Abstract
Mycobacterium tuberculosis is an important respiratory pathogen the growth of which is controlled primarily by cytokine-activated macrophages. One of the principal mediators of this control is nitric oxide; however, superoxide has recently been shown to be protective in systemic mycobacterial infections. To determine whether superoxide is important in controlling M. tuberculosis during primary pulmonary infection, mice lacking the cytosolic p47phox gene (which is essential for effective superoxide production by the NADPH oxidase) were infected aerogenically. The lack of superoxide during an aerosol infection with M. tuberculosis resulted in a significant increase in bacterial growth over the early period of infection. Once antigen-specific gamma interferon-producing lymphocytes were detected in the draining lymph nodes, however, bacterial growth in the lung stopped. One interesting consequence of the lack of superoxide was an increase in neutrophilic infiltrates within the granuloma. This may be a consequence of increased tissue damage due to more rapid bacterial growth or may reflect a role for superoxide in controlling inflammation.
Chronic granulomatous disease (CGD) is an inherited defect of the NADPH oxidase in which phagocytes are unable to generate superoxide anion and its downstream metabolites. Because of the defect in this key host defense pathway, patients with CGD suffer from recurrent life-threatening bacterial and fungal infections. Based on case reports, CGD patients may also have increased susceptibility to mycobacterial infection and disease (2, 6, 9, 21, 27).
Both the gp91phox (phox encodes phagocyte oxidase) and p47phox proteins are critical components of the NADPH oxidase and have been targeted by gene disruption approaches, resulting in mice whose phagocytes are unable to produce superoxide (16, 23). These mice have been proven to be useful animal models of CGD in children but more recently have been used to study the immunopathology of mycobacteria infections (1, 26). These models, which have to date consisted of high-dose intravenous infections, have suggested differences in the susceptibility to reactive oxygen intermediates (ROI) between experimental Mycobacterium tuberculosis and M. avium infections.
Because M. tuberculosis is an important respiratory pathogen, we used the p47phox gene knockout (p47-KO) mouse model to determine whether such mice differed in susceptibility to an experimental pulmonary infection with M. tuberculosis. The results of this study indicate that the p47-KO mice undergo an early but transient period of increased susceptibility that is not apparent once acquired specific immunity is expressed. These results thus suggest that ROI may play a protective role against pulmonary tuberculosis but does so only during a brief early phase of the pulmonary immune response.
MATERIALS AND METHODS
Mice.
p47-KO mice were generated as previously described (16) and maintained in the animal facility at Colorado State University. They were given sterile food and water ad libitum and housed under specific-pathogen-free conditions in microisolator cages. Both p47-KO and wild-type littermates were derived from heterozygote parents and backcrossed five times onto the C57/BL6 lineage.
Infection.
A virulent strain of M. tuberculosis (Erdman) was grown from a low-passage-number seed lot in Proskauer-Beck liquid media to mid-log phase, aliquoted, and frozen at −70°C. Mice were infected using a Glas-Col (Terre Haute, Ind.) aerosol generator such that 100 bacteria were deposited in the lungs of each animal (8). The number of viable bacteria in the lungs of each of four mice was determined at various time points by plating serial dilutions of partial organ homogenates on nutrient Middlebrook 7H11 agar and counting colonies after 20 days of incubation at 37°C. A day 1 count was performed to determine the infecting dose.
Analysis of cellular response.
Thoracic lymph nodes from infected wild-type and p47-KO mice were harvested throughout infection. Nodes were gently disrupted and washed twice. The single-cell suspension was counted, and cells were plated at 2 × 105/ml in 96-well plates. All manipulations were performed in HEPES-buffered Dulbecco's minimal essential medium containing 10% fetal calf serum (Summit Biotechnology, Fort Collins, Colo.) and 2 mM l-glutamine (media and additives were obtained from Sigma, St. Louis, Mo.). Cells were grown with culture filtrate proteins from M. tuberculosis (25 μg/ml; obtained from J. T. Belisle) for 4 days, whereupon supernatants were frozen prior to analysis for cytokine content. Enzyme-linked immunosorbent assays (ELISAs) for gamma interferon (IFN-γ) were performed using antibody pairs from Pharmingen (San Diego, Calif.) according to the manufacturer's protocol.
Histological analysis.
The lower left lobe of the lung of each mouse was inflated with 10% neutral buffered formal saline and processed routinely for light microscopy. Sections were then stained with hematoxylin and eosin. Slides were examined by a veterinary pathologist without knowledge of experimental group and were subjectively graded for both quantity and quality of cellular accumulation. Repeat evaluations were performed to confirm that grading was reproducible.
RESULTS
p47-KO mice were initially more susceptible to an aerogenic infection with M. tuberculosis.
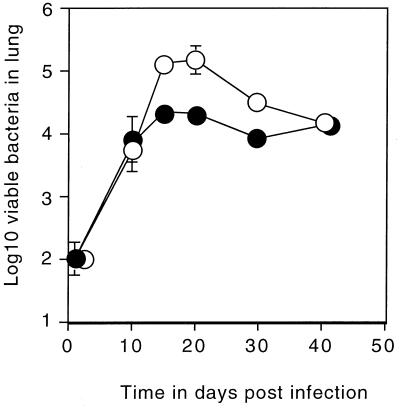
Figure 1 shows a representative growth curve for bacteria in the lungs of both wild-type and p47-KO mice. The wild-type animals demonstrated the expected ability to limit bacterial numbers to 104 per lung and maintained that control up to day 40. In contrast, in p47-KO mice a 10-fold-greater bacterial number could be detected. The difference was significant by day 14 (P < 0.05) and remained so until day 30. After that time, the infection was apparently contained and did not result in the death of any animals.
FIG. 1.
Wild-type (closed circles) and p47-KO (open circles) mice were infected with M. tuberculosis via aerosol (n = 4). p47-KO mice were more susceptible to aerogenic infection from days 15 to 30 (P ≤ 0.05). The results show data from one experiment representative of a total of three experiments.
p47-KO mice are able to generate a strong antigen-specific IFN-γ response.
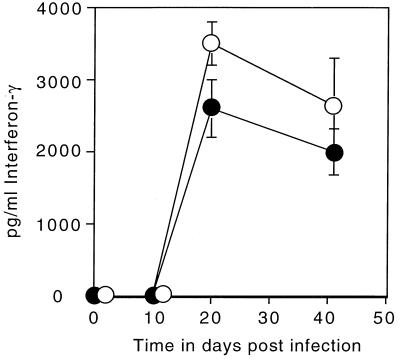
Oxygen-derived radicals such as superoxide have recently been implicated as signal transduction messengers involved in growth and apoptosis of nonphagocytic cells (10). It was therefore possible that antigen-specific cellular responses could be disrupted in the p47-KO mice. To address this question, we assessed the recruitment of antigen-specific, IFN-γ-producing lymphocytes to the lymph nodes of the infected animals. The number of cells recruited to the draining lymph node of the p47-KO mice was equivalent to the number recruited to the lymph nodes from the control mice (data not shown). More importantly, the p47-KO antigen-specific IFN-γ response was similar in kinetics and magnitude to the response from the wild-type mice (Fig. 2). The slightly increased IFN-γ response in the p47-KO mice may have resulted from the increased stimulation mediated by higher bacterial numbers in the lungs of these mice.
FIG. 2.
Thoracic lymph node cells were cultured with M. tuberculosis antigen, and the amount of IFN-γ produced was measured by ELISA. Cells from both wild-type (closed circles) and p47-KO (open circles) thoracic nodes responded to antigen on days 20 and 40. No IFN-γ was detectable in nodes taken from uninfected (day 0) or 10-day-infected mice (n = 4). The results show data from one experiment representative of a total of two experiments.
Lung tissues from p47-KO mice exhibited increased pyogranulomatous responses compared to control mice.
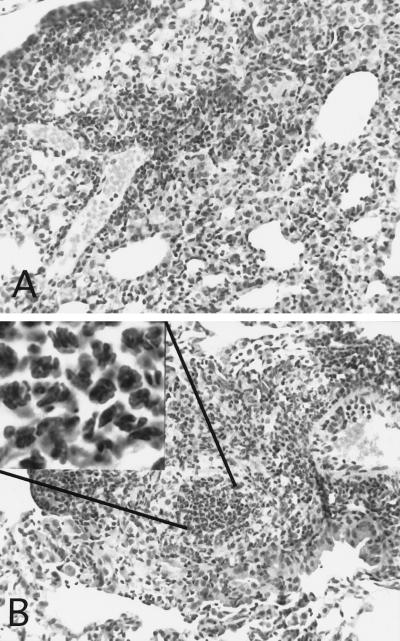
The histological appearance of lung tissues was monitored over the course of infection. In wild-type mice, mild perivascular, lymphocytic cuffing was observed at 20 days, followed at 40 days by the emergence of the traditional granulomatous response characterized by fields of epithelioid macrophages and associated lymphocytes. The response was similar in the p47-KO mice except that the day 20 response was more severe, with increased numbers of macrophages. At both day 20 and day 40, some granulomata in the p47-KO mice had increased numbers of neutrophils within the lesions (Fig. 3).
FIG. 3.
Photomicrograph of representative pulmonary lesions. (A) Lung tissue from a wild-type mouse infected 40 days previously, with typical epithelioid macrophage and lymphocyte accumulations (magnification, ×200). (B) Lung tissue from p47-KO mice 40 days after infection, with macrophage and lymphocyte accumulations as well as aggregates of neutrophils (magnification, ×200; insert magnification, ×1,000; stained with hematoxylin and eosin).
DISCUSSION
The production of ROI in tuberculosis is thought to be less important than the production of the reactive nitrogen intermediate, nitric oxide (NO). This view is supported by the results of intravenous infection models using mice in which the gene responsible for the production of large amounts of NO has been disrupted (1, 17). In addition, in vitro experiments suggest that M. tuberculosis is more readily killed by NO than by ROI (5). Although this holds true for the laboratory strains, the effects of NO on clinical isolates are more variable (24).
The results of this study suggest, however, that after aerosol infection there is a transient phase where ROI production contributes to bacterial control, in that an increased bacterial load can be observed in the lungs of the p47-KO mice. This deficit in initial control is no longer apparent when the acquired response emerges and increased IFN-γ production mediates an increase in macrophage activation (20). The presumptive mediators of the increased control are the increased expression of the inducible NO synthase (iNOS) enzyme and the acidification of the phagosome (1, 4, 11, 25). It is also possible that the lack of superoxide results in increased lymphocyte proliferation and therefore increased IFN-γ production (10) (in the present study, we noted a moderate increase in IFN-γ production in p47-KO mice) and that this masks a more profound susceptibility of p47-KO mice.
The failure to generate ROI was associated in this study with a pyogranulomatous response in the lung tissues, with noticeable accumulations of neutrophils and some abscess formation. During the early emergence of interstitial pneumonia in the lungs of aerosol-infected mice, only a few neutrophils can usually be found, despite the suggestion that they contribute to protection since their removal by monoclonal antibody administration transiently reduces resistance (22). One explanation for the accumulation seen in the current model is that the transient loss of resistance resulting from the absence of ROI results in increased local lung tissue damage, thus further promoting neutrophil influx. A second explanation relates to an intrinsic defect in controlling acute inflammatory responses in CGD. For example, in an experimental skin window model, neutrophil exudate was increased in CGD patients compared to healthy volunteers (12). In addition, in both the p47phox−/− (16) and the gp91phox−/− (23) mouse models of CGD, the KO mice exhibit increased peritoneal leukocytosis in response to thioglycolate compared to wild-type controls. Moreover, in the X-linked mouse model of CGD, enhanced neutrophil accumulation accompanied intratracheal inoculation of heat-killed Aspergillus fumigatus (19). Such increased inflammatory responses may be related to defective metabolism of the proinflammatory mediators leukotriene B4, C5a, and N-formyl peptide (7, 13, 14).
The results of the present study are highly consistent with the report by Adams and colleagues that gp91-KO mice were less resistant than wild-type mice to high-dose intravenous inoculation with M. tuberculosis but more resistant than the iNOS-KO mice (1). In the gp91-KO mice, as in the p47-KO mice used here, the disease in the lungs was associated with a pyogranulomatous response.
Tuberculosis bacteria are generally thought to be quite resistant to ROI, due to their production of both superoxide dismutase (15) and catalase (18), as well as their possession of the cell wall lipoarabinomannan, which is an excellent scavenger of oxygen radicals (3). The transient loss of resistance described above occurred very early during the pulmonary infection, when both alveolar macrophages and possibly recruited monocytes are contributing to the early development of the interstitial inflammation but long before the development of the granuloma becomes evident. At this time the bacilli are apparently vulnerable to ROI directly or to other antimycobacterial moieties that are dependent on ROI production. The latter could include factors dependent on ROI-mediated signaling or on ROI interaction with other entities such as NO. Therefore, ROI appear to have an early role in control of M. tuberculosis in the lung, helping to contain growth until specific immunity is generated.
ACKNOWLEDGMENTS
This work was supported by grant AI40488 from the National Institute of Allergy and Infectious Disease, National Institutes of Health.
We gratefully acknowledge the provision of culture filtrate protein antigens by John Belisle under NIH contract AI75320.
REFERENCES
- 1.Adams L B, Dinauer M C, Morgenstern D E, Krahenbuhl J L. Comparison of the roles of reactive oxygen and nitrogen intermediates in the host response to Mycobacterium tuberculosis using transgenic mice. Tuberc Lung Dis. 1997;78:237–246. doi: 10.1016/s0962-8479(97)90004-6. [DOI] [PubMed] [Google Scholar]
- 2.Allen D M, Chng H H. Disseminated Mycobacterium flavescens in a probable case of chronic granulomatous disease. J Infect. 1993;26:83–86. doi: 10.1016/0163-4453(93)97000-n. [DOI] [PubMed] [Google Scholar]
- 3.Chan J, Fan X, Hunter S W, Brennan P J, Bloom B R. Lipoarabinomannan, a possible virulence factor involved in the persistence of Mycobacterium tuberculosis within macrophages. Infect Immun. 1991;59:1755–1761. doi: 10.1128/iai.59.5.1755-1761.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chan J, Tanaka K, Carroll D, Flynn J L, Bloom B R. Effects of nitric oxide synthase inhibitors on murine infection with Mycobacterium tuberculosis. Infect Immun. 1995;63:736–740. doi: 10.1128/iai.63.2.736-740.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chan J, Xing Y, Magliozzo R, Bloom B R. Killing of virulent Mycobacterium tuberculosis by reactive nitrogen intermediates produced by activated murine macrophages. J Exp Med. 1992;175:1111–1122. doi: 10.1084/jem.175.4.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chusid M J, Parillo J E, Fauci A S. Chronic granulomatous disease. Diagnosis in a 27-year-old man with Mycobacterium fortuitum. JAMA. 1975;233:1295–1296. doi: 10.1001/jama.233.12.1295. [DOI] [PubMed] [Google Scholar]
- 7.Clark R A, Klebanoff S J. Chemotactic factor inactivation by the myeloperoxidase-hydrogen peroxide-halide system. J Clin Investig. 1979;64:913–920. doi: 10.1172/JCI109557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cooper A M, Callahan J E, Griffin J P, Roberts A R, Orme I M. Old mice are able to control low-dose aerogenic infections with Mycobacterium tuberculosis. Infect Immun. 1995;63:3259–3265. doi: 10.1128/iai.63.9.3259-3265.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Esterly J R, Sturner W Q, Esterly N B, Windhorst D B. Disseminated BCG in twin boys with presumed chronic granulomatous disease of childhood. Pediatrics. 1971;48:141–144. [PubMed] [Google Scholar]
- 10.Finkel T. Signal transduction by reactive oxygen species in non-phagocytic cells. J Leukoc Biol. 1999;65:337–340. doi: 10.1002/jlb.65.3.337. [DOI] [PubMed] [Google Scholar]
- 11.Flynn J L, Chan J, Triebold K J, Dalton D K, Stewart T A, Bloom B R. An essential role for interferon gamma in resistance to Mycobacterium tuberculosis infection. J Exp Med. 1993;178:2249–2254. doi: 10.1084/jem.178.6.2249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gallin J I, Buescher E S, Seligmann B E, Nath J, Gaither T, Katz P. NIH conference. Recent advances in chronic granulomatous disease. Ann Intern Med. 1983;99:657–674. doi: 10.7326/0003-4819-99-5-657. [DOI] [PubMed] [Google Scholar]
- 13.Hamasaki T, Sakano T, Kobayashi M, Sakura N, Ueda K, Usui T. Leukotriene B4 metabolism in neutrophils of patients with chronic granulomatous disease: phorbol myristate acetate decreases endogenous leukotriene B4 via NADPH oxidase-dependent mechanism. Eur J Clin Investig. 1989;19:404–411. doi: 10.1111/j.1365-2362.1989.tb00249.x. [DOI] [PubMed] [Google Scholar]
- 14.Henderson W R, Klebanoff S J. Leukotriene production and inactivation by normal, chronic granulomatous disease and myeloperoxidase deficient neutrophils. J Biol Chem. 1983;258:13522–13527. [PubMed] [Google Scholar]
- 15.Jackett P S, Aber V R, Lowrie D B. Virulence and resistance to superoxide, low pH and hydrogen peroxide among strains of Mycobacterium tuberculosis. J Gen Microbiol. 1978;104:37–45. doi: 10.1099/00221287-104-1-37. [DOI] [PubMed] [Google Scholar]
- 16.Jackson S H, Gallin J I, Holland S M. The p47phox mouse knock-out model of chronic granulomatous disease. J Exp Med. 1995;182:751–758. doi: 10.1084/jem.182.3.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.MacMicking J D, North R J, LaCourse R, Mudgett J, Shah S K, Nathan C F. Identification of NOS2 as a protective locus against tuberculosis. Proc Natl Acad Sci USA. 1997;94:5243–5248. doi: 10.1073/pnas.94.10.5243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Middlebrook G. Isoniazid-resistance and catalase activity of tubercle bacilli. Am Rev Tuberc. 1954;69:471–472. doi: 10.1164/art.1954.69.3.471. [DOI] [PubMed] [Google Scholar]
- 19.Morgenstern D E, Gifford M A, Li L L, Doerschuk C M, Dinauer M C. Absence of respiratory burst in X-linked chronic granulomatous disease mice leads to abnormalities in both host defense and inflammatory response to Aspergillus fumigatus. J Exp Med. 1997;185:207–218. doi: 10.1084/jem.185.2.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nathan C F. Interferon gamma and macrophage activation in cell mediated immunity. In: Steinman R M, North R J, editors. Mechanisms of host resistance to infectious agents, tumours and allografts. New York, N.Y: The Rockefeller University Press; 1986. pp. 165–184. [Google Scholar]
- 21.Ohga S, Ikeuchi K, Kadoya R, Okada K, Miyazaki C, Suita S, Ueda K. Intrapulmonary Mycobacterium avium infection as the first manifestation of chronic granulomatous disease. J Infect. 1997;34:147–150. doi: 10.1016/s0163-4453(97)92509-3. [DOI] [PubMed] [Google Scholar]
- 22.Pedrosa J, Saunders B M, Appelberg R, Orme I M, Silva M T, Cooper A M. Neutrophils play a protective, nonphagocytic, role in systemic Mycobacterium tuberculosis infection of mice. Infect Immun. 2000;68:577–583. doi: 10.1128/iai.68.2.577-583.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pollock J D, Williams D A, Gifford M A C, Lui L L, Du X, Fisherman J, Orkni S H, Doerschuk C M, Dinauer M C. Mouse model of X-linked chronic granulomatous disease, an inherited defect in phagocyte superoxide production. Nat Genet. 1995;9:202–209. doi: 10.1038/ng0295-202. [DOI] [PubMed] [Google Scholar]
- 24.Rhoades E R, Orme I M. Susceptibility of a panel of virulent strains of Mycobacterium tuberculosis to reactive nitrogen intermediates. Infect Immun. 1997;65:1189–1195. doi: 10.1128/iai.65.4.1189-1195.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schaible U E, Sturgill-Koszycki S, Schlesinger P H, Russell D G. Cytokine activation leads to acidification and increases maturation of Mycobacterium avium-containing phagosomes in murine macrophages. J Immunol. 1998;160:1290–1296. [PubMed] [Google Scholar]
- 26.Segal B H, Doherty T M, Wynn T A, Cheever A W, Sher A, Holland S M. The p47phox−/− mouse model of chronic granulomatous disease has normal granuloma formation and cytokine responses to Mycobacterium avium and Schistosoma mansoni eggs. Infect Immun. 1999;67:1659–1665. doi: 10.1128/iai.67.4.1659-1665.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Smith P A, Wittenberg D F. Disseminated BCG infection in a child with chronic granulomatous disease. A case report. S Afr Med J. 1984;65:821–822. [PubMed] [Google Scholar]