Abstract
BACKGROUND:
Post-tonsillectomy hemorrhage (PTH) affects around 4% of patients after tonsillectomy. We hypothesized that preoperative thromboelastography (TEG) might identify patients at higher risk of PTH.
OBJECTIVE:
Investigate whether evaluation of coagulation function by preoperative TEG might help to predict PTH after tonsillectomy by coblation tonsillectomy (TE).
DESIGN:
Post-hoc analysis of randomized controlled study.
SETTING:
Otolaryngology Department between January 2017 and August 2019.
PATIENTS AND METHODS:
This post-hoc analysis included adults who underwent coblation TE for benign tonsillar disorders. Routine blood tests and TEG were performed preoperatively. The TEG parameters evaluated included coagulation reaction time (R) and maximum thrombus amplitude (MA).
MAIN OUTCOME MEASURES:
The main outcome was PTH during the 4-week postoperative period.
SAMPLE SIZE AND CHARACTERISTICS:
284
RESULTS:
The 19 patients (6.7%) that experienced PTH had a higher prevalence of diabetes mellitus, lower use of intraoperative suturing, fewer patients with grade I and II tonsillar enlargement, a higher white blood cell count, lower platelet count, lower fibrinogen level, lower R value, and a lower MA value than patients without PTH (all P<.05). Multivariate logistic regression revealed that diabetes mellitus (P<.053), fibrinogen level ≤2.735 g/L (P<.027), R≤6.55 min (P<.011) and MA≤59.15 mm (P<.012) were independently associated with PTH. A regression model incorporating these four factors predicted PTH with a sensitivity of 73.7% and specificity of 83.8%.
CONCLUSION:
Preoperative evaluation of diabetes mellitus history, fibrinogen level, and TEG parameters might help to identify patients at higher risk of PTH after coblation TE.
LIMITATIONS:
Single-center study with a small sample size; possibly underpowered statistically. TEG measurements might not accurately reflect coagulation function, and a validation cohort was unavailable.
CONFLICT OF INTEREST:
None.
CHINESE CLINICAL TRIAL REGISTRY NUMBER OF STUDY USED IN THIS ANALYSIS:
ChiCTR2000032171. http://www.chictr.org.cn/showprojen.aspx?proj=52553
INTRODUCTION
Tonsillectomy (TE) is used in the management of chronic tonsillitis, benign tonsillar tumors, and obstructive sleep apnea-hypopnea syndrome caused by tonsillar hypertrophy.1,2 Coblation TE involves the passing of radiofrequency energy through a conductive medium (e.g., saline) to generate a plasma field that breaks tissue bonds and coagulates blood vessels at much lower temperatures (40-70°C) than electrosurgery (>100°C).3 Coblation TE has advantages over conventional tonsillectomy, including a shorter operation time, less intraoperative blood loss and tissue injury, less postoperative pain, earlier restoration of normal diet and activities, and lower recurrence rate.3-7
Severe complications are uncommon after tonsillectomy but include post-tonsillectomy hemorrhage (PTH), infection, and excessive wound scarring. PTH affects ∼4% of patients after coblation TE.8 Furthermore, PTH incidence may be higher for coblation TE than for conventional TE,3,6,9 although not all studies support that finding.10,11 Risk factors for PTH include age ≥12 years or ≥15 years (depending on the study), male sex, operator experience, and coblation TE as the technique.12,13 The development of a method to evaluate PTH risk preoperatively would help identify patients requiring closer postoperative monitoring and facilitate earlier intervention if PTH were to occur.
Thromboelastography (TEG) can identify individuals with hypercoagulable blood and facilitate hemostasis and transfusion management in major surgery and trauma.14 TEG provides a comprehensive functional analysis of coagulation factors, fibrinogen, platelets, and fibrinolytic processes15 and is considered a useful tool for the perioperative monitoring of coagulation function in surgical patients.16 TEG measures several coagulation-related features such as the velocity of blood clot formation and blood clot stability and amplitude. TEG yields information regarding risk factors for bleeding that is not provided by conventional tests of coagulation factor levels.17 Furthermore, the sensitivity and specificity of TEG for bleeding and thrombogenesis are higher than conventional tests of coagulation function.18 TEG has been applied as a point-of-care viscoelastic test of hemostasis in various types of surgery.19
We hypothesized that preoperative TEG might identify patients at higher risk of PTH. Therefore, the aim of this study was to evaluate whether parameters measured by preoperative TEG were associated with PTH in patients who underwent coblation TE.
PATIENTS AND METHODS
This article documents a post-hoc analysis of a previous randomized controlled study assessing whether suture of the palatopharyngeal arch, palatoglossal arch, and tonsillar fossa reduced PTH incidence after coblation TE.20 This post-hoc analysis included adult patients who underwent coblation TE for benign disorders, including chronic tonsillitis, benign tonsillar tumor, and tonsillar hypertrophy, at the Otolaryngology Department, between January 2017 and August 2019. The Ethics Committee approved this study. The original study20 had been approved by the Ethics Committee of our hospital (No. LL-KY-2020035), and all patients had provided informed written consent. The original trial was registered on the Chinese Clinical Trial Registry as ChiCTR2000032171.
The inclusion criteria were age ≥18 years old, diagnosis of chronic tonsillitis, tonsillar hypertrophy, or benign tonsil mass according to postoperative pathology, and being scheduled to undergo coblation TE. The exclusion criteria were pregnancy or menstruation, acute tonsillitis or pharyngeal inflammation during the previous 2 weeks (based on the inquiry of medical history, clinical examination, and laboratory blood routine test), use of aspirin during the week before surgery, or personal or family history of coagulation disorders. The withdrawal criteria were diagnosis of malignant tumor according to postoperative pathology or failure to complete follow-up.
Surgical methods
The operations were performed by the same surgeon, who had >10 years of professional experience. Surgery was performed under orotracheal intubation and intravenous combined anesthesia with the patient in the supine position. The oropharynx was exposed with a mouth gag, and coblation TE was performed using a G33E51 disposable scalpel low-temperature plasma surgical system (Jiexiehuizhong Company, Beijing, China). The energy power was set at level 7 for intraoperative resection and level 3 for hemostasis. A nasal endoscope was used for illumination and visualization. For patients who did not undergo suture, the plasma knife was positioned close to the tonsillar capsule to cut the mucosa, and the tonsil was completely resected from the superior pole to the inferior pole. Subsequently, a nasal endoscope was used to carefully examine the wound surface, and the surgical cavity was rinsed with normal saline after hemostasis had been achieved with a plasma knife. Surgery was completed after re-examination of the wound surface to exclude bleeding.
For patients who underwent suture, the tonsil was removed as described above, and the palatine arch and extra-capsular tissues were protected during surgery to avoid substantial injury. Once hemostasis had been achieved, Vicryl 3-0 thread was used for three-spicule interrupted suture of the full-layer tissues of the palatopharyngeal arch, palatoglossal arch, and tonsillar fossa from the inferior pole to the superior pole of the tonsillar fossa. Then, the surgical cavity was rinsed with normal saline and carefully examined to exclude bleeding.
All patients were fasted for 6 h after surgery and then given a soft diet for 2 weeks. Patients were recommended to use mouthwash regularly and painkillers or antibiotics as needed. All patients were observed in the hospital for 1 week after surgery and followed up at the outpatient department once weekly for 3 weeks after discharge. Wound bleeding status and the growth and fall-off of the pseudo-membrane on the tonsillar fossa wound were evaluated at each follow-up visit.
Investigations and outcome measures
Routine blood tests and coagulation function tests, including red blood cell count (RBC; reference range, 4.0-5.5×1012/L), white blood cell count (WBC; reference range, 4-10×109/L), platelet count (PLT; reference range, 100-300×109/L), hemoglobin concentration (reference range, 120-160 g/L) and fibrinogen level (reference range, 2-4 g/L), were performed preoperatively.
The TEG parameters evaluated preoperatively, within 3 days before surgery, were coagulation reaction time (R), the kinetics of clot development (K), alpha angle (α), and maximum thrombus amplitude (MA) (Figure 1). R (time to initiation of coagulation; reference range, 5-10 min) was measured as the latency from the start of the test to the first sign of clot formation and reflects the functions of coagulation factors. K (time to blood clot formation; reference range, 1-3 min) was measured as the time taken from the end of R to reach an amplitude of 20 mm and reflects fibrinogen function. α (velocity of blot clot aggregation; reference range, 53-72°) was measured as the angle between the tangent to the maximum radian of the thromboelastogram curve and the horizontal and is influenced by the fibrinogen level. MA (maximal strength of the clot; reference range, 50-70 mm) was measured as the largest amplitude of the trace and reflected fibrinogen and platelet function. The main outcome was PTH. PTH was defined as any bloody secretions appearing in the surgical cavity within 4 weeks after surgery.20 As a secondary outcome, pain intensity during the first 3 days after surgery was evaluated using a visual analog scale.
Figure 1.
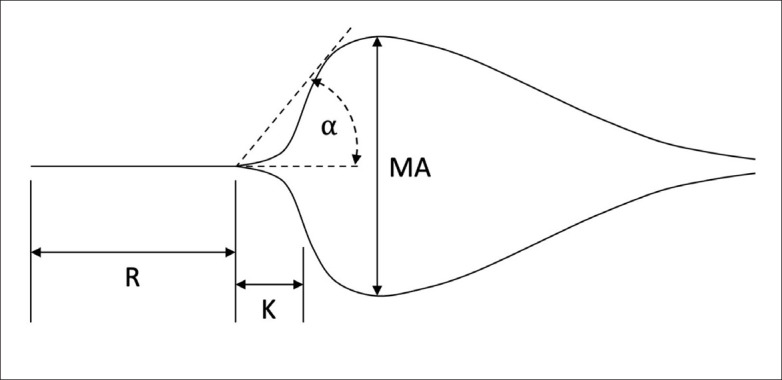
Schematic diagram illustrating the measurement of thromboelastography parameters from a thromboelastogram. R: coagulation reaction time; K: kinetics of clot development; α: alpha angle; MA: maximum thrombus amplitude.
Statistical analysis
IBM SPSS version 22.0 (IBM, Armonk, NY, USA) was used for the post-hoc analysis. PTH patients were compared with non-PTH patients. Normally distributed quantitative data are described as mean and standard deviation and were compared between groups using the t test for independent samples. Non-normally distributed quantitative data are described as median (range) and were compared between groups using the Mann-Whitney U test. Qualitative data are expressed as n (%) and were compared between groups using the chi-squared test or the Fisher exact test (expected frequency <5). Receiver operating characteristic (ROC) curve analyses were performed to assess the association of various parameters with PTH. The area under the ROC curve (AUC), optimal cut-off value (based on the Youden index), sensitivity, and specificity were calculated. Univariate and multivariate logistic regression analyses with forward (likelihood ratio) selection were used to identify factors associated with PTH. Odds ratios (OR) and 95% confidential intervals (95% CIs) were calculated. P<.1 was considered statistically significant.
RESULTS
Nineteen of the 284 patients (6.7%) experienced PTH. There were no significant differences between the PTH and non-PTH patients in gender, age, disease course, body mass index, smoking status, alcohol consumption, the prevalence of hypertension, medical history other than hypertension/diabetes mellitus (DM), or indication for coblation TE (Table 1). However, the PTH patients had a higher prevalence of DM (P=.048), lower use of intraoperative suturing (P=.009), and fewer patients with grade I/II tonsillar enlargement (P=.027) than the non-PTH patients. None of the patients received preoperative drugs that could affect coagulation (e.g., tranexamic acid). The PTH patients had a significantly higher WBC (P=.042), lower PLT (P=.065), lower fibrinogen level (P=.063), lower R (P=.014) and lower MA (P=.031) than the non-PTH patients (Table 2). There were no significant differences between groups in RBC, hemoglobin level, K or angle α the PTH patients had significantly higher pain scores on postoperative day 1 P=.029), postoperative day 2 (P=.009) and postoperative day 3 (P=.033).
Table 1.
Baseline clinical characteristics of the study participants.
| Characteristic | Posttonsillectomy hemorrhage (n=19) | No post-tonsillectomy hemorrhage (n=265) | P |
|---|---|---|---|
| Age (years) | 43 (26–57) | 44 (18–71) | .606 |
| Gender | |||
| Female | 10 (52.6) | 121 (45.7) | .556 |
| Male | 9 (47.4) | 144 (54.3) | |
| Disease course (years) | 6 (2–11) | 6 (1–15) | .966 |
| Body mass index (kg/m2) | 21.6 (19.0–25.0) | 21.6 (19.0–26.0) | .434 |
| Current smoker | 9 (47.4) | 89 (33.6) | .222 |
| Alcohol consumption | 1 (5.3) | 26 (9.8) | .999 |
| Hypertension | 5 (26.3) | 36 (13.6) | .167 |
| Diabetes mellitus | 4 (21.1) | 18 (6.8) | .048 |
| Other disease history | 2 (10.5) | 4 (1.5) | .054 |
| Intraoperative suture | |||
| Yes | 4 (21.1) | 138 (52.1) | .009 |
| No | 15 (78.9) | 127 (47.9) | |
| Diagnosis | |||
| Chronic tonsillitis | 15 (78.9) | 194 (73.2) | .828 |
| Tonsillar hypertrophy | 4 (21.1) | 55 (20.8) | |
| Benign tumor of the tonsila | 0 | 16 (6.0) | |
| Degree of tonsillar enlargement | |||
| I | 2 (10.5)b | 89 (33.6) | .027 |
| II | 16 (84.2)b | 139 (52.5) | |
| III | 1 (5.3) | 37 (14.0) | |
Data are expressed as median (range) or n (%). Post-tonsillectomy hemorrhage.
Including tonsillar cysts, polyps and papillomas.
Represented a significant difference between PTH group and non-PTH group.
Table 2.
Comparison of preoperative blood test results, preoperative thromboelastography parameters and postoperative pain intensity between patients with and without post-tonsillectomy hemorrhage.
| Parameter | Posttonsillectomy hemorrhage (n=19) | No post-tonsillectomy hemorrhage (n=265) | P |
|---|---|---|---|
| Blood test results | |||
| Red blood cell count (×1012/L) | 4.62 (3.66–5.79) | 4.64 (3.27–5.96) | .706 |
| White blood cell count (×109/L) | 6.76 (4.21–10.92) | 5.97 (3.1–11.79) | .042 |
| Hemoglobin (g/L) | 131.88 (99.26–170.11) | 136.23 (83.59–206.85) | .786 |
| Platelet count (×109/L) | 161.18 (104.23–295.26) | 172.82 (99.88–354.37) | .065 |
| Fibrinogen (g/L) | 2.33 (2.05–4.09) | 2.65 (1.94–4.19) | .063 |
| Thromboelastography parameters | |||
| Coagulation reaction time (R)(min) | 5.70 (5.20–9.60) | 6.50 (5.00–9.20) | .014 |
| Kinetics of clot development (K)(min) | 1.70 (1.20–2.60) | 1.70 (1.00–2.90) | .767 |
| α angle (°) | 61.20 (52.20–71.40) | 62.30 (51.00–71.90) | .473 |
| Maximum thrombus amplitude (mm) | 57.10 (51.20–65.10) | 59.60 (50.10–71.60) | .031 |
| Postoperative pain | |||
| Day 1 | 3.02 (1.00–4.00) | 2.48 (1.00–4.00) | .029 |
| Day 2 | 6.30 (4.00–8.00) | 5.56 (2.00–8.00) | .009 |
| Day 3 | 4.19 (1.00–6.00) | 3.01 (1.00–7.00) | .033 |
Data are expressed as median (range).
ROC curve analyses established optimal cut-off values for WBC, PLT, fibrinogen level, R, and MA (Table 3). However, the AUC values for all these parameters were <0.7, suggesting that these indicators have poor predictive value when used alone.
Table 3.
Receiver operating characteristic curve analyses.
| AUC | SE | P | 95% CI | Cut-off | Youden index | Sensitivity | Specificity | |
|---|---|---|---|---|---|---|---|---|
| White blood cell count | 0.639 | 0.064 | .042 | 0.514-0.765 | 6.485 | 0.303 | 68.4% | 61.9% |
| Platelet count | 0.627 | 0.065 | .065 | 0.492-0.762 | 135.9 | 0.247 | 42.1% | 82.6% |
| Fibrinogen | 0.628 | 0.071 | .063 | 0.488-0.767 | 2.735 | 0.302 | 84.2% | 46.0% |
| Coagulation reaction time (R) | 0.669 | 0.056 | .014 | 0.559-0.779 | 6.55 | 0.359 | 89.5% | 46.4% |
| Maximum thrombus amplitude (MA) | 0.648 | 0.056 | .031 | 0.538-0.758 | 59.15 | 0.347 | 78.9% | 55.8% |
AUC: area under the receiver operating characteristic curve; SE: standard error
Univariate logistic regression indicated that history of DM, intraoperative suturing, tonsillar enlargement grade, WBC, PLT, fibrinogen level, R, and MA were associated with PTH (Table 4). Multivariate logistic regression revealed that history of DM (P=.053), fibrinogen level ≤2.735 g/L (P=.027), R≤6.55 min (P=.011) and MA ≤59.15 mm (P=.012) were independently associated with PTH (Table 4).
Table 4.
Univariate and multivariate logistic regression analyses of factors associated with post-tonsillectomy hemorrhage (n=284).
| Univariate analysis | Multivariate analysis | ||||||
|---|---|---|---|---|---|---|---|
| Odds ratio | 95% CI | P | Odds ratio | 95% CI | P | ||
| Diabetes | Yes vs. No | 3.659 | 1.100–12.177 | .034 | 3.707 | 0.984–13.967 | .053 |
| Suture | Yes vs. No | 0.245 | 0.079–0.759 | .015 | |||
| Tonsil enlargement | II/III vs. I | 4.298 | 0.971–19.018 | .055 | |||
| Blood parameters | |||||||
| White blood cell count (×109/L) | >6.485 vs. ≤6.485 | 3.518 | 1.296–9.55 | .014 | |||
| Platelet count (×109/L) | ≤135.9 vs. >135.9 | 3.462 | 1.32–9.085 | .012 | |||
| Fibrinogen (g/L) | ≤2.735 vs. >2.735 | 4.55 | 1.295–15.986 | .018 | 4.375 | 1.185–16.16 | .027 |
| Thromboelastography parameters | |||||||
| Coagulation (min) | ≤6.55 vs. >6.55 | 7.363 | 1.668–32.503 | .008 | 7.036 | 1.551–31.923 | .011 |
| Maximum thrombus amplitude (mm) | ≤59.15 vs. >59.15 | 4.744 | 1.533–14.674 | .007 | 4.5 | 1.394–14.53 | .012 |
Model summary measures: deviance: 13.0, Nagelkerke R square: 0.488, Overall model test: .098).
Diagnostic accuracy data was generated based on the factors identified in the multivariate analysis (history of DM, fibrinogen level ≤2.735 g/L, R ≤6.55 min, and MA ≤59.15 mm). ROC curve analysis (Figure 2) showed that the AUC of the four-parameter model was 0.828 (95% CI=0.746-0.911), indicating that the model had a good predictive ability. At an optimal cut-off value of 0.151, the sensitivity and specificity were 73.7% and 83.8%, respectively.
Figure 2.
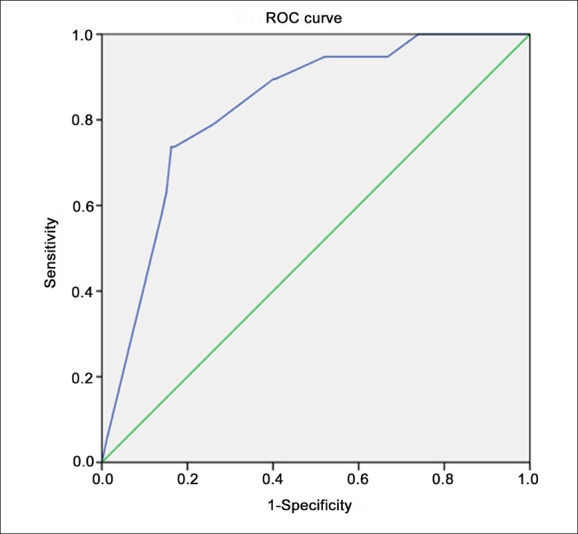
Receiver operating characteristic curve for the prediction of post-tonsillectomy hemorrhage by a regression model that included four factors: history of diabetes mellitus, preoperative fibrinogen level ≤2.735 g/L, preoperative coagulation reaction time (R) ≤6.55 min and preoperative maximum thrombus amplitude (MA) ≤59.15 mm. Diagonal segments were produced by ties.
DISCUSSION
PTH occurred in 6.7% (19/284) of adults who underwent coblation TE for benign tonsillar disorders. Multivariate logistic regression revealed that a history of DM, fibrinogen level ≤2.735 g/L, R≤6.55 min, and MA ≤59.15 mm were independently associated with PTH. A regression model incorporating these four parameters predicted PTH with an AUC of 0.828, a sensitivity of 73.7%, and a specificity of 83.8%. Our findings indicate that preoperative evaluation of DM history, fibrinogen level, and TEG parameters might help identify patients at higher risk of postoperative hemorrhage after coblation TE.
The incidence of PTH in this study is consistent with previously reported incidences of 0.3%-10% after tonsillectomy.8,21,22 Coblation TE has several advantages over conventional tonsillectomy including minimal invasiveness, clearer surgical field, reduced tissue injury and less intraoperative blood loss.6 However, the incidence of PTH, especially secondary PTH, may be higher for coblation TE than for conventional tonsillectomy,3,6,9,23 although not all investigators agree.10,11 Postoperative hemorrhage often occurs during the night24 and at about 7 days after surgery, which is usually after discharge from the hospital. Therefore, PTH can catch clinicians and patients unprepared, which increases the risks to patients. Identifying risk factors for PTH would help to screen out patients requiring closer observation.
Our multivariate analysis identified four factors independently associated with PTH: a history of DM, fibrinogen level ≤2.735 g/L, R≤6.55 min, and MA ≤59.15 mm. The latter three parameters directly reflect the function of the coagulation system. A previous investigation reported that PTH occurrence was associated with hyperactivation of fibrinolysis, which might inhibit blood clot formation in the wound and cause clots to dissolve more rapidly.25 In agreement with our finding, prior studies have demonstrated that a low plasma fibrinogen level may be associated with PTH.26,27 Although not all investigators believe that a comprehensive assessment of preoperative coagulation function can identify patients at risk of PTH,28,29 this may reflect the limitations of conventional tests of coagulation factor levels. TEG is thought to provide more detailed information about risk factors for bleeding than conventional tests of coagulation.17,18 TEG has been used to evaluate hemorrhage risk after knee surgery,30 pediatric cardiac surgery,31 and other procedures.19 We found that R≤6.55 min and MA ≤59.15 mm were independently associated with increased PTH risk. R measures the time to initiation of coagulation and reflects the functions of various coagulation factors, while MA measures maximal clot strength and reflects the integrated functions of fibrinogen and platelets. A recent pilot study suggested that TEG might be a useful coagulation test in patients undergoing invasive interventions, including tonsillectomy.32 Nevertheless, our study is the first to show that preoperative TEG parameters potentially could be used to facilitate PTH risk prediction in adults undergoing coblation TE. Indeed, a regression model incorporating a history of DM, preoperative fibrinogen level, R, and MA predicted PTH with an AUC of 0.828, a sensitivity of 73.7%, and a specificity of 83.8%.
Intraoperative suture to close damaged blood vessels is an effective method of hemostasis. All tonsillectomy techniques predispose to bleeding by damaging tissues and blood vessels in the tonsillar fossa.33 Postoperatively, a protective pseudo-membrane forms over the wound and then gradually falls off during recovery. However, wound infection, early intake of solid/hot food, and frequent coughing can induce pseudo-membrane instability and hemorrhage.34 Some patients in this study underwent intraoperative suturing to close the wound in the tonsillar fossa with the aim of reducing wound exposure to food, infection risk, and postoperative pain. Previously published data in this cohort showed that intraoperative suture reduced secondary PTH incidence and PTH severity, with no patients in the suture group experiencing primary PTH.20 Therefore, intraoperative suturing may help reduce PTH incidence, especially in patients at higher risk of hemorrhage after coblation TE.
TEG is widely used in emergency trauma, intensive care, gynecology, obstetrics, cardiovascular surgery, hepatobiliary surgery, and other surgical departments. As the main means to detect the coagulation function of perioperative patients, it is accepted and applied by an increasing number of clinicians because of its simple application, intuitive detection parameters, and graphics, and can simulate the whole process from the formation of blood clots to the dissolution of blood clots. This technology is the same as a routine blood test. In China, there are unified normal value standards for testing, and there are regular calibration requirements. Still, whether the values and threshold are the same across ethnicities is unknown and will be to be examined.
This study has limitations. This was a single-center study, and all operations were performed by the same surgeon. Hence, external validity is limited, and the generalizability of the findings remains unknown. Second, the sample size was quite small, so the study may have been insufficiently powered to detect some real differences between groups. Patients with obstructive sleep apnea were excluded because they also have abnormalities in coagulation, weight, blood pressure, cardiac function, respiratory status, and blood lipids. In addition, they often undergo soft palate ablation, while the present study only included TE. Third, TEG was conducted within 3 days before surgery but not on the day of surgery. Therefore, the TEG measurements might not reflect coagulation function on the day of surgery. Fourth, the performance of our four-factor regression model was not assessed in a validation cohort. Fifth, the PT and APTT of the patients were detected in the preoperative routine coagulation function test, and the normal values were confirmed before they were included in the study groups. Because the PT and APTT values of the patients were normal, we did not carry out a correlation analysis between them and TEG detection parameters. Some parameters like AEC and CRP were not measured. Finally, the study aimed to examine preoperative parameters that could help predict PTH after coblation TE, but the surgery itself will introduce factors that can change the patient's coagulation status and hence the risk of PTH, especially considering that the recovery period after TE is 2 weeks. Future studies could also include intraoperative or immediately postoperative parameters. Preoperative evaluation of DM history, fibrinogen level, and TEG parameters might help to identify patients at risk of PTH after coblation TE. Additionally, intraoperative suturing may help to reduce PTH incidence, particularly in patients at higher risk of PTH.
ACKNOWLEDGMENTS
We would like to thank the library of our Hospital for help with data analysis.
Funding Statement
Funding: This work was supported by the Basic Research Project of the Shenzhen Knowledge Innovation Program (grant number: JCYJ20170307095642983).
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study were included in this published article.
AUTHORS' CONTRIBUTIONS
Qian Liu and Yanping Zhang conceived and supervised the study; Qian Liu and Yanping Zhang designed experiments; Qian Liu performed experiments; Qian Liu provided new tools and reagents; Qian Liu developed new software and performed simulation studies; Qian Liu and Yanlu Lyu analyzed data; Qian Liu, Yanping Zhang and Yanlu Lyu wrote the manuscript; Qian Liu made manuscript revisions. All authors reviewed the results and approved the final version of the manuscript.
REFERENCES
- 1.Mitchell RB, Archer SM, Ishman SL, Rosenfeld RM, Coles S, Finestone SA, et al. Clinical Practice Guideline: Tonsillectomy in Children (Update)-Executive Summary. Otolaryngol Head Neck Surg. 2019; 160 (2):187-205. [DOI] [PubMed] [Google Scholar]
- 2.Isaacson G. Pediatric tonsillectomy: an evidence-based approach. Otolaryngol Clin North Am. 2014; 47 (5):673-90. [DOI] [PubMed] [Google Scholar]
- 3.Pynnonen M, Brinkmeier JV, Thorne MC, Chong LY, Burton MJ. Coblation versus other surgical techniques for tonsillectomy. Cochrane Database Syst Rev. 2017; 8 (8):Cd004619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zuo HX, Niu YM, Zhang C, Huang JJ, Wang L, Du SM. [The clinical efficacy of coblation tonsillectomy and conventional tonsillectomy in China: A Meta analysis]. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2018; 32 (4):269-78. [DOI] [PubMed] [Google Scholar]
- 5.Windfuhr JP, Chen YS, Propst EJ, Güldner C. The effect of dexamethasone on post-tonsillectomy nausea, vomiting and bleeding. Braz J Otorhinolaryngol. 2011; 77 (3):373-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.El-Taher M, Aref Z. Coblation Versus Conventional Tonsillectomy: A Double Blind Randomized Controlled Trial. Indian J Otolaryngol Head Neck Surg. 2019; 71 (Suppl 1):172-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Izny Hafiz Z, Rosdan S, Mohd Khairi MD. Coblation tonsillectomy versus dissection tonsillectomy: a comparison of intraoperative time, intraoperative blood loss and postoperative pain. Med J Malaysia. 2014; 69 (2):74-8. [PubMed] [Google Scholar]
- 8.Mösges R, Hellmich M, Allekotte S, Albrecht K, Böhm M. Hemorrhage rate after coblation tonsillectomy: a meta-analysis of published trials. Eur Arch Otorhinolaryngol. 2011; 268 (6):807-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Betancourt AR, López C, Zerpa V, Carrasco M, Dalmau J. Does surgical technique influence post-tonsillectomy haemorrhage? Our experience. Acta Otorrinolaringol Esp. 2015; 66 (4):218-23. [DOI] [PubMed] [Google Scholar]
- 10.Belloso A, Chidambaram A, Morar P, Timms MS. Coblation tonsillectomy versus dissection tonsillectomy: postoperative hemorrhage. Laryngoscope. 2003; 113 (11):2010-3. [DOI] [PubMed] [Google Scholar]
- 11.Reusser NM, Bender RW, Agrawal NA, Albright JT, Duncan NO, Edmonds JL. Post-tonsillectomy hemorrhage rates in children compared by surgical technique. Ear Nose Throat J. 2017; 96 (7):E7-e11. [DOI] [PubMed] [Google Scholar]
- 12.Tomkinson A, Harrison W, Owens D, Harris S, McClure V, Temple M. Risk factors for postoperative hemorrhage following tonsillectomy. Laryngoscope. 2011; 121 (2):279-88. [DOI] [PubMed] [Google Scholar]
- 13.Heidemann CH, Wallén M, Aakesson M, Skov P, Kjeldsen AD, Godballe C. Post-tonsillectomy hemorrhage: assessment of risk factors with special attention to introduction of coblation technique. Eur Arch Otorhinolaryngol. 2009; 266 (7):1011-5. [DOI] [PubMed] [Google Scholar]
- 14.Bischof D, Dalbert S, Zollinger A, Ganter MT, Hofer CK. Thrombelastography in the surgical patient. Minerva Anestesiol. 2010; 76 (2):131-7. [PubMed] [Google Scholar]
- 15.Kim E, Shim HS, Kim WH, Lee SY, Park SK, Yang JH, et al. Predictive Value of Intraoperative Thromboelastometry for the Risk of Perioperative Excessive Blood Loss in Infants and Children Undergoing Congenital Cardiac Surgery: A Retrospective Analysis. J Cardiothorac Vasc Anesth. 2016; 30 (5):1172-8. [DOI] [PubMed] [Google Scholar]
- 16.Meesters MI, Burtman D, van de Ven PM, Boer C. Prediction of Postoperative Blood Loss Using Thromboelastometry in Adult Cardiac Surgery: Cohort Study and Systematic Review. J Cardiothorac Vasc Anesth. 2018; 32 (1):141-50. [DOI] [PubMed] [Google Scholar]
- 17.Gerlinger I, Török L, Nagy A, Patzkó A, Losonczy H, Pytel J. [Frequency of coagulopathies in cases with post-tonsillectomy bleeding]. Orv Hetil. 2008; 149 (10):441-6. [DOI] [PubMed] [Google Scholar]
- 18.Spiezia L, Vasques F, Behr A, Campello E, Maggiolo S, Berizzi A, et al. Perioperative coagulation assessment of patients undergoing major elective orthopedic surgery. Intern Emerg Med. 2016; 11 (6):793-801. [DOI] [PubMed] [Google Scholar]
- 19.Whiting D, DiNardo JA. TEG and ROTEM: technology and clinical applications. Am J Hematol. 2014; 89 (2):228-32. [DOI] [PubMed] [Google Scholar]
- 20.Liu Q, Zhang Y, Lyu Y. Postoperative hemorrhage following coblation tonsillectomy with and without suture: A randomized study in Chinese adults. Am J Otolaryngol. 2021; 42 (1):102760. [DOI] [PubMed] [Google Scholar]
- 21.Söderman AC, Odhagen E, Ericsson E, Hemlin C, Hultcrantz E, Sunnergren O, et al. Post-tonsillectomy haemorrhage rates are related to technique for dissection and for haemostasis. An analysis of 15734 patients in the National Tonsil Surgery Register in Sweden. Clin Otolaryngol. 2015; 40 (3):248-54. [DOI] [PubMed] [Google Scholar]
- 22.Ikoma R, Sakane S, Niwa K, Kanetaka S, Kawano T, Oridate N. Risk factors for post-tonsillectomy hemorrhage. Auris Nasus Larynx. 2014; 41 (4):376-9. [DOI] [PubMed] [Google Scholar]
- 23.Kim JW, Mun SJ, Lee WH, Mo JH. Post-tonsillectomy hemorrhage in children: a single surgeon's experience with coblation compared to diathermy. Eur Arch Otorhinolaryngol. 2013; 270 (1):339-44. [DOI] [PubMed] [Google Scholar]
- 24.Kim SJ, Walsh J, Tunkel DE, Boss EF, Ryan M, Lee AH. Frequency of post-tonsillectomy hemorrhage relative to time of day. Laryngoscope. 2020; 130 (7):1823-27. [DOI] [PubMed] [Google Scholar]
- 25.Bitar M, Dunya G, Khalifee E, Muwakkit S, Barazi R. Risk of postoperative hemorrhage after adenoidectomy and tonsillectomy: Value of the preoperative determination of partial thromboplastin time and prothrombin time. Int J Pediatr Otorhinolaryngol. 2019; 116:62-64. [DOI] [PubMed] [Google Scholar]
- 26.Aoki M, Osaka Y, Ando K, Morita Y. Anesthesia Experience for Tonsillectomy in a Patient With Hypofibrinogenemia: A Case Report. A A Pract. 2019; 12 (8):259-60. [DOI] [PubMed] [Google Scholar]
- 27.Kontorinis G, Schwab B. Significance of advanced haemostasis investigation in recurrent, severe post-tonsillectomy bleeding. J Laryngol Otol. 2011; 125 (9):952-7. [DOI] [PubMed] [Google Scholar]
- 28.Scheckenbach K, Bier H, Hoffmann TK, Windfuhr JP, Bas M, Laws HJ, et al.. [Risk of hemorrhage after adenoidectomy and tonsillectomy. Value of the preoperative determination of partial thromboplastin time, prothrombin time and platelet count]. Hno. 2008; 56 (3):312-20. [DOI] [PubMed] [Google Scholar]
- 29.Howells RC, 2nd, Wax MK, Ramadan HH. Value of preoperative prothrombin time/partial thromboplastin time as a predictor of postoperative hemorrhage in pediatric patients undergoing tonsillectomy. Otolaryngol Head Neck Surg. 1997; 117 (6):628-32. [DOI] [PubMed] [Google Scholar]
- 30.Wang J, Zhu HL, Shi ZJ, Zhang Y. The Application of Thromboelastography in Understanding and Management of Ecchymosis After Total Knee Arthroplasty. J Arthroplasty. 2018; 33 (12):3754-58. [DOI] [PubMed] [Google Scholar]
- 31.Emani S, Sleeper LA, Faraoni D, Mulone M, Diallo F, DiNardo JA, et al. Thromboelastography Is Associated With Surrogates for Bleeding After Pediatric Cardiac Operations. Ann Thorac Surg. 2018; 106 (3):799-806. [DOI] [PubMed] [Google Scholar]
- 32.Durila M, Jonas J, Durilova M, Rygl M, Skrivan J, Vymazal T. Thromboelastometry as an Alternative Method for Coagulation Assessment in Pediatric Patients Undergoing Invasive Procedures: A Pilot Study. Eur J Pediatr Surg. 2019; 29 (3):298-301. [DOI] [PubMed] [Google Scholar]
- 33.Wulu JA, Chua M, Levi JR. Does suturing tonsil pillars post-tonsillectomy reduce postoperative hemorrhage?: A literature review. Int J Pediatr Otorhinolaryngol. 2019; 117:204-09. [DOI] [PubMed] [Google Scholar]
- 34.Grasl S, Janik S, Vyskocil E, Kadletz L, Grasl MC, Erovic BM. Preoperative plasma fibrinogen as a predictive factor for post-tonsillectomy haemorrhage. Clin Otolaryngol. 2019; 44 (6):935-41. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study were included in this published article.


