Case Presentation
An acute exacerbation of interstitial lung disease (ILD) is an acute deterioration that can occur at any time and is associated with significant morbidity and mortality rates. We herein report three patients with ILD who experienced acute respiratory failure after SARS-CoV-2 messenger RNA vaccination. All the patients were male; the mean age was 77 years. They had a smoking history that ranged from 10 to 30 pack-years. Duration from the vaccination to the onset of respiratory failure was 1 day in two patients and 9 days in one patient. In an autopsied case, lung pathologic evidence indicated diffuse alveolar damage superimposed on usual interstitial pneumonia. In the other two cases, CT scans showed diffuse ground-glass opacities and subpleural reticulation, which suggests acute exacerbation of ILD. Two patients were treated successfully with high-dose methylprednisolone. Although benefits of vaccination outweigh the risks associated with uncommon adverse events, patients with chronic lung diseases should be observed carefully after SARS-CoV-2 vaccination.
COVID-19 remains a global health concern, and several SARS-CoV-2 vaccines have been developed for the prevention of infection or aggravation and lowering mortality rates.1 However, some concerns about adverse effects that are related to SARS-CoV-2 vaccination have been raised. Recently, various adverse effects have been reported, but there are little data on adverse effects on the lungs, such as exacerbation of preexisting lung disease. In this report, we describe three cases of acute respiratory failure subsequent to SARS-CoV-2 messenger RNA (mRNA) vaccination, which include an autopsied case.
Case Reports
Case 1
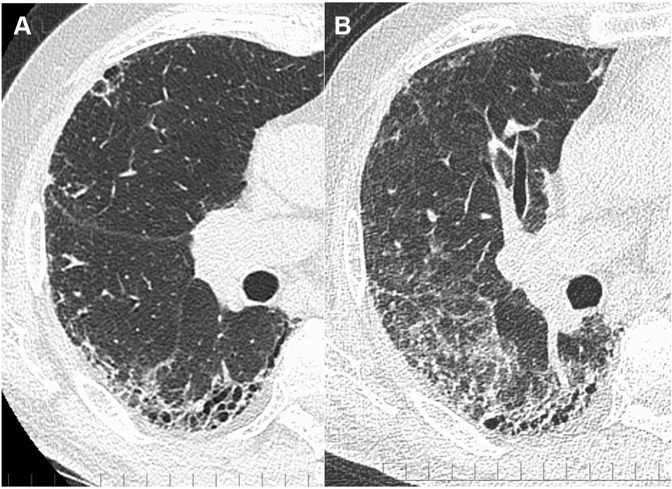
A 67-year-old man who had a previous diagnosis of metastatic colon cancer and who received chemotherapy with S-1 plus oxaliplatin experienced the development of interstitial lung disease (ILD) in July 2021. He had a former smoking history of 30 pack-years. Because his ILD was mild and nonprogressive, no corticosteroids or antifibrotic agents were introduced. In August 2021, he received his first dose of BNT162b2 vaccine (Comirnaty; BioNTech/Pfizer). From the next day, he had fever and dyspnea on exertion. Because of the worsening symptoms, he visited the ED and was admitted to our hospital 3 days after the vaccination. His chest high-resolution CT (HRCT) scan revealed diffuse ground-glass opacities (GGO) superimposed on preexisting subpleural reticulation and traction bronchiectasis (Fig 1 ).
Figure 1.
A-C, High resolution CT scan findings of Case 1. High-resolution CT scan taken B, 1 month before the exacerbation showed subpleural reticulation and traction bronchiectasis A, that had not been observed 15 months before. On admission, C, high-resolution CT scan revealed diffuse ground-glass opacities superimposed on the preexisting interstitial changes.
On admission, his vital signs were a temperature of 38.0°C, pulse rate of 150 beats/min, respiratory rate of 30 breaths/min, and peripheral oxygen saturation of 52% in ambient air. Physical examination revealed bilateral fine crackles and no heart murmur, leg edema, or clubbed fingers. Arterial blood gas analysis on admission revealed hypoxemia and respiratory alkalosis (pH 7.493; Paco 2, 15.8 mm Hg; Pao 2, 51.5 mm Hg; HCO3 –, 9.2 mM) with 8 L/min of supplemental oxygen via face mask. Blood tests revealed elevated levels of C-reactive protein (CRP) (5.17 mg/dL), lactate dehydrogenase (LDH) (607 units/L), Krebs von den Lungen-6 (506 units/mL), and procalcitonin (0.25 ng/mL). Before the vaccination, the levels of CRP (0.32 mg/dL) and LDH (271 units/mL) were within normal range. Antinuclear antibody and other autoantibodies were negative. WBC count was 12,550/μL, and the differential count was neutrophils 78.5%, lymphocytes 12.1%, and eosinophils 0.3%. SARS-CoV-2 antigen test and polymerase chain reaction were both negative. Cultures of his sputum and tracheal secretions were also negative. During the transfer to the ICU, he suddenly had cardiopulmonary arrest and died.
The autopsy results revealed the following data: The lungs had subpleural dense fibrosis with alternating areas of normal lung, which suggests temporal heterogeneity of fibrosis. At the boundary between normal lung and dense fibrosis, fibroblastic foci were observed, which suggests usual interstitial pneumonia (UIP) (Fig 2A). The lung pathologic examination also showed diffuse alveolar damage that was characterized by infiltration of inflammatory cells and hyaline membranes with protein-rich edema fluid (Fig 2B). These findings were consistent with the clinical diagnosis of acute exacerbation of UIP. Although atherosclerosis was observed, there was no pathologic changes in the cardiovascular system that could be responsible for sudden death.
Figure 2.
A and B, Autopsy findings from Case 1. A, The lungs had subpleural dense fibrosis with alternating areas of normal lung suggest temporal heterogeneity of fibrosis. As shown in the inset, scattered fibroblastic foci were also observed, which was suggestive of usual interstitial pneumonia. B, The lung pathology report also showed diffuse alveolar damage that was characterized by infiltration of inflammatory cells and hyaline membranes with protein-rich edema fluid.
Case 2
An 82-year-old man who had received his third dose of BNT162b2 vaccine 16 days before was referred to our hospital because of severe respiratory failure. He presented with fever, nonproductive cough, and progressive dyspnea over a week. He had a former smoking history of 10 pack-years. When he visited the ED of a local hospital, his peripheral oxygen saturation was as low as 75%, and the chest roentgenogram showed bilateral pulmonary infiltrates. On admission, his vital signs were a body temperature of 37.5°C, pulse rate of 108 beats/min, respiratory rate of 30 breaths/min, and peripheral oxygen saturation of 87% with 10 L/min of supplemental oxygen through a rebreather mask. Physical examination revealed fine crackles on the lungs and clubbed fingers. Arterial blood gas analysis on admission revealed hypoxemia and respiratory alkalosis (pH 7.456; Paco 2, 31.6 mm Hg; Pao 2, 53.2 mm Hg; HCO3 –, 21.9 mM) with 10 L/min of supplemental oxygen via face mask. Laboratory findings revealed the following data: WBCs 15,080/μL (neutrophils, 81.2%; lymphocytes, 11.3%); CRP, 20.66 mg/dL; LDH, 669 units/L; Krebs von den Lungen-6, 1,069 units/mL, and procalcitonin, 0.25 ng/mL. None of autoantibodies were positive. SARS-CoV-2 polymerase chain reaction was negative.
Because HRCT on admission showed diffuse GGO with subpleural reticulation and traction bronchiectasis (Fig 3 ), we considered acute exacerbation of preexisting interstitial pneumonia and introduced high-dose steroid therapy with 500 mg of methylprednisolone for 3 days followed by 1 mg/kg of prednisolone that was slowly tapered. After 5 days of mechanical ventilation, the patient was extubated and received high-flow nasal cannula oxygen therapy. HRCT revealed resolution of GGO, but subpleural reticulation and traction bronchiectasis remained. The reticular shadow persisted on the ventral subpleural region where minimal GGO were observed. Considering this HRCT findings and the presence of clubbing, it was suggested that ILD might have been present before the vaccination. On the day 29 from admission, he was discharged home while receiving 25 mg of oral prednisolone and supplemental oxygen.
Figure 3.
A-C, High-resolution CT scan findings from Case 2. A and B, High-resolution CT scan on admission show diffuse ground-glass opacities with subpleural reticulation and traction bronchiectasis. C, After high-dose steroid therapy, high-resolution CT scan on the day 27 from admission revealed resolution of ground-glass opacities; however, subpleural reticulation and traction bronchiectasis remained.
Case 3
An 81-year-old man, who had been diagnosed with interstitial pneumonia 13 months before received his third dose of BNT162b2 vaccine. He had a former smoking history of 20 pack-years. HRCT revealed honeycombing with subpleural reticulation predominantly in the lung bases, which indicated a clinical diagnosis of idiopathic pulmonary fibrosis (IPF). Because a significant decline in vital capacity was observed, antifibrotic treatment with 300 mg/d of nintedanib had been introduced 7 months before. On the next day of the vaccination, he had fever and dyspnea and was transferred to our hospital. His HRCT revealed wide-spread GGO, which was superimposed on preexisting honeycombing and subpleural (Fig 4 ).
Figure 4.
A and B, High-resolution CT scan findings from Case 3. A, High-resolution CT scan 4 months before the admission shows honeycombing with subpleural reticulation predominantly in the lung bases, which indicates a clinical diagnosis of idiopathic pulmonary fibrosis. B, On admission, his high-resolution CT scan showed wide-spread ground-glass opacity, which was superimposed on preexisting honeycombing and subpleural reticulation.
On admission, his vital signs were a temperature of 38.9°C, pulse rate of 104 beats/min, respiratory rate of 33 breaths/min, and peripheral oxygen saturation of 80% in ambient air. Laboratory findings revealed the following data: WBCs 9,010/μL (neutrophils, 81.7%; lymphocytes, 9.7%); CRP, 11.41 mg/dL; LDH, 465 U/L; Krebs von den Lungen-6, 1,579 units/mL, and procalcitonin, 0.03 ng/mL. No autoantibodies were detected. SARS-CoV-2 polymerase chain reaction was negative. Because we considered acute exacerbation of IPF (AE-IPF), we introduced high-dose steroid therapy with 1 g of methylprednisolone for 3 days followed by 1 mg/kg of prednisolone that was slowly tapered. His respiratory status improved gradually.
Discussion
We experienced three cases of acute respiratory failure subsequent to SARS-CoV-2 mRNA vaccination, including an autopsied case. Two cases had received a diagnosis of ILD, and one case was considered to have preexisting ILD based on the HRCT finding. We speculated that acute respiratory failure in these patients might be associated with SARS-CoV-2 vaccination.
AE-IPF has been defined as an acute, clinically significant deterioration that develops within <1 month without obvious clinical cause such as fluid overload, left heart failure, or pulmonary embolism.2 Diffuse alveolar damage is the histopathologic feature of AE-IPF, which is characterized by diffuse, bilateral GGO on HRCT. A growing body of evidence now focuses on acute exacerbations of ILD (AE-ILD) other than IPF.3 Based on a shared pathophysiologic evidence, it is accepted generally that AE-ILD can affect various types of ILD but apparently occurs more frequently in patients with an underlying UIP pattern.3 , 4 A recent meta-analysis showed that the UIP pattern was observed predominantly in elderly men with a history of smoking, whereas nonspecific interstitial pneumonia occurred in a younger patient population.5 All the patients were elderly men with a history of smoking. In two cases, the UIP pattern of ILD was indicated pathologically in Case 1 and radiologically in Case 3. We considered that the UIP pattern might be a risk factor of AE-ILD subsequent to SARS-CoV-2 vaccination.
The cause and pathogenesis of AE-ILD remains unclear, but there are distinct risk factors such as infection, mechanical stress, aspiration, and drug toxicity that may trigger the development of acute exacerbation.2 There have been some reports of AE-IPF after influenza A vaccination,6 which suggests that vaccinations may serve as a trigger of acute exacerbation. According to the World Health Organization global pharmacovigilance database, 678 cases of suspected ILD, 159 cases of suspected organizing pneumonia, 12 cases of acute lung injury, and two cases of acute interstitial pneumonitis after BNT162b2 vaccine were reported, whereas no case of AE-ILD was included.7 In addition, a clinical trial of BNT162b2 vaccine showed no case of AE-ILD.1 To the best of our knowledge, four cases of AE-ILD subsequent to SARS-CoV-2 vaccination have been reported.8, 9, 10 All the patients were men who had a smoking history and in whom AE-ILD developed within 9 days after the vaccination, which coincides with the cases described in this report. AE-ILD may develop over weeks.2 Development of respiratory failure over the course of several days may be a hallmark of the postvaccination exacerbation of ILD.
Amiya et al10 reported two cases of AE-ILD after SARS-CoV-2 vaccination. In their report, previous CT scan images showed only slight fibrosis of the lung base in one case and no significant abnormalities in the other. The two cases might not have been exacerbation of chronic fibrotic ILD, which is different from these patients.
In general, AE-ILD has a poor prognosis and is associated with a high mortality rate within 6 to 12 months.2 Although there has been no sufficient evidence-based data, in clinical practice, AE-ILD is often treated with a high-dose corticosteroid therapy, which was administered to each of the four patients previously reported.8, 9, 10 Although the mechanism of mRNA vaccine-induced exacerbation of ILD remains unclear, recent reports suggest that an innate inflammatory response could be induced by the mRNA or by SARS-CoV-2 spike protein.11 , 12 It has also been reported that cross-reactivity of spike proteins with lung surfactants and related proteins may induce pulmonary inflammation.13 Two of the patients received high-dose corticosteroid therapy, and clinical improvement was observed in both, which indicated that the excessive activation of immune system might contribute to the exacerbation.
In two patients, we could not perform lung pathologic testing or BAL, which made the exclusion of various causes of acute respiratory failure such as viral or bacterial infection difficult. In Case 2, the patient became symptomatic 9 days after receiving the vaccination. In addition, the possibility of drug-induced ILD because of oxaliplatin should be considered in Case 1. Drug-induced ILD can also develop acute exacerbation.14 Because CRP and LDH levels were not elevated even after the last dose of oxaliplatin, at least it is unlikely that active oxaliplatin-induced pneumonia was underlying. Taken together, although the vaccination might result in AE-ILD, it cannot be denied that infection or other factors may have been involved.
In conclusion, we experienced three cases of acute respiratory failure in patients with ILD, which could be associated with SARS-CoV-2 mRNA vaccination. Because ILD is considered to be a risk factor for severe COVID-19,15 the benefits of vaccination should outweigh the risks associated with uncommon adverse events. However, clinicians should note the risk and carefully observe patients with ILD after SARS-CoV-2 mRNA vaccination.
Acknowledgments
Financial/nonfinancial disclosures: None declared.
Other contributions:CHEST worked with the authors to ensure that the Journal policies on patient consent to report information were met.
References
- 1.Polack F.P., Thomas S.J., Kitchin N., et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383(27):2603–2615. doi: 10.1056/NEJMoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Collard H.R., Ryerson C.J., Corte T.J., et al. Acute exacerbation of idiopathic pulmonary fibrosis: an international working group report. Am J Respir Crit Care Med. 2016;194(3):265–275. doi: 10.1164/rccm.201604-0801CI. [DOI] [PubMed] [Google Scholar]
- 3.Park I.N., Kim D.S., Shim T.S., et al. Acute exacerbation of interstitial pneumonia other than idiopathic pulmonary fibrosis. Chest. 2007;132(1):214–220. doi: 10.1378/chest.07-0323. [DOI] [PubMed] [Google Scholar]
- 4.Miyazaki Y., Tateishi T., Akashi T., et al. Clinical predictors and histologic appearance of acute exacerbations in chronic hypersensitivity pneumonitis. Chest. 2008;134(6):1265–1270. doi: 10.1378/chest.08-0866. [DOI] [PubMed] [Google Scholar]
- 5.Ebner L., Christodoulidis S., Stathopoulou T., et al. Meta-analysis of the radiological and clinical features of usual interstitial pneumonia (UIP) and nonspecific interstitial pneumonia (NSIP) PLoS One. 2020;15(1) doi: 10.1371/journal.pone.0226084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Umeda Y., Morikawa M., Anzai M., et al. Acute exacerbation of idiopathic pulmonary fibrosis after pandemic influenza A (H1N1) vaccination. Intern Med. 2010;49(21):2333–2336. doi: 10.2169/internalmedicine.49.3890. [DOI] [PubMed] [Google Scholar]
- 7.VigiAccess. WHO Collaborating Centre for International Drug Monitoring. http://www.vigiaccess.org
- 8.Bando T., Takei R., Mutoh Y., et al. Acute exacerbation of idiopathic pulmonary fibrosis after SARS-CoV-2 vaccination. Eur Respir J. 2022;59(3) doi: 10.1183/13993003.02806-2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ghincea A., Ryu C., Herzog E.L. An acute exacerbation of idiopathic pulmonary fibrosis after BNT162b2 mRNA COVID-19 vaccination: a case report. Chest. 2022;161(2):e71–e73. doi: 10.1016/j.chest.2021.07.2160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Amiya S., Fujimoto J., Matsumoto K., et al. Case report: acute exacerbation of interstitial pneumonia related to messenger RNA COVID-19 vaccination. Int J Infect Dis. 2022;116:255–257. doi: 10.1016/j.ijid.2022.01.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lu Q., Liu J., Zhao S., et al. SARS-CoV- 2 exacerbates proinflammatory responses in myeloid cells through C-type lectin receptors and Tweety family member 2. Immunity. 2021;54(6):1304–1319. doi: 10.1016/j.immuni.2021.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhao Y., Kuang M., Li J., et al. SARS-CoV-2 spike protein interacts with and activates TLR41. Cell Res. 2021;31(7):818–820. doi: 10.1038/s41422-021-00495-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kanduc D., Shoenfeld Y. On the molecular determinants of the SARS-CoV-2 attack. Clin Immunol. 2020;215:108426. doi: 10.1016/j.clim.2020.108426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Usui Y., Kaga A., Sakai F., et al. A cohort study of mortality predictors in patients with acute exacerbation of chronic fibrosing interstitial pneumonia. BMJ Open. 2013;3(7) doi: 10.1136/bmjopen-2013-002971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Drake T.M., Docherty A.B., Harrison E.M., et al. Outcome of hospitalization for COVID-19 in patients with interstitial lung disease: an international multicenter study. Am J Respir Crit Care Med. 2020;202(12):1656–1665. doi: 10.1164/rccm.202007-2794OC. [DOI] [PMC free article] [PubMed] [Google Scholar]