Abstract
Background: We present the case of a patient with a Merkel cell carcinoma (MCC) of the left preauricular area.
Case presentation:A 84-year-old Greek man was examined at the outpatient ENT Department of our clinic with a lesion in the preauricular area that had appeared four months ago. The patient history included antihypertensive and antihyperlipidemic therapy as well as treatment for dementia. The excision of the skin lesion was performed under local anesthesia. The histological examination revealed a Merkel cell carcinoma. The patient underwent a computed tomography (CT) scan that showed a lesion with clear limits in the left parotid gland and lymph nodes. Under general anesthesia, he underwent a left superficial parotidectomy, left submandibular gland excision and radical neck dissection. Histological preparations were analyzed using an intraoperative flow cytometry (iFC) protocol. A radiation therapy concluded the patient's treatment.
Conclusion:Even if MCC appears as a less common and more aggressive skin cancer type, a clinician always has to include it in the differential diagnosis of a skin lesion. We found the use of iFC very useful for the diagnosis of this skin cancer.
Keywords:Merkel cell carcinoma, head neck tumors, preauricular area, skin cancer, case report, intraoperative flow cytometry.
INTRODUCTION
Merkel cell carcinoma (MCC), that was first described by Toker (1972) as trabecular carcinoma, is a primary cutaneous tumor of neuroendocrine origin. It has a propensity for local recurrence and regional lymph node metastases. As the head and neck is a sun-exposed area, the skin lesion most commonly appears on the head and neck than on the trunk, genitalia, and perianal region. Except significant ultraviolet exposure, the main factors involved in MCC pathogenesis seem to be the history of malignancy, immune compromise as well as Merkel cell polyomavirus (MCPyV) DNA. Merkel cell carcinoma has a high local recurrence rate, which is much higher than that reported for melanoma. Surgical excision of the primary tumor, adjuvant therapy and postoperative radiotherapy or chemotherapy are proposed as treatment of the MCC and the choice of treatment depends on the staging (1-9).
CASE PRESENTATION
A 84-year-old male patient was examined in the outpatient ENT clinic of our hospital because of a skin lesion in the preauricular area that had appeared four months ago. He reported an injury in the skin lesion area that followed a regional edema. According to the patient's medical history, he was receiving antihypertensive therapy, atorvastatin as antihyperlipidemic treatment as well as donepezil as dementia therapy. The skin lesion appeared as a painless, pinkish purple cutaneous nodule without ulceration with diameter dimensions smaller than 1 cm. The clinical examination revealed no lymph nodes in the neck area and no pathological findings through the otoscopy, nasal endoscopy and laryngoscopy. An ultrasound evaluation confirmed the absence of nodal disease. An operation for the excision of the skin lesion was prepared under local anesthesia. The histological examination of the lesion concluded that it referred to a malignant neoplasm with morphological and immunohistochemical analysis compatible with neuroendocrine neoplasm, a Merkel cell carcinoma. The patient was informed about the type and prognosis of malignancy and agreed to proceed to further investigation. Ten days later, he underwent a CT scanning using a multislice computed tomograph with slice thickness of 2 mm before and after providing contrast agent. The CT scan revealed a lesion with clear limits and maximum diameter 24 mm in the left parotid gland. Lymph nodes with maximum diameter 8 mm were also found in the left parotid area. The CT scan of the brain, chest and abdomen did not reveal any pathological finding. An operation under general anesthesia was performed after obtaining the patient's consent. We proceeded to a left submandibular gland excision. We also carried out a left superficial parotidectomy, removing the part of the gland that was infiltrated by the tumor, separately. The facial nerve was identified and controlled by continuous facial nerve monitoring with a neurostimulator (Figure 1). The following neck dissection included the lymph nodes of the levels: Ib, IIa, IIb. In order to achieve the perioperative evaluation of the tumor progression, histological examination of the submandibular lymph nodes and the skin margins was held through periprocedural quick biopsies. The tissue sections that were removed were sent for histological preparation and examination. Histological preparations were analyzed using an intraoperative flow cytometry (iFC) protocol, which was optimized for head and neck lesions, as a means for tumor margin evaluation (10). The results of iFC corroborated those of the histological examination (Figures 2, 3). In detail, peripheral blood mononuclear cells (PBMCs) were used as a control to determine physiological DNA content and G0/G1 cell cycle peak. The percentage of dividing cells present in lymph nodes and a tumor margin are indicative of pathological cell populations with a tumor index (S and G2/M cell fractions) above 5% (Figure 4).
The patient did not show up other postoperative complications and was discharged home ten days after surgery. The results of the histological examination revealed that all examined tissues were infiltrated by the tumor. The patient's treatment was completed with adjuvant radiotherapy.
DISCUSSION
Although the pathogenesis of Merkel carcinoma is not yet fully understood, the most frequently mentioned hypothesis states that MCC derives from intraepidermal Merkel cells, which belong to the amine precursor uptake and decarboxylation system (APUD). Merkel cell carcinoma has an estimated mortality rate of 33%, which is definitely higher than that of melanoma, while satellite metastases can occur rather early. The presence of nodal disease is a poor prognostic indicator, highlighting the importance of early diagnosis and treatment. In our case, because all examined tissues were infiltrated by the tumor, maybe it was an indication for adjuvant concomitant radio chemotherapy as an early therapy. But oncologists proposed adjuvant radiotherapy after the extensive removal of the tumor. As half of MCC cases are located in the head and neck region, the head and neck surgeon should be familiar with this disease entity, diagnosis and treatment strategies. Merkel cell carcinoma can occur anywhere in the head and neck, more often in sun-exposed skin areas. It can rarely arise from the nasal and oral mucosa. Because MCC presentation is non-specific, the clinician must have a high index of suspicion. The differential diagnosis of MCC includes other "blue-cell" appearing tumors such as lymphoma, but can also include a poorly differentiated SCC, basal cell carcinoma, and melanoma (1-9).
CONCLUSION
We report the case of a cutaneous MCC of the preauricular left area. Despite the fact that the ultrasound evaluation revealed the absence of nodular disease, the course of the disease changed. The CT scan revealed intraparotid and cervical lymph node metastasis. We avoided proceeding to a sentinel lymph node biopsy (SLNB), as it is associated with more complications, because the nodes are not always easy to reach. We found that the iFC was very useful, as histological preparation, following cytometry and the results were available in half an hour. Considering the poor prognosis and evolution of the MCC, a clinician always has to include it in the differential diagnosis of a skin lesion, even if it has the appearance of a less common and more aggressive skin cancer type. In cases of locoregional metastasis, it is extremely useful to use intraoperative flow cytometry, in order to determine the filtration limits of the tumor as best as possible.
Conflict of interests: none declared.
Financial support: This research did not receive specific funding, but was performed as part of the employment of the authors; name of employer: Vasilios Ragos.
Authors' contributions: AZ analyzed and interpreted the patient data. AT, IK, VR performed the surgical treatment. AP, EL the histological examination, EV the intraoperative flow cytometry. AZ and AL were major contributors in writing the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate: The work described has been carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans.
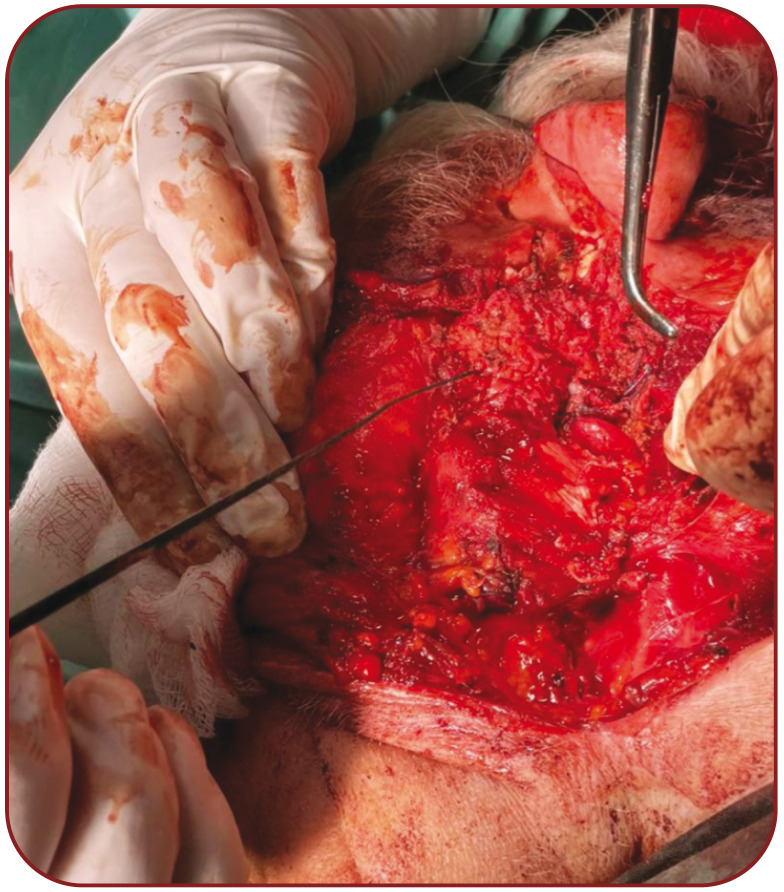
FIGURE 1.
Facial nerve monitoring with a neurostimulator
FIGURE 2.
Infiltrating Merkel cell carcinoma (right), metastatic to lymph node (left) (hematoxylin-eosin staining, magnificationx200). Immunohistochemically, the neoplastic cells were highlighted by synaptophysin, a neuroendocrine marker (DAB, magnificationx100)
FIGURE 3.
Parotid gland parenchyma infiltration by Merkel cell carcinoma (a. hematoxylin-eosin staining, magnificationx100). Areas of necrosis were noted (b. hematoxylin-eosin staining, magnificationx200). By immunohistochemistry, the neoplastic cells were positive for synaptophysin (c. DAB, magnificationX100) and cytokeratin 20 (d. DAB, magnificationx600). Cytokeratin 20 staining pattern was dot-like perinuclear
FIGURE 4.
Intraoperative Flow cytometry analysis of DNA content, following propidium iodide staining. (A) analysis of PBMCs, (B) analysis of lymph node, (C) analysis of tumor margin
Contributor Information
Athina ZARACHI, Department of Otorhinolaryngology, Head and Neck Surgery, Faculty of Medicine, School of Health Sciences, University of Ioannina, Ioannina, Greece.
Evrysthenis VARTHOLOMATOS, Neurosurgical Institute, Medical School, University of Ioannina, Ioannina, Greece.
Alkistis TSIKOU, Department of Otorhinolaryngology, Head and Neck Surgery, Faculty of Medicine, School of Health Sciences, University of Ioannina, Ioannina, Greece.
Ioannis KOMNOS, Department of Otorhinolaryngology, Head and Neck Surgery, Faculty of Medicine, School of Health Sciences, University of Ioannina, Ioannina, Greece.
Alexandra PAPOUDOU-BAI, Department of Pathology, Faculty of Medicine, School of Health Sciences, University of Ioannina, Greece.
Evangelia LAMPRI, Department of Pathology, Faculty of Medicine, School of Health Sciences, University of Ioannina, Greece.
Angelos LIONTOS, Faculty of Medicine, Department of Internal Medicine, School of Health Sciences, University of Ioannina, Ioannina, Greece.
Vasilios RAGOS, Department of Maxillofacial Surgery, Medical School, University of Ioannina, Ioannina, Greece.
References
- 1.Marek L, Grzanka A, Chmielowska E, et al. Letter to the Editor Merkel cell carcinoma: an illustrative case and review. Adv Dermatol Allergol. 2014;5:325–328. doi: 10.5114/pdia.2014.40797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Swann MH, Yoon J. Merkel Cell Carcinoma. S. eminars in Oncology. 2007;34:51–56. doi: 10.1053/j.seminoncol.2006.10.021. [DOI] [PubMed] [Google Scholar]
- 3.Boyle F, Pendelebury S, Bell D. Further insights into the natural history and management of primary cutaneous neuroendocrine (Mercel Cell carcinoma) carcinoma. Int J Radiat Oncol Biol Phys. 1995;31:315–323. doi: 10.1016/0360-3016(93)E0110-R. [DOI] [PubMed] [Google Scholar]
- 4.Schwartz RA, Lambert WC. The Merkel cell carcinoma: a 50-year retrospect. J Surg Oncol. 2005;89:5. doi: 10.1002/jso.20165. [DOI] [PubMed] [Google Scholar]
- 5.Medina-Franco H, Urist MM, Fiveash J, et al. Multimodality Treatment of Merkel Cell Carcinoma: Case Series and Literature Review of 1024 Cases. Ann Surg Oncol. 2001;8:204–208. doi: 10.1007/s10434-001-0204-4. [DOI] [PubMed] [Google Scholar]
- 6.Londino III AV, Miles BA. The Role of Free Tissue Transfer in Merkel Cell Carcinoma of the Head and Neck. J Skin Cancer, 2012. [DOI] [PMC free article] [PubMed]
- 8.Andea AA, Coit DG, Amin B, Busam KJ. Merkel cell carcinoma: histologic features and prognosis. Cancer. 2008;9:2549–2558. doi: 10.1002/cncr.23874. [DOI] [PubMed] [Google Scholar]
- 9.Lemos BD, Storer BE, Iyer JG, et al. Pathologic nodal evaluation improves prognostic accuracy in Merkel cell carcinoma: analysis of 5823 cases as the basis of the first consensus staging system. J Am Acad Dermatol. 2010;63:751–761. doi: 10.1016/j.jaad.2010.02.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vartholomatos G, Basiari L, Exarchakos G, et al. Intraoperative flow cytometry for head and neck lesions. Assessment of malignancy and tumour-free resection margins. Oral Oncol. 2019;99:104344. doi: 10.1016/j.oraloncology.2019.06.025. [DOI] [PubMed] [Google Scholar]