Abstract
Motivation
Natural language processing (NLP) tasks aim to convert unstructured text data (e.g. articles or dialogues) to structured information. In recent years, we have witnessed fundamental advances of NLP technique, which has been widely used in many applications such as financial text mining, news recommendation and machine translation. However, its application in the biomedical space remains challenging due to a lack of labeled data, ambiguities and inconsistencies of biological terminology. In biomedical marker discovery studies, tools that rely on NLP models to automatically and accurately extract relations of biomedical entities are valuable as they can provide a more thorough survey of all available literature, hence providing a less biased result compared to manual curation. In addition, the fast speed of machine reader helps quickly orient research and development.
Results
To address the aforementioned needs, we developed automatic training data labeling, rule-based biological terminology cleaning and a more accurate NLP model for binary associative and multi-relation prediction into the MarkerGenie program. We demonstrated the effectiveness of the proposed methods in identifying relations between biomedical entities on various benchmark datasets and case studies.
Availability and implementation
MarkerGenie is available at https://www.genegeniedx.com/markergenie/. Data for model training and evaluation, term lists of biomedical entities, details of the case studies and all trained models are provided at https://drive.google.com/drive/folders/14RypiIfIr3W_K-mNIAx9BNtObHSZoAyn?usp=sharing.
Supplementary information
Supplementary data are available at Bioinformatics Advances online.
1 Introduction
Relations of biomedical entities (bioentities) are critical to biomedical studies and are hidden in a large number of biomedical articles. In this work, the main goal is to rapidly and accurately identify associative relations between a pair of biomedical entities present in the literature. We consider two entities to be associative in a context when they are described to be correlated directly, causal or non-causal. Most biomedical entity relations such as a biomarker and a disease are associative. Determining such a relation is typically an important first step to guide additional wet-lab or clinical studies to verify the diagnostic, predictive, prognostic, predisposing and treatment relation. Without being exhaustive, a bioentity may refer to a disease, a gene, a metabolite or a microbial taxa.
Many biomedical text-mining methods have been used to identify associations of diseases and biomarkers. These methods can digest research articles more efficiently and comprehensively compared to human researchers and can help prioritize the targets in diagnoses and drug target discovery. In the following, we provide a brief overview of the current status of biomarker relation database curation and text-mining methods.
Bioentity relation databases are typically manually curated and serve as the ground truth for the research community. For example, Ma et al. (2017) manually extracted 292 microbe–microbe, 39 disease–disease and 483 microbe–disease associations from microbiome-related articles. Janssens et al. (2018) established a disease–microbiome database by querying PUBMED database using criteria ([(’microbiota’ OR ’microbiome) and (’health’ OR ’disease’)] and [microbiome alterations]). Then, the disease, microbiome terms and their relations were extracted manually. Noronha et al. (2019) created a database that relates human metabolism with genetics, microbial metabolism, nutrition and diseases.
To automate and expand the scope of entity relation extraction, a few methods, including PolySearch2 (Liu et al., 2015), BEST (Lee et al., 2016), GenCLiP 3 (Wang et al., 2020), STRING (Szklarczyk et al., 2021), IBDDB (Khan et al., 2021) and DrugShot (Kropiwnicki et al., 2022), have been introduced. With a common assumption that the strength of entity association is positively correlated with their co-occurring frequency in the same context, these methods first identified frequently co-occurring entities of interest, then refined the entity relation with different scoring and filtering criteria. However, there are some limitations. A pair of entities with low co-occurring frequency can be reliable but would be missed. For example, recently discovered relations would have few mentions in the literature. Meanwhile, high co-occurrence counts can include many false positives that require ad hoc and complex rules to eliminate.
These limitations have been addressed by supervised machine learning (ML)-based methods (Hsieh et al., 2017; Hua and Quan, 2016; Xu et al., 2015). To curate CIViC database (Lever et al., 2019), published literature was parsed and sentences containing a pair of target entities were identified via exact string matching. A support vector machine-based classifier was then trained using 800 labeled sentences. Ahmed et al. (2019) proposed a novel neural network architecture for identifying protein–protein interactions (PPIs) from biomedical text using a tree long-short-term memory (LSTM) network with structured attention to traverse the dependency tree of a sentence through a child sum tree LSTM. Meanwhile, structural information was learned through a parent selection mechanism by modeling non-projective dependency trees. The main challenge for the application of ML methods is the lack of labeled training data. Although distant supervision (Mintz et al., 2009) can be used to acquire additional training data with positive labels, negative training data cannot be generated and this method requires a high-quality knowledge database that is typically hard to curate.
In this article, we treat finding relevant biomedical entities as a sentence-level binary/multiple relation classification task. During entity extraction, we introduced rule-based strategies to reduce false positive extractions as the existing bioentity terminologies still contain a large number of ambiguities and sometimes, errors. To address the lack of training data and the labor-intensive manual labeling process, we proposed an automated training data generation using co-occurrence frequency matrix and demonstrated its practical use. We then developed a new model, SBGT (SciBERT+Gumbel Tree-GRU), for relation classification that uses SciBERT (Beltagy et al., 2019) to encode the context features of words and Gumbel Tree-GRU (Hong et al., 2020) to encode the syntactic structures of sentences.
We provide MarkerGenie as an online text-mining tool. The current release includes the following entities: diseases, microbiomes, genes and metabolites. The corpus currently includes the free-text and tables of articles in PubMed and PubMed Central. The overview of MarkerGenie is given in Figure 1 that includes four main components: user query processing, article retrieving and sentence filtering, model-based classification and results reporting. The implementation details of MarkerGenie are provided in Supplementary Section S1.
Fig. 1.
MarkerGenie online workflow. MarkerGenie is a text-mining system for identifying biomarker relations with diseases. Given a query disease term, MarkerGenie first identifies relevant disease terms through fuzzy matching. Then, it retrieves articles according to the synonyms of the disease, and select the sentences that contain both the disease and biomarkers through entity extraction. Afterwards, the filtered sentences are classified by NLP models. Finally, the system returns the biomarkers related to the disease extracted from the literature in detail that including the source sentences, tables and articles. To improve speed, result caching was used
2 Materials and methods
2.1 SBGT model
In the proposed SBGT model, we used SciBERT (Beltagy et al., 2019) to extract the contextual features of words given the input sentence. SciBERT can improve the handling of unseen and rare words by using subword tokenizer in between words and characters. It had been experimentally shown to outperform BERT-Base and Bio-BERT in relation extraction of biomedical text (Beltagy et al., 2019). Then, we used the Gumbel Tree-GRU (Hong et al., 2020) to encode the syntactic structure. The encoded vectors were concatenated and fed into a fully connected layer for prediction. As shown in Figure 1, given a sentence, SciBERT extracts the contextual features of each word. Each word is encoded as a 1*768 vector. Then Gumbel Tree-GRU is used to organize those words into a vector to represent the sentence. Afterward, the vector as well as the contextual features of Entity1 and Entity2 are concatenated to indicate their relation. Finally, a fully connected layer is applied to predict the probability of the relation falling within each category.
2.2 Unsupervised training data generation for binary relation classification
To generate the training data, a co-occurrence frequency matrix of the bioentities from sentences was first constructed from free-text in PubMed and PubMed Central. We chose entity pairs with the most co-occurrence counts and used two thresholds, ‘minimum co-occurrences t1’ and ‘truncating quantity t2’, to generate the positive data. Particularly, sentences containing a pair of entities co-occurring times were considered. At most t2 of these sentences were retained to prevent the bias toward high frequency entity pairs. The default values of t1 = 10 and t2 = 50 were empirically set and used in all current experiments. To generate negative data, sentences containing entity pairs with the frequency of one in the matrix were included except the ones that contain a single disease term and biomarker term; because we found that the latter was more likely to be a positive case. Note that the negative sample means no direct association between two biomedical entities in a sentence. Same as co-occurrence-based methods, some rarer and possibly more relevant biomedical associations may be missed by ignoring low-occurring data. However, the samples generated here were used as the labeled data to train a model rather than used as the final result. The associative relation between a pair of bioentities is extracted by the trained model regardless of their co-occurrence frequency in the actual prediction stage. An example of the positive and negative data generation process is given in Figure 2. The complete training data was generated subject to a 6:4 ratio for positive and negative instances. The ratio is consistent with the fraction of positive and negative instances observed in the literature. The generated training data were further divided by an 8:2 ratio into training and validation sets for model parameter optimization on F1-score. The model performance was measured on independent datasets.
Fig. 2.
Example of unsupervised training data generation. The heatmap of the co-occurrence of 20 diseases and 20 microbes in the literature is shown in the left part of this figure. Gastroenteritis and Vibrio parahaemolyticus co-occur most frequently: more than 400 times, greater than a predefined threshold, so they are considered related and the corresponding sentences in the articles were selected as positive samples. On the contrary, the low co-occurrence couples, e.g. Liver Cirrhosis and Alkaline xylosoxidans, tend to be irrelevant and the corresponding sentences formed the negative samples
2.3 Entity extraction
Entity extraction is a pre-requisite step of relation extraction. The entities of interest were curated in term lists in advance. Currently, the following entity term lists have been curated—disease, microbiome, metabolite and gene as detailed in Supplementary Section S2. First, spaCy (Neumann et al., 2019) was used for sentence splitting and tokenization. Then, similar to CIViCmine, exact string matching was applied to the tokenized sentences to extract entities. To achieve this, we first constructed a trie on all synonyms and then located the most extended term in sentences by traversing the trie. This strategy has a run time complexity of O(n) for a length-n sentence.
To improve the accuracy of entity recognition, rule-based filtering was further applied. If a disease term had a prefix of a letter followed by a dot, like ‘s. pneumonia’, the term was disregarded; we also removed term with a length less than four characters unless it was determined to be an abbreviation, conforming the pattern of ‘synonym + (entity)’.
2.4 Relation extraction from tables
Different from the classification-based relation extraction used for text, we used rule-based methods on tables. A table was first extracted and stored as a tuple (caption, table – head, table – body: list of data rows). The bioentity relations generally appear in two different patterns in a table as illustrated by the disease–microbiome relation extraction example: when a disease term and a collective term of the microbiome (e.g. ‘microbiome’, ‘bacteria’) co-occurred in the caption of a table and the microbiome terms were present in the body of the table, all microbiome terms in the table body were considered to be related to the disease (Fig. 3A). When a disease term and a specific microbiome term co-occurred in a row or caption of the table, they were considered to be related (Fig. 3B).
Fig. 3.
Illustration of relation extraction from tables. (A) CRC and a collective terms of the microbiome (‘bacterium’) co-occur in the caption of the table, so all microbes in the table body are considered to be related to CRC. (B) CRC and a specific microbiome term (‘Fusobacterium nucleatum’) co-occur in a row of the table. They are considered as related
2.5 Granular relation classification between a disease and bioentities
When a disease and a bioentity were determined by binary classification to be associative, MarkerGenie can further predict them to be one of the five granular relation types—Predictive, Prognostic, Diagnostic, Predisposingor Treatment if there is a potential specific relation between them judged by CIViCmine’s search terms (Fig. 4). The training sentences of this classification task were first generated via distant supervision method using knowledge databases of CBD (Zhang et al., 2018), MarkerDB (Wishart et al., 2021) and Oncomx (Dingerdissen et al., 2020). Then we used a term list (e.g. ‘risk’ and ‘survival’) provided by CIViCmine to screen sentences that potentially contain one of the five specific relations. In addition, we expanded the term list by using pre-trained word vectors to include synonyms to increase the size of training data.
Fig. 4.
The workflow of MarkerGenie for classifying the granular relation types between a disease and bio-entities. When a disease and a bio-entity are determined by binary classification to be associative, MarkerGenie judges if there is a potential specific relation between them by using CIViCmine’s search terms. Then, the trained model is applied to predict the granular relation types (Predictive, Prognostic, Diagnostic, Predisposing or Treatment) of the filtered sentences
3. Results
In this section, we first demonstrate the improved accuracy of SBGT model by applying it on the curated benchmark datasets that were used by previous methods—the binary relation classification of PPI (Pyysalo et al., 2007) and the multi-relation classification of drug–drug interaction (DDI’13) (Herrero-Zazo et al., 2013). Next, we demonstrate the validity of automatic training data generation by applying MarkerGeine to disease–biomarker binary associative relation classification. This task does not require any prior knowledge or curated databases. However, when curated databases are available, MarkerGenie would generate training data via distant supervision strategy and produce multi-relation classification. This was demonstrated on disease–gene multi-relation extraction task as carried out in CIViCmine (Lever et al., 2019). Finally, we demonstrate how MarkerGenie can aid biomarker discovery with a few case studies.
3.1 Binary relation classification
The SBGT model was first validated on the PPI corpora (Pyysalo et al., 2007), which was used as a benchmark dataset by prior methods. The dataset information and hyper-parameters of SBGT are summarized in Table 1. To ensure the generalization of the learned model, we replaced the pair of proteins in each sentence with ‘PROTEIN1’ and ‘PROTEIN2’. In addition, all sentences were truncated or padded to a maximum length of 100. The performance of SBGT was compared with seven other state-of-the-art models—sdpCNN (Hua and Quan, 2016), sdpLSTM (Xu et al., 2015), Bert (Devlin et al., 2019), BioBERT (Lee et al., 2020), DRCNN (Zhang et al., 2019), Bi-LSTM (Hsieh et al., 2017) and BioKGLM (Fei et al., 2021). The evaluation scheme and parameters of the compared algorithms were all set per the original papers. The F1-scores of these methods are given in Table 2, where SBGT achieved 3.2% improvement over the runner up. Since some of the methods were evaluated with macro F1-score in the corresponding references, we also included this metric in Table 2, where SBGT showed consistent superiority to the compared models, including DCNN (Choi, 2018), Att-sdpLSTM (Yadav et al., 2019), tLSTM (Ahmed et al., 2019) and DRCNN.
Table 1.
PPI and DDI’13 dataset information and hyper-parameters of SBGT
| Dataset | PPI | DDI’13 |
|---|---|---|
| No. classes | 2 | 5 |
| No. samples | 9666 | 15 861 |
| Evaluation scheme | 10-fold cross-validation | 77% training, 23% test |
| SBGT miniBatch-size | 16 | 32 |
| SBGT learning rate | 1e−5 | 3e−5 |
| SBGT num of epoch | 50 | 10 |
| SBGT optimizer | AdamW | AdamW |
Note: The evaluation schemes were selected to be consistent with the methods under comparison.
Table 2.
Comparison of SBGT and other methods on PPI dataset in terms of F1 score and macro-F1 score
| Method | F1 score (%) | Method | Macro-F1 scores (%) |
|---|---|---|---|
| sdpCNN | 75.2 | DCNN | 74.7 |
| sdpLSTM | 77.3 | Att-sdpLSTM | 81.7 |
| Bert | 82.3 | tLSTM | 89.1 |
| BioBERT | 84.6 | DRCNN | 91.1 |
| DRCNN | 86.9 | — | — |
| Bi-LSTM | 87.2 | — | — |
| BioKGLM | 89.3 | — | — |
| SBGT | 92.4 | SBGT | 94.9 |
3.2 Multi-relation classification
We applied SBGT to the DDI’13 dataset (Herrero-Zazo et al., 2013) where the goal was to determine specific relations (defined as {NA, ADVICE, EFFECT, MECHANISM, INT}) given two drugs. Like binary classification, we replaced the pair of drugs in each sentence with ‘<ent1 >’ and ‘<ent2 >’ and all sentences were truncated or padded to a maximum length of 100. On this dataset, SBGT were trained with the hyper-parameters shown in Table 1. SBGT was compared with the seven other state-of-the-art models, including SCNN (Zhao et al., 2016), CNN-bioWE (Liu et al., 2016), MCCNN (Quan et al., 2016), Joint AB-LSTM (Sahu and Anand, 2018), RvNN (Lim et al., 2018), Position-aware LSTM (Zhou et al., 2018) and BERE (Hong et al., 2020) in terms of precision, recall and F1-score. As shown in Table 3, SBGT attained the best trade-off of precision and recall. In terms of F1 score, SBGT obtained a score of 77.1% that is ∼3% higher than that of the second best model.
Table 3.
Comparison of SBGT and other methods on DDI’13 dataset in terms of Precision, Recall and F1 score
| Method | P (%) | R (%) | F (%) |
|---|---|---|---|
| SCNN | 69.1 | 65.1 | 67.0 |
| CNN-bioWE | 75.7 | 64.7 | 69.8 |
| MCCNN | 75.9 | 65.2 | 70.2 |
| Joint AB-LSTM | 73.4 | 69.6 | 71.5 |
| RvNN | 74.4 | 69.3 | 71.7 |
| PM-BLSTM | 75.8 | 70.4 | 73.0 |
| BERE | 76.9 | 71.3 | 73.9 |
| SBGT | 80.3 | 74.2 | 77.1 |
3.3 Disease–biomarker associative binary classification with automatic training data generation
We selected three major biomarker types—microbiome, metabolite and gene—to study their associative relations with diseases from publicly available articles of PubMed and PubMed Central. The labeled training data for these tasks are scarce or even missing though some have been manually curated (Lever et al., 2019; Liu et al., 2015). We introduced an unsupervised method that can automatically generate the labeled training data in Section 2.
Admittedly, the automatic label generation can include many false positive instances—upon manual inspection, around 15–20% of the positive samples are incorrectly labeled. Yet, we can obtain a large amount of data within a few hours’ run time. We have obtained around 6000 disease–microbiome and 10 000 disease–metabolite or disease–gene training samples. The data of this size would be more suitable for deep learning strategies compared to the typical curated data size in the hundreds scale (Lever et al., 2019). Though trained using noisy data increased the model bias, the overall model performance improved along with the size of training samples on the test data. As the example of disease–microbiome shown in Figure 5, the F1 value generally increased as more data became available.
Fig. 5.

An illustration of the impact of automatically acquired training data on model performance. The SBGT model was trained on different sizes of disease–microbiome training dataset and F1 scores were obtained on the independent 477 test samples. Each experiment was repeated three times and the F1 score was the average
To evaluate the performance of MarkerGenie on the above tasks, we manually curated 477 disease–microbiome samples and 610 annotated disease–metabolite samples. For disease–gene prediction, 382 labeled disease–gene samples were directly obtained from Liu et al. (2015). MarkerGenie predicted disease–microbiome, disease–metabolite and disease–gene relations with precisions of 83.28%, 85.26% and 82.01%, respectively (Fig. 6A, the corresponding F1 scores and precision–recall curves are shown in Fig. 6B and C). Empirically, around 60–70% instances of disease–biomarker pairs co-occurring in the same sentence have a true positive relation, MarkerGenie therefore removed over 10–20% of the false positive instances. For these three tasks, MarkerGenie recalled 84.92%, 89.73% and 88.43% of the relations, respectively. We note that, the reported performance from Liu et al. (2015) on this disease–gene relation dataset obtained from the same study had both higher precision (∼5%) and recall (∼2%), yet its generalizability cannot be independently evaluated. Also note that, Liu et al. (2015) used the rule-based method that factors in the prior knowledge of validated disease–gene relations, which is generally unknown to the model.
Fig. 6.
Performance of MarkerGenie on disease–biomarker relation identification. (A) Precision and recall of disease–biomarkers’ associative binary classification. (B) F1 scores of disease–biomarkers’ associative binary classification. (C) Precision-recall curves, due to the high threshold at the beginning, there are few samples marked as positive examples, so the upper left part of the curve fluctuates greatly. (D–G) Precision-recall curves of MarkerGenie and CIViCmine on four specific relation extraction, i.e. predictive, prognostic, diagnostic and predisposing
3.4 Granular relation extraction via distant supervision
Following binary associative relation prediction, MarkerGenie can rely on disease–biomarker relation knowledge-bases to automatically generate training data via distant supervision, then yield more deterministic relation predictions. In this part, the performance of MarkerGenie was verified with the 250 test samples from Lever et al. (2019) that contains four granular relation types, Diagnostic, Predictive, Predisposing and Prognostic between cancers and genes. MarkerGenie was compared with CIViCmine (Lever et al., 2019) in terms of precision and recall, where the precision–recall curves of the two methods are shown in Figure 6D–G. MarkerGenie obtained better precision and recall than CIViCmine.
In the following, we demonstrate how MarkerGenie can be applied to biomarker discoveries with different case studies.
3.4.1 Identification of colorectal cancer-related microbes
Colorectal cancer (CRC) is the third most common cancer worldwide and one of the primary causes of cancer-related deaths (Rawla et al., 2019). The association between CRC and the human gut microbiome is a focus of the current CRC research (Abdulla et al., 2021; Chattopadhyay et al., 2021; Sánchez-Alcoholado et al., 2020). In this study, we used MarkerGenie to find the microbes related to CRC from the literature and manually verified the results.
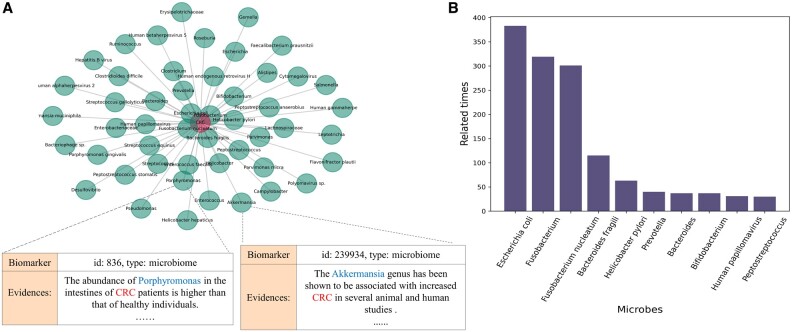
In searching for microbes related to CRC, MarkerGenie returned a total of 2257 sentences that included 264 microbes. Among these 2257 sentences, 2118 were correctly predicted, whereas 98 were wrongly predicted and 41 were difficult to judge via manual inspection. The overall sentence’s binary classification precision is 93.8%. For microbes, 247 out of 264 microbes are associated with CRC. In Figure 7, an example list of microbes and the corresponding sentences is shown in A. The top 10 microbes with the highest occurrences are shown in B, among these, eight of them have been previously shown to be significantly associated with CRC in the meta-analysis study (Thomas et al., 2019). The remaining two microbes ‘Helicobacter pylori’ and ‘Human papillomavirus’ also have been shown to be strongly related to CRC in more recent work (Chao et al., 2020; Wang et al., 2021). These results should provide a good reference to researchers studying CRC and the microbiome.
Fig. 7.
Statistics of microbes related to CRC returned by MarkerGenie. (A) Example of microbes and the corresponding sentences. (B) The top 10 microbes returned by MarkerGenie
As discussed earlier, upon a positive prediction of binary associative relation between CRC and a microbe, we can further generate more deterministic relation classification via distant supervision (see Section 2.5). Here, MarkerGenie produced 185 predisposing, 181 predictive, 154 prognostic, 67 treatment and 33 diagnostic relations.
3.4.2 Identification of breast cancer-related genes
Identifying relevant genes is valuable for the early diagnosis, prevention and treatment of breast cancer (Kazmi et al., 2022; Schettini et al., 2021; Zhang et al., 2022). We used MarkerGenie to search and rank the importance of the genes associated with breast cancer. Similar to BEST (Lee et al., 2016), we presented the top 10 genes found in MarkerGenie along with the ones identified by BEST, Polysearch2 and CIViCmine in Figure 8. Eight of them were identified in at least one of the other methods and reported to be associated with breast cancer in CIViC knowledge-base (https://civicdb.org) or NCBI’s GENE database (https://www.ncbi.nlm.nih.gov/gene/). The remaining two genes ‘ITK’ and ‘NAC’ were false positives upon inspection. Specifically, the term ‘NAC’ refers to a type of therapy for breast cancer. For ‘ITK’, the term identified in association with breast cancer is ‘EMT’, which is an alias of ‘ITK’ gene. However, ‘EMT’ refers to ‘epithelial-mesenchymal transition’ that is a process linked to breast cancer. Both false positives are valid entries in the gene list but had different meanings in the text. To further improve accuracy, ambiguities of terms in the list need to be resolved.
Fig. 8.
Top 10 genes retrieved with the query ‘Breast cancer’ by different systems
3.4.3 Disease–miRNA association inference
The output of MarkerGenie can also be directly used for other applications such as association prediction. We select the disease–miRNA association inference as a suitable application as it involves three-way interactions—disease–disease, disease–miRNA and miRNA–miRNA. The details of the inference method and experimental results are provided in Supplementary Section S3. Based on miRNA–miRNA functional similarity, disease–disease semantic similarity and the disease–miRNA associations identified by MarkerGenie, we can infer unknown disease–miRNA associations as accurately as the methods based on curated databases like HMDD (Huang et al., 2019). MarkerGenie can serve as a surrogate for the laboriously curated databases.
4 Conclusions
In this work, we proposed a text-mining system, MarkerGenie, to identify bioentity relations from texts and tables of publications in PubMed and PubMed Central. The identification problem was formulated as a relation classification task. A new unsupervised training data generation method and new classification model SBGT were introduced and tested with benchmark datasets and real-world case studies. The experimental results demonstrated the effectiveness of the system. There are further rooms for improvement, including cross-sentence relations extraction, improving negative samples selection, and better ways to handle ambiguities of short entity terms such as gene symbols. It is also favorable to recognize the context (e.g. conditions of experiments and biology relevance) in which the biomarkers are identified and to improve the entity extraction with text-mining methods (e.g. PubTator and NER models).
Supplementary Material
Acknowledgments
We would like to express our gratitude toward the editor and the anonymous reviewers whose valuable comments greatly contributed to this manuscript.
Author contributions
X.Y. and W.G. conceived the experiments. W.G. and M.Y. conducted the experiments. K.H. and W.P. analyzed the results. Z.Z., X.Y., K.H. and W.G. wrote and reviewed the manuscript.
Funding
This work was partially supported by National Key Research and Development Project [2019YFE0109600] and the National Natural Science Foundation of China [61871272 and 61911530218], the Guangdong Provincial Key Laboratory [2020B121201001], the Shenzhen Fundamental Research Program [JCYJ20190808173617147], and the open project of BGIShenzhen [BGIRSZ20200002].
Conflict of Interest: none declared.
References
- Abdulla M.-H. et al. (2021) Association of the microbiome with colorectal cancer development. Int. J. Oncol., 58, 1–12. [DOI] [PubMed] [Google Scholar]
- Ahmed M. et al. (2019) Identifying protein-protein interaction using tree LSTM and structured attention. In: 2019 IEEE 13th International Conference on Semantic Computing, Newport Beach, CA, USA. pp. 224–231.
- Beltagy I. et al. (2019) SciBERT: a pretrained language model for scientific text. In: Proceedings of the 2019 Conference on Empirical Methods in Natural Language Processing and the 9th International Joint Conference on Natural Language Processing, Hong Kong, China. pp. 3615–3620.
- Chao G. et al. (2020) The prevalence of human papillomavirus in colorectal cancer and adenoma: a Meta-analysis. J. Cancer Res. Ther., 16, 1656. [DOI] [PubMed] [Google Scholar]
- Chattopadhyay I. et al. (2021) Exploring the role of gut microbiome in Colon cancer. Appl. Biochem. Biotechnol., 193, 1780–1799. [DOI] [PubMed] [Google Scholar]
- Choi S.-P. (2018) Extraction of protein–protein interactions (PPIs) from the literature by deep convolutional neural networks with various feature embeddings. J. Inf. Sci., 44, 60–73. [Google Scholar]
- Devlin J. et al. (2019) BERT: pre-training of deep bidirectional transformers for language understanding. In: Proceedings of the 2019 Conference of the North American Chapter of the Association for Computational Linguistics: Human Language Technologies, Minneapolis, MN, USA. Vol. 1, pp. 4171–4186. [Google Scholar]
- Dingerdissen H.M. et al. (2020) OncoMX: a knowledgebase for exploring cancer biomarkers in the context of related cancer and healthy data. JCO Clin. Cancer Inform., 4, 210–220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fei H. et al. (2021) Enriching contextualized language model from knowledge graph for biomedical information extraction. Brief. Bioinform., 22, bbaa110. [DOI] [PubMed] [Google Scholar]
- Herrero-Zazo M. et al. (2013) The DDI corpus: an annotated corpus with pharmacological substances and drug–drug interactions. J. Biomed. Inform., 46, 914–920. [DOI] [PubMed] [Google Scholar]
- Hong L. et al. (2020) A novel machine learning framework for automated biomedical relation extraction from large-scale literature repositories. Nat. Mach. Intell., 2, 347–355. [Google Scholar]
- Hsieh Y.-L. et al. (2017) Identifying protein-protein interactions in biomedical literature using recurrent neural networks with long short-term memory. In: Proceedings of the Eighth International Joint Conference on Natural Language Processing, Taipei, Taiwan. Vol. 2, pp. 240–245. [Google Scholar]
- Hua L., Quan C. (2016) A shortest dependency path based convolutional neural network for protein-protein relation extraction. Biomed. Res. Int., 2016, 8479587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Z. et al. (2019) HMDD v3.0: a database for experimentally supported human microRNA–disease associations. Nucleic Acids Res., 47, D1013–D1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janssens Y. et al. (2018) Disbiome database: linking the microbiome to disease. BMC Microbiol., 18, 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kazmi N. et al. (2022) Rho GTPase gene expression and breast cancer risk: a Mendelian randomization analysis. Sci. Rep., 12, 1463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan F. et al. (2021) IBDDB: a manually curated and text-mining-enhanced database of genes involved in inflammatory bowel disease. Database, 2021, 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kropiwnicki E. et al. (2022) DrugShot: querying biomedical search terms to retrieve prioritized lists of small molecules. BMC Bioinformatics, 23, 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J. et al. (2020) BioBERT: a pre-trained biomedical language representation model for biomedical text mining. Bioinformatics, 36, 1234–1240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S. et al. (2016) BEST: next-generation biomedical entity search tool for knowledge discovery from biomedical literature. PLoS One, 11, e0164680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lever J. et al. (2019) Text-mining clinically relevant cancer biomarkers for curation into the CIViC database. Genome Med., 11, 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim S. et al. (2018) Drug drug interaction extraction from the literature using a recursive neural network. PLoS One, 13, e0190926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S. et al. (2016) Drug-Drug interaction extraction via convolutional neural networks. Comput. Math. Methods Med., 2016, 6918381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y. et al. (2015) Polysearch2: a significantly improved text-mining system for discovering associations between human diseases, genes, drugs, metabolites, toxins and more. Nucleic Acids Res., 43, W535–W542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma W. et al. (2017) An analysis of human microbe-disease associations. Brief. Bioinform., 18, 85–97. [DOI] [PubMed] [Google Scholar]
- Mintz M. et al. (2009) Distant supervision for relation extraction without labeled data. In: Proceedings of the Joint Conference of the 47th Annual Meeting of the ACL and the 4th International Joint Conference on Natural Language Processing of the AFNLP, Singapore. pp. 1003–1011.
- Neumann M. et al. (2019) ScispaCy: Fast and robust models for biomedical natural language processing. In: Proceedings of the 18th BioNLP Workshop and Shared Task, Florence, Italy. pp. 319–327.
- Noronha A. et al. (2019) The virtual metabolic human database: integrating human and gut microbiome metabolism with nutrition and disease. Nucleic Acids Res., 47, D614–D624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pyysalo S. et al. (2007) BioInfer: a corpus for information extraction in the biomedical domain. BMC Bioinformatics, 8, 50–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quan C. et al. (2016) Multichannel convolutional neural network for biological relation extraction. Biomed Res. Int., 2016, 1850404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawla P. et al. (2019) Epidemiology of colorectal cancer: incidence, mortality, survival, and risk factors. Prz. Gastroenterol., 14, 89–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahu S.K., Anand A. (2018) Drug-drug interaction extraction from biomedical texts using long short-term memory network. J. Biomed. Inform., 86, 15–24. [DOI] [PubMed] [Google Scholar]
- Sánchez-Alcoholado L. et al. (2020) The role of the gut microbiome in colorectal cancer development and therapy response. Cancers, 12, 1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schettini F. et al. (2021) Clinical, pathological, and PAM50 gene expression features of HER2-low breast cancer. NPJ Breast Cancer, 7, 1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szklarczyk D. et al. (2021) The STRING database in 2021: customizable protein–protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res., 49, D605–D612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas A.M. et al. (2019) Metagenomic analysis of colorectal cancer datasets identifies cross-cohort microbial diagnostic signatures and a link with choline degradation. Nat. Med., 25, 667–678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C. et al. (2021) Hp-positive Chinese patients should undergo colonoscopy earlier and more frequently: the result of a cross-sectional study based on 13,037 cases of gastrointestinal endoscopy. Front. Oncol., 11, 698898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J.-H. et al. (2020) Genclip 3: mining human genes’ functions and regulatory networks from pubmed based on co-occurrences and natural language processing. Bioinformatics, 36, 1973–1975. [DOI] [PubMed] [Google Scholar]
- Wishart D.S. et al. (2021) MarkerDB: an online database of molecular biomarkers. Nucleic Acids Res., 49, D1259–D1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Y. et al. (2015) Classifying relations via long short term memory networks along shortest dependency paths. In: Proceedings of the 2015 Conference on Empirical Methods in Natural Language Processing, Lisbon, Portugal. pp. 1785–1794.
- Yadav S. et al. (2019) Feature assisted stacked attentive shortest dependency path based Bi-LSTM model for protein–protein interaction. Knowl. Based Syst., 166, 18–29. [Google Scholar]
- Zhang H. et al. (2019) Deep residual convolutional neural network for protein-protein interaction extraction. IEEE Access, 7, 89354–89365. [Google Scholar]
- Zhang W. et al. (2022) Epigenetic study of early breast cancer (EBC) based on DNA methylation and gene integration analysis. Sci. Rep., 12, 1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X. et al. (2018) CBD: a biomarker database for colorectal cancer. Database, 2018, 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Z. et al. (2016) Drug drug interaction extraction from biomedical literature using syntax convolutional neural network. Bioinformatics, 32, 3444–3453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou D. et al. (2018) Position-aware deep multi-task learning for drug–drug interaction extraction. Artif. Intell. Med., 87, 1–8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.