Abstract
Background
Subclinical tuberculosis (TB) is accidentally detected by radiologic and microbiologic findings. Transmission by those with subclinical TB could delay prevention effort. However, our study demonstrated positive aspect of COVID-19 outbreak as it could allow subclinical TB to be detected faster through a chest X-Ray (CXR).
Methods
This cross-sectional cohort study aimed to report demographics, comorbidities, and outcomes related to early detection of TB among COVID-19 patients, and to elaborate the association between SARS-CoV-2 and pulmonary TB. Data of patients with SARS-CoV-2 co-infection with Mycobacterium tuberculosis (MTB) diagnosed between March 2020 – March 2022 was collected.
Results
Out of 12,275 COVID-19 patients, 26 were definitively diagnosed with MTB infection (mean age 48.16 ± 20.17 years). All cases that had suspicious CXR that were not typical for COVID-19, were tested for MTB. On average, pulmonary TB was diagnosed after admission 5(3−10) days, the treatment initiation period was 3(1−5) days from the TB diagnosis.
Conclusions
This suggests an early detection of tuberculosis among COVID-19 patients by quicker screening CXR and sputum comparing to previous symptom guided screening. Thereby reducing the chance of TB transmission demonstrated during COVID-19 pandemic. So, clinicians should be aware of pulmonary tuberculosis in COVID-19 patients with atypical radiologic findings.
Keywords: Mycobacterium tuberculosis, Subclinical tuberculosis, SARS-CoV-2, COVID-19
1. Introduction
Since 2020, the coronavirus disease-2019 (COVID-19) has still been an ongoing pandemic with 430 million cases and over six million deaths reported until February 2022 [15]. Simultaneously, tuberculosis (TB) also continues to be a global burden with 1.3 million deaths, ranking as the second leading cause of death after COVID-19 pandemic (World Health Organization, 2020) [13]. However, there are limited case reports or case series describing TB and COVID-19 co-infection, mostly reported in TB high burden countries such as India, China, and Bangladesh [10]. A meta-analysis by Song et al. [8] suggested that patients with TB and COVID-19 coinfection were 2.21 and 2.27 times at risk of death and developing severe disease, respectively, more than COVID-19 patients without TB. Owing to the worse prognosis and overlapping clinical symptoms of these two diseases, routine screening for Mycobacterium tuberculosis (MTB) among COVID-19 cases was recommended in high-TB burden countries such as India, China, Indonesia, and Philippines. Thailand is also ranked 19th in TB high burden countries, yet no study was conducted. Therefore, this is the first study on COVID-19 associated pulmonary tuberculosis in Thailand and Southeast Asia. In addition, there is still a knowledge gap regarding the association between these two diseases whether tuberculosis just developed after COVID-19 or formerly dormant in the asymptomatic patients. This study will present the epidemiology, clinical presentation, diagnostic test, and treatment data of patients unexpectedly diagnosed with pulmonary TB after COVID-19 diagnosis.
2. Methods
2.1. Definitions
To define COVID-19 cases, all patients with suspected clinical symptoms and/or compatible imaging findings were confirmed by nasopharyngeal and throat swab using reverse transcription polymerase chain reaction (RT-PCR) for SARS-CoV-2. FDA emergency-use authorized PCR kits used in this study including Cobas ® SARS-CoV-2 Duo Test and Fosun ® COVID19 RT-PCR detection kit.
For TB diagnosis, patients with radiographic findings that were atypical for COVID-19 but suspicious for TB, were screened with acid fast bacilli (AFB) smear of respiratory specimens, and subsequently confirmed by mycobacterium culture, RT-PCR for Mycobacterium tuberculosis complex, or Xpert MTB/RIF assay. Suspicious chest x-ray findings can be pulmonary consolidation shown as homogeneous or patchy opacities mostly in the middle and lower lobes, with or without hilar lymphadenopathy. Other findings suggestive of active pulmonary TB may include miliary opacities, pleural effusion, and pulmonary edema [1].
2.2. Study design
This was a retrospective study of epidemiological and clinical data from patients with SARS-CoV-2 coinfection with MTB within 6 months after the diagnosis of COVID-19. Data were collected during March 2020 to March 2022 at King Chulalongkorn Memorial Hospital (KCMH), Bangkok, Thailand.
2.3. Study procedures
Patients' information was collected using electronic medical records and a case report form. The information included demographic data, underlying conditions, history of upper respiratory infection symptoms, diagnostic tests for SARS-CoV-2 infection, history of closed contact TB, history of previous TB diseases and current TB symptoms, along with clinical course and medication history. Participants underwent chest radiographs, which were interpreted by radiologists. Those testing positive on symptom screening or chest radiographs were proceeded for further sputum smear microscopy, culture for M. tuberculosis and/or GeneXpert MTB/Rifampin (RIF) (Xpert; Cepheid, Sunnyvale, CA). Participants diagnosed with SARS-CoV-2 infection were admitted at King Chulalongkorn Memorial Hospital or underwent home/community isolation during the infectious period. Participants also underwent blood screening of HIV antibodies using 4th generation enzyme-linked immunoassay (ELISA) method. In case of positive anti-HIV screening, one another confirmatory test with HIV viral load would be also tested during admission.
2.4. Data analysis
Medical data were transferred in electronic format. Data were compiled and analysed in Microsoft Excel. Mean, standard deviation (SD), median, interquartile range (IQR) and frequencies (%) were used to describe individuals’ characteristics in this study.
3. Results
According to a cross-sectional data collected at King Chulalongkorn Memorial Hospital, Bangkok, Thailand during March 2020 – March 2022, there were 12,275 COVID-19 patients diagnosed at our centre. Out of these, twenty-six cases demonstrated SARS-CoV-2 with MTB coinfection, 17 (65.38%) were male and 9 (34.62%) were female patients. Mean age was 48.16 years (range 17 – 85;SD 20.17). Five (19.23%) patients had history of smoking while 14 (53.85%) ones had chronic medical conditions.
Among all patients, the symptoms that can be associated with COVID-19 were cough (73.7%), low grade fever (57.69%), rhinorrhoea (23.08%), sore throat (34.32%), anosmia (15.38%), and myalgia (7.69%). Mean duration of COVID-19 symptom onset was 4.15 days (SD 3.44) before seeking medical care. Nearly half of them (46.15%) did not report symptoms compatible with pulmonary tuberculosis. However, for symptomatic patients, symptoms that associated with MTB infection were chronic cough; persisted more than three weeks, unrecognized and did not affect their daily activities (38.46%), and unintentional significant weight loss; defined as weight loss more than 5% in 30 days (15.38%). All patients were tested negative for anti-HIV. History of household contact with a pulmonary tuberculosis patient, was found in two cases. And two other patients previously served sentence in a correctional facility in Bangkok, which also constituted a risk factor of MTB contact. No drug-resistant tuberculosis was found in any patient by both genotypic and phenotypic susceptibility testing. The demographic data, clinical symptoms, laboratory investigations, radiological findings, treatments, and outcomes of each patient were described in Table 1 .
Table 1.
Characteristics and outcomes of twenty-six patients with subclinical tuberculosis (TB) and coronavirus disease (COVID-19) co-infection.
| Case No. | Age /Gender |
Underlying conditions | COVID-19 onset (days) | COVID-19 symptoms | Imaging diagnosis | COVID-19 treatment | History of pulmonary TB | TB symptoms | History of TB contact | Laboratory for TB confirmation | Period of TB diagnosis after COVID-19 (days) | TB treatment initiation after diagnosis (days) | TB treatment regimen | Outcome at 6 months |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 31/F | – | 3 | cough, low grade fever | cavitary lesion at left upper lung zone | Favipiravir for 5 days with supportive treatment | no | asymptomatic | household contact pulmonary TB 1 year PTA | RT-PCR for MTBC | 4 | 3 | 2HRZE/4HR | loss F/U |
| 2 | 22/F | – | 14 | sore throat | reticulonodular opacity with cavitary lesion at right upper lung zone | Favipiravir for 5 days with supportive treatment | no | subacute cough for 2 weeks | no history of contact | MTB C/S | 5 | 3 | 2HRZE/4HR | cured |
| 3 | 18/M | – | 2 | cough, rhinorrhea, fever, sore throat | reticulopatchy opacity at right upper lung and left apex | supportive treatment | no | chronic cough for 6 months | no history of contact | RT-PCR for MTBC and MTB C/S | 3 | 3 | 2HRZE/4HR | cured |
| 4 | 50/M | HCV infection without cirrhosis, | 7 | fever, sore throat, rhinorrhea | nodular opacities at apical regions of both lungs | Favipiravir for 5 days with supportive treatment | no | asymptomatic | no history of contact | RT-PCR for MTBC | 2 | 3 | 2HRZE/4HR | cured |
| 5 | 60/M | – | 7 | anosmia, cough with few sputum | reticulonodulopatchy infiltration at left upper lung zone | Favipiravir for 5 days with supportive treatment | no | asymptomatic | no history of contact | MTB C/S | 4 | 2 | 2HRZE/4HR | cured |
| 6 | 33/M | – | 14 | cough, rhinorrhea, anosmia, fever | cavitary lesion at right upper lung zone | Favipiravir for 5 days with supportive treatment | no | chronic cough for 1 month | no history of contact | RT-PCR for MTBC | 6 | 3 | 2HRZE/4HR | cured |
| 7 | 54/F | DM, HT, active smoker 30-pack-year |
1 | cough, rhinorrhea, anosmia | mixed patchy and nodular opacity at right lung | Favipiravir for 10 days with supportive treatment | no | asymptomatic | no history of contact | RT-PCR for MTBC | 3 | 0 | 2HRZE/4HR | loss F/U |
| 8 | 26/M | Aggressive behaviour with history of amphetamine usage | 2 | fever, cough with sputum | ground glass opacity at right lung | Favipiravir for 10 days with supportive treatment | no | asymptomatic | no history of contact | RT-PCR for MTBC and MTB C/S | 4 | 1 | 2HRZE/4HR | loss F/U |
| 9 | 76/M | – | 4 | cough, sore throat, fever, myalgia | ground glass opacity at periphery of left upper lung zone | Favipiravir for 10 days with supportive treatment | no | asymptomatic | no history of contact | RT-PCR for MTBC | 5 | 1 | 2HRZE/4HR | loss F/U |
| 10 | 17/M | – | 3 | cough with sputum, right chest pain due to cough | reticulonodular infiltration at apical region of both lungs with cavitary lesion at right upper lung | Favipiravir for 10 days with supportive treatment | no | asymptomatic | no history of contact | RT-PCR for MTBC and MTB C/S | 1 | 3 | 2HRZE/4HR | transferred to other center |
| 11 | 44/M | – | 8 | cough, sore throat, fever, myalgia | ground glass opacity at the bilateral lower lung | Favipiravir for 5 days with supportive treatment | no | chronic cough with no sputum for 1 month, weight loss 1–2 kg/month for 2 months |
correctional facility 2 years PTA | RT-PCR for MTBC and MTB C/S | 6 | 2 | 2HRZE/4HR | cured |
| 12 | 59/F | DM | 4 | asymptomatic | patchy opacity in left upper lung zone | Favipiravir for 5 days with dexamethasone 6 mg/day for 5 days |
yes | whitish sputum, chronic cough, low grade fever at night, unintentional weight loss 4–5 kg/months for 3 months |
no history of contact | RT-PCR for MTBC | 12 | 3 | 2HRZE/4HR | treatment completed |
| 13 | 32/F | – | 6 | cough, sore throat, rhinorrhea | ground glass and patchy opacity in both lungs | Favipiravir for 10 days with Prednisolone 20 mg/day for 3 days then 10 mg/day for 3 days | yes | asymptomatic | no history of contact | RT-PCR for MTBC | 8 | 17 | 2HRZE/4HR | loss F/U |
| 14 | 79/M | pulmonary HT, COPD, PV/ET, CKD stage 3 | 3 | fever, rhinorrhea, cough with sputum | massive pleural effusion of both lungs | Favipiravir for 2 days then Remdesivir for 8 days, with dexamethasone 6 mg/day for 3 days then dexamethasone 10 mg/day for 5 days then dexamethasone 6 mg/day for 4 days then prednisolone 20 mg/day for 1 days then dexamethasone 6 mg/day for 4 days then prednisolone 20 mg/day for 6 days |
no | asymptomatic | no history of contact | RT-PCR for MTBC and MTB C/S | 4 | 1 | 2HRZE/4HR | died |
| 15 | 33/F | DM | 2 | anosmia, cough | recticular infiltration at both upper lungs | supportive treatment | no | unintentional weight loss 3 kg/months for 2 months | no history of contact | RT-PCR for MTBC and MTB C/S | 8 | 6 | 2HRZE/4HR | cured |
| 16 | 66/M | – | 3 | cough with diarrhea | reticulonodular infiltration at both upper lungs | Remdesivir for 10 days | no | chronic cough with no sputum for 1 months, weight loss | no history of contact | RT-PCR for MTBC and MTB C/S | 1 | 1 | 2HRZE/4HR | cured |
| 17 | 85/M | – | 2 | cough, sore throat, fever | reticulopatchy opacity at both lungs | Favipiravir for 10 days, then Remdesivir for 2 days with Dexamethasone 6 mg 3 days, 10 mg 1 days, methylprednisolone 250 mg 3 days, Dexamethasone 20 mg 4 days, 15 mg 1 day | no | none | no history of contact | MTB C/S | 4 | 1 | 2HRZE/4HR | treatment completed |
| 18 | 51/F | DM, HT, DLP, Hyperthyroidism, Chronic HF | 3 | fever, dyspnea | recticular infiltration at both upper lungs | Favipiravir for 10 days | no | chronic cough | household contact pulmonary TB 10 years PTA | RT-PCR for MTBC and MTB C/S | 30 | 6 | 2HRZE/4HR | treatment completed |
| 19 | 44/M | – | 5 | cough, sore throat, fever | normal lungs | supportive treatment | no | chronic cough, unintentional weight loss 2 kg/months for 3 months | correctional facility 6 months PTA | RT-PCR for MTBC and MTB C/S | 35 | 12 | 2HRZE/4HR | cured |
| 20 | 79/M | HT, COPD | 2 | fever, dyspnea | recticular infiltration at both upper lungs | Favipiravir for 10 days, then Remdesivir for 2 days with Dexamethasone 6 mg 3 days, 10 mg 1 days, methylprednisolone 250 mg 3 days, Dexamethasone 20 mg 4 days, 15 mg 1 day |
no | chronic cough | no history of contact | MTB C/S | 60 | 5 | 2HRZE/4HR | treatment completed |
| 21 | 75/M | COPD, BPH | 3 | cough, sore throat | reticulonodular opacity in both upper lungs zone | Favipiravir for 10 days | no | chronic cough | household contact pulmonary TB 6 years PTA | RT-PCR for MTBC | 10 | 1 | 2HRZE/4HR | cured |
| 22 | 51/F | epilepsy, hypothyroidism | 1 | fever, dyspnea | patchy opacity in left lower lung zone, with pleural effusion | Remdesivir for 5 days with dexamethasone for 3 days | yes | none | no history of contact | RT-PCR for MTBC | 20 | 5 | 2HRZE/4HR | cured |
| 23 | 60/M | DM, ESRD, HT, DLP, TVD | 2 | cough, fever, dyspnea | perihilar infiltration | Remdesivir 1 dose then Favipiravir for 5 days | yes | palpable supraclavicular lymph node, subacute fever for 2 weeks | no history of contact | RT-PCR for MTBC and MTB C/S | 21 | 5 | 2HRZE/4HR | cured |
| 24 | 29/F | – | 3 | fever | reticulonodular opacity in both upper lungs zone | supportive treatment | yes | chronic cough for 4 months | no history of contact | MTB C/S | 0* | 2 | 2HRZE/4HR | cured |
| 25 | 40/M | – | 2 | cough, atypical chest pain | patchy opacity with lucency in the left lung | supportive treatment | no | unintentional weight loss 4 kg/months for 3 months | no history of contact | RT-PCR for MTBC and MTB C/S | 8 | 3 | 2HRZE/4HR | cured |
| 26 | 38/M | s/p CDKT | 2 | cough | recticular infiltration at left upper lung | Remdesivir for 6 days | yes | none | no history of contact | MTB C/S | 1 | 8 | 2HRZE/4HR | cured |
CDKT; Cadaveric kidney transplantations
CKD; Chronic Kidney disease
COPD: Chronic obstructive pulmonary disease
DLP; Dyslipidemia
DM; Diabetes mellitus
ESRD; End stage renal disease
HF; Heart failure
HT; Hypertension
PV/ET; Polycythemia vera with essential thrombocythemia
TVD; Triple vessels disease
C/S; Microorganism culture and sensitivity
H; Isoniazid
R; Rifampicin
Z; Pyrazinamide
E; Ethambutol
F/U; follow-up
Treatment completed; microorganism negative on culture or RT-PCR, but lesion was still found in chest radiograph
Cured; treatment completed, and no lesion found on chest radiograph
Tested potitive for COVID during TB treatment
All cases had suspicious CXR that were not typical for solely SARS-CoV-2 infection, which made the attending physicians suspected other respiratory diseases, namely reticulonodular opacity (19.2%), ground glass appearance (15.4%), patchy infiltration (15.4%), reticular infiltration (15.4%), cavitary lesions (7.7%). All patients were confirmed of MTB infection by RT-PCR detection, or microorganism culture and sensitivity. Subsequently, appropriate tuberculosis treatment regimens were initiated with an average period of 3.50 days (SD 3.54) from COVID-19 diagnosis.
All patients received standard symptomatic care. Favipiravir, which was a standard antiviral for mild to moderate COVID-19 of Thai national guideline during that time, was distributed to 15 patients (57.69%, mean duration 7.13 days, SD 2.87). While 6 patients (23.08%) received remdesivir (mean duration days 4.00, SD 2.75) because of more severe symptom or having medical risk of COVID-19 severity progression.
There are 6 patients who received corticosteroid as adjunctive treatment. Dexamethasone, prednisolone, and methylprednisolone were used in varying dosage and duration as described in Table 1 .
On average, TB was diagnosed median 5 days (IQR 3–10 days) after admission. Anti-TB medications were initiated shortly after the diagnosis median 3 days (IQR 1–5 days). Twenty-five patients recovered without respiratory complication or hypoxia needing oxygen supplementation. Only one required ambulatory oxygen canula. All patients had no drug adverse reaction, nor drug interaction between favipiravir and anti-TB drugs including isoniazid (H), rifampicin(R), pyrazinamide (Z) and ethambutol (E).
The outcome of every patient was documented after TB treatment. Among 26 TB and COVID-19 coinfected patients, 18 patients (69%) had completed the HRZE treatment with clinically full recovery, sputum AFB conversion, and improved CXR. While 4 patients (15%) completed the treatment with minimal residual lung lesion such as minor focal atelectasis and minor consolidation at left upper lung. One patient with disseminated TB also completed treatment with full recovery, which makes 84% treatment completion rate. In addition, there was no drug resistance reported among these patients. However, one patient (3.8%) with TB pleuritis following massive pleural effusion both lungs was later dead with congestive heart failure. Unfortunately, 3 (12%) patients had lost follow-up from the study hospital, probably due to COVID-19 situations.
We hereby presented three index cases of COVID-19 patients that piqued our attending physician interests, one of them was MTB infection shortly after discharge from COVID-19 admission (case #13), the other was a newly diagnosed MTB infection (case#14), and the third case was disseminated TB (case #23).
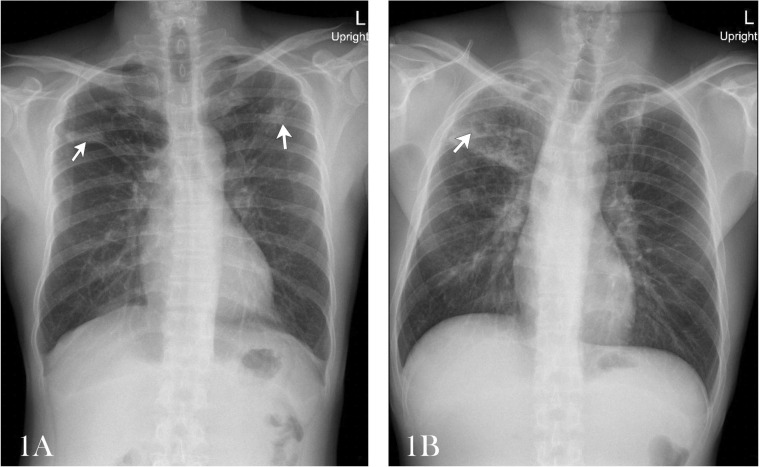
Case #13: A 32-year-old female with a history of pulmonary tuberculosis for 5 years ago. She had no underlying conditions. Her COVID-19 presented with cough, sore throat, and rhinorrhoea. After RT-PCR for SARS-CoV-2 was confirmed, oral favipiravir was prescribed for 10 days. Owing to her hypoxia, prednisolone tapering dose 0.2–0.4 mg/kg/day prescribed for 6 days also. After a-week, she recovered from COVID-19 symptoms and was discharged without any complication. Seventeen days after COVID-19 diagnosis, chest radiograph shows ground glass opacity at both lungs. This makes the attending physician suspect pulmonary TB. Afterwards, RT-PCR for MTB complex was performed, and detected. Pulmonary TB treatment were administered consisting of 2-month induction of 2HRZE and then 4-month maintenance therapy with 4HR. She now resolved from pulmonary TB.( Fig. 1).
Fig. 1.
A Chest X-Ray of case #4 shows nodular opacity in both lungs. B Chest X-Ray of case #10 showed reticular reticulonodular infiltration at apical region of both lungs, and cavitary lesion at right upper lung.
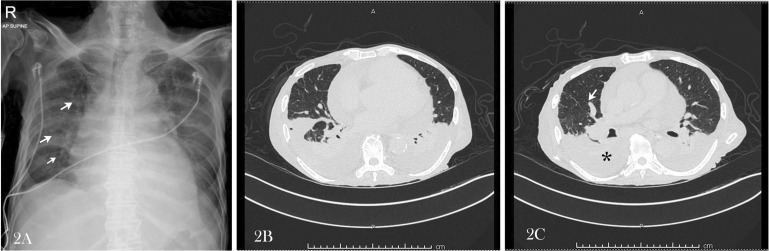
Case #14: 79-year-old Thai male, with pulmonary hypertension, chronic obstructive pulmonary disease, and chronic kidney disease stage 3, had fever, rhinorrhoea, and cough for 3 days as the initial symptoms of COVID-19. He had no history of previous MTB infection. Chest radiograph showed massive pleural effusion in both lungs, which made the attending physician suspect pulmonary TB infection. The imaging was shown in the Fig. 2. RT-PCR for MTB complex was subsequently done and respectively detected. After 2-day of oral favipiravir oxygen desaturation was progressed, so he was given intravenous remdesivir for 8 days later. High dose dexamethasone was also administered intravenously for 5 days. On fourth day of dexamethasone, his oxygenation became worse, with lowest at 81% saturation. He complained of shortness of breath, so he received step-up oxygen supplementation by oxygen cannular, and then high flow nasal cannula. He also developed sharp chest pain which cardiac rubbing sound. Chest computed tomography (CT) showed bilateral pleural thickening with pleural effusion. Needle thoracocentesis was proceeded which later showed positive for AFB staining of pleural effusion suggesting TB pleurisy. Dexamethasone was tapered to oral prednisolone with total 23-day duration. His TB was treated with 2HRZE and 4HR continuum. He was markedly improved since a couple week of TB treatment and now his CXR returned to previous background with resolved effusion. Unfortunately, he died 3 months after the treatment was completed due to congestive heart failure.
Fig. 2.
A Chest X-Ray of case #14 showed multiple lungs nodules in the right lung, along with reticular infiltration in the right lower lung field. B and 2 C Chest computed tomography of case #14 showed multiple hyperdensity lesions in both lungs (arrow), and bilateral pleural effusion (star).
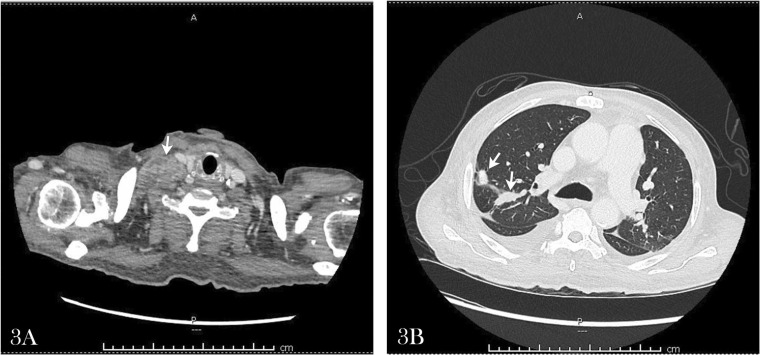
Case #23: Thai male, aged 60 years, with end stage renal disease, hypertension, triple vessels disease, and type II diabetes mellitus, was admitted to our hospital due to COVID-19 RT-PCR detection two days after symptoms of cough, low grade fever, and mild dyspnoea. His CXR revealed perihilar infiltration in both lungs. He had no previous history of MTB infection also. Oral Favipiravir was administrated. He did not need corticosteroid because of mild COVID-19. On the fourth day of admission, the physician noticed the palpable mass at right supraclavicular area. Fine needle aspiration was done at the lesion, then AFB and PCR for MTB complex both detected. The imaging was shown in the Fig. 3 . Sputum RT-PCR for MTB complex was detected also. His disseminated TB treatment consisted of 2HRZE and 4HR regimen. The patient was discharged after 14 days of admission with clinical improvement and no adverse drug reaction found.
Fig. 3.
A Neck computed tomography of case #23 showed enlarged right supraclavicular lymph node (arrow). B Chest computed tomography of case #23 showed multiple hyperdensity lesions (arrows) in the right lung.
4. Discussion
In the year 2021, Thailand is one of the thirty countries with highest burden for tuberculosis with an incidence of 150 per 100,000 people. According to our results, the incidence of newly diagnosed TB infection was 26 of the 12,275 COVID patients, equalled to 205 per 100,000 people, which was little higher than the incident rate among general population without COVID-19. The possible explanation could be that the included patients might be subclinical TB with revealed their abnormal CXR during COVID-19 diagnosis.
Twenty-one (21/26, 80.8%) patients were diagnosed with TB coinfection within 2 weeks after COVID-19 diagnosis. It could possibly be subclinical TB as we discussed before, or TB reactivation that just occur after COVID-19. Nevertheless, this implied a promising possibility of early detection of TB infection, which leads to early effective treatment. Consequently, early diagnosis helps cut down transmission within households and communities, decrease burden of TB, and leads to better outcomes, which include curation (culture conversion among bacteriologically confirmed pulmonary TB patients), and treatment completion (treatment completed without evidence of failure, but without sputum smear or culture results (World Health Organization, 2020) [14].
Regarding other five TB cases diagnosed more than 2 weeks after COVID-19 diagnosis, these findings point out a possible association between TB and COVID-19 infection. SARS-CoV-2 virus as well as its treatment, potentially had an influence on the immune system through various mechanisms which finally resulted in the reactivation of tuberculosis. Firstly, there was direct viral damage to the cells of the respiratory system on which epithelium expresses angiotensin-converting enzyme 2 (ACE2), the main target receptor of SARS-CoV-2 [5]. Secondly, there is cell-mediated immune damage by excessive inflammatory cell infiltration, activation of complement cascade and release of biologically active substances in the lung parenchyma, aggravating diffuse alveolar damage [7]. Thirdly, in severe cases of COVID-19, most patients might develop lymphopenia. Numbers of B-cells, CD4 + & CD8 + T-cells, and NK T-cells would be decreased, especially T-cells, which were the key adaptive immunity to fight against tuberculosis [16]. To summarize, both long-lasting lung damage and impaired immune function following COVID-19 possibly predisposed patients to progression, reactivation, or acceleration of MTB infection.
Apart from viral infection and host immune responses, other medications prescribed during COVID-19 treatment, especially anti-inflammatory agents, also play an important role in immune disturbance, might further predispose to secondary infections. Generally, corticosteroids use could elevate risk of developing tuberculosis by regulation of numerous genes responsible for immune response that is required in tuberculosis control; for example, inhibition of lymphokine effect, monocyte chemotaxis, and Fc-receptor binding, circulated lymphocytes, especially T-cells. [3].
For COVID-19 patients, a prospective meta-analysis of 7 clinical trials reported reduction in all-cause mortality among those with severe and critically ill conditions, regardless of whether they required mechanical ventilation. As a result, corticosteroids were recommended as a part of standard care for critically ill COVID-19 patients [4]. On the other hand, corticosteroids administration was not supported in mild or less severe patients. World Health Organization (WHO) guideline, launched in 2020, suggested systemic corticosteroids, administered either orally or intravenously, at a dose of 6 mg of dexamethasone, which is equivalent to 150 mg of hydrocortisone, or 40 mg of prednisone, or 32 mg of methylprednisolone, for up to 7–10 days in severe and critical COVID-19 patients (World Health Organization, 2020) [12]. However, the previous study from American Thoracic Society (ATS) and US Center for Disease Control and Prevention (CDC) (2000) revealed that the risk of TB reactivation increased with oral prednisolone for more than 15 mg per day, or equivalent, for 2–4 weeks [9]. It is remarkable that the dose proposed for COVID-19 treatment substantially exceeds the dose at risk for enhance TB reactivation. So, TB reactivation should be monitored especially among severe to critical COVID-19 who received high and prolonged corticosteroids.
The TB/COVID-19 Global Study Group (2022) had reported 767 TB and COVID-19 co-infected patients from 34 countries [10]. There were 74% of patients with history of previous TB, 16.5% diagnosed within the same week from suspicious imaging, and 9.5% initially diagnosed with COVID-19. Interestingly, among the last group, 35 out of 71 patients were diagnosed COVID-19 more than 30 days prior to TB, which is considered sufficient time for TB disease development, thus the study discussed that COVID-19 may not play a major role in TB progression. Another cohort study from South Africa by Western Cape Department of Health in collaboration with the National Institute for Communicable Diseases SA (2019), included 22,308 COVID-19 patients, of which 2128 (27%) got TB and COVID-19 coinfection. Among this group, 22% had history of previous TB and 5% also known as current MTB infection [11]. However, more evidence will be needed to fill gaps in knowledge about the role of COVID-19 in TB reactivation.
Favipiravir and remdesivir are the RNA-dependent RNA polymerase inhibitors which were not used widely until the COVID-19 pandemic. From previous meta-analysis, both drugs were associated with clinical and laboratory improvement in COVID-19 patients with no serious adverse drug reactions [6]. Favipiravir is mainly metabolised in the liver by aldehyde oxidase which has a short half-life of 2–5.5 h and can cause asymptomatic liver injury with increased liver transaminases [2]. Remdesivir, half-life of 20 h, is also metabolised by the liver, causing transient elevation of aminotransferase which is not usually linked to clinical injury in COVID-19 patients. Also, anti-TB drugs are well known for drug-induced liver toxicity ranging from asymptomatic to severe toxicity. However, nearly all patients in our study, had no significant elevation of liver function tests during treatment with anti-TB drugs shortly after favipiravir or remdesivir. The possible explanation of these findings could be rapid clearance and short half-life of the COVID-19 medications that might not significantly raise liver toxicity. The interactions between COVID-19 treatment and anti-TB drugs are still limited, thus further studies are needed, and safety monitoring should be observed.
The outcome of patient TB treatment shows that 69% had completed the HRZE treatment and fully recovered. While 15% completed the course of treatment with minimal residual lung lesions. The mortality rate was 0%.
We also compared the outcome with COVID-19 patients without TB sub-grouped into clinical spectrum defined by US-CDC; mild/moderate, severe and critical COVID-19. In our center, the mortality rate of the critical group, who had respiratory failure or multiorgan system dysfunction, thus required intensive care unit admission, was 170 in 390 (43.6%). While the mortality rate of the severe patients, who were complicated with pneumonia, acute respiratory distress syndrome, sepsis, acute kidney injury, or secondary bacterial infections, was 40 in 274 (14.6%). In contrast, the mortality rate of mild/moderate groups who did not need hospitalization, was only 15 in 7654 (0.2%). The overall mortality rate was 2% among patients without MTB coinfection.
While data collected between 2018 and 2021 in our center showed success treatment 446 in 538 (83%)) of patient with isolated TB infection; 111 (25%) were cured, and 335 (75%) received full course of medication. The overall mortality rate among solely TB infection was 31 in 538 (6%) that more than half found in complicated medical underlying hosts.
However, TB and COVID-19 coinfection in our study mostly revealed among mild/moderate COVID-19. We could not conclude that TB has no effect on COVID-19 mortality. This was probably due to small number of index cases, and under-detection.
Our study also has several strengths to guide novel information and policy making. Firstly, it is the first study in Southeast Asia that reports the incidence of COVID-19 and TB coinfection and outcomes after receiving medications. Secondly, characteristics, symptoms, and findings of each case were elaborately described. Every patient was diagnosed with COVID-19 and TB infection by using RT-PCR, which reported within 3 days, resulting in early detection, early treatment, and reduction of transmission. Thirdly, the literature regarding mechanisms and impacts of COVID-19 infection as well as corticosteroids therapy on tuberculosis infection were delicately reviewed. This study, by reporting cases with COVID-19 and TB coinfection, emphasized the importance of further research to obtain more evidence and clearer understanding on both topics. Also, health care providers should be more aware of tuberculosis infection when treating COVID-19 patients with atypical radiographic findings especially in high TB burden countries.
However, our study was a retrospective study and had a small number of index cases. We are aware that our index TB cases mainly triggered by using CXR imaging, which might result in under-reported cases. However, we collected data for two years and followed patients who received steroids whether they develop MTB infection after 6 months of treatment. Although about 5–10% of patients with latent TB could progress to active disease within the first two years in general, this number might be shorter among immune dysregulated hosts. So we suggest further research and authority could implement an active screening of tuberculosis among COVID-19 patients to obtain more cases, which might result in higher early detection and good treatment outcomes. Extended period of follow-up in post COVID-19 patients could also be implemented to obtain clearer association between COVID-19/COVID-19 treatment with corticosteroids and TB reactivation. Furthermore, additional laboratory investigation, involving the immune mechanisms such as certain proinflammatory cytokines and immune cells might be collected to analyse possible correlation, and to serve as a biomarker to predict disease severity and risk of coinfection in the future.
Moreover, our study reported an incidence of tuberculosis on COVID patients in high burden areas. These results could not be applicable for low burden countries, thus further studies will be needed.
5. Conclusion
Tuberculosis is an epidemic in the Asia-Pacific region. The current COVID-19 pandemic might help speed up the diagnosis process as it allows patients to be screened quicker than before. Clinicians should be aware of pulmonary tuberculosis in COVID-19 patients with atypical radiologic findings, especially in high TB burden countries. The association between SARS-CoV-2 and TB remains unclear, and needs further studies.
Ethical Approval statement
Informed consents were obtained from the patients for the publication of their information and radiographic images.
Funding Source
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
None.
Declaration of Competing Interest
We have no conflict of interest to declare.
References
- 1.Burrill J., Williams C.J., Bain G., Conder G., Hine A.L. Misra RR. tuberculosis: a radiologic review. Radiographics. 2007;27(5):1255–1273. doi: 10.1148/rg.275065176. [DOI] [PubMed] [Google Scholar]
- 2.Du Y.X., Chen X.P. Favipiravir: pharmacokinetics and concerns about clinical trials for 2019-nCoV infection. Clin Pharm Ther. 2020;108(2):242–247. doi: 10.1002/cpt.1844. [DOI] [PubMed] [Google Scholar]
- 3.Fauci A.S., Dale D.C., Balow J.E. Glucocorticosteroid therapy: mechanisms of action and clinical considerations. Ann Intern Med. 1976;84(3):304–315. doi: 10.7326/0003-4819-84-3-304. [DOI] [PubMed] [Google Scholar]
- 4.Sterne J.A.C., Murthy S., Diaz J.V., Slutsky A.S., Villar J., Angus D.C., et al. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis. Jama. 2020;324(13):1330–1341. doi: 10.1001/jama.2020.17023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lin L., Lu L., Cao W., Li T. Hypothesis for potential pathogenesis of SARS-CoV-2 infection-a review of immune changes in patients with viral pneumonia. Emerg Microbes Infect. 2020;9(1):727–732. doi: 10.1080/22221751.2020.1746199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rezagholizadeh A., Khiali S., Sarbakhsh P., Entezari-Maleki T. Remdesivir for treatment of COVID-19; an updated systematic review and meta-analysis. Eur J Pharm. 2021;897 doi: 10.1016/j.ejphar.2021.173926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Short K.R., Kroeze E., Fouchier R.A.M., Kuiken T. Pathogenesis of influenza-induced acute respiratory distress syndrome. Lancet Infect Dis. 2014;14(1):57–69. doi: 10.1016/S1473-3099(13)70286-X. [DOI] [PubMed] [Google Scholar]
- 8.Song W.M., Zhao J.Y., Zhang Q.Y., Liu S.Q., Zhu X.H., An Q.Q., et al. COVID-19 and tuberculosis coinfection: an overview of case reports/case series and meta-analysis. Front Med (Lausanne) 2021;8 doi: 10.3389/fmed.2021.657006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Targeted tuberculin testing and treatment of latent tuberculosis infection. Am Therac Soc MMWR Recomm Rep. 2000;49(Rr-6):1–51. [PubMed] [Google Scholar]
- 10.Tuberculosis and COVID-19 co-infection: description of the global cohort. Eur Respir J. 2022;59(3) doi: 10.1183/13993003.02538-2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Risk Factors for coronavirus disease 2019 (COVID-19) death in a population cohort study from the Western Cape Province. South Afr Clin Infect Dis. 2021;73(7) doi: 10.1093/cid/ciaa1198. e2005-e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.World Health Organization. Corticosteroids for COVID-19: living guidance, 2 September 2020; Geneva; 2020.
- 13.World Health Organization. Definitions and reporting framework for tuberculosis [2013 revision: updated December 2014 and January 2020]. https://www.who.int/publications/i/item/9789241505345, 2020 (accessed 22 February 2022).
- 14.World Health Organization. Global tuberculosis report 2020. Geneva; 2020.
- 15.World Health Organization. WHO COVID-19 Dashboard. https://covid19.who.int/, 2020 (accessed 22 February 2022).
- 16.Zheng H.Y., Zhang M., Yang C.X., Zhang N., Wang X.C., Yang X.P., et al. Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients. Cell Mol Immunol. 2020;17(5):541–543. doi: 10.1038/s41423-020-0401-3. [DOI] [PMC free article] [PubMed] [Google Scholar]