Abstract
Background
Upfront surgery followed by systemic treatment is recommended to treat clinical stage I–IIA small cell lung cancer (SCLC), but data on the clinical outcomes are sparse. Thus, this study evaluated the stage migration and long-term prognosis of surgically treated clinical stage I–IIA SCLC.
Methods
We retrospectively reviewed 49 patients with clinical stage I–IIA SCLC who underwent upfront surgery between 2000 and 2020. Additionally, we re-evaluated the TNM (tumor-node-metastasis) staging according to the eighth edition of the American Joint Committee on Cancer staging system for lung cancer.
Results
The clinical stages of SCLC were cIA in 75.5%, cIB in 18.4%, and cIIA in 6.1% of patients. A preoperative histologic diagnosis was made in 65.3% of patients. Lobectomy and systematic lymph node dissection were performed in 77.6% and 83.7% of patients, respectively. The pathological stages were pI in 67.3%, pII in 24.5%, pIII in 4.1%, and pIV in 4.1% of patients. The concordance rate between clinical and pathological stages was 44.9%, and the upstaging rate was 49.0%. The 5-year overall survival (OS) rate was 67.8%. No significant difference in OS was found between stages pI and pII. However, the OS for stages pIII/IV was significantly worse than for stages pI/II (p<0.001).
Conclusion
In clinical stage I–IIA SCLC, approximately half of the patients were pathologically upstaged, and OS was favorable after upfront surgery, particularly in pI/II patients. The poor prognosis of pIII/IV patients indicates the necessity of intensive preoperative pathologic mediastinal staging.
Keywords: Small cell lung carcinoma, Surgery, Neoplasm staging, Survival analysis, Chemotherapy
Introduction
Small cell lung cancer (SCLC) accounts for around 15% of lung cancers. The rapid progression of SCLC means that it is usually detected at an advanced stage, and the prognosis is therefore poor [1,2]. Therefore, chemoradiation therapy was considered the standard treatment for SCLC, regardless of the stage, and surgery was only offered to highly selected patients. For decades, the aggressive nature of SCLC and the limited treatment options have led to SCLC being staged as simply “limited” or “extensive” disease with consideration of radiation therapy. However, the accumulation of clinical data and the development of more precise staging modalities have altered the landscape of SCLC staging [3]. Furthermore, recent empirical data have shown a good prognosis after surgery-based treatment in early-stage SCLC. Thus, a sophisticated tumor-node-metastasis (TNM) staging classification, similar to that of non-small cell lung cancer (NSCLC), should be applied to SCLC to select resectable cases. Currently, the International Association for the Study of Lung Cancer recommends applying the TNM staging classification for lung cancer to SCLC [4,5]. Additionally, the National Comprehensive Cancer Network (NCCN) guidelines recommend upfront surgery for clinical stage I–IIA SCLC, followed by systemic treatment [6]. Nevertheless, data on clinical outcomes after upfront surgery for SCLC based on these current recommendations are sparse because patients with clinical stage I–IIA comprise only <5% of SCLC cases, and previous studies have not considered the current staging classifications.
We conducted this study to clarify the clinical outcomes of surgery-based treatment in early-stage SCLC according to the current recommendations.
Methods
Patients
From November 2000 to November 2020, 93 patients underwent surgery for SCLC at Seoul National University Hospital. Of them, 74 underwent upfront curative surgery. After applying the exclusion criteria, the final study cohort comprised 49 patients with clinical stage I–IIA SCLC (Fig. 1). Patients with combined histological findings of SCLC and NSCLC were also included. The clinical stages were reclassified according to the eighth edition of the American Joint Committee on Cancer staging system for lung cancer [4].
Fig. 1.
Study cohort and exclusion criteria. SCLC, small cell lung cancer; TNM, tumor-node-metastasis.
This study was reviewed and approved by the Institutional Review Board of Seoul National University Hospital (IRB approval no., H-1811-058-983). Due to the retrospective design of this study, the requirement for informed consent from individual patients was omitted.
Preoperative evaluation
All patients underwent a chest X-ray and contrast computed tomography (CT) scan, and all but 1 underwent a positron emission tomography (PET)/CT scan for stage evaluation. Preoperative histologic diagnosis of SCLC was made in some of the patients. Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) or mediastinoscopy were used for mediastinal staging in patients with a preoperative diagnosis of SCLC or if imaging studies showed suspected lesions. Brain magnetic resonance imaging (MRI) was performed in patients with a preoperative diagnosis of SCLC or a clinical stage ≥IB. All patients underwent pulmonary function tests, including carbon monoxide lung diffusion capacity.
Surgery
The standard surgery was lobectomy with systematic lymph node dissection. The surgical technique was modified according to the preoperative histologic diagnosis, pulmonary function, and tumor location. The primary approach has been minimally invasive since 2008, using video-assisted or robot-assisted thoracoscopic surgery.
Adjuvant treatment and surveillance
Depending on the patient’s performance status, they received adjuvant systemic therapy consisting of 4–6 cycles of etoposide/carboplatin or cisplatin. Prophylactic cerebral irradiation (PCI) of 25 Gy in 10 fractions was administered to patients with a pathologically advanced stage. Patients were followed up for 5 years with contrast chest CT every 6 months and PET-CT annually to monitor postoperative recurrence. All follow-up data were retrieved by reviewing the patients’ electronic medical records. Survival data of patients who were lost to follow-up were obtained from Statistics Korea, the Korean national statistical agency, and the cause of death was identified using the Korean Standard Classification of Diseases codes. The last follow-up date was the end of October 2021.
Statistical analysis
IBM SPSS ver. 24.0 (IBM Corp., Armonk, NY, USA) was used for all statistical analyses. Survival was calculated from the date of surgery to the date of the last follow-up visit or death. Subgroup characteristics were compared using the t-test for continuous variables and the chi-square test for categorical variables. Time-dependent variables, such as overall survival (OS) and freedom from recurrence (FFR), were assessed using Kaplan-Meier analysis. The log-rank test was performed to compare time-dependent variables between subgroups. A Cox proportional hazards model was used to assess risk variables. Following univariable analysis, those with a p-value <0.2 were included in the multivariable analysis. A p-value <0.05 was considered to indicate statistical significance.
Results
Of the 49 patients in the study cohort, 46 (93.9%) were men, and 44 (89.8%) were ex-smokers or current smokers. The median age of patients was 68.0 years (interquartile range [IQR], 62.0–75.0 years).
Preoperative workup
A preoperative histological diagnosis was made in 32 patients (65.3%) (SCLC/high-grade neuroendocrine carcinoma, 28; NSCLC, 3; and malignancy not otherwise specified, 1). A preoperative biopsy was not performed in 17 patients (highly risky biopsy, 7; radiologically indolent nodule, 4; clinical impression of intrapulmonary recurrence, 6). PET/CT and brain MRI were performed in 48 (98.0%) and 38 (77.6%) patients, respectively (Table 1). EBUS-TBNA was performed in 11 (39.3%) and mediastinoscopy in 3 (10.7%) of the 28 patients diagnosed preoperatively with SCLC. The clinical stage was cIA in 37 (75.5%), cIB in 9 (18.4%), and cIIA in 3 (6.1%) patients.
Table 1.
Preoperative patient characteristics (n=49)
| Characteristic | Value |
|---|---|
| Age (yr) | 68.0 (62.0–75.0) |
| Male sex | 46 (93.9) |
| Smoking | |
| Nonsmoker | 5 (10.2) |
| Ex-smoker | 30 (61.2) |
| Current smoker | 14 (28.6) |
| Smoking (pack-years) | 42.5 (30.0–50.0) |
| FEV1 (% predicted) | 97.0 (88.0–109.0) |
| DLCO (% predicted) | 92.0 (79.0–102.0) |
| Preoperative evaluation | |
| CT | 49 (100.0) |
| PET/CT | 48 (98.0) |
| EBUS-TBNA | 11 (22.4) |
| Brain MRI | 38 (77.6) |
| Clinical T stage | |
| cT1 | 37 (75.5) |
| cT2a | 9 (18.4) |
| cT2b | 3 (6.1) |
| Preoperative biopsy | |
| None | 17 (34.7) |
| SCLC or neuroendocrine carcinoma | 28 (57.1) |
| NSCLC | 3 (6.1) |
| Non-specific | 1 (2.0) |
| Comorbidity | |
| Diabetes mellitus | 11 (22.4) |
| Renal deficiency | 3 (6.1) |
| Underlying lung disease | 10 (20.4) |
| Cardiovascular disease | 22 (44.9) |
| Previous cancer history | 14 (28.6) |
Values are presented as median (interquartile range) or number (%).
FEV1, forced expiratory volume in 1 second; DLCO, carbon monoxide lung diffusion capacity; CT, computed tomography; PET/CT, positron emission tomography-computed tomography; EBUS-TBNA, endobronchial ultrasound-guided transbronchial needle aspiration; MRI, magnetic resonance imaging; SCLC, small cell lung cancer; NSCLC, non-small cell lung cancer.
Surgery, pathology, and adjuvant treatment
A minimally invasive approach was used in 38 patients (77.6%). Lobectomy or bilobectomy was performed in 40 cases (81.6%), and none of the patients underwent pneumonectomy. Sublobar resection was performed in 9 cases (18.4%) (compromised, 2; intentional, 5; and unanticipated metastasis, 2), and all cases of intentional sublobar resection were due to an incorrect intraoperative frozen section diagnosis. Following sublobar resection, systemic treatment was offered directly without a secondary completion of lobectomy (Table 2). The median number of dissected lymph nodes was 25.0 (IQR, 16.0–37.0), and systematic lymph node dissection was conducted in 41 cases (83.7%). Lymph node sampling was performed in patients with sublobar resection or severely calcified lymph nodes. Complete resection was achieved in 46 patients (93.9%). There were no cases of in-hospital mortality.
Table 2.
Intraoperative and postoperative data (n=49)
| Characteristic | Value |
|---|---|
| Surgical approach | |
| Thoracoscopic surgery | 38 (77.6) |
| Thoracotomy | 11 (22.4) |
| Extent of resection | |
| Sublobar resection | 9 (18.4) |
| Lobectomy | 38 (75.6) |
| Bilobectomy | 2 (4.1) |
| Surgical margin | |
| R0 resection | 46 (93.9) |
| R1/2 resection | 3 (6.1) |
| Histology | |
| Pure type | 40 (81.6) |
| Mixed type | 9 (18.4) |
| Pathologic T stage | |
| pT1 | 26 (53.1) |
| pT2a | 17 (34.7) |
| pT2b | 0 |
| pT3 | 6 (12.2) |
| Harvested lymph nodes | 25.0 (16.0–37.0) |
| Pathologic N stage | |
| pN0 | 39 (79.6) |
| pN1 | 8 (16.3) |
| pN2 | 2 (4.1) |
| Pathologic stage | |
| pIA | 21 (42.9) |
| pIB | 12 (24.5) |
| pIIA | 0 |
| pIIB | 12 (24.5) |
| pIIIA | 2 (4.1) |
| pIVA | 2 (4.1) |
| Adjuvant treatment | |
| Adjuvant chemotherapy | 39 (79.6) |
| Adjuvant CCRT | 1 (2.0) |
| Prophylactic cranial irradiation | 7 (14.3) |
| 90-Day mortality | 0 |
| Follow-up duration (mo) | 41.1 (23.1–70.1) |
Values are presented as number (%) or median (interquartile range).
CCRT, concurrent chemoradiation therapy.
The pathologic stages were pIA in 21 (42.9%), pIB in 12 (24.5%), pIIB in 12 (24.5%), and pIII/IV in 4 (8.2%) patients. Forty-one patients (83.7%) had pure SCLC and 8 patients (16.3%) had mixed SCLC combined with an NSCLC component. The concordance rate between the clinical and pathological stages was 44.9% (n=22). The rate of upstaging was 49.0% (n=24), while the rate of downstaging was 6.1% (n=3). The contributing factors for upstaging were the T factor in 12 (50%) (tumor size, 6; visceral pleural invasion, 5; satellite nodule in the same lobe, 1), the N factor in 10 (41.7%) (pN1, 8; pN2, 2), and the M factor in 2 patients (8.3%).
Regarding systemic adjuvant treatment, 39 patients (79.6%) completed the course, 4 (8.1%) discontinued treatment due to adverse effects, and 1 developed metastasis. Adjuvant therapy was not administered to 2 patients due to a lack of follow-up and to 3 due to old age and poor postoperative lung function. Adjuvant radiation therapy was performed in 1 patient (2.0%) with incomplete resection. PCI was performed in 7 patients (14.3%).
Prognosis
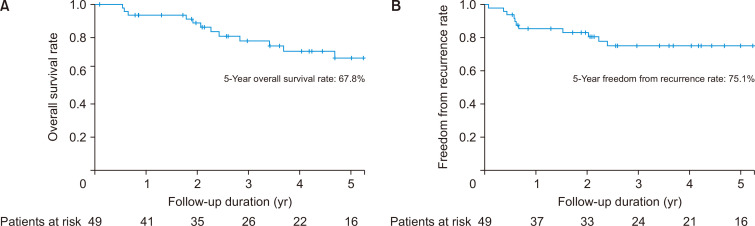
The median follow-up duration was 41.1 months (IQR, 23.1–70.1 months). The median survival duration and 5-year OS rate were 56.3 months and 67.8%, respectively (Fig. 2A), and the median duration of FFR and 5-year FFR rate were 50.9 months and 75.1%, respectively (Fig. 2B). Eleven patients (22.5%) experienced recurrence (locoregional, 3; pleura, 1; extrathoracic, 6; and combined, 1), and 14 patients died during the follow-up period (cancer-related, 8; treatment-related, 1; noncancer related, 3; second primary lung cancer, 1; and unknown, 1). The median time to recurrence of patients with recurrence was 7.5 months.
Fig. 2.
Kaplan-Meier curves for overall survival (A) and freedom from recurrence (B).
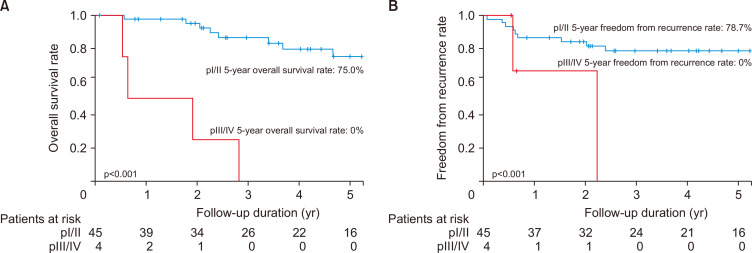
There were no significant differences in OS between stages c1A, c1B, and cIIA or between stages pI and pII (70.6% versus 88.9%, p=0.29). However, significant differences were identified between stages pI/II and pIII/IV in OS (5-year OS: 75.0% for pI/II versus 0% for pIII/IV, p<0.001) (Fig. 3A) and FFR (5-year FFR: 78.7% versus 0%, p<0.001) (Fig. 3B).
Fig. 3.
Kaplan-Meier curves for overall survival (A) and freedom from recurrence in pI/II and pIII/IV (B).
Univariable analyses revealed that the extent of resection, pathological stage, and adjuvant chemotherapy were significant prognostic factors for OS. In the multivariable analysis, pathological stage III/IV was the only significant prognostic factor for OS, with an unfavorable impact (Table 3).
Table 3.
Univariable and multivariable analysis for mortality
| Variable | Univariable analysis | Multivariable analysis | |||
|---|---|---|---|---|---|
|
|
|
||||
| Hazard ratio (95% CI) | p-value | Hazard ratio (95% CI) | p-value | ||
| Age | 1.023 (0.964–1.086) | 0.460 | |||
| FEV1 (% predicted) | 0.992 (0.953–1.032) | 0.695 | |||
| DLCO (% predicted) | 0.972 (0.943–1.002) | 0.064 | 0.980 (0.949–1.012) | 0.212 | |
| Current smoker | 0.698 (0.232–2.067) | 0.692 | |||
| Diabetes mellitus | 1.270 (0.352–4.583) | 0.715 | |||
| Underlying lung disease | 2.538 (0.847–7.604) | 0.096 | 0.908 (0.150–5.493) | 0.917 | |
| Cardiovascular disease | 1.140 (0.393–3.303) | 0.809 | |||
| cTNM stage (cIA vs.) | 0.841 | ||||
| cIB | 0.705 (0.156–3.171) | 0.649 | |||
| cIIA | 1.400 (0.178–11.013) | 0.749 | |||
| Thoracoscopic surgery (vs. thoraocotomy) | 0.967 (0.314–2.975) | 0.953 | |||
| Lobectomy/bilobectomy (vs. sublobar resection) | 0.202 (0.066–0.617) | 0.005 | 0.963 (0.124–8.558) | 0.972 | |
| Complete resection | 0.250 (0.053–1.183) | 0.080 | 1.262 (0.114–8.109) | 0.838 | |
| Mixed histologic type | 1.090 (0.301–3.943) | 0.895 | |||
| pTNM stage (I vs.) | <0.001 | <0.001 | |||
| II | 0.348 (0.043–2.782) | 0.319 | 0.348 (0.43–2.782) | 0.319 | |
| III/IV | 13.216 (3.382–51.646) | <0.001 | 13.216 (3.382–51.646) | <0.001 | |
| Adjuvant chemotherapy | 0.129 (0.104–0.941) | 0.039 | 0.554 (0.163–1.885) | 0.344 | |
| Prophylactic cranial irradiation | 1.881 (0.585–6.052) | 0.289 | |||
CI, confidence interval; FEV1, forced expiratory volume in 1 second; DLCO, carbon monoxide lung diffusion capacity; TNM, tumor-node-metastasis; cTNM, clinical TNM; pTNM, pathological TNM.
Discussion
Our study findings revealed favorable long-term survival following upfront surgery in clinical stage I–IIA SCLC patients, even though pathologic upstaging occurred in a substantial proportion. Pathological stage III/IV was a significant poor prognostic factor for OS.
The 5-year OS rate of the patients in our study who underwent upfront surgery in accordance with NCCN guidelines was 67.8%, which was compatible with previous reports of 48%–63% (Table 4) [7-10]. Given that the 5-year OS rate after surgery in early NSCLC was reported to be 78.1% [11], the prognosis after upfront surgery for early SCLC in our study is encouraging. Additionally, the performance of upfront surgery in early-stage SCLC in our study was safe, with no surgical mortality, and did not hinder adjuvant systemic treatment. Therefore, upfront surgery should be considered in cases of cI–IIA SCLC.
Table 4.
Reported prognoses of small cell lung cancer patients who underwent upfront surgery
| Authors | Period | Stage | No. of patients | 5-Year overall survival (%) |
|---|---|---|---|---|
| Combs et al. [7] (2015) | 1998–2011 | pIA | 407 | 50.0a) |
| pIB | 130 | 45.0 | ||
| Takei et al. [8] (2014) | 2004 | cIA | 132 | 63.3 |
| cIB | 36 | 45.7 | ||
| Takenaka et al. [9] (2015) | 1947–2011 | cI | 44 | 62.0 |
| Yang et al. [10] (2018) | 2003–2011 | cTI–II | 681 | 48.1 |
a)Lobectomy only.
Upstaging, especially nodal upstaging, is inevitable in lung cancer surgery because of the innate limitations of preoperative evaluation modalities. In our study, the upstaging rate of clinically early-stage SCLC was 49%, which was much higher than previous reports of 23%–25% [8,12]. Nodal upstaging occurred in approximately 20% of our study cohort. The rate of lymph node metastasis in early SCLC was reported as approximately 20%, which is significantly higher than the rate of 10% for early NSCLC [13,14]. It is believed that the higher rate of upstaging in SCLC is due to its aggressive nature. A study on data obtained from the National Cancer Database showed that the pN stage of SCLC patients who underwent surgery at cT1–2N0M0 was pN0 in 79.4%, pN1 in 15.5%, and pN2 in 5% [10], similar to the pathological stage distribution observed in our study. In our study, post hoc analysis revealed that the mean interval between the date of the PET/CT scan and the date of surgery was 36.2 and 23.6 days in the nodal and non-nodal upstaging groups, respectively (p=0.086).
However, nodal upstaging did not result in a poor prognosis in the pN1 subgroup in our study. Additionally, there was no significant difference in OS between pN0 and pN1 (80% versus 69% at 5 years, respectively; p=0.505). Several studies reported the 5-year survival rates of pN0 SCLC as 66%–72%, whereas those of pN1 were 10%–25%, reflecting a significant difference from the results of our study [15-17]. This can be attributed to 2 factors. First, the relatively superior prognosis of the pN1 subgroup in our study may be related to the selection of appropriate patients for surgery and accurate staging. The pN1 subgroup comprised highly selected patients with limited nodal metastasis who underwent a comprehensive evaluation for nodal metastasis by PET/CT and/or EBUS-TBNA prior to surgery, and those with clinical evidence of N1 disease were referred for nonsurgical treatment. The second factor may be the effect of extensive lymph node dissection and more accurate pathological staging. In our study, the median number of dissected and pathologically evaluated nodes was 25.0 (IQR, 16.0–37.0). Thus, the chance of undetected pN2 disease in the pN1 subgroup was minimal.
In contrast, the prognosis of pN2 in our study was extremely dismal, and there were no long-term survivors in the pN2 subgroup. This finding emphasizes once again the importance of comprehensive preoperative mediastinal staging in SCLC to avoid unnecessary surgery. Among 2 patients with pN2 disease, one had metastasis at station 5, which was inaccessible for EBUS, and the other had a false-negative EBUS-TBNA result at station 7. The sensitivity of EBUS-TBNA is around 49% (95% CI, 41%–57%) [18], and delayed surgery may affect pathologic upstaging [19]. Moreover, mediastinal lymph node dissection should be aggressively performed to achieve accurate pathological staging [20,21].
Postoperative systemic treatment was administered to 79.6% of our study cohort, indicating a high compliance rate. Univariable analysis revealed that adjuvant chemotherapy was a significant prognostic factor of OS. Zhou et al. [22] demonstrated that adjuvant chemotherapy increased survival in stage I–III SCLC (hazard ratio, 0.49; 95% CI, 0.29–0.80; p=0.005).
PCI in early-stage SCLC remains a matter of debate. In our study, univariate analysis did not identify PCI as a prognostic factor. Zhou et al. [22] reported that the prognostic effect of PCI was unclear. Several studies have demonstrated that PCI improves survival rates and the time to progression in limited stages [23,24]. However, the NCCN guidelines do not recommend PCI because patients in stage I–IIA who have undergone surgery are unlikely to develop brain metastasis [4]. In our study, brain metastasis occurred in 18.1% of patients who did not receive PCI.
Our study has some limitations. First, the number of patients was insufficient for the multivariable analysis to generate additional significant results. Further research on data from a larger population of patients with SCLC is expected to yield more significant results. Second, the retrospective nature of our study may have introduced selection bias. Therefore, randomized studies should be performed to validate our study findings. The lack of a control group made it impossible to determine whether early-stage SCLC could be effectively treated with upfront surgery. Nevertheless, because upfront surgery has been demonstrated to be beneficial for early-stage SCLC in other trials, the survival rates shown in our study indicate a significant improvement [8,9].
In conclusion, in clinical stages I–IIA of SCLC, upfront surgery followed by systemic treatment resulted in a favorable long-term prognosis in patients with clinical stage I–IIA SCLC, even though pathologic upstaging occurred in a substantial proportion of patients. Comprehensive mediastinal staging is crucial to rule out N2 disease prior to surgery since the prognosis of pIII/IV patients is dismal.
Article information
Author contributions
Conceptualization: HSW, JWS, IKP. Data curation: HSW, JWS. Formal analysis: HSW, IKP. Methodology: HSW, IKP. Project administration: HSW, JWS, IKP. Visualization: HSW, IKP. Writing–original draft: HSW, IKP. Writing–review & editing: HSW, IKP. Final approval of the manuscript: HSW, JWS, SP, IKP, CHK, YTK.
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- 1.Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2018. National Cancer Institute; Bethesda (MD): 2021. [Google Scholar]
- 2.Breitling LP, Rinke A, Gress TM. Recent survival trends in high-grade neuroendocrine neoplasms and lung cancer. Neuroendocrinology. 2020;110:225–33. doi: 10.1159/000500883. https://doi.org/10.1159/000500883. [DOI] [PubMed] [Google Scholar]
- 3.Shepherd FA, Crowley J, Van Houtte P, et al. The International Association for the Study of Lung Cancer lung cancer staging project: proposals regarding the clinical staging of small cell lung cancer in the forthcoming (seventh) edition of the tumor, node, metastasis classification for lung cancer. J Thorac Oncol. 2007;2:1067–77. doi: 10.1097/JTO.0b013e31815bdc0d. https://doi.org/10.1097/JTO.0b013e31815bdc0d. [DOI] [PubMed] [Google Scholar]
- 4.Amin MB, Edge SB, Greene FL, et al. Lung. In: Amin MB, Edge SB, Greene FL, et al., editors. AJCC cancer staging manual. 8th ed. Springer; Cham: 2017. pp. 431–55. [DOI] [Google Scholar]
- 5.Nicholson AG, Chansky K, Crowley J, et al. The International Association for the Study of Lung Cancer Lung Cancer Staging Project: proposals for the revision of the clinical and pathologic staging of small cell lung cancer in the forthcoming eighth edition of the TNM Classification for Lung Cancer. J Thorac Oncol. 2016;11:300–11. doi: 10.1016/j.jtho.2015.10.008. https://doi.org/10.1016/j.jtho.2015.10.008. [DOI] [PubMed] [Google Scholar]
- 6.Ettinger DS, Wood DE, Aisner DL, et al. NCCN guidelines insights: non-small cell lung cancer, version 2.2021. J Natl Compr Canc Netw. 2021;19:254–66. doi: 10.6004/jnccn.2021.0013. https://doi.org/10.6004/jnccn.2021.0013. [DOI] [PubMed] [Google Scholar]
- 7.Combs SE, Hancock JG, Boffa DJ, Decker RH, Detterbeck FC, Kim AW. Bolstering the case for lobectomy in stages I, II, and IIIA small-cell lung cancer using the National Cancer Data Base. J Thorac Oncol. 2015;10:316–23. doi: 10.1097/JTO.0000000000000402. https://doi.org/10.1097/JTO.0000000000000402. [DOI] [PubMed] [Google Scholar]
- 8.Takei H, Kondo H, Miyaoka E, et al. Surgery for small cell lung cancer: a retrospective analysis of 243 patients from Japanese Lung Cancer Registry in 2004. J Thorac Oncol. 2014;9:1140–5. doi: 10.1097/JTO.0000000000000226. https://doi.org/10.1097/JTO.0000000000000226. [DOI] [PubMed] [Google Scholar]
- 9.Takenaka T, Takenoyama M, Inamasu E, et al. Role of surgical resection for patients with limited disease-small cell lung cancer. Lung Cancer. 2015;88:52–6. doi: 10.1016/j.lungcan.2015.01.010. https://doi.org/10.1016/j.lungcan.2015.01.010. [DOI] [PubMed] [Google Scholar]
- 10.Yang CJ, Chan DY, Shah SA, et al. Long-term survival after surgery compared with concurrent chemoradiation for node-negative small cell lung cancer. Ann Surg. 2018;268:1105–12. doi: 10.1097/SLA.0000000000002287. https://doi.org/10.1097/SLA.0000000000002287. [DOI] [PubMed] [Google Scholar]
- 11.Shewale JB, Corsini EM, Correa AM, et al. Time trends and predictors of survival in surgically resected early-stage non-small cell lung cancer patients. J Surg Oncol. 2020;122:495–505. doi: 10.1002/jso.25966. https://doi.org/10.1002/jso.25966. [DOI] [PubMed] [Google Scholar]
- 12.Thomas DC, Arnold BN, Rosen JE, et al. Defining outcomes of patients with clinical stage I small cell lung cancer upstaged at surgery. Lung Cancer. 2017;103:75–81. doi: 10.1016/j.lungcan.2016.11.016. https://doi.org/10.1016/j.lungcan.2016.11.016. [DOI] [PubMed] [Google Scholar]
- 13.Haruki T, Takagi Y, Kubouchi Y, et al. Comparison between robot-assisted thoracoscopic surgery and video-assisted thoracoscopic surgery for mediastinal and hilar lymph node dissection in lung cancer surgery. Interact Cardiovasc Thorac Surg. 2021;33:409–17. doi: 10.1093/icvts/ivab112. https://doi.org/10.1093/icvts/ivab112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kneuertz PJ, Cheufou DH, D'Souza DM, et al. Propensity-score adjusted comparison of pathologic nodal upstaging by robotic, video-assisted thoracoscopic, and open lobectomy for non-small cell lung cancer. J Thorac Cardiovasc Surg. 2019;158:1457–66. doi: 10.1016/j.jtcvs.2019.06.113. https://doi.org/10.1016/j.jtcvs.2019.06.113. [DOI] [PubMed] [Google Scholar]
- 15.Casiraghi M, Sedda G, Del Signore E, et al. Surgery for small cell lung cancer: when and how. Lung Cancer. 2021;152:71–7. doi: 10.1016/j.lungcan.2020.12.006. https://doi.org/10.1016/j.lungcan.2020.12.006. [DOI] [PubMed] [Google Scholar]
- 16.Leuzzi G, Lococo F, Alessandrini G, et al. Prognostic impact of node-spreading pattern in surgically treated small-cell lung cancer: a multicentric analysis. Lung. 2017;195:107–14. doi: 10.1007/s00408-016-9954-4. https://doi.org/10.1007/s00408-016-9954-4. [DOI] [PubMed] [Google Scholar]
- 17.Yang H, Mei T. The prognostic value of lymph node ratio in patients with surgically resected stage I-III small-cell lung cancer: a propensity score matching analysis of the SEER database. Eur J Cardiothorac Surg. 2021;60:1212–20. doi: 10.1093/ejcts/ezab287. https://doi.org/10.1093/ejcts/ezab287. [DOI] [PubMed] [Google Scholar]
- 18.Leong TL, Loveland PM, Gorelik A, Irving L, Steinfort DP. Preoperative staging by EBUS in cN0/N1 lung cancer: systematic review and meta-analysis. J Bronchology Interv Pulmonol. 2019;26:155–65. doi: 10.1097/LBR.0000000000000545. https://doi.org/10.1097/LBR.0000000000000545. [DOI] [PubMed] [Google Scholar]
- 19.Bott MJ, Patel AP, Crabtree TD, et al. Pathologic upstaging in patients undergoing resection for stage I non-small cell lung cancer: are there modifiable predictors? Ann Thorac Surg. 2015;100:2048–53. doi: 10.1016/j.athoracsur.2015.05.100. https://doi.org/10.1016/j.athoracsur.2015.05.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lad T, Piantadosi S, Thomas P, Payne D, Ruckdeschel J, Giaccone G. A prospective randomized trial to determine the benefit of surgical resection of residual disease following response of small cell lung cancer to combination chemotherapy. Chest. 1994;106(6 Suppl):320S–323S. doi: 10.1378/chest.106.6_supplement.320s. https://doi.org/10.1378/chest.106.6_supplement.320s. [DOI] [PubMed] [Google Scholar]
- 21.Badzio A, Kurowski K, Karnicka-Mlodkowska H, Jassem J. A retrospective comparative study of surgery followed by chemotherapy vs. non-surgical management in limited-disease small cell lung cancer. Eur J Cardiothorac Surg. 2004;26:183–8. doi: 10.1016/j.ejcts.2004.04.012. https://doi.org/10.1016/j.ejcts.2004.04.012. [DOI] [PubMed] [Google Scholar]
- 22.Zhou N, Bott M, Park BJ, et al. Predictors of survival following surgical resection of limited-stage small cell lung cancer. J Thorac Cardiovasc Surg. 2021;161:760–71. doi: 10.1016/j.jtcvs.2020.10.148. https://doi.org/10.1016/j.jtcvs.2020.10.148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Patel S, Macdonald OK, Suntharalingam M. Evaluation of the use of prophylactic cranial irradiation in small cell lung cancer. Cancer. 2009;115:842–50. doi: 10.1002/cncr.24105. https://doi.org/10.1002/cncr.24105. [DOI] [PubMed] [Google Scholar]
- 24.Eze C, Roengvoraphoj O, Niyazi M, et al. Treatment response and prophylactic cranial irradiation are prognostic factors in a real-life limited-disease small-cell lung cancer patient cohort comprehensively staged with cranial magnetic resonance imaging. Clin Lung Cancer. 2017;18:e243–9. doi: 10.1016/j.cllc.2016.11.005. https://doi.org/10.1016/j.cllc.2016.11.005. [DOI] [PubMed] [Google Scholar]