Abstract
A 8-week feeding trial was conducted to determine the effects of dietary supplementation with curcumin on growth, intestinal health, and gills resistance to ammonia stress in juvenile Seriola dumerili. Three isonitrogenous and isolipidic test diets were prepared by supplementing incremental levels of dietary curcumin at 0% (CUR0%, control), 0.01% (CUR0.01%), 0.02% (CUR0.02%), respectively. Fish were fed with experimental diet. Recovery and protection capacity after ammonia challenge assay was adopted to test the effect of curcumin. At the end of the feeding trial, the results showed that dietary supplementation with proper curcumin level had a significant positive effect on fish survival and intestinal histology structure. Meanwhile, dietary supplementation with proper curcumin level can improve intestinal health by increasing immune enzyme activity, up-regulating the expression of anti-inflammatory cytokines, down-regulating the expression of pro-inflammatory cytokines and regulating other immune-related genes. Subsequently, after the ammonia challenge and recovery experiment, the results of antioxidant-related genes and antioxidant enzymes showed that dietary supplementation with proper curcumin level can improve the gill protective and recovery capacity from ammonia stress through increasing antioxidant capacity. These results suggest that dietary supplementation with proper curcumin can promote the growth, intestinal health, gill resistance and recovery to ammonia stress of S. dumerili.
Keywords: Growth performance, Intestinal histology, Intestinal immune responses, Ammonia stress, Protective effect, Recovery capacity
Growth performance; Intestinal histology; Intestinal immune responses; Ammonia stress; Protective effect; Recovery capacity.
1. Introduction
The greater amberjack (Seriola dumerili) is considered a popular fish due to its thick and tender flesh, rich in protein and fat, it has recently received some attention in the aquaculture industry [1, 2]. Intensive farming has become more and more popular with the increased demand for S. dumerili, but high-density farming also brings a lot of stress to the farming process, such as the rise in the frequency of disease leading to huge economic losses [3, 4]. In addition, intensive farming causes an inevitable increase in ammonia concentration in the water environment, eventually accumulating to unsafe levels [5, 6]. When the organism is exposed to high concentrations of ammonia, abnormal oxidative reactions in aerobic metabolic pathways are caused, resulting in excess singlet oxygen and other reactive oxygen species (ROS). This may lead to different histopathological changes and a series of physiological effects, even triggering diseases and death, ultimately causing huge economic losses [7, 8]. The research about how to improve the immunity of aquaculture animals to deal with the occurrence of diseases has become a hot topic, with the most common solution being to control and prevent the disease with antibiotics and chemical drugs [9, 10]. In recent years, natural feed supplements that promote growth and disease resistance have been effectively used in aquafeeds with people's awareness of food safety and environmental protection has improved. In particular, extracts of Chinese herbs have been proven in many studies to improve the immune capacity and health of aquatic animals [11, 12, 13]. For example, the antioxidant capacity of the liver and kidney of tilapia (Oreochromis niloticus) can be increased when garlic was added to the diet [14]. Similarly, Nile tilapia showed a significant rise in lysozyme activity and phagocytosis when the diet was supplemented with a mixture of Astragalus and Lonicera extracts [15]. The Chinese herbs extract we chose for this experiment was curcumin, which is cheaper and more available than other Chinese herb extracts.
Curcumin (CM) is a yellow acidic phenol extracted from the turmeric rhizome, which is commonly used as a spice, colorant, and food preservative [16]. Previous studies have shown that CM has many biological activities, such as antioxidant [17], anti-bacterial [18], and anti-inflammatory effects [19, 20]. lysozyme and alternative hemolytic complement activity in rainbow trout serum were significantly improved when 400 mg/kg of curcumin was added to the diet [21]. Manju et al [22] also declared that feeding curcumin can inhibit liver lipid peroxidation of climbing perch. These results suggest that curcumin supplementation in the diet is effective in promoting the innate immune response of fish serum and liver to improve fish health. However, little information is available on the effects of dietary curcumin supplementation on the fish intestinal structure and immune capacity. The intestinal tract is a major site of fish digestion and immunity, which determines the absorption and health of fish, intestinal mucosal damage and inflammatory disease are important causes of fish mortality [23, 24].
Fish gill is sensitive to the concentration of ammonia in the aquaculture water, which is the main organ of fish respiration, osmoregulation, and ammonia excretion, its immune capacity defines the growth and survival of the fish [25]. Ammonia nitrogen is one of the main pollutants in aquaculture, and ammonia nitrogen concentrations could be raised in intensive aquaculture with high-density farming and high feeding rates [26]. Excessive ammonia in the farmed water may lead to the destruction of non-specific immune defenses in the gill [27, 28]. Due to the immunomodulatory effects of curcumin, its supplementation in the diet may be a potential method to improve the resistance of high concentrations on fish, and to reduce the use of antibiotics and chemical drugs in aquaculture.
Thus, we investigated the effects of dietary curcumin supplementation on growth, intestinal structure, intestinal immune enzymes, and relative expression of intestinal immune genes of S. dumerili. Subsequently, the ammonia nitrogen challenge experiment and recovery experiment were conducted. The protective and recovery capacity of dietary curcumin supplementation on gill tissue was evaluated by the relative expression of gill tissue immune genes. The aim of the present research was whether curcumin could positively improve the health capacity of S. dumerili and thus increase the production efficiency of practical aquaculture farming. It also provides reference information for the application of herbs as dietary immune additive in aquaculture.
2. Materials and methods
2.1. Experimental diet preparation
The curcumin (purity>95%) used in the experiment was supplied by Xi'an Feida biotechnology Co., Ltd. In this study, three experimental feeds with isonitrogenous and isolipid were prepared by adding 0%, 0.01%, and 0.02% curcumin to the feeds. Then, a dietary feeding experiment was conducted on S. dumerili. The experimental feeds were designed and produced by Lingshui Tropical Aquatic Research and Development Center. All ingredients were crushed and screened through a 0.2mm mesh and subsequently mixed with oil using a commercial food mixer (Guangdong Li feng., LTD., China), and finally passed through a granulator to produce 4 mm diameter pellets that were air dried at room temperature (25 °C). The prepared feed was sealed in plastic bags and stored at -20 °C until used for the feeding trial. Experimental feed formula information is shown in Table 1.
Table 1.
Formulation and proximate composition of experimental diets.
| Ingredients | Diets |
||
|---|---|---|---|
| CUR0% | CUR0.01% | CUR0.02% | |
| Fish meal | 8850.0 | 8850.0 | 8850.0 |
| Corn gluten meal | 1050.0 | 1050.0 | 1050.0 |
| Soybean meal | 1200.0 | 1200.0 | 1200.0 |
| maize starch | 1200.0 | 1200.0 | 1200.0 |
| Microcrystalline Cellulose | 750.0 | 750.0 | 750.0 |
| Fish oil | 1050.0 | 1050.0 | 1050.0 |
| Lecithin | 150.0 | 150.0 | 150.0 |
| Vitamin premixa | 75.0 | 75.0 | 75.0 |
| Mineral premixb | 75.0 | 75.0 | 75.0 |
| Choline chloride | 75.0 | 75.0 | 75.0 |
| Betaine | 75.0 | 75.0 | 75.0 |
| Carboxymethyl cellulose | 450.0 | 450.0 | 450.0 |
| Curcumin |
0.0 |
1.5 |
3.0 |
|
Analyzed nutrients (%) | |||
| Dry matter | 87.9 | 87.9 | 87.9 |
| Crude protein | 49.7 | 49.7 | 49.7 |
| Crude lipids | 12.7 | 12.7 | 12.7 |
| Crude ash | 10.7 | 10.7 | 10.7 |
According to each parallel 15000g feed, the weight of each component of the feed is shown in the table. The CUR0%, the CUR0.01% and the CUR0.02% represented the experimental groups fed with 0 (control), 100 mg/kg and 200 mg/kg curcumin, respectively, that is, the amount of curcumin in each 15000g feed was 0, 1.5 and 3g, respectively.
Vitamin premix (mg kg−1 diet): vitamin A 9000000 (IU kg−1 diet), vitamin K3 600 (IU kg−1 diet), vitamin D 2500000 (IU kg−1 diet), vitamin E 500 (IU kg−1 diet), vitamin B (B1 3200, B2 10900, B5 20000, B6 5000, B12 1160), vitamin C 50000, phaseomannite 1500, calcium pantothenate 200, niacin 400, folic acid 50, biotin 2.
Mineral premix (mg kg−1 diet): KCl 70; KI 1.5; MgSO4·7H2O 300; MnSO4·4H2O 3; CuCl2 5; ZnSO4·7H2O 14; CoCl2·6H2O 0.5; FeSO4·7H2O 15; CaCl2 2.8 (g kg−1 diet); KH2 PO4·H2O 4.5 (g kg−1 diet). The dietary energy was calculated as carbohydrate: 17.15 MJ kg−1, protein: 23.64 MJ kg−1,120 lipid: 39.54 MJ kg −1.
2.2. Experimental fish and feeding experiment
The S. dumerili used in the experiment were provided by the Tropical Fisheries Research and Development Center, South China Sea Fisheries Research and Development Center, South China Sea Fisheries Research Institute, Chinese Academy of Fishery Science (Lingshui, Hainan, China). In experiment Ⅰ, before the start of the feeding experiment, healthy fish which transported to the experimental conditions. A total of 135 fish with an initial weight of 151.44 ± 7.16 g were randomly distributed into 9 circulating mariculture tanks filled with 200L water at the stocking rate of 15 fish per tank. The fish were acclimated in the experimental system for a week when transferred to the experimental tank. Each experimental diet was randomly assigned to three replicates. Nine tanks were assigned to three groups feeding different diets (curcumin supplementation at 0, 100, and 200 mg/kg, respectively), and no curcumin supplementation group as the control group. Fish from each groups were weighed before the start of the experiment. Visual satiation was achieved by hand feeding twice daily (8:00 am and 4:00 pm). Dead fish within 72 h of stocking were replaced by similar sized fish. Feces were siphoned out to avoid water pollution at 1h after feeding. During the experiment, the water temperature, salinity, pH of cultured seawater were maintained at 27–31 °C, 35 g/L, and 7.5–8.0, respectively. Aeration was provided to maintain dissolved oxygen levels near saturation (6.5–7 mg/L). The nitrite content was kept lower than 0.02 mg/L, and ammonia nitrogen content was lower than 0.1 mg/L. The feeding experiment lasted for 8 weeks. Experiment Ⅱ was conducted to determine whether the dietary supplement with curcumin could relieve the stress induced by NH3–N and enhance the ability of recover. Ammonia challenge experiment was conducted on the fish after 8 weeks of feeding experiments. Ammonium chloride (NH4Cl) was added into each pool to prepare the culture water at a concentration of 1 g/L. When all the fish show a visible negative effects, such as manic agitation or faint, the duration was approximately 6 min according to our pre-experiments. Then, two fish were randomly selected from each pool, one of the fish was placed in normal seawater for recovery and subsequently sampled in the fainting state, and the other was sampled as the ammonia nitrogen challenge sample. A schematic representation of the design of the experiment Ⅰ, experiment Ⅱ, and measured parameters with different diet feeding is presented in Figure 1.
Figure 1.
A schematic representation of the rearing system and measured parameters in S. dumerili feeding different diet.
2.3. Sampling
At the end of the experiment Ⅰ, fish were starved 24h before sampling, then anesthetized with Eugenol at 50 mg/L. All fish were measured for final body weight (FBW). The front intestine of eight fish was collected from each treatment group, three intestines were fixed in 10% buffered formalin for the analysis of histological observation, and five intestines were immediately aliquoted into the sterile tube, subsequently frozen using liquid nitrogen and finally stored at -80 °C for the analysis of immune enzymes activities and intestinal immune gene expression. At the end of the experiment Ⅱ, the gill of fish from different treatment groups were sampled and placed in separate centrifuge tubes, then rapidly snap-frozen in liquid nitrogen and stored at -80 °C until analysis.
2.4. Histological observation of intestine
The fixed intestine samples were dehydrated in a graded ethanol series, embedded in paraffin, and sectioned. The sections were stained with hematoxylin and eosin (H&E). Finally, the samples were photographed with a light microscope (100×). We referred to previous studies on fish intestinal histological structure [10, 29]. The degree of variation in the intestinal fold height, enterocyte height, and muscular thickness was evaluated by Caseviewer 2.3. Applying the scale in the bottom right corner of the picture as the standard, five complete folds were selected for each section, their height (μm), enterocyte height (μm), and muscle thickness (μm) were measured.
2.5. Assay of enzyme activities
Intestines and gill tissues were homogenized in ice-cold saline solution (0.86%) in the proportion of 1:9 (w/v). After centrifugation (2500∗g, 10min, 4 °C), the supernatants were separated and kept at 4 °C for further analysis. The Acid phosphatase (ACP), Alkaline phosphatase (AKP), Lysozyme (LZM), Superoxide dismutase (SOD) and Glutathione peroxidase (GSH-Px) were analyzed according to the instructions of commercially available kits (Nanjing Jiancheng biotech. Co, Nanjing, China). The protein concentration of the enzyme extracts was measured by using the Bradford method [30].
2.6. Gene expression
The total RNA was isolated from frozen tissue by Trizol Reagent (Invitrogen, Thermo Fisher Scientific Co., Ltd., Shanghai, China). The micro ultraviolet spectrophotometer (ND5000, Biotech Corporation, Beijing, China) and agarose gel electrophoresis were used to determine the concentration and purity of RNA. It was considered effective when the OD260/280 (optical density) was in the range of 1.9–2.1μg. According to the manufacturer's instructions of One-Step gDNA Removal and cDNA Synthesis SuperMix (EasyScript, Beijing TransGen Biotech., Ltd., Beijing, China), total RNA was extracted for reverse transcription and the and synthesize the first-strand cDNA accordingly. Quantitative Real-time PCR analysis was conducted by the Real-Time PCR System (Q1000, Hangzhou LongGene Scientific Instruments Co., Ltd., Hangzhou, China). Running conditions were performed as follow: initial denaturation at 95 °C for 15 min, 40 cycles of 95 °C for 10s, 60 °C for 20s, 72 °C for 30s. All PCR amplifications were conducted in triplicate with a final volume of 20μL, where the reactions include 2×RealUniversal PreMix (10μL), each PCR primers (0.6μL, 10 μmol/L), template cDNA (2μL), and RNase-free ddH2O. EF1 α and β-Actin were selected as the reference genes after pre-experimentation. The absence of non-specific products was determined by performing dissociation analysis at the end of each PCR reaction. A single peak was observed in the melt curve analysis, indicating a single PCR product. Serial dilutions of 10-fold were made for each primer pair of cDNA to establish a standard curve. PCR reaction efficiencies in the range of 90%–110% were achieved. Table 2 showed the primers used for the analysis. The relative expression of C4, C3, Hepc, IFN-γ, IgT, IL-10, IL-8, IL-1β, NF-κB1, Mx, TGF-β1, and TNF-α were determined in intestine tissue. The relative expression of Keap1, Hsp70, Cyt C, Cu-SOD, and GSH-Px were determined in gill tissue.
Table 2.
Primers used for real-time PCR.
| Gene abbreviation | Primer sequence (5′-3′) | Amplicon size (bp) | Accession number |
|---|---|---|---|
| C4 | F:ACATCGCAATGGAGGAGAAC | 170 | XM_022768450.1 |
| R:CAGTCCCGTGATAGGCTTTA | |||
| C3 | F:CATCGTTCCGCATCATAGC | 81 | XM_022755728 |
| R:AGTCCTTGACATCCACCCA | XM_022755434 | ||
| Hepc | F:GATGATGCCGAATCCCGTCAGG | 99 | XM_022764299.1 |
| R:CAGAAACCGCAGCCCTTGTTGGC | |||
| IFN-γ | F:TCTGTCTGACCCTCTGGTTTTC | 136 | LC146385.1 |
| R:AAGATGGGCTTCCCGCTA | |||
| 2IgT | F:TGGACCAGTCGCCATCTGAG | 196 | XM_022756471.1 |
| R:GGGAAACGGCTTTGAAAGGA | |||
| IL-10 | F:CAGTGTTGTCGTTTCGTGGAG | 173 | XM_022745101.1 |
| R:TGCTGTTCATTGCGTGGC | |||
| 1IL-8 | F:GAAGCCTGGGAGTAGAGCTG | 164 | XM_022758559.1 |
| R:GGGGTCTAGGCAGACCTCTT | |||
| 1IL-1β | F:TGATGGAGAACATGGTGGAA | 205 | XM_022753745.1 |
| R:GTCGACATGGTCAGATGCAC | |||
| NF-κB1 | F:CACAGACAGTTCGCCATCG | 185 | XM_022761336.1 |
| R:AGCGTCTTCTGCCTCTTCC | |||
| Mx | F:GACTTGGCTCTACCTGCTATCG | 177 | XM_022744797.1 |
| R:GCTTATCTTTCCGTACCACTCC | |||
| TGF-β1 | F:CGGAGCTGCGGATGTTAA | 111 | XM_022738547.1 |
| R:TGGTGATGAAGCGGGAAG | |||
| 1TNF-α | F:GAAAACGCTTCATGCCTCTC | 212 | XM_022746377.1 |
| R:GTTGGTTTCCGTCCACAGTT | |||
| 3EF1α | F:ATCGTTGCCGCTGGTGTT | 134 | XM_022744048.1 |
| R:TCGGTGGAGTCCATCTTGTT | |||
| 1,3β-Actin | F:TCTGGTGGGGCAATGATCTTGATCTT | 212 | XM_022757055.1 |
| R:CCTTCCTTCCTCGGTATGGAGTCC | |||
| Keap1 | F:CCTCCATAAACCCACCAAAG | 203 | XM_022766859.1 |
| R:CAGCGTAGAAAAGCCCACT | |||
| HSP70 | F:CACGTATTCTTGCGTTGGG | 146 | XM_022741879.1 |
| R:TCATGGCGACCTGGTTCT | |||
| Cyt C | F:AGGCAGCGGGCTACTCTTA | 89 | XM_022748189.1 |
| R:GGGGTTCTCCAGGTACTCCA | |||
| Cu-SOD | F:AGGACCTCACTTCAACCCC | 93 | XM_022738876.1 |
| R:GCTCCAGCAGTCACATTCC | |||
| GSH-Px | F:ACCAGCGGTACTCCAGCAA | 118 | XM_022745698.1 |
| R:CCAGGACGGACATACTTCAGA |
2.7. Calculations and statistical analysis
The weight gain and survival of fish were calculated based on the following formulae:
The weight gain (WG, %) = 100∗(final weight-initial weight) /initial weight
Survival (%) = 100∗(final amount of fish) /(initial amount of fish)
The relative quantity of gene expression was calculated according to the 2−△△Ct method [31]. All data are expressed as the standard deviation (mean ± SD). All data were firstly examined for homogeneity of variance using SPSS Statistic 23.0 software. One-way analysis of variance (ANOVA) was used to test the effect of dietary manipulation when the data had homogeneous variance. The tukey test was used to determine significant differences among treatment groups, and probability values of P < 0.05 were deemed to be statistically different.
3. Results
3.1. Survival and growth performance after dietary curcumin supplementation
The final body weight, weight gain, and survival increased first and then decreased with the increase of dietary curcumin supplementation level. Both final body weight and weight gain were higher in the curcumin-containing than that of control group, but there were no significant differences (P>0.05) among the groups. The survival of CUR0.01% group was significantly higher (P<0.05) than that of control group, while the survival of CUR0.02% had no significant difference (P>0.05) (Table 3).
Table 3.
Growth performance and survival of S.dumerili fed with diets having different levels of curcumin for 8 weeks.
| Items | Diets |
||
|---|---|---|---|
| CUR0% | CUR0.01% | CUR0.02% | |
| IBW (g) | 149.02 ± 5.17 | 153.51 ± 4.09 | 151.78 ± 3.85 |
| FBW(g) | 286.54 ± 33.21 | 324.01 ± 27.88 | 299.48 ± 36.60 |
| WG (%) | 92.28 ± 21.12 | 111.07 ± 18.16 | 97.31 ± 24.11 |
| Survival (%) | 75.56 ± 6.29a | 91.11 ± 8.31b | 77.78 ± 8.31a |
Values presented as mean ± SD of samples (n = 3).
Values of each parameter in the same row with different superscripts are significantly different (ANOVA, P<0.05).
3.2. Intestinal histology structure after dietary curcumin supplementation
The intestinal structure of each groups was shown in Figure 2 (a, b, c). The fold height, muscular thickness, and enterocyte height of intestine all increased and then decreased with the increase of dietary curcumin supplementation level, and the significantly higher fold height, muscular thickness and enterocyte height was found in the CUR0.01% group than that of control group (P<0.05). When the curcumin content continued to increase, the fold height in the CUR0.02% group was not significantly (P>0.05) different compared to the control group, while the muscular thickness and enterocyte height were significantly higher (P<0.05) (Table 4).
Figure 2.
Intestinal segments obtained from fish fed with different diet groups (letters a, b, and c represent the CUR0% diet group, CUR0.01% diet group, and CUR0.02% diet group, respectively). The hF means fold height, hE means enterocyte height and Mt means muscular thickness. Measuring stick = 100μm.
Table 4.
Histological structure of the intestine of S. dumerili fed different experimental diets for 8 weeks.
| Parameters | Diet group |
||
|---|---|---|---|
| CUR0% | CUR0.01% | CUR0.02% | |
| Fold height (μm) | 439.71 ± 60.31b | 591.54 ± 52.93a | 414.07 ± 33.69b |
| Muscular thickness (μm) | 143.73 ± 11.89c | 190.80 ± 14.44a | 174.98 ± 11.74b |
| Enterocyte height (μm) | 11.94 ± 1.35b | 17.14 ± 1.52a | 16.68 ± 1.97a |
Values are means of triplicate groups±SD (n = 3). Within a row, means with different letters are significantly different (ANOVA, P<0.05).
3.3. Intestinal immune enzyme activity after dietary curcumin supplementation
Dietary curcumin supplementation level were positively correlated with AKP activity, with the significantly difference (P<0.05) was found in CUR0.01% and CUR0.02% group. The activity of ACP and LZM all increased and then decreased with the increase of dietary curcumin supplementation level. Compared to the control group, the significantly higher (P<0.05) ACP and LZM activity was observed in CUR0.01% group, the significantly lower (P<0.05) LZM activity was observed in CUR0.02% group (Table 5).
Table 5.
The immune enzyme activities in the intestine of S.dumerili fed different experimental diets for 8 weeks.
| Enzyme activity | Diets |
||
|---|---|---|---|
| CUR0% | CUR0.01% | CUR0.02% | |
| AKP (U/gprot) | 34482.37 ± 746.10b | 56316.33 ± 4490.40a | 60379.50 ± 664.71a |
| ACP (U/gprot) | 8813.94 ± 589.72b | 10957.19 ± 1583.26a | 9491.80 ± 1477.91 ab |
| LZM (U/ml) | 21.70 ± 0.75b | 32.96 ± 1.72a | 15.95 ± 1.76c |
Values presented as mean ± SD of samples (n = 3).
Values of each parameter in the same row with different superscripts are significantly different (ANOVA, P<0.05).
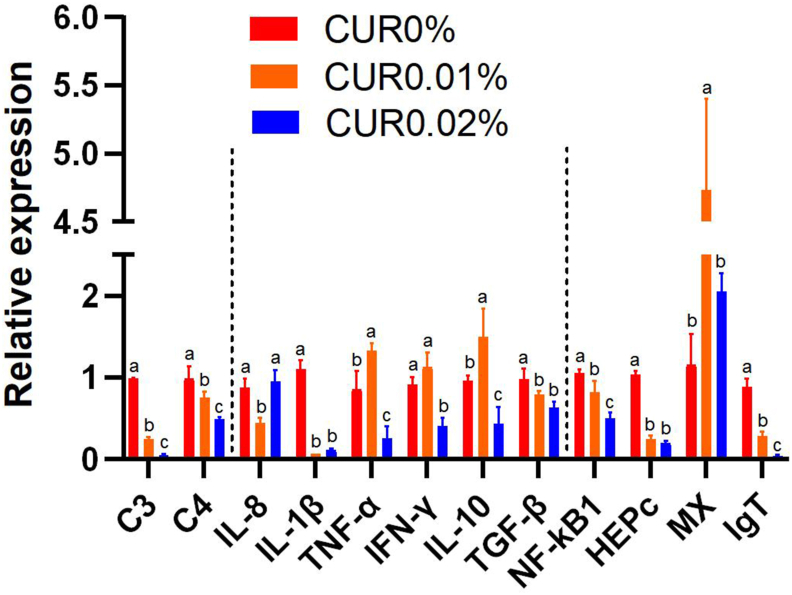
3.4. Intestinal immune gene expression after dietary curcumin supplementation
The effects of curcumin on immune gene expression were determined by q-RT-PCR in intestine tissues. Q-RT-PCR results suggested that significantly lower (P<0.05) relative expression of C4, C3, Hepc, IgT, IL-1β, NF-κB1 and, TGF-β was found in the curcumin-containing groups compared to the control group (CUR0%). Among them, the relative expression of C4, C3, IgT and NF-κB1 was negatively correlated with the dietary curcumin supplementation level, and there was no significant difference (P>0.05) in the relative expression of Hepc, IL-1β and TGF-β between the two groups containing curcumin. The relative expression of IFN-γ, IL-10, TNF-α and MX in intestine tissues first increased and then decreased with the increase of dietary curcumin supplementation level. And the relative expression of IFN-γ was not significantly different in the CUR0.01% group compared to the control group, while the relative expression of IL-10, TNF-α and MX was significantly higher (P<0.05). When the dietary curcumin supplementation level continued to rise, the significantly lower (P<0.05) relative expression of IFN-γ, IL-10, and TNF-α was found in the CUR0.02% group than that of control group while the relative expression of MX have not significant difference (P>0.05). In addition, the relative expression of IL-8 in the intestine first decreased and then increased with the increase of dietary curcumin supplementation level. And the significantly lower (P<0.05) relative expression of IL-8 was found in the CUR0.01% group compared to the control group (Figure 3)
Figure 3.
Effects of different content of curcumin supplementation on relative gene expression of C4, C3, Hepc, IFN-γ, IgT, IL-10, IL-8, IL-1β, NF-κB1, MX, TGF-β and TNF-α in the intestine of S. dumerili (n = 3). Column bars, within the same gene, sharing a common letter (a,b,c) are not significantly different (ANOVA, P>0.05).
3.5. The activity of antioxidant enzymes in the gill of S. dumerili after ammonia challenge experiment and recovery experiment
The activity of GSH-Px and SOD enzyme in gill was analysed after ammonia challenge and recovery experiment, respectively (Figure 4 a, b). After ammonia challenge, the significantly higher (P<0.05) GSH-Px and SOD activity were only found in the CUR0.02% group. After recovery experiment, both GSH-Px and SOD activity were positively correlated with dietary curcumin supplementation level. The significantly higher (P<0.05) GSH-Px activity was only found in the CUR0.02% group, and significantly higher SOD was found in the curcumin-containing groups. Compared to ammonia challenge phase, the GSH-Px activity increased significantly (P<0.05) in all groups after recovery, while SOD activity decreased significantly (P<0.05).
Figure 4.
The activity of antioxidant-related enzymes (letter a and b represent GSH-Px and SOD, respectively) in gill tissue of S. dumerili subjected to ammonia challenge experiment and recovery experiment (n = 3). Different letters above bars indicate significant differences (ANOVA, P>0.05).
3.6. Relative expression of immune-related genes in the gill of S. dumerili after ammonia challenge experiment and recovery experiment
The immune-related genes were quantified in the gill tissue of S. dumerili after ammonia challenge experiment and recovery experiment, respectively (Figure 5 a–e). After ammonia challenge, the significantly lower relative expression of Keap1 was found in curcumin-containing group than that of control group, and the significantly higher (P<0.05) relative expression of Hsp70 and Cu-SOD was only found in the CUR0.02% group. There was no significant difference (P>0.05) in the relative expression of Cyt C between all groups, and significantly higher (P<0.05) relative expression of GSH-Px was only found in the CUR0.02% group. The trend in the relative expression of Keap1 gene after the recovery experiment was consistent with that after the ammonia challenge experiment, but there were some changes in the remaining genes. The relative expression of HSP70 genes in the curcumin-supplemented group was not significantly different (P>0.05) compared to the control group after the recovery trial, with the maximum of HSP70 was found in CUR0.01% group and the minimum of HSP70 was found in CUR0.02% group. The significantly higher (P<0.05) relative expression of Cyt C was only found in CUR0.02% group. And the significantly higher (P<0.05) relative expression of Cu-SOD and GSH-Px was found in the curcumin-containing groups than that of control group, with the maximum value was found in the CUR0.01% group. Compared to the ammonia challenge phase, the relative expression of Keap1 in all groups, HSP70 in the CUR0.02% group, Cyt C in the CUR0% and CUR0.01% groups, GSH-Px in the CUR0% group and CUR0.02% group were significantly lower (P<0.05) after recovery, while the relative expression of Cu-SOD and GSH-Px in the CUR0.01% group were significantly higher (P<0.05).
Figure 5.
The relative expression of antioxidant-related genes (letters a, b, c, d and e represent Keap1, HSP70, Cyt C, Cu-SOD and GSH-Px, respectively) in gill tissue of S. dumerili subjected to ammonia challenge experiment and recovery experiment (n = 3). Different letters above bars indicate significant differences (ANOVA, P>0.05).
4. Discussion
4.1. The effects on growth performance after dietary supplementation with curcumin
In this experiment, dietary curcumin supplementation can not significantly improve weight gain in S. dumerili. Previous studies have shown inconsistent results on whether curcumin can significantly improve the growth of aquatic animals. Specific growth rate and daily growth coefficient of pacific white shrimp (Litopenaeus vannamei) were significantly improved when curcumin-loaded chitosan nanoparticles were supplemented in the diet [32]. Dietary supplementation with 5 g/kg curcumin significantly improved the final body weight (FBW), percent weight gain (PWG), and feed efficiency (FE) of crucian carp (Carassius auratus) [33]. In contrast, dietary supplementation with curcumin had no significant effect on the weight gain (WG), specific growth rate (SGR), feed conversion ratio (FCR) of rainbow trout (Oncorhynchus mykiss) [21]. We suspect that this may be related to the species of aquatic animal, the breeding cycle of S. dumerili is usually 1–2 years and the 8-week experiment was short, therefore the difference in growth performance between the treatment groups was not significant. In addition, the results showed that dietary supplementation with the proper rate of curcumin significantly increased the survival rate of fish. This data is consistent with the results of Bhoopathy et al. that dietary supplementation with curcumin-loaded chitosan nanoparticles significantly improved the survival rate of L. vannamei [21]. However, the survival rate of fish did not improve significantly when dietary supplementation with curcumin was above the optimum level, this may be related to the reduction in food intake. When excess curcumin is added to the diet, curcumin produces odor and acts as a phytoestrogen, both of these factors can reduce the amount of food taken by fish [34, 35].
4.2. The effects on histology of the intestine after dietary supplementation with curcumin
This experiment showed that dietary supplementation with extracts of Chinese herbs positively improved the intestinal structure of S. dumerili. This is consistent with previous studies that higher intestinal villi height and crypt depth were found in early-weaned pigs which dietary supplementation with a plant extract combination [36]. As the primary site of food digestion, nutrient uptake, and transformation, the intestine is central to physiological functioning, and optimum utilization of dietary nutrients depends on their functional effectiveness [37]. In the result it can be visualized that in the treatment containing curcumin, the intestinal folds appeared significantly curved, resulting in an increase of the contact area of nutrients with the intestinal epithelium as fold height and enterocyte height rises, while the intestinal thickness is a key factor in intestinal motility and absorption [38]. This suggests that curcumin enhances the absorption of nutrients by improving intestinal integrity. This is also consistent with the results of our other experiment in which dietary supplementation with curcumin significantly improved intestinal digestive enzymes activity (lipase and trypsin) [39].
4.3. The effects on immune response of the intestine after dietary supplementation with curcumin
In addition, curcumin supplementation also enhances intestinal immune enzyme activity. Firstly, the intestinal integrity is critical to fish's health, intestinal epithelium can produce a natural physical barrier and express a large amount of antimicrobial peptides that defend invasion by pathogens [40]. Secondly, curcumin stimulates the innate immune system of fish, which is the mainline of defense against invading pathogens and is more important for fish than mammals [41]. Our results show that dietary supplementation with the right proportion of curcumin significantly increases the activity of AKP, ACP, and LZM in the intestine, which are important indicators of the immune function and health status of the organism. Metabolism in aquatic organisms is regulated in part by phosphorylation and dephosphorylation, and the completion of these processes is mainly catalyzed by different phosphatases. ACP and AKP are involved in the transfer and metabolism of phosphate groups and the hydrolysis of metabolites to phosphate and ethanol [42, 43]. And lysozyme activity is considered to be a major indicator of innate immune defense in fish, with the potential to hydrolyze N-acetylglutamate and N-acetylglucosamine in the peptidoglycan layer of the cell wall of Gram-positive and Gram-negative bacteria, and it also triggers the complement system and phagocytosis through modulators [44]. The ability of curcumin to increase immune enzymes activity has been confirmed in previous studies. For example, the AKP activity in the intestine of crucian carp was significantly increased when curcumin was supplemented at 5 g/kg in the diet [33]. And dietary curcumin supplementation significantly increased lysozyme and malondialdehyde activity in Nile tilapia [45]. We suggested that the ability of dietary curcumin supplementation to improve intestinal immunity is related to the fact that curcumin has antimicrobial effects. Many studies have analyzed the function of curcumin, with Tyagi et al. demonstrating a correlation between bacterial killing and membrane damage caused by curcumin [46] and Baldissera et al. suggesting that curcumin could improve the health of aquatic animals against bacterial pathogens [47].
At the gene level, some inflammation markers (Cytokine and complements) genes were chosen to determine the immune response after dietary supplementation with curcumin by their relative expression. Complement plays an important role in the fish immune system, which can promote phagocytosis, lyses and clears antigen-antibody aggregates, releases bioactive peptides to promote inflammatory responses, and enhances the sensitivity of B cell to antigens [48, 49, 50]. Complement protein C3 and C4 are central roles in the complement system which are classified as acute phase reactants, and it is usually accompanied by upregulation of the relative expression of C3 and C4 during inflammation [51]. The results showed that dietary curcumin supplementation was negatively correlated with the relative expression of C3, C4 gene mRNA, which may indicate that dietary curcumin supplementation significantly alleviates the inflammatory response of the fish intestines.
Dietary supplementation with the proper proportion of curcumin can improve intestinal health conditions by positively regulating cytokines. Cytokines are mainly composed of interleukins (ILs), transforming growth factor-β family (TGF-β), tumor necrosis (TNFs), and interferon (IFN), etc. [52]. Cytokines can also be divided into two types, pro-inflammatory and anti-inflammatory cytokines, the balance of which plays a vital role in maintaining the physiological and immune health of the fish [53]. Previous studies suggested that the organism releases pro-inflammatory cytokines when regulated by physiological adaptive mechanism, which can induce various immune responses by triggering inflammation. However, the excessive inflammatory response may lead to the damage of glial cells and eventually to the death of the organism. Comparatively, the anti-inflammatory cytokine is responsible for eliminating the inflammation reaction response and restoring the organism's return to the normal situation [54, 55, 56]. The results in this experiment showed that dietary supplementation with appropriate levels of curcumin demonstrated the ability to inhibit pro-inflammatory cytokine IL-8, IL-1β, TNF-α, IFN-γ expression and promote anti-inflammatory cytokine IL-10 expression in the fish intestine. The ability of curcumin to against inflammatory responses has been widely reported. Cao, et al. [57] showed that 0.5% and 1.0% curcumin reduced liver damage in Cyprinus carpio by inhibiting the expression of IL-1β, TNF-α, and NF-κB1.
Dietary supplementation with curcumin maintains the healthy intestine by inhibiting the relative expression of NF-κB1 in the intestine. Transcription factor nuclear factor kappa B (NF-κB1) is an important intracellular nuclear transcription factor. It is involved in the organism's inflammatory response, immune response, and stress response, but over-activation expression of NF-κB1 is often accompanied by diseases such as inflammation and cancer [58, 59]. Previous studies have shown that dietary supplementation with curcumin can block the NF-κB1 pathway to inhibit cytokine expression [60]. Curcumin downregulates the relative expression of NF-κB1 in mice brain induced to oxidative stress by ethanol [61]. Another test on Oncorhynchus Mykiss showed the relative expression of NF-κB1 was suppressed after curcumin supplementation in the diet [60]. In this experiment, the relative expression of NF-κB1 in the fish intestine was negatively correlated with the content of curcumin in the diet. Perhaps the persistence of the inflammatory response in the fish intestine could be avoided more rapidly by dietary curcumin supplementation.
Some other immune-related genes (Hepc, Mx, IgT) were also identified in this study. Hepcidin (Hepc) is a member of antimicrobial peptides which have good antimicrobial activity, and the action of pathogenic bacteria often causes an increase of its expression [62, 63, 64]. Immunoglobulin T (IgT) plays an essential role in the mucosal immunity of teleost fish, which is mainly exists in the intestinal mucosa, and intestinal inflammation in teleost fish was able to promote the relative expression of IgT [65]. The results showed that the relative expression of Hepc and IgT was negatively correlated with the curcumin level. The protein encoded by Mx gene is an antiviral protein downstream of the interferon system (IFN) signaling pathway, with multiple antiviral activities and broad-spectrum antibacterial effects, inhibiting a variety of negative-strand RNA viruses [66]. The results showed that the appropriate level of curcumin could significantly increase the relative expression of Mx. This may indicate that dietary curcumin supplementation can improve intestinal immune status by inhibiting pathogenic bacteria and inflammatory responses, as well as improving the ability of the fish intestine to resist viruses. In addition, curcumin is more available and cheaper than astaxanthin and antibiotics. So combining the results of intestinal immune enzymes and immune-related gene expression, dietary supplementation with curcumin supplementation may be a low-cost and effective method to improve the health condition of fish.
4.4. The effects on gill's resistance to ammonia stress and recovery after dietary supplementation with curcumin
The fish gill is very sensitive to external stimuli, as it is directly involved in the exchange of substances between the external environment and the interior of the organism. Many studies have shown that the growth, tissue integrity, and immune capacity are compromised when ammonia concentrations exceed the maximum tolerated of the organism [25, 67]. Dietary supplementation with curcumin can improve the antioxidant capacity of S. dumerili gills to reduce damage caused by acute high concentrations of ammonia. Excess reactive oxygen species (ROS) produced by acute ammonia stress may lead to reduced antioxidant capacity and even death in aquatic animals. ROS are primarily synthesized as a part of the immune response against invading pathogens, but the massive accumulation of ROS and the reduced ability of the organism to mitigate them lead to oxidative damage of the host biological molecules including DNA [8, 68]. The ability of mitigating ROS can be demonstrated by antioxidant enzymes, with both Cu/Zn-SOD (Cu/Zn-Superoxide Dismutase) and GSH-Px (Glutathione peroxidase) being the most important ones [69]. SOD converts superoxide anion into hydrogen peroxide, which is subsequently decomposed by GSH-Px to protect cells from severe damage [70, 71]. And transcription factors can be activated by ROS to control antioxidant enzymes, such as Nrf2/Keap1, which is a key signaling pathway of antioxidant damage in the organisms [72]. Under physiological conditions, it binds to the cytosolic chaperone Keap1 to maintain a relatively inhibitory activity. Upon exposure to oxidative stressors, Nrf2 activates a range of downstream antioxidant enzymes such as SOD and CAT, thereby protecting the organism from stress-induced or exacerbated diseases [73]. The results of antioxidant-related genes and antioxidant enzyme activities demonstrate that dietary supplementation with curcumin can maintain health conditions by scavenging ROS and inducing antioxidant reactions directly or indirectly through antioxidant activity in acute with high ammonia concentrations. During the recovery phase, the group containing curcumin still maintained high antioxidant capacity. Similar results were reported that curcumin could alleviate ethanol-induced oxidative stress in the brain of rats by enhancing antioxidant capacity [61]. Curcumin reduces CCl4-induced oxidative stress by enhancing the liver antioxidant capacity of Jian carp [57]. These experiments confirmed that dietary curcumin supplementation could reduce oxidative stress to the organism. But it should be noted that the curcumin content was not proportional to the antioxidant capacity of the gills. During the recovery phase, the antioxidant capacity of the fish gill supplemented with high concentrations of curcumin was lower than the treatment group with low concentrations of curcumin, and more research about the reasons for this phenomenon is necessary.
In addition, dietary supplementation with the 0.02% concentration of curcumin significantly enhanced the resistance of fish gill against ammonia stress and enabled organisms to maintain the high antioxidant capacity and stability during recovery experiments. Hsp70 is a stress protein induced in tissues following various stimuli, which relative expression increases significantly and rapidly. Hsp70 binds to denatured proteins, preventing them from folding further and promoting their recovery or accelerating their degradation to maintain tissue function [74, 75]. And Cyt C is not only essential factors involved in apoptosis but also strongly inhibit the production of H2O2 [76,77]. The relative expression of Cyt C gene rises rapidly to maintain the organism's stability and increase its antioxidant capacity. Our results show that dietary supplementation with 0.02% curcumin not only protects against ammonia stress-induced damage in gill by significantly increasing the relative expression of Hsp70 but also improves the antioxidant capacity and stability of the organism by increasing the relative expression of Cyt C during the recovery phase. However, another of our studies has shown that the optimal content of curcumin supplementation will change due to different tissues of fish, which may be related to the function of different tissues.
5. Conclusion
In conclusion, we assessed the effect of dietary curcumin supplementation on S. dumerili by growth performance, intestinal histology structure, intestinal immune enzyme activity, the relative expression of intestinal immune-related genes and resistance of the gill to ammonia stress. The results showed that dietary supplementation with curcumin could have a positive effects on survival rate, intestinal structure and immune capacity, gill resistance to ammonia stress and stress recovery. We believe that our research may contribute to reducing the use of chemical drugs and antibiotics for healthy fish farming, and improving the production efficiency of practical aquaculture farming. However, the assessment of intestinal health is still not comprehensive enough, so further studies are needed such as the effect on the resistance to pathogenic bacteria, the effect on antioxidant capacity and intestinal microbial diversity. This will provide a better reference for practical application.
Ethics statement
No protected species were used during the experiment. This study was conducted in strict accordance with the recommendation in the Animal Welfare Committee of Chinese Academy of Fishery Sciences (approval code: 2020TD55).
Declarations
Author contribution statement
Yuhang He and Zhenhua Ma - Conceived and Designed the experiments; Wrote the paper.
Shiming Dai - Performed the experiments.
Zhengyi Fu - Analyzed and interpreted the data.
Gang Yu - Contributed reagents, materials, analysis tools.
Xiaomei Wang - Wrote the paper.
Funding statement
This work was supported by Central Public-interest Scientific Institution Basal Research Fund, Chinese Academy of Fishery Sciences [CAFS NO.2020TD55], Guangxi Innovation-Driven Development Project [Guike AA18242031].
Data availability statement
Data will be made avasilable on request.
Declaration of interest's statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
Contributor Information
Zhenhua Ma, Email: zhenhua.ma@hotmail.com.
Xiaomei Wang, Email: xiaomei_wang328@126.com.
References
- 1.Robles C.M.A.N. Rearing of greater amberjack (Seriola dumerili) 2019;44:18–25. [Google Scholar]
- 2.Mazzola A., Favaloro E., Sarà G. Cultivation of the mediterranean amberjack, Seriola dumerili (risso, 1810), in submerged cages in the western mediterranean sea. Aquaculture. 2000;181:257–268. [Google Scholar]
- 3.Anderson D.P. Environmental factors in fish health: immunological aspects. Fish Immun. Syst.: Organ. Pathog. Environ. 1996;15:289–310. [Google Scholar]
- 4.Luis A.I.S., Campos E.V.R., Oliveira J.L., Fraceto L.F. Trends in aquaculture sciences: from now to use of nanotechnology for disease control. Rev. Aquacult. 2019;11:119–132. [Google Scholar]
- 5.Randall D.J., Tsui T.K.N. Ammonia toxicity in fish. Mar. Pollut. Bull. 2002;45:17–23. doi: 10.1016/s0025-326x(02)00227-8. [DOI] [PubMed] [Google Scholar]
- 6.Wilson R.P. In: Fish Nutrition. third ed. Halver J.E., Hardy R.W., editors. Academic Press; San Diego: 2003. 3 - amino acids and proteins; pp. 143–179. [Google Scholar]
- 7.Hillaby B.A., Randall D.J. Acute ammonia toxicity and ammonia excretion in rainbow trout (Salmo gairdneri) J. Fish. Res. Board Can. 1979;36:621–629. [Google Scholar]
- 8.Qiu J., Wang W.N., Wang L.J., Liu Y.F., Wang A.L. Oxidative stress, DNA damage and osmolality in the Pacific white shrimp, Litopenaeus vannamei exposed to acute low temperature stress. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2011;154:36–41. doi: 10.1016/j.cbpc.2011.02.007. [DOI] [PubMed] [Google Scholar]
- 9.Bager F., Aarestrup F.M., Wegener H.C. Dealing with antimicrobial resistance – the Danish experience. Can. J. Anim. Sci. 2000;80:223–228. [Google Scholar]
- 10.Hossain M.S., Koshio S., Ishikawa M., Yokoyama S., Sony N.M., Kader M.A., Maekawa M., Fujieda T. Effects of dietary administration of inosine on growth, immune response, oxidative stress and gut morphology of juvenile amberjack, Seriola dumerili. Aquaculture. 2017;468:534–544. [Google Scholar]
- 11.Wang W., Sun J., Liu C., Xue Z. Application of immunostimulants in aquaculture: current knowledge and future perspectives. Aquacult. Res. 2017;48:1–23. [Google Scholar]
- 12.Khanna D., Sethi G., Ahn K.S., Pandey M.K., Kunnumakkara A.B., Sung B., Aggarwal A., Aggarwal B.B. Natural products as a gold mine for arthritis treatment. Curr. Opin. Pharmacol. 2007;7:344–351. doi: 10.1016/j.coph.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 13.Divyagnaneswari M., Christybapita D., Michael R.D. Enhancement of nonspecific immunity and disease resistance in Oreochromis mossambicus by Solanum trilobatum leaf fractions. Fish Shellfish Immunol. 2007;23:249–259. doi: 10.1016/j.fsi.2006.09.015. [DOI] [PubMed] [Google Scholar]
- 14.El-Barbary, Manal I. Detoxification and antioxidant effects of garlic and curcumin in Oreochromis niloticus injected with aflatoxin B-1 with reference to gene expression of glutathione peroxidase (GPx) by RT-PCR. Fish Physiol. Biochem. 2016;42:617–629. doi: 10.1007/s10695-015-0164-4. [DOI] [PubMed] [Google Scholar]
- 15.Ardó L., Yin G., Xu P., Váradi L., Szigeti G., Jeney Z., Jeney G. Chinese herbs (Astragalus membranaceus and Lonicera japonica) and boron enhance the non-specific immune response of Nile tilapia (Oreochromis niloticus) and resistance against Aeromonas hydrophila. Aquaculture. 2008;275:26–33. [Google Scholar]
- 16.Prasad S., Gupta S.C., Tyagi A.K., Aggarwal B.B. Curcumin, a component of golden spice: from bedside to bench and back. Biotechnol. Adv. 2014;32:1053–1064. doi: 10.1016/j.biotechadv.2014.04.004. [DOI] [PubMed] [Google Scholar]
- 17.Boonla O., Kukongviriyapan U., Pakdeechote P., Kukongviriyapan V., Pannangpetch P., Prachaney P., Greenwald S.E. Curcumin improves endothelial dysfunction and vascular remodeling in 2K-1C hypertensive rats by raising nitric oxide availability and reducing oxidative stress. Nitric Oxide. 2014;42:44–53. doi: 10.1016/j.niox.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 18.Bellio P., Brisdelli F., Perilli M., Sabatini A., Bottoni C., Segatore B., Setacci D., Amicosante G., Celenza G. Curcumin inhibits the SOS response induced by levofloxacin in Escherichia coli. Phytomedicine. 2014;21:430–434. doi: 10.1016/j.phymed.2013.10.011. [DOI] [PubMed] [Google Scholar]
- 19.Fu Y., Gao R., Cao Y., Guo M., Wei Z., Zhou E., Li Y., Yao M., Yang Z., Zhang N. Curcumin attenuates inflammatory responses by suppressing TLR4-mediated NF-kappaB signaling pathway in lipopolysaccharide-induced mastitis in mice. Int. Immunopharm. 2014;20:54–58. doi: 10.1016/j.intimp.2014.01.024. [DOI] [PubMed] [Google Scholar]
- 20.Shi X., Zheng Z., Li J., Xiao Z., Qi W., Zhang A., Wu Q., Fang Y. Curcumin inhibits Abeta-induced microglial inflammatory responses in vitro: involvement of ERK1/2 and p38 signaling pathways. Neurosci. Lett. 2015;594:105–110. doi: 10.1016/j.neulet.2015.03.045. [DOI] [PubMed] [Google Scholar]
- 21.Kohshahi A.J., Sourinejad I., Sarkheil M., Johari S.A. Dietary cosupplementation with curcumin and different selenium sources (nanoparticulate, organic, and inorganic selenium): influence on growth performance, body composition, immune responses, and glutathione peroxidase activity of rainbow trout (Oncorhynchus mykiss) Fish Physiol. Biochem. 2018;45:793–804. doi: 10.1007/s10695-018-0585-y. [DOI] [PubMed] [Google Scholar]
- 22.Manju M., Akbarsha M.A., Oommen O.V. In vivo protective effect of dietary curcumin in fish Anabas testudineus (Bloch) Fish Physiol. Biochem. 2012;38:309–318. doi: 10.1007/s10695-011-9508-x. [DOI] [PubMed] [Google Scholar]
- 23.Surai P. Antioxidant-prooxidant balance in the intestine: applications in chick placement and pig weaning. J. Vet. Sci. Med. 2015;3:1–16. [Google Scholar]
- 24.Ellis A.E. Immunity to bacteria in fish. Fish Shellfish Immunol. 1999;9:291–308. [Google Scholar]
- 25.Dutra F.M., Rönnau M., Sponchiado D., Forneck S.C., Freire C.A., Ballester E.L.C. Histological alterations in gills of Macrobrachium amazonicum juveniles exposed to ammonia and nitrite. Aquat. Toxicol. 2017;187:115–123. doi: 10.1016/j.aquatox.2017.04.003. [DOI] [PubMed] [Google Scholar]
- 26.Chen J.C., Ting Y.Y., Lin J.N., Lin M.N. Lethal effects of ammonia and nitrite on Penaeus chinensis juveniles. Mar. Biol. 1990;107:427–431. [Google Scholar]
- 27.Zhao Q., Feng K., Zhang L., Bai Y., Yao W. Effects of Acute Ammonia Stress on Antioxidant Responses, Histopathology and Ammonia Detoxification Metabolism in Triangle Sail Mussels (Hyriopsis cumingii). Water. 2021;13(425) [Google Scholar]
- 28.Yang W., Sun H., Xiang F., Yang Z., Chen Y. Response of juvenile crucian carp (Carassius auratus) to long-term ammonia exposure: feeding, growth, and antioxidant defenses. J. Freshw. Ecol. 2011;26:563–570. [Google Scholar]
- 29.Chen G., Feng L., Kuang S., Liu Y., Jiang J., Hu K., Jiang W., Li S., Tang L., Zhou X. Effect of dietary arginine on growth, intestinal enzyme activities and gene expression in muscle, hepatopancreas and intestine of juvenile Jian carp (Cyprinus carpio var. Jian) Brit. J. Nutr. 2012;108:195–207. doi: 10.1017/S0007114511005459. [DOI] [PubMed] [Google Scholar]
- 30.Bradford M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 31.Livak K.J., Schmittgen T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods (San Diego, CA, U. S.) 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 32.Bhoopathy S., Inbakandan D., Rajendran T., Chandrasekaran K., Prabha S B., Reddy B.A., Kasilingam R., Rameshkumar V., Dharani G. Dietary supplementation of curcumin-loaded chitosan nanoparticles stimulates immune response in the white leg shrimp Litopenaeus vannamei challenged with Vibrio harveyi. Fish Shellfish Immunol. 2021;117:188–191. doi: 10.1016/j.fsi.2021.08.002. [DOI] [PubMed] [Google Scholar]
- 33.Jiang J., Wu X., Zhou X., Feng L., Liu Y., Jiang W., Wu P., Zhao Y. Effects of dietary curcumin supplementation on growth performance, intestinal digestive enzyme activities and antioxidant capacity of crucian carp Carassius auratus. Aquaculture. 2016;463:174–180. [Google Scholar]
- 34.Folwarczna J. In: Nutrition and Diet in Menopause. Hollins Martin C.J., Watson R.R., Preedy V.R., editors. Humana Press; Totowa, NJ: 2013. Curcumin and its potential effects on the development of postmenopausal osteoporosis; pp. 165–180. [Google Scholar]
- 35.Hosseini-Vashan S., Golian A., Yaghobfar A., Zarban A., Afzali N., Esmaeilinasab P. Antioxidant status, immune system, blood metabolites and carcass characteristic of broiler chickens fed turmeric rhizome powder under heat stress. 2012;11:16118–16125. [Google Scholar]
- 36.Manzanilla E.G., Nofrarías M., Anguita M., Castillo M., Perez J.F., Martín-Orúe S.M., Kamel C., Gasa J. Effects of butyrate, avilamycin, and a plant extract combination on the intestinal equilibrium of early-weaned pigs1. J. Anim. Sci. 2006;84:2743–2751. doi: 10.2527/jas.2005-509. [DOI] [PubMed] [Google Scholar]
- 37.Caballero M.J., Izquierdo M.S., Kjørsvik E., Montero D., Socorro J., Fernández A.J., Rosenlund G. Morphological aspects of intestinal cells from gilthead seabream (Sparus aurata) fed diets containing different lipid sources. Aquaculture. 2003;225:325–340. [Google Scholar]
- 38.Pereira-Fantini P.M., Thomas S.L., Taylor R.G., Nagy E., Sourial M., Fuller P.J., Bines J.E. Colostrum supplementation restores insulin-like growth factor -1 levels and alters muscle morphology following massive small bowel resection. Jpen.-Parenter. Enter. 2008;32:266–275. doi: 10.1177/0148607108316197. [DOI] [PubMed] [Google Scholar]
- 39.Yang J., Hong J., Fu Z., Ma Z. Effects of dietary curcumin on growth and digestive physiology of Seriola dumerili. Front. Mar. Sci. 2022;9 [Google Scholar]
- 40.Abreu M.T., Fukata M., Arditi M. TLR signaling in the gut in health and disease. J. Immunol. 2005;174:4453. doi: 10.4049/jimmunol.174.8.4453. [DOI] [PubMed] [Google Scholar]
- 41.Saurabh S., Sahoo P.K. Lysozyme: an important defence molecule of fish innate immune system. Aquacult. Res. 2008;39:223–239. [Google Scholar]
- 42.Pinoni S.A., López Mañanes A.A. Alkaline phosphatase activity sensitive to environmental salinity and dopamine in muscle of the euryhaline crab Cyrtograpsus angulatus. J. Exp. Mar. Biol. Ecol. 2004;307:35–46. [Google Scholar]
- 43.Pipe R.K. Hydrolytic enzymes associated with the granular haemocytes of the marine mussel Mytilus edulis. Histochem. J. 1990;22:595–603. doi: 10.1007/BF01072941. [DOI] [PubMed] [Google Scholar]
- 44.Lien C. Lysozymes in the animal kingdom. J. Biosci. (Tueb.) 2010;35 doi: 10.1007/s12038-010-0015-5. [DOI] [PubMed] [Google Scholar]
- 45.Mahmoud H.K., Al-Sagheer A.A., Reda F.M., Mahgoub S.A., Ayyat M.S. Dietary curcumin supplement influence on growth, immunity, antioxidant status, and resistance to Aeromonas hydrophila in Oreochromis niloticus. Aquaculture. 2017;475:16–23. [Google Scholar]
- 46.Namitosh T., D D., Rashmi S. Curcumin inhibits paraquat induced lung inflammation and fibrosis by extracellular matrix modifications in mouse model. Inflammopharmacology. 2016;24 doi: 10.1007/s10787-016-0286-z. [DOI] [PubMed] [Google Scholar]
- 47.Baldissera M.D., Souza C.F., Zeppenfeld C.C., Descovi S., Machado V.S., Santos R.C.V., Baldisserotto B. Efficacy of dietary curcumin supplementation as bactericidal for silver catfish against Streptococcus agalactiae. Microb. Pathog. 2018;116 doi: 10.1016/j.micpath.2018.01.044. [DOI] [PubMed] [Google Scholar]
- 48.Carroll M.C. The role of complement and complement receptors in induction and regulation of immunity. Annu. Rev. Immunol. 1998;16 doi: 10.1146/annurev.immunol.16.1.545. [DOI] [PubMed] [Google Scholar]
- 49.Dodds M.W., Law S.K.A. The phylogeny and evolution of the thioester bond-containing proteins C3, C4 and α2–macroglobulin. Immunol. Rev. 1998:166. doi: 10.1111/j.1600-065x.1998.tb01249.x. [DOI] [PubMed] [Google Scholar]
- 50.Fearon D.T., Carter R.H. The CD19/CR2/TAPA-1 complex of B lymphocytes: linking natural to acquired immunity. Annu. Rev. Immunol. 1995;13 doi: 10.1146/annurev.iy.13.040195.001015. [DOI] [PubMed] [Google Scholar]
- 51.M W., L M.B., M B.C. Immune responses of teleost fish. Aust. Vet. J. 2001;79 doi: 10.1111/j.1751-0813.2001.tb10753.x. [DOI] [PubMed] [Google Scholar]
- 52.Savan R., Sakai M. Genomics of fish cytokines. Comp. Biochem. Physiol. - Part D: Genom. Proteonomi. 2005;1 doi: 10.1016/j.cbd.2005.08.005. [DOI] [PubMed] [Google Scholar]
- 53.Jiawei H., Shengjie Z., Gang Y., Chuanxin Q., Tao Z., Zhenhua M. Effects of transporting stress on the immune responses of Asian seabass Lates calcarifer fry. Aquacult. Res. 2020:52. [Google Scholar]
- 54.Bémeur C., Qu H., Desjardins P., Butterworth R.F. IL-1 or TNF receptor gene deletion delays onset of encephalopathy and attenuates brain edema in experimental acute liver failure. Neurochem. Int. 2009;56 doi: 10.1016/j.neuint.2009.11.010. [DOI] [PubMed] [Google Scholar]
- 55.S B., J Z., J S.C. Advances in fish cytokine biology give clues to the evolution of a complex network. Curr. Pharmaceut. Des. 2006;12 doi: 10.2174/138161206777947434. [DOI] [PubMed] [Google Scholar]
- 56.Secombes C.J., Wang T., Hong S., Peddie S., Crampe M., Laing K.J., Cunningham C., Zou J. Cytokines and innate immunity of fish. Dev. Comp. Immunol. 2001;25 doi: 10.1016/s0145-305x(01)00032-5. [DOI] [PubMed] [Google Scholar]
- 57.Cao L., Ding W., Du J., Jia R., Liu Y., Zhao C., Shen Y., Yin G. Effects of curcumin on antioxidative activities and cytokine production in Jian carp (Cyprinus carpio var. Jian) with CCl4-induced liver damage. Fish Shellfish Immunol. 2015;43:150–157. doi: 10.1016/j.fsi.2014.12.025. [DOI] [PubMed] [Google Scholar]
- 58.Mahmoud A., R. F.M., A. A.S., O. D.M.A., S. E.S., Kuldeep D. Curcumin and its different forms: a review on fish nutrition. Aquaculture. 2020:532. [Google Scholar]
- 59.Amit A., K T.B., M R.M.S., S R.D., K T.A., B A.B. Synthesis, characterization and in vitro anticancer activity of C-5 curcumin analogues with potential to inhibit TNF-α-induced NF-κB activation. BioMed Res. Int. 2014;2014 doi: 10.1155/2014/524161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Akdemir F., Orhan C., Tuzcu M., Sahin N., Juturu V., Sahin K. The efficacy of dietary curcumin on growth performance, lipid peroxidation and hepatic transcription factors in rainbow trout Oncorhynchus Mykiss (Walbaum) reared under different stocking densities. Aquacult. Res. 2017;48 [Google Scholar]
- 61.Ikram M., Saeed K., Khan A., Muhammad T., Khan M.S., Jo M.G., Rehman S.U., Kim M.O. Natural dietary supplementation of curcumin protects mice brains against ethanol-induced oxidative stress-mediated neurodegeneration and memory impairment via nrf2/TLR4/RAGE signaling. Nutrients. 2019;11(1082) doi: 10.3390/nu11051082. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 62.Song-Lin C., Wei L., Liang M., Zhen-Xia S., Zhi-Jian W., Guo-Cheng R. Molecular cloning and expression analysis of a hepcidin antimicrobial peptide gene from turbot (Scophthalmus maximus) Fish Shellfish Immunol. 2007;22 doi: 10.1016/j.fsi.2006.04.004. [DOI] [PubMed] [Google Scholar]
- 63.Cuesta A., Meseguer J., Esteban M.Á. The antimicrobial peptide hepcidin exerts an important role in the innate immunity against bacteria in the bony fish gilthead seabream. Mol. Immunol. 2007;45 doi: 10.1016/j.molimm.2007.11.007. [DOI] [PubMed] [Google Scholar]
- 64.Lehrer R.I., Ganz T. Antimicrobial peptides in mammalian and insect host defence. Curr. Opin. Immunol. 1999;11 doi: 10.1016/s0952-7915(99)80005-3. [DOI] [PubMed] [Google Scholar]
- 65.Yong-An Z., Irene S., Jun L., David P., Sarah B., Zhen X., E L.S., Jerri B., Oriol S.J. IgT, a primitive immunoglobulin class specialized in mucosal immunity. Nat. Immunol. 2010;11 doi: 10.1038/ni.1913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Haller O., Kochs G. Interferon-induced Mx proteins: dynamin-like GTPases with antiviral activity. Traffic. 2002;3:710–717. doi: 10.1034/j.1600-0854.2002.31003.x. [DOI] [PubMed] [Google Scholar]
- 67.Wilson R.C.H. Acute toxicity of spent sulphite liquor to atlantic salmon (Salmo salar) J. Fisher. Board Canada. 1972;29 [Google Scholar]
- 68.Wang W., Zhou J., Wang P., Tian T., Zheng Y., Liu Y., Mai W., Wang A. Oxidative stress, DNA damage and antioxidant enzyme gene expression in the Pacific white shrimp, Litopenaeus vannamei when exposed to acute pH stress. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009;150:428–435. doi: 10.1016/j.cbpc.2009.06.010. [DOI] [PubMed] [Google Scholar]
- 69.W R.S., A D.D., S K., L C.B. Physiological (antioxidant) responses of estuarine fishes to variability in dissolved oxygen. Comp. Biochem. Physiol. Toxicol. Pharmacol. : CB (Curr. Biol.) 2001:130. doi: 10.1016/s1532-0456(01)00243-5. [DOI] [PubMed] [Google Scholar]
- 70.Francisca S., J M.L., Braga F.S., João P.M., C C.L.F., Isabel C., de Almeida Ozório Rodrigo Otávio, Antunes M.F., Magalhães G.J.F. Dietary oil source and selenium supplementation modulate Fads2 and Elovl5 transcriptional levels in liver and brain of meagre (Argyrosomus regius) Lipids. 2016;51 doi: 10.1007/s11745-016-4157-6. [DOI] [PubMed] [Google Scholar]
- 71.Takahashi L.S., Biller-Takahashi J.D., Mansano C.F.M., Urbinati E.C., Gimbo R.Y., Saita M.V. Long-term organic selenium supplementation overcomes the trade-off between immune and antioxidant systems in pacu (Piaractus mesopotamicus) Fish Shellfish Immunol. 2017;60 doi: 10.1016/j.fsi.2016.11.060. [DOI] [PubMed] [Google Scholar]
- 72.Sykiotis G.P., Bohmann D. Keap1/Nrf2 signaling regulates oxidative stress tolerance and lifespan in Drosophila. Dev. Cell. 2008;14:76–85. doi: 10.1016/j.devcel.2007.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Motohashi H., Yamamoto M. Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol. Med. 2004;10:549–557. doi: 10.1016/j.molmed.2004.09.003. [DOI] [PubMed] [Google Scholar]
- 74.Heredia-Middleton P., Brunelli J., Drew R.E., Thorgaard G.H. Heat shock protein (HSP70) RNA expression differs among rainbow trout (Oncorhynchus mykiss) clonal lines. Comp. Biochem. Physiol., B. 2008:149. doi: 10.1016/j.cbpb.2007.05.012. [DOI] [PubMed] [Google Scholar]
- 75.E D.E., S W.N.Y. Cloning and characterization of the hsp70 multigene family from silver sea bream: modulated gene expression between warm and cold temperature acclimation. Biochem. Bioph. Res. Co. 2005:330. doi: 10.1016/j.bbrc.2005.03.039. [DOI] [PubMed] [Google Scholar]
- 76.Liu L., Peng J., Liu K., Yang H., Li Y., Hong H. Influence of cytochrome c on apoptosis induced by Anagrapha (Syngrapha) falcifera multiple nuclear polyhedrosis virus (AfMNPV) in insect Spodoptera litura cells. Cell Biol. Int. 2007;31 doi: 10.1016/j.cellbi.2007.03.011. [DOI] [PubMed] [Google Scholar]
- 77.Barros M.H., Netto L.E.S., Kowaltowski A.J. H2O2 generation in Saccharomyces cerevisiae respiratory pet mutants: effect of cytochrome c. Free Radical Biol. Med. 2003;35:179–188. doi: 10.1016/s0891-5849(03)00307-1. [DOI] [PubMed] [Google Scholar]
- 78.á. F.M., S. T., F. A., T. K., J. B., J. S., J. R., A. M., J. D., M. C., et al. Aquaculture; 2020. Improving greater amberjack (Seriola dumerili) Defenses dgainst Monogenean Parasite Neobenedenia Girellae infection through functional dietary additives. [Google Scholar]
- 79.Fernández-Montero Á., Torrecillas S., Izquierdo M., Caballero M.J., Milne D.J., Secombes C.J., Sweetman J., Silva P.D., Acosta F., Montero D. Increased parasite resistance of greater amberjack (Seriola dumerili Risso 1810) juveniles fed a cMOS supplemented diet is associated with upregulation of a discrete set of immune genes in mucosal tissues. Fish Shellfish Immunol. 2018;86 doi: 10.1016/j.fsi.2018.10.034. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be made avasilable on request.