Key Points
This study describes a novel drug treatment to prevent vascular calcification in patients with CKD and on dialysis.
For the first time, we describe a potential therapeutic to treat renal osteodystrophy and vascular calcification in CKD.
This study describes a novel therapeutic to prevent and possibly to treat calciphylaxis in patients with CKD and on dialysis.
Keywords: chronic kidney disease, ASARM, basic science, calciphylaxis, calciprotein particles, CKD-MBD, DMP1, FGF23, matrix vesicles, MEPE, osteopontin, vascular calcification
Visual Abstract
Abstract
Background
Abnormalities in calcium, phosphorus, PTH, vitamin D metabolism, bone, and vascular calcification occur in chronic kidney disease mineral bone disorder (CKD-MBD). Calciphylaxis, involving painful, ulcerative skin lesions, is also a major problem associated with CKD-MBD. There are no quality medical interventions to address these clinical issues. Bone ASARM peptides are strong inhibitors of mineralization and induce hypophosphatemia by inhibiting phosphate uptake from the gut. We hypothesize treatment of CKD-MBD rats with ASARM peptides will reverse hyperphosphatemia, reduce soft-tissue calcification, and prevent calciphylaxis.
Methods
To test our hypothesis, we assessed the effects of synthetic ASARM peptide in rats that had undergone a subtotal 5/6th nephrectomy (56NEPHREX), a rodent model of CKD-MBD. All rats were fed a high phosphate diet (2% Pi) to worsen mineral metabolism defects. Changes in serum potassium, phosphate, BUN, creatinine, PTH, FGF23, and calcium were assessed in response to 28 days of ASARM peptide infusion. Also, changes in bone quality, soft-tissue calcification, and expression of gut Npt2b (Slc34a2) were studied following ASARM peptide treatment.
Results
Rats that had undergone 56NEPHREX treated with ASARM peptide showed major improvements in hyperphosphatemia, blood urea nitrogen (BUN), and bone quality compared with vehicle controls. Also, ASARM-infused 56NEPHREX rats displayed improved renal, brain, and cardiovascular calcification. Notably, ASARM peptide infusion prevented the genesis of subdermal medial blood vessel calcification and calciphylaxis-like lesions in 56NEPHREX rats compared with vehicle controls.
Conclusions
ASARM peptide infusion corrects hyperphosphatemia and improves vascular calcification, renal calcification, brain calcification, bone quality, renal function, and skin mineralization abnormalities in 56NEPHREX rats. These findings confirm our hypothesis and support the utility of ASARM peptide treatment in patients with CKD-MBD.
Introduction
Abnormalities in blood chemistries, parathyroid hyperplasia, renal osteodystrophy (ROD), and mineral vascular or soft-tissue calcification occur in CKD mineralization bone disorder (CKD-MBD) (1–3). In 2000, an estimated 26.3 million US adults had stages 1–4 CKD, with 5%–10% of the world’s population affected by CKD (2,4,5). Roughly 15% of adult Americans currently have CKD, and nearly 800,000 of these have ESKD (5). In patients receiving dialysis, cardiovascular events account for nearly one third of hospitalizations and one half of all deaths (5). Importantly, a dramatic increased risk of an adverse major cardiovascular event also occurs, with “non-ESKD” patients showing small reductions in kidney function (6,7). Most individuals with mild to moderate CKD will progress to ESKD requiring dialysis or transplantation and are 10–20 times more likely to die of cardiovascular causes (8). Traditional risk factors such as diabetes and hypertension account for some of the high rate of cardiovascular morbidity and mortality in patients with CKD. Of note, there are unique risk factors promoting cardiovascular pathology in the CKD population (9,10). For example, a high prevalence of vascular calcification (VC) occurs in CKD patients, and this contributes to reduced arterial compliance, hypertension, left ventricular hypertrophy (LVH) (11–13), cognitive impairment, and neurologic disorders (14,15). Notably, VC and impaired mineral homeostasis dynamically link to the abnormal bone disorders, hypocalcemia, secondary hyperparathyroidism, and increased FGF23 levels characteristic of CKD-MBD (16).
VC and increased morbidity and mortality in patients with CKD-MBD or on dialysis correlate strongly with hyperphosphatemia (17–23). However, there are no randomized controlled trials confirming a causal link of hyperphosphatemia to patient outcomes. Current drug therapies for VC chiefly rely on inhibitors that directly target phosphate lowering or “indirectly target” imbalanced mineralization promoters (e.g., MGP activation with vitamin K) (22,24). There is a clear and urgent need to develop treatment strategies to control hyperphosphatemia and prevent VC progression in CKD-MBD and ESKD patients on dialysis. This study examines the utility of a small 2.2 kDa peptide (ASARM peptide) to prevent hyperphosphatemia and VC in a rat model of CKD-MBD. The ASARM peptide derives from the C-terminal ASARM motif of matrix extracellular phospho-glycoprotein (MEPE) (25). The ASARM motif, when cleaved and released as a phosphorylated peptide (ASARM peptide), inhibits mineralization, renal phosphate uptake, and intestinal phosphate uptake (26–48). Specifically, ASARM peptide binds strongly to calcium phosphate mineral complexes (including hydroxyapatite) and is a potent inhibitor of mineralization and crystal nucleation uptake (in vivo, ex vivo, and in vitro) (25,26,35,38,40–42,47–49). Also, the ASARM motif is important for regulating expression of FGF23 (a phosphaturic cytokine) in vitro and in vivo, and ASARM peptides suppress osteogenesis in vivo and in vitro (26–28,32,37,38). Increased circulating ASARM peptides in X-linked and autosomal inherited hypophosphatemic rickets are chiefly responsible for the mineralization changes in these diseases (32,38,43). Moreover, we have bioengineered a 4.2-kDa synthetic PHEX-peptide (SPR4) that specifically binds and neutralizes ASARM peptides (38). This peptide (SPR4) corrects the mineralization defect in vitro and in vivo and has marked anabolic effects on bone (27,28,33,38,50).
Because ASARM peptides inhibit mineralization and suppress osteogenic phenotypic transition and inhibit intestinal and renal phosphate uptake, we examined the utility of ASARM treatment in renal disease. Specifically, this study investigated whether a synthetic 2.2-kDa ASARM peptide infusion is useful for preventing disease progression in a rat model of CKD-MBD. The data show that ASARM peptide infusion reduces serum phosphate levels, corrects BUN levels, suppresses brain, renal, and cardiovascular calcification, improves bone quality, and prevents calciphylaxis-like lesions. These results show for the first time that synthetic ASARM peptide has potential as a treatment for CKD-MBD sequelae, including hyperphosphatemia, VC, and loss of renal function.
Materials and Methods
Animal Housing, Protocols, ASARM Peptide, and Diet
All animal protocols were in accordance with institutional guidelines of the Kansas University Medical Center (KUMC) as detailed in the Guide for the Care and Use of Laboratory Animals (51). The Animal Care and Use Project numbers for this study were #2017–2422 and #2020–2591. Rat experiments were conducted with male Wistar rats (weighing 285 g and aged 60–63 days old at the start of surgery) purchased from Charles River Laboratories (Wilmington, MA). Mouse experiments were carried out with male Alport mice (Col4a3−/− and Col4a3+/+; male, 12 weeks old; strain 129-Col4a3tm1De) as reported previously and maintained as a breeding colony in the animal care facility at KUMC (52). Alport mice genotypes were confirmed by PCR analysis of DNA from tail clippings (52). Rats were housed one per cage and mice five per cage with enrichment and free access to food and water. The temperature and the humidity of the air were controlled and varied between 40% and 60%; a light cycle of 7–19 hours was carried out. Several daily passages of competent personnel were carried out in the animal facility to control the welfare of the animals and for the identification of the end points. ASARM peptide (NH2-RDDSSESSDSGS[PO3H2]-SS-[PO3H2]ES-[PO3H2]DGD-OH) was synthesized using standard techniques and purchased from Peptides International (Louisville, KY) as described previously (32,38,41). The control diet (NPO4) consisted of 0.33% Pi, 2000 IU vitamin D, and 0.8% Ca (#170497; Teklad) and the high phosphate diet (HPO4) consisted of 2% Pi, 2000 IU vitamin D, and 0.8% Ca (#170496; Teklad). Both diets (NPO4 and HPO4) were formulated to have identical compositions except for the phosphate and can be purchased from Envigo (Indianapolis, IN).
Induction of CKD in Rats Using Subtotal 5/6th Nephrectomy
Surgical 5/6th nephrectomy (56NEPHREX) and sham surgery Wistar male rats were purchased from Charles River Laboratories and transferred to the KUMC and Laboratory Animal Resource Center. The 56NEPHREX procedure consisted of a two-part surgery, with 5–7 days between surgeries as reported in the literature and listed as surgery code “56NEPHREX” by Charles River Laboratories (53,54). The first 56NEPHREX surgery removes two thirds of one kidney, and the second surgery removes the entire other kidney. Specifically, for stage 1 surgery, a ventral midline incision into the abdomen was made to expose the animal’s left kidney. The exposed kidney was then freed from the surrounding tissue, and ligatures applied to the upper and lower third poles. The upper and lower one third sections were then excised, and the abdominal incision closed. After 5–7 days, stage 2 of the surgical procedure was carried out. Briefly, the animal was placed in ventral recumbency, and an incision was made parallel to the midline. The abdominal cavity was then entered, and the right kidney freed from the surrounding tissue. The kidney was then pulled gently out from the incision, and the adrenal gland was freed and replaced into the abdominal cavity. The renal blood vessels and the ureter were then ligated or cauterized. The kidney was then removed by transecting the vessels and ureter just distal to the ligature or cauterized section. Finally, the skin incision was closed with wound clips. Rats were first maintained on a control diet (NPO4; 0.33% Pi, 2000 IU vitamin D, and 0.8% Ca; #170497; Teklad). Ten days after surgery and 3 days before osmotic pump implantation, the diet was switched to a high phosphate diet (HPO4; 2% Pi, 2000 IU vitamin D, and 0.8% Ca; #170496; Teklad; Envigo). The 0.33% and 2% Pi diets reflect accurately the available Pi (nonphytates) and are compositionally identical except for the Pi content. The 2% phosphate (nonphytate) diet emulates the upper range of high phosphate intake in the Western diet (55–58). A summary of the study protocol and the number of rats used per group is shown in Figure 1. Baseline sera were collected 2 days before switching to a high phosphate diet (8 days post surgery), and osmotic pumps were implanted 3 days after switching to a high phosphate diet (HPO4; 13 days post surgery). Confirmation of CKD induction and standardization for the 56NEPHREX rats was carried out by first measuring serum phosphate, serum creatinine, and BUN (sera drawn 8 days post surgery). Specifically, sera drawn before pump implantation were used for the serum measurements, and the values compared with the sham rats. All three of the 56NEPHREX serum metrics (serum PO4, BUN, and serum creatinine) were significantly different to sham rats and consistent with CKD disease induction. Grubb’s test, also called the extreme studentized deviate method, was then used to determine outliers for all 56NEPRHEX rats (59,60). Notably, no outliers were detected for the serum metrics using Grubb’s test. A random generator was then used to divide the 24 56NEPHREX rats into the vehicle- and ASARM-treated groups (see Figures 1 and 2). Note, two ASARM peptide 56NEPHREX rats did not recover from anesthesia following ASARM pump implantation. Male rats were used for the experiment (weighing 285 g and aged 60–63 days at the start of sham and 56NEPHREX surgeries).
Figure 1.
Experimental design. (A) Protocol and rat numbers for osmotic pump infusion of ASARM peptide. (B) Timeline of experiment. Rats were first maintained on a control diet (NPO4; 0.33% P, 2000 IU vitamin D, and 0.8% Ca; #170497; Teklad). Baseline sera was collected 2 days before switching to a high phosphate diet, and osmotic pumps were implanted 3 days after switching to a high phosphate diet (HPO4; 2% P, 2000 IU vitamin D, and 0.8% Ca; #170496; Teklad). The subcutaneous implantation of Alzet pumps was carried out 2 weeks post surgery. Pump infusion with ASARM peptide or vehicle was then carried out for 28 days, and rats were maintained on the high phosphate diet. The timing and number of serum and urine collections are highlighted with arrows and text. Male rats were used for the experiment (weight 285 g; aged 60–63 days at start of sham and 56NEPHREX surgeries). Note: two ASARM peptide 56NEPHREX rats did not recover from anesthesia following ASARM pump implantation. 56NEPHREX, subtotal 5/6th nephrectomy.
Figure 2.
ASARM infusion corrects key serum chemistries in subtotal 56NX rats fed a high phosphate diet (HPO4; 2% P, 2000 IU vitamin D, and 0.8% Ca; #170496; Teklad). Measurements were taken at the start and end of the experiment (28 days infusion). See Figure 1 for experimental design and rat numbers per group (n). For the changes between groups over time, the data were analyzed using two-way ANOVA with Tukey’s multiple comparison test (α=0.05)—see narrative below. For statistical analyses and comparisons at the start and end of the experiment, a one-way ANOVA and Tukey’s multi comparison test were carried out; ns, not significant; *P<0.01, significant. (A) Serum PO4: Error bars represent SD. For 56NX VEH versus 56NX ASARM, ASARM treatment has a significant effect at all values of time, with the interaction accounting for approximately 30% of the total variance (F=34.09, DFn=1, and Dfd=43; P<0.001). For SHAM VEH versus 56NX ASARM, time has no significant effect, with the interaction accounting for approximately <0.1% of the total variance (F=0.03, DFn=1, and Dfd=30; P=0.87). For SHAM VEH versus 56NX VEH, time has a significant effect with the interaction, accounting for approximately 13% of the total variance (F=24.03, DFn=1, and Dfd=31; P<0.001). (B) Serum Ca2+: Error bars represent SD. Interactions show significant serum Ca2+ changes over time for all groups (P<0.001). (C) Serum BUN: Error bars represent SEM. For 56NX VEH versus 56NX ASARM, ASARM treatment has a significant effect at all values of time, with the interaction accounting for approximately 23% of the total variance (F=32.32, DFn=1, and Dfd=42; P<0.001). For SHAM VEH versus 56NX ASARM, treatment time has no significant effect, with the interaction accounting for approximately 2% of the total variance (F=1.22, DFn=1, and Dfd=32; P=0.28). For SHAM VEH versus 56NX VEH, treatment time has a significant effect with the interaction, accounting for approximately 25% of the total variance (F=41.14, DFn=1, and Dfd=32; P<0.001). (D) Serum creatinine: Error bars represent SD. Interactions show significant serum creatinine changes over time for all groups (P<0.001). (E) Serum K+: Error bars represent SEM. Interactions show significant serum K+ changes over time for all groups (P<0.001). (F) Serum Na+: Error bars represent SEM. (G) Serum HCO3: Error bars represent SD. (H) Serum albumin: Error bars represent SEM. SHAM VEH, sham-operated vehicle; 56NX VEH, subtotal 5/6th nephrectomy vehicle; 56NX ASARM, subtotal 5/6th nephrectomy ASARM peptide.
Infusion of ASARM Peptide and Experimental Protocol
Following 56NEPHREX or sham operations, rats were subjected to the following protocols (see Figure 1A for design and Figure 1B for timeline). Continuous infusion (28 days) of ASARM peptide and vehicle were undertaken using subcutaneous transplantation of Alzet osmotic pumps (Durect Corp., Cupertino, CA). Pump model 2ML4 (28-day model; infusion rate 2.5 μl/h) was used. Three days before implantation of the osmotic pumps, the diet was switched to the high phosphate diet (HPO4). The subcutaneous implantation of the Alzet pumps was carried out 13 days post surgery. The pumps were implanted in rats anesthetized with isoflurane. An ASARM peptide pump infusion rate of 8.4 nmol/kg per hour or 5.1 × 1015 molecules/kg per hour was used for 28 days. This flow rate is based on our published results and consistent with known circulating levels of ASARM peptides in HYP and wild-type rodents (26–28,32,37,40,43,50,61,62). Specifically, this regimen is an excellent compromise for duplicating the human clinical context of CKD-MBD in rats. Animals were housed individually, and food intake monitored to assess whether treatments affected appetite.
Serum Analysis, Histology, and Immunohistologic Staining
Blood samples obtained by retro-orbital bleed (base control; 8 days post surgery) and cardiac exsanguination (final bleed; 28th day of infusion) were collected in separator tubes, and serum prepared as described previously (32). Serum analysis results are shown in Figures 2 and 3 and were carried out using commercial kits as described previously (26–28,32,37,38,40,41,50). ASARM peptides in sera were measured using our extensively published and developed competitive ELISA (26,32,37,38,40,43,62). Immunohistochemistry and histologic analyses were carried out as described previously (37,38,43). Briefly, tissues were suspended in paraformaldehyde for 24 hours followed by fixation in 70% ethanol for 7 days and then embedded in paraffin. Using a microtome (Leica, Wetzlar, Germany), 20-μM sections were attached to microscope slides and then stained for mineral using Von Kossa as described previously (37). Also, fixed jejunum samples were screened with anti-NT2B (slc334a2) antibodies and filamentous F-Actin antibodies (Invitrogen, Carlsbad, CA).
Figure 3.
ASARM infusion corrects key serum chemistries in subtotal 56NX rats fed a high phosphate diet (HPO4; 2% P, 2000 IU vitamin D, and 0.8% Ca; #170496; Teklad). Measurements were taken at the start and end of the experiment (28 days infusion). See Figure 1 for experimental design and rat numbers per group (n). For the changes between groups over time, the data were analyzed using two-way ANOVA with Tukey’s multiple comparison test (α=0.05)—see narrative below. For statistical analyses and comparisons at the start and end of the experiment, a one-way ANOVA and Tukey’s multiple comparison test were carried out; ns, not significant; *P<0.01, significant. (A) Serum protein: Error bars represent SEM. For SHAM VEH versus 56NX ASARM, treatment time has no significant effect, with the interaction accounting for approximately 2% of the total variance (F=0.79, DFn=1, and Dfd=25; P=0.38). For SHAM VEH versus 56NX VEH, treatment time has no significant effect, with the interaction accounting for approximately 7% of the total variance (F=2.18, DFn=1, and Dfd=24; P=0.15). For 56NX VEH versus 56NX ASARM, ASARM treatment time has a significant effect, with the interaction accounting for approximately 12% of the total variance (F=4.19, DFn=1, and Dfd=29; P=0.05). (B) Serum PTH: Error bars represent SEM. Treatment time (vehicle or ASARM) has a significant effect for all groups, with the interaction accounting for approximately 55% of the total variance (F=57.12, DFn=1, and Dfd=45; P<0.001. However, no significant treatment effects occurred between the three groups. (C) SERUM ALT: Error bars represent SD. No significant treatment or temporal effects occurred between the three groups. (D) Serum glucose: Error bars represent SD. For SHAM VEH versus 56NX VEH, there is a significant effect at all values of time, with the interaction accounting for approximately 21% of the total variance (F=20.69, DFn=1, and Dfd=23; P<0.001). For SHAM VEH versus 56NX ASARM, there is no significant effect at all values of time, with the interaction accounting for <0.1% of the total variance (F=0.01, DFn=1, and Dfd=22; P=0.91). For 56NX VEH versus 56NX ASARM, ASARM treatment has a significant effect at all values of time, with the interaction accounting for approximately 19% of the total variance (F=30.91, DFn=1, and Dfd= 25; P<0.001). (E) Serum FGF23: Error bars represent SEM. For SHAM VEH versus 56NX ASARM, there is a significant effect at all values of time, with the interaction accounting for approximately 14% of the total variance (F=12.81, DFn=1, and Dfd=28; P=0.001). For SHAM VEH versus 56NX VEH, there is no significant effect at all values of time, with the interaction accounting for approximately 9% of the total variance (F=4.21, DFn=1, and Dfd=27; P=0.05). For 56NX VEH versus 56NX ASARM, ASARM treatment no significant effect at all values of time, with the interaction accounting for 0.57% of the total variance (F=0.36, DFn=1, and Dfd=35; P=0.55). (F) Serum ALK PHOSPHATASE: Error bars represent SEM. For SHAM VEH versus 56NX VEH, there is a significant effect at all values of time, with the interaction accounting for approximately 38% of the total variance (F=30.37, DFn=1, and Dfd=23; P<0.001). For SHAM VEH versus 56NX ASARM, there is a significant effect at all values of time, with the interaction accounting for approximately 33% of the total variance (F=43.49, DFn=1, and Dfd=30; P<0.001). For 56NX VEH versus 56NX ASARM, ASARM treatment has a significant effect at all values of time, with the interaction accounting for approximately 31% of the total variance (F=34.6, DFn=1, and Dfd=33; P<0.001). (G) Serum uric acid: Error bars represent SEM. Interactions show no significant serum Ca2+ changes at all values of time for all groups. (H) Serum cholesterol: Error bars represent SEM. For SHAM VEH versus 56NX VEH, there is a significant effect at all values of time, with the interaction accounting for approximately 46% of the total variance (F=32.2, DFn=1, and Dfd=23; P<0.001). For SHAM VEH versus 56NX ASARM, there is a significant effect at all values of time, with the interaction accounting for approximately 27% of the total variance (F=12.96, DFn=1, and Dfd=27; P=0.001). For 56NX VEH versus 56NX ASARM, ASARM treatment has a significant effect at all values of time, with the interaction accounting for approximately 11% of the total variance (F=6.87, DFn=1, and Dfd=30; P=0.01). ALK-PHOSPHATASE, serum alkaline phosphatase; SERUM ALT, alanine aminotransferase. Note, results for serum aspartate aminotransferase (AST) were similar to serum alanine aminotransferase (ALT) results (C), with no significant difference between groups (data not shown for ALT).
Microcomputed Tomography
Femurs, kidneys, the heart, ascending aortae whole brain, and skin were removed at necropsy and screened using a μCT40 system (Scanco, Brüttisellen, Switzerland) as described previously (26–28,32,37,38,40,41,50) (see Figure 1B for timeline). Rat brain anatomic structure and mineral composition were defined using Omnipaque iodine (iohexitol) contrast treatment and microcomputed tomography (μCT) scanning (63).
Results
ASARM Prevents Hyperphosphatemia and Corrects Renal Function, Hypoglycemia, and Hypercholesterolemia
As expected, abnormally high serum phosphate levels occurred with 56NEPHREX rats at the start of the study and before vehicle or ASARM peptide infusion. The hyperphosphatemia increased dramatically with the vehicle-treated 56NEPHREX rats as the experiment progressed over 28 days of infusions. In contrast, increased hyperphosphatemia was prevented in 56NEPHREX rats that received ASARM peptide (Figure 2A). The corrected hyperphosphatemia occurred even though serum FGF23 levels remained high with the ASARM-treated 56NEPHREX rats (Figure 3E). Similar positive effects occurred with serum calcium, BUN, serum creatinine, potassium, bicarbonate, and protein (Figures 2, B, C, D, E, G, and 3A). A striking improvement in serum alkaline phosphatase also occurred with ASARM-treated 56NEPHREX rats compared with vehicle-treated controls (Figure 3F). Because of the high phosphate diet (HPO4; 2% Pi), elevated serum parathryroid hormone (PTH) levels also occurred with sham and 56NEPHREX rats treated with vehicle or ASARM peptide (Figure 3B). Moreover, although hypocalcemia was improved (not significant) with the 56NEPHREX ASARM-treated rats compared with 56NEPHREX vehicle rats, both 56NEPHREX groups presented with significant hypocalcemia compared with the sham rats (Figure 2B). Increased serum uric acid levels occurred for all groups after 28 days, with no significant differences (Figure 3G). Serum cholesterol levels increased strikingly, and hypoglycemia developed with 56NEPHREX vehicle rats over the 28 days. In contrast, the ASARM-treated 56NEPHREX rat serum cholesterol and serum glucose remained unchanged and were not significantly different from sham vehicle rats (Figure 3, D and H). No significant differences between the three groups occurred for serum aspartate aminotransferase (AST) and alanine aminotransferase, Figure 3C (data for AST not shown). Both enzymes (alanine aminotransferase and AST) are clinical indicators of liver health.
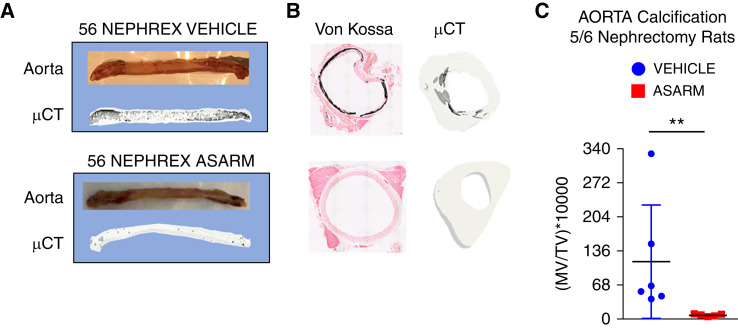
ASARM Peptide Prevents Calcification of the Heart and Medial Calcification of the Aorta
Measurements of calcification of the hearts and aortae using μCT and Von Kossa staining of histology sections revealed major improvements. The HPO4 diet caused severe cardiac calcification with 56NEPHREX rats infused with vehicle (Figure 4). With sham rats, no cardiac calcification was observed. Low-level tracheal calcification occurred with all three groups. A significant and dramatic reduction in cardiac calcification occurred with 56NEPHREX rats infused with ASARM peptide when compared with vehicle-treated 56NEPHREX rats (Figure 4). Severe medial calcification of the aorta also occurred with 56NEPHREX rats infused with vehicle. With sham rats, no calcification of the aorta occurred (Figure 5). A significant and dramatic reduction in medial calcification of the aortae occurred with the 56NEPHREX rats infused with ASARM peptide (Figure 5). No measurable aorta calcification occurred with the sham vehicle rats (data not shown). The dramatically suppressed medal calcification of the aortae in ASARM-treated 56NEPHREX rats is consistent with the observed suppression of cardiac calcification (Figures 4 and 5).
Figure 4.
μCT scans of rat hearts, showing significantly reduced heart calcification in ASARM infused subtotal 56NEPHREX rats fed a high phosphate diet (HPO4; 2% P, 2000 IU vitamin D, and 0.8% Ca; #170496; Teklad). (A) Representative heart μCT scans. (B) Graphical output of heart mineral volume over total tissue volume (MV/TV). No measurable calcification occurred in sham vehicle rats (data not shown). SHAMX, sham operated. One-way ANOVA with Tukey multiple comparison; *P=0.003; ns, not significant—as shown on the graph. Note with an unpaired t test (two-tailed), the SHAM group and 56NEPHREX ASARM group were significantly different (P=0.03). Also, the 56NEPHREX vehicle versus 56NEPHREX ASARM group remained significantly different with an unpaired t test (two-tailed; P=0.004). μCT, microcomputed tomography.
Figure 5.
ASARM infusion prevents medial calcification of the aorta. (A) Representative aorta photos and corresponding μCT scans of vehicle and ASARM-treated subtotal 56NEPHREX rats. (B) Aorta histology sections (7 μM) stained with Von Kossa (black color, mineral). Note extensive medial calcification is prevented in rats infused with ASARM peptide. (C) Graph showing aorta MV/TV calculated using μCT analysis is reduced in rats infused with ASARM. No measurable calcification occurred in SHAM vehicle rats (data not shown). Unpaired t test (two-tailed). The 56NEPHREX vehicle versus 56NEPHREX ASARM were significantly different P=0.02. No detectable mineral deposition occurred with the SHAM-treated rats (not shown). Rats were fed the high phosphate diet (HPO4; 2% P, 2000 IU vitamin D, and 0.8% Ca; #170496; Teklad).
ASARM Peptide Suppresses Renal Calcification in CKD-MBD rats
Kidneys removed at necropsy after 28 days were scanned using μCT and Von Kossa staining of histologic sections. Significant calcification of the kidneys occurred in sham and both 56NEPHREX groups fed the HPO4 diet (Figure 6). Specifically, the sham rats fed the high-phosphate diet maintained normophosphatemia by presumably increasing renal phosphate excretion that predisposed to extensive renal mineralization (Figure 2A). Consistent with the improved renal function observed in 56NEPHREX rats treated with ASARM peptide, a significant reduction in renal calcification occurred (Figure 6). Given ASARM peptide corrected the hyperphosphatemia, the decrease in renal calcification is a significant finding and consistent with the improved renal function (Figures 2, A, C, D, and 6). Notably, the significant decrease in ASARM-treated rat renal mineralization and relative hypophosphatemia compared with vehicle-treated rats suggest an ASARM-mediated inhibition of intestinal phosphate uptake may play a major role (Figure 6).
Figure 6.
ASARM peptide infusion decreases renal mineralization in 56NEPHREX rats fed a high phosphate diet ((HPO4; 2% P, 2000 IU Vitamin D and 0.8% Ca; Teklad #170496)). (A) Representative μCT scans of SHAM-operated and 56NEPHREX rats. (B) Graphical presentation of μCT quantification of renal total MV/TV. (C) Renal histology section stained with Von Kossa showing mineral deposition (black color) in 7-μM section of 56NEPHREX rat vehicle-treated kidney (×20). Note significant decreased calcification in ASARM-treated 56NEPHREX rats. Results for the one-way ANOVA with Tukey multiple comparison test were: 56NEPHREX vehicle versus 56NEPHREX ASARM P<0.001 and 56NEPHREX vehicle versus SHAM P<0.001. Statistics shown on the graph represent unpaired t test (two tailed). No significant difference occurred between 56NEPHREX ASARM versus SHAM groups for both tests.
ASARM Peptide Suppresses Nodular Ulcerative Skin Lesions in CKD-MBD Rats Fed a High Phosphate Diet
The HPO4 diet used in this study (2% PO4; nonphytates) induced skin abnormalities that appeared as indurations, nodular plaques (“peau d’orange” aspect), and pre-ulcerative lesions in vehicle-treated 56NEPHREX rats (Figure 7). The lesions occurred on the flank, abdomen, and rear area of the affected rats (Figure 7C). Strikingly, no lesions were detected with sham rats treated with vehicle or 56NEPHREX rats treated with ASARM peptide. Analysis of 1 cm−2 sections of skin sections by μCT and histology showed heavy medial mineralization of skin blood vessels (Figure 7, A, B, D, E, and F). Quantitative analysis by μCT confirmed a significant dermal calcification with 56NEPHREX rats treated with vehicle versus sham vehicle and 56NEPHREX rats treated with ASARM (Figure 7G). Calcification of skin blood vessels is characteristic of calcific uremic arteriolopathy (calciphylaxis) but not sufficient to diagnose the disease definitively (64). Specifically, calciphylaxis skin biopsy sections typically show the presence of fine arteriolar and interstitial calcifications. Importantly, a combination of medial calcification and thrombosis in ESKD is considered diagnostic for calciphylaxis and occurs rarely in individuals without kidney disease (64). Also, 70%–80% of lesions in patients with ESKD have a central distribution (abdomen and thighs) (65,66) compared with approximately 50% of lesions in patients who do not have ESKD (66,67). Of note, multiple skin vascular lesions occurring in the 56NEPHREX vehicle rats consisted of thrombi or arteriolar occlusion with extensive atherosclerotic occlusions with a central distribution (abdomen and thighs) (Figure 7, Supplemental Figure 1, A and B). Extravascular dermal calcifications also occurred with significantly increased expression of TGF-β (Supplemental Figures 1C and 2). These lesions did not occur with sham or 56NEPHREX rats treated with ASARM peptide, and TGF-β expression was not significantly different between ASARM-treated 56NEPHREX rats and sham vehicle (Supplemental Figures 1 and 2). TGF-β plays a key role in VC and regulates the osteoblastic differentiation of vascular smooth-muscle cells (VSMCs) (68). Also, TGF-β signaling cascade induces fibrosis. Thus, ASARM peptide inhibition of TGF-β may partly explain the improved renal function (69). No significant difference in expression of TIMP or α smooth-muscle actin occurred between each of the groups (Supplemental Figure 3). Given the context of renal failure and the clinical pathology, the skin abnormalities occurring in 56NEPHREX vehicle rats are likely due to calcific uremic arteriolopathy or calciphylaxis.
Figure 7.
Skin subdermal VC is significantly reduced in 56NEPHREX rats infused with ASARM—calciphylaxis-like lesions are absent. (A) μCT skin scan showing subdermal calcified blood vessels. (B) Corresponding photograph showing “under skin” surface and blood vessels scanned in (A). (C) Photo showing upper skin surface with “peau d’orange” lesions that resemble calciphylaxis lesions. (D) Cross-sectional μCT x-ray “dicom” image showing evidence of medial VCs. (E and F) Corresponding histology—skin cross-section (7 μm) stained for mineral with Von Kossa (black color, mineral) ×20. (G) Graphical presentation of μCT quantification of skin total MV/TV. Results for the one-way ANOVA with Tukey multiple comparison test were: 56NEPHREX vehicle versus 56NEPHREX ASARM P=0.02 and SHAM versus 56NEPHREX vehicle P=0.04. Unpaired t test (two tailed) for the 56NEPHREX vehicle versus 56NEPHREX ASARM were also significantly different (P=0.02). No significant difference between SHAM and 56NEPHREX ASARM groups were measured for both tests. Rats were fed the high phosphate diet (HPO4; 2% P, 2000 IU vitamin D, and 0.8% Ca; #170496; Teklad). VC, vascular calcification.
ASARM Peptide Suppresses Rat Jejunum NPT2B Phosphate Transporter Expression
Progressive systemic and hormonal changes in CKD-MBD should result in a reduced fractional intestinal phosphorus absorption that compensates for impaired renal phosphorus excretion. However, CKD animal models show a failure of the intestines to compensate for hyperphosphatemia. Indeed, intestinal phosphate uptake is not statistically different between CKD rats and controls, with no change in NPT2B mRNA expression (70,71). Of relevance, a marked correction in CKD-associated hyperphosphatemia occurs in “adenine-induced” uremic Npt2b-knockout mice compared with uremic wild-type control mice (72). The same study showed an improved survival of the uremic Npt2b knockout mice compared with uremic wild-type mice (72). Our studies suggest synthetic ASARM peptide is a useful treatment for the intestinal maladaptation by inhibiting intestinal phosphate uptake and VC (Supplemental Figure 4). Also, our studies and others show administration of MEPE protein or ASARM peptide inhibits renal and intestinal phosphate. ASARM inhibition of phosphate uptake occurs in rats and mice in vitro, ex vivo, and in vivo (32,38,42,44–46,73). Of relevance, we show for the first time phosphorylated synthetic ASARM peptide (2.2 kDa) specifically inhibits intestinal phosphate uptake (Supplemental Figure 4). The ASARM peptide inhibition of intestinal phosphate uptake is consistent with previous studies that showed intact full-length recombinant MEPE protein (60 kDa) inhibits intestinal phosphate uptake (MEPE protein contains an ASARM motif) (45). Thus, our research has discovered the defined MEPE protein sequence responsible for the inhibition of intestinal phosphate uptake (C-terminal ASARM peptide/motif). The role of PiT1 and PiT2 transporters is addressed in the discussion.
ASARM Peptide Suppresses Brain Calcification in CKD-MBD Rats Fed a High Phosphate Diet
Vehicle-treated 56NEPHREX rats developed brain calcifications (Supplemental Figures 5 and 6). Several calcified regions were identified using Allen’s Brain Atlas. These included the globus pallidus, dentate nucleus, pons, putamen, superior colliculus, primary motor area, angular insular area, entorhinal area, vermal regions, and frontal lobes. Using an iodine, iohexitol contrast agent (Omnipaque) and μCT 3D high resolution (6 μM) imaging, regions of calcification were mapped to specific brain structures. Sham-operated and ASARM-treated NEPHX rats showed major and significant reductions in brain mineral deposits (Supplemental Figures 5 and 6). Of note, phosphate is reported to play a major role in the cognitive impairment and other neurologic disorders occurring in CKD (14,15).
ASARM Infusion Corrects ROD in 56NEPHREX Rats
56NEPHREX rats fed a high phosphate diet (HPO4) develop significant bone mineralization defects with reduced bone volume/total volume ratios (Supplemental Figure 7). Also, significantly increased serum alkaline phosphatase occurs consistent with progressive renal failure and CKD-MBD sequelae (Figure 3F). In contrast, significant corrections in bone quality and serum alkaline phosphatase levels occurred with 56NEPHREX rats infused with ASARM peptide (Figure 3F, Supplemental Figure 7). Of note, PTH levels increased for all three groups, including the sham vehicle rats, and no significant differences occurred (Figure 3B). The increased expression of PTH in the sham vehicle group is due to the high phosphate diet.
Alport Mice and Patients with ESKD Have Decreased Serum ASARM Peptide
ASARM peptides derived from osteocyte-expressed extracellular matrix SIBLING proteins (MEPE and DMP1) play a key role in mineralization and FGF23 expression. In CKD-MBD, downregulation of DMP1 and MEPE occurs and correlates with FGF23. Notably, transgenic DMP1 mice crossed with Alport mice (Col4a3−/−/trgDMP1) show corrected CKD-MBD abnormalities (74). Using ELISA, we measured circulating ASARM peptides in patients with ESKD (increased FGF23) and Alport mice (an established murine CKD-MBD model). In both cases, a significant decrease in ASARM peptides occurred compared with healthy, sex- and age-matched controls (Table 1). Consistent with the reduced levels of ASARM peptide, PHEX, a zinc metalloendopeptidase, is reportedly markedly upregulated in CKD (75). Specifically, ASARM peptide is the only known physiologic substrate for PHEX (32,38,40,41,47,48,76,77).
Table 1.
Decreased serum ASARM peptides in Alport mice (Col4a3−/−) and ESKD patients
| Serum ASARM peptide ng/ml | |||
|---|---|---|---|
| Mice | Humans | ||
| WT Mice (Col4a3+/+) | Alport Mice (Col4a3−/−) | Control Subjects | ESKD Patients |
| 388.2±174.3a | 166.2±93.04a | 213.7±30.46b | 176.3±15.32b |
Data shown as mean±SD. Decreased serum ASARM peptides occur in mice and patients with CKD-MBD. Sera from 12-wk-old male Alport mice (Col4a−/− and Col4a+/+, strain 129-Col4a3tm1De) (52) and CKD-MBD patients on dialysis with ESKD were assayed for ASARM peptides. ESKD patients consisted of 14 men (65±12 yr old; White), and human controls consisted of seven men (58±15 yr old; White). Murine wild type versus null serum values for Pi, Ca, FGF23, and BUN were all significantly different, confirming advanced renal disease. Specifically, values for wild-type mice (Col4a+/+) were Pi=6.7±0.5 mg/dl, Ca2+=8.7±0.5 mg/dl, FGF23=236.3±23.2 pg/dl, creatinine=0.44±0.05 mg/dl, and BUN=62.3±3.9 mg/dl. Serum values for null mice (Col4a−/−) were Pi=17.8±2.4 mg/dl, Ca2+=7.3±0.3 mg/dl, FGF23=5407±1269 pg/dl, creatinine=1.27±0.23 mg/dl, and BUN=126.9±50.8 mg/dl. CKD-MBD, CKD mineralization bone disorder.
P<0.04 Alport (Col4a3−/−; n=5) versus wild type (Col4a3+/+; n=5).
P=0.001 ESKD patients (n=14) versus control subjects (Col4a3+/+; n=7).
Discussion
Abnormal and progressive changes in serum phosphorus, calcium, PTH, and FGF23 occur in CKD-MBD (1–3). The clinical management of these changes are key in preventing VC, ROD, and related debilitating clinical sequelae such as hypertension and LVH. Hyperphosphatemia is a key abnormality occurring at a later end stage point of the renal disease. The appearance of hyperphosphatemia correlates strongly with VC and an increased morbidity/mortality in patients with CKD (17–21). However, there are no randomized controlled trials confirming a causal link of hyperphosphatemia to patient outcomes. Once CKD progresses to an advanced stage, the current treatments available are dialysis or renal transplantation. Both treatment choices reduce quality of life and/or involve major health care costs (78). Importantly, therapies aimed at lowering phosphate uptake (phosphate binders) do not prevent progression of early stage CKD to ESKD. Indeed, there is no cure to prevent CKD progression other than identifying and controlling the disorders that caused the kidney damage in individual patients. So, it is important to identify a novel therapeutic target to treat elderly and early/late stage CKD patients to prevent nephron loss and VC.
Of relevance, abnormal protease activity occurs in patients with CKD and likely impairs the release of ASARM peptides from DMP1 and MEPE ASARM motifs (79–82). The ASARM motif is cleaved and released as a phosphorylated peptide (ASARM peptide) from a family of proteins (including MEPE, DMP1, osteopontin, DSPP, and statherin) called SIBLING proteins (26–48). Also, released ASARM peptides are highly resistant to proteases, and a marked upregulation of the only enzyme shown to cleave ASARM peptide, “PHEX” (32,38,40,41,47,48,76,77), occurs in CKD (75). Further, ASARM peptide bioactivity requires serine phosphorylation and FAM20C bone kinase plays an important role in this regard (83–85). Of relevance to treating CKD sequelae, ASARM peptide binds to mineral (hydroxyapatite and calciprotein complexes), inhibits mineralization, inhibits osteogenesis, and inhibits renal and intestinal phosphate uptake (in vivo, ex vivo, and in vitro) (25–48). Further, ASARM peptides likely inhibit VC by their potent anti-osteogenic properties (38). Notably, ASARM peptide significantly decreases osteoblast numbers and marker expression (runx2, osterix, msx2, alkaline phosphatase, and osteocalcin) in murine bone marrow stromal cell culture experiments (38). Co-incubating with an inhibitor of ASARM peptide (SPR4) reverses ASARM peptide bioactivity (38). Importantly, our in vivo studies using MEPE transgenic and HYP mice show ASARM peptide(s) are potent anti-(re)modeling molecules involved actively in skeletogenesis and induce bone-renal angiogenesis by stimulating VEGF secretion (37). Also, the ASARM motif is important for regulating expression of FGF23 (a phosphaturic cytokine) (86) in vitro and in vivo; however, its role in CKD-MBD is unclear. Of relevance, a reduced expression of DMP1 (an ASARM motif containing SIBLING protein) occurs in Alport mice (Col4a3−/−) (74), an established model of CKD-MBD with increased FGF23 (74). Notably, Alport transgenic mice that overexpress DMP1 (Col4a3−/−/trgDMP1) have reduced FGF23 with improved CKD-MBD pathology (74). Further, in this study, we show Alport mice and patients with ESKD develop significantly reduced levels of ASARM peptides (Table 1). Therefore, the data compellingly support that abnormal post-translational processing and expression of ASARM peptide/motif occurring in CKD-MBD is likely etiologic to the procalcific phenotype.
Several studies show LVH associates with increased levels of FGF23 and cardiovascular disease in CKD-MBD patients (87–90). Of note, X-linked hypophosphatemic rickets patients and mice (XLH) have high levels of FGF23 (91,92). The definition of LVH in XLH remains controversial because of abnormalities in body surface area and height, and it is difficult to differentiate primary defects in XLH from complications of treatment (91,92). Also, some clinical studies conclude there is no relationship between circulating FGF23 and cardiovascular outcomes for CKD-MBD and XLH (93,94). Moreover, XLH-mice with excessively high levels of FGF23, increased serum ASARM peptides, and hypophosphatemia do not develop LVH and cardiovascular hypertrophy (95,96). This suggests FGF23 and associated cardiovascular/LVH effects are likely co-dependent on ASARM peptide and or hyperphosphatemia. Further support for the involvement of ASARM peptide is our finding that serum ASARM peptide levels are reduced in CKD-MBD patients and Alport mice (Table 1). Also, ASARM peptide treatment of 56NEPHREX rats corrects cardiovascular abnormalities and hyperphosphatemia without reducing the increased FGF23 levels (Figures 2 and 3). Specifically, although speculative, the findings suggest that a combination of high FGF23 and low ASARM peptide with high or inadequately controlled levels of serum Pi are optimal for the progression of cardiovascular disease and LVH in CKD-MBD.
Another possible drug target for ASARM peptides in CKD-MBD is calciprotein particles (CPP1 and CPP2). Calciprotein precipitates (CPP) are cytotoxic to the vascular endothelium and induce a phenotypic transformation of VSMC to osteoblast-like (bone-forming) cells. The CPP-induced osteoblast-like cells deposit hydroxyapatite in the arterial wall and are thus an early step in the pathophysiology of VC in CKD (97–100). Importantly, CPP concentrations increase with CKD progression and correlate with arterial calcification burden and overall vessel stiffness (101,102). Because CPPs contain amorphous mineral (CPP1) and crystalline hydroxyapatite (CPP2), ASARM peptide likely binds to CPP1 and CPP2 particles. Binding of ASARM peptide to CPPs may prevent CPP transition (CPP1 to CPP2) and thus sterically inhibit CPP2-induced VC. Further studies are needed to confirm ASARM peptide CPP interactions.
In summary, the data suggests the positive effects of ASARM peptide treatment on VC and CKD disease progression are mediated by (1) binding to and inhibiting mineral and calciprotein particles (CPP1/2), (2) directly suppressing CPP-mediated osteogenic phenotypic transition of VSMCs, and (3) inhibiting intestinal and renal phosphate uptake. Figure 8 illustrates a model that explains how ASARM peptide(s) likely prevents renal disease and VC. Specifically, the inappropriately low ASARM peptide levels in CKD likely contributes to the VC and disease sequelae. The positive effects of ASARM on CKD-MBD bone quality are serendipitous but require further study to confirm if the beneficial effect occurs under normal phosphate diet conditions.
Figure 8.
Role of ASARM peptides in CKD. CKD causes progressive hyperphosphatemia, predisposing to nanocrystal or calciprotein particle (CPP) formation. CPPs and high Pi interact with vascular smooth muscle cells (VSMC), inducing proliferation and transition to an osteoprogenitor phenotype. Osteogenic transition continues with the genesis of matrix vesicles and microcrystalline nidi of hydroxyapatite. VC proceeds through intimal and medial mineral deposition and growth. ASARM peptides inhibit renal and intestinal Pi uptake, reducing serum Pi (A), bind to CPPs (B), inhibit osteogenesis and VSMC phenotypic transition (C), and binding to and preventing the growth of hydroxyapatite and mineral (D). In CKD, defective expression of SIBLING proteins, altered proteolytic processing to ASARM peptides, altered phosphorylation and/or altered degradation (colored arrow) results in reduced levels of active ASARM peptides, thus promoting VC. For example, ASARM peptide is the only known physiologic substrate for PHEX (32,38,40,41,47,48,76,77) and PHEX is highly expressed in CKD (75). The reduced levels of ASARM peptides in CKD predisposes to VC because of the reduced inhibition of VC pathways.
The pathways regulating VC and bone tissue mineralization are similar, and this poses a problem when defining targets to prevent cardiovascular disease. Specifically, preventing the undesirable VC outcome in CKD-MBD might have adverse effects on bone or worsen the ROD. Notably, ASARM peptide is an effective VC inhibitor but also induces osteomalacia, inhibits osteogenesis, and suppresses bone turnover. Serendipitously, this study shows ASARM peptide treatment of 56NEPHREX rats on a high phosphate diet improves bone quality. The dissonant finding of improvement in bone quality and reduced VC is not unprecedented and occurs with bone morphogenetic protein 7 treatment of murine models of CKD and atherosclerotic VC (103,104). Notably, bone morphogenetic protein 7 treatment of CKD rats reduces intimal calcification through lowering phosphate by stimulating bone formation. Importantly, orthotopic bone mineralization patterned by decreased bone formation or increased bone formation has marked effects for heterotopic bone formation in CKD-MBD. Notably, the bone mineral abnormalities in CKD-MBD are complex and consist of high bone turnover and low bone turnover disease. Specifically, a dynamic continuum between bone turnover, mineralization, and bone volume orchestrates bone quality. The composite outcomes for renal osteodystrophy ranges from adynamic bone disease, osteomalacia, osteitis fibrosa, mixed uremic osteodystrophy, or mild hyperparathyroid-related bone disease. Thus, for 56NEPHREX rats fed a high phosphate diet, ASARM treatment may improve bone quality by reducing high turnover disease. The precise mechanism of ASARM peptide positive effects on bone in CKD-MBD will require further studies.
ASARM treatment significantly corrected the hypoglycemia and hypercholesterolemia (Figure 3, D and H) that occurred with the 56NEPHREX vehicle rats and there were no significant differences in food intake (data not shown). Intriguingly, a markedly decreased fat mass and increased serum ASARM peptides occur in X-linked hypophosphatemic mice (HYP) (28). Also, infusion of HYP mice with SPR4 peptide, a peptide that neutralizes ASARM peptide reverses the low-fat mass phenotype (28). Notably, SPR4 treatment (an ASARM peptide inhibitor) reverses HYP mice hyperglycemia, hypoleptinemia, hypoinsulinemia, and reduced serum adiponectin (28,105–107). Of relevance to cardiovascular disease, a reduction in adipose differentiation-related protein (Plin2) occurs in HYP mice and plays a major role in atherosclerotic intimal plaque production from VSMCs (108,109).
In summary, our study shows administration of ASARM peptide suppresses VC, calciphylaxis-like lesions, and kidney disease progression in CKD-MBD rats by preventing soft-tissue mineralization (brain, kidneys, and skin). Also, ASARM peptide treatment significantly improves bone quality in 56NEPHREX rats fed a high phosphate diet. Although ASARM treatment in 56NEPHREX rats fed a high phosphate diet improved CKD-MBD sequelae, we did not test a normal phosphate diet. Another limitation is the 56NEPHREX rat model induces an acute form of CKD-MBD and may not replicate fully the human disease. Future clinical studies will determine whether progressive changes in ASARM levels and post-translational modifications play a major role in CKD-MBD and ESKD pathology. Although the molecular model presented is compelling, the signaling pathways involved remain unknown. Published studies implicate sclerostin and the Wnt/β-catenin pathway in transducing ASARM peptide activity (26,27,33,37,39). Our study measured jejunum NPT2b (slc34a2) but not Pit1 or Pit2 Na+-dependent phosphate cotransporter expression. Specifically, future functional analyses of phosphate uptake that includes NPT2b, PiT1, and PiT2 across the entire intestinal structure will provide mechanistic answers on ASARM intestinal phosphate uptake in CKD-MBD. Also, previous studies showed wild-type mice treated with ASARM peptide develop a renal phosphate leak with increased fractional excretion of phosphate and urinary phosphate clearance (32). Importantly, we did not directly measure fractional excretion of phosphate, urinary phosphate clearance, or directly measure intestinal phosphate uptake (jejunum). The significant decrease in ASARM-treated rat renal mineralization compared with vehicle-treated rats suggests ASARM-mediated inhibition of intestinal phosphate may play a major role (Figure 6). Also, the extensive renal calcification in the sham vehicle rats is due to the high phosphate diet. Specifically, we do not know whether the ASARM-suppressed hyperphosphatemia is due to inhibition of intestinal phosphate uptake, an increased renal phosphate leak, altered bone turnover, or a combination of all three.
In conclusion, this study provides the foundation for preventing adverse cardiac, bone, and renal clinical outcomes that are the primary drivers of morbidity and mortality in CKD patients. Importantly, no effective therapies exist for preventing VC and CKD progression. Because VC occurs in all forms of progressive CKD, effective therapies for preventing VC could have a significant impact on patient care and clinical outcomes.
Disclosures
E.M. McCarthy reports research funding from Sparsentan. P.S. Rowe reports patents or royalties from the University of Kansas Medical Center. A.L. Yu reports consultancy for Calico, George Clinical, Navitor, Otsuka, Palladio, Reata, and Regulus; ownership interest in Amgen, CVS, Dialysis Associates, Express Scripts, Gilead, Ionis, Prothena, Pfizer, and Vertex; research funding from Regulus and Sanofi; honoraria from Elsevier and Wolters Kluwer; and other interests or relationships with the PKD Foundation (scientific advisory board). The remaining author has nothing to disclose.
Funding
This work was supported by funds from the National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases (R01DK111693) to P.S. Rowe and the National Institute of Diabetes and Digestive and Kidney Diseases (R01DK122212) to J.R. Stubbs.
Author Contributions
E.M. McCarthy and P.S. Rowe were responsible for visualization; E.M. McCarthy, P.S. Rowe, and J.R. Stubbs were responsible for validation; P.S. Rowe was responsible for conceptualization, funding acquisition, methodology, project administration, resources, software, and supervision, and wrote the original draft of the manuscript; P.S. Rowe and J.R. Stubbs were responsible for data curation; P.S. Rowe, J.R. Stubbs, and A.L. Yu were responsible for the investigation; and all authors were responsible for the formal analysis and reviewed and edited the manuscript.
Supplemental Material
This article contains the following supplemental material online at http://kidney360.asnjournals.org/lookup/suppl/doi:10.34067/KID.0002782022/-/DCSupplemental.
ASARM treatment prevents intimal and medial vascular mineral deposition with atherosclerotic occlusion and thrombi in 56NEPHREX rats. Download Supplemental Figure 1, PDF file, 610 KB (609.7KB, pdf) .
ASARM treatment reduces TGF-β dermal expression in 56NEPHREX rats. Download Supplemental Figure 2, PDF file, 610 KB (609.7KB, pdf) .
Skin immunohistology analyses for tissue inhibitor metalloprotease (TIMP), α smooth-muscle actin, and TGF-β expression. Download Supplemental Figure 3, PDF file, 610 KB (609.7KB, pdf) .
Decreased serum phosphate and gut jejunum Na+-dependent phosphate co-transporter NPT2B (Slc34A2) occurs with 56NEPHREX rats infused with ASARM peptide compared with rats infused with vehicle. Download Supplemental Figure 4, PDF file, 610 KB (609.7KB, pdf) .
Decreased brain calcification occurs in 56NEPHREX rats treated with ASARM peptide. Download Supplemental Figure 5, PDF file, 610 KB (609.7KB, pdf) .
Brain mineral deposits identified using μCT iodine contrast imaging (Omnipaque, iohexitol perfusion) and ATLAS brain atlas mapping. Download Supplemental Figure 6, PDF file, 610 KB (609.7KB, pdf) .
Representative μCT scans of femur diaphysis. Download Supplemental Figure 7, PDF file, 610 KB (609.7KB, pdf) .
References
- 1.Moe S, Drüeke T, Cunningham J, Goodman W, Martin K, Olgaard K, Ott S, Sprague S, Lameire N, Eknoyan G; Kidney Disease: Improving Global Outcomes (KDIGO) : Definition, evaluation, and classification of renal osteodystrophy: A position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 69: 1945–1953, 2006. 10.1038/sj.ki.5000414 [DOI] [PubMed] [Google Scholar]
- 2.Coresh J, Selvin E, Stevens LA, Manzi J, Kusek JW, Eggers P, Van Lente F, Levey AS: Prevalence of chronic kidney disease in the United States. JAMA 298: 2038–2047, 2007. 10.1001/jama.298.17.2038 [DOI] [PubMed] [Google Scholar]
- 3.Kidney Disease: Improving Global Outcomes CKD-MBD Work Group : KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int S1–S130, 2009. 10.1038/ki.2009.188 [DOI] [PubMed] [Google Scholar]
- 4.Eknoyan G, Lameire N, Barsoum R, Eckardt KU, Levin A, Levin N, Locatelli F, MacLeod A, Vanholder R, Walker R, Wang H: The burden of Kidney Disease: Improving Global Outcomes. Kidney Int 66: 1310–1314, 2004. 10.1111/j.1523-1755.2004.00894.x [DOI] [PubMed] [Google Scholar]
- 5.Johansen KL, Chertow GM, Foley RN, Gilbertson DT, Herzog CA, Ishani A, Israni AK, Ku E, Kurella Tamura M, Li S, Li S, Liu J, Obrador GT, O’Hare AM, Peng Y, Powe NR, Roetker NS, St Peter WL, Abbott KC, Chan KE, Schulman IH, Snyder J, Solid C, Weinhandl ED, Winkelmayer WC, Wetmore JB: US Renal Data System 2020 annual data report: Epidemiology of kidney disease in the United States. Am J Kidney Dis 77: A7–A8, 2021. 10.1053/j.ajkd.2021.01.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, de Jong PE, Coresh J, Gansevoort RT; Chronic Kidney Disease Prognosis Consortium : Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. Lancet 375: 2073–2081, 2010. 10.1016/S0140-6736(10)60674-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY: Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 351: 1296–1305, 2004. 10.1056/NEJMoa041031 [DOI] [PubMed] [Google Scholar]
- 8.Keith DS, Nichols GA, Gullion CM, Brown JB, Smith DH: Longitudinal follow-up and outcomes among a population with chronic kidney disease in a large managed care organization. Arch Intern Med 164: 659–663, 2004. 10.1001/archinte.164.6.659 [DOI] [PubMed] [Google Scholar]
- 9.Fox CS, Matsushita K, Woodward M, Bilo HJ, Chalmers J, Heerspink HJ, Lee BJ, Perkins RM, Rossing P, Sairenchi T, Tonelli M, Vassalotti JA, Yamagishi K, Coresh J, de Jong PE, Wen CP, Nelson RG; Chronic Kidney Disease Prognosis Consortium : Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without diabetes: A meta-analysis. Lancet 380: 1662–1673, 2012. 10.1016/S0140-6736(12)61350-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mahmoodi BK, Matsushita K, Woodward M, Blankestijn PJ, Cirillo M, Ohkubo T, Rossing P, Sarnak MJ, Stengel B, Yamagishi K, Yamashita K, Zhang L, Coresh J, de Jong PE, Astor BC; Chronic Kidney Disease Prognosis Consortium : Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without hypertension: A meta-analysis. Lancet 380: 1649–1661, 2012. 10.1016/S0140-6736(12)61272-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Anaya P, Blomquist GA, Davenport DL, Monier-Faugere MC, Sorrell VL, Malluche HH: Coronary artery calcification in CKD-5D patients is tied to adverse cardiac function and increased mortality. Clin Nephrol 86: 291–302, 2016. 10.5414/CN108940 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen J, Budoff MJ, Reilly MP, Yang W, Rosas SE, Rahman M, Zhang X, Roy JA, Lustigova E, Nessel L, Ford V, Raj D, Porter AC, Soliman EZ, Wright JT Jr, Wolf M, He J; CRIC Investigators : Coronary artery calcification and risk of cardiovascular disease and death among patients with chronic kidney disease. JAMA Cardiol 2: 635–643, 2017. 10.1001/jamacardio.2017.0363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sigrist M, Bungay P, Taal MW, McIntyre CW: Vascular calcification and cardiovascular function in chronic kidney disease. Nephrol Dial Transplant 21: 707–714, 2006. 10.1093/ndt/gfi236 [DOI] [PubMed] [Google Scholar]
- 14.Rroji M, Figurek A, Viggiano D, Capasso G, Spasovski G: Phosphate in the context of cognitive impairment and other neurological disorders occurrence in chronic kidney disease. Int J Mol Sci 23: 7362, 2022. 10.3390/ijms23137362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Miglinas M, Cesniene U, Janusaite MM, Vinikovas A: Cerebrovascular disease and cognition in chronic kidney disease patients. Front Cardiovasc Med 7: 96, 2020. 10.3389/fcvm.2020.00096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Elias RM, Dalboni MA, Coelho ACE, Moysés RMA: CKD-MBD: From the pathogenesis to the identification and development of potential novel therapeutic targets. Curr Osteoporos Rep 16: 693–702, 2018. 10.1007/s11914-018-0486-0 [DOI] [PubMed] [Google Scholar]
- 17.Chue CD, Edwards NC, Moody WE, Steeds RP, Townend JN, Ferro CJ: Serum phosphate is associated with left ventricular mass in patients with chronic kidney disease: A cardiac magnetic resonance study. Heart 98: 219–224, 2012. 10.1136/heartjnl-2011-300570 [DOI] [PubMed] [Google Scholar]
- 18.Mathew S, Tustison KS, Sugatani T, Chaudhary LR, Rifas L, Hruska KA: The mechanism of phosphorus as a cardiovascular risk factor in CKD. J Am Soc Nephrol 19: 1092–1105, 2008. 10.1681/ASN.2007070760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jono S, Shioi A, Ikari Y, Nishizawa Y: Vascular calcification in chronic kidney disease. J Bone Miner Metab 24: 176–181, 2006. 10.1007/s00774-005-0668-6 [DOI] [PubMed] [Google Scholar]
- 20.Block GA, Klassen PS, Lazarus JM, Ofsthun N, Lowrie EG, Chertow GM: Mineral metabolism, mortality, and morbidity in maintenance hemodialysis. J Am Soc Nephrol 15: 2208–2218, 2004. 10.1097/01.ASN.0000133041.27682.A2 [DOI] [PubMed] [Google Scholar]
- 21.Block GA, Hulbert-Shearon TE, Levin NW, Port FK: Association of serum phosphorus and calcium×phosphate product with mortality risk in chronic hemodialysis patients: A national study. Am J Kidney Dis 31: 607–617, 1998. 10.1053/ajkd.1998.v31.pm9531176 [DOI] [PubMed] [Google Scholar]
- 22.Xu C, Smith ER, Tiong MK, Ruderman I, Toussaint ND: Interventions to attenuate vascular calcification progression in chronic kidney disease: A systematic review of clinical trials. J Am Soc Nephrol 33: 1011–1032, 2022. 10.1681/ASN.2021101327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shroff RC, McNair R, Figg N, Skepper JN, Schurgers L, Gupta A, Hiorns M, Donald AE, Deanfield J, Rees L, Shanahan CM: Dialysis accelerates medial vascular calcification in part by triggering smooth muscle cell apoptosis. Circulation 118: 1748–1757, 2008. 10.1161/CIRCULATIONAHA.108.783738 [DOI] [PubMed] [Google Scholar]
- 24.Ruderman I, Holt SG, Hewitson TD, Smith ER, Toussaint ND: Current and potential therapeutic strategies for the management of vascular calcification in patients with chronic kidney disease including those on dialysis. Semin Dial 31: 487–499, 2018. 10.1111/sdi.12710 [DOI] [PubMed] [Google Scholar]
- 25.Rowe PS, de Zoysa PA, Dong R, Wang HR, White KE, Econs MJ, Oudet CL: MEPE, a new gene expressed in bone marrow and tumors causing osteomalacia. Genomics 67: 54–68, 2000. 10.1006/geno.2000.6235 [DOI] [PubMed] [Google Scholar]
- 26.Zelenchuk LV, Hedge AM, Rowe PS: Age dependent regulation of bone-mass and renal function by the MEPE ASARM-motif. Bone 79: 131–142, 2015. 10.1016/j.bone.2015.05.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zelenchuk LV, Hedge AM, Rowe PS: SPR4-peptide alters bone metabolism of normal and HYP mice. Bone 72: 23–33, 2015. 10.1016/j.bone.2014.11.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zelenchuk LV, Hedge AM, Rowe PS: PHEX mimetic (SPR4-peptide) corrects and improves HYP and wild type mice energy-metabolism (published correction appears in PLoS One 9: e101192, 2014). PLoS One 9: e97326, 2014. 10.1371/journal.pone.0097326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Salmon B, Bardet C, Khaddam M, Naji J, Coyac BR, Baroukh B, Letourneur F, Lesieur J, Decup F, Le Denmat D, Nicoletti A, Poliard A, Rowe PS, Huet E, Vital SO, Linglart A, McKee MD, Chaussain C: MEPE-derived ASARM peptide inhibits odontogenic differentiation of dental pulp stem cells and impairs mineralization in tooth models of X-linked hypophosphatemia. PLoS One 8: e56749, 2013. 10.1371/journal.pone.0056749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Staines KA, Mackenzie NC, Clarkin CE, Zelenchuk L, Rowe PS, MacRae VE, Farquharson C: MEPE is a novel regulator of growth plate cartilage mineralization. Bone 51: 418–430, 2012. 10.1016/j.bone.2012.06.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Opsahl Vital S, Gaucher C, Bardet C, Rowe PS, George A, Linglart A, Chaussain C: Tooth dentin defects reflect genetic disorders affecting bone mineralization. Bone 50: 989–997, 2012. 10.1016/j.bone.2012.01.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.David V, Martin A, Hedge AM, Drezner MK, Rowe PS: ASARM peptides: PHEX-dependent and -independent regulation of serum phosphate. Am J Physiol Renal Physiol 300: F783–F791, 2011. 10.1152/ajprenal.00304.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Atkins GJ, Rowe PS, Lim HP, Welldon KJ, Ormsby R, Wijenayaka AR, Zelenchuk L, Evdokiou A, Findlay DM: Sclerostin is a locally acting regulator of late-osteoblast/preosteocyte differentiation and regulates mineralization through a MEPE-ASARM-dependent mechanism. J Bone Miner Res 26: 1425–1436, 2011. 10.1002/jbmr.345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Boukpessi T, Gaucher C, Léger T, Salmon B, Le Faouder J, Willig C, Rowe PS, Garabédian M, Meilhac O, Chaussain C: Abnormal presence of the matrix extracellular phosphoglycoprotein-derived acidic serine- and aspartate-rich motif peptide in human hypophosphatemic dentin. Am J Pathol 177: 803–812, 2010. 10.2353/ajpath.2010.091231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Boskey AL, Chiang P, Fermanis A, Brown J, Taleb H, David V, Rowe PS: MEPE’s diverse effects on mineralization. Calcif Tissue Int 86: 42–46, 2010. 10.1007/s00223-009-9313-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gaucher C, Boukpessi T, Septier D, Jehan F, Rowe PS, Garabédian M, Goldberg M, Chaussain-Miller C: Dentin noncollagenous matrix proteins in familial hypophosphatemic rickets. Cells Tissues Organs 189: 219–223, 2009. 10.1159/000151382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.David V, Martin A, Hedge AM, Rowe PS: Matrix extracellular phosphoglycoprotein (MEPE) is a new bone renal hormone and vascularization modulator. Endocrinology 150: 4012–4023, 2009. 10.1210/en.2009-0216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Martin A, David V, Laurence JS, Schwarz PM, Lafer EM, Hedge AM, Rowe PS: Degradation of MEPE, DMP1, and release of SIBLING ASARM-peptides (minhibins): ASARM-peptide(s) are directly responsible for defective mineralization in HYP. Endocrinology 149: 1757–1772, 2008. 10.1210/en.2007-1205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rowe PS, Liu S, Vierthaler L, Zhou J, Quarles LD: Phosphorylated acidic serine-aspartate-rich MEPE-associated motif peptide from matrix extracellular phosphoglycoprotein inhibits phosphate regulating gene with homologies to endopeptidases on the X-chromosome enzyme activity. J Endocrinol 192: 261–267, 2007. 10.1677/joe.1.07059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rowe PS, Matsumoto N, Jo OD, Shih RN, Oconnor J, Roudier MP, Bain S, Liu S, Harrison J, Yanagawa N: Correction of the mineralization defect in hyp mice treated with protease inhibitors CA074 and pepstatin. Bone 39: 773–786, 2006. 10.1016/j.bone.2006.04.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rowe PSN, Garrett IR, Schwarz PM, Carnes DL, Lafer EM, Mundy GR, Gutierrez GE: Surface plasmon resonance (SPR) confirms that MEPE binds to PHEX via the MEPE-ASARM motif: A model for impaired mineralization in X-linked rickets (HYP). Bone 36: 33–46, 2005. 10.1016/j.bone.2004.09.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rowe PS, Kumagai Y, Gutierrez G, Garrett IR, Blacher R, Rosen D, Cundy J, Navvab S, Chen D, Drezner MK, Quarles LD, Mundy GR: MEPE has the properties of an osteoblastic phosphatonin and minhibin. Bone 34: 303–319, 2004. 10.1016/j.bone.2003.10.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bresler D, Bruder J, Mohnike K, Fraser WD, Rowe PSN: Serum MEPE-ASARM-peptides are elevated in X-linked rickets (HYP): Implications for phosphaturia and rickets. J Endocrinol 183: R1–R9, 2004. 10.1677/joe.1.05989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shirley DG, Faria NJ, Unwin RJ, Dobbie H: Direct micropuncture evidence that matrix extracellular phosphoglycoprotein inhibits proximal tubular phosphate reabsorption. Nephrol Dial Transplant 25: 3191–3195, 2010. 10.1093/ndt/gfq263 [DOI] [PubMed] [Google Scholar]
- 45.Marks J, Churchill LJ, Debnam ES, Unwin RJ: Matrix extracellular phosphoglycoprotein inhibits phosphate transport. J Am Soc Nephrol 19: 2313–2320, 2008. 10.1681/ASN.2008030315 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dobbie H, Unwin RJ, Faria NJ, Shirley DG: Matrix extracellular phosphoglycoprotein causes phosphaturia in rats by inhibiting tubular phosphate reabsorption. Nephrol Dial Transplant 23: 730–733, 2008. 10.1093/ndt/gfm535 [DOI] [PubMed] [Google Scholar]
- 47.Addison WN, Masica DL, Gray JJ, McKee MD: Phosphorylation-dependent inhibition of mineralization by osteopontin ASARM peptides is regulated by PHEX cleavage. J Bone Miner Res 25: 695–705, 2010 [DOI] [PubMed] [Google Scholar]
- 48.Addison WN, Nakano Y, Loisel T, Crine P, McKee MD: MEPE-ASARM peptides control extracellular matrix mineralization by binding to hydroxyapatite: An inhibition regulated by PHEX cleavage of ASARM. J Bone Miner Res 23: 1638–1649, 2008. 10.1359/jbmr.080601 [DOI] [PubMed] [Google Scholar]
- 49.Hoyer JR, Asplin JR, Otvos L: Phosphorylated osteopontin peptides suppress crystallization by inhibiting the growth of calcium oxalate crystals. Kidney Int 60: 77–82, 2001. 10.1046/j.1523-1755.2001.00772.x [DOI] [PubMed] [Google Scholar]
- 50.Rowe PS, Zelenchuk LV, Laurence JS, Lee P, Brooks WM, McCarthy ET: Do ASARM peptides play a role in nephrogenic systemic fibrosis? Am J Physiol Renal Physiol 309: F764–F769, 2015. 10.1152/ajprenal.00201.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.National Research Council : Guide for the Care and Use of Laboratory Animals, Washington, DC, National Institutes of Health, 2011 [Google Scholar]
- 52.Stubbs JR, He N, Idiculla A, Gillihan R, Liu S, David V, Hong Y, Quarles LD: Longitudinal evaluation of FGF23 changes and mineral metabolism abnormalities in a mouse model of chronic kidney disease. J Bone Miner Res 27: 38–46, 2012. 10.1002/jbmr.516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Griffin KA, Picken M, Bidani AK: Method of renal mass reduction is a critical modulator of subsequent hypertension and glomerular injury. J Am Soc Nephrol 4: 2023–2031, 1994. 10.1681/ASN.V4122023 [DOI] [PubMed] [Google Scholar]
- 54.Zhang Y, Kompa AR: A practical guide to subtotal nephrectomy in the rat with subsequent methodology for assessing renal and cardiac function. Nephrology (Carlton) 19: 552–561, 2014. 10.1111/nep.12279 [DOI] [PubMed] [Google Scholar]
- 55.Chang AR, Anderson C: Dietary phosphorus intake and the kidney. Annu Rev Nutr 37: 321–346, 2017. 10.1146/annurev-nutr-071816-064607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lee AW, Cho SS: Association between phosphorus intake and bone health in the NHANES population. Nutr J 14: 28, 2015. 10.1186/s12937-015-0017-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Calvo MS, Uribarri J: Public health impact of dietary phosphorus excess on bone and cardiovascular health in the general population. Am J Clin Nutr 98: 6–15, 2013. 10.3945/ajcn.112.053934 [DOI] [PubMed] [Google Scholar]
- 58.Larsson TE, Olauson H, Hagström E, Ingelsson E, Arnlöv J, Lind L, Sundström J: Conjoint effects of serum calcium and phosphate on risk of total, cardiovascular, and noncardiovascular mortality in the community. Arterioscler Thromb Vasc Biol 30: 333–339, 2010. 10.1161/ATVBAHA.109.196675 [DOI] [PubMed] [Google Scholar]
- 59.Barnett V, Lewis T: Outliers in Statistical Data, 3rd Ed., New York, John Wiley & Sons, Inc., 1994 [Google Scholar]
- 60.Iglewicz B, Hoaglin DC: How to Detect Outliers, Vol. 16, Wisconsin, ASQ Quality Press, 1993 [Google Scholar]
- 61.Jain A, Fedarko NS, Collins MT, Gelman R, Ankrom MA, Tayback M, Fisher LW: Serum levels of matrix extracellular phosphoglycoprotein (MEPE) in normal humans correlate with serum phosphorus, parathyroid hormone and bone mineral density. J Clin Endocrinol Metab 89: 4158–4161, 2004. 10.1210/jc.2003-032031 [DOI] [PubMed] [Google Scholar]
- 62.Yuan B, Takaiwa M, Clemens TL, Feng JQ, Kumar R, Rowe PS, Xie Y, Drezner MK: Aberrant Phex function in osteoblasts and osteocytes alone underlies murine X-linked hypophosphatemia. J Clin Invest 118: 722–734, 2008. 10.1172/JCI32702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Saito S, Murase K: Ex vivo imaging of mouse brain using micro-CT with non-ionic iodinated contrast agent: A comparison with myelin staining. Br J Radiol 85: e973–e978, 2012. 10.1259/bjr/13040401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ellis CL, O’Neill WC: Questionable specificity of histologic findings in calcific uremic arteriolopathy. Kidney Int 94: 390–395, 2018. 10.1016/j.kint.2018.03.016 [DOI] [PubMed] [Google Scholar]
- 65.Nigwekar SU, Zhao S, Wenger J, Hymes JL, Maddux FW, Thadhani RI, Chan KE: A nationally representative study of calcific uremic arteriolopathy risk factors. J Am Soc Nephrol 27: 3421–3429, 2016. 10.1681/ASN.2015091065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Weenig RH, Sewell LD, Davis MD, McCarthy JT, Pittelkow MR: Calciphylaxis: Natural history, risk factor analysis, and outcome. J Am Acad Dermatol 56: 569–579, 2007. 10.1016/j.jaad.2006.08.065 [DOI] [PubMed] [Google Scholar]
- 67.Nigwekar SU, Wolf M, Sterns RH, Hix JK: Calciphylaxis from nonuremic causes: A systematic review. Clin J Am Soc Nephrol 3: 1139–1143, 2008. 10.2215/CJN.00530108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kanno Y, Into T, Lowenstein CJ, Matsushita K: Nitric oxide regulates vascular calcification by interfering with TGF- signalling. Cardiovasc Res 77: 221–230, 2008. 10.1093/cvr/cvm049 [DOI] [PubMed] [Google Scholar]
- 69.Zeisberg M, Bottiglio C, Kumar N, Maeshima Y, Strutz F, Müller GA, Kalluri R: Bone morphogenic protein-7 inhibits progression of chronic renal fibrosis associated with two genetic mouse models. Am J Physiol Renal Physiol 285: F1060–F1067, 2003. 10.1152/ajprenal.00191.2002 [DOI] [PubMed] [Google Scholar]
- 70.Vorland CJ, Biruete A, Lachcik PJ, Srinivasan S, Chen NX, Moe SM, Hill Gallant KM: Kidney disease progression does not decrease intestinal phosphorus absorption in a rat model of chronic kidney disease-mineral bone disorder. J Bone Miner Res 35: 333–342, 2019. 10.1002/jbmr.3894 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Marks J, Churchill LJ, Srai SK, Biber J, Murer H, Jaeger P, Debnam ES, Unwin RJ; Epithelial Transport and Cell Biology Group : Intestinal phosphate absorption in a model of chronic renal failure. Kidney Int 72: 166–173, 2007. 10.1038/sj.ki.5002292 [DOI] [PubMed] [Google Scholar]
- 72.Schiavi SC, Tang W, Bracken C, O’Brien SP, Song W, Boulanger J, Ryan S, Phillips L, Liu S, Arbeeny C, Ledbetter S, Sabbagh Y: Npt2b deletion attenuates hyperphosphatemia associated with CKD. J Am Soc Nephrol 23: 1691–1700, 2012. 10.1681/ASN.2011121213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Marks J, Churchill LJ, Debnam ES, Unwin RJ: Matrix extracellular phosphoglycoprotein inhibits phosphate transport. J Am Soc Nephrol 19: 2313–2320, 2008. 10.1681/ASN.2008030315PMC2588094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Dussold C, Gerber C, White S, Wang X, Qi L, Francis C, Capella M, Courbon G, Wang J, Li C, Feng JQ, Isakova T, Wolf M, David V, Martin A: DMP1 prevents osteocyte alterations, FGF23 elevation and left ventricular hypertrophy in mice with chronic kidney disease. Bone Res 7: 12, 2019. 10.1038/s41413-019-0051-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Graciolli FG, Neves KR, Barreto F, Barreto DV, Dos Reis LM, Canziani ME, Sabbagh Y, Carvalho AB, Jorgetti V, Elias RM, Schiavi S, Moysés RMA: The complexity of chronic kidney disease-mineral and bone disorder across stages of chronic kidney disease. Kidney Int 91: 1436–1446, 2017. 10.1016/j.kint.2016.12.029 [DOI] [PubMed] [Google Scholar]
- 76.Rowe PS: Regulation of bone-renal mineral and energy metabolism: The PHEX, FGF23, DMP1, MEPE ASARM pathway. Crit Rev Eukaryot Gene Expr 22: 61–86, 2012. 10.1615/CritRevEukarGeneExpr.v22.i1.50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Campos M, Couture C, Hirata IY, Juliano MA, Loisel TP, Crine P, Juliano L, Boileau G, Carmona AK: Human recombinant endopeptidase PHEX has a strict S1′ specificity for acidic residues and cleaves peptides derived from fibroblast growth factor-23 and matrix extracellular phosphoglycoprotein. Biochem J 373: 271–279, 2003. 10.1042/bj20030287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Levin A, Tonelli M, Bonventre J, Coresh J, Donner JA, Fogo AB, Fox CS, Gansevoort RT, Heerspink HJL, Jardine M, Kasiske B, Köttgen A, Kretzler M, Levey AS, Luyckx VA, Mehta R, Moe O, Obrador G, Pannu N, Parikh CR, Perkovic V, Pollock C, Stenvinkel P, Tuttle KR, Wheeler DC, Eckardt KU; ISN Global Kidney Health Summit participants : Global kidney health 2017 and beyond: A roadmap for closing gaps in care, research, and policy. Lancet 390: 1888–1917, 2017. 10.1016/S0140-6736(17)30788-2 [DOI] [PubMed] [Google Scholar]
- 79.Owens EP, Vesey DA, Kassianos AJ, Healy H, Hoy WE, Gobe GC: Biomarkers and the role of mast cells as facilitators of inflammation and fibrosis in chronic kidney disease. Transl Androl Urol 8[Suppl 2]: S175–S183, 2019. 10.21037/tau.2018.11.03 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wolke C, Teumer A, Endlich K, Endlich N, Rettig R, Stracke S, Fiene B, Aymanns S, Felix SB, Hannemann A, Lendeckel U: Serum protease activity in chronic kidney disease patients: The GANI_MED renal cohort. Exp Biol Med (Maywood) 242: 554–563, 2017. 10.1177/1535370216684040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Filip S, Pontillo C, Peter Schanstra J, Vlahou A, Mischak H, Klein J: Urinary proteomics and molecular determinants of chronic kidney disease: Possible link to proteases. Expert Rev Proteomics 11: 535–548, 2014. 10.1586/14789450.2014.926224 [DOI] [PubMed] [Google Scholar]
- 82.Tan RJ, Liu Y: Matrix metalloproteinases in kidney homeostasis and diseases. Am J Physiol Renal Physiol 302: F1351–F1361, 2012. 10.1152/ajprenal.00037.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Christensen B, Schytte GN, Scavenius C, Enghild JJ, McKee MD, Sørensen ES: FAM20C-mediated phosphorylation of MEPE and its acidic serine- and aspartate-rich motif. JBMR Plus 4: e10378, 2020. 10.1002/jbm4.10378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Tagliabracci VS, Engel JL, Wen J, Wiley SE, Worby CA, Kinch LN, Xiao J, Grishin NV, Dixon JE: Secreted kinase phosphorylates extracellular proteins that regulate biomineralization. Science 336: 1150–1153, 2012. 10.1126/science.1217817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Ishikawa HO, Xu A, Ogura E, Manning G, Irvine KD: The Raine syndrome protein FAM20C is a Golgi kinase that phosphorylates bio-mineralization proteins. PLoS One 7: e42988, 2012. 10.1371/journal.pone.0042988 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rowe PS: A unified model for bone-renal mineral and energy metabolism. Curr Opin Pharmacol 22: 64–71, 2015. 10.1016/j.coph.2015.03.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Grabner A, Amaral AP, Schramm K, Singh S, Sloan A, Yanucil C, Li J, Shehadeh LA, Hare JM, David V, Martin A, Fornoni A, Di Marco GS, Kentrup D, Reuter S, Mayer AB, Pavenstädt H, Stypmann J, Kuhn C, Hille S, Frey N, Leifheit-Nestler M, Richter B, Haffner D, Abraham R, Bange J, Sperl B, Ullrich A, Brand M, Wolf M, Faul C: Activation of cardiac fibroblast growth factor receptor 4 causes left ventricular hypertrophy. Cell Metab 22: 1020–1032, 2015. 10.1016/j.cmet.2015.09.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Gutiérrez OM, Wolf M, Taylor EN: Fibroblast growth factor 23, cardiovascular disease risk factors, and phosphorus intake in the health professionals follow-up study. Clin J Am Soc Nephrol 6: 2871–2878, 2011. 10.2215/CJN.02740311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, Isakova T, Gutiérrez OM, Aguillon-Prada R, Lincoln J, Hare JM, Mundel P, Morales A, Scialla J, Fischer M, Soliman EZ, Chen J, Go AS, Rosas SE, Nessel L, Townsend RR, Feldman HI, St John Sutton M, Ojo A, Gadegbeku C, Di Marco GS, Reuter S, Kentrup D, Tiemann K, Brand M, Hill JA, Moe OW, Kuro-O M, Kusek JW, Keane MG, Wolf M: FGF23 induces left ventricular hypertrophy. J Clin Invest 121: 4393–4408, 2011. 10.1172/JCI46122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Gutiérrez OM, Januzzi JL, Isakova T, Laliberte K, Smith K, Collerone G, Sarwar A, Hoffmann U, Coglianese E, Christenson R, Wang TJ, deFilippi C, Wolf M: Fibroblast growth factor 23 and left ventricular hypertrophy in chronic kidney disease. Circulation 119: 2545–2552, 2009. 10.1161/CIRCULATIONAHA.108.844506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Daniels SR, Kimball TR, Morrison JA, Khoury P, Witt S, Meyer RA: Effect of lean body mass, fat mass, blood pressure, and sexual maturation on left ventricular mass in children and adolescents. Statistical, biological, and clinical significance. Circulation 92: 3249–3254, 1995. 10.1161/01.CIR.92.11.3249 [DOI] [PubMed] [Google Scholar]
- 92.Nehgme R, Fahey JT, Smith C, Carpenter TO: Cardiovascular abnormalities in patients with X-linked hypophosphatemia. J Clin Endocrinol Metab 82: 2450–2454, 1997. 10.1210/jcem.82.8.4181 [DOI] [PubMed] [Google Scholar]
- 93.Faul C: FGF23 effects on the heart-levels, time, source, and context matter. Kidney Int 94: 7–11, 2018. 10.1016/j.kint.2018.03.024 [DOI] [PubMed] [Google Scholar]
- 94.Covic A, Vervloet M, Massy ZA, Torres PU, Goldsmith D, Brandenburg V, Mazzaferro S, Evenepoel P, Bover J, Apetrii M, Cozzolino M: Bone and mineral disorders in chronic kidney disease: Implications for cardiovascular health and ageing in the general population. Lancet Diabetes Endocrinol 6: 319–331, 2018. 10.1016/S2213-8587(17)30310-8 [DOI] [PubMed] [Google Scholar]
- 95.Liu ES, Thoonen R, Petit E, Yu B, Buys ES, Scherrer-Crosbie M, Demay MB: Increased circulating FGF23 does not lead to cardiac hypertrophy in the male Hyp mouse model of XLH. Endocrinology 159: 2165–2172, 2018. 10.1210/en.2018-00174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Latic N, Zupcic A, Frauenstein D, Erben RG: Activation of RAAS signaling contributes to hypertension in aged Hyp mice. Biomedicines 10: 1691, 2022. 10.3390/biomedicines10071691 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Shuto E, Taketani Y, Tanaka R, Harada N, Isshiki M, Sato M, Nashiki K, Amo K, Yamamoto H, Higashi Y, Nakaya Y, Takeda E: Dietary phosphorus acutely impairs endothelial function. J Am Soc Nephrol 20: 1504–1512, 2009. 10.1681/ASN.2008101106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Di Marco GS, Hausberg M, Hillebrand U, Rustemeyer P, Wittkowski W, Lang D, Pavenstädt H: Increased inorganic phosphate induces human endothelial cell apoptosis in vitro. Am J Physiol Renal Physiol 294: F1381–F1387, 2008. 10.1152/ajprenal.00003.2008 [DOI] [PubMed] [Google Scholar]
- 99.Steitz SA, Speer MY, Curinga G, Yang HY, Haynes P, Aebersold R, Schinke T, Karsenty G, Giachelli CM: Smooth muscle cell phenotypic transition associated with calcification: Upregulation of Cbfa1 and downregulation of smooth muscle lineage markers. Circ Res 89: 1147–1154, 2001. 10.1161/hh2401.101070 [DOI] [PubMed] [Google Scholar]
- 100.Jono S, McKee MD, Murry CE, Shioi A, Nishizawa Y, Mori K, Morii H, Giachelli CM: Phosphate regulation of vascular smooth muscle cell calcification. Circ Res 87: E10–E17, 2000. 10.1161/01.RES.87.7.e10 [DOI] [PubMed] [Google Scholar]
- 101.Smith ER, Ford ML, Tomlinson LA, Rajkumar C, McMahon LP, Holt SG: Phosphorylated fetuin-A-containing calciprotein particles are associated with aortic stiffness and a procalcific milieu in patients with pre-dialysis CKD. Nephrol Dial Transplant 27: 1957–1966, 2012. 10.1093/ndt/gfr609 [DOI] [PubMed] [Google Scholar]
- 102.Hamano T, Matsui I, Mikami S, Tomida K, Fujii N, Imai E, Rakugi H, Isaka Y: Fetuin-mineral complex reflects extraosseous calcification stress in CKD. J Am Soc Nephrol 21: 1998–2007, 2010. 10.1681/ASN.2009090944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Davies MR, Lund RJ, Hruska KA: BMP-7 is an efficacious treatment of vascular calcification in a murine model of atherosclerosis and chronic renal failure. J Am Soc Nephrol 14: 1559–1567, 2003. 10.1097/01.ASN.0000068404.57780.DD [DOI] [PubMed] [Google Scholar]
- 104.Hruska KA, Mathew S, Lund RJ, Memon I, Saab G: The pathogenesis of vascular calcification in the chronic kidney disease mineral bone disorder: The links between bone and the vasculature. Semin Nephrol 29: 156–165, 2009. 10.1016/j.semnephrol.2009.01.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Xie W, Méchin MC, Dubois SG, Lajeunesse D, van de Werve G: Up-regulation of liver glucose-6-phosphatase in x-linked hypophosphatemic mice. Horm Metab Res 34: 288–292, 2002. 10.1055/s-2002-33256 [DOI] [PubMed] [Google Scholar]
- 106.Hruska KA, Rifas L, Cheng SL, Gupta A, Halstead L, Avioli L: X-linked hypophosphatemic rickets and the murine Hyp homologue. Am J Physiol 268: F357–F362, 1995. 10.1152/ajprenal.1995.268.3.F357 [DOI] [PubMed] [Google Scholar]
- 107.Capparelli AW, Roh D, Dhiman JK, Jo OD, Yanagawa N: Altered proximal tubule glucose metabolism in X-linked hypophosphatemic mice. Endocrinology 130: 328–334, 1992. 10.1210/endo.130.1.1309337 [DOI] [PubMed] [Google Scholar]
- 108.Russo M, Montone RA, D’Amario D, Camilli M, Canonico F, Santamaria C, Iannaccone G, Pedicino D, Pidone C, Galli M, Trani C, Severino A, Liuzzo G, Niccoli G, Crea F: Role of perilipin 2 in microvascular obstruction in patients with ST-elevation myocardial infarction. Eur Heart J Acute Cardiovasc Care 10: 633–642, 2021. 10.1093/ehjacc/zuaa004 [DOI] [PubMed] [Google Scholar]
- 109.Zhao H, Han T, Hong X, Sun D: Adipose differentiation-related protein knockdown inhibits vascular smooth muscle cell proliferation and migration and attenuates neointima formation. Mol Med Rep 16: 3079–3086, 2017. 10.3892/mmr.2017.6997 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
ASARM treatment prevents intimal and medial vascular mineral deposition with atherosclerotic occlusion and thrombi in 56NEPHREX rats. Download Supplemental Figure 1, PDF file, 610 KB (609.7KB, pdf) .
ASARM treatment reduces TGF-β dermal expression in 56NEPHREX rats. Download Supplemental Figure 2, PDF file, 610 KB (609.7KB, pdf) .
Skin immunohistology analyses for tissue inhibitor metalloprotease (TIMP), α smooth-muscle actin, and TGF-β expression. Download Supplemental Figure 3, PDF file, 610 KB (609.7KB, pdf) .
Decreased serum phosphate and gut jejunum Na+-dependent phosphate co-transporter NPT2B (Slc34A2) occurs with 56NEPHREX rats infused with ASARM peptide compared with rats infused with vehicle. Download Supplemental Figure 4, PDF file, 610 KB (609.7KB, pdf) .
Decreased brain calcification occurs in 56NEPHREX rats treated with ASARM peptide. Download Supplemental Figure 5, PDF file, 610 KB (609.7KB, pdf) .
Brain mineral deposits identified using μCT iodine contrast imaging (Omnipaque, iohexitol perfusion) and ATLAS brain atlas mapping. Download Supplemental Figure 6, PDF file, 610 KB (609.7KB, pdf) .
Representative μCT scans of femur diaphysis. Download Supplemental Figure 7, PDF file, 610 KB (609.7KB, pdf) .