Abstract
Few cases have been reported to date, in which a massive rhabdomyolysis causes a cardiac arrest in a male adult suffering from undiagnosed McArdle disease. Veno‐arterial extracorporeal membrane oxygenation and cytokine adsorption filter (CytoSorb®) were required to reach a complete and successful recovery.
Keywords: Cardiac arrest, CytoSorb®, ECMO, Exercise intolerance, Myoglobinuria, Myophosphorylase deficiency, Renal failure, Rhabdomyolysis
Introduction
Metabolic myopathies are genetic disorders that cause an impairment in glycogen storage, transport, and oxidation of fatty acids and mitochondrial defects. 1 Glycogen storage disease Type V, also known as McArdle disease, results from myophosphorylase deficiency and usually manifests as exercise intolerance with myalgia and cramps. 2 Herein, we report a case of McArdle disease that manifested as cardiac arrest.
Case report
A 26‐year‐old male presented an abrupt cardiac syncope after one‐and‐a‐half‐hour medium‐intensity exercise. At admission to the emergency room, he appeared to be conscious, with progressive worsening of his general appearance and tendency to hypotension. An electrocardiogram (EKG) was performed, showing QRS widening with bizarre QRS morphology and high peaked T waves (Figure 1 ). After 10 min under continuous rhythm monitoring, the patient presented a cardiac arrest, identifying asystole as the first rhythm.
Figure 1.
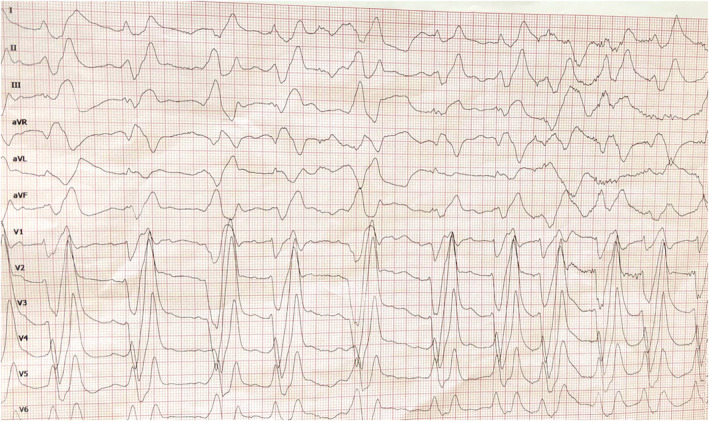
ECG performed in emergency department previous to asystole rhythm, showing QRS widening with bizarre QRS morphology and high peaked T waves.
Cardiopulmonary resuscitation manoeuvres were initiated, requiring orotracheal intubation and endovenous adrenaline. During resuscitation manoeuvres, he developed pulseless electrical activity and ventricular tachycardia, needing defibrillation in order to return to spontaneous circulation. At admission to the coronary care unit, he deteriorated as a consequence of another episode of pulseless electrical activity, and a veno‐arterial extracorporeal membrane oxygenation (VA‐ECMO) through right femoral vein and left femoral artery was implanted due to refractory cardiac arrest, after 50 min of resuscitation manoeuvres.
The patient was in good health before the described event. He had two siblings that were professional hockey players, and his parents had no known diseases. The parents mentioned the patient had complained about several episodes of dark‐brown urine and muscle weakness after exercise since he was 20 years old, reporting two or three previous episodes of syncope in this context. He had never received a medical check‐up.
Admission examination revealed remarkable oedema in the neck and upper right extremity muscles. Laboratory testing showed normal glucose and sodium, potassium 5.7 mEq/L (N:3.5–5 mEq/L) that reached levels of 8.9 mEq/L while resuscitation, blood urea nitrogen (BUN) 65 mg/dL (N:12–54 mg/dL), creatinine 1.14 mg/dL (N:0.7–1.3 mg/dL), creatine phosphokinase (CPK) 100×000 U/L (N:30–170 U/L), and high sensitivity cardiac troponin (hscTn) 700 ng/L (N:<5 ng/L), together with severe metabolic acidosis (pH 7.15) and lactate level of 9.8 mmol/L. The urine test was positive for blood, proteins, and myoglobin and negative for drugs. To stabilize the patient, a VA‐ECMO flow of 4.2 l/min (3 400 revolutions per minute [rpm]) was needed. Also, the use of high doses of vasopressor and inotropic drugs (noradrenaline 2.24 mcg/kg/min, dobutamine 22 mcg/kg/min, adrenaline 0.39 mcg/kg/min, and terlipressin 0.05 mcg/kg/h) was mandatory. A trans‐oesophageal echocardiogram (TOE) was performed, showing severe left ventricular dysfunction with no regional wall motion abnormalities or valve involvement, and right ventricular function was preserved (Figure 2 A and 2 B ). Tissue oxygenation was monitored by near‐infrared spectroscopy measuring symmetric values referring to frontal cortical oxygenation and both lower extremities.
Figure 2.
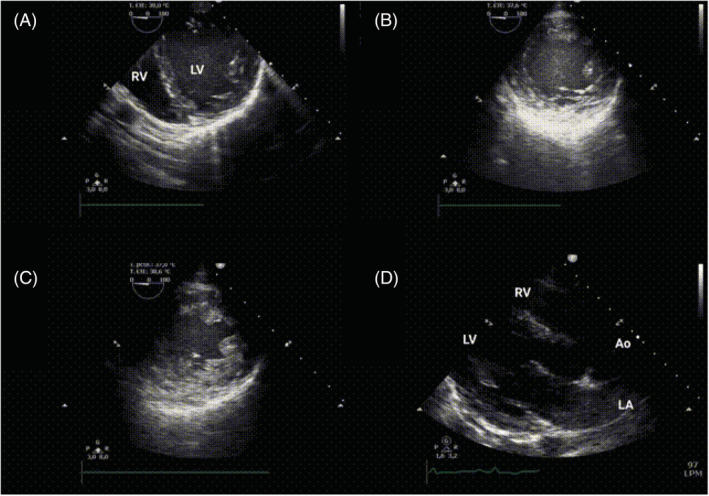
Echocardiogram. (A) TOE (transgastric view) performed during resuscitation manoeuvres. (B) TOE performed after VA‐ECMO implantation. It shows severe left ventricular dysfunction with no regional wall motion abnormalities or hypertrophy. (C) TOE performed after 24 h of VA‐ECMO support revealed significative left ventricular oedema. (D) TTR (parasternal long axis) showed normal biventricular function after VA‐ECMO discontinuation and previous to discharge from hospital.
Progressive worsening of acute renal injury with severe hypocalcaemia and elevated levels of CPK with maximum values of 314 000 U/L were detected, requiring daily extracorporeal haemodialysis with associated CytoSorb® adsorption filter during 48 h. Further tests were performed, such as muscle echography describing an asymmetric and bilateral pattern, with predominance in right upper extremity and proximal region, in relation to exercise‐induced rhabdomyolysis with probable basal metabolic disorder. Also, right biceps and deltoid muscle biopsy (Figure 3 ) resulted in early metabolic necrosis. All serological and immunological tests were negative. Several findings were demonstrated by genetic testing (Table 1 ): An homozygous mutation for the PYGM gene (p.Arg50* variant) led to diagnosis of Type V glucogenosis, and an acylcarnitine assay determined an elevated proportion C12 + C16/C2 that suggested concomitant Type II carnitine palmitoyltransferase deficiency (CPT‐II). Moreover, homozygous mutation for Val368Ile variant and heterozygous for Met647Val variant were found, in relation to CPT‐II. Other structural myopathies and cardiomyopathies were discarded by negative next‐generation sequencing panel, which included 133 and 204 genes, respectively. After these results, 1 g of L‐carnitine every 8 h and glucose–polymer supplements were administered in order to prevent further muscle injury.
Figure 3.
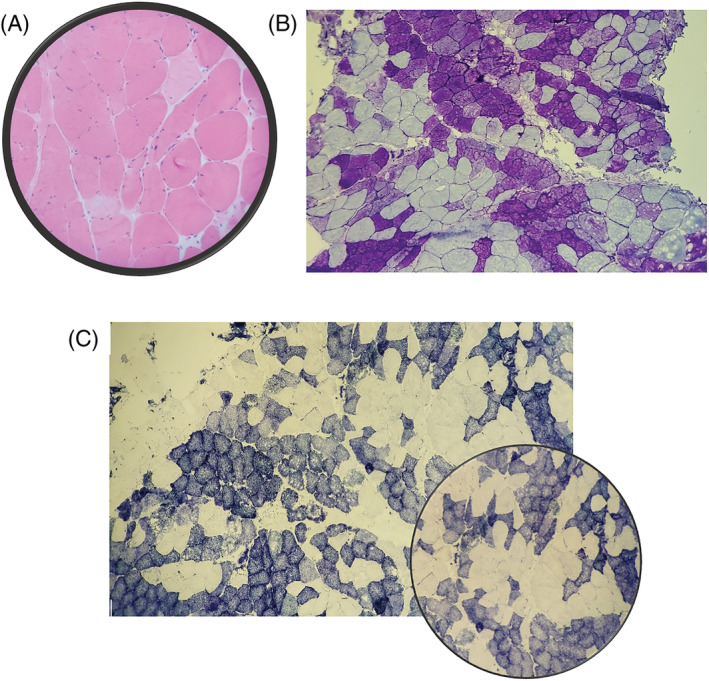
Skeletal muscle: Biopsy of right deltoids and biceps muscle: histopathological and enzymohistochemical changes lead to early (metabolic) necrosis. (A) Haematoxylin–eosin: moderately affected muscle, made up of muscle fibres of discretely variable sizes; (B) PAS: fibres with absence of staining; (C) DPNH: negative fibres.
Table 1.
Genetic testing findings following next‐generation sequencing panel in relation to cardiomyopathy and myopathy
| Genetic testing results | ||||
|---|---|---|---|---|
| Genetic testing for cardiomyopathy a | ||||
| Gene | Variant | Results | Pathogenic | Frequency in gnomAD b |
| CASZ1 |
NP_001073312.1:p.Ala1141Thr NM_001079843.2:c.3421G>A NC_000001.10:g.10707934C>T |
Heterozygosity | Variant of uncertain significance | 0.004% |
| RYR2 |
NM_001035.2:c.9067+63A>G NC_000001.10:g.237850867A>G |
Heterozygosity | Probably benign | 0.006% |
| Genetic testing for myopathy c | ||||
| PYGM |
NP_005600.1:p.Arg50* NM_005609.2:c.148C>T NC_000011.9:g.64527223G>A |
Homozygosity | Pathogenic (+++) | 0.150% |
204 genes related to genetic testing panel for cardiomyopathy.
Genome Aggregation Database.
133 genes related to genetic testing panel for myopathy.
The patient had optimal clinical recovery. VA‐ECMO was discontinued after two favourable protocols for weaning by Day 4 of support. Transthoracic echocardiogram (TTR) confirmed a normal biventricular function (Figure 2 D ). His renal function gradually returned to normal, and he was finally discharged from hospital after 33 days. A medical evaluation was performed 1 week later, providing several recommendations about nutritional supplements, aerobic training, and prevention of myoglobinuria crisis. He had progressive physical recuperation, with excellent renal and neurological recovery (Cerebral Performance Category 1).
Discussion
McArdle disease is an inherited condition resulting from myophosphorylase deficiency because of a mutation in a gene located at chromosome 11 (11q13). 3 This enzyme initiates the breakdown of glycogen in skeletal muscle, and its absence consequently causes an inability to use it as a source of energy. It is often first shown in the first two decades of life, 2 and it usually manifests as exercise intolerance with myalgia, rapid fatigue, and cramps, precipitated by sustained aerobic or isometric exercise. 4 McArdle disease patients commonly improve their exercise tolerance by the ‘second‐wind’ phenomenon, occurring after a few minutes of exercise and with a sudden decrease in heart rate. 5
Cardiac muscle is not often significantly affected in this metabolic myopathy. This might be explained by the presence of three myophosphorylase isoenzymes in cardiac human tissue. McArdle patients only lack the isoenzyme found in skeletal muscle; therefore, it remains an adequate myocardial myophosphorylase activity. 6 The most frequent cardiac involvement is electrocardiographic changes, hypertrophic cardiomyopathy, and coronary artery disease. 7 Moustafa et al. 8 have also reported degenerative vascular disease secondary to excessive circulatory responses to moderate exercise loads.
Exercise intolerance that characterized this pathology usually causes mild rhabdomyolysis, often with an associated contributing factor (alcohol, statin intake). In the described case, the patient developed a massive rhabdomyolysis, resulting in cardiac arrest, which required support with VA‐ECMO. Minimal neurological damage and reduced mortality have been reported in prolonged cardiac arrest due to reversible aetiology, in which ECMO is used as a rescue therapy. 9 Al‐Takrouri et al. 9 described a case of an 11‐year‐old male who had an unknown myopathy, developing cardiac arrest secondary to severe rhabdomyolysis and hyperkalaemia after succinylcholine administration, requiring ECMO and haemodialysis. In the Spanish national registry 10 in 2014, acute kidney injury was present in 4% of patients with McArdle disease, all of them male.
Rhabdomyolysis is a life‐threatening condition that can produce hypovolaemia, hyperkalaemia, severe metabolic acidosis, acute kidney injury, and disseminated intravascular coagulation. Its management includes aggressive fluid resuscitation and early correction of electrolyte disturbances caused by cell decay. 11 Renal replacement therapy (RRT) is seldom needed in rhabdomyolysis, but it should be considered if severe hyperkalaemia, persistent metabolic acidosis, or progressive renal failure persist in spite of medical treatment. In addition, an accumulation of creatine kinase (CK) and myoglobin in plasma occurs. Maximum value of serum CK levels is reached within 24 h after muscle injury and can further increase if damage persists. Also, 12 h after muscle damage, the maximum myoglobin value in blood is reached and is rapidly eliminated by the kidneys. 11 An accumulation of high levels of myoglobin might lead to acute renal injury by direct toxicity of myoglobin on the tubule cells. 4 The molecular weight of myoglobin is approximately 17 kDa, and because of this, it is poorly eliminated by standard dialysis membrane used in RRT.
Extracorporeal systems are currently designed in order to effectively adsorb proinflammatory mediators that have a molecular weight between 10 and 60 kDa from the blood. These devices contain a large haemoadsorption surface made up of beads of a biocompatible porous polymer capable of removing cytokines from the plasma (TNF‐α, IL‐1β, IL‐6, etc.) as well as other molecules that enhance inflammatory response (bilirubin, myoglobin, etc.). 12 Cytokine filters such as CytoSorb® are intended for the elimination of cytokines in hyperinflammatory conditions and are relatively simple to use. They can be inserted in devices as continuous veno‐venous haemodiafiltration (CVVHDF) or short‐term mechanical circulatory support (VA‐ECMO or VV‐ECMO). This allows a reduction in the severity of systemic inflammatory response syndrome and consequently the metabolic acidosis, risk of acute kidney and lung failure, peripheral vasodilation, and lastly the overall mortality. 11 Although CytoSorb® has been recently approved within the European Union for removing myoglobin, bilirubin, and bile acids from blood, according to in vitro and experimental data, there is a lack of available clinical studies to confirm the clinical benefits in these indications. 13 Myoglobin elimination with cytokine adsorber integrated into high‐flux dialyzer in critically ill patients secondary to rhabdomyolysis can be recommended for routine clinical use due to its easy application and absence of side effects.
There is no current treatment for McArdle disease, but individuals usually benefit from a carbohydrate‐rich diet and moderate‐intensity aerobic training to increase muscle oxidative capacity and cardiorespiratory fitness. 14 Moreover, there are several genes that can act as modulators of clinical phenotype, for example, AMPD1 or angiotensin‐converting enzyme. In patients with D/D angiotensin‐converting enzyme genotype, treatment with low doses of ramipril should be considered in order to improve exercise tolerance.
To our knowledge, this is the first case reported in the literature where CytoSorb® has been used together with CVVHDF and VA‐ECMO in a patient suffering from cardiac arrest secondary to severe rhabdomyolysis in relation to undiagnosed metabolic disease. This could be triggered by intense exercise activity and lack of optimal hydration and energy supplements, after alcohol intake or under high temperature conditions.
Conflict of interest
No conflict of interest.
Soria‐Navarro, R. , Burgos Palacios, V. , Castrillo Bustamante, C. , Gallardo Agromayor, E. , Marcos González, S. , and González‐Lamuño Leguina, D. (2022) Cardiac arrest as a manifestation of unknown Type V glycogenosis: a case report. ESC Heart Failure, 9: 3625–3629. 10.1002/ehf2.14060.
References
- 1. Tarnopolsky MA. Metabolic myopathies. Contin Lifelong Learn Neurol 2016; 22: 1829–1851. [DOI] [PubMed] [Google Scholar]
- 2. Gordon N. Glycogenosis type V or McArdle's disease. Dev Med Child Neurol 2003; 45: 640–644. [DOI] [PubMed] [Google Scholar]
- 3. Faigel HC. Carnitine palmitoyltransferase deficiency in a college athlete: A case report and literature review. J Am Coll Health Assoc 1995; 44: 43–49. [DOI] [PubMed] [Google Scholar]
- 4. Loupy A, Pouchot J, Hertig A, Bonnard G, Bouvard E, Rondeau E. Rhabdomyolyse massive révélant une maladie de McArdle. Rev Med Interne 2007; 28: 501–503. [DOI] [PubMed] [Google Scholar]
- 5. Porcelli S, Marzorati M, Belletti M, Bellistri G, Morandi L, Grassi B. The “second wind” in McArdle's disease patients during a second bout of constant work rate submaximal exercise. J Appl Physiol 2014; 116: 1230–1237. [DOI] [PubMed] [Google Scholar]
- 6. Nicholls DP, Campbell NPS, Stevenson HP, Patterson VH. Angina in McArdle's disease. Br Heart J 1996; 76: 372–373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Jones DM, Lopes L, Quinlivan R, Elliott PM, Khanji MY. Cardiac manifestations of McArdle disease. Eur Heart J 2019; 40: 397–398. [DOI] [PubMed] [Google Scholar]
- 8. Moustafa S, Patton DJ, Connelly MS. Unforeseen cardiac involvement in McArdle's disease. Heart Lung Circ 2013; 22: 769–771. [DOI] [PubMed] [Google Scholar]
- 9. Al‐Takrouri H, Martin TW, Mayhew JF. Hyperkalemic cardiac arrest following succinylcholine administration: The use of extracorporeal membrane oxygenation in an emergency situation. J Clin Anesth 2004; 16: 449–451. [DOI] [PubMed] [Google Scholar]
- 10. Lucia A, Ruiz JR, Santalla A, Nogales‐Gadea G, Rubio JC, García‐Consuegra I, Cabello A, Pérez M, Teijeira S, Vieitez I, Navarro C, Arenas J, Martin MA, Andreu AL. Genotypic and phenotypic features of McArdle disease: Insights from the Spanish national registry. J Neurol Neurosurg Psychiatry 2012; 83: 322–328. [DOI] [PubMed] [Google Scholar]
- 11. Scharf C, Liebchen U, Paal M, Irlbeck M, Zoller M, Schroeder I. Blood purification with a cytokine adsorber for the elimination of myoglobin in critically ill patients with severe rhabdomyolysis. Crit Care 2021; 25: 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Carballo B, Alonso Á, Sánchez J. Novedades en cardiología crítica y cirugía cardíaca: utilización de filtros de adsorción extracorpórea de citoquinas (Citosorb®). Cirugia Cardiovasc 2019; 26: 293–295. [Google Scholar]
- 13. Poli EC, Rimmelé T, Schneider AG. Hemoadsorption with CytoSorb®. Intensive Care Med 2019; 45: 236–239. [DOI] [PubMed] [Google Scholar]
- 14. Quinlivan R, Martinuzzi A, Schoser B. Pharmacological and nutritional treatment for McArdle disease (glycogen storage disease type V). Cochrane Database Syst Rev 2014; 2014: CD003458. [DOI] [PMC free article] [PubMed] [Google Scholar]


