Abstract
This study aims to investigate health behaviors and psychological burdens in adolescent children of cancer parents. We compared health behaviors and mental health outcomes between 266 adolescent children with a parent diagnosed with cancer and 3163 control adolescents aged 12–19 years using data from the Korean National Health and Nutrition Examination Survey (KNHANES) from 2010 to 2018. Alcohol use of adolescents increased between 2 and 5 years after parental cancer diagnosis (adjusted odds ratio [aOR], 1.72; 95% confidence interval [CI], 1.01–2.94) but decreased after 5 years. Parental cancer was associated with increased vaccination uptake in adolescents within 1 year of diagnosis (aOR, 3.19; 95% CI, 1.55–6.54), but after 2 years, there was no difference from rates in their peers. Maternal cancer was associated with increased depression among adolescents (aOR, 1.73; 95% CI, 1.10 − 2.73). Although the risks of suicidal thoughts/plans/attempts increased within 1 year after parental cancer diagnosis (aOR, 2.96; 95% CI, 1.00 − 8.83), it reduced 2 years after diagnosis, leading to no significant difference from the frequency in peers. Within five years after the parent was diagnosed with cancer, support for their adolescent children's health behaviors and mental health is necessary in the community.
Subject terms: Cancer, Health care, Oncology, Risk factors, Human behaviour
Introduction
Cancer is a leading cause of death worldwide, with nearly 10.0 million deaths in 20201. Cancer development is a complex multi-stage process involving molecular and cellular changes over many years with various risk factors. Accordingly, the incidence of most cancers increases with age, usually more dramatically after middle age2. However, a significant proportion of the child-bearing-aged population is still affected. According to the statistical data of the Korean Central Cancer Registry, 10% of male cancer patients and 26% of female cancer patients were in the child-bearing age group between the ages of 30–49 in 20183.
Parental cancer can affect all family members physically, emotionally, socially, and culturally. The patient’s regular routine is disrupted by treatment, potential job loss, and financial problems. Unplanned admission, frequent clinic visits, and decreased function in cancer parents make it difficult for them to care for their children so they can participate in school activities. Higher levels of anxiety and depression in cancer patients are also associated with an increased risk of emotional and behavioral problems and lower quality of life in their children4–6. Among children of all ages, adolescents are particularly at risk of psychological problems7–9.
Adolescent children show the first signs of puberty in their early teens, approximately between 9 and 13 years10,11. They experience a period of dramatic change with rapid physical growth, onset of sexual maturation, and increased self-consciousness and social challenges. Although adolescence is one of the physically healthiest periods in one’s lifetime, there are a notably high proportion of deaths from preventable causes, such as interpersonal violence, suicide, and accidents such as drowning or road traffic accidents12. In addition, lifestyle risk factors that dominate life are formed during this period and affect mortality rates later in life. These adolescent health problems are disproportionately influenced by family stability and the social environment13,14.
Although the health of children with parental cancer has been studied steadily in Western countries, there is still scarce evidence in Asian cultures. In the present study, we used data from the Korean National Health and Nutrition Survey (KNHANES) to assess the association between factors of parental cancer and health risks of adolescent children after a parental cancer diagnosis.
Materials and methods
Study population
The Korea National Health and Nutrition Examination Survey (KNHANES) is a nationwide survey of the health and nutritional status of community-dwelling Koreans. A stratified multi-stage clustered probability design was used to select a representative sample of civilian, non-institutionalized Koreans. The fifth (2010–2012), sixth (2013–2015), and seventh (2016–2018) KNHANES included comprehensive information on the health status, health behavior, and sociodemographic characteristics of the population in 576 national districts. The health interview survey was conducted for all study participants, but the health examination survey and nutritional survey were conducted by randomly selecting about a third of the participants in the health interview survey. The health interview survey in KNHANES was conducted through face-to-face interviews at subjects’ homes by trained interviewers.
When parents and children from one family participated in the survey, the ID of the father and/or mother was coded, and their data were merged for further analysis. The initial participants included 5686 adolescents aged between 12 and 19 years who had the ID code of their parents during the fifth, sixth, and seventh KNHANES surveys, and completed the health interview and health examination surveys. Among them, 2257 participants whose parents did not have a history of cancer were excluded, yielding 3429 adolescents as the final study population (Fig. 1).
Figure 1.
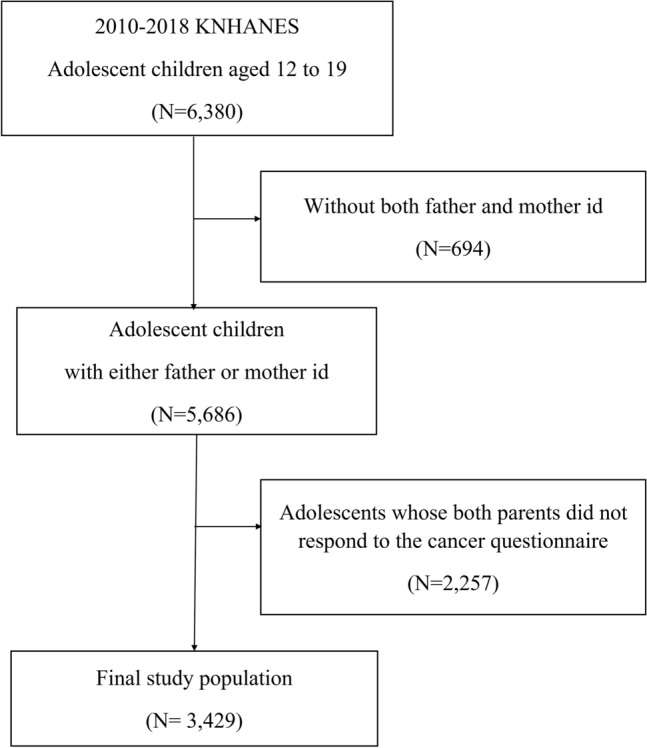
Study population.
Associated factors
Adolescents’ variables
From 2010 to 2018, adolescent participants completed the Health Interview Survey, including age, sex, residential area (rural or urban), and education level. Household income was divided into three levels (< 2000, 2000–5000, or > 5000 thousand won).
Height and weight measurements of adolescent participants were obtained using a standard protocol with an automatic and portable height and weight measuring device. Their weight-for-age was divided into three groups (< 5th, 5th − < 95th, and ≥ 95th percentile) in reference to the 2017 Korean National Growth Chart15.
Adolescents' smoking status was assessed by a question about lifetime ever-smoking experience aged from 12 to 18 years (yes or no) while another question about lifetime smoking measures their lifetime smoking classified into (1) less than 100 cigarettes (2) 100 cigarettes or more (3) never for adolescents aged 19 years or older. For the purpose of analysis, we dichotomously divided them into ever-smoker and nonsmoker. Lifetime alcohol consumption was assessed “Have you ever drank more than one drink in your life?” and the option was yes or no.
The physical activity in the past week was assessed among adolescents using IPAQ (International Physical Activity Questionnaire) between 2010 and 2013, GPAQ (Global physical activity questionnaire) between 2014 and 2016, and hours of moderate or more physical activity between 2017 and 2018. GPAQ reflects leisure-time, worktime, and transport-related PA, allowing accurate evaluation of PA for each area, and the rest of the questionnaires do not distinguish them. Although there was difficulty in simple comparison, we set as a dichotomous variable whether moderate-intensity PA was performed for 150 min or more per week, vigorous-intensity PA more than 75 min, or the equivalent dose of moderate and vigorous physical activity according to the American College of Sports Medicine (ACSM) exercise guideline.
Influenza vaccination during the past 1 year was assessed using a self-reported questionnaire, “Have you been vaccinated against influenza (seasonal flu) in the past year?” And participants who answered “yes” to this question were defined as having received an influenza vaccination. Currently, influenza and Human Papilloma Virus vaccines are recommended in a Korean national vaccination program for adolescents.
Self-rated health consists of the response to the single item, "Would you say your health, in general, is very good, good, fair, poor, or very poor?" We dichotomously categorized “very bad” or “bad” into the “bad participant health” group and “very good,” “good,” or “average” into the “good participant health” group.
Adolescent participants were asked about their stress, “How much stress do you feel in your daily life?” and they rated their stress level as “very high,” “high,” “low,” or “very low.” We dichotomously categorized “very high” or “high” into the “high stress” group and “very low” or “low,” into the “low stress” group16.
Depression was screened through a single question, “Over the last 1 year, have you ever had two weeks or more where nearly every day you felt sad or hopeless?” which could be answered yes or no. Adolescents’ suicidal thoughts, plans, and attempts were assessed by asking about the occurrence of those experiences in the last 1 year. An adolescent could answer yes or no to such questions about suicide as “Have you seriously considered suicide in the past year?” “Have you made any concrete plans to commit suicide in the past year?” and “Have you actually attempted suicide in the past year?”.
Parental cancer variables
The type of cancer was identified from the medical history section of the questionnaire from the parental survey. From 2010 to 2018, the seven most common types of cancer in Korea were identified through cancer questionnaires (breast, colon, stomach, liver, uterine cervix, lung, and thyroid cancer) and cancer from other sites, including prostate, pancreas, bladder, esophagus, and ovary, among others, was categorized as “other cancer types.” Parental cancer was classified as maternal or paternal. The time after parental cancer diagnosis was calculated by subtracting the age in years at which the participant reportedly was when a parent first had cancer from the age in years at the time of the questionnaire.
Data analysis
Descriptive statistical methods were used to describe the basic characteristics of the study population, and the results from the chi-squared test for categorical variables and the unpaired t − test for continuous variables are reported. We conducted univariate logistic regression analysis to measure the association between parental cancer and adolescents’ development, tobacco and alcohol use, physical activity, vaccination, subjective health status, stress, depressive mood, and suicidal thought/plan/attempt. To assess the adjusted odds ratios for aforementioned adolescent health behaviors and psychological factors, we used the following covariates in the multivariate logistic regression analysis: sex, age, and household income. A p-value of less than 0.05 was considered significant, and 95% confidence intervals are shown. No weighting was applied. All statistical tests were performed using STATA version 14.0 (Stata Corp., College Station, TX, USA).
Ethical Approval
This survey complies with the principles of the Helsinki Declaration. The methods were carried out following the relevant guidelines and regulations. All participants agreed to fill out an informed consent form. The KNHANES database is anonymized with the use of its data guided by strict confidentiality guidelines17. This present study was approved by the ethics boards of the Seoul National University Hospital (IRB number E-2112–007-1284).
Results
Baseline characteristics
Baseline characteristics of the study population are presented in Table 1. The mean age of adolescents with parental cancer was 15.6 ± 2.3 years, which was older than those without parental cancer (15.3 ± 2.3 years). The proportion of elementary school students was higher among adolescents with parental cancer than among adolescents without parental cancer. The difference in sex distribution was not significant. The weight for age for both groups and proportion of smokers and drinkers were not significantly different. Household income and residential area of adolescents also did not differ between the two groups. The proportion of having single parents was 15.8% among adolescents without cancer patients and 11.0% among adolescents with cancer patients.
Table 1.
Baseline characteristics of adolescent aged from 12 to 19 (n = 3429).
| Characteristic | Adolescents without parental cancer | Adolescents with parental cancer | df | Pearson chi-square value | Cramer's V or phi value | pa |
|---|---|---|---|---|---|---|
| 3163 (92.2) | 266 (7.8) | |||||
| Age (years) | 15.3 ± 2.3 | 15.6 ± 2.3 | 0.039 | |||
| Sex | ||||||
| Male | 1627 (51.4) | 134 (50.4) | 1 | 0.111 | 0.006 | 0.739 |
| Female | 1536 (48.6) | 132 (49.6) | ||||
| Weight for age | ||||||
| < 5 percentile | 154 (6.5) | 12 (5.7) | 2 | 0.329 | 0.011 | 0.848 |
| 5- < 95 percentile | 1,858 (78.8) | 168 (80.4) | ||||
| ≥ 95 | 346 (14.7) | 29 (13.9) | ||||
| Smoking | ||||||
| Nonsmoker | 2,306 (86.3) | 194 (87.0) | 1 | 0.076 | −0.005 | 0.782 |
| Ever-smoker | 365 (13.7) | 29 (13.0) | ||||
| Alcohol intake | ||||||
| Nondrinker | 2084 (71.8) | 168 (67.5) | 1 | 2.096 | 0.026 | 0.148 |
| Drinker | 819 (28.2) | 81 (32.5) | ||||
| Education level | ||||||
| ≤ elementary school | 688 (17.9) | 66 (26.7) | 2 | 3.162 | 0.032 | 0.003 |
| Middle school | 2,182 (56.8) | 126 (51.0) | ||||
| ≥ High school | 970 (25.3) | 55 (22.3) | ||||
| Household monthly income | ||||||
| < 2000 | 368 (11.7) | 30 (11.3) | 2 | 0.186 | 0.007 | 0.911 |
| 2,000–5000 | 1596 (50.5) | 138 (51.9) | ||||
| > 5000 | 1196 (37.9) | 98 (36.8) | ||||
| Having single parent | ||||||
| No | 2383 (84.2) | 211 (89.0) | 1 | 0.447 | −0.036 | 0.047 |
| Yes | 448 (15.8) | 26 (11.0) | ||||
| Parental cancer | ||||||
| Maternal cancer | 201 (75.6) | |||||
| Paternal cancer | 65 (24.4) | |||||
| Number of parental cancers | ||||||
| 1 | 235 (94.8) | |||||
| 2 | 13 (5.2) | |||||
| Cancer type of parents | ||||||
| Stomach cancer | 23 (8.6) | |||||
| Liver cancer | 4 (1.5) | |||||
| Colorectal cancer | 13 (4.9) | |||||
| Breast cancer | 57 (21.4) | |||||
| Cervical cancer | 29 (10.9) | |||||
| Thyroid cancer | 97 (36.5) | |||||
| Other cancer | 48 (18.0) | |||||
| Time since parental cancer diagnosis | ||||||
| ≤ 1 year | 20 (8.1) | |||||
| 2– < 5 years | 95 (38.3) | |||||
| ≥ 5 years | 133 (53.6) | |||||
Data are presented number and percentage (%) or mean ± S.D.
ap value unpaired t-test for continuous variables or from chi-square test for categorical variables.
Among adolescents with cancer parents, 75.6% had maternal cancer, while 24.4% had paternal cancer. None of the patients had both paternal and maternal cancer. Most cases of parental cancer only involved a single cancer; 5.2% of parents with cancer had a second primary cancer. Among cancer survivors, thyroid cancer was the most common cancer (36.5%), followed by breast (21.4%), uterine cervix (10.9%), stomach (8.6%), colorectal (4.9%), liver (1.5%), and other cancers (18.0%). Among the cancer survivors, ≥ 5 years had elapsed since diagnosis in 53.6% of subjects, 2 − < 5 years since diagnosis in 38.3%, and ≤ 1 year since diagnosis in 8.1%.
Adolescent health behaviors
Table 2 shows the health behaviors of adolescents with and without parents with cancer. The proportion of adolescents with abnormal weight for their age was similar between adolescents with and without a parent with cancer. The proportion of current smokers and adolescents who performed physical activity over the recommended levels was similar in adolescents with and without parents with cancer. In an unadjusted model, the proportion of any alcohol use among adolescents whose mother was diagnosed with cancer was higher (odds ratio 1.30, 95% confidence interval [CI], 0.96–1.78); however, the significance was weakened after multivariate adjustment. Interestingly, the adolescents who had any parent with cancer and who had paternal cancer were more likely to have an influenza vaccination than adolescents without parental cancer. However, adolescents with maternal cancer did not show a difference in influenza vaccination rates from that in adolescents without a parent with cancer.
Table 2.
Behavioral risk factors and psychological factors among adolescent with or without parental cancer.
| Characteristic | Adolescents without parental cancer | Adolescents with parental cancer | Adolescents with paternal cancer | Adolescents with maternal cancer |
|---|---|---|---|---|
| Abnormal weight for agea | ||||
| Proportion (%) | 20.9 | 19.5 | 16.3 | 20.7 |
| OR | 1.00 | 0.91 (0.64 1.29) | 0.72 (0.34 1.48) | 1.00 (0.67 1.49) |
| aORb | 1.00 | 0.83 (0.57 1.20) | 0.69 (0.33 1.46) | 0.89 (0.59 1.37) |
| Ever-smoker, yes | ||||
| Proportion (%) | 13.7 | 11.6 | 10.9 | 13.7 |
| OR | 1.00 | 0.94 (0.63 1.42) | 0.87 (0.37 2.06) | 1.00 (0.64 1.58) |
| aORb | 1.00 | 0.87 (0.56 1.37) | 0.87 (0.34 2.22) | 0.90 (0.55 1.47) |
| Any alcohol use, yes | ||||
| Proportion (%) | 28.2 | 32.5 | 28.3 | 33.9 |
| OR | 1.00 | 1.23 (0.93 1.62) | 1.07 (0.60 1.89) | 1.30 (0.96 1.78) |
| aORb | 1.00 | 1.11 (0.78 1.57) | 1.08 (0.53 2.23) | 1.12 (0.76 1.66) |
| Moderate/vigorous intensity physical activity | ||||
| Proportion (%) | 67.3 | 65.3 | 66.7 | 64.9 |
| OR | 1.00 | 0.91 (0.66 1.25) | 0.95 (0.49 1.82) | 0.89 (0.63 1.27) |
| aORb | 1.00 | 0.95 (0.69 1.30) | 0.99 (0.51 1.92) | 0.93 (0.65 1.33) |
| Influenza vaccination, yes | ||||
| Proportion (%) | 26.3 | 32.4 | 40.7 | 29.8 |
| OR | 1.00 | 1.34 (1.00 1.80)* | 1.86 (1.07 3.24)* | 1.16 (0.83 1.64) |
| aORb | 1.00 | 1.45 (1.07 1.97)* | 1.96 (1.11 3.47)* | 1.28 (0.90 1.83) |
| Subjective health status (very bad/bad) | ||||
| Proportion | 40.0 | 35.3 | 28.3 | 37.6 |
| OR | 1.00 | 0.81 (0.62–1.07) | 0.61 (0.35–1.09) | 0.91 (0.67–1.23) |
| aORb | 1.00 | 0.78 (0.59–1.03) | 0.60 (0.34–1.07)† | 0.86 (0.63–1.18) |
| Very high/high level stress | ||||
| Proportion | 24.9 | 27.3 | 21.7 | 29.1 |
| OR | 1.00 | 1.13 (0.85 1.52) | 0.84 (0.45–1.57) | 1.27 (0.92 1.76) |
| aOR b | 1.00 | 1.11 (0.83 1.49) | 0.83 (0.44–1.55) | 1.24 (0.90 1.72) |
| Depressive mood more than two weeksb | ||||
| Proportion | 8.0 | 10.5 | 1.75 | 13.3 |
| OR | 1.00 | 1.35 (0.87 2.09) | 0.20 (0.03–1.47) | 1.81 (1.15 2.84)* |
| aORb | 1.00 | 1.29 (0.83 2.01) | 0.19 (0.03–1.42) | 1.73 (1.10 2.73)* |
| Suicidal thought/plan/attempt in the past year | ||||
| Proportion | 4.1 | 5.0 | 1.75 | 6.1 |
| OR | 1.00 | 1.25 (0.68 2.30) | 0.47 (0.06 3.43) | 1.62 (0.86 3.08) |
| aORb | 1.00 | 1.20 (0.65 2.23) | 0.44 (0.06 3.25) | 1.58 (0.83 3.01) |
Data are presented as percentages (%) and odds ratio (95% CI).
OR odds ratio; CI confidence interval.
aAbnormal weight for age was defined as ≥ 95percentile or < 5percentile of weight for age of Korean adolescent population.
bAdjusted for age, sex, and household monthly income.
cRecommended physical activity (150 min/week of moderate-intensity physical activity, or 75 min/week of vigorous-intensity physical activity, or the equivalent dose of moderate and vigorous physical activity).
*P < 0.05.
Table 3 shows the health behavior factors according to time since parental cancer diagnosis compared to that in the control group with no parents with cancer. The crude proportion showed that health risk factors, such as abnormal weight for age, smoking, and alcohol use, were highest at 2–5 years after parental cancer diagnosis (23.8%, 18.4%, and 36.2%, respectively). Desirable health behavior such as influenza vaccination were as high 48.5%, immediately after a parental cancer diagnosis at ≤ 1 year and decreased with an increasing number of years after cancer diagnosis to levels similar to those in the control group.
Table 3.
Behavioral risk factors and psychological factors among adolescent according to time since parental cancer diagnosis.
| Characteristic | Adolescents without parental cancer | Adolescents with parental cancer | P trend | ||
|---|---|---|---|---|---|
| Time since parental cancer diagnosis | |||||
| ≤ 1 year | 2– < 5 years | ≥ 5 years | |||
| Abnormal weight for agea | |||||
| Proportion (%) | 20.9 | 21.4 | 23.8 | 16.4 | |
| OR | 1.00 | 0.99 (0.40 2.47) | 1.02 (0.62 1.68) | 0.72 (0.40 1.29) | 0.416 |
| aORb | 1.00 | 0.91 (0.35 2.32) | 0.98 (0.58 1.64) | 0.63 (0.34 1.15) | 0.306 |
| Ever-smoker, yes | |||||
| Proportion (%) | 13.7 | 12.9 | 18.4 | 7.5 | |
| OR | 1.00 | 1.04 (0.36 2.99) | 1.58 (0.93 2.68)* | 0.56 (0.28 1.23) | 0.137 |
| aORb | 1.00 | 0.95 (0.31 2.90) | 1.46 (0.80 2.66) | 0.56 (0.25 1.26) | 0.209 |
| Any alcohol use, yes | |||||
| Proportion (%) | 28.2 | 33.3 | 36.2 | 28.7 | |
| OR | 1.00 | 1.34 (0.67 2.70) | 1.52 (1.01 2.29)* | 1.07 (0.70 1.66) | 0.397 |
| aORb | 1.00 | 0.98 (0.41 2.34) | 1.72 (1.01 2.94)* | 0.88 (0.52 1.49) | 0.294 |
| Physical activity above recommendationc | |||||
| Proportion (%) | 67.7 | 60.7 | 65.9 | 66.3 | |
| OR | 1.00 | 0.74 (0.34 1.59) | 0.92 (0.58 1.48) | 0.94 (0.58 1.51) | 0.657 |
| aORb | 1.00 | 0.87 (0.39 1.91) | 0.94 (0.58 1.52) | 0.98 (0.60 1.60) | 0.697 |
| Influenza vaccination, yes | |||||
| Proportion (%) | 26.3 | 48.5 | 31.2 | 28.1 | |
| OR | 1.00 | 2.55 (1.28 5.10)* | 1.22 (0.78 1.93) | 1.06 (0.67 1.68) | 0.056 |
| aORb | 1.00 | 3.19 (1.55 6.54)* | 1.33 (0.83 2.11) | 1.15 (0.72 1.84) | 0.046 |
| Subjective health status (very bad/bad) | |||||
| Proportion | 40.0 | 31.4 | 34.3 | 37.7 | |
| OR | 1.00 | 0.72 (0.35–1.47) | 0.82 (0.54–1.47) | 0.94 (0.35–1.47) | 0.461 |
| aORb | 1.00 | 0.65 (0.31–1.35) | 0.80 (0.53–1.22) | 0.90 (0.59–1.35) | 0.500 |
| Very high/high level stress | |||||
| Proportion | 24.9 | 22.2 | 27.6 | 28.7 | |
| OR | 1.00 | 0.89 (0.40–1.95) | 1.18 (0.76 1.83) | 1.25 (0.81 1.92) | 0.502 |
| aORb | 1.00 | 0.87 (0.39–1.93) | 1.20 (0.77 1.86) | 1.23 (0.80 1.89) | 0.579 |
| Depressive mood more than two weeksb | |||||
| Proportion | 8.0 | 5.9 | 11.0 | 11.5 | |
| OR | 1.00 | 0.72 (0.17–3.04) | 1.43 (0.74–2.73) | 1.50 (0.80 2.81) | 0.428 |
| aORb | 1.00 | 0.65 (0.15–2.77) | 1.42 (0.74–2.72) | 1.43 (0.76 2.69) | 0.331 |
| Suicidal thought/plan/attempt in the past year | |||||
| Proportion | 4.1 | 11.8 | 4.0 | 3.9 | |
| OR | 1.00 | 3.43 (1.18 10.01)* | 1.07 (0.38 2.99) | 1.03 (0.37 2.88) | 0.138 |
| aORb | 1.00 | 2.96 (1.00 8.83)* | 1.04 (0.37 2.91) | 0.95 (0.34 2.66) | 0.153 |
Data are presented as percentages (%) and odds ratio (95% CI).
OR odds ratio; CI confidence interval.
aAbnormal weight for age was defined as ≥ 95 percentile or < 5 percentile of weight for age of Korean adolescent population.
bAdjusted for age, sex, and household monthly income.
cRecommended physical activity (150 min/week of moderate-intensity physical activity, or 75 min/week of vigorous-intensity physical activity, or the equivalent dose of moderate and vigorous physical activity).
*P < 0.05.
In the multivariate logistic model, the proportion of adolescents with abnormal weight for age differed between groups according to time. The smoking rate of adolescents was significantly higher 2 − 5 years after a parental cancer diagnosis with an odds ratio of 1.58 (95% CI, 0.93–2.68), but this association was weakened after multivariate adjustment. Alcohol use was significantly increased 2–5 years after a parental cancer diagnosis with an adjusted odds ratio (aOR) of 1.79 (95% CI, 1.05–3.03). The influenza vaccination rate increased considerably at ≤ 1 year with an aOR of 3.17 (95% CI, 1.54–6.50); however, it decreased 2 or more years after cancer diagnosis to a level that was no different from that in the control group.
Adolescent mental health
The subjective health status and stress levels did not differ between adolescents with and without parental cancer (Table 2). The multivariate model showed marginal significance that adolescents with paternal cancer were not likely to feel very bad or bad about their own health. Depressive symptoms in adolescents were particularly high when their mothers were diagnosed with cancer (aOR, 1.74; 95% CI, 1.50–2.75). Although suicidal thoughts were more common with a crude proportion of 6.1% vs. 4.1% when mothers had cancer, these associations were non-significant after multivariate adjustment.
Depressive mood was more common 2 years after diagnosis, but this association was also weakened in the multivariate model (Table 3). Approximately 11.8% of adolescents whose parents had cancer had suicidal thoughts/plans/attempts within 1 year after a parental cancer diagnosis. Although the risks of suicidal thoughts/plans/attempts increased within 1 year after parental cancer diagnosis (aOR, 2.96; 95% CI, 1.00 − 8.83), it reduced 2 years after diagnosis, leading to no significant difference from the frequency in peers.
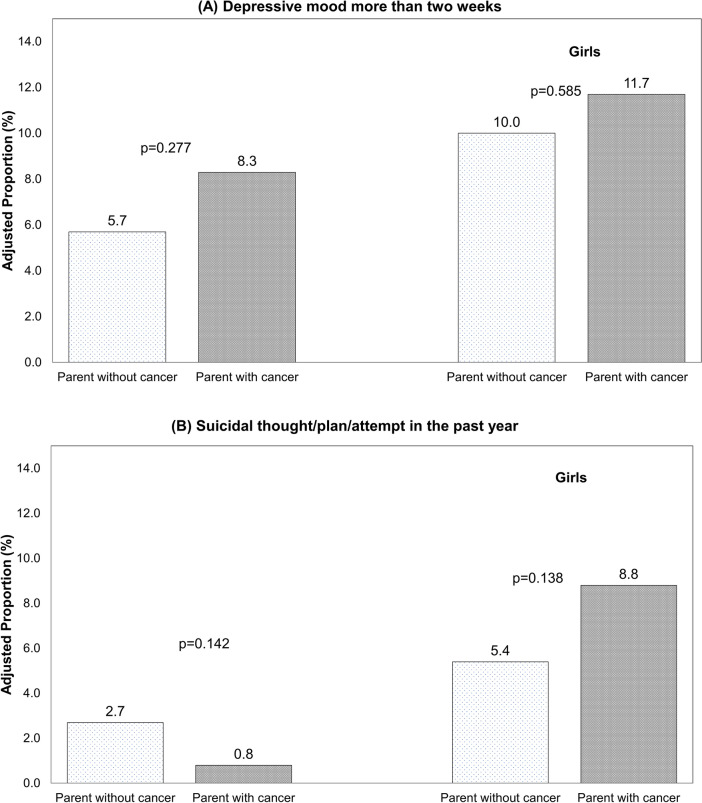
Depression and suicidal ideation were more prevalent in girls than boys, with or without cancer parents (Fig. 2). In general, the raw proportions increased in boys and girls with cancer parents compared to boys and girls without cancer parents, but there was no statistically significant difference. When the multivariate analysis was conducted, the adjusted proportion of depression was 8.3% for boys with cancer parents and 5.7% for boys without cancer parents, and there was no statistically significant difference (Fig. 2). In addition, girls with cancer parents had a proportion of depression of 11.7%, and girls without cancer patients had a 10.0%, but there was also no significant statistical difference. There was no statistical difference in suicidal thought/plan/attempt between 0.8% of boys with cancer patients' parents and 2.7% of boys without cancer patients. In addition, 8.8% of girls with cancer patients' parents and 5.4% of girls without cancer patients had suicidal thought/plan/attempt in the past year, but there was no statistical difference in the multivariate model (Fig. 2).
Figure 2.
Comparison of depression and suicidal thought among adolescent boys and girls with and without cancer parents. (A) Depressive mood more than two weeks. (B) Suicidal thought/plan/attempt in the past year.
Discussion
Our study shows that, although adolescent children have high behavioral changes and psychological stress in the early stages of their parents' cancer diagnosis, they adapt over time and there is no difference from the general peer group after 5 years in the community setting. Their negative behavior such as alcohol use increased and positive behavior such as vaccination increased for a period of time after a parental cancer diagnosis. There was no significant difference in their subjective health status or stress, but adolescents were more likely to be depressed when their mother was diagnosed with cancer and have suicidal thoughts/plans/attempts immediately after a parent was diagnosed with cancer. However, this vulnerability decreased five years after diagnosis.
Our data showed similar prevalence of adolescent psychiatric problems compared to previous Western studies. Adolescent with parental cancer showed depressive mood in 10.5%, significant stress in 27.3%, and suicidality in 5%. Filial piety, ‘hyo’, is still regarded as an adult children’s duty to care for their old and sick parents in Korea and many Asian countries. Caregiving is strongly aligned with Confucian concept of family and family participation18,19. Although studies in other Asian county shows filial piety may lessen caregiver burden20,21, it does not mean that the family caregivers do not experience burden or stress22,23 In norms of filial piety, there also has been a gender difference for the roles of sons and daughters, and daughters are more likely to serve as caregivers for their parents21. As Korean society modernizes, the idea of filial piety is gradually disappearing24, and in this study, Korean adolescents with cancer parents show a similar magnitude of risks by gender to those of previous Western studies. ADDIN EN.CITE25,26.
Our result corresponds to previous studies that girls reported more psychological symptoms than boys when their parents had a cancer diagnosis27,28. In the present study, 8.3% of boys and 11.7% of girls had depressive symptoms. In a Finnish birth cohort study, among 13–17-year-old adolescents, 7.2% of boys and 15.7% of girls with cancer mothers received outpatient psychiatric care27. Previous studies suggested that girls are more likely to suffer from distress due to the genetic nature of breast cancer, and girls are likely to have internalizing problems while boys have externalizing problems6,29.Stressful life events, such as death, divorce or separation, or unemployment of parents, are known to increase drinking and smoking rates in adolescents30,31. Previous studies have suggested that adolescents with family history of cancer have similar health behaviors, such as smoking, alcohol consumption, and nutrition, compared to those without family history of cancer, despite predisposed genetic risks32,33. In the present study, the adolescents with parents with cancer did not show differences in their physical development. or health behaviors, compared to those without parental cancer, which is in accordance with previous studies. Their smoking and drinking rates tend to increase at some point after their parents are diagnosed with cancer. Their parents, cancer survivors are recommended to reduce alcohol and smoking cessation to increase their survival, reduce cancer recurrence, and decrease second primary cancer34,35. A family-level health behavior intervention program can be considered after a parental cancer diagnosis.
Our study also shows that parental cancer diagnosis can positively influence adolescent children's health behaviors. Previously, there was no evidence of the effect of parental cancer on children's vaccination uptake. However, parental attitudes and social factors are well known to be associated with childhood vaccination36,37. Our study showed that the positive influence on vaccination uptake was especially high in adolescents with paternal cancer. It is most likely that not only did the adolescents change vaccination behavior, but their parents also increased their knowledge and had a positive attitude toward vaccination in families caring for fathers with cancer who often had chemotherapy and immunocompromised. Unfortunately, it lasted less than 1 year after the parent’s cancer diagnosis and did not last if the parent survived for more than 5 years. Strategies are needed to ensure that these healthy behaviors are maintained38.
Our study corresponds to previous studies that a mother’s illness may have a more substantial influence on adolescents’ emotional well-being than a father’s illness because mothers are more often involved in caring for their children than father’s due to cultural norms39. In a previous interview with breast cancer patients revealed that they struggled to be responsible for raising their children while receiving treatment and could not focus on being sick40. Moreover, surviving mothers often feel guilty about their children’s abandonment during treatment40. This influence is often bidirectional and mutual, and other family members can interact. One study found that children's anxiety and depression increased when their fathers were unable to adjust to bereavement and when families had closed communication about their mother's cancer41. The burden on families could be reduced by emphasizing social support for enhancing expression and empathy between sick parents and children.
A previous study reported that the children of cancer patients experienced post-traumatic stress syndrome in the first 4 months of the parental cancer diagnosis29. Our study also showed that although they showed a notable increase in suicidal thoughts, plans, or attempts in the first year, they did not show increased suicidality after the second year of a parental cancer diagnosis. Their risky health behaviors increased between 2 and 5 years but decreased from 5 years after parental cancer diagnosis. Although there may be individual differences according to their temperament, stage or unpredictability of parental cancer, or family functioning, the majority of adolescents experience a restoration in psychological and physical function over time42–44. Social support may enable them to overcome these crises more easily; adapt to significant physical, cognitive, social, and emotional changes in adolescence45–47; and be a catalyst for their posttraumatic growth after negative life events48,49.
A major strength of our study was the utilization of nationwide data sources, which enabled us to compare results across the general adolescent population. It is possible to better reflect the reality of families with members with cancer through a community-based design rather than a hospital-centered study. Other strengths include the availability of detailed information on the physical and psychosocial conditions of adolescents.
The significance of this study may serve as reference data for future interventions. Screening for adolescent mental health immediately after the parent's cancer diagnosis is the top priority, and it is also necessary to evaluate their health behaviors. Regular evaluation is required for adolescent use of alcohol and smoking within five years of a parental cancer diagnosis. At the same time, it will be possible to make the crisis act as a positive factor by encouraging physical activity and vaccination of adolescent children. Future research is needed to investigate family-oriented interventions for cancer survivorship to reduce cancer parents' mortality and the long-term risk for their adolescent children.
Some limitations of this study should be considered. First, because this was a cross-sectional study, the causes of differences between cancer survivors and the general population could not be established. Second, several pieces of information were collected from self-reported questionnaires, so reporting bias cannot be excluded. In particular, since the mental health status of adolescents was evaluated through a single question, it was not possible to classify and evaluate the correct psychiatric diagnoses such as major depressive disorder, bipolar disorder, anxiety disorder, affective disorder, or etc. Third, parental cancer stage, previous or ongoing treatment, prognostic factors, and other comorbid chronic diseases were not considered. Fourth, predisposed adolescent history, such as psychiatric diseases other chronic illnesses, school absence, and academic achievement could not be acquired. Fifth, this study mainly investigated cancer survivors, and we cannot generalize our results to all adolescents with a parent with cancer. In addition, we only considered adolescent children of long-term survivors. However, considering that the overall cancer survival rate has improved up to 70% with early detection and treatment, we still need to address the growing problem of cancer survivorship.
Conclusion
Our study suggests that most adolescent children of cancer survivors did not differ in their smoking, alcohol use, physical activity, and risk of being underweight or overweight compared to general peers. However, alcohol use and vaccination rates somewhat increased immediately after parental diagnosis. These behavioral changes occurred mainly within 5 years of diagnosis, and after 5 years of parental survival, behaviors were the same as those in the general population. There was no significant difference in their subjective health status or stress, but adolescents were likely to become depressed when the mother was diagnosed and have suicidal thoughts/plans/attempts immediately after a parent was diagnosed with cancer. However, this vulnerability decreased from five years after diagnosis. Overall, our study showed that most adolescent children of cancer survivors are resilient to distress associated with parental cancer diagnosis over time. Tailored community-based social support will be required for adolescents to make positive psychological adjustments and grow into adults.
Abbreviations
- KNHANES
Korean National Health and Nutrition Survey
- ACSM
American College of Sports Medicine
Author contributions
K. H. K. designed and performed the analyses and drafted the manuscript. S. M. P. and M. S. K. supported the conceptualization of the study and the definition of the concrete analyses' contents. The process was accompanied by S. C. and S. M. K. provided technic and scientific support for the analyses. The final manuscript has been critically revised and approved by all authors.
Data availability
The data that support the findings of this study are openly available in the official website of KNHANES at https://knhanes.kdca.go.kr/knhanes/sub03/sub03_02_05.do.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Sung H, et al. Global cancer statistics 2020 GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.White MC, et al. Age and cancer risk: A potentially modifiable relationship. Am. J. Prev. Med. 2014;46:S7–S15. doi: 10.1016/j.amepre.2013.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Korea Central Cancer Registry, National. Cancer. Center, Annual report of cancer statistics in Korea in 2018, 60–61 (2020).
- 4.Ainuddin HA, Loh SY, Low WY, Sapihis M, Roslani AC. Quality of life of multiethnic adolescents living with a parent with cancer. Asian Pac. J. Cancer Prev. 2012;13:6289–6294. doi: 10.7314/apjcp.2012.13.12.6289. [DOI] [PubMed] [Google Scholar]
- 5.Gotze H, Ernst J, Brahler E, Romer G, von Klitzing K. Predictors of quality of life of cancer patients, their children, and partners. Psychooncology. 2015;24:787–795. doi: 10.1002/pon.3725. [DOI] [PubMed] [Google Scholar]
- 6.Huizinga GA, et al. Family-oriented multilevel study on the psychological functioning of adolescent children having a mother with cancer. Psychooncology. 2011;20:730–737. doi: 10.1002/pon.1779. [DOI] [PubMed] [Google Scholar]
- 7.Huizinga GA, et al. Stress response symptoms in adolescent and young adult children of parents diagnosed with cancer. Eur. J. Cancer. 2005;41:288–295. doi: 10.1016/j.ejca.2004.10.005. [DOI] [PubMed] [Google Scholar]
- 8.Welch AS, Wadsworth ME, Compas BE. Adjustment of children and adolescents to parental cancer parents' and children's perspectives. Cancer. 1996;77:1409–1418. doi: 10.1002/(SICI)1097-0142(19960401)77:7<1409::AID-CNCR28>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 9.Visser A, et al. Emotional and behavioural functioning of children of a parent diagnosed with cancer: A cross-informant perspective. Psycho-Oncol. J. Psychol. Soc. Behav. Dimens. Cancer. 2005;14:746–758. doi: 10.1002/pon.902. [DOI] [PubMed] [Google Scholar]
- 10.Park MJ, Lee I-S, Shin E-K, Joung H, Cho S-I. The timing of sexual maturation and secular trends of menarchial age in Korean adolescents. Korean J. Pediatr. 2006;49:610–616. doi: 10.3345/kjp.2006.49.6.610. [DOI] [Google Scholar]
- 11.Lee PA, Guo SS, Kulin HE. Age of puberty: Data from the United States of America. APMIS. 2001;109:81–88. doi: 10.1034/j.1600-0463.2001.d01-107.x. [DOI] [PubMed] [Google Scholar]
- 12.Strong KL, et al. Patterns and trends in causes of child and adolescent mortality 2000–2016: Setting the scene for child health redesign. BMJ Glob. Health. 2021;6:e004760. doi: 10.1136/bmjgh-2020-004760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fowler PJ, Henry DB, Marcal KE. Family and housing instability: Longitudinal impact on adolescent emotional and behavioral well-being. Soc. Sci. Res. 2015;53:364–374. doi: 10.1016/j.ssresearch.2015.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cunningham RM, Walton MA, Carter PM. The major causes of death in children and adolescents in the United States. N. Engl. J. Med. 2018;379:2468–2475. doi: 10.1056/NEJMsr1804754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim JH, et al. The 2017 Korean national growth charts for children and adolescents: Development, improvement, and prospects. Korean J. Pediatr. 2018;61:135–149. doi: 10.3345/kjp.2018.61.5.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kwak Y, Kim Y. Quality of life and subjective health status according to handgrip strength in the elderly: A cross-sectional study. Aging Ment. Health. 2019;23:107–112. doi: 10.1080/13607863.2017.1387766. [DOI] [PubMed] [Google Scholar]
- 17.Korea Disease Control and Preventing Agency. Guidebook for the use of raw data from Korea national health and nutrition examination survey, (2021).
- 18.Park M. Filial piety and parental responsibility: An interpretive phenomenological study of family caregiving for a person with mental illness among Korean immigrants. BMC Nurs. 2012;11:28. doi: 10.1186/1472-6955-11-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jang SN, Avendano M, Kawachi I. Informal caregiving patterns in Korea and European countries: A cross-national comparison. Asian Nurs. Res. 2012;6:19–26. doi: 10.1016/j.anr.2012.02.002. [DOI] [PubMed] [Google Scholar]
- 20.Pan Y, Chen R, Yang D. The relationship between filial piety and caregiver burden among adult children: A systematic review and meta-analysis. Geriatr. Nurs. 2022;43:113–123. doi: 10.1016/j.gerinurse.2021.10.024. [DOI] [PubMed] [Google Scholar]
- 21.Lee WK-M, Hong-kin K. Differences in expectations and patterns of informal support for older persons in Hong Kong: Modification to filial piety. Ageing Int. 2005;30:188–206. doi: 10.1007/s12126-005-1011-1. [DOI] [Google Scholar]
- 22.Lai DWL. Filial Piety, caregiving appraisal, and caregiving burden. Res. Aging. 2010;32:200–223. doi: 10.1177/0164027509351475. [DOI] [Google Scholar]
- 23.Chun M. Effects of Filial Piety values on the coping styles in family caregiver burden and depression. J. Korean Gerontol. Soc. 2006;26:665–680. [Google Scholar]
- 24.Hwang-ki K. Effects of the parent’s filial piety education on the educational culture of teenagers(3)—with a focus on the acceptance of traditional filial piety culture and modern filial piety culture. J. Youth Filial Piety Culture. 2018;32:145–175. [Google Scholar]
- 25.Ernst J, Weißflog G, Brähler E, Romer G, Götze H. Depressivität bei 11 bis 17 jährigen Kindern krebskranker Eltern im Verlauf und Abhängigkeit zur elterlichen Belastung. Prax. Kinderpsychol. Kinderpsychiatr. 2012;61:432–446. doi: 10.13109/prkk.2012.61.6.432. [DOI] [PubMed] [Google Scholar]
- 26.Annunziato RA, Rakotomihamina V, Rubacka J. Examining the effects of maternal chronic illness on child well-being in single parent families. J. Dev. Behav. Pediatr. 2007;28:386–391. doi: 10.1097/DBP.0b013e3181132074. [DOI] [PubMed] [Google Scholar]
- 27.Niemela M, et al. The prevalence of children affected by parental cancer and their use of specialized psychiatric services: The 1987 Finnish birth cohort study. Int. J. Cancer. 2012;131:2117–2125. doi: 10.1002/ijc.27466. [DOI] [PubMed] [Google Scholar]
- 28.Bultmann JC, et al. Parental cancer: Health-related quality of life and current psychosocial support needs of cancer survivors and their children. Int. J. Cancer. 2014;135:2668–2677. doi: 10.1002/ijc.28905. [DOI] [PubMed] [Google Scholar]
- 29.Visser A, Huizinga GA, van der Graaf WT, Hoekstra HJ, Hoekstra-Weebers JE. The impact of parental cancer on children and the family: A review of the literature. Cancer Treat Rev. 2004;30:683–694. doi: 10.1016/j.ctrv.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 30.Simantov E, Schoen C, Klein JD. Health-compromising behaviors: Why do adolescents smoke or drink? identifying underlying risk and protective factors. Arch. Pediatr. Adolesc. Med. 2000;154:1025–1033. doi: 10.1001/archpedi.154.10.1025. [DOI] [PubMed] [Google Scholar]
- 31.Wills TA, Resko JA, Ainette MG, Mendoza D. Smoking onset in adolescence: A person-centered analysis with time-varying predictors. Health Psychol. 2004;23:158–167. doi: 10.1037/0278-6133.23.2.158. [DOI] [PubMed] [Google Scholar]
- 32.Schwartz LA, et al. Preventative health and risk behaviors among adolescent girls with and without family histories of breast cancer. J. Adolesc. Health. 2019;64:116–123. doi: 10.1016/j.jadohealth.2018.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jantzer V, et al. Risk behaviors and externalizing behaviors in adolescents dealing with parental cancer—a controlled longitudinal study. Psychooncology. 2013;22:2611–2616. doi: 10.1002/pon.3327. [DOI] [PubMed] [Google Scholar]
- 34.Tabuchi T, et al. Tobacco smoking and the risk of subsequent primary cancer among cancer survivors: A retrospective cohort study. Ann. Oncol. 2013;24:2699–2704. doi: 10.1093/annonc/mdt279. [DOI] [PubMed] [Google Scholar]
- 35.Park SM, et al. Prediagnosis smoking, obesity, insulin resistance, and second primary cancer risk in male cancer survivors: National health insurance corporation study. J. Clin. Oncol. 2007;25:4835–4843. doi: 10.1200/JCO.2006.10.3416. [DOI] [PubMed] [Google Scholar]
- 36.Sales JM, et al. Rural parents' vaccination-related attitudes and intention to vaccinate middle and high school children against influenza following educational influenza vaccination intervention. Hum. Vaccin. 2011;7:1146–1152. doi: 10.4161/hv.7.11.17891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bennett P, Smith C. Parents attitudinal and social influences on childhood vaccination. Health Educ. Res. 1992;7:341–348. doi: 10.1093/her/7.3.341. [DOI] [PubMed] [Google Scholar]
- 38.Terry MB, Forman MR. Empowering pediatricians to prevent chronic disease across generations. Pediatrics. 2016;138:S92–S94. doi: 10.1542/peds.2015-4268M. [DOI] [PubMed] [Google Scholar]
- 39.Woodhouse SS, Ramos-Marcuse F, Ehrlich KB, Warner S, Cassidy J. The role of adolescent attachment in moderating and mediating the links between parent and adolescent psychological symptoms. J. Clin. Child Adolesc. Psychol. 2010;39:51–63. doi: 10.1080/15374410903401096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kim S, Ko YH, Jun EY. The impact of breast cancer on mother–child relationships in Korea. Psychooncology. 2012;21:640–646. doi: 10.1002/pon.1941. [DOI] [PubMed] [Google Scholar]
- 41.Cohen MM, Wellisch DK, Ormseth SR, Yarema VG. The father-daughter relationship in the wake of maternal death from breast cancer. Palliat. Support Care. 2018;16:741–748. doi: 10.1017/s1478951517000906. [DOI] [PubMed] [Google Scholar]
- 42.Landi G, et al. Illness unpredictability and psychosocial adjustment of adolescent and young adults impacted by parental cancer: The mediating role of unmet needs. Support. Care Cancer. 2021 doi: 10.1007/s00520-021-06379-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lindqvist B, Schmitt F, Santalahti P, Romer G, Piha J. Factors associated with the mental health of adolescents when a parent has cancer. Scand. J. Psychol. 2007;48:345–351. doi: 10.1111/j.1467-9450.2007.00573.x. [DOI] [PubMed] [Google Scholar]
- 44.Osborn T. The psychosocial impact of parental cancer on children and adolescents: A systematic review. Psychooncology. 2007;16:101–126. doi: 10.1002/pon.1113. [DOI] [PubMed] [Google Scholar]
- 45.Rodríguez L. An exploration of resilience in adolescents facing maternal cancer. Cancer Rep. 2020;3:e1208. doi: 10.1002/cnr2.1208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Askeland KG, et al. Life events and adolescent depressive symptoms: Protective factors associated with resilience. PLoS One. 2020;15:e0234109. doi: 10.1371/journal.pone.0234109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rodriguez L, Groarke A, Dolan P. Individual predictors of adolescent adjustment to maternal cancer: The role of perceived stress, coping, social support, attachment, and self-efficacy. Cancer Rep. 2019;2:e1145. doi: 10.1002/cnr2.1145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Palacio GC, Krikorian A, Gómez-Romero MJ, Limonero JT. Resilience in caregivers: A systematic review. Am. J. Hosp. Palliat. Med. 2019;37:648–658. doi: 10.1177/1049909119893977. [DOI] [PubMed] [Google Scholar]
- 49.Hirooka K, Fukahori H, Akita Y, Ozawa M. Posttraumatic growth among Japanese parentally bereaved adolescents: A web-based survey. Am. J. Hosp. Palliat. Med. 2017;34:442–448. doi: 10.1177/1049909115627776. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are openly available in the official website of KNHANES at https://knhanes.kdca.go.kr/knhanes/sub03/sub03_02_05.do.