Abstract
Sepsis represents a serious risk to the life of any patient, which is why it is crucial to start an effective treatment in all its extremes as soon as possible, that is, the chosen antibiotics must have activity against the pathogen that produces the condition and, in addition, they must be dosed considering the patient’s situation in all its extremes. It should be considered that it will be necessary to adjust the dose when there is edema (drugs with reduced volume of distribution), hypoproteinemia (drugs bound to proteins in a high proportion), obesity, and also when they require the use of external techniques such as ECMO or any of the different types of hemodialysis and hemofiltration.
Keywords: antibiotics, treatment, sepsis
The optimization of antimicrobial treatment in patients with sepsis becomes a priority since the correct choice of drug will lead to beneficial effects, while errors often have dire consequences for the patient. The list of criteria to consider in choosing the drug is extensive and often strictly followed by the prescribing physician, and despite this, it is not uncommon for an inadequate dosage regimen to be prescribed, generally because not all adjustment criteria have been considered and especially, those related to the PK/PD relationship.
Currently antibiotics tend to be grouped into one of the PK/PD models; fCmax/MIC, fT>MIC and fAUC/MIC requiring a different dosage plan. In the case of fCmax/MIC adjustment, a high dose should be administered that allows Cmax values of free drug to be 10-12 times the MIC (aminoglycosides and fluoroquinolones). The drugs whose ajsute corresponds to the efficacy time fT>CMI; Basically, beta-lactams are administered in short intervals or extended and even continuous perfusions, so that the value of the PK/PD parameter is close to 100%. Finally, it is recommended to prescribe doses and intervals that facilitate reaching fAUC/MIC values between 80 and 100 (linezolid, daptomycin) or greater than 400, in the case of vancomycin [1,2].
This type of dosage adjustment must also take into account the multitude of situations specific to the patient that alter the volume of distribution and/or the clearance of drugs, which affects the production of different plasmatic concentrations; high or low, which may justify inefficacy or adverse effects. The patient in a situation of sepsis is an example in which these types of alterations are almost constant. Next, the alterations that generate changes in the volume of distribution will be briefly described; edema, hypoproteinemia, obesity and/ or clearance; use of external techniques; ECMO and other purification systems. Renal or hepatic failure will not be included in this description since there is abundant information on the dose adjustment of each of the drugs used in the treatment of patients with sepsis [3].
Edema. Edema is characterized by the increase in water in the extravascular, interstitial space and with it the increase in the volume in which some drugs are dissolved. The impact on drug concentrations is very different depending on the type of distribution that characterizes them. A simple numerical example allows us to appreciate these differences. A drug that has a volume of distribution of 20L and is administered intravenously at a dose of 1 g will reach a maximum plasma concentration of 50 mg/L (1000 mg divided by 20 L). If this drug is administered to a patient with edema, for example 10L of water in the form of edema, the concentration reached will be clearly lower; 1000 mg divided by 30 L; 20+10 L of edema. In this case, the maximum plasma concentration will be 33 mg/L, that is, approximately 60% lower, hence, to reach the same concentration as if there were no edema, it is necessary to increase the dose.
If we repeat the calculations administering the same dose of another drug that, in relation to its chemical structure, has a greater volume of distribution; for example, 100 L, the plasma concentration that will initially be 10 mg/L, will become, in the patient with 10 L of edema, 9.09 mg/L, that is to say, practically only 10% lower, hence no require dose adjustment.
Therefore, the aforementioned dose adjustment will only apply to those antibiotics that have low distribution volumes, approximately 0.5 L/kg or less; ie beta-lactams, aminoglyco-sides, glycopeptides, lipopeptides, polypeptides. In this case, estimating the edema volume to add it to the conventional distribution volume of the drug can be a very useful tool to avoid sub-therapeutic concentrations.
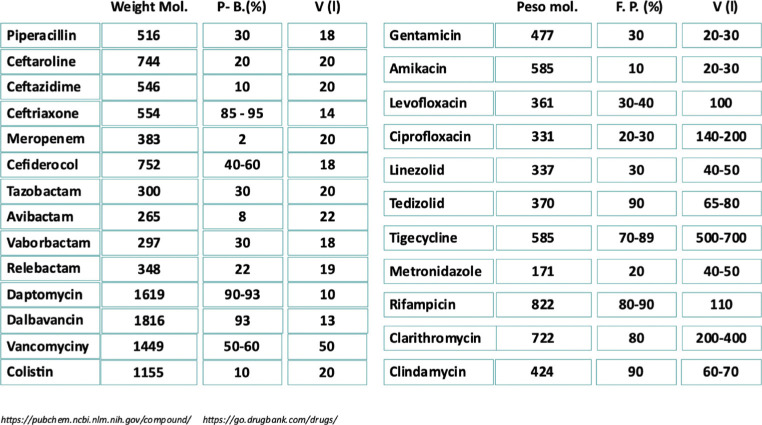
Plasma proteins. Some antibiotics circulate in plasma bound to proteins in a high proportion. The protein that most frequently participates in this transport is albumin and, much less frequently, alpha-glycoprotein. The presence of a reduced concentration of this type of protein in plasma means that the free fraction of circulating fixed drugs increases and contrary to what could be deduced, the overall balance is a reduction in concentrations since the increase in the free fraction It implies greater distribution and greater clearance. There is no adequate method to adjust the dose since there is no methodology to estimate the impact of this phenomenon on the concentration, which, on the other hand, only becomes relevant in drugs with very high protein binding (Figure 1); Daptomycin, dalbavancin, tedizolid, ceftriaxone, tigecycline, and rifampin are the most notable examples of drugs with potential use in patients with sepsis [4].
Figure 1.
Bound protein
Obesity. It is probably the disease that entails the greatest difficulties in adjusting the dose of any drug. In a generic way, it is usually pointed out that the most fat-soluble drugs are the ones that need to be adjusted to the real weight of people, while the water-soluble ones could be administered at the ideal weight, sometimes called lean weight, that is, the one corresponding to the sex, age and height of the person. person. To assess the degree of lipid solubility of a molecule, it is necessary to know its partition coefficient, which is estimated through LogP; quotient between the concentration of non-ionized solutes in octanol and that in water, both expressed in natural logarithms. The presence of a negative number expresses water solubility, while a positive value indicates lipid solubility (Figure 2). Following this criterion, fat-soluble drugs should be administered according to the actual weight of the patient, but accumulated experience indicates that this type of adjustment carries a risk of overdosing, since the volume of distribution of these drugs is not directly related to weight gain. In fact, the tendency is to make a less aggressive adjustment, using the so-called adjusted weight, which is calculated using the ideal weight, to which is added a proportion of overweight, which is usually 0.3-0.4 [5-8]. Figure 2 includes the description of the type of adjustment that is currently considered appropriate for each of the antibiotics.
Figure 2.

Log P and adjustment of dose to weight
ECMO (Extracorporeal Membrane Oxygenation). Extracorporeal membrane oxygenation (ECMO) is an extrapulmonary gas exchange system that allows oxygenation and CO2 extraction through a membrane connected to an external venovenous or venoarterial bypass system.
There are multiple factors inherent to the circuit, the drugs and the patient’s situation that make it difficult to estimate the impact on the drugs and thus the type of adjustment to be made.
On the part of the drug, it seems that lipid solubility leads to an increase in the volume of distribution, and a reduction in clearance, probably due to sequestration of the drug in the membrane. In addition, protein binding is a factor associated with reduced clearance and also volume of distribution in patients on ECMO. Based on their protein binding (>70%) and their partition coefficient (>2), tedizolid, rifampicin, clarithromycin or clindamycin are the antibiotics that are most likely to be affected by membrane sequestration (Figures 1 and 2). Other drugs affected in relation to high protein binding are cloxacillin, ceftriaxone, dalbavancin, daptomycin, and tigecycline. It is likely that all these drugs require the administration of higher doses and more frequently, but at the moment there are no clear criteria for making this type of adjustment [9-12].
External debugging techniques. This type of technique is of great importance in the maintenance of critical patients with or without sepsis. Its impact on the pharmacokinetics of antibiotics can become very evident and again this impact depends on a large number of circumstances. For antibiotics, volume of distribution, protein binding, and molecular weight are inversely related to the efficacy of elimination techniques. In other words, the higher the values of these parameters, the lower the amount of drug eliminated. The vast majority of beta-lactam antibiotics (except ceftriaxone, cefonid or ertapenem), vancomycin, amino-glycosides and colistin are found in this situation, drugs that will require the administration of doses higher than the conventional adjustment in relation to the patient’s renal function [13,14].
CONFLICT OF INTEREST
Authors declare no conflict of interest
References
- 1.Mensa J, Barberán J, Soriano A, Llinares P, Marco F, Cantón R, et al. Antibiotic selection in the treatment of acute invasive infections by Pseudomonas aeruginosa: Guidelines by the Spanish Society of Chemotherapy. Rev Esp Quimioter. 2018. Feb;31(1):78-100. [PMC free article] [PubMed] [Google Scholar]
- 2.Phe K, Heil EL, Tam VH. Optimizing Pharmacokinetics-Pharmaco-dynamics of Antimicrobial Management in Patients with Sepsis: A Review. J Infect Dis. 2020. Jul 21;222(Suppl 2):S132-S141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Póvoa P, Moniz P, Pereira JG, Coelho L. Optimizing Antimicrobial Drug Dosing in Critically Ill Patients. Microorganisms. 2021. Jun 28;9(7):1401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ulldemolins M, Roberts JA, Rello J, Paterson DL, Lipman J. The effects of hypoalbuminaemia on optimizing antibacterial dosing in critically ill patients. Clin Pharmacokinet. 2011. Feb;50(2):99-110. [DOI] [PubMed] [Google Scholar]
- 5.Janson B, Thursky K. Dosing of antibiotics in obesity. Curr Opin Infect Dis. 2012. Dec;25(6):634-49. [DOI] [PubMed] [Google Scholar]
- 6.Alobaid AS, Hites M, Lipman J, Taccone FS, Roberts JA. Effect of obesity on the pharmacokinetics of antimicrobials in critically ill patients: A structured review. Int J Antimicrob Agents. 2016. Apr;47(4):259-68. [DOI] [PubMed] [Google Scholar]
- 7.Pai MP. Anti-infective Dosing for Obese Adult Patients: A Focus on Newer Drugs to Treat Methicillin-resistant Staphylococcus aureus Acute Bacterial Skin and Skin Structure Infections. Clin Ther. 2016. Sep;38(9):2032-44. [DOI] [PubMed] [Google Scholar]
- 8.Meng L, Mui E, Holubar MK, Deresinski SC. Comprehensive Guidance for Antibiotic Dosing in Obese Adults. Pharmacotherapy. 2017. Nov;37(11):1415-1431. [DOI] [PubMed] [Google Scholar]
- 9.Ha MA, Sieg AC. Evaluation of Altered Drug Pharmacokinetics in Critically Ill Adults Receiving Extracorporeal Membrane Oxygenation. Pharmacotherapy. 2017. Feb;37(2):221-235. [DOI] [PubMed] [Google Scholar]
- 10.Gomez F, Veita J, Laudanski K. Antibiotics and ECMO in the Adult Population-Persistent Challenges and Practical Guides. Antibiotics (Basel). 2022. Mar 4;11(3):338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shekar K, Roberts JA, Barnett AG, et al. Can physicochemical properties of antimicrobials be used to predict their pharmacokinetics during extracorporeal membrane oxygenation? Illustrative data from ovine models. Crit Care. 2015. Dec 15;19:437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gomez F, Veita J, Laudanski K. Antibiotics and ECMO in the Adult Population-Persistent Challenges and Practical Guides. Antibiotics (Basel). 2022. Mar 4;11(3):338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hoff BM, Maker JH, Dager WE, Heintz BH. Antibiotic Dosing for Critically Ill Adult Patients Receiving Intermittent Hemodialysis, Prolonged Intermittent Renal Replacement Therapy, and Continuous Renal Replacement Therapy: An Update. Ann Pharmacother. 2020. Jan;54(1):43-55. [DOI] [PubMed] [Google Scholar]
- 14.Choi G, Gomersall CD, Tian Q, Joynt GM, Freebairn R, Lipman J. Principles of antibacterial dosing in continuous renal replacement therapy. Crit Care Med. 2009. Jul;37(7):2268-82. [DOI] [PubMed] [Google Scholar]