Abstract
Feeding 10 billion people sustainably by 2050 in the era of slow genetic progress has spurred urgent calls to bring more crops per unit time. Over the last century, crop physiologists and breeders have been trying to alter plant biology to investigate and intervene in developmental processes under controlled chambers. Accelerating the breeding cycle via “speed breeding” was the outcome of these experiments. Speed breeding accelerates the genetic gain via phenome and genome-assisted trait introgression, re-domestication, and plant variety registration. Furthermore, early varietal release through speed breeding offers incremental benefits over conventional methods. However, a lack of resources and species-specific protocols encumber the technological implementation, which can be alleviated by reallocating funds to establish speed breeding units. This review discusses the limitations of conventional breeding methods and various alternative strategies to accelerate the breeding process. It also discusses the intervention at various developmental stages to reduce the generation time and global impacts of speed breeding protocols developed so far. Low-cost, field-based speed breeding protocol developed by Punjab Agricultural University, Ludhiana, Punjab, India to harvest at least three generations of wheat in a year without demanding the expensive greenhouses or growth chambers is also discussed.
Keywords: Rapid generation advancement, Conventional breeding, Doubled haploid, In-vitro nursery, Field-based speed breeding, And economic assessment
Introduction
Rising human population, depleting arable land, and fluctuating climates have received a great deal of devotion as an influence on world demand for food. With demand outweighing supply, the number of malnourished individuals will increase, and even if, on average, supply matches demand, abrupt climatic changes and surging diseases may cause irreparable economic shock, thus threatening the food security (Tanin et al. 2022). However, the environmental impacts and trade-offs of these alternative paths of agricultural expansion are unclear (Godfray et al. 2010).
Since the beginning of agriculture, conventional breeding approaches have brought together the co-adapted gene complexes to deliver high-yielding crop varieties which can be mechanically harvested. “Green revolution based on conventional approaches was a temporary success in man's war against hunger giving man a breathing space” (Borlaug 2007). However, the current pace of crop improvement for important field crops, including rice, wheat, and maize, is insufficient to get rid of hunger (Pennisi 2008; Ray et al. 2012). Since most crop plants have long generation time, achieving higher genetic gain and feeding unfed stomachs is challenging (Li et al. 2018). Plant breeders are concerned to increase the genetic gain by developing climate-smart, higher-yielding, nutritious crop varieties which are resilient to prevailing stresses (Gudi et al. 2022).
Genetic variability, heritability, selection intensity, and most importantly, the breeding time will influence the genetic gain, which is the improvement in the average performance over cycles of selection (Hazel and Lush 1942). Integrating conventional breeding techniques with advanced molecular and omics tools will facilitate the recovery of lost genetic variation (Gudi et al. 2020; Singh et al. 2022). Refinements in the scale and precision of high-throughput genotyping and phenotyping tools can significantly increase the selection intensity (Xu et al. 2020). Overall, the combined use of genotyping, phenotyping, and envirotyping with conventional breeding can deal with all the factors influencing genetic gain except the reduction in breeding time, which is the critical game-changer to maximize the genetic gain. Shortening the breeding cycle will be a promising way to enhance genetic gain without greatly increasing population size. Therefore, a rapid paradigm shifts in science-based advances in crop breeding, will be essential to reduce the breeding cycle of the crops via rapid generation advancement (RGA).
Australian scientists, enkindled by the National Aeronautics and Space Administration’s (NASA's) research to grow wheat in space, have developed an RGA technology, also known as “speed breeding,” to reduce the generation time and accelerate the breeding process (Hickey et al. 2019). Unlike the doubled haploid (DH) technique for the production of homozygous lines, speed breeding is not limited by genotype specificity and the requirement of well-equipped laboratories. However, the technology needs to deal with plant phenology and its physiology to shorten the generation time. Speed breeding accelerates trait introgression and the progression of high priority crosses to achieve homozygosity. This opportunity for trait selection in parallel with the fast development of inbred lines provides opportunity to achieve higher genetic gain (Li et al. 2018). Integrating speed breeding with advanced tools such as high-throughput phenotyping (Speed-phenomics), marker-assisted selection (Speed-MAS), marker-assisted backcrossing (Speed-MABC), marker-assisted recurrent selection (Speed-MARS), genomic selection (Speed-GS), de novo domestication (Speed-domestication), mutation breeding (Speed-MUT), and genome editing (Express edit) will reduce the length of the breeding cycle and increase genetic gain per unit time (Fels et al. 2019).
Conventional breeding: slow approach in view of today’s challenges
Conventional breeding is still a promising mean to develop cultivars through manipulating plant genomes within the natural genetic boundaries of the crop species. Conventional breeding has played a significant role in developing hundreds of thousands of crop varieties that consumers encounter in the markets.
The major limitations of conventional breeding methods are lengthy breeding cycles (10–15 years) to disseminate improved varieties by: (i) crossing selected parents; (ii) selfing in segregating generations (3–7 years); (iii) multilocational evaluation for yield, stress tolerance, and quality traits (4–5 years); and (iv) seed multiplication and release of variety (1–3 years) (Breseghello and Coelho 2013). Similar timelines exist for developing hybrids in cross-pollinated crops, where parental selection and inbreeding can take up to 10 years (Shimelis and Laing 2012). This sluggish crop improvement rate is partly attributed to the long generation time of crops (i.e., only 1 or 2 generations per year). Integrating novel omics tools with the traditional breeding methods has already hastened the selection procedures, however, it failed to achieve the expected genetic gain. This spurred an urgent call to adopt novel breeding techniques to hasten the breeding procedures by reducing the time required to develop new varieties.
Various strategies to speed up the breeding processes
The duration of crop breeding cycle can be reduced by using the following methods (Fig. 1).
Fig. 1.
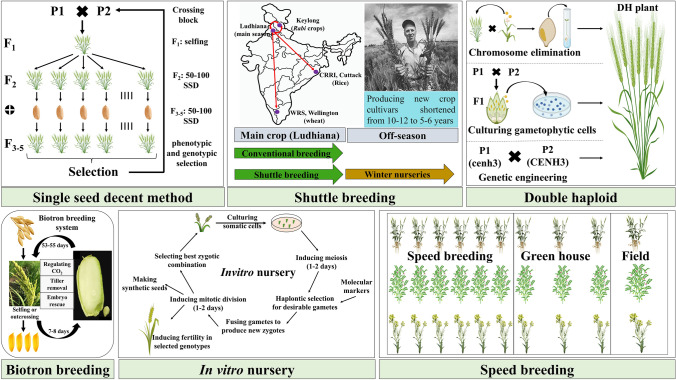
Various strategies to speed up the breeding process: (a) rapid generation advancement with single seed descent (RGA-SSD) method; (b) shuttle breeding to harvest two generations in a year; (c) double haploid (DH) production through chromosome elimination (Bulbosum technique and wheat × maize system), gametophytic cell (anther, pollen, and ovary) culture, and genetic engineering (cenh3); (d) biotron breeding system (BBS) for generation advancement in rice; (e) in-vitro nurseries to induce meiosis and mitosis to accelerate the generation advancement; (f) comparison of generation advancement using speed breeding, greenhouse, and conventional (field) breeding methods in wheat, chickpea, and brassica
Rapid generation advancement with single seed descent (RGA-SSD) method
Rapid generation advancement (RGA) is a technique of growing segregating generations in a greenhouse to accelerate breeding cycles. The Japanese rice breeders used the RGA technique for the first time in 1930 to advance the F2 generations to recombinant inbred lines (RILs) (Maruyama 1989) (Fig. 1a). The single seed descent (SSD) method in connection with RGA was proposed by the Goulden (1939). It is an effective tool to speed up the breeding cycles by producing homozygous lines and reducing the cost by requiring less space and labour (Goulden 1939). Since its proposal, the RGA-SSD is becoming popular to advance the breeding cycles among many crop plants, including rice (Collard et al. 2019), wheat (Tanio et al. 2006), soybean (Carandang et al. 2006), chickpea (Gaur et al. 2007), tomato (Bhattarai et al. 2009), sorghum (Rizal et al. 2014), cotton (Wang et al. 2011), etc.
A famous rice cultivar, ‘Nipponbare,’ released in 1963, was the first commercial variety bred by RGA-SSD (Koumura 1972b). He reported reduction in generation time by regulating temperature and photoperiod so as to harvest three generations per year. Since then, RGA-SSD technique has been utilized to develop cultivars, RILs (Janwan et al. 2013), backcross inbred lines (Robin et al. 2003), chromosome segment substitution lines (Toshio Yamamoto, personal communication) etc., The major limitation of RGA- SSD is the screening of miniature plants grown in dense conditions, where only highly heritable traits are amenable for seedling evaluation. Depending on crop species and the availability of resources, RGA-SSD will be able to achieve a maximum of three generations in a year (Koumura 1972a).
Shuttle breeding: double harvest season
Shuttle breeding relies on the successful evaluation of segregating generations in two distinctly different geographic sites and seasons in the same year. The concept was introduced by the 1970’s Nobel Peace Prize Laureate, Dr. Norman E. Borlaug, where he used two specific sites in Mexico to harvest two generations of wheat in a year, when other breeders harvested only one generation in a year (Ortiz et al. 2007). In this, selected plant material from one location were shifted to another location after each generation, thereby halving the time required to develop a new cultivar (from 10–12 to 5–6 years) (Borlaug 2007). Shuttle breeding motivated breeders across the globe, including India, to adopt double harvest seasons to advance the segregating generations.
Scientists from the Punjab Agricultural University (PAU), a premier agricultural university of India, have adopted shuttle breeding programs since 1990 for major Kharif and Rabi crops (Fig. 1b). The main season is harvested in Ludhiana (i.e., mid-June to mid-October for the Kharif crops and November to the end of April for the Rabi crops, while the additional season is taken at the National Rice Research Institute, Cuttack, Odisha, for the rice (December to May), and at high altitudes such as Wellington (Tamil Nadu) and Lahaul-Spiti (Himachal Pradesh) for the Rabi crops (May to September).
Though the main focus of shuttle breeding is to speed up the breeding, Borlaug perceived the supplementary rewards of shuttling genotypes between the two geographical locations. Firstly, the broader adaptability of breeding material due to continued exposure of genotypes to contrasting environments involving different disease spectra, soil types, and environmental stresses. Secondly, the photo- and thermo-sensitive genotypes were selectively eliminated from the breeding population, allowing the development of semi dwarf wheat varieties across the globe (Ortiz et al. 2007). In addition, photo and thermo-insensitive genotypes reduce the generation time by allowing early flowering. This gave sufficient time to shift breeding material from one place to another during shuttle breeding. The major limitations of shuttle breeding are the cost for transport of material, resources, labour etc., chance of losing genetic material during transportation and the short time between crop cycles.
Doubled haploid (DH): single recombination to attain homozygosity
Producing homozygous lines is time-consuming part of breeding, taking years in a traditional breeding program (Santra et al. 2017) and can be overcome by using DH technique, which is the two-step approach to homozygosity in one round of recombination (Forster and Thomas 2005). In the first step, haploid plants will be generated by culturing gametophytic cells (Kasha and Kao 1970); making crosses to induce chromosome elimination (i.e., Bulbosum technique, wheat × maize system, etc.) (Laurie and Bennett 1988) and using haploid inducer stocks (indeterminate gametophyte 1 (ig1) in maize and CENH3 in Arabidopsis thaliana) (Guo et al. 2004; Ravi and Chan 2010) (Fig. 1c). In the second step, haploid plants will be treated with colchicine to induce chromosome doubling to develop homozygous diploid plants. Cell culture and chromosome elimination are the most promising techniques for haploid production. However, the genotype specificity and the requirement of tedious tissue culture for these techniques restrict their widespread application. In maize, haploid plants (~ 3%) can be produced by crossing female plants having ig1 mutations with male plants carrying mutations for the dmp (domain of unknown function 679 membrane protein) gene (Guo et al. 2004). CENH3 is the centromere-specific histone-3 variant that plays an important role in assembling the kinetochores. The null mutants of CENH3 (ex., cenh3-1) interfere with the assembly of kinetochore in the centromeres, thereby affecting the chromosomal separation during cell division (Jiang et al. 2003). The mutant forms of CENH3 (i.e., cenh3) were utilized to induce haploid production in A. thaliana (Ravi and Chan 2010). By crossing CENH3 mutants with their wild type, paternal (25–45%) or maternal (4–5%) haploid plants depending on whether the mutants were used as female or male parents, respectively, were produced (Ravi and Chan 2010). Several attempts were made to produce haploid plants by editing the N-terminal tail of CENH3 in rice (0.3–1%), tomato (0.2–2.3%) (Kalinowska et al. 2019), and maize (0.065–0.86%) (Kelliher et al. 2016). In addition to N-terminal editing, efforts were also made to induce haploid production by editing the C-terminal region of CENH3 (Ashtiyani et al. 2015; Kuppu et al. 2015) or by replacing the crop CENH3 with its wild relatives (Maheshwari et al. 2015). The C-terminal edited and CENH3 replaced point mutants are viable and fully fertile with an improved haploid recovery rate.
In addition to the above techniques, PAU, Ludhiana has deployed the detached tiller culture technique for DH production. This technique has dramatically reduced the time required to develop homozygous lines, thereby speeding up the breeding process in wheat (Srivastava and Bains 2018). Reduced time and increased efficiency of wheat breeders to develop new cultivars can bring about high-yielding, better-adapted seed options at a faster pace (Bains et al. 1995). Technological limitations involving-tissue culture laboratories encumber the widespread application of DH technology. Furthermore, acute genotypic specificity, low frequency of haploid production, contamination of DH's with aneuploids and mixed haploid-diploid types, depleting genetic variation, and daunting selection efficiency are challenges of DH technology.
Biotron breeding system (BBS): an artificial growth chamber for rice breeding
Biotron breeding system (BBS) is a rapid and reliable method of producing advanced generations of rice using artificial growth chambers supplemented with controlled temperature and CO2, accompanied by tiller removal and embryo rescue (Ohnishi et al. 2011) (Fig. 1d). Programmed lighting and temperature systems inside the biotron significantly shorten the life cycle of famous rice variety "Nipponbare" to about 2 months. However, tiller removal and embryo rescue are limiting the application of BBS to large breeding populations.
Tanaka and co-workers introduced the simplified-BBS (sBBS) that eliminates the need for tiller removal and embryo rescue but controls CO2 levels and day-length inside the growth chambers (Tanaka et al. 2016). Compared to 2 months in BBS, the sBBS could reduce the interval between generations in "Nipponbare" to less than 3 months. In addition, sBBS helped to develop the isogenic lines of "Oborozuki" and "Akidawara" for Wx1-1, a low amylose allele by allowing four generations of rice in a year. To accomplish many generations per year, this approach can also be applied to other crops.
In-vitro nursery: rapid cycles of meiosis and mitosis to reduce generation time
This is the concept of inducing meiosis in single somatic cells and fusing a desirable combination of gametes to yield appropriate genotypes (Fig. 1e). Here, the generation time is governed by how fast new gametes are produced and unified to form zygotes (Gerald et al. 2013). These zygotes could be induced to undergo further meiosis to produce new gametes (Kranz’ and Lorz 1993). This contrasts with the in-vivo system, where the breeder needs to wait until flowering and seed maturity to produce the next generation. Finally, selected gametes or cell lines could be transformed into mature plants.
The benefit of in-vitro nurseries is the accelerated development of inbred lines by completing 12 cycles of recombination and selection in a year. In addition, these nurseries help in: (i) overcoming self-incompatibility through bypassed pollination; (ii) MABC for targeted gene introgression; (iii) gametic selection to improve the selection efficiency; and (iv) producing artificial seed through encapsulated somatic embryos. This system has broad applicability, including trees, photo, thermo-sensitive species, apomictic species, and annual crops. Although the proposed idea is novel and innovative, it has the several drawbacks. Some of them includes: (i) ability to induce meiosis and produce gametes in vitro; (ii) genotype specificity for regeneration; (iii) unintended selection for loci; and (iv) the phenomenon of somaclonal variation, which counteracts homozygous and homogeneous line production (Gerald et al. 2013).
Speed breeding: NASA’s inspiration to curtail generation time
Just imagine that, in an era of creeping genetic progress, a breeder thinks of harvesting 6 or 7 generations of a crop in a year to have F4-derived lines within 12 months, thus, releasing variety in 6–7 years instead of 10–12 years (Fig. 1e). Transforming this fantasy into reality necessitates technological improvement and intervention in the various developmental stages of crop plants. Over the last century, scientists from various disciplines are consistently engaged in technological improvements to harvest six or seven generations in a year (Fig. 2). For the first time, plant physiologists showed the ability of plants to grow under artificial light using carbon arc lamps (Siemens 1880). Later, the effects of continuous light on plant growth and flowering were investigated in many crop plants (Arthur et al. 1930). In the early 1980s, NASA partnered with Utah State University to evaluate world germplasm collections of wheat in search of suitable genotypes for food production in space conditions (Bugbee and Koerner 1997) leading to development of "USU-Apogee," a high-yielding, full-dwarf hard red spring wheat variety. The major revolution came with use of light-emitting diodes (LEDs) with reduced operational costs to bolster indoor propagation (Bula et al. 1991; Stutte 2015). Inspiration from NASA experiments and the reduced cost of LEDs help the scientists from the University of Queensland to achieve the vision of harvesting six or seven generations in a year (Watson et al. 2018) (Fig. 2).
Fig. 2.
Step-wise evolution of speed breeding technology and its integration with high-throughput phenotyping, high-throughput, genomic selection, and genome editing tools
Speed breeding involves a set of protocols that use the optimal light quality, intensity, photoperiod, and temperatures to accelerate flowering in crop plants. In addition, speed breeding also uses the immature seed harvesting to curtail the generation time (Hickey et al. 2019). Species specific protocols are available to induce flowering under specific environmental cues. Speed breeding has been successfully used to accomplish up to eight generations per year in lentil, seven generations in faba bean (Mobini et al. 2015), six generations in spring wheat, durum wheat, barley, chickpea, and pea (Watson et al. 2018), five generations in winter wheat (Cha et al. 2021), pea (Mobini and Warkentin 2016), soybean, rice, and amaranth (Jähne et al. 2020), four generations in canola (Watson et al. 2018) and pigeon pea (Saxena et al. 2019), and 3–4 generations in groundnut (O’Connor et al. 2013). High-density planting in speed breeding units reduces the space requirement and costs involved in developing inbred lines (Ghosh et al. 2018).
Speed breeding systems
Since the introduction of speed breeding various protocols and speed breeding units have been adapted depending on the species. Watson et al (2018) used the sophisticated growth chambers to reduce the generation time. It needs the exposure of long-day crops to protracted photoperiod (i.e., 22 h of light), elevated temperature (22 °C during day and 17 °C at night), and high-humidity (> 70%). White LED bars, far-red LED lamps, and quartz iodide lamps were used as light sources and were optimized to ramp-up and down to mimic natural dawn and dusk conditions. The light quality (i.e., blue/red light ratio) was set to 360–380 and 490–500 µmol/m2/s at the bench-height and adult plant height stages, respectively. They also proposed high-pressure sodium lamps as a light source in the temperature-regulated glasshouses. This system does not include changes in light and temperature to create a ramping up and down. Similarly, scientists at ICRISAT, Hyderabad (International Crops Research Institute for the Semi-Arid Tropics, Hyderabad), proposed a low-cost greenhouses facilitated with natural light and evaporative air coolers for generation advancement in pigeon pea (Saxena et al. 2019). Watson et al (2018) also suggested the low-cost, homemade rooms (insulated) fitted with LB-8 LED light-boxes (140 cm above the bench) and domestic air conditioners (21 °C during the day and 18 °C at night). In this system, lighting was set to run a 12 h photoperiod for 4 weeks and then increased to an 18 h photoperiod. The lights did not ramp up and down during the day and night switch.
By using the LED-controlled lighting system, Jahne et al. (2020) designed the simple speed breeding protocol for accelerating generation advancement in short-day crops. It includes the speed breeding boxes with reflecting surfaces fixed inside the growth chambers and different LED modules (CompLED, Relumity, and Growking) emitting wavelength-specific light spectra (viz., blue-light or far-red enriched light). The growing conditions inside the box were well maintained with 28 °C day and night temperatures, 80% humidity, and 10 h of light.
Adopting sophisticated growth chambers and glasshouses as amenities are too expensive, but given the importance of generation advancement, (Chiurugwi et al. 2019) proposed alternative approaches (i.e., speed breeding capsules and centres) for accelerated breeding of orphan crops. Firstly, they have supposed to build “speed breeding capsules” out of waste containers (typically refrigerated or steel-coated) by providing a source of light, temperature, and irrigation. Speed breeding components could also be arranged in ‘kit’ form and assembled in-situ to further reduce the cost incurred in setting-up the capsules for poorly funded areas. These capsules can be used to grow plants of different heights in both warm (ventilator) and cold (insulator) climates. The second approach was to establish “speed breeding centres” at the regional stations to provide training and trait integration services. These centres would be equipped with the necessary facilities for carrying out speed breeding. Scientists at the ICRISAT, Hyderabad, the IITA, Nigeria (International Institute of Tropical Agriculture, Nigeria), and the ICARDA, Beirut (International Centre for Agricultural Research in the Dry Areas, Beirut) intend to develop speed breeding units and such alliances may benefit the improvement of critical orphan crops via generation advancement.
Speed breeding for long-day and short-day crops
Long-day crops, including wheat, barley, chickpea, mustard, and pea, are cool-season crops require a longer light period of 14–16 h for flowering. In contrast, short-day crops flower only when the light hours fall below the critical day length (i.e., 8–10 h). Some of the agriculturally important short-day crops includes cereals (maize, rice, and millets), legumes (pigeon pea), and oilseeds (soybean and groundnut). The number of light hours plays an important role in flower induction by altering the activity of phytochromes. During the daytime, plants accumulate higher amount of far-red light-absorbing active phytochromes (Pfr). During the dark period, Pfr will slowly convert to its inactive, red-light absorbing form (Pr). Extending photoperiod above the threshold hours will prevent the conversion of Pfr to Pr and will affect the flower initiation in photoperiod sensitive crops. For instance, accumulation of Pfr above the threshold level will trigger the early flowering in long-day crops (Carre et al. 2008). This characteristic feature of long-day crops has been exploited to reduce their generation time. For instance, by exposing the crops to 22 h of light, a group of scientists from the University of Queensland achieved early flowering in spring wheat (6 generations per year), winter wheat (5 generations per year), durum wheat (6 generations per year), barley (6 generations per year), chickpea (6 generations per year), pea (6 generations per year), and canola (four generations per year) (Watson et al. 2018) (Table 1).
Table 1.
List of speed breeding protocols developed in long-day and short-day crops
| Crop | Field/greenhouse generation | Speed breeding generations | Methodology | References | |
|---|---|---|---|---|---|
| Days | Cycles | ||||
| Long-day crops | |||||
| Lentil (Lens culinaris) | 102–107 | 45 | 8 | Optimum flurprimidol, indole-3-acetic acid, zeatin and light intensity | Mobini et al. (2015) |
| – | 3–4 | 22 h light with 25–28 °C and 2 h dark with 12–18 °C under SSD | Idrissi (2020) | ||
| Pea (Pisum sativum) | 60–80 | – | 5.3 | Flurprimidol, photoperiod (20 h light with 500 μM m−2 s−1 light intensity), temperature (21 °C at light and 16 °C at dark), 266 plants/m2, and hydroponics with fertigation | Mobini and Warkentin (2016) |
| – | 6 | 22 h light/2 h dark with 22/17 °C temperature, immature seed harvest and drying | Watson et al. (2018) | ||
| – | 4–5 | Hydroponics, temperature (20 ± 2 °C), photoperiod (22 h light provided with T5 fluorescent tubes), flurprimidol antigiberelin and immature seed harvest | Cazzola et al. (2020) | ||
| Sugarcane (Saccharum officinarum) | > 365 | – | – | Short-day and constant supply of nutrients to induce synchronous flowering | Hale et al. (2017) |
| Spring wheat (Triticum aestivum) | 145–155/105 | 62 | 6 | 22 h light/2 h dark with 22/17 °C temperature, immature seed harvest and drying | Watson et al. (2018) |
| Durum wheat (Triticum durum) | 150–155/105 | 62 | 6 | ||
| Barley (Hordeum vulgare) | 130–150/110 | 55 | 6 | ||
| Chickpea (Cicer arietinum) | 150–160/90 | 58 | 6 | ||
| Canola (Brassica napus) | 135–150/120 | 75 | 4 | ||
| Model grass (Brachypodium distachyon) | 75 | 45 | 4–6 | ||
| Oat (Avena sativa) | 114 | 72 | 3.2–4.9 | 22 h of photoperiod, controlled temperature and early seed harvest with SSD | Gonzalez-Huitron et al. (2021) |
| Winter wheat (Triticum aestivum) | 160–180 | – | 5 | Expose wheat seeds to protracted photoperiod (22 h of light) and shift to speed breeding units | Cha et al. (2021) |
| Faba bean (Vicia faba) | 104 | 54 | 7 | Optimum flurprimidol, indole-3-acetic acid, zeatin and light intensity | Mobini et al. (2015) |
| 80 | – | 6-benzylaminopurine (BAP) (cytokinin) application and cold treatment | Mobini et al. (2020) | ||
| Potato (Solanum tuberosum) | 140 | – | – | Speed breeding using protracted photoperiod is under development | James Hutton Institute |
| Short-day crops | |||||
| Groundnut (Arachis hypogaea) | 125–145 | 89 | 3–4 | Continuous light, with 28/17 °C temperature, high-intensity light and SSD | O’Connor et al. (2013) |
| Tomato (Solanum lycopersicum) | 80 | – | – | Introgression of continuous light tolerance gene chlorophyll a/b binding protein 13 (CAB-13) to increase productivity under continuous light | Gonzalez-Huitron et al. (2021) |
| Sorghum (Sorghum bicolor) | 100–120 | 88 | – | Split culm (which produces crossed and selfed seeds in the same panicle of the uniculm plant) and embryo rescue | Rizal et al. (2014b) |
| Rice (Oryza sativa) | 90–120 | – | 4–6 | Regulation of CO2, day-length and maintains an appropriate root volume | Tanaka et al. (2016b) |
| 95–105 | – | Field-based RGA with < 40 cm3 soil/plant | Richard et al. (2018) | ||
| 78–85 | 5 | CO2 (560–800 ppm) supplementation, 10 h light (far-red), 27/25 °C temperature, 260 cm3 soil/plant | Jähne et al. (2020) | ||
| 60–65 | 6 | CO2 regulation, tiller removal, and embryo rescue | Richard et al. (2018) | ||
| Amaranth (Amaranthus spp.) | 95–100 | – | 6 | 8 h light and 30 °C | Richard et al. (2018) |
| – | 5 | CO2 (560–800 ppm) supplementation, 10 h light (far-red), 27/25 °C temperature, 260 cm3 soil/plant | Jähne et al. (2020) | ||
| Soybean (Glycine max) | 100–140 | 70 | 5 | Fluorescent lamps with 14 h photoperiod, 30/25 °C day/night temperature, CO2 regulation (400–600 ppm), and improved crossing efficacy | Tanaka et al. (2016b) |
| 77 | 5 | CO2 (560–800 ppm) regulation, 10 h light (blue light enriched) (480–1511 nm), 27/25 °C temperature, immature seed harvest, and 260 cm3 soil/plant | Jähne et al. (2020) | ||
| 63–81 | 5 | Red–blue light coupled with photothermal conditions | Harrison et al. (2021) | ||
| Pigeonpea (Cajanus cajan) | 120–190 | – | 4 | Immature seed germination for RGA and a “single pod descent” | Saxena et al. (2019) |
Short-day crops are sensitive to Pfr accumulation and will flower only when the duration of dark hours are sufficient to convert most of the Pfr to Pr (Carre et al. 2008). Therefore, the principle of using extended photoperiod is not an appropriate technique to induce early flowering in photoperiod-sensitive, short-day crops (Stetter et al. 2016). However, preliminary evaluation and selection of photoperiod-insensitive and phytochrome-deficient accessions of rice, maize, pigeon pea, soybean, etc. is an optional opportunity to grow short-day crops under extended photoperiod (Izawa et al. 2000). The conversion of the active form of phytochromes to an inactive form using specific light intensities (i.e., far-red light) will induce early flowering under extended photoperiod. Such opportunities were exploited to reduce generation time in groundnut (O’Connor et al. 2013) and soybean (Jähne et al. 2020).
Such opportunities are not for all short-day crops and their genotypes. Therefore, it is advised to devise crop-specific speed breeding protocols for these crops. Concerning these requirements, German scientists demonstrated the LED-based speed breeding protocols for soybean, rice, and amaranthus (Jähne et al. 2020). Their initial experiments noticed the delayed and heterogeneous flowering among the soybean genotypes evaluated for different light regimes with an increased photoperiod (low red; 600–700 nm to far-red; 700–800 nm light). Finally, they established a protocol to achieve five generations of rice, soybean, and amaranthus in a year by adjusting the 10 h of light period with enhanced blue-light (450–490 nm) without providing far-red (> 700 nm) light. Far-red light helps to induce early flowering in some accessions of rice (by 20 days) and amaranthus (by 10 days). These results highlight that speed breeding protocols developed for one crop cannot be readily interchanged with another crop. Specifically, light quality needs to be taken into account for the best genotype-independent speed breeding techniques.
Speed breeding for high-biomass crops
High-biomass crops, including sugarcane, potato, sweet potato, etc., are propagated vegetatively. The commercial cultivars of these crops (called clones) are becoming more susceptible to prevailing biotic and abiotic stresses. This can be overcome by developing climate-resilient and pest and disease-resistant clones. The conventional techniques of hybridization and clonal selection are inefficient in meeting these demands, as it requires at least 10–14 years to release the best clone as a variety (Mahadevaiah et al. 2021). Speed breeding offers an excellent opportunity to shorten the breeding cycle by performing faster crosses and clonal selection, thereby accelerating varietal release. There are no reports on the application of speed breeding in these crops, except little effort made in sugarcane to induce flowering by providing artificial light (Hale et al. 2017). High-speed potato breeding (HISPOB) has been proposed to develop new potato varieties within 2 years (Horizon 2020; https://cordis.europa.eu/project/id/666729). The proposed strategy may bring high-yielding clones/varieties with resistance to prevailing diseases. Therefore, establishing speed breeding protocols for high-biomass crops may accelerate their breeding cycle and also helps in sustainable production.
Speed breeding for fruit tree crops
Fruit tree crops are challenged by several stresses which affect the quality and quantity of fruit production. Genetic improvement of fruit tree crops is the best solution to overcome these challenges but is hampered by long juvenile periods, dormancy, alternative bearing, complex genetic architecture and reproductive biology, a high level of heterozygosity, linkage drag, and field costs (Flachowsky et al. 2011). Conventional breeding approaches requires 15–20 years to develop a variety in these crops. Opportunities are available to reduce the breeding cycle, specifically by reducing the juvenile periods through manipulation of temperature and photoperiod; application of growth retardants; trunk ringing and root pruning; micro-budding; grafting; and creation of early flowering mutants. Efforts were made to induce early flowering by engineering flowering genes, FT in citrus, LFY in citrus and apple (Pena and Séguin 2001), AP1 in citrus and Sweet Kumquat (Duan et al. 2010), and BpMADS4 in apple (Flachowsky et al. 2011). Such efforts helped to reduce the juvenile period from 5 to 10 months in apple (Nocker and Gardiner 2014) and from 7 to only 2 years in chestnut (Baier et al. 2012). This technique of reducing the juvenile period by engineering early flowering genes is popularly called "FastTrack" breeding. In addition to reducing the juvenile period, FastTrack breeding also accelerates the trait introgression from wild species without the linkage drag. For instance, overexpression of the mutant MADS4 transcription factor helped to develop fire blight resistant apple lines (i.e., F5 generation) within 7 years (Flachowsky et al. 2011). Integrating speed breeding with FastTrack breeding may further reduce the breeding cycle by harvesting multiple generations in a year. However, the plant architecture of fruit tree crops is the major constraint which prevents them from growing under controlled growth chambers. Further efforts are needed to reduce the plant height or to develop special growth chambers that can fit tree crops which may help to reduce the juvenile periods, thus accelerating the varietal developmental in trees.
Intervening key developmental stages to reduce generation time
Growth and development are the fundamental and conspicuous characteristics of crop plants. During this process, plants pass through various developmental stages, including germination/emergence, vegetative growth, budding and flowering, grain-filling, ripening, and senescence. Each species has specific periodicity of transit from one stage to another, which is influenced by many intrinsic and extrinsic factors. Reducing the transition phase, specifically from the vegetative to the reproductive, needs in-depth knowledge regarding the developmental processes of species, so that breeders can effectively reduce the breeding cycle (Fig. 3).
Fig. 3.
Intervening the key developmental stages of plants to reduce generation time
Breaking seed dormancy and inducing faster germination is the foremost step of physiological intervention to reduce the breeding cycle. Seed dormancy can be overwhelmed by germination-promoting factors such as light, cold treatment (stratification), and chemicals such as sulphuric acid (scarification), gibberellins, KNO3, and NO (Penfield 2017). Immature seed harvesting, drying, and cold stratification breaks the seed dormancy and thereby reduce the generation time by 15 days in wheat, barley, soybean, and lentil (Watson et al. 2018; Jähne et al. 2020). The creation of null mutants of LEC1, LEC2, FUS3, and ABI3, genes coupled with embryo rescue helped to overcome the dormancy and enhance the germination of immature wheat seeds (harvested between 8 and 12 days after pollination) (Raz et al. 2001).
Crop plants usually shun flowering immediately after germination and display a short juvenile phase of reproductive incompetence. Transition period from vegetative to reproductive stage can be reduced by encountering the early genotypes, altering the plant growth regulators (PGRs; Auxin, Cytokinin, and flurprimodal), creating the stress (viz., drought, heat, and high-density planting), controlling temperature and photoperiod conditions. Genotypes like "USU-Apogee" and “Fast-Flowering Mini-Maize (FFMM)” with a very short life cycle can be used to accelerate the breeding cycles by reducing vegetative phase (Bugbee and Koerner 1997; McCaw et al. 2016). Exogenous application of PGRs will improve plant growth and development. For instance, application of (i) IAA (indole-3-acetic acid) and 4-CL-IAA (4-chloro-IAA) enhanced the growth and seed setting in green gram (Ali et al. 2008); (ii) auxins and cytokinins induced early flowering in white lupin (Saeid et al. 2011) and lentil (Khalil et al. 2006); (iii) flurprimidol reduced the internodal length and vegetative growth in field pea and grass pea (Ochatt et al. 2002); and (iv) flurprimidol and 4-CL-IAA enhanced the anthesis and pod setting in faba bean (Mobini et al. 2015). Exposing to stress conditions, such as drought and heat at key developments stages will induce early flowering as a stress escape mechanism (Wada and Takeno 2010; Hatfield and Prueger 2015). High-density planting (> 1000 plants/m2) may also induce the early flowering by restricting the availability of nutrients and room for root growth (Watson et al. 2018). Many plant species, including economically important cereals such as wheat and barley, the model plant A. thaliana, and the legume plant lupin (Lupinus angustifolius L.), require prolonged cold exposure (vernalization) to induce flowering (Trevaskis et al. 2007; Bielska et al. 2020). Manipulating various genetic (viz., VRN1, VRN2, and VRN3) and epigenetic (viz., DNA methylation and histone modifications) mechanisms of vernalization could hasten the flower induction in these species (Trevaskis et al. 2007).
The developmental processes of the plants can also be accelerated by optimizing the light quality and duration. Extended photoperiod (22 h light) promotes early flowering in long-day crops like wheat, barley, chickpea, pea, and mustard (Watson et al. 2018). However, except for some phytochrome-deficient or insensitive genotypes of rice, sorghum, and soybean, most of the short-day plants are sensitive long day light (i.e., > 12 h) (Jähne et al. 2020). Therefore, it is of foremost importance to optimize the duration and quality of light to induce early flowering in short-day crops. For instance, by adjusting the photoperiod and appropriate combination of red to blue light ratios researchers were able to induce early flowering in soybean (by 23 days), rice (by 60 days), and amaranth (by 35 days).
Applications of speed breeding
We have discussed the efficacy of speed breeding to advance the generations of several crop plants by shortening the breeding cycle. In addition, speed breeding can also favour adult plant phenotyping, rapid trait introgression and pyramiding, synchronous flowering, crop domestication, and genetic diversity mining.
Speed breeding enables the phenotyping of large segregating generations for stable and highly heritable traits such as resistance to yellow rust (Hickey et al. 2012), brown rust (Riaz et al. 2016, 2018), stem rust (Riaz and Hickey 2017), yellow spot (Dinglasan et al. 2016), crown rot (Alahmad et al. 2018), and Fusarium head blight (Watson et al. 2018). It also helps phenotype agronomic traits, including root angle, number (Richard et al. 2018), cuticular wax (Alahmad et al. 2018), flowering, height, and pre-harvest sprouting (Watson et al. 2018). Combining high-throughput phenotyping platforms with speed breeding may speed up the gene discovery and their introgression into elite cultivars. The modified backcross method, which combine speed breeding and high-throughput phenotyping, has successfully transferred multiple resistance in elite barley cultivars (Hickey et al. 2017), pre-harvest sprouting in spring wheat (Hickey et al. 2019), and salt tolerance in rice (Rana et al. 2019). Rapid trait stacking through “Speed-MAS” has been proposed to develop super wheat pyramided lines by combining tolerant genes for preharvest sprouting, rust, Fusarium head blight, and salinity (Hickey et al. 2019). Speed breeding enables the synchronous flowering by shifting the plants in step-wise from long-day conditions to short-day conditions, which facilitate the flower production at different stages (Jähne et al. 2020). In addition, speed breeding facilitates accelerated re-domestication through the production of synthetic polyploids. Synthetics polyploids can be utilized in breeding program to increase the genetic diversity or to transfer the desirable alleles in cultivar background.
Inducing the targeted (using CRISPR-CA9) or random mutations (using chemical or physical mutagens) will produce the M0 plants with novel phenotypes. The M0 plants need to be advanced to next generation and evaluated before release as a variety which requires at least 6–8 years to release a mutant variety. The lengthy breeding process can be alleviated by integrating speed breeding with mutation breeding (i.e., SpeedMUT) or genome editing (i.e., Express edit). In this strategy, one can grow the initial mutant generations (i.e., M0–M5) or Cas9 constructed plants in the speed breeding units to advance their generation or to achieve rapid introgression of mutant alleles. SpeedMUT and Express Edit can also be combined with high-throughput phenotyping and MAS to further improve the efficiency of breeding process. This will help to develop the mutant variety or a variety with a mutant allele in a short span of time (i.e., 5–6 years instead of 8–10 years).
Speed DUS testing: hasten the registration of new plant varieties
Plant Variety Rights (PVRs) are the intellectual property prerogatives designed by the International Union for the Protection of New Varieties of Plants (UPOV) to safeguard the substantial economic investment of the plant breeders (Helfer 2004). New plant varieties will be protected under PVR only when they pass the DUS (Distinctness, Uniformity, and Stability) and VCU (Value for Cultivation and Use) tests (Yang et al. 2021). DUS testing evaluates the morphological traits in the field, thereby providing the foundation for seed certification, plant varietal protection (PVP), and plant breeder's rights (PBR) (Jamali et al. 2019).
The dissemination of improved crop varieties to the farmer’s field is delayed by the arduous and time-consuming process of varietal registration. Currently, depending on the country and the crop type, the overall timeframe for variety registration might range from 2 to 4 years (Jamali et al. 2020). However, it is conceivable that alterations to the current registration procedure could hasten the delivery of improved varieties to farmers. Until this happens, the lengthy procedures of DUS testing will prevent the utilization of speed breeding techniques that attempt to accelerate the development of new crop varieties.
"Speed-DUS testing” is an excellent approach to reduce the DUS testing duration without violating the standard protocols proposed for DUS testing (Jamali et al. 2020). In this technique, DUS testing will be carried out under RGA in a shorter time period, replacing the labour-intensive field evaluation of crop varieties. However, it is anticipated that all DUS traits are not amenable for evaluation under speed breeding units. In addition to this, the lack of facilities is another major drawback, limiting its versatility. Even if speed-DUS testing cannot be implemented to save time, the method would still offer significant advantages to improve the efficiency of DUS testing, which is the key requirement for varietal registration and protection.
Global impact of speed breeding
As an economic assessment, conventional breeding methods have benefited from improved varieties for their investments. Information on economic returns from the breeding of new varieties is available for the conventional (Brennan et al. 2007), DH (Brennan 1989), and RGA methods (Lenaerts et al. 2018). However, the economic benefit assessments such as cost–benefit ratio, net present value, and internal rate of returns are not available for speed breeding techniques. Even though many crop scientists are willing to adopt speed breeding systems, the high relative cost of establishment and maintenance of speed breeding units (i.e., 3930 USD or 720.72 USD per annum) and the lack of economic assessment encumber its implementation (Lenaerts et al. 2018). Thus, a vigilant assessment of the different variables impacting the relative costs and returns of speed breeding is required before the economics of its usage can be ascertained.
While studying the global impact of RGA, Lenaerts and colleagues calculated the “multiplicator factors” of RGA on the profits earned from conventional breeding methods and applied them in a meta-analysis (Lenaerts et al. 2018). Their models relied on the assumption of similar costs and benefits from conventional and RGA methods. They showed the higher economic benefits from adopting accelerated breeding procedures. Such multiplicator factors can be used for making the economic assessment of adopting accelerated breeding techniques by any researchers.
For instance, by using the Eqs. 1 and 5 of Lenaerts et al. we predicted the annual benefits and percentage of incremental benefits for shuttle breeding (~ 2 years reduction), RGA (~ 3 years reduction), DH (~ 3 years reduction), speed breeding (~ 4 years reduction), speed-GS (~ 5 years reduction), speed-GS + speed-DUS (~ 6 years reduction) over the conventional breeding method at different discount rates (i.e., 1, 5 and 10%) (Figs. 4 and 5). In addition, we used the 13 economic assessment studies done in rice and wheat for meta-analysis (Eq. 7 of Lenaerts et al.) to compute the incremental benefits (Table 2). These results showed that, percentage of incremental benefits are directly proportional to the number of years of breeding cycle reduction (Fig. 5). Maximum incremental benefits were achieved in speed breeding techniques integrated with GS and DUS. Future economic assessments must include the cost incurred in each breeding methods to assess their economic benefits.
Fig. 4.
Time scale of varietal development and the annual benefits achieved through conventional breeding, shuttle breeding, double haploid (DH) or rapid generation advancement (RGA), speed breeding, speed breeding with genomic selection (SpeedGS), and SpeedGS with SpeedDUS (speed breeding with varietal registration)
Fig. 5.
Incremental benefits of accelerated breeding methods over conventional breeding approach at 1% (yellow), 5% (orange), and 10% (blue) discount rates
Table 2.
Incremental benefits (in million USD) of adopting different breeding schemes (with “n” year reduction) revealed by meta-analysis
| Sl.No | Crop/technology | Discount rate | Benefits in conventional method (reported) | Incremental benefits (imputed) | References | ||||
|---|---|---|---|---|---|---|---|---|---|
| Shuttle breeding (n = 2) | DH/RGA n = 3) | Speed breeding (n = 4) | SpeedGS (n = 5) | SpeedGS + SpeedDUS (n = 6) | |||||
| 1 | Wheat | 0.05 | 843.20 | 86.43 | 132.91 | 181.71 | 232.96 | 286.77 | Barkley and Reno (1997) |
| 2 | Bt Rice | 0.05 | 618.80 | 63.43 | 97.54 | 133.36 | 170.96 | 210.45 | Mamaril (2002) |
| 3 | Upland rice | 0.04 | 289.18 | 23.60 | 36.11 | 49.12 | 62.65 | 76.72 | Pardey (2004) |
| 4 | Golden Rice | 0.03 | 51.75 | 3.15 | 4.80 | 6.50 | 8.24 | 10.04 | Zimmermann and Qaim (2004) |
| 5 | Rice | 0.05 | 30323.97 | 3108.21 | 4779.82 | 6535.01 | 8377.95 | 10313.05 | Fan et al. (2005) |
| Rice | 0.05 | 69491.02 | 7122.83 | 10953.52 | 14975.75 | 19199.09 | 23633.59 | ||
| 6 | Rice | 0.05 | 50.55 | 5.18 | 7.97 | 10.89 | 13.97 | 17.19 | Singh et al. (2005) |
| Cold-tolerant rice | 0.05 | 91.82 | 9.41 | 14.47 | 19.79 | 25.37 | 31.23 | ||
| 7 | Golden Rice | 0.03 | 5087.50 | 309.83 | 471.75 | 638.53 | 810.31 | 987.24 | Stein et al. (2006) |
| 8 | Rice | 0.05 | 454.86 | 46.62 | 71.70 | 98.03 | 125.67 | 154.70 | Jaroensathapornkul (2007) |
| 9 | Drought tolerant rice | 0.05 | 0.41 | 0.04 | 0.06 | 0.09 | 0.11 | 0.14 | Gautam (2009) |
| 0.05 | 0.89 | 0.09 | 0.14 | 0.19 | 0.25 | 0.30 | |||
| 0.05 | 0.90 | 0.09 | 0.14 | 0.19 | 0.25 | 0.31 | |||
| 10 | Rice | 0.05 | 3001.40 | 307.64 | 473.10 | 646.82 | 829.23 | 1020.76 | Brennan and Malabayabas (2011) |
| 0.05 | 7894.51 | 809.19 | 1244.37 | 1701.32 | 2181.11 | 2684.89 | |||
| 0.05 | 11129.16 | 1140.74 | 1754.23 | 2398.40 | 3074.78 | 3784.98 | |||
| 11 | Rice | 0.05 | 859.31 | 88.08 | 135.45 | 185.19 | 237.41 | 292.25 | Raitzer et al. (2015) |
| 0.05 | 2132.43 | 218.57 | 336.12 | 459.55 | 589.15 | 725.23 | |||
| 0.05 | 10655.30 | 1092.17 | 1679.54 | 2296.28 | 2943.86 | 3623.82 | |||
| 12 | Wheat | 0.05 | 5.35 | 0.55 | 0.84 | 1.15 | 1.48 | 1.82 | Asadi et al. (2017) |
| 0.05 | 4.99 | 0.51 | 0.79 | 1.08 | 1.38 | 1.70 | |||
| 0.05 | 11.11 | 1.14 | 1.75 | 2.39 | 3.07 | 3.78 | |||
| 13 | Frost tolerant wheat | 0.05 | 3510.21 | 359.80 | 553.30 | 756.47 | 969.81 | 1193.81 | Mushtaq et al. (2017) |
Low-cost, field-based speed breeding system developed at PAU, Ludhiana, Punjab, India
PAU is the prestigious agricultural university in India, which is devoted to helping millions of farmers to grow nutritious crops through research and developments. Being the "Mother of the green revolution," PAU has been dedicatedly engaged to solve the challenges encountered in the farmers field since its establishment. However, fast-evolving diseases and pests (Ex., wheat rust and blast), fluctuating climates (heat and drought), and rising population force us to adopt innovative, science-based technologies to accelerate crop improvement. Having discussed above the studies that led to the development of the speed breeding technique as a potential tool to cut the generation time, we describe the research to implement this system to the needs of a wheat breeding program at PAU Ludhiana, India (Fig. 6).
Fig. 6.
Field-based, low-cost, speed breeding protocol developed by PAU, Ludhiana for generation advancement in wheat
Early sowing in germination trays (at the end of September) under controlled temperature (~ 16 °C) is the first step of our field-based speed breeding protocol. Field transplanting of 15 days old seedlings (at two-leaf stage) during mid-October demands special care for irrigation, weeding, pest and disease control as they grow in unfavourable seasons. Depending on the intensity and persistence of cold in the early months of winter, plants confront anthesis at the end of November or at the first week of December, which means they are ready for harvesting at 15–20 days after flowering (first generation). Following the artificial drying of spikes (at 37 °C for four days) and giving the cold shock (at 4 °C for two days), seeds will be ready for the next sowing (i.e., end of December or early January). December planting commences flowering in the hot months (i.e., March or April), necessitating frequent irrigation to alleviate the thermal effects. High temperature coupled with frequent irrigation hasten the crop growth and brings maturity by the last week of April to the first week of May (second generation), which is an appropriate time to sow wheat in Himalayan foothills in Keylong, Lahaul-Spiti, Himachal Pradesh (third generation).
The field-based speed breeding protocol helped to harvest three generations (two in Ludhiana and one in Keylong) of wheat in a year without demanding expensive growth chambers and greenhouses, which are mostly unaffordable in many developing and under-developed countries (O’Connor et al. 2013). In this protocol, segregating generations are consistently experiencing the challenges encountered in the field, which provides the additional benefit of not losing superior genotypes in the later generations (due to ‘field shock’) (Saxena et al. 2019). Since there are no space restrictions as in growth chambers, our protocol favours the simultaneous advancement of large numbers of crosses (about 30–40 crosses involving different combinations), which is impossible in speed breeding chambers. Further opportunities to improve the protocol to achieve an additional generation (i.e., 4 generations in a year) by implementing embryo rescue or immature seed harvest (Raz et al. 2001) provided with lighting facilities can be exploited. Plant breeders with limited resources can benefit by adopting our field-based, low-cost speed breeding technique to advance segregating generations and achieve a higher genetic gain.
Challenges and future prospects
Speed breeding techniques offer an excellent opportunity to accelerate the breeding process by reducing generation time. Besides their potential applications, the technique has certain limitations, as others do. These includes: (i) lack of trained breeders to handle the growth chambers; (ii) lack of financial and infrastructure facilities; (iii) inadequate electricity and water sources; (iv) requirement of genotype and species-specific protocols; (v) inadequate phenotyping; and (vi) probable loss of superior genotypes in growth chambers or/and while transferring in to the field.
Growth chambers supplied with electricity (to control temperature and lighting) make the speed breeding units unaffordable for many researchers. Greenhouses (Watson et al. 2018) or refrigerated shipping containers (Chiurugwi et al. 2019) fitted with energy-efficient lighting (low-cost LEDs) and temperature control systems (i.e., inverter-based air-conditioners) supplemented with solar power can be utilized in the less resourced countries. Regional research stations and agricultural universities must collaborate with national and international organizations to gather resources and build speed breeding infrastructures. In addition, governments need to regulate their policies and practices to reallocate funds to train the plant breeders and to establish speed breeding units.
Since each crop species and their genotypes are unique in growth and development, currently devised speed breeding protocols are not for all crop species. Researchers must devote resources towards establishing species or genotype-independent speed breeding protocols by optimizing growing conditions such as light, temperature, and growing media (Jähne et al. 2020).
Shorter generation time keeps the breeders busy in preparing seed material, sowing, and harvesting. In the meantime, they cannot perform for trait selection. Introducing high-throughput phenotyping specialist equipment’s, such as drones fitted with cameras and other sensors, into speed breeding units permit multi-trait phenotyping (speed-phenomics) (Hickey et al. 2019). In addition, speed breeding can also be integrated with genomic tools, such as MAS (Speed-MAS), MABC (Speed-MABC), MARS (Speed-MARS), and GS (Speed-GS). These genomic tools circumvent laborious phenotyping and facilitate multi-trait selection under growth chambers (Hickey et al. 2019).
Elevated temperatures and extended photoperiods may reduce the population size, resulting in the loss of valuable genetic variability required for later generation selection (Saxena et al. 2019). Field transfer of speed-bred advanced breeding lines may experience a sudden 'field shock,' which may further reduce the size of the breeding population or may eliminate the superior genotypes. It can be overcome by growing the generations alternatively in the field (in the main season) and speed breeding units (in the off-season). Though it may not fully exploit the speed breeding technique, but there will be the benefit for field adaptive traits.
Conclusion
Growing human population and changing climatic conditions demands for the accelerated crop improvement to meet the world's food requirement. Speed breeding accelerates the breeding process by reducing generation time and, thereby, helps to harvest multiple generations in a year. Integration of speed breeding with other breeding tools, such as high-throughput phenotyping, MAS, GS, MABC, MARS, mutation breeding, and genome editing may further accelerate the genetic gain per unit time. A field-based, low-cost, speed breeding protocol developed by PAU, Ludhiana, may help breeders to harvest at least three generations of wheat in a year without demanding the expensive greenhouses or growth chambers.
Acknowledgements
Special thanks to the Minority Welfare Department, Government of Karnataka, India for providing PhD fellowship to SG and to Head, Department of Plant Breeding and Genetics, Punjab Agricultural University, Ludhiana, (India) for providing necessary facilities.
Abbreviations
- RGA
Rapid generation advancement
- DH
Doubled haploid
- MAS
Marker-assisted selection
- MABC
Marker-assisted backcrossing
- MARS
Marker-assisted recurrent selection
- GS
Genomic selection
Author contributions
Conceptualization, SG and AS; writing-original draft preparation, SG, PK and MJT; writing-review and editing, SG, SS, and AS; supervision, AS. All authors have read and agreed to this version of the manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Availability of data and materials
Relevant data are included in this paper.
Code availability
Not applicable.
Declarations
Conflict of interest
The authors of this manuscript declare no conflicts of interest.
Ethical approval
The manuscript has not been submitted to more than one journal for simultaneous consideration.
Consent to participate
All authors have approved the article to be submitted to your journal.
Consent for publication
All authors have approved the article to be submitted to your journal for publication.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Alahmad S, Dinglasan E, Leung KM, et al. Speed breeding for multiple quantitative traits in durum wheat. Plant Methods. 2018 doi: 10.1186/S13007-018-0302-Y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ali B, Hayat S, Aiman Hasan S, Ahmad A. A comparative effect of IAA and 4-Cl-IAA on growth, nodulation and nitrogen fixation in Vigna radiata (L.) Wilczek. Acta Physiol Plant. 2008;30:35–41. doi: 10.1007/S11738-007-0088-4. [DOI] [Google Scholar]
- Arthur JM, Guthrie JD, Newell JM. Some effects of artificial climates on the growth and chemical composition of plants. Am J Bot. 1930;17:416. doi: 10.2307/2435930. [DOI] [Google Scholar]
- Asadi H, Zamanian G, Tash MNS, et al. An economic analysis of wheat breeding programs for some iranian irrigated bread wheat varieties. Agric Econ Rev. 2017;18(1):5–18. [Google Scholar]
- Baier KM, MaynardPowell CW. Early flowering in chestnut species induced under high dose light in growth chambers. J Am Chestnut Found. 2012;26:8–10. [Google Scholar]
- Bains NS, Singh J, Ravi GSS. Production of wheat haploids through embryo rescue from wheat × maize crosses. Curr Sci. 1995;69:621–623. [Google Scholar]
- Barkley AP, Reno (1997) Kansas wheat breeding: An economic analysis (No. 1839-2016-152179)
- Bhattarai SP, De La Pena RC, Midmore DJ, Palchamy K. In vitro culture of immature seed for rapid generation advancement in tomato. Euphytica. 2009;167:23–30. doi: 10.1007/S10681-008-9855-6. [DOI] [Google Scholar]
- Bielska S, Plewiński P, Kozak B, et al. Photoperiod and vernalization control of flowering-related genes: a case study of the narrow-leafed Lupin (Lupinus angustifolius L.) Front Plant Sci. 2020;11:1576. doi: 10.3389/FPLS.2020.572135/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borlaug NE. Sixty-two years of fighting hunger: personal recollections. Euphytica. 2007;157:287–297. doi: 10.1007/S10681-007-9480-9. [DOI] [Google Scholar]
- Brennan JP, Peter AE, Martin J. Returns to investment in new breeding technologies. Euphytica. 2007 doi: 10.1007/s10681-007-9378-6. [DOI] [Google Scholar]
- Brennan JP (1989) An analysis of the economic potential of some innovations in a wheat breeding programme. https://blog.jqueryui.com/2013/05/jquery-ui-1-10-3/
- Brennan JP, Malabayabas A (2011) International Rice Research Institute’s contribution to rice varietal yield improvement in South-East Asia. Canberra, ACT: Australian Centre for International Agricultural Research
- Breseghello F, Coelho ASG. Traditional and modern plant breeding methods with examples in Rice (Oryza sativa L.) J Agric Food Chem. 2013;61:8277–8286. doi: 10.1021/JF305531J. [DOI] [PubMed] [Google Scholar]
- Bugbee B, Koerner G. Yield comparisons and unique characteristics of the dwarf wheat cultivar ‘USU-Apogee’. Adv Sp Res. 1997;20:1891–1894. doi: 10.1016/S0273-1177(97)00856-9. [DOI] [PubMed] [Google Scholar]
- Bula RJ, Morrow RC, Tibbitts TW, et al. Light-emitting diodes as a radiation source for plants. HortScience. 1991;26:203–205. doi: 10.21273/HORTSCI.26.2.203. [DOI] [PubMed] [Google Scholar]
- Carandang FM, Shanmugasundaram S, Carpena AL. Rapid generation advancement in soybeans using immature seeds. Philipp J Crop Sci. 2006;31:53. [Google Scholar]
- Carre I, Coupland G, Putterill J (2008) Photoperiodic responses and the regulation of flowering. In: Hall AJW, McWatters HG (Eds.) Endogenous plant rhythms (Annual Plant Reviews). pp 167–190
- Cazzola F, Bermejo CJ, Guindon MF, Cointry E. Speed breeding in pea (Pisum sativum L.), an efficient and simple system to accelerate breeding programs. Euphytica. 2020;216(1):1–11. [Google Scholar]
- Cha J-K, O’Connor K, Alahmad S et al (2021) A new protocol for speed vernalisation of winter cereals. bioRxiv. 10.1101/2021.12.01.470717
- Chiurugwi T, Kemp S, Powell W, Hickey LT. Speed breeding orphan crops. Theor Appl Genet. 2019;132:607–616. doi: 10.1007/s00122-018-3202-7. [DOI] [PubMed] [Google Scholar]
- Collard BCY, Gregorio GB, Thomson MJ, et al. Transforming rice breeding: re-designing the irrigated breeding pipeline at the international Rice Research Institute (IRRI) Crop Breed Genet Genom. 2019 doi: 10.20900/cbgg20190008. [DOI] [Google Scholar]
- Dinglasan E, Godwin ID, Mortlock MY, Hickey LT. Resistance to yellow spot in wheat grown under accelerated growth conditions. Euphytica. 2016;209:693–707. doi: 10.1007/S10681-016-1660-Z. [DOI] [Google Scholar]
- Duan YX, Fan J, Guo WW. Regeneration and characterization of transgenic kumquat plants containing the Arabidopsis APETALA1 gene. Plant Cell, Tissue Organ Cult. 2010;100(3):273–281. doi: 10.1007/s11240-009-9646-3. [DOI] [Google Scholar]
- Fan S, Chan-Kang C, Qian K, Krishnaiah K. National and international agricultural research and rural poverty: the case of rice research in India and China. Agric Econ. 2005;33(s3):369–379. doi: 10.1111/j.1574-0864.2005.00077.x. [DOI] [Google Scholar]
- Flachowsky H, Le Roux PM, Peil A, et al. Application of a high-speed breeding technology to apple (Malus× domestica) based on transgenic early flowering plants and marker-assisted selection. New Phytol. 2011;192(2):364–377. doi: 10.1111/j.1469-8137.2011.03813.x. [DOI] [PubMed] [Google Scholar]
- Forster PB, Thomas TBW. Doubled haploids in genetics and plant breeding. Plant Breed Rev. 2005;25:57. [Google Scholar]
- Gaur P, Samineni S, Gowda C, Rao B. Rapid generation advancement in chickpea. J SAT Agric Res. 2007;3:1–3. [Google Scholar]
- Gautam A (2009) Impact evaluation of drought tolerant rice technologies through participatory approaches in eastern India. Rutgers University, Graduate School, New Brunswick
- Gerald NDLF, Ursula KF, Thomas L. Accelerating plant breeding. Trends Plant Sci. 2013;18:667–672. doi: 10.1016/j.tplants.2013.09.001. [DOI] [PubMed] [Google Scholar]
- Ghosh S, Watson A, Gonzalez-Navarro OE, et al. Speed breeding in growth chambers and glasshouses for crop breeding and model plant research. Nat Protoc. 2018;13:2944–2963. doi: 10.1038/s41596-018-0072-z. [DOI] [PubMed] [Google Scholar]
- Godfray HCJ, Beddington JR, Crute IR, et al. Food security: the challenge of feeding 9 billion people. Science. 2010;327:812–818. doi: 10.1126/SCIENCE.1185383. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Huitron V, León-Borges JA, Rodriguez-Mata AE, et al. Disease detection in tomato leaves via CNN with lightweight architectures implemented in Raspberry Pi 4. Comput Electron Agric. 2021;181:105951. doi: 10.1016/j.compag.2020.105951. [DOI] [Google Scholar]
- Goulden CH (1939) Problems in plant selection. Burn RC Proc 7th Int Genet Congr
- Gudi S, Atri C, Goyal A, et al. Physical mapping of introgressed chromosome fragment carrying the fertility restoring (Rfo) gene for Ogura CMS in Brassica juncea L. Czern & Coss. Theor Appl Genet. 2020;133(10):2949–2959. doi: 10.1007/s00122-020-03648-3. [DOI] [PubMed] [Google Scholar]
- Gudi S, Saini DK, Singh G, et al. Unravelling consensus genomic regions associated with quality traits in wheat using meta-analysis of quantitative trait loci. Planta. 2022;255(6):1–19. doi: 10.1007/s00425-022-03904-4. [DOI] [PubMed] [Google Scholar]
- Guo F, Huang BQ, Han Y, Zee SY. Fertilization in maize indeterminate gametophyte1 mutant. Protoplasma. 2004;223:111–120. doi: 10.1007/s00709-004-0045-7. [DOI] [PubMed] [Google Scholar]
- Hale AL, White PM, Webber CL, Todd JR. Effect of growing media and fertilization on sugarcane flowering under artificial photoperiod. PLoS ONE. 2017 doi: 10.1371/JOURNAL.PONE.0181639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison D, Silva M, Wu C, et al. Effect of light wavelength on soybean growth and development in a context of speed breeding. Crop Sci. 2021;61:917–928. doi: 10.1002/CSC2.20327. [DOI] [Google Scholar]
- Hatfield JL, Prueger JH. Temperature extremes: effect on plant growth and development. Weather Clim Extrem. 2015;10:4–10. doi: 10.1016/J.WACE.2015.08.001. [DOI] [Google Scholar]
- Hazel LN, Lush JL. The efficiency of three methods of selection. J Hered. 1942;33:393–399. doi: 10.1093/OXFORDJOURNALS.JHERED.A105102/2/33-11-393.PDF.GIF. [DOI] [Google Scholar]
- Helfer LR (2004) Intellectual property rights in plant varieties: international legal regimes and policy options for national governments. 104
- Hickey LT, Wilkinson PM, Knight CR, et al. Rapid phenotyping for adult-plant resistance to stripe rust in wheat. Plant Breed. 2012;131:54–61. doi: 10.1111/j.1439-0523.2011.01925.x. [DOI] [Google Scholar]
- Hickey LT, Germán SE, Pereyra SA, et al. Speed breeding for multiple disease resistance in barley. Euphytica. 2017 doi: 10.1007/s10681-016-1803-2. [DOI] [Google Scholar]
- Hickey LT, Hafeez AN, Robinson H, et al. Breeding crops to feed 10 billion. Nat Biotechnol. 2019;37:744–754. doi: 10.1038/s41587-019-0152-9. [DOI] [PubMed] [Google Scholar]
- Horizon HISPOB- High speed potato breeding: securing healthy food for the future. CORDIS EU research results. 2020 doi: 10.3030/666729. [DOI] [Google Scholar]
- Idrissi O (2020) Application of extended photoperiod in lentil: towards accelerated genetic gain in breeding for rapid improved variety development. Moroccan J Agric Sci 1(1)
- Izawa T, Oikawa T, Tokutomi S, et al. Phytochromes confer the photoperiodic control of flowering in rice (a short-day plant) Plant J. 2000;22:391–399. doi: 10.1046/J.1365-313X.2000.00753.X. [DOI] [PubMed] [Google Scholar]
- Jähne F, Hahn V, Würschum T, Leiser WL. Speed breeding short-day crops by LED-controlled light schemes. Theor Appl Genet. 2020;133:2335–2342. doi: 10.1007/s00122-020-03601-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamali SH, Cockram J, Hickey LT. Insights into deployment of DNA markers in plant variety protection and registration. Theor Appl Genet. 2019;132:1911–1929. doi: 10.1007/S00122-019-03348-7. [DOI] [PubMed] [Google Scholar]
- Jamali SH, Cockram J, Hickey LT. Is plant variety registration keeping pace with speed breeding techniques? Euphytica. 2020;216:1–13. doi: 10.1007/s10681-020-02666-y. [DOI] [Google Scholar]
- Janwan M, Sreewongchai T, Sripichitt P. Rice breeding for high yield by advanced single seed descent method of selection. J Plant Sci. 2013;8:24–30. doi: 10.3923/JPS.2013.24.30. [DOI] [Google Scholar]
- Jaroensathapornkul J. The Economic Impact of Public Rice Research in Thailand. Chulalongkorn J Econ. 2007;19(2):111–134. [Google Scholar]
- Jiang J, Birchler JA, Parrott WA, Dawe RK. A molecular view of plant centromeres. Trends Plant Sci. 2003;8(12):570–575. doi: 10.1016/j.tplants.2003.10.011. [DOI] [PubMed] [Google Scholar]
- Kalinowska K, Chamas S, Unkel K, et al. State-of-the-art and novel developments of in vivo haploid technologies. Theor Appl Genet. 2019;132:593–605. doi: 10.1007/s00122-018-3261-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashtiyani R, Ishii T, Niessen M, et al. Point mutation impairs centromeric CENH3 loading and induces haploid plants. Proc Natl Acad Sci. 2015;112(36):11211–11216. doi: 10.1073/pnas.1504333112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasha KJ, Kao KN. High frequency haploid production in barley (Hordeum vulgare L.) Nature. 1970;225:874–876. doi: 10.1038/225874a0. [DOI] [PubMed] [Google Scholar]
- Kelliher T, Starr D, Wang W, et al. Maternal haploids are preferentially induced by CENH3-tailswap transgenic complementation in maize. Front Plant Sci Plant Sci. 2016;7:414. doi: 10.3389/fpls.2016.00414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khalil S, El-saeid H, Shalaby M. The role of kinetin in flower abscission and yield of lentil plant. J Appl Sci Res. 2006;2:587. [Google Scholar]
- Koumura T. Breeding of new rice variety ‘Nipponbare’ (1) Agric Tech. 1972;27:112–116. [Google Scholar]
- Koumura T. Breeding of new rice variety “Nipponbare” (2) Agric Tech. 1972;27:159–161. [Google Scholar]
- Kranz E, Lorz H. In vitro fertilization with isolated, single gametes results in zygotic embryogenesis and fertile maize plants. Plant Cell. 1993;5:739–746. doi: 10.1105/TPC.5.7.739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuppu S, Tan EH, Nguyen H, et al. Point mutations in centromeric histone induce post-zygotic incompatibility and uniparental inheritance. PLoS Genet. 2015;11(9):e100. doi: 10.1371/journal.pgen.1005494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laurie DA, Bennett MD. The production of haploid wheat plants from wheat x maize crosses. Theor Appl Genet. 1988;76:393–397. doi: 10.1007/BF00265339. [DOI] [PubMed] [Google Scholar]
- Lenaerts B, de Mey Y, Demont M. Global impact of accelerated plant breeding: evidence from a meta-analysis on rice breeding. PLoS ONE. 2018 doi: 10.1371/journal.pone.0199016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Rasheed A, Hickey LT, He Z. Fast-forwarding genetic gain. Trends Plant Sci. 2018;23:184–186. doi: 10.1016/j.tplants.2018.01.007. [DOI] [PubMed] [Google Scholar]
- Mahadevaiah C, Appunu C, Aitken K, et al. Genomic selection in sugarcane: current status and future prospects. Front Plant Sci. 2021 doi: 10.3389/fpls.2021.708233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maheshwari S, Tan EH, West A, et al. Naturally occurring differences in CENH3 affect chromosome segregation in zygotic mitosis of hybrids. PLoS Genet. 2015;11(1):e100. doi: 10.1371/journal.pgen.1004970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mamaril CBC (2002) Transgenic pest resistant rice: an economic evaluation of an adoption impact pathway inthe philippines and Vietnam for bt rice (Doctoral dissertation, MS thesis, Virginia tech)
- Maruyama K (1989) Using rapid generation advance with single seed descent in rice breeding. https://blog.jqueryui.com/2013/05/jquery-ui-1-10-3/
- McCaw ME, Wallace JG, Albert PS, et al. Fast-flowering mini-maize: seed to seed in 60 days. Genetics. 2016;204:35–42. doi: 10.1534/genetics.116.191726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mobini SH, Warkentin TD. A simple and efficient method of in vivo rapid generation technology in pea (Pisum sativum L.) Vitr Cell Dev Biol Plant. 2016;52:530–536. doi: 10.1007/s11627-016-9772-7. [DOI] [Google Scholar]
- Mobini S, Khazaei H, Warkentin TD, Vandenberg A. Shortening the generation cycle in faba bean (Viciafaba) by application of cytokinin and cold stress to assist speed breeding. Plant Breeding. 2020;139(6):1181–1189. doi: 10.1111/pbr.12868. [DOI] [Google Scholar]
- Mobini SH, Lulsdorf M, Warkentin TD, Vandenberg A. Plant growth regulators improve in vitro flowering and rapid generation advancement in lentil and faba bean. Vitr Cell Dev Biol Plant. 2015;51:71–79. doi: 10.1007/s11627-014-9647-8. [DOI] [Google Scholar]
- Mushtaq S, An-Vo DA, Christopher M, et al. Economic assessment of wheat breeding options for potential improved levels of post head-emergence frost tolerance. Field Crops Res. 2017;213:75–88. doi: 10.1016/j.fcr.2017.07.021. [DOI] [Google Scholar]
- O’Connor DJ, Wright GC, Dieters MJ, et al. Development and application of speed breeding technologies in a commercial peanut breeding program. Peanut Sci. 2013;40:107–114. doi: 10.3146/ps12-12.1. [DOI] [Google Scholar]
- Ochatt SJ, Sangwan RS, Marget P, et al. New approaches towards the shortening of generation cycles for faster breeding of protein legumeshh. Plant Breed. 2002;121:436–440. doi: 10.1046/J.1439-0523.2002.746803.X. [DOI] [Google Scholar]
- Ohnishi T, Yoshino M, Yamakawa H, Kinoshita T. The biotron breeding system: a rapid and reliable procedure for genetic studies and breeding in rice. Plant Cell Physiol. 2011;52:1249–1257. doi: 10.1093/pcp/pcr066. [DOI] [PubMed] [Google Scholar]
- Ortiz R, Trethowan R, Ferrara GO, et al. High yield potential, shuttle breeding, genetic diversity, and a new international wheat improvement strategy. Euphytica. 2007;157:365–384. doi: 10.1007/s10681-007-9375-9. [DOI] [Google Scholar]
- Pardey PG (2004) Assessing and attributing the benefits from varietal improvement research in Brazil. Washington DC: International Food Policy Research Institute (Research report). Report No.: 136
- Pena L, Séguin A. Recent advances in the genetic transformation of trees. Trends Biotechnol. 2001;19(12):500–506. doi: 10.1016/S0167-7799(01)01815-7. [DOI] [PubMed] [Google Scholar]
- Penfield S. Seed dormancy and germination. Curr Biol. 2017;27:R874–R878. doi: 10.1016/J.CUB.2017.05.050. [DOI] [PubMed] [Google Scholar]
- Pennisi E. The blue revolution, drop by drop, gene by gene. Science. 2008;320:171–173. doi: 10.1126/science.320.5873.171. [DOI] [PubMed] [Google Scholar]
- Raitzer DA, Sparks AH, Huelgas Z et al (2015) Is rice improvement still making a difference? Assessing the economic, poverty and food security impacts of rice varieties released from 1989 to 2009 in Bangladesh, Indonesia and the Philippines. Standing Panel on Impact Assessment (SPIA), CGIAR Independent Science and Partnership Council (ISPC)
- Rana MM, Takamatsu T, Baslam M, et al. Salt tolerance improvement in rice through efficient SNP marker-assisted selection coupled with speed-breeding. Int J Mol Sci. 2019 doi: 10.3390/ijms20102585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ravi M, Chan SWL. Haploid plants produced by centromere-mediated genome elimination. Nature. 2010;464:615–618. doi: 10.1038/NATURE08842. [DOI] [PubMed] [Google Scholar]
- Ray DK, Ramankutty N, Mueller ND, et al. (2012) Recent patterns of crop yield growth and stagnation. Nat Commun. 2012;31(3):1–7. doi: 10.1038/ncomms2296. [DOI] [PubMed] [Google Scholar]
- Raz V, Bergervoet JHW, Koornneef M. Sequential steps for developmental arrest in Arabidopsis seeds. Development. 2001;128:243–252. doi: 10.1242/DEV.128.2.243. [DOI] [PubMed] [Google Scholar]
- Riaz A, Hickey LT. Rapid phenotyping adult plant resistance to stem rust in wheat grown under controlled conditions. Methods Mol Biol. 2017;1659:183–196. doi: 10.1007/978-1-4939-7249-4_16. [DOI] [PubMed] [Google Scholar]
- Riaz A, Periyannan S, Aitken E, Hickey L. A rapid phenotyping method for adult plant resistance to leaf rust in wheat. Plant Methods. 2016;12:1–10. doi: 10.1186/s13007-016-0117-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riaz A, Athiyannan N, Periyannan SK, et al. Unlocking new alleles for leaf rust resistance in the Vavilov wheat collection. Theor Appl Genet. 2018;131:127–144. doi: 10.1007/S00122-017-2990-5. [DOI] [PubMed] [Google Scholar]
- Richard C, Christopher J, Chenu K, et al. Selection in early generations to shift allele frequency for seminal root angle in wheat. Plant Genome. 2018;11:170071. doi: 10.3835/PLANTGENOME2017.08.0071. [DOI] [PubMed] [Google Scholar]
- Rizal G, Karki S, Alcasid M, et al. Shortening the breeding cycle of sorghum, a model crop for research. Crop Sci. 2014;54:520–529. doi: 10.2135/cropsci2013.07.0471. [DOI] [Google Scholar]
- Robin S, Pathan MS, Courtois B, et al. Mapping osmotic adjustment in an advanced back-cross inbred population of rice. Theor Appl Genet. 2003;107:1288–1296. doi: 10.1007/S00122-003-1360-7. [DOI] [PubMed] [Google Scholar]
- Saeid HM, Abouziena HF, AbdAlla MSA. Effect of some bioregulators on white lupine (lupinus termis) seed yield and its components and on endogenous hormones content in seeds. Electron J Polish Agric Univ. 2011;14:2. [Google Scholar]
- Santra M, Wang H, Seifert S, Haley S. Doubled haploid laboratory protocol for wheat using wheat–Maize wide hybridization. Methods Mol Biol. 2017;1679:235–249. doi: 10.1007/978-1-4939-7337-8_14. [DOI] [PubMed] [Google Scholar]
- Saxena KB, Saxena RK, Hickey LT, Varshney RK. Can a speed breeding approach accelerate genetic gain in pigeonpea? Euphytica. 2019;215:1–7. doi: 10.1007/s10681-019-2520-4. [DOI] [Google Scholar]
- Shimelis H, Laing M. Timelines in conventional crop improvement: pre-breeding and breeding procedures. Aust J Crop Sci. 2012;6:1542–1549. [Google Scholar]
- Siemens CW. III. On the influence of electric light upon vegetation, and on certain physical principles involved. Proc R Soc London. 1880;30:210–219. doi: 10.1098/RSPL.1879.0108. [DOI] [Google Scholar]
- Singh G, Kaur N, Khanna R, et al. 2022) 2Gs and plant architecture: breaking grain yield ceiling through breeding approaches for next wave of revolution in rice (Oryza sativa L.) Crit Rev Biotechnol. 2022 doi: 10.1080/07388551.2022.2112648. [DOI] [PubMed] [Google Scholar]
- Singh R, Brennan JP, Farrell T et al (2005) Economic analysis of improving cold tolerance in rice in Australia. University of Sydney
- Srivastava P, Bains NS. Accelerated wheat breeding: doubled haploids and rapid generation advance. Biotechnol Crop Improv. 2018;1:437–461. doi: 10.1007/978-3-319-78283-6_13. [DOI] [Google Scholar]
- Stein AJ, Sachdev HP, Qaim M. Potential impact and cost-effectiveness of Golden Rice. Nat Biotech. 2006;24:1200–1201. doi: 10.1038/nbt1006-1200b. [DOI] [PubMed] [Google Scholar]
- Stetter MG, Zeitler L, Steinhaus A, et al. Crossing methods and cultivation conditions for rapid production of segregating populations in three grain amaranth species. Front Plant Sci. 2016 doi: 10.3389/fpls.2016.00816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stutte GW. Commercial transition to LEDs: a pathway to high-value products. HortScience. 2015;50:1297–1300. doi: 10.21273/HORTSCI.50.9.1297. [DOI] [Google Scholar]
- Tanaka J, Hayashi T, Iwata H. A practical, rapid generation-advancement system for rice breeding using simplified biotron breeding system. Breed Sci. 2016;66:542–551. doi: 10.1270/jsbbs.15038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanin MJ, Saini DK, Sandhu KS, et al. Consensus genomic regions associated with multiple abiotic stress tolerance in wheat and implications for wheat breeding. Sci Rep. 2022;12(1):1–17. doi: 10.1038/s41598-022-18149-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanio M, Kato K, Ishikawa N, et al. Effect of shuttle breeding with rapid generation advancement on heading traits of Japanese wheat. Breed Sci. 2006;56:311–320. doi: 10.1270/jsbbs.56.311. [DOI] [Google Scholar]
- Trevaskis B, Tadege M, Hemming MN, et al. Short vegetative phase-like MADS-Box genes inhibit floral meristem identity in barley. Plant Physiol. 2007;143:225–235. doi: 10.1104/PP.106.090860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Nocker S, Gardiner SE. Breeding better cultivars, faster: applications of new technologies for the rapid deployment of superior horticultural tree crops. Hortic Res. 2014;1:14022. doi: 10.1038/hortres.2014.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voss-Fels KP, Herzog E, Dreisigacker S, et al. “SpeedGS” to accelerate genetic gain in spring wheat. Amsterdam: Elsevier; 2019. [Google Scholar]
- Wada KC, Takeno K. Stress-induced flowering. Plant Signal Behav. 2010;5:944–947. doi: 10.4161/PSB.5.8.11826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Wang Y, Zhang G, Ma Z. An integrated breeding technology for accelerating generation advancement and trait introgression in cotton. Plant Breed. 2011;130:569–573. doi: 10.1111/j.1439-0523.2011.01868.x. [DOI] [Google Scholar]
- Watson A, Ghosh S, Williams MJ, et al. Speed breeding is a powerful tool to accelerate crop research and breeding. Nat Plants. 2018;4:23–29. doi: 10.1038/s41477-017-0083-8. [DOI] [PubMed] [Google Scholar]
- Xu Y, Liu X, Fu J, et al. Enhancing genetic gain through genomic selection: from livestock to plants. Plant Commun. 2020;1:100005. doi: 10.1016/J.XPLC.2019.100005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang C, Russell J, Ramsay L, et al (2021) Overcoming barriers to the registration of new plant varieties under the DUS system. https://nature.com/ [DOI] [PMC free article] [PubMed]
- Zimmermann R, Qaim M. Potential health benefits of Golden Rice: a Philippine case study. Food Policy. 2004;29(2):147–168. doi: 10.1016/j.foodpol.2004.03.001. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Relevant data are included in this paper.
Not applicable.