and BMI were found for pwCF with versus without OSA (all p > 0.05). Eight pwCF (15%) had an ESS score > 10 (indicating EDS). OSA was best predicted by awake pO2 (area under the curve [AUC] 0.66, p = 0.048), while nocturnal hypoxemia was best predicted by ppFEV1 (AUC 0.74, p = 0.009), awake pO2 (AUC 0.76, p = 0.006) and awake SpO2 (AUC 0.71; p = 0.025).
Conclusion
OSA, nocturnal hypoxemia and EDS were common in adult pwCF, but no strong predictors were identified. Therefore, we suggest regular PSG and ESS scoring in adult pwCF, regardless of disease severity.
Keywords: Cystic fibrosis, Adults, Polysomnography, Excessive daytime sleepiness, Apnea-hypopnea index, Obstructive sleep apnea, Nocturnal hypoxemia
Background
Cystic fibrosis (CF), an autosomal recessive monogenetic disorder, is caused by mutations in the CF transmembrane conductance regulator (CFTR) gene on chromosome 7 [1]. This results in disturbed anion transport (Cl− and HCO3−) through epithelial cell membranes and therefore to the formation of highly viscous secretions in all exocrine organs. The organs that are primarily affected are the lungs and the gastro-intestinal tract, leading to progressive lung damage and malnutrition [2]. Mortality and morbidity are mainly caused by lung involvement with progressive obstructive lung disease, hyperinflation, impaired gas exchange and end-stage respiratory failure [3]. Due to advances in the treatment of people with cystic fibrosis (pwCF), life expectancy has increased to more than 50 years of age and the number of adults with CF now exceeds the number of children with the disease [4].
As with other chronic obstructive lung diseases, such as chronic obstructive pulmonary disease (COPD) and asthma [5, 6], pwCF of all ages may also have sleep-disordered breathing (SDB), mainly obstructive sleep apnea (OSA) and nocturnal hypoxemia [7–9]. The gold standard technique for diagnosing SDB is overnight, in-laboratory polysomnography (PSG), as recommended by the American Academy of Sleep Medicine (AASM) [10]. An apnea-hypopnea index (AHI) of ≥ 5/h is required for diagnosis of OSA. The prevalence of OSA in the general population is up to 38%, with (overweight) males and elderly people most likely to be affected [11]. The reported prevalence of OSA in pwCF varies widely, being as high as 70% in children [12] and up to 3.9% in adults [13].
Nocturnal hypoxemia is also a common finding in pwCF [14–16]. Sleep studies in pwCF of all ages showed a direct correlation between the presence of nocturnal hypoxemia and lung disease severity [13, 15, 17]. In addition, nocturnal hypoxemia may be present even in children with mild lung disease [12]. Furthermore, hypoventilation with hypercapnia requiring non-invasive ventilation is common in individuals with advanced CF [18, 19].
Poor subjective and objective sleep quality is a well-known comorbidity in pwCF across all age groups [20–22]. In addition to disease-specific causes of disturbed sleep, such as coughing or nocturnal PEG feeding, SDB is also likely to play an important role. Impaired sleep and SDB may have a variety of consequences in pwCF, including reduced health-related quality of life (HRQoL) [20, 23], increased daytime sleepiness [20, 24], reduced neurocognitive function [24], development of pulmonary hypertension [25], or reduced physical activity [26]. Despite the knowledge that SDB is a common comorbidity in pwCF and its negative impact on different aspects of the disease, most CF centers do not screen routinely for SDB [27]. Recent reviews highlighted the significant impact of SDB for pwCF. Further research with larger sample sizes was requested to better define SDB in pwCF and to avoid nonattention of this relevant comorbitiy.
The aims of this study were to determine the presence of OSA and nocturnal hypoxemia in a large cohort of adult pwCF, to assess the clinical characteristics of pwCF with and without OSA, nocturnal hypoxemia and excessive daytime sleepiness (EDS), and to evaluate possible clinical parameters for predicting OSA and nocturnal hypoxemia in pwCF.
Methods
Study design
This prospective, observational, and descriptive-analytical study included adult pwCF from the Adult Cystic Fibrosis Unit of the Ruhrlandklinik Essen, Germany, who were recruited between September and December 2020. The study was approved by the local ethics committee of the University Hospital Essen (19-8961-BO) and followed the Declaration of Helsinki Ethical Principles for Medical Research Involving Human Subjects. All pwCF provided written informed consent for participation in the study.
Participants
All participants were ≥ 18 years of age and had a diagnosis of CF based on the presence of two defining mutations in the CFTR gene. All participants were clinically stable without signs of respiratory exacerbation, on stable medication and had a stable percent predicted forced expiratory volume in 1 s (ppFEV1) for a minimum of 4 weeks prior to the study assessments.
Polysomnography and Epworth Sleepiness Scale (ESS)
In-lab PSG was performed using a digital polygraph (Nox Medical, Iceland). Two electroencephalograms (EEG), two electrooculograms (EOG), submental and tibialis electromyogram (EMG), rib cage and abdominal inductance pneumograms, pulse oximeter (Nonin, Minnesota, USA), nasal cannula (measurement of flow at a sample frequency of 20 Hz), and body position were recorded. Using the AASM standard [28], an apnea was defined as a flow cessation for ≥ 10 s, and a hypopnea was defined as a ≥ 50% reduction in flow amplitude or a ≥ 30% decrease in flow amplitude accompanied by a > 3% reduction in oxygen saturation lasting ≥ 10 s. Relevant oxygen desaturation during sleep was defined as an oxygen saturation (SpO2) of < 90% for ≥ 5% of total sleep time (TST) with a nadir of at least 85%, as described elsewhere [16].
All signals were recorded automatically and subsequently analyzed blinded by the same German Sleep Society (DGSM) trained investigator (SDT) to prevent inter-rater variability. The AHI was defined as the number of apneas and hypopneas per hour of sleep. Depending on whether they occurred in rapid eye movement (REM) or non-REM (NREM) sleep phases, AHI was further classified as AHI REM or AHI NREM.
After diagnostic PSG, participants were asked to answer the ESS questionnaire, a self-report instrument that addresses the possibility of falling asleep in daily life. The score consists of 8 items (0–3 points each) ranging from 0 to 24. A score of > 10 indicates daytime sleepiness [29].
Pulmonary function testing (PFT) and body mass index
Calculation of the body mass index (BMI) and pulmonary function testing (PFT) were performed on the day of, or one day after, the diagnostic sleep study. Forced vital capacity (FVC), forced expiratory volume in 1 s (FEV1) and residual volume (RV) were measured with a JAEGER MasterScreen Body (CareFusion, Hoechberg, Germany) according to ATS guidelines [30]. Global Lung Function Initiative reference values were used [31]. Arterialized ear lobe blood gas samplings were used to assess pH, partial pressure of oxygen (pO2), partial pressure of carbon dioxide (pCO2), base excess (BE), and bicarbonate (HCO3-). This analysis was performed before PFT.
Statistical analysis
Statistical analysis was performed using the SPSS statistics package version 27 (SPSS Inc., Chicago, USA). Data are presented as mean ± standard deviation. The Shapiro-Wilk test was used to evaluate the data for normal distribution. Student’s t-test or Mann-Whitney U-test were used to assess between-group differences, as appropriate. A p-value of < 0.05 was considered statistically significant.
Receiver operator characteristics (ROC) analysis was performed to identify predictors of OSA and nocturnal hypoxemia using ppFEV1, BMI, awake SpO2 and pO2, ESS score and age as variables. GraphPad Prism version 9.3 (GraphPad Software, San Diego, USA) was used for plotting ROC curves and ROC analysis, including determination of the area under the ROC curve (AUC), specificity and sensitivity for the single variables.
Results
Study population
A total of 64 pwCF were recruited, all of whom underwent full PSG. Twelve patients were excluded from statistical analysis due to having a TST < 180 min or an insufficient sleep data quality or missing PFT data. The remaining 52 pwCF were included in the analysis (Table 1). Two patients used nocturnal oxygen supplementation, which was paused during the diagnostic night. No patient was using nocturnal continuous or bilevel positive airway pressure (CPAP/BiPAP) therapy.
Table 1.
Patient clinical and demographic characteristics
| Characteristics | Patients (n = 52) |
|---|---|
| Age, years | 30.7 ± 8.0 (20–49) |
| Female, n (%) | 18 (35) |
| Genotype, n (%) | |
| F508del homozygous | 38 (73) |
| F508del heterozygous | 14 (27) |
| CFTR modulator therapy, n (%) | |
| None | 16 (31) |
| Tezacaftor/ivacaftor | 33 (63) |
| Lumacaftor/ivacaftor | 3 (6) |
| Body mass index, kg/m2 | 21.5 ± 3.3 (15.6–31.2) |
| FEV1, L | 2.1 ± 0.8 (1.0-4.8) |
| FEV1, % predicted | 52.1 ± 14.8 (30.0–96.0) |
| FVC, L | 3.4 ± 1.1 (1.5–6.2) |
| FVC, % predicted | 69.5 ± 16.2 (37.0-105.0) |
| Residual volume, L | 3.0 ± 0.9 (1.1–5.6) |
| Residual volume, % predicted | 179.9 ± 41.9 (99.0-283.0) |
| Pancreatic insufficiency, n (%) | 51 (98) |
| Pseudomonas aeruginosa positive, n (%) | 28 (54) |
| Cystic fibrosis-related diabetes, n (%) | 12 (23) |
| Oxygen supplementation, n (%) | 2 (4) |
Data are mean ± standard deviation (range) or number of patients (%)
CFTR, cystic fibrosis transmembrane conductance regulator; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity
Mean age of the study population was 30.7 ± 8.0 years with a mean ppFEV1 of 52.1 ± 14.8 (Table 1). BMI ranged from 15.6 to 31.2 kg/m2 (mean 21.5 ± 3.3) and 15% of patients had a BMI > 25 kg/m2 (Table 1). The ESS score was 6.7 ± 3.8 overall, and 15% of patients had an ESS score of > 10 (Table 1).
Respiratory events and nocturnal gas exchange
Overall, the AHI was in the normal range (4.5 ± 4.0 events/h), and was higher during REM sleep (Table 2). Awake capillary blood gas analysis revealed normal values. Mean and minimum nocturnal SpO2 values were 92.1 ± 2.2% and 87.0 ± 3.8% respectively (Table 2). Thirteen patients (25%) had significant nocturnal hypoxemia (SpO2 < 90% for more than 5% of TST and a nadir of at least 85%), and mean sleep time spent with SpO2 < 90% was 41.3 min (maximum 321.3 min) (Table 2).
Table 2.
Polysomnographic data
| Patients (n = 52) | |
|---|---|
| AHI, events/h | 4.5 ± 4.0 (0-15.5) |
| AHI > 5 events/h, n (%) | 21 (40) |
| AHI REM, events/h | 10.2 ± 10.2 (0-38.5) |
| AHI NREM, events/h | 3.1 ± 3.4 (0-17.3) |
| Arousal index, events/h | 18.8 ± 10.2 (0.5–46.4) |
| ESS score | 6.7 ± 3.8 (0–22) |
| ESS score > 10, n (%) | 8 (15) |
| TST, min | 298.1 ± 43.8 (180–400) |
| Sleep efficiency, % | 74.9 ± 10.2 (48.6–93.9) |
| Sleep latency, min | 69.6 ± 37.4 (8.8-151.2) |
| WASO, min | 33.3 ± 27.7 (0.5-145.5) |
| Sleep stages, % TST | |
| N1 | 3.1 ± 1.8 (0.5-9.0) |
| N2 | 51.0 ± 7.9 (31.3–68.7) |
| N3 | 25.5 ± 8.9 (11.1–49.8) |
| REM | 17.7 ± 6.6 (5.2–34.3) |
| ODI, events/h | 4.2 ± 3.8 (0-15.5) |
| ODI REM, events/h | 11.2 ± 13.8 (0–80.0) |
| ODI NREM, events/h | 2.9 ± 3.1 (0-15.7) |
| Nocturnal mean SpO2, % | 92.1 ± 2.2 (84–96) |
| Nocturnal minimum SpO2, % | 87.0 ± 3.8 (77–92) |
| SpO2 < 90%, % TST | 13.4 ± 27.3 (0.0-99.9) |
| SpO2 < 90%, min | 41.3 ± 85.5 (0.0-321.3) |
| Nocturnal respiratory rate, breaths/min | 21.4 ± 3.9 (15.0-31.2) |
| Nocturnal heart rate, beats/min | 67.9 ± 12.5 (49.9-102.3) |
| Awake SpO2, % | 95.8 ± 1.7 (91.0–98.0) |
| Awake pO2, mmHg | 78.6 ± 9.4 (57.0–97.0) |
| Awake pCO2, mmHg | 38.9 ± 2.9 (33.0–45.0) |
| Awake pH | 7.4 ± 0.3 (7.29–7.50) |
| Awake HCO3−, mmol/L | 24.9 ± 2.0 (19.1–30.5) |
Values are mean ± standard deviation (range) or number of patients (%)
AHI, apnea-hypopnea index; ESS, Epworth Sleepiness Scale; HCO3−, bicarbonate; NREM, non-rapid eye movement sleep; ODI, oxygen desaturation index; pCO2, partial pressure of carbon dioxide pressure; pO2, partial pressure of oxygen; REM, rapid eye movement sleep; SpO2, oxygen saturation; TST, total sleep time; WASO, wake after sleep onset
Sleep structure
Adult pwCF showed decreased sleep efficiency 74.9 ± 10.2%) and increased sleep latency (69.6 ± 37.4 min) (Table 2). Sleep architecture was in the normal range for time spent in N1-3 and REM sleep (Table 2).
Profiles of pwCF with and without OSA
Twenty-one of 52 pwCF (40%) fulfilled polysomnographic criteria for OSA (AHI > 5/h). There were no significant differences between pwCF with and without OSA with respect to age, lung function, BMI, ESS score, sleep architecture and sleep quality (Table 3). Those with versus without OSA had significantly lower nocturnal oxygen levels (Table 3). Respiratory rates did not differ between the two groups, whereas pwCF with OSA had a slightly higher nocturnal heart rate than those without OSA (p < 0.049) (Table 3). Only two of the 21 pwCF with OSA has an ESS score > 10 (Table 3).
Table 3.
Comparison of clinical characteristics and polysomnographic data in people with cystic fibrosis with and without obstructive sleep apnea
| OSA (n = 21) | No OSA (n = 31) | p value | |
|---|---|---|---|
| Age, years | 32.6 ± 9.6 | 29.4 ± 6.5 | 0.318 |
| AHI, events/h | 8.6 ± 3.4 | 1.9 ± 1.3 | 0.000 |
| AHI REM, events/h | 18.3 ± 11.2 | 5.2 ± 4.7 | 0.000 |
| AHI NREM, events/h | 6.0 ± 3.8 | 1.2 ± 1.0 | 0.000 |
| ODI, events/h | 8.4 ± 3.6 | 1.9 ± 1.1 | 0.000 |
| ODI REM, events/h | 25.5 ± 30.1 | 5.5 ± 4.8 | 0.000 |
| ODI NREM, events/h | 5.8 ± 4.0 | 1.2 ± 0.9 | 0.000 |
| Arousal index, events/h | 22.4 ± 9.8 | 16.4 ± 10.1 | 0.039 |
| ESS score | 6.8 ± 4.3 | 6.7 ± 3.6 | 0.888 |
| TST, min | 302.5 ± 35.5 | 298.5 ± 48.4 | 0.621 |
| Sleep efficiency, % | 76.3 ± 9.3 | 74.5 ± 10.9 | 0.539 |
| Sleep latency, min | 61.5 ± 32.9 | 72.7 ± 39.5 | 0.520 |
| WASO, min | 34.2 ± 17.9 | 33.6 ± 33.4 | 0.176 |
| Sleep stages, % TST | |||
| N1 | 3.0 ± 1.9 | 3.1 ± 1.7 | 0.608 |
| N2 | 49.4 ± 6.2 | 51.9 ± 8.7 | 0.261 |
| N3 | 25.2 ± 7.4 | 25.1 ± 9.8 | 0.668 |
| REM | 19.8 ± 5.4 | 17.1 ± 6.9 | 0.134 |
| Nocturnal mean SpO2, % | 91.0 ± 2.8 | 92.8 ± 1.5 | 0.025 |
| Nocturnal minimum SpO2, % | 83.8 ± 3.6 | 89.1 ± 2.1 | 0.000 |
| spO2 < 90%, % TST | 30.6 ± 36.7 | 1.7 ± 4.4 | 0.000 |
| spO2 < 90%, min | 94.9 ± 115.6 | 5.0 ± 11.8 | 0.000 |
| Nocturnal respiratory rate, breaths/min | 21.0 ± 4.3 | 21.6 ± 3.8 | 0.613 |
| Nocturnal heart rate, beats/min | 71.8 ± 12.9 | 64.4 ± 10.2 | 0.049 |
| BMI, kg/m2 | 21.9 ± 3.7 | 21.2 ± 3.0 | 0.737 |
| FEV1, L | 2.0 ± 0.8 | 2.1 ± 0.8 | 0.608 |
| FEV1, % predicted | 49.1 ± 14.8 | 54.2 ± 14.7 | 0.233 |
| FVC, L | 3.3 ± 1.1 | 3.4 ± 1.0 | 0.920 |
| FVC, % predicted | 66.4 ± 17.9 | 71.7 ± 14.9 | 0.252 |
| RV, L | 3.2 ± 0.9 | 2.9 ± 0.8 | 0.303 |
| RV, % predicted | 179.9 ± 37.7 | 180.0 ± 45.1 | 0.996 |
| Awake SpO2, % | 95.4 ± 1.8 | 96.0 ± 1.5 | 0.136 |
| Awake pO2, mmHg | 75.7 ± 8.9 | 80.9 ± 8.4 | 0.048 |
| Awake pCO2, mmHg | 39.5 ± 2.7 | 38.5 ± 2.8 | 0.256 |
| Awake pH | 7.42 ± 0.02 | 7.40 ± 0.03 | 0.023 |
| Awake HCO3−, mmol/L | 25.6 ± 1.6 | 24.1 ± 1.7 | 0.003 |
Values are mean ± standard deviation
AHI, apnea-hypopnea index; ESS, Epworth Sleepiness Scale; BMI, body mass index; HCO3−, bicarbonate; NREM, non-rapid eye movement sleep; pCO2, partial pressure of carbon dioxide pressure; pO2, partial pressure of oxygen; ppFEV1, percent predicted forced expiratory volume in 1 s; ppFVC, percent predicted forced vital capacity; ODI, oxygen desaturation index; OSA, obstructive sleep apnea; REM, rapid eye movement sleep; RV, residual volume; SpO2, oxygen saturation; TST, total sleep time; WASO, wake after sleep onset
Bold values denote statistical significance at the p < 0.05 level
Profiles of pwCF with and without nocturnal hypoxemia
Thirteen (25%) pwCF had relevant nocturnal hypoxemia with SpO2 < 90% for ≥ 5% of TST with a nadir of at least 85%. Those with versus without nocturnal hypoxemia had significantly lower ppFEV1, ppFVC and markers of awake oxygenation, and significantly higher AHI, AHI REM and AHI NREM (Table 4). There were no significant differences between patient groups in RV, age, BMI, ESS score, nocturnal respiratory and heart rates, awake pCO2, and sleep quality and architecture (Table 4). Only one of the thirteen patients with nocturnal hypoxemia had an ESS score > 10 (Table 4).
Table 4.
Comparison of clinical characteristics and polysomnographic data in people with cystic fibrosis with and without nocturnal hypoxemia
| Hypoxemia (n = 13) | No hypoxemia (n = 39) | p value | |
|---|---|---|---|
| Age, years | 34.4 ± 10.2 | 29.4 ± 6.8 | 0.120 |
| AHI, events/h | 8.8 ± 3.8 | 3.2 ± 3.1 | 0.000 |
| AHI REM, events/h | 20.3 ± 11.8 | 7.2 ± 7.3 | 0.001 |
| AHI NREM, events/h | 5.8 ± 4.3 | 2.3 ± 2.6 | 0.000 |
| ODI, events/h | 9.0 ± 4.0 | 3.1 ± 2.9 | 0.000 |
| ODI REM, events/h | 21.5 ± 10.6 | 7.4 ± 7.3 | 0.000 |
| ODI NREM, events/h | 6.1 ± 4.8 | 2.1 ± 2.4 | 0.000 |
| Arousal index, events/h | 20.9 ± 9.8 | 18.1 ± 10.5 | 0.409 |
| ESS score | 5.9 ± 3.0 | 6.8 ± 4.1 | 0.400 |
| TST, min | 308.3 ± 21.5 | 297.3 ± 48.4 | 0.575 |
| Sleep efficiency, % | 78.0 ± 7.6 | 74.4 ± 10.9 | 0.279 |
| Sleep latency, min | 59.9 ± 27.2 | 70.9 ± 39.7 | 0.634 |
| WASO, min | 36.8 ± 17.0 | 32.8 ± 30.9 | 0.139 |
| Sleep stages, % TST | |||
| N1 | 3.1 ± 1.9 | 3.1 ± 1.7 | 0.916 |
| N2 | 48.2 ± 5.3 | 51.7 ± 8.4 | 0.159 |
| N3 | 26.9 ± 6.3 | 24.6 ± 9.5 | 0.148 |
| REM | 19.1 ± 5.7 | 17.9 ± 6.7 | 0.565 |
| Nocturnal mean SpO2, % | 89.6 ± 2.6 | 92.9 ± 1.5 | 0.000 |
| Nocturnal minimum SpO2, % | 81.4 ± 2.2 | 88.8 ± 2.0 | 0.000 |
| Nocturnal respiratory rate, breaths/min | 22.7 ± 4.6 | 20.1 ± 3.7 | 0.172 |
| Nocturnal heart rate, beats/min | 72.9 ± 13.0 | 65.5 ± 11.0 | 0.064 |
| BMI, kg/m2 | 21.8 ± 3.9 | 21.4 ± 3.1 | 0.983 |
| FEV1, L | 1.7 ± 0.6 | 2.2 ± 0.8 | 0.014 |
| FEV1, % predicted | 43.5 ± 13.0 | 55.0 ± 14.4 | 0.014 |
| FVC, L | 2.9 ± 0.1 | 3.5 ± 1.0 | 0.046 |
| FVC, % predicted | 60.8 ± 16.1 | 72.5 ± 15.2 | 0.023 |
| RV, L | 3.4 ± 1.0 | 2.9 ± 0.8 | 0.068 |
| RV, % predicted | 196.9 ± 43.2 | 174.3 ± 40.5 | 0.092 |
| Awake SpO2, % | 95.0 ± 1.8 | 96.1 ± 1.6 | 0.021 |
| Awake pO2, mmHg | 72.5 ± 8.4 | 80.8 ± 8.2 | 0.003 |
| Awake pCO2, mmHg | 40.2 ± 2.5 | 38.4 ± 2.7 | 0.051 |
| Awake pH | 7.42 ± 0.02 | 7.41 ± 0.03 | 0.175 |
| Awake HCO3−, mmol/L | 25.7 ± 1.7 | 24.4 ± 1.7 | 0.017 |
Values are mean ± standard deviation
AHI, apnea-hypopnea index; ESS, Epworth Sleepiness Scale; BMI, body mass index; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; HCO3−, bicarbonate; NREM, non-rapid eye movement sleep; ODI, oxygen desaturation index; pCO2, partial pressure of carbon dioxide pressure; pO2, partial pressure of oxygen; RDI, respiratory disturbance index; REM, rapid eye movement sleep; RV, residual volume; SpO2, oxygen saturation; TST, total sleep time; WASO, wake after sleep onset
Bold values denote statistical significance at the p < 0.05 level
Profiles of pwCF with and without daytime sleepiness
The proportion of adult pwCF with an ESS score > 10, indicating EDS, was 15% (Table 5). The only significant difference between pwCF with and without EDS was for BMI (p = 0.040) (Table 5).
Table 5.
Comparison of clinical characteristics and polysomnographic data in people with cystic fibrosis with or without daytime sleepiness (Epworth Sleepiness Scale score > 10 versus ≤ 10)
| ESS score ≥ 10 (n = 8) | ESS score < 10 (n = 44) | p value | |
|---|---|---|---|
| Age, years | 28.9 ± 4.8 | 31.0 ± 8.4 | 0.718 |
| AHI, events/h | 4.1 ± 3.2 | 4.7 ± 4.2 | 0.833 |
| AHI REM, events/h | 7.7 ± 5.4 | 11.0 ± 10.9 | 0.970 |
| AHI NREM, events/h | 3.2 ± 3.5 | 3.2 ± 3.5 | 0.872 |
| ODI, events/h | 3.7 ± 2.7 | 4.7 ± 4.2 | 0.891 |
| ODI REM, events/h | 7.6 ± 5.4 | 14.4 ± 23.7 | 0.694 |
| ODI NREM, events/h | 2.7 ± 2.8 | 3.1 ± 3.6 | 0.911 |
| Arousal index, events/h | 22.4 ± 8.2 | 18.1 ± 10.6 | 0.292 |
| TST, min | 313.4 ± 29.0 | 297.7 ± 45.3 | 0.276 |
| Sleep efficiency, % | 79.2 ± 8.4 | 74.6 ± 10.4 | 0.245 |
| Sleep latency, min | 51.3 ± 20.7 | 71.2 ± 38.7 | 0.155 |
| WASO, min | 30.8 ± 21.5 | 34.4 ± 29.2 | 0.813 |
| Sleep stages, % TST | |||
| N1 | 2.8 ± 0.9 | 3.1 ± 1.9 | 0.970 |
| N2 | 54.4 ± 5.4 | 50.2 ± 8.1 | 0.167 |
| N3 | 21.9 ± 4.6 | 25.8 ± 9.3 | 0.375 |
| REM | 18.6 ± 5.6 | 18.1 ± 6.6 | 0.833 |
| Nocturnal mean SpO2, % | 92.4 ± 1.9 | 92.0 ± 2.4 | 0.718 |
| Nocturnal minimum SpO2, % | 88.4 ± 3.0 | 86.7 ± 3.9 | 0.187 |
| SpO2 < 90%, % TST | 7.9 ± 21.0 | 14.4 ± 28.3 | 0.133 |
| SpO2 < 90, min | 23.0 ± 60.2 | 44.6 ± 89.5 | 0.097 |
| Nocturnal respiratory rate, breaths/min | 20.5 ± 3.2 | 21.6 ± 4.1 | 0.478 |
| Nocturnal heart rate, beats/min | 60.9 ± 7.0 | 68.5 ± 12.2 | 0.108 |
| BMI, kg/m2 | 23.4 ± 2.6 | 21.1 ± 3.1 | 0.040 |
| FEV1, L | 2.1 ± 0.8 | 2.1 ± 0.8 | 0.990 |
| FEV1, % predicted | 52.0 ± 15.3 | 52.2 ± 14.9 | 0.978 |
| FVC, L | 3.4 ± 1.2 | 3.4 ± 1.0 | 0.897 |
| FVC, % predicted | 70.9 ± 18.8 | 69.3 ± 15.9 | 0.803 |
| RV, L | 2.7 ± 0.6 | 3.1 ± 0.9 | 0.329 |
| RV, % predicted | 172.8 ± 40.5 | 181.3 ± 42.5 | 0.603 |
| Awake SpO2, % | 96.4 ± 1.1 | 95.7 ± 1.7 | 0.276 |
| Awake pO2, mmHg | 78.0 ± 6.1 | 78.9 ± 9.4 | 0.789 |
| Awake pCO2, mmHg | 38.4 ± 2.9 | 29.0 ± 2.8 | 0.591 |
| Awake pH | 7.4 ± 0.02 | 7.4 ± 0.03 | 0.335 |
| Awake HCO3−, mmol/L | 25.1 ± 1.3 | 24.6 ± 1.9 | 0.543 |
Values are mean ± standard deviation
AHI, apnea-hypopnea index; ESS, Epworth Sleepiness Scale; BMI, body mass index; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; HCO3−, bicarbonate; NREM, non-rapid eye movement sleep; ODI, oxygen desaturation index; pCO2, partial pressure of carbon dioxide pressure; pO2, partial pressure of oxygen; RDI, respiratory disturbance index; REM, rapid eye movement sleep; RV, residual volume; SpO2, oxygen saturation; TST, total sleep time; WASO, wake after sleep onset
Bold values denote statistical significance at the p < 0.05 level
Prediction of OSA and sleep hypoxemia
ROC curves and ROC analysis (see Table 6) were performed to compare the accuracy of ppFEV1, age, BMI, ESS score, awake pO2 and SpO2 to predict OSA (see Fig. 1) or nocturnal hypoxemia (see Fig. 2).
Table 6.
Receiver operator characteristics (ROC) analysis for predicting obstructive sleep apnea and nocturnal hypoxemia
| OSA | Nocturnal hypoxemia | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| AUC | p | Cut-off value | Sensitivity (95% CI) | Specificity (95% CI) | AUC | p | Cut-off value | Sensitivity (95% CI) | Specificity (95% CI) | |
| BMI, kg/m2 | 0.53 | 0.737 | > 21 | 47.6 (28.3–67.6) | 51.6 (34.8–68.0) | 0.50 | 0.983 | > 21 | 46.2 (23.2–70.9) | 51.3 (36.2–66.1) |
| FEV1, % predicted | 0.59 | 0.225 | < 49.5 | 57.1 (36.6–75.5) | 64.5 (47.0-79.9) | 0.74 | 0.009 | < 49.5 | 76.9 (49.7–91.8) | 66.7 (51.0-79.4) |
| Awake pO2, mmHg | 0.66 | 0.048 | < 78.5 | 66.7 (45.4–82.8) | 71.0 (53.4–84.0) | 0.76 | 0.006 | < 78.5 | 76.9 (49.7–91.8) | 66.7 (51.0-79.4) |
| Age, years | 0.58 | 0.318 | > 28.5 | 66.7 (45.4–82.1) | 48.4 (32.0-65.2) | 0.65 | 0.120 | > 30.5 | 61.5 (35.5–82.3) | 61.5 (46.0-75.1) |
| ESS score | 0.51 | 0.888 | < 5.5 | 52.4 (32.4–71.7) | 54.8 (37.8–70.9) | 0.58 | 0.404 | < 6.5 | 61.5 (35.5–82.3) | 41.0 (27.1–56.6) |
| Awake SpO2, % | 0.62 | 0.148 | < 96.5 | 66.7 (45.4–82.8) | 45.2 (29.2–62.2) | 0.71 | 0.025 | < 95.5 | 61.5 (35.5–82.3) | 79.5 (64.5–89.2) |
AUC, area under the ROC curve; BMI, body mass index; CI, confidence interval; ESS, Epworth Sleepiness Scale; FEV1, forced expiratory volume in 1 s; OSA, obstructive sleep apnea; pO2, partial pressure of oxygen; SpO2, oxygen saturation
Bold values denote statistical significance at the p < 0.05 level
Fig. 1.
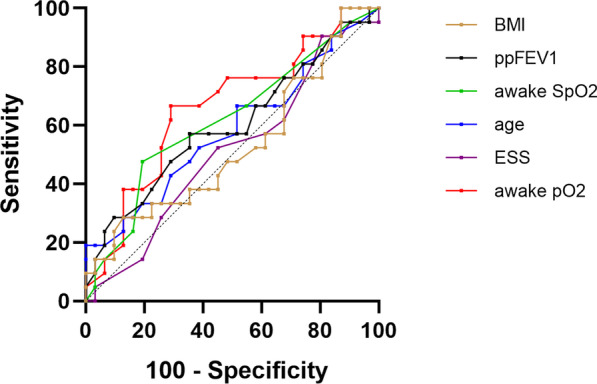
Receiver operating characteristic (ROC) curves for prediction of obstructive sleep apnea using percent predicted forced expiratory volume in 1 s (ppFEV1), body mass index (BMI), age, awake oxygen saturation (SpO2), awake partial pressure of oxygen (pO2) and Epworth Sleepiness Scale (ESS) score
Fig. 2.

Receiver operating characteristic (ROC) curves for prediction of nocturnal hypoxemia using percent predicted forced expiratory volume in 1 s (ppFEV1), body mass index (BMI), age, awake oxygen saturation (SpO2), awake partial pressure of oxygen (pO2) and Epworth Sleepiness Scale (ESS) score
The only significant predictor of OSA was awake pO2 (AUC 0.66, p = 0.048) (Fig. 1; Table 6). The best predictors of nocturnal hypoxemia in this population were pFEV1, awake pO2 and SpO2 (Fig. 2; Table 6). BMI, age and ESS score were not good predictors of nocturnal hypoxemia (Fig. 2; Table 6).
The pO2 cut-off value with the greatest ROC for predicting AHI was < 78.5 mmHg with a sensitivity of 67% and specificity of 71%. For predicting nocturnal hypoxemia, cut-off values were < 49.5 for ppFEV1 (sensitivity 76.9%, specificity 66.7%), < 78.5 mmHg for awake pO2 (sensitivity 76.9%, specificity 66.7%), and < 95.5% for awake SpO2 (sensitivity 61.5%, specificity 79.5%).
Discussion
To the best of our knowledge, this is one of the largest sleep studies evaluating adult pwCF. The main finding of our study is a high prevalence of OSA and nocturnal hypoxemia in this population, and that ability of clinical markers to predict OSA is very limited, whereas the incidence of nocturnal hypoxemia is dependent on lung function parameters and awake oxygenation markers (SpO2 and pO2). EDS with an ESS score > 10 was also a common finding, SDB did not significantly influence the occurrence of EDS.
Although the AHI of the entire population in this study was normal, consistent with existing literature [13, 21, 24, 32], there was a high prevalence of OSA (AHI ≥ 5/h). Comparing our data with the available literature is challenging, because study designs and patient selection are heterogenous with respect to age, lung function, BMI and sample size, and PSG studies examining an exclusively adult CF population are rare [7, 33]. Published data regarding the prevalence of OSA in adult pwCF report a prevalence of 3.2–3.9% [13, 32], substantially lower than the 40% in our study. However, our data are more in line with adult patients with non-CF-bronchiectasis, which showed a similarly high prevalence of OSA [34, 35]. Due to the diversity of studies and patient characteristics, it must be assumed that the prevalence of OSA is often underestimated, and further studies are needed to confirm our findings. This assumption is supported by the fact that the CF population is aging due to improved treatment options and obesity is an increasing problem [4, 36].
Outside the field of CF, the co-existence of OSA and chronic obstructive airflow limitation (OSA-COPD overlap syndrome) has received increasing attention in recent years. The prevalence of OSA-COPD overlap is 10–65%, depending on study design and patient selection [6]. There is growing evidence that patients with OSA-COPD overlap experience more pulmonary exacerbations and have a higher mortality than patients with COPD alone [37, 38]. In addition, patients with OSA-COPD and concomitant bronchiectasis had higher AHI and lower nocturnal oxygen levels than patients with OSA or COPD alone [39]. Comparable data do not exist for pwCF. Whether this knowledge can be transferred to pwCF should be investigated in further longitudinal studies.
Another poorly discussed question regarding OSA in adult pwCF is the role of the upper airways. CF results in impaired mucociliary clearance in the upper respiratory tract as well as the lungs, which may lead to the development of chronic rhinosinusitis (CRS) [40]. CRS may narrow the upper airways and impair breathing through the nose especially during sleep. Combining questionnaires and standard otolaryngology examination, alterations of the upper airways in children and adolescents with CF due to nasal polyposis and chronic infection are related to the development of OSA syndrome [41]. However, radiologic staging of upper airway patency is poorly standardized. In a study by Veronezi et al. [37], the Lund-Mackay score was used to assess upper airways in adolescents and young adults with CF. In their analysis, there was no association between the involvement of upper airways and AHI. Corresponding data for adult pwCF are lacking. The presence of CRS seems does not affect the AHI [42], but patients with CRS objectively report poor sleep quality [43]. Prevalence data on the co-occurrence of CRS and OSA in non-CF patients vary widely, between 15% and 64.7% [44, 45].
Our ROC analysis showed that classical clinical markers such as ppFEV1, BMI, age, and awake pO2 and SpO2 were poor predictors of the AHI. This is another area where data in adult pwCF are rare. In a significantly younger and less affected CF patient group, Veronezi et al. showed that nutritional status, awake SpO2 and daytime sleepiness were closely associated with the presence of OSA [46]. We were not able to confirm these findings in our analysis because we found only a weak association between awake pO2 and the presence of OSA. All other factors, including ESS score, BMI, ppFEV1 and awake SpO2 were not significantly associated with the presence of OSA in adult pwCF in the ROC analysis.
Nocturnal hypoxia is a common finding in pwCF. As with in other sleep studies in pwCF [16, 17, 47, 48], our work also showed a correlation between the severity of lung involvement in CF and the occurrence of nocturnal hypoxia. Detection and correction of nocturnal hypoxia can have a major impact on disease progression and the person’s well-being. Chronic nocturnal hypoxia can cause of sleep disturbance, impaired glucose regulation, decreased quality of life, development of pulmonary hypertension, impaired neurocognitive function, and daytime sleepiness [24, 49, 50]. Therefore, early detection of nocturnal hypoxemia is important. Our data support the findings by others that l awake oxygenation (SpO2 and pO2) seem to be the most important clinical predictors of nocturnal hypoxemia in adult pwCF, whereas ppFEV1 has been shown to be a good predictor of nocturnal hypoxemia in children with CF [13, 15, 48, 51].
Most current data regarding sleep quality and sleep architecture in pwCF come from studies in children and adolescents. Consistent with existing literature in adult pwCF [22, 24, 52, 53], we confirmed that these patients have reduced sleep quality (total sleep time, sleep efficiency) and increased sleep latency and wake after sleep onset. There are multiple potential contributors to impaired subjective and objective sleep in pwCF, including nocturnal coughing, pain, chronic rhinosinusitis, CF-related diabetes, and PEG-feeding [54]. However, sleep architecture (N1, N2, N3 and REM stages) was preserved despite reduced sleep quality. These findings are consistent with data from other studies in children and adult pwCF [13, 19, 23, 53].
Although EDS is one of the main symptoms of OSA, the relationship between AHI and EDS in individuals without CF is inconsistent [55]. This is in line with our findings showing that the overall ESS score was in the normal range even though the prevalence of OSA and nocturnal hypoxia was high. In a study by Bouka et al., clinically stable adult pwCF showed elevated ESS scores compared with healthy individuals, indicating a higher level of daytime sleepiness [20]. In their study, nearly 20% of the examined adult pwCF had an ESS score of > 10, similar to the 15% in our study. In addition, both our study and the one by Bouka et al. reported that the overall ESS score was in the normal range.
In our study, there were no differences between patients with high vs. low ESS scores in terms of SDB, sleep architecture, sleep quality or respiratory markers. This suggests that there must be other factors that influence daytime sleepiness besides sleep and respiratory markers. Depression and anxiety are well known comorbidities in pwCF [56], and these can impact on sleep quality [24, 54]. We can only speculate that there is an association between daytime sleepiness and depression/anxiety in pwCF. However, data from individuals without CF show that the presence of daytime sleepiness is more associated with depression than with SDB [55].
Our study has a number of strengths, including a large number of patients with a wide range of disease severity, but there are also some limitations to note. The main limitation is that we do not have data on nocturnal hypoventilation to provide a complete picture of SDB in adult pwCF. With progression of the disease and a further decline in lung function, nocturnal hypoventilation with consecutive hypercapnia is detectable with possible need for noninvasive ventilation [57]. In this context, nocturnal carbon dioxide levels, preferably measured transcutaneously, are part of a comprehensive sleep assessment. Furthermore, as we know from numerous other studies, there is an association between sleep and HRQoL [49, 54]. However, we did not have HRQoL data for our patients and were therefore unable to investigate associations between HRQoL and objective sleep parameters.
Conclusion
In summary, our data show a high prevalence of OSA, nocturnal hypoxemia and EDS in adult pwCF. OSA and nocturnal hypoxemia were mainly detected in REM sleep. There was no difference between patients with and without OSA (AHI ≥ 5/h) did not differ significantly with respect to age, lung function and weight, but had significantly lower awake oxygen levels. The occurrence and duration of nocturnal hypoxemia were dependent on lung function and awake oxygenation. None of the clinical markers assessed was a significant predictor of OSA, whereas ppFEV1, awake SpO2 and pO2 were good predictors of the occurrence of nocturnal hypoxemia. Neither the presence of OSA nor nocturnal hypoxemia had any influence on the ESS score. Based on our data, we suggest regular PSG screening to detect OSA and nocturnal hypoxemia in adult pwCF, regardless of disease severity. This could help to prevent medical deterioration due to undetected SDB in pwCF.
Acknowledgements
We thank the sleep lab technicians for their excellent support.
Abbreviations
- AHI
Apnea-hypopnea index
- AUC
Area under the curve
- BMI
Body mass index
- CF
Cystic fibrosis
- CFTR
Cystic fibrosis transmembrane conductance regulator
- Cl−
Chloride
- COPD
Chronic obstructive pulmonary disease
- EDS
Excessive daytime sleepiness
- ESS
Epworth Sleepiness Scale
- FEV1
Forced expiratory volume in 1 s
- FVC
Forced vital capacity
- HCO3−
Bicarbonate
- OSA
Obstructive sleep apnea
- PFT
Pulmonary function testing
- pCO2
Partial pressure of carbon dioxide
- pO2
Partial pressure of oxygen
- PSG
Polysomnography
- ppFEV1
Percent predicted forced expiratory volume in 1 s
- pwCF
People with cystic fibrosis
- REM
Rapid eye movement
- ROC
Receiver operator characteristics
- RV
Residual volume
- SDB
Sleep-disordered breathing
- SpO2
Oxygen saturation
- TST
Total sleep time
Author contributions
MW, SDT, CS, SS: Conceptualization, Investigation, Supervision, Validation, Writing – review & editing. TS: Data curation, UN and FEG: Data curation, Writing – review & editing. CT: Supervision, Writing – review & editing. SST: Recruitment, Investigation, Validation. GW: Data curation, Statistics, Writing – review & editing. All authors reviewed and approved the final manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data availability
The data used to support the current findings are available from the corresponding author upon request.
Declarations
Ethics approval and consent to participate
The study was approved by the local ethics committee of the University Hospital Essen (19-8961-BO) and followed the Declaration of Helsinki Ethical Principles for Medical Research Involving Human Subjects. All pwCF provided written informed consent for participation in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no conflicts of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kerem B, Rommens J, Buchanan J, Markiewicz D, Cox T, Chakravarti A, et al. Identification of the cystic fibrosis gene: genetic analysis. Science. 1989;245:1073–80. doi: 10.1126/science.2570460. [DOI] [PubMed] [Google Scholar]
- 2.Shteinberg M, Haq IJ, Polineni D, Davies JC. Cystic fibrosis. The Lancet. 2021;397:2195–211. doi: 10.1016/S0140-6736(20)32542-3. [DOI] [PubMed] [Google Scholar]
- 3.Kerem E, Reisman J, Corey M, Canny GJ, Levison H. Prediction of mortality in patients with cystic fibrosis. N Engl J Med. 1992;326:1187–91. doi: 10.1056/NEJM199204303261804. [DOI] [PubMed] [Google Scholar]
- 4.McKone EF, Ariti C, Jackson A, Zolin A, Carr SB, Orenti A, et al. Survival estimates in european cystic fibrosis patients and the impact of socioeconomic factors: a retrospective registry cohort study. Eur Respir J 2021. doi:10.1183/13993003.02288-2020. [DOI] [PubMed]
- 5.Lurie A, Roche N. Obstructive sleep apnea in patients with chronic obstructive Pulmonary Disease: facts and perspectives. COPD. 2021;18:700–12. doi: 10.1080/15412555.2021.1950663. [DOI] [PubMed] [Google Scholar]
- 6.Brennan M, McDonnell MJ, Walsh SM, Gargoum F, Rutherford R. Review of the prevalence, pathogenesis and management of OSA-COPD overlap. Sleep Breath. 2022 doi: 10.1007/s11325-021-02540-8. [DOI] [PubMed] [Google Scholar]
- 7.Reiter J, Gileles-Hillel A, Cohen-Cymberknoh M, Rosen D, Kerem E, Gozal D, Forno E. Sleep disorders in cystic fibrosis: a systematic review and meta-analysis. Sleep Med Rev. 2020;51:101279. doi: 10.1016/j.smrv.2020.101279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shakkottai A, Nasr SZ, Hassan F, Irani S, O’Brien LM, Chervin RD. Sleep-disordered breathing in cystic fibrosis. Sleep Med. 2020;74:57–65. doi: 10.1016/j.sleep.2020.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lumertz MS, Pinto LA. Sleep-disordered breathing in cystic fibrosis pediatric subjects. Sleep Sci. 2019;12:165–70. doi: 10.5935/1984-0063.20190079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kapur VK, Auckley DH, Chowdhuri S, Kuhlmann DC, Mehra R, Ramar K, Harrod CG. Clinical practice Guideline for Diagnostic Testing for Adult Obstructive Sleep Apnea: an American Academy of Sleep Medicine Clinical Practice Guideline. J Clin Sleep Med. 2017;13:479–504. doi: 10.5664/jcsm.6506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Senaratna CV, Perret JL, Lodge CJ, Lowe AJ, Campbell BE, Matheson MC, et al. Prevalence of obstructive sleep apnea in the general population: a systematic review. Sleep Med Rev. 2017;34:70–81. doi: 10.1016/j.smrv.2016.07.002. [DOI] [PubMed] [Google Scholar]
- 12.Spicuzza L, Sciuto C, Leonardi S, La Rosa M. Early occurrence of obstructive sleep apnea in infants and children with cystic fibrosis. Arch Pediatr Adolesc Med. 2012;166:1165–9. doi: 10.1001/archpediatrics.2012.1177. [DOI] [PubMed] [Google Scholar]
- 13.Perin C, Fagondes SC, Casarotto FC, Pinotti AFF, Menna Barreto SS, Dalcin PdTR. Sleep findings and predictors of sleep desaturation in adult cystic fibrosis patients. Sleep Breath. 2012;16:1041–8. doi: 10.1007/s11325-011-0599-5. [DOI] [PubMed] [Google Scholar]
- 14.Muller NL, Francis PW, Gurwitz D, Levison H, Bryan AC. Mechanism of hemoglobin desaturation during rapid-eye-movement sleep in normal subjects and in patients with cystic fibrosis. Am Rev Respir Dis. 1980;121:463–9. doi: 10.1164/arrd.1980.121.3.463. [DOI] [PubMed] [Google Scholar]
- 15.Isaiah A, Daher A, Sharma PB, Naqvi K, Mitchell RB. Predictors of sleep hypoxemia in children with cystic fibrosis. Pediatr Pulmonol. 2019;54:273–9. doi: 10.1002/ppul.24233. [DOI] [PubMed] [Google Scholar]
- 16.Ramos RTT, Santana MAP, Almeida PdC, MachadoAdS, Araújo-Filho JB, Jr, Salles C. Nocturnal hypoxemia in children and adolescents with cystic fibrosis. J Bras Pneumol. 2013;39:667–74. doi: 10.1590/S1806-37132013000600005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Barbosa RRB, Liberato FMG, de Freitas Coelho P, Vidal PDR, de Carvalho RBCO, Donadio MVF. Sleep-disordered breathing and markers of morbidity in children and adolescents with cystic fibrosis. Pediatr Pulmonol. 2020;55:1974–83. doi: 10.1002/ppul.24780. [DOI] [PubMed] [Google Scholar]
- 18.Milross MA, Piper AJ, Norman M, Becker HF, Willson GN, Grunstein RR, et al. Low-flow oxygen and bilevel ventilatory support: effects on ventilation during sleep in cystic fibrosis. Am J Respir Crit Care Med. 2001;163:129–34. doi: 10.1164/ajrccm.163.1.2005130. [DOI] [PubMed] [Google Scholar]
- 19.Young AC, Wilson JW, Kotsimbos TC, Naughton MT. Randomised placebo controlled trial of non-invasive ventilation for hypercapnia in cystic fibrosis. Thorax. 2008;63:72–7. doi: 10.1136/thx.2007.082602. [DOI] [PubMed] [Google Scholar]
- 20.Bouka A, Tiede H, Liebich L, Dumitrascu R, Hecker C, Reichenberger F, et al. Quality of life in clinically stable adult cystic fibrosis out-patients: Associations with daytime sleepiness and sleep quality. Respir Med. 2012;106:1244–9. doi: 10.1016/j.rmed.2012.06.010. [DOI] [PubMed] [Google Scholar]
- 21.Íscar-Urrutia M, Madrid-Carbajal CJ, Rubinos-Cuadrado G, Fernández-Álvarez R, Vázquez-López MJ, Hernández-González C, et al. Objective and subjective sleep efficiency in adult patients with cystic fibrosis and impact on quality of life. Lung. 2018;196:761–7. doi: 10.1007/s00408-018-0167-x. [DOI] [PubMed] [Google Scholar]
- 22.Shakkottai A, O’Brien LM, Nasr SZ, Chervin RD. Sleep disturbances and their impact in pediatric cystic fibrosis. Sleep Med Rev. 2018;42:100–10. doi: 10.1016/j.smrv.2018.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Forte GC, Barni GC, Perin C, Casarotto FC, Fagondes SC, Dalcin PdTR. Relationship between clinical variables and health-related quality of life in young adult subjects with cystic fibrosis. Respir Care. 2015;60:1459–68. doi: 10.4187/respcare.03665. [DOI] [PubMed] [Google Scholar]
- 24.Dancey DR, Tullis ED, Heslegrave R, Thornley K, Hanly PJ. Sleep quality and daytime function in adults with cystic fibrosis and severe lung disease. Eur Respir J. 2002;19:504–10. doi: 10.1183/09031936.02.00088702. [DOI] [PubMed] [Google Scholar]
- 25.Fraser KL, Tullis DE, Sasson Z, Hyland RH, Thornley KS, Hanly PJ. Pulmonary hypertension and cardiac function in adult cystic fibrosis: role of hypoxemia. Chest. 1999;115:1321–8. doi: 10.1378/chest.115.5.1321. [DOI] [PubMed] [Google Scholar]
- 26.Cox NS, Pepin V, Holland AE. Greater Sleep Fragmentation is Associated with Less Physical activity in adults with cystic fibrosis. J Cardiopulm Rehabil Prev. 2019;39:E11–4. doi: 10.1097/HCR.0000000000000363. [DOI] [PubMed] [Google Scholar]
- 27.Thomas CS, Brown RF. Sleep screening for cystic fibrosis patients: a survey of cystic Fibrosis Programs. Pediatr Pulmonol. 2020 doi: 10.1002/ppul.25051. [DOI] [PubMed] [Google Scholar]
- 28.Berry RB, Brooks R, Gamaldo CE, Harding SM, Lloyd RM, Marcus CL, Vaughn BV. The AASM Manual for the Scoring of Sleep and Associated events: rules, terminology and technical specifications, Version 2.2. www.aasmnet.org. Darien. Illinois: American Academy of Sleep Medicine; 2015. [Google Scholar]
- 29.Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14:540–5. doi: 10.1093/sleep/14.6.540. [DOI] [PubMed] [Google Scholar]
- 30.Miller MR, Hankinson J, Brusasco V, Burgos F, Casaburi R, Coates A, et al. Standardisation of spirometry. Eur Respir J. 2005;26:319–38. doi: 10.1183/09031936.05.00034805. [DOI] [PubMed] [Google Scholar]
- 31.Quanjer PH, Stanojevic S, Cole TJ, Baur X, Hall GL, Culver BH, et al. Multi-ethnic reference values for spirometry for the 3-95-yr age range: the global lung function 2012 equations. Eur Respir J. 2012;40:1324–43. doi: 10.1183/09031936.00080312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Milross MA, Piper AJ, Norman M, Willson GN, Grunstein RR, Sullivan CE, Bye PT. Predicting sleep-disordered breathing in patients with cystic fibrosis. Chest. 2001;120:1239–45. doi: 10.1378/chest.120.4.1239. [DOI] [PubMed] [Google Scholar]
- 33.Jagpal SK, Jobanputra AM, Ahmed OH, Santiago TV, Ramagopal M. Sleep-disordered breathing in cystic fibrosis. Pediatr Pulmonol. 2020 doi: 10.1002/ppul.25028. [DOI] [PubMed] [Google Scholar]
- 34.Faria Júnior NS, Urbano JJ, Santos IR, Silva AS, Perez EA, Souza ÂH, et al. Evaluation of obstructive sleep apnea in non-cystic fibrosis bronchiectasis: a cross-sectional study. PLoS ONE. 2017;12:e0185413. doi: 10.1371/journal.pone.0185413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Borekci S, Hacoğlu Y, Musellim B. Investigation of Obstructive Sleep Apnea in Adult Non-Cystic Fibrosis Bronchiectasis Patients. In: ERS International Congress 2019 abstracts: European Respiratory Society; 09282019. PA631. doi:10.1183/13993003.congress-2019.PA631.
- 36.Kutney KA, Sandouk Z, Desimone M, Moheet A. Obesity in cystic fibrosis. J Clin Transl Endocrinol. 2021;26:100276. doi: 10.1016/j.jcte.2021.100276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Marin JM, Soriano JB, Carrizo SJ, Boldova A, Celli BR. Outcomes in patients with chronic obstructive pulmonary disease and obstructive sleep apnea: the overlap syndrome. Am J Respir Crit Care Med. 2010;182:325–31. doi: 10.1164/rccm.200912-1869OC. [DOI] [PubMed] [Google Scholar]
- 38.Shawon MSR, Perret JL, Senaratna CV, Lodge C, Hamilton GS, Dharmage SC. Current evidence on prevalence and clinical outcomes of co-morbid obstructive sleep apnea and chronic obstructive pulmonary disease: a systematic review. Sleep Med Rev. 2017;32:58–68. doi: 10.1016/j.smrv.2016.02.007. [DOI] [PubMed] [Google Scholar]
- 39.Yang X, Tang X, Cao Y, Dong L, Wang Y, Zhang J, Cao J. The Bronchiectasis in COPD-OSA Overlap Syndrome Patients. Int J Chron Obstruct Pulmon Dis. 2020;15:605–11. doi: 10.2147/COPD.S243429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Krajewska J, Zub K, Słowikowski A, Zatoński T. Chronic rhinosinusitis in cystic fibrosis: a review of therapeutic options. Eur Arch Otorhinolaryngol. 2022;279:1–24. doi: 10.1007/s00405-021-06875-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ramos RTT, Salles C, Gregório PB, Barros AT, Santana A, Araújo-Filho JB, Acosta AX. Evaluation of the upper airway in children and adolescents with cystic fibrosis and obstructive sleep apnea syndrome. Int J Pediatr Otorhinolaryngol. 2009;73:1780–5. doi: 10.1016/j.ijporl.2009.09.037. [DOI] [PubMed] [Google Scholar]
- 42.Mahdavinia M, Hui JW, Zitun M, Lastra A, Herdegen JJ, Codispoti CD, et al. Patients with chronic rhinosinusitis and obstructive sleep apnea have increased paroxysmal limb movement. Am J Rhinol Allergy. 2018;32:94–7. doi: 10.1177/1945892418762843. [DOI] [PubMed] [Google Scholar]
- 43.Alt JA, Smith TL, Mace JC, Soler ZM. Sleep quality and disease severity in patients with chronic rhinosinusitis. Laryngoscope. 2013;123:2364–70. doi: 10.1002/lary.24040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Alt JA, DeConde AS, Mace JC, Steele TO, Orlandi RR, Smith TL. Quality of life in patients with chronic rhinosinusitis and sleep dysfunction undergoing endoscopic sinus surgery: a Pilot Investigation of Comorbid Obstructive Sleep Apnea. JAMA Otolaryngol Head Neck Surg. 2015;141:873–81. doi: 10.1001/jamaoto.2015.1673. [DOI] [PubMed] [Google Scholar]
- 45.Jiang R-S, Liang K-L, Hsin C-H, Su M-C. The impact of chronic rhinosinusitis on sleep-disordered breathing. Rhinology. 2016;54:75–9. doi: 10.4193/Rhino15.204. [DOI] [PubMed] [Google Scholar]
- 46.Veronezi J, Carvalho AP, Ricachinewsky C, Hoffmann A, Kobayashi DY, Piltcher OB, et al. Sleep-disordered breathing in patients with cystic fibrosis. J Bras Pneumol. 2015;41:351–7. doi: 10.1590/S1806-37132015000004468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.van der Giessen L, Bakker M, Joosten K, Hop W, Tiddens H. Nocturnal oxygen saturation in children with stable cystic fibrosis. Pediatr Pulmonol. 2012;47:1123–30. doi: 10.1002/ppul.22537. [DOI] [PubMed] [Google Scholar]
- 48.Castro-Silva C de, Bruin VMS de, Cavalcante AGM, Bittencourt LRA, Bruin PFC de. Nocturnal hypoxia and sleep disturbances in cystic fibrosis. Pediatr Pulmonol. 2009;44:1143–50. doi:10.1002/ppul.21122. [DOI] [PubMed]
- 49.Young AC, Wilson JW, Kotsimbos TC, Naughton MT. The impact of nocturnal oxygen desaturation on quality of life in cystic fibrosis. J Cyst Fibros. 2011;10:100–6. doi: 10.1016/j.jcf.2010.11.001. [DOI] [PubMed] [Google Scholar]
- 50.Suratwala D, Chan JSH, Kelly A, Meltzer LJ, Gallagher PR, Traylor J, et al. Nocturnal saturation and glucose tolerance in children with cystic fibrosis. Thorax. 2011;66:574–8. doi: 10.1136/thx.2010.142141. [DOI] [PubMed] [Google Scholar]
- 51.Frangolias DD, Wilcox PG. Predictability of oxygen desaturation during sleep in patients with cystic fibrosis: clinical, spirometric, and exercise parameters. Chest. 2001;119:434–41. doi: 10.1378/chest.119.2.434. [DOI] [PubMed] [Google Scholar]
- 52.Vandeleur M, Walter LM, Armstrong DS, Robinson P, Nixon GM, Horne RSC. What keeps children with cystic fibrosis awake at night? J Cyst Fibros. 2017 doi: 10.1016/j.jcf.2017.04.012. [DOI] [PubMed] [Google Scholar]
- 53.Reiter J, Breuer O, Cohen-Cymberknoh M, Forno E, Gileles-Hillel A. Sleep in children with cystic fibrosis: more under the covers. Pediatr Pulmonol. 2021 doi: 10.1002/ppul.25462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Vandeleur M, Walter LM, Armstrong DS, Robinson P, Nixon GM, Horne RSC. Quality of life and mood in children with cystic fibrosis: Associations with sleep quality. J Cyst Fibros. 2018;17:811–20. doi: 10.1016/j.jcf.2017.11.021. [DOI] [PubMed] [Google Scholar]
- 55.Bixler EO, Vgontzas AN, Lin H-M, Calhoun SL, Vela-Bueno A, Kales A. Excessive daytime sleepiness in a general population sample: the role of sleep apnea, age, obesity, diabetes, and depression. J Clin Endocrinol Metab. 2005;90:4510–5. doi: 10.1210/jc.2005-0035. [DOI] [PubMed] [Google Scholar]
- 56.Quittner AL, Goldbeck L, Abbott J, Duff A, Lambrecht P, Solé A, et al. Prevalence of depression and anxiety in patients with cystic fibrosis and parent caregivers: results of the International Depression Epidemiological Study across nine countries. Thorax. 2014;69:1090–7. doi: 10.1136/thoraxjnl-2014-205983. [DOI] [PubMed] [Google Scholar]
- 57.Jobanputra A, Jagpal S, Marulanda P, Ramagopal M, Santiago T, Naik S. An overview of noninvasive ventilation in cystic fibrosis. Pediatr Pulmonol. 2021 doi: 10.1002/ppul.25753. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the current findings are available from the corresponding author upon request.


