Abstract
Study Objectives:
To test the hypothesis that children taking trazodone have less leg movements during sleep (LMS) and higher rapid eye movement (REM) sleep atonia than children taking selective serotonin reuptake inhibitors (SSRIs) but more than normal controls.
Methods:
Fifteen children (9 girls and 6 boys, mean age 11.7 years, standard deviation [SD] 3.42) taking trazodone (median dosage 50 mg/d, range 25–200 mg) for insomnia and 19 children (11 girls and 8 boys, mean age 13.7 years, SD 3.07) taking SSRIs for depression, anxiety, or both were consecutively recruited, as well as an age- and sex-matched group of 25 control children (17 girls and 8 boys, mean age 13.7 years, SD 3.11). LMS were scored and a series of parameters was calculated, along with the analysis of their time structure. The Atonia Index was then computed for each non-REM sleep stage and for REM sleep.
Results:
Children taking trazodone exhibited slightly higher leg movement indices than controls but lower than those found in children taking SSRIs and their time structure was different. Chin electromyogram atonia in all sleep stages was not significantly altered in children taking trazodone but was decreased in children taking SSRIs, especially during non-REM sleep.
Conclusions:
In children, SSRIs but not trazodone are associated with a significantly increased number of LMS, including periodic LMS, and increased chin tone in all sleep stages. The assessment of periodic limb movement disorder and REM sleep without atonia might not be accurate when children are taking SSRIs because of their significant impact.
Citation:
DelRosso LM, Mogavero MP, Bruni O, Schenck CH, Fickenscher A, Ferri R. Trazodone affects periodic leg movements and chin muscle tone during sleep less than selective serotonin reuptake inhibitor antidepressants in children. J Clin Sleep Med. 2022;18(12):2829–2836.
Keywords: periodic leg movements during sleep, leg movement activity during sleep, REM sleep without atonia, atonia index, chin EMG tone, trazodone, SSRI antidepressants, children
BRIEF SUMMARY
Current Knowledge/Study Rationale: Antidepressants, such as selective serotonin reuptake inhibitors (SSRIs) and trazodone, are often used also in children. As already reported for adults, SSRIs can decrease rapid eye movement (REM) sleep atonia and increase leg movements during sleep (LMS) also in children. With this study, we show that this is not true for trazodone, which showed a much less important impact than SSRIs on LMS and a negligible effect on muscle atonia in all sleep stages.
Study Impact: These findings have important practical implications and suggest that the assessment of periodic limb movement disorder or REM sleep without atonia should be made with particular attention in children taking SSRIs but not in children taking trazodone.
INTRODUCTION
Antidepressants are a diverse group of medications used mainly to treat major depressive disorder but also commonly used to treat generalized anxiety disorder, obsessive-compulsive disorder, bulimia, and post-traumatic stress disorder. Antidepressants are commonly used in children and the prevalence of major depressive disorder in children aged 3–17 years is 3.2%, while the prevalence of anxiety is 7.2%.1 Comorbid depression and anxiety are also common, with 3 out of 4 children with depression having comorbid anxiety while 1 in 3 children with anxiety are diagnosed with comorbid depression.1 Selective serotonin reuptake inhibitors (SSRIs) are the most frequently used antidepressants in children,2 especially fluoxetine, but other SSRIs include escitalopram and fluvoxamine.2
Trazodone is an antidepressant usually used as second-line agent. Trazodone belongs to the category of serotonin (5-HT) receptor antagonists and reuptake inhibitors (SARIs) that has antagonistic properties to serotonin type 2 receptors (5-HT2) and α-adrenoreceptors, as well as inhibitory properties to 5-HT reuptake, which play a role in its antidepressant and anxiolytic effects.3 Although the mechanism of action of trazodone is not completely understood, antagonism of 5-HT2 receptors seems to exert the most potent pharmacological effect of trazodone, in contrast to SSRIs, especially fluoxetine, for which the most potent pharmacological action is the inhibition of 5-HT uptake.4 Indeed, as an example, the inhibition of 5-HT uptake by trazodone is more than an order of magnitude less potent than that of fluoxetine.4 Due to its sedative properties, trazodone is also often used off label in cases of insomnia.5
Approximately 8.3% of children with a psychiatric disorder are evaluated in sleep clinics and often require a sleep study.6 Current guidelines for polysomnography (PSG) in children do not recommend tapering off antidepressants prior to a sleep study. Therefore, it is of utmost importance to understand the impact that antidepressants have in sleep and sleep architecture. In prior studies we have demonstrated that SSRI antidepressants can affect the polysomnographic sleep findings, rapid eye movement (REM) sleep atonia,7 and periodic leg movements during sleep in children.8
In this study we aimed to (1) to evaluate the characteristics of leg movements during sleep (LMS) in children taking trazodone, (2) to compare LMS in children taking trazodone with those of children taking SSRIs and controls, and (3) to specifically assess and compare the REM sleep atonia in the same groups of individuals. Based on our previous findings with SSRIs7,8 and on the differences in mechanisms of actions between them and trazodone,4 we hypothesized that children taking trazodone have less leg movement activity during sleep and higher REM sleep atonia than children taking SSRIs but more leg movements and less REM sleep atonia than normal controls.
METHODS
Subjects
For this study, a convenience sample of 15 children (9 girls and 6 boys, mean age 11.7 years, standard deviation [SD] 3.42) taking trazodone (median dosage 50 mg/d, range 25–200 mg) for insomnia and 19 children (11 girls and 8 boys, mean age 13.7 years, SD 3.07) taking SSRIs for depression, anxiety, or both were consecutively recruited at the Seattle Children’s Hospital, Seattle, Washington. None of the children had another medical or psychiatric disorder. PSG recording had been ordered because of snoring, fatigue, restless sleep, or sleepiness. From the group of children taking trazodone, 8 were initiated on trazodone by a sleep provider (5 of whom for sleep maintenance insomnia and 3 for sleep-onset insomnia) and 7 were initiated on trazodone by a primary care physician. Eight of the children on SSRIs were taking sertraline (75–200 mg/day), 4 were taking escitalopram and 1 was taking citalopram (10–30 mg/day), 5 were taking fluoxetine (10–30 mg/day), and 1 was taking duloxetine (30 mg/day). Children taking SSRIs were only taking 1 SSRI and were not taking a combination of other medications. Three children only took the SSRI in the evening and the rest took it in the morning. All children taking trazodone took it in the evening. Treatment duration ranged between 2 and 12 months. None of the children in these groups was diagnosed with restless legs syndrome.
Finally, an age- and sex-matched group of 25 control children (17 girls and 8 boys, mean age 13.7 years, SD 3.11) was selected from our database (including children recruited by the authors at their respective centers, who have been successfully used for previous studies9). None of these children were taking drugs or were affected by another medical, psychiatric, or sleep disorder other than snoring. In all 3 groups, physical and neurological examinations were done.
The body mass index or sex composition and age of the groups were not significantly different (chi-square = 0.53, nonsignificant [NS]) and (Kruskal-Wallis analysis of variance [ANOVA] H2,59 = 3.966, NS), respectively. All children had an apnea-hypopnea index < 1 event/h. In addition, sleep apnea, hypoventilation, and hypoxemia were ruled out in each case, independently by 2 different sleep experts. No sample size/power analysis was possible because this was a convenience sample. The study was approved by the local ethics committee and all children or their parents/guardians had provided informed written consent.
Polysomnographic recording
All participants underwent a routine full-night PSG recording in the sleep laboratory, including electromyogram (EMG) of the submentalis and both tibialis anterior muscles and electrocardiogram. Sleep stages and arousals were visually scored following standard criteria.10
Scoring and quantification of leg movements during sleep
LMS was detected and scored according to the most recent criteria from the World Association of Sleep Medicine,11 followed by the calculation of a series of parameters including the following:
(1) Total LMS index, n/hour;
(2) Periodic limb movements of sleep (PLMS) index, n/hour, LMS included in regular and noninterrupted sequences of at least 4 with onset-to-onset inter-movement interval (IMI) 10–90 seconds;
(3) PLMS/arousal index, n/hour, an arousal event and a PLMS event were considered to be associated with each other when there was less than 0.5 seconds between the end of 1 event and the onset of the other event, regardless of which was first;
(4) Short-interval LMS (SILMS) index, n/hour, LMS with preceding IMI < 10 seconds;
(5) Isolated LMS (ISOLMS) index, n/hour, LMS with IMI > 90 seconds and LMS with IMI 10–90 seconds not meeting all the criteria for PLMS;
(6) Percentage of bilateral PLMS, PLMS formed by 2 to 4 monolateral LMS from the 2 legs overlapping each other within 0.5-second windows with a combined total duration < 15 seconds;
(7) Periodicity Index, PLMS to total LMS ratio;
(8) PLMS duration, seconds;
(9) SILMS duration, seconds;
(10) ISOLMS duration, seconds;
(11) PLMS index in REM sleep, n/hour; and
(12) PLMS index in non-REM (NREM) sleep, n/hour.
All sleep leg movement onset-to-onset IMIs, from each recording, were counted, in each participant, for 2-second classes (0.5 < IMI ≤ 2, 2 < IMI ≤ 4, 4 < IMI ≤ 6, …, 98 < IMI ≤ 100) and group grand averages were obtained, which were used for statistical analysis. Finally, hourly, night-distribution histograms of the number of PLMS, during the first 8 recording hours, were obtained for each group of participants.
Quantification of the submentalis muscle EMG amplitude
We set a minimum amount of 15 minutes of REM sleep, in order to reliably evaluate chin EMG atonia in all recordings; however, none of the recordings was excluded for this reason. LMS were detected and scored according with the most recent criteria by the World Association of Sleep Medicine.11
Similarly to the method we have previously reported,12–14 the submentalis muscle EMG signal was digitally band-pass filtered at 10–100 Hz, with a notch filter at 50 Hz, and rectified; subsequently, all sleep epochs were included in the analysis and subdivided into 30 1-second mini-epochs. The average amplitude of the rectified submentalis muscle EMG signal was then obtained for each mini-epoch. Then, a noise reduction method was implemented13 by subtracting from the average rectified EMG amplitude in each mini-epoch the minimum value found in a moving window of ±30 mini-epochs (assuming that the minimum value in this 60-second period is a good estimate of the local level of noise affecting all sampled signal values in such a period). The values of the average noise-reduced EMG signal amplitude in each mini-epoch were used to draw distribution histograms of the percentage of values in the following 20 amplitude (amp) classes (expressed in μV): amp ≤ 1, 1 < amp ≤ 2, …, 18 < amp ≤ 19, amp ≥ 20. The Atonia Index was then computed, for each sleep stage, as follows:
which reflects the degree of preponderance of the first column in these graphs (amp ≤ 1) in relation to the cumulative value of all columns with amp > 2 and amp ≤ 19 (the column 1 < amp ≤ 2 is not taken into account because it might reflect both atonia and EMG activation). Mathematically, this index can vary from 0 (absence of mini-epochs with amp ≤ 1), which means complete absence of EMG atonia, to 1 (all mini-epochs with amp ≤ 1) or continuous stable EMG atonia during each sleep stage.
Subsequently, we also counted all sequences of consecutive mini-epochs exceeding the value of 2 μV (which we will call “EMG activations”) and calculated their number per hour of sleep.
Statistical analysis
The nonparametric Kruskal-Wallis ANOVA was used for between-group comparisons, followed by post hoc comparisons by the Mann-Whitney test. Frequencies were compared with the chi-square test. The slope and significance of the number of PLMS per hour of the night recording were analyzed with the Pearson’s linear regression coefficient. Following the indications by Cohen,15 we considered correlations 0.10, 0.30, and 0.50 as corresponding to small, medium, and large sizes, respectively.
The commercially available software STATISTICA, version 6 (StatSoft, Inc Tulsa, OK), was used for the statistical analysis, and the significance level was set at P < .05.
RESULTS
Table 1 shows the comparison between sleep staging parameters found in the 3 groups of children. Children taking SSRIs had longer sleep latency and higher number of stage shifts than both controls and children taking trazodone; in addition, they had a higher number of awakenings and decreased sleep efficiency and sleep stage N2, with respect to controls only. Arousals were also highest in children taking SSRIs and lowest in controls. Children taking trazodone differed significantly from controls only in their higher number of stage shifts and their reduced NREM stage 2 percentage. Of note, NREM sleep stage 3 was highest in children on trazodone.
Table 1.
Sleep parameters found in the 3 groups of participants.
| 1. Controls (n = 25) | 2. Trazodone (n = 15) | 3. SSRIs (n = 19) | Kruskal-Wallis ANOVA | Mann-Whitney Test, P < | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | H2,59 | P < | 1 vs 2 | 2 vs 3 | 1 vs 3 | |
| Time in bed, min | 509.6 | 57.84 | 524.8 | 29.03 | 520.0 | 40.24 | 2.728 | NS | |||
| Sleep period time, min | 477.2 | 48.63 | 482.0 | 49.02 | 473.0 | 41.15 | 1.384 | NS | |||
| Total sleep time, min | 442.2 | 66.41 | 424.6 | 67.69 | 396.7 | 78.79 | 4.086 | NS | |||
| Sleep latency, min | 19.3 | 17.12 | 40.2 | 50.49 | 47.2 | 26.26 | 12.839 | 0.0016 | NS | NS | .00065 |
| Stage R latency, min | 129.8 | 66.81 | 165.6 | 121.91 | 143.3 | 94.11 | 0.601 | NS | |||
| Stage shifts/h | 7.6 | 3.93 | 11.6 | 7.06 | 15.2 | 6.27 | 17.497 | .0002 | .025 | .048 | .00008 |
| Awakenings/h | 2.3 | 2.24 | 3.4 | 3.39 | 5.5 | 3.86 | 9.936 | .007 | NS | NS | .0033 |
| Sleep efficiency, % | 86.9 | 10.68 | 81.0 | 12.63 | 76.2 | 13.13 | 9.137 | .01 | NS | NS | .004 |
| Stage W, % | 7.4 | 10.29 | 11.6 | 12.83 | 16.5 | 12.65 | 5.682 | NS | |||
| Stage N1, % | 5.8 | 3.18 | 6.8 | 5.53 | 10.2 | 9.45 | 2.706 | NS | |||
| Stage N2, % | 48.3 | 9.16 | 38.9 | 11.55 | 39.4 | 10.17 | 10.479 | .0053 | .016 | NS | .003 |
| Stage N3, % | 21.5 | 5.24 | 26.4 | 10.28 | 20.1 | 7.78 | 3.390 | NS | |||
| Stage R, % | 17.0 | 5.67 | 16.4 | 5.69 | 13.8 | 6.95 | 2.160 | NS | |||
| Arousals/h | 6.1 | 4.88 | 8.5 | 6.07 | 18.4 | 13.40 | 20.815 | .00001 | NS | .0055 | .000006 |
ANOVA = analysis of variance, NS = nonsignificant, SD = standard deviation, SSRI = selective serotonin reuptake inhibitor.
LMS parameters of the same groups are compared in Table 2; children taking trazodone showed PLMS (during both NREM and REM sleep), PLMS/arousal, total LMS, SILMS, and ISOLMS indexes intermediate between those controls (lowest) and children taking SSRIs (highest). Also, the Periodicity Index was highest in children on SSRIs, followed by those taking trazodone and then by controls. The post hoc between-group comparisons showed that children taking trazodone differed from controls only in their higher total LMS and SILMS index, while children taking SSRIs had higher total LMS, PLMS, PLMS/arousal, and Periodicity Indexes than both children taking trazodone and controls.
Table 2.
Leg movement activity during sleep parameters found in the 3 groups of participants.
| 1. Controls (n = 25) | 2. Trazodone (n = 15) | 3. SSRIs (n = 19) | Kruskal-Wallis ANOVA | Mann-Whitney Test, P < | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | H2,59 | P < | 1 vs 2 | 2 vs 3 | 1 vs 3 | |
| Total LMS number | 89.0 | 42.93 | 167.4 | 118.61 | 246.5 | 141.41 | 23.577 | .00001 | NS | NS | .00001 |
| PLMS number | 9.6 | 10.10 | 33.8 | 46.81 | 86.8 | 95.08 | 29.512 | .00001 | NS | .002 | .00001 |
| SILMS number | 20.9 | 17.36 | 46.1 | 40.83 | 53.4 | 45.20 | 9.375 | .0092 | NS | NS | .0024 |
| ISOLMS number | 58.4 | 23.33 | 87.5 | 55.58 | 106.5 | 49.09 | 13.403 | .0012 | NS | NS | .00015 |
| Total LMS index, n/h | 12.2 | 5.87 | 23.0 | 16.21 | 39.6 | 27.62 | 28.207 | .00001 | .017 | .0093 | .00001 |
| PLMS index, n/h | 1.4 | 1.40 | 4.5 | 6.17 | 14.3 | 19.01 | 31.202 | .00001 | NS | .0008 | .00001 |
| NREM PLMS index, n/h | 1.4 | 1.60 | 4.9 | 6.86 | 15.9 | 21.30 | 30.090 | .00001 | NS | NS | .00001 |
| REM PLMS index, n/h | 0.8 | 1.93 | 2.3 | 4.49 | 4.6 | 5.93 | 9.598 | .008 | NS | .001 | .012 |
| PLMS/arousal index, n/h | 0.1 | 0.25 | 0.5 | 0.85 | 1.4 | 1.53 | 22.582 | .00001 | NS | .002 | .00001 |
| SILMS index, n/h | 2.9 | 2.29 | 6.4 | 5.42 | 8.7 | 7.93 | 12.014 | .0025 | .033 | NS | .001 |
| ISOLMS index, n/h | 8.0 | 3.18 | 12.2 | 7.57 | 16.6 | 8.01 | 17.816 | .0001 | NS | NS | .000021 |
| Periodicity index | 0.1 | 0.09 | 0.2 | 0.19 | 0.4 | 0.18 | 24.164 | .00001 | NS | .001 | .000002 |
| PLMS duration, s | 2.8 | 1.08 | 2.6 | 0.97 | 2.8 | 0.87 | 0.193 | NS | |||
| SILMS duration, s | 2.6 | 0.88 | 2.7 | 0.92 | 3.1 | 0.91 | 3.199 | NS | |||
| ISOLMS duration, s | 3.1 | 0.83 | 3.0 | 1.15 | 3.5 | 1.05 | 2.409 | NS | |||
ANOVA = analysis of variance, ISOLMS = isolated leg movements during sleep, LMS = leg movements during sleep, NREM = non–rapid eye movement, NS = nonsignificant, PLMS = periodic leg movements during sleep, REM = rapid eye movement, SD = standard deviation, SILMS = short-interval leg movements during sleep, SSRI = selective serotonin reuptake inhibitor.
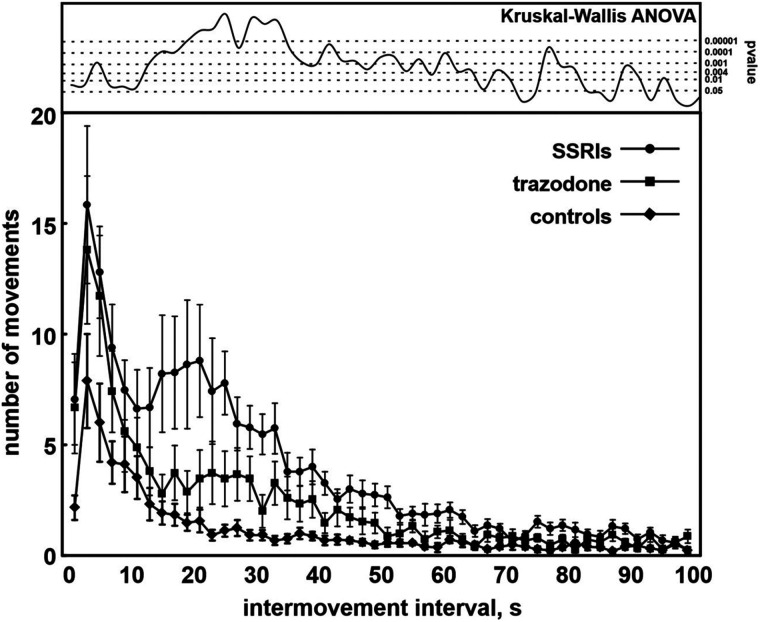
The LMS IMI distribution graphs (Figure 1) showed the presence, in children taking SSRIs, of 2 well-defined peaks, the first between 2 and 10 seconds, with a maximum at around 4 seconds, and the second, corresponding to PLMS, between approximately 10 and 60 seconds, with a maximum at around 20–22 seconds. This second peak was much less evident in children taking trazodone and not detectable at all in controls, while the peak at around 4 seconds was very similar in children taking SSRIs and those taking trazodone, but lower in controls. The differences between the groups were generally highly significant in correspondence to the 10–60-second peak.
Figure 1. Distribution of intervals between consecutive leg movements during sleep in children taking trazodone or SSRIs and controls.
Data are shown as mean (circles, squares, and diamonds) and standard errors (whiskers). The H2,59 values obtained with the Kruskal-Wallis ANOVA, comparing the 3 groups and computed for all points of the graphs, are shown at the top, along with their significance level (P value). ANOVA = analysis of variance, SSRI = selective serotonin reuptake inhibitor.
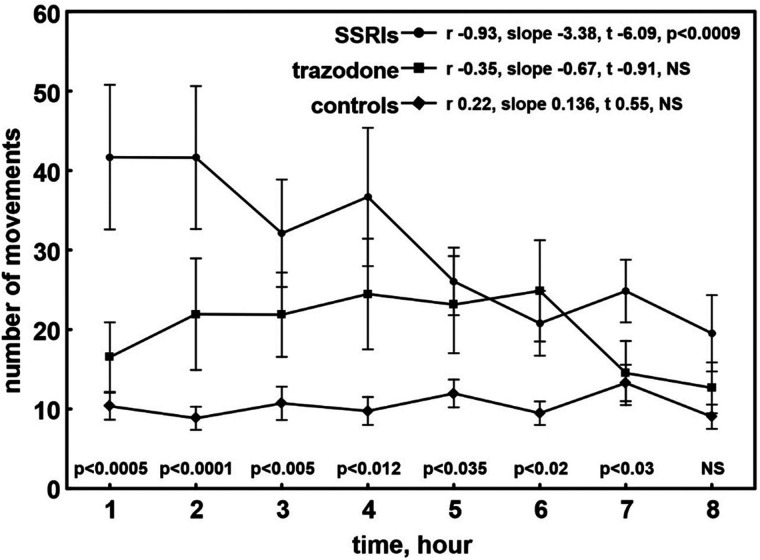
The hourly distribution of PLMS (Figure 2) showed a decrease during the night in children taking SSRIs only, with a statistically significant slope; children taking trazodone had a generally higher number of PLMS than both controls, throughout the night, but without significant variations along the night. The Pearson correlation coefficient of the decrease in children taking SSRIs was large, significantly higher than that of children taking trazodone (z = –3.39, P < .0007) or controls (z = –5.73, P < .000001); there was no significant difference between children taking trazodone and controls for this comparison (z = 1.64, NS).
Figure 2. Number of PLMS recorded during the first 8 hours of sleep in children taking trazodone or SSRIs and controls.
Data are shown as mean (circles, squares, and diamonds) and standard errors (whiskers). The P values obtained with the Kruskal-Wallis ANOVA, comparing the 3 groups and computed for all points of the graphs, are also shown. ANOVA = analysis of variance, NS = nonsignificant, PLMS = periodic limb movements of sleep, SSRI = selective serotonin reuptake inhibitor.
The parameters related to the chin EMG tone during sleep found in controls, participants taking SSRIs, and those taking trazodone are reported in Table 3. Atonia Index in all sleep stages was not significantly different between children taking trazodone and controls, or the number of chin muscle EMG activations. On the contrary, Atonia Index was found to be reduced in all sleep stages in children taking SSRIs, reaching statistical significance during NREM sleep stages N2 and N3 with respect to both children taking trazodone and controls. Similar results were found for the increase in the number of EMG activations for which, however, a statistically significant difference was found during NREM sleep stages N1 (with respect to both children taking trazodone and controls) and N2 (with respect to only children taking trazodone).
Table 3.
Chin EMG tone during sleep parameters found in the 3 groups of participants.
| 1. Controls (n = 25) | 2. Trazodone (n = 15) | 3. SSRIs (n = 19) | Kruskal-Wallis ANOVA | Mann-Whitney test, P < | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | H2,59 | P < | 1 vs 2 | 2 vs 3 | 1 vs 3 | |
| Atonia Index, R | 0.922 | 0.053 | 0.899 | 0.120 | 0.878 | 0.099 | 2.728 | NS | |||
| Atonia Index, N1 | 0.567 | 0.257 | 0.598 | 0.214 | 0.410 | 0.252 | 5.878 | NS | |||
| Atonia Index, N2 | 0.763 | 0.158 | 0.818 | 0.173 | 0.559 | 0.281 | 8.831 | .012 | NS | .013 | .025 |
| Atonia Index, N3 | 0.826 | 0.160 | 0.856 | 0.160 | 0.588 | 0.332 | 6.789 | .034 | NS | .023 | .034 |
| EMG activations, R | 66.7 | 37.64 | 78.3 | 57.27 | 100.9 | 68.63 | 2.462 | NS | |||
| EMG activations, N1 | 204.7 | 92.76 | 203.7 | 105.94 | 286.3 | 94.12 | 10.100 | .006 | NS | .009 | .006 |
| EMG activations, N2 | 172.8 | 109.54 | 136.7 | 100.58 | 246.2 | 134.96 | 6.535 | .038 | NS | .017 | NS |
| EMG activations, N3 | 149.1 | 135.23 | 140.3 | 155.51 | 272.9 | 202.67 | 5.835 | NS | |||
ANOVA = analysis of variance, EMG = electromyogram, NS = nonsignificant, SD = standard deviation, SSRI = selective serotonin reuptake inhibitor.
DISCUSSION
The findings of this study support our initial hypothesis that children taking trazodone exhibit higher leg movement indices than controls but lower than those found in children taking SSRIs, without any clinical correlate. This might be explained by the different sets of neurotransmitters influenced by trazodone and SSRIs. Although the full mechanism of action of trazodone on neurotransmission is not fully understood, it is generally accepted that it blocks histaminergic and alpha-1-adrenergic receptors but also inhibits the reuptake of serotonin, although to a lesser degree than SSRIs. In adults, at lower doses (25–100 mg), trazodone usually acts on the 5-HT2A, alpha-1 and H1 receptors (hypnotic effect), while at higher doses (150–600 mg) blocking of 5-HT2A and serotonin transporter occurs (antidepressant effect).16 Our children were taking a median dose of trazodone 50 mg/d (only 4 adolescents out of the 15 children were taking > 50 mg/d: one, aged 15 years, 75 mg; two, both aged 15 years, 100 mg; and one, aged 16 years, 200 mg), which places them in the low-dose range, also considering their age.
This is, to our knowledge, the first study assessing LMS in children taking trazodone. Studies in adults with insomnia have been few and inconsistent, and have assessed only the acute effects of trazodone on leg movements during PSG.17,18 They have shown a reduction in periodic leg movement (PLM) index during wakefulness only, but not during sleep, in 1 study,18 while in a second study no distinction was made between PLM during wakefulness and sleep, but the authors reported a marginally significant reduction, which could have been due to the simple reduction of the wake time due to the hypnotic effect of trazodone.17 However, the International Restless Legs Syndrome Study Group (IRLSSG) PLMS scoring criteria were not used in these studies but nonstandard criteria set up by the same authors and automatically applied via a computerized analysis were used.17,18
Using the IRLSSG scoring criteria, we have clearly identified a significant difference in leg movement activity between children taking trazodone and children taking SSRIs. Children taking SSRIs show increased indices of PLMS, LMS, Periodicity Index, and PLMS with arousal; children taking trazodone only exhibit increased LMS and SILMS. This is very important when diagnosing periodic limb movement disorder (PLMD) in children taking antidepressants, allowing the diagnosis of PLMD only in the presence of genuine PLMS when trazodone is used and considering PLMS secondary to medication when SSRI antidepressants are taken. The IMI peak between 10 and 30 seconds seen in children taking SSRIs corroborates the dopaminergic-mediated mechanism underlying these PLMS since the increase in serotonin can increase dopaminergic inhibition.19 Dopaminergic-mediated PLMS have been demonstrated to exhibit an IMI of 10–90 seconds (10–40 seconds, in particular) and constitute the basis of the IRLSSG scoring criteria of PLMS.20
It should also be noted that children taking SSRIs showed a clear and significant decline in LMS during the night, another feature making their leg movement activity during sleep very similar to that of adult restless legs syndrome8,21,22 and also probably connected with the circadian changes in dopamine levels.23 On the contrary, the same decline was not observed in children taking trazodone. This further emphasizes the different effects of SSRIs and trazodone on LMS, which might be explained by their substantial different level of serotonin reuptake inhibition.
Also, the mechanism by which SSRIs affect sleep atonia can be postulated to be a serotonin-mediated decrease in monoaminergic inhibitory activity over the brainstem and spinal cord during sleep, especially REM sleep. We have previously shown that reduced sleep atonia seen in children taking SSRIs is comparable to the increased chin tone found in children with narcolepsy and involves all sleep stages, not only REM sleep.7 It is important to note that, in children, REM without atonia can be a marker of narcolepsy24 and it has also been reported in other neurodevelopmental or neurologic disorders.25 However, the mechanism by which SSRIs reduce chin atonia during sleep might be more complex or different from that of the reduction in REM sleep atonia in adult REM sleep behavior disorder (RBD) and in children or adults with RBD associated with narcolepsy, as suggested by the different patterns found by measuring the Atonia Index in the different sleep stages.7,26 Indeed, although the current understanding of the regulation of the muscle tone during sleep assigns a principal role to glutamatergic, glycinergic, and GABAergic neurons within the sublaterodorsal tegmental (or subceruleus) and the ventral gigantocellular reticular nucleus,27 serotonergic and dopaminergic pathways, as well as noradrenergic and hypocretinergic pathways, also seem to be able to influence motoneuron activity during sleep.28–31 Thus, the complexity of the muscle tone neurochemical regulation might explain the differential effects of antidepressants such as SSRIs and trazodone, which differentially impact the pathways underlying muscle tone during sleep.
Finally, regarding the PSG findings, compared with controls, children taking trazodone present with lower stage N2, higher stage N3, and prolonged sleep latency (probably connected with their insomnia and not completely reversed by trazodone). Studies in adults have demonstrated increased slow-wave sleep and decreased sleep stages N1 and N2.32,33 Conversely, children taking SSRIs have increased arousal index, increased awakenings, and decreased sleep efficiency, as also previously described8; this might lead to thinking that sleep disturbance was more important in these individuals and that this might have influenced, at least to some extent, the occurrence of a higher number of PLMS. Vice versa, it might be also speculated that more PLMS contribute to the sleep architecture involvement in individuals taking SSRIs. However, it has already been reported that PLMS and arousals are indeed correlated,34 but it is difficult to prove a direct cause-effect relationship between them. Dopamine agonists can inhibit PLMS very effectively but do not decrease sleep instability in restless legs syndrome; conversely, clonazepam significantly reduces sleep instability but does not modify PLMS.35 In addition, experimentally induced arousals are unable to trigger PLMS.36 These data seem to indicate that synchronization does occur between sleep instability and PLMS but the latter emerge essentially because of their own pathophysiological mechanisms.
Limitations to this study include the single-center design, limited number of patients, and inability to assess dose-dependent changes. While all patients taking trazodone were taking it at bedtime, only 3 out of 19 patients taking SSRIs were taking them at bedtime; the remaining patients were taking them in the morning. However, SSRIs have a much longer half-life, ranging from 12 hours for duloxetine to 82 hours for fluoxetine; thus, it can be inferred that their effects were still significant during the night.
In conclusion, our findings demonstrate that pediatric use of SSRIs but not trazodone is associated with a significantly increased number of LMS, including PLMS, that have dopaminergic features, and increased chin tone in all sleep stages. These findings have practical considerations for the assessment of PLMD and REM sleep without atonia; these do not seem to be affected by trazodone, but our findings indicate that diagnosis could be problematic in children taking SSRIs because of their significant impact. Finally, as previously advocated, the effects on REM atonia (and other sleep motor parameters) should be evaluated for all new antidepressant and other psychotropic medications, in both children and adults, for the reasons mentioned above.37
ABBREVIATIONS
- ANOVA
analysis of variance
- EMG
electromyogram
- IMI
inter-movement interval
- ISOLMS
isolated leg movements during sleep
- LMS
leg movements during sleep
- NS
nonsignificant
- PLMS
periodic limb movements of sleep
- PSG
polysomnography
- REM
rapid eye movement
- SD
standard deviation
- SILMS
short-interval leg movements during sleep
- SSRI
selective serotonin reuptake inhibitor
DISCLOSURE STATEMENT
All authors have seen and approved the manuscript. The authors report no conflicts of interest.
REFERENCES
- 1. Ghandour RM , Sherman LJ , Vladutiu CJ , et al . Prevalence and treatment of depression, anxiety, and conduct problems in US children . J Pediatr. 2019. ; 206 : 256 – 267.e3 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Dwyer JB , Bloch MH . Antidepressants for pediatric patients . Curr Psychiatr. 2019. ; 18 ( 9 ): 26 – 42F . [PMC free article] [PubMed] [Google Scholar]
- 3. Khouzam HR . A review of trazodone use in psychiatric and medical conditions . Postgrad Med. 2017. ; 129 ( 1 ): 140 – 148 . [DOI] [PubMed] [Google Scholar]
- 4. Marek GJ , McDougle CJ , Price LH , Seiden LS . A comparison of trazodone and fluoxetine: implications for a serotonergic mechanism of antidepressant action . Psychopharmacology (Berl). 1992. ; 109 ( 1-2 ): 2 – 11 . [DOI] [PubMed] [Google Scholar]
- 5. Oggianu L , Ke AB , Chetty M , et al . Estimation of an appropriate dose of trazodone for pediatric insomnia and the potential for a trazodone-atomoxetine interaction . CPT Pharmacometrics Syst Pharmacol. 2020. ; 9 ( 2 ): 77 – 86 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. DelRosso LM , Ferri R . The prevalence of restless sleep disorder among a clinical sample of children and adolescents referred to a sleep centre . J Sleep Res. 2019. ; 28 ( 6 ): e12870 . [DOI] [PubMed] [Google Scholar]
- 7. Ferri R , Mogavero MP , Bruni O , Plazzi G , Schenck CH , DelRosso LM . Increased chin muscle tone during all sleep stages in children taking SSRI antidepressants and in children with narcolepsy type 1 . Sleep. 2021. ; 44 : zsab147 . [DOI] [PubMed] [Google Scholar]
- 8. Ferri R , Mogavero MP , Bruni O , Picchietti DL , Kapoor V , Del Rosso LM . Leg movements during sleep in children treated with serotonergic antidepressants . Sleep. 2022. ; 45 ( 3 ): zsab236 . [DOI] [PubMed] [Google Scholar]
- 9. Ferri R , DelRosso LM , Aricò D , et al . Leg movement activity during sleep in school-age children and adolescents: a detailed study in normal controls and participants with restless legs syndrome and narcolepsy type 1 . Sleep. 2018. ; 41 ( 4 ): zsy010 . [DOI] [PubMed] [Google Scholar]
- 10. Berry RB , Quan SF , Abreu AR , et al. for the American Academy of Sleep Medicine . The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications. Version 2.6 . Darien, IL: : American Academy of Sleep Medicine; ; 2020. . [Google Scholar]
- 11. Ferri R , Fulda S , Allen RP , et al. International and European Restless Legs Syndrome Study Groups (IRLSSG and EURLSSG) . World Association of Sleep Medicine (WASM) 2016 standards for recording and scoring leg movements in polysomnograms developed by a joint task force from the International and the European Restless Legs Syndrome Study Groups (IRLSSG and EURLSSG) . Sleep Med. 2016. ; 26 : 86 – 95 . [DOI] [PubMed] [Google Scholar]
- 12. Ferri R , Manconi M , Plazzi G , et al . A quantitative statistical analysis of the submentalis muscle EMG amplitude during sleep in normal controls and patients with REM sleep behavior disorder . J Sleep Res. 2008. ; 17 ( 1 ): 89 – 100 . [DOI] [PubMed] [Google Scholar]
- 13. Ferri R , Rundo F , Manconi M , et al . Improved computation of the atonia index in normal controls and patients with REM sleep behavior disorder . Sleep Med. 2010. ; 11 ( 9 ): 947 – 949 . [DOI] [PubMed] [Google Scholar]
- 14. Ferri R , Bruni O , Fulda S , Zucconi M , Plazzi G . A quantitative analysis of the submentalis muscle electromyographic amplitude during rapid eye movement sleep across the lifespan . J Sleep Res. 2012. ; 21 ( 3 ): 257 – 263 . [DOI] [PubMed] [Google Scholar]
- 15. Cohen J . Statistical Power Analysis for the Behavioral Sciences. 2nd ed. Hillsdale, NJ: : Erlbaum; ; 1988. . [Google Scholar]
- 16. Bossini L , Casolaro I , Koukouna D , Cecchini F , Fagiolini A . Off-label uses of trazodone: a review . Expert Opin Pharmacother. 2012. ; 13 ( 12 ): 1707 – 1717 . [DOI] [PubMed] [Google Scholar]
- 17. Saletu B , Prause W , Anderer P , et al . Insomnia in somatoform pain disorder: sleep laboratory studies on differences to controls and acute effects of trazodone, evaluated by the Somnolyzer 24 x 7 and the Siesta database . Neuropsychobiology. 2005. ; 51 ( 3 ): 148 – 163 . [DOI] [PubMed] [Google Scholar]
- 18. Saletu-Zyhlarz GM , Abu-Bakr MH , Anderer P , et al . Insomnia related to dysthymia: polysomnographic and psychometric comparison with normal controls and acute therapeutic trials with trazodone . Neuropsychobiology. 2001. ; 44 ( 3 ): 139 – 149 . [DOI] [PubMed] [Google Scholar]
- 19. Hashemi P , Dankoski EC , Lama R , Wood KM , Takmakov P , Wightman RM . Brain dopamine and serotonin differ in regulation and its consequences . Proc Natl Acad Sci USA. 2012. ; 109 ( 29 ): 11510 – 11515 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Manconi M , Ferri R , Feroah TR , Zucconi M , Ferini-Strambi L . Defining the boundaries of the response of sleep leg movements to a single dose of dopamine agonist . Sleep. 2008. ; 31 ( 9 ): 1229 – 1237 . [PMC free article] [PubMed] [Google Scholar]
- 21. Ferri R , DelRosso LM , Silvani A , et al . Peculiar lifespan changes of periodic leg movements during sleep in restless legs syndrome . J Sleep Res. 2020. ; 29 ( 3 ): e12896 . [DOI] [PubMed] [Google Scholar]
- 22. Ferri R , Manconi M , Lanuzza B , et al . Age-related changes in periodic leg movements during sleep in patients with restless legs syndrome . Sleep Med. 2008. ; 9 ( 7 ): 790 – 798 . [DOI] [PubMed] [Google Scholar]
- 23. Clemens S , Rye D , Hochman S . Restless legs syndrome: revisiting the dopamine hypothesis from the spinal cord perspective . Neurology. 2006. ; 67 ( 1 ): 125 – 130 . [DOI] [PubMed] [Google Scholar]
- 24. Bin-Hasan S , Videnovic A , Maski K . Nocturnal REM sleep without atonia is a diagnostic biomarker of pediatric narcolepsy . J Clin Sleep Med. 2018. ; 14 ( 2 ): 245 – 252 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Lloyd R , Tippmann-Peikert M , Slocumb N , Kotagal S . Characteristics of REM sleep behavior disorder in childhood . J Clin Sleep Med. 2012. ; 8 ( 2 ): 127 – 131 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Ferri R , Zucconi M , Marelli S , Plazzi G , Schenck CH , Ferini-Strambi L . Effects of long-term use of clonazepam on nonrapid eye movement sleep patterns in rapid eye movement sleep behavior disorder . Sleep Med. 2013. ; 14 ( 5 ): 399 – 406 . [DOI] [PubMed] [Google Scholar]
- 27. Peever J , Luppi PH , Montplaisir J . Breakdown in REM sleep circuitry underlies REM sleep behavior disorder . Trends Neurosci. 2014. ; 37 ( 5 ): 279 – 288 . [DOI] [PubMed] [Google Scholar]
- 28. Lai YY , Kodama T , Siegel JM . Changes in monoamine release in the ventral horn and hypoglossal nucleus linked to pontine inhibition of muscle tone: an in vivo microdialysis study . J Neurosci. 2001. ; 21 ( 18 ): 7384 – 7391 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Burgess C , Lai D , Siegel J , Peever J . An endogenous glutamatergic drive onto somatic motoneurons contributes to the stereotypical pattern of muscle tone across the sleep-wake cycle . J Neurosci. 2008. ; 28 ( 18 ): 4649 – 4660 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Fenik VB , Davies RO , Kubin L . REM sleep-like atonia of hypoglossal (XII) motoneurons is caused by loss of noradrenergic and serotonergic inputs . Am J Respir Crit Care Med. 2005. ; 172 ( 10 ): 1322 – 1330 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Trulson ME , Jacobs BL , Morrison AR . Raphe unit activity during REM sleep in normal cats and in pontine lesioned cats displaying REM sleep without atonia . Brain Res. 1981. ; 226 ( 1-2 ): 75 – 91 . [DOI] [PubMed] [Google Scholar]
- 32. Yamadera H , Suzuki H , Nakamura S , Endo S . Effects of trazodone on polysomnography, blood concentration and core body temperature in healthy volunteers . Psychiatry Clin Neurosci. 1999. ; 53 ( 2 ): 189 – 191 . [DOI] [PubMed] [Google Scholar]
- 33. Roth AJ , McCall WV , Liguori A . Cognitive, psychomotor and polysomnographic effects of trazodone in primary insomniacs . J Sleep Res. 2011. ; 20 ( 4 ): 552 – 558 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Ferri R , Rundo F , Zucconi M , et al . An evidence-based analysis of the association between periodic leg movements during sleep and arousals in restless legs syndrome . Sleep. 2015. ; 38 ( 6 ): 919 – 924 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Manconi M , Ferri R , Zucconi M , et al . Dissociation of periodic leg movements from arousals in restless legs syndrome . Ann Neurol. 2012. ; 71 ( 6 ): 834 – 844 . [DOI] [PubMed] [Google Scholar]
- 36. Ferri R , Manconi M , Aricò D , Punjabi NM , Zucconi M . Experimentally induced arousals do not elicit periodic leg motor activity during sleep in normal subjects . Sleep Med. 2013. ; 14 ( 1 ): 85 – 90 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Schenck CH . Preface: RBD in a Nutshell and Suggested Ways To Read This Book . In: Schenck CH , Högl B , Videnovic A , eds. Rapid-Eye-Movement Sleep Behavior Disorder. Cham, Switzerland: : Springer Nature Switzerland AG; ; 2018. : vii – xv . [Google Scholar]