Keywords: cachexia, muscle wasting, noncoding RNA, pancreatic cancer, tissue biopsy
Abstract
Cachexia is an acute syndrome that is very commonly observed in patients with cancer. Cachexia is the number one cause of death in patients with metastatic disease and is also the major factor for physical toxicity and financial burden. More importantly, the majority of patients with advanced-stage pancreatic ductal adenocarcinoma (PDAC) cancer undergo cachexia. Pancreatic cancer causes deaths of ∼50,000 Americans and about 400,000 people worldwide every year. The high mortality rates in metastatic PDAC are due to systemic pathologies and cachexia, which quickens death in these patients. About 90% of all patients with PDAC undergo wasting of muscle causing mobility loss and leading to a number of additional pathological conditions. PDAC-associated cancer cachexia emanates from complex signaling cues involving both mechanical and biological signals. Tumor invasion is associated with the loss of pancreatic function-induced digestive disorders and malabsorption, which causes subsequent weight loss and eventually promotes cachexia. Besides, systemic inflammation of patients with PDAC could release chemical cues (e.g., cytokine-mediated Atrogin-1/MAFbx expression) that participate in muscle wasting. Our understanding of genes, proteins, and cytokines involved in promoting cancer cachexia has evolved considerably. However, the role of epigenetic factors, particularly the role of noncoding RNAs (ncRNAs) in regulating PDAC-associated cachexia is less studied. In this review article, the most updated knowledge on the various ncRNAs including microRNAs (miRs), long noncoding RNA (lncRNAs), piwi interacting RNAs (PiwiRNAs), small nucleolar RNA (snoRNAs), and circular RNAs (circRNA) and their roles in cancer cachexia are described.
INTRODUCTION
According to global epidemiology studies, pancreatic cancer kills ∼50,000 Americans and around 400,000 people worldwide annually (1). Pancreatic ductal adenocarcinoma (PDAC) constitutes over 95% of pancreatic cancer and is a complex systemic disease. The high morbidity is not just due to pancreatic organ failure; rather, a complex set of events in the host including paracrine signaling mediated systemic dysfunction causes an overall reduction in the patient’s quality of life that quickens death.
Cancer-associated cachexia is one of the major systemic syndromes observed in patients with PDAC (2). Cachexia affects ∼90% of all patients with PDAC both in the United States and the European Union causing loss of mobility and several additional pathological conditions leading to death (3). According to standard definition, muscle wasting is defined as loss of 5% or more of body weight over the preceding 6 mo, weight loss > 2% in the host body mass index (BMI) <20 kg/m2, or indications of sarcopenia that is linked with weight loss > 2% (4). However, different criteria were used to categorize patients with PDAC-associated cachexia in different studies published from different countries (Table 1). Recently, a comprehensive guideline for the management of cancer cachexia has been published (14). The biological mechanisms initiating cancer cachexia have been intensively studied. The heterogeneity of PDAC and the intertwined complex signaling pathways prompted through tumor and microenvironment interactions guide cachexia development (15). For example, the production of the tumor necrosis factor (TNF)-α and cytokines, which are proinflammatory factors commonly observed in PDAC, are considered to be among the drivers of cachexia in metastatic disease (16). Patients with PDAC progressively show loss of appetite, undergo chronic weight loss, and have continuing pain, alongside subdued gastrointestinal function that is caused by tumor invasion or side effects of combination regimens such as gemcitabine-nab-paclitaxel or FOLFIRINOX chemotherapy (17). These complex biological signals emanating from tumor, tumor-microenvironment, and tumor-host interactions render an environment conducive for disuse and untenable for proper muscle function leading to wasting (18). Aside from genetic signals, emerging evidence shows that both muscle development and cancer cachexia pathways are also regulated by epigenetic factors (19). Among these include the noncoding RNAs that have been proposed to influence cachexia initiation, sustenance, and also may be used as biomarkers for cachexia detection (20). Majority of the research on epigenetic interactors of cachexia has been focused on microRNAs. As the field on noncoding RNA evolves, aside from microRNAs, newer classes of noncoding RNAs are being studied in the context of cachexia. In this review, we discuss the various classes of noncoding RNAs such as long noncoding RNA (lncRNAs), small nucleolar RNA (snoRNAs), and circular RNAs (circRNA) for their role in PDAC-associated cachexia.
Table 1.
Diagnosis criteria, tumor stages, and prevalence of pancreatic cancer associated cachexia analyzed in different observational studies (3)
| Criteria for Cachexia Diagnosis | Tumor Stage | Age (yr) | Date of Study | Number of Patients | Cachexia Frequency in Patients (%) | Location of the Study | References |
|---|---|---|---|---|---|---|---|
| WL > 5% or BMI < 20 and WL 2%–5% | 86% UICC III/IV | 63 ± 13 | 2012–2014 | 94 | 74 | Italy | (5) |
| WL ≥ 5% at time of operation | 40% UICC III/IV | 57–70 | 2004–2005 | 227 | 41 | Germany | (6) |
| Diagnosis or BMI < 20 and/or WL ≥ 5% | Not reported | 63 ± 12 | 2002–2009 | 60 | 27 | Germany | (7) |
| WL ≥ 5% in last 6 mo | Not reported | 30–80 | 1976–1980 | 111 | 54 | USA | (8) |
| Diagnosis, cachexia medication, WL ≥ 5% | 40% metastatic | 64 ± 12 | 1999–2004 | 221 | 35 | USA | (9) |
| BMI < 18.5 + <75 yr or BMI < 21 + ≥75 yr and/or WL > 10% | 48% metastatic | 59 ± 13 | 2013 | 42 | 67 | France | (10) |
| Any WL | 64% metastatic | 30–80 | 1990 | 39 | 51 | USA | (11) |
| Any WL | 100% UICC III/IV | 21–80 | 1981–1983 | 63 | 89 | USA | (12) |
| Any WL before commencing chemotherapy | 100% UICC III/IV | 16–84 | 1990–1996 | 162 | 72 | UK | (13) |
BMI, body mass index; WL, weight loss.
PANCREATIC DUCTAL ADENOCARCINOMA AND CACHEXIA
Metastatic PDAC remains a deadly disease and is considered incurable. In general, <20% of patients with PDAC are eligible for surgical resection, which remains the only curative option. Majority of patients present when the disease has metastasized, and chemotherapy remains the only option. Over the years, much has been known about the sequential development of PDAC starting from PanIN lesions to PanIN (II), PanIN (III), and ductal adenocarcinoma (21). PDAC tumors arise from a sequential accumulation of mutations in Kirsten rat sarcoma virus (KRAS) (>80%), p53 (>50%), Suppressor of Mothers Against Decapentaplegic (SMAD), and CDKN2A leading to a transition from a PanIN lesion to ductal adenocarcinoma (22). PDAC tumors are heterogeneous carrying multiple populations of cell types including cancer cells embedded in the sea of cancer-associated fibroblasts (CAFs), stellate cells, induced pluripotent cells, cancer stem-like progenitor cells among others (23). These tumors are considered immunologically cold with reduced tumor-infiltrating lymphocytes and the predominant fraction of immune cells include macrophages and regulatory T cells that blunt the development of antitumor response (24). PDAC tumors thrive on the cross talk between different types of cells. For example, CAF-mediated signaling is known to promote tumor cell growth (18). At the same time tumor growth cell secretory factors have been shown to activate CAF tumor-promoting function. Excessive presence of CAFs in the surrounding microenvironment alongside the shielding effect of collagen reduces tumor vasculature creating a hypoxic microenvironment that does not allow proper penetration of chemotherapeutic drugs (25). Aside from genetic and cellular interactions, studies have also indicated epigenetic players responsible for pancreatic cancer development, subsistence, and therapy resistance. The noncoding RNAs (covered later in this review) have been identified to play critical roles in carcinogenesis, tumor sustenance, and therapy resistance (26).
The complex architecture of PDAC tumors along with intricate cross talk between genetic and epigenetic signaling has rendered the majority of targeted therapeutics and immunotherapies ineffective and a major fraction of patients are treated by chemotherapies. The Multi-chemo combination FOLFIRINOX is given to patients with PDAC with better performance status and has been shown to improve the median overall survival to little over 11 mo when compared with gemcitabine (6.4 mo) or gemcitabine plus nab-paclitaxel (8.6 mo; 27). Nevertheless, the majority of patients with PDAC have poor performance status and are only eligible for gemcitabine-nab-paclitaxel. No matter which therapy is given, these chemo combinations (both gemcitabine-nab-paclitaxel and FOLFIRINOX) have been shown to promote cachexia in the host (28). The consequent reduction in overall performance status and quality of life is dose-limiting and often does not allow completion of full cycle of chemo treatment (29). Due in part to cachexia, patients with advanced-stage PDAC often become ineligible for further continuation on the chemotherapy, have reduced appetite, reduced muscle function, resulting in inability to perform regular body functions. Therefore, understanding the underlying biology of higher incidence of cachexia in patients with PDAC and management of systems would not only help in the design of new treatment strategies, but it will also help in better treatment outcomes for patients with this deadly disease.
PANCREATIC DUCTAL ADENOCARCINOMA PHYSICAL AND CHEMICAL SIGNALS PROMOTING CACHEXIA
Mechanical and biological signals are responsible for PDAC-associated cachexia (30). Tumor invasion directly impacts pancreatic function and that leads to digestive disorders. Such digestive problems can lead to loss of appetite and reduction of food intake that in turn can exacerbate weight loss (31). Patients with PDAC universally present with symptoms of pancreas insufficiency such as pain, nausea, constipation, and malabsorption.
Aside from organ structural issues, the chemical cues from PDAC tumors have been shown to promote cachexia signaling. It is recognized that patients with PDAC have systemic inflammation that results in excessive release of cytokines (IL-10 and IL-6) as well as high C-reactive proteins, which are all recognized to be responsible for poor performance status, weight loss, and cachexia (32, 33). Excessive cytokine signals can also interfere with hypothalamus function and deregulating the sensors guiding energy intake thereby reducing appetite. Despite starvation, PDAC tumor-derived cytokines such as IL-1 have been shown to enhance the basal expression of energy sensor leptin thereby giving “do not eat” signals (34). In addition, studies have shown that excessive serotonin release, often found in PDAC tumors, can interfere with appetite sensors and hypothalamus function by directly interacting with pro-opiomelanocortin (POMC)/cocaine and amphetamine-regulated transcript (CART) neurons that are known to reduce food intake (35). The tumor necrosis factor-alpha (TNF-α) is elevated in PDAC and can induce cachexia. TNF-α has been shown to promote the expression of major muscle atrophy gene Atrogin-1/muscle atrophy F-box (MAFbx) in skeletal muscles leading to wasting (36). Studies have shown that tumor-derived factors such as lipid mobilizing factor (LMF) and proteolysis-inducing factor (PIF) are found elevated in urine samples in mouse models and patients with PDAC undergoing cachexia (37). LMF and PIF can promote fat loss in mouse models linking its role to overall body weight loss. There are many complex interactive signaling involved in the development of PDAC cachexia for which there are several excellent reviews. Here we focus on noncoding RNA signaling and its role in PDAC cachexia as described in the forthcoming passages.
PDAC METASTASIS SIGNALING CROSS TALK TO CACHEXIA
In most instances, cachexia occurs in end stage PDAC when the disease has metastasized. Therefore, the convergence of pancreatic cancer metastasis signaling and cachexia has been a topic that is intensively investigated. This connection becomes more relevant, as the development and progression of PDAC are fostered by inflammatory cells and cytokines, which are recognized to be central in promoting cachexia. Among the different interleukins, the interleukin-6 (IL-6) is a well-recognized cytokine that promotes protumorigenic microenvironment and is involved in PDAC angiogenesis and metastasis (38). IL-6 has been shown to be elevated in the serum of patients with pancreatic cancer and correlates with cachexia, advanced tumor stage, and poor survival (38). Further, along this line, other groups have investigated the role of interleukin-13 receptor α2 chain (IL-13Rα2) in PDAC metastasis and cachexia. Using a PDAC experimental in vivo model, it was shown that L-13Rα2-positive tumor metastasized more frequently to the lymph nodes, liver, and peritoneum at a significantly higher rate compared with IL-13Rα2-negative tumors (39). More importantly, IL-13Rα2-positive and metastatic models displayed cachexia. In yet another example, Underwood et al. (40) studied the role of nicotine (smoking being a risk factor in PDAC) on interleukins and cachexia. Using an experimental patient-derived tumor model, this group showed that nicotine-induced secretion of interleukin 8 (IL-8) by tumor-associated stroma cells in an extracellular signal-regulated kinase (ERK)-dependent fashion, resulting in enhanced tumor growth, metastasis, and more severe cachexia (40).
Work has also been done to evaluate the impact of PDAC tumor secretory factors on cachexia. For example, Yoo et al. (41) evaluated the role of PDAC-secreted pancreatic adenocarcinoma upregulated factor (PAUF) on cachexia development. PAUF is over-expressed in advanced PDAC and is recognized as an angiogenesis and immune evasion marker in PDAC and other solid tumors (42, 43). In view of these observations, Yoo et al. (41), further evaluated the link between PAUF and cachexia. Their studies showed that Panc-1 tumors supplemented with PAUF (Panc-1/PAUF) have superior body weight loss and cachectic symptoms compared with Panc-1/Mock tumors. Further, recombinant rPAUF introduction to muscle recapitulated muscle atrophy (41). Conversely, inhibiting PAUF through PAUF-neutralizing antibody abrogated muscle wasting in Panc-1/PAUF tumor harboring mice (41). In addition, these authors also utilized a C2C12 myotubes as a muscle atrophy model, which showed similar results with recombinant PAUF i.e., activation of Atrogin-1/MAFbx through Akt/Fork head box O (FOXO) pathway (41). Earlier studies showed that metal ion transporter Slc39a14 (Zip14) causes muscle wasting in a metastatic model of cancer. It is interesting to note that Zip14 is positively regulated by TNF-α and TGB-β (44); two markers that have been extensively evaluated for their role in PDAC metastasis as well as cachexia (45–47). Building on these studies, Shakri et al. (48) showed aberrant expression of ZIP14 and increased zinc ion levels in cachectic muscles in experimental metastasis models involving the murine PDAC cell lines, Pan02 and FC1242. This group further corroborated their studies demonstrating that in patients with advanced PDAC, high levels of ZIP14 in muscles correlated with the presence of cachexia (48). Collectively, the above studies clearly show that paracrine signaling from metastatic PDAC microenvironment can promote and exacerbate cachexia thereby contributing to this complex syndrome and associated morbidity.
NONCODING RNAs IN PDAC
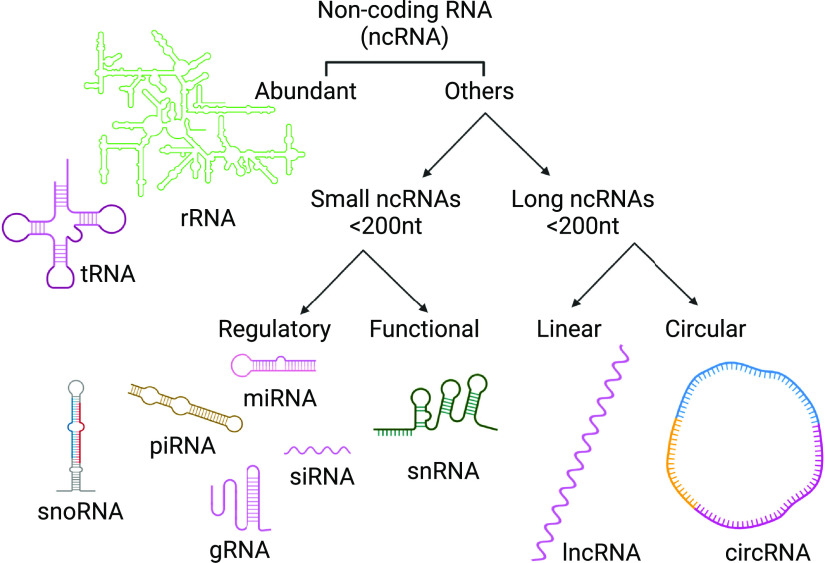
Cells carry a long list of ribonucleic acid (RNA) species. Messenger RNAs encode for proteins (transcription) and transfer RNAs help in the translation of mRNA messages to proteins (translation). Majority of the RNA in the cells is either ribosomal RNA (rRNA) or transfer RNA (tRNA). Besides coding RNAs, cells also carry noncoding RNAs that do not take part in protein synthesis. These include long noncoding RNAs (lncRNAs), microRNAs, piwi interacting RNAs (PiwiRNAs), small nucleolar RNA (snoRNAs), small nuclear RNAs (snRNAs), circular RNAs (circRNAs), and small interfering RNAs (siRNAs; 26). Majority of these RNA species are shorter than 200 nucleotides with the exception of lncRNAs that are larger in size. The size of circRNA ranges from ∼100 nucleotides to more than 4 kb (49). The ncRNAs are recognized to mediate gene regulation by either attaching to the 3' untranslated region (UTR) of mRNAs (by microRNAs, lncRNAs, piwiRNAs, snoRNAs, and snRNAs) or through sponging of mRNAs at different sites (by circRNAs). Various ncRNAs and their broad categories are shown in Fig. 1.
Figure 1.
Overview of noncoding RNAs (ncRNAs). Among ncRNAs, ribosomal RNA (rRNA), and transfer RNA (tRNA) are most abundant. Other ncRNAs can be categorized based on their size. Small ncRNA consists of <200 nucleotides (nt), whereas long ncRNA consists of more than 200 nt. The ncRNA further can be divided into linear (long ncRNA or lncRNA) and circular (circRNA) types. Small ncRNA such as microRNA (miRNA), guide RNA (gRNA), small interfering RNA (siRNA), small nucleolar RNA (snoRNA) plays a regulatory function. Small nuclear RNA (snRNA) shown to perform a functional role in a cell. The figure created with BioRender.com.
NcRNAs have been implicated in various stages of pancreatic tumor development. Majority of the studies have been done on microRNAs that have been found to be altered in PDAC (26). Besides tumor ncRNAs, studies have shown that ncRNAs from CAFs and other nontumor cells can influence PDAC tumor growth (50–52). These ncRNAs are also described for their role in regulating immune suppression that is commonly observed in PDAC (53). ncRNAs have been proposed to be important as diagnostic and prognostic markers of PDAC and are being explored for their utility as predictors of therapy response. These ncRNAs are stable in blood and therefore become ideal for monitoring disease progression over the course of treatment (54). Nevertheless, there is no specific set of ncRNAs that can be used for early detection or as signatures for response to therapies and such studies are intensively being pursued by the research community. As with PDAC signaling, nc-RNAs have been implicated in cancer cachexia and muscle wasting. A search on PubMed using keywords “muscle wasting non-coding RNAs” and “cachexia non-coding RNAs” yielded 545 and 94 results. In addition, a search using keywords “muscle wasting microRNAs” yielded 338 papers. This shows that there is ample evidence on the role of ncRNAs in the biology of cachexia and muscle wasting which is discussed Noncoding RNAs in cachexia section.
NONCODING RNAs IN CACHEXIA
A number of noncoding RNAs have been implicated in cachexia and muscle wasting. In fact, there are a series of ncRNAs specifically enriched in muscles and have been termed myomiRs. The initial studies were focused on investigating the differential expression of ncRNAs in muscle undergoing wasting. In a meta-analysis using nine different muscle-wasting studies, Freire et al. (55) identified several microRNA-mRNA interactions (miR-27a/Foxo1, miR-27a/Mef2c, miR-27b/Cxcl12, miR-27b/Mef2c, miR-140/Cxcl12, miR-199a/Cav1, and miR-199a/Junb), which were proposed to be involved in muscle wasting. In another early study using transcriptomics analysis in anx/anx anorexia mouse models showed certain microRNAs to be deregulated in the hypothalamus and cortex thereby linking microRNA machinery to anorexia and cachexia (56). Kulyté et al. (57) performed microRNA profiling of adipose tissue obtained from gastrointestinal cancer patients with or without cachexia. Their results indicated upregulation of 4 microRNAs (miR-483-5p/-23a/-744/-99b) and downregulation of mir-378 specifically in patients with cachexia (57). In another study, 22 cachectic patients were compared with 20 noncachectic patients for the differential expression of noncoding RNAs. Their results identified eight differentially expressed microRNAs specific in patients with cachexia that were confirmed to be linked to myogenesis and inflammatory pathways (58). Soares et al.(59) showed that microRNA-206 and -21 were induced in denervated muscles and were sufficient to induce muscle atrophy through downstream modulation of transcription factor YY1 and the translational initiator factor eIF4E3. In another study using Lewis lung carcinoma cells (LLC) tumor and cachexia models in C57BL/6J371 mice, Lee et al. (60) showed that there were 371 miRNAs to be statistically enhanced in the muscle tissue compared with mice without tumors. In a genetic mouse model, Chacon-Cabrera et al. (61) showed that PARP knockout could reverse lower microRNAs (miR-1, -133, -206, and -486) acetylation, and reverse cachexia symptoms. Yang et al. (62) showed that circRNAs (circFgfr2, circQrich1, circMettl9, and circCamta1) were differentially expressed that targeted JAK/STAT pathway in pig cachexia model (62). Similarly, exosomal/microvesicle-enclosed microRNA-21 (mir-21) has been shown to exert muscle loss through deregulation of JAK/STAT signaling (63).
PANCREATIC CANCER CACHEXIA ASSOCIATED NcRNAs
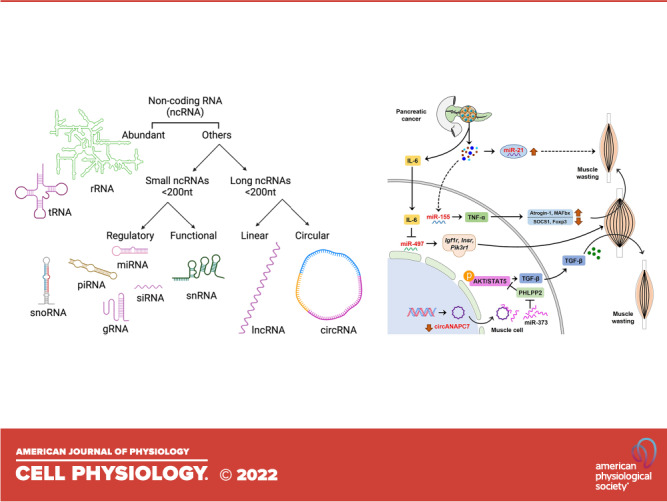
There are several studies linking aberrant ncRNAs in pancreatic cancer cachexia (Table 2). In an in vitro model, IMO-8503, a TLR7, 8, and 9 antagonist could resolve cachexia symptoms through inhibition of mir-21 in a mouse model of cachexia (66). In a case-control study in pancreatic cancer and non-small cell lung cancer (NSCLC), the expression of mir-155 was higher in cachexia and was linked to the severity in the cachectic group as compared with normal controls (64). This study showed that miR-155 mediated modulation of TNF-α, a well-recognized promoter of muscle wasting. Besides microRNAs, very recently, Shi and colleagues (67) showed that circANAPC7 acted as a sponge for miR-373, which inhibited tumor growth and muscle wasting in vitro and in vivo through PHLPP2-AKT-TGF-β signaling axis in pancreatic cancer. Roles of ncRNAs in pancreatic cancer-associated cachexia are illustrated in Fig. 2.
Table 2.
Noncoding RNAs involved in pancreatic cancer associated muscle wasting
| ncRNAs | Type of Study | Expression | Biological Significance | References |
|---|---|---|---|---|
| miR-21 | In vitro: myoblasts from TLR7−/− and TLR7+/+ mice | Overexpressed in microvesicles secreted by lung and pancreatic cancer cells | Involved in TLR7+/+ myoblast cell death | (63) |
| miR-3184-3p | In vivo: Rectus abdominis from patients with pancreatic and colorectal cancer | Upregulated | Plays critical roles in adipogenesis, myogenesis, signal transduction pathways, inflammation, and innate immune response | (58) |
| miR-423-5p | ||||
| let-7d-3p | ||||
| miR-1296-5p | ||||
| miR-345-5p | ||||
| miR-532-5p | ||||
| miR-423-3p | ||||
| miR-199a-3p | ||||
| miR-155 | Clinical: a case-control study involving 203 patients with pancreatic and lung cancer with mean age 51.45 ± 9.7 ranging from 20 to 77 yr | Highly overexpressed in patients with cachexia (424-fold increase) | miR-155 act as a regulator for TNF-α and its downstream targets SOCS1, TAB2, and Foxp3 | (64) |
| miR-497-5p | In silico meta-analysis: NCBI Gene Expression Omnibus (GEO) data sets for pancreatic (GSE51931) and colon cancers (GSE48363, GSE63032, and GSE24112) | Downregulated | miR-497-5p level decreased by IL-6 under atrophic condition thus fail to induce its target genes Igf1r, Insr, and Pik3r1 associated with skeletal muscle hypertrophy | (65) |
| circANAPC7 | Preclinical/clinical: pancreatic cancer cells, three-dimensional spheroids and organoids, mouse models, and clinical specimens | Downregulated | circANAPC7 act as sponge for miR-373 and functions through the CREB-miR-373-PHLPP2 axis that causes AKT dephosphorylation, and cyclin D1 and TGF-β downregulation leading to suppression of muscle wasting in pancreatic cancer | (61) |
CREB, cAMP response element-binding; ncRNAs, noncoding RNAs; TNF, tumor necrosis factor.
Figure 2.
Noncoding RNAs involved in pancreatic cancer associated cachexia. Secretory microvesicles from pancreatic cancer cells are enriched in miR-21 causing death to myoblast via a currently unknown mechanism that leads to muscle wasting. Micro RNA miR-155 is highly overexpressed in patients with cachexia, which regulate TNF-α leading to muscle wasting via modulation of several downstream molecules. Pancreatic tumor cells release IL-6, which in turn inhibit miR-497. MiR-497 is associated with skeletal muscle hypertrophy via induction of its target genes Igf1r, Insr, and Pik3r1. MiR-373 causes AKT and STAT5 phosphorylation by inhibiting PHLPP2. This results in upregulation of TGF-β and promotes muscle wasting. Circular RNA circANAPC7 acts as a sponge for miR-373, however, in pancreatic cancer this circular RNA is downregulated. Dashed line indicates multiple currently unknown steps; arrows indicate promotion; and blunt ended lines indicate inhibition. TNF, tumor necrosis factor.
TARGETING NcRNAs TO REVERSE CACHEXIA
The best approach to deal with cachexia is the curative treatment of cancer, however, this is not possible for many types of malignancies including pancreatic cancer (68, 69). NcRNAs control many signaling pathways associated with the pathophysiology of cancer cachexia making them suitable targets for therapy. Studies have shown that ncRNAs can be targeted to reverse cachexia symptoms at least in cellular models of muscle atrophy. For example, recently it was shown that targeting miR-497-5p using miR inhibitors can prevent muscle IL-6-induced atrophy in C2C12 cells (65). It has been demonstrated that circular RNA circANAPC7 can inhibit pancreatic cancer-associated cachexia via downregulation of CREB-miR-373-PHLPP2 axis (67). Targeting circANAPC7-mediated regulation of cachexia could lead to the development of a novel treatment strategy for cachexia as well as pancreatic cancer.
NcRNAs AS BIOMARKERS FOR PDAC ASSOCIATED CACHEXIA
NcRNAs specially circulating microRNAs have been regarded as suitable biomarkers for assessing risk of cancer cachexia development (70). For example, the myomiRs could be used as biomarkers for cachectic patient’s follow-up due to its association with resistance exercise, which could be deleterious for the cachectic patients (71). Despite the importance of ncRNAs, none has been described in pancreatic cancer-associated cachexia so far. However, a number of ncRNA-regulated genes and their corresponding proteins shown to be critical in the regulation of cachexia in pancreatic cancer. Studies in the preclinical and clinical settings demonstrated that uncoupling protein 1 (UCP1) upregulation in subcutaneous adipose tissue (SAT), could serve as a biomarker of cachectic and newly hyperglycemic PDAC patients (72). A significant difference in UPC1 expression can be detected as early as 6 mo before the diagnosis of PDAC. The study detected UCP1 overexpression in SAT treated with PDAC exosomes in vitro as well as in vivo PDAC mouse model. Among patients with PDAC, all five showed UCP1 overexpression compared with only one out of four controls (72). IL-13Rα2 has been shown to promote cachexia in PDAC orthotropic nude mice xenograft model system and silencing of IL-13Rα2 in HS766T cells was able to prevent cachexia in this mice model suggesting its important role (39). There is a scarcity of blood-based biomarkers to determine muscle mass change (e.g., myostatin and arginine; 73–75). Considering the stability, the noninvasive collection method, and sensitive quantitative detection, ncRNAs could be the ideal biomarkers for cachexia in PDAC. A preclinical study predicted the potential involvement of miRNAs such as miR-9 and miR-338-3p in the modulation of aforementioned UCP1 expression by targeting 3'-UTRs of UCP1 mRNA (76). Other studies showed the involvement of multiple ncRNAs namely miR-27b, miR-494-3p, MiR‐92a, miR-30b/c, miR-106b, and miR-93 in differential regulation of UCP1 (77–81). Moreover, a clinical trial is (NCT05275075) ongoing to identify microRNAs associated with pancreatic cancer-related cachexia. Further study should demonstrate the novel role of ncRNAs in regulating cachexia in pancreatic cancers.
CONCLUSIONS
Cancer-associated cachexia is one of the major obstacles for the treatment of patients with cancer particularly many patients with pancreatic cancer. Moreover, the scarcity of efficient therapies against cachexia worsens the quality of patients’ lives. Currently, there is a lack of knowledge on deeper understanding of molecular mechanisms involved in cachexia. Such unmet need in turn has delayed the discovery of appropriate biomarkers to determine cachexia at different stages of pancreatic cancer. Noncoding RNAs are stable and involved in many crucial biological processes, which can serve as promising biomarkers for diagnosis, prognosis, and follow-ups. Recent study confirms the involvement of a few ncRNAs including miRNA and circRNA in pancreatic cancer-associated cachexia. There are hints that in addition IL6, proteasome, certain novel signaling [e.g., Wnt signaling, natural killer (NK) cells] pathways are involved (82). These findings are preliminary and further large-scale exploration is necessary to find suitable ncRNAs for proper diagnosis and monitoring of cachexia associated with pancreatic cancer. It will also be interesting to decipher the molecular difference between pancreatic cancer and cachexia stages and their cross talk. Understanding pathophysiology of these parallel events in relation to ncRNAs could lead to effective therapeutic targets and identification of biomarkers.
GRANTS
This work was supported by National Cancer Institute Grants 5R37CA215427 and 5R01CA240607 (to A.S. Azmi).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
M.H.U., A.S.A., and I.M. conceived and designed research; M.H.U. prepared figures; M.H.U., R.M.M., A.S.A., and I.M. drafted manuscript; M.H.U., R.M.M., P.A.P., A.S.A., and I.M. edited and revised manuscript; M.H.U., R.M.M., P.A.P., A.S.A., and I.M. approved final version of manuscript.
REFERENCES
- 1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71: 209–249, 2021. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2. Poulia KA, Antoniadou D, Sarantis P, Karamouzis MV. Pancreatic cancer prognosis, malnutrition risk, and quality of life: a cross-sectional study. Nutrients 14: 442, 2022. doi: 10.3390/nu14030442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Anker MS, Holcomb R, Muscaritoli M, von Haehling S, Haverkamp W, Jatoi A, Morley JE, Strasser F, Landmesser U, Coats AJS, Anker SD. Orphan disease status of cancer cachexia in the USA and in the European Union: a systematic review. J Cachexia Sarcopenia Muscle 10: 22–34, 2019. doi: 10.1002/jcsm.12402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Fram J, Vail C, Roy I. Assessment of cancer-associated cachexia - how to approach physical function evaluation. Curr Oncol Rep 24: 751–761, 2022. doi: 10.1007/s11912-022-01258-4. [DOI] [PubMed] [Google Scholar]
- 5. Muscaritoli M, Lucia S, Farcomeni A, Lorusso V, Saracino V, Barone C, Plastino F, Gori S, Magarotto R, Carteni G, Chiurazzi B, Pavese I, Marchetti L, Zagonel V, Bergo E, Tonini G, Imperatori M, Iacono C, Maiorana L, Pinto C, Rubino D, Cavanna L, Di Cicilia R, Gamucci T, Quadrini S, Palazzo S, Minardi S, Merlano M, Colucci G, Marchetti P; PreMiO Study Group. Prevalence of malnutrition in patients at first medical oncology visit: the PreMiO study. Oncotarget 8: 79884–79896, 2017. doi: 10.18632/oncotarget.20168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Bachmann J, Heiligensetzer M, Krakowski-Roosen H, Büchler MW, Friess H, Martignoni ME. Cachexia worsens prognosis in patients with resectable pancreatic cancer. J Gastrointest Surg 12: 1193–1201, 2008. doi: 10.1007/s11605-008-0505-z. [DOI] [PubMed] [Google Scholar]
- 7. Barkhudaryan A, Scherbakov N, Springer J, Doehner W. Cardiac muscle wasting in individuals with cancer cachexia. ESC Heart Fail 4: 458–467, 2017. doi: 10.1002/ehf2.12184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Dewys WD, Begg C, Lavin PT, Band PR, Bennett JM, Bertino JR, Cohen MH, Douglass HO, Engstrom PF, Ezdinli EZ, Horton J, Johnson GJ, Moertel CG, Oken MM, Perlia C, Rosenbaum C, Silverstein MN, Skeel RT, Sponzo RW, Tormey DC. Prognostic effect of weight loss prior to chemotherapy in cancer patients. Eastern Cooperative Oncology Group. Am J Med 69: 491–497, 1980. doi: 10.1016/s0149-2918(05)80001-3. [DOI] [PubMed] [Google Scholar]
- 9. Fox KM, Brooks JM, Gandra SR, Markus R, Chiou CF. Estimation of cachexia among cancer patients based on four definitions. J Oncol 2009: 693458, 2009. doi: 10.1155/2009/693458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Hebuterne X, Lemarié E, Michallet M, de Montreuil CB, Schneider SM, Goldwasser F. Prevalence of malnutrition and current use of nutrition support in patients with cancer. JPEN J Parenter Enteral Nutr 38: 196–204, 2014. doi: 10.1177/0148607113502674. [DOI] [PubMed] [Google Scholar]
- 11. Krech RL, Walsh D. Symptoms of pancreatic cancer. J Pain Symptom Manage 6: 360–367, 1991. doi: 10.1016/0885-3924(91)90027-2. [DOI] [PubMed] [Google Scholar]
- 12. Wachtel T, Allen-Masterson S, Reuben D, Goldberg R, Mor V. The end stage cancer patient: terminal common pathway. Hosp J 4: 43–80, 1988. doi: 10.1080/0742-969x.1988.11882634. [DOI] [PubMed] [Google Scholar]
- 13. Andreyev HJ, Norman AR, Oates J, Cunningham D. Why do patients with weight loss have a worse outcome when undergoing chemotherapy for gastrointestinal malignancies? Eur J Cancer 34: 503–509, 1998. doi: 10.1016/s0959-8049(97)10090-9. [DOI] [PubMed] [Google Scholar]
- 14. Roeland EJ, Bohlke K, Baracos VE, Bruera E, Del Fabbro E, Dixon S, Fallon M, Herrstedt J, Lau H, Platek M, Rugo HS, Schnipper HH, Smith TJ, Tan W, Loprinzi CL. Management of cancer cachexia: ASCO guideline. J Clin Oncol 38: 2438–2453, 2020. doi: 10.1200/JCO.20.00611. [DOI] [PubMed] [Google Scholar]
- 15. Webster JM, Kempen L, Hardy RS, Langen RCJ. Inflammation and skeletal muscle wasting during cachexia. Front Physiol 11: 597675, 2020. doi: 10.3389/fphys.2020.597675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Paval DR, Patton R, McDonald J, Skipworth RJE, Gallagher IJ, Laird BJ; Caledonian Cachexia Collaborative. A systematic review examining the relationship between cytokines and cachexia in incurable cancer. J Cachexia Sarcopenia Muscle 13: 824–838, 2022. doi: 10.1002/jcsm.12912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kays JK, Shahda S, Stanley M, Bell TM, O'Neill BH, Kohli MD, Couch ME, Koniaris LG, Zimmers TA. Three cachexia phenotypes and the impact of fat-only loss on survival in FOLFIRINOX therapy for pancreatic cancer. J Cachexia Sarcopenia Muscle 9: 673–684, 2018. doi: 10.1002/jcsm.12307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Shukla SK, Markov SD, Attri KS, Vernucci E, King RJ, Dasgupta A, Grandgenett PM, Hollingsworth MA, Singh PK, Yu F, Mehla K. Macrophages potentiate STAT3 signaling in skeletal muscles and regulate pancreatic cancer cachexia. Cancer Lett 484: 29–39, 2020. doi: 10.1016/j.canlet.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Barreiro E, Tajbakhsh S. Epigenetic regulation of muscle development. J Muscle Res Cell Motil 38: 31–35, 2017. doi: 10.1007/s10974-017-9469-5. [DOI] [PubMed] [Google Scholar]
- 20. Liu Q, Deng J, Qiu Y, Gao J, Li J, Guan L, Lee H, Zhou Q, Xiao J. Non-coding RNA basis of muscle atrophy. Mol Ther Nucleic Acids 26: 1066–1078, 2021. doi: 10.1016/j.omtn.2021.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Noë M, Hong SM, Wood LD, Thompson ED, Roberts NJ, Goggins MG, Klein AP, Eshleman JR, Kern SE, Hruban RH. Pancreatic cancer pathology viewed in the light of evolution. Cancer Metastasis Rev 40: 661–674, 2021. doi: 10.1007/s10555-020-09953-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Maitra A, Hruban RH. Pancreatic cancer. Annu Rev Pathol 3: 157–188, 2008. doi: 10.1146/annurev.pathmechdis.3.121806.154305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Herting CJ, Karpovsky I, Lesinski GB. The tumor microenvironment in pancreatic ductal adenocarcinoma: current perspectives and future directions. Cancer Metastasis Rev 40: 675–689, 2021. doi: 10.1007/s10555-021-09988-w. [DOI] [PubMed] [Google Scholar]
- 24. Schmiechen ZC, Stromnes IM. Mechanisms governing immunotherapy resistance in pancreatic ductal adenocarcinoma. Front Immunol 11: 613815, 2020. doi: 10.3389/fimmu.2020.613815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Carvalho TMA, Di Molfetta D, Greco MR, Koltai T, Alfarouk KO, Reshkin SJ, Cardone RA. Tumor microenvironment features and chemoresistance in pancreatic ductal adenocarcinoma: insights into targeting physicochemical barriers and metabolism as therapeutic approaches. Cancers (Basel) 13: 6135, 2021. doi: 10.3390/cancers13236135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Li Y, Al Hallak MN, Philip PA, Azmi AS, Mohammad RM. Non-coding RNAs in pancreatic cancer diagnostics and therapy: focus on lncRNAs, circRNAs, and piRNAs. Cancers (Basel) 13: 4161, 2021. doi: 10.3390/cancers13164161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. De Dosso S, Siebenhüner AR, Winder T, Meisel A, Fritsch R, Astaras C, Szturz P, Borner M. Treatment landscape of metastatic pancreatic cancer. Cancer Treat Rev 96: 102180, 2021. doi: 10.1016/j.ctrv.2021.102180. [DOI] [PubMed] [Google Scholar]
- 28. Mitsunaga S, Kasamatsu E, Machii K. Incidence and frequency of cancer cachexia during chemotherapy for advanced pancreatic ductal adenocarcinoma. Support Care Cancer 28: 5271–5279, 2020. doi: 10.1007/s00520-020-05346-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Kiriukova M, de la Iglesia Garcia D, Panic N, Bozhychko M, Avci B, Maisonneuve P, de-Madaria E, Capurso G, Sandru V. Pancreatic cancer malnutrition and pancreatic exocrine insufficiency in the course of chemotherapy in unresectable pancreatic cancer. Front Med (Lausanne) 7: 495, 2020. doi: 10.3389/fmed.2020.00495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Kordes M, Larsson L, Engstrand L, Löhr JM. Pancreatic cancer cachexia: three dimensions of a complex syndrome. Br J Cancer 124: 1623–1636, 2021. doi: 10.1038/s41416-021-01301-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Santos I, Mendes L, Mansinho H, Santos CA. Nutritional status and functional status of the pancreatic cancer patients and the impact of adjacent symptoms. Clin Nutr 40: 5486–5493, 2021. doi: 10.1016/j.clnu.2021.09.019. [DOI] [PubMed] [Google Scholar]
- 32. Falconer JS, Fearon KC, Plester CE, Ross JA, Carter DC. Cytokines, the acute-phase response, and resting energy expenditure in cachectic patients with pancreatic cancer. Ann Surg 219: 325–331, 1994. doi: 10.1097/00000658-199404000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Moses AG, Maingay J, Sangster K, Fearon KC, Ross JA. Pro-inflammatory cytokine release by peripheral blood mononuclear cells from patients with advanced pancreatic cancer: relationship to acute phase response and survival. Oncol Rep 21: 1091–1095, 2009. doi: 10.3892/or_00000328. [DOI] [PubMed] [Google Scholar]
- 34. Lipsey CC, Harbuzariu A, Daley-Brown D, Gonzalez-Perez RR. Oncogenic role of leptin and Notch interleukin-1 leptin crosstalk outcome in cancer. World J Methodol 6: 43–55, 2016. doi: 10.5662/wjm.v6.i1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Gullo L, De Giorgio R, D'Errico A, Grigioni W, Parenti M, Corinaldesi R. Pancreatic exocrine carcinoma producing adrenocorticotropic hormone. Pancreas 7: 172–176, 1992. doi: 10.1097/00006676-199203000-00007. [DOI] [PubMed] [Google Scholar]
- 36. Patel HJ, Patel BM. TNF-alpha and cancer cachexia: molecular insights and clinical implications. Life Sci 170: 56–63, 2017. doi: 10.1016/j.lfs.2016.11.033. [DOI] [PubMed] [Google Scholar]
- 37. Wigmore SJ, Todorov PT, Barber MD, Ross JA, Tisdale MJ, Fearon KC. Characteristics of patients with pancreatic cancer expressing a novel cancer cachectic factor. Br J Surg 87: 53–58, 2000. doi: 10.1046/j.1365-2168.2000.01317.x. [DOI] [PubMed] [Google Scholar]
- 38. Holmer R, Goumas FA, Waetzig GH, Rose-John S, Kalthoff H. Interleukin-6: a villain in the drama of pancreatic cancer development and progression. Hepatobiliary Pancreat Dis Int 13: 371–380, 2014. doi: 10.1016/s1499-3872(14)60259-9. [DOI] [PubMed] [Google Scholar]
- 39. Fujisawa T, Joshi B, Nakajima A, Puri RK. A novel role of interleukin-13 receptor alpha2 in pancreatic cancer invasion and metastasis. Cancer Res 69: 8678–8685, 2009. doi: 10.1158/0008-5472.CAN-09-2100. [DOI] [PubMed] [Google Scholar]
- 40. Underwood PW, Zhang DY, Cameron ME, Gerber MH, Delitto D, Maduka MU, Cooper KJ, Han S, Hughes SJ, Judge SM, Judge AR, Trevino JG. Nicotine induces IL-8 secretion from pancreatic cancer stroma and worsens cancer-induced cachexia. Cancers (Basel) 12: 329, 2020. doi: 10.3390/cancers12020329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Yoo W, Choi H, Son YH, Lee J, Jo S, Jung D, Kim YJ, Koh SS, Yang YR, Kwon E-S, Lee K-P, Noh KH, Kim KW, Ko Y, Jun E, Kim SC, Kim S. Pancreatic cancer induces muscle wasting by promoting the release of pancreatic adenocarcinoma upregulated factor. Exp Mol Med 53: 432–445, 2021. doi: 10.1038/s12276-021-00582-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Cho I-R, Koh SS, Min H-J, Kim SJ, Lee Y, Park E-H, Ratakorn S, Jhun BH, Oh S, Johnston RN, Chung Y-H. Pancreatic adenocarcinoma up-regulated factor (PAUF) enhances the expression of beta-catenin, leading to a rapid proliferation of pancreatic cells. Exp Mol Med 43: 82–90, 2011. doi: 10.3858/emm.2011.43.2.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Kim SA, Lee Y, Jung DE, Park KH, Park JY, Gang J, Jeon SB, Park EC, Kim Y-G, Lee B, Liu Q, Zeng W, Yeramilli S, Lee S, Koh SS, Song SY. Pancreatic adenocarcinoma up-regulated factor (PAUF), a novel up-regulated secretory protein in pancreatic ductal adenocarcinoma. Cancer Sci 100: 828–836, 2009. doi: 10.1111/j.1349-7006.2009.01106.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Wang G, Biswas AK, Ma W, Kandpal M, Coker C, Grandgenett PM, Hollingsworth MA, Jain R, Tanji K, Lόpez-Pintado S, Borczuk A, Hebert D, Jenkitkasemwong S, Hojyo S, Davuluri RV, Knutson MD, Fukada T, Acharyya S. Metastatic cancers promote cachexia through ZIP14 upregulation in skeletal muscle. Nat Med 24: 770–781, 2018. doi: 10.1038/s41591-018-0054-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Waning DL, Mohammad KS, Reiken S, Xie W, Andersson DC, John S, Chiechi A, Wright LE, Umanskaya A, Niewolna M, Trivedi T, Charkhzarrin S, Khatiwada P, Wronska A, Haynes A, Benassi MS, Witzmann FA, Zhen G, Wang X, Cao X, Roodman GD, Marks AR, Guise TA. Excess TGF-β mediates muscle weakness associated with bone metastases in mice. Nat Med 21: 1262–1271, 2015. doi: 10.1038/nm.3961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Balkwill F. TNF-alpha in promotion and progression of cancer. Cancer Metastasis Rev 25: 409–416, 2006. doi: 10.1007/s10555-006-9005-3. [DOI] [PubMed] [Google Scholar]
- 47. Kang Y, He W, Tulley S, Gupta GP, Serganova I, Chen C-R, Manova-Todorova K, Blasberg R, Gerald WL, Massagué J. Breast cancer bone metastasis mediated by the Smad tumor suppressor pathway. Proc Natl Acad Sci USA 102: 13909–13914, 2005. [Erratum in Proc Natl Acad Sci USA 103: 8570, 2006]. doi: 10.1073/pnas.0506517102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Shakri AR, Zhong TJ, Ma W, Coker C, Kim S, Calluori S, Scholze H, Szabolcs M, Caffrey T, Grandgenett PM, Hollingsworth MA, Tanji K, Kluger MD, Miller G, Biswas AK, Acharyya S. Upregulation of ZIP14 and altered zinc homeostasis in muscles in pancreatic cancer cachexia. Cancers (Basel) 12: 3, 2019. doi: 10.3390/cancers12010003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Szabo L, Salzman J. Detecting circular RNAs: bioinformatic and experimental challenges. Nat Rev Genet 17: 679–692, 2016. doi: 10.1038/nrg.2016.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Donahue TR, Nguyen AH, Moughan J, Li L, Tatishchev S, Toste P, Farrell JJ. Stromal microRNA-21 levels predict response to 5-fluorouracil in patients with pancreatic cancer. J Surg Oncol 110: 952–959, 2014. doi: 10.1002/jso.23750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Chen S, Chen X, Shan T, Ma J, Lin W, Li W, Kang Y. MiR-21-mediated metabolic alteration of cancer-associated fibroblasts and its effect on pancreatic cancer cell behavior. Int J Biol Sci 14: 100–110, 2018. doi: 10.7150/ijbs.22555. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 52. Fang Y, Zhou W, Rong Y, Kuang T, Xu X, Wu W, Wang D, Lou W. Exosomal miRNA-106b from cancer-associated fibroblast promotes gemcitabine resistance in pancreatic cancer. Exp Cell Res 383: 111543, 2019. doi: 10.1016/j.yexcr.2019.111543. [DOI] [PubMed] [Google Scholar]
- 53. Xiao X, Cheng W, Zhang G, Wang C, Sun B, Zha C, Kong F, Jia Y. Long noncoding RNA: shining stars in the immune microenvironment of gastric cancer. Front Oncol 12: 862337, 2022. doi: 10.3389/fonc.2022.862337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Toden S, Goel A. Non-coding RNAs as liquid biopsy biomarkers in cancer. Br J Cancer 126: 351–360, 2022. doi: 10.1038/s41416-021-01672-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Freire PP, Fernandez GJ, Cury SS, de Moraes D, Oliveira JS, de Oliveira G, Dal-Pai-Silva M, dos Reis PP, Carvalho RF. The pathway to cancer cachexia: microRNA-regulated networks in muscle wasting based on integrative meta-analysis. IJMS 20: 1962, 2019. doi: 10.3390/ijms20081962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Mercader JM, González JR, Lozano JJ, Bak M, Kauppinen S, Sumoy L, Dierssen M, Fernández-Aranda F, Visa J, Gratacòs M, Estivill X. Aberrant brain microRNA target and miRISC gene expression in the anx/anx anorexia mouse model. Gene 497: 181–190, 2012. doi: 10.1016/j.gene.2012.01.057. [DOI] [PubMed] [Google Scholar]
- 57. Kulyté A, Lorente-Cebrián S, Gao H, Mejhert N, Agustsson T, Arner P, Rydén M, Dahlman I. MicroRNA profiling links miR-378 to enhanced adipocyte lipolysis in human cancer cachexia. Am J Physiol Endocrinol Physiol 306: E267–E274, 2014. doi: 10.1152/ajpendo.00249.2013. [DOI] [PubMed] [Google Scholar]
- 58. Narasimhan A, Ghosh S, Stretch C, Greiner R, Bathe OF, Baracos V, Damaraju S. Small RNAome profiling from human skeletal muscle: novel miRNAs and their targets associated with cancer cachexia. J Cachexia Sarcopenia Muscle 8: 405–416, 2017. doi: 10.1002/jcsm.12168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Soares RJ, Cagnin S, Chemello F, Silvestrin M, Musaro A, De Pitta C, Lanfranchi G, Sandri M. Involvement of microRNAs in the regulation of muscle wasting during catabolic conditions. J Biol Chem 289: 21909–21925, 2014. doi: 10.1074/jbc.M114.561845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Lee DE, Brown JL, Rosa-Caldwell ME, Blackwell TA, Perry RA Jr, Brown LA, Khatri B, Seo D, Bottje WG, Washington TA, Wiggs MP, Kong B-W, Greene NP. Cancer cachexia-induced muscle atrophy: evidence for alterations in microRNAs important for muscle size. Physiol Genomics 49: 253–260, 2017. doi: 10.1152/physiolgenomics.00006.2017. [DOI] [PubMed] [Google Scholar]
- 61. Chacon-Cabrera A, Fermoselle C, Salmela I, Yelamos J, Barreiro E. MicroRNA expression and protein acetylation pattern in respiratory and limb muscles of Parp-1(−/−) and Parp-2(−/−) mice with lung cancer cachexia. Biochim Biophys Acta 1850: 2530–2543, 2015. doi: 10.1016/j.bbagen.2015.09.020. [DOI] [PubMed] [Google Scholar]
- 62. Yan J, Yang Y, Fan X, Liang G, Wang Z, Li J, Wang L, Chen Y, Adetula AA, Tang Y, Li K, Wang D, Tang Z. circRNAome profiling reveals circFgfr2 regulates myogenesis and muscle regeneration via a feedback loop. J Cachexia Sarcopenia Muscle 13: 696–712, 2022. doi: 10.1002/jcsm.12859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. He WA, Calore F, Londhe P, Canella A, Guttridge DC, Croce CM. Microvesicles containing miRNAs promote muscle cell death in cancer cachexia via TLR7. Proc Natl Acad Sci USA 111: 4525–4529, 2014. doi: 10.1073/pnas.1402714111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Yehia R, Schaalan M, Abdallah DM, Saad AS, Sarhan N, Saleh S. Impact of TNF-α gene polymorphisms on pancreatic and non-small cell lung cancer-induced cachexia in adult Egyptian patients: a focus on pathogenic trajectories. Front Oncol 11: 783231, 2021. doi: 10.3389/fonc.2021.783231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Freire PP, Cury SS, Lopes LO, Fernandez GJ, Liu J, de Moraes LN, de Oliveira G, Oliveira JS, de Moraes D, Cabral-Marques O, Dal-Pai-Silva M, Hu X, Wang D-Z, Carvalho RF. Decreased miR-497-5p suppresses IL-6 induced atrophy in muscle cells. Cells 10: 3527, 2021. doi: 10.3390/cells10123527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Calore F, Londhe P, Fadda P, Nigita G, Casadei L, Marceca GP, Fassan M, Lovat F, Gasparini P, Rizzotto L, Zanesi N, Jackson D, Mehta S, Nana-Sinkam P, Sampath D, Pollock RE, Guttridge DC, Croce CM. The TLR7/8/9 antagonist IMO-8503 inhibits cancer-induced cachexia. Cancer Res 78: 6680–6690, 2018. doi: 10.1158/0008-5472.CAN-17-3878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Shi X, Yang J, Liu M, Zhang Y, Zhou Z, Luo W, Fung KM, Xu C, Bronze MS, Houchen CW, Li M. Circular RNA ANAPC7 inhibits tumor growth and muscle wasting via PHLPP2-AKT-TGF-β signaling axis in pancreatic cancer. Gastroenterology 162: 2004–2017.e2, 2022. doi: 10.1053/j.gastro.2022.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Inui A. Cancer anorexia-cachexia syndrome: current issues in research and management. CA Cancer J Clin 52: 72–91, 2002. doi: 10.3322/canjclin.52.2.72. [DOI] [PubMed] [Google Scholar]
- 69. Mueller TC, Bachmann J, Prokopchuk O, Friess H, Martignoni ME. Molecular pathways leading to loss of skeletal muscle mass in cancer cachexia—can findings from animal models be translated to humans? BMC Cancer 16: 75, 2016. doi: 10.1186/s12885-016-2121-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Santos JMO, Peixoto da Silva S, Gil da Costa RM, Medeiros R. The emerging role of microRNAs and other non-coding RNAs in cancer cachexia. Cancers (Basel) 12: 1004, 2020. doi: 10.3390/cancers12041004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Molinari F, Pin F, Gorini S, Chiandotto S, Pontecorvo L, Penna F, Rizzuto E, Pisu S, Musarò A, Costelli P, Rosano G, Ferraro E. The mitochondrial metabolic reprogramming agent trimetazidine as an 'exercise mimetic' in cachectic C26-bearing mice. J Cachexia Sarcopenia Muscle 8: 954–973, 2017. doi: 10.1002/jcsm.12226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Sah RP, Sharma A, Nagpal S, Patlolla SH, Sharma A, Kandlakunta H, Anani V, Angom RS, Kamboj AK, Ahmed N, Mohapatra S, Vivekanandhan S, Philbrick KA, Weston A, Takahashi N, Kirkland J, Javeed N, Matveyenko A, Levy MJ, Mukhopadhyay D, Chari ST. Phases of metabolic and soft tissue changes in months preceding a diagnosis of pancreatic ductal adenocarcinoma. Gastroenterology 156: 1742–1752, 2019. doi: 10.1053/j.gastro.2019.01.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Scherbakov N, Knops M, Ebner N, Valentova M, Sandek A, Grittner U, Dahinden P, Hettwer S, Schefold JC, von Haehling S, Anker SD, Joebges M, Doehner W. Evaluation of C-terminal Agrin Fragment as a marker of muscle wasting in patients after acute stroke during early rehabilitation. J Cachexia Sarcopenia Muscle 7: 60–67, 2016. doi: 10.1002/jcsm.12068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Nishikawa H, Enomoto H, Ishii A, Iwata Y, Miyamoto Y, Ishii N, Yuri Y, Hasegawa K, Nakano C, Nishimura T, Yoh K, Aizawa N, Sakai Y, Ikeda N, Takashima T, Takata R, Iijima H, Nishiguchi S. Elevated serum myostatin level is associated with worse survival in patients with liver cirrhosis. J Cachexia Sarcopenia Muscle 8: 915–925, 2017. doi: 10.1002/jcsm.12212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Calvani R, Marini F, Cesari M, Tosato M, Anker SD, von Haehling S, Miller RR, Bernabei R, Landi F, Marzetti E; SPRINTT consortium. Biomarkers for physical frailty and sarcopenia: state of the science and future developments. J Cachexia Sarcopenia Muscle 6: 278–286, 2015. doi: 10.1002/jcsm.12051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Zheng Z, Liu X, Zhao Q, Zhang L, Li C, Xue Y. Regulation of UCP1 in the browning of epididymal adipose tissue by β3-adrenergic agonist: a role for microRNAs. Int J Endocrinol 2014: 530636, 2014. doi: 10.1155/2014/530636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Wang X, Chen S, Lv D, Li Z, Ren L, Zhu H, Xie X, Liu Y. Liraglutide suppresses obesity and promotes browning of white fat via miR-27b in vivo and in vitro. J Int Med Res 49: 3000605211055059, 2021. doi: 10.1177/03000605211055059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Lemecha M, Morino K, Imamura T, Iwasaki H, Ohashi N, Ida S, Sato D, Sekine O, Ugi S, Maegawa H. MiR-494-3p regulates mitochondrial biogenesis and thermogenesis through PGC1-α signalling in beige adipocytes. Sci Rep 8: 15096, 2018. doi: 10.1038/s41598-018-33438-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Zhang Z, Jiang H, Li X, Chen X, Huang Y. MiR-92a regulates brown adipocytes differentiation, mitochondrial oxidative respiration, and heat generation by targeting SMAD7. J Cell Biochem 121: 3825–3836, 2020. doi: 10.1002/jcb.29539. [DOI] [PubMed] [Google Scholar]
- 80. Hu F, Wang M, Xiao T, Yin B, He L, Meng W, Dong M, Liu F. miR-30 promotes thermogenesis and the development of beige fat by targeting RIP140. Diabetes 64: 2056–2068, 2015. doi: 10.2337/db14-1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Wu Y, Zuo J, Zhang Y, Xie Y, Hu F, Chen L, Liu B, Liu F. Identification of miR-106b-93 as a negative regulator of brown adipocyte differentiation. Biochem Biophys Res Commun 438: 575–580, 2013. doi: 10.1016/j.bbrc.2013.08.016. [DOI] [PubMed] [Google Scholar]
- 82. Narasimhan A, Shahda S, Kays JK, Perkins SM, Cheng L, Schloss KNH, Schloss DEI, Koniaris LG, Zimmers TA. Identification of potential serum protein biomarkers and pathways for pancreatic cancer cachexia using an aptamer-based discovery platform. Cancers (Basel) 12: 3787, 2020. doi: 10.3390/cancers12123787. [DOI] [PMC free article] [PubMed] [Google Scholar]