Abstract
The kidney is a mitochondria-rich organ, and kidney diseases are recognized as mitochondria-related pathologies. Intact mitochondrial DNA (mtDNA) maintains normal mitochondrial function. Mitochondrial dysfunction caused by mtDNA damage, including impaired mtDNA replication, mtDNA mutation, mtDNA leakage, and mtDNA methylation, is involved in the progression of kidney diseases. Herein, we review the roles of mtDNA damage in different setting of kidney diseases, including acute kidney injury (AKI) and chronic kidney disease (CKD). In a variety of kidney diseases, mtDNA damage is closely associated with loss of kidney function. The level of mtDNA in peripheral serum and urine also reflects the status of kidney injury. Alleviating mtDNA damage can promote the recovery of mitochondrial function by exogenous drug treatment and thus reduce kidney injury. In short, we conclude that mtDNA damage may serve as a novel biomarker for assessing kidney injury in different causes of renal dysfunction, which provides a new theoretical basis for mtDNA-targeted intervention as a therapeutic option for kidney diseases.
Keywords: mitochondrial DNA, kidney diseases, mtDNA replication, mtDNA mutation, mtDNA leakage, mtDNA methylation
1. Introduction
The kidney removes waste metabolic products through the glomerular filtration barrier and maintains water–electrolyte balance via renal tubular reabsorption. As an organ with high energy demand, the kidney has abundant mitochondria to generate ATP to sustain its internal homeostasis. An increasing number of studies have revealed that mitochondrial dysfunction plays a vital role in the occurrence and progression of kidney diseases, including acute kidney injury (AKI) and chronic kidney disease (CKD) [1,2]. However, the mechanism of mitochondrial dysfunction remains undefined.
Mitochondrial DNA (mtDNA) is a double-stranded circular DNA that is independent of nuclear DNA. Normally, mtDNA with a length of 16,596 base pairs is located in the mitochondrial matrix. mtDNA possesses its own transcriptional and translational system and encodes 2 rRNAs, 22 tRNAs, and 13 polypeptides. These polypeptides include ND1-6, ND4L, COXⅠ-Ⅲ, cyt-b, ATPase6, and ATPase8, which are involved in the composition of the mitochondrial respiratory complexes to maintain the integrity of the electron transport chain (ETC) and the stability of oxidative phosphorylation (OXPHOS) [3]. The functioning of OXPHOS provides physiologically required energy to cells and organs, and the ETC is the main source of reactive oxygen species (ROS) production. mtDNA damage leads to inefficient mitochondrial function, such as ROS overproduction, decreased ATP generation and altered metabolite profiles [4]. Damaged mtDNA was accompanied by oxidative stress activation and reduced mitochondrial mass [5]. Importantly, mtDNA integrity is closely related to mitochondrial function. In addition to determining mitochondrial function directly, mtDNA can also act as an endogenous pathogenic factor. Curiously, the latest studies have reported that mtDNA leaking into the cytoplasm can act as an inflammatory mediator and activate the natural immune inflammatory response [6,7].
The process of DNA transcription and replication is often accompanied by mutations, deletions, insertions, translocations and strand breaks. These damages could be rapidly reversed by the nuclear DNA repair system to promote genomic integrity [8]. However, mtDNA is absent of mature damage-sensing signaling and protective histones. Excessive ROS production mediates oxidative stress damage in cells and can also exacerbate mtDNA mutations, strand breaks and deletions, creating a vicious cycle of mtDNA damage [9]. Therefore, it is more sensitive to be impaired by internal and external unfavorable factors.
The roles of mtDNA damage in multiple diseases, such as cancers, cardiovascular diseases, liver diseases, and neurological diseases, have attracted much attention [10,11,12,13]. Similarly, it has been reported that mtDNA damage, including impaired mtDNA replication, mtDNA mutations, mtDNA leakage, and mtDNA modification, plays an important role in the progression of kidney diseases (Figure 1). In this review, we summarize the roles of mtDNA damage in kidney diseases and highlight the potential value for diagnostic and therapeutic targets.
Figure 1.
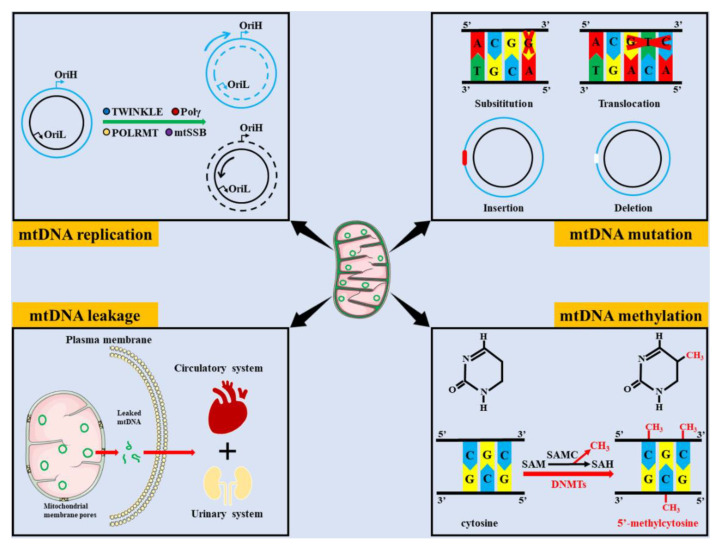
Common types of mtDNA damage. mtDNA damage includes impaired mtDNA replication, mtDNA mutations, mtDNA leakage and mtDNA methylation. mtDNA replication operates in a semi-conserved manner and contains multiple enzymes, such as TWINKLE, Pol γ, POLRMT, and mtSSB, which may hinder mtDNA replication when they are disrupted. The common types of mtDNA mutation includes substitution, translocation, insertion, and deletion. Leaked mtDNA can be transferred to peripheral plasma and urine via the circulatory and urinary systems, respectively. Under the action of DNMTs, the methyl donor compounds derived from SAM are transferred to CpG islands to form 5′-methylcytosine. (OriH, the origin of heavy strand replication; OriL, the origin of light strand replication; Pol γ, polymerase gamma; POLRMT, mitochondrial RNA polymerase; mtSSB, mitochondrial single-strand binding protein; SAM, S-adenosyl-L-methionine; SAH, S-adenosyl-L-homocysteine; SAMC, S-adenosylmethionine carrier; DNMTs, and DNA methyltransferases).
2. Common Types of mtDNA Damage
2.1. Impaired mtDNA Replication
The stability of mtDNA is essential for maintaining a healthy function of mitochondria within cells. mtDNA replication and distribution within mitochondrial networks play an important role in sustaining mitochondrial homeostasis. Similar to nuclear DNA replication, mtDNA replication operates in a semiconserved manner and entails multiple types of mechanisms, including strand-displacement and strand-coupled models [14]. In each cell cycle, mtDNA replicates multiple times, and the replication of both strands, the heavy strand and light strand, is not synchronized. mtDNA replication is closely related to mitochondrial metabolism, and its activity could be affected by alterations in specific mitochondrial metabolites, such as nucleotides and NAD+. Interestingly, exogenous supplementation with the NAD+ precursor, beta-nicotinamide mononucleotide, increased the pool of mitochondrial nucleotides, and promoted mtDNA replication [15]. Decreased mtDNA copy number is associated with increased oxidative stress through ROS overproduction, which further results in mitochondria-related metabolic disorders and apoptosis [16].
A variety of related enzymes and regulatory factors are involved in mtDNA replication, such as mtDNA polymerase γ (POL γ), mitochondrial single-strand binding protein (mtSSB), mitochondrial helicase TWINKLE, topoisomerase, and mitochondrial transcription factor A (TFAM) [17,18]. All of these factors are mainly encoded by nuclear genes. Therefore, mtDNA replication is regulated by both its own and nuclear DNA. POL γ-deficient cells experience severe deletion of mtDNA, which is rescued by enhancing mitochondrial deoxyribonucleoside triphosphate production [19]. Activating transcription factor associated with stress 1 (ATFS-1) is absent in healthy mitochondria owing to its degradation by the mtDNA-bound protease Lon peptidase 1 (LONP-1), but it accumulates in damaged mitochondria. LONP-1 inhibition increases ATFS-1 and PLO γ binding to mtDNA and promotes mtDNA replication thereby improving the mtDNA heteroplasmy ratio and restoring OXPHOS [20]. mtSSB is critically required for restricting transcription initiation to optimize RNA primer formation at two origins of mtDNA replication and its mutations affect mtDNA replication and induce mtDNA deletion [21,22]. TFAM deficiency aggravates reduction of mtDNA copy number and OXPHOS [23]. In summary, complete mtDNA replication is an orderly process. When any one of these steps is disrupted, it may cause impaired mtDNA replication.
2.2. mtDNA Mutations
mtDNA appears to be more frequent and susceptible to mutations than nuclear DNA. mtDNA mutations are commonly found in maternally inherited diseases, and unfavorable environmental factors can also cause sporadic mtDNA mutations [24]. Both the gene-coding region and displacement loop (D-loop), which plays a role in transcriptional regulation, can be mutated in mtDNA, thus affecting the vital regions of the genome.
Because each mitochondrion contains a different amount mtDNA copies, the types of mtDNA mutations can be divided into homogeneous and heterogeneous mutations. Homogeneous mutations refer to the mutation of all mtDNA in mitochondria, whereas heterogeneous mutations refer to the coexistence of mutant and wild-type mtDNA. mtDNA mutations have the cumulative effect of defective mitochondrial function, causing impairment of a cellular energy supply [25]. The biological consequences of mutations depend on the proportion of mutant mtDNA and the type of mutation carried by the cells. The minimum number of copies of mtDNA mutations that cause dysfunction in specific tissues and organs is called the threshold value, and the lower the threshold value, the greater is the probability of disease occurrence [26]. The mutation threshold value has important implications for the clinical manifestation of energy dependence and the disease and the threshold value are different between individual tissues and organs.
With the continuous improvement of detection technologies, mtDNA mutation-related diseases are being discovered and increasingly gain the attention of scholars and clinicians. Studies have found that mtDNA mutations are associated with the development of numerous diseases, including cardiovascular diseases, kidney diseases, tumors, and aging [27,28,29,30]. Currently, there is a lack of effective treatment for mtDNA mutation-related diseases, which mainly focuses on improving clinical symptoms. Therefore, it is particularly important to accurately detect the sites of mtDNA mutations.
2.3. mtDNA Leakage
The mitochondrial membrane is similar to the cell membrane and has a bilayer structure to maintain the integrity of mitochondria. Lipids and proteins are the main components of the inner mitochondrial membrane (IMM) and outer mitochondrial membrane (OMM). The difference in their composition determines that the IMM and OMM have different physiological functions. The OMM contains a large number of integral membrane proteins that regulates mitochondrial permeability. In contrast, the IMM is composed of a variety of carrier proteins and metabolism-related enzymes that are responsible for complex mitochondrial biochemical reactions. Normally, mtDNA is encapsulated in the mitochondrial matrix. Once the structural integrity of mitochondrial membrane is disrupted, mtDNA is released into the cytoplasm. Defective mitochondrial membrane structure and increased permeability are key causes of mtDNA leakage. Altered lipid composition in the mitochondrial membrane leads to increased mitochondrial permeability and mtDNA leakage [31]. Mitochondrial damage caused by multiple factors is often accompanied by mtDNA leakage. Recent studies revealed that mtDNA can be released into the cytoplasm through several pathways, such as the BAK/BAX pore, voltage-dependent anion channel (VDAC) oligomer pore and mitochondrial permeability transition pore (mPTP) [32,33,34], as shown in Figure 2. However, these processes have been studied to an inconsistent extent among different diseases.
Figure 2.
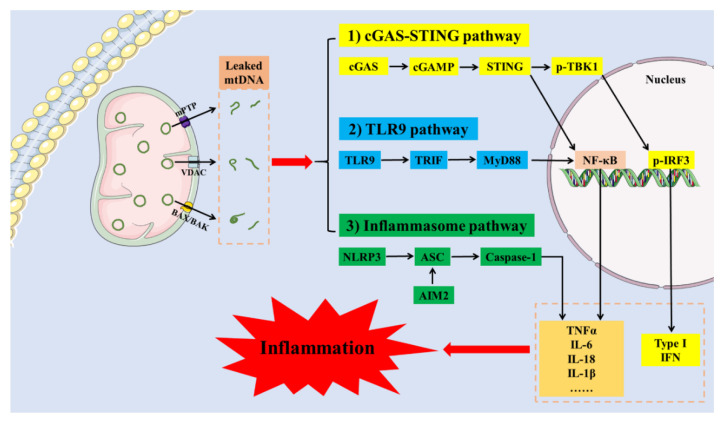
mtDNA leakage induces inflammation activation. mtDNA is released into the cytoplasm through several pathways, such as the BAK/BAX pore, VDAC oligomer pore, and mPTP. mtDNA released into the cytoplasm activates inflammatory responses through multiple signaling pathways, including cGAS-STING, TLR9, NLRP3, and AIM2 inflammasome. (cGAS, cyclic GMP-AMP synthase; STING, stimulator of interferon genes; cGAMP, cyclic guanosine monophosphate-adenosine monophosphate; p-TBK1, phospho-TANK-binding kinase-1; TLR9, toll-like receptor 9; TRIF, TIR domain-containing adapter-inducing IFN β; MyD88, myeloid differentiation protein 88; NLRP3, nod-like receptor pyrin 3; ASC, apoptosis associated speck like protein; AIM2, absent in melanoma 2; VDAC, voltage-dependent anion channel; mPTP, mitochondrial permeability transition pore; TNFα, tumor necrosis factor α; IL-6, interleukin-6; IL-18, interleukin-18; IL-1β, interleukin-1β; NF-κB, nuclear factor kappa-B; p-IRF3, phospho-interferon regulatory factor-3; IFN, interferon).
Accumulated mtDNA in the cytoplasm is recognized as an endogenous pathogen and activates innate immune and inflammatory responses [12,35]. mtDNA released into the cytoplasm can activate inflammatory responses through multiple signaling pathways, including the cyclic GMP-AMP synthase (cGAS)-stimulator of interferon genes (STING), toll-like receptor 9 (TLR9), nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) and absent in melanoma (AIM2) inflammasome signaling pathways [36]. cGAS is a member of the nucleotidyl transferase family and contains a DNA-binding region, which recognizes endogenous and exogenous DNA, including viral DNA, mtDNA, chromosomal terminal telomeric repeat sequence DNA, and cytoplasmic chromatin fragments. The recognition of DNA by cGAS is length-dependent, and DNA can effectively bind to cGAS when the length of dsDNA is >45 bp [37]. The phosphoribose backbone of DNA binds to cGAS in a non-sequence-dependent manner and induces conformational changes [38]. mtDNA released into the cytoplasm or peripheral circulation is recognized by cGAS and catalyzes the generation of the second messenger cyclic guanosine monophosphate-adenosine monophosphate (cGAMP), which further activates STING-related pathways, including the type 1 interferon (IFN) response and the classical NF-κB inflammatory pathway, thereby activating the immune inflammation [39]. TLR9 is a transmembrane protein mainly located in the endoplasmic reticulum, which contains an extracellular region that recognizes pathogen-associated molecular patterns (PAMPs) and an intracellular region including a toll/interleukin-1 receptor (TIR) structure for downstream signaling. TLR9 can act as a DNA recognition receptor and participate in the natural immune response in human body. A growing number of studies suggest that TLR9 plays an important role in the development of autoimmune diseases [40,41]. TLR9 is a member of the TLR family of proteins that are most closely related to mtDNA [42]. mtDNA activates TLR9 and is involved in cytokine production, splenic apoptosis and renal injury in sepsis [43]. TLR9 mediates the formation of inflammatory responses by activating the NF-κB inflammatory pathway via myeloid differentiation factor 88 (MyD88) [44]. NLRP3 activates caspase-1 and gasdermin D (GSDMD), releasing large amounts of inflammatory factors and initiating pyroptosis, a new orderly mode of cell death [45]. NLRP3 can recognize both PAMPs and danger-associated molecular patterns (DAMPs), and activate NLRP3 inflammasome, which consists of NLRP3 receptor protein, apoptosis-associated speck-like protein (ASC) and caspase-1 precursor protein (pro-caspase-1). A recent study found that mtDNA leaking into the cytoplasm activated NLRP3 inflammasome in brown adipose tissue, which is engaged in obesity-induced insulin resistance and impaired thermogenesis [46]. The double-stranded DNA receptor AIM2 also recognizes mtDNA released through BAK/BAX pores, and triggers IL-1β secretion and pyroptosis [47].
2.4. mtDNA Methylation
DNA methylation is one of the most widely studied epigenetic mechanisms. Under the action of DNA methyltransferases (DNMTs), the methyl donor compounds derived from S-adenosylmethionine (SAM) are transferred to CpG islands to form 5′-methylcytosine (5′-mC), which is the most common type of DNA methylation. DMNTs include two major groups, DNMT3a and DNMT3b, which catalyze the de novo methylation of unmethylated DNA duplexes, and DNMT1, which maintains the methylation state of DNA after semiconserved replication. It is reported that DNMTs plays a significant role in regulating tricarboxylic acid metabolites, mitochondrial respiration and oxidative stress [48,49].
Recent studies have revealed that mtDNA can also be methylated and methylated mtDNA underlies disease progression [50]. The levels of mtDNA methylation may be influenced by a number of intracellular or extracellular factors. Significant abnormalities in mtDNA methylation occur in a variety of diseases, particularly in mitochondria-related diseases [51]. Methylation of mitochondrial genome could lead to the etiology of human disease and altered methylation levels of mtDNA were assessed in animal models and human tissue from patients with obesity, diabetes, cancer, and cardiovascular and neurodegenerative diseases [14]. Hypermethylation of mtDNA genes also induces systemic insulin resistance and a related metabolic disorder [52].
mtDNA methylation is an emerging and incompletely understood phenomenon that regulates mitochondrial function. mtDNA methylation drift at different sites of coding gene loci results in decreased mtDNA copy number and altered gene expression [53]. The degree of mtDNA methylation was negatively correlated with mtDNA content [54]. mtDNA methylation can be considered as an early molecular event and a potential biomarker for effective disease prediction and diagnosis. mtDNA methylation is a reversible epistatic modification, making it an important therapeutic target. Gene editing of mtDNA methylation is still in the basic research and early clinical stage and, as such, safety issues cannot be ignored. It is hoped that with the development of mitochondrial gene editing technology, it will help us to further understand how mtDNA is methylated.
3. mtDNA Distribution in Kidney Diseases
Generally, intact mtDNA is present in the mitochondrial matrix but not in the cellular matrix, peripheral blood, or urine. However, mitochondrial damage contributes to cell injury in multiple diseases and is often followed by mtDNA leakage from mitochondria. When leaked mtDNA are insufficient to clean by cellular repair and phagocytosis system, they could be released into the peripheral circulation. mtDNA in the peripheral circulation filtrates through the glomerular filtration barrier and participates in the formation of urine. Cell detachment in the urinary system, such as the bladder and ureter, can also lead to the presence of mtDNA in the urine. Therefore, mtDNA can be detected in both peripheral plasma and urine. The levels of mtDNA in peripheral blood and urine can be used to assess mitochondrial function and the status of some organs. There is also an increasing number of studies on the correlations of mtDNA distribution and kidney function (Figure 3).
Figure 3.
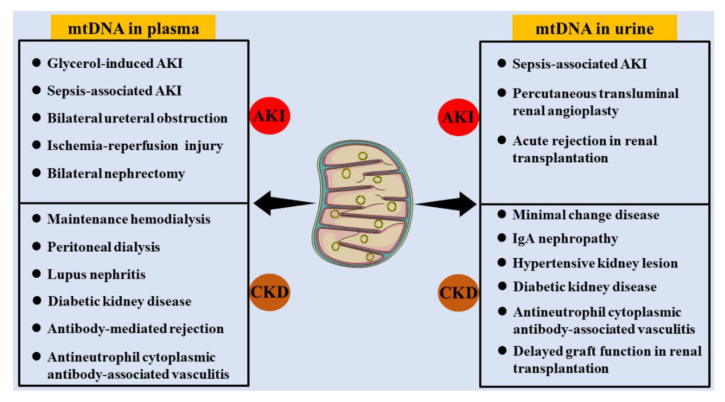
mtDNA distribution in kidney diseases. mtDNA can be detected in both peripheral plasma and urine of multiple kidney diseases including AKI and CKD. (AKI, acute kidney injury; CKD, chronic kidney disease; IgA, Immunoglobulin A).
3.1. mtDNA in Peripheral Serum
The levels of mtDNA in peripheral serum is relatively low under normal physiological conditions, and its concentration is increased with damage to several organs or tissues, such as the kidney, heart, liver, brain, and muscle [55,56,57,58]. A correlation between plasma mtDNA and kidney diseases, including AKI and CKD, has been reported. In addition, plasma mtDNA has been considered as an indicator for assessing kidney injury.
Numerous factors can trigger the occurrence of AKI, including bilateral ureteral obstruction, sepsis-associated AKI, glycerol-induced AKI, ischemia-reperfusion injury (IRI) and bilateral nephrectomy [59]. The predictive role of plasma mtDNA in AKI has been taken into consideration. For example, the plasma mtDNA level was increased in AKI patients with sepsis [60]. In glycerol-induced AKI rats, the concentration of plasma mtDNA was increased after 3 h, indicating that plasma mtDNA could be an early and sensitive biomarker of AKI [61].
A later study in chronic renal insufficiency cohort reported that the lower mtDNA copy number was correlated with a higher risk of CKD progression, independent of established risk factors in CKD patients [62]. mtDNA release in platelets driven by a receptor for immune complex FcγRIIA is the key source of mitochondrial antigens in systemic lupus erythematosus [7]. By pumping excess mtDNA into the circulation of mice, a high level of serum mtDNA can trigger inflammation and induce kidney damage [63]. Plasma mtDNA is a strong predictor of cardiovascular events as well as the need for hospitalization in patients with peritoneal dialysis [64]. In patients undergoing maintenance hemodialysis (MHD), circulating mtDNA contents were significantly higher in sarcopenia patients, together with higher TLR9 and IL-6 expression, which demonstrated that mtDNA could be involved in the pathogenesis of MHD-related sarcopenia [65]. As one of the indispensable proteins encoded by mtDNA, serum ND6 was increased in active antineutrophil cytoplasmic antibody-associated vasculitis, and ND6 concentration was negatively correlated with the percentage of normal glomeruli in kidney biopsies [66]. These studies revealed that serum mtDNA reflected immune inflammatory status and kidney injury.
Serum mtDNA-mediated immune rejection determines the efficacy of renal transplantation. The higher the level of serum mtDNA in the kidney donor, the more likely kidney transplant recipient is to experience antibody-mediated rejection. Therefore, donor serum mtDNA can be used as a predictive marker for antibody-mediated rejection and for validated donor organ evaluation [67]. Accordingly, donor plasma mtDNA was an independent risk factor for delayed graft function (DGF) of renal recipient, which was valuable in organ evaluation [68].
3.2. mtDNA in Urine
mtDNA in urine can be used as an indicator to assess kidney function. An acute rise in urinary mtDNA by percutaneous transluminal renal angioplasty reflects renal mitochondrial damage and thus inhibits renal recovery [69]. In patients with sepsis, elevated urinary mtDNA levels were associated with mitochondrial dysfunction and renal injury, suggesting that sepsis causes renal mitochondrial injury. Therefore, urinary mtDNA may be considered a valuable biomarker for determining the development of AKI and mitochondria-targeted therapy following sepsis-induced AKI [70]. Compared to healthy controls, the expression of STING in kidney was increased and urinary mtDNA levels were elevated in minimal change disease (MCD) patients, which could be used as a valuable prognostic marker in MCD [71]. Urinary mtDNA levels were significantly elevated in both diabetic patients and mice, which was negatively correlated with glomerular filtration rate and positively correlated with interstitial fibrosis [63,72]. mtDNA was also readily detected in the urinary supernatant of nondiabetic CKD and its level correlated with the rate of decline in renal function and predicted the risk of elevated serum creatinine and the need for dialysis in CKD patients [73]. Low urinary mtDNA was significantly correlated with favorable renal outcomes at 6 months follow-up, indicating the novel prognostic role of mtDNA for renal outcome in CKD patients [74]. In renovascular hypertensive patients, elevated urinary mtDNA copy numbers were correlated with mitochondrial dysfunction and renal injury, including increased urinary neutrophil gelatinase-associated lipocalin, kidney injury molecular-1 (KIM-1) levels and decreased estimated glomerular filtration [75,76]. Higher urinary mtDNA copy numbers and higher mean annual rates of estimated glomerular filtration rate (eGFR) decline were displayed in minor glomerular abnormalities and IgA nephropathy (IgAN) patients, and mitochondrial injury could be prior to pathological changes and increased proteinuria [77,78]. Urinary mtDNA was elevated in antineutrophil cytoplasmic autoantibodies-associated vasculitis (ANCA-AAV) patients suffering from abnormal kidney function and its level correlated with the severity of renal injury and pathological neutrophil infiltration [79]. Urinary mtDNA level was corelated with cold ischemia time and renal function in human renal transplant recipients, which was associated with renal allograft function and the diagnosis of DGF following renal transplantation [80]. The urinary mtDNA level was significantly higher in patients with acute rejection and DGF, which could be predictive of short-term post-transplant renal function [81]. Taken together, the above studies demonstrate that mtDNA in urine is closely associated with alterations in renal function in a variety of kidney diseases and a high level of urinary mtDNA is an unfavorable factor.
4. mtDNA Damage in Kidney Diseases
4.1. Impaired mtDNA Replication
mtDNA replicates in cultured cells from kidney through the asynchronous mechanism [82]. Decreased mtDNA copy number in blood specimens was associated with abnormal levels of serum creatinine, which indicated impaired kidney function [83]. During mtDNA replication, the role of mtSSB1 is to protect displaced single-stranded DNA from damage, to prevent the formation of DNA secondary structures and the binding of inappropriate DNA synthesis and catabolic enzymes. Gustafson et al. reported a case of young CKD patient harboring a mtSSB1 mutation (p.E27K) accompanied by a single large-scale mtDNA deletion [84]. mtDNA content of the kidney was also significantly decreased in SSBP1 mutation (p.R107Q) patients who displayed impaired OXPHOS and kidney function insufficiency requiring transplantation [85]. Except for mtDNA of brain compartments, kidney mtDNA was the most vulnerable to accumulation of age-related damage and the copy number of mtDNA in kidney of aged rats was significantly increased [86]. Furthermore, mtDNA levels were significantly higher in the proximal and distal tubules than in the glomerular and collecting duct epithelium of kidney. With increasing age, mtDNA content was decreased in renal tubules, which was consistent with the gradual decline of kidney function and could be reversed by caloric restriction [87]. Overall, mtDNA replication is a biomarker of mitochondrial function, which is associated with increased mortality and morbidity in age-related diseases [88].
Recent studies have revealed that impaired mtDNA replication contributed to AKI. mtDNA replication and content were decreased with enhanced mitophagy in kidney, which contributed to the occurrence of AKI and increased mortality in liver transplantation rats [89]. mtDNA copy number was decreased in renal fibrosis models, including unilateral ureteral obstruction (UUO) and IRI, accompanied with mitochondrial dysfunction and oxidative stress [90]. Hypoxia inducible factor-1α (HIF-1α)-BCL2/adenovirus E1B 19 kDa protein-interacting protein 3 (BNIP3)-mediated mitophagy regulated mtDNA copy number and ROS production and inhibited cell apoptosis in renal tubular cells of IRI model [91].
Various causes of CKD exist with abnormal mtDNA replication. The reduced mtDNA copy number in peripheral blood mononuclear cells of MHD patients predicted poor clinical outcomes [92]. Consistently, mtDNA copy number was decreased in kidneys of diabetic mice, accompanied by downregulated TFAM expression and ATP production [93]. Similarly, our previous study demonstrated that impaired mtNDA replication in podocytes contributed to kidney injury in diabetic kidney disease (DKD) [94]. mtDNA deletion also aggravated podocyte injury and depletion, which was involved in the pathogenesis of focal segmental glomerulosclerosis (FSGS) [95]. A decrease in mtDNA content was the main cause of reduced OXPHOS in chromophobe renal cancer (ChRCC) [96]. The latest study showed that mtDNA replication defects led to the formation of mtDNA linear deletion, which triggered an immune response and led to progressive kidney disease in aging animals [97].
4.2. mtDNA Mutations
The kidney is not only an organ with high mtDNA replication. It also contains multiple mtDNA mutation sites that can be mutated [98]. The downstream effect of mtDNA mutations is mitochondrial dysfunction. mtDNA mutations usually cause systemic diseases, which may also be associated with the progression of AKI and CKD [28]. The kidney diseases caused by mtDNA mutations at different loci are summarized in Table 1.
Table 1.
mtDNA mutations in kidney diseases.
| Mutation | Gene | Clinical Characters | Diagnosis | Reference |
|---|---|---|---|---|
| m.616T>C | MT-tRNAPhe | Recurrent swelling and pain | CKD | [28] |
| Renal insufficiency | Hyperuricemia | |||
| m.3243A>G | MT-TL1 | Chest tightness and shortness of breath | Membranous nephropathy | [99] |
| Hyperlactatemia | ||||
| Hyperuricemia | ||||
| Proteinuria | ||||
| m.6145G>A | MT-CO1 | Chronic tubulointerstitial changes | Rhabdomyolysis | [100] |
| Elevated serum creatinine level Dark-colored urine Mitochondrial abnormalities | ||||
| m.616T>C | MT-tRNAPhe | Developmental delay Epilepsy Hypertension Electrolyte disturbance Chronic renal insufficiency |
Autosomal-dominant tubulointerstitial kidney disease | [101] |
| m.13513G>A m.13514G>A |
MT-ND5 | Anuric AKI | Tubulo-interstitial kidney disease | [102] |
| Acute pulmonary edema | ||||
| Hyperlactatemia with metabolic acidosis | ||||
| Proteinuria | ||||
| Hypertension | ||||
| m.4216T>C | MT-ND1 | Higher levels of fasting glucose | DKD | [103] |
| Decreased renal function | ||||
| 3571_3572insC | MT-ND1 | - | Renal oncocytoma | [104] |
| 3571delC | ||||
| 10952_10953insC | MT-ND4 | |||
| 11038delA | ||||
| 12384_12385insT | MT-ND5 | |||
| 12390_12391insC | ||||
| m.13493T>C | ||||
| m.3243A>G | MT-TL1 | |||
| m.3565T>AC | MT-ND1 | - | ChRCC | [105] |
| m.3922G>A | ||||
| m.4569G>A | MT-ND2 | |||
| m.4969G>C | ||||
| m.10806G>A | MT-ND4 | |||
| m.11866A>AC | ||||
| m.12384TC>T | MT-ND5 | |||
| m.12417C>CA | ||||
| m.13127AC>A | ||||
| m.13206CTG>C | ||||
| m.13230CA>C | ||||
| m.14159C>A | MT-ND6 | |||
| m.6490T>C | MT-CO1 | |||
| m.9651C>T | MT-CO3 | |||
| m.3243A>G | MT-TL1 | Proteinuria Decreased eGFR Hyperuricemia |
FSGS | [106] |
| Nephrosclerosis | ||||
| DKD | ||||
| Tubulointerstitial nephropathy | ||||
| Minor glomerular abnormality | ||||
| m.3243A>G | MT-TL1 | Osteoporosis | Nephrolithiasis | [107] |
| Bilateral sensorineural deafness | ||||
| Sensory axonal neuropathy | ||||
| m.6129G>A | MT-CO1 | - | Von Hippel-Lindau renal oncocytoma | [108] |
| m.8993T>G | MT-ATP6 | Proteinuria Decreased eGFR |
Neuropathy, ataxia and retinitis pigmentosa syndrome | [109] |
| End-stage renal disease | ||||
| m.3243A>G | MT-TL1 | Wolff-Parkinson-White syndrome Proteinuria |
Chronic progressive external ophthalmoplegia | [110] |
| FSGS | ||||
| m.547A>T | MT-HSP | Interstitial fibrosis | Tubulointerstitial kidney disease | [111] |
| m.616T>C | MT-tRNAPhe | Tubular atrophy | ||
| m.09155A>G | MT-ATP6 | Central obesity Proteinuria Impaired glucose tolerance |
Maternally inherited deafness and diabetes FSGS |
[112] |
| m.5540G>A | MT-TW | Proteinuria Hypertension |
Cataract | [113] |
| Basal ganglia calcification | ||||
| Retinitis pigmentosa | ||||
| m.9267G>C m.5913G>A |
MT-CO3 MT-CO1 |
Hypertension | Mitochondrial diabetes DKD |
[114] |
| Nephropathy | ||||
| Hyperglycemia | ||||
| Insulin resistance | ||||
| Deafness | ||||
| m.7501T>A | MT-tRNASer | Proteinuria | Glomerulosclerosis Diabetes mellitus |
[115] |
| Hypertension | ||||
| Hyperglycemia |
Abbreviations: eGFR, estimated glomerular filtration rate; FSGS, focal segmental glomerulosclerosis; DKD, diabetic kidney disease; AKI, acute kidney injury; ChRCC, chromophobe renal cancer; CKD, chronic kidney disease.
An adenine to guanine substitution at nucleotide 3243 of the mtDNA (m.3243A>G), which affects the mitochondrial MT-TL1 gene, has been shown to cause mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS) syndrome. The most striking characteristics of renal biopsy were FSGS and arteriolar hyaline thickening [116]. Cai et al. recently reported that a patient with m.3243A>G mutation was diagnosed with membranous nephropathy, and AKI complicated by hyperuricemia might be attributed to mtDNA mutations [99].
A novel heteroplasmic nonsense mtDNA mutation m.6145G>A in the mitochondrial cytochrome c oxidase subunit I (MTCO1) was also identified in a patient who exhibited mitochondrial abnormalities, chronic tubulointerstitial changes and recurrent episodes of rhabdomyolysis [100]. Mitochondrial tubulointerstitial nephropathy (MITKD) is a tubulointerstitial nephropathy caused by mutations in mtDNA. m.616T>C is one of the mutations that lead to MITKD, the main symptoms of which are chronic renal insufficiency and epilepsia [101]. It has also been reported that two mt-ND5 pathogenic variants m.13513G>A and m.13514A>G caused mitochondrial dysfunction in tubulo-interstitial kidney disease [102]. Aristolochic acid elevated the levels of mutagenic 8-oxo-2′-deoxyguanosine and 7-(deoxyadenosine-N6-yl)-aristolactam adduct on mtDNA isolated from human HEK293 cells, which shed light on a potentially important causative role of mtDNA mutations and mitochondrial dysfunction in the etiology of aristolochic acid nephropathy [117]. Patients with type 2 diabetes diagnosed with the m.4216T>C mtDNA mutation are more likely to have poor glycemic control, which triggers the progression of DKD [103]. However, an mtDNA mutation m.8344A>G in mitochondrial tRNALys gene causing myoclonic epilepsy with ragged-red fibers (MERRF) syndrome has no influence on mtDNA copy number and kidney function [118].
Comprehensive mtDNA resequencing has been applied for detecting mutations in clinical samples and several kinds of tumor tissues have been examined for mtDNA mutations, including that of renal cell carcinoma (RCC) [119]. Mutation analysis of mtDNA showed mutations in the gene of mitochondrial complex Ⅰ subunit ND1, ND5, and ND6, which contributes to respiratory chain deficiency and the occurrence of renal oncocytoma [104]. ChRCC is a subtype of RCC, which is accompanied by a high rate of mtDNA mutations [120]. mtDNA sequencing analysis revealed that mtDNA mutations led to function deficiency of NADH dehydrogenase subunits, which further promoted the transformation of the metabolic pattern in ChRCC [105].
4.3. mtDNA Leakage
The kidney is an organ rich in mtDNA. When the kidney is stimulated by harmful factors, mtDNA can be released from mitochondrial matrix to cytoplasm. Cisplatin induced mtDNA leakage into the cytosol, likely through BAX pores in the mitochondrial outer membrane in kidney tubules with subsequent activation of the cGAS-STING pathway, thereby triggering inflammation and AKI progression [121]. The receptor-interacting protein kinase 3 translocates to mitochondria and interacts with mitofilin, resulting in increased mtDNA release and activation of cGAS-STING-p65 pathway in renal IRI [122]. In mice with tubule-specific knockout of TFAM, aberrant packaging of mtDNA resulted in cytosolic translocation, which further activated the cytosolic cGAS-STING pathway and recruited cytokine and immune cells to aggravate kidney fibrosis [123]. In DKD, the downregulation of superoxide dismutase 2 mediated mitochondrial dysfunction and mtDNA leakage, which could activate TLR9 in macrophages [124]. In summary, mtDNA leakage may be a concomitant phenomenon of kidney injury and mediates the development of multiple inflammatory responses.
4.4. mtDNA Methylation
Current studies on mtDNA methylation in kidney are still a blind spot. Only a single study on the correlation between renal tumor metastasis and mtDNA methylation was reported. It was demonstrated that, compared to primary RCC cells, the D-loop region of mtDNA was markedly hypermethylated in bone metastatic RCC cells, which provided a direct link between hypermethylation of mtDNA in RCC and tumor growth in bone metastases [125]. Due to technology limitations and cost challenges in detecting mtDNA methylation, the study progress in this area is relatively poor. With sustained attention and dedication against investigation to this field, the role of mtDNA methylation in kidney diseases will be continuously explored.
5. Pharmacological Intervention of mtDNA Damage in Kidney Diseases
As discussed in the previous section, the content and integrity of mtDNA in the kidney is impaired in the etiology of a variety of kidney diseases. mtDNA damage can directly affect mitochondrial function. Therefore, targeted interventions against mtDNA damage may have a therapeutic effect on kidney diseases.
A wide variety of agents are currently applied to intervene in renal mtDNA damage, as shown in Table 2. We summarized the pharmacological mechanisms of these agents, and found that most of them alleviate kidney injury and cellular apoptosis by improving mitochondrial functions, such as increased mtDNA content, peroxisome proliferator-activated receptor gamma coactivator-1α (PGC-1α) and peroxisome proliferator-activated receptor γ (PPAR γ) expression, which promote mitochondrial OXPHOS to facilitate energy supply and reduce oxidative stress, ROS production, and inflammation. There are also a few therapeutic agents, such as l-carnitine and sacubitril/valsartan, which attenuate the inflammatory response activated by mtDNA leakage via suppressing inflammation-related pathways, such as the TLR9 and cGAS-STING signaling pathways [124,126].
Table 2.
Therapeutic intervention for mtDNA in different kidney models.
| Therapeutic Interventions |
Models | Main Effects on mtDNA and Mitochondrial Function |
Reference |
|---|---|---|---|
| Fluorofenidone | UUO and IRI | Increased mtDNA copy number Increased TFAM and PGC-1α expression Maintained mitochondrial structure Reduced mitochondrial oxidative stress |
[90] |
| l-carnitine | DKD | Decreased circulating mtDNA content Reduced mtROS production Suppressed inflammation |
[124] |
| Sacubitril/valsartan | DKD | Albuminuria Inhibited cGAS-STING signaling Decreased oxidative response |
[126] |
| Coenzyme Q10 | IRI | Alleviated mtDNA damage Suppressed inflammatory and oxidative responses |
[127] |
| Treprostinil | IRI | Increased mtDNA copy number Increased PGC-1α expression |
[128] |
| Increased ATP level | |||
| Reduced mitochondrial oxidative injury | |||
| Roxadustat | IRI | Increased ATPβ and PPARγ expression, and mtDNA | [129] |
| Alleviated DNA damage | |||
| Celastrol | Cisplatin-induced AKI | Increased mtDNA copy number Increased MMP Restored OXPHOS activity |
[130] |
| H151 | Cisplatin-induced AKI | Restored mtDNA content Reversed mitochondrial gene expression Suppressed inflammation |
[131] |
| Adiponectin | DKD | Increased mtDNA content Increased TFAM and PGC-1α expression Increased mitochondrial mass Increased MMP |
[132] |
| Salidroside | DKD | Increased mtDNA copy number Enhanced ETC proteins Increased PGC-1α expression |
[133] |
| Artemether | Adriamycin nephropathy | Restored redox imbalance Increased mtDNA copy number |
[134] |
| Improved mitochondrial function |
Abbreviations: UUO, unilateral ureteral obstruction; IRI, ischemia-reperfusion injury; PGC-1α, peroxisome proliferator-activated receptor gamma coactivator-1α; ATPβ, ATPase β subunit; PPARγ, peroxisome proliferator-activated receptor γ; MMP, mitochondrial membrane potential; DKD, diabetic kidney disease; mtROS, mitochondrial reactive oxygen species; cGAS, cyclic GMP-AMP synthase; STING, stimulator of interferon genes.
A number of other alternative therapies have also been reported. For instance, extracellular vesicles (EVs) derived from mesenchymal stem cells (MSCs) contain functional mitochondrial components, such as mtDNA, mitochondrial proteins, and energy-related proteins from the tricarboxylic acid cycle [135]. MSC-EV-mediated TFAM mRNA transfer restored TFAM expression, mtDNA deletion, and OXPHOS defects in renal tubular cells of AKI [136]. The latest research shows that mitochondrial transplantation might be a novel treatment for mitochondrial diseases. Direct exogenous supplementation of mitochondria can replace damaged mtDNA, restore mitochondrial function, and inhibit oxidative stress, thus reducing apoptosis [137,138]. Moreover, mitochondrial replacement therapy can be used for maternally inherited diseases caused by mutations in mtDNA [139].
6. Conclusions and Future Perspectives
Intact mtDNA is closely related to mitochondrial function. mtDNA lacks a sophisticated self-repair system and is susceptible to a variety of external and internal factors, including drugs, infections, immune system disorders, hypertension, diabetes, and aging. All these etiologies can lead to mtDNA damage, which further augments mitochondrial dysfunction, manifested as defective ETC, reduced OXPHOS, and oxidative stress and inflammatory response, thus participating in the process of kidney injury. Therefore, it is imperative to explore the role of mtDNA damage in kidney disease.
In this review, we have scrutinized the common types of mtDNA damage, including impaired mtDNA replication, mtDNA mutations, mtDNA leakage, and mtDNA methylation. The mechanisms of these damage types were comprehensively described, and the studies related to mtDNA damage in kidney diseases were also summarized in detail. It has been revealed that mtDNA damage plays an important role in kidney diseases and the levels of mtDNA in peripheral plasma and urine have indicated a marker role in kidney diseases. Pharmacological treatment or exogenous mtDNA transplantation can improve damaged mtDNA, restore mitochondrial function, or directly inhibit mtDNA-induced inflammatory response, thus providing a theoretical basis and new avenues for the treatment of kidney diseases. In conclusion, mtDNA damage serve as a key biomarker of kidney diseases.
Author Contributions
J.F., Z.C. and G.D. put forward the concept. J.F. performed a literature review and wrote the manuscript. W.L. and Z.W. contributed to language editing and content adjustment. J.F. and G.D. participated in the revision of this manuscript. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This study was supported by the grants from National Natural Science Foundation of China (82070713 to G.D., 82100704 to Z.C. and 81970631 to W.L.).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Tang C., Cai J., Yin X.M., Weinberg J.M., Venkatachalam M.A., Dong Z. Mitochondrial quality control in kidney injury and repair. Nat. Rev. Nephrol. 2021;17:299–318. doi: 10.1038/s41581-020-00369-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bhatia D., Capili A., Choi M.E. Mitochondrial dysfunction in kidney injury, inflammation, and disease: Potential therapeutic approaches. Kidney Res. Clin. Pr. 2020;39:244–258. doi: 10.23876/j.krcp.20.082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gilea A.I., Ceccatelli B.C., Magistrati M., di Punzio G., Goffrini P., Baruffini E., Dallabona C. Saccharomyces cerevisiae as a Tool for Studying Mutations in Nuclear Genes Involved in Diseases Caused by Mitochondrial DNA Instability. Genes. 2021;12:1866. doi: 10.3390/genes12121866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hershberger K.A., Rooney J.P., Turner E.A., Donoghue L.J., Bodhicharla R., Maurer L.L., Ryde I.T., Kim J.J., Joglekar R., Hibshman J.D., et al. Early-life mitochondrial DNA damage results in lifelong deficits in energy production mediated by redox signaling in Caenorhabditis elegans. Redox Biol. 2021;43:102000. doi: 10.1016/j.redox.2021.102000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.van der Slikke E.C., Star B.S., van Meurs M., Henning R.H., Moser J., Bouma H.R. Sepsis is associated with mitochondrial DNA damage and a reduced mitochondrial mass in the kidney of patients with sepsis-AKI. Crit. Care. 2021;25:36. doi: 10.1186/s13054-020-03424-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jin L., Yu B., Armando I., Han F. Mitochondrial DNA-Mediated Inflammation in Acute Kidney Injury and Chronic Kidney Disease. Oxid Med. Cell Longev. 2021;2021:9985603. doi: 10.1155/2021/9985603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Melki I., Allaeys I., Tessandier N., Levesque T., Cloutier N., Laroche A., Vernoux N., Becker Y., Benk-Fortin H., Zufferey A., et al. Platelets release mitochondrial antigens in systemic lupus erythematosus. Sci. Transl. Med. 2021;13:eaav5928. doi: 10.1126/scitranslmed.aav5928. [DOI] [PubMed] [Google Scholar]
- 8.Kockler Z.W., Osia B., Lee R., Musmaker K., Malkova A. Repair of DNA Breaks by Break-Induced Replication. Annu. Rev. Biochem. 2021;90:165–191. doi: 10.1146/annurev-biochem-081420-095551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang X., Wu X., Hu Q., Wu J., Wang G., Hong Z., Ren J. Mitochondrial DNA in liver inflammation and oxidative stress. Life Sci. 2019;236:116464. doi: 10.1016/j.lfs.2019.05.020. [DOI] [PubMed] [Google Scholar]
- 10.Lin Y.H., Lim S.N., Chen C.Y., Chi H.C., Yeh C.T., Lin W.R. Functional Role of Mitochondrial DNA in Cancer Progression. Int. J. Mol. Sci. 2022;23:1659. doi: 10.3390/ijms23031659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lechuga-Vieco A.V., Latorre-Pellicer A., Calvo E., Torroja C., Pellico J., Acin-Perez R., Garcia-Gil M.L., Santos A., Bagwan N., Bonzon-Kulichenko E., et al. Heteroplasmy of Wild-Type Mitochondrial DNA Variants in Mice Causes Metabolic Heart Disease With Pulmonary Hypertension and Frailty. Circulation. 2022;145:1084–1101. doi: 10.1161/CIRCULATIONAHA.121.056286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhong W., Rao Z., Xu J., Sun Y., Hu H., Wang P., Xia Y., Pan X., Tang W., Chen Z., et al. Defective mitophagy in aged macrophages promotes mitochondrial DNA cytosolic leakage to activate STING signaling during liver sterile inflammation. Aging Cell. 2022;21:e13622. doi: 10.1111/acel.13622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nie Y., Murley A., Golder Z., Rowe J.B., Allinson K., Chinnery P.F. Heteroplasmic mitochondrial DNA mutations in frontotemporal lobar degeneration. Acta Neuropathol. 2022;143:687–695. doi: 10.1007/s00401-022-02423-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stoccoro A., Coppede F. Mitochondrial DNA Methylation and Human Diseases. Int. J. Mol. Sci. 2021;22:4594. doi: 10.3390/ijms22094594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nomiyama T., Setoyama D., Yasukawa T., Kang D. Mitochondria metabolomics reveals a role of beta-nicotinamide mononucleotide metabolism in mitochondrial DNA replication. J. Biochem. 2022;171:325–338. doi: 10.1093/jb/mvab136. [DOI] [PubMed] [Google Scholar]
- 16.Castellani C.A., Longchamps R.J., Sun J., Guallar E., Arking D.E. Thinking outside the nucleus: Mitochondrial DNA copy number in health and disease. Mitochondrion. 2020;53:214–223. doi: 10.1016/j.mito.2020.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Roy A., Kandettu A., Ray S., Chakrabarty S. Mitochondrial DNA replication and repair defects: Clinical phenotypes and therapeutic interventions. Biochim. Biophys. Acta Bioenerg. 2022;1863:148554. doi: 10.1016/j.bbabio.2022.148554. [DOI] [PubMed] [Google Scholar]
- 18.Manini A., Abati E., Comi G.P., Corti S., Ronchi D. Mitochondrial DNA homeostasis impairment and dopaminergic dysfunction: A trembling balance. Ageing Res. Rev. 2022;76:101578. doi: 10.1016/j.arr.2022.101578. [DOI] [PubMed] [Google Scholar]
- 19.Blazquez-Bermejo C., Carreno-Gago L., Molina-Granada D., Aguirre J., Ramon J., Torres-Torronteras J., Cabrera-Perez R., Martin M.A., Dominguez-Gonzalez C., de la Cruz X., et al. Increased dNTP pools rescue mtDNA depletion in human POLG-deficient fibroblasts. FASEB J. 2019;33:7168–7179. doi: 10.1096/fj.201801591R. [DOI] [PubMed] [Google Scholar]
- 20.Yang Q., Liu P., Anderson N.S., Shpilka T., Du Y., Naresh N.U., Li R., Zhu L.J., Luk K., Lavelle J., et al. LONP-1 and ATFS-1 sustain deleterious heteroplasmy by promoting mtDNA replication in dysfunctional mitochondria. Nat. Cell Biol. 2022;24:181–193. doi: 10.1038/s41556-021-00840-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Piro-Megy C., Sarzi E., Tarres-Sole A., Pequignot M., Hensen F., Quiles M., Manes G., Chakraborty A., Senechal A., Bocquet B., et al. Dominant mutations in mtDNA maintenance gene SSBP1 cause optic atrophy and foveopathy. J. Clin. Investig. 2020;130:143–156. doi: 10.1172/JCI128513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jiang M., Xie X., Zhu X., Jiang S., Milenkovic D., Misic J., Shi Y., Tandukar N., Li X., Atanassov I., et al. The mitochondrial single-stranded DNA binding protein is essential for initiation of mtDNA replication. Sci. Adv. 2021;7:eabf8631. doi: 10.1126/sciadv.abf8631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Otten A., Kamps R., Lindsey P., Gerards M., Pendeville-Samain H., Muller M., van Tienen F., Smeets H. Tfam Knockdown Results in Reduction of mtDNA Copy Number, OXPHOS Deficiency and Abnormalities in Zebrafish Embryos. Front. Cell Dev. Biol. 2020;8:381. doi: 10.3389/fcell.2020.00381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sercel A.J., Carlson N.M., Patananan A.N., Teitell M.A. Mitochondrial DNA Dynamics in Reprogramming to Pluripotency. Trends Cell Biol. 2021;31:311–323. doi: 10.1016/j.tcb.2020.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sato T., Goto-Inoue N., Kimishima M., Toyoharu J., Minei R., Ogura A., Nagoya H., Mori T. A novel ND1 mitochondrial DNA mutation is maternally inherited in growth hormone transgenesis in amago salmon (Oncorhynchus masou ishikawae) Sci. Rep. 2022;12:6720. doi: 10.1038/s41598-022-10521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.McMillan R.P., Stewart S., Budnick J.A., Caswell C.C., Hulver M.W., Mukherjee K., Srivastava S. Quantitative Variation in m.3243A > G Mutation Produce Discrete Changes in Energy Metabolism. Sci. Rep. 2019;9:5752. doi: 10.1038/s41598-019-42262-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Liu H., Liu X., Zhou J., Li T. Mitochondrial DNA Is a Vital Driving Force in Ischemia-Reperfusion Injury in Cardiovascular Diseases. Oxid. Med. Cell Longev. 2022;2022:6235747. doi: 10.1155/2022/6235747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Xu C., Tong L., Rao J., Ye Q., Chen Y., Zhang Y., Xu J., Mao X., Meng F., Shen H., et al. Heteroplasmic and homoplasmic m.616T>C in mitochondria tRNAPhe promote isolated chronic kidney disease and hyperuricemia. JCI Insight. 2022;7:e157418. doi: 10.1172/jci.insight.157418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ji X., Guo W., Gu X., Guo S., Zhou K., Su L., Yuan Q., Liu Y., Guo X., Huang Q., et al. Mutational profiling of mtDNA control region reveals tumor-specific evolutionary selection involved in mitochondrial dysfunction. Ebiomedicine. 2022;80:104058. doi: 10.1016/j.ebiom.2022.104058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nunn C.J., Goyal S. Contingency and selection in mitochondrial genome dynamics. Elife. 2022;11:e76557. doi: 10.7554/eLife.76557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hancock-Cerutti W., Wu Z., Xu P., Yadavalli N., Leonzino M., Tharkeshwar A.K., Ferguson S.M., Shadel G.S., De Camilli P. ER-lysosome lipid transfer protein VPS13C/PARK23 prevents aberrant mtDNA-dependent STING signaling. J. Cell Biol. 2022;221:e202106046. doi: 10.1083/jcb.202106046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McArthur K., Whitehead L.W., Heddleston J.M., Li L., Padman B.S., Oorschot V., Geoghegan N.D., Chappaz S., Davidson S., San C.H., et al. BAK/BAX macropores facilitate mitochondrial herniation and mtDNA efflux during apoptosis. Science. 2018;359:eaao6047. doi: 10.1126/science.aao6047. [DOI] [PubMed] [Google Scholar]
- 33.Kim J., Gupta R., Blanco L.P., Yang S., Shteinfer-Kuzmine A., Wang K., Zhu J., Yoon H.E., Wang X., Kerkhofs M., et al. VDAC oligomers form mitochondrial pores to release mtDNA fragments and promote lupus-like disease. Science. 2019;366:1531–1536. doi: 10.1126/science.aav4011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yu C.H., Davidson S., Harapas C.R., Hilton J.B., Mlodzianoski M.J., Laohamonthonkul P., Louis C., Low R., Moecking J., De Nardo D., et al. TDP-43 Triggers Mitochondrial DNA Release via mPTP to Activate cGAS/STING in ALS. Cell. 2020;183:636–649. doi: 10.1016/j.cell.2020.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li J.S., Hao Y.Z., Hou M.L., Zhang X., Zhang X.G., Cao Y.X., Li J.M., Ma J., Zhou Z.X. Development of a Recombinase-aided Amplification Combined With Lateral Flow Dipstick Assay for the Rapid Detection of the African Swine Fever Virus. Biomed. Environ. Sci. 2022;35:133–140. doi: 10.3967/bes2022.018. [DOI] [PubMed] [Google Scholar]
- 36.Harapas C.R., Idiiatullina E., Al-Azab M., Hrovat-Schaale K., Reygaerts T., Steiner A., Laohamonthonkul P., Davidson S., Yu C.H., Booty L., et al. Organellar homeostasis and innate immune sensing. Nat. Rev. Immunol. 2022;9:539–545. doi: 10.1038/s41577-022-00682-8. [DOI] [PubMed] [Google Scholar]
- 37.Luecke S., Holleufer A., Christensen M.H., Jonsson K.L., Boni G.A., Sorensen L.K., Johannsen M., Jakobsen M.R., Hartmann R., Paludan S.R. cGAS is activated by DNA in a length-dependent manner. Embo. Rep. 2017;18:1707–1715. doi: 10.15252/embr.201744017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kato K., Omura H., Ishitani R., Nureki O. Cyclic GMP-AMP as an Endogenous Second Messenger in Innate Immune Signaling by Cytosolic DNA. Annu. Rev. Biochem. 2017;86:541–566. doi: 10.1146/annurev-biochem-061516-044813. [DOI] [PubMed] [Google Scholar]
- 39.Luteijn R.D., Zaver S.A., Gowen B.G., Wyman S.K., Garelis N.E., Onia L., McWhirter S.M., Katibah G.E., Corn J.E., Woodward J.J., et al. SLC19A1 transports immunoreactive cyclic dinucleotides. Nature. 2019;573:434–438. doi: 10.1038/s41586-019-1553-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ding P., Tan Q., Wei Z., Chen Q., Wang C., Qi L., Wen L., Zhang C., Yao C. Toll-like receptor 9 deficiency induces osteoclastic bone loss via gut microbiota-associated systemic chronic inflammation. Bone Res. 2022;10:42. doi: 10.1038/s41413-022-00210-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Honke N., Lowin T., Opgenoorth B., Shaabani N., Lautwein A., Teijaro J.R., Schneider M., Pongratz G. Endogenously produced catecholamines improve the regulatory function.n of TLR9-activated B cells. PLoS Biol. 2022;20:e3001513. doi: 10.1371/journal.pbio.3001513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hepokoski M., Singh P. Mitochondria as mediators of systemic inflammation and organ cross talk in acute kidney injury. Am. J. Physiol. Ren. Physiol. 2022;6:F589–F596. doi: 10.1152/ajprenal.00372.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tsuji N., Tsuji T., Ohashi N., Kato A., Fujigaki Y., Yasuda H. Role of Mitochondrial DNA in Septic AKI via Toll-Like Receptor. J. Am. Soc. Nephrol. 2016;27:2009–2020. doi: 10.1681/ASN.2015040376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pradhan P., Toy R., Jhita N., Atalis A., Pandey B., Beach A., Blanchard E.L., Moore S.G., Gaul D.A., Santangelo P.J., et al. TRAF6-IRF5 kinetics, TRIF, and biophysical factors drive synergistic innate responses to particle-mediated MPLA-CpG co-presentation. Sci. Adv. 2021;7:eabd4235. doi: 10.1126/sciadv.abd4235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.De Gaetano A., Solodka K., Zanini G., Selleri V., Mattioli A.V., Nasi M., Pinti M. Molecular Mechanisms of mtDNA-Mediated Inflammation. Cells. 2021;10:2898. doi: 10.3390/cells10112898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Huang Y., Zhou J.H., Zhang H., Canfran-Duque A., Singh A.K., Perry R.J., Shulman G.I., Fernandez-Hernando C., Min W. Brown adipose TRX2 deficiency activates mtDNA-NLRP3 to impair thermogenesis and protect against diet-induced insulin resistance. J. Clin. Invest. 2022;132:e148852. doi: 10.1172/JCI148852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang L., Liu T., Yang S., Sun L., Zhao Z., Li L., She Y., Zheng Y., Ye X., Bao Q., et al. Perfluoroalkyl substance pollutants activate the innate immune system through the AIM2 inflammasome. Nat. Commun. 2021;12:2915. doi: 10.1038/s41467-021-23201-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Shen J., Wang C., Li D., Xu T., Myers J., Ashton J.M., Wang T., Zuscik M.J., McAlinden A., O’Keefe R.J. DNA methyltransferase 3b regulates articular cartilage homeostasis by altering metabolism. JCI Insight. 2017;2:e93612. doi: 10.1172/jci.insight.93612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Damal V.S., Ebert S.M., Lim H.W., Kim J., You D., Jung B.C., Palacios H.H., Tcheau T., Adams C.M., Kang S. A necessary role of DNMT3A in endurance exercise by suppressing.g ALDH1L1-mediated oxidative stress. Embo. J. 2021;40:e106491. doi: 10.15252/embj.2020106491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Liu Q., Li H., Guo L., Chen Q., Gao X., Li P.H., Tang N., Guo X., Deng F., Wu S. Effects of short-term personal exposure to air pollution on platelet mitochondrial DNA methylation levels and the potential mitigation by L-arginine supplementation. J. Hazard. Mater. 2021;417:125963. doi: 10.1016/j.jhazmat.2021.125963. [DOI] [PubMed] [Google Scholar]
- 51.Wang Y., Gao J., Wu F., Lai C., Li Y., Zhang G., Peng X., Yu S., Yang J., Wang W., et al. Biological and epigenetic alterations of mitochondria involved in cellular replicative and hydrogen peroxide-induced premature senescence of human embryonic lung fibroblasts. Ecotoxicol. Env. Saf. 2021;216:112204. doi: 10.1016/j.ecoenv.2021.112204. [DOI] [PubMed] [Google Scholar]
- 52.Cao K., Lv W., Wang X., Dong S., Liu X., Yang T., Xu J., Zeng M., Zou X., Zhao D., et al. Hypermethylation of Hepatic Mitochondrial ND6 Provokes Systemic Insulin Resistance. Adv. Sci (Weinh) 2021;8:2004507. doi: 10.1002/advs.202004507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Liu Y., Song F., Yang Y., Yang S., Jiang M., Zhang W., Ma Z., Gu X. Mitochondrial DNA methylation drift and postoperative delirium in mice. Eur. J. Anaesthesiol. 2022;39:133–144. doi: 10.1097/EJA.0000000000001620. [DOI] [PubMed] [Google Scholar]
- 54.Zhang J., Shang J., Wang F., Huo X., Sun R., Ren Z., Wang W., Yang M., Li G., Gao D., et al. Decreased mitochondrial D-loop region methylation mediates an increase in mitochondrial DNA copy number in CADASIL. Clin. Epigenetics. 2022;14:2. doi: 10.1186/s13148-021-01225-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cushen S.C., Ricci C.A., Bradshaw J.L., Silzer T., Blessing A., Sun J., Zhou Z., Scroggins S.M., Santillan M.K., Santillan D.A., et al. Reduced Maternal Circulating Cell-Free Mitochondrial DNA Is Associated With the Development of Preeclampsia. J. Am. Heart Assoc. 2022;11:e21726. doi: 10.1161/JAHA.121.021726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wei R., Ni Y., Bazeley P., Grandhi S., Wang J., Li S.T., Hazen S.L., Wilson T.W., LaFramboise T. Mitochondrial DNA Content Is Linked to Cardiovascular Disease Patient Phenotypes. J. Am. Heart Assoc. 2021;10:e18776. doi: 10.1161/JAHA.120.018776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhong W., Rao Z., Rao J., Han G., Wang P., Jiang T., Pan X., Zhou S., Zhou H., Wang X. Aging aggravated liver ischemia and reperfusion injury by promoting STING-mediated NLRP3 activation in macrophages. Aging Cell. 2020;19:e13186. doi: 10.1111/acel.13186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gonzalez-Freire M., Moore A.Z., Peterson C.A., Kosmac K., McDermott M.M., Sufit R.L., Guralnik J.M., Polonsky T., Tian L., Kibbe M.R., et al. Associations of Peripheral Artery Disease With Calf Skeletal Muscle Mitochondrial DNA Heteroplasmy. J. Am. Heart Assoc. 2020;9:e15197. doi: 10.1161/JAHA.119.015197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Homolova J., Janovicova L., Konecna B., Vlkova B., Celec P., Tothova L., Babickova J. Plasma Concentrations of Extracellular DNA in Acute Kidney Injury. Diagnostics. 2020;10:152. doi: 10.3390/diagnostics10030152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wu J., Ren J., Liu Q., Hu Q., Wu X., Wang G., Hong Z., Ren H., Li J. Effects of Changes in the Levels of Damage-Associated Molecular Patterns Following Continuous Veno-Venous Hemofiltration Therapy on Outcomes in Acute Kidney Injury Patients With Sepsis. Front. Immunol. 2018;9:3052. doi: 10.3389/fimmu.2018.03052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Jancuska A., Potocarova A., Kovalcikova A.G., Podracka L., Babickova J., Celec P., Tothova L. Dynamics of Plasma and Urinary Extracellular DNA in Acute Kidney Injury. Int. J. Mol. Sci. 2022;23:3402. doi: 10.3390/ijms23063402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.He W.J., Li C., Huang Z., Geng S., Rao V.S., Kelly T.N., Hamm L.L., Grams M.E., Arking D.E., Appel L.J., et al. Association of Mitochondrial DNA Copy Number with Risk of Progression of Kidney Disease. Clin. J. Am. Soc. Nephrol. 2022;17:966–975. doi: 10.2215/CJN.15551121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cao H., Wu J., Luo J., Chen X., Yang J., Fang L. Urinary mitochondrial DNA: A potential early biomarker of diabetic nephropathy. Diabetes Metab. Res. Rev. 2019;35:e3131. doi: 10.1002/dmrr.3131. [DOI] [PubMed] [Google Scholar]
- 64.Szeto C.C., Lai K.B., Chow K.M., Kwan B.C., Cheng P.M., Kwong V.W., Choy A.S., Leung C.B., Li P.K. Plasma Mitochondrial DNA Level is a Prognostic Marker in Peritoneal Dialysis Patients. Kidney Blood Press Res. 2016;41:402–412. doi: 10.1159/000443442. [DOI] [PubMed] [Google Scholar]
- 65.Fan Z., Guo Y., Zhong X.Y. Circulating Cell-Free Mi.itochondrial DNA: A Potential Blood-Based Biomarker for Sarcopenia in Patients Undergoing Maintenance Hemodialysis. Med. Sci. Monit. 2022;28:e934679. doi: 10.12659/MSM.934679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tian S.L., Bai X., Xu P.C., Chen T., Gao S., Hu S.Y., Wei L., Jia J.Y., Yan T.K. Circulating nicotinamide adenine dinucleotide-ubiquinone oxidoreductase chain 6 is associated with disease activity of anti-neutrophil cytoplasmic antibody-associated vasculitis. Clin. Chim. Acta. 2020;511:125–131. doi: 10.1016/j.cca.2020.10.009. [DOI] [PubMed] [Google Scholar]
- 67.Han F., Sun Q., Huang Z., Li H., Ma M., Liao T., Luo Z., Zheng L., Zhang N., Chen N., et al. Donor plasma mitochondrial DNA is associated with antibody-mediated rejection in renal allograft recipients. Aging (Albany NY) 2021;13:8440–8453. doi: 10.18632/aging.202654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Han F., Wan S., Sun Q., Chen N., Li H., Zheng L., Zhang N., Huang Z., Hong L., Sun Q. Donor Plasma Mitochondrial DNA Is Correlated with Posttransplant Renal Allograft Function. Transplantation. 2019;103:2347–2358. doi: 10.1097/TP.0000000000002598. [DOI] [PubMed] [Google Scholar]
- 69.Eirin A., Herrmann S.M., Saad A., Abumoawad A., Tang H., Lerman A., Textor S.C., Lerman L.O. Urinary mitochondrial DNA copy number identifies renal mitochondrial injury in renovascular hypertensive patients undergoing renal revascularization: A Pilot Study. Acta Physiol. (Oxf) 2019;226:e13267. doi: 10.1111/apha.13267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hu Q., Ren J., Ren H., Wu J., Wu X., Liu S., Wang G., Gu G., Guo K., Li J. Urinary Mitochondrial DNA Identifies Renal Dysfunction and Mitochondrial Damage in Sepsis-Induced Acute Kidney Injury. Oxid Med. Cell Longev. 2018;2018:8074936. doi: 10.1155/2018/8074936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Yu B.C., Moon A., Lee K.H., Oh Y.S., Park M.Y., Choi S.J., Kim J.K. Minimal Change Disease Is Associated with Mitochondrial Injury and STING Pathway Activation. J. Clin. Med. 2022;11:577. doi: 10.3390/jcm11030577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wei P.Z., Kwan B.C., Chow K.M., Cheng P.M., Luk C.C., Li P.K., Szeto C.C. Urinary mitochondrial DNA level is an indicator of intra-renal mitochondrial depletion and renal scarring in diabetic nephropathy. Nephrol. Dial. Transpl. 2018;33:784–788. doi: 10.1093/ndt/gfx339. [DOI] [PubMed] [Google Scholar]
- 73.Wei P.Z., Kwan B.C., Chow K.M., Cheng P.M., Luk C.C., Lai K.B., Li P.K., Szeto C.C. Urinary mitochondrial DNA level in non-diabetic chronic kidney diseases. Clin. Chim. Acta. 2018;484:36–39. doi: 10.1016/j.cca.2018.05.036. [DOI] [PubMed] [Google Scholar]
- 74.Chang C.C., Chiu P.F., Wu C.L., Kuo C.L., Huang C.S., Liu C.S., Huang C.H. Urinary cell-free mitochondrial and nuclear deoxyribonucleic acid correlates with the prognosis of chronic kidney diseases. BMC Nephrol. 2019;20:391. doi: 10.1186/s12882-019-1549-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Eirin A., Saad A., Tang H., Herrmann S.M., Woollard J.R., Lerman A., Textor S.C., Lerman L.O. Urinary Mitochondrial DNA Copy Number Identifies Chronic Renal Injury in Hypertensive Patients. Hypertension. 2016;68:401–410. doi: 10.1161/HYPERTENSIONAHA.116.07849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Eirin A., Saad A., Woollard J.R., Juncos L.A., Calhoun D.A., Tang H., Lerman A., Textor S.C., Lerman L.O. Glomerular Hyperfiltration in Obese African American Hypertensive Patients Is Associated With Elevated Urinary Mitochondrial-DNA Copy Number. Am. J. Hypertens. 2017;30:1112–1119. doi: 10.1093/ajh/hpx103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yu B.C., Cho N.J., Park S., Kim H., Gil H.W., Lee E.Y., Kwon S.H., Jeon J.S., Noh H., Han D.C., et al. Minor Glomerular Abnormalities are Associated with Deterioration of Long-Term Kidney Function and Mitochondrial Injury. J. Clin. Med. 2019;9:33. doi: 10.3390/jcm9010033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yu B.C., Cho N.J., Park S., Kim H., Choi S.J., Kim J.K., Hwang S.D., Gil H.W., Lee E.Y., Jeon J.S., et al. IgA nephropathy is associated with elevated urinary mitochondrial DNA copy numbers. Sci. Rep. 2019;9:16068. doi: 10.1038/s41598-019-52535-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wu S.J., Yang X., Xu P.C., Chen T., Gao S., Hu S.Y., Wei L., Yan T.K. Urinary mitochondrial DNA is a useful biomarker for assessing kidney injury of antineutrophil cytoplasmic antibody -associated vasculitis. Clin. Chim. Acta. 2020;502:263–268. doi: 10.1016/j.cca.2019.11.014. [DOI] [PubMed] [Google Scholar]
- 80.Jansen M., Pulskens W., Uil M., Claessen N., Nieuwenhuizen G., Standaar D., Hau C.M., Nieuwland R., Florquin S., Bemelman F.J., et al. Urinary mitochondrial DNA associates with delayed graft function following renal transplantation. Nephrol. Dial. Transpl. 2020;35:1320–1327. doi: 10.1093/ndt/gfy372. [DOI] [PubMed] [Google Scholar]
- 81.Kim K., Moon H., Lee Y.H., Seo J.W., Kim Y.G., Moon J.Y., Kim J.S., Jeong K.H., Lee T.W., Ihm C.G., et al. Clinical relevance of cell-free mitochondrial DNA during the early postoperative period in kidney transplant recipients. Sci. Rep. 2019;9:18607. doi: 10.1038/s41598-019-54694-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Herbers E., Kekalainen N.J., Hangas A., Pohjoismaki J.L., Goffart S. Tissue specific differences in mitochondrial DNA maintenance and expression. Mitochondrion. 2019;44:85–92. doi: 10.1016/j.mito.2018.01.004. [DOI] [PubMed] [Google Scholar]
- 83.Longchamps R.J., Yang S.Y., Castellani C.A., Shi W., Lane J., Grove M.L., Bartz T.M., Sarnowski C., Liu C., Burrows K., et al. Genome-wide analysis of mitochondrial DNA copy number reveals loci implicated in nucleotide metabolism, platelet activation, and megakaryocyte proliferation. Hum. Genet. 2022;141:127–146. doi: 10.1007/s00439-021-02394-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Gustafson M.A., McCormick E.M., Perera L., Longley M.J., Bai R., Kong J., Dulik M., Shen L., Goldstein A.C., McCormack S.E., et al. Mitochondrial single-stranded DNA binding protein novel de novo SSBP1 mutation in a child with single large-scale mtDNA deletion (SLSMD) clinically manifesting as Pearson, Kearns-Sayre, and Leigh syndromes. PLoS ONE. 2019;14:e221829. doi: 10.1371/journal.pone.0221829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Del D.V., Ullah F., Di Meo I., Magini P., Gusic M., Maresca A., Caporali L., Palombo F., Tagliavini F., Baugh E.H., et al. SSBP1 mutations cause mtDNA depletion underlying a complex optic atrophy disorder. J. Clin. Investig. 2020;130:108–125. doi: 10.1172/JCI128514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Gureev A.P., Andrianova N.V., Pevzner I.B., Zorova L.D., Chernyshova E.V., Sadovnikova I.S., Chistyakov D.V., Popkov V.A., Semenovich D.S., Babenko V.A., et al. Dietary restriction modulates mitochondrial DNA damage and oxylipin profile in aged rats. FEBS J. 2022;289:5697–5713. doi: 10.1111/febs.16451. [DOI] [PubMed] [Google Scholar]
- 87.Chen J., Zheng Q., Peiffer L.B., Hicks J.L., Haffner M.C., Rosenberg A.Z., Levi M., Wang X.X., Ozbek B., Baena-Del V.J., et al. An in Situ Atlas of Mitochondrial DNA in Mammalian Tissues Reveals High Content in Stem and Proliferative Compartments. Am. J. Pathol. 2020;190:1565–1579. doi: 10.1016/j.ajpath.2020.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Fukunaga H. Mitochondrial DNA Copy Number and Developmental Origins of Health and Disease (DOHaD) Int. J. Mol. Sci. 2021;22:6634. doi: 10.3390/ijms22126634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Liu Q., Krishnasamy Y., Rehman H., Lemasters J.J., Schnellmann R.G., Zhong Z. Disrupted Renal Mitochondrial Homeostasis after Liver Transplantation in Rats. PLoS ONE. 2015;10:e140906. doi: 10.1371/journal.pone.0140906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Liao X., Lv X., Zhang Y., Han Y., Li J., Zeng J., Tang D., Meng J., Yuan X., Peng Z., et al. Fluorofenidone Inhibits UUO/IRI-Induced Renal Fibrosis by Reducing Mitochondrial Da.amage. Oxid Med. Cell Longev. 2022;2022:2453617. doi: 10.1155/2022/2453617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Fu Z.J., Wang Z.Y., Xu L., Chen X.H., Li X.X., Liao W.T., Ma H.K., Jiang M.D., Xu T.T., Xu J., et al. HIF-1alpha-BNIP3-mediated mitophagy in tubular cells protects against renal ischemia/reperfusion injury. Redox Biol. 2020;36:101671. doi: 10.1016/j.redox.2020.101671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rao M., Li L., Demello C., Guo D., Jaber B.L., Pereira B.J., Balakrishnan V.S. Mitochondrial DNA injury and mortality in hemodialysis patients. J. Am. Soc. Nephrol. 2009;20:189–196. doi: 10.1681/ASN.2007091031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Akhtar S., Siragy H.M. Pro-renin receptor suppresses mitochondrial biogenesis and function via AMPK/SIRT-1/ PGC-1alpha pathway in diabetic kidney. PLoS ONE. 2019;14:e225728. doi: 10.1371/journal.pone.0225728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Feng J., Chen Z., Ma Y., Yang X., Zhu Z., Zhang Z., Hu J., Liang W., Ding G. AKAP1 contributes to impaired mtDNA replication and mitochondrial dysfunction in podocytes of diabetic kidney disease. Int. J. Biol Sci. 2022;10:4026–4042. doi: 10.7150/ijbs.73493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kaneko S., Usui J., Hagiwara M., Shimizu T., Ishii R., Takahashi-Kobayashi M., Kageyama M., Nakada K., Hayashi J.I., Yamagata K. Mitochondrial DNA deletion-dependent podocyte injuries in Mito-miceDelta, a murine model of mitochondrial disease. Exp. Anim. 2022;71:14–21. doi: 10.1538/expanim.21-0054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Xiao Y., Clima R., Busch J., Rabien A., Kilic E., Villegas S.L., Timmermann B., Attimonelli M., Jung K., Meierhofer D. Decreased Mitochondrial DNA Content Drives OXPHOS Dysregulation in Chromophobe Renal Cell Carcinoma. Cancer Res. 2020;80:3830–3840. doi: 10.1158/0008-5472.CAN-20-0754. [DOI] [PubMed] [Google Scholar]
- 97.Milenkovic D., Sanz-Moreno A., Calzada-Wack J., Rathkolb B., Veronica A.O., Gerlini R., Aguilar-Pimentel A., Misic J., Simard M.L., Wolf E., et al. Mice lacking the mitochondrial exonuclease MGME1 develop inflammatory kidney disease with glomerular dysfunction. PLoS Genet. 2022;18:e1010190. doi: 10.1371/journal.pgen.1010190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Samuels D.C., Li C., Li B., Song Z., Torstenson E., Boyd C.H., Rokas A., Thornton-Wells T.A., Moore J.H., Hughes T.M., et al. Recurrent tissue-specific mtDNA mutations are common in humans. PLoS Genet. 2013;9:e1003929. doi: 10.1371/journal.pgen.1003929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Cai M., Yu Q., Bao J. A case report of mitochondrial myopathy with membranous nephropathy. BMC Nephrol. 2022;23:87. doi: 10.1186/s12882-022-02710-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Fervenza F.C., Gavrilova R.H., Nasr S.H., Irazabal M.V., Nath K.A. CKD Due to a Novel Mitochondrial DNA Mutation: A Case Report. Am. J. Kidney Dis. 2019;73:273–277. doi: 10.1053/j.ajkd.2018.06.032. [DOI] [PubMed] [Google Scholar]
- 101.Lorenz R., Ahting U., Betzler C., Heimering S., Borggrafe I., Lange-Sperandio B. Homoplasmy of the Mitochondrial DNA Mutation m.616T>C Leads to Mitochondrial Tubulointerstitial Kidney Disease and Encephalopathia. Nephron. 2020;144:156–160. doi: 10.1159/000504412. [DOI] [PubMed] [Google Scholar]
- 102.Bakis H., Trimouille A., Vermorel A., Redonnet I., Goizet C., Boulestreau R., Lacombe D., Combe C., Martin-Negrier M.L., Rigothier C. Adult onset tubulo-interstitial nephropathy in MT-ND5-related phenotypes. Clin. Genet. 2020;97:628–633. doi: 10.1111/cge.13670. [DOI] [PubMed] [Google Scholar]
- 103.Diaz-Morales N., Lopez-Domenech S., Iannantuoni F., Lopez-Gallardo E., Sola E., Morillas C., Rocha M., Ruiz-Pesini E., Victor V.M. Mitochondrial DNA Haplogroup JT is Related to Impaired Glycaemic Control and Renal Function in Type 2 Diabetic Patients. J. Clin. Med. 2018;7:220. doi: 10.3390/jcm7080220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Mayr J.A., Meierhofer D., Zimmermann F., Feichtinger R., Kogler C., Ratschek M., Schmeller N., Sperl W., Kofler B. Loss of complex I due to mitochondrial DNA mutations in renal oncocytoma. Clin. Cancer Res. 2008;14:2270–2275. doi: 10.1158/1078-0432.CCR-07-4131. [DOI] [PubMed] [Google Scholar]
- 105.Davis C.F., Ricketts C.J., Wang M., Yang L., Cherniack A.D., Shen H., Buhay C., Kang H., Kim S.C., Fahey C.C., et al. The somatic genomic landscape of chromophobe renal cell carcinoma. Cancer Cell. 2014;26:319–330. doi: 10.1016/j.ccr.2014.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Imasawa T., Hirano D., Nozu K., Kitamura H., Hattori M., Sugiyama H., Sato H., Murayama K. Clinicopathologic Features of Mitochondrial Nephropathy. Kidney Int. Rep. 2022;7:580–590. doi: 10.1016/j.ekir.2021.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Bargagli M., Primiano G., Primiano A., Gervasoni J., Naticchia A., Servidei S., Gambaro G., Ferraro P.M. Recurrent kidney stones in a family with a mitochondrial disorder due to the m.3243A>G mutation. Urolithiasis. 2019;47:489–492. doi: 10.1007/s00240-018-1087-1. [DOI] [PubMed] [Google Scholar]
- 108.De Luise M., Guarnieri V., Ceccarelli C., D’Agruma L., Porcelli A.M., Gasparre G. A Nonsense Mitochondrial DNA Mutation Associates with Dysfunction of HIF1alpha in a Von Hippel-Lindau Renal Oncocytoma. Oxid Med. Cell Longev. 2019;2019:8069583. doi: 10.1155/2019/8069583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Lemoine S., Panaye M., Rabeyrin M., Errazuriz-Cerda E., Mousson D.C.B., Petiot P., Juillard L., Guebre-Egziabher F. Renal Involvement in Neuropathy, Ataxia, Retinitis Pigmentosa (NARP) Syndrome: A Case Report. Am. J. Kidney Dis. 2018;71:754–757. doi: 10.1053/j.ajkd.2017.09.020. [DOI] [PubMed] [Google Scholar]
- 110.Narumi K., Mishima E., Akiyama Y., Matsuhashi T., Nakamichi T., Kisu K., Nishiyama S., Ikenouchi H., Kikuchi A., Izumi R., et al. Focal Segmental Glomerulosclerosis Associated with Chronic Progressive External Ophthalmoplegia and Mitochondrial DNA A3243G Mutation. Nephron. 2018;138:243–248. doi: 10.1159/000485109. [DOI] [PubMed] [Google Scholar]
- 111.Connor T.M., Hoer S., Mallett A., Gale D.P., Gomez-Duran A., Posse V., Antrobus R., Moreno P., Sciacovelli M., Frezza C., et al. Mutations in mitochondrial DNA causing tubulointerstitial kidney disease. PLoS Genet. 2017;13:e1006620. doi: 10.1371/journal.pgen.1006620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Adema A.Y., Janssen M.C., van der Heijden J.W. A novel mutation in mitochondrial DNA in a patient with diabetes, deafness and proteinuria. Neth. J. Med. 2016;74:455–457. [PubMed] [Google Scholar]
- 113.Ng Y.S., Hardy S.A., Shrier V., Quaghebeur G., Mole D.R., Daniels M.J., Downes S.M., Freebody J., Fratter C., Hofer M., et al. Clinical features of the pathogenic m.5540G>A mitochondrial transfer RNA tryptophan gene mutation. Neuromuscul. Disord. 2016;26:702–705. doi: 10.1016/j.nmd.2016.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Tabebi M., Mkaouar-Rebai E., Mnif M., Kallabi F., Ben M.A., Ben S.W., Charfi N., Keskes-Ammar L., Kamoun H., Abid M., et al. A novel mutation MT-COIII m.9267G>C and MT-COI m.5913G>A mutation in mitochondrial genes in a Tunisian family with maternally inherited diabetes and deafness (MIDD) associated with severe nephropathy. Biochem. Biophys. Res. Commun. 2015;459:353–360. doi: 10.1016/j.bbrc.2015.01.151. [DOI] [PubMed] [Google Scholar]
- 115.Imasawa T., Tanaka M., Yamaguchi Y., Nakazato T., Kitamura H., Nishimura M. 7501 T > A mitochondrial DNA variant in a patient with glomerulosclerosis. Ren Fail. 2014;36:1461–1465. doi: 10.3109/0886022X.2014.945181. [DOI] [PubMed] [Google Scholar]
- 116.Seidowsky A., Hoffmann M., Glowacki F., Dhaenens C.M., Devaux J.P., de Sainte F.C., Provot F., Gheerbrant J.D., Hummel A., Hazzan M., et al. Renal involvement in MELAS syndrome—A series of 5 cases and review of the literature. Clin. Nephrol. 2013;80:456–463. doi: 10.5414/CN107063. [DOI] [PubMed] [Google Scholar]
- 117.Chan W., Ham Y.H. Probing the Hidden Role of Mitochondrial DNA Damage and Dysfunction in the Etiology of Aristolochic Acid Nephropathy. Chem. Res. Toxicol. 2021;34:1903–1909. doi: 10.1021/acs.chemrestox.1c00175. [DOI] [PubMed] [Google Scholar]
- 118.Brinckmann A., Weiss C., Wilbert F., von Moers A., Zwirner A., Stoltenburg-Didinger G., Wilichowski E., Schuelke M. Regionalized pathology correlates with augmentation of mtDNA copy numbers in a patient with myoclonic epilepsy with ragged-red fibers (MERRF-syndrome) PLoS ONE. 2010;5:e13513. doi: 10.1371/journal.pone.0013513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Jakupciak J.P., Maragh S., Markowitz M.E., Greenberg A.K., Hoque M.O., Maitra A., Barker P.E., Wagner P.D., Rom W.N., Srivastava S., et al. Performance of mitochondrial DNA mutations detecting early stage cancer. BMC Cancer. 2008;8:285. doi: 10.1186/1471-2407-8-285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Yuan Y., Ju Y.S., Kim Y., Li J., Wang Y., Yoon C.J., Yang Y., Martincorena I., Creighton C.J., Weinstein J.N., et al. Comprehensive molecular characterization of mitochondrial genomes in human cancers. Nat. Genet. 2020;52:342–352. doi: 10.1038/s41588-019-0557-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Maekawa H., Inoue T., Ouchi H., Jao T.M., Inoue R., Nishi H., Fujii R., Ishidate F., Tanaka T., Tanaka Y., et al. Mitochondrial Damage Causes Inflammation via cGAS-STING Signaling in Acute Kidney Injury. Cell Rep. 2019;29:1261–1273. doi: 10.1016/j.celrep.2019.09.050. [DOI] [PubMed] [Google Scholar]
- 122.Feng Y., Imam A.A., Tombo N., Draeger D., Bopassa J.C. RIP3 Translocation into Mitochondria Promotes Mitofilin Degradation to Increase Inflammation and Kidney Injury after Renal Ischemia-Reperfusion. Cells-Basel. 2022;11:1894. doi: 10.3390/cells11121894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Chung K.W., Dhillon P., Huang S., Sheng X., Shrestha R., Qiu C., Kaufman B.A., Park J., Pei L., Baur J., et al. Mitochondrial Damage and Activation of the STING Pathway Lead to Renal Inflammation and Fibrosis. Cell Metab. 2019;30:784–799. doi: 10.1016/j.cmet.2019.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Ito S., Nakashima M., Ishikiriyama T., Nakashima H., Yamagata A., Imakiire T., Kinoshita M., Seki S., Kumagai H., Oshima N. Effects of L-Carnitine Treatment on Kidney Mitochondria and Macrophages in Mice with Diabetic Nephropathy. Kidney Blood Press Res. 2022;47:277–290. doi: 10.1159/000522013. [DOI] [PubMed] [Google Scholar]
- 125.Liu Z., Tian J., Peng F., Wang J. Hypermethylation of mitochondrial DNA facilitates bone metastasis of renal cell carcinoma. J. Cancer. 2022;13:304–312. doi: 10.7150/jca.62278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Myakala K., Jones B.A., Wang X.X., Levi M. Sacubitril/valsartan treatment has differential effects in modulating diabetic kidney disease in db/db mice and KKAy mice compared with valsartan treatment. Am. J. Physiol Ren. Physiol. 2021;320:F1133–F1151. doi: 10.1152/ajprenal.00614.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Liu Z., Li Y., Li C., Yu L., Chang Y., Qu M. Delivery of coenzyme Q10 with mitochondria-targeted nanocarrier attenuates renal ischemia-reperfusion injury in mice. Mater. Sci. Eng. C Mater. Biol. Appl. 2021;131:112536. doi: 10.1016/j.msec.2021.112536. [DOI] [PubMed] [Google Scholar]
- 128.Ding M., Tolbert E., Birkenbach M., Gohh R., Akhlaghi F., Ghonem N.S. Treprostinil reduces mitochondrial injury during rat renal ischemia-reperfusion injury. Biomed. Pharmacother. 2021;141:111912. doi: 10.1016/j.biopha.2021.111912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zhang M., Dong R., Yuan J., Da J., Zha Y., Long Y. Roxadustat (FG-4592) protects against ischaemia/reperfusion-induced acute kidney injury through inhibiting the mitochondrial damage pathway in mice. Clin. Exp. Pharm. Physiol. 2022;49:311–318. doi: 10.1111/1440-1681.13601. [DOI] [PubMed] [Google Scholar]
- 130.Yu X., Meng X., Xu M., Zhang X., Zhang Y., Ding G., Huang S., Zhang A., Jia Z. Celastrol ameliorates cisplatin nephrotoxicity by inhibiting NF-kappaB and improving mitochondrial function. Ebiomedicine. 2018;36:266–280. doi: 10.1016/j.ebiom.2018.09.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Gong W., Lu L., Zhou Y., Liu J., Ma H., Fu L., Huang S., Zhang Y., Zhang A., Jia Z. The novel STING antagonist H151 ameliorates cisplatin-induced acute kidney injury and mitochondrial dysfunction. Am. J. Physiol. Ren. Physiol. 2021;320:F608–F616. doi: 10.1152/ajprenal.00554.2020. [DOI] [PubMed] [Google Scholar]
- 132.Chen Y., Yang Y., Liu Z., He L. Adiponectin promotes repair of renal tubular epithelial cells by regulating mitochondrial biogenesis and function. Metabolism. 2022;128:154959. doi: 10.1016/j.metabol.2021.154959. [DOI] [PubMed] [Google Scholar]
- 133.Xue H., Li P., Luo Y., Wu C., Liu Y., Qin X., Huang X., Sun C. Salidroside stimulates the Sirt1/PGC-1alpha axis and ameliorates diabetic nephropathy in mice. Phytomedicine. 2019;54:240–247. doi: 10.1016/j.phymed.2018.10.031. [DOI] [PubMed] [Google Scholar]
- 134.Han P., Cai Y., Wang Y., Weng W., Chen Y., Wang M., Zhan H., Yu X., Wang T., Shao M., et al. Artemether ameliorates kidney injury by restoring redox imbalance and improving mitochondrial function in Adriamycin nephropathy in mice. Sci. Rep. 2021;11:1266. doi: 10.1038/s41598-020-80298-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Zorova L.D., Kovalchuk S.I., Popkov V.A., Chernikov V.P., Zharikova A.A., Khutornenko A.A., Zorov S.D., Plokhikh K.S., Zinovkin R.A., Evtushenko E.A., et al. Do Extracellular Vesicles Derived from Mesenchymal Stem Cells Contain Functional Mitochondria? Int. J. Mol. Sci. 2022;23:7408. doi: 10.3390/ijms23137408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Olsen G.M., Rinder H.M., Tormey C.A. De novo acquired hemophilia as an immune dysregulation phenomenon following SARS-CoV-2 infection. Transfusion. 2021;61:989–991. doi: 10.1111/trf.16254. [DOI] [PubMed] [Google Scholar]
- 137.Liu Z., Sun Y., Qi Z., Cao L., Ding S. Mitochondrial transfer/transplantation: An emerging therapeutic approach for multiple diseases. Cell Biosci. 2022;12:66. doi: 10.1186/s13578-022-00805-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Hernandez-Cruz E.Y., Amador-Martinez I., Aranda-Rivera A.K., Cruz-Gregorio A., Pedraza C.J. Renal damage induced by cadmium and its possible therapy by mitochondrial transplantation. Chem. Biol. Interact. 2022;361:109961. doi: 10.1016/j.cbi.2022.109961. [DOI] [PubMed] [Google Scholar]
- 139.Fan X.Y., Guo L., Chen L.N., Yin S., Wen J., Li S., Ma J.Y., Jing T., Jiang M.X., Sun X.H., et al. Reduction of mtDNA heteroplasmy in mitochondrial replacement therapy by inducing forced mitophagy. Nat. Biomed. Eng. 2022;6:339–350. doi: 10.1038/s41551-022-00881-7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.