Abstract
Antigen-specific interleukin-5 (IL-5), gamma interferon (IFN-γ), and granulocyte-macrophage colony-stimulating factor (GM-CSF) responses in individuals living in an area of hyperendemicity for onchocerciasis in Cameroon were examined. The responses against antigens prepared from Onchocerca volvulus third-stage larvae (L3), molting L3 (mL3), and crude extract from adult males (M-OvAg) were compared to the responses against antigens from adult female worms and skin microfilariae. Cytokine responses for the putatively immune individuals (PI) and the infected individuals (INF) were compared. A differential cytokine profile of IL-5 (Th2 phenotype) and IFN-γ (Th1 phenotype) was found in these individuals in response to the antigens. In both the PI and the INF, Th2 responses against all the antigens tested were dominant. However, in the PI group as a whole, there was an enhanced Th2 response against the larval antigens and the adult male and adult female antigens, and a Th1 response in a subgroup of the PI (27 to 54.5%) against L3, mL3, and M-OvAg antigens was present. While the PI produced significantly higher levels of GM-CSF against L3, mL3, and M-OvAg antigens than the INF, there was no difference in the GM-CSF responses of the groups against the other antigens. The present study indicated that, in comparison to the INF, the PI have distinct larva-specific and adult male-specific cytokine responses, thus supporting the premise that immunological studies of the PI would lead to the identification of immune mechanisms and the target genes that play a role in protective immunity.
Our present knowledge of antionchocerca immunity comes from three basic approaches: correlative and immunoepidemiological studies of human populations living in areas in which onchocerciasis is endemic, in vitro studies, and in vivo studies using defined animal models. The studies of human populations have provided evidence that naturally acquired immunity against Onchocerca volvulus infection can occur in humans. First, within regions where onchocerciasis is endemic, 1 to 5% of the population who have been exposed to high transmission rates of infection have no clinical manifestations of the disease and are thus distinguished as the putatively immune individuals (PI) (12, 16, 48). Second, in infected individuals (INF) the number of skin microfilariae (Smf) tends to level off between 20 and 40 years of age, which suggests an acquired means of limiting infection (10). Third, sera from both the PI and the INF have been shown to promote in vitro opsonization and killing of the infective third-stage larvae (L3) and to limit molting of L3 to fourth-stage larvae (18, 20). Experimental studies using animal models have shown that antigens from L3 and the subsequent developmental stages of molting L3 (mL3) are promising sources for protective antigens and the targets for control of filarial infections (11, 30, 31, 41). Partial protection against O. volvulus L3 challenge has been achieved by immunization of mice with irradiated L3 (25, 30, 39, 45) and vaccination with recombinant O. volvulus larval antigens (19, 23, 46). In the Acanthocheilonema viteae Jird model protection was elicited by irradiated L3, supernatants from L3 cultured in vitro, and live mL3 but not by live L3 (11, 31). In addition, in the cat-Brugia pahangi model, animals that were exposed to a repeated infection with L3 developed immunity against an L3 challenge infection (7, 8). Protective responses in humans against the infective stages of the parasite would prevent the development of adult worms and microfilariae (mf) that cause the disease.
In efforts to define the mechanisms of immunity against O. volvulus in humans, the cellular and humoral immune responses in the PI have been compared with those in the INF. Because of the scarcity of larval antigens, previous studies have been limited to antigens of O. volvulus adult worms, which contain microfilarial antigens as well. In most of these studies the humoral and cellular immune responses in PI were found to differ from those in INF, providing more acceptance that the observed clinical difference between the two groups is associated with immune system-mediated resistance (3–5, 12, 13, 32, 36, 37, 38, 43, 44, 47, 48). Some of these studies have concluded that protective immunity in the PI appeared to be accompanied by diminished specific immunoglobulin E (IgE), IgG, and IgG subclass responses and an enhanced production of interleukin-2 (IL-2) and gamma interferon (IFN-γ) in response to adult worm antigens, a profile suggesting protective Th1-type responses (24, 32, 37, 43, 44). However, in recent studies done in Benin, the PI were found to produce prominently IL-2, IL-5, and granulocyte-macrophage colony-stimulating factor (GM-CSF) in response to adult worm antigens and thus exhibited no predominance of the Th1-type cytokine response or had a strict Th1 or Th2 dichotomy (3).
In the onchocerca mouse model, where the challenge infection consists of L3 implanted in diffusion chambers, immune responses of the Th2 type including the production of IL-4, IL-5, IgE, and eosinophil-mediated effector reactivity were shown to account for the partial protection induced by immunization with irradiated O. volvulus L3 (25). The importance of Th2-type responses for mediating protection against L3 in mice was subsequently determined by cytokine depletion experiments (26) and after using IL-4 and IFN-γ knockout mice for immunization (22).
Given that T cells play a critical role in regulating the immune response, determining the stage specificity of the T-cell responses is essential for understanding protective immunity against the parasite. While previous cellular studies of onchocerciasis were restricted to the use of antigens from the adult worms, in this study we have examined for the first time the cytokine responses (IL-5, IFN-γ, and GM-CSF) produced by peripheral blood mononuclear cells (PBMC) from the PI and the INF to antigens from L3 and mL3, the stages shown to be more relevant to protective immunity (1, 25, 45), and crude extracts prepared from adult male worms (M-OvAg). These responses were compared to those induced by antigens from crude extracts prepared from adult female worms (F-OvAg) which contained microfilarial antigens, and from Smf. It is the adult female and Smf stages that result in patent infection and disease.
MATERIALS AND METHODS
Study population.
The study was performed in the Kumba region, an area of hyperendemicity for onchocerciasis in southwest Cameroon. A total of 1,440 individuals who were either born or had lived in this region for at least 10 years received thorough physical, parasitological, and ophthalmological examinations, and skin biopsies taken from right and left hips and calves determined the number of O. volvulus mf. One hundred sixty-eight of these individuals (11.7%) were negative for Smf and were classified mf negative (mf−), whereas individuals positive for Smf were grouped as INF. In order to identify the PI, the biopsies of the mf− individuals were tested for the presence of a tandem repeat DNA specific for O. volvulus. The O. volvulus 150-mer DNA repeats were amplified by PCR and identified by Southern blotting using a specific internal O. volvulus probe (35). This confirmatory test resulted in a subgroup of 51 individuals (3.6% of the total 1,440 surveyed, 30% of those mf−) that were negative for Smf and negative by PCR (mf− PCR−). Furthermore, this subgroup did not exhibit any clinical signs of the disease and was thus classified as the PI group (12). The PI were examined every 6 months for clinical manifestations of onchocerciasis, and four skin biopsies were taken from each individual. All maintained their mf− PCR− status and had no new clinical signs of onchocerciasis for an additional 2 years. Fifty percent of the INF and 90% of the PI were 15 years of age or less. Twelve PI (5 to 15 years of age) and 26 INF (with Smf ranging from 0.25 to 174 per skin snip) matched for age and sex consented to participate in this study. The individuals were from villages around Kumba: Marumba I, Marumba II, Boa Bakundu, Bombanda, and Bombele.
Antigens.
All parasite material was collected in our research facility at the Tropical Medicine Research Station, Kumba, Cameroon. Crude antigen extracts were prepared from different stages of O. volvulus including L3 (obtained from infected black flies), mL3, adult female worms, adult male worms, and Smf. mL3 were generated by incubating L3 in vitro in solution containing a 1:1 mixture of Iscove modified Dulbecco medium and NCTC-135, 20% fetal calf serum, and antibiotic-antimycotic solution (GIBCO BRL Life Technologies, Gaithersburg, Md.) for 3 days at 37°C (6). Larvae were collected after 1, 2, or 3 days in culture, washed in phosphate-buffered saline (PBS), and quick-frozen in liquid nitrogen. Ultrastructural examination of such larvae by electron microscopy confirmed that the cultured larvae had started the molting process, as the separation between the cuticle of L3 and the newly synthesized cuticle of the fourth-stage larvae was evident in the cross sections (33). The mL3 antigen preparation was made from a pooled mixture of similar numbers of larvae that were collected on day 1, 2, or 3 in culture. Crude L3, mL3, and Smf antigens and antigens from F-OvAg and M-OvAg were prepared as described before (48). Briefly, the worms were ground to a powder using a Bessman tissue pulverizer (Spectrum Lab Products, Houston, Tex.) and further disrupted by sonication before extraction in PBS containing 10 mM 3-(3-cholamidopropyl)-dimethylammonio-2-hydroxy-1-propanesulfonate (Calbiochem, San Diego, Calif.) and protease inhibitors (Sigma, St. Louis, Mo.; 2 mM phenylmethylsulfonyl fluoride, 0.2 mM N-α-p-tosyl-l-lysine chloromethyl ketone, 0.2 nM N-tosyl-l-phenylalanine chloromethyl ketone, 25 μg of leupeptin/ml, 10 mM EDTA). The insoluble material was extracted twice in the same buffer for 12 h at 4°C. The pooled soluble extracts of each stage-specific preparation were then dialyzed against PBS, centrifuged at 4°C, and filter sterilized.
Lymphocyte stimulation.
PBMC from each individual were isolated by density gradient centrifugation over Ficoll (Sigma). Cells were cultured at 2 × 106/ml in RPMI 1640 medium containing 10% heat-inactivated fetal calf serum, 25 mM HEPES, 2 mM l-glutamine, and 50 μg of gentamicin (BioWhittaker, Walkersville, Md.)/ml (13). The cells were cultured for 5 days in the presence of the following final concentrations of antigen preparations: F-OvAg at 5 μg/ml; L3 antigen at 0.5 μg/ml (equivalent to 50 L3/ml); mL3 antigen at 0.36 μg/ml (equivalent to 50 mL3/ml); Smf antigen at 0.25 μg/ml (equivalent to 500 Smf/ml), and M-OvAg at 2 μg/ml. The antigens were used at concentrations determined to give optimal responses in the exposed individuals. Similar antigen concentrations have been shown to induce significant proliferation in PI and INF in comparison to controls from areas where the disease is not endemic (data not shown). The cytokine response to a nonparasite antigen was obtained by using streptolysin-O (1:100; Difco, Detroit, Mich.). In addition, the PBMC were cultured in the presence of a mitogenic stimulus of phorbol myristate acetate (50 ng/ml) plus ionomycin (1 μg/ml) (Calbiochem) (13). The supernatants were harvested at day 5 and stored at −70°C until analyzed for IL-5 (marker for Th2 phenotype), IFN-γ (marker for Th1 phenotype), and GM-CSF cytokine production. We determined the antigen-specific GM-CSF production because GM-CSF is known to enhance the activity of neutrophils, basophils, monocytes, and eosinophils (17). These cellular components of the immune response can participate in antibody-dependent cellular cytotoxicity, an effector mechanism that was shown to be important for protective immunity against filarial and other helminth diseases (34, 40).
Cytokine production and statistical analysis.
The quantities of the cytokines IL-5, IFN-γ, and GM-CSF present in the culture supernatants were measured by using commercial sandwich enzyme-linked immunosorbent assay kits (R & D Systems, Minneapolis, Minn.). Cytokine levels were expressed in picograms per milliliter. The net antigen-specific production of a cytokine was calculated by subtracting the quantity of the cytokine produced by PBMC cultured without antigen from that produced by PBMC cultured with a specific antigen. Because the distribution of the data was not normal, comparisons between PI and INF were made by the nonparametric Mann-Whitney U test. Fisher's exact test (F test) was used for comparison of the response rates, responders versus nonresponders, in the different study groups. Cytokine production was considered positive if the individual had a net production above 10 pg/ml.
RESULTS
Enhanced Th1- and Th2-type responses in PI in response to L3 and mL3 antigens.
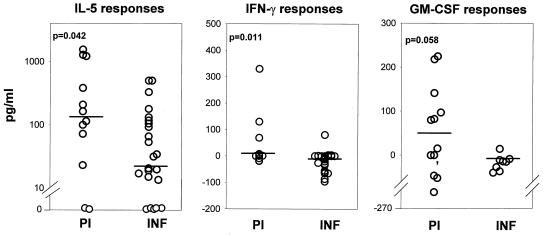
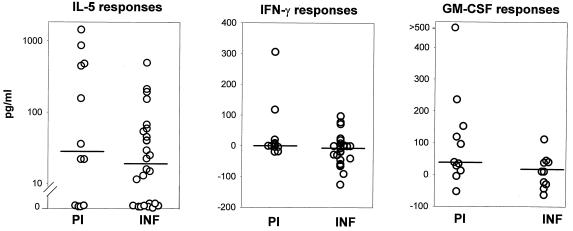
The PBMC from both the PI and the INF produced IL-5 in response to the L3 antigens; however, the PI produced significantly more of the cytokine than the INF (Fig. 1). Second, significantly more PI produced IL-5 (83.3%) than IFN-γ (27%) (P = 0.012; F test) and significantly more INF produced IL-5 (75%) than IFN-γ (4%) (P ≤ 0.0001; F test), indicating that Th2 responses against L3 antigens are predominant in both groups. Interestingly, in the PI, L3 antigens also induced significantly higher levels of IFN-γ than in the INF, albeit low median levels in both groups (Fig. 1). Although the IFN-γ levels were statistically higher in the PI than in the INF, it should be noted that the significance of this difference is based on only 27% of the PI producing the cytokine in comparison to 4% of the INF. It seems therefore that the PI group is divided into two distinct immunological subgroups when stimulated with L3 antigens: PBMC of eight individuals produced only IL-5 and no IFN-γ, while PBMC of three produced both. Third, PBMC from the PI produced higher levels of GM-CSF than PBMC from the INF after stimulation with L3 antigens (median values: PI, 47.6 pg/ml; INF, −14.5 pg/ml) although the GM-CSF levels in the PI are not quite significantly higher than those in the INF (Fig. 1; P = 0.058). The INF on the other hand exhibited only a Th2 phenotype (IL-5) in response to the L3 antigens.
FIG. 1.
Cytokine responses to L3 antigens in PI and INF. Values for IL-5 (PI, n = 12; INF, n = 24), IFN-γ (PI, n = 11; INF, n = 24), and GM-CSF (PI, n = 12; INF, n = 9) levels are depicted. The statistical significance between the groups is indicated by the P value. The Mann-Whitney U test was used to test the significance by comparing the medians, which are indicated by horizontal lines.
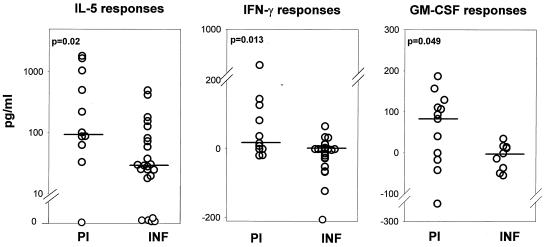
In response to mL3 antigens, PBMC from the PI produced significantly higher quantities of IL-5, IFN-γ, and GM-CSF than those from the INF (Fig. 2). The significant Th1 response in the PI is attributed to 54.5% of the group, again implying the presence of two distinct subgroups, one that has only a Th2 response and the other that has a mixed Th1/Th2 response. In comparison, the INF exhibited only a Th2 phenotype in response to the mL3 antigens, as significantly more INF produced IL-5 (78%) than IFN-γ (21.7%) (P = 0.0003; F test).
FIG. 2.
Cytokine responses to mL3 antigens in PI and INF. Values for IL-5 (PI, n = 12; INF, n = 23), IFN-γ (PI, n = 11; INF, n = 23), and GM-CSF (PI, n = 12; INF, n = 9) levels are depicted. The statistical significance between the groups is indicated by the P value. The Mann-Whitney U test was used to test the significance by comparing the medians, which are indicated by horizontal lines.
Enhanced Th1- and Th2-type responses in PI in response to antigens from adult male worms (M-OvAg).
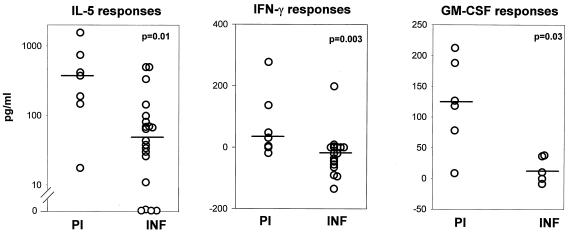
Our interest in the possible contribution of adult male worms to protective immunity arose after we found by using Western blot analysis that sera from the PI had recognized unique proteins (molecular masses of 250, 57, 45, 36, 34, and 28 kDa) in adult male worm extracts not recognized by sera from the INF (21). These results suggested that male-specific antigens could probably induce antimale cellular and/or humoral responses that promote the killing or block the development of male worms. This will impede the reproduction and release of mf by fertilized female worms and result in an occult and nonpatent infection that might also account for the clinical status of the PI: lack of Smf and clinical manifestations of the disease (12). When the cytokine responses in response to M-OvAg were evaluated, the PBMC from the PI were found to produce significantly higher quantities of IL-5, IFN-γ, and GM-CSF in response to M-OvAg than the INF (Fig. 3). The elevated Th1 response in the PI is attributed to 54.5% of the group, once more implying the presence of two distinct subgroups, one that has only a Th2 response and the other that has a mixed Th1/Th2 response. In comparison, the INF exhibited only a Th2 phenotype in response to the M-OvAg, as significantly more INF produced IL-5 (80%) than IFN-γ (5%) (P = 0.0001; F test).
FIG. 3.
Cytokine responses to M-OvAg antigens in PI and INF. Values for IL-5 (PI, n = 7; INF, n = 20), IFN-γ (PI, n = 7; INF, n = 20), and GM-CSF (PI, n = 6; INF, n = 5) levels are depicted. The statistical significance between the groups is indicated by the P value. The Mann-Whitney U test was used to test the significance by comparing the medians, which are indicated by horizontal lines.
Enhanced Th2-type response in PI in response to antigens from adult female worms (F-OvAg).
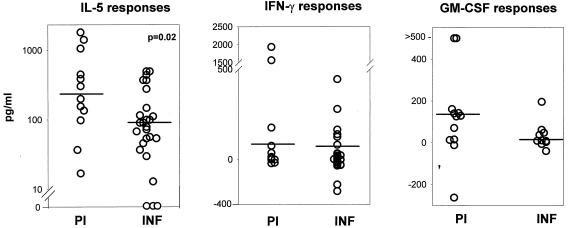
In response to F-OvAg, PBMC from both the PI and the INF produced IL-5; however, the PI produced significantly more of the cytokine than the INF (Fig. 4). Moreover, significantly more PI and INF produced IL-5 than produced IFN-γ. One hundred percent of the PI produced IL-5 versus 54.5% that produced IFN-γ (P = 0.013; F test), and 88% of the INF produced IL-5 versus 38% that produced IFN-γ (P = 0.0004; F test). Thus, in both the PI and the INF, F-OvAg antigens induced a Th2-type response, with the PI exhibiting an elevated Th2 response. PBMC from the PI and the INF produced comparable levels of GM-CSF and IFN-γ in response to F-OvAg (Fig. 4).
FIG. 4.
Cytokine responses to F-OvAg antigens in PI and INF. Values for IL-5 (PI, n = 12; INF, n = 26), IFN-γ (PI, n = 11; INF, n = 26), and GM-CSF (PI, n = 12; INF, n = 10) levels are depicted. The statistical significance between the groups is indicated by the P value. The Mann-Whitney U test was used to test the significance by comparing the medians, which are indicated by horizontal lines.
Th2-type response in PI and INF in response to antigens from Smf.
Analysis of the IL-5, IFN-γ, and GM-CSF levels produced in response to Smf antigens indicated that the PBMC from both the PI and the INF produced similar amounts of each of the cytokines (Fig. 5). The number of individuals producing IL-5 was compared with the number of individuals producing IFN-γ, and it seems that more PI produced IL-5 (66.6%) than IFN-γ (27%) and that significantly more INF produced IL-5 (65%) than IFN-γ (15%) (P = 0.009; F test) in response to Smf antigens. Thus, qualitatively, both the PI and the INF exhibited a predominant Th2 response to Smf antigens.
FIG. 5.
Cytokine responses to Smf antigens in PI and INF. Values for IL-5 (PI, n = 12; INF, n = 26), IFN-γ (PI, n = 11; INF, n = 26), and GM-CSF (PI, n = 11; INF, n = 10) levels are depicted. The statistical significance between the groups is indicated by the P value. The Mann-Whitney U test was used to test the significance by comparing the medians, which are indicated by horizontal lines.
Cytokine responses induced by a mitogen and a nonparasitic antigen.
The PBMC from both the PI and the INF produced similarly high levels of IL-5 and IFN-γ (more than 500 pg/ml) in response to a mitogenic stimulus (phorbol myristate acetate plus ionomycin) or streptolysin-O, a nonparasitic antigen (data not shown). Mitogen and nonparasitic antigen stimulation of the PBMC revealed that, while the cells of both groups had equivalent capacities to produce both cytokines, the inability of PBMC from the INF and some of the PI to produce IFN-γ was O. volvulus antigen specific.
DISCUSSION
Although the existence of immunity to O. volvulus infection is supported by the presence of PI individuals in areas where onchocerciasis is highly endemic, there has been no investigation of the underlying mechanisms of specific antilarval cellular responses, which could prevent the establishment of infective larvae in these individuals. To date all of the correlative and immunoepidemiological studies of human populations living in areas of endemicity for onchocerciasis were done using adult worm antigens based on their ready availability rather than on their ability to protect against incoming infective larvae. PI from Ecuador and Togo were shown to secrete predominately IFN-γ in response to adult worm antigens, which supported the theory that Th1-type responses are correlated with protection (13, 43). However, in a study from Benin, PBMC from PI secreted only the IL-5 and GM-CSF cytokines and furthermore the PI produced significantly more of both cytokines than the INF (3), implying a Th2 type of response. Thus, no clear and consistent correlation between distinct cellular immune responses to adult antigens and immunity has been established.
This is the first study describing the cytokine responses of human T cells to larval and adult male antigens of O. volvulus in individuals who live in a region of hyperendemicity for O. volvulus. When the PBMC from the PI and the INF groups of individuals were cultured in the presence of two distinct larval antigens (L3 and mL3 antigens) and adult worm antigens (M-OvAg), a differential cytokine profile was found (Fig. 1 to 3). First, although an IL-5 response (Th2) was present in both the PI and the INF in response to L3, mL3, and M-OvAg antigens, the PI group as a whole had a significantly elevated IL-5 response to these antigens. Second, in comparison to the INF, a subgroup of the PI also had significantly elevated IFN-γ responses to L3 (27%), mL3 (54.5%), and M-OvAg (54.5%) antigens. Third, the PI secreted higher levels of GM-CSF in response to L3, mL3, and M-OvAg antigens. In contrast, the F-OvAg and Smf antigens induced only a Th2-type response in the PI group. The INF conversely exhibited only a Th2 phenotype in response to the L3, mL3, and M-OvAg antigens as well as in response to F-OvAg and the Smf antigen. The levels of other cytokines, IL-4 (Th2) and IL-2 (Th1), were negligible in both groups against all antigens (data not shown).
The importance of the predominant Th2 responses for protective immunity in humans against the O. volvulus infective-stage larvae is corroborated by the Th2 responses seen in mice that were protected against an L3 challenge following immunization with irradiated O. volvulus larvae (22, 26). One possible Th2-mediated protective mechanism in the PI could be through the elevated and specific IL-5 and GM-CSF cytokine responses against L3 and mL3 antigens observed in this study. Both cytokines are able to augment the number of cells from the granulocyte-macrophage lineage (neutrophils, eosinophils, and basophils) and enhance their activity (17). GM-CSF in conjunction with IL-5 was also shown to induce eosinophil-specific chemotaxis (27). As a result, these major Th2-associated effector cells, in combination with larva-specific antibodies, could participate in antibody-dependent antilarval cell-mediated cytotoxicity reactions. This is corroborated by previous studies where we found that human neutrophils were capable of promoting antibody-mediated killing of O. volvulus L3 as well as the inhibition of L3 molting in vitro (20, 21). Furthermore, in vitro studies by Leke et al. (29) have shown specific serum-mediated leucocyte adherence to O. volvulus L3 in uninfected individuals in comparison to no significant adherence in highly infected individuals. In the O. volvulus mouse model, maximal levels of eosinophils and the presence of IL-5 in the diffusion chambers were coincident with the time of parasite killing in protected animals (26).
In addition to the PI having a significant and elevated Th2 response as a whole when stimulated by L3, mL3, and M-OvAg antigens there was a subgroup that also had a distinct Th1 response, albeit in small quantities. The presence of both Th1 and Th2 responses in this subgroup suggests that not all PI have a complete bias towards a Th2 response. Even though the Th1 response in the present study was only apparent in a subgroup of the PI (27 to 54.5%) and the mean net production of IFN-γ in the group as a whole was mostly less than 30 pg/ml for any antigen, it still points to the possibility that some of the PI have potentially more than one mechanism of protective immunity against larvae and adult male worms. In a study conducted in Liberia (14), a mixed Th1/Th2 type of immune response was observed in response to the S1 protein, a distinct protective recombinant larval protein of O. volvulus (2, 19). The S1 protein was preferentially recognized by IgG3 antibodies of the PI in that study and induced both IFN-γ and IL-5 secretion by S1 antigen-specific CD4+ T cells from the PI (9). Those authors have suggested that the induction of both IFN-γ and IL-5 responses by the S1 protein may trigger in vivo macrophage- and eosinophil-dependent killing of L3. Th1 cytokines can stimulate the macrophage cells (15), which, together with antigen-specific antibodies, could also participate in antibody-dependent cellular cytotoxicity reactions against larval stages. Th1 cells can produce IgG1, and Th2 cells can produce IgE and IgG3 cytophilic antibodies. Specific killing of the O. volvulus larvae would prevent the establishment of an infection. Although the mechanisms by which either Th1 or Th2 cells and their products mediate specific killing of the filarial parasite remain to be elucidated, there is evidence for both Th1- and Th2-type responses being protective in many helminthic infections, depending on the particular model used (13, 34). In the Brugia malayi mouse model it was clearly shown that the host protective immune responses were not dependent on the predominate Th2-type response that was elicited by the filarial parasite, as T-cell populations other than Th2 cells were also capable of conferring protection against L3 (28). However, to confirm the role of Th1-type responses in protective immunity against O. volvulus would require additional investigations using a larger PI study group, optimal antigen concentrations for stimulation, and more sensitive detection assays for secreted or intracellular IFN-γ.
Of particular interest are our results regarding the IL-5, IFN-γ, and GM-CSF responses in the PI to male antigens (M-OvAg), which were similar to those observed in response to L3 and mL3 antigens (Fig. 3). These responses may account for the induction of antimale cellular and humoral responses that could promote the killing, or block the development, of male worms. This would impede the reproduction and release of mf by the female worms and result in an occult and nonpatent infection. In onchocerciasis and lymphatic filariasis, it is believed that PI and healthy individuals from areas of endemicity more likely represent a heterogeneous group of individuals capable of controlling the development of infective larvae or of having an occult filarial infection (12). Absent the ability to distinguish those that are truly immune to infection from those who may have an occult infection, further analysis of the immune responses of the PI to the different stage-specific recombinant antigens versus the responses to crude antigens may further elucidate the nature of stage-specific protective immunity. It would be important in future studies to include PI who are older than 15 years of age, thus allowing a more appropriate immunological representation of such a group.
In the INF, the response against the larval stages was predominantly of the Th2 type, as significantly more of the INF produced IL-5 than IFN-γ. It seemed that the IFN-γ response in the INF group as a whole was down regulated in the presence of O. volvulus antigens, as many of the INF had antigen-specific levels of the cytokine below zero (Fig. 1 to 5). The down regulation of the Th1-type response in the INF to parasite antigens was previously shown to be correlated with the appearance of mf in the skin and is believed to be induced by the mf (42). The Th2 response in the INF, although significantly lower than in the PI, could provide the INF with antilarval protective immunity as well, immunity that can protect the already-infected individuals from new infections with L3. Such immunity, which is termed concomitant immunity, has been shown to be present in lymphatic filariasis and other helminthic infections (34).
In conclusion, our observations showed for the first time that in comparison to the INF the PI have distinct and specific antilarval and antimale cytokine responses. The PI as a whole produced enhanced GM-CSF and IL-5 cytokine levels and had a predominant Th2 type of response, with a subgroup of PI also producing IFN-γ (Th1), while the INF produced only IL-5 (Th2). Although the mechanisms of immunity are not clear, this study provides evidence that both Th2-type and GM-CSF-mediated responses are important for protective immunity. The Th1-mediated responses require more studies. This study also pointed out the possibility that adult male worms may provide an additional new target for vaccine development. These results are important not only because they contribute to the understanding of the parasite stage-specific immune responses in humans but also because of their relevance to current investigations of potentially protective recombinant antigens. Studies to determine the T-cell responses in humans to defined antigens derived from different stages of the parasite and to identify those antigens that are capable of inducing protective immune responses are under way. As the responses in humans are highly heterogeneous, a viable vaccine might need to consist of a pool of antigens from different stages of the parasite that could elicit a broad range of responses providing protection to all individuals who live in areas of endemicity for onchocerciasis.
ACKNOWLEDGMENTS
We thank the people of the villages around Kumba, Marumba I, Marumba II, Boa Bakundu, Bombanda and Bombele, Cameroon, that have participated in the study and the personnel at the Tropical Medicine Research Station for their help throughout the course of the study. We thank David Abraham, Amy Klion, and Cohava Gelber for critical review of the manuscript.
The study was partially supported by grant RO1 AI 42328-02 from the National Institutes of Health.
REFERENCES
- 1.Bancroft A J, Grencis R K, Else K J, Devaney E. Cytokine production in BALB/c mice immunized with radiation attenuated third stage larvae of the filarial nematode, Brugia pahangi. J Immunol. 1993;150:1395–1402. [PubMed] [Google Scholar]
- 2.Bradley J E, Elson L, Tree T I, Stewart G, Guderian R, Calvopina M, Paredes W, Araujo E, Nutman T B. Resistance to Onchocerca volvulus: differential cellular and humoral responses to a recombinant antigen, OvMBP20/11. J Infect Dis. 1995;172:831–837. doi: 10.1093/infdis/172.3.831. [DOI] [PubMed] [Google Scholar]
- 3.Brattig N, Nietz C, Hounkpatin S, Lucius R, Seeber F, Pichlmeier U, Pogonka T. Differences in cytokine responses to Onchocerca volvulus extract and recombinant Ov33 and OvL3-1 proteins in exposed subjects with various parasitologic and clinical states. J Infect Dis. 1997;176:838–842. doi: 10.1086/517317. [DOI] [PubMed] [Google Scholar]
- 4.Brattig N W, Tischendorf F W, Albiez E J, Buttner D W, Berger J. Distribution pattern of peripheral lymphocyte subsets in localized and generalized form of onchocerciasis. Clin Immunol Immunopathol. 1987;44:149–159. doi: 10.1016/0090-1229(87)90062-6. [DOI] [PubMed] [Google Scholar]
- 5.Brattig N W, Krawietz I, Abakar A Z, Erttmann K D, Kruppa T F, Massougbodji A. Strong IgG isotypic antibody response in sowdah type onchocerciasis. J Infect Dis. 1994;170:955–961. doi: 10.1093/infdis/170.4.955. [DOI] [PubMed] [Google Scholar]
- 6.Cupp E W, Bernardo M J, Kiszewski A E, Trpis M, Taylor H R. Large scale production of the vertebrate infective stage (L3) of Onchocerca volvulus (Filarioidea: Onchocerciadae) Am J Trop Med Hyg. 1988;38:596–600. doi: 10.4269/ajtmh.1988.38.596. [DOI] [PubMed] [Google Scholar]
- 7.Denham D A, McGreevy P B, Suswillo R R, Rogers R. The resistance to re-infection of cats repeatedly inoculated with infective larvae of Brugia pahangi. Parasitology. 1983;86:11–18. doi: 10.1017/s0031182000057127. [DOI] [PubMed] [Google Scholar]
- 8.Denham D A, Fletcher C. The cat infected with Brugia pahangi as a model of human filariasis. Ciba Found Symp. 1987;127:225–235. doi: 10.1002/9780470513446.ch15. [DOI] [PubMed] [Google Scholar]
- 9.Doetze A, Erttmann K D, Gallin M Y, Fleischer B, Hoerauf A. Production of both IFN-gamma and IL-5 by Onchocerca volvulus S1 antigen-specific CD4+ T cells from putatively immune individuals. Int Immunol. 1997;9:721–729. doi: 10.1093/intimm/9.5.721. [DOI] [PubMed] [Google Scholar]
- 10.Duke B O L, Moore P R. The contribution of different age groups to the transmission of onchocerciasis in a Cameroon forest village. Trans R Soc Trop Med Hyg. 1968;62:22–29. doi: 10.1016/0035-9203(68)90026-6. [DOI] [PubMed] [Google Scholar]
- 11.Eisenbeiss W F, Apfel H, Meyer T F. Protective immunity linked with distinct developmental stage of filarial parasite. J Immunol. 1994;152:735–742. [PubMed] [Google Scholar]
- 12.Elson L H, Guderian R H, Araujo E, Bradley J E, Days A, Nutman T B. Immunity to onchocerciasis: identification of a putatively immune population in a hyperendemic area of Ecuador. J Infect Dis. 1994;169:588–594. doi: 10.1093/infdis/169.3.588. [DOI] [PubMed] [Google Scholar]
- 13.Elson L H, Manuel Calvopina H, Wilson Paredes Y, Edmundo Araujo N, Bradley J E, Guderian R H, Nutman T B. Immunity to onchocerciasis: putative immune persons produce a Th1-like response to Onchocerca volvulus. J Infect Dis. 1995;171:652–658. doi: 10.1093/infdis/171.3.652. [DOI] [PubMed] [Google Scholar]
- 14.Erttmann K D, Buttner D W, Gallin M Y. A putative protein related to human chemokines encoded antisense to the cDNA of an Onchocerca volvulus antigen. Trop Med Parasitol. 1995;46:123–130. [PubMed] [Google Scholar]
- 15.Flieger D, Spengler U, Beier I, Kleinschmidt R, Hoff A, Varvenne M, Sauerbruch T, Schmidt-Wolf I. Enhancement of antibody dependent cellular cytotoxicity (ADCC) by combination of cytokines. Hybridoma. 1999;18:63–68. doi: 10.1089/hyb.1999.18.63. [DOI] [PubMed] [Google Scholar]
- 16.Gallin M Y, Edmonds K, Ellner J J, Erttmann K D, White A T, Newland H S, Taylor H R, Greene B M. Cell-mediated immune responses in human infection with Onchocerca volvulus. J Immunol. 1988;146:1999–2007. [PubMed] [Google Scholar]
- 17.Gasson J C, Fraser J K, Nimer S D. Human granulocyte-macrophage colony-stimulating factor (GM-CSF): regulation of expression. Prog Clin Biol Res. 1990;338:27–41. [PubMed] [Google Scholar]
- 18.Greene B M, Taylor H R, Aikawa M. Humoral and cellular immune responses to Onchocerca volvulus infection in humans. Rev Infect Dis. 1985;7:789–795. doi: 10.1093/clinids/7.6.789. [DOI] [PubMed] [Google Scholar]
- 19.Jenkins R E, Taylor M J, Gilvary N, Bianco A E. Characterization of a secreted antigen of Onchocerca volvulus with host-protective potential. Parasite Immunol. 1996;18:29–42. doi: 10.1046/j.1365-3024.1996.d01-10.x. [DOI] [PubMed] [Google Scholar]
- 20.Johnson E H, Irvine M, Kass P H, Browne J, Abdullai M, Prince A M, Lustigman S. Onchocerca volvulus: in vitro cytotoxic effects of human neutrophils and serum on third-stage larvae. Trop Med Parasitol. 1994;45:331–335. [PubMed] [Google Scholar]
- 21.Johnson E H, Lustigman S, Kass P H, Irvine M, Browne J, Prince A M. Onchocerca volvulus: a comparative study of in vitro neutrophil killing of microfilariae and humoral responses in infected and endemic normals. Exp Parasitol. 1995;81:9–19. doi: 10.1006/expr.1995.1087. [DOI] [PubMed] [Google Scholar]
- 22.Johnson E H, Schynder-Candrian S, Rajan T V, Nelson F K, Lustigman S, Abraham D. Immune responses to third stage larvae of Onchocerca volvulus in interferon-gamma and interleukin-4 knockout mice. Parasite Immunol. 1998;20:319–324. doi: 10.1046/j.1365-3024.1998.00148.x. [DOI] [PubMed] [Google Scholar]
- 23.Joseph G T, Huima T, Lustigman S. Characterization of an Onchocerca volvulus L3-specific larval antigen, Ov-ALT-1. Mol Biochem Parasitol. 1998;96:177–183. doi: 10.1016/s0166-6851(98)00094-2. [DOI] [PubMed] [Google Scholar]
- 24.King C L, Nutman T B. Regulation of the immune response in lymphatic filariasis and onchocerciasis. Immunol Today. 1991;12:A54–A58. doi: 10.1016/S0167-5699(05)80016-7. [DOI] [PubMed] [Google Scholar]
- 25.Lange A M, Yutanawiboonchai W, Lok J B, Trpis M, Abraham D. Induction of protective immunity against larval Onchocerca volvulus in a mouse model. Am J Trop Med Hyg. 1993;49:783–788. doi: 10.4269/ajtmh.1993.49.783. [DOI] [PubMed] [Google Scholar]
- 26.Lange A M, Yutanawiboonchai W, Scott P, Abraham D. IL-4 and IL-5 dependent protective immunity to Onchocerca volvulus infective larvae in BALB/cBYJ mice. J Immunol. 1994;153:205–211. [PubMed] [Google Scholar]
- 27.Lantero S, Sacco O, Scala C, Rossi G A. Stimulation of blood mononuclear cells of atopic children with the relevant allergen induces the release of eosinophil chemotaxins such as IL-3, IL-5, and GM-CSF. J Asthma. 1997;34:141–152. doi: 10.3109/02770909709075659. [DOI] [PubMed] [Google Scholar]
- 28.Lawrence R A, Allen J E, Gregory W F, Kopf M, Maizels R M. Infection of IL-4-deficient mice with the parasitic nematode Brugia malayi demonstrates that host resistance is not dependent on a T helper 2-dominated immune response. J Immunol. 1995;154:5995–6001. [PubMed] [Google Scholar]
- 29.Leke R G, Boto W M, Lando G, Ngu J L. Immunity to Onchocerca volvulus. Serum mediated leucocyte adherence to infective larvae in vitro. Trop Med Parasitol. 1989;40:39–41. [PubMed] [Google Scholar]
- 30.Lok J B, Abraham D. Animal models for the study of immunity in human filariasis. Parasitol Today. 1992;8:168–171. doi: 10.1016/0169-4758(92)90014-s. [DOI] [PubMed] [Google Scholar]
- 31.Lucius R, Textor G, Kern A, Kirsten C. Acanthocheilonema viteae: vaccination of Jirds with irradiation-attenuated stage-3 larvae and with exported larval antigens. Exp Parasitol. 1991;73:184–196. doi: 10.1016/0014-4894(91)90021-n. [DOI] [PubMed] [Google Scholar]
- 32.Luder C G, Schulz-Key H, Banla M, Pritze S, Soboslay P T. Immuno-regulation in onchocerciasis: predominance of Th1-type responsiveness to low molecular weight antigens of Onchocerca volvulus in exposed individuals without microfilaridermia and clinical disease. Clin Exp Immunol. 1996;105:245–253. doi: 10.1046/j.1365-2249.1996.d01-747.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lustigman S, Huima T, Brotman B, Miller K, Prince A M. Onchocerca volvulus: biochemical and morphological characteristics of the surface of third- and fourth-stage larvae. Exp Parasitol. 1990;71:489–495. doi: 10.1016/0014-4894(90)90075-n. [DOI] [PubMed] [Google Scholar]
- 34.Maizels R M, Bundy D A, Selkirk M E, Smith D F, Anderson R M. Immunological modulation and evasion by helminth parasites in human populations. Nature. 1993;365:797–805. doi: 10.1038/365797a0. [DOI] [PubMed] [Google Scholar]
- 35.Meredith S E, Lando G, Gbakima A A, Zimmerman P A, Unnasch T R. Onchocerca volvulus: application of the polymerase chain reaction to identification and strain differentiation of the parasite. Exp Parasitol. 1991;73:335–344. doi: 10.1016/0014-4894(91)90105-6. [DOI] [PubMed] [Google Scholar]
- 36.Ngu J L, Tume C, Lando G, Ndumbe P, Leke R G, Titanji V, Asonganyi T. Comparative studies of clinical groups of patients in an onchocerciasis endemic area for evidence of immune-mediated protection. Trop Med Parasitol. 1989;40:460–463. [PubMed] [Google Scholar]
- 37.Ottesen E A. Immune responsiveness and pathogenesis of human onchocerciasis. J Infect Dis. 1995;171:659–671. doi: 10.1093/infdis/171.3.659. [DOI] [PubMed] [Google Scholar]
- 38.Plier D A, Awadzi K, Freedman D O. Immunoregulation in onchocerciasis: persons with ocular inflammatory disease produce a Th2-like response to Onchocerca volvulus antigen. J Infect Dis. 1996;174:380–386. doi: 10.1093/infdis/174.2.380. [DOI] [PubMed] [Google Scholar]
- 39.Prince A M, Brotman B, Johnson E H, Smith A, Pasqual D, Lustigman S. Onchocerca volvulus: immunization of chimpanzees with X-irradiated third stage larvae. Exp Parasitol. 1992;74:239–250. doi: 10.1016/0014-4894(92)90147-3. [DOI] [PubMed] [Google Scholar]
- 40.Rainbird M A, MacMillan D, Meeusen E N. Eosinophil-mediated killing of Haemonchus contortus larvae: effect of eosinophil activation and role of antibody, complement and interleukin-5. Parasite Immunol. 1998;20:93–103. doi: 10.1046/j.1365-3024.1998.00132.x. [DOI] [PubMed] [Google Scholar]
- 41.Selkirk M E, Maizels R M, Yazdanbaksh M. Immunity and the prospects for vaccination against filariasis. Immunobiology. 1992;184:263–281. doi: 10.1016/S0171-2985(11)80479-1. [DOI] [PubMed] [Google Scholar]
- 42.Soboslay P T, Dreweck C M, Taylor H R, Brotman B, Wenk P, Greene B M. Experimental onchocerciasis in chimpanzees. Cell-mediated immune responses, and production and effects of IL-1 and IL-2 with Onchocerca volvulus infection. J Immunol. 1991;147:346–353. [PubMed] [Google Scholar]
- 43.Soboslay P T, Geiger S M, Weiss N, Banla M, Luder C G, Dreweck C M, Batchassi E, Boatin B A, Stadler A, Schulz-Key H. The diverse expression of immunity in humans at distinct states of Onchocerca volvulus infection. Immunology. 1997;90:592–599. doi: 10.1046/j.1365-2567.1997.00210.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Steel C, Nutman T B. Regulation of IL-5 in onchocerciasis. A critical role for IL-2. J Immunol. 1993;150:5511–5518. [PubMed] [Google Scholar]
- 45.Taylor M J, Van Es R P, Shay K, Folkard S G, Townson S, Bianco A E. Protective immunity against Onchocerca volvulus and Onchocerca lienalis infective larvae in mice. Trop Med Parasitol. 1994;45:17–23. [PubMed] [Google Scholar]
- 46.Taylor M J, Jenkins R E, Bianco A E. Protective immunity induced by vaccination with Onchocerca volvulus tropomyosin in rodents. Parasite Immunol. 1998;18:219–225. doi: 10.1046/j.1365-3024.1996.d01-93.x. [DOI] [PubMed] [Google Scholar]
- 47.Tsang V C, Boyer A E, Pilcher J B, Eberhard M L, Reimer C B, Zea-Flores G, Zhou W, Richards F O. Guatemalan human onchocerciasis. Systemic analysis of patient populations, nodular antigens and specific isotypic reactions. J Immunol. 1991;146:3993–4000. [PubMed] [Google Scholar]
- 48.Ward D J, Nutman T B, Zea-Flores G, Portocarrero C, Lujan A, Ottesen E A. Onchocerciasis and immunity in humans: enhanced T cell responsiveness to parasite antigen in putatively immune individuals. J Infect Dis. 1988;157:536–543. doi: 10.1093/infdis/157.3.536. [DOI] [PubMed] [Google Scholar]