Abstract
Introduction
Motion preserving atlas ring osteosynthesis (C1-RO) for unstable Jefferson burst fractures (JBF) with insufficiency of the transverse atlantal ligament (TAL) is under debate. There is controversy about when to apply C1-RO and when further stabilization is needed.
Research question:
Is intraoperative stability testing after C1-RO with restoration of secondary stabilizers feasible, and what are mid-to long-term results of posterior C1-RO vs. C1–C2 ORIF in unstable Jefferson burst fractures with Dickman type I or II transverse atlantal ligament lesions based on intraoperative decision using this stability testing?
Material and methods
Five consecutive patients with unstable JBF were treated with posterior C1-RO or C1–C2 ORIF based on the findings after intraoperative reduction and posterior C1-RO and stability testing. This newly developed intraoperative stability test based on the findings of biomechanical studies is a fluoroscopically controlled manual C1–C2 test with a force of approximately 50 N posterior-anterior stress and a tilting maneuver after C1-RO with repositioning. Clinical and radiological results of the cases with C1-RO were analyzed 3.5–21 months postoperatively.
Results
Posterior C1-RO was performed in four patients. One case required C1–C2 fixation due to significant instability. In cases of C1-RO, stable bony fusions of the atlas ring were observed within a year. In flexion-extension views, the anterior atlanto-dental interval (AADI) did not increase until the latest follow-up. No complications were observed.
Discussion and conclusion
The described intraoperative stability test after posterior C1-RO in unstable JBF enables the determination if C1-RO is sufficient or C1–C2 ORIF is necessary for treatment.
Keywords: Motion preserving, Intraoperative stability test, C1-ring osteosynthesis, Unstable jefferson burst fracture, C1-RO
Highlights
-
•
Intraoperative stability testing after atlas ring osteosynthesis (C1-RO) with restoration of secondary stabilizers as a useful concept in treating unstable JBF operatively.
-
•
Intraoperative stability testing to gain better confidence of sufficient stability or proving insufficient stability after C1-RO in treatment of unstable JBF.
-
•
New concept to further evaluate instability of unstable JBF beyond theoretical instability for specific treatment.
1. Introduction
Conservative treatment of stable JBF has a role, as long Halo treatment in geriatric patients can be avoided. For conservative treatment of unstable JBF though, Halo treatment after closed reduction is necessary but has a high complication rate (Kandziora et al., 2017) and shows inferior results compared with operative management (Dvorak et al., 2005), especially when persisting instability or non-union results in deformity extending from C0 to C2. When modern operative treatment is not available or feasible, Halo treatment remains an option that may restore at least partly C1–C2 function.
In cases of unstable Jefferson burst fractures (JBF), posterior atlas-ring osteosynthesis (C1-RO) is an operative treatment option that enables C1–C2 functional preservation. Ruf et al. (2004) firstly described anterior C1-RO in 2004 and, since then, posterior (Abeloos et al., 2011; Li-Jun et al., 2011; Jo et al., 2011) and combined posterior-anterior (Böhm et al., 2006) C1-RO have also been reported. To date, the only commonly accepted indications for C1-RO are Dickman type II transverse atlantal ligament (TAL) lesions in unstable JBF (Kandziora et al., 2017), as there are concerns that a Dickman type I lesion might not heal after C1-RO, thus leading to late C1–C2 instability (Kandziora et al., 2017; Dickman et al., 2004).
On the other hand, a report of long-term outcomes (average 17 months) in 12 cases of mixed type Dickman TAL lesions revealed, that C1-RO procedures were not followed by any late sequelae of C1–C2 instability in Dickman type I or Dickman type II lesions (Shatsky et al., 2016). Based on these findings, in a 2017 review (Kandziora et al., 2017), Kandziora et al. remarked that “the current state-of-the-art concept of performing an atlantoaxial fusion in unstable atlas fractures with a Dickman Type I rupture of the TAL has to be challenged. Maybe, an isolated motion preserving atlas osteosynthesis might be sufficient in these cases. Further studies have to prove whether atlas osteosynthesis has the potential to shift a paradigm”.
Indeed a very recent (2022) study from China by Yan et al. (Yan et al., 2022) has achieved a prospective randomized controlled study to compare 37 C1- RO with 36 C1-2 fixation fusions for unstable atlas fractures and found C1-RO superior over C1–C2 fixation fusion for neck pain, operation time, intraoperative blood loss, radiation dose, bedridden period, hospital stay, with higher neck Disability index and better function in flexion-extension and axial rotation in a 5 year follow-up with a follow-up rate of more than 70%.
So it seems, that there is already evidence to shift the paradigm towards C1-RO. But there is a detail that this study has not taken into account: in the 2010 biomechanical study by Koller et al. (2010) and a 2014 biomechanical study of the longitudinal ligaments by Li-Jun et al. (2014) it was proven that secondary stabilizers in C1-RO with reduction can provide sufficient stability under physiologic loads, as a rationale for C1-RO. Due to the biomechanical compressive nature of the trauma in JBF, secondary longitudinal stabilizers would remain generally intact. However, Koller et al. (2010) also stated that “the results derived from the current study do not preclude individual assessment of the strength of the secondary stabilizers in unstable JBF with TAL disruption; neither vertical stability when conservative treatment nor a C1-RO is considered.”
This leads to the remaining question whether C1-RO with reduction of unstable JBF achieves sufficient immediate stability in the individual case, or if it is necessary to perform further stabilization via C1–C2 fixation and/or fusion or even C0–C2 stabilization and if this procedure then provides long-term stability, regardless of the Dickman type TAL lesion.
In 2017, we introduced an intraoperative stability assessment test for use during C1-RO (Ottenbacher and Bettag, 2017) for unstable JBF. Since then, five patients with this rather rare condition have been treated in our institution, with the intraoperative stability assessment used to decide whether a C1-RO was sufficient or if further stabilization was needed.
In the present study, we aimed to evaluate the mid-to long-term results of C1-RO based on the findings of the intraoperative stability test, in cases of unstable JBF with Dickman type I and II or indetermined TAL lesions.
2. Methods
2.1. Intraoperative stability test
Analogous to the biomechanical study of Koller et al. (2010), we have developed an intraoperative stability test. In the study by Koller at al (Koller et al., 2010), they translationally applied a physiological range of forces from 10 to 50 N to C1–C2, as well as a force of 100 N, assumed to be the worst case. Therefore, we chose to intraoperatively test approximately 50 N, applied bimanually by the surgeon, with anterior atlanto-dental interval (AADI) controlled by dynamic fluoroscopy. Our case report from 2017 (Ottenbacher and Bettag, 2017) includes a brief description of the intraoperative stability test. Here we provide a detailed description of the intraoperative stability test, with recommendations for reduction.
-
1.
Preoperatively, MRI or CT scanning (including angio-investigations) is used to evaluate the spinal canal width at the C1 level, to assess the reserve space anterior and posterior of the myelon, to exclude relevant intraspinal hematoma, and to evaluate the patency of vertebral arteries. In cases of suspected Dickman type I lesion, a preoperative MRI (Dickman et al., 1991) is advised for verification.
-
2.
In cases with severely displaced, tangled, or locked fractures, preoperative distraction may be an option to attempt to reduce those fractures before the operation. However, all necessary investigations (mainly MRI) must be completed before application of Crutchfield or Halo ring distraction.
-
3.
In the OR, the Mayfield clamp is applied. Heart rate and blood pressure must be monitored, and testing should be paused upon any suspicious observations. With the patient in the supine position, dosed distraction is applied under dynamic fluoroscopic control. Longitudinal instability of the C0–C1–C2 complex should be excluded, and an attempt to reduce the fracture undertaken. Simultaneous extension and distraction and manual pressure to the transverse processes of the atlas may be applied. The patient should then be turned to the prone position for ideal positioning for screw insertion. Then, the Mayfield clamp is locked and the patient's head will rest in the Mayfield clamp during the whole procedure.
-
4.
Typical open and limited preparation is performed for the entry points of the lateral mass screws, and fluoroscopic or navigated (3D-scan) insertion of half-thread 3.5-mm polyaxial screws is conducted, ideally bicortically (Eck et al., 2007). At this point, no further preparation should be undertaken for possible C2 screws or an occipital plate. A good bony screw purchase is advised for further intraoperative repositioning attempts and, finally, for the stability test. Monoaxial screws have been proven to show better ability for reduction (Chung et al., 2011; Gumpert et al., 2017; Zhang et al., 2018; Gelinas-Phaneuf et al., 2018). However, 3.5-mm or 4-mm monoaxial screws have not yet been available to us. Therefore, we attempted to reduce the typically anterolaterally displaced lateral mass with the use of a “monoaxial” thread cutter in a closing anterior wedge maneuver. Koller (2019) has described another maneuver to reduce a displaced unstable JBF with the aid of a hinged rod. Posterior compression over the transversely applied rod, with the use of a compression plier to reduce the posterior C1 arch, completes reduction. Complete reduction often cannot be achieved; however, still stability testing may be continued. If available, a 3D scan may be utilized to assess bony screw purchase and/or achieved intraoperative repositioning.
-
5.
A Kocher clamp is attached to the spinous process of C2, while another clamp grasps the transverse rod of the C1-RO. Under dynamic fluoroscopic control, C1 is held in place, while C2 is pushed downwards to a mechanical stop, thus identifying the minimum AADI. Then C1 is held in place, while C2 is lifted upwards. Separate testing with a forcemeter and two Kocher clamps has demonstrated that by using this bimanual maneuver in the OR setting, a surgeon may comfortably test a range of 10–50 N with submaximal effort. After several practice attempts, the surgeon was able to repeatedly reproduce a load of approximately 50 N bimanually and blindly. The AADI is fluoroscopically controlled, while the surgeon experiences haptic feedback of the secondary stabilizers (Video 3A, 4A and 5A). This procedure requires careful evaluation of AADI and posterior atlanto-dental interval (PADI) with respect to the assessed reserve space anterior and posterior of the myelon. Another method of testing is to reversely push on the transverse rod while holding C2 in place.
-
6.
Destructive forces should be avoidable as long as the surgeon closely observes the fluoroscopy, and force application is slowly increased to a maximum of approximately 50 N. In our series, we observed no fracture of the C2 spinous process, and no cutting or pulling out of the lateral mass screws.
-
7.
Tilting of the C1 construct over C2 should be tested next, as tilting has been observed as a possible failure pattern (Koller et al., 2010). For this purpose, C2 is held in place while a downward tilting of C1 is carried out under dynamic fluoroscopic control (Video 3B, 4B and 5B in the supplemental material). Eventually the transverse rod might touch the C2 lamina. In pathologic cases, C1 might slide anteriorly with signs of an anterior gapping of the AADI.
-
8.
Videorecording and replay of the fluoroscopy may be also helpful.
-
9.
Maximum AADI should be expected to be 1.5 mm greater than in TAL intact conditions (Koller et al., 2010), until secondary longitudinal stabilizers come into full effect. Thus, an AADI of up to 5 mm is acceptable as long as the C1 reserve spaces are large enough.
-
10.
If failure patterns are observed – such as tilting of C1–C2 with anterior gapping, an AADI of >5 mm with translational force, or other suspect motion – the surgery should be converted to C1–C2 ORIF, ideally with a cross connector at the C1 level (Tessitore et al., 2008), preferably without fusion.
The following are the supplementary data related to this article:
2
3
4
5
2.1.1. Validation
The method could yet not be validated in our setting, as a sterile force meter for measuring the actually applied posterior-anterior C1–C2 force would be necessary, which was not available for us. Furthermore there would be problems with coupling the two directions in one forcemeter. Validating the tilting of C1–C2 is even harder to realize. Therefore this study lacks a formal control group, but the first author has on many occaisons before in operating on “stable” C1–C2 conditions assessed the tightness of C1–C2 with the aid of 2 Kocher clamps intraoperatively to get an idea of the remaining overall mobility. Additionaly the fluoroscopically controlled anterior atlantodental interval (AADI) and the tilting angles in the operative seting is not measurable exactly without calibrated instruments and x-rays.
Therefore the stability testing should be considered a “surgical skill” similar to testing the achieved tightness of the knee after orthopaedic crucial ligament repair for example.
The forces in the biomechanical study of Koller et al. though were controlled quite well, and our intraoperative stability test is a direct translation of these findings to the OR.
All radiologic measurements were measured using our PACS software (Sectra Workstation IDS7 Version 22.2.4.4150 (x64) patch 2020, Sectra AB, Sweden).
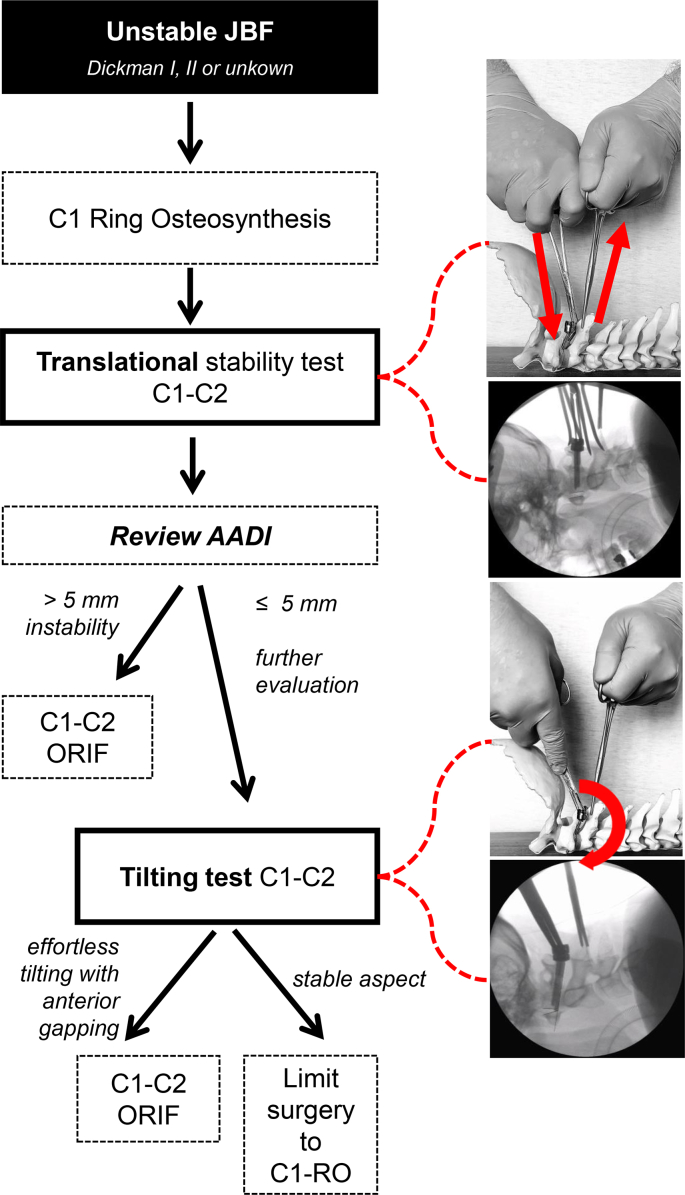
2.2. Algorithm in the treatment of unstable JBF
Graphic 1 presents the graphic summary for algorithmic use in the treatment of unstable JBF in our study.
Graphic 1.
Algorithm of intraoperative stability testing in the treatment of unstable JBF.
Additional information is provided in illustrative cases and supplemental material. Video 3, Video 4, Video 5 show the intraoperative stability testing of patients #3, #4, and #5.
3. Results
3.1. Subjects and indications for surgery
Between 2017 and 2019, five consecutive patients with unstable JBF underwent posterior C1-RO after intraoperative reduction. Intraoperative stability assessment (see Methods) was conducted applying the previously described technique (Ottenbacher and Bettag, 2017). The stability test findings were used to determine whether the operation was finished as C1-RO or required conversion to further stabilization (Graphic 1).
Patients with 2-, 3-, or 4-part atlas fractures (Jefferson burst fractures or Jefferson-like burst fractures, both abbreviated here as JBF), who were diagnosed at or referred to our institution, were initially immobilized using a rigid collar. Then the patients underwent neurological evaluation. None of the five patients had any neurologic deficit with respect to the JBF. One patient (#3) exhibited mild transient myelopathic deficits (ASIA Grade D) related to a myelon contusion at the C5/6 level, which didn't require subaxial operative treatment. Each patient also underwent vascular examination, by Doppler flow sonography and/or angio-MRI and/or angio-CT, to evaluate the vertebral arteries. One patient (#1) exhibited an occlusion/dissection of the left vertebral artery in the V3 segment, and this patient was initially treated with therapeutic perfusion of unfractionated heparin, as previously described in a case report (Ottenbacher and Bettag, 2017).
Within 2–7 days (mean, 4.2 days), all patients underwent radiological evaluation with an upright anteroposterior (AP) dens view in the rigid collar to measure lateral mass displacement (LMD) bilaterally for calculation of the rule of Spence (Spence et al., 1970) (ROS). TAL insufficiency was assumed at a threshold of ≥8.1 mm (Heller et al., 1993; Woods et al., 2018), and the fracture was classified as an unstable JBF. CT scans revealed that three of the five patients (patients #2, #3, and #4) showed a bony avulsion of the TAL, and thus these patients’ TAL lesions were classified as Dickman type II lesions. Patient #1 underwent MRI, which confirmed an intraligamentous (midsubstance) tear of the TAL, which was classified as a Dickman type I lesion. Patient #4 initially received conservative treatment for 10 weeks in a rigid collar with initial LMD and ROS of 3.3 mm, but was readmitted for surgery due to increasing pain and LMD resulting in an ROS of 8.9 mm. In this case, the alar ligaments also showed bony avulsions at the condyles. Patient #5 suffered from a more distorted and locked dislocation of his 2-part JBF, and was placed under halo-distraction of ≥8 kg traction for 2 days to achieve reduction. Distraction was continued into the OR and, therefore, MRI was no longer feasible. Due to the wide displacement of fragments without signs of bony avulsion, patient #5 was assumed to have a TAL Dickmann type I lesion. Table 1 presents the patient data and fracture characteristics.
Table 1.
Patient and fracture characteristics.
| Pt. | Age/Gen | JBF | TAL lesion (Dg. Mod.) | ROS | LMD right | LMD left |
|---|---|---|---|---|---|---|
| 1 | 70 m | 4 part | Dickman I (MRI) | 12.6 mm | 6.2 mm | 6.4 mm |
| 2 | 47 m | 3 part | Dickman II (CT) | 11.4 mm | 6.4 mm | 5.0 mm |
| 3 | 47 m | 2 part | Dickman II (CT) | 13.5 mm | 5.0 mm | 8.5 mm |
| 4 | 75 f | 3 part | Dickman II (CT) | 8.9 mm | 5.8 mm | 3.1 mm |
| 5 | 65 m | 2 part | Dickman I (ROS) | 8.2 mm | 2.4 mm | 5.8 mm |
Pt = patient #; Gen = gender; m = male; f = female; JBF = Jefferson burst fracture; TAL = transverse atlantal ligament; ROS = rule of Spence; LMD = lateral mass displacement; MRI = magnetic resonance imaging; CT = computed tomography.
3.2. Surgical results
Of the 5 consecutive patients (mean age, 61 years; age range 47–75 years; 4 male and 1 female), the operation time and blood loss was recorded. No patient suffered from any minor or major surgical or medical adverse event during or after the operation. Mean operation time of the C1-ring osteosyntheses, including stability testing, was 106.5 min. The mean blood loss during C1-ring osteosyntheses was 500 ml. Table 2 presents operative and postoperative surgical and medical results.
Table 2.
Operative and postoperative surgical and medical results.
| Pt. | Operation | Adverse Events | Operation Time (min) | Blood loss (ml) |
|---|---|---|---|---|
| 1 | C1-RO | none | 136 | 200 |
| 2 | C1-RO | none | 83 | 100 |
| 3 | C1–C2 ORIF | none | 148 | 400 |
| 4 | C1-RO | none | 107 | 1200 |
| 5 | C1-RO | none | 110 | 500 |
Pt. = patient #; C1-RO = atlas-ring osteosynthesis; C1–C2 ORIF = open reduction and internal fixation.
3.3. Course and results of intraoperative stability testing after C1-RO with reposition
For each patient, intraoperative stability testing was performed under dynamic fluoroscopic control. Patients #1 and #2 underwent operations in our former operation theatre. In August 2017 we moved to our new facilities, in which videorecording of the dynamic fluoroscopy was possible. Thus video recordings are available for patients #3, #4, and #5, which can be viewed in the supplemental material section online (Video 3A, 3B, 4A, 4B, 5A and 5B). For patients #1 and #2, we have only one static view of the testing for each. Table 3 displays the results of the intraoperative testing.
Table 3.
Results of intraoperative stability testing after posterior C1-RO.
| Pt. | C1–C2 Translation test | C1–C2 Tilting test | |
|---|---|---|---|
| AADI max | AADI min | Tilting C1 over C2 ( degree) | |
| 1 | 2 mm | 1 mm | minimal – stable aspect |
| 2 | 2 mm | 1 mm | minimal – stable aspect |
| 3 | 3 mm | 2 mm | 6 with 3 mm anterior gapping |
| 4 | 3 mm | 2 mm | minimal – stable aspect |
| 5 | 5 mm | 3 mm | minimal – stable aspect |
Pt = patient #; C1-RO = atlas-ring osteosynthesis; AADI = anterior atlanto-dental interval.
Based on the C1–C2 Translation tests, all patients were classified as stable, with acceptance of an AADI of up to 5 mm. Patients #1, #2, #4, and #5 exhibited no pathologic tilting. In patient #3, we observed a tilting of 6 of the C1 construct over C2, with a concomitant anterior wedge-shaped atlanto-dental gapping of 3 mm. The observed tilting instability was reproducible by applying minimal force. Therefore patient #3 recieved C1-C2 ORIF (see treatment algorithm (Graphic 1)).
3.4. Radiological results
All patients underwent a postoperative CT scan for evaluation of implants, and a postoperative upright AP dens view without a collar to measure LMD bilaterally for calculation of the remaining ROS (Spence et al., 1970), i.e., radiologic reduction/repositioning of the lateral masses (Table 4).
Table 4.
Postoperative findings—radiologic reposition in upright anteroposterior dens view.
| Pt. | Operation | ROS p.o. | ROS pre-op/p.o. | LMD right p.o. | LMD left p.o. |
|---|---|---|---|---|---|
| 1 | C1-RO | 5.3 mm | −7.3 mm | 2.3 mm | 3.0 mm |
| 2 | C1-RO | 5.0 mm | −6.4 mm | 2.8 mm | 2.3 mm |
| 3 | C1–C2 ORIF | 6.9 mm | −6.6 mm | 3.0 mm | 3.9 mm |
| 4 | C1-RO | 3.1 mm | −5.8 mm | 1.6 mm | 1.5 mm |
| 5 | C1-RO | 5.4 mm | −2.8 mm | 1.0 mm | 4.4 mm |
Pt = patient #; ROS = rule of Spence; LMD = lateral mass displacement; p.o. = postoperative; C1-RO = atlas-ring osteosynthesis; C1–C2 ORIF = open reduction and internal fixation.
Patients underwent radiologic evaluation with postoperative flexion-extension views at 3–7 days postoperatively (mean, 4.8 days). These flexion-extension views were obtained both postoperatively and at the time of most recent follow-up. Patient #2, a construction worker, showed up only once after discharge. At 3.5 months after the accident, flexion-extension views, an upright AP dens view, and a CT scan were carried out, and then this patient was lost to follow-up. The other patients had follow-up examinations at 10–21 months postoperatively. Clinical findings, namely pain and ability to rotate the head in degrees, were also analyzed at 3.5–21 months postoperatively. Postoperatively and at the time of latest follow-up (3.5–21 months postoperatively), upright AP dens views and CT scans were obtained for measurement of LMD and signs of fusion.
Patients #1, #2, and #4 had incomplete but satisfactory reduction of the fracture. Patient #3 had an acceptable reduction with C1–C2 ORIF. Patient #5 had a rather irreducible fracture anteriorly at the left side.
Flexion-extension views were obtained at 3–7 days postoperatively (mean, 4.8 days), and at the latest follow-up (mean, 11.6 months; range, 3.5–21 months). These images were analyzed for AADI in flexion and extension. CT scans were also obtained at latest follow-up for analysis of stable bony fusions of the atlas ring (Table 5).
Table 5.
Postoperative and follow-up findings—radiologic AADI in flexion-extension (flex/ex) views and findings of CT scans.
| Pt. | Operation | AADI flex/ex p.o. (days) | AADI flex/ex follow-up (months) | Stable fusion (months) |
|---|---|---|---|---|
| 1 | C1-RO | 2 mm/1 mm (3) | 2 mm/1 mm (21) | yes (21) |
| 2 | C1-RO | 2 mm/1 mm (3) | 1 mm/1 mm (3.5) | yes (3.5) |
| 3 | C1–C2 ORIF | -/- | -/- | yes (12) |
| 4 | C1-RO | 1 mm/1 mm (6) | 2 mm/1 mm (12) | yes (12) |
| 5 | C1-RO | 5 mm/3 mm (7) | 4 mm/3 mm (10) | yes (10) |
AADI = anterior atlanto-dental interval; Pt. = patient #; p.o. = postoperative; CT = computed tomography; C1-RO = atlas-ring osteosynthesis; C1–C2 ORIF = open reduction and internal fixation.
The flexion-extension views obtained postoperatively and at latest follow-up revealed that AADI in flexion and extension at these two time-points remained within a maximal difference of 1 mm compared to the corresponding intraoperative measurements of maximum and minimum AADI. In flexion, the values were identical or lower than the intraoperative AADI maximum (see Discussion). The CT scans revealed stable fusions of the C1 ring in all cases of C1-RO. In patient #4, an anterior gap remained at the left side, but the posterior gap of the 2-part fracture was reduced and fused and, therefore, the C1 ring was classified as stable overall.
3.5. Clinical results
We assessed the clinical findings at the latest follow-up (Table 6). The overall ability to rotate the head in a sitting position with the shoulders fixed was measured in degrees (, using a protractor viewed from above, with the nose as the pointing reference. The patients were mostly free of pain at follow-up. Patient #2 reported minor pain at 3.5 months postoperatively, but pain medication was not necessary, he then continued working as a construction worker. Patient #3, who underwent C1–C2 ORIF, also had minor pain at the latest follow-up, but also did not require pain medication.
Table 6.
Clinical findings—clinical assessments.
| Pt. | Operation | Head rotation at follow-up (months) | Neck Pain at follow-up (months) |
|---|---|---|---|
| 1 | C1-RO | 70 (21) | no (21) |
| 2 | C1-RO | 40 (3.5) | minor (3.5) |
| 3 | C1–C2 ORIF | not assessed | minor (12) |
| 4 | C1-RO | 48 (12) | no (12) |
| 5 | C1-RO | 55 (10) | no (10) |
Pt. = patient #; C1-RO = atlas-ring osteosynthesis; C1–C2 ORIF = open reduction and internal fixation.
3.6. Illustrative case C1-RO (patient #4)
After reduction of the 3-part fracture, and stabilization with bicortical lateral mass screws with posterior compression, a Kocher clamp was attached to the spinous process of C2. Another Kocher clamp grasped the transverse rod of the C1-RO. While C1 was held in place, C2 was pushed down to a mechanic stop. The resulting AADI under dynamic fluoroscopic control was noted as the minimum AADI (Fig. 1). Then C1 was held in place, while C2 was lifted up with a slowly increasing effort under dynamic fluoroscopic control (Video 4A in the supplemental material). Maximum AADI was estimated to be 3 mm (Fig. 2), at which point greater force did not further increase the AADI. There was a haptic feeling of a soft stop that resisted the increasing force. The estimated maximum effort was 50 N, as previously trained by the surgeon with a forcemeter. Tilting of the construct over C2 was within limits, and without anterior gapping (Video 4B in the supplemental material). Hence, the operation was finalized with wound closure.
Fig. 1.
Translational intraoperative stability test C1–C2 in patient #4 (screenshots of dynamic fluoroscopy). Minimum anterior atlanto-dental interval (AADI).
Fig. 2.
Translational intraoperative stability test C1–C2 in patient #4 (screenshots of dynamic fluoroscopy). Maximum AADI.
3.7. Illustrative Case C1-2 ORIF (Patient#3)
After closed reduction of the 2-part fracture, and stabilization with bicortical lateral mass screws with posterior compression, a Kocher clamp was attached to the spinous process of C2. Rod-holding forceps were used to grasp the transverse rod of the C1-RO. C1 was held in place, while C2 was pushed down to a mechanic stop. The resulting AADI under dynamic fluoroscopic control was noted as the minimum AADI (Fig. 3). Then C1 was held in place, while C2 was lifted up with a slowly increasing effort under dynamic fluoroscopic control (Video 3A in the supplemental material). The maximum AADI was estimated to be 3 mm (Fig. 4), at which point additional force did not further increase the AADI. There was a haptic feeling of a soft stop that resisted the increasing force. The estimated maximum effort was 50 N, as previously trained by the surgeon with a forcemeter.
Fig. 3.
Translational intraoperative stability test C1–C2 in patient #3 (screenshots of dynamic fluoroscopy). Minimum AADI.
Fig. 4.
Translational intraoperative stability test C1–C2 in patient #3 (screenshots of dynamic fluoroscopy). Maximum AADI.
The maximum AADI would have been acceptable for C1-RO, but we then observed a tilting instability of the atlas over the axis of Δ6° with 2 mm AADI at minimum (neutral) tilt (Fig. 5), and 3 mm anterior atlanto-dental wedge-shaped gapping at maximum tilt (Fig. 6). This tilting instability was reproducible by applying minimal force (Video 3B in the supplemental material). Thus, this instability was then treated by conversion to C1–C2 ORIF with a cross connector, as proposed by Tessitore et al. (2008).
Fig. 5.
Tilting intraoperative stability test C1–C2 in patient #3. Minimum tilt.
Fig. 6.
Tilting intraoperative stability test C1–C2 in patient #3. Maximum tilt.
4. Discussion
4.1. Conservative (non-operative) treatment
The role of conservative treatment has been lined out in the introduction section. When modern operative therapy is not available, conservative treatment is a viable option, but should include Halo-traction after closed reduction with timely follow-ups to avoid the high complication rates and persisting instability respectively non-union under this treatment. It has to be accepted though, that operative methods like the aforementioned stability testing and C1-RO with the aid of sophisticated implants and tools and maybe navigation is yet far from being widespread in most parts of the world.
4.2. Radiological and clinical results
Our present radiological and clinical results are similar to previous findings (Li-Jun et al., 2011; Jo et al., 2011; Shatsky et al., 2016; Gelinas-Phaneuf et al., 2018; Bednar and Almansoori, 2016) in cases of posterior C1-RO, with excellent outcomes, typical achievement of fusion, and widely restored function. Although difficulty of complete reduction is a well-known problem in these patients, it was demonstrated that even incomplete restoration of physiologic alignment still often provides sufficient stability and function. Nevertheless, complete reduction should be strived for. Our present description of the intraoperative stability test provides tips and insights regarding better tools and implants to achieve this goal. Rating of the functional outcome in traumatic scenarios has limited statistical value, as the pretraumatic function is only quantified by self-estimation of the patient. Furthermore we did not measure C1–C2 rotation directly. Therefore the functional outcome will not be addressed in this discussion.
4.3. The concept and application of intraoperative stability testing and its rationale
In this article, we provide a detailed description of an intraoperative stability test that is based on and inspired by the biomechanical findings of Koller et al., in 2010 (Koller et al., 2010). Our test is a direct translation of the set-up of that biomechanical study. Therefore, the translational forces applied in that study are valid for the intraoperative setting. In the 2014 study of Li-Jun et al. (2014), eccentric rotational forces were applied that are similar to the physiologic stress present in flexion-extension views, which produced comparable but somewhat smaller AADI values than in the study of Koller et al. (2010). This finding was explained by a more physiologic manner of loading. We also observed this tendency when comparing our intraoperative translational AADI (max) values (Table 3) to the flexion-extension AADI (flex) values during the follow-up period, as the AADI (flex) values were identical to or lower than the intraoperative values of translational AADI max (Table 5).
We would like to point out, that exact meassurement of AADI, especially in the OR-setting is not possible, even the measurement of screenshots of fluoroscopy with sophisticated tools fails accuracy. Therefore the stability test is not validated (see methods). Neither is it necessary to obtain exact meassurement in our opinion. More important is the tactile sensation of stability during the testing as long the gross limit of 5 mm is not violated and there is enough reserve space anterior and posterior to the myelon as assessed beforehand. Because of this we have only given integer values of the obtained minimum and maximum AADI in mm.
The bimanual application of posterior-anterior stress by the surgeon we could not measure so far. Therefore it was “trained” by the surgeon beforehand with a forcemeter. But the necessary range of 10–50 N is an everyday range of force as we all have an idea about how 1–5 kg feel when we lift it up. Again in the described setting it is not necessary to apply an exact amount of force, but to feel the resistance of the tissue within the mentioned gross limits and at the same time watch the resulting dynamic fluoroscopy. In the study of Koller et al. (2010) failure of the C1-RO always occurred with a small “peak with sudden decline in the load-displacement curve” and was described with shearing, tilting, bending and stretching characteristics. Hence, with the aid of haptic and optic control, the surgeon should be able to detect insufficiency. Therefore we suggest to refer to the testing as a ‘surgical skill’.
The possibility of stability testing provides a surgeon who attempts a posterior C1-RO with a biomechanically reasoned conceptual measure for determining the quality of the restoration of stability, within a certain margin of confidence, in each individual case. Our intraoperative stability testing uncovered instability in patient #3, that was addressed by conversion to C1–C2 ORIF (Fig. 7). Prior to the findings of our intraoperative stability assessment, there was no methodological assessment that would have identified this case as not suitable for C1-RO. Our stability testing therefore addresses the real stability as opposed to a theoretical or radiologically derived or assumed stability.
Fig. 7.
C1–C2 open reduction and internal fixation (ORIF) with a cross connector.
4.4. Limitations
This is a very small case series with no statistical power. It is rather the introduction of a new concept with initial success, illustrated by a few cases. We are aware that the described stability testing in addition to an already complex operation is yet not ready to use for general practice for most surgeons. The stability testing is a subjective method literally in the hand of the surgeon. It is not validated and mabe it never will be. But once accepted as an idea, a surgeon operating around C1–C2 (for other conditions) may grasp the atlas and the Lamina of C2 with Kocher clamps and feel how stable C1–C2 is in posterior-anterior stress. This will give the surgeon an idea what is to expect and compare this feeling to a reduced and fixed C1 with an insufficient TAL whenever a C1-RO is attempted.
In our opinion though there is yet another problem that needs to be addressed, before the C1–C2 stability test may bloom: there have to be better tools for a sufficient, complete and safe repositioning of 2,3 or 4 part Jefferson fractures from posterior. A good to perfect reduction is the key to reconstitute the secondary stabilizers of the (C0) C1–C2 complex.
4.5. Relevance of the Dickman classification for choice of surgical technique
It is crucial to know whether the surgically achieved stability after posterior C1-RO is preserved over time, especially in cases of Dickman Type I TAL lesions.
In 2016, Shatsky et al. (2016) showed that Dickman Type I and Dickman Type II lesions were equally unlikely to develop late sequelae of atlantoaxial instability (at a mean of 17 months postoperatively) after posterior C1-RO for unstable JBF.
A very recent (2022) randomized multicenter study from China by Yan et al. (Yan et al., 2022) has compared C1-RO with C1–C2 fixation fusion for unstable JBF in 37 respectively 36 patients, with almost equally distributed TAL Dickman type I and II lesions in both subgroups with superiority of the C1-RO over C1–C2 fixation fusion. Therefore, although it was not specifically stated by the authors, there is indeed evidence, that the TAL Dickman type lesion does not matter in when performing a C1-RO in unstable JBF.
Another recent (2021) work (preprint) by Tu et al. (2021) described the use of an anterior reduction plate to facilitate repositioning of the anterior arch of unstable JBF, with excellent realignment results. After 3 months, they reported a symptomless increase of AADI of >4 mm in three patients with Dickman type I lesion patients (out of 22 total patients treated), and thus stated that Dickman type I lesions might not be an indication for anterior C1-RO. However, based on the findings of Koller et al. (2010) and our own results, we think that an AADI of even up to 5 mm may be acceptable (as according to Koller et al. (2010) a maximum AADI of 1.5 mm more than in TAL intact conditions have to be expected until secondary stabilizers come into full effect), as long as there is sufficient reserve space anterior and posterior to the myelon, and stability that prevents further AADI progression. This may have been the situation in the three patients in the study of Tu et al. (2021), since they were asymptomatic. Intraoperative stability testing in anterior C1-RO has not yet been described and is probably not feasible. Therefore, in our opinion, the outcome of anterior-only C1-RO for unstable JBF will remain uncertain.
Among our four patients treated with C1-RO, the two patients with Dickman type I TAL lesions and the two patients with Dickman type II TAL lesions showed similarly good clinical results after a mean observation time of 1 year, and none of these patients developed radiological atlantoaxial instability that exceeded the intraoperative findings. We have not yet been able to directly evaluate (by MRI) the TAL in the two cases with Dickman type I lesions in the long-term; however, this may be achieved in the future on a larger scale, especially once more patients with Dickman type I TAL lesions have undergone C1-RO with intraoperatively assessed stability. Still the fate of the ligament in Dickman type I lesions would not be decisive as long as atlantoaxial instability does not develop in the long-term.
4.6. Prospective for future research applying the stability testing
The size of our sample is small due to rarity of unstable JBF, and the fact that even fewer patients qualify for this surgery. However, a rising number of institutions with a higher caseload are capable of treating patients with this technique due to the increasingly widespread availability of intraoperative 3D navigation/scanning, suitable implants and tools.
With the possibility to intraoperatively determine the achieved stability after C1-RO, as well as the potential to extend the stabilization to C2 or C0 from posterior in the same setting if necessary, more surgeons may strive for a function-preserving/restoring posterior C1-RO using the presently described controlled method. In the future, studies like the above mentioned study by Yan et al. (2022) implementing the stability testing and the algorithm shown in Graphic 1 may obtain short- and long-term data regarding the value of individual evaluation of the stability during posterior C1-RO for unstable JBF. On top of that, patients that recieved C1-C2 ORIF might be reevaluated intraoperatively after 3-6 months again using the stability testing after partial implant removal, if function restoration still is the goal.
5. Conclusions
The assessment of intraoperative stability after posterior C1-RO for unstable JBF, where the longitudinal stabilizers are restored by lifting up C0 through repositioning and reducing the C1-ring, represents an important upgrade of the surgeon's armamentarium. We were able to show that the cases we considered stable continued to be stable over time with function restoring C1-RO. This method might shift the treatment of unstable JBF towards posterior C1-RO whenever possible, and will also provide confidence and a margin of safety by uncovering cases that are not suitable for C1-RO, which previously would have been missed. Our present findings may serve as a blueprint for developing treatment algorithms (Graphic 1) for unstable JBF in the future, when the described stability testing is adopted as a surgical skill.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bas.2022.101668.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- Abeloos L., De Witte O., Walsdorff M., Delpierre I., Bruneau M. Posterior osteosynthesis of the atlas for nonconsolidated Jefferson fractures: a new surgical technique. Spine. 2011;36(20):E1360–E1363. doi: 10.1097/BRS.0b013e318206cf63. [DOI] [PubMed] [Google Scholar]
- Bednar D.A., Almansoori K.A. Solitary C1 posterior fixation for unstable isolated atlas fractures: case report and systematic review of the literature. Global Spine J. Jun. 2016;6(4):375–382. doi: 10.1055/s-0035-1564806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böhm H., Kayser R., El Saghir H., Heyde C.E. Die direkte Osteosynthese instabiler Atlasfrakturen Gehweiler Typ III. Präsentation einer dorsoventralen funktionserhaltenden Osteosynthese instabiler Atlasfrakturen [Direct osteosynthesis of instable Gehweiler Type III atlas fractures. Presentation of a dorsoventral osteosynthesis of instable atlas fractures while maintaining function] Unfallchirurg. Sep. 2006;109(9):754–760. doi: 10.1007/s00113-006-1081-x. German, PMID: 16874483. [DOI] [PubMed] [Google Scholar]
- Chung S.K., Park J.T., Lim J., Park J. Open posterior reduction and stabilization of a C1 burst fracture using mono-axial screws. Spine. 2011;36(5):E301–E306. doi: 10.1097/BRS.0b013e31820644cd. [DOI] [PubMed] [Google Scholar]
- Dickman C.A.C., Mamourian A.A., Sonntag V.K.V., et al. Magnetic resonance imaging of the transverse atlantal ligament for the evaluation of atlantoaxial instability. J. Neurosurg. 1991;75:221–227. doi: 10.3171/jns.1991.75.2.0221. [DOI] [PubMed] [Google Scholar]
- Dickman C., Re Ruf M., Melcher R., Harms J. Transoral reduction and osteosynthesis C1 as a function-preserving option in the treatment of unstable Jefferson fractures. Spine. 2004;29(19):2196. doi: 10.1097/01.brs.0000116984.42466.7e. author reply 2197. [DOI] [PubMed] [Google Scholar]
- Dvorak M.F., Johnson M.G., Boyd M., et al. Long-term health-related quality of life outcomes following Jeffersun-type burst fractures of the atlas. J. Neurosurg. Spine. 2005;2005(2):411–417. doi: 10.3171/spi.2005.2.4.0411. [DOI] [PubMed] [Google Scholar]
- Eck J.C., Walker M.P., Currier B.L., et al. Biomechanical comparison of unicortical versus bicortical C1 lateral mass screw fixation. J. Spinal Disord. Tech. 2007;20:505–508. doi: 10.1097/BSD.0b013e318031af8b. [DOI] [PubMed] [Google Scholar]
- Gelinas-Phaneuf N., Stienen M.N., Park J. Posterior open reduction and internal fixation of C1 fractures: the C-clamp technique. Acta Neurochir. 2018;160:2451–2457. doi: 10.1007/s00701-018-3710-x. [DOI] [PubMed] [Google Scholar]
- Gumpert R., Poglitsch T., Krassnig R., Pranzl R., Puchwein P. Reduction and ring fixation of instable C1 fractures with monoaxial pedicle screws. Arch. Orthop. Trauma. Surg. 2017;137:1253–1259. doi: 10.1007/s00402-017-2737-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heller J.G., Viroslav S., Hudson T. Jefferson fractures: the role of magnification artifact in assessing transverse ligament integrity. J. Spinal Disord. 1993;6:392–396. [PubMed] [Google Scholar]
- Jo K.-W., Park I.-S., Hong J.T. Motion-preserving reduction and fixation of C1 Jefferson fracture using a C1 lateral mass screw construct. J. Clin. Neurosci. 2011;18(5):695–698. doi: 10.1016/j.jocn.2010.08.033. [DOI] [PubMed] [Google Scholar]
- Kandziora F., Chapman J.R., Vaccaro A.R., Schroeder G.D., Scholz M. Atlas fractures and atlas osteosynthesis: a comprehensive narrative review. J Orthop Trauma. Sep. 2017;31(Suppl. 4):S81–S89. doi: 10.1097/BOT.0000000000000942. PMID: 28816879. [DOI] [PubMed] [Google Scholar]
- Koller H. vol. 2019. 2019. Hinged-rod technique for JBF-reduction & C1-ring osteosynthesis CSRS-E. (Annual Meeting). [Google Scholar]
- Koller H., Resch H., Tauber M., et al. A biomechanical rationale for C1 –ring osteosynthesis as treatment for displaced Jefferson burst fractures with incompetency of the transverse atlantal ligament. Eur. Spine J. 2010;19(8):1288–1298. doi: 10.1007/s00586-010-1380-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li-Jun L., Ying-Chao H., Ming-Jie Y., et al. Biomechanical analysis of the longitudinal ligament of upper cervical spine in maintaining atlantoaxial stability. Spinal Cord. 2014;52:342–347. doi: 10.1038/sc.2014.8. [DOI] [PubMed] [Google Scholar]
- Li-Jun, Teng H., Pan J., et al. Direct posterior C1 lateral mass screws compression reduction and osteosynthesis in the treatment of unstable Jefferson fractures. Spine. 2011;36(15):E1046–E1051. doi: 10.1097/BRS.0b013e3181fef78c. [DOI] [PubMed] [Google Scholar]
- Ottenbacher A., Bettag M. Resolution of traumatic vertebral artery dissection and occlusion after repositioning and posterior C1-ring osteosynthesis of a displaced Jefferson burst fracture. Acta Neurochir (Wien). Aug. 2017;159(8):1561–1564. doi: 10.1007/s00701-017-3241-x. Epub 2017 Jun 29. PMID: 28660396. [DOI] [PubMed] [Google Scholar]
- Ruf M., Melcher R., Harms J. Transoral reduction and osteosynthesis C1as a function-preserving option in the treatment of unstable Jefferson fractures. Spine. 2004;29:823–827. doi: 10.1097/01.brs.0000116984.42466.7e. [DOI] [PubMed] [Google Scholar]
- Shatsky J., Bellabarba C., Nguyen Q., Bransford R.J. A retrospective review of fixation of C1 ring fractures--does the transverse atlantal ligament (TAL) really matter? Spine J. Mar; 2016;16(3):372–379. doi: 10.1016/j.spinee.2015.11.041. [DOI] [PubMed] [Google Scholar]
- Spence K.F., Jr., Decker S., Sell K.W. Bursting atlantal fracture associated with rupture of the transverse ligament. J Bone Joint Surg Am. Apr. 1970;52(3):543–549. PMID: 5425648. [PubMed] [Google Scholar]
- Tessitore E., Momjian A., Payer M. Posterior reduction and fixation of an unstable Jefferson fracture with C1 lateral mass screws, C2 isthmus screws, and crosslink fixation: technical case report. Neurosurgery. 2008;63 doi: 10.1227/01.neu.0000335022.91240.c9. discussion ONSE101. [DOI] [PubMed] [Google Scholar]
- Tu Q., Chen H., Li Z., et al. Anterior reduction and C1-ring osteosynthesis with jefferson-fracture reduction plate (JeRP) via transoral approach for unstable Atlas fractures. Research Square. 2021 doi: 10.21203/rs.3.rs-155382/v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woods R.O., Inceoglu S., Akpolat Y.T., Cheng W.K., Jabo B., Danisa O. C1 lateral mass displacement and transverse atlantal ligament failure in jefferson's fracture: a biomechanical study of the "rule of spence. Neurosurgery. Feb 1. 2018;82(2):226–231. doi: 10.1093/neuros/nyx194.PMID:28431136. [DOI] [PubMed] [Google Scholar]
- Yan L., Du J., Yang J., et al. C1-ring osteosynthesis versus C1-2 fixation fusion in the treatment of unstable atlas fractures: a multicenter,prospective, randomized controlled study with 5-year follow-up. J. Neurosurg. Spine. 2022;2022 doi: 10.3171/2021.12SPINE211063. [DOI] [PubMed] [Google Scholar]
- Zhang Y.S., Zhang J.X., Yang Q.G., Li W., Tao H., Shen C.L. Posterior osteosynthesis with monoaxial lateral mass screw-rod system for unstable C1 burst fractures. Spine J. Jan. 2018;18(1):107–114. doi: 10.1016/j.spinee.2017.06.029. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
2
3
4
5