Abstract
Objective
This study was designed to analyze the clinical characteristics, etiological characteristics, drug resistance, and empirical use of antibiotics for community-acquired pyogenic liver abscess (PLA) to provide a basis for rational and effective empirical treatment of PLA in the local area.
Methods
The clinical data, etiological characteristics, drug resistance, and empirical anti-infective therapy schemes of 606 patients with PLA were collected and analyzed retrospectively.
Results
The included patients were mainly males, with a male-to-female ratio of 1.3:1. The average age of the patients was 60.3 ± 14.1 years. The underlying diseases were diabetes and biliary tract disease, accounting for 38.7% and 22.3%, respectively. The main clinical manifestations were fever (92.9%), abdominal pain (44.7%), and nausea (33.3%). Imaging findings: the proportion of patients with a single lesion was 74.7%, and 67% of the patients had involvement in the right lobe of the liver. The main pathogen was Klebsiella pneumoniae accounted for 74.9% in blood culture and 84.1% in pus culture, mainly extended-spectrum β-lactamase. In 272 strains negative for extended-spectrum β-lactamase (ESBLs), 100% were resistant to ampicillin and less than 50% were sensitive to nitrofurantoin. Only 36 ESBL-positive strains had higher than 80% sensitivity to carbapenems, β-lactamase inhibitor compound, and amikacin. Patients treated with different treatment methods showed significantly different average length of hospital stay (14 [9–21] vs 13 [8–18]). Empirical anti-infective therapy: Beta-lactamase complex, carbapenems, cephalosporins, and quinolones were used in 280 (37.6%), 180 (29.7%), 180 (29.7%), and 147 (24.3%) patients, respectively.
Conclusion
Patients with community-acquired PLA in this area are mainly males, and the underlying diseases are mainly diabetes and hepatobiliary system disease. The main clinical manifestation is fever, so patients with fever of unknown cause should pay attention to possible liver abscesses. Based on drug sensitivity tests, the empirical use of antibiotics is somewhat unreasonable.
Keywords: liver abscess, Klebsiella pneumoniae, epidemiological, community
Introduction
Pyogenic liver abscess (PLA) is an infectious disease caused by pyogenic bacteria invading the liver through various channels, accounting for 13% of abdominal infectious diseases and 48% of visceral abscesses.1,2 In recent years, the overall incidence rate of PLA has been increasing gradually, and the incidence rate is higher in Asian countries.3–5 In the past 20 years, the infection rate of Klebsiella pneumoniae (K. pneumoniae) in the Asian population has gradually increased. At present, it has become the main pathogen causing PLA infection.6–8 Patients with PLA may have nonspecific manifestations such as chills, fever, shivers, and pain in the liver area. When the infection is poorly controlled, PLAs, especially those caused by K. pneumoniae, can lead to sepsis, multiple organ dysfunction, and death; it has a mortality rate of 2%–31%.9 However, due to the nonspecific manifestations of some patients, there are some misdiagnoses and missed diagnoses in clinics. To improve the diagnosis rate of PLA in the local area, effective empirical treatment must begin as soon as possible to improve the prognosis of patients. The selection of antimicrobial drugs requires the combination with local PLA pathogenesis and clinical manifestations. In this retrospective study, we aimed to analyze the clinical characteristics, etiological characteristics, drug resistance, and empirical use of antibiotics for community-acquired pyogenic liver abscess (PLA), and therefore to provide a basis for rational and effective empirical treatment of PLA in the local area.
Data and Methods
Subjects
PLA met the diagnostic criteria proposed by Foo et al10 in 2010. Exclusion criteria: (1) mixed infection, combined with fungal or parasitic infection; (2) the pathogenic culture results of the same patient were not the same; (3) the culture results were considered as contaminated bacteria. The study was approved by the ethics committee of the hospital.
Research Methods
A total of 606 patients with PLA, diagnosed in Yantai Yuhuangding Hospital from 2015 to 2020, were analyzed retrospectively. The basic data, laboratory results, microbiological results, and treatment methods (eg, empirical anti-infective therapy scheme and drainage) were collected.
Result Interpretation Criteria
The curative effect was evaluated after two weeks of hospitalization. (1) Effective: the symptoms were relieved, and the abscess shrunk after treatment. (2) Ineffective: the symptoms were not alleviated or worsened after treatment, the size of the pus cavity did not change, increases upon imaging examination (abdominal ultrasound, computed tomography [CT] or magnetic resonance imaging) were noted, or the death of the patient.
Bacterial Isolation and Culture
Culture of Drainage Fluid and Blood
The patient’s ultrasound-guided percutaneous drainage fluid was immediately sent to the bacterial room for culture. An inoculating nutrient broth was used to increase the amount of the bacteria. After 24 h, the bacteria were transferred to a blood plate and a MacConkey agar plate at the same time and cultured at 35°C for 24 h. A blood culture was carried out in strict accordance with the relevant methods in the BACTEC9050 blood culture user manual. After the positive alarm of the LED display screen of the instrument, smearing and seed transfers were performed at the same time. The method of seed transfer is the same as that of ordinary culture. For the pus culture, the pus was directly inoculated in a blood plate and a MacConkey agar plate and cultured at 35°C for 24 h.
Pathogen Identification and Drug Sensitivity Test
The separated bacteria were dissolved evenly in the identification culture medium. The turbidimeter was used to adjust the turbidity (0.5 Michaelis turbidity), and 25 μL of the sample was taken from the dissolved identification culture medium into the drug sensitivity inoculation culture medium and placed in the corresponding well of the positive plate or negative plate. A full-automatic microbial analyzer, with its own bacterial identification/drug sensitivity system, was used for pathogen identification and drug sensitivity detection.
Statistical Analysis
The original data of the patients were collected and sorted with Excel. The data were statistically analyzed using statistical software SPSS 22.0. Normally distributed and approximately normally distributed measurement data were expressed as the mean ± standard deviation and compared between groups using t-tests. Non-normally distributed measurement data were expressed as the median and interquartile and compared between groups using non-parametric tests. The count data were expressed as numbers (proportion) and compared using two independent sample Chi-square tests or Fisher exact probability methods. P < 0.05 was considered statistically significant.
Results
Basic Data of Patients with PLA
Of the 606 patients with PLA, 345 were males (56.7%), with a male-to-female ratio of 1.3:1. The age range of these patients was 18–97 years, with an average age of 60.3 ± 14.1 years. The main underlying diseases were diabetes, cerebrovascular disease, and benign biliary disease, accounted for 38.7%, 27.9%, and 22.3% respectively (Table 1).
Table 1.
Clinical Features of Pyogenic Liver Abscess
| Item | Number (Percentage) | |
|---|---|---|
| Gender | Male | 346(57.1%) |
| Female | 260(42.9%) | |
| Average age (years old) | 60.3±14.1 | |
| Underlying disease | Hypertension | 89(14.7%) |
| Diabetes mellitus | 234(38.6%) | |
| Coronary heart disease | 36(5.9%) | |
| Cerebrovascular disease | 169(27.9%) | |
| Malignant tumors | 67(11.1%) | |
| Benign biliary disease or operation | 135(22.3%) | |
| Clinical manifestations | Fever | 563(92.9%) |
| Fear of cold, shiver | 453(74.7%) | |
| Abdominal pain | 271(44.7%) | |
| Nausea, vomiting | 202(33.3%) | |
| Fatigue, poor appetite | 110(18.2%) | |
| Jaundice | 38(6.3%) | |
| Dyspnea | 35(5.7%) | |
| Headache dizziness | 25(4.1%) | |
| Unconsciousness | 11(1.8%) | |
| Peak body temperature (°C) | >39.0 | 323(57.3%) |
| ≤39.0 | 240(42.7%) |
Clinical and Laboratory Data of PLA
Clinical Manifestations
Fever (563 patients, 92.9%) was the most common clinical manifestation, followed by chills and chills (74.7%). Of all patients, 323 had a peak body temperature of >39°C, accounting for 57.3% of the patients with fever. Abdominal pain was a clinical manifestation in 44.7% of patients, 33.3% had vomiting or nausea, 18.2% had fatigue and poor appetite, and 6.3% had jaundice. Some patients had other atypical clinical symptoms (Table 1).
Laboratory Tests
Elevated leukocytes were found in 408 patients (68.5%). There were 76.8% of the patients with elevated neutrophils; 289 (99.0%) of them had elevated C-reactive protein; and 202 (76.5%) had elevated procalcitonin. Other common abnormal laboratory indicators were as follows: 338 patients (56.8%) with anemia, 179 patients with albumin reduction (30.2%), 423 patients (71.4%) with elevated γ-glutamyltransferase, 324 patients (54.7%) with elevated alkaline phosphatase, 301 patients (50.6%) with elevated alanine aminotransferase, 270 patients (45.4%) with elevated aspartate aminotransferase, 278 patients (47.2%) with elevated fasting blood glucose, and 462 (79.9%) patients with elevated lactate dehydrogenase.
Ultrasonic or CT Results
Of these 606 patients, 451 patients (74.4%) had a single abscess. In 406 patients (67%), the abscess was in the right lobe.
Analysis of Etiological Characteristics of PLA
Blood cultures were administered to 404 patients, and the positive rate was 26.1%. Among them, K. pneumoniae was dominant (79 patients, 74.5%), followed by Escherichia coli (E. coli; 13 patients, 12.3%). Pus cultures were performed on 409 patients, and the positive rate was 69.4% (284 patients). Among them, K. pneumoniae was dominant (233 patients, 82.1%), followed by E. coli (29 patients, 10.2%). The detection rate of positive cocci was very low in both the blood and pus cultures (Table 2).
Table 2.
Results of Blood Culture and Pus Culture
| Item | Blood Culture (n=414) | Pus Culture (n=409) |
|---|---|---|
| Positive (n (%)) | 106 (25.6) | 284(69.4) |
| KPN | 79(19.1) | 233(56.3) |
| ESBL (+) | 1(0.2) | 13(3.1) |
| ESBL (-) | 78(18.8) | 220(53.1) |
| Escherichia coli | 13(3.1) | 29(7.0) |
| ESBL (+) | 4(1.0) | 18(4.3) |
| ESBL (-) | 9(2.2) | 9(2.2) |
| Enterococcus faecalis | 3(0.7) | 3(0.7) |
| Morganella morganii | 1(0.2) | 2(0.5) |
| Staphylococcus aureus | 0 | 3(0.7) |
| Pseudomonas aeruginosa | 1(0.2) | 1(0.2) |
| Citrobacter freundii | 0 | 1(0.2) |
| Others | 9(2.2) | 12(2.9) |
| Negative | 308(76.4) | 125(30.6) |
Drug Resistance of Pathogens
In the 272 strains negative for extended-spectrum β-lactamase (ESBLs), in addition to 100% resistance to ampicillin and less than 50% sensitivity to nitrofurantoin, their sensitivity to the commonly used cephalosporins III, cephalosporins and monocyclic β-Lactams, quinolones (ciprofloxacin, levofloxacin), aminoglycosides (tobramycin, gentamicin, amikacin), tetracyclines (tigecycline) β-lactamase inhibitor compound (Cefoperazone/sulbactam, amoxicillin/clavulanic acid, piperacillin/tazobactam), and carbapenems was higher than 90%, whereas the sensitivity rate to amikacin and meropenem was 100%. However, the susceptibility of K. pneumoniae to cotrimoxazole and levofloxacin was better than that of E. coli.
Thirty-three ESBL (+) strains had higher than 80% sensitivity to carbapenems (imipenem, meropenem, ertapenem, and biapenem), tetracyclines (tigecycline), β-lactamase inhibitor compound (cefoperazone/sulbactam, piperacillin/tazobactam), and amikacin. Only two strains were multidrug resistance and all of them were E. coli (Table 3).
Table 3.
Analysis of Drug Resistance of Enterobacteriaceae Bacteria
| 271 Cases with K. pneumoniae | 35 Cases with E. coli | |||||||
|---|---|---|---|---|---|---|---|---|
| 18 ESBL+Sensitivity | 18 ESBL+Drug Resistance | 253 ESBL-Sensitivity | 253 ESBL- Drug Resistance | 17 ESBL+Sensitivity | 17 ESBL+Drug Resistance | 18 ESBL- Sensitivity | 18 ESBL-Drug Resistance | |
| N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | |
| Amikacin | 16 (88.9) | 2 (11.1) | 253 (100.0) | 0 (0.0) | 14 (82.4) | 3 (17.6) | 18 (100.0) | 0 (0.0) |
| Imipenem | 18(100.0) | 0(0.0) | 251 (99.2) | 0 (%) | 15 (88.2) | 1 (5.9) | 16 (88.9) | 2 (11.1) |
| Tigecycline | 18(100.0) | 0(0.0) | 240 (94.9) | 10(3.9) | 17 (100.0) | 0 (0.0) | 18 (100.0) | 0 (0.0) |
| Ertapenem | 18(100.0) | 0(0.0) | 250 (98.8) | 0(0.0) | 15 (88.2) | 2 (11.8) | 16 (88.9) | 2 (11.1) |
| Cefoxitin | 13(72.2) | 5(27.8) | 240 (94.9) | 8(3.2) | 10 (58.8) | 5 (29.4) | 13 (72.2) | 1 (5.6) |
| Piperacillin/Tazobactam | 17(94.4) | 1(5.6) | 250 (98.8) | 2(0.8) | 16 (94.1) | 0 (0.0) | 15 (88.2) | 2 (11.1) |
| Cefoperazone-Sulbactam | 16(88.9) | 2(11.1) | 251 (99.2) | 2(0.8) | 17 (100.0) | 0 (0.0) | 17 (94.4) | 1 (5.6) |
| Cefepime | 17(94.4) | 1(5.6) | 252 (99.6) | 0(0.0) | 12 (70.6) | 5 (29.4) | 16 (88.9) | 2 (11.1) |
| Ceftriaxone | 8(44.4) | 10(55.6) | 249 (98.4) | 4(1.6) | 3 (17.6) | 14 (82.4) | 15 (83.3) | 2 (11.1) |
| Amoxicillin clavulanate | 12(66.7) | 6(33.3) | 250 (98.8) | 1(0.4) | 8 (47.1) | 4 (23.5) | 8 (44.4) | 4 (22.2) |
| Levofloxacin | 14(77.8) | 4(22.2) | 250 (98.8) | 3(11.9) | 5 (29.4) | 12 (70.6) | 11 (61.1) | 6 (33.3) |
| Trimethoprim/Sulfamethoxazole | 13(77.2) | 5(22.8) | 243 (96.0) | 10(3.9) | 2 (11.8) | 15 (88.2) | 9 (50.0) | 9 (50.0) |
Empirical Anti-Infective Therapy
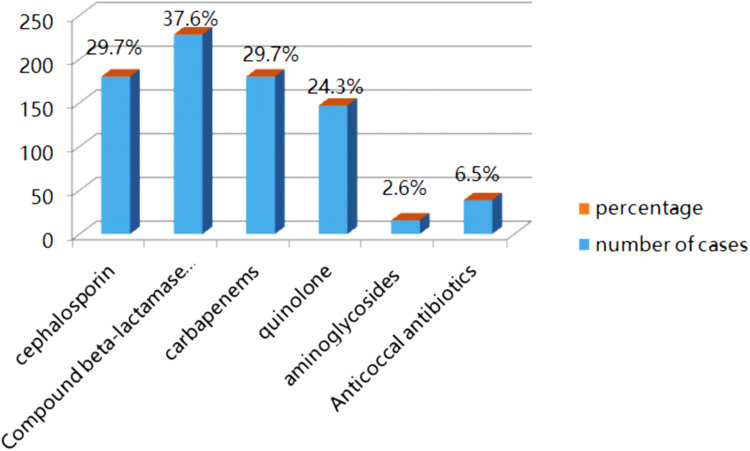
Of all patients, 228 were empirically treated with β-lactamase inhibitor compound for anti-infection treatment, and the proportion was the highest (37.6%). Of the total, 180 patients (29.7%) were empirically treated with cephalosporin, and 180 patients (29.7%) were empirically treated with carbapenems. In addition, 147 patients (24.3%) were empirically treated with quinolones, and 135 patients (22.3%) were treated with a single type of antibiotic. The remaining patients were treated with combined anti-infective therapy (cephalosporins combined with quinolones or aminoglycosides or nitroimidazoles, cefoperazone/sulbactam, or piperacillin/ sulbactam combined with nitroimidazoles). In 75 patients (12.3%), the anti-infection treatment was effective, and these patients underwent step-down treatment. A total of 105 patients (17.3%) received an upgraded treatment of carbapenem anti-infection. In 60 (9.9%) of these patients, the treatment was upgraded to antibiotics because of their serious condition, experience, or poor anti-infection treatment effect according to the drug sensitivity results (Figure 1).
Figure 1.
Empirical use of antibiotics.
Efficacy Evaluation of Different Treatment Methods
Of all patients, 154 were treated with antibiotics alone and 452 were treated with antibiotics combined with puncture and drainage. Of the 154 patients treated with antibiotics alone, 143 patients (92.9%) achieved effective outcomes, and 11 patients (7.1%) achieved ineffective outcomes. In the combined treatment group, 423 patients (93.6%) achieved effective outcomes, and 29 patients (6.4%) achieved ineffective outcomes. A comparison revealed that there was no significant difference in the efficacy between the two treatment methods. However, the average length of the hospital stays of the patients who received combined treatment was significantly shorter than that of the patients who were treated with antibiotics alone (Table 4).
Table 4.
Comparison of Curative Effect Between the Two Groups with Different Treatment Methods
| Item | Antibiotic Group (n=154) | Combined Group* (n=452) | P-value |
|---|---|---|---|
| Curative effect (n%) | |||
| Effective | 143 (92.9%) | 423 (93.6) | 0.527 |
| Ineffective | 11 (7.1%) | 29 (6.4%) | |
| Length of hospital stay | 14(9~21) | 13(8~18) | 0.029 |
Note: *Combined group: antibiotics combined with puncture and drainage.
Discussion
Pyogenic liver abscess is a serious infectious disease that threatens human health. It can develop into a life-threatening severe infection in patients with immunodeficiencies and older patients with underlying diseases. This study revealed that PLA usually occurs in males (the male-to-female ratio was 1.3:1). The average age was 60.3 ± 14.1 years. These results are consistent with the results of previous studies. The main underlying diseases were diabetes and biliary tract disease, similar to those of previous studies. This is mainly because the liver has two sets of blood supply, namely the hepatic artery and the portal vein. The portal vein is connected to the gastrointestinal tract, and patients with underlying diseases of the biliary tract and intestinal tract will have an increased chance of an incidence of liver abscess.11–16 In this study, 563 patients with PLA (92.9%) had a fever as the main clinical symptom and 323 patients (57.3%) had a high fever (>39.0°C). This is basically consistent with that reported in studies both locally and globally.17–19 Therefore, in patients with a fever of an unknown cause, PLA should be carefully considered, especially for patients with a high fever. Other clinical symptoms of PLA can be jaundice, fatigue, poor appetite, and fading consciousness, suggesting that the clinical manifestation of PLA is not typical, so clinicians should be vigilant to avoid misdiagnoses and missed diagnoses that prolong the condition and affect the prognosis. This study revealed that patients with a single bacterial infection accounted for 93.0% of the patients. Among them, the detection rate of K. pneumoniae was 73.1%, slightly higher than that reported in the literature (64.0%).20 The reason for this may be related to the patients’ underlying disease.21 The literature reported that diabetes mellitus was an independent risk factor for K. pneumoniae liver abscess.22 In this study, the proportion of diabetes in the underlying diseases was the highest.23 In our study, empirical anti-infection therapy was mainly compound β-lactamase inhibitor (36.0%), carbapenems (31.5%), and cephalosporins (31.3%). Singapore scholars studied patients with PLA with similar basic data treated with oral ciprofloxacin and injection of levofloxacin for 28 days. The results revealed that there was no difference in the treatment effects between the two groups.24 Scholars in China’s Taiwan region revealed that25 there was no significant difference in the treatment effect of patients with PLA and even severe infection between quinolones and compound β-lactamase inhibitors. Quinolones can also shorten the time of administration of intravenous antibiotics and hospital stay. The reason may be that the main pathogen of PLA in China’s Taiwan region and Singapore is K. pneumoniae.
The limitation of this study should also be acknowledged. In terms of the selection of empirical antibiotics, there was a lack of evaluation of the efficacy of different antibiotics in patients with the same basic data. In future research, pairing research will be conducted to provide the basis for a more reasonable and effective laboratory selection of antibiotics.
Conclusion
The present study revealed that empirical treatment of patients with community-acquired liver abscess without risk factors of ESBL infection should also adopt compound β-lactamase inhibitors and even carbapenems. These findings may provide a basis for rational and effective empirical treatment of PLA in this region.
Acknowledgments
We are particularly grateful to all the people who have given us help on our article.
Funding Statement
This study was supported by the Yantai Science and technology innovation development plan (2022YD015).
Data Sharing Statement
All data generated or analysed during this study are included in this article. Further enquiries can be directed to the corresponding author.
Ethics Approval and Consent to Participate
The study was conducted in accordance with the Declaration of Helsinki (as was revised in 2013). The study was approved by Ethics Committee of the Yuhuangding Hospital. Written informed consent was obtained from all participants.
Disclosure
The authors declare that they have no competing interests.
References
- 1.Chan DS, Archuleta S, Llorin RM, Lye DC, Fisher D. Standardized outpatient management of Klebsiella pneumoniae liver abscesses. Int J Infect Dis. 2013;17(3):e185–e188. doi: 10.1016/j.ijid.2012.10.002 [DOI] [PubMed] [Google Scholar]
- 2.Rahimian J, Wilson T, Oram V, Holzman RS. Pyogenic liver abscess: recent trends in etiology and mortality. Clin Infect Dis. 2004;39(11):1654–1659. doi: 10.1086/425616 [DOI] [PubMed] [Google Scholar]
- 3.Fung CP, Chang FY, Lee SC, et al. A global emerging disease of Klebsiella pneumoniae liver abscess: is serotype K1 an important factor for complicated endophthalmitis? Gut. 2002;50(3):420–424. doi: 10.1136/gut.50.3.420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Meddings L, Myers RP, Hubbard J, et al. A population-based study of pyogenic liver abscesses in the United States: incidence, mortality, and temporal trends. Am J Gastroenterol. 2010;105(1):117–124. doi: 10.1038/ajg.2009.614 [DOI] [PubMed] [Google Scholar]
- 5.Horiguchi K, Tsurutani Y, Sasaki T, et al. Hypervirulent Klebsiella pneumoniae infection in an elderly patient with diabetes mellitus. Geriatr Gerontol Int. 2021;21(7):590–591. doi: 10.1111/ggi.14179 [DOI] [PubMed] [Google Scholar]
- 6.Tseng CW, Chen YT, Lin CL, Liang JA. Association between chronic pancreatitis and pyogenic liver abscess: a nationwide population study. Curr Med Res Opin. 2017;33(3):505–510. doi: 10.1080/03007995.2016.1266312 [DOI] [PubMed] [Google Scholar]
- 7.Jepsen P, Vilstrup H, Schønheyder HC, Sørensen HT. A nationwide study of the incidence and 30-day mortality rate of pyogenic liver abscess in Denmark, 1977–2002. Aliment Pharmacol Ther. 2005;21(10):1185–1188. doi: 10.1111/j.1365-2036.2005.02487.x [DOI] [PubMed] [Google Scholar]
- 8.Qian Y, Wong CC, Lai S, et al. A retrospective study of pyogenic liver abscess focusing on Klebsiella pneumoniae as a primary pathogen in China from 1994 to 2015. Sci Rep. 2016;6:38587. doi: 10.1038/srep38587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Santos-Rosa OM, Lunardelli HS, Ribeiro-Junior MA. PYOGENIC LIVER ABSCESS: DIAGNOSTIC AND THERAPEUTIC MANAGEMENT. Arq Bras Cir Dig. 2016;29(3):194–197. doi: 10.1590/0102-6720201600030015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Foo NP, Chen KT, Lin HJ, Guo HR. Characteristics of pyogenic liver abscess patients with and without diabetes mellitus. Am J Gastroenterol. 2010;105(2):328–335. doi: 10.1038/ajg.2009.586 [DOI] [PubMed] [Google Scholar]
- 11.Chen N, Jin TT, Liu WN, et al. Gastric microbiota alteration in Klebsiella pneumoniae-caused liver abscesses mice. Pol J Microbiol. 2019;68(2):247–254. doi: 10.33073/pjm-2019-026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shi SH, Feng XN, Lai MC, Kong HS, Zheng SS. Biliary diseases as main causes of pyogenic liver abscess caused by extended-spectrum beta-lactamase-producing Enterobacteriaceae. Liver Int. 2017;37(5):727–734. doi: 10.1111/liv.13267 [DOI] [PubMed] [Google Scholar]
- 13.Shi S, Xia W, Guo H, Kong H, Zheng S. Unique characteristics of pyogenic liver abscesses of biliary origin. Surgery. 2016;159(5):1316–1324. doi: 10.1016/j.surg.2015.11.012 [DOI] [PubMed] [Google Scholar]
- 14.Chen SC, Lee YT, Yen CH, et al. Pyogenic liver abscess in the elderly: clinical features, outcomes and prognostic factors. Age Ageing. 2009;38(3):271–276. doi: 10.1093/ageing/afp002 [DOI] [PubMed] [Google Scholar]
- 15.Lok KH, Li KF, Li KK, Szeto ML. Pyogenic liver abscess: clinical profile, microbiological characteristics, and management in a Hong Kong hospital. J Microbiol Immunol Infect. 2008;41(6):483–490. [PubMed] [Google Scholar]
- 16.Shen DX, Wang J, Li DD. Klebsiella pneumoniae liver abscesses. Lancet Infect Dis. 2013;13(5):390–391. doi: 10.1016/S1473-3099(13)70068-9 [DOI] [PubMed] [Google Scholar]
- 17.Serraino C, Elia C, Bracco C, et al. Characteristics and management of pyogenic liver abscess: a European experience. Medicine. 2018;97(19):e0628. doi: 10.1097/MD.0000000000010628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang WJ, Tao Z, Wu HL. Etiology and clinical manifestations of bacterial liver abscess: a study of 102 cases. Medicine. 2018;97(38):e12326. doi: 10.1097/MD.0000000000012326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu L, Chen W, Lu X, Zhang K, Zhu C. Pyogenic liver abscess: a retrospective study of 105 cases in an emergency department from East China. J Emerg Med. 2017;52(4):409–416. doi: 10.1016/j.jemermed.2016.09.026 [DOI] [PubMed] [Google Scholar]
- 20.Ruiz-Hernández JJ, León-Mazorra M, Conde-Martel A, Marchena-Gómez J, Hemmersbach-Miller M, Betancor-León P. Pyogenic liver abscesses: mortality-related factors. Eur J Gastroenterol Hepatol. 2007;19(10):853–858. doi: 10.1097/MEG.0b013e3282eeb53b [DOI] [PubMed] [Google Scholar]
- 21.Lin L, Su LL, Li HH, et al. Study on quality markers and action mechanisms of inulae flos on anti-hepatitis through network pharmacology and high-performance liquid chromatography fingerprints. World J Tradit Chin Med. 2022;8:426–435. doi: 10.4103/wjtcm.wjtcm_1_22 [DOI] [Google Scholar]
- 22.Kong H, Yu F, Zhang W, Li X. Clinical and microbiological characteristics of pyogenic liver abscess in a tertiary hospital in East China. Medicine. 2017;96(37):e8050. doi: 10.1097/MD.0000000000008050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li W, Chen H, Wu S, Peng J. A comparison of pyogenic liver abscess in patients with or without diabetes: a retrospective study of 246 cases. BMC Gastroenterol. 2018;18(1):144. doi: 10.1186/s12876-018-0875-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Molton JS, Chan M, Kalimuddin S, et al. Oral vs intravenous antibiotics for patients with Klebsiella pneumoniae liver abscess: a randomized, controlled noninferiority study. Clin Infect Dis. 2020;71(4):952–959. doi: 10.1093/cid/ciz881 [DOI] [PubMed] [Google Scholar]
- 25.Chuang C, Chou SH, Wang FD, Huang YH, Tsai TH, Lin YT. Fluoroquinolones as an alternative treatment for Klebsiella pneumoniae liver abscess and impact on hospital length of stay. Int J Antimicrob Agents. 2020;56(4):106120. doi: 10.1016/j.ijantimicag.2020.106120 [DOI] [PubMed] [Google Scholar]