Abstract
Background
There is still uncertainty on whether ionizing radiation from CT scans can increase the risks of cancer. This study aimed to identify the association of cumulative ionizing radiation from CT scans with pertaining cancer risks in adults.
Methods
Five databases were searched from their inception to November 15, 2020. Observational studies reporting cancer risks from CT scans in adults were included. The main outcome included quantified cancer risks as cancer case numbers in exposed/unexposed adult participants with unified converted measures to odds ratio (OR) for relative risk, hazard ratio. Global background radiation (2.4 mSv per year) was used as control for lifetime attribution risk (LAR), with the same period from incubation after exposure until survival to 100 years.
Results
25 studies were included with a sum of 111,649,943 participants (mean age: 45.37 years, 83.4% women), comprising 2,049,943 actual participants from 6 studies with an average follow-up period as 30.1 years (range, 5 to 80 years); 109,600,000 participants from 19 studies using LAR. The cancer risks for adults following CT scans were inordinately increased (LAR adults, OR, 10.00 [95% CI, 5.87 to 17.05]; actual adults, OR, 1.17 [95%CI, 0.89 to 1.55]; combined, OR, 5.89 [95%CI, 3.46 to 10.35]). Moreover, cancer risks elevated with increase of radiation dose (OR, 33.31 [95% CI, 21.33 to 52.02]), and multiple CT scan sites (OR, 14.08 [95% CI, 6.60 to 30.05]). The risk of solid malignancy was higher than leukemia. Notably, there were no significant differences for age, gender, country, continent, study quality and studying time phrases.
Conclusions
Based on 111.6 million adult participants from 3 continents (Asia, Europe and America), this meta-analysis identifies an inordinately increase in cancer risks from CT scans for adults. Moreover, the cancer risks were positively correlated with radiation dose and CT sites. The meta-analysis highlights the awareness of potential cancer risks of CT scans as well as more reasonable methodology to quantify cancer risks in terms of life expectancy as 100 years for LAR.
Prospero trial registration number
CRD42019133487.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12885-022-10310-2.
Keywords: Computed tomography, Adult, Cancer risk, Radiation exposure, Meta-analysis
Background
As an auxiliary diagnostic imaging modality, CT scans play an important role in the diagnosis of various complicated diseases [1]. As new clinical indications continue to be discovered, the use of CT scan has increased rapidly over the past decade worldwidely [2–5]. Although CT scans are of great diagnostic benefits to individual patients, relatively higher radiation doses are delivered compared with other conventional imaging modalities [1, 6, 7]..
Substantial follow-up works are now underway to determine whether patients experiencing ionizing radiation from CT scans can significantly increase the risk of cancer. Indeed, many lines of evidence have shown that ionizing radiation from CT scans is associated with cancer/tumor risks [8–12]. Whereas recently epidemiological studies have examined potential disease risks from pediatric CT scanning, studies of cancer risk from CT scans during adulthood are also of critical important for public health. Adults receive over 10 times more CT scans than children [13–15] and most radiation induced cancers occur during middle or older age.
However, researchers questioned the validity of the earlier indirect estimates based on uncertain risk projections for radiation [16], whereas others declared that the findings of recent CT scan studies need to be interpreted with caution, due to the possibility of reverse causation [17]. Therefore, we cannot necessarily attribute all excess cases of cancer to ionizing radiation during the period of follow-up after patients undergoing CT scans, since the decision of performing CT scans is not allocated randomly but based on medical indications. For example, patients with precancerous symptoms or early symptoms that might prompt their physician to perform a CT scan, which may lead to the possibility of reverse causation. Until now, there is still uncertainty on whether ionizing radiation from CT scans can increase the risks of cancer. It can be hard for the medical community to design and conduct studies to validate previous reports. Prior studies have presented the evidence on cancer and mortality risks of young scoliosis patients from cumulative radiographic radiation (spinal radiographs) [18] and tentatively proposed low radiation X-ray methodology [19], focusing on the issue dynamically with the medical community [20–24] with the aims for benefiting global patients and the public. Consequently, this work aims to evaluate whether adult CT scan exposure can increase the risks of cancer during the follow-up observation, based on systematic review and meta-analysis of global observational studies.
Methods
Protocol
This systematic review and meta-analysis was successfully registered in the International Prospective Register of Systematic reviews (PROSPERO) on 28 April 2019, with the Registration number CRD42019133487. The initial enrollment was to study the relationship between CT screening and cancer risk in children and adolescents. We revised the registry for adult CT scans and cancer risks due to a published study by another group during our studying process [25]..
Study types
Observational studies, cohort studies, and case-control studies were included in this systematic review and meta-analysis, while studies assessed review articles, proceedings, case series and case reports were excluded. No restrictions to the language of publication were applied to select primary studies. We added extra studies by additional hand search of the reference lists of related articles.
Population
Patients meeting the following requirements were included in this study: 1) Patients were older than 18 years for their first CT scan, 2) Patients did not have any malignant diseases (such as precancerous symptoms or early symptoms of the cancer) prior to their first CT scan, 3) Patients with follow-up of more than 1 year after their first scan. 4) Patients underwent at least one CT scan. Those with the following conditions were excluded: 1) Patients with incomplete demographic data or data errors, 2) Patients lost follow-up for a variety of factors.
Interventions
Included patients received one or more electronically archived CT scans. The absorbed dose from a CT scan mainly depends on factors including age, sex, examination site, year of scan (2007 as a milestone for CT scans in terms of dose reduction in Annals of the ICRP [26]), and machine parameters.
Outcome measures
The study included two outcome measures.
Cancer risks, including each part of the human body with at least 1 year lag period after first exposure.
The potential associations between adult CT scan exposure radiation doses and cancer risks, including lifetime attribute risk (LAR).
Search methods for identification of studies
A thorough search was conducted in the following global databases, PubMed, Medline, EMBASE, Web of Science, Springer Link, Cochrane Library, from inception through Nov 15th 2020 with terms including “Adult”, “computerized tomography, X ray”, and “Cancer”. Search strategies were designed by an experienced librarian and revised by another librarian according to the PRESS (Peer Review of Electronic Search Strategies) checklist [27]. Three reviewers performed the selection process independently, and disagreements were discussed and resolved by a fourth reviewer.
Assessment of risk of bias (ROBs) in included studies
The ROBs of each study were assessed independently by two authors according to Newcastle-Ottawa Scale [28]..
Dealing with missing data
When missing data were encountered in included articles, we contacted the corresponding authors of relevant studies for these data by sending electronic mails twice with time interval of 1 month. When the study did not provide the number of cancer cases in the case/exposed group or control/unexposed group, the numbers were calculated by the formula defined as Re/Ru for RR (Relative Risk), Re (1- Ru)/[Ru (1- Re)] for OR (the rate in exposed persons, denoted Re; the rate in unexposed persons, denoted Ru), and (Or/Oe)/(Cr/Ce) for HR (experimental group, denoted O; control group, denoted C; actual persons, denoted r; theoretical persons, denoted e) [29].
Assessment of publication bias
Publication bias were explored by a funnel plot (i.e., plots of study results against precision), Egger’s tests [30]..
Subgroup analysis
In studies using LAR to estimate cancer risks, control group was defined according to the global incidence of cancer at 2.4 mSv of background radiation dose, i.e., lifetime baseline risk (LBR) [31]. We used the radiation risk models for sex- and organ-specific cancer incidence developed by the National Research Council’s BEIR VII committee [32]. LAR calculates the risk of cancer from an incubation period after radiation exposure (5 years for solid cancer and 2 years for leukemia) to survival to 100 years. LAR and LBR were calculated with the same method. For an age with a mantissa of 5: 45 years, divide by 2 the sum of the LAR values for 40 years and 50 years. For example, the LAR at the age of 45 years is (141 + 70)/2 = 105.5 at full 100 mSv (141 cases per 100,000 at 40-year-old and 70 cases per 100,000 at 50-year-old by BEIR VII). If less than 100 mSv, if it was 2.4 mSv, the LAR at the age of 40 years, 45 years, and 50 years are 41(2.4/100), 105.5(2.4/100), and 70(2.4/100), respectively.
Sensitivity analysis
Sensitivity analysis was performed by removing relevant studies to observe whether the homogeneity and the results change significantly. If the heterogeneity was too large to be analyzed, descriptive analyses were presented.
Assessment of heterogeneity
All analyses were performed using Stata V.16.0 software (Stata Corp LP, College Station, Texas, USA). Heterogeneity among primary studies were analyzed using standard Chi-squared tests (P value) and the Ι2 statistic as recommended by the Cochrane Handbook for Systematic Reviews of Interventions [33]. We interpreted Ι2 values according to Deeks [34]. Meta-regression was used to explore prior factors that may be important sources of heterogeneity. P < 0.05 was considered as a statistically significant.
Results
Hallmarks of included studies
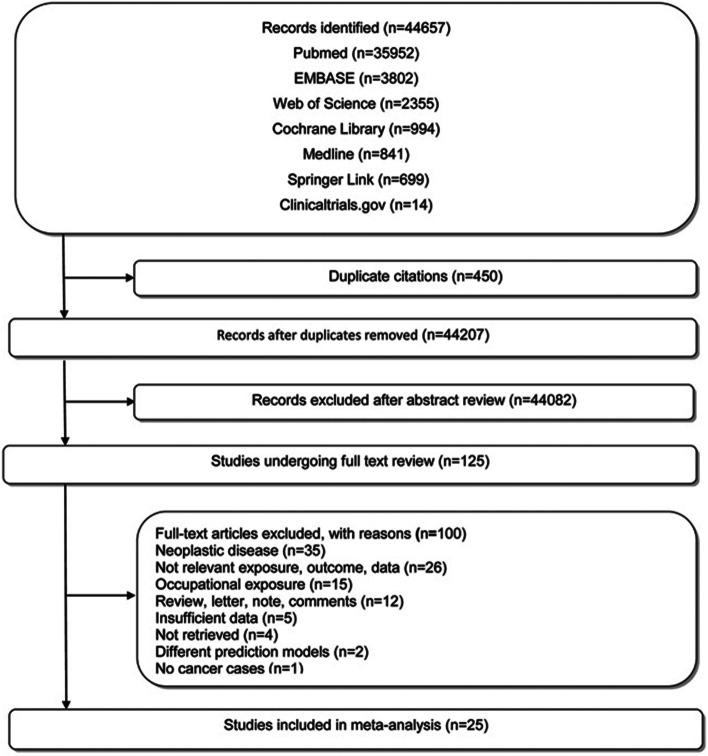
The literature-retrieving strategy and pertaining results were shown in Fig. 1 and Table S1. A total of 44,657 relevant studies were preliminarily reviewed. In total, 125 studies satisfied the eligibility criteria for full-text screening and 25 were eventually included for this meta-analysis [2, 8–11, 35–54]. Of these 25 studies, 5 reported actual cancer cases [8, 10, 11, 35, 36], 19 estimated cancer cases by calculating LAR [2, 9, 37–53], 1 reported expected cancer cases in control group [54], and all were published since 2007. Four studies were excluded due to incomplete data [12, 55–57]. Additional data were requested by contacting the authors of 4 studies via repeated electronic mails with a time interval of 1 month (Table S2). However, no replies and requested data were received.
Fig. 1.
Flowchart of literature screening process
Participant hallmarks
Studies were predominantly from the US (n = 11), followed by China (n = 4), Sweden (n = 3), Greece (n = 2), and Canada, Italy, Thailand, Croatia, and Denmark (n = 1 each); One study [42] included populations from China (Hong Kong) and the US. Based on the LAR base number of 100,000 persons, 25 studies were included with a sum of 111,649,943 participants, comprising 2,049,943 actual participants (111,449 cases VS 1938494 controls) from 6 studies; 109,600,000 estimated participants (54,912,051 participants received at least one CT radiation) from 19 studies. The average age was 51.3 years in CT scan exposed group (range, 20.0 to 94.0 years) and 40.5 years in control group (range, 25 to 84 years). The gender was reported in 19 studies of CT scan exposed group and 5 studies of unexposed group. The proportion of women was even higher (53.0%, 140,305 participants in CT group; 85.6%, 1,914,381 participants in unexposed group). The average follow-up time was 12.1 years in actual group (range, 5.9 to 22.5 years), 48.0 years (range, 5 to 80 years) in LAR group. The radiation dose per capita was 66.7 mSv (range:5.15 mSv to 122 mSv). A detailed list of study hallmarks was shown in Table 1 and Table 2.
Table 1.
Detailed information and characteristics of participants
| Study | Country | Participants | Age (Y) | Gender (F/M) | Cancer Case | Participants/Cancer case | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| E | U | E | U | E | U | E | U | LAR | LBR | ||
| Burton [ 35], et al. 2018 | Canada | 5859 | 1,292,059 | 30.4 ± 5.8 | 28.7 ± 5.7 | F:5859 | F:1292059 | 27 | 10,080 | – | – |
| Nordenskjöld [ 10], et al. 2017 | Sweden | 15,534 | 51,398 | 41.27 | 38.88 |
7786 7748 |
26,296 25,102 |
23 | 45 | – | – |
| Hung [ 36], et al. 2013 | China | 18,697 | 19,109 | 62.5 ± 13.9 | 60.2 ± 14.8 |
8330 10,367 |
10,799 8310 |
954 | 885 | – | – |
| Olsen [ 54], et al. 2014 | Denmark | 15,104 | 15,104 | NA | NA | NA | NA | 37 | NA | – | – |
| Shao [ 8], et al. 2020 | China | 56,050 | 560,491 | > 25 | > 25 |
31,857 24,193 |
309,561 241,921 |
2552 | 17,613 | – | – |
| Davis [ 11], et al. 2011 | America | 205 | 333 | 48.5 ± 12.3 | 49.8 ± 12.5 |
91 114 |
178 155 |
56 | 72 | – | – |
| Rampinelli [ 9], et al. 2017 | Italy | 5203 | NA | > 50 | NA |
1764 3439 |
NA | – | – |
1,600,000 653 |
1,600,000 136.9 |
| Kritsaneepaiboon [ 37], et al. 2016 | Thailand | 328 | NA | 40.10 ± 15.81 | NA |
73 255 |
NA | – | – |
100,000 140 |
100,000 18.4 |
| Griffey [ 38], et al. 2009 | America | 130 | NA | 56 | NA |
82 48 |
NA | – | – |
100,000 1219.5 |
100,000 14.4 |
| Einstein [ 39], et al. 2008 | America | 50 | NA | 61 ± 12 | NA |
20 30 |
NA | – | – |
100,000 62.7 |
100,000 12.9 |
| Faletra [ 40], et al. 2010 | Sweden | 749 | NA | 52–70 | NA |
280 469 |
NA | – | – |
2,100,000 5306.9 |
2,100,000 253.1 |
| Niemann [ 41],et al 2013 | Sweden | 691 | NA | 66 | NA |
339 352 |
NA | – | – |
8,700,000 538.6 |
8,700,000 143.3 |
| Huang [ 42], et al. 2009 | China and America | NA | NA | 20–80 | NA | NA | NA | – | – |
23,200,000 19,493 |
23,200,000 404.1 |
| Perisinakis [ 43], et al. 2015 | Greece | 136 | NA |
F:59.4 M:60.6 |
NA |
41 95 |
NA | – | – |
100,000 71.5 |
100,000 27 |
| Smith-Bindman [ 2], et al. 2009 | America | 1119 | NA | 59 | NA |
535 584 |
NA | – | – |
200,000 423 |
200,000 51.6 |
| Sodickson [ 44], et al. 2009 | America | 31,462 | NA | 56.9 ± 17.5 | NA | 13,843 17,619 | NA | – | – |
100,000 300 |
100,000 28.9 |
| Huang [ 45], et al. 2009 | China | NA | NA | 20–80 | NA | NA | NA | – | – |
12,500,000 8690 |
12,500,000 204.1 |
| Huda [ 46], et al. 2010 | America | 100 | NA | 59 ± 11 | NA |
60 40 |
NA | – | – |
200,000 231 |
200,000 29.5 |
| Perisinakis [ 47], et al. 2012 | Greece | 62 | NA |
F:61 ± 10 M:59 ± 13 |
NA |
31 31 |
NA | – | – |
200,000 59 |
200,000 25.8 |
| Einstein [ 48], et al. 2007 | America | NA | NA | 61 ± 12 | NA | NA | NA | – | – |
800,000 1702.1 |
800,000 134.9 |
| Kim [ 49], et al. 2009 | America | NA | NA |
F:45–75 M:55–75 |
NA | NA | NA | – | – |
1,300,000 102 |
1,300,000 112.7 |
| Majer [ 50], et al. 2018 | Croatia | NA | NA | NA | NA | NA | – | – |
2,200,000 1410 |
2,200,000 55.3 |
|
| Salibi [ 51], et al. 2014 | America | 67 | NA | 34 ± 14 | NA |
54 13 |
NA | – | – |
100,000 810 |
100,000 39.4 |
| Shah [ 52], et al. 2013 | America | 24,393 | NA | 23–84 | NA | NA | NA | – | – |
200,000 1163.5 |
200,000 32.7 |
| Wylie [53], et al. 2018 | America | 3863 | NA | 20–60 | NA |
3434 429 |
NA | – | – |
1,000,000 880 |
1,000,000 199.7 |
Abbreviations: E Exposed, U Unexposed, NA Not available, Y Years, F Female, M Male, LAR Lifetime attributable risks, LBR Lifetime background risks
Table 2.
Detailed information and characteristics of included studies
| Study | Study Type | Reason | CT scanner | NO. of CT scan (CT sites, participants) |
Effective dose (mSv) | Cumulative dose (mSv) | Observation time | Follow-up (Y) | |
|---|---|---|---|---|---|---|---|---|---|
| E | U | ||||||||
| Burton [ 35], et al. 2018 | A | NA | NA |
1, 5655 ≥ 2, 204 |
10–44 | NA | 1995–2004 | 5.9 | 11.1 |
| Nordenskjöld [ 10], et al. 2017 | A | NA | NA | NA | NA | NA | 1973–1992 | 22.5 | 21.9 |
| Hung [ 36], et al. 2013 | A | CAD | NA |
1–5, 657 ≥ 6, 297 |
NA | NA | 1997–2010 | 12 | 12 |
| Olsen [ 54], et al. 2014 | A | CHD | NA | NA | NA | NA | 1977–2008 | 8 | 8 |
| Shao [ 8], et al. 2020 | B | NA | NA |
1–3, 17,423 ≥ 4, 2742 |
0.5–17.0 | 0.5–71.3 | 2000–2013 |
9.3- 9.9 |
9.3- 9.9 |
| Davis [ 11], et al. 2011 | B | NA | NA |
1–2, 98 ≥ 3, 30 |
2 | 1.5–10 | 2003.04–2007.12 | 2 | 2 |
| Rampinelli [ 9], et al. 2017 | C | LCS | NA | NA |
M: 1.0 F: 1.4 |
M: 9.3 F: 13 |
2004–2015 | 10 | 10 |
| Kritsaneepaiboon [ 37], et al. 2016 | NA | Multiple-injury | 64-multislice and 16-multislice CT, Phillips Brilliance | NA | NA | 19.43 ± 21.31 |
2013.01.01- 2013.12.31 |
1 | NA |
| Griffey [ 38], et al. 2009 | C | Emergency | 64-multislice CT | NA | NA | 122 | 2005.06.01–2006.05.31 | 7.7 | NA |
| Einstein [ 39], et al. 2008 | C | NA | Siemens AG 16-slice | NA | 8.8 ± 2.9 | NA | NA | NA | NA |
| Faletra [ 40], et al. 2010 | C | Coronary disease | 64-slice CTCA | NA |
Low: M 3.9 ± 1.7, F 3.1 ± 1.4 High: M 23.3 ± 4.0, F 22.3 ± 2.3 |
NA | NA | NA | NA |
| Niemann [ 41],et al 2013 | C | PE | Siemens Sensation 64;Siemens Sensation 16;Siemens Sensation 10 | NA | 4.35 ± 0.31 | NA | NA | 1 | NA |
| Huang [ 42], et al. 2009 | NA | NA | 64-detector CT system (Discovery PET/CT,GE Healthcare, Milwaukee, Wis) | NA |
M: 13.65, 24.80, 32.18 F: 13.45, 24.79, 31.91 |
100 | NA | NA | NA |
| Perisinakis [ 43], et al. 2015 | D | NA | 256-slice CT | NA | 8.9 ± 0.9 | NA | NA | NA | NA |
| Smith-Bindman [ 2], et al. 2009 | E | Various | NA | NA | 2–31 | NA | 2018.01.01–2018.05.30 | NA | NA |
| Sodickson [ 44], et al. 2009 | A | Cysticfibrosis | NA | NA | NA | 54.3 | 1985–2007 | NA | NA |
| Huang [ 45], et al. 2009 | D | NA | NA | NA | 45–73 | 100 | NA | NA | NA |
| Huda [ 46], et al. 2010 | C | CAD | 64-MDCT scanner (Siemens Healthcare) | NA | 25.1 ± 4.9 | NA | NA | NA | NA |
| Perisinakis [ 47], et al. 2012 | NA | NA | 256-slice TRO-CTA | NA |
M: 3.8 ± 0.7 F: 6.5 ± 1.0 |
NA | NA | NA | NA |
| Einstein [ 48], et al. 2007 | NA | NA |
64-slice scanner (SOMATOM Sensation 64, Siemens AG, Munich, Germany) |
NA | NA |
lungs: 42–91 breast: 50–80 |
NA | NA | NA |
| Kim [ 49], et al. 2009 | NA | CAC | NA | NA | 3.1 | NA | 2000–2005 | NA | NA |
| Majer [ 50], et al. 2018 | NA | NA | NA | NA | NA | 16–94 | NA | NA | NA |
| Salibi [ 51], et al. 2014 | E | TBI | NA | NA | 87 ± 45 | NA | NA | NA | |
| Shah [ 52], et al. 2013 | C | Emergency | NA | 83.4 | NA | 2001.01–2007.12 | NA | NA | |
| Wylie [53], et al. 2018 | C | Hip Pain | NA | 30 | NA | 2015.01–2016.12 | NA | NA | |
ROBs of included studies
The quality scores of 5 studies with actual participants and cancer cases ranged from 7 to 9 (Table S3A). Twenty of the studies were estimated cancer incidence and not case-control or cohort studies, the quality scores ranged from 2 to 6 (Table S3B).
Cancer risk from CT scans
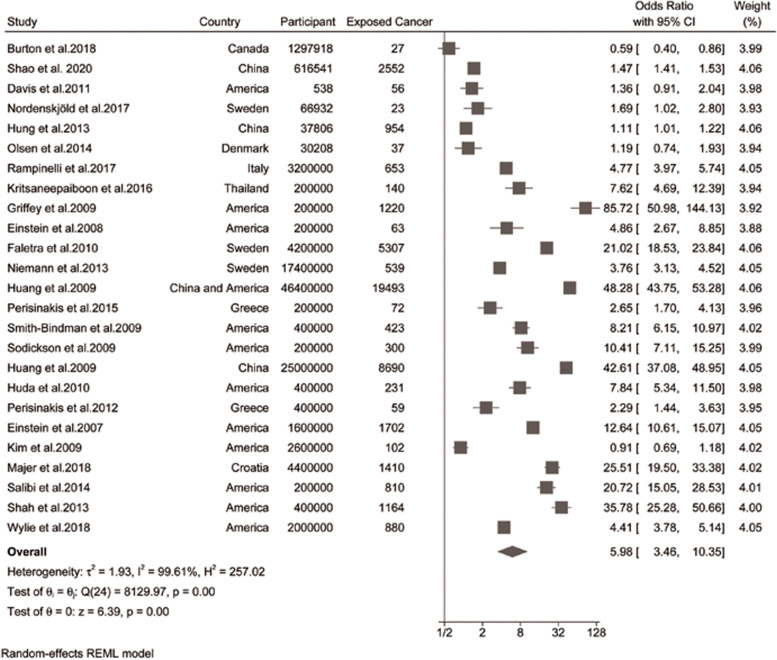
The cancer risks for adults following CT scans were inordinately increased (LAR adults, OR, 10.00 [95% CI, 5.87 to 17.05], Ι2 = 99.14; actual adults, OR, 1.17 [95%CI, 0.89 to 1.55], Ι2 = 94.18; combined, OR, 5.89 [95%CI, 3.46 to 10.35], Ι2 = 99.61%; Fig. 2). Notably, cancer incidence for actual studies was significantly different due to the inclusion (OR, 1.17 [95% CI, 0.89 to 1.55]) or exclusion (OR, 1.32 [95% CI, 1.11 to 1.56]) of Bruton’s study [35] with pregnant women participants (Table 3). Subgroup analysis indicated that cancer risks from LAR studies were significantly higher than actual studies (P = 0.00; Table 3), due to a longer life expectancy (to 100 years) used for calculation.
Fig. 2.
Forest plot of cancer risk from CT scans versus unexposed group
Table 3.
Subgroup analyses of CT exposure radiation and the risks of cancer incidence
| n | OR (95% CI) | I [2](%), Pheterogeneity | Psubgroup | |
|---|---|---|---|---|
| All studies | 25 | 5.98 (3.46 to 10.35) | 99.61, 0.00 | |
| Sex | 0.71 | |||
| Men | 18 | 4.89 (2.40 to 9.96) | 99.43, 0.00 | |
| Women | 18 | 5.88 (2.96 to 11.68) | 99.52, 0.00 | |
| Age | 0.93 | |||
| < 45 years | 11 | 5.79 (2.08 to 16.14) | 99.64, 0.00 | |
| 45 to 65 years | 11 | 5.04 (2.02 to 12.57) | 99.47, 0.00 | |
| > 65 years | 8 | 6.66 (2.07 to 21.38) | 98.00, 0.00 | |
| Scanning year | 0.88 | |||
| Before 2007 | 8 | 3.83 (1.05 to 13.91) | 98.86, 0.00 | |
| After 2007 | 5 | 3.39 (1.60 to 7.72) | 99.54, 0.00 | |
| Data type | 0.00,0.00 | |||
| Actual | 6 | 1.17 (0.89 to 1.55) | 94.18, 0.00 | |
| Actual (Except Burton et al. 2018) | 5 | 1.32 (1.11 to 1.56) | 80.79, 0.00 | |
| Estimate | 19 | 10.00 (5.87 to 17.05) | 99.14, 0.00 | |
| Country | 0.19 | |||
| NonAmerica | 14 | 4.65 (2.15 to 10.05) | 99.73, 0.00 | |
| America | 12 | 9.56 (4.48 to 20.40) | 99.03, 0.00 | |
| Continent | 0.58 | |||
| Asia | 5 | 7.81 (1.57 to 38.89) | 99.91, 0.00 | |
| Europe | 8 | 4.37 (2.00 to 9.56) | 98.74, 0.00 | |
| America | 14 | 7.72 (3.42 to 17.42) | 99.19, 0.00 | |
| Cancer type | 0.01 | |||
| Nonleukemia | 14 | 5.57 (2.36 to 13.16) | 99.67, 0.00 | |
| Leukemia | 5 | 1.66 (1.29 to 2.15) | 19.62, 0.21 | |
| CT scan sites | 0.00 | |||
| < 3 | 15 | 3.38 (1.80 to 6.33) | 99.23, 0.00 | |
| ≥ 3 | 10 | 14.08 (6.60 to 30.05) | 99.53, 0.00 | |
| Quality scores | 0.25 | |||
| < 5 | 14 | 8.04 (4.38 to 14.75) | 99.23, 0.00 | |
| ≥ 5 | 11 | 4.12 (1.57 to 10.86) | 99.69, 0.00 | |
Abbreviations: OR Odds ratio, CI Confidence interval
There was no evidence of publication bias (egger, P = 0.58). Cancer risks of exposed men and women were not significantly different (P = 0.71, men, OR, 4.89 [95% CI, 2.40 to 9.96]; Ι2 = 99.43%; women, OR, 5.88 [95% CI, 2.96 to 11.68], Ι2 = 99.52%; Table 3). There were no significantly different cancer risks (P = 0.93) for adults in terms of age (Table 3, less than 45 years, OR, 5.79 [95% CI, 2.08 to 16.14], Ι2 = 99.64%; 45 to 65 years, OR, 5.04 [95% CI, 2.02 to 12.57], Ι2 = 99.47%; older than 65 years, OR, 6.66 [95% CI, 2.07 to 21.38], Ι2 = 98.00%).
The LAR estimation included the 2006 National Academy of Sciences BEIR VII. We extracted data in terms of scanning year (before and after 2007, 26] and countries (US or not) to explore the impact of scanning year and countries on cancer risks. Scanning year, countries, and continents of studies did not alter cancer risks (Ι2 = 99.61%, P = 0.88; Ι2 = 99.61%, P = 0.19; Ι2 = 99.61%, P = 0.58, Table 3). There was no significant difference in study quality scores (Ι2 = 99.61%, P = 0.25; Table 3).
Dose-response analysis showed that the cancer risks significantly increased linearly with radiation dose of CT scans (Coefficient = 0.03, P = 0.00) (Fig. S1). In details, the cancer risks were lowest for radiation dose of less than 15 mSv (OR, 2.51 [95% CI, 1.55 to 4.06], Ι2 = 94.46%), highest for dose greater than 55 mSv (OR, 33.31 [95% CI, 21.33 to 52.02], Ι2 = 97.09%), intermediate for dose of 15 to 55 mSv (OR, 5.48 [95% CI, 2.74 to 10.97]), with significant differences between triple groups (P < 0.001, dose grouping according to the overall radiation dose distribution included in the studies) (Fig. S2). Moreover, the relationship between cancer risk and radiation dose was more significant in the < 45 years group (P = 0.00), while there was no significant difference in 45–65 years groups (P = 0.08, Fig. S3).
For cancer types, a significantly higher risk of non-leukemic cancer was noted (P = 0.01; Table 3), in comparison with leukemic cancer. Cancer risks were significantly increased when more than 3 sites were scanned (P = 0.00; Table 3).
Heterogeneity and sensitivity analyses
Sensitivity analyses did not alter the results, by excluding 1 study at a time from each meta-analysis (Table S4). Heterogeneity was present in all analyses (> 99% for all pooled estimates). Meta-regression (Table S5) identified data type, CT sites, and radiation dose contributing to heterogeneity for cancer risk, with no factors substantially reducing heterogeneity between studies.
Discussion
Main findings and the significance of using LBR for LAR studies
The meta-analysis presents novel and complete evidence for the common and important issue concerned by clinicians, patients and the public, i.e., exposed CT scans and pertinent cancer risks for adults in their remaining lives. Based on 2,049,943 actual and 109,600,000 LAR/LBR participants from 9 countries, cancer risks for adults undergoing CT scans were successfully identified. Importantly, studies using LAR have been neglected in previous synthesized reports; partly due to the paucity of an appropriate control/unexposed population in quantify OR for these studies.
Indeed, selecting a control group (LBR) for LAR studies was challenging for such meta-analysis with global included studies. Participants unexposed to CT scans receive cosmic radiation (background radiation) in the earth, with various radiation dose range in different regions [31]. When background radiation was greater than 20 mSv per year, the area is designated as high natural background radiation (HNBR) region [58]. An ideal LBR should estimate cancer incidence based on various background radiation doses in different regions. Since HNBR areas were not covered in LAR studies for our meta-analysis, it is appropriate to use the global mean of 2.4 mSv per year as control group to estimate cancer risks. Our data showed that the incidence of cancer in the LBR group (42.7/100000 for men and 65.7/100000 for women) was lower than that in the CT exposed group (68.8/100000 for men and 91.9/100000 for women) and the updated annual global incidence of cancer [59]..
Sample size strengths and profound analysis for cancer risks from CT scans
So far, no studies have quantified the cancer risk of low-dose radiation. One of the most important reasons is insufficient sample size. According to the evaluation of a total of 1000 mSv for a sample of 1000 people, the sample size needed to evaluate 100 mSv would be 100,000, 10 mSv would be 10 million [60]. In this meta-analysis, the sample size was sufficient with strengths, including 24,000,538 participants for CT radiation exposure < 15 mSv, 1,056,402,315 for 15 to 55 mSv, and 76,600,000 for dose > 55 mSv. Cancer risks increased with cumulative radiation dose from CT scans, consistent with previous evidence [61]. Meanwhile, the results showed that cancer risks increased slowly during radiation dose below 55 mSv, and rapidly for those above 55 mSv.
Ample evidence from Japanese atomic bomb survivors indicates that children are more susceptible than adults to the deleterious effects of ionizing radiation [62]. Children have longer life expectancy since exposure to ionizing radiation, providing more time for a cancer to manifest. This point supports our results with higher cancer risks for LAR than actual studies, since life expectancy used for LAR was 100 years [63]. Notably, there was no significant difference of age on cancer incidence when we divided adults into young, middle-aged and older (age groups according to the 2010 WHO Recommendations on physical activity for health) [64]. Similarly, age did not significantly alter cancer risks for children and adolescents exposed for CT scans in Huang’s study [25] (0 to 5 years (relative risk [RR], 1.35), 6 to 15 years (RR, 1.14), and > 15 years (RR, 1.24) [25]. Mavragani et al. found that DNA damage from low radiation doses was not severe enough to cause cell death, but could trigger a red flag [65, 66]. One of those red flags is the body’s immune system. Increasing evidence suggests that radiation exposure of low dose may not only have immunosuppressive effects, but likely to be associated with a radiation-induced enhancement in the immune system [67–69]. However, immune systems of older adults are not as well functioning as those of younger people. This factor may underlie the results that the risk of cancer from CT radiation does not continue to decrease with age. This may also explain why cancer risks in the 45–65 years group are less sensitive to radiation exposure than those in the < 45 years group. Despite the age differences were insignificant, there was a significant increase in cancer risks for exposed adults compared with the background radiation group.
In 2007, the International Commission on Radiological Protection (ICPR) published a guide to the basic principles of appropriate radiation Protection based on radiation exposure. Since then, almost all international standards and national regulations dealing with radiation protection have been based on the recommendations of the Committee. Consequently, 2007 should be a milestone for CT scan in terms of dose reduction. Even in this study, no significant difference was found between radiation and cancer risk compared with studies published before or after 2007. What is clear is that researchers will adjust their protective strategies to a higher level in the plethora of exposure situations, as ICRP itself states.
Limitations
Firstly, There are various confounders for cancer incidence [70], including genetics [71], hormone levels, [72, 73] tobacco, alcohol, overweight, physical activity [74, 75], socio-economic status, [76] and infectious agents [77, 78]. These confounders were not included in this meta-analysis, due to the unavailability of information from original studies. Secondly, the numbers of participants in this study are mostly derived from estimated group and tend to be theoretical cancer risks from CT scan. Nevertheless, evidence from these LAR studies should not be ignored for meta-analysis (have been ignored in previous systematic review). Thirdly, over 80% participants in this study were women, with the risk of cancer in women undergoing CT scans during pregnancy remains unclear. Fourthly, the actual and estimated participants were combined. The difference in age at the end of follow-up between the two groups may amplify the actual cancer incidence. Nevertheless, current follow-up time period from actual studies was relatively limited, with incomplete reflected cancer risks from CT scans. Fifth, it is apparent that CT scans were correlated with increased risk of solid cancers, i.e., cancer risk increased with radiation dose and sites scanned. However, the relative increased quantification of scans may be biased due to different weight factors of various organs/tissues of the body. Finally, heart disease was the most common reason for CT scans in included studies, with variation in terms of the severity of patients, the treatment plan, and the duration of follow-up, thus contributing to the inconsistency of included studies.
Conclusion
Based on 111.6 million adult participants from 3 continents (Asia, Europe and America), this meta-analysis identifies an inordinately increase in cancer risks from CT scans for adults (Additional Video). Moreover, the cancer risks were positively correlated with radiation dose and CT sites. The meta-analysis highlights the awareness of potential cancer risks of CT scans as well as more reasonable methodology to quantify cancer risks in terms of life expectancy as 100 years for LAR.
Supplementary Information
Additional file 1 Table S1. Search strategy and results
Additional file 2 Table S2. Contacting authors for additional information
Additional file 3 Table S3A. Newcastle-Ottawa Quality Assessment Scale – Case–control studies. (.docx). Table S3B. Newcastle-Ottawa Quality Assessment Scale – Cohort studies
Additional file 4 Table S4. Ionizing radiation and cancer risks sensitivity analysis
Additional file 5 Table S5. Meta regression analysis
Additional file 6 Fig. S1. Bubble plot depicting the relationship between “dose” as radiation to CT scans and “response” as cancer risks
Additional file 7 Fig. S2. Forest plot of cancer risk at different doses from CT scans
Additional file 8 Fig. S3. Forest plot of cancer risks at radiation doses from CT radiation exposure in age groups (A: <45 years; B: 45 to 65 years)
Additional file 9 Video. The association of cumulative ionizing radiation from CT scans with pertinent cancer risks in adults. (.mp4) (The source of the images included in the video are original.)
Acknowledgements
Not application.
Availability of data and material
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Abbreviations
- LAR
Lifetime attribution risk
- OR
Odds ratio
- CI
Confidence interval
- BEIR
Biological Effects of Ionizing Radiation
- Lbr
Lifetime baseline risk
- RR
Relative risk
- HBV
Hepatitis B virus
- HPV
Human papilloma virus
- HCV
Hepatitis C virus
- LNT
Linear non-threshold
- HDI
Human development index
Authors’ contributions
HQW had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: CFC and HQW. Acquisition of data: CFC, HS, SQZ, and TFL. Analysis and interpretation of data: CFC, YW, and HQW. Drafting of the manuscript: CFC, KLM, and JZ. Critical revision of the manuscript for important intellectual content: HQW. Administrative, technical, or material support: HQW. All authors have read and approved the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (Grant number: 81572182 to HQW) and Shaanxi provincial key research and development program (Grant number: 2021SF-353 to JZ). The funding bodies played no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Mettler FA, Jr, Huda W, Yoshizumi TT, Mahesh M. Effective doses in radiology and diagnostic nuclear medicine: a catalog. Radiology. 2008;248(1):254–263. doi: 10.1148/radiol.2481071451. [DOI] [PubMed] [Google Scholar]
- 2.Smith-Bindman R, Lipson J, Marcus R, Kim KP, Mahesh M, Gould R, Berrington de Gonzalez A, Miglioretti DL. Radiation dose associated with common computed tomography examinations and the associated lifetime attributable risk of cancer. Arch Intern Med. 2009;169(22):2078–2086. doi: 10.1001/archinternmed.2009.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hricak H, Brenner DJ, Adelstein SJ, Frush DP, Hall EJ, Howell RW, McCollough CH, Mettler FA, Pearce MS, Suleiman OH, et al. Managing radiation use in medical imaging: a multifaceted challenge. Radiology. 2011;258(3):889–905. doi: 10.1148/radiol.10101157. [DOI] [PubMed] [Google Scholar]
- 4.Smith-Bindman R, Miglioretti DL, Johnson E, Lee C, Feigelson HS, Flynn M, Greenlee RT, Kruger RL, Hornbrook MC, Roblin D, et al. Use of diagnostic imaging studies and associated radiation exposure for patients enrolled in large integrated health care systems, 1996-2010. JAMA. 2012;307(22):2400–2409. doi: 10.1001/jama.2012.5960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pola A, Corbella D, Righini A, Torresin A, Colombo PE, Vismara L, Trombetta L, Maddalo M, Introini MV, Tinelli D, et al. Computed tomography use in a large Italian region: trend analysis 2004-2014 of emergency and outpatient CT examinations in children and adults. Eur Radiol. 2018;28(6):2308–2318. doi: 10.1007/s00330-017-5225-x. [DOI] [PubMed] [Google Scholar]
- 6.Schauer DA, Linton OW. NCRP Report No. 160, Ionizing Radiation Exposure of the Population of the United States, medical exposure--are we doing less with more, and is there a role for health physicists? Health Phys. 2009;97(1):1–5. doi: 10.1097/01.HP.0000356672.44380.b7. [DOI] [PubMed] [Google Scholar]
- 7.Rehani MM, Berry M. Radiation doses in computed tomography. The increasing doses of radiation need to be controlled. BMJ. 2000;320(7235):593–594. doi: 10.1136/bmj.320.7235.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shao YH, Tsai K, Kim S, Wu YJ, Demissie K. Exposure to Tomographic Scans and Cancer Risks. JNCI Cancer Spectr. 2020;4(1):pkz072. doi: 10.1093/jncics/pkz072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rampinelli C, De Marco P, Origgi D, Maisonneuve P, Casiraghi M, Veronesi G, Spaggiari L, Bellomi M. Exposure to low dose computed tomography for lung cancer screening and risk of cancer: secondary analysis of trial data and risk-benefit analysis. BMJ. 2017;356:j347. doi: 10.1136/bmj.j347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nordenskjold AC, Bujila R, Aspelin P, Flodmark O, Kaijser M. Risk of Meningioma after CT of the Head. Radiology. 2017;285(2):568–575. doi: 10.1148/radiol.2017161433. [DOI] [PubMed] [Google Scholar]
- 11.Davis F, Il'yasova D, Rankin K, McCarthy B, Bigner DD. Medical diagnostic radiation exposures and risk of gliomas. Radiat Res. 2011;175(6):790–796. doi: 10.1667/RR2186.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cohen S, Liu A, Gurvitz M, Guo L, Therrien J, Laprise C, Kaufman JS, Abrahamowicz M, Marelli AJ. Exposure to Low-Dose Ionizing Radiation From Cardiac Procedures and Malignancy Risk in Adults With Congenital Heart Disease. Circulation. 2018;137(13):1334–1345. doi: 10.1161/CIRCULATIONAHA.117.029138. [DOI] [PubMed] [Google Scholar]
- 13.Hansen J, Jurik AG. Analysis of Current Practice of CT examinations. Acta Oncol. 2009;48(2):295–301. doi: 10.1080/02841860802266714. [DOI] [PubMed] [Google Scholar]
- 14.Berrington de Gonzalez A, Mahesh M, Kim KP, Bhargavan M, Lewis R, Mettler F, Land C. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169(22):2071–2077. doi: 10.1001/archinternmed.2009.440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mettler FA., Jr Medical Radiation Exposure in the United States: 2006-2016 Trends. Health Phys. 2019;116(2):126–128. doi: 10.1097/HP.0000000000000996. [DOI] [PubMed] [Google Scholar]
- 16.Cucinotta FA, Schimmerling W, Wilson JW, Peterson LE, Saganti PB, Dicello JF. Uncertainties in estimates of the risks of late effects from space radiation. Adv Space Res. 2004;34(6):1383–1389. doi: 10.1016/j.asr.2003.10.053. [DOI] [PubMed] [Google Scholar]
- 17.Hendee WR, O'Connor MK. Radiation risks of medical imaging: separating fact from fantasy. Radiology. 2012;264(2):312–321. doi: 10.1148/radiol.12112678. [DOI] [PubMed] [Google Scholar]
- 18.Luan FJ, Wan Y, Mak KC, Ma CJ, Wang HQ. Cancer and mortality risks of patients with scoliosis from radiation exposure: a systematic review and meta-analysis. Eur Spine J. 2020;29(12):3123–3134. doi: 10.1007/s00586-020-06573-7. [DOI] [PubMed] [Google Scholar]
- 19.Luan F-J, Zhang J, Mak K-C, Liu Z-H, Wang H-Q. Low Radiation X - rays: Benefiting People Globally by Reducing Cancer Risks. Int J Med Sci. 2021;18(1):73–80. doi: 10.7150/ijms.48050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu TF, Shan H, Wang HQ, Luan FJ. Ionizing Radiation Exposure and Cancer Risks: Matter or not Matter? Spine (Phila Pa 1976) 2021;46(4):E286. doi: 10.1097/BRS.0000000000003848. [DOI] [PubMed] [Google Scholar]
- 21.Luan FJ, Zhang J, Wang HQ. Epidemiological study of adolescent idiopathic scoliosis using low/non-radiation screening methodology. J Rehabil Med. 2018;50(8):765–766. doi: 10.2340/16501977-2355. [DOI] [PubMed] [Google Scholar]
- 22.Wang HQ, Zhang J, Ma CJ. Triple Issues Underlying Lung Injury for Adolescent Idiopathic Scoliosis Surgery. Lung. 2018;196(4):381–382. doi: 10.1007/s00408-018-0131-9. [DOI] [PubMed] [Google Scholar]
- 23.Zhang J, Ma CJ, Liu ZH, Wang HQ. Defining the Pros and Cons of AIS Surgery: Bringing Truth to the Neurosurgery Community and the Public. World Neurosurg. 2018;113:393–394. doi: 10.1016/j.wneu.2018.01.209. [DOI] [PubMed] [Google Scholar]
- 24.Zhang J, Ma CJ, Xin XF, Wang HQ. Critical issues underlying expenditures for adolescent idiopathic scoliosis surgery: questioning the surgical treatment motivation. J Pediatr. 2018;198:326–327. doi: 10.1016/j.jpeds.2018.03.035. [DOI] [PubMed] [Google Scholar]
- 25.Huang R, Liu X, He L, Zhou PK. Radiation Exposure Associated With Computed Tomography in Childhood and the Subsequent Risk of Cancer: A Meta-Analysis of Cohort Studies. Dose Response. 2020;18(2):1559325820923828. doi: 10.1177/1559325820923828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wrixon AD. New ICRP recommendations. J Radiol Prot. 2008;28(2):161–168. doi: 10.1088/0952-4746/28/2/R02. [DOI] [PubMed] [Google Scholar]
- 27.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 Guideline Statement. J Clin Epidemiol. 2016;75:40–46. doi: 10.1016/j.jclinepi.2016.01.021. [DOI] [PubMed] [Google Scholar]
- 28.Wells GA, Shea B, O’Connell D, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. Secondary The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. 2011.
- 29.Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med. 1998;17(24):2815–2834. doi: 10.1002/(SICI)1097-0258(19981230)17:24<2815::AID-SIM110>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 30.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mc Laughlin JP. Some characteristics and effects of natural radiation. Radiat Prot Dosim. 2015;167(1–3):2–7. doi: 10.1093/rpd/ncv206. [DOI] [PubMed] [Google Scholar]
- 32.Committee to Assess Health Risks from Exposure to Low Levels of Ionizing Radiation, Board on Radiation Effects, Research Division on Earth and Life Studies, National Research Council of the National Academies. Health risks from exposure to low levels of ionizing radiation: BEIR VII phase 2. Washington, DC: National Academy of Sciences; 2006.
- 33.Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Deeks JJ, Higgins JPT, Altman DG. Analysing Data and Undertaking Meta-Analyses. In: Higgins JPT, editor. Cochrane Handbook for Systematic Review of Interventions. Hoboken (NJ): Wiley-Blackwell; 2011. pp. 243–296. [Google Scholar]
- 35.Burton KR, Park AL, Fralick M, Ray JG. Risk of early-onset breast cancer among women exposed to thoracic computed tomography in pregnancy or early postpartum. J Thromb Haemost. 2018;16(5):876–885. doi: 10.1111/jth.13980. [DOI] [PubMed] [Google Scholar]
- 36.Hung MC, Hwang JJ. Cancer risk from medical radiation procedures for coronary artery disease: a nationwide population-based cohort study. Asian Pac J Cancer Prev. 2013;14(5):2783–2787. doi: 10.7314/APJCP.2013.14.5.2783. [DOI] [PubMed] [Google Scholar]
- 37.Kritsaneepaiboon S, Jutiyon A, Krisanachinda A. Cumulative radiation exposure and estimated lifetime cancer risk in multiple-injury adult patients undergoing repeated or multiple CTs. Eur J Trauma Emerg Surg. 2018;44(1):19–27. doi: 10.1007/s00068-016-0665-6. [DOI] [PubMed] [Google Scholar]
- 38.Griffey RT, Sodickson A. Cumulative radiation exposure and cancer risk estimates in emergency department patients undergoing repeat or multiple CT. AJR Am J Roentgenol. 2009;192(4):887–892. doi: 10.2214/AJR.08.1351. [DOI] [PubMed] [Google Scholar]
- 39.Einstein AJ, Sanz J, Dellegrottaglie S, Milite M, Sirol M, Henzlova M, Rajagopalan S. Radiation dose and cancer risk estimates in 16-slice computed tomography coronary angiography. J Nucl Cardiol. 2008;15(2):232–240. doi: 10.1016/j.nuclcard.2007.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Faletra FF, D'Angeli I, Klersy C, Averaimo M, Klimusina J, Pasotti E, Pedrazzini GB, Curti M, Carraro C, Diliberto R, et al. Estimates of lifetime attributable risk of cancer after a single radiation exposure from 64-slice computed tomographic coronary angiography. Heart. 2010;96(12):927–932. doi: 10.1136/hrt.2009.186973. [DOI] [PubMed] [Google Scholar]
- 41.Niemann T, Zbinden I, Roser HW, Bremerich J, Remy-Jardin M, Bongartz G. Computed tomography for pulmonary embolism: assessment of a 1-year cohort and estimated cancer risk associated with diagnostic irradiation. Acta Radiol. 2013;54(7):778–784. doi: 10.1177/0284185113485069. [DOI] [PubMed] [Google Scholar]
- 42.Huang B, Law MW, Khong PL. Whole-body PET/CT scanning: estimation of radiation dose and cancer risk. Radiology. 2009;251(1):166–174. doi: 10.1148/radiol.2511081300. [DOI] [PubMed] [Google Scholar]
- 43.Perisinakis K, Seimenis I, Tzedakis A, Papadakis AE, Damilakis J. Individualized assessment of radiation dose in patients undergoing coronary computed tomographic angiography with 256-slice scanning. Circulation. 2010;122(23):2394–2402. doi: 10.1161/CIRCULATIONAHA.109.935346. [DOI] [PubMed] [Google Scholar]
- 44.Sodickson A, Baeyens PF, Andriole KP, Prevedello LM, Nawfel RD, Hanson R, Khorasani R. Recurrent CT, cumulative radiation exposure, and associated radiation-induced cancer risks from CT of adults. Radiology. 2009;251(1):175–184. doi: 10.1148/radiol.2511081296. [DOI] [PubMed] [Google Scholar]
- 45.Huang B, Li J, Law MW, Zhang J, Shen Y, Khong PL. Radiation dose and cancer risk in retrospectively and prospectively ECG-gated coronary angiography using 64-slice multidetector CT. Br J Radiol. 2010;83(986):152–158. doi: 10.1259/bjr/29879495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Huda W, Schoepf UJ, Abro JA, Mah E, Costello P. Radiation-related cancer risks in a clinical patient population undergoing cardiac CT. AJR Am J Roentgenol. 2011;196(2):W159–W165. doi: 10.2214/AJR.10.4981. [DOI] [PubMed] [Google Scholar]
- 47.Perisinakis K, Seimenis I, Tzedakis A, Papadakis AE, Damilakis J. Triple-rule-out computed tomography angiography with 256-slice computed tomography scanners: patient-specific assessment of radiation burden and associated cancer risk. Investig Radiol. 2012;47(2):109–115. doi: 10.1097/RLI.0b013e31822d0cf3. [DOI] [PubMed] [Google Scholar]
- 48.Einstein AJ, Henzlova MJ, Rajagopalan S. Estimating risk of cancer associated with radiation exposure from 64-slice computed tomography coronary angiography. JAMA. 2007;298(3):317–323. doi: 10.1001/jama.298.3.317. [DOI] [PubMed] [Google Scholar]
- 49.Kim KP, Einstein AJ, Berrington de Gonzalez A. Coronary artery calcification screening: estimated radiation dose and cancer risk. Arch Intern Med. 2009;169(13):1188–1194. doi: 10.1001/archinternmed.2009.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Majer M, Knezevic Z, Popic J, Hrsak H, Miljanic S. Organ doses and associated cancer risks for computed tomography examinations of the thoracic region. Nuclear Technol Rad Protect. 2018;33(1):100–105. doi: 10.2298/NTRP1801100M. [DOI] [Google Scholar]
- 51.Salibi PN, Agarwal V, Panczykowski DM, Puccio AM, Sheetz MA, Okonkwo DO. Lifetime attributable risk of cancer from CT among patients surviving severe traumatic brain injury. AJR Am J Roentgenol. 2014;202(2):397–400. doi: 10.2214/AJR.12.10294. [DOI] [PubMed] [Google Scholar]
- 52.Shah KH, Slovis BH, Runde D, Godbout B, Newman DH, Lee J. Radiation exposure among patients with the highest CT scan utilization in the emergency department. Emerg Radiol. 2013;20(6):485–491. doi: 10.1007/s10140-013-1142-8. [DOI] [PubMed] [Google Scholar]
- 53.Wylie JD, Jenkins PA, Beckmann JT, Peters CL, Aoki SK, Maak TG. Computed Tomography Scans in Patients With Young Adult Hip Pain Carry a Lifetime Risk of Malignancy. Arthroscopy: The Journal of Arthroscopic & Related Surgery. 2018;34(1):155–163.e153. doi: 10.1016/j.arthro.2017.08.235. [DOI] [PubMed] [Google Scholar]
- 54.Olsen M, Garne E, Svaerke C, Sondergaard L, Nissen H, Andersen HO, Hjortdal VE, Johnsen SP, Videbaek J. Cancer risk among patients with congenital heart defects: a nationwide follow-up study. Cardiol Young. 2014;24(1):40–46. doi: 10.1017/S1047951112002144. [DOI] [PubMed] [Google Scholar]
- 55.Meulepas JM, Hauptmann M, Lubin JH, Shuryak I, Brenner DJ. Is there Unmeasured Indication Bias in Radiation-Related Cancer Risk Estimates from Studies of Computed Tomography? Radiat Res. 2018;189(2):128–135. doi: 10.1667/RR14807.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wu TH, Lin WC, Chen WK, Chang YC, Hwang JJ. Predicting cancer risks from dental computed tomography. J Dent Res. 2015;94(1):27–35. doi: 10.1177/0022034514554226. [DOI] [PubMed] [Google Scholar]
- 57.Bosch de Basea M, Morina D, Figuerola J, Barber I, Muchart J, Lee C, Cardis E. Subtle excess in lifetime cancer risk related to CT scanning in Spanish young people. Environ Int. 2018;120:1–10. doi: 10.1016/j.envint.2018.07.020. [DOI] [PubMed] [Google Scholar]
- 58.Hendry JH, Simon SL, Wojcik A, Sohrabi M, Burkart W, Cardis E, Laurier D, Tirmarche M, Hayata I. Human exposure to high natural background radiation: what can it teach us about radiation risks? J Radiol Prot. 2009;29(2A):A29–A42. doi: 10.1088/0952-4746/29/2A/S03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021. [DOI] [PubMed]
- 60.Brenner DJ, Doll R, Goodhead DT, Hall EJ, Land CE, Little JB, Lubin JH, Preston DL, Preston RJ, Puskin JS, et al. Cancer risks attributable to low doses of ionizing radiation: assessing what we really know. Proc Natl Acad Sci U S A. 2003;100(24):13761–13766. doi: 10.1073/pnas.2235592100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hauptmann M, Daniels RD, Cardis E, Cullings HM, Kendall G, Laurier D, Linet MS, Little MP, Lubin JH, Preston DL, et al. Epidemiological Studies of Low-Dose Ionizing Radiation and Cancer: Summary Bias Assessment and Meta-Analysis. J Natl Cancer Inst Monogr. 2020;2020(56):188–200. doi: 10.1093/jncimonographs/lgaa010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pierce DA, Shimizu Y, Preston DL, Vaeth M, Mabuchi K. Studies of the mortality of atomic bomb survivors. Report 12, Part I. Cancer: 1950-1990. Radiat Res. 1996;146(1):1–27. doi: 10.2307/3579391. [DOI] [PubMed] [Google Scholar]
- 63.Siegel JA, Greenspan BS, Maurer AH, Taylor AT, Phillips WT, Van Nostrand D, Sacks B, Silberstein EB. The BEIR VII Estimates of Low-Dose Radiation Health Risks Are Based on Faulty Assumptions and Data Analyses: A Call for Reassessment. J Nucl Med. 2018;59(7):1017–1019. doi: 10.2967/jnumed.117.206219. [DOI] [PubMed] [Google Scholar]
- 64.In: Global Recommendations on Physical Activity for Health. edn. Geneva; 2010. [PubMed]
- 65.Mavragani IV, Laskaratou DA, Frey B, Candeias SM, Gaipl US, Lumniczky K, Georgakilas AG. Key mechanisms involved in ionizing radiation-induced systemic effects. A current review. Toxicol Res (Camb) 2016;5(1):12–33. doi: 10.1039/c5tx00222b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mavragani IV, Nikitaki Z, Souli MP, Aziz A, Nowsheen S, Aziz K, et al. Complex DNA Damage: A Route to Radiation-Induced Genomic Instability and Carcinogenesis. Cancers (Basel). 2017;9(7). [DOI] [PMC free article] [PubMed]
- 67.Hellweg CE. The Nuclear Factor kappaB pathway: A link to the immune system in the radiation response. Cancer Lett. 2015;368(2):275–289. doi: 10.1016/j.canlet.2015.02.019. [DOI] [PubMed] [Google Scholar]
- 68.Cui J, Yang G, Pan Z, Zhao Y, Liang X, Li W, et al. Hormetic Response to Low-Dose Radiation: Focus on the Immune System and Its Clinical Implications. Int J Mol Sci. 2017;18(2). [DOI] [PMC free article] [PubMed]
- 69.Alexakhin RM. Radiation, United Nations Scientific Committee on the Effects of Atomic 2008. Radiats Biol Radioecol. 2015;55(5):548–549. [PubMed] [Google Scholar]
- 70.Arem H, Loftfield E. Cancer Epidemiology: A Survey of Modifiable Risk Factors for Prevention and Survivorship. Am J Lifestyle Med. 2018;12(3):200–210. doi: 10.1177/1559827617700600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Metcalfe KA, Poll A, Royer R, Llacuachaqui M, Tulman A, Sun P, Narod SA. Screening for founder mutations in BRCA1 and BRCA2 in unselected Jewish women. J Clin Oncol. 2010;28(3):387–391. doi: 10.1200/JCO.2009.25.0712. [DOI] [PubMed] [Google Scholar]
- 72.Hunter DJ, Colditz GA, Hankinson SE, Malspeis S, Spiegelman D, Chen W, Stampfer MJ, Willett WC. Oral contraceptive use and breast cancer: a prospective study of young women. Cancer Epidemiol Biomark Prev. 2010;19(10):2496–2502. doi: 10.1158/1055-9965.EPI-10-0747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.La Vecchia C, Tavani A. Female hormones and benign liver tumours. Dig Liver Dis. 2006;38(8):535–536. doi: 10.1016/j.dld.2006.04.012. [DOI] [PubMed] [Google Scholar]
- 74.Katzke VA, Kaaks R, Kuhn T. Lifestyle and cancer risk. Cancer J. 2015;21(2):104–110. doi: 10.1097/PPO.0000000000000101. [DOI] [PubMed] [Google Scholar]
- 75.Song M, Giovannucci E. Preventable Incidence and Mortality of Carcinoma Associated With Lifestyle Factors Among White Adults in the United States. JAMA Oncol. 2016;2(9):1154–1161. doi: 10.1001/jamaoncol.2016.0843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pearce MS, Salotti JA, McHugh K, Kim KP, Craft AW, Lubin J, Ron E, Parker L. Socio-economic variation in CT scanning in Northern England, 1990-2002. BMC Health Serv Res. 2012;12:24. doi: 10.1186/1472-6963-12-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.White MK, Pagano JS, Khalili K. Viruses and human cancers: a long road of discovery of molecular paradigms. Clin Microbiol Rev. 2014;27(3):463–481. doi: 10.1128/CMR.00124-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Munoz N, Castellsague X, de Gonzalez AB, Gissmann L. Chapter 1: HPV in the etiology of human cancer. Vaccine. 2006;24(Suppl 3):S3/1–S310. doi: 10.1016/j.vaccine.2006.05.115. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1 Table S1. Search strategy and results
Additional file 2 Table S2. Contacting authors for additional information
Additional file 3 Table S3A. Newcastle-Ottawa Quality Assessment Scale – Case–control studies. (.docx). Table S3B. Newcastle-Ottawa Quality Assessment Scale – Cohort studies
Additional file 4 Table S4. Ionizing radiation and cancer risks sensitivity analysis
Additional file 5 Table S5. Meta regression analysis
Additional file 6 Fig. S1. Bubble plot depicting the relationship between “dose” as radiation to CT scans and “response” as cancer risks
Additional file 7 Fig. S2. Forest plot of cancer risk at different doses from CT scans
Additional file 8 Fig. S3. Forest plot of cancer risks at radiation doses from CT radiation exposure in age groups (A: <45 years; B: 45 to 65 years)
Additional file 9 Video. The association of cumulative ionizing radiation from CT scans with pertinent cancer risks in adults. (.mp4) (The source of the images included in the video are original.)
Data Availability Statement
All data generated or analyzed during this study are included in this published article and its supplementary information files.