Abstract
Background
Childhood cancer survivors (CCSs) show relevant cardiac morbidity and mortality throughout life. Early detection is key for optimal support of patients at risk. The aim of this study was to evaluate 2‐dimensional speckle‐tracking echocardiography strain analysis during semisupine exercise stress in CCSs for detection of subclinical left ventricular dysfunction after cancer treatment.
Methods and Results
Seventy‐seven CCSs ≥1‐year postchemotherapy were prospectively examined at rest, low, and submaximal stress level and compared with a cohort of healthy adolescents and young adults (n=50). Global longitudinal strain (GLS), short axis circumferential strain, and corresponding strain rates were analyzed using vendor‐independent software. CCSs at median 7.8 years postchemotherapy showed comparable left ventricular GLS, circumferential strain, and strain rate values at all stress stages to healthy controls. Yet, prevalence of abnormal GLS (defined as <2 SD of controls reference) in CCSs was 1.3% at rest, 2.7% at low, and 8.6% at submaximal stress. In CCSs, relative change of circumferential strain from rest to submaximal stress was lower than in healthy controls, median 16.9 (interquartile range [IQR], 3.4; 28.8) % versus 23.3 (IQR, 11.3; 33.3) %, P=0.03, most apparent in the subgroups of CCSs after high‐dose anthracycline treatment and cancer diagnosis before the age of 5 years.
Conclusions
In this prospective 2‐dimensional speckle tracking echocardiography strain study, prevalence of abnormal left ventricular GLS increased with stress level reflecting impaired cardiac adaptation to exercise stress in some CCSs. However, relatively early after last chemotherapy, this did not result in significant differences of mean GLS‐, circumferential strain‐, and strain rate values between CCSs and controls at any stress level.
Keywords: 2D‐speckle‐tracking strain echocardiography, cardio‐oncology, childhood cancer survivor, stress echocardiography
Subject Categories: Echocardiography, Exercise Testing, Imaging, Cardio-Oncology, Cardiotoxicity
Nonstandard Abbreviations and Acronyms
- 2D
2‐dimensional
- a2c
apical 2 chamber
- a3c
apical 3 chamber
- a4c
apical 4 chamber
- CCSs
childhood cancer survivors
- CS
circumferential strain
- DICOM
Digital Imaging and Communications in Medicine
- fps
frames per second
- GLS
global longitudinal strain
- GLSR
global longitudinal strain rate
- LS
longitudinal strain
- SJLIFE
St. Jude Lifetime Cohort Study
- SR
strain rate
- STE
speckle tracking echocardiography
Clinical Perspective.
What Is New?
Overall, childhood cancer survivors show comparable response to exercise stress compared with healthy controls measured by 2‐dimensional‐speckle‐tracking strain analysis a median of 7.8 years after last chemotherapy.
Yet, prevalence of abnormal global longitudinal strain increases with stress level, reflecting impaired cardiac adaptation to exercise stress in some childhood cancer survivors.
What Are the Clinical Implications?
Cardiac events are the leading nonmalignant cause of death in childhood cancer survivors, and lifelong cardiac surveillance is necessary to pursue early identification of potential (subclinical) cardiac impairment.
With outcome data lacking, routine application of 2‐dimensional‐speckle tracking strain stress testing for cardio‐oncologic surveillance does not seem reasonable at this point, but we recommend consideration of this low side‐effect, noninvasive method in select patients with borderline or inconclusive findings to obtain more diagnostic information and possibly shorten the follow‐up interval.
Childhood cancer survivors (CCSs) show an elevated risk for cardiac morbidity and mortality, which increases even long after cancer therapy. 1 Cardiac events are the leading nonmalignant cause of death in CCSs. 2 Cardiotoxicity in CCSs is mainly attributable to anthracycline and radiation therapy. 3 Therefore, lifelong cardiac surveillance is recommended.
Echocardiography is the cornerstone in cardio‐oncologic follow‐up. In a recent consensus statement, 2‐dimensional (2D)‐speckle‐tracking echocardiography (STE) has been introduced as the imaging technique of choice to detect subclinical left ventricular (LV) dysfunction. 4
2D‐STE imaging can help to quantify deformation of the myocardium and objectify regional as well as global LV function. Altered strain in CCSs despite normal LV‐ejection fraction (EF) has been described. 5 , 6 Yet, clinical relevance of the method in CCSs has to be determined. 7
Conventional stress echocardiographic findings in CCSs are controversial. 8 , 9 Exercise echocardiography has not been recommended in routine follow‐up for asymptomatic CCSs. 4
Combining the potential of stress echocardiography with the strength of 2D‐STE might be a promising approach for early detection of cardiac malfunction.
The aim of this study was to evaluate 2D‐STE global longitudinal strain (GLS), circumferential strain (CS), and corresponding strain rates (SR) during semisupine exercise stress testing as a potential method for detection of early LV dysfunction in CCSs. We hypothesized that CCSs may have subclinical impairment of cardiac function manifested in a deteriorated cardiac response to exercise stress measured by GLS, CS, and corresponding SR compared with healthy controls.
METHODS
The data that support the findings of this study are available from the corresponding author upon reasonable request.
In preparation to this study in CCSs, our workgroup has published a standardized protocol and reference values for 2D‐STE‐derived LV‐strain and LV‐SR during semisupine bicycle stress testing in adolescents and young adults. 10 Methodical aspects are in excerpts included in this article.
Study Population
CCSs were prospectively recruited at our institution during routine cardio‐oncologic follow‐up. Inclusion criteria were age <18 years at cancer diagnosis, currently in remission, time since last chemotherapy >1 year, body size >140 cm to reach bicycle pedals. Exclusion criteria were congenital heart disease (except patent foramen ovale or small atrial septal defect).
The control group consisted of healthy adolescents and young adults. Details on the control group have been previously published by our workgroup. 10
Written informed consent for study participation was obtained from all study participants and their parents or legal guardians if the patient was underage. The study protocol conforms to the ethical guidelines of the Declaration of Helsinki and was approved by the local ethics committee (approval number 101/16).
Clinical Characteristics
All CCSs underwent routine cardiac examination including ECG and echocardiography before the stress test. Height and weight were measured. Disease history was obtained from patient records at our institution's archive. We assessed cancer type, age at diagnosis, time since last chemotherapy, cancer therapy regimen, cumulative anthracycline and radiation doses, as well as current health issues and medication.
Cumulative anthracycline doses are presented as doxorubicin‐equivalent doses. Dose conversion was conducted according to the Children's Oncology Group Long‐Term Follow‐Up Guidelines Version 5.0 (Conversion factors: doxorubicin x1, daunorubicin x0.5, idarubicin x5, and mitoxantrone x4). 11
Cardiomyopathy risk was stratified according to a predescribed recommendation. 12 High cardiotoxic risk is defined as cumulative equivalent anthracycline dose ≥250 mg/m2 or chest radiation dose ≥35 gray or anthracycline dose ≥100 mg/m2 and chest radiation ≥15 gray. Moderate cardiotoxic risk represents anthracycline dose of 100 to <250 mg/m2 or chest radiation dose ≥15 to <35 gray. Low cardiotoxic risk is assumed for anthracycline dose <100 mg/m2.
Stress Protocol and Echocardiographic Image Acquisition
Stress tests were performed with patients on a bicycle ergometer (ergoselect 1200; ergoline GmbH, Bitz, Germany) in semisupine body position. Patients were tilted up to 45° leftwards for optimal acoustic window. Workload was increased every 3 minutes by 20 W (body weight <50 kg) or 25 W (body weight >50 kg). Target pedaling speed was 55 to 65 revolutions/min. Resting images were acquired after 3 minutes in still semisupine position. Exercise images were recorded at low and submaximal stress level, defined as heart rates of 60%, respectively 75% to 85% of age based on maximal exercise heart rate (according to 220−age formula). During image recording, the corresponding workload was not altered and pedaling was continued. Poststress images were recorded after a cool‐down phase when heart rate approached resting values.
Echocardiographic studies were conducted by experienced stress echocardiography investigators on a Philips Epiq 5G (Philips, Amsterdam, the Netherlands) ultrasound machine with a S5‐2 transducer. In all exercise stages, loops of apical 4‐ (a4c), 3‐ (a3c), and 2‐ (a2c) chamber views and in midpapillary short axis were saved.
Image sets of 2 ECG‐gated cardiac cycles were retrospectively recorded. Four subsets for every image plane, each containing 2 cardiac cycles, were instantly assessed on the screen of the ultrasound machine. Image quality was evaluated and the best of the 4 subsets was saved for offline analysis. If image quality was unsatisfactory, up to 3 recording attempts were repeated pursuing optimal image quality even during exercise stress. The correspondent stress stage was elongated if necessary. Images were optimized for STE strain analysis by narrowing the field of view and optimizing image depth according to LV size, thereby increasing the frame rate. Eighty‐five frames per second (fps) or higher were approached. In addition, LV diameters, LV‐fractional shortening, and LV‐EF (Teichholz) were measured in M‐mode in a standard parasternal long‐axis view.
Strain Analysis
Images were transferred in Digital Imaging and Communications in Medicine (DICOM) format to a workstation. Strain analysis was performed offline using Tomtec 2D‐CPA analysis software, version TTA 2.30 (Tomtec Imaging Systems, Unterschleissheim, Germany). Image quality for every loop was reviewed offline using a modified predescribed model. 13 Image quality was rated excellent for 95% to 100%, good for 70% to 95%, and substandard for <70% visualization of LV‐endocardial and myocardial structures. Loops with insufficient endocardial visualization were excluded from analysis.
For every heart cycle, end‐diastole and end‐systole were defined. Endocardial border was traced manually at end‐systole followed by a semiautomatic recognition of end‐diastolic endocardial contour. In case of inadequate tracing, manual adjustments were made until achieving a visually sufficient tracking or excluding the image from analysis.
Strain is defined as peak‐systolic strain in this study. Longitudinal strain (LS) of a4c‐, a3c‐, a2c‐views and CS at the midpapillary short‐axis level as well as corresponding SRs were measured. GLS and GL‐SR were derived from the 3 longitudinal image planes. We describe more negative strain and SR values as “higher” (defying mathematical logic) because more negative values represent increased myocardial contraction.
Abnormal strain was defined as strain values <2 SD of controls reference. Abnormality thresholds for GLS were −17.8% at rest, −19.4% at low stress, −21.5% at submaximal stress and for CS −19.0% at rest, −22.2% at low stress, and −24.3% at submaximal stress level.
Statistical Analysis
Values are given as median with interquartile range or frequencies as appropriate. The Shapiro–Wilk test was performed to test for data normality. Differences between CCSs and controls were compared using the Mann–Whitney U test. Subgroup analysis was performed to test for differences considering patient sex and patient age at cancer diagnosis. P<0.05 was considered statistically significant. Statistical analyses were performed using SPSS Statistics for Windows, version 25 (IBM Corp., Armonk, NY).
RESULTS
Patient Characteristics
Seventy‐seven patients and 50 healthy controls were included in the study. Median age in CCSs was slightly lower than in the control group. Details are shown in Table 1.
Table 1.
Clinical Characteristics
| CCSs | Controls | P value | |
|---|---|---|---|
| Age at study, y (IQR) | 16.7 (13.9; 20.9) | 19.0 (15.4; 20.3) | 0.286 |
| Female sex, n | 31/77 (40.3%) | 22/50 (44%) | 0.677 |
| Weight, kg (IQR) | 63.0 (52; 77) | 64.5 (48; 72) | 0.445 |
| Height, cm (IQR) | 168 (159; 175) | 168 (161; 183) | 0.454 |
| BMI, kg/m2 (IQR) | 21.5 (19.5; 24.9) | 20.6 (18.8; 23.5) | 0.081 |
| BSA, m2 (IQR) | 1.72 (1.55; 1.94) | 1.73 (1.47; 1.90) | 0.555 |
| Cancer specification | CCSs |
|---|---|
| Acute lymphoblastic leukemia, n | 39 (50.6%) |
| Hodgkin lymphoma, n | 12 (15.6%) |
| Nephroblastoma, n | 10 (13%) |
| Non‐Hodgkin lymphoma, n | 3 (3.9%) |
| Neuroblastoma, n | 3 (3.9%) |
| Ewing sarcoma, n | 3 (3.9%) |
| Osteosarcoma, n | 2 (2.6%) |
| Rhabdomyosarcoma, n | 2 (2.6%) |
| Soft tissue sarcoma, n | 1 (1.3%) |
| Hepatoblastoma, n | 1 (1.3%) |
| Acute myelogenous leukemia, n | 1 (1.3%) |
| Cancer diagnosis and treatment | CCSs |
|---|---|
| Age at diagnosis, y (IQR) | 7.7 (3.4; 14.8) |
| Time since last chemotherapy, y (IQR) | 7.8 (4.3; 10.7) |
| Anthracycline therapy, n | 67 (87.0%) |
| Cumulative anthracycline equivalent, mg/m2 (IQR)* | 180 (160; 210) |
| Chest radiation, n | 6 (7.8%) |
| Radiation dose, Gy (IQR) | 19.8 (19.8; 22.3) |
| Cardiomyopathy risk* | CCSs |
|---|---|
| High, n | 20 (26.0%) |
| Moderate, n | 43 (55.8%) |
| Low, n | 14 (18.2%) |
Values as median (IQR). BMI indicates body mass index; BSA, body surface area; CCSs, childhood cancer survivors; and IQR, interquartile range.
Cardiomyopathy risk according to Children's Oncology Group Long‐Term Follow‐Up Guidelines Version 5.0.
Eighty‐seven percent of CCSs had received anthracycline derivatives. The therapeutic regimen contained solely doxorubicin in 43.3% (29/67), solely daunorubicin in 1.5% (1/67), a combination of doxorubicin with daunorubicin in 52.2% (35/67), a combination of doxorubicin, daunorubicin with idarubicin in 1.5% (1/67), and a combination of daunorubicin, idarubicin, and mitoxantrone in 1.5% (1/67) of cases.
One patient had bone marrow transplantation for relapse 14 months after initial diagnosis of acute lymphoblastic leukemia.
All CCSs were asymptomatic when being asked for cardiac symptoms. Comorbidities in the CCSs group were arterial hypertension (n=2) treated with angiotensin‐converting enzyme inhibitor and angiotensin‐II‐receptor blocker, respectively, small atrial septal defect (n=1), trisomy 21 (n=1), hypothyroidism (n=6) treated with levothyroxine, mild asthma (n=1) treated with inhaled corticosteroids, epilepsy (n=1) on levetiracetam treatment, and congenital adrenal hyperplasia (n=1) with accordant hormone substitution.
Stress Test Characteristics
Exercise duration was shorter and workload lower to achieve target heart rates in CCSs compared with controls. Conventional stress echocardiographic parameters did not differ substantially between groups (Table S1). None of the controls and 1 CCS showed an impaired LV‐EF (<53%) at rest.
LV Strain and SR
The main findings are summarized in Figure 1. LV‐GLS and CS did not differ between CCSs and healthy controls at any stress stage. In CCSs with high cardiomyopathy risk, average GLS and CS values were lower at rest, low and submaximal stress, thus given the small subgroup sample size not reaching significance (Table 2).
Figure 1. Exercise stress 2‐dimensional‐speckle tracking echocardiography strain analysis in childhood cancer survivors.
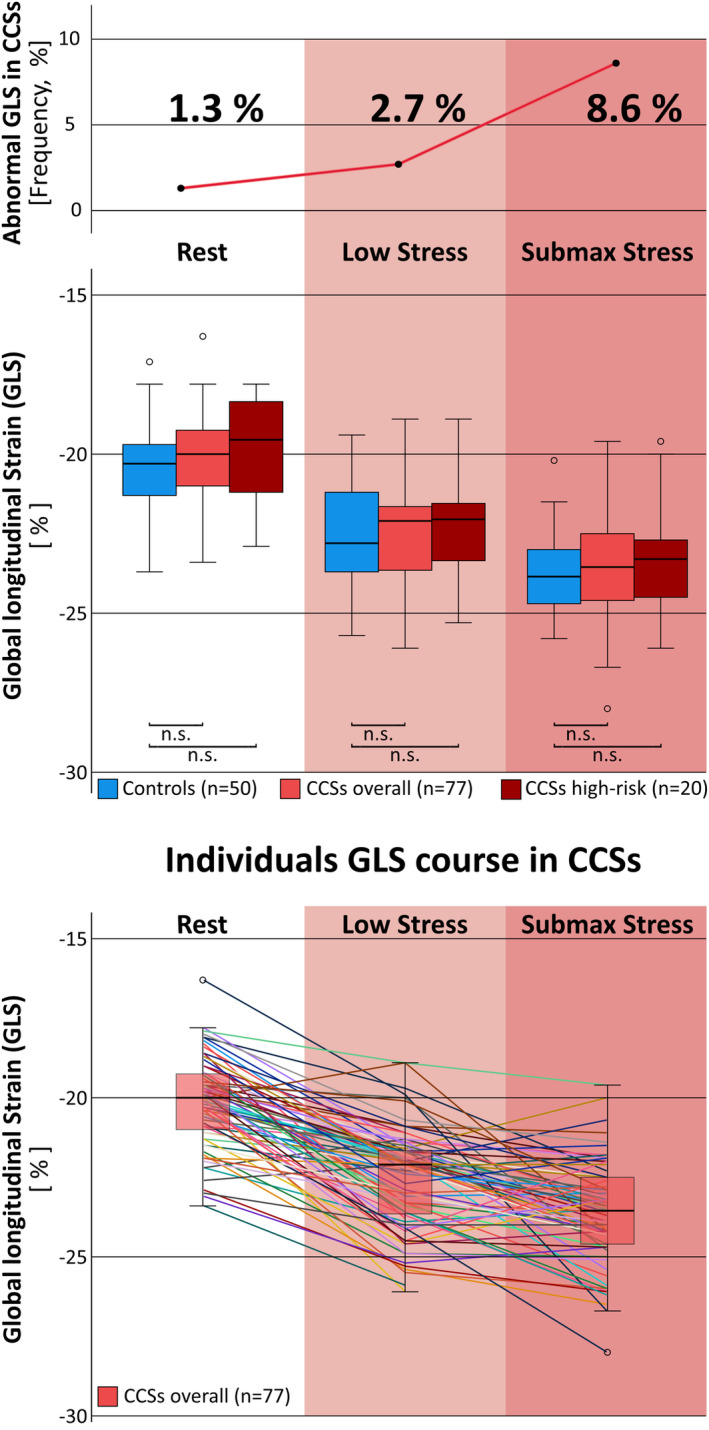
Childhood cancer survivors (CCSs), median 7.8 years postchemotherapy, showed an increasing frequency of abnormal global longitudinal strain (GLS) with higher exercise level, reflecting an impaired cardiac adaption in up to 8.6% of CCSs at submaximal stress (top). Yet, there was no relevant difference in mean GLS between CCSs and healthy controls at any stress level neither considering all CCSs (n=77) nor when stratifying for CCSs with high cardiomyopathy risk (n=20), defined as cumulative equivalent anthracycline dose ≥250 mg/m2 or chest radiation dose ≥35 Gy or anthracycline dose ≥100 mg/m2 and chest radiation ≥15 Gy (middle). Individuals GLS course at different stress levels is shown at the bottom. CCSs indicates childhood cancer survivors; GLS, global longitudinal strain; and n.s., not significant.
Table 2.
Strain Parameters
| Rest | Low stress | Submaximal stress | Poststress | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P value | P value | % ∆ to rest | P value | P value | % ∆ to rest | P value | P value | |||||
| GLS, % (IQR) | ||||||||||||
| CCS | ||||||||||||
| Total | −20.0 (−19.2; −21.1) | 0.19 | −22.1 (−21.6; −23.7) | 0.96 | 10.7 (5.6; 19.7) | 0.28 | −23.6 (−22.5; −24.6) | 0.45 | 17.6 (9.4; 24.8) | 0.81 | −20.7 (−19.5; −21.6) | 0.98 |
| High risk | −19.6 (−18.3; −21.3) | 0.16 | −22.1 (−21.5; −23.4) | 0.62 | 12.6 (5.7; 20.6) | 0.33 | −23.3 (−22.5; −24.7) | 0.38 | 15.2 (8.9; 26.0) | 0.96 | −20.9 (−19.5; −21.6) | 0.80 |
| Moderate risk | −20.0 (−19.6; −20.9) | 0.27 | −22.0 (−21.4; −23.2) | 0.42 | 9.1 (5.3; 16.4) | 0.99 | −23.6 (−22.5; −24.3) | 0.45 | 17.9 (8.6; 23.0) | 0.92 | −20.5 (−19.4; −21.6) | 0.80 |
| Controls | −20.3 (−19.6; −21.3) | −22.8 (−21.2; −23.8) | 9.8 (5.8; 15.4) | −23.9 (−23.0; −24.7) | 17.3 (13.5; 21.3) | −20.7 (−19.7; −21.5) | ||||||
| SAX‐CS, % (IQR) | ||||||||||||
| CCS | ||||||||||||
| Total | −26.2 (−23.4; −29.6) | 0.41 | −27.9 (−26.5; −30.3) | 0.14 | 6.2 (−0.1; 18.0) | 0.025* | −31.4 (−27.9; −33.1) | 0.28 | 16.9 (3.4; 28.8) | 0.028* | −26.0 (−24.2; −28.6) | 0.19 |
| High risk | −25.4 (−23.9; −29.7) | 0.89 | −27.3 (−26.3; −30.1) | 0.12 | 4.9 (−4.0; 15.5) | 0.059 | −29.9 (−27.2; −32.6) | 0.17 | 5.7 (3.0; 28.3) | 0.032* | −25.6 (−24.4; −28.5) | 0.30 |
| Moderate risk | −26.4 (−23.1; −28.5) | 0.90 | −27.9 (−26.6; −30.2) | 0.24 | 7.8 (0.9; 20.1) | 0.23 | −31.6 (−28.0; −33.3) | 0.42 | 18.0 (5.3; 33.9) | 0.32 | −25.4 (−23.6; −28.4) | 0.13 |
| Controls | −25.4 (−23.7; −28.0) | −29.0 (−26.6; −32.1) | 10.7 (5.0; 18.5) | −31.3 (−29.0; −34.2) | 23.3 (11.3; 33.3) | −27.8 (−23.7; −29.5) | ||||||
Values as median (IQR). Differences between CCSs and controls were compared using Mann–Whitney U test. %∆ indicates percentage change; CCSs, childhood cancer survivors; GLS, global longitudinal strain; IQR, interquartile range; and SAX‐CS, parasternal short axis circumferential strain.
P‐values < 0.05.
Relative change in GLS between different stress stages was comparable between groups. Relative change in CS was lower in CCSs (Table 2), most apparent in the subgroup of CCSs diagnosed with cancer <5 years of age (n=28) where GLS and CS were unaltered compared with controls but a reduced relative increase of CS from rest to low stress (median 1.5% versus 10.7%, P<0.001) and from rest to submaximal stress level (median 12.1% versus 23.2%, P=0.007) was noticeable.
Abnormal strain (defined as strain values <2 SD of controls) at rest/low stress/submaximal stress was present in 1.3%/2.7%/8.6% of CCSs regarding GLS and in 1.4%/3.0%/3.4% of CCSs regarding CS.
Six of 70 (8.6%) CCSs showed maladaptive strain at submaximal stress level, including the only individual with abnormal LV‐EF at rest. Four of 6 were female. Tumor types were acute lymphoblastic leukemia in 4/6, rhabdomyosarcoma in 1/6, and hepatoblastoma in 1/6. Considering oncological therapy details in this subgroup, 3/6 patients are considered to belong the high cardiomyopathy risk group, 2/6 to the intermediate, and 1/6 to the low‐risk group. Five of 6 patients were younger than 5 years upon cancer diagnosis. Time since last chemotherapy was median 8.1 years, ranging from 6.8 to 11.8 years.
SR increased concordantly to heart rate. There was no difference in absolute values or change of SR between groups (Table 3).
Table 3.
Strain Rate Parameters
| Rest | Low stress | Submaximal stress | Poststress | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P value | P value | % ∆ to rest | P value | P value | % ∆ to rest | P value | P value | |||||
| GLSR, 1/s (IQR) | ||||||||||||
| CCSs | ||||||||||||
| Total | −1.1 (−1.0; −1.2) | 0.69 | −1.4 (−1.3; −1.5) | 0.20 | 30.0 (20.9; 41.5) | 0.47 | −1.8 (−1.7; 2.0) | 0.21 | 72.6 (51.5; 86.9) | 0.58 | −1.2 (−1.1; −1.3) | 0.51 |
| High risk | −1.1 (−1.0; −1.2) | 0.83 | −1.4 (−1.3; −1.5) | 0.21 | 24.3 (18.2; 35.2) | 0.16 | −1.8 (−1.6; −2.0) | 0.18 | 64.9 (50.0; 76.5) | 0.22 | −1.3 (−1.1; −1.3) | 0.44 |
| Moderate risk | −1.0 (−1.0; −1.2) | 0.71 | −1.4 (−1.3; −1.5) | 0.25 | 31.0 (21.9; 40.3) | 0.48 | −1.8 (−1.7; −2.0) | 0.36 | 75.4 (51.9; 86.9) | 0.70 | −1.2 (−1.1; −1.3) | 0.37 |
| Controls | −1.1 (−1.0; −1.2) | −1.4 (−1.3; −1.6) | 33.3 (22.4; 43.7) | −1.9 (−1.8; −2.0) | 75.0 (58.8; 93.5) | −1.2 (−1.2; −1.4) | ||||||
| SAX‐CSR, 1/s (IQR) | ||||||||||||
| CCSs | ||||||||||||
| Total | −1.6 (−1.4; −1.8) | 0.92 | −2.0 (−1.8; −2.2) | 0.41 | 23.1 (5.9; 50.0) | 0.33 | −2.8 (−2.5; −3.1) | 0.56 | 63.2 (38.9; 92.9) | 0.39 | −1.7 (−1.5; −1.8) | 0.59 |
| High risk | −1.6 (−1.4; −1.8) | 0.85 | −2.0 (−1.8; −2.3) | 0.88 | 26.8 (5.9; 55.7) | 0.75 | −2.8 (−2.6; −3.0) | 0.50 | 64.7 (30.0; 100.0) | 0.58 | −1.8 (−1.5; −2.0) | 0.79 |
| Moderate risk | −1.6 (−1.4; −1.7) | 0.77 | −2.0 (−1.8; −2.2) | 0.43 | 24.9 (5.6; 55.2) | 0.68 | −2.7 (−2.5; −3.2) | 0.41 | 71.4 (42.8; 97.2) | 0.95 | −1.7 (−1.5; −1.9) | 0.88 |
| Controls | −1.6 (−1.4; −1.8) | −2.0 (−1.8; 2.4) | 30.3 (17.6; 46.2) | −2.6 (−2.4; −3.0) | 81.3 (44.4; 100.0) | −1.7 (−1.5; −1.9) | ||||||
Values as median (IQR). Differences between CCSs and controls were compared using Mann–Whitney U test. %∆ indicates percentage change; CCSs, childhood cancer survivors; GLSR, global longitudinal strain rate; IQR, interquartile range; and SAX‐CSR, parasternal short axis circumferential strain rate.
GLS was similar between female CCSs (n=31) and female controls (n=22), CS was lower at low stress level (median −27.2 versus −29.4, P=0.014), not prevailing at submaximal stress level in this subgroup.
Figure 2 shows a representative example of a4c strain curves at different stress stages from a CCS. Corresponding video loops are provided online (Video S1).
Figure 2. Strain curves of a CCS.
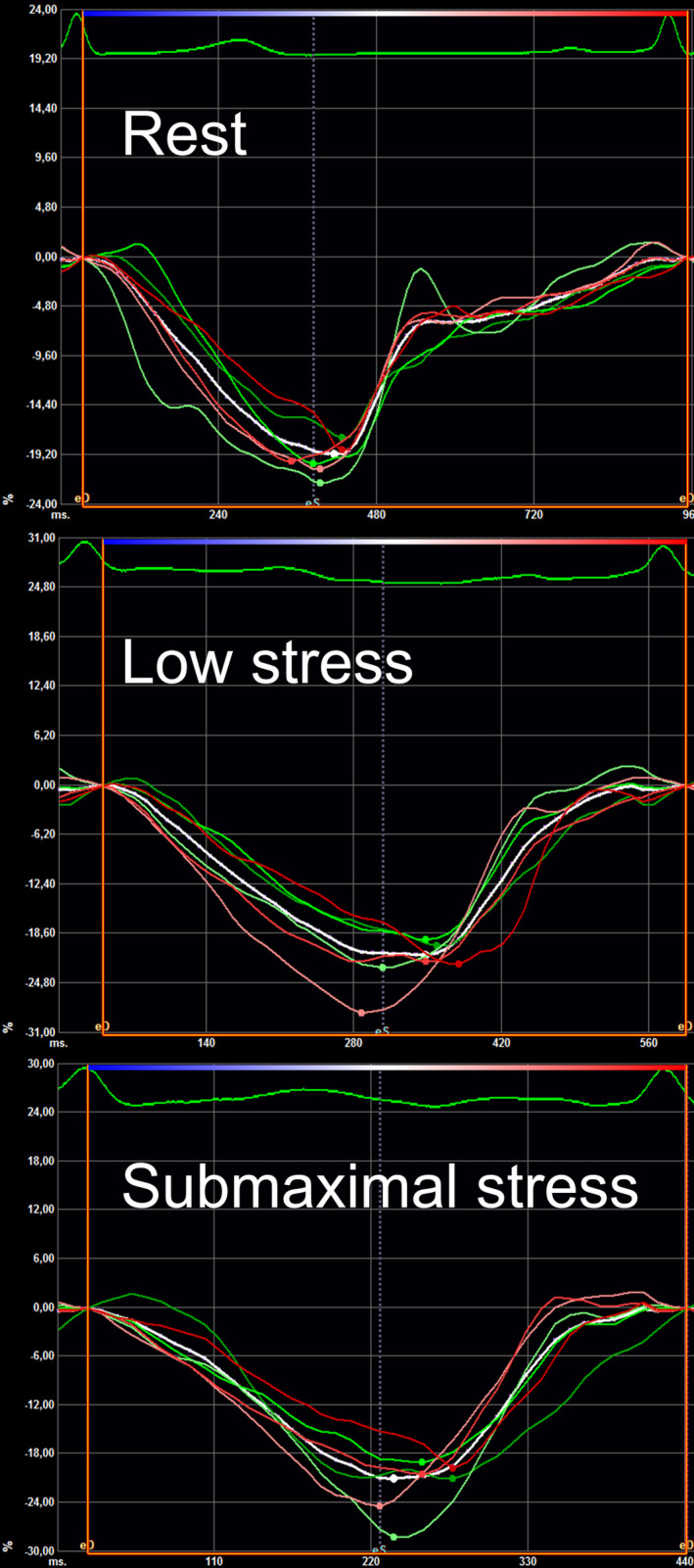
Apical 4‐chamber view strain curves from a CCS at rest (top), low (middle), and submaximal stress level (bottom). White curve indicates mean longitudinal strain. CCS indicates childhood cancer survivor.
Technical Aspects
Image quality assessment for CCSs is shown in Figure S1. Image quality dropped with increasing stress level, especially in a3c‐view. GLS in CCSs was determinable in 75/77 (97.4%) at rest, 75/77 (97.4%) at low stress, and in 70/77 (90.9%) cases at submaximal stress.
Frame rates in CCSs were 97.1±12.7 frames per second (fps) at rest, 98.4±11.0 fps at low stress, and 99.2±11.5 fps at submaximal stress, which are sufficient for strain analysis at high heart rates.
DISCUSSION
To the best of our knowledge, this is the first prospective semisupine bicycle exercise stress study in CCSs to investigate 2D‐STE strain and SR as primary end points. We found a rising prevalence of abnormal GLS with increasing stress level, suggesting impaired cardiac adaptation in individual CCSs. Mean GLS, CS, and corresponding SRs were similar in CCSs (median age at cancer diagnosis 7.7 years, median 7.8 years postchemotherapy) compared with healthy controls.
Strain and SR Measurement at Rest in CCSs
LV‐GLS is a recommended parameter in cardio‐oncologic assessment. 4 Impaired myocardial deformation measured by strain imaging precedes significant change in LV‐EF. 14
Especially during or early (<1 year) after chemotherapy, strain abnormalities have been described. 5 , 15
Considering an intermediate interval (>1 to <10 years) postchemotherapy, as best comparable to our CCSs cohort, there seems to be some compensation potential of cardiac function. Most CCSs have no apparent cardiac symptoms during that period of time post cancer therapy. Studies regarding subclinical cardiac impairment measured by strain imaging are contradictory.
In a retrospective study, Moon et al found lower GLS, CS, and circumferential strain rate in patients a mean 3.9 years after cancer diagnosis with anthracycline treatment compared with controls. Despite including only subjects with normal fractional shortening (>28%), relative difference in fractional shortening (−5.2%) was similar to relative difference in GLS (−7.4%) and CS (−8.6%) between CCSs and controls. Compared with our study, anthracycline doses were higher and heart rate differed between their groups, which might distort SR measurement. 16
Çetin et al reported subtle changes of LV strain in CCSs at a mean 21.9 months after chemotherapy compared with controls. They found impaired LS for all apical (a4c‐, a3c‐, a2c‐) views but not for GLS, which is surprising because GLS is derived from the aforementioned views. They did not find a difference in CS. The time interval since last chemotherapy in this study was shorter, partly <1 year. 17
Yu et al reported strain findings from 32 CCSs at a median 6.9 years postchemotherapy compared with healthy controls. In their study, LS (a4c) did not differ between groups, concordant with our findings. In contrast to that, they reported lower CS in CCSs. 18
Akam‐Venkata et al reported lower GLS and CS in CCSs compared with controls in a retrospective study at a median follow‐up 4.7 years after chemotherapy. 19
A cardiac magnetic resonance strain study reported 46 CCSs exposed to ≥200 mg/m2 anthracycline doses a of median 9.6 years postchemotherapy. They found lower CS and LS determined by magnetic resonance imaging in CCSs compared with controls. In their echocardiographic strain evaluation, only a difference in CS but not in LS was evident. 20
A large multicenter study by Slieker et al compared 546 CCSs to 134 healthy controls. The median age at cancer diagnosis was 3.6 years, and the median time since last anthracyclines was 7.9 years. CS was higher in CCSs than in controls. They measured CS in the mid‐ and epicardial layer, not at the subendocardial level as we did. GLS was lower in CCSs, but the absolute difference was small (0.73%) between groups. They reported abnormal LV‐EF in 0.8% of patients, similar to our findings. Abnormal LS (defined as Z‐score < −2) at rest was present in 7.7%. Yet, they reported prevalence of abnormality for a4c‐LS instead of the more robust parameter GLS. 7
Cardiac morbidity increases over a long time in CCSs. 1 Accordingly, Armstrong et al reported higher prevalence for cardiac dysfunction in a large cohort of 1820 CCSs from the SJLIFE (St. Jude Lifetime Cohort Study) with a median age of 31 years and median time since diagnosis of 23 years. Prevalence of reduced 3D‐LV‐EF was 5.8%, of impaired GLS (defined as <2 SD of mean using sex‐, age‐, and vendor‐specific strain values) 31.8%, and of impaired CS 23.1%. 6 Even when stressing the heart, we found a considerably lower abnormality prevalence in our cohort.
Cumulative anthracycline dose and cardiac radiation exposure are the most powerful predictors of cardiotoxicity. 1 , 21 Regarding a multivariate association analysis provided in the SJLIFE study, the rate ratio (RR) of reduced 3D‐LV‐EF increased stepwise accordingly to cumulative anthracycline dose (RR 1.74 for anthracycline dose <100 mg/m2, RR 7.71 for doses >400 mg/m2). This effect was much less pronounced for abnormal GLS (RR 1.38 for anthracycline dose <100 mg/m2, RR 1.73 for doses >400 mg/m2) and inconclusive for abnormal CS. 6
In a study investigating strain in 111 CCSs ≈15 years after last anthracycline therapy, lower GLS and global longitudinal strain rate were also evident, but no significant relation between GLS and high‐dose anthracycline nor between elevated NT‐pro‐BNP (N‐terminal pro‐B‐type natriuretic peptide) and lower strain values was found. 22
Outcome studies investigating strain in CCSs are still lacking but ultimately essential for implementation of STE strain echocardiography into therapeutic decision‐making in CCSs. A GLS‐guided interventional study in adult patients with cancer deciding upon initiation of cardioprotective therapy during cancer treatment by change in GLS instead of change in LV‐EF did not reveal an advantage for the GLS group at 1‐year follow‐up, 23 thus questioning the predictive value of strain measurement in cancer cohorts.
Cardiac Response to Exercise Stress in CCSs
Stress echocardiography has been performed in CCSs. 8 , 9 , 24 It is not routinely recommended for cardio‐oncologic surveillance. 4 , 9 , 25 Investigation of strain and SR in CCSs during exercise stress is scarce.
Ryerson et al performed a stress study with 80 CCSs. Patients age at diagnosis was lower and time postchemotherapy longer than in our study. They reported comparable resting and stress values for fractional shortening and EF and no remarkable findings for strain and SR at rest in patients after anthracycline therapy compared with anthracycline‐naïve controls. Exercise strain data were unavailable in their study because frame rates were insufficient for analysis at exercise heart rates. 9
Cifra et al published stress data of 100 CCSs at a mean 10.3 years postchemotherapy. Demographics including anthracycline doses and exercise heart rate were similar to our study. Concordantly to our findings, LS was unchanged at rest and during exercise between groups. They conclude that at 10‐year follow‐up CCSs have overall LV and RV systolic and diastolic myocardial exercise response comparable with that of control subjects. 26 Remarkable is the large SD for LS (determined solely from a4c‐view) in their CCSs group at peak exercise, which might point towards more heterogeneous exercise stress strain values than in our study, which was primarily focusing on the assessment of 2D‐STE LV strain.
In a study evaluating exercise response with strain imaging in 22 CCSs a median 6 years after anthracycline exposure, there was no difference in relative change of CS compared with controls. Yet, they found an impaired circumferential strain rate exercise reserve in CCSs. 27 Exercise heart rate in the study was higher compared with our study. STE SR measurement at high heart rate is at the edge of technical feasibility because high temporal resolution is crucial to avoid undersampling. 28 Therefore, STE SR stress measurements have to be interpreted carefully. Assessment of SR did not reveal relevant differences in our study.
We did not see differences in relative change for GLS but found a tendency towards impaired relative change of CS in CCSs (Table 2), especially in the subgroup of CCSs with therapy that began at <5 years of age. Assessing relative change of strain during exercise stress seems promising taking the patient's own baseline into consideration. This is especially valuable for CS where normal values are widely scattered and may improve intervendor comparability. The diagnostic value of reduced relative increase of CS in CCSs during exercise stress needs further investigation in the future.
Prevalence of Abnormality
Because prevalence of cardiac dysfunction in CCSs is low during early years and an individual's susceptibility to cardiac dysfunction after cancer treatment varies, indicating the prevalence of abnormality seems more meaningful than searching for differences by comparing mean values of groups.
A major issue when defining “abnormal” strain remains the appropriate abnormality threshold. Strain analysis, despite all harmonization efforts, still is vulnerable to technical aspects and intervendor differences. 29 , 30 Implementing a control group and using vendor‐independent software as in our study might help to reduce this bias.
Regarding CCSs with abnormal GLS at submaximal stress level (n=6) in our study, they disproportionately often belong to the subgroup with the highest therapy intensity (50% in GLS abnormality subgroup versus 26% in CCSs overall), age <5 years at cancer diagnosis (83.3% in GLS abnormality subgroup versus 36.4% in CCSs overall), and female sex (66.7% in GLS abnormality subgroup versus 40.2% in CCSs overall). All are known risk factors for development of cancer therapy–related cardiomyopathy. 31
Study Limitations
Limitation by the small number of cases, especially considering the high cardiomyopathy risk subgroup, is obvious. One could underestimate anthracycline exposure in this study because equivalent anthracycline doses were calculated according to Children's Oncology Group Long‐Term Follow‐Up Guidelines Version 5 where daunorubicin dose is factored by ×0.5 instead of ×1 as in most earlier studies.
The highest stress level in this study was restricted to submaximal exercise stress, because of physical (breathing, motion artifacts) and technical aspects (frame rate), to guarantee adequate image quality for strain analysis. Thus subtle cardiac impairment might be underestimated because cardiac reserve was not fully challenged.
To guarantee as reliable as possible strain data, we applied a prospective study design with strain as primary end point, adhered to a standardized acquisition protocol, used vendor‐independent analysis software, and meticulously optimized technical aspects such as image quality and frame rate to reduce bias.
A mean frame rate of 99.2±11.5 fps at submaximal stress can be considered sufficient for strain analysis at the submaximal stress heart rates in this study, exceeding the recommended 30 frames per heart cycle. 32 Yet, strain rate measurements at the highest stress stage in our study are at the edge of technical feasibility with the risk of undersampling because of a limited temporal resolution and therefore have to be interpreted carefully. 28
In this study, several statistical hypotheses were tested, with explorative character in the subgroup comparisons. Therefore, statistical significance, especially in the subgroup comparisons, has to be interpreted with caution.
CONCLUSIONS
In this prospective 2D‐STE strain study, we found a rising prevalence of abnormal GLS with increasing stress level in CCSs, reflecting an impaired cardiac adaptation in 8.6% of CCSs at submaximal stress. There were no major differences in mean GLS, CS, and corresponding SRs between CCSs and healthy controls during semisupine submaximal exercise stress.
With outcome data lacking routine application of 2D‐STE, strain stress testing for cardio‐oncologic surveillance does not seem reasonable at this point. In clinical routine this low side‐effect, noninvasive method may be considered in select patients with borderline or inconclusive findings to obtain more diagnostic information and possibly shorten the follow‐up interval.
Further research is needed, preferably in cohorts with higher pretest probability (ie, subgroups with highest anthracycline/radiation exposure, younger age at diagnosis, or later postchemotherapy) to identify cohorts potentially benefiting most from the method and correlate findings with outcome data.
Additionally, investigating intraindividual relative change of strain either between different stress stages or in longitudinal follow‐up should be a special focus of further research because it incorporates the patient's own baseline, possibly indicating subtle impairment before absolute values are considered pathologic. This also could help to improve intervendor comparability.
Sources of Funding
This study was supported by German Society of Pediatric Cardiology (DGPK), Willy Robert Pitzer Foundation, and Stiftung Valentina.
Disclosures
None.
Supporting information
Table S1
Figure S1
Video S1
Acknowledgments
We acknowledge Marion Frengel (MD) and Judit Kappe (MD), Division of Pediatric Oncology, University Children's Hospital Ulm, for their advice regarding oncologic questions.
Presented in part at the 54th Annual Meeting of the German Society for Pediatric Cardiology (DGPK), in Hamburg, Germany, February 19 to 22, 2022, and published in abstract form [Thorac Cardiovasc Surg. 2022;70(S 02):S67–S103 or 10.1055/s‐0042‐1743015].
For Sources of Funding and Disclosures, see page 11.
References
- 1. Mulrooney DA, Yeazel MW, Kawashima T, Mertens AC, Mitby P, Stovall M, Donaldson SS, Green DM, Sklar CA, Robison LL, et al. Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: retrospective analysis of the childhood cancer survivor study cohort. BMJ. 2009;339:b4606. doi: 10.1136/bmj.b4606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Mertens AC, Liu Q, Neglia JP, Wasilewski K, Leisenring W, Armstrong GT, Robison LL, Yasui Y. Cause‐specific late mortality among 5‐year survivors of childhood cancer: the childhood cancer survivor study. J Natl Cancer Inst. 2008;100:1368–1379. doi: 10.1093/jnci/djn310 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Adams MJ, Lipshultz SE. Pathophysiology of anthracycline‐ and radiation‐associated cardiomyopathies: implications for screening and prevention. Pediatr Blood Cancer. 2005;44:600–606. doi: 10.1002/pbc.20352 [DOI] [PubMed] [Google Scholar]
- 4. Plana JC, Galderisi M, Barac A, Ewer MS, Ky B, Scherrer‐Crosbie M, Ganame J, Sebag IA, Agler DA, Badano LP, et al. Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2014;15:1063–1093. doi: 10.1093/ehjci/jeu192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Pignatelli RH, Ghazi P, Reddy SC‐B, Thompson P, Cui Q, Castro J, Okcu MF, Jefferies JL. Abnormal myocardial strain indices in children receiving anthracycline chemotherapy. Pediatr Cardiol. 2015;36:1610–1616. doi: 10.1007/s00246-015-1203-8 [DOI] [PubMed] [Google Scholar]
- 6. Armstrong GT, Joshi VM, Ness KK, Marwick TH, Zhang N, Srivastava D, Griffin BP, Grimm RA, Thomas J, Phelan D, et al. Comprehensive echocardiographic detection of treatment‐related cardiac dysfunction in adult survivors of childhood cancer. J Am Coll Cardiol. 2015;65:2511–2522. doi: 10.1016/j.jacc.2015.04.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Slieker MG, Fackoury C, Slorach C, Hui W, Friedberg MK, Fan CS, Manlhiot C, Dillenburg R, Kantor P, Mital S, et al. Echocardiographic assessment of cardiac function in pediatric survivors of anthracycline‐treated childhood cancer. Circ Cardiovasc Imaging. 2019;12:e008869. doi: 10.1161/CIRCIMAGING.119.008869 [DOI] [PubMed] [Google Scholar]
- 8. Smibert E, Carlin JB, Vidmar S, Wilkinson LC, Newton M, Weintraub RG. Exercise echocardiography reflects cumulative anthracycline exposure during childhood. Pediatr Blood Cancer. 2004;42:556–562. doi: 10.1002/pbc.20016 [DOI] [PubMed] [Google Scholar]
- 9. Ryerson AB, Border WL, Wasilewski‐Masker K, Goodman M, Meacham L, Austin H, Mertens AC. Assessing anthracycline‐treated childhood cancer survivors with advanced stress echocardiography. Pediatr Blood Cancer. 2015;62:502–508. doi: 10.1002/pbc.25328 [DOI] [PubMed] [Google Scholar]
- 10. von Scheidt F, Kiesler V, Kaestner M, Bride P, Kramer J, Apitz C. Left ventricular strain and strain rate during submaximal semisupine bicycle exercise stress echocardiography in healthy adolescents and young adults: systematic protocol and reference values. J Am Soc Echocardiogr. 2020;33:848–857.e841. doi: 10.1016/j.echo.2019.12.015 [DOI] [PubMed] [Google Scholar]
- 11. Long‐term follow‐up guidelines for survivors of childhood, adolescent, and young adult cancers, version 5.0. Children's Oncology Group; 2018. Available at: http://www.survivorshipguidelines.org. Accessed July 26, 2021. [Google Scholar]
- 12. Armenian SH, Hudson MM, Mulder RL, Chen MH, Constine LS, Dwyer M, Nathan PC, Tissing WJE, Shankar S, Sieswerda E, et al. Recommendations for cardiomyopathy surveillance for survivors of childhood cancer: a report from the international late effects of childhood cancer guideline harmonization group. Lancet Oncol. 2015;16:e123–e136. doi: 10.1016/s1470-2045(14)70409-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Johri AM, Chitty DW, Hua L, Marincheva G, Picard MH. Assessment of image quality in real time three‐dimensional dobutamine stress echocardiography: an integrated 2D/3D approach. Echocardiography. 2015;32:496–507. doi: 10.1111/echo.12692 [DOI] [PubMed] [Google Scholar]
- 14. Thavendiranathan P, Poulin F, Lim KD, Plana JC, Woo A, Marwick TH. Use of myocardial strain imaging by echocardiography for the early detection of cardiotoxicity in patients during and after cancer chemotherapy: a systematic review. J Am Coll Cardiol. 2014;63:2751–2768. doi: 10.1016/j.jacc.2014.01.073 [DOI] [PubMed] [Google Scholar]
- 15. Mavinkurve‐Groothuis AM, Marcus KA, Pourier M, Loonen J, Feuth T, Hoogerbrugge PM, de Korte CL, Kapusta L. Myocardial 2D strain echocardiography and cardiac biomarkers in children during and shortly after anthracycline therapy for acute lymphoblastic leukaemia (ALL): a prospective study. Eur Heart J Cardiovasc Imaging. 2013;14:562–569. doi: 10.1093/ehjci/jes217 [DOI] [PubMed] [Google Scholar]
- 16. Moon TJ, Miyamoto SD, Younoszai AK, Landeck BF. Left ventricular strain and strain rates are decreased in children with normal fractional shortening after exposure to anthracycline chemotherapy. Cardiol Young. 2014;24:854–865. doi: 10.1017/S1047951113001182 [DOI] [PubMed] [Google Scholar]
- 17. Cetin S, Babaoglu K, Basar EZ, Deveci M, Corapcioglu F. Subclinical anthracycline‐induced cardiotoxicity in long‐term follow‐up of asymptomatic childhood cancer survivors: assessment by speckle tracking echocardiography. Echocardiography. 2018;35:234–240. doi: 10.1111/echo.13743 [DOI] [PubMed] [Google Scholar]
- 18. Yu W, Li SN, Chan GC, Ha SY, Wong SJ, Cheung YF. Transmural strain and rotation gradient in survivors of childhood cancers. Eur Heart J Cardiovasc Imaging. 2013;14:175–182. doi: 10.1093/ehjci/jes143 [DOI] [PubMed] [Google Scholar]
- 19. Akam‐Venkata J, Kadiu G, Galas J, Lipshultz SE, Aggarwal S. Left ventricle segmental function in childhood cancer survivors using speckle‐tracking echocardiography. Cardiol Young. 2019;29:1494–1500. doi: 10.1017/S1047951119002622 [DOI] [PubMed] [Google Scholar]
- 20. Toro‐Salazar OH, Gillan E, O'Loughlin MT, Burke GS, Ferranti J, Stainsby J, Liang B, Mazur W, Raman SV, Hor KN. Occult cardiotoxicity in childhood cancer survivors exposed to anthracycline therapy. Circ Cardiovasc Imaging. 2013;6:873–880. doi: 10.1161/CIRCIMAGING.113.000798 [DOI] [PubMed] [Google Scholar]
- 21. Armstrong GT, Liu Q, Yasui Y, Neglia JP, Leisenring W, Robison LL, Mertens AC. Late mortality among 5‐year survivors of childhood cancer: a summary from the childhood cancer survivor study. J Clin Oncol. 2009;27:2328–2338. doi: 10.1200/JCO.2008.21.1425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Mavinkurve‐Groothuis AM, Groot‐Loonen J, Marcus KA, Bellersen L, Feuth T, Bokkerink JP, Hoogerbrugge PM, de Korte C, Kapusta L. Myocardial strain and strain rate in monitoring subclinical heart failure in asymptomatic long‐term survivors of childhood cancer. Ultrasound Med Biol. 2010;36:1783–1791. doi: 10.1016/j.ultrasmedbio.2010.08.001 [DOI] [PubMed] [Google Scholar]
- 23. Thavendiranathan P, Negishi T, Somerset E, Negishi K, Penicka M, Lemieux J, Aakhus S, Miyazaki S, Shirazi M, Galderisi M, et al. Strain‐guided management of potentially cardiotoxic cancer therapy. J Am Coll Cardiol. 2021;77:392–401. doi: 10.1016/j.jacc.2020.11.020 [DOI] [PubMed] [Google Scholar]
- 24. De Souza AM, Potts JE, Potts MT, De Souza ES, Rowland TW, Pritchard SL, Sandor GG. A stress echocardiography study of cardiac function during progressive exercise in pediatric oncology patients treated with anthracyclines. Pediatr Blood Cancer. 2007;49:56–64. doi: 10.1002/pbc.21122 [DOI] [PubMed] [Google Scholar]
- 25. Sieswerda E, Kremer LC, Vidmar S, De Bruin ML, Smibert E, Sjoberg G, Cheung MM, Weintraub RG. Exercise echocardiography in asymptomatic survivors of childhood cancer treated with anthracyclines: a prospective follow‐up study. Pediatr Blood Cancer. 2010;54:579–584. doi: 10.1002/pbc.22371 [DOI] [PubMed] [Google Scholar]
- 26. Cifra B, Chen CK, Fan CS, Slorach C, Manlhiot C, McCrindle BW, Dragulescu A, Redington AN, Friedberg MK, Nathan PC, et al. Dynamic myocardial response to exercise in childhood cancer survivors treated with anthracyclines. J Am Soc Echocardiogr. 2018;31:933–942. doi: 10.1016/j.echo.2018.02.003 [DOI] [PubMed] [Google Scholar]
- 27. Kaneko S, Tham EB, Haykowsky MJ, Spavor M, Khoo NS, Mackie AS, Smallhorn JF, Thompson RB, Nelson MD. Impaired left ventricular reserve in childhood cancer survivors treated with anthracycline therapy. Pediatr Blood Cancer. 2016;63:1086–1090. doi: 10.1002/pbc.25933 [DOI] [PubMed] [Google Scholar]
- 28. Lord RN, George K, Jones H, Somauroo J, Oxborough D. Reproducibility and feasibility of right ventricular strain and strain rate (SR) as determined by myocardial speckle tracking during high‐intensity upright exercise: a comparison with tissue Doppler‐derived strain and SR in healthy human hearts. Echo Res Pract. 2014;1:31–41. doi: 10.1530/ERP-14-0011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Nagata Y, Takeuchi M, Mizukoshi K, Wu VC, Lin FC, Negishi K, Nakatani S, Otsuji Y. Intervendor variability of two‐dimensional strain using vendor‐specific and vendor‐independent software. J Am Soc Echocardiogr. 2015;28:630–641. doi: 10.1016/j.echo.2015.01.021 [DOI] [PubMed] [Google Scholar]
- 30. Voigt JU, Pedrizzetti G, Lysyansky P, Marwick TH, Houle H, Baumann R, Pedri S, Ito Y, Abe Y, Metz S, et al. Definitions for a common standard for 2D speckle tracking echocardiography: consensus document of the EACVI/ASE/Industry Task Force to standardize deformation imaging. Eur Heart J Cardiovasc Imaging. 2015;16:1–11. doi: 10.1093/ehjci/jeu184 [DOI] [PubMed] [Google Scholar]
- 31. Chow EJ, Chen Y, Kremer LC, Breslow NE, Hudson MM, Armstrong GT, Border WL, Feijen EAM, Green DM, Meacham LR, et al. Individual prediction of heart failure among childhood cancer survivors. J Clin Oncol. 2015;33:394–402. doi: 10.1200/jco.2014.56.1373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Rosner A, Barbosa D, Aarsaether E, Kjonas D, Schirmer H, D'Hooge J. The influence of frame rate on two‐dimensional speckle‐tracking strain measurements: a study on silico‐simulated models and images recorded in patients. Eur Heart J Cardiovasc Imaging. 2015;16:1137–1147. doi: 10.1093/ehjci/jev058 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1
Figure S1
Video S1


