Abstract
Pleural mesothelioma (PM) is an aggressive malignancy with poor prognosis. Although histology and pathologic stage are important prognostic factors, better prognostic biomarkers are needed. The ribosomal protein S6 is a downstream target of the phosphatidylinositol 3-kinase (PI3K) pathway involved in protein synthesis and cell proliferation. In previous studies, low phosphorylated S6 (pS6) immunoreactivity was significantly correlated with longer progression-free survival (PFS) and overall survival (OS) in PM patients. We aimed to correlate pS6 expression to clinical data in a large multi-centre PM cohort as part of the European Thoracic Oncology Platform (ETOP) Mesoscape project. Tissue Micro Arrays (TMAs) of PM were constructed and expression of pS6 was evaluated by a semi-quantitatively aggregate H-score. Expression results were correlated to patient characteristics as well as OS/PFS. pS6 IHC results of 364 patients from 9 centres, diagnosed between 1999 and 2017 were available. The primary histology of included tumours was epithelioid (70.3%), followed by biphasic (24.2%) and sarcomatoid (5.5%). TMAs included both treatment-naïve and tumour tissue taken after induction chemotherapy. High pS6 expression (181 patients with H-score>1.41) was significantly associated with less complete resection. In the overall cohort, OS/PFS were not significantly different between pS6-low and pS6-high patients. In a subgroup analysis non-epithelioid (biphasic and sarcomatoid) patients with high pS6 expression showed a significantly shorter OS (p < 0.001, 10.7 versus 16.9 months) and PFS (p < 0.001, 6.2 versus 10.8 months). In subgroup analysis, in non-epithelioid PM patients high pS6 expression was associated with significantly shorter OS and PFS. These exploratory findings suggest a clinically relevant PI3K pathway activation in non-epithelioid PM which might lay the foundation for future targeted treatment strategies.
Introduction
Pleural mesothelioma (PM) originates from the mesothelial cell lining of the pleura surface. PM is a rare1 and aggressive disease with a median survival of 7 months for untreated patients2. There are three main histologic subtypes of PM. The epithelioid subtype is the most common (50–60%) and is known to have longer survival than sarcomatoid and biphasic PMs3. Most PM cases can be linked to asbestos exposure with a latency of 20–40 years4. Although histology and pathologic stage are important prognostic factors, there is still significant variability in the survival of patients with similar characteristics5. Therefore, better prognostic biomarkers are needed.
The phosphatidylinositol 3-kinase (PI3K) pathway is considered a hallmark of cancer and emerged as a potential prognostic marker6. Activation of the PI3K pathway is stimulated by diverse oncogenes and growth factor receptors resulting in cell growth and proliferation, making it a potential prognostic biomarker and therapeutic target7. The PI3K pathway consists of the PI3K, a heterodimeric lipid kinase, as well as several downstream signalling proteins including AKT, mTOR, PTEN, 4E-BP1 and ribosomal protein S6. Alteration of the PI3K pathway proteins in PM, particularly AKT and PTEN, is described in cell lines8. The prognostic impact of PTEN expression was also investigated in large cohorts using PM tissue9,10. Phosphorylated ribosomal protein S6 (pS6) expression in PM was investigated in three studies before. While two claimed pS6 immunoreactivity in PM has a prognostic impact11,12, a more recent study could not confirm this finding13.
The present study aimed to elucidate the prognostic significance of pS6 expression in a large international multi-centre cohort of PM patients, the European Thoracic Oncology Platform (ETOP) Mesoscape virtual biobank.
Materials and methods
Mesoscape—study design
ETOP Mesoscape was designed as an innovative platform to address the challenges of studying the molecular epidemiology of PM and to expedite our knowledge of current and evolving clinical and molecular biomarkers. As the basis of this work, a centralized biobank and clinical database were created with currently 499 cases, with most of these represented on TMAs. The ETOP Mesoscape 001 pS6 project is using health-related data and biological samples from patients, which were collected retrospectively in the framework of Mesoscape. The research was conducted according to the Mesoscape master and Mesoscape 001 pS6 substudy protocols with adherence to country-specific ethics, regulatory requirements, and Reporting Recommendations for Tumour Marker Prognostic Studies. The study was approved and waivers of consent were granted by the Ethics Committees of the participating centres. ETOP Mesoscape was performed in accordance with the Declaration of Helsinki.
Case selection: clinical data capture
The ETOPdata central electronic database was built for annotated comprehensive clinical data collection from each participating site on patients with PM and a minimum follow-up of 2 years or until death, whichever occurred first. According to predefined criteria, eligible patients had an adequate quantity and quality of formalin-fixed paraffin-embedded tumour for analysis and available clinical, demographic, treatment, and outcome data. To enable quality assurance of tissue analysed and pathologic staging data, it was mandatory to upload an anonymized pathology report to the ETOPdata system. All submitted data were independently medically reviewed to ensure the adequacy of clinical data.
Study population
PM samples from 10 institutions (University Hospital Zurich, The Netherlands Cancer Institute Amsterdam, University Hospital Leuven, University Hospital of Parma, University Health Network, University Hospital Centre Zagreb, ICO Hospitalet (Bellvitge), Sotiria General Hospital, St James’s Hospital, Erasmus MC) were retrospectively collected. All patients had a histologically proven diagnosis of PM. The tumour stage was defined by tumour-node-metastasis (TNM) classification (8th edition) developed by the American Joint Commission on Cancer (AJCC) and the Union for International Cancer Control (UICC)14,15. Histological subclassification was done according to WHO classification16. Follow-up of patients was performed according to local policy.
Tissue microarray construction and Immunohistochemistry
A total of 13 tissue microarrays (TMAs) with three to eight punches per patient were prepared at each institution and send to the central lab at the Department of Pathology and Molecular Pathology, University Hospital Zurich. TMA blocks were sectioned and stained with haematoxylin and eosin for morphologic assessment. Deparaffinised 2-μm-thick TMA sections were automatically stained with BenchMark (Ventana, Tucson, AZ) using the iView diamino benzidine detection kit (Ventana). The primary antibody was a rabbit monoclonal antibody against Phospho-S6 Ribosomal Protein (pS6, Ser240/244, Cell Signaling Technology) at a 1:50 dilution.
TMA spots with a lack of tumour tissue or damaged tissue were excluded from the analysis. Immunohistochemical evaluation of the TMAs was conducted by two independent observers (JHR, MH) in a blinded manner. The cytoplasmatic staining intensity was semi quantitatively scored 0 (negative), 1 (weak), 2 (moderate), or 3 (strong). Furthermore, the percentage of cells having any positivity was proportionally scored 0 (0%), 0.1 (1–9%), 0.5 (10–49%), or 1.0 (50% and more) as previously described17. The H-score was obtained by multiplying intensity with percentage staining (final range, 0 to 3, per core). The final semi-quantitative H-score was determined by averaging the H-scores of all the cores from the same patient. Slides were digitalized (Nanozoomer NDP digital slide scanner C9600-12) and scored with the Hamamatsu NDP.view 2.8.24 Software. Intratumoral heterogeneity was assessed by the deviations of the intensity scores between the cores stained for each sample. Additionally, 5% of cases were selected and compared to corresponding whole-sections.
Statistical analysis
The analysis focused on comparing cohort characteristics and outcomes, between the cohorts of “pS6 high” versus “pS6 low” expression. The classification of patients as high/low was based on the median of the overall “H-scores” from all patients.
Differences in baseline characteristics by pS6 status were explored via the Fisher’s exact test for categorical characteristics and the Mann–Whitney test for continuous variables.
Clinical outcome was evaluated as overall survival (OS) and progression-free survival (PFS), estimated respectively, as time from diagnosis date to time to death from any cause, and time to progression/relapse or death from any cause. Median follow-up time was estimated using the reverse censoring method for OS. Both time-to-event endpoints (OS and PFS) were graphically depicted via Kaplan–Meier curves for the pS6 high and pS6 low patients, while median times and rates at 1- and 2-year time points were estimated based on the product-limit Kaplan–Meier method. Log-rank tests were performed to explore the difference in OS and PFS between pS6 high and pS6 low patients (overall as well as for prespecified groups by histology and treatment strategy). To further assess the effect of pS6 on OS/PFS, Cox proportional hazards models were fitted: univariate, as well as multivariable Cox models, adjusting for several factors of clinical interest: gender (“male” vs. “female”), ethnicity (“Caucasian” vs. “Other”), age at diagnosis (with an age cut-off of <64 and ≥64 years), ECOG performance status (“0” vs. “≥1” vs. “Unknown/Missing”), exposure to asbestos (“Yes/Possible” vs. “No” vs. “Unknown/Missing”), smoking history (“Former/Current” vs. “Never”), histology (“Epithelioid” vs. “Non-epithelioid”), localization (“Right” vs. “Left”), stage (“I” vs. “II” vs. “III” vs. “IV”) and treatment strategy (“Palliative” vs. “Complete resection”). In addition, multivariable Cox models, including also the interaction of pS6 status with each factor, were applied. The backward elimination method, with a removal criterion at 10%, was used to conclude on the significant prognostic factors. The hazard ratios (HRs) and the corresponding 95% confidence intervals (CIs) for all significant predictors are presented. The proportional hazards assumption was verified, visualizing the Schoenfeld residuals and testing the time-dependent covariates of the interaction of patients’ groups with survival time. Further exploratory subgroup analysis was performed according to the type of TMA tissue (treatment-naive or pre-treated) that was used for the assessment of pS6 expression as well as by diagnosis timing (patients diagnosed between 1999–2010 versus those diagnosed between 2011–2017). A sensitivity survival analysis was performed by splitting our cohort in three pS6 subgroups, according to 33% and 67% H-score percentiles.
Data were analysed using the SAS software package version 9.4 (SAS Institute, Cary, NC). All p-values (p) presented are 2-sided, and a p < 0.05 is considered statistically significant. In the case of the multiple comparisons of baseline characteristics a False Discovery Rate (FDR) adjustment was also taken into account.
Results
Analysis cohort
As of 9th of November 2020, 499 patients diagnosed with mesothelioma from 1999 to 2017 in 10 centres have been included in the ETOP Mesoscape database, while staining results for pS6 evaluation were available for 364 patients from 9 centres (Supplementary Tables S1–S2). Staining results were missing due to delayed tissue shipping from one centre, lack and damage of tumour tissue on the TMAs (Supplementary Fig. S1).
The pS6 analysis cohort, consisted primarily of males (86.5%) with median age at diagnosis of 64 years, while the vast majority was of Caucasian ethnicity (99.1%). Former and current smokers represented 48.0% and 11.5% of the cohort, respectively, while 40.4% were never smokers. Most of the patients (76.8%) were definitely or possibly exposed to asbestos, and for 45 of them (29.0%), asbestos fibres were detected in the lung. 114 (47.1% among those with available information) and 128 (52.9%) patients had ECOG performance status 0 and ≥1, respectively.
The majority of the cases (70.3%) were of epithelioid histology, while 24.2% were biphasic and 5.5% sarcomatoid (i.e., 29.7% non-epithelioid). The epithelioid and sarcomatoid components in biphasic PMs were evaluated in a combined score. For dubious sarcomatoid and desmoplastic PMs a cytokeratin and calretinin stain were available for cases from Zurich. Unclear external cases with a sarcomatoid/ desmoplastic histology were excluded.
In 204 (56.0%) patients tumour was detected on the right side. The distribution by clinical stage was 13.8%, 31.5%, 40.8% and 13.8% for stages I, II, III and IV respectively (based on patients with available information).
In addition, among the total 364 patients of the analysis cohort, 113 (31.0%) patients received palliative treatment as a first intention strategy (90 of them (80%) palliative chemotherapy), 249 (68.4%) patients had a macroscopic complete resection and only 2 (0.5%) patients did not receive any treatment (treatment details in Supplementary Table S3). Tumour tissue from biopsies at diagnosis and surgical resections were available. Of the 364 patients with tissue included in the pS6 analysis, 188 (51.6%) were treatment-naïve, 93 (25.5%) were pre-treated, while 83 (22.9%) could not be classified.
Baseline patient, tumour and other clinico-pathological characteristics for the pS6 analysis cohort (n = 364), as well as all registered Mesoscape patients (n = 499), are provided in Supplementary Table S4.
According to available IHC evaluations, calretinin (97.6%), CK5/6 (90.0%) and WT1 (88.6%) were detected in the vast majority of patients tested (Supplementary Table S5).
pS6 ribosomal protein expression
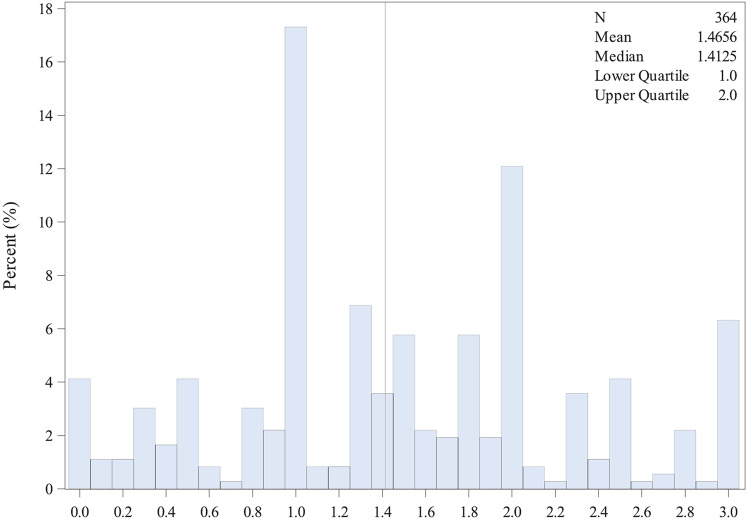
Cytoplasmic immunohistochemical pS6 ribosomal protein expression could be evaluated in TMA cores of 364 patients (Fig. 1). The distribution of the semi-quantitative H-scores (ranging from 0 to 3) is presented in Fig. 2. The median value of H-scores, used for the classification of patients in pS6 low versus high was 1.41. Corresponding distribution by histologic subtype is available in the supplement (Fig. S2)
Fig. 1. Immunohistochemical staining of pS6 ribosomal protein in pleural mesothelioma (PM).
A Hematoxylin and eosin stained TMA punches of epithelioid PM and corresponding immunohistochemical pS6 ribosomal protein staining. The immunohistochemical staining intensity reaching from 1 (weak), 2 (moderate) to 3 (strong). B Hematoxylin and eosin stained TMA punches of sarcomatoid PM and corresponding immunohistochemical pS6 ribosomal protein staining. TMA tissue micro array, PM pleural mesothelioma. Scale bar 200 µm.
Fig. 2. Distribution of H-scores.
Grey vertical line represents the median value of H-scores.
Furthermore, the percentage of pS6 high in the cohort of 188 treatment-naive patients was 66.0% (95% CI: 58.7–72.7%), while for the 93 pre-treated patients, pS6 high was significantly lower: 12.9% (95% CI: 6.9–21.5%) (p < 0.001).
Assessment of intratumoral heterogeneity
In our cohort, 153 cases (42%) showed a homogenous intensity score among the TMA punches, 151 cases (41%) revealed a minor heterogeneity in intensity (one intensity score difference) and 60 cases (16%) had a major heterogeneity (two or three intensity scores difference) in pS6 staining. Comparison of heterogeneity evaluated in TMAs and corresponding whole sections (n = 19) revealed a very good level of consistency with a non-significant difference in the derived H-score (Table S6).
Association of pS6 expression with histopathologic parameters
Patient, tumour and treatment characteristics, for the sub-cohorts of “pS6 high” versus “pS6 low” patients are presented in Table 1.
Table 1.
Baseline characteristics, by pS6 status and overall.
| Characteristic | pS6 high (n = 181) | pS6 low (n = 183) | All patients (N = 364) | p-value |
|---|---|---|---|---|
| Patient characteristics | ||||
| Gender - n (%) | ||||
| Male | 156 (86.2) | 159 (86.9) | 315 (86.5) | 0.88* |
| Female | 25 (13.8) | 24 (13.1) | 49 (13.5) | |
| Ethnicity - n (%) | ||||
| African | 1 (0.6) | – | 1 (0.3) | – |
| Caucasian | 168 (98.8) | 174 (99.4) | 342 (99.1) | |
| East Asian | 1 (0.6) | – | 1 (0.3) | |
| Other | – | 1 (0.6) | 1 (0.3) | |
| Unknown/Missing | 11 | 8 | 19 | |
| ECOG Performance status - n (%) | ||||
| 0 | 62 (45.9) | 52 (48.6) | 114 (47.1) | 0.86*,~ |
| 1 | 59 (43.7) | 46 (43.0) | 105 (43.4) | |
| ≥2 | 14 (10.4) | 9 (8.4) | 23 (9.5) | |
| Unknown/Missing | 46 | 76 | 122 | |
| Smoking history - n (%) | ||||
| Current | 18 (10.1) | 23 (12.9) | 41 (11.5) | 0.70*,~ |
| Former | 88 (49.4) | 83 (46.6) | 171 (48.0) | |
| Never | 72 (40.4) | 72 (40.4) | 144 (40.4) | |
| Unknown/Missing | 3 | 5 | 8 | |
| Exposure to asbestos - n (%) | ||||
| Yes | 83 (52.5) | 90 (50.6) | 173 (51.5) | 0.95*,~ |
| Possible | 39 (24.7) | 46 (25.8) | 85 (25.3) | 0.90*,~,$ |
| No | 36 (22.8) | 42 (23.6) | 78 (23.2) | |
| Unknown/Missing | 23 | 5 | 28 | |
| Asbestos fibers detected in lung¥ - n (%) | ||||
| Yes | 9 (12.3) | 36 (43.9) | 45 (29.0) | – |
| No | 2 (2.7) | 1 (1.2) | 3 (1.9) | |
| Not tested | 62 (84.9) | 45 (54.9) | 107 (69.0) | |
| Unknown/Missing | 49 | 54 | 103 | |
| Age at diagnosis (years) | ||||
| n | 181 (100.0) | 183 (100.0) | 364 (100.0) | 0.21§ |
| Mean (95% CI) | 63.7 (62.4 – 65.0) | 62.7 (61.5 – 64.0) | 63.2 (62.3 - 64.1) | |
| Median (Min-Max) | 65 (35 - 89) | 63 (33 - 89) | 64 (33 - 89) | |
| Tumour characteristics | ||||
| Histology - n (%) | ||||
| Epithelioid | 136 (75.1) | 120 (65.6) | 256 (70.3) | 0.051*,† |
| Non-epithelioid, incl: | 45(24.9) | 63 (34.4) | 108 (29.7) | 0.0024*,^ |
| Biphasic | 31 (17.1) | 57 (31.1) | 88 (24.2) | |
| Sarcomatoid | 14 (7.7) | 6 (3.3) | 20 (5.5) | |
| Localization - n (%) | ||||
| Right | 111 (61.3) | 93 (50.8) | 204 (56.0) | 0.034*,& FDR adj: 0.14 |
| Left | 67 (37.0) | 89 (48.6) | 156 (42.9) | |
| Both | 3 (1.7) | 1 (0.5) | 4 (1.1) | |
| Clinical T stage - n (%) | ||||
| 1 | 23 (15.9) | 31 (20.5) | 54 (18.2) | 0.14*,~ |
| 2 | 57 (39.3) | 69 (45.7) | 126 (42.6) | |
| 3 | 44 (30.3) | 40 (26.5) | 84 (28.4) | |
| 4 | 21 (14.5) | 11 (7.3) | 32 (10.8) | |
| Unknown/Missing | 36 | 32 | 68 | |
| Clinical N stage - n (%) | ||||
| 0 | 101 (70.6) | 97 (64.2) | 198 (67.3) | 0.11*,~ |
| 1 | 12 (8.4) | 27 (17.9) | 39 (13.3) | |
| 2 | 22 (15.4) | 21 (13.9) | 43 (14.6) | |
| 3 | 8 (5.6) | 6 (4.0) | 14 (4.8) | |
| Unknown/Missing | 38 | 32 | 70 | |
| Clinical M stage - n (%) | ||||
| 0 | 138 (95.2) | 145 (99.3) | 283 (97.3) | 0.036*,~ FDR adj: 0.14 |
| 1 | 7 (4.8) | 1 (0.7) | 8 (2.7) | |
| Unknown/Missing | 36 | 37 | 73 | |
| Clinical staging - n (%) | ||||
| I | 15 (10.5) | 25 (17.1) | 40 (13.8) | 0.081*,~ |
| II | 47 (32.9) | 44 (30.1) | 91 (31.5) | |
| III | 55 (38.5) | 63 (43.2) | 118 (40.8) | |
| IV | 26 (18.2) | 14 (9.6) | 40 (13.8) | |
| Unknown/Missing | 38 | 37 | 75 | |
| Treatment strategy | ||||
| Palliative | 69 (38.1) | 44 (24.0) | 113 (31.0) | 0.0032*,# |
| Complete Resection | 110 (60.8) | 139 (76.0) | 249 (68.4) | FDR adj: 0.039 |
| None | 2 (1.1) | – | 2 (0.5) | |
(*)Fisher’s exact test, (~) Category “Unknown/Missing” is excluded, ($) Categories “Yes” and “Possible” are combined, (¥) All percentages are over the total number of patients who were exposed to asbestos (yes/possible), (§) Mann–Whitney U test, (†) epithelioid vs. non-epithelioid, (^) epithelioid vs. biphasic vs. sarcomatoid, (&) Category “Both” is excluded, (#) Category ‘None’ is excluded.
Statistically significant p-values are in bold.
The association of pS6 status with treatment strategy was found to be statistically significant. Complete resection was less common in pS6 high patients (60.8%) compared to pS6 low (76.8%) (p = 0.0032) (the % of pS6 high expression was 44% for patients with complete resection versus 61% for patients receiving palliative treatment).
Right side localization of the disease was also more common among pS6 high patients (61.3%) compared to 50.8% in pS6 low (the % of pS6 high expression was 43% for patients with left localization versus 54% for patients with right localization) p = 0.034, but we note that this observed difference was not significant after FDR adjustment. In addition, even though only 8 cases in our analysis cohort were of clinical M stage 1, an association emerges, with 7 of the 8 cases belonging to the pS6 high group (p = 0.036, not significant after FDR adjustment).
Finally, with respect to histology the observed difference was not significant (p = 0.051) (Fig. 3).
Fig. 3. Bar plot of pS6 high prevalence by subgroups of interest.
*Category “Both” is excluded. All p-values refer to Fisher’s exact test.
More particularly, among pS6 high patients, 75.1% were epithelioid and 24.9% non-epithelioid (17.1% biphasic; 7.7% sarcomatoid), versus 65.6% epithelioid and 34.4% non-epithelioid (31.1% biphasic; 3.3% sarcomatoid) for pS6 low. The percentage of pS6 high expression was 53.1% for the epithelioid and 41.7% for the non-epithelioid cases (35% in the biphasic and 70% for the few sarcomatoid cases). Overall, pS6 high expression was not significantly different between epithelioid and non-epithelioid patients (p = 0.051), while a significant differentiation emerges when assessing separately biphasic and sarcomatoid cases (p = 0.0024). Of note, in our cohort, the non-epitheloiod group primarily consists of biphasic patients.
In a post-hoc exploratοry analysis, it is found that in the more recent cases a higher percentage of pS6 high patients was detected (60% for diagnosis in 2011–2017 vs 40% in 1999–2010, p < 0.001).
Prognostic value of pS6
The clinical outcome of the patients (OS, PFS) was evaluated at a median follow-up of 53.8 months (interquartile range: 43.2–77.1 months), comparable between the two sub-cohorts (p = 0.32). Most of the patients had died with disease (71.4%), 15.1% died without evidence of disease or with unknown disease status while only 4.7% of the patients were alive and disease-free at their last follow-up and 8.8% were alive with disease or unknown status.
A total of 155 (85.6%) deaths were observed in pS6 high patients, with median OS 18.3 months (95% CI: 16.4–20.9) and 160 (87.4%) deaths in pS6 low patients, with corresponding median OS 21.7 months (95% CI: 16.7–23.7), not significantly different (p = 0.52, Fig. S3). The 1-year OS estimates, along with the corresponding 95% CIs for pS6 high and low patients were 71.1% (63.9–77.2%) and 68.3% (61.0–74.5%), respectively. The OS estimates at 2-years were 37.1% (30.0–44.1%) for pS6 high patients and 41.5% (34.3–48.5%) for the pS6 low patients. Overall, the effect of pS6 was not found significant in Cox models, univariable (p = 0.52) or adjusting for baseline characteristics of interest (p = 0.28, with significant covariates: gender, histology and treatment strategy).
Subgroup analysis for OS has been performed by histology (epithelioid/non-epithelioid Fig. 4) and by treatment strategy (Fig. S4A, B). A statistically significant difference in OS for the pS6 status was observed only in non-epithelioid patients (p < 0.001), with median OS 10.7 months (95% CI: 7.4–13.6) for pS6 high versus 16.9 months (95% CI: 11.5–22.7) for patients with pS6 low expression.
Fig. 4. Overall survival (OS) according to histological subtype.
A OS by pS6 status; epithelioid patients. B OS by pS6 status; non-epithelioid patients. Interaction p-value (from Cox model including pS6 status with histology interaction): <0.001. Overall median OS was 23.1 months (95% CI: 19.9–25.2) for epithelioid patients and 13.7 months (95% CI: 10.7–15.7) for non-epithelioid. Log-rank p-value comparing pS6 high vs. low: 0.88 for epithelioid; <0.001 for non-epithelioid.
The significant effect of pS6 status in non-epithelioid patients was also verified in the multivariable Cox analysis, where the interaction of pS6 status with histology was significant (p < 0.001), with HRhigh vs. low = 2.12 (95% CI: 1.42–3.17); p < 0.001 for the non-epithelioid group (Table 2). In addition, in both high and low pS6 groups, histology was a significant prognostic factor with favourable outcome for epithelioid patients: HRepithelioid vs. non-epithelioid = 0.26 (95% CI: 0.18–0.38); p < 0.001 in pS6 high group and HRepithelioid vs. non-epithelioid = 0.64 (95% CI: 0.46–0.89); p = 0.0075 in pS6 low group. Furthermore, female patients (HRfemale vs. male = 0.55 (95% CI: 0.39–0.79); p = 0.0011), as well as patients who underwent complete resection (HRcomplete resections vs. palliative = 0.50 (95% CI: 0.39–0.64); p < 0.001), exhibited significantly lower risk of death.
Table 2.
Multivariable Cox proportional hazards model for overall survival (OS).
| No. of patients = 362* No. of deaths = 313 | Hazard Ratio | 95% CI | p-value |
|---|---|---|---|
| Interaction effects | |||
| pS6 status*Histology | <0.001 | ||
| High vs. Low | |||
| In Epithelioid | 0.87 | (0.66–1.14) | 0.31 |
| In Non-epithelioid | 2.12 | (1.42–3.17) | <0.001 |
| Epithelioid vs. Non-epithelioid | |||
| In High pS6 | 0.26 | (0.18–0.38) | <0.001 |
| In Low pS6 | 0.64 | (0.46–0.89) | 0.0075 |
| Main effects | |||
| Gender | |||
| Female vs. Male | 0.55 | (0.39–0.79) | 0.0011 |
| Treatment strategy | |||
| Complete Resection vs. Palliative | 0.50 | (0.39–0.64) | <0.001 |
(*) 2 patients who haven’t received treatment are excluded.
Note1: Variables of interest initially included in the model: Gender, Ethnicity, ECOG performance status, Smoking history, Exposure to asbestos, Age at diagnosis, Histology, Localization of tumour, Clinical staging, Treatment strategy.
Note2: P-values corresponding to the non-significant variables: Age at diagnosis: 0.87; Exposure to asbestos: 0.58; Clinical staging: 0.61; Ethnicity: 0.68; Smoking history: 0.54; ECOG performance status: 0.31; Localization of tumour: 0.21.
Note3: Ethnicity: Categories African, East Asian and Other are combined; Smoking history: Categories Current and Former are combined; Exposure to asbestos: Categories Yes and Possible are combined.
Analogous were the results for PFS. A total of 340 PFS events were recorded (166 (91.7%) in pS6 high patients and 174 (95.1%) in pS6 low patients, with a median PFS estimate of 12.4 months for both pS6 high and low patients and no statistically significant difference (p = 0.97) (Fig. S5). The PFS estimates at 1 and 2-years along with the corresponding 95% CIs for the pS6 high patients were 51.6% (44.1–58.7%) and 23.4% (17.5–29.9%), respectively. The corresponding estimates for the pS6 low patients were 50.3% (42.8–57.3%) and 21.8% (16.2–28.1%). Overall, the effect of pS6 on PFS was not found significant in Cox models, univariable (p = 0.97) or adjusting for baseline characteristics of interest (p = 0.96).
Subgroup analysis for PFS has also been performed by histology (Fig. 5A, B) and by treatment strategy (Fig. S6A, B). As with OS, a statistically significant PFS difference between high/low pS6 was observed in non-epithelioid patients (p < 0.001), with median PFS 6.2 months (95% CI: 4.1–8.9) for pS6 high versus 10.8 months (95% CI: 7.8–14.8) for patients with pS6 low expression.
Fig. 5. Progression-free survival (PFS) by pS6 status and histological subtype.
A PFS by pS6 status; epithelioid patients. B PFS by pS6 status; non-epithelioid patients. Interaction p-value (from Cox model including pS6 status with histology interaction): <0.001. Overall median PFS was 14.3 months (95% CI: 12.4–15.9) for epithelioid patients and 8.8 months (95% CI: 6.3–10.4) for non-epithelioid. Log-rank p-value comparing pS6 high vs. low: 0.18 for epithelioid; <0.001 for non-epithelioid.
In the multivariable Cox analysis, the interaction of pS6 status with histology was also found to be significant (p < 0.001), with HRhigh vs. low = 2.06 (95% CI: 1.39–3.07); p < 0.001 for the non-epithelioid group, while the reverse effect on PFS was observed for the epithelioid patients; HRhigh vs. low = 0.74 (95%CI: 0.57–0.97); p = 0.028 (Table 3). The favourable effect of epithelioid compared to non-epithelioid patients was detected in the group of pS6 high patients: HRepithelioid vs. non-epithelioid = 0.32 (95% CI: 0.23–0.46); p < 0.001. In addition, significantly lower risk of PFS event was observed for female patients (HRfemale vs. male = 0.61 [95% CI: 0.43–0.85]; p = 0.0034), and those who underwent complete resection (HRcomplete resections vs. palliative = 0.55 [95% CI: 0.44–0.70]; p < 0.001).
Table 3.
Multivariable Cox proportional hazards model for progression-free survival (PFS).
| No. of patients = 362* No. of PFS events = 338 | Hazard Ratio | 95% CI | p-value |
|---|---|---|---|
| Interaction effects | |||
| pS6 status*Histology | <0.001 | ||
| High vs. Low | |||
| In Epithelioid | 0.74 | (0.57–0.97) | 0.028 |
| In Non-epithelioid | 2.06 | (1.39–3.07) | <0.001 |
| Epithelioid vs. Non-epithelioid | |||
| In High ps6 | 0.32 | (0.23–0.46) | <0.001 |
| In Low ps6 | 0.89 | (0.65–1.22) | 0.48 |
| Main effects | |||
| Gender | |||
| Female vs. Male | 0.61 | (0.43–0.85) | 0.0034 |
| Treatment strategy | |||
| Complete Resection vs. Palliative | 0.55 | (0.44–0.70) | <0.001 |
(*) 2 patients who haven’t received treatment are excluded.
Note1: Variables of interest initially included in the model: Gender, Ethnicity, ECOG performance status, Smoking history, Exposure to asbestos, Age at diagnosis, Histology, Localization of tumour, Clinical staging, Treatment strategy.
Note2: P-values corresponding to the non-significant variables: Ethnicity: 0.85; Age at diagnosis: 0.50; ECOG performance status: 0.49; Exposure to asbestos: 0.53; Clinical staging: 0.33; Smoking history: 0.36; Localization of tumour: 0.22.
Note3: Ethnicity: Categories African, East Asian and Other are combined; Smoking history: Categories Current and Former are combined; Exposure to asbestos: Categories Yes and Possible are combined.
In a further exploratory subgroup analysis according to the type of TMA tissue (treatment-naive or pre-treated), the significant effect of pS6 in non-epithelioiod patients was confirmed in the subgroup of treatment-naïve tissue (n = 50 patients; with 26 being pS6 high), with median OS 8.6 months (95% CI: 5.7–10.8) for pS6 high versus 15.1 months (95% CI: 5.1–27.0) for pS6 low (p = 0.014), while in the smaller group of “pre-treated” non-epitheliod patients (37 patients; with only 5 being pS6 high) pS6 effect was not significant (p = 0.17). Analogous were the results for the PFS endpoint (for the 50 non-epithelioid patients with treatment-naïve tissue), with median PFS 4.5 months (95% CI: 2.4–6.1) for pS6 high versus 9.2 months (95% CI: 5.0–26.1) for pS6 low (p = 0.017).
In the post-hoc exploratory analysis by diagnosis timing, no significant difference was detected between the two time-groups of patients, neither for OS (medians in months (95% CI): 17.4 (14.5–21.7) vs 21.3 (18.0–24.3), log-rank p = 0.23) nor for PFS (medians in months: 11.8 (9.7–13.7) vs 12.8 (10.8–14.9), p = 0.72). In addition, the interaction effect of diagnosis time with pS6 expression was not found significant, in unadjusted (p = 0.78, p = 0.82, respectively for OS/PFS) as well as in multivariable Cox models (p = 0.93, 0.87).
Finally, the sensitivity survival analysis, based on three pS6 subgroups (140 cases with low pS6 H-score ≤ 1, 102 cases with intermediate pS6 H-score between 1 and 1.84, and 122 cases with high pS6 H-score ≥ 1.84) confirmed our primary findings, that is, in the overall cohort no difference in OS/PFS was found between the three pS6 subgroups (Figs. S7, S8), while the interaction with histology was significant in multivariable analysis (Tables S7, S8).
Discussion
In the present study, the expression of pS6 was analysed in a large TMA-based PM cohort consisting of 364 patients. An overexpression of pS6 in non-epithelioid PM patients revealed a significantly shorter PFS and OS. Additionally, less complete resections were found in the pS6 high subgroup.
New prognostic biomarkers for PM are urgently needed. However, suggested prognostic biomarkers are mostly screened only on small cohorts18–21. In 2012 Cedres et al. reported a low pS6 immunoreactivity significantly correlated with longer PFS and OS in a relatively small cohort of 26 analysed treatment-naïve PM patients11. Interestingly, in 2016 Cedres et al. investigated another cohort of 23 mesotheliomas and found pAKT, FOXO3a and PD-L1 significantly associated with OS but not pS6 expression13. Another study by Bitanihirwe et al. detected a high pS6 expression associated with shorter PFS in a cohort of 74 treatment naïve patients12.
Altogether, in this study TMA-based tissue of 364 patients was included, with available clinical and outcome data, comprising epithelioid (n = 256, 70.3%), biphasic (n = 88, 24.2%) and sarcomatoid (n = 20, 5.5%) PMs.
All studies investigating pS6 expression in PM used the same commercially available pS6 antibody. Whereas Cedres et al.11 investigated the original material received for making the diagnosis, Bitanihirwe et al.12 and the present study investigated TMAs with at least 3 (up to 8) spots representing the tumour of each patient. For evaluation, an H-score was calculated by multiplying intensity (0–3+ ) with the corresponding percentage of positive cells. Although Cedres et al. used a continuous scale of positive cells (0–100%), in Bitanihirwe’s and this study, the percentage of positive cells was proportionally scored (0%, 1–9%, 10–49% and 50% and more). Concerning pre-treatment, Cedres and Bitanihirwe et al. only used treatment-naïve PM tissue11,12. Our study included TMAs consiting of treatment-naïve (n = 188, 51.6%) and pre-treated tumour tissue (n = 93, 25.5%). Bitanihirwe et al. also investigated immunhistochemical expression changes in matched pre- and postchemotherapy samples of different PI3K pathway members. A significant reduction of pS6 expression was detected after chemotherapy12. This finding is confirmed in our cohort where pS6 high cases occurred significantly more often in treatment-naïve (66%) than in pre-treated (12.9%) samples (p < 0.001).
The prognostic value of PI3K pathway members in PM is increasingly investigated. PTEN loss was associated with reduced overall survival in a large study by Opitz et al. whereas Agarwal et al. reported no relationship with survival9,10. Elevated pAKT expression is described in a large cohort of PM22, and also a relationship to longer OS was found13. Additionally, Cedres et al. described the expression level of FOXO3a and PD-L1 to be related to OS in PM11,13.
Overexpression of pS6 ribosomal protein has also been reported to be related to worse overall, shorter metastatic-free and disease-free survival in lung, ovarian and breast cancer as well as in renal cell carcinomas23–26.
The ribosomal protein S6 is a downstream signalling protein in the PI3K pathway involved in protein synthesis and cell proliferation27 and it has been recently shown to be correlated with proliferation marker ki67 in mesothelioma28. Although phosphorylation of S6 can also be regulated by enhanced RAS/RAF/ERK/mTORC1 activity some lines of evidence support the link between pS6 and PI3K/Akt/mTOR in mesothelioma. They include the recent finding of loss of PTEN expression in sarcomatoid mesothelioma and combined deletion of PTEN and Tp53 leds to non-epithelioid development29. Therefore, the PI3K pathway is also a potential therapeutic target. PM cell lines and mouse xenografts were successfully treated with a combination of CDK4/6 and PI3K/mTOR inhibitors30 and MET and PI3K/mTOR inhibitors22,31. However, treatment targeting the PI3K pathway in unselected PM patient cohorts did not bring up encouraging results up until now32,33.
In this study, only in non-epithelioid PMs an overexpression of pS6 was associated with shorter PFS and OS, indicating that in non-epithelioid PMs, the PI3K pathway is activated in a biologically relevant way. This activation of the PI3K pathway is further supported by findings in our previous studies, where we could show that similar to pS6 overexpression, higher expression of the upstream regulator pmTOR is also associated with shorter OS (in a histologically heterogeneous cohort), while at the same time the short survivors also show a downregulation of the pathway suppressor PTEN9,12. Cedres and colleagues further show an elevation of another PI3K pathway component, pAKT, however they do not observe the expected negative association with OS11,13. Taken together these findings are supportive of an activation of the entire pathway in sarcomatoid PM. It is known that tumours displaying sarcomatoid features (including biphasic) are associated with worse prognosis and higher chemoresistance34,35. Thus, new therapy options for this subtype are even more needed. Interestingly, in sarcomatoid mesotheliomas Marques et al. described a response to combined MEK and PI3K inhibition in vitro31. Interestingly, the RAS/MEK/ERK pathway is also known to phosphorylate pS6, however exclusively at Ser235/23636, not at Ser240/244, which is targeted by the antibody used in this study. Together, this might point to the consideration that the sarcomatoid subtype is a predictive biomarker for the response to targeted PI3K inhibition.
Our study has some limitations, including the retrospective approach. The TMAs consisted of 3 to 8 punches per patient which might not fully represent the heterogeneous nature of each tumour. However, an internal comparison of TMAs to corresponding whole slides revealed H-scores with a non-significant difference. Although a significant higher percentage of pS6 high patients was detected in more recent cases (1999–2010 vs 2011–2017, p < 0.001), no interaction effect of diagnosis timing and pS6 expression on the clinical outcome was detected.
To the best of our knowledge, this study represents the largest immunohistochemical screening of mesothelioma specimens for the expression of pS6 ribosomal protein with correlation to clinical data. In the primary analysis of all-histologies cohort no association of pS6 expression with outcome is detected. However, subgroup analysis indicates high pS6 expression as a prognostic biomarker associated with significantly shorter OS and PFS in non-epithelioid PM patients.
These exploratory, hypothesis-generating, results suggest a relevant PI3K pathway activation in non-epithelioid PM which might lay the foundations for future targeted treatment strategies.
Supplementary information
Acknowledgements
The European Thoracic Oncology Platform (ETOP) coordinated Mesoscape and is responsible for the collection and management of data, data analysis, reporting, and for study design together with the University Hospital of Zurich (USZ). We thank Susanne Dettwiler, Fabiola Prutek and Christiane Mittmann for tissue processing and immunohistochemical staining’s. We thank Bart Vrugt, Alex Soltermann for their contribution to the Mesoscape development and for tumour identification. We acknowledge the Biobanks of the participating centres, and particularly the Department of Pathology and Molecular Pathology at the University Hospital of Zurich, in their role as central Mesoscape laboratory. We thank Martina Friess and Alessandra Matter for their support.
Author contributions
JHR, RK, SP, RAS, PB, EFB and IO performed study concept and design; JHR, MH, KN, MDP, LB, EN, ST, SGG, LA, JA, MBK, KM, BW, FBZ, MS, RL, SPF, ES, JVDT, PB and IO provided data acquisition; JHR, MH, ZT, KV and IO provided analysis and interpretation of data; JHR, ZT, KN, EN, SGG, JA, MBK, JVDT, NM, KV, RK, SP, PB and IO performed writing; All Authors read and approved the final paper, and confirmed their integrity.
Funding
This work was supported by ETOP, Oncosuisse [KFS-3626-02-2015], Bristol-Myers Squibb, and Merck, who supplied support for sample handling and assays. Bristol-Myers Squibb and Merck did not have a role in the reporting or interpretation of the study. Open access funding provided by University of Zurich.
Data availability
Access to de-identified individual participant data used in this study may be requested by researchers by submitting a research proposal (Mesoscape@etop-eu.org), which will be reviewed for scientific merit and feasibility in accordance with the ETOP Biobank Policy.
Competing interests
JHR, MH, ZT, KN, MDP, ST, SGG, LA, EFB, MBK, KM, BW, FBZ, MS, RL, SPF, ES, NM, RK, RAS declared no conflict of interest. EN declares his participation in the following advisoriy boards and lectures: Roche, BMS, MSD, Merck-Serono, Pfizer, Lilly, Amgen, Boehringer-Ingelheim, AstraZeneca, Takeda, Sanofi and Bayer and his research support from: Roche, Pfizer, Merck-Serono and BMS. JA declares: Adv board BMS, Eli-Lilly, MSD, Bayer, Amphera, Roche/Stock owner Amphera/Patent on allogenic tumor lysate, combination immunotherapy. JVDT declares: Adv board and lectures: AstraZeneca, BMS, Eli-Lilly, Johnson&Johnson, MSD, Pfizer, Roche. SP delclares: I have received education grants, provided consultation, attended advisory boards and/or provided lectures for the following organizations, from whom I have received honoraria (all fees to institution): Consultation / Advisory role: AbbVie, Amgen, AstraZeneca, Bayer, Beigene, Biocartis, Boehringer Ingelheim, Bristol-Myers Squibb, Clovis, Daiichi Sankyo, Debiopharm, ecancer, Eli Lilly, Elsevier, Foundation Medicine, Illumina, Imedex, IQVIA, Incyte, Janssen, Medscape, Merck Sharp and Dohme, Merck Serono, Merrimack, Novartis, OncologyEducation, Pharma Mar, Phosplatin Therapeutics, PER, Pfizer, PRIME, Regeneron, RMEI, Roche/Genentech, RTP, Sanofi, Seattle Genetics, Takeda.Talk in a company’s organized public event: AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, ecancer, Eli Lilly, Illumina, Imedex, Medscape, Merck Sharp and Dohme, Novartis, PER, Pfizer, Prime, Roche/Genentech, RTP, Sanofi, Takeda.Receipt of grants/research supports: (Sub)investigator in trials (institutional financial support for clinical trials) sponsored by Amgen, AstraZeneca, Biodesix, Boehringer Ingelheim, Bristol-Myers Squibb, Clovis, GSK, Illumina, Lilly, Merck Sharp and Dohme, Merck Serono, Mirati, Novartis, and Pfizer, Phosplatin Therapeutics, Roche/Genentech. PB declares: Consultancy roel for BMS, Advisory Board for MSD, Beigene, Pfizer, Takeda and Boehringer Ingelheim. Research Grants from AZ and BMS. IO declares: Consultation / Advisory role: AstraZeneca, Bristol-Myers Squibb, MSD Merck Sharp & Dohme AG. Talk in a company’s organized public event: AstraZeneca, Roche. Receipt of grants/research supports: Institutional grants for fellowships from Medtronic, Roche. Proctorship for Intuitive.
Ethics approval and consent to participate
The study protocol and procedures adhere to the principles outlined in the International Conference on Harmonisation Good Clinical Practice (ICH GCP) guidelines. The study was conducted in compliance with the protocol and performed in accordance with the ethical principles in the Declaration of Helsinki, and regulations on the use of human tissue for research in each participating country. The protocol and the use of biological samples from patients have the approval of a properly constituted committee or committees responsible for approving clinical studies.
Footnotes
The original online version of this article was revised: Due to an error in an author name.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A list of authors and their affiliations appears at the end of the paper.
Change history
10/17/2022
A Correction to this paper has been published: 10.1038/s41379-022-01170-z
Contributor Information
Isabelle Opitz, Email: isabelle.schmitt-opitz@usz.ch.
for the ETOP Mesoscape consortium:
Rolf Stahel, Anita Hiltbrunner, Rosita Kammler, Nesa Marti, Patrick Vagenknecht, Barbara Ruepp, Urania Dafni, Zoi Tsourti, Panagiota Zygoura, Katerina Vervita, Georgia Dimopoulou, Charitini Andriakopoulou, Androniki Stavrou, Jan H. Rüschoff, Martina Haberecker, Susanne Dettwiler, Fabiola Prutek, and Christiane Mittmann
Supplementary information
The online version contains supplementary material available at 10.1038/s41379-022-01145-0.
References
- 1.Carbone M, Adusumilli PS, Alexander HR, Baas P, Bardelli F, Bononi A, et al. Mesothelioma: Scientific clues for prevention, diagnosis, and therapy. CA Cancer J Clin 69, 402–429 (2019) [DOI] [PMC free article] [PubMed]
- 2.Taioli E, Wolf AS, Camacho-Rivera M, Kaufman A, Lee DS, Nicastr D, et al. Determinants of survival in malignant pleural mesothelioma: a surveillance, epidemiology, and end results (SEER) study of 14,228 patients. PLoS One 10 (2015) [DOI] [PMC free article] [PubMed]
- 3.Tolani B, Acevedo LA, Hoang NT, Biao H. Heterogeneous Contributing Factors in MPM Disease Development and Progression: Biological Advances and Clinical Implications. Int J Mol Sci 19 (2018) [DOI] [PMC free article] [PubMed]
- 4.Carbone M, Kratzke RA, Testa JR. The pathogenesis of mesothelioma. Sem Oncol 29, 2–17 (2002) [DOI] [PubMed]
- 5.Flores RM, Zakowski M, Venkatraman E, Krug L, Rosenzweig K, Dycoco J, et al. Prognostic factors in the treatment of malignant pleural mesothelioma at a large tertiary referral center. J Thorac Oncol 2, 957–965 2007) [DOI] [PubMed]
- 6.Fruman DA, Chiu H, Hopkins BD, Bagrodia S, Cantley LC, Abraham RT. The PI3K Pathway in Human Disease. Cell 170, 605–635 (2017) [DOI] [PMC free article] [PubMed]
- 7.Katso R, Okkenhaug K, Ahmadi K, white S, Timms J, Waterfield MD. Cellular function of phosphoinositide 3-kinases: implications for development, homeostasis, and cancer. Annu Rev Cell Dev Biol 17, 615–675 (2001) [DOI] [PubMed]
- 8.Suzuki Y, Murakami H, Kawaguchi K, Tanigushi T, Fujii M, Shinjo K, et al. Activation of the PI3K-AKT pathway in human malignant mesothelioma cells. Mol Med Rep 2, 181–188 (2009) [DOI] [PubMed]
- 9.Opitz I, Soltermann A, Abaecherli M, Hinterberger M, Probst-Hensch N, Stahel R, et al. PTEN expression is a strong predictor of survival in mesothelioma patients. Eur J Cardiothorac Surg 33, 501–505 (2008) [DOI] [PubMed]
- 10.Agarwal V, Campbell A, Beaumont KL, Cawkwell L, Lind ML. PTEN protein expression in malignant pleural mesothelioma. Tumour Biol 34, 847–851 (2013) [DOI] [PubMed]
- 11.Cedres S, Montero MA, Martinez P, Martinez A, Rodríguez-Freixinós V, Torrejon D, et al. Exploratory analysis of activation of PTEN-PI3K pathway and downstream proteins in malignant pleural mesothelioma (MPM). Lung Cancer 77, 192–198 (2012) [DOI] [PubMed]
- 12.Bitanihirwe BK, Meerang M, Friess F, Soltermann A, Frischknecht L, Thies S, et al. PI3K/mTOR signaling in mesothelioma patients treated with induction chemotherapy followed by extrapleural pneumonectomy. J Thorac Oncol 9, 239–247 (2014) [DOI] [PubMed]
- 13.Cedres S, Ponce-Aix S, Pardo-Aranda N, Navarro-Mendivil A, Martinez-Marti A, Zugazagoitia J, et al. Analysis of expression of PTEN/PI3K pathway and programmed cell death ligand 1 (PD-L1) in malignant pleural mesothelioma (MPM). Lung Cancer 96, 1–6 (2016) [DOI] [PubMed]
- 14.Brierley J, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours, 8th edn. (John Wiley & Sons, Inc. Hoboken, NJ, USA, 2017)
- 15.Amin MB, Edge SB, Greene FL, Byrd DR, Brookland RK, Washington MK, et al. (Eds.), AJCC cancer staging manual, 8th edn. (Springer International Publishing AG: Cham, CH, 2017)
- 16.Travis WD, Brambilla E, Müller-Hermelink HK, Harris CC. (Eds.), Pathology and genetics of tumours of the lung, pleura, thymus and heart; World Health Organization classification of tumours, 3rd edn. (IARC Press: Lyon, FR, 2015) p 344
- 17.Sidi R, Pasello G, Opitz I, Soltermann A, Tutic M, Rehrauer H, et al. Induction of senescence markers after neo-adjuvant chemotherapy of malignant pleural mesothelioma and association with clinical outcome: an exploratory analysis. Eur J Cancer 47, 326–332 (2011) [DOI] [PubMed]
- 18.Destro A, Ceresoli GL, Zucali PA, Morenghi E, Bianchi P, Pellegrini C, et al. EGFR overexpression in malignant pleural mesothelioma. An immunohistochemical and molecular study with clinico-pathological correlations. Lung Cancer 51, 207–215 (2006) [DOI] [PubMed]
- 19.Dazzi H, Hasleton PS, Thatcher N, Wilkes S, Swindell R, Chatterjee AK. Malignant pleural mesothelioma and epidermal growth factor receptor (EGF-R). Relationship of EGF-R with histology and survival using fixed paraffin embedded tissue and the F4, monoclonal antibody. Br J Cancer 61, 924–926 (1990) [DOI] [PMC free article] [PubMed]
- 20.O’Byrne KJ, Edwards JG, Waller DA. Clinico-pathological and biological prognostic factors in pleural malignant mesothelioma. Lung Cancer 45(Suppl 1), S45–48 (2004) [DOI] [PubMed]
- 21.Gordon GJ, Mani M, Mukhopadhyay L, Dong L, Edenfield HR, Glickman JN, et al. Expression patterns of inhibitor of apoptosis proteins in malignant pleural mesothelioma. J Pathol 211, 447–454 (2007) [DOI] [PubMed]
- 22.Kanteti R, Dhanasingh I, Kawada I, Lennon FE, Arif Q, Bueno R, et al. MET and PI3K/mTOR as a potential combinatorial therapeutic target in malignant pleural mesothelioma. PLoS One 9 (2014) [DOI] [PMC free article] [PubMed]
- 23.McDonald JM, Pelloski CE, Ledoux A, Sun M, Raso G, Komaki R, et al. Elevated phospho-S6 expression is associated with metastasis in adenocarcinoma of the lung. Clin Cancer Res 14, 7832–7837 (2008) [DOI] [PMC free article] [PubMed]
- 24.Pantuck AJ, Seligson DB, Klatte T, Yu H, Leppert JT, Moore L, et al. Prognostic relevance of the mTOR pathway in renal cell carcinoma: implications for molecular patient selection for targeted therapy. Cancer 109, 2257–2267 (2007) [DOI] [PubMed]
- 25.Liu Z, Yun R, Yu X, Hu H, Huang G, Tan B, et al. Overexpression of Notch3 and pS6 Is Associated with Poor Prognosis in Human Ovarian Epithelial Cancer. Mediators Inflamm (2016) [DOI] [PMC free article] [PubMed]
- 26.Cuperjani F, Gashi L, Kurshumliu F, Dreshaj S, Selimi F. Relationship between Ribosomal Protein S6-pS240 Expression and other Prognostic Factors in Non-Special Type Invasive Breast Cancer. Breast Care 14, 171–175 (2019) [DOI] [PMC free article] [PubMed]
- 27.Volarevic S, Thomas G. Role of S6 phosphorylation and S6 kinase in cell growth. Prog Nucleic Acid Res Mol Biol 65, 101–127 (2001) [DOI] [PubMed]
- 28.Grosso S, Marini A, Gyuraszova K, Vande Voorde J, Sfakianos A, Garland GD, et al. The pathogenesis of mesothelioma is driven by a dysregulated translatome. Nat Commun 12 (2021) [DOI] [PMC free article] [PubMed]
- 29.Marqués M, Tranchant R, Risa-Ebrí B, Suárez-Solís ML, Fernández LC, Carrillo-de-Santa-Pau E, et al. Combined MEK and PI3K/p110beta Inhibition as a Novel Targeted Therapy for Malignant Mesothelioma Displaying Sarcomatoid Features. Cancer Res 80, 843–856 (2020) [DOI] [PubMed]
- 30.Bonelli M, Terenziani R, Zoppi S, Fumarola C, La Monica S, Cretella D, et al. Dual Inhibition of CDK4/6 and PI3K/AKT/mTOR Signaling Impairs Energy Metabolism in MPM Cancer Cells. Int J Mol Sci 21 (2020) [DOI] [PMC free article] [PubMed]
- 31.Kanteti R, Riehm JJ, Dhanasingh I, Lennon FE, Mirzapoiazova T, Mambetsariev B et al. PI3 Kinase Pathway and MET Inhibition is Efficacious in Malignant Pleural Mesothelioma. Sci Rep 6 (2016) [DOI] [PMC free article] [PubMed]
- 32.Stahel RA, Weder W, Felley-Bosco E, Petrausch U, Curioni-Fontecedro A, Schmitt-Opitz I, et al. Searching for targets for the systemic therapy of mesothelioma. Ann Oncol 26, 1649–1660 (2015) [DOI] [PubMed]
- 33.Remon J, Reguart N, Corral J, Lianes P. Malignant pleural mesothelioma: new hope in the horizon with novel therapeutic strategies. Cancer Treat Rev 41, 27–34 (2015) [DOI] [PubMed]
- 34.Rusch VW. Extrapleural pneumonectomy and extended pleurectomy/decortication for malignant pleural mesothelioma: the Memorial Sloan-Kettering Cancer Center approach. Ann Cardiothorac Surg 1, 523–531 (2012) [DOI] [PMC free article] [PubMed]
- 35.Rusch VW, Giroux D, Kennedy C, Ruffini E, Cangir AK, Rice D, et al. Initial analysis of the international association for the study of lung cancer mesothelioma database. J Thorac Oncol 7, 1631–1639 (2012) [DOI] [PubMed]
- 36.Roux PP, Shahbazian D, Vu H, Holz MK, Cohen MS, Taunton J, et al. RAS/ERK signaling promotes site-specific ribosomal protein S6 phosphorylation via RSK and stimulates cap-dependent translation. J Biol Chem 282, 14056–14064 (2007) [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Access to de-identified individual participant data used in this study may be requested by researchers by submitting a research proposal (Mesoscape@etop-eu.org), which will be reviewed for scientific merit and feasibility in accordance with the ETOP Biobank Policy.