Abstract
Background
Remote patient monitoring (RPM) for automated peritoneal dialysis (APD) may improve clinical outcomes. Paediatric data, however, remain extremely scarce.
Methods
We conducted a prospective observational study of children (0–18 years) receiving APD with cloud-based RPM over two 24-week periods (pre- and post-RPM). Primary outcomes were unplanned hospitalizations and fluid management. Children receiving APD without RPM (non-RPM) were included as control.
Results
Seven patients (6 females) receiving APD were enrolled in the RPM programme at 11.3 years (IQR 2.6–17.1). Main indications for RPM included history of fluid overload (n = 3) and non-adherence (n = 2). Ten children were included in the non-RPM group (6 females; 16.9 years, IQR 12.8–17.6). Four patients (57.1%, 95% CI 22.5–100%) experienced fewer unplanned hospitalizations and 5 patients (71.4%, 95% CI 34.1–100%) had shorter hospital stays during the post-RPM period. The hospitalization rates and length of stay were reduced by 45% and 42%, respectively. The higher hospitalization rates among the RPM group, compared to the non-RPM group, were no longer observed following implementation of RPM. There was a significant increase in ultrafiltration (565.6 ± 248.7 vs. 501.7 ± 286.6 ml/day, p = 0.03) and reduction in systolic blood pressure (114.1 ± 12.6 vs. 119.9 ± 11.19 mmHg, p = 0.02) during the post-RPM period. All patients demonstrated satisfactory adherence. Although quality of life (PedsQL 3.0 ESRD module) was not different pre- and post-RPM, all patients agreed in the questionnaires that the use of RPM improved their quality of life and sense of security.
Conclusions
In conclusion, RPM in children receiving APD is associated with fewer and shorter unplanned hospitalizations, improved fluid management and favourable adherence to PD.
Graphical abstract

A higher resolution version of the Graphical abstract is available as Supplementary information
Supplementary information
The online version contains supplementary material available at 10.1007/s00467-022-05828-3.
Keywords: Claria, Remote patient monitoring, Peritoneal dialysis, Children
Introduction
Peritoneal dialysis (PD) is an important modality of kidney replacement therapy, especially among young children [1–3]. Hong Kong adopts a PD-first policy and 70% of children and young people are offered automated PD (APD) as initial modality [2]. While PD as a home therapy facilitates normal schooling, it is associated with hospitalizations due to mechanical or infectious complications [4]. There are also concerns with non-adherence, as well as stress and poor quality of life for patients and their families [5, 6].
APD systems equipped with cloud-based, remote patient monitoring (RPM) have been developed to enhance patient-centred care. This system allows active monitoring of PD-related parameters, such as blood pressure and ultrafiltration, and remote adjustment of PD prescription. Limited trials involving adult patients generally showed improved patient satisfaction, but the benefits in clinical outcomes and reduction of medical visits remained controversial [7, 8]. In the only available paediatric series, Bakkaloglu et al. focused on the perceptions of RPM from families and health care providers [9]. The aim of this study was to provide prospective data on important clinical outcome measures, such as hospitalization rates and blood pressure control, and evaluate patient quality of life among children receiving APD with RPM.
Methods
We conducted a prospective observational study of children (0–18 years) receiving APD, who were enrolled in a pilot RPM programme, at the Paediatric Nephrology Centre, Hong Kong Children’s Hospital, Hong Kong. The Paediatric Nephrology Centre is the territory-wide, designated site providing chronic kidney replacement therapy (KRT) in Hong Kong. Due to limited APD machines with cloud-based RPM function available, patients were recruited into the pilot RPM programme according to specific indications such as history of fluid overload and treatment non-adherence. The programme was led by a dedicated team comprised of a paediatric nephrologist and two dialysis nurses, and was launched on 1 September 2021. The study was conducted over two 24-week periods, before (March 2021 to August 2021) and after (September 2021 to February 2022) implementation of the RPM programme (pre- and post-RPM). Patients who received standard APD without RPM (non-RPM group) during the same study period were also included as control. All patients who did not complete the observation periods were excluded. Data pertaining to clinical and PD-related parameters, alarms, details of RPM and interventions, and quality of life of patients and caregivers were collected and evaluated. Questionnaires were given to all caregivers or adolescent patients (> 12 years) who received PD following the completion of the 6-month post-RPM period (Table 4). In addition, a questionnaire on perception of the RPM programme was distributed to the dialysis team 1 year after programme implementation (Table 5).
Table 4.
Results from post-RPM implementation questionnaires distributed to patients and carers who performed automated peritoneal dialysis at homea
| Questions | Strongly disagree | Disagree | Neutral | Agree | Strongly agree |
|---|---|---|---|---|---|
| I feel safer to perform peritoneal dialysis at home under frequent monitoring by health care workers | 0 | 0 | 0 | 1 (17%) | 5 (83%) |
| I feel less burden with shared responsibility of home treatment | 0 | 0 | 0 | 0 | 6 (100%) |
| I feel my/my child’s quality of life has improved with the remote patient monitoring program | 0 | 0 | 0 | 2 (33%) | 4 (67%) |
| I have become more adherent to the peritoneal dialysis treatment | 0 | 0 | 0 | 0 | 6 (100%) |
| I feel that that there are less unplanned/ unexpected peritoneal dialysis-related admissions with remote patient monitoring | 0 | 0 | 0 | 0 | 6 (100%) |
| I perceive that the number of peritoneal dialysis-related complications are either reduced or prevented | 0 | 0 | 0 | 1 (17%) | 5 (83%) |
| I feel that there are less medical visits with remote patient monitoring program | 0 | 0 | 0 | 2 (33%) | 4 (67%) |
| I feel that the number of phone calls or interventions received from the health care team is reasonable | 0 | 0 | 0 | 1 (17%) | 5 (83%) |
| I think the remote patient monitoring program is beneficial to children on peritoneal dialysis | 0 | 0 | 0 | 0 | 6 (100%) |
Data are expressed in number (%)
aA total of 7 questionnaires were distributed to patients/caregivers who performed peritoneal dialysis following the post-RPM period. Six responses (86%) were received (4 caregivers and 2 patients)
Table 5.
Perception of the dialysis team towards RPM 1-year after programme implementationa
| Questions | Strongly disagree | Disagree | Neutral | Agree | Strongly agree |
|---|---|---|---|---|---|
| The burden of frequent patient reviews is reasonable | 0 | 0 | 0 | 5 (83%) | 1 (17%) |
| I often need to spend additional work time on patient reviews | 0 | 0 | 1 (17%) | 4 (67%) | 1 (17%) |
| My routine clinical duty is not affected by the remote patient monitoring programme | 0 | 1 (17%) | 0 | 3 (50%) | 2 (33%) |
| The program should be extended to all children receiving peritoneal dialysis | 0 | 0 | 1 (17%) | 1 (17%) | 4 (67%) |
| Additional manpower is needed for expanding the remote patient monitoring service | 0 | 0 | 0 | 1 (17%) | 5 (83%) |
| The overall efficiency in data analysis improves with gaining experience on the Sharesource® system | 0 | 0 | 0 | 1 (17%) | 5 (83%) |
| More frequent reviews are required during the initial phase of enrolment | 0 | 1 (17%) | 0 | 1 (17%) | 4 (67%) |
| The efficiency in individual patient review often improves with time | 0 | 0 | 0 | 5 (83%) | 1 (17%) |
| Patient/family are more adhered to dialysis treatments | 0 | 0 | 0 | 4 (67%) | 2 (33%) |
| The dialysis team is more engaged with the patient/family | 0 | 0 | 0 | 2 (33%) | 4 (67%) |
| The program helps patients to reach target body weight and improve blood pressure control | 0 | 0 | 0 | 4 (67%) | 2 (33%) |
| The program improves patient/ family understanding of their disease condition and management | 0 | 0 | 0 | 3 (50%) | 3 (50%) |
| The joint reviews with physician and technicians on Sharesource® system are helpful and beneficial | 0 | 0 | 0 | 1 (17%) | 5 (83%) |
| In your opinion, what is the optimal nurse to patient ratio is? | 1:5 (100%), 1:10 (0%), 1:15 (0%), 1:20 (0%) | ||||
aA total of 6 questionnaires were distributed to 3 dialysis nurses, 2 paediatric nephrologists and 1 technical supporting staff for the cloud-based Sharesource®platform
Homechoice Claria® cycler equipped with the cloud-based Sharesource® platform (Baxter Healthcare, Deerfield, IL) was used in all patients. Through the Sharesource® platform, anonymized data (e.g. ultrafiltration volume, dwell time, alarms) and self-entered information (e.g. body weight and blood pressure) were accessible from the hospital. PD prescription could also be changed remotely. Dry weight and target blood pressure were set according to clinical and bioimpedance evaluation. Flag rules were set as follows (yellow flag and red flag, respectively): lost dwell time (45 min and 75 min), lost therapy volume (none and 10%), initial drain variance (none and 50%), adjusted peritoneal volume (none and 2) and events during treatment (5 and 10). Patient data were screened by the dialysis nurse at least twice a week. Detailed reviews were performed in patients with abnormal findings. Alternatively, patients might contact the team if they experienced problems. A case conference with the nephrologist was held once weekly during the first 2 weeks, bi-weekly during the third to twelfth week and monthly thereafter following programme launch. Dialysis interventions were communicated through phone calls and text messages by kidney nurses and the PD programme was adjusted online directly by the nephrologist.
Primary outcomes were unplanned hospitalization rates related to PD and fluid management, including daily ultrafiltration volume and blood pressure control. Secondary outcomes included the frequency of clinic visits, reviews and interventions performed through RPM, PD-related anthropometric and laboratory parameters, treatment compliance, and quality of life, which was assessed by the PedsQL 3.0 ESRD module pre- and post-RPM [10].
The study was performed according to the Declaration of Helsinki and was approved by the institutional review board of the Hong Kong Children’s Hospital, Hospital Authority, Hong Kong (HKCH-REC-2020–006).
IBM SPSS statistics version 26 software was used for all statistical analysis. Data were expressed in mean, standard deviation, median, number, percentage and 95% confidence intervals (95% CI) when appropriate. The mean difference of the continuous variables was analysed by paired t-test. A p-value less than 0.05 in two tails was treated as significant.
Results
At the beginning of the pre-RPM observation period, 8 out of 26 patients receiving APD were enrolled in the RPM programme. One patient received a kidney transplant 4 days after programme initiation and was excluded from the analysis. Seven patients (6 females; 6 Chinese and 1 Pakistani) were included. The median age at initiation of KRT and RPM enrolment was 9.7 years (IQR 2.3–14.9) and 11.3 years (IQR 2.6–17.1), respectively. The time on dialysis at programme enrolment was 17.2 months (IQR 7.6–27.2). Indications for RPM were history of significant fluid overload (n = 3), non-adherence (n = 2), repeated unplanned hospitalizations (n = 1) and social reason due to language barrier (n = 1). Ten patients who underwent APD without RPM (non-RPM group) during the pre- and post-RPM periods were included for analysis. Patients in the non-RPM group were older in age, on dialysis for a longer duration and had a lower prevalence of development delay. Details of the baseline demographics are presented in Table 1.
Table 1.
Baseline demographics for children receiving automated peritoneal dialysis with or without remote patient monitoring during the study period (March 2021 to February 2022)
| All (n = 17)a | RPM (n = 7) | Non-RPM (n = 10) | |
|---|---|---|---|
| Female | 12 (71) | 6 (86) | 6 (60) |
| Ethnicity | |||
| Chinese | 14 (82) | 6 (86) | 8 (80) |
| Pakistani | 2 (12) | 1 (14) | 1 (10) |
| White | 1 (6) | 0 (0) | 1 (10) |
| Age at enrolment, years | 14.9 (7.8–17.4) | 11.3 (2.6–17.1) | 16.9 (12.8–17.6) |
| Causes of kidney failure | |||
| Hereditary | 7 (41) | 3b (43) | 4d (40) |
| CAKUT | 4 (24) | 1 (14) | 3 (30) |
| Miscellaneous | 4 (24) | 1c (14) | 3e (30) |
| Glomerulopathy | 2 (12) | 2 (29) | 0 (0) |
| Age at KRT initiation, years | 13.2 (5.9–15.5) | 9.7 (2.3–14.9) | 14.3 (8.7–16.01) |
| Dialysis vintage, m | 27.2 (8.2–41.4) | 17.2 (7.6–27.2) | 34.6 (8.8–46.7) |
| Delayed development | 7 (41) | 4 (57) | 3 (30) |
Data expressed as number (&), or median (interquartile range), as appropriate
CAKUT, congential anomalies of kidney and urinary tract; KRT, kidney replacement therapy
aReasons for exclusion from the analysis (RPM group, one patient due to transplant; Non-RPM group, n = 8; transplant, n = 4; Transition, n = 2; migration, n = 1; death, n = 1)
bCongenital nephrotic syndrome due to NPHS1 variant, genetic podocytopathy due to PLCE1 variant and nephronophthisis
cIschaemic nephropathy following major cardiac surgery for congential heart disease
dRenal cystic disease, nephronophthisis, autosomal recessive polycystic kidney disease and autosomal dominant tubulointerstitial disease
eAtypical HUS, mitochrondial disease and transplant-associated thrombotic microangiopathy
Patient reviews and interventions
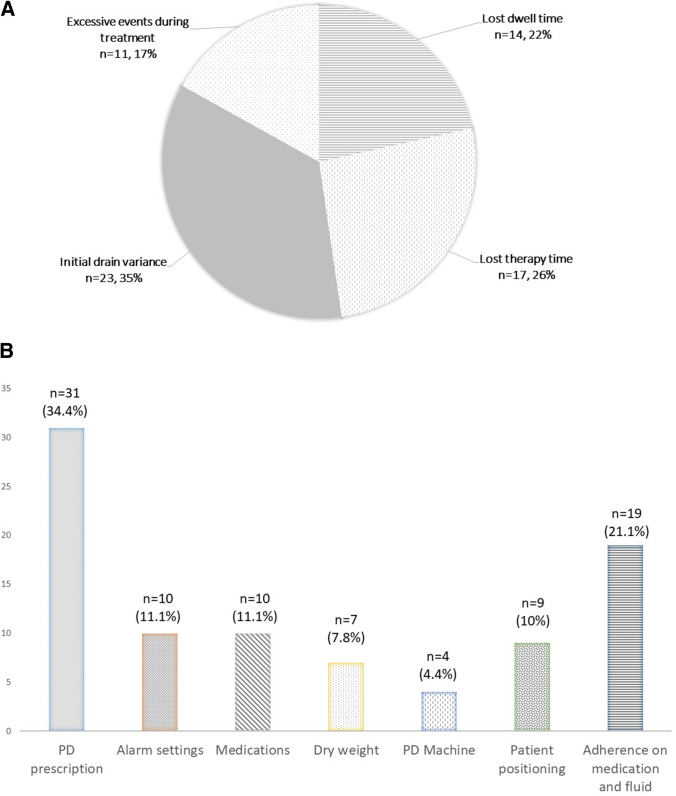
Over the 24-week post-RPM period, there were 72 regular screenings performed by nurses and 11 case conferences were held. There were 202 patient episodes of detailed reviews. Total time spent and phone consults performed were 222.9 min and 8.1 phone consultations per patient/24 weeks, respectively. There were 265 yellow flag and 65 red flag alarms. The leading reasons for red flag alarms were initial drain variance (n = 23, 35.4%) and lost therapy time (n = 17, 26.2%) (Fig. 1A). A total of 90 interventions were recorded (Fig. 1B), including change in PD prescription (n = 31, 34.4%), alarm settings (n = 10, 11.1%), medications (n = 10, 11.1%), dry weight (n = 7, 7.8%), PD machine (n = 4, 4.4%) and advice on patient positioning (n = 9, 10%) and adherence of medication and fluid restriction (n = 19, 21.1%). While there were 5 changes in PD prescription made at clinic during both pre- and post-RPM periods, 31 remote adjustments were performed through RPM. Compared to the pre-RPM period, there was a significant increase in PD regimen adjustments during the post-RPM period (0.7 to 5.1 adjustments per patient/24 weeks, p = 0.05).
Fig. 1.
A Reasons for red flag alarms during remote patient monitoring. B Interventions performed through remote patient monitoring programme
All children performed PD daily and did not miss PD treatment during the post-RPM period. However, one adolescent experienced the lost therapy time alarm for 13 episodes. The patient admitted that he decided for early termination of PD due to issues with schooling. With repeated counselling and support, the patient became adherent to PD treatments with no further issues.
Patient outcomes
Compared to the pre-RPM period, 4 patients (57.1%, 95% CI 22.5–100%) experienced fewer unplanned hospitalizations and 5 patients (71.4%, 95% CI 34.1–100%) had shorter hospital stays. The overall unplanned hospitalization rate and length of stay were reduced by 45% and 42%, respectively (Table 2). While the RPM group had a higher unplanned hospitalization rate than the non-RPM group at baseline (1 vs. 0.27 episode per patient/24 weeks, p = 0.03), following the implementation of RPM the hospitalization rates became similar between the two groups (0.55 vs. 0.27 episode per patient/24 weeks) (Table 3).
Table 2.
Outcomes of patients receiving automated peritoneal dialysis with remote patient monitoring
| Pre-RPM | Post-RPM | |
|---|---|---|
| Total no. of unplanned hospitalizations | 7 | 4 |
| Causes of hospitalization | ||
| Fluid retention | 2 (28.6) | 2 (50.0) |
| Leakage | 1 (14.3) | 0 (0.0) |
| Wet contamination | 1 (14.3) | 0 (0.0) |
| Drainage pain | 2 (28.6) | 0 (0.0) |
| Hypercalcemia | 1 (14.3) | 0 (0.0) |
| Malnutrition | 0 (0.0) | 1 (25.0) |
| Non-specific abdominal pain | 0 (0.0) | 1 (25.0) |
| Unplanned hospitalization rates, episode per patient/24-week | 1.0 | 0.55 |
| Unplanned hospitalization days, days per patient/24-week | 9.43 | 5.43 |
| Ultrafiltration volume, ml per day | 501.7 ± 286.6* | 565.6 ± 248.7* |
| Systolic blood pressure, mmHg | 119.9 ± 11.2** | 114.1 ± 12.6** |
| Diastolic blood pressure, mmHg | 79.3 ± 7.4 | 75.4 ± 10.8 |
| No. of anti-hypertensive medications | 1.9 ± 1.0 | 2.0 ± 0.6 |
| Dialysis adequacy (n = 6) | ||
| Kt/V (total) | 2.4 ± 0.7 | 2.3 ± 0.6 |
| Kt/V (dialysate) | 2.1 ± 0.7 | 2.1 ± 0.6 |
| Kt/V (urine) | 0.4 ± 0.6 | 0.3 ± 0.4 |
| QOL (parent) (n = 7) | 61.2 ± 12.2 | 57.3 ± 13.1 |
| QOL (patient) (n = 4) | 72.1 ± 6.7 | 62.5 ± 12.3 |
*p = 0.03; **p = 0.02
QOL, quality of life measure by PedsQL 3.0 ESRD module; RPM, remote patient monitoring
Table 3.
Outcomes during pre-RPM period (March 2021 to August 2021) between patients who were and were not enrolled into RPM
| Pre-RPM period | Post-RPM period | |||
|---|---|---|---|---|
| RPM | Non-RPM | RPM | Non-RPM | |
| Total No. of unplanned hospitalizations | 7 | 3 | 4 | 3 |
| Causes of hospitalization | ||||
| Fluid retention/hypertension | 2 (28.6) | 3 (100) | 2 (50) | 2 (66) |
| Leakage | 1 (14.3) | 0 (0) | (0) (0) | 0 (0) |
| Wet contamination | 1 (14.3) | 0 (0) | (0) (0) | 0 (0) |
| Drainage pain | 2 (28.6) | 0 (0) | (0) (0) | 0 (0) |
| Hypercalcemia | 1 (14.3) | 0 (0) | (0) (0) | 0 (0) |
| Malnutrition | 0 (0) | 0 (0) | (0) (25) | 0 (0) |
| Non-specific abdominal pain | 0 (0) | 0 (0) | (0) (25) | 0 (0) |
| Hyponatraemia | 0 (0) | 0 (0) | (0) (0) | 1 (33) |
| Unplanned hospitalization rates, episode per patient/24-week | 1* | 0.27* | 0.55 | 0.27 |
| Unplanned hospitalization days, days per patient/24-week | 9.43 | 2.36 | 5.43 | 1.91 |
| Systolic blood pressure, mmHg | 119.9 ± 11.2 | 121.1 ± 11.4 | 114.1 ± 12.6 | 123.4 ± 10.9 |
| Diastolic blood pressure, mmHg | 79.3 ± 7.4 | 78 ± 8.68 | 75.4 ± 10.8 | 79.4 ± 8.21 |
*p = 0.03
Data presented as number (%), mean ± SD, as appropriate
RPM, remote patient monitoring
There was a significant increase in daily ultrafiltration (565.6 ± 248.7 vs. 501.7 ± 286.6 ml/day, p = 0.03) and reduction in systolic blood pressure (114.1 ± 12.6 vs. 119.9 ± 11.19 mmHg, p = 0.02) during the post-RPM period. There was also a trend of reduction in diastolic blood pressure (75.4 ± 10.8 vs. 79.3 ± 7.4 mmHg, p = 0.09). The number of anti-hypertensives prescribed was not different during the two periods. There was no difference between the number of clinic visits (7.4 ± 3.0 vs. 7.1 ± 2.2 visits per patient/24 weeks), as well as Kt/V, haemoglobin, calcium, phosphate and parathyroid hormone levels during the two periods. Details on the primary and secondary outcomes are presented in Table 2. None of the patients on RPM required conversion to haemodialysis.
Change of RPM over time
Compared to the first 3 months following the implementation of the RPM programme, the average time spent for reviews was significantly reduced by 41.1% (46.8 to 27.5 min per patient month, p < 0.001) during the second half of the post-RPM period. The number of red alarms per patient was also reduced by 36.8% (1.9 to 1.2 episodes per patient, p = 0.22). The rate of PD regimen adjustment was the same at 0.7 adjustments per patient month.
Quality of life and perceptions
Although the quality of life measured by the PedsQL 3.0 ESRD module was not different between the pre- and post-RPM periods, all patients agreed in the questionnaires that the use of RPM was beneficial, which improved their quality of life, sense of security, adherence and reduced PD-related clinic visits, admissions and related complications (Table 4). A follow-up questionnaire distributed to the dialysis team also demonstrated positive perceptions towards patient adherence, engagement, disease understanding and fluid management. Despite an improved efficiency in data interpretation with time, a significant proportion of dialysis team members (> 80%) expressed concerns about the burden of workload which might have affected their regular clinical duties. Nonetheless, most members (83%) agreed that the programme should be extended to all patients on APD with additional manpower support, preferably with an improved nurse-to-patient ratio of 1:5 (Table 5).
Cost savings related to reduction in hospitalizations
We estimated the total medical expenditure related to unplanned hospitalizations for management of complications arising from PD. The average nominal cost for hospitalization in public hospital was USD 653.8/day. The reduced bed days in our cohort was 8.0 days/patient year, resulting in a cost saving of USD 5230.4/patient year.
Discussion
In this prospective study, children on APD benefited from RPM with fewer unplanned hospitalizations and shorter hospital stays. In addition, fluid management improved with increased daily ultrafiltration and lower systolic blood pressure. There was satisfactory treatment adherence and favourable perception to the RPM programme. Over time, our data demonstrated an improved PD performance as evidenced by fewer number of alarms, and consequently the time required for RPM service also was significantly reduced.
Hospitalizations are common among children receiving PD [2, 4]. The main objective of RPM is to identify and resolve problems early, in order to reduce PD-related complications and avoid unnecessary admissions. Our data showed that 57% and 71% children had fewer unplanned admissions and shorter hospital stays, respectively. This may be attributable to the intensification of patient monitoring, frequent communications and timely interventions. In accordance with previous reports [11], there was enhanced blood pressure control following counselling on fluid restriction and proactive PD adjustments optimizing ultrafiltration. This postulation is supported by the fact that a similar number of anti-hypertensive medications were prescribed between the pre- and post-RPM periods. Furthermore, all our patients demonstrated excellent adherence to their PD treatments following the implementation of RPM. Non-compliance is well known to be prevalent among PD patients [12, 13], and may lead to higher rates of peritonitis, hospitalizations and mortality. Specifically, non-adherence was correctly identified in an adolescent and favourable outcome was observed following appropriate counselling. Minimizing hospital visits is important to facilitate schooling, and to reduce infection risk especially during the COVID-19 pandemic [11]. Indeed, questionnaires distributed to patients and dialysis teams showed that RPM was associated with better patient engagement, adherence and disease awareness.
The success of RPM relies much on dedicated nurses who have spent hours on patient reviews, communications and interventions. Fortunately, the hours required for RPM tend to decrease over time because patients’ clinical status often improves. There were also fewer red flag alarms (reduced by 37%) during the second half of the post-RPM period. Smoother procedures may lead to better quality of sleep for patients at night [9]. Importantly, frequent patient reviews pose a significant burden on the dialysis team. Appropriate manpower provision and facilitation should be offered to ensure sustainability and quality of the RPM programme.
The use of RPM is associated with improved quality of life in children and adults [8, 9]. A recent survey showed that 90% of patients/caregivers felt safe under RPM [9], although the scales on quality of life did not reflect an improvement in our cohort. There are a few explanations. First, the number of patients was too small to detect any significant difference. Second, initial frequent communications with patients/caregivers might have raised the awareness of their poor physical health and induced stress. However, as shown in our questionnaires, all patients perceived a better quality of life and sense of security. We believe that the benefit of improved quality of life would be more apparent with an extended observational period on RPM.
Although we have a small sample size, our study provides important prospective outcome data with minimal missing data. However, there is potential selection bias as the most complicated patients were enrolled in the RPM programme since only limited PD machines in our unit had RPM function. Second, while there was a reduction in hospitalizations following implementation of RPM, improvement in certain hospitalizations, such as wet contamination, might not be truly measurable by the RPM programme.
In conclusion, cloud-based RPM in children receiving APD is associated with fewer and shorter unplanned hospital visits, improved fluid management and excellent adherence to PD. The programme also improves patient engagement and disease awareness, and may potentially save medical expenditures due to fewer hospitalizations. Further well-designed studies with larger paediatric cohorts are required to evaluate patient-centred outcomes.
Supplementary information
Below is the link to the electronic supplementary material.
Acknowledgements
We would love to thank Ms. Ivy Yu from Baxter Healthcare who provided staff training and technical support on the use of the Sharesource® platform. We would also love to thank occupational therapist Ms. Phoebe Chan for performing the quality of life assessment.
Declarations
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Chan Y, Ma AL, Tong P, Lai W, Tse NK. Chronic peritoneal dialysis in Chinese infants and children younger than two years. Hong Kong Med J. 2016;22:365–371. doi: 10.12809/hkmj154781. [DOI] [PubMed] [Google Scholar]
- 2.Chan EYH, Yap DYH, Wong WHS, Ho TW, Tong PC, Lai WM, Chan TM, Ma ALT. Demographics and long-term outcomes of children with end-stage kidney disease: a 20-year territory-wide study. Nephrology (Carlton) 2022;27:171–180. doi: 10.1111/nep.14007. [DOI] [PubMed] [Google Scholar]
- 3.Chan EY, Borzych-Duzalka D, Alparslan C, Harvey E, Munarriz RL, Runowski D, Vidal E, Coccia PA, Jankauskiene A, Principi I. Colostomy in children on chronic peritoneal dialysis. Pediatr Nephrol. 2020;35:119–126. doi: 10.1007/s00467-019-04372-x. [DOI] [PubMed] [Google Scholar]
- 4.Neu AM, Sander A, Borzych-Dużałka D, Watson AR, Vallés PG, Ha IS, Patel H, Askenazi D, Bałasz-Chmielewska I, Lauronen J. Comorbidities in chronic pediatric peritoneal dialysis patients: a report of the International Pediatric Peritoneal Dialysis Network. Perit Dial Int. 2012;32:410–418. doi: 10.3747/pdi.2012.00124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chua AN, Warady BA. Adherence of pediatric patients to automated peritoneal dialysis. Pediatr Nephrol. 2011;26:789–793. doi: 10.1007/s00467-011-1792-2. [DOI] [PubMed] [Google Scholar]
- 6.Lai W-M. Quality of life in children with end-stage renal disease: does treatment modality matter? Perit Dial Int. 2009;29:190–191. doi: 10.1177/089686080902902S38. [DOI] [PubMed] [Google Scholar]
- 7.Jung H-Y, Jeon Y, Kim YS, Kim DK, Lee JP, Yang CW, Ko EJ, Ryu D-R, Kang S-W, Park JT. Outcomes of remote patient monitoring for automated peritoneal dialysis: a randomized controlled trial. Nephron. 2021;145:702–710. doi: 10.1159/000518364. [DOI] [PubMed] [Google Scholar]
- 8.Uchiyama K, Morimoto K, Washida N, Kusahana E, Nakayama T, Itoh T, Kasai T, Wakino S, Itoh H. Effects of a remote patient monitoring system for patients on automated peritoneal dialysis: a randomized crossover controlled trial. Int Urol Nephrol. 2022;54:2673–2681. doi: 10.1007/s11255-022-03178-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Uzun Kenan B, Demircioglu Kilic B, Akbalık Kara M, Taktak A, Karabay Bayazit A, Yuruk Yildirim ZN, Delibas A, Aytac MB, Conkar S, Kaya Aksoy G. Evaluation of the Claria sharesource system from the perspectives of patient/caregiver, physician, and nurse in children undergoing automated peritoneal dialysis. Pediatr Nephrol. 2022;14:1–7. doi: 10.1007/s00467-022-05563-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Goldstein SL, Graham N, Warady BA, Seikaly M, McDonald R, Burwinkle TM, Limbers CA, Varni JW. Measuring health-related quality of life in children with ESRD: performance of the generic and ESRD-specific instrument of the Pediatric Quality of Life Inventory (PedsQL) Am J Kidney Dis. 2008;51:285–297. doi: 10.1053/j.ajkd.2007.09.021. [DOI] [PubMed] [Google Scholar]
- 11.Bunch A, Ardila F, Castaño R, Quiñonez S, Corzo L. Through the storm: automated peritoneal dialysis with remote patient monitoring during COVID-19 pandemic. Blood Purif. 2021;50:279–282. doi: 10.1159/000511407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Griva K, Lai AY, Lim HA, Yu Z, Foo MWY, Newman SP. Non-adherence in patients on peritoneal dialysis: a systematic review. PLoS ONE. 2014;9:e89001. doi: 10.1371/journal.pone.0089001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bernardini J, Nagy M, Piraino B. Pattern of noncompliance with dialysis exchanges in peritoneal dialysis patients. Am J Kidney Dis. 2000;35:1104–1110. doi: 10.1016/S0272-6386(00)70047-3. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.