Summary
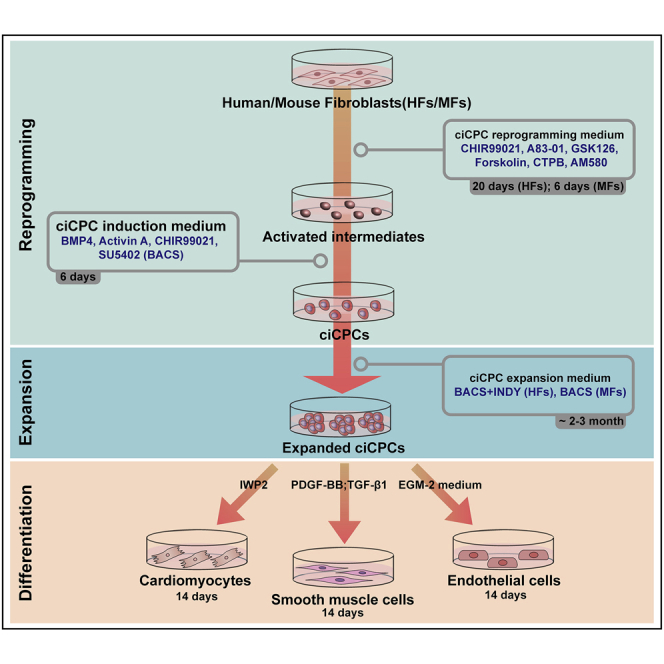
Here, we present a protocol to reprogram mouse and human fibroblasts into expandable cardiovascular progenitor cells (CPCs) via a defined small-molecule treatment. We describe steps to prepare fibroblasts and generate the chemically induced CPCs (ciCPCs), followed by expansion and differentiation of the ciCPCs. These cells can self-renew in the long term, faithfully retaining the CPC phenotype and cardiovascular differentiation capacity. This protocol provides an autologous and expandable cardiovascular cell source, which may find uses in cardiovascular disease modelling, drug discovery, and cardiac cell therapy.
For complete details on the use and execution of this protocol, please refer to Wang et al. (2022).1
Subject areas: Cell Biology, Cell culture, Cell-based Assays, Stem Cells, Cell Differentiation
Graphical abstract
Highlights
-
•
Direct conversion of fibroblasts into CPCs via a defined small-molecule treatment
-
•
Detailed procedures for the expansion and differentiation of ciCPCs
-
•
ciCPCs can be long-term cultured without losing the differentiation capacity
-
•
Protocol can be used with human and mouse fibroblasts
Publisher’s note: Undertaking any experimental protocol requires adherence to local institutional guidelines for laboratory safety and ethics.
Here, we present a protocol to reprogram mouse and human fibroblasts into expandable cardiovascular progenitor cells (CPCs) via a defined small-molecule treatment. We describe steps to prepare fibroblasts and generate the chemically induced CPCs (ciCPCs), followed by expansion and differentiation of the ciCPCs. These cells can self-renew in the long term, faithfully retaining the CPC phenotype and cardiovascular differentiation capacity. This protocol provides an autologous and expandable cardiovascular cell source, which may find uses in cardiovascular disease modelling, drug discovery, and cardiac cell therapy.
Before you begin
Institutional permissions
Human embryonic stem cells and fibroblasts are commercially obtained and all relevant experiments were approved by the Ethical Committee of Sun Yat-sen University and University of Health and Rehabilitation Sciences and comply with the 2021 Guidelines for Stem Cell Research and Clinical Translation (issued by the International Society for Stem Cell Research, ISSCR). All mouse work in this study was done in accordance with the Guide for the Care and Use of Laboratory Animals (National Institutes of Health, Publication No. 85-23, Revised) and carried out under the supervision of the Sun Yat-sen University Animal Care and Use Committee.
Preparations
Timing: 2–3 h
All cells were cultured in a humidified 37°C incubator supplemented with 5% CO2. The following medium should be prepared before the initiation of each step of the protocol and pre-warmed at 37°C for 20 min before use. Please refer to key resources table for a comprehensive list of reagents and resources. Please refer to materials and equipment for tables of medium recipes.
Key resources table
| REAGENT or RESOURCE | SOURCE | IDENTIFIER |
|---|---|---|
| Antibodies | ||
| Mouse monoclonal anti-GATA4 (Clone: G-4) Dilution 1:400 | Santa Cruz Biotechnology | Cat#SC25310; RRID: AB_627667 |
| Mouse monoclonal anti-ISL1 (Clone: 39.4D5) Dilution 1:50 | Developmental Studies Hybridoma Bank | Cat#39.4D5; RRID: AB_2314683 |
| Rabbit monoclonal anti-MEF2C (Clone: D80C1) Dilution 1:200 | Cell Signalling Technology | Cat# 5030; RRID: AB_10548759 |
| Purified Mouse anti-Ki67 (Clone: B56) Dilution 1:400 | BD Biosciences | Cat# 550609; RRID: AB_393778 |
| Goat anti-Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 Dilution 1:1000 | Life Technologies | Cat# A11008; RRID: AB_143165 |
| Goat anti-Mouse IgG2a Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 Dilution 1:1000 | Life Technologies | Cat# A21131; RRID: AB_2535771 |
| Goat anti-Mouse IgG2a Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 647 Dilution 1:1000 | Life Technologies | Cat# A21241; RRID: AB_2535810 |
| Goat anti-Mouse IgG2b Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 Dilution 1:1000 | Life Technologies | Cat# A21141; RRID: AB_2535778 |
| Goat anti-Mouse IgG2b Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 594 Dilution 1:1000 | Life Technologies | Cat# A21145; RRID: AB_2535781 |
| Goat anti-Mouse IgG1 Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 Dilution 1:1000 | Life Technologies | Cat# A21121; RRID: AB_2535764 |
| Chemicals, peptides, and recombinant proteins | ||
| CHIR99021 | Selleck | S2924; CAS:1797989-42-4 |
| A83-01 | Stemgent | 04-0014; CAS: 909910-43-6 |
| GSK126 | Selleck | S7061; CAS: 1346574-57-9 |
| Forskolin | Sigma | F6886; CAS: 66575-29-9 |
| CTPB | Sigma | EPS001; CAS: 586976-24-1 |
| AM580 | Selleck | S2933; CAS:102121-60-8 |
| INDY | MedChemExpress | HY-108476; CAS: 1169755-45-6 |
| SU5402 | Selleck | S7667; CAS:215543-92-3 |
| L-ascorbic acid 2-phosphate magnesium | Sigma | A8960; CAS: 1713265-25-8 |
| IWP2 | Selleck | S7085; CAS: 686770-61-6 |
| Monothioglycerol (MTG) | Sigma | M6145; CAS: 96-27-5 |
| Heparin sodium salt | Sigma | H3149; CAS:9041-08-1 |
| Insulin solution human | Sigma | I9278; CAS:11061-68-0 |
| Recombinant Human FGF2 | PeproTech | 100-18B |
| Recombinant Human BMP4 | R&D Systems | 314-BP |
| Recombinant Human Activin A | R&D Systems | 338-AC |
| Recombinant Human TGF-β1 | R&D Systems | 240-B |
| Recombinant Human PDGF-BB | R&D Systems | 220-BB |
| B27 supplement without vitamin A | Gibco | 12587-010 |
| Knockout serum replacement | Gibco | 10828-010 |
| DMEM (4.5 g/l glucose) | Gibco | 11965-092 |
| Knockout DMEM (4.5 g/l glucose) | Gibco | 10829-018 |
| DMEM/F12 | Gibco | 11320-033 |
| TeSR™-E8™ Kit for hESC/hiPSC Maintenance | Stem Cell Technologies | 05990 |
| IMDM (4.5 g/l glucose) | Gibco | 12440-053 |
| F12 medium | Gibco | 11765-054 |
| EGM-2 BulletKit | Lonza | CC-3162 |
| Fetal bovine serum (FBS) | Gibco | 10099-141 |
| Phosphate-buffered saline (PBS) | Gibco | 10010-049 |
| GlutaMAX | Gibco | 35050-061 |
| Nonessential amino acids (NEAA) | Gibco | 11140-050 |
| β-mercaptoethanol | Sigma | M6250 |
| Insulin-transferrin-sodium selenite (ITS) | Gibco | 41400-045 |
| Chemically defined lipid concentrates | Gibco | 11905-031 |
| Recombinant human albumin | Sigma | NIST2925 |
| Xeno-free knockout serum replacement | Gibco | 12618-012 |
| Collagenase B | Roche | 11088815001 |
| Accutase | Stem Cell Technologies | 07920 |
| Vitronectin | Gibco | CTS279S3 |
| Matrigel | BD Biosciences | 354277 |
| Gelatin | Sigma | V900863 |
| Experimental models: Cell lines | ||
| Mouse embryonic fibroblasts: C57BL/6J | Primary cells | N/A |
| Mouse tail-tip fibroblasts: C57BL/6J | Primary cells | N/A |
| Human foreskin fibroblasts line CRL2097 | American Type Culture Collection | CRL2097 |
| hESC line H1 | WiCell | N/A |
| Experimental models: Organisms/strains | ||
| Mouse: C57BL/6J (Male: 8 weeks) | Sun Yat-Sen University Laboratory Animal Center | N/A |
| Mouse: C57BL/6J (Female: 10 weeks) | Sun Yat-Sen University Laboratory Animal Center | N/A |
| Other | ||
| 0.22-μm Millex-GP filter unit | Millipore | SLGP033RB |
Materials and equipment
All medium is prepared using sterile techniques. We recommend that cell culture is performed in a sterile environment without the use of antibiotics.
Fibroblast medium
| Reagent | Final concentration | Amount |
|---|---|---|
| DMEM (4.5 g/l glucose) | N/A | 44 mL |
| FBS | 10% | 5 mL |
| NEAA | 1% | 0.5 mL |
| GlutaMAX | 1% | 0.5 mL |
| Total | N/A | 50 mL |
Note: Fibroblast medium should be stored at 4°C for up to 1 month and pre-warmed at 37°C for at least 20 min prior to use.
Mouse ciCPC reprogramming medium
| Reagent | Final concentration | Amount |
|---|---|---|
| Knockout DMEM (4.5 g/l glucose) | N/A | 39 mL |
| FBS | 10% | 5 mL |
| Knockout serum replacement | 10% | 5 mL |
| NEAA | 1% | 0.5 mL |
| GlutaMAX | 1% | 0.5 mL |
| FGF2 | 100 ng/mL | 5 μg |
| β-mercaptoethanol | 0.1 mM | 0.4 mg |
| L-ascorbic acid 2-phosphate magnesium | 50 μg/mL | 2.5 mg |
| CHIR99021 | 10 μM | 0.25 mg |
| A83-01 | 1 μM | 21 μg |
| GSK126 | 1 μM | 26 μg |
| Forskolin | 50 μM | 1 mg |
| CTPB | 5 μM | 0.14 mg |
| AM580 | 0.03 μM | 527 ng |
| Total | N/A | 50 mL |
Human ciCPC reprogramming medium
| Reagent | Final concentration | Amount |
|---|---|---|
| Knockout DMEM | N/A | 43 mL |
| Xeno-free knockout serum replacement | 10% | 5 mL |
| NEAA | 1% | 0.5 mL |
| GlutaMAX | 1% | 0.5 mL |
| ITS | 1% | 0.5 mL |
| Chemically defined lipid concentrates | 1% | 0.5 mL |
| FGF2 | 10 ng/mL | 0.5 μg |
| β-mercaptoethanol | 0.1 mM | 0.4 mg |
| Recombinant human albumin | 500 μg/mL | 25 mg |
| L-ascorbic acid 2-phosphate magnesium | 50 μg/mL | 2.5 mg |
| CHIR99021 | 10 μM | 0.25 mg |
| A83-01 | 1 μM | 21 μg |
| GSK126 | 1 μM | 26 μg |
| Forskolin | 50 μM | 1 mg |
| CTPB | 5 μM | 0.14 mg |
| AM580 | 0.03 μM | 527 ng |
| Total | N/A | 50 mL |
Note: Store FGF2 stock solution at −80°C for up to 6 months. Then prepare enough 0.1 mL diluted aliquots and store them at −20°C until use. Avoid repeated freezing and thawing. Prepare fresh mouse and human reprogramming medium and filter-sterilize through a 0.2 μm filter before use.
Mouse ciCPC induction medium
| Reagent | Final concentration | Amount |
|---|---|---|
| DMEM/F12 | N/A | 48.5 mL |
| B27 supplement without vitamin A | 1% | 0.5 mL |
| NEAA | 1% | 0.5 mL |
| GlutaMAX | 1% | 0.5 mL |
| β-mercaptoethanol | 0.1 mM | 0.4 mg |
| CHIR99021 | 3 μM | 75 μg |
| SU5402 | 2 μM | 29.6 μg |
| BMP4 | 5 ng/mL | 0.25 μg |
| Activin A | 10 ng/mL | 0.5 μg |
| Total | N/A | 50 mL |
Human ciCPC induction medium
| Reagent | Final concentration | Amount |
|---|---|---|
| DMEM/F12 | N/A | 48 mL |
| NEAA | 1% | 0.5 mL |
| GlutaMAX | 1% | 0.5 mL |
| ITS | 1% | 0.5 mL |
| Chemically defined lipid concentrates | 1% | 0.5 mL |
| β-mercaptoethanol | 0.1 mM | 0.4 mg |
| Recombinant human albumin | 500 μg/mL | 25 mg |
| CHIR99021 | 3 μM | 75 μg |
| SU5402 | 2 μM | 29.6 μg |
| BMP4 | 5 ng/mL | 0.25 μg |
| Activin A | 10 ng/mL | 0.5 μg |
| Total | N/A | 50 mL |
Note: Store BMP4 and Activin A stock solution at −80°C for up to 6 months. Then prepare enough 0.1 mL diluted aliquots and store them at −20°C until use. Avoid repeated freezing and thawing. Prepare fresh mouse and human induction medium and filter-sterilize through a 0.2 μm filter before use.
Mouse and human ciCPC expansion medium
| Reagent | Final concentration | Amount |
|---|---|---|
| DMEM/ F12 | N/A | 48 mL |
| NEAA | 1% | 0.5 mL |
| GlutaMAX | 1% | 0.5 mL |
| ITS | 1% | 0.5 mL |
| Chemically defined lipid concentrates | 1% | 0.5 mL |
| L-ascorbic acid 2-phosphate magnesium | 64 μg/mL | 3.2 mg |
| CHIR99021 | 3 μM | 75 μg |
| SU5402 | 2 μM | 29.6 μg |
| BMP4 | 5 ng/mL | 0.25 μg |
| Activin A | 10 ng/mL | 0.5 μg |
| INDY (human ciCPC expansion medium only) | 0.2 μM | 2.35 μg |
| Total | N/A | 50 mL |
Note: Make fresh ciCPC expansion medium and filter-sterilize through a 0.2 μm filter. For long-term expansion of human ciCPCs, INDY should be added to promote cell growth.
Mouse cardiomyocyte (CM) differentiation medium
| Reagent | Final concentration | Amount |
|---|---|---|
| IMDM (4.5 g/l glucose): F12 | 3:1 | 49 mL |
| B27 supplement without vitamin A | 1% | 0.5 mL |
| GlutaMAX | 1% | 0.5 mL |
| L-ascorbic acid 2-phosphate magnesium | 50 μg/mL | 2.5 mg |
| MTG | 0.4 mM | 2.2 mg |
| IWP2 | 5 μM | 0.12 mg |
| Total | N/A | 50 mL |
Note: Mouse CM differentiation medium should be stored at 4°C for up to 1 week and pre-warmed at 37°C for at least 20 min prior to use.
Mouse smooth muscle cell (SMC) differentiation medium
| Reagent | Final concentration | Amount |
|---|---|---|
| IMDM (4.5 g/l glucose): F12 | 3:1 | 49 mL |
| B27 supplement without vitamin A | 1% | 0.5 mL |
| GlutaMAX | 1% | 0.5 mL |
| L-ascorbic acid 2-phosphate magnesium | 50 μg/mL | 2.5 mg |
| MTG | 0.4 mM | 2.2 mg |
| PDGF-BB | 10 ng/mL | 0.5 μg |
| TGF-β1 | 2 ng/mL | 0.1 μg |
| Total | N/A | 50 mL |
Note: Store PDGF-BB and TGF-β1 stock solution at −80°C for up to 6 months. Then prepare enough 0.1 mL diluted aliquots and store them at −20°C until use. Avoid repeated freezing and thawing. SMC differentiation medium should be filter-sterilized through a 0.2 μm filter before use and stored at 4°C for up to 1 week.
Human ciCPC CM differentiation medium
-
•
Conditioned medium from human embryonic stem cell (hESC)-derived CMs supplemented with 5 μM IWP2.
Note: Conditioned medium should be stored at −80°C until use and should be used within 2 weeks after thawing.
CRITICAL: Detailed procedures of conditioned medium collection will be described in the “step-by-step method details: 27. Cardiac differentiation of hESCs and conditioned medium preparation” section as listed below.
hESC CM differentiation medium
| Reagent | Final concentration | Amount |
|---|---|---|
| E8 basal medium | N/A | 49.5 mL |
| Chemically defined lipid concentrates | 1% | 0.5 mL |
| CHIR99021 (added during day 0-day 1) | 5 μM | 0.13 mg |
| IWP2 (added during day 2-day 5) | 3 μM | 70 μg |
| Heparin (added during day 2-day 5) | 3 μg/mL | 0.15 mg |
| Insulin (day 7 onwards) | 20 μg/mL | 1 mg |
| Total | N/A | 50 mL |
Note: hESC CM differentiation medium should be stored at 4°C for up to 1 week.
Human CM culture medium
| Reagent | Final concentration | Amount |
|---|---|---|
| DMEM/ F12 | N/A | 48 mL |
| ITS | 1% | 0.5 mL |
| Chemically defined lipid concentrates | 1% | 0.5 mL |
| L-ascorbic acid 2-phosphate magnesium | 64 μg/mL | 3.2 mg |
| Total | N/A | 50 mL |
Note: Human CM culture medium should be stored at 4°C for up to 2 weeks.
Step-by-step method details
Fibroblast preparation
Timing: 4 days
This step describes methods to prepare and culture mouse and human fibroblasts.
-
1.Prepare mouse and human fibroblast cells.
-
a.To obtain healthy and proliferative mouse fibroblast cells for reprogramming, mouse embryonic fibroblasts (MEFs) or tail-tip fibroblasts (TTFs) are derived from 13.5-day mouse embryos or 8-weeks adult mice, respectively, according to a standard procedure.2 MEFs and TTFs are maintained in fibroblast medium and cells that have been passaged for less than 3 times are used.
-
b.Human foreskin fibroblast (HFF) line CRL2097 are used for reprogramming and are passaged onto the gelatin-coated dishes in fibroblast medium (troubleshooting 1).
-
a.
CRITICAL: For MEF preparation, remove head, spinal cords, and all developing organs from mouse embryos to avoid lineage precursor cell contamination.
ciCPC induction
Timing: 15–27 days
This step describes the stepwise procedure to directly generate ciCPCs from mouse or human fibroblasts.
-
2.
Coat a 12-well plate with matrigel (for mouse ciCPC induction) or a 6-well plate with vitronectin (for human ciCPC induction) and place the plate in a 37°C, 5% CO2 tissue culture incubator for at least 1 h (troubleshooting 1).
-
3.
Carefully remove the coating reagent and plate 10,000 (MEFs), 20,000 (TTFs), or 40,000 (HFFs) cells per well, respectively, in pre-warmed fibroblast medium.
-
4.
Place the plate in a 37°C, 5% CO2 tissue culture incubator for at least 8 h.
CRITICAL: Fibroblasts are likely to aggregate. As evenly distributed cells and appropriate cell confluence are both important for reprogramming, therefore rock the plate gently to evenly distribute fibroblast cells.
-
5.
Carefully aspirate the fibroblast medium without touching the bottom of the plate and wash with PBS twice.
-
6.
Add 2 mL of fresh mouse ciCPC reprogramming medium or 2.5 mL of human ciCPC reprogramming per well, respectively, which includes a combination of 6 compounds (6C) comprising CHIR99021, A83-01, GSK126, Forskolin, CTPB, and AM580 for CPC induction.
CRITICAL: It is important not to disturb the attached cells which may result in low reprogramming efficiency. Therefore, gently and slowly add medium along the side wall of the well and avoid adding medium directly onto the cultured cells.
-
7.
Place the plate in a 37°C, 5% CO2 tissue culture incubator. Replace with fresh reprogramming medium every 2 days.
-
8.
After 8 (for mouse ciCPC induction) or 20 days (for human ciCPC induction) of incubation, carefully aspirate the reprogramming medium from the plates and wash with PBS twice (troubleshooting 3).
-
9.
Add 2 mL of fresh mouse or human ciCPC induction medium per well, respectively, and return the plates to the incubator.
-
10.
Change medium every 2 days.
-
11.
After 6 days, mouse (Figure 1) or human (Figure 2) ciCPC colonies are formed and can be picked for further expansion (troubleshooting 4).
CRITICAL: Compared to mouse ciCPC reprogramming condition, HFFs are executed to chemical-treatment for continuous 20 days due to slower growth rate and reprogramming kinetics of human fibroblasts. 6C-treating time may vary from 15–30 days for different fibroblast lines and optimal treating duration for each line may require experimental validation.
Figure 1.
Reprogramming of MEFs into CPCs via defined small-molecule treatment
(A) Morphology of 6C-induced cell colonies derived from MEFs at day 14. Scale bars, 100 μm.
(B) Immunofluorescence analyses of Gata4 and Isl1 in 6C-induced cell colonies at day 14. Scale bars, 100 μm.
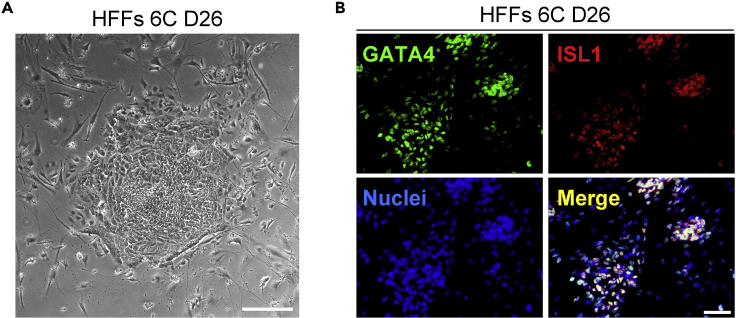
Figure 2.
Reprogramming of HFFs into CPCs via defined small-molecule treatment
(A) Morphology of 6C-induced cell colonies derived from HFFs at day 26. Scale bars, 100 μm.
(B) Immunofluorescence analyses of GATA4 and ISL1 in 6C-induced cell colonies at day 26. Scale bars, 100 μm.
ciCPC clone picking and expansion
Timing: 2–3 h
This step describes the methods for picking the ciCPC clones and subsequent long-term expansion of them in a chemically defined xeno-free condition.
-
12.
Pick mouse or human ciCPC colonies into a 1.5 mL-tube.
-
13.
Centrifuge the picked colonies at 800 g for 3 min at 25°C.
-
14.
Carefully remove the supernatant, resuspend the pellet in 0.1 mL collagenase B, and digest for 10 min at 37°C.
-
15.
Quench the enzyme by adding 0.1 mL of fresh ciCPC expansion medium and centrifuge the digested cells at 800 g for 3 min at 25°C.
-
16.
Carefully remove the supernatant and resuspend the picked colonies in 0.1 mL of accutase and digest for 5 min at 37°C.
-
17.
Repeat procedure 15. Then carefully remove the supernatant and resuspend the digested cells in 0.1 mL of fresh ciCPC expansion medium and seed them into a well of a vitronectin-coated 96-well plate.
CRITICAL: Do not rock the plate as cells in 96-well plate tend to gather in the well center upon shaking.
-
18.
Cultured ciCPCs are ready for passaging when grow to confluence. For passaging, aspirate the ciCPC expansion medium, and wash the cells twice with PBS. Add 0.1 mL collagenase B per well of 96-well plate and digest for 10 min at 37°C (troubleshooting 5).
-
19.
Carefully remove collagenase B without dislodge the cells, and add 0.1 mL accutase per well of 96-well plate for 5 min at 37°C.
-
20.
Quench the enzyme by adding 0.1 mL of fresh ciCPC expansion medium into each well.
-
21.
Collect all undetached cells from the well and transfer them into a 15-mL tube. Then centrifuge the cells at 800 g for 3 min at 25°C.
-
22.
Carefully remove the supernatant and resuspend the cells in 1 mL of fresh expansion medium. Pipette the cells up and down thoroughly.
-
23.
Add the single-cell suspensions into the vitronectin-coated 24-well plate. Gently rock the plate to evenly distribute the ciCPCs and then place the plate in a 37°C, 5% CO2 tissue culture incubator (troubleshooting 1).
-
24.
For long-term expansion, cells can be routinely passaged every 3–4 days (for mouse ciCPCs) or 4–6 day (for human ciCPCs), respectively, at a density of 5×105 cells per well of a 12 well plate. The culture medium should be changed every 2 days (troubleshooting 5).
-
25.
In this defined culture condition, mouse or human ciCPCs can long-term propagate for more than 20 or 12 passages, respectively, with stable growth rate and undifferentiated morphology maintained (Figure 3).
Pause point: Mouse and human ciCPCs can be frozen and long-term stored in liquid nitrogen.
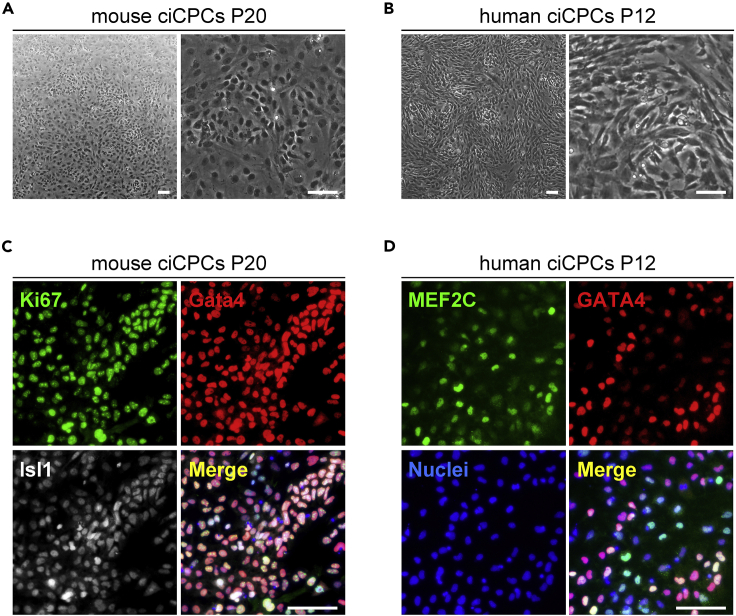
Figure 3.
ciCPCs are long-term expandable in fully chemically defined conditions
(A) Morphology of P20 mouse ciCPCs. P, passage. Scale bars, 100 μm.
(B) Morphology of P12 human ciCPCs. Scale bars, 100 μm.
(C) Immunofluorescence analyses of Ki67, Gata4, and Isl1 in P20 mouse ciCPCs. Scale bars, 100 μm.
(D) Immunofluorescence analyses of MEF2C and GATA4 in P12 human ciCPCs. Scale bars, 100 μm.
ciCPC differentiation
Timing: 7–14 days
This step describes the methods for targeted differentiation of ciCPCs into cardiomyocytes (CMs), smooth muscle cells (SMCs), endothelial cells (ECs).
-
26.Procedures of mouse ciCPC differentiation in vitro (troubleshooting 1 and 6).
-
a.For CM differentiation, plate accutase-dissociated mouse ciCPCs onto the vitronectin-coated plate in fresh mouse CM differentiation medium for continuous 7 days (at a density of 3×105 cells per cm2). Change medium every 2 days.
-
b.CMs display synchronized contraction usually at days 7.
-
c.For SMC differentiation, suspend accutase-dissociated mouse ciCPCs with fresh SMC differentiation medium.
-
d.Add the suspension to the vitronectin-coated plate at a density of 1×104 cells per cm2.
-
e.Culture for 7 days. Change medium every 2 days. Differentiated SMCs can be monitored at day 7.
-
f.For EC differentiation, plate accutase-dissociated mouse ciCPCs onto the vitronectin-coated plate in EGM-2 medium for continuous 7 days (at a density of 1×104 cells per cm2). Change medium every 2 days.
-
g.Differentiated ECs can be monitored at day 7.
-
a.
Pause point: Cells can be fixed and stored in PBS at 4°C for 2–3 weeks.
-
27.Cardiac differentiation of hESCs and conditioned medium preparation.
-
a.hESC line H1 is cultured on the vitronectin-coated plate in TeSR™-E8™ medium (troubleshooting 1).
-
b.Cardiac differentiation of H1 hESCs is initiated by seeding hESCs in monolayer in TeSR™-E8™ medium and allowing them to grow to 80%–90% confluence.
-
c.Switch to hESC CM differentiation medium and name this day as day 0. The medium should be changed daily until day 7 and every 2–3 days thereafter.
-
d.Needed supplements in CM differentiation medium are varied in different differentiation days. Add correct supplement at the indicated dosages and times following the recipe listed above.
-
e.On differentiation day 20, reseed cells onto the gelatin-coated plate in human CM culture medium.
-
f.Collect the conditioned medium of the differentiated CMs from differentiation day 30 to day 60. Medium is harvested every 2 days.
-
a.
CRITICAL: Split the conditioned medium into 15-mL tubes. It should be stored at −80°C until use and should be used within 2 weeks after thawing.
-
28.Procedures of human ciCPC differentiation in vitro (troubleshooting 1 and 6).
-
a.For CM differentiation, plate accutase-dissociated human ciCPCs onto the vitronectin-coated plate in fresh human ciCPC CM differentiation medium for 14 days (at a density of 3×105 cells per cm2). Culture medium is renewed every 2 days.
-
b.Procedures of the SMC and EC differentiation from human ciCPCs are the same as the mouse ciCPCs except extending the treating time to 14 days.
-
a.
CRITICAL: Quality of conditioned medium of hESC-derived CMs is very important for cardiac differentiation of human ciCPCs due to its paracrine factors. Use freshly thawed conditioned medium for CM differentiation of human ciCPCs.
Pause point: Cells can be fixed and stored in PBS at 4°C for 2–3 weeks.
Expected outcomes
CPCs are lineage-restricted progenitors that have extensive proliferative capacity and multi-potency towards cardiovascular lineages (including CMs, SMCs and ECs).3 Compared to CMs, CPCs display a greater potency for heart repairing after transplantation into the mouse myocardial infarction heart.4,5,6,7 In this protocol, we describe an efficient method for generation of expandable CPCs from fibroblasts with defined small molecules.
By stimulation with the reprogramming small molecule cocktail, mouse and human fibroblasts are initially induced to an epigenetically unstable cell state with increased plasticity. Then the activated cells are specifically directed towards a CPC state with the treatment of defined small molecules and cytokines. Following this protocol, fibroblasts undergo morphology changes and convert into Gata4+/Isl1+ ciCPCs (Figures 1 and 2). In terms of long-term expansion, ciCPCs can robustly propagate for more than 20 passages (mouse) and 12 passages (human), respectively, in the optimized culture condition. Further characterization assays reveal that late passage ciCPCs retain undifferentiated morphology, stable growth rate, and uniform expression of CPCs markers (Figure 3). Passaged ciCPCs can efficiently differentiate into cardiovascular lineages cells.
In summary, this protocol enables ciCPC self-renewal long-term in vitro. Expanded ciCPCs can be frozen stored and thawed for rapid use.
Limitations
This protocol provides a method for reprogramming of fibroblasts into expandable CPCs without introducing genetic manipulation.1 Nevertheless, there are still limitations that should be overcome before the method can be widely used for future clinical translation.
First, human CPC reprogramming is relatively inefficient (∼5% based on key marker expression) and time consuming, which may result from the slower growth rate and reprogramming kinetics of HFFs. The current protocol should be optimized through additional boosting molecule screening to improve human reprogramming efficiency.
Secondly, in this protocol human fetal fibroblasts are used as the starting population for reprogramming, as they are more epigenetically amenable. Generating ciCPCs from adult human fibroblasts are still challenging. Identification of additional small molecules that enables adult human somatic cells is a prerequisite for further optimization of the current reprogramming protocol and future clinical application.
Troubleshooting
Problem 1
Poor attachment of cells in the coated plate (related to steps 1, 2, 23, 26, 27, and 28).
Potential solution
It may be caused by imperfect substrate coating. We recommend restarting with a new properly coated plate. For matrigel coating, it is important to work on ice and use cold tips. Besides, perform medium changing carefully and gently.
Problem 2
Cultures become contaminated (related to each step).
Potential solution
To avoid contamination, operation should be always performed in the sterile culture hood. All the medium should be filter-sterilized through 0.2 μm filter before use.
Problem 3
Mouse ciCPC colonies fail to appear during the reprogramming process (related to step 8).
Potential solution
It may be caused by poor quality of the starting fibroblasts which may affect the reprogramming efficiency and final outcomes. To ensure a healthy and proliferative starting population, we don’t recommend using frozen cells or fibroblasts passaged greater than 3 times for mouse ciCPCs reprogramming. In addition, fine-tuning of the concentration of each reprogramming chemical may be required for some type of mouse fibroblasts.
Problem 4
Human ciCPC colonies are not observed after induction (related to step 11).
Potential solution
There are two common reasons for this problem: 1) Freshly thawed HFFs are used immediately for reprogramming; 2) Small molecules or cytokines used in the medium are improperly stored.
To initial with an active starting population, we recommend use HFFs passaged at least one time after thawing, and HFFs passaged greater than 10 should not be used for ciCPC reprogramming. Secondly, small molecules or cytokines aliquots should be stored at the correct temperature and used within the indicated time. Avoid repeated freeze-thaw cycles and use freshly prepared medium.
Problem 5
Low survival rate during ciCPC seeding and expansion (related to steps 18 and 24).
Potential solution
Rho-associated coiled-coil containing protein kinase (ROCK) inhibitors, such as thiazovivin (1 μM, Stemgent) or Y27632 (10 μM, Stemgent), can be added to the expansion medium to improve cell survival.
Problem 6
Low differentiation efficiency of mouse and human ciCPCs in vitro (related to steps 26 and 28).
Potential solution
The most common reason for this problem is the unsuitable cell density during initial cell seeding which may negatively affect the differentiation efficiency. To harvest specifically differentiated cells maximally, we strongly recommend fine-tuning the initial cell seeding number around the standard density.
Resource availability
Lead contact
Further information and requests for resources and reagents should be directed to and will be fulfilled by the lead contact, Nan Cao (caon3@mail.sysu.edu.cn).
Materials availability
This study did not generate any unique reagents.
Acknowledgments
This study was funded by the National Key R&D Program of China (2018YFA0109600 and 2018YFA0109100) and the National Natural Science Foundation of China (92057113 and 82061148011).
Author contributions
N.C. conceptualized and supervised the project. J.W. performed the experiments and wrote the protocol. Q.W. assisted the experiments and edited the protocol.
Declaration of interests
The authors declare no competing interests.
Contributor Information
Jia Wang, Email: jiawang@uor.edu.cn.
Nan Cao, Email: caon3@mail.sysu.edu.cn.
Data and code availability
This study did not generate/analyze any datasets or code.
References
- 1.Wang J., Gu S., Liu F., Chen Z., Xu H., Liu Z., Cheng W., Wu L., Xu T., Chen Z., et al. Reprogramming of fibroblasts into expandable cardiovascular progenitor cells via small molecules in xeno-free conditions. Nat. Biomed. Eng. 2022;6:403–420. doi: 10.1038/s41551-022-00865-7. [DOI] [PubMed] [Google Scholar]
- 2.Wang H., Cao N., Spencer C.I., Nie B., Ma T., Xu T., Zhang Y., Wang X., Srivastava D., Ding S. Small molecules enable cardiac reprogramming of mouse fibroblasts with a single factor, Oct4. Cell Rep. 2014;6:951–960. doi: 10.1016/j.celrep.2014.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Qyang Y., Martin-Puig S., Chiravuri M., Chen S., Xu H., Bu L., Jiang X., Lin L., Granger A., Moretti A., et al. The renewal and differentiation of Isl1+ cardiovascular progenitors are controlled by a Wnt/beta-catenin pathway. Cell Stem Cell. 2007;1:165–179. doi: 10.1016/j.stem.2007.05.018. [DOI] [PubMed] [Google Scholar]
- 4.Menasché P., Vanneaux V., Hagège A., Bel A., Cholley B., Cacciapuoti I., Parouchev A., Benhamouda N., Tachdjian G., Tosca L., et al. Human embryonic stem cell-derived cardiac progenitors for severe heart failure treatment: first clinical case report. Eur. Heart J. 2015;36:2011–2017. doi: 10.1093/eurheartj/ehv189. [DOI] [PubMed] [Google Scholar]
- 5.Lalit P.A., Salick M.R., Nelson D.O., Squirrell J.M., Shafer C.M., Patel N.G., Saeed I., Schmuck E.G., Markandeya Y.S., Wong R., et al. Lineage reprogramming of fibroblasts into proliferative induced cardiac progenitor cells by defined factors. Cell Stem Cell. 2016;18:354–367. doi: 10.1016/j.stem.2015.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhang Y., Cao N., Huang Y., Spencer C.I., Fu J.D., Yu C., Liu K., Nie B., Xu T., Li K., et al. Expandable cardiovascular progenitor cells reprogrammed from fibroblasts. Cell Stem Cell. 2016;18:368–381. doi: 10.1016/j.stem.2016.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhao J., Ghafghazi S., Khan A.R., Farid T.A., Moore J.B., 4th Recent developments in stem and progenitor cell therapy for cardiac repair. Circ. Res. 2016;119:e152–e159. doi: 10.1161/CIRCRESAHA.116.310257. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This study did not generate/analyze any datasets or code.