Abstract
Bicuspid aortic valve (BAV) patients develop ascending aortic (AAo) dilation. The pathogenesis of BAV aortopathy (genetic vs. haemodynamic) remains unclear. This study aims to identify regional changes around the AAo wall in BAV patients with aortopathy, integrating molecular data and clinical imaging. BAV patients with aortopathy (n = 15) were prospectively recruited to surgically collect aortic tissue and measure molecular markers across the AAo circumference. Dilated (anterior/right) vs. non-dilated (posterior/left) circumferential segments were profiled for whole-genomic microRNAs (next-generation RNA sequencing, miRCURY LNA PCR), protein content (tandem mass spectrometry), and elastin fragmentation and degeneration (histomorphometric analysis). Integrated bioinformatic analyses of RNA sequencing and proteomic datasets identified five microRNAs (miR-128-3p, miR-210-3p, miR-150-5p, miR-199b-5p, and miR-21-5p) differentially expressed across the AAo circumference. Among them, three miRNAs (miR-128-3p, miR-150-5p, and miR-199b-5p) were predicted to have an effect on eight common target genes, whose expression was dysregulated, according to proteomic analyses, and involved in the vascular-endothelial growth-factor signalling, Hippo signalling, and arachidonic acid pathways. Decreased elastic fibre levels and elastic layer thickness were observed in the dilated segments. Additionally, in a subset of patients n = 6/15, a four-dimensional cardiac magnetic resonance (CMR) scan was performed. Interestingly, an increase in wall shear stress (WSS) was observed at the anterior/right wall segments, concomitantly with the differentially expressed miRNAs and decreased elastic fibres. This study identified new miRNAs involved in the BAV aortic wall and revealed the concomitant expressional dysregulation of miRNAs, proteins, and elastic fibres on the anterior/right wall in dilated BAV patients, corresponding to regions of elevated WSS.
Keywords: aortic segmentation, microRNAs, proteins, wall shear stress
1. Introduction
Bicuspid aortic valve (BAV) is the most common congenital heart disease with 1–2% prevalence in the general population, and its heterogenous nature commonly affects both the aortic valve and the aorta, leading to aortic valve stenosis/regurgitation and progressive ascending aortic (AAo) dilation [1,2]. The association between AAo dilation and BAV has been thoroughly confirmed, yet the exact molecular and haemodynamic mechanisms leading to BAV-associated aortopathy remain unclear [3].
MicroRNAs are small non-coding RNAs, which canonically repress the expression of their “target gene” at the post-transcriptional level, and are abundantly expressed in the vasculature, guiding vascular development and remodelling [4]. They have been implicated in aneurysm formation in the ascending aorta [5] and have been proposed as circulating biomarkers in this clinical setting [6]. Increasing evidence indicates that miRNAs play a role in mechanotransduction events [7], suggesting that structural alterations in aortopathy could be partially mediated by miRNAs sensitive to cyclic biomechanical stress (“mechanomiRs”).
We therefore conducted a detailed prospective study in a population of patients with BAV aortopathy, collecting tissue biopsies and imaging data to study local, segmental molecular, and haemodynamic changes around the aortic circumference. This study proposes a methodological framework to conduct segmental analyses on different molecular levels, complemented by additional preliminary haemodynamic observations, to study the complex nature of BAV aortopathy through dynamic and likely intertwined changes. In the literature, two studies [8,9] compared media degeneration, including elastic degradation to wall shear stress (WSS), while another study [10] compared miRNA changes between the inner and outer curvature of the AAo. Combining miRNA and proteomic analyses with observations on histomorphometric and haemodynamic components across different segments of the aortic circumference is promising in the unravelling of the complex mechanisms underlying BAV aortopathy.
The overarching aim of this study was to propose an approach of aortic segmentation to generate new knowledge on the mechanisms underlying aortic dilation in BAV patients, derived from comparisons of the most likely vs. the least likely aortic segments to be involved. The specific objectives were: (a) to identify and validate changes in miRNA and protein expression between dilated and non-dilated aortic segments; (b) to bioinformatically identify the association between dysregulated miRNAs and proteins and the wider molecular pathways they could control; (c) to measure the amount and thickness of elastin in the same aortic segments as a marker of adverse remodelling due to aortopathy; and (d) to measure WSS in the AAo and elucidate links between dysregulated miRNAs and WSS levels.
2. Materials and Methods
2.1. Patient Population
Patients scheduled for aortic replacement (i.e., Ross procedure, interposition aortic graft/bypass, aortic valve sparing, Ozaki procedure, and elephant trunk/whole arch replacement) were identified for biopsy collection based on their valve morphology (i.e., bicuspid) and invited for a pre-surgical research cardiovascular magnetic resonance (CMR) scan. The decision for aortic replacement followed ACC/AHA Guidelines 2020 [11]. Exclusion criteria are reported in the Appendix A. Informed consent for the research use of images and the use of waste aortic tissue collected during surgery was obtained from the participating patients. The study received Health Research Authority approval following the Research Ethics Committee (REC) review (REC references: 17/NI/0147, 15/LO/1064). The investigation of human tissue conforms to the principles outlined in the Declaration of Helsinki.
2.2. Data Collection
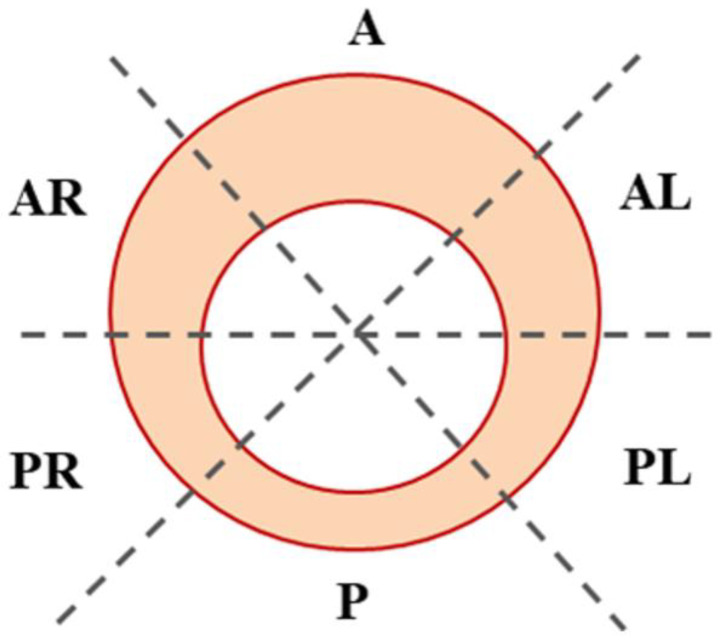
Aortic segments were collected at the time of surgery and cut into six segments (Figure 1), namely the anterior (A), anterior-right (AR), posterior-right (PR), posterior (P), posterior-left (PL), and anterior-left (AL). Each segment was further divided into three pieces to be stored as follows: (i) one piece in 500 μL RNA later for miRNA analysis, stored at −80 °C; (ii) a second piece flash-frozen for proteomic analysis, stored at −80 °C; and (iii) a third piece in 4 mL of 0.1M phosphate-buffered saline (PBS) to be subsequently fixed in a 4% formaldehyde solution and embedded in paraffin wax for histomorphometric analysis.
Figure 1.
Map of the ascending aorta segmentation. Six segments were cut for each aortic sample, i.e., anterior (A), anterior-right (AR), posterior-right (PR), posterior (P), posterior-left (PL), and anterior-left (AL), and preserved for analyses.
When possible and when patients consented to the additional imaging, research CMR scans were performed at 3T (Magnetom Skyra, Siemens Healthineers, Erlangen, Germany), including an aortic 4D flow sequence for haemodynamic assessment.
Demographic (i.e., sex, age at time of study, and body surface area (BSA)) and clinical data were collected from the patients’ clinical records (see Supplementary Methods).
2.3. Molecular Analyses
2.3.1. Next-Generation RNA Sequencing and Automated miRCURY LNA miRNA PCR Assay
Aortic tissue (20–30 mg) preserved in RNA was later homogenised in 700 μL QIAzol Lysis Reagent with 2.8 mm ceramic beads, using a Bertin homogeniser at 4 °C. An MiRNeasy Mini Kit was used for total miRNA extraction (No. 217004, Qiagen, Hilden, Germany), which was performed for n = 8 patients (Table 1: P2, 3, 6, 7, 8, 9, 14, and 15). As per clinical assessment, the anterior-right wall is dilated in the case of asymmetric BAV dilation; thus, two opposite segments per patient were selected, i.e., AR (side of dilation) vs. PL (diametrically opposite anatomical side). These patients were selected based on the presence of asymmetric (AR) dilation, no severe mix valve disease, and no severe calcification, and an equal number of males and females were selected to avoid gender bias. The RNA extract was sent to Qiagen for NGS analysis and selected markers were then validated by an automated miRCURY LNA miRNA PCR assay (Qiagen) [12] by expanding the initial cohort to n = 15 patients, including an available n = 3 patients with overall aortic dilation (rather than one-sided, asymmetrical) for comparison. Additional patients are also listed in Table 1. This analysis was performed on all 6 aortic segments per patient to explore the full spectrum of anatomical changes; therefore, a combination of adjacent segments, i.e., A + AR vs. P + PL, was also included to validate results on the most involved vs. the least involved segments and to extend the observations, taking into account possible human error in segmentation. The miRNA data analyses in the Supplementary Materials show the calculations used.
Table 1.
Patient population characteristics.
| Patient | Sex | Age at the Date of Study (yrs) | BSA (m2) | Type of Surgery | Type of Dilation | Degree of Dilation (echo mm) | Valve Phenotype | Aortic Regurgitation Severity | Aortic Stenosis Severity | Presence of Calcification | NGS, Proteomics, Histology | miRCURY LNA Assay | Haemodynamic Data |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P2 | F | 69 | 1.86 | AVR, AAo, AF | Anterior-Right | SoV:42 AAo:49 | RL | Moderate | None | None | ✓ | ✓ | ✓ |
| P3 | M | 68 | 1.98 | AVR, AAo | Anterior-Right | SoV:38 AAo:42 | RNC | None | Severe | Yes | ✓ | ✓ | ✓ |
| P6 | M | 58 | 2.18 | AVR, AAo | Anterior-Right | SoV:33 AAo:40 | RL | None | Moderate | Yes | ✓ | ✓ | |
| P7 | F | 66 | 1.84 | AVR, Arch | Anterior-Right | SoV:23 AAo:44 | RNC | Mild | Mild | None | ✓ | ✓ | |
| P8 | M | 59 | 1.88 | AVR, AAo/Root | Anterior-Right | SoV:44 AAo:41 | RL | None | Severe | Yes | ✓ | ✓ | |
| P9 | M | 65 | 2.35 | AVR, AAo, CAN | Anterior-Right | SoV:44 AAo:49 | RL | Moderate | None | None | ✓ | ✓ | |
| P14 | F | 57 | 1.55 | Ross | Anterior-Right | SoV:31 AAo:36 | RL | None | Severe | None | ✓ | ✓ | ✓ |
| P15 | F | 30 | 1.65 | Ozaki, AAo | Anterior-Right | SoV:37 AAo:37 | RL | Moderate | Mild | None | ✓ | ✓ | ✓ |
| P1 | M | 55 | 2.19 | AVR, AAo, AF, LAA | Overall | SoV:38 AAo:46 | RNC | Moderate | Moderate | None | ✓ | ✓ | |
| P4 | F | 32 | 1.9 | Ross | Anterior-Right | SoV:39 AAo:38 | RNC | Severe | Severe | Yes | ✓ | ||
| P5 | M | 34 | 1.92 | AVR, AAo | Anterior-Right | SoV:34 AAo:45 | RNC | Mild | Severe | Yes | ✓ | ||
| P10 | M | 70 | 1.78 | AVR, AAo | Overall | SoV:40 AAo:46 | RL | Mild | Severe | Yes | ✓ | ||
| P11 | M | 75 | 2.18 | Root | Anterior-Right | SoV:42 AAo:51 | RNC | Moderate | Moderate | Yes | ✓ | ||
| P12 | M | 56 | 1.99 | AVR, AAo | Anterior-Right | SoV:36 AAo:45 | N/A | Moderate | Severe | Yes | ✓ | ||
| P13 | M | 58 | 2.19 | Ozaki, AAo | Overall | SoV:46 AAo:53 | RNC | None | Severe | Yes | ✓ | ✓ |
Baseline characteristics for patients (P), including data collection. AAo = ascending aorta; AF = atrial fibrillation; AVR = aortic valve replacement; CAN = coronary artery bypass; LAA = left atrial appendage; N/A = non-applicable; RL = right-left; RNC = right-non-coronary; SoV = sinus of Valsalva.
2.3.2. Mass-Spectrometry Analysis
Flash-frozen aortic tissue (20–30 mg) from the same n = 8 patients chosen for NGS (two segments per patient, i.e., AR vs. PL) was homogenised, RIPA-buffered (i.e., using gentleMACS Dissociator), and centrifuged to collect the supernatant. The homogenised samples were shaken on ice for 30 min and then centrifuged at 13,000× g for 10 min at 4 °C. The aliquoted supernatant was sent to the University of Bristol Proteomics Facility for tandem mass spectrometric analysis.
2.4. Bioinformatic Analysis
Using miRWalk suite (v.2.0, University of Heidelberg, Heidelberg, Germany,) and selecting the TargetScan and miRDB databases, as well as a confidence score of 0.90, target prediction was carried out for the validated miRNA markers (which were significantly dysregulated between the anatomically opposite aortic segments, i.e., AR vs. PL, considering adjacent segments, thus comparing A + AR vs. P + PL). The predicted targets were then compared to significantly dysregulated proteins derived from two analyses of the same patients. Downregulated gene targets were selected for upregulated miRNAs, and vice versa. Gene ontology and protein pathways were extracted from Webgestalt for gene targets with >0.5 and <−0.5 log2 (fold change).
2.5. Histomorphometric Analysis
Paraffin-embedded samples for the same n = 8 patients (two segments per patient, i.e., AR vs. PL) were cut on a microtome (5 μm thickness) and stained with elastin van Gieson (EVG) using an autostainer machine. Microphotograph imaging and analyses were performed blinded to the clinical diagnosis. Two serial sections of each tissue were prepared to obtain technical duplicates per tissue sample, and 8 arbitrary fields were imaged per section at 40 and 200× magnifications to analyse both the thickness of the elastic layer and the content of the elastic fibres, in the intima and the media layers of the aortic wall, respectively. The contents of the elastic fibres were quantified using Image J software (National Institutes of Health, Bethesda, MD, USA). The thickness was analysed by averaging the length of 5 transversal sites of the area comprised of elastic fibres.
2.6. Haemodynamic Analysis
Haemodynamic parameters were analysed using Caas MR 4D flow software (v5.2.1, Pie Medical Imaging, Maastricht, The Netherlands). The magnitude and phase x, y, z components were selected for analysis. The aortic centreline was automatically set, as was a threshold for the aortic volumes, which were manually adjusted to fit the centreline at the point of the highest flow through the aorta in systole. The plane for the analysis was set at the level of the mid-AAo (i.e., the same level of surgical biopsy). Wall shear stress on the aortic wall and flow streamlines, velocity, and flow displacement in the aorta were measured. The values of WSS were derived as 90 vectors on the analysis plane and then combined into six regions, corresponding to the six surgical segments.
2.7. Statistical Analysis
Statistical analysis (R, Vienna, Austria) was carried out to compare the level of miRNAs, proteins, elastin (% and thickness), and WSS between the dilated vs. the non-dilated segments of the aorta. Non-parametric paired analysis was carried out (Wilcoxon signed-rank test). Data were reported as medians and interquartile ranges (IQRs) or proportions, as appropriate. Where the associations between variables were assessed, linear regression analysis was performed. A p-value < 0.05 indicated statistical significance.
3. Results
3.1. Patient Population
The baseline characteristics of all the recruited patients (n = 15) are reported in Table 1. The patients’ age was 58 (56, 67) years (range: 30–75 years), 67% were male, and they had a BSA of 1.92 (1.85, 2.18) m2. The patients predominantly had anterior-right AAo dilation (80 vs. 20% overall dilated). The patients had varying degrees of aortic regurgitation (47% moderate/severe) and aortic stenosis (73% moderate/severe). Molecular data were collected in all patients, of which n = 8 were collected for full molecular characterisation. Haemodynamic data were available in n = 6 patients aged 58 (56, 66) years, 50% of whom were male, with a BSA of 1.92 (1.70, 2.13) m2 (Table 1).
3.2. Molecular Analysis
In the NGS analysis (n = 8), a paired comparison between AR and PL segments revealed 92 differentially expressed miRNAs based on a p-value < 0.05 (see Supplementary Materials Table S1), of which 11 with an FDR < 0.1 were chosen as targets for qPCR-based validation (Table 2). Highly sensitive quantification by miRCURY LNA assays in the whole cohort (n = 15) were performed to confirm differentially expressed miRNA candidates. Table 3 reports the results for the detectable miRNAs from the analysis. A paired comparison between the AR vs. the PL and the A + AR vs. the P + PL segments highlighted five differentially expressed miRNAs (miR-128-3p, miR-210-3p, miR-150-5p, miR-199b-5p, and miR-21-5p, see Table 3). Additionally, the results for the overall-dilated patient shown in Table 3 indicate no miRNA difference from the segmental comparison, compared to the one-side-dilated patients.
Table 2.
Next-generation sequencing chosen targets for miRCURY LNA assay validation.
| MiRNA Name | Dilated vs. Non-Dilated—FC | Dilated vs. Non-Dilated—Log2FC | Dilated vs. Non-Dilated—p-Value | Dilated vs. Non-Dilated—FDR p-Value |
|---|---|---|---|---|
| hsa-miR-1247-5p | 3.5892 | 1.8437 | <0.0001 | 0.0003 |
| hsa-miR-21-5p | 0.6667 | −0.5849 | <0.0001 | 0.0003 |
| hsa-miR-483-3p | 2.1387 | 1.0967 | <0.0001 | 0.0024 |
| hsa-miR-211-5p | 0.3308 | −1.5960 | <0.0001 | 0.0095 |
| hsa-miR-21-3p | 0.6068 | −0.7206 | <0.0001 | 0.0095 |
| hsa-miR-199b-5p | 2.1820 | 1.1256 | <0.0001 | 0.0095 |
| hsa-miR-128-3p | 0.6975 | −0.5197 | <0.0001 | 0.0095 |
| hsa-miR-150-5p | 1.8983 | 0.9247 | 0.0002 | 0.0431 |
| hsa-miR-488-3p | 2.7460 | 1.4573 | 0.0003 | 0.0615 |
| hsa-miR-375-3p | 2.5455 | 1.3480 | 0.0005 | 0.0872 |
| hsa-miR-210-3p | 0.6465 | −0.6294 | 0.0006 | 0.0996 |
FC = fold change; FDR = false discovery rate.
Table 3.
miRCURY LNA validated targets from segmental analysis.
| AR vs. PL | A + AR vs. P + PL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Anterior-Right (N = 12) | Overall (N = 3) | Anterior-Right (N = 12) | Overall (N = 3) | |||||||
| MiRNA Targets NGS | p-Value | FC | Log2FC | FDR | p-Value | p-Value | FC | Log2FC | FDR | p-Value |
| miR-1247-5p | 0.110 | 1.647 | 0.720 | 0.154 | 0.750 | 0.470 | 1.291 | 0.368 | 0.470 | 0.470 |
| miR-21-5p | 0.339 | 0.919 | −0.121 | 0.396 | 0.250 | 0.034 | 0.835 | −0.261 | 0.060 | 0.250 |
| miR-21-3p | 0.791 | 1.030 | 0.043 | 0.791 | 0.500 | 0.380 | 0.869 | −0.202 | 0.444 | 0.500 |
| miR-199b-5p | 0.012 | 1.354 | 0.437 | 0.021 | 0.500 | 0.233 | 1.173 | 0.230 | 0.327 | 1.000 |
| miR-128-3p | 0.012 | 0.724 | −0.466 | 0.021 | 0.250 | <0.001 | 0.665 | −0.588 | 0.002 | 0.750 |
| miR-150-5p | 0.007 | 1.736 | 0.796 | 0.021 | 0.750 | 0.012 | 1.418 | 0.504 | 0.028 | 1.000 |
| miR-210-3p | 0.009 | 0.773 | −0.371 | 0.021 | 0.250 | <0.001 | 0.722 | −0.471 | 0.002 | 0.750 |
Reporting two comparisons of aortic segments (anterior-right vs. posterior-left, AR vs. PL, and a more extensive comparison combining adjacent segments, anterior + anterior-right vs. posterior + posterior-left, A + AR vs. P + PL) for one-side- and overall-dilated patients. In bold, p-value < 0.05 and FDR < 0.1, which were used as threshold of significance.
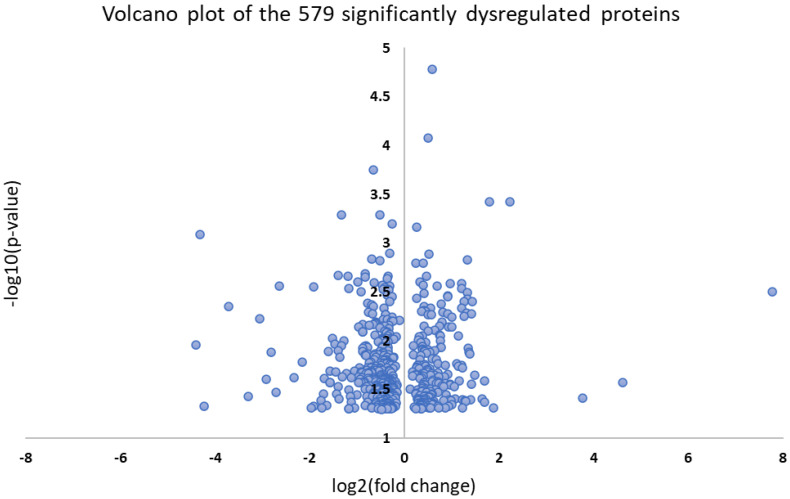
Proteomic analysis in the same cohort (n = 8) revealed 579 significantly dysregulated proteins based on a p-value < 0.05 in the AR vs. PL segment comparison (see Supplementary Material Table S2).
3.3. Bioinformatic Analysis
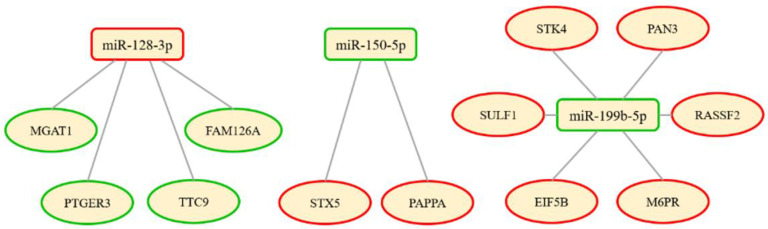
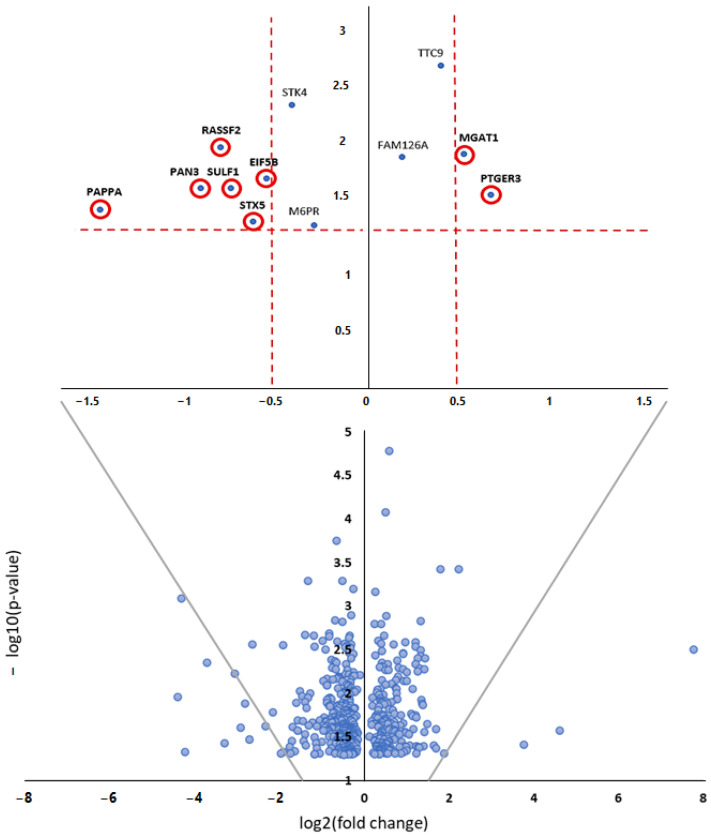
Bioinformatic analysis linking the above five miRNAs (validated by miRCURY LNA assays) and significantly dysregulated proteins revealed a potential network link between three miRNAs (miR-128-3p, miR-150-5p, miR-199b-5p) and twelve proteins (Figure 2). Gene ontology and relevant protein pathways were identified for the eight most differentially expressed proteins, as shown on the volcano plot (Figure 3; Table 4 and Table 5), based on −0.5 > log2FC > 0.5 and −log10(p-value) > 1.3.
Figure 2.
MiRNA and protein network link. The networks indicate predicted interactions between validated miRNAs and the significantly dysregulated proteins. Green = upregulation; red = downregulation.
Figure 3.
Volcano plot for the selection of the differentially expressed proteins. The 579 proteins with a p-value < 0.05 identified from the proteomic analysis (see Supplementary Material) are displayed. Extracted and plotted alone on top of the plot are the significantly dysregulated proteins (highlighted in red) as determined by cut-off values of −0.5 > log2FC > 0.5 and −log10(p-value) > 1.3. Proteins with log2FC < 0 are downregulated in dilated vs. non-dilated segments, while proteins with log2FC > 0 are upregulated.
Table 4.
Gene ontology analysis for the eight most significantly dysregulated proteins.
| Gene Target | Description | Functional Database | p-Value |
|---|---|---|---|
| MGAT1 | peptidyl-asparagine modification | Biological_Process | 0.0139 |
| PAPPA | response to gonadotropin | Biological_Process | 0.0144 |
| MGAT1; SULF1 | glycoprotein metabolic process | Biological_Process | 0.0150 |
| SULF1 | vascular-endothelial growth-factor production | Biological_Process | 0.0169 |
| PTGER3; SULF1 | muscle system process | Biological_Process | 0.0176 |
| MGAT1 | nucleotide sugar metabolic process | Biological_Process | 0.0184 |
| MGAT1 | amino sugar metabolic process | Biological_Process | 0.0188 |
| SULF1 | animal organ formation | Biological_Process | 0.0301 |
| SULF1 | nerve development | Biological_Process | 0.0379 |
| SULF1 | vascular-endothelial growth-factor receptor-signalling pathway | Biological_Process | 0.0427 |
| PTGER3 | phospholipase C-activating G-protein-coupled receptor-signalling pathway | Biological_Process | 0.0470 |
| STX5 | organelle disassembly | Biological_Process | 0.0470 |
| MGAT1 | catalytic activity, acting on a glycoprotein | Molecular_Function | 0.0114 |
| STX5 | SNAP receptor activity | Molecular_Function | 0.0149 |
| MGAT1 | manganese ion binding | Molecular_Function | 0.0301 |
| EIF5B | translation factor activity, RNA binding | Molecular_Function | 0.0427 |
| STX5 | SNARE binding | Molecular_Function | 0.0500 |
| STX5 | SNARE complex | Cellular_Component | 0.0233 |
Bioinformatic tools were used to extract gene ontology analysis for the eight most differentially expressed proteins associated with the three miRNA targets, (p-value < 0.05).
Table 5.
Pathway analysis for the eight most significantly dysregulated proteins.
| Gene Target | Description | p-Value |
|---|---|---|
| MGAT1 | N-glycan trimming and elongation in the cis-Golgi | 0.0027 |
| MGAT1; STX5 | transport to the Golgi and subsequent modification | 0.0039 |
| PTGER3 | prostanoid ligand receptors | 0.0048 |
| PTGER3 | eicosanoid ligand-binding receptors | 0.0080 |
| PTGER3 | small ligand GPCRs | 0.0102 |
| MGAT1; STX5 | asparagine N-linked glycosylation | 0.0104 |
| PTGER3 | relationship between inflammation, COX-2, and EGFR | 0.0133 |
| PAN3 | deadenylation of mRNA | 0.0133 |
| RASSF2 | Hippo signalling pathway | 0.0155 |
| STX5 | Cargo concentration in the ER | 0.0176 |
| STX5 | SNARE interactions in vesicular transport | 0.0181 |
| STX5 | intra-Golgi traffic | 0.0234 |
| PTGER3 | prostaglandin synthesis and regulation | 0.0239 |
| MGAT1 | N-glycan biosynthesis | 0.0260 |
| EIF5B | translation factors | 0.0286 |
| PTGER3 | regulation of lipolysis in adipocytes | 0.0286 |
| PAN3 | deadenylation-dependent mRNA decay | 0.0297 |
| STX5 | COPII-mediated vesicle transport | 0.0359 |
| PAN3 | RNA degradation | 0.0416 |
Bioinformatic tools were used to extract pathway analysis for the eight most differentially expressed proteins associated with the three miRNA targets (p-value < 0.05).
3.4. Histomorphometric Analysis
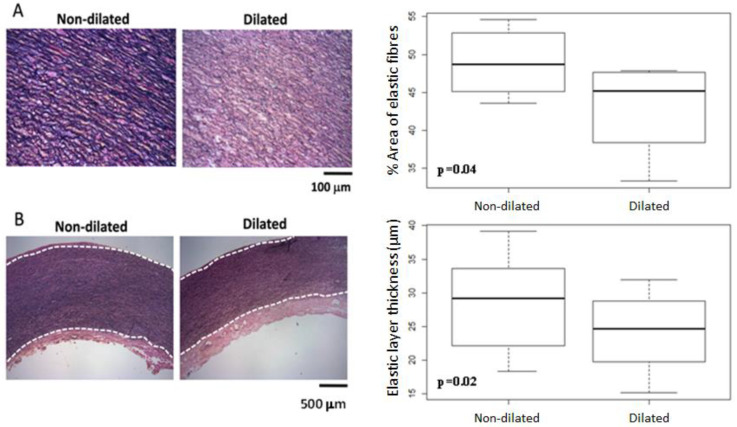
Histomorphometric analysis (same cohort, n = 8) showed that the dilated aortic segments (i.e., AR) had decreased elastic fibres [average % area of elastic fibres: AR = 45.21 (39.71, 47.64) vs. PL = 48.74 (45.22, 52.64), p = 0.039] and a thinner elastic layer [AR = 24.71 (20.26, 28.69) vs. PL = 29.21 (22.35, 33.36) um, p = 0.016] (Figure 4).
Figure 4.
Quantitative histomorphometry of the elastic component. (A) Representative images (200× magnification) for non-dilated and dilated segments, and differences (% area) of elastic fibres showing decreased elastic fibres in dilated segments (p = 0.04); (B): example of measurements of elastic layer (40× magnification) and differences in thickness showing a thinner layer in dilated segments (p = 0.02).
3.5. Haemodynamic Analysis
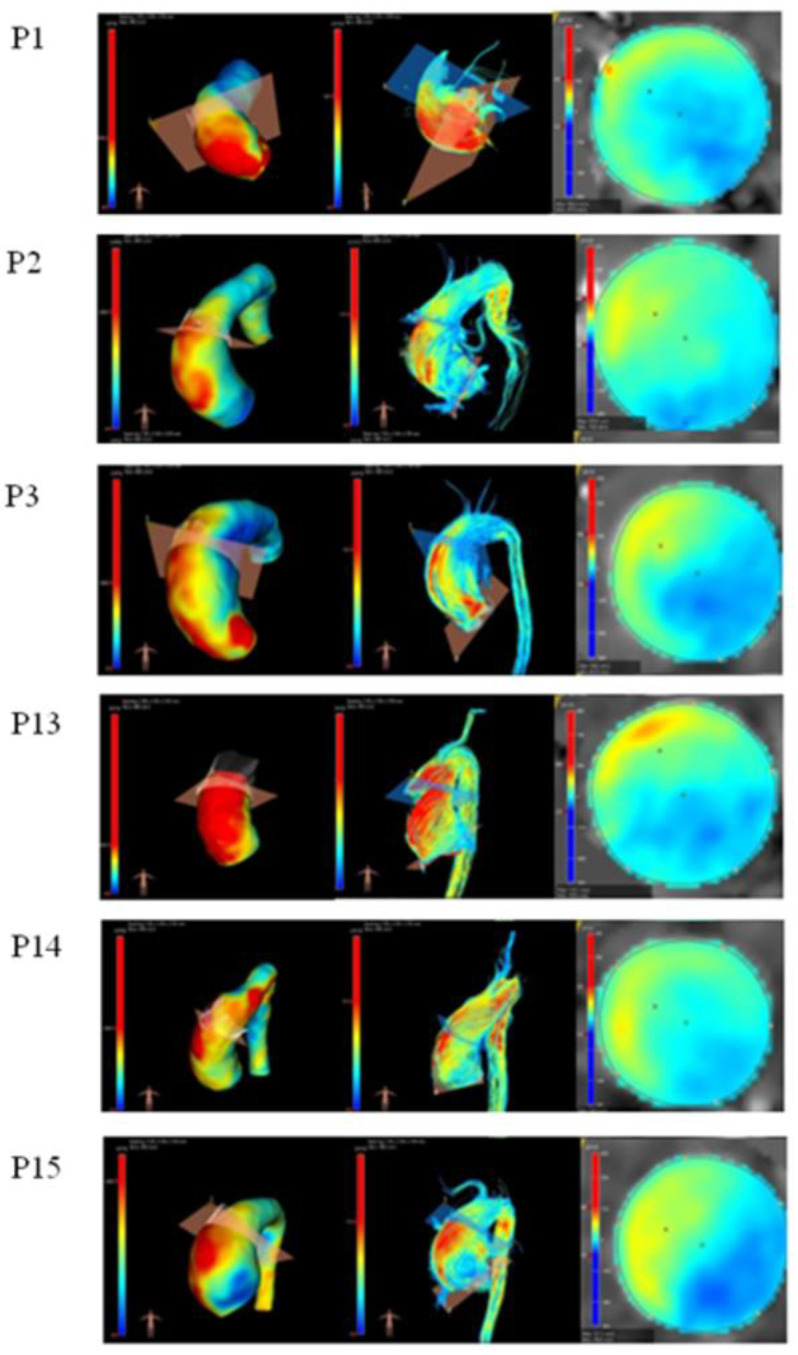
Where available (n = 6), a visual assessment of WSS distribution, velocity streamlines, and flow-velocity distribution (Figure 5) revealed the following: (i) an increased WSS and velocity at the anterior-right side in patients with anterior-right dilation, with helical right-handed flow and eccentric flow velocity towards the anterior-right wall; and (ii) a further increased WSS on the AAo wall in patients with overall dilation, helical aortic flow, and a higher flow velocity.
Figure 5.
4D flow analysis in patients with both molecular and haemodynamic data. First column: wall shear stress (WSS, range = 0–2000 mPAŸs); second column: velocity streamlines (range = −122–262 cm/s); third column: flow displacement.
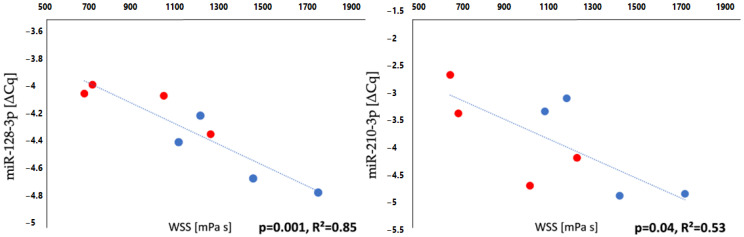
Interestingly, a correlation between increased WSS and decreased miRNA expression was found for miR-128-3p and miR-210-3p (Figure 6), with these miRNAs identified as being downregulated on the side of dilation (Table 3, see log2FC). In patients with both histomorphometric and haemodynamic data (n = 4), the dilated segments (i.e., those with less elastic fibres and a thinner layer) also exhibited double WSS values on average, compared to the non-dilated segments [AR vs. PL: 1523.43 (1141.55, 1934.00) vs. 786.73 (514.25, 1005.93) mPa s].
Figure 6.
Correlation between WSS and miRNA. Relationship between WSS and miRNA expression in n = 4 patients with anterior-right dilation. Red = average of posterior segments; blue = average of anterior segments. Both graphs show less miRNA expression and higher WSS in the dilated segments.
4. Discussion
In order to study the complex nature of BAV aortopathy, a segmental approach can be beneficial to uncover detailed local information about changes in the aortic wall. We conducted a prospective study, collecting tissue biopsies for (a) NGS and miRCURY LNA PCR for the identification and validation of miRNA markers, (b) proteomic analysis, (c) bioinformatic network analysis, (d) histomorphometric analysis of elastic fibres, and (e) preliminary 4D flow CMR data for haemodynamic assessment. The NGS and miRCURY LNA PCR validation showed that five miRNAs had significant differences between the anterior-right and posterior-left sides of the aorta, i.e., miR-128-3p, miR-210-3p, miR-150-5p, miR-199b-5p, and miR-21-5p. Our bioinformatic network analysis showed that, amongst those identified by proteomics, eight gene targets, i.e., MGAT1, PTGER3, STX5, PAPPA, SULF1, EIF5B, RASSF2, and PAN3, are linked to miR-128-3p, miR-150-5p, and miR-199b-5p. Based on a strict cut-off of ±0.5 log2FC (also considering the FDR), two miRNAs were significantly dysregulated, i.e., miR-128-3p (downregulated) and miR-150-5p (upregulated), while miR-210-3p (downregulated) and miR-199b-5p (upregulated) could be considered borderline.
MiR-128-3p has been characterised in the literature as a novel regulator of vascular smooth-muscle-cell (SMC) phenotypic switches, regulated by epigenetic modifications under stress [13]. While SMCs have been abundantly studied in the aneurysmal process, the role of miR-128-3 on SMCs should be further exploited for therapeutics. Based on our network analysis, miR-128-3p is linked to PTGER3 gene expression, which in turn is associated with prostanoid ligand receptors (p = 0.005), eicosanoid ligand-binding receptors (p = 0.008), the relationship between inflammation, COX-2 and EGFR (p = 0.013), and prostaglandin synthesis and regulation (p = 0.024). This shows its involvement in arachidonic acid metabolic pathways, activated by inflammation, as previously reported in aortic aneurysm formation in Marfan syndrome [14], which is in turn suggested to share similarities with BAV aortopathy [15]. Additionally, miR-150-5p is referred to in the literature as being involved in the regulation of genes with a role in eicosanoid synthesis, such as PTGIS and the MMP/TIMP pathway, which are implicated in the pathogenesis of abdominal aortic aneurysm (AAA) [16]. There is also speculation that miR-21 regulates the PTGS2/PGE2 pathway in the AAA context [16]. All this may imply an interconnected pathway of the above miRNAs and prostaglandin biosynthesis in the aneurysmal process that should be further studied. Furthermore, miR-150-5p and miR-21-5p have been previously mentioned as both plasma and tissue biomarkers of AAA [17], and the role of miR-21-5p in BAV aortopathy has been well-established in the literature [18,19,20,21].
Based on a study of thoracic aneurysmatic aortopathy in Marfan syndrome, miR-199b-5p was significantly dysregulated and associated with extracellular matrix (ECM)–receptor interaction and the Hippo signalling pathway [22]. Based on our network analysis, miR-199b-5p might be associated with Hippo signalling through RASSF2 (p = 0.015) [23]. The Hippo signalling pathway is involved in ECM production and degradation, as well as in the growth, death, and migration of vascular SMCs and endothelial cells, which contribute to vascular remodelling in cardiovascular diseases, such as aortic aneurysms, pulmonary hypertension, and atherosclerosis, and in cardiovascular processes, such as restenosis and angiogenesis [24]. Additionally, our network analysis showed miR-199b-5p was associated with SULF1, which has in turn been associated with vascular-endothelial growth-factor (VEGF) production and receptor-signalling pathways (p = 0.017 and p = 0.043, respectively) [25,26]. VEGF has a central role in promoting angiogenesis as it contributes to the proliferation, survival, and migration of endothelial cells and their progenitors, whilst also mediating the migration of circulating inflammatory monocytes and tissue macrophage precursors to target organs [27]. Additionally, pathway cross-talking is supported by the literature (e.g., TNF-a and RASSF–Hippo signalling, and TGF-β1 and VEGF, together with the Notch signalling pathways involved in the regulation of vascular morphogenesis). Additionally, BAV aortopathy may reflect the pathophysiological implications of BAV disease, thereby involving multiple integrated cell-signalling pathways [23,28]. These mechanisms should be further examined through in vitro cell-culturing studies. Additionally, because flow-sensitive miRNAs, known as mechanomiRs, can regulate endothelial gene expression and, consequently, endothelial dysfunction [29], the above network involving VEGF should be further examined through the relationship of miRNAs influenced by blood-flow changes.
Whether the pathogenesis of BAV aortopathy is related to genetic or haemodynamic factors is still a cause for debate [30]. A “genetic theory” supports that the brittleness of the aortic wall is caused by the developmental abnormalities of the wall and the aortic valve, due to heritable gene mutations [31,32]. In contrast, a “haemodynamic theory” focuses on abnormal wall shear stress (WSS) progressively acting on the aortic wall, with emerging evidence showing that even a ‘clinically normal’ BAV is associated with abnormal flow patterns and asymmetrically increased WSS in the proximal aorta [31,33]. Changes in WSS in BAV have been studied using CMR [34,35,36]. Our results confirmed that BAV patients had right-handed helical and more turbulent, eccentric blood flow (i.e., flow displacement from the centre towards the side of the aortic wall with increased velocity). These results concur with the literature, as BAV aortas, compared to TAV, show increased helicity in the mid-AAo and increased flow angle, associated with a rotating jet along the aortic wall that eventually leads to dilation [37]. It has also been reported that right-handed helical flow was the most common flow abnormality in BAV, associated with large AAo diameters and a high rotational flow and systolic flow angle, irrespective of valve morphotype [37]. Furthermore, BAV cusp-fusion morphology is also reported in the literature as a factor altering flow patterns, WSS, and the expression of aortopathy [38]. This could not be examined in this study due to the small available sample, but it should be further examined in larger cohort studies considering segmental alterations.
One study examined miRNA differences between the convexity and concavity in mildly aortic-dilated BAV patients, TAV patients, and non-dilated heart transplant donors [10]. Here, aortic RNA was analysed through PCR arrays, profiling the expression of 84 microRNAs involved in cardiovascular disease, target prediction, and pathway-enrichment analysis. The study showed that miRNA dysregulation, potentially affecting mechanotransduction pathways, exhibits a higher prevalence in BAV convexity, with reference to TGF-β1, Hippo, and PI3K/Akt/FoxO. BAV convexity vs. BAV concavity showed, among others, a significant upregulation of hsa-miR-150-5p and hsa-miR-21-5p, and a significant downregulation of hsa-miR-199a-5p, as well as a significant involvement in Hippo signalling pathways, all in agreement with our results. However, the direction of miRNA dysregulation (i.e., either upregulation or downregulation) can be controversial in the literature, as in the case of miR-21 [39]. Additionally, a pathway-enrichment analysis from an miRNome-profiling study in BAV aortopathy identified a link with the Hippo signalling pathway, ErbB signalling, TGF-beta signalling, and focal adhesion, verifying the involvement of these pathways in the disease and their link with miRNA dysregulation [40].
In our population, the highest miRNA, WSS, and elastin dysregulation (decreased levels of elastic fibres and a thinner layer) was observed at the dilated anterior-right side of the aortic wall, suggesting a correspondence between molecular and haemodynamic changes. Whether these miRNAs are directly involved in the aortic wall pathogenesis of BAV disease, or whether they are a consequence of the increased aortic shear stress generated by the anomalous aortic flow caused by BAV, should be studied in larger cohorts. A key study by Guzzardi et al. [8] reported the relationship between altered WSS and regional aortic tissue remodelling (i.e., elastic fibre quantification, fibre thickness, and fibre distance, and MMP and TGF-β concentration) in BAV aortopathy in regions of the AAo (adjacent vs. distal to the right pulmonary artery) in a similarly sized group (n = 20). The presence of thin, fragmented elastic fibres, reduced fibrillin-1 content, decreased type I and III collagen, and MMP/TIMP ratio dysregulation have been well-studied in the literature, suggesting an inherent developmental defect underlying the dysregulation of the ECM in the aortic medial layer as a main cause of aortic dilatation [30,41]. Additionally, a recent study [9] correlated SMC loss, elastic degradation, and aortic wall strength to WSS, showing an increased dysregulation on the outer curve of the AAo. However, it has not yet been explained whether the WSS-mediated ECM dysregulation is a consequence of the dilatation itself, rather than a determinant or risk factor. Our detailed, segment-by-segment analysis advances some of these observations within an overall framework of exploring the relationship between altered haemodynamics and aortic tissue remodelling. More studies, such as the one by Zhang et al. [42] which shows that the inhibition of miRNA-29 enhances elastin levels in cells, ought to be carried out to elucidate the interaction between mechanomiRs and elastin and demonstrate that miRNAs can be used in therapeutics and the discovery of novel pharmacological targets [43].
Further studies are needed to prove that vascular remodelling could be a biomarker in clinical imaging and miRNAs could be the effectors of flow-mediated changes. Here, we have proposed a specific segmental comparison of A + AR vs. P + PL (segments with the most vs. the least molecular dysregulation, as derived from our analyses) and have shown segmental correspondence between miRNAs and WSS. We have also provided additional information on proteins and elastin changes, suggesting miRNAs as mechanistic mediators of the vascular changes induced by the altered flow in BAV. However, we do not know whether these miRNA changes have a transcriptional or post-transcriptional origin. It would therefore be interesting to understand this through a follow-up study, as it could provide information on cellular processes involved in the dysregulation and, ultimately, on BAV aortopathy and its genetic vs. WSS drivers.
From a methodological standpoint, we note extending observations from AR vs. PL segments to A + AR vs. P + PL was based on (a) ensuring to cover the full spectrum of the dilated area, and (b) accounting for potential human error in the process of segmentation in the surgical theatre, although the latter was carefully guided by and discussed with surgeons. The study, due to the prospective design, has the disadvantage of not all the recruited patients having a full molecular and haemodynamic dataset. The lack of these data was mainly caused either by issues in collecting the biopsies at the time of surgery or patients not consenting to the additional CMR scan.
5. Conclusions
This study merged segmental molecular and histological data, as well as clinical imaging, shedding light on local changes across the aortic wall circumference in BAV patients. MiRNA dysregulation linked to inflammatory pathways, changes in aortic wall remodelling, and WSS were concomitantly observed on the anterior-right wall in BAV patients, emphasizing the requirement to further study regional changes in the aorta.
Acknowledgments
The authors acknowledge the generous support of the British Heart Foundation, the NIHR Bristol Biomedical Research Centre, and Above & Beyond.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cells11233721/s1, Table S1: Whole list of miRNAs from NGS analysis; Table S2: Whole list of proteins from Tandem Mass Spectrometer analysis. Table S3 STORBE Statement—Checklist of items that should be included in reports of cohort studies.
Appendix A
Patient Population
This is a single-centre prospective study. Patients scheduled for aortic replacement (i.e., Ross procedure, interposition aortic graft/bypass, aortic valve sparing, Ozaki procedure, and elephant trunk/whole arch replacement) were identified from the surgical list for biopsy collection based on their valve morphology (i.e., bicuspid), while patients with previous AVR or aortic surgical intervention and syndromes (e.g., Turner) or other connective tissue disorders (e.g., Marfan) were excluded. As patients were also invited for a CMR scan, additional exclusion criteria included the following: claustrophobia; permanent metal fragments or body implants; pregnancy; and severe kidney dysfunction (defined as an estimated glomerular filtration rate <60 mL/min/1.73 m2), in light of the administration of intravenous contrast in the presence of a cardiologist. Patients were invited by telephone and were posted the patient information sheet in advance. They were invited for a pre-surgical research CMR scan at our centre and consented to both the research use of the images and the use of aortic tissue waste collected during surgery. The study received Health Research Authority approval following the Research Ethics Committee (REC) review (REC reference: 17/NI/0147).
Data Collection
The research CMR scans were performed at 3T (Magnetom Skyra, Siemens Healthineers, Erlangen, Germany) and mainly included an aortic 4D flow sequence for haemodynamic assessment. (Example of sequence settings: 4D Flow venc 400_Ipat2_asMM_native_P, TR: 38.6, TE: 2.1.)
At the time of surgery, aortic segments were collected and cut in the surgical theatre, dividing the ascending aorta into six segments, i.e., anterior (A), anterior-right (AR), posterior-right (PR), posterior (P), posterior-left (PL), and anterior-left (AL) (see Central Illustration Figure). Each segment was further divided into three pieces. The first piece of each segment was collected in a 1.5 mL RNase-free Eppendorf tube and later in 500 μL RNA for miRNA analysis. It was kept at 4 °C overnight before being discarded the next day and stored at −80 °C. The second piece was flash-frozen in 1.6 mL CryoPure tubes in liquid nitrogen and kept at −80 °C for proteomic analysis. The third piece was collected in a 7 mL bijou tube in 4 mL of 0.1M phosphate-buffered saline (PBS) and transferred to a 4% formaldehyde solution for 24 h fixation, and then embedded in paraffin wax for histomorphometric analysis.
Demographic (i.e., sex, age at time of study, BSA) and clinical data were collected from clinical patient records. The clinical data included: type of surgery; type of dilation (one-sided vs. overall); degree of dilation at SoV and proximal AAo; valve phenotype (i.e., right-left (RL) or right-non-coronary (RNC), or left-non-coronary (LNC) cusps non-separation), the degree of aortic regurgitation and/or aortic stenosis severity; and presence of calcification.
MiRNA Data Analysis
Below are shown the calculations used for miRNAs to calculate (a) the normalised difference for each miRNA using the Cq-derived values from the RNA analysis. Five miRNAs were chosen from the NGS analysis, as the most stable miRNAs (i.e., has-let-7i-5p, has-miR-191-5p, has-miR-107, has-miR-let-7a-5p, and has-miR-151a-3p), to be used as normalisers in miRCURY LNA assay. Their values have been averaged for the following calculations: (b) the segmental difference using the normalised ΔCq calculated values; and the fold change for either (c) positive or (d) negative ΔΔCq.
-
(1)
ΔCq = Cq(averaged normalisers)—Cq(miRNA of interest)
-
(2)
ΔΔCq = ΔCq(dilated segment)—ΔCq(non-dilated segment)
-
(3)
If ΔΔCq is positive, then FC = 2ΔΔCq
-
(4)
If ΔΔCq is negative, then FC = −[2(−ΔΔCq)]
Whole List of miRNAs from NGS Analysis
The 92 differentially expressed miRNAs based on a p-value < 0.05 are shown below: (see Supplementary Material Table S1).
Whole List of Proteins from Tandem Mass Spectrometer Analysis
The 579 significantly dysregulated proteins based on a p-value < 0.05 are shown below (see Supplementary Material Table S2).
Figure A1.
The volcano plot of proteins.
Author Contributions
Conceptualization, C.R., C.B.-D., M.C., C.E. and G.B.; Methodology, F.S., M.C.K.H. and G.B.; Formal analysis, F.S., M.G.B., M.A., E.J. and A.C.-J.; Investigation, F.S., E.D.G., K.M., V.D.F., A.W., S.S., A.P., U.B., P.C., F.G., M.C.K.H., C.B.-D. and G.B.; Resources, M.C.; Data curation, F.S. and E.D.G.; Writing—original draft, F.S.; Writing—review & editing, M.A., A.C.-J., C.R., S.S., C.B.-D., M.C., C.E. and G.B.; Supervision, C.R., M.C., C.E. and G.B.; Funding acquisition, C.B.-D. and M.C. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Research Ethics Committee (REC references: 17/NI/0147, 15/LO/1064).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Conflicts of Interest
Chiara Bucciarelli-Ducci is the Chief Executive Officer (part-time) of the Society for Cardiovascular Magnetic Resonance. The remaining authors have nothing to disclose.
Funding Statement
This work has been generously supported by the British Heart Foundation (AA/18/1/34219, CH/17/1/32804, CH/15/1/31199, RG/20/9/35101), National Institute for Health Research (NIHR) Bristol Biomedical Research Centre (BRC), and Above & Beyond.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Verma S., Siu S.C. Aortic Dilatation in Patients with Bicuspid Aortic Valve. N. Engl. J. Med. 2014;370:1920–1929. doi: 10.1056/NEJMra1207059. [DOI] [PubMed] [Google Scholar]
- 2.Baumgartner H., De Backer J., Babu-Narayan S.V., Budts W., Chessa M., Diller G.P., Lung B., Kluin J., Lang I.M., Meijboom F., et al. 2020 ESC Guidelines for the management of adult congenital heart disease. Eur. Heart J. 2021;42:563–645. doi: 10.1093/eurheartj/ehaa554. [DOI] [PubMed] [Google Scholar]
- 3.Abdulkareem N., Smelt J., Jahangiri M. Bicuspid aortic valve aortopathy: Genetics, pathophysiology and medical therapy. Interact. Cardiovasc. Thorac. Surg. 2013;17:554–559. doi: 10.1093/icvts/ivt196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gao Y., Peng J., Ren Z., He N.Y., Li Q., Zhao X.S., Wang M.M., Wen H.Y., Tang Z.H., Jiang Z.S., et al. Functional regulatory roles of microRNAs in atherosclerosis. Clin. Chim. Acta. 2016;460:164–171. doi: 10.1016/j.cca.2016.06.044. [DOI] [PubMed] [Google Scholar]
- 5.Alajbegovic A., Holmberg J., Albinsson S. Molecular Regulation of Arterial Aneurysms: Role of Actin Dynamics and microRNAs in Vascular Smooth Muscle. Front. Physiol. 2017;8:569. doi: 10.3389/fphys.2017.00569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhou S.S., Jin J.P., Wang J.Q., Zhang Z.G., Freedman J.H., Zheng Y., Cai L. miRNAS in cardiovascular diseases: Potential biomarkers, therapeutic targets and challenges. Acta Pharmacol. Sin. 2018;39:1073–1084. doi: 10.1038/aps.2018.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mohamed J.S., Hajira A., Lopez M.A., Boriek A.M. Genome-wide Mechanosensitive MicroRNA (MechanomiR) Screen Uncovers Dysregulation of Their Regulatory Networks in the mdm Mouse Model of Muscular Dystrophy. J. Biol. Chem. 2015;290:24986–25011. doi: 10.1074/jbc.M115.659375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Guzzardi D.G., Barker A.J., van Ooij P., Malaisrie S.C., Puthumana J.J., Belke D.D., Mewhort H.E., Svystonyuk D.A., Kang S., Verma S., et al. Valve-Related Hemodynamics Mediate Human Bicuspid Aortopathy: Insights from Wall Shear Stress Mapping. J. Am. Coll. Cardiol. 2015;66:892–900. doi: 10.1016/j.jacc.2015.06.1310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kiema M., Sarin J.K., Kauhanen S.P., Torniainen J., Matikka H., Luoto E.S., Jaakkola P., Saari P., Liimatainen T., Vanninen R., et al. Wall Shear Stress Predicts Media Degeneration and Biomechanical Changes in Thoracic Aorta. Front. Physiol. 2022;13:934941. doi: 10.3389/fphys.2022.934941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Albinsson S., Della Corte A., Alajbegovic A., Krawczyk K.K., Bancone C., Galderisi U., Cipollaro M., De Feo M., Forte A. Patients with bicuspid and tricuspid aortic valve exhibit distinct regional microrna signatures in mildly dilated ascending aorta. Heart Vessel. 2017;32:750–767. doi: 10.1007/s00380-016-0942-7. [DOI] [PubMed] [Google Scholar]
- 11.Otto C.M., Nishimura R.A., Bonow R.O., Carabello B.A., Erwin J.P., 3rd, Gentile F., Jneid H., Krieger E.V., Mack M., McLeod C., et al. 2020 ACC/AHA Guideline for the Management of Patients with Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143:e72–e227. doi: 10.1161/CIR.0000000000000923. [DOI] [PubMed] [Google Scholar]
- 12.Mestdagh P., Hartmann N., Baeriswy L., Andreasen D., Bernard N., Chen C., Cheo D., D’Andrade P., DeMayo M., Dennis L., et al. Evaluation of quantitative miRNA expression platforms in the microRNA quality control (miRQC) study. Nat. Methods. 2014;11:809–815. doi: 10.1038/nmeth.3014. [DOI] [PubMed] [Google Scholar]
- 13.Farina F.M. miR-128-3p Is a Novel Regulator of Vascular Smooth Muscle Cell Phenotypic Switch and Vascular Diseases (vol 126, e120, 2020) Circ. Res. 2020;127:e120–e135. doi: 10.1161/CIRCRESAHA.120.316489. [DOI] [PubMed] [Google Scholar]
- 14.Soto M.E., Guarner-Lans V., Herrera-Morales K.Y., Perez-Torres I. Participation of Arachidonic Acid Metabolism in the Aortic Aneurysm Formation in Patients with Marfan Syndrome. Front. Physiol. 2018;9:77. doi: 10.3389/fphys.2018.00077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hiratzka L.F., Bakris G.L., Beckman J.A., Bersin R.M., Carr V.F., Casey D.E., Jr., Eagle K.A., Hermann L.K., Isselbacher E.M., Kazerooni E.A., et al. 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with Thoracic Aortic Disease: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, American Association for Thoracic Surgery, American College of Radiology, American Stroke Association, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society of Interventional Radiology, Society of Thoracic Surgeons, and Society for Vascular Medicine. Circulation. 2010;121:e266–e369. doi: 10.1161/CIR.0b013e3181d4739e. [DOI] [PubMed] [Google Scholar]
- 16.Araujo N.N.F., Lin-Wang H.T., Germano J.F., Farsky P.S., Feldman A., Rossi F.H., Izukawa N.M., Higuchi M.L., Savioli Neto F., Hirata M.H., et al. Dysregulation of microRNAs and target genes networks in human abdominal aortic aneurysm tissues. PLoS ONE. 2019;14:e0222782. doi: 10.1371/journal.pone.0222782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zalewski D.P., Ruszel K.P., Stepniewski A., Galkowski D., Bogucki J., Komsta L., Kolodziej P., Chmiel P., Zubilewicz T., Feldo M., et al. Dysregulation of microRNA Modulatory Network in Abdominal Aortic Aneurysm. J. Clin. Med. 2020;9:1974. doi: 10.3390/jcm9061974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ikonomidis J.S., Ivey C.R., Wheeler J.B., Akerman A.W., Rice A., Patel R.K., Stroud R.E., Shah A.A., Hughes C.G., Ferrari G., et al. Plasma biomarkers for distinguishing etiologic subtypes of thoracic aortic aneurysm disease. J. Thorac. Cardiovasc. Surg. 2013;145:1326–1333. doi: 10.1016/j.jtcvs.2012.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Girdauskas E., Neumann N., Petersen J., Sequeira-Gross T., Naito S., von Stumm M., von Kodolitsch Y., Reichenspurner H., Zeller T. Expression Patterns of Circulating MicroRNAs in the Risk Stratification of Bicuspid Aortopathy. J. Clin. Med. 2020;9:276. doi: 10.3390/jcm9010276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mohamed S.A. MicroRNA detection in the pathogenesis of BAV-associated aortopathy-mediated vascular remodelling through EndMT/EMT. J. Intern. Med. 2019;285:115–117. doi: 10.1111/joim.12856. [DOI] [PubMed] [Google Scholar]
- 21.Li Y., Maegdefessel L. Non-coding RNA Contribution to Thoracic and Abdominal Aortic Aneurysm Disease Development and Progression. Front. Physiol. 2017;8:429. doi: 10.3389/fphys.2017.00429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.D’Amico F., Doldo E., Pisano C., Scioli M.G., Centofanti F., Proietti G., Falconi M., Sangiuolo F., Ferlosio A., Ruvolo G., et al. Specific miRNA and Gene Deregulation Characterize the Increased Angiogenic Remodeling of Thoracic Aneurysmatic Aortopathy in Marfan Syndrome. Int. J. Mol. Sci. 2020;21:6886. doi: 10.3390/ijms21186886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Oceandy D., Amanda B., Ashari F.Y., Faizah Z., Aziz M.A., Stafford N. The Cross-Talk Between the TNF- and RASSF-Hippo Signalling Pathways. Int. J. Mol. Sci. 2019;20:2346. doi: 10.3390/ijms20092346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.He J., Bao Q., Yan M., Liang J., Zhu Y., Wang C., Ai D. The role of Hippo/yes-associated protein signalling in vascular remodelling associated with cardiovascular disease. Br. J. Pharmacol. 2018;175:1354–1361. doi: 10.1111/bph.13806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Korf-Klingebiel M., Reboll M.R., Grote K., Schleiner H., Wang Y., Wu X.K., Klede S., Mikhed Y., Bauersachs J., Klintschar M., et al. Heparan Sulfate-Editing Extracellular Sulfatases Enhance VEGF Bioavailability for Ischemic Heart Repair. Circ. Res. 2019;125:787–801. doi: 10.1161/CIRCRESAHA.119.315023. [DOI] [PubMed] [Google Scholar]
- 26.Du P.Z., Dai F.J., Chang Y.W., Wei C.Y., Yan J., Li J.M., Liu X.B. Role of miR-199b-5p in regulating angiogenesis in mouse myocardial microvascular endothelial cells through HSF1/VEGF pathway. Environ. Toxicol. Pharmacol. 2016;47:142–148. doi: 10.1016/j.etap.2016.09.007. [DOI] [PubMed] [Google Scholar]
- 27.Xu B., Iida Y., Glover K.J., Ge Y., Wang Y., Xuan H., Hu X., Tanaka H., Wang W., Fujimura N., et al. Inhibition of VEGF (Vascular Endothelial Growth Factor)-A or its Receptor Activity Suppresses Experimental Aneurysm Progression in the Aortic Elastase Infusion Model. Arterioscler. Thromb. Vasc. Biol. 2019;39:1652–1666. doi: 10.1161/ATVBAHA.119.312497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Holderfield M.T., Hughes C.C.W. Crosstalk between vascular endothelial growth factor, notch, and transforming growth factor-beta in vascular morphogenesis. Circ. Res. 2008;102:637–652. doi: 10.1161/CIRCRESAHA.107.167171. [DOI] [PubMed] [Google Scholar]
- 29.Kumar S., Kim C.W., Simmons R.D., Jo H. Role of Flow-Sensitive microRNAs in Endothelial Dysfunction and Atherosclerosis Mechanosensitive Athero-miRs. Arterioscler. Thromb. Vasc. Biol. 2014;34:2206–2216. doi: 10.1161/ATVBAHA.114.303425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sophocleous F., Milano E.G., Pontecorboli G., Chivasso P., Caputo M., Rajakaruna C., Bucciarelli-Ducci C., Emanueli C., Biglino G. Enlightening the Association between Bicuspid Aortic Valve and Aortopathy. J. Cardiovasc. Dev. Dis. 2018;5:21. doi: 10.3390/jcdd5020021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Girdauskas E., Borger M.A., Secknus M.A., Girdauskas G., Kuntze T. Is aortopathy in bicuspid aortic valve disease a congenital defect or a result of abnormal hemodynamics? A critical reappraisal of a one-sided argument. Eur. J. Cardio-Thorac. Surg. 2011;39:809–814. doi: 10.1016/j.ejcts.2011.01.001. [DOI] [PubMed] [Google Scholar]
- 32.Padang R., Bannon P.G., Jeremy R., Richmond D.R., Semsarian C., Vallely M., Wilson M., Yan T.D. The genetic and molecular basis of bicuspid aortic valve associated thoracic aortopathy: A link to phenotype heterogeneity. Ann. Cardiothorac. Surg. 2013;2:83–91. doi: 10.3978/j.issn.2225-319X.2012.11.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Atkins S.K., Sucosky P. Etiology of bicuspid aortic valve disease: Focus on hemodynamics. World J. Cardiol. 2014;6:1227–1233. doi: 10.4330/wjc.v6.i12.1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Saikrishnan N., Mirabella L., Yoganathan A.P. Bicuspid aortic valves are associated with increased wall and turbulence shear stress levels compared to trileaflet aortic valves. Biomech. Model. Mechanobiol. 2015;14:577–588. doi: 10.1007/s10237-014-0623-3. [DOI] [PubMed] [Google Scholar]
- 35.Barker A.J., Markl M., Burk J., Lorenz R., Bock J., Bauer S., Schulz-Menger J., von Knobelsdorff-Brenkenhoff F. Bicuspid Aortic Valve Is Associated with Altered Wall Shear Stress in the Ascending Aorta. Circ.-Cardiovasc. Imaging. 2012;5:457–466. doi: 10.1161/CIRCIMAGING.112.973370. [DOI] [PubMed] [Google Scholar]
- 36.Cosentino F., Scardulla F., D’Acquisto L., Agnese V., Gentile G., Raffa G., Bellavia D., Pilato M., Pasta S. Computational modeling of bicuspid aortopathy: Towards personalized risk strategies. J. Mol. Cell Cardiol. 2019;131:122–131. doi: 10.1016/j.yjmcc.2019.04.026. [DOI] [PubMed] [Google Scholar]
- 37.Bissell M.M., Hess A.T., Biasiolli L., Glaze S.J., Loudon M., Pitcher A., Davis A., Prendergast B., Markl M., Barker A.J., et al. Aortic Dilation in Bicuspid Aortic Valve Disease Flow Pattern Is a Major Contributor and Differs with Valve Fusion Type. Circ.-Cardiovasc. Imaging. 2013;6:499–507. doi: 10.1161/CIRCIMAGING.113.000528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mahadevia R., Barker A.J., Schnell S., Entezari P., Kansal P., Fedak P.W., Malaisrie S.C., McCarthy P., Collins J., Carr J., et al. Bicuspid aortic cusp fusion morphology alters aortic three-dimensional outflow patterns, wall shear stress, and expression of aortopathy. Circulation. 2014;129:673–682. doi: 10.1161/CIRCULATIONAHA.113.003026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jones J.A., Stroud R.E., O’Quinn E.C., Black L.E., Barth J.L., Elefteriades J.A., Bavaria J.E., Gorman J.H., Gorman R.C., Spinale F.G., et al. Selective MicroRNA Suppression in Human Thoracic Aneurysms Relationship of miR-29a to Aortic Size and Proteolytic Induction. Circ.-Cardiovasc. Genet. 2011;4:605–613. doi: 10.1161/CIRCGENETICS.111.960419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Borghini A., Foffa I., Pulignani S., Vecoli C., Ait-Ali L., Andreassi M.G. miRNome Profiling in Bicuspid Aortic Valve-Associated Aortopathy by Next-Generation Sequencing. Int. J. Mol. Sci. 2017;18:2498. doi: 10.3390/ijms18112498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yassine N.M., Shahram J.T., Body S.C. Pathogenic Mechanisms of Bicuspid Aortic Valve Aortopathy. Front. Physiol. 2017;8:687. doi: 10.3389/fphys.2017.00687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhang P., Huang A., Ferruzzi J., Mecham R.P., Starcher B.C., Tellides G., Humphrey J.D., Giordano F.J., Niklason L.E., Sessa W.C. Inhibition of MicroRNA-29 Enhances Elastin Levels in Cells Haploinsufficient for Elastin and in Bioengineered Vessels-Brief Report. Arterioscler. Thromb. Vasc. Biol. 2012;32:756–759. doi: 10.1161/ATVBAHA.111.238113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Christopher A.F., Kaur R.P., Kaur G., Kaur A., Gupta V., Bansal P. MicroRNA therapeutics: Discovering novel targets and developing specific therapy. Perspect. Clin. Res. 2016;7:68–74. doi: 10.4103/2229-3485.179431. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.