Abstract
BACKGROUND
Labor is a physiological process triggered by mechanical and hormonal events that promote uterine contractions to expel the fetus.
OBJECTIVE
This study aimed to evaluate the effectiveness of a nonpharmacologic childbirth care protocol in women in the active phase of labor in improving obstetrical and perinatal outcomes.
STUDY DESIGN
This was a randomized trial with concealed allocation, assessor blinding, and intention-to-treat analysis. A total of 80 low-risk primigravida women at the end of pregnancy admitted at the beginning of the active phase of labor participated in the study. The participants were divided into an experimental group (n=40) and a control group (n=40). Women in the experimental group received 4 interventions: ambulation at 4 to 6 cm of cervical dilation, alternation of maternal postures, transcutaneous electrical nerve stimulation at 6 to 7 cm, and a warm shower bath at >7 cm. The control group received only routine obstetrical care during labor. The parturient could request pharmacologic analgesia at any time during the study. The main outcome measures were the duration of the active phase of the first stage of labor, the duration of the expulsive phase of labor, and the prevalence of labor dystocia as assessed by the partograph. The researchers collected other maternal and neonatal data from official birth records.
RESULTS
The parturients who received the nonpharmacologic protocol had a shorter active phase of the first stage of labor (444 minutes in the control group and 373 minutes in the experimental group; P=.02), presented rupture of membranes later in labor (7 cm in the control group and 8 cm in the experimental group; P<.01), requested pharmacologic analgesia with more significant cervical dilation (5 cm in the control group and 8 cm in the experimental group; P<.01), requested fewer additional doses of analgesics, and had lower labor dystocia rates than the patients in the control group. In addition, there was no difference in other maternal and perinatal variables between the control group and the experimental group.
CONCLUSION
The implementation of a sequential nonpharmacologic protocol composed of ambulation, transcutaneous electrical nerve stimulation with change of upright positions, and warm shower bath has the potential to reduce labor pain, as reflected in decreased and delayed use of pharmacologic analgesia and reduced duration of the active phase of labor and dystocia rates. Maternity hospitals should provide this protocol, and women should be encouraged to request this childbirth care protocol.
Key Word: dystocia, labor pain, active phase of labor, nonpharmacologic resources, pharmacologic analgesia, randomized controlled trial
AJOG Global Reports at a Glance.
Why was this study conducted?
There is no physiotherapeutic protocol of nonpharmacologic resources that reduces the duration of the active phase of labor and the prevalence of labor dystocia, therefore constituting a need for study.
Key findings
The protocol adopted in this study reduced the duration of the active phase of labor and the incidence of dystocia.
What does this add to what is known?
This nonpharmacologic protocol can reduce the length of labor, delay pharmacologic analgesia, and decrease the need for additional doses of analgesics and dystocia rates without perinatal compromise.
Introduction
Labor is a physiological process triggered by mechanical and hormonal events that promote uterine contractions to expel the fetus.1 Body and emotional preparations through respectful maternity care with evidence-based interventions2, 3, 4, 5 are necessary for pregnant women to better accept vaginal delivery within this painful scenario. Studies have shown that labor pain can be alleviated by nonpharmacologic resources (NPRs) that are safe, easily applicable, noninvasive, and harmless to the pregnant woman and her fetus.6, 7, 8, 9, 10
Parturients can use several NPRs, such as relaxation and breathing exercises, continuous support, movements, pelvic exercises on a birth ball, massage, shower bath, and transcutaneous electrical nerve stimulation (TENS). These NPRs can be applied in isolation, combined or sequentially, depending on the parturient's desire, the healthcare team's training, and the availability of resources in the scenario.1,10, 11, 12, 13, 14, 15
The pain relief provided by NPRs is a benefit known and described by several studies.16, 17, 18 However, some pieces of evidence show that these resources may improve myometrial contraction quality, increase the cervical dilation speed and pelvic diameters, and facilitate the fetus's descent and cardinal movements in the birth canal.15 Therefore, they could influence the mechanism of labor and modify its progression and thus improve maternal and perinatal outcomes. These results represent good indicators of obstetrical care quality and help choose which evidence-based interventions should be used to improve outcomes.19,20 In contrast, few randomized controlled trials (RCTs) have evaluated the effects of various physical therapy resources used in different labor stages.
Although there are relevant studies addressing the use of nonpharmacologic resources for pain relief and correction of dystocia during labor, there are few randomized and controlled articles, and more research is needed on the subject and the effects of these methods on maternal and perinatal outcomes. Therefore, it is necessary to establish and use a care protocol for parturients to compare the groups (control and intervention) and to demonstrate the effectiveness of these methods in the different moments of labor and delivery.
Hence, the objectives of this study were to evaluate if (1) a physical therapy protocol modifies the duration of the active phase of labor, the duration of the expulsive phase of labor, and the prevalence of labor dystocia and (2) a physiotherapy protocol modifies the prevalence of intrapartum fetal compromise and improves neonatal outcomes.
Methods
Study design
This study was an RCT with concealed allocation, assessor blinding, and intention-to-treat analysis. The researchers registered the study protocol on the website ClinicalTrials.gov (identifier NCT01601860). After selecting the parturients, the primary researcher randomized them to an experimental group (EG) or a control group (CG) according to computer-generated random assignments (Randomization.com). The primary researcher applied the NPR protocol to the EG by cervical dilation, as explained below. A second researcher, who remained blinded to the participants’ group, evaluated women from the EG and CG and assessed the outcomes. Parturients in the CG received routine maternity care and were assessed by the blinded researcher simultaneously with the pregnant women in the EG. The blinded researcher also collected obstetrical and perinatal outcome data from the participants’ medical records.
Participants, researchers, and center
All participants were recruited from among the parturients admitted to the Reference Centre of Women's Health of Ribeirão Preto-MATER, São Paulo, Brazil, from May 2012 to July 2015. The inclusion criteria were low-risk primigravida at > 37 weeks of gestation with a single fetus in the cephalic position, spontaneous onset of labor, cervical dilation of 4 to 5 cm and ≥2 uterine contractions, intact ovular membranes, and the ability to read the consent form and understand the study. The exclusion criteria were the use of analgesic medications or other drugs that interfere with uterine contraction from hospital admission to randomization, cognitive or psychiatric problems, or other risk factors (gestational hypertension or gestational diabetes mellitus). The reasons to stop the intervention protocol were as follows: the participant desired to stop or had an intolerance to the NPR application because of pain, fetal compromise; cesarean delivery performed before the end of the research protocol, and an inability to obtain relevant data from maternal and neonatal medical records. The participants were free to withdraw from the study at any stage.
Although the primary researcher was a physiotherapist with 4 years of clinical experience, there was a standardization of the methods to evaluate labor pain to minimize the researcher's interference. The therapists assumed the same role (ie, the primary researcher performed randomization and application of the protocol or routine care, whereas the blinded researcher measured the outcomes). The baby's heart rate was evaluated every 30 minutes by the doctor as a way of identifying any fetal distress.
Intervention protocol
The EG received 4 NPRs: ambulation, free alternation of maternal postures, TENS, and shower bath. The primary researcher instructed the participant to walk at the beginning of the active phase of labor during 4 to 6 cm of cervical dilation. This intervention was intended to be delivered for 30-minute periods with stops for 10 minutes for assessment. If a participant progressed to the next level of cervical dilation, the researcher proceeded to the next intervention. The researcher attended participants in the CG for 30-minute periods, as performed for the EG, although they were only present for observation and to answer questions; they did not know which group the participant belonged to alternate positions with the use of electrodes.
The next 2 interventions were administered when the participant reached 6 cm of cervical dilation. The primary researcher proposed alternate upright positions to the participants every 15 minutes in the following sequence: squatting, sitting, supine, standing, and “on-all-fours” position in association with TENS. Moreover, 2 pairs of electrodes measuring 5×9 cm were fixed on the participants’ paravertebral regions using surgical tape. The paired electrodes were placed 1 cm laterally on either side of the spine at the T10 to L1 and S2 to S4 levels. The parameters used were 80-Hz frequency and pulse width of 75 μs, and the parturients individually titrated the intensity according to their sensitivity. The fourth intervention was applied when cervical dilation was at >7 cm. The primary researcher led the participant to take a warm shower for 40 minutes with the water at 37°C and with the primary researcher's supervision. The parturient could request pharmacologic analgesia or interruption of the intervention at any time during the study.
Experimental and control groups
Participants in both groups received all routine obstetrical care based on the World Health Organization (WHO) recommendations. In addition, the participants in the CG were allowed to choose the most comfortable position. Oxytocin could be used when indicated by the partograph, and pharmacologic analgesia was available to all pregnant women if they wished.
Outcome measures
Primary outcomes
The primary outcomes were the duration of the active phase of labor, the duration of the expulsive phase of labor, and labor dystocia. The instrument used to measure these outcomes was the partograph of the WHO. The healthcare team that evaluated the participant and filled out the partograph was blind to the participant group. The active phase of labor was from 4 cm to complete cervical dilation. The length of the expulsive phase of labor was from complete cervical dilation to fetal expulsion. Labor dystocia may range from “abnormally” slow dilation of the cervix during active labor or the fetus's slow descent during the expulsive phase of labor. “First-stage protraction” was diagnosed in pregnant women at ≥4 cm who were dilating less than approximately 1 cm/hour. “First-stage arrest” was diagnosed at cervical dilation of ≥4 cm in a patient with ruptured membranes and no cervical change for ≥2 hours despite adequate contractions. “Prolonged second stage” was considered when a nulliparous woman without epidural anesthesia had pushed for 3 hours or a multiparous woman without epidural anesthesia had pushed for 2 hours. If a woman had epidural anesthesia, there was an additional hour in the expulsive phase of labor. “Second-stage arrest” was diagnosed in a patient with ruptured membranes and no fetal descent for ≥2 hours after complete cervical dilation. The researchers considered the first-stage protraction and prolonged second stage together as functional dystocia.21
Secondary outcomes
The obstetrical outcomes included time taken for the parturient to request neuraxial labor analgesia, cervical dilation in which rupture of membranes occurred, the need to stimulate labor with oxytocin, mode of birth, maternal hemorrhaging (requiring surgical approach or blood transfusion), and postpartum infection. The perinatal outcomes included meconium in the amniotic fluid, intrapartum fetal compromise, 1-minute and 5-minute Apgar scores, and neonatal intensive care unit admission.
Sample size and data analysis
The researchers used the study by Ragnar et al22 to calculate the sample size that showed a difference in the duration of the expulsive phase of labor approximately 25% between the groups (CG and EG). They calculated 40 participants for each group with a significance level of 5% and a test power of 95%. The participants’ baseline characteristics and the outcome measures at each assessment point were summarized as mean (±SD) or number (percentage) as appropriate. Independent t test and Mann-Whitney U test were used to compare continuous variables, and the chi-square test was used to compare nominal variables. The survival analysis by Kaplan-Meier method was performed to highlight the differences in the active phase of labor and duration of the expulsive phase of labor between the groups.
Results
Flow and characteristics of participants throughout the study
The flow of participants throughout the trial is shown in Figure 1. A total of 378 parturients were assessed, and 298 parturients did not meet the inclusion criteria. A total of 80 participants were included and enrolled in the study's 2 arms: the EG (n=40) and the CG (n=40). There was no loss to follow-up of participants, and no participant left the study. Baseline characteristics (Table 1) showed that the groups were balanced for features, including maternal age, pregestational and current body mass index, educational status, marital status, presence of a person during labor, participation in a childbirth preparation course, and uterine dynamics at the enrollment into the study.
Figure 1.
Flow of participants throughout the study
Some participants were excluded by more than 1 criterion.
TENS, transcutaneous electrical nerve stimulation.
Santana. Physical therapy during labor and obstetrical outcomes. Am J Obstet Gynecol Glob Rep 2022.
Table 1.
Baseline characteristics of the participants in the control and experimental groups
| Groups |
||
|---|---|---|
| Maternal characteristics | Control (n=40) | Experimental (n=40) |
| Age (y), mean (±SD) | 21.8 (±4.22) | 22 (±4.49) |
| PPW (kg), mean (±SD) | 60.5 (±13.00) | 61.1 (±14.40) |
| PDW (kg), mean (±SD) | 72.6 (±14.70) | 73.6 (±14.40) |
| Height (cm), mean (±SD) | 1.6 (±0.07) | 16.0 (±0.05) |
| BMI (Kg/m2),mean (±SD) | 28.4 (±5.36) | 28.6 (±4.94) |
| Marital status, n (%) | ||
| Single | 6 (15.0) | 13 (32.5) |
| Married | 11 (27.5) | 8 (20.0) |
| With partner | 23 (57.5) | 19 (47.5) |
| Educational level, n (%) | ||
| Elementary school | 13 (32.5) | 9 (22.5) |
| Middle school | 24 (60.0) | 27 (67.5) |
| High school | 3 (7.5) | 4 (10.0) |
| Paid employment, n (%) | 17 (42.5) | 12 (30.0) |
BMI, body mass index; PDW, predelivery weight; PPW, prepregnancy weight; SD, standard deviation.
Santana. Physical therapy during labor and obstetrical outcomes. Am J Obstet Gynecol Glob Rep 2022.
The obstetrical features of the participants are shown in Table 2. The CG and EG were similar regarding gestational age at birth (P=.23), the need for oxytocin to stimulate or correct labor (relative risk [RR], 1.28; 95% confidence interval [CI], 0.96–1.70), and request for neuraxial labor analgesia (RR, 1.06; 95% CI, 0.91–1.22).
Table 2.
Obstetrical and neonatal outcomes of the participants in the control and experimental groups
| Groups |
Effecta,b or P valuec |
|||
|---|---|---|---|---|
| Outcomes | Control (n=40) | Experimental (n=40) | Control relative to experimental | |
| GA at birth (wk), mean (±SD) | 39.2 (±1.34) | 39.6 (±1.33) | −0.42 (−1.02 to 0.17)a | |
| Need of oxytocin, n (%) | 32 (80) | 25 (62.5) | 1.280 (0.96–1.70)b | |
| Pharmacologic analgesia, n (%) | 37 (92.5) | 35 (87.5) | 1.057 (0.91–1.22)b | |
| CD at which analgesia was requested (cm), median (IQR)c | 5 (4–10) | 8 (5–10) | <.0001 | |
| Additional doses of analgesics, n (%)d | 30 (81.0) | 17 (48.5) | 1.669 (1.15–2.42)b | |
| Length of active phase of labor (min), mean (±SD)c | 444 (±188) | 373 (±134) | 70.8 (−2.22 to 144.00)a | |
| Expulsive period (min), median (IQR)c | 26.0 (7–135) | 18.5 (5–140) | .1674 | |
| CD at which rupture of membranes occurred (cm), median (IQR)c | 7 (5–10) | 8 (6–10) | .0033 | |
| Dystocia, n (%) | 34 (85) | 26 (65) | 1.308 (1.02–1.69)b | |
| Cesarean delivery, n (%) | 10 (25) | 6 (15) | 1.308 (0.67–4.15)b | |
| Fetal compromise, n (%) | 2 (5.0) | 1 (2.5) | 2.00 (0.18–21.1)b | |
| Newborn weight (kg), mean (±SD) | 3.14 (±0.37) | 3.17 (±0.38) | −0.03 (0.20–0.13)a | |
| Newborn length (cm), mean (±SD) | 49 (±1.84) | 49 (±1.76) | −0.11 (−0.91 to 0.69)a | |
| Cephalic perimeter (cm), mean (±SD) | 33 (±1.50) | 34 (±1.41) | 0.65 (−1.290 to −0.002)a | |
| Thoracic perimeter (cm), mean (±SD) | 32 (±1.55) | 32 (±1.56) | −0.36 (−1.05 to 0.32)a | |
| Apgar score, n (%) | ||||
| >7 at 1 min | 31 (77.5) | 30 (75.0) | 1.03 (0.80–1.32b | |
| >7 at 5 min | 40 (100.0) | 40 (100.0) | NA | |
| Meconium in the amniotic fluid, n (%) | 8 (20.0) | 13 (32.5) | 0.61 (0.28–1.32)b | |
| Postpartum complication, n (%) | ||||
| Hemorrhage | 2 (5) | 0 (0) | NA | |
CD, cervical dilation; CI, confidence interval; GA, gestational age; IQR, interquartile range; NA, not applicable (n=zero or 100%); SD, standard deviation.
Mean between-group difference (95% CI)
Relative risk (95% CI)
Mann-Whitney U test
Only among women who had analgesia.
Santana. Physical therapy during labor and obstetrical outcomes. Am J Obstet Gynecol Glob Rep 2022.
Effect of intervention
Parturients in the CG had significantly higher dystocia rates than those in the EG (RR, 1.31; 95% CI, 1.02–1.69). However, the types of dystocias, meaning functional dystocia (70.5% for CG vs 65.3% for EG), first-stage arrest (17.6% for CG vs 11.5% for EG), and second-stage arrest (11.7% for CG vs 23% for EG), were in similar proportions in the groups.
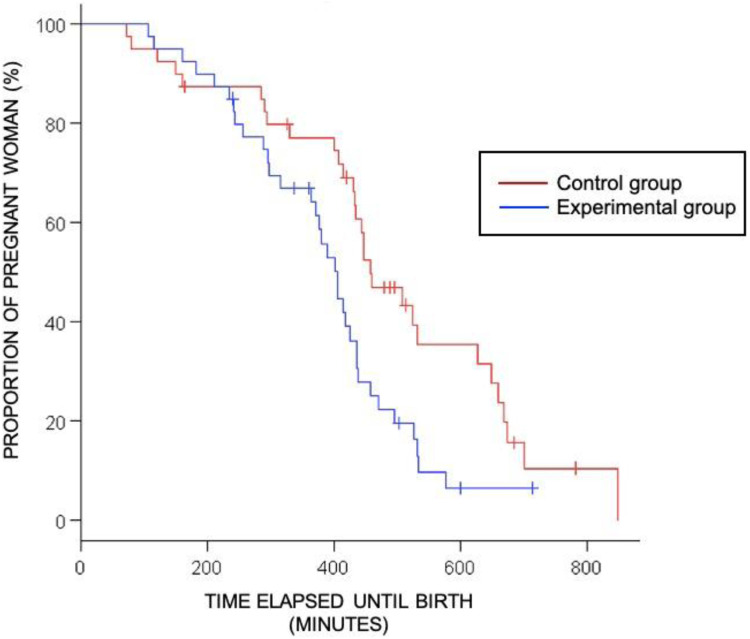
The mean durations of the active phase of labor were 444±188 minutes and 373±134 minutes in CG and EG, respectively. Therefore, the active phase of labor was significantly faster in the EG by a mean of 71 minutes. Figure 2 demonstrates that half of the patients in the CG reached the expulsive phase of labor at the end of 458 minutes, although this occurred after 405 minutes for the EG. In addition, it is possible to notice that only 2 patients gave birth in the EG after 600 minutes, whereas the number of parturients that gave birth after this time is much higher in the CG. In contrast, the average length of the expulsive phase of labor was 7.5 minutes less in the EG, but this was not significantly different between the groups (P=.16).
Figure 2.
Analysis of the length of the active phase of labor
Santana. Physical therapy during labor and obstetrical outcomes. Am J Obstet Gynecol Glob Rep 2022.
The participants in the CG requested pharmacologic analgesia earlier than the participants in the EG (5 cm vs 8 cm, respectively; P<.0001). Therefore, the experimental protocol postponed the need for pharmacologic analgesia for pain relief. This event resulted in a significantly higher number of additional doses of analgesia after the initial dose in the CG (RR, 1.67; 95% CI, 1.15–2.24). Moreover, 5 parturients (12.5%) in the EG and 3 (7.5%) parturients in the CG did not use any analgesic medication.
The rupture of amniotic membranes was significantly earlier in the CG than in the EG. Cervical dilation was 1.0 cm greater in the EG than in the CG before the rupture of membranes (P=.003). However, the prevalence of the opportune rupture of membranes (>6 cm) was similar in both groups (Table 2).
Among the women who had a vaginal delivery, there was no significant difference in the episiotomy rates between the groups (P=.19). In addition, the postpartum complication rates were similar between the groups (P=.15), although 2 women in the CG had hemorrhaging. However, this result should be interpreted with caution as no patient presented any hemorrhagic complication in the EG. No patient had a postpartum infection.
There was no significant difference among any neonatal outcomes, as shown in Table 2. All of the newborns had 5-minute Apgar scores of >7 in both groups, and respiratory distress rates were similar. Only 2 newborns in the CG and 1 newborn in the EG had respiratory distress and were admitted to an intermediate care unit (P=.56).
Discussion
Main findings
This study showed that the parturients who received the NPRs had a shorter active phase of labor, presented rupture of membranes later, and had lower dystocia rates than the patients in the CG. Moreover, they requested analgesia with more significant cervical dilation and fewer additional doses of analgesics. In addition, there was no difference in other maternal and perinatal variables between the groups. The protocol used in this study included NPRs designed to promote these outcomes individually and safely.
Comparison with literature
Some authors have evaluated the effect of walking on the duration of the active phase of labor. Randomized studies by Bio et al,8 Mamede et al,23 Regaya et al,19 and Mathew et al24 demonstrated significant and similar reductions to that observed in our study in the EG. Mamede et al23 showed an average reduction in the active phase of labor of 2.04 hours in the group of parturients who walked, similar to the data from our study (112 minutes). Furthermore, Ben Regaya et al19 showed a significant reduction (34%) in the duration of the active phase of labor in primigravidae of the EG, whereas our results demonstrated a reduction of 23% in the EG. These articles only differ from the current study concerning the application time of the resource. Note that the protocol was started in our patients when they were in labor with 4 to 5 cm dilation, a criterion used in the institution to allow studies with patients; however, in a recent prospective study, the WHO indicates 6 cm as a definer of the activation phase of labor.25
The benefits of upright positions on labor duration seem to be related to the augmented uterine blood volume, effect of gravity, and pelvic mobility.20 The increase in uterine blood flow allows a concentration of endogenous oxytocin to reach the myometrium and promote more intense and coordinated contractions.20 Furthermore, this birth position increases cervical dilation because of the pressure that the fetal head exerts on the cervix.19 In addition, the movement increases pelvic diameters and facilitates fetal descent.20 Therefore, walking seems to be a beneficial intervention in reducing the active phase of labor as long as there is no maternal or perinatal adverse outcome. Previous studies did not show negative outcomes, such as the meta-analysis published by Lawrence et al20 (odds ratio, 0.20; 95% CI, 0.04–0.89). The experimental protocol used in this study also did not demonstrate adverse maternal or perinatal outcomes.
Walking and alternating upright positions could reduce the duration of the expulsive phase of labor for the same reasons that reduce the first stage of labor. The mean difference in the expulsive phase of labor in this study was 7.5 minutes shorter in the EG than in the CG.20 This result did not quite reach statistical significance, probably because of a lack of study power for this outcome; however, it represents almost a 30% reduction in the expulsive phase of labor.
Few studies have evaluated the effect of TENS on the stages of labor duration, and no study has presented similar interventions to this study. The studies by Orange et al,6 Ratna and Rekha,26 and Chao et al27 investigated the effect of TENS applied at the beginning of the active phase on the duration of the first stage of labor, and none of them showed differences between the CG and EG.
In contrast, the clinical trial conducted by Shahoei et al28 demonstrated that primigravidae who received TENS at the beginning of the active phase of labor requested analgesia later than the parturients in the CG (P<.001). This study reaffirmed the benefit of TENS in childbirth care protocols. Studies that evaluated the effects of TENS in parturients did not show any adverse perinatal outcomes.26
The last intervention used as part of the protocol was the warm shower. Studies evaluating the effect of hydrotherapy on the progression of labor are scarce and controversial. A study by Chaichian et al28 revealed a shorter active phase of labor for women who remained immersed in warm water throughout labor than for those who received conventional care.29
There was no similar study that analyzed labor dystocia or rupture of membranes and NPRs applied during labor. Cephalopelvic disproportion seems to be the leading cause for the greater incidence of dystocia in the CG (P=.04). However, there was no significant difference in the birthweight (P=.75) or cephalic perimeter of the newborns (P=.07) between the EG and CG. The incidence of functional dystocia and the need for oxytocin were higher in the CG than in the EG. There were 2 possible explanations for these higher incidences of labor dystocia in the CG. First, the parturients in the EG requested pharmacologic analgesia with more significant cervical dilation and demanded fewer additional analgesic doses. The earlier pharmacologic analgesia may have increased the risk of dystocia in the CG.30 Second, delaying the use of analgesic medication with this NPR protocol could mean sufficient pain relief to comfort the parturient to more actively deal with labor and with more position changes.31 These movements have the potential to accelerate labor.
Although the protocol adopted in this study reduced the active phase of labor and incidence of dystocia, there was no reduction in the cesarean delivery rates. These data are also consistent with other studies.20,32 A similar RCT by Gallo et al15 included parturients who received 3 interventions. The authors also did not show the differences in the cesarean delivery rates between the EG and CG. However, the childbirth care protocol in the EG was different from ours. The number of participants may be insufficient to analyze the cesarean delivery rates and maternal postpartum complications. It is relevant to consider whether higher analgesic doses and a more prolonged first stage of labor in the CG could increase the risk of hemorrhage.
Strengths and limitations
Although this was an RCT with a blinded researcher who evaluated maternal and perinatal outcomes, along with strict and carefully respected inclusion criteria, some limitations need to be highlighted. First, it was not possible to blind the participants to the interventions included in the protocol. Second, the CG received continuous support that can lead to labor pain relief and better obstetrical outcomes. However, although these parturients could move freely, this is not comparable to the systematic and consistent use of a protocol applied by a physiotherapist. Third, the external validity of this study was restricted to low-risk parturients.
Conclusion
Our study supported the benefits of an NPR protocol in the active phase of labor, including walking followed by TENS applied in different maternal upright positions and a shower bath at the end. This protocol enabled a reduction in the labor duration, delayed pharmacologic analgesia, and decreased the number of additional analgesic doses and dystocia rates without perinatal compromise. The protocol may consequently result in lower costs, and this facilitates increased patient turnover and a positive childbirth experience. This study reinforced the importance of a multiprofessional healthcare team with the physiotherapist in evidence-based obstetrical care. We suggest implementing the protocol in scenarios with low-risk parturients and performing new studies to investigate the protocol's safety and effects in high-risk pregnancies.
Acknowledgments
We thank all the women who consented to participate in the trial and the entire staff of the Reference Centre of Women's Health of Ribeirão Preto-MATER, São Paulo, Brazil, for contributing to developing this study.
Footnotes
The authors report no conflict of interest.
The Ethics Committee of the University Hospital of the Ribeirão Preto Medical School approved this study under the protocol HCRP number 9147/2011. All participants gave written informed consent before data collection (approval date: August 17, 2011; reference number 2954/2011).
This study was supported by the National Council for Scientific and Technological Development, Brazil.
The lead researcher, L.S.S., had full access to all study data, and she takes responsibility for the data integrity and the data analysis accuracy.
Patient consent was not required because no personal information or detail was included.
This study has been registered on ClinicalTrials.gov (identifier NCT01601860; registered May 18, 2012; https://clinicaltrials.gov/ct2/show/NCT01601860).
Cite this article as: Santana LS, Gallo RBS, Quintana SM, et al. Applying a physiotherapy protocol to women during the active phase of labor improves obstetrical outcomes: a randomized clinical trial. Am J Obstet Gynecol Glob Rep 2022;XX:x.ex–x.ex.
References
- 1.Santana LS, Gallo RB, Ferreira CH, Duarte G, Quintana SM, Marcolin AC. Transcutaneous electrical nerve stimulation (TENS) reduces pain and postpones the need for pharmacological analgesia during labour: a randomised trial. J Physiother. 2016;62:29–34. doi: 10.1016/j.jphys.2015.11.002. [DOI] [PubMed] [Google Scholar]
- 2.Van der Gucht N, Lewis K. Women's experiences of coping with pain during childbirth: a critical review of qualitative research. Midwifery. 2015;31:349–358. doi: 10.1016/j.midw.2014.12.005. [DOI] [PubMed] [Google Scholar]
- 3.da Saúde do Brasil Ministério. Ministério da Saúde; Brasília, Brazil: 2017. Secretaria de ciência, tecnologia e insumos estratégicos. Departamento de gestão e incorporação de tecnologias em saúde. Diretriz Nacional de Assistência ao parto normal. [Google Scholar]
- 4.World Health Organization . World Health Organization; Geneva, Switzerland: 2018. WHO recommendations: intrapartum care for a positive childbirth experience. [PubMed] [Google Scholar]
- 5.766: approaches to limit intervention during labor and birth. ACOG Committee Opinion. 2019;133:e164–e173. doi: 10.1097/AOG.0000000000003074. Obstet Gynecol. [DOI] [PubMed] [Google Scholar]
- 6.Orange FAD, Amorim MMRD, Lima L. Uso da eletroestimulação transcutânea para alívio da dor durante o trabalho de parto em uma maternidade-escola: ensaio clínico controlado. Rev Bras Ginecol Obstet. 2003;25:45–52. [Google Scholar]
- 7.Simkin P, Bolding A. Update on nonpharmacologic approaches to relieve labor pain and prevent suffering. J Midwifery Womens Health. 2004;49:489–504. doi: 10.1016/j.jmwh.2004.07.007. [DOI] [PubMed] [Google Scholar]
- 8.Bio E, Bittar RE, Zugaib M. Influência da mobilidade materna na duração da fase ativa do trabalho de parto. Rev Bras Ginecol Obstet. 2006;28:671–679. [Google Scholar]
- 9.Carraro TE, Knobel R, Radunz V, et al. Cuidado e conforto durante o trabalho de parto e parto: na busca pela opinião das mulheres. Enfermagem. 2006;15:95–104. [Google Scholar]
- 10.Gupta JK, Sood A, Hofmeyr GJ, Vogel JP. Position in the second stage of labour for women without epidural anaesthesia. Cochrane Database Syst Rev. 2017;5 doi: 10.1002/14651858.CD002006.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Taghavi S, Barband S, Khaki A. Effect of hydrotherapy on pain of labor process. Baltica. 2015;28:116–121. [Google Scholar]
- 12.Hensley JG, Collins MR, Leezer CL. Pain management in obstetrics. Crit Care Nurs Clin North Am. 2017;29:471–485. doi: 10.1016/j.cnc.2017.08.007. [DOI] [PubMed] [Google Scholar]
- 13.Smith CA, Levett KM, Collins CT, Dahlen HG, Ee CC, Suganuma M. Massage, reflexology and other manual methods for pain management in labour. Cochrane Database Syst Rev. 2018;3 doi: 10.1002/14651858.CD009290.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Caughey AB. UpToDate; Waltham, MA: 2019. Nonpharmacologic approaches to management of labour pain. [Google Scholar]
- 15.Gallo RBS, Santana LS, Marcolin AC, Duarte G, Quintana SM. Sequential application of non-pharmacological interventions reduces the severity of labour pain, delays use of pharmacological analgesia, and improves some obstetric outcomes: a randomised trial. J Physiother. 2018;64:33–40. doi: 10.1016/j.jphys.2017.11.014. [DOI] [PubMed] [Google Scholar]
- 16.Bonapace J, Gagné GP, Chaillet N, Gagnon R, Hébert E, No Buckley S. 355-Physiologic basis of pain in labour and delivery: an evidence-based approach to its management. J Obstet Gynaecol Can. 2018;40:227–245. doi: 10.1016/j.jogc.2017.08.003. [DOI] [PubMed] [Google Scholar]
- 17.Báez Suárez A, Martín Castillo E, García Andújar J, García Hernández JÁ, Quintana Montesdeoca MP, Loro Ferrer JF. Evaluation of the effectiveness of transcutaneous nerve stimulation during labor in breech presentation: a case series. J Matern Fetal Neonatal Med. 2021;34:24–30. doi: 10.1080/14767058.2019.1572110. [DOI] [PubMed] [Google Scholar]
- 18.Thomson G, Feeley C, Moran VH, Downe S, Oladapo OT. Women's experiences of pharmacological and non-pharmacological pain relief methods for labour and childbirth: a qualitative systematic review. Reprod Health. 2019;16:71. doi: 10.1186/s12978-019-0735-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ben Regaya L, Fatnassi R, Khlifi A, et al. [Role of deambulation during labour: a prospective randomized study] J Gynecol Obstet Biol Reprod (Paris) 2010;39:656–662. doi: 10.1016/j.jgyn.2010.06.007. [DOI] [PubMed] [Google Scholar]
- 20.Lawrence A, Lewis L, Hofmeyr GJ, Styles C. Maternal positions and mobility during first stage labour. Cochrane Database Syst Rev. 2013 doi: 10.1002/14651858.CD003934.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Royal College of Obstetricians and Gynaecologists. Shoulder dystocia (Greentop Guideline No. 42). 2011. Available at:https://www.rcog.org.uk/guidance/browse-all-guidance/green-top-guidelines/shoulder-dystocia-green-top-guideline-no-42/. Accessed in 11.02.2020.
- 22.Ragnar I, Altman D, Tydén T, Olsson SE. Comparison of the maternal experience and duration of labour in two upright delivery positions–a randomised controlled trial. BJOG. 2006;113:165–170. doi: 10.1111/j.1471-0528.2005.00824.x. [DOI] [PubMed] [Google Scholar]
- 23.Mamede FV, Almeida AM, Souza L, Mamede MV. A dor durante o trabalho de parto: o efeito da deambulação. Rev Lat Am Enferm. 2007;15:1157–1162. [Google Scholar]
- 24.Mathew A, Nayak S, Vandana K. A comparative study on effect of ambulation and birthing ball on maternal and newborn outcome among primigravida mothers in selected hospitals in Mangalore. Nitte Univ J Health Sci. 2012;2:2–5. [Google Scholar]
- 25.World Health Organization . World Health Organization; Geneva, Switzerland: 2018. WHO recommendations on intrapartum care for a positive childbirth experience. [PubMed] [Google Scholar]
- 26.Ratna T, Rekha P. Comparative study of transcutaneous electrical nerve stimulation (TENS) and tramadol hydrochloride for pain relief in labor. J Obstet Gynecol India. 2004;54:346–350. [Google Scholar]
- 27.Chao AS, Chao A, Wang TH, et al. Pain relief by applying transcutaneous electrical nerve stimulation (TENS) on acupuncture points during the first stage of labor: a randomized double-blind placebo-controlled trial. Pain. 2007;127:214–220. doi: 10.1016/j.pain.2006.08.016. [DOI] [PubMed] [Google Scholar]
- 28.Chaichian S, Akhlaghi A, Rousta F, Safavi M. Experience of water birth delivery in Iran. Arch Iran Med. 2009;12:468–471. [PubMed] [Google Scholar]
- 29.Samadzadeh S, Rezavand N, Yari M, Rezaei M, Faizmahdavi H, Hematti M. Comparison of entonox and transcutaneous electrical nerve stimulation (TENS) in labor pain. J Med Biomed Sci. 2017;6:11–16. [Google Scholar]
- 30.Sng BL, Leong WL, Zeng Y, et al. Early versus late initiation of epidural analgesia for labour. Cochrane Database Syst Rev. 2014 doi: 10.1002/14651858.CD007238.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Thuvarakan K, Zimmermann H, Mikkelsen MK, Gazerani P. Transcutaneous electrical nerve stimulation as a pain-relieving approach in labor pain: a systematic review and meta-analysis of randomized controlled trials. Neuromodulation. 2020;23:732–746. doi: 10.1111/ner.13221. [DOI] [PubMed] [Google Scholar]
- 32.Cluett ER, Burns E. Immersion in water in labour and birth. Cochrane Database Syst Rev. 2009 doi: 10.1002/14651858.CD000111.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]