Abstract
A 60-year-old female presented with headaches, blurry vision, diplopia, and dizziness for six weeks. Her workup revealed an elevated hematocrit, thrombocytosis, high ferritin, and normal erythropoietin. She was diagnosed with polycythemia vera with the JAK2 V617F mutation. The patient underwent magnetic resonance venography, which showed left-sided sigmoid venous thrombosis. She was placed on low-molecular-weight heparin, with a plan to transition to oral anticoagulation after four weeks and repeat imaging in three months to assess for resolution. Thrombotic events may occur in patients with polycythemia vera, and a JAK2 mutation further heightens that risk. Even so, intracranial venous thrombosis is not among the most common events, and it should be kept in the differential for any patient with myeloproliferative neoplasms presenting with new neurological symptoms.
1. Introduction
The risk of venous thromboembolism in myeloproliferative neoplasms (MPN), including polycythemia vera, is well established and not uncommon. However, primary venous thromboembolism occurring intracranially is rare. A large retrospective study found a disease-specific frequency of 1.2% for cerebral venous thrombosis (CVT) in patients with polycythemia vera [1]. Women seem to be at higher risk for those events, as are patients with the Janus kinase 2 (JAK2) mutation, and those presenting with leukocytosis [2–4]. Headaches are the leading symptom, but visual disturbances, nausea and vomiting, hemiparesis, seizures, and vertigo have also been reported [1, 5].
Here we report a case of sigmoid venous thrombosis in a patient recently diagnosed with polycythemia vera with the JAK2 V617F mutation, causing headaches and diplopia.
2. Case Presentation
A 60-year-old female with a past medical history of ductal carcinoma in situ after lumpectomy and radiation and currently on hormonal therapy with an aromatase inhibitor presented with six weeks of pinpoint frontal headaches, blurry vision, diplopia, and dizziness. Her symptoms worsened over the past two weeks prior to her presentation. Her past medical history is also remarkable for tremors, for which she was seeing a neurologist, obesity, and obstructive sleep apnea on CPAP (continuous positive airway pressure). There are no additional cardiovascular risk factors in her past medical history, and she has never smoked. She is a frequent blood donor, as she claims that her blood counts are always on the higher side.
She started taking topiramate and a trial of indomethacin for the headaches, with only modest improvement. As an outpatient, her hematocrit was found to be elevated at 49.8% (Table 1), and she was referred to a hematologist. Her erythropoietin level was 4.4 mIU/mL (reference range 2.6–18.5). She was found to have a mutation on JAK2 V617F, measured at 16% of total JAK2 DNA (Table 2). Retrospectively, it was discovered that the patient has a sister with polycythemia vera.
Table 1.
Baseline complete blood count.
| Hemoglobin | 16.6 g/dL |
| Hematocrit | 49.8% |
| RBCs | 5.28 M/uL |
| MCV | 94.3 fL |
| MCH | 31.4 pg |
| MCHC | 33.3 g/dL |
| Platelets | 497 K/uL |
| MPV | 10.7 fI |
| RDW | 13.8% |
| WBC | 10.17 K/uL |
RBC: red blood cell; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; MPV: mean platelet volume; RDW: red cell distribution width; WBC: white blood cell.
Table 2.
Hematology workup.
| Ferritin | 263 ug/L |
| Iron | 119 ug/dL |
| Iron saturation | 51% |
| TIBC | 234 ug/dL |
| PT | 13 sec |
| PT-INR | 1.0 |
| PTT | 26.1 sec |
| EPO | 4.4 mIU/mL |
| JAK2V617F | Positive; 16% |
TIBC: total iron binding capacity; PT: prothrombin time; INR: international normalized ratio; PTT: partial thromboplastin time; EPO: erythropoietin; JAK2: Janus kinase 2.
The patient was scheduled to initiate phlebotomy treatment with a plan to start hydroxyurea afterwards. She underwent one session of phlebotomy without complications.
On arrival at the hospital, the patient's vital signs were normal. On physical examination, she was awake, alert, and oriented. Extraocular movements were intact with no nystagmus. There was no rest tremor, no dysmetria, and no intention tremor on finger-nose-finger testing. Ambulation was normal. No lymphadenopathy was found. There was no pruritus, no erythromelalgia, no ecchymosis, petechiae, or rash. Respiratory, cardiovascular, and abdominal exams were also unremarkable.
A brain magnetic resonance imaging was ordered and failed to reveal an acute change or finding to explain her symptoms. Consequently, magnetic resonance venography (MRV) was obtained, revealing T1 hyperintensity with a lack of enhancement in the lateral aspect of the left proximal sigmoid sinus, compatible with dural venous sinus thrombosis, with likely extension to the left jugular bulb (Figure 1).
Figure 1.
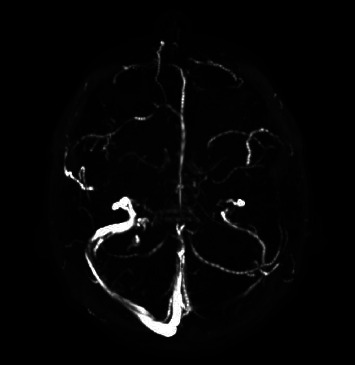
Magnetic resonance venography: T1 hyperintensity with lack of enhancement in the lateral aspect of the left proximal sigmoid sinus.
The patient was admitted and started on low-molecular-weight heparin. It was suggested that her double vision might have been a partial CN VI palsy secondary to increased intracranial pressure, and a trial of IV acetazolamide (250 mg BID) was given. The patient's headaches and visual disturbances gradually improved, and she was discharged on enoxaparin for four weeks, planning to transition to oral anticoagulation with either direct oral agents or warfarin for three to twelve months, at the discretion of her hematologist. There was also a plan to repeat the MRV in three months to assess its resolution. She had a follow-up two weeks after discharge, and the patient was still experiencing some symptoms, even though they were now milder.
3. Discussion
The most common venous thrombotic events in patients with MPNs are deep vein thrombosis, pulmonary embolism, splanchnic vein thrombosis, and superficial thrombophlebitis. Those events are usually found shortly after the diagnosis of either polycythemia vera (PV) or essential thrombocythemia (ET) is made [6]. Patients at risk for thrombosis are those over 60 years of age with a previous history of thrombosis and the JAK2V617F mutation. Additionally, patients with severe thrombocytosis may be at risk for bleeding, as it can be associated with acquired von Willebrand syndrome [7]. Our patient also has a sister with a diagnosis of PV, which raises the possibility of familial MPNs. This is of importance because in patients with either familial PV or ET, the JAK2V617F mutation has been associated with a higher risk of thrombotic events than in those with nonfamilial MPNs, despite bearing a similar prognosis [8]. Finally, in our case, the patient was prescribed an aromatase inhibitor for the treatment of breast cancer. Those agents are not usually associated with thrombotic events, and hence there is a less likely chance that this therapy is associated with the patient's current presentation [9].
3.1. Pathogenesis
The pathogenesis leading up to thrombotic events in polycythemia vera is not entirely clear. Blood hyperviscosity is thought to be one of the causes of thrombotic events both in neoplastic and in nonclonal exaggerated erythropoiesis [10]. However, there are likely other implicating factors, given that polycythemia itself would not explain higher rates of thrombosis seen in other MPNs such as essential thrombocythemia. It is postulated that patients with PV present with higher prothrombotic gene expression, including F3, IL10, VEGFA, LDHA, and SLC2A1. These genes were found to correlate positively with a JAK2 mutation in PV [11]. Leukocytosis may also be an implicating cause. The biological plausibility lies in the fact that patients with PV express higher levels of beta-2 integrin, CD11b, and leukocyte alkaline phosphatase, ultimately leading to increased activation of neutrophils. These activated polymorphonuclear cells interact with platelets, impairing normal hemostasis [12]. A meta-analysis confirmed that leukocytosis is associated with increased thrombotic events in PV, but a subgroup analysis revealed that this was true for arterial rather than venous events. Furthermore, the study could not establish a cutoff for white blood cell counts that portended a higher risk for thrombosis [13].
3.2. Epidemiology, Clinical Manifestations, and Diagnosis
A large observational study found the prevalence of venous thrombotic events, regardless of location, in patients with polycythemia vera to be 9% [2]. Another study identified a prevalence of 1.2% for CVT in patients with PV. The most common sites for cerebral sinus thrombosis in this series of cases were the transverse sinus, the sagittal sinus, and the sigmoid sinus, respectively [1] (Figure 2). Headache and blurry vision are among the leading symptoms; however, nausea and vomiting, diplopia, and seizures have also been reported [1, 5]. CVT should also raise concern for signs and symptoms of intracranial hypertension, such as associated papilledema [14]. Most recent guidelines recommend screening oncologic patients for cerebral venous thrombosis. Furthermore, they state that either magnetic resonance or computed tomography angiography are the tests of choice to diagnose CVT [15].
Figure 2.

Anatomy of the cerebral venous system and sigmoid sinus thrombosis.
3.3. Management and Prognosis
Few studies address treatment of CVT, specifically in the subgroup of patients with MPNs. In general, low molecular weight heparin (LMWH) seems to be a better option for acute anticoagulation than unfractionated heparin (UFH) when considering adverse events such as major bleeding and thrombotic complications [16]. Heparin should be used in the acute setting, defined as a period short after the diagnosis, when intracranial hypertension and brain herniation may occur [17]. There is no high-quality evidence assessing the duration of anticoagulation. Hence, it is reasonable to maintain oral therapy for a period of 3 to 12 months [15]. The use of direct oral anticoagulants (DOACs) in CVT appears to be a safe choice when compared to coumarins [18, 19]. A large multicenter retrospective study assessed DOACs for treatment of CVT of any cause. It showed no statistically significant difference between DOACs and warfarin regarding outcomes such as recurrence, death, and venous recanalization. It also showed a higher risk of major hemorrhage with warfarin when compared to DOACs [19]. Even though cerebral sinus thrombosis does not seem to affect mortality rates, it might be associated with morbidity as some patients may present persistent neurological symptoms [1]. As for both arterial and venous thromboembolic episodes in the MPN population, observational studies suggest that DOACs seem both effective and safe when compared with vitamin K antagonists [20].
This is a case of cerebral sinus thrombosis in a patient with the JAK2 V617F mutation of polycythemia vera. Even though thrombosis is a well-established complication of myeloproliferative neoplasms, sigmoid thrombosis is not particularly common. The patient presented with headaches and diplopia, signs of intracranial hypertension, and was diagnosed using the gold-standard method, a magnetic resonance venography. Patients with CVT should be treated with anticoagulation for three to 12 months, starting with a heparinoid product in the acute setting and transitioning to oral anticoagulation for the long-term outpatient treatment. This case reinforces that CVT should be kept on the differential for patients with MPNs and new neurological symptoms.
Abbreviations
- CVT:
Cerebral venous thrombosis
- DOAC:
Direct oral anticoagulant
- JAK2:
Janus kinase 2
- LMWH:
Low-molecular-weight heparin
- MPN:
Myeloproliferative neoplasm
- MRV:
Magnetic resonance venography
- PV:
Polycythemia vera
- UFH:
Unfractionated heparin.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Gangat N., Guglielmelli P., Betti S., et al. Cerebral venous thrombosis and myeloproliferative neoplasms: a three-center study of 74 consecutive cases. American Journal of Hematology . 2021;96(12):1580–1586. doi: 10.1002/ajh.26336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tefferi A., Rumi E., Finazzi G., et al. Survival and prognosis among 1545 patients with contemporary polycythemia vera: an international study. Leukemia . 2013;27(9):1874–1881. doi: 10.1038/leu.2013.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Landolfi R., Di Gennaro L., Barbui T., et al. Leukocytosis as a major thrombotic risk factor in patients with polycythemia vera. Blood . 2007;109(6):2446–2452. doi: 10.1182/blood-2006-08-042515. [DOI] [PubMed] [Google Scholar]
- 4.Carobbio A., Thiele J., Passamonti F., et al. Risk factors for arterial and venous thrombosis in WHO-defined essential thrombocythemia: an international study of 891 patients. Blood . 2011;117(22):5857–5859. doi: 10.1182/blood-2011-02-339002. [DOI] [PubMed] [Google Scholar]
- 5.Shen H. X., Huang X. Q., Fan C. Q., Cao Y. X., Zhao H., Duan J. G. Cerebral venous sinus thrombosis in polycythemia vera patients with JAK2V617F mutation. Chinese Medical Journal . 2021;134(10):1251–1253. doi: 10.1097/CM9.0000000000001484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Griesshammer M., Kiladjian J. J., Besses C. Thromboembolic events in polycythemia vera. Annals of Hematology . 2019;98(5):1071–1082. doi: 10.1007/s00277-019-03625-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tefferi A., Barbui T. Polycythemia vera and essential thrombocythemia: 2021 update on diagnosis, risk-stratification and management. American Journal of Hematology . 2020;95(12):1599–1613. doi: 10.1002/ajh.26008. [DOI] [PubMed] [Google Scholar]
- 8.Malak S., Labopin M., Saint-Martin C., Bellanne-Chantelot C., Najman A., French Group of Familial Myeloproliferative Disorders Long term follow up of 93 families with myeloproliferative neoplasms: life expectancy and implications of JAK2V617F in the occurrence of complications. Blood Cells, Molecules, and Diseases . 2012;49(3-4):170–176. doi: 10.1016/j.bcmd.2012.06.004. [DOI] [PubMed] [Google Scholar]
- 9.Dombernowsky P., Smith I., Falkson G., et al. Letrozole, a new oral aromatase inhibitor for advanced breast cancer: double-blind randomized trial showing a dose effect and improved efficacy and tolerability compared with megestrol acetate. Journal of Clinical Oncology . 1998;16(2):453–461. doi: 10.1200/jco.1998.16.2.453. [DOI] [PubMed] [Google Scholar]
- 10.Marchioli R., Finazzi G., Specchia G., et al. Cardiovascular events and intensity of treatment in polycythemia vera. New England Journal of Medicine . 2013;368(1):22–33. doi: 10.1056/nejmoa1208500. [DOI] [PubMed] [Google Scholar]
- 11.Gangaraju R., Song J., Kim S. J., et al. Thrombotic, inflammatory, and HIF-regulated genes and thrombosis risk in polycythemia vera and essential thrombocythemia. Blood Advances . 2020;4(6):1115–1130. doi: 10.1182/bloodadvances.2019001379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Landolfi R., Di Gennaro L., Falanga A. Thrombosis in myeloproliferative disorders: pathogenetic facts and speculation. Leukemia . 2008;22(11):2020–2028. doi: 10.1038/leu.2008.253. [DOI] [PubMed] [Google Scholar]
- 13.Carobbio A., Ferrari A., Masciulli A., Ghirardi A., Barosi G., Barbui T. Leukocytosis and thrombosis in essential thrombocythemia and polycythemia vera: a systematic review and meta-analysis. Blood Advances . 2019;3(11):1729–1737. doi: 10.1182/bloodadvances.2019000211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Biousse V., Ameri A., Bousser M. G. Isolated intracranial hypertension as the only sign of cerebral venous thrombosis. Neurology . 1999;53(7):1537–1542. doi: 10.1212/wnl.53.7.1537.10534264 [DOI] [PubMed] [Google Scholar]
- 15.Ferro J. M., Bousser M. G., Canhão P., et al. European stroke organization guideline for the diagnosis and treatment of cerebral venous thrombosis—endorsed by the European academy of neurology. European Journal of Neurology . 2017;24(10):1203–1213. doi: 10.1111/ene.13381. [DOI] [PubMed] [Google Scholar]
- 16.Silvis S. M., de Sousa D. A., Ferro J. M., Coutinho J. M. Cerebral venous thrombosis. Nature Reviews Neurology . 2017;13(9):555–565. doi: 10.1038/nrneurol.2017.104. [DOI] [PubMed] [Google Scholar]
- 17.Ulivi L., Squitieri M., Cohen H., Cowley P., Werring D. J. Cerebral venous thrombosis: a practical guide. Practical Neurology . 2020;20(5):356–367. doi: 10.1136/practneurol-2019-002415. [DOI] [PubMed] [Google Scholar]
- 18.Ferro J. M., Coutinho J. M., Dentali F., et al. Safety and efficacy of dabigatran etexilate vs. dose-adjusted warfarin in patients with cerebral venous thrombosis: a randomized clinical trial. JAMA Neurology . 2019;76(12):1457–1465. doi: 10.1001/jamaneurol.2019.2764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yaghi S., Shu L., Bakradze E., et al. Direct oral anticoagulants versus warfarin in the treatment of cerebral venous thrombosis (ACTION-CVT): a multicenter international study. Stroke . 2022;53(3):728–738. doi: 10.1161/STROKEAHA.121.037541. [DOI] [PubMed] [Google Scholar]
- 20.Hamulyák E. N., Daams J. G., Leebeek F. W. G., et al. A systematic review of antithrombotic treatment of venous thromboembolism in patients with myeloproliferative neoplasms. Blood Advances . 2021;5(1):113–121. doi: 10.1182/bloodadvances.2020003628. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


