Abstract
Background:
Sepsis-associated acute kidney injury (SA-AKI) is associated with significant morbidity and mortality. Immune dysregulation is a hallmark of sepsis, with important contributions to organ dysfunction including injury and repair mechanisms in AKI. Macrolide antibiotics, such as azithromycin, have previously demonstrated in pre-clinical models a myriad of immunomodulatory effects that may benefit critically ill patients with SA-AKI. The aim of this study was to determine if early receipt of azithromycin in SA-AKI is associated with a reduction in major adverse kidney events (MAKE) at hospital discharge.
Methods:
This was a single center, retrospective cohort study of critically ill adult patients with SA-AKI. Early exposure to azithromycin was defined as receipt of one or more doses within 48 hours of a hospital admission with SA-AKI. The primary outcome of MAKE assessed at hospital discharge was the composite of death, requirement for kidney replacement therapy, or a decline in estimated glomerular filtration rate of 25% or more. Multivariable logistic regression was used to account for potential confounders in the assessment.
Results:
Of 737 included patients with SA-AKI, 152 (20.6%) received azithromycin. Patients that received early azithromycin were less likely to experience MAKE at hospital discharge when compared to those patients not receiving azithromycin: 38.8% versus 48.4% (p=0.035). In multivariable logistic regression, receipt of azithromycin was independently associated with a decreased odds of MAKE at hospital discharge (aOR 0.62, 95% CI 0.41–0.93).
Conclusions:
Early exposure to azithromycin in SA-AKI is independently associated with lower odds of MAKE at hospital discharge.
Keywords: acute kidney injury, major adverse kidney event, azithromycin, macrolide, sepsis, critical care
Introduction
Sepsis is a leading contributor to acute kidney injury (AKI) in hospitalized patients, identified as a risk factor in nearly 50% of AKI cases (1–4). Patients with sepsis-associated AKI (SA-AKI) often present with more severe AKI and are at higher risk of mortality compared to other etiologies of AKI (1). For those surviving hospitalization, SA-AKI is also associated with significant short- and long-term impairments in kidney function (5, 6).
SA-AKI is multifactorial, with potential intra-renal hemodynamic, metabolic, and immune pathologies influencing kidney injury and repair (7–10). Macrolide antibiotics, including azithromycin, have demonstrated a myriad of immunomodulatory effects in pre-clinical models that may prove beneficial for syndromes of immune dysregulation in critically ill patients such as SA-AKI (11). Azithromycin is a Food and Drug Administration approved antibiotic and widely used clinically in various respiratory conditions that trigger an inflammatory response including cystic fibrosis, community-acquired pneumonia, and chronic obstructive pulmonary disease (11).
Given its significant morbidity and lack of therapeutic agents for SA-AKI, we sought to evaluate the effect of early azithromycin exposure on kidney outcomes and mortality in a cohort of critically ill patients with SA-AKI.
Materials and Methods
Study Design
This was a retrospective, single center, cohort study conducted at a tertiary care, academic medical center. Patients admitted from January 1, 2013 to July 31, 2020 were screened for study eligibility and included if they were 18 years of age or older, initially admitted to the medical intensive care unit (ICU), and diagnosed with sepsis or septic shock and AKI at the time of ICU admission. Sepsis and septic shock diagnoses were identified based on International Classification of Diseases (ICD)-9 and ICD-10 codes. AKI was defined as Kidney Disease: Improving Global Outcomes (KDIGO) stage 1 or higher based on the serum creatinine component (12). Exclusion criteria consisted of the following: outside hospital transfer, death within 24 hours of admission, end-stage kidney disease, or history of kidney transplantation. For patients with multiple ICU admissions during the study period, only the first ICU admission was evaluated.
Demographic data and baseline characteristics were collected on ICU admission including severity of illness indicators such as the Sepsis-related Organ Failure Assessment (SOFA) score (13). Baseline comorbidities were identified using ICD-9 and ICD-10 codes. The presence of septic shock was classified based on the need for vasopressor support within the first 48 hours of admission. To assess baseline kidney function, the most recent serum creatinine within six months prior to admission was collected when available. Estimated glomerular filtration rate (eGFR) was calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation (14).
Exposure to azithromycin was defined as one or more doses of oral or intravenous azithromycin within 48 hours of ICU admission. In order to distinguish if sepsis was secondary to a suspected community or hospital source, antibiotic administration within the first 48 hours of ICU admission was assessed. Patients receiving anti-Pseudomonal antibiotic coverage (cefepime, piperacillin/tazobactam, or meropenem) for more than one day were classified as a suspected hospital source and all other patients were classified as a suspected community source. The study was approved by the University of Kentucky Institutional Review Board (#61174) with a waiver of informed consent due to the retrospective nature.
Study Outcomes
The primary outcome was Major Adverse Kidney Events (MAKE) at hospital discharge, a composite of death, need for kidney replacement therapy (KRT), or decrease in eGFR ≥ 25% from baseline (15). Secondary outcomes included KRT or death within 1 week of admission, development of acute kidney disease (AKD), serum creatinine and eGFR at discharge in survivors not requiring KRT, ICU length of stay, and hospital length of stay. AKD was defined per the Acute Disease Quality Initiative consensus definition (16).
Statistical Analysis
Continuous data are presented as medians with interquartile range and compared using the Wilcoxon rank-sum test. Categorical data are presented as counts with associated percentages and compared using a chi-square test. For patients who did not have a baseline serum creatinine available prior to admission, multiple imputations were conducted using SAS 9.4 Proc MI (SAS Institute Inc), fully conditional specification, with 50 imputed data sets. Variables included for imputation were age, sex, race, diabetes and hypertension as they have been validated in previous studies of serum creatinine imputation (17, 18). The average creatinine value from the 50 imputations was used to classify screened patients with sepsis or septic shock as having AKI or not.
Multivariable logistic regression was used to model MAKE at hospital discharge. Variables for the model were defined a priori and hypothesized by the investigators to influence both the receipt of azithromycin and the MAKE outcome. These variables included: age, sex, baseline eGFR, diabetes, hypertension, heart failure, liver disease, chronic kidney disease, KDIGO AKI stage 1–3 on admission, serum lactate on admission, admission SOFA, and anti-Pseudomonal antibiotic coverage as a surrogate for suspect hospital vs. community-acquired infection. Four separate sensitivity analyses of this model were conducted: 1) examination of an alternative MAKE definition using a 50% reduction in eGFR rather than a 25% reduction; 2) examination of patients with a measured (non-imputed) baseline serum creatinine; 3) examination of a composite outcome of death or KRT (removing eGFR component from MAKE); and 4) examination of patients with severe SA-AKI limited to those with KDIGO stage 2 or 3 AKI. An E-value was calculated to determine the minimum strength of association required by an unmeasured confounder to explain away the relationship observed between azithromycin and MAKE (19). A p-value <0.05 was considered statistically significant. Statistical analyses were performed in Stata (StataCorp. 2019. Stata Statistical Software: Release 16; StataCorp LLC, College Station, Texas, USA).
Results
Over the study period, 4,810 patients were admitted to the medical ICU with a diagnosis of sepsis or septic shock. After exclusion criteria were applied, 1,205 patients remained, and of those, 737 had AKI present on admission (Figure 1). One-hundred fifty-two patients received early azithromycin and the remaining 585 were considered non-exposed or control patients (10 of these patients received azithromycin at some point later in the admission beyond 48 hours). Based on the timing of the evaluation period, three patients tested positive for severe acute respiratory coronavirus 2 (SARS-CoV-2). All three patients had bacterial co-infections and were included in the cohort of SA-AKI patients.
Figure 1.
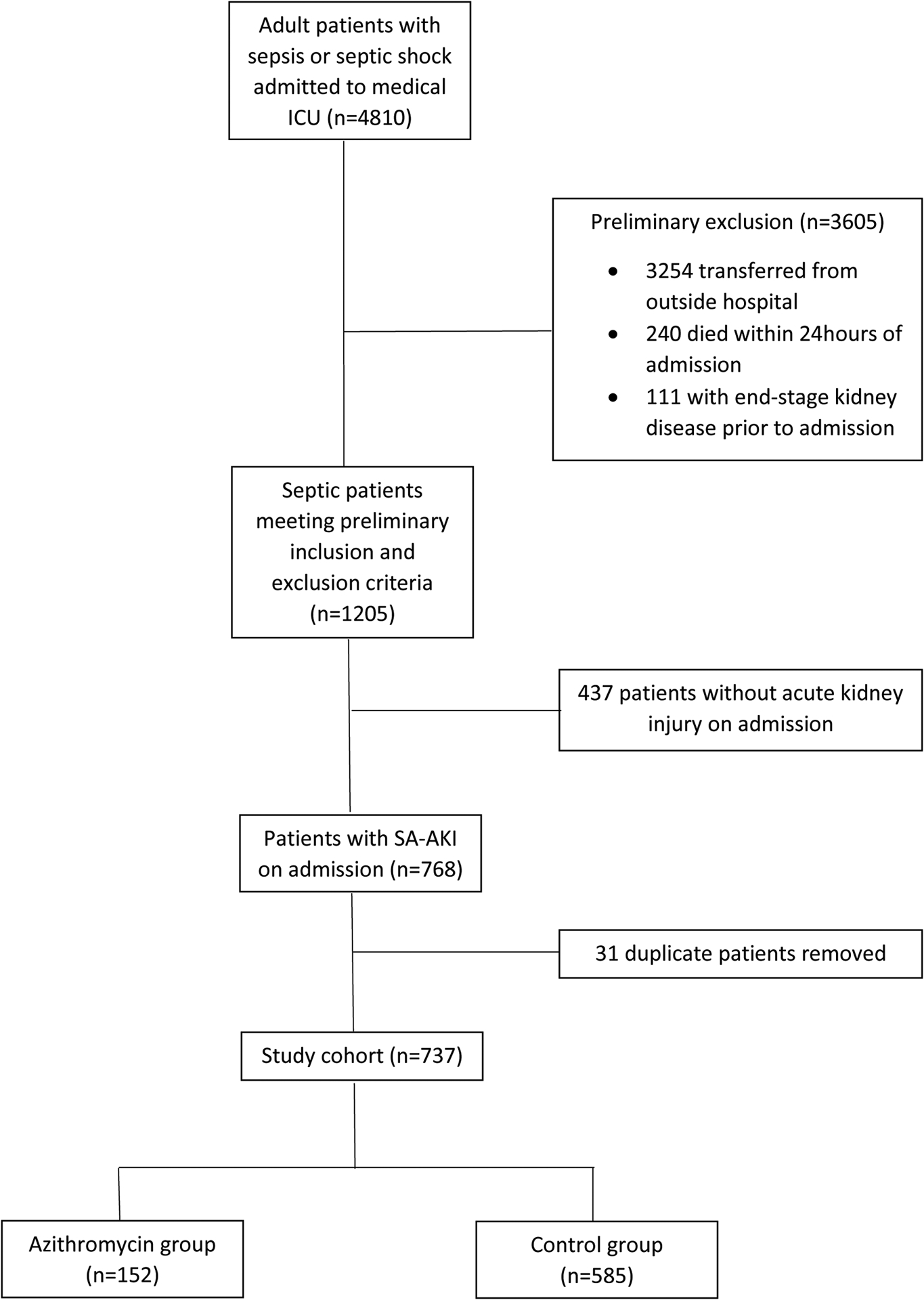
Study Flow Diagram
Baseline demographics are presented in Table 1. Patients in the azithromycin group were slightly older, more commonly male, and with a lower baseline eGFR. Comorbidities were similar between groups except for heart failure which was more common in the azithromycin group. Patients prescribed azithromycin tended to have less severe AKI on admission with more stage 1 and less stage 3 AKI compared to the non-exposed group. Patients receiving azithromycin were also less likely to receive anti-Pseudomonal antibiotic coverage compared to the non-exposed group. Both groups demonstrated similar severity of illness as assessed by admission SOFA score and vasopressor requirement in the first 48 hours of ICU admission. The median duration of receipt of azithromycin was 3 (2–5) days.
Table 1.
Patient Demographics
| Demographic | Azithromycin (n=152) | Control (n=585) | p-value |
|---|---|---|---|
| Prior to admission | |||
| Age (years) | 61 (50–70) | 56 (45–66) | 0.001 |
| Sex (% male) | 98 (64.5%) | 313 (53.5%) | 0.015 |
| Race (% white) | 132 (86.8%) | 519 (88.7%) | 0.685 |
| Height (cm) | 173 (165–180) | 173 (163–178) | 0.185 |
| Weight (kg) | 83 (70–100) | 81 (68–100) | 0.239 |
| Baseline serum creatinine (mg/dl) | 1.0 (0.8–1.1) | 0.9 (0.7–1.0) | 0.011 |
| Baseline eGFR (ml/min/1.73 m2) | 77 (64–97) | 87 (67–105) | 0.016 |
| Diabetes (%) | 48 (31.6%) | 202 (34.5%) | 0.494 |
| Hypertension (%) | 99 (65.1%) | 376 (64.3%) | 0.844 |
| Coronary artery disease (%) | 54 (35.5%) | 183 (31.3%) | 0.318 |
| Heart failure (%) | 48 (31.6%) | 132 (22.6%) | 0.021 |
| Liver disease (%) | 40 (26.3%) | 182 (31.1%) | 0.251 |
| Chronic kidney disease (%) | 39 (25.7%) | 127 (21.7%) | 0.299 |
| Hospital admission | |||
| KDIGO AKI stage | <0.001 | ||
| 1 | 84 (55.3%) | 223 (38.1%) | |
| 2 | 37 (24.3%) | 156 (26.7%) | |
| 3 | 31 (20.4%) | 206 (35.2%) | |
| Serum lactate (mmol/l) | 0.9 (0.4–1.9) | 1.0 (0.5–3.3) | 0.03 |
| SOFA score | 9 (6–13) | 9 (6–13) | 0.393 |
| Anti-pseudomonal antibiotic coverage (%) | 103 (67.8%) | 444 (75.9%) | 0.041 |
| Vasopressor requirement within 48 hours of admission (%) | 109 (71.7%) | 438 (74.9%) | 0.427 |
| Duration of azithromycin treatment (days) | 3 (2–5) | 0 (0–0) | <0.001 |
eGFR=estimated glomerular filtration rate; KDIGO=Kidney Disease Improving Global Outcomes; SOFA=Sequential Organ Failure Assessment
The primary outcome of MAKE at hospital discharge occurred in 59 (38.8%) patients in the azithromycin group and in 283 (48.4%) patients in the non-exposed group (p=0.035) (Table 2). The individual components of the MAKE outcome were all numerically less frequent in the azithromycin group (Figure 2) but were not statistically significant as a single criterion. There were no differences in any of the secondary outcomes assessed.
Table 2.
Study Outcomes
| Outcome | Azithromycin (n=152) | Control (n=585) | p-value |
|---|---|---|---|
| Major Adverse Kidney Events (%) | 59 (38.8%) | 283 (48.4%) | 0.035 |
| Hospital mortality | 41 (27.0%) | 189 (32.3%) | 0.206 |
| KRT at discharge | 1 (0.7%) | 14 (2.4%) | 0.177 |
| eGFR decrease ≥ 25% at discharge | 17 (11.2%) | 80 (13.7%) | 0.418 |
| KRT within one week of admission (%) | 19 (12.5%) | 85 (14.5%) | 0.522 |
| Death within one week of admission (%) | 23 (15.1%) | 103 (17.6%) | 0.470 |
| Incident Acute Kidney Disease (%) | 37 (24.3%) | 157 (26.8%) | 0.534 |
| Death or Incident Acute Kidney Disease (%) | 60 (39.5%) | 260 (44.4%) | 0.271 |
| ICU mortality (%) | 30 (19.7%) | 147 (25.1%) | 0.166 |
| Serum creatinine at discharge, survivors without KRT (mg/dl) | 0.9 (0.7–1.1) | 0.8 (0.7–1.2) | 0.888 |
| eGFR at discharge, survivors without KRT (ml/min/1.73 m2) | 92 (64–108) | 89 (61–110) | 0.777 |
| ICU length of stays (days) | 5.8 (2.9–12.1) | 5.5 (2.9–11.1) | 0.499 |
| Hospital length of stay (days) | 10.3 (5.2–18.6) | 9.5 (5.1–18.7) | 0.879 |
KRT=kidney replacement therapy; eGFR=estimated glomerular filtration rate; ICU=intensive care unit
Figure 2.
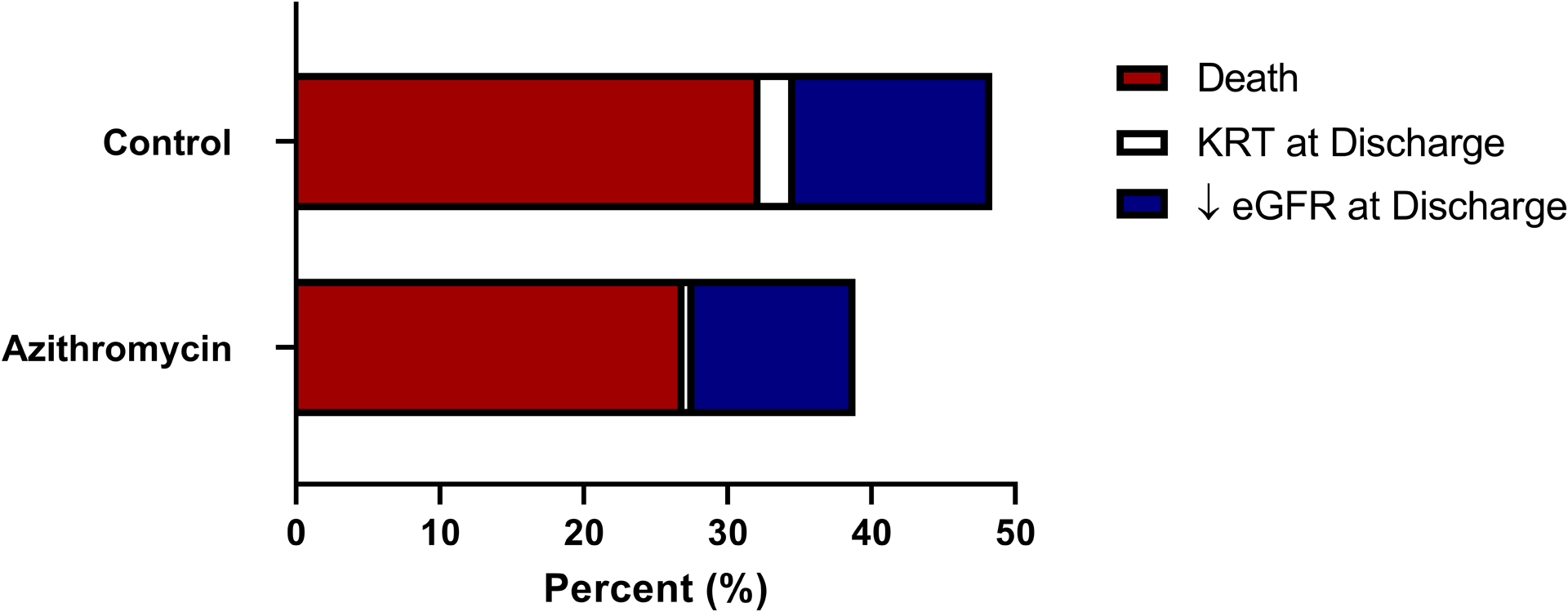
Component Outcomes of Major Adverse Kidney Events Between Cohorts
When controlling for potential confounders in multivariable logistic regression, exposure to azithromycin was independently associated with a reduction in MAKE at hospital discharge (aOR 0.62 [95% CI 0.41–0.93]; p=0.020) (Table 3). In sensitivity analysis (eTables 1–4) azithromycin retained a statistically significant association on reducing the odds of MAKE at discharge when the eGFR threshold for the MAKE outcome was changed to 50% reduction from baseline and when analyzing the composite outcome of death or KRT at discharge without accounting for the eGFR reduction. Azithromycin exposure was not significantly associated with a reduction of MAKE at discharge when limited to patients with a documented (non-imputed) baseline serum creatinine or when limited to patients with KDIGO stage 2–3 AKI. Using the adjusted odds ratio for MAKE at discharge from the primary model, the E-value was determined to be 1.86, indicating an unmeasured confounder would need an odds ratio of 1.86 to explain away the observed benefit of azithromycin exposure seen in the primary analysis (eFigure 1).
Table 3.
Multivariable Logistic Regression for Major Adverse Kidney Events at Discharge
| Variable | Odds Ratio (95% Confidence Interval | p-value |
|---|---|---|
| Azithromycin | 0.62 (0.41–0.93) | 0.020 |
| Age (per 1-year) | 1.01 (0.99–1.02) | 0.224 |
| Sex (male vs. female) | 1.43 (1.03–1.98) | 0.031 |
| Baseline eGFR (per 1 ml/min/1.73 m2) | 0.99 (0.98–1.00) | 0.155 |
| Diabetes | 0.83 (0.58–1.18) | 0.299 |
| Hypertension | 0.62 (0.42–0.92) | 0.016 |
| Heart failure | 1.09 (0.75–1.59) | 0.641 |
| Liver disease | 1.93 (1.35–2.77) | <0.001 |
| Chronic kidney disease | 2.29 (1.51–3.49) | <0.001 |
| KDIGO AKI stage (vs. stage 1) | ||
| Stage 2 | 0.99 (0.66–1.47) | 0.946 |
| Stage 3 | 1.33 (0.90–1.97) | 0.146 |
| Lactate (per 1 mmol/l) | 1.02 (0.98–1.07) | 0.261 |
| SOFA (per 1-unit) | 1.10 (1.06–1.14) | <0.001 |
| Anti-pseudomonal antibiotic coverage | 0.76 (0.53–1.10) | 0.148 |
eGFR=estimated glomerular filtration rate; KDIGO=Kidney Disease Improving Global Outcomes; SOFA=Sequential Organ Failure Assessment score
Discussion
To our knowledge, this is the first study to assess the clinical effects of azithromycin on kidney-related outcomes in critically ill patients with SA-AKI. We observed an independent association between receipt of azithromycin within the first 48-hours of ICU admission and a reduction in the risk of MAKE at hospital discharge in critically ill adults with SA-AKI. Results were consistent when adjusted for identified confounders and across several sensitivity analyses.
Macrolides have previously been studied as anti-infective or potential immunomodulatory adjuncts in critically ill patients, with a focus on primarily respiratory conditions including pneumonia and acute respiratory distress syndrome yielding mixed results (20–33). In the largest randomized trial of macrolides in critically ill patients (n=600 patients), Giamarellos-Bourboulis et al evaluated clarithromycin versus placebo in gram-negative sepsis (34). Although there was no difference in 28-day mortality, of the 54 patients with septic shock and multiple organ dysfunction, 15/28 (53.6%) randomized to clarithromycin died compared to 19/26 (73.1%) receiving placebo (p=0.020) (34). Although this was a subset of the entire trial population, the results are consistent with analysis from a smaller pilot trial performed by the same group of investigators studying clarithromycin versus placebo in ventilator-associated pneumonia (27). In this smaller trial of 200 patients, clarithromycin was also associated with mortality reduction compared to placebo in the subgroup of patients with septic shock and multiple organ dysfunction (27). These subgroup analyses from prior trials of macrolides in critical illness suggest that any potential benefit present may be driven by those patients with septic shock and a significant degree of organ dysfunction. With nearly 3 out of 4 patients requiring vasopressor support within 48 hours of admission, a median SOFA score of 9, and all patients in our cohort with SA-AKI, the patients from our study represent this type of acute illness phenotype.
Although the design of our study does not allow for evaluation of any potential immunomodulatory or other mechanisms for the protective association observed with azithromycin, pre-clinical data suggest several possible mechanisms whereby azithromycin may be beneficial in patients with infection or sepsis, including: reversal of immune suppression and endotoxin tolerance in sepsis and facilitating return of innate immune cell function (11, 28, 29), induction of a shift to more tolerogenic immune cell phenotypes such as T-helper-2 and M2-macrophages (35, 36), alteration of the microbiome and its interaction with the immune system (37), impaired Toll-like receptor signaling and reduced inflammasome activation (38–40), reduction of inflammatory chemokines and cytokines (41), inhibition of CD4 T-cell proliferation (42), enhancement of phagocytosis (43) and efferocytosis (44), enhancement of apoptosis in neutrophils contributing to excessive inflammation (45), enhancement of bacterial killing (46) and/or synergism with other antimicrobials, and inhibition of quorum sensing (47), among other potential mechanisms. Readers are referred to an excellent review by Reijnders and colleagues for an in-depth discussion of these potential mechanisms (11).
Potential advantages of azithromycin include: 1.) It is a commonly prescribed antibiotic available in multiple formulations and with a well-defined safety profile; 2.) It has extensive tissue distribution (48), with tissue concentrations exceeding those observed in serum anywhere from 10–100 fold, including lung and kidney tissue (49); and 3.) It achieves rapid, high, and prolonged intracellular concentrations in many cell lines (50, 51).
Our study has several strengths. First, we included a relatively large sample size with consideration of the many potential confounders in critical care nephrology research. Our E-value of 1.86 suggests that an unmeasured confounder of this magnitude could theoretically move the confidence interval to the null, but the unmeasured confounder would have to be approximately similar in magnitude to CKD or liver disease (Table 3), which we anticipate being unlikely. Second, the outcome of MAKE is clinically relevant, accounts for the competing risk of mortality, and is garnering increased support as a universal endpoint in critical care nephrology research (15). Lastly, we conducted several sensitivity analyses including more specific GFR thresholds for the MAKE outcome that also demonstrated a protective association of azithromycin.
Our study also has notable limitations. First, as a single center, retrospective, observational study, there may be inherent biases including information bias and selection bias that affected our results. Second, culture data were not available for all patients in our study, so we were unable to assess what proportion of patients had a respiratory versus non-respiratory source of infection. Third, baseline serum creatinine was missing for approximately half of the patient population. Although imputation was used to estimate missing values and define our cohort of SA-AKI patients, this may have biased the true baseline kidney function for a significant portion of our cohort. The sensitivity analysis in documented (non-imputed) baseline serum creatinine was not significant for azithromycin’s association with MAKE, but was also reduced in power by approximately 50%. Fourth, azithromycin is predominantly used for community-acquired infections or to provide antibiotic coverage for atypical pathogens which may have introduced indication bias in our study with less illness severity in the azithromycin group. Although we used anti-Pseudomonal antibiotic coverage as a surrogate for hospital versus community source of infection as well as adjusted for numerous other relevant potential confounders, we may have been unable to fully control for any potential bias. Fifth, over 50% of patients in the azithromycin group were diagnosed with KDIGO Stage 1 AKI on admission. Although we controlled for KDIGO severity stage, and even though the azithromycin group had lower baseline eGFR, this could indicate less kidney injury on admission in the azithromycin group that may not be adequately controlled for with multivariable regression modeling which may have led to more favorable outcomes in patients receiving azithromycin. Finally, the design of our study did not allow the opportunity to study immune parameters and specific immunomodulatory effects of azithromycin in SA-AKI. While we hypothesize that the immunomodulatory properties shown in other conditions may be beneficial in SA-AKI (52–54), further research is required.
Our study found that early exposure to azithromycin in critically ill adult patients with SA-AKI was associated with a reduction in MAKE at hospital discharge compared to patients not receiving azithromycin. Additional investigation of macrolide antibiotics or derivatives in SA-AKI is warranted.
Supplementary Material
Conflicts of Interest and Source of Funding:
The authors have no relevant conflicts of interest to disclose. The project described was supported by the NIH National Center for Advancing Translational Sciences through grant number UL1TR001998. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
References:
- 1.Bagshaw SM, George C, Bellomo R: Early acute kidney injury and sepsis: a multicentre evaluation. Crit Care 12(2):R47, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bagshaw SM, Uchino S, Bellomo R, Morimatsu H, Morgera S, Schetz M, Tan I, Bouman C, Macedo E, Gibney N, et al. : Septic acute kidney injury in critically ill patients: clinical characteristics and outcomes. Clin J Am Soc Nephrol 2(3):431–9, 2007. [DOI] [PubMed] [Google Scholar]
- 3.Cruz DN, Bolgan I, Perazella MA, Bonello M, de Cal M, Corradi V, Polanco N, Ocampo C, Nalesso F, Piccinni P, et al. : North East Italian Prospective Hospital Renal Outcome Survey on Acute Kidney Injury (NEiPHROS-AKI): targeting the problem with the RIFLE Criteria. Clin J Am Soc Nephrol 2(3):418–25, 2007. [DOI] [PubMed] [Google Scholar]
- 4.Uchino S, Kellum JA, Bellomo R, Doig GS, Morimatsu H, Morgera S, Schetz M, Tan I, Bouman C, Macedo E, et al. : Acute renal failure in critically ill patients: a multinational, multicenter study. JAMA 294(7):813–8, 2005. [DOI] [PubMed] [Google Scholar]
- 5.Peerapornratana S, Priyanka P, Wang S, Smith A, Singbartl K, Palevsky PM, Chawla LS, Yealy DM, Angus DC, Kellum JA: Sepsis-Associated Acute Kidney Disease. Kidney Int Rep 5(6):839–850, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Flannery AH, Li X, Delozier NL, Toto RD, Moe OW, Yee J, Neyra JA: Sepsis-Associated Acute Kidney Disease and Long-term Kidney Outcomes. Kidney Med 3(4):507–514, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nakano D: Septic acute kidney injury: a review of basic research. Clin Exp Nephrol 24(12):1091–1102, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Prowle JR, Bellomo R: Sepsis-associated acute kidney injury: macrohemodynamic and microhemodynamic alterations in the renal circulation. Semin Nephrol 35(1):64–74, 2015. [DOI] [PubMed] [Google Scholar]
- 9.Wan L, Bellomo R, Di Giantomasso D, Ronco C: The pathogenesis of septic acute renal failure. Curr Opin Crit Care 9(6):496–502, 2003. [DOI] [PubMed] [Google Scholar]
- 10.Gomez H, Ince C, De Backer D, Pickkers P, Payen D, Hotchkiss J, Kellum JA: A unified theory of sepsis-induced acute kidney injury: inflammation, microcirculatory dysfunction, bioenergetics, and the tubular cell adaptation to injury. Shock 41(1):3–11, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Reijnders TDY, Saris A, Schultz MJ, van der Poll T: Immunomodulation by macrolides: therapeutic potential for critical care. Lancet Respir Med 8(6):619–630, 2020. [DOI] [PubMed] [Google Scholar]
- 12.Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group: KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney inter., Suppl 2: 1–138, 2012. [Google Scholar]
- 13.Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, Bruining H, Reinhart CK, Suter PM, Thijs LG: The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22(7):707–10, 1996. [DOI] [PubMed] [Google Scholar]
- 14.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, et al. : A new equation to estimate glomerular filtration rate. Ann Intern Med 150(9):604–12, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Billings FT, Shaw AD: Clinical trial endpoints in acute kidney injury. Nephron Clin Pract 127(1–4):89–93, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chawla LS, Bellomo R, Bihorac A, Goldstein SL, Siew ED, Bagshaw SM, Bittleman D, Cruz D, Endre Z, Fitzgerald RL, et al. : Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol 13(4):241–257, 2017. [DOI] [PubMed] [Google Scholar]
- 17.Laszczyńska O, Severo M, Correia S, Azevedo A: Estimation of Missing Baseline Serum Creatinine for Acute Kidney Injury Diagnosis in Hospitalized Patients. Nephron 145(2):123–132, 2021. [DOI] [PubMed] [Google Scholar]
- 18.Siew ED, Peterson JF, Eden SK, Moons KG, Ikizler TA, Matheny ME: Use of multiple imputation method to improve estimation of missing baseline serum creatinine in acute kidney injury research. Clin J Am Soc Nephrol 8(1):10–8, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Linden A, Mathur MB, VanderWeele TJ: Conducting sensitivity analysis for unmeasured confounding in observational studies using E-values: The evalue package. Stata J 20(1):162–175, 2020. [Google Scholar]
- 20.Ceccato A, Cilloniz C, Martin-Loeches I, Ranzani OT, Gabarrus A, Bueno L, Garcia-Vidal C, Ferrer M, Niederman MS, Torres A: Effect of Combined β-Lactam/Macrolide Therapy on Mortality According to the Microbial Etiology and Inflammatory Status of Patients With Community-Acquired Pneumonia. Chest 155(4):795–804, 2019. [DOI] [PubMed] [Google Scholar]
- 21.Lorenzo MJ, Moret I, Sarria B, Cases E, Cortijo J, Méndez R, Molina J, Gimeno A, Menéndez R: Lung inflammatory pattern and antibiotic treatment in pneumonia. Respir Res 16(1):15, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cilloniz C, Albert RK, Liapikou A, Gabarrus A, Rangel E, Bello S, Marco F, Mensa J, Torres A: The Effect of Macrolide Resistance on the Presentation and Outcome of Patients Hospitalized for Streptococcus pneumoniae Pneumonia. Am J Respir Crit Care Med 191(11):1265–72, 2015. [DOI] [PubMed] [Google Scholar]
- 23.van Delden C, Köhler T, Brunner-Ferber F, François B, Carlet J, Pechère JC: Azithromycin to prevent Pseudomonas aeruginosa ventilator-associated pneumonia by inhibition of quorum sensing: a randomized controlled trial. Intensive Care Med 38(7):1118–25, 2012. [DOI] [PubMed] [Google Scholar]
- 24.Laserna E, Sibila O, Fernandez JF, Maselli DJ, Mortensen EM, Anzueto A, Waterer G, Restrepo MI: Impact of macrolide therapy in patients hospitalized with Pseudomonas aeruginosa community-acquired pneumonia. Chest 145(5):1114–1120, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Restrepo MI, Mortensen EM, Waterer GW, Wunderink RG, Coalson JJ, Anzueto A: Impact of macrolide therapy on mortality for patients with severe sepsis due to pneumonia. Eur Respir J 33(1):153–9, 2009. [DOI] [PubMed] [Google Scholar]
- 26.Afshar M, Foster CL, Layden JE, Burnham EL: Azithromycin use and outcomes in severe sepsis patients with and without pneumonia. J Crit Care 32:120–5, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Giamarellos-Bourboulis EJ, Pechère JC, Routsi C, Plachouras D, Kollias S, Raftogiannis M, Zervakis D, Baziaka F, Koronaios A, Antonopoulou A, et al. : Effect of clarithromycin in patients with sepsis and ventilator-associated pneumonia. Clin Infect Dis 46(8):1157–64, 2008. [DOI] [PubMed] [Google Scholar]
- 28.Spyridaki A, Raftogiannis M, Antonopoulou A, Tsaganos T, Routsi C, Baziaka F, Karagianni V, Mouktaroudi M, Koutoukas P, Pelekanou A, et al. : Effect of clarithromycin in inflammatory markers of patients with ventilator-associated pneumonia and sepsis caused by Gram-negative bacteria: results from a randomized clinical study. Antimicrob Agents Chemother 56(7):3819–25, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tsaganos T, Raftogiannis M, Pratikaki M, Christodoulou S, Kotanidou A, Papadomichelakis E, Armaganidis A, Routsi C, Giamarellos-Bourboulis EJ: Clarithromycin Leads to Long-Term Survival and Cost Benefit in Ventilator-Associated Pneumonia and Sepsis. Antimicrob Agents Chemother 60(6):3640–6, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kawamura K, Ichikado K, Takaki M, Eguchi Y, Anan K, Suga M: Adjunctive therapy with azithromycin for moderate and severe acute respiratory distress syndrome: a retrospective, propensity score-matching analysis of prospectively collected data at a single center. Int J Antimicrob Agents 51(6):918–924, 2018. [DOI] [PubMed] [Google Scholar]
- 31.Walkey AJ, Wiener RS: Macrolide antibiotics and survival in patients with acute lung injury. Chest 141(5):1153–1159, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Simonis FD, de Iudicibus G, Cremer OL, Ong DSY, van der Poll T, Bos LD, Schultz MJ: Macrolide therapy is associated with reduced mortality in acute respiratory distress syndrome (ARDS) patients. Ann Transl Med 6(2):24, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pons S, Timsit JF, Ruckly S, Schwebel C, Papazian L, Azoulay E, Reignier J, Zafrani L: Impact of macrolide therapy in critically ill patients with acute respiratory failure: a desirability of outcome ranking analysis to investigate the OUTCOMEREA database. Intensive Care Med 45(7):1043–1045, 2019. [DOI] [PubMed] [Google Scholar]
- 34.Giamarellos-Bourboulis EJ, Mylona V, Antonopoulou A, Tsangaris I, Koutelidakis I, Marioli A, Raftogiannis M, Kopterides P, Lymberopoulou K, Mouktaroudi M, et al. : Effect of clarithromycin in patients with suspected Gram-negative sepsis: results of a randomized controlled trial. J Antimicrob Chemother 69(4):1111–8, 2014. [DOI] [PubMed] [Google Scholar]
- 35.Matsui K, Tamai S, Ikeda R: Effects of Macrolide Antibiotics on Th1 Cell and Th2 Cell Development Mediated by Langerhans Cells. J Pharm Pharm Sci 19(3):357–366, 2016. [DOI] [PubMed] [Google Scholar]
- 36.Murphy BS, Sundareshan V, Cory TJ, Hayes D Jr., Anstead MI, Feola DJ: Azithromycin alters macrophage phenotype. J Antimicrob Chemother 61(3):554–60, 2008. [DOI] [PubMed] [Google Scholar]
- 37.Ruiz VE, Battaglia T, Kurtz ZD, Bijnens L, Ou A, Engstrand I, Zheng X, Iizumi T, Mullins BJ, Müller CL, et al. : A single early-in-life macrolide course has lasting effects on murine microbial network topology and immunity. Nat Commun 8(1):518, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fan LC, Lin JL, Yang JW, Mao B, Lu HW, Ge BX, Choi AMK, Xu JF: Macrolides protect against Pseudomonas aeruginosa infection via inhibition of inflammasomes. Am J Physiol Lung Cell Mol Physiol 313(4):L677–l686, 2017. [DOI] [PubMed] [Google Scholar]
- 39.Lendermon EA, Coon TA, Bednash JS, Weathington NM, McDyer JF, Mallampalli RK: Azithromycin decreases NALP3 mRNA stability in monocytes to limit inflammasome-dependent inflammation. Respir Res 18(1):131, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nujić K, Banjanac M, Munić V, Polančec D, Eraković Haber V: Impairment of lysosomal functions by azithromycin and chloroquine contributes to anti-inflammatory phenotype. Cell Immunol 279(1):78–86, 2012. [DOI] [PubMed] [Google Scholar]
- 41.Tong J, Liu ZC, Wang DX: Azithromycin acts as an immunomodulatory agent to suppress the expression of TREM-1 in Bacillus pyocyaneus-induced sepsis. Immunol Lett 138(2):137–43, 2011. [DOI] [PubMed] [Google Scholar]
- 42.Ratzinger F, Haslacher H, Poeppl W, Hoermann G, Kovarik JJ, Jutz S, Steinberger P, Burgmann H, Pickl WF, Schmetterer KG: Azithromycin suppresses CD4(+) T-cell activation by direct modulation of mTOR activity. Sci Rep 4:7438, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hodge S, Hodge G, Brozyna S, Jersmann H, Holmes M, Reynolds PN: Azithromycin increases phagocytosis of apoptotic bronchial epithelial cells by alveolar macrophages. Eur Respir J 28(3):486–95, 2006. [DOI] [PubMed] [Google Scholar]
- 44.Hodge S, Hodge G, Jersmann H, Matthews G, Ahern J, Holmes M, Reynolds PN: Azithromycin improves macrophage phagocytic function and expression of mannose receptor in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 178(2):139–48, 2008. [DOI] [PubMed] [Google Scholar]
- 45.Aoshiba K, Nagai A, Konno K: Erythromycin shortens neutrophil survival by accelerating apoptosis. Antimicrob Agents Chemother 39(4):872–7, 1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dey S, Bishayi B: Riboflavin along with antibiotics balances reactive oxygen species and inflammatory cytokines and controls Staphylococcus aureus infection by boosting murine macrophage function and regulates inflammation. J Inflamm (Lond) 13:36, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kanoh S, Rubin BK: Mechanisms of action and clinical application of macrolides as immunomodulatory medications. Clin Microbiol Rev 23(3):590–615, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pfizer Labs Inc. Zithromax (azithromycin) [package insert]. Published online 2013.
- 49.Jain R, Danziger LH: The macrolide antibiotics: a pharmacokinetic and pharmacodynamic overview. Curr Pharm Des 10(25):3045–53, 2004. [DOI] [PubMed] [Google Scholar]
- 50.Bosnar M, Kelnerić Z, Munić V, Eraković V, Parnham MJ: Cellular uptake and efflux of azithromycin, erythromycin, clarithromycin, telithromycin, and cethromycin. Antimicrob Agents Chemother 49(6):2372–7, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Foulds G, Shepard RM, Johnson RB: The pharmacokinetics of azithromycin in human serum and tissues. J Antimicrob Chemother 25 Suppl A:73–82, 1990. [DOI] [PubMed] [Google Scholar]
- 52.Al-Darraji A, Haydar D, Chelvarajan L, Tripathi H, Levitan B, Gao E, Venditto VJ, Gensel JC, Feola DJ, Abdel-Latif A: Azithromycin therapy reduces cardiac inflammation and mitigates adverse cardiac remodeling after myocardial infarction: Potential therapeutic targets in ischemic heart disease. PLoS One 13(7):e0200474, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Feola DJ, Garvy BA, Cory TJ, Birket SE, Hoy H, Hayes D, Murphy BS: Azithromycin alters macrophage phenotype and pulmonary compartmentalization during lung infection with Pseudomonas. Antimicrob Agents Chemother 54(6):2437–47, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhang B, Bailey WM, Kopper TJ, Orr MB, Feola DJ, Gensel JC: Azithromycin drives alternative macrophage activation and improves recovery and tissue sparing in contusion spinal cord injury. J Neuroinflammation 12:218, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


