Abstract
The Plasmodium falciparum rhoptry-associated proteins 1 and 2 (RAP1 and RAP2) are candidate antigens for a subunit malaria vaccine. The design of the study, which looks at the acquisition of immunity to malaria from childhood to old age, has allowed us to document the interaction of HLA and age on levels of antibody to specific malarial antigens. Antibodies reach maximum levels to RAP1 after the age of 15 but to RAP2 only after the age of 30. The effect of HLA-DRB1 and -DQB1 and age on levels of antibody to rRAP1 and rRAP2 was analyzed with a multiple regression model in which all HLA alleles and age were independent variables. DQB1*0301 and -*03032 showed an age-dependent association with levels of antibody to rRAP1, being significant in children 5 to 15 years (P < 0.001) but not in individuals over 15 years of age. DRB1*03011 showed an age-dependent association with antibody levels to rRAP2; however, this association was in adults over the age of 30 years (P < 0.01) but not in individuals under the age of 30 years. No associations were detected between DRB1 alleles and RAP1 antibody levels or between DQB1 alleles and RAP2 antibody levels. Thus, not only the HLA allele but also the age at which an interaction is manifested varies for different malarial antigens. The interaction may influence either the rate of acquisition of antibody or the final level of antibody acquired by adults.
The rhoptry-associated proteins 1 and 2 (RAP1 and RAP2) form a low-molecular-mass complex located in the rhoptries of Plasmodium falciparum (5, 9). The rhoptries are a pair of organelles at the apical end of the parasite that are involved in the invasion of erythrocytes; thus, molecules in the rhoptries were among the first components identified as potential candidates for a subunit vaccine. Subsequent studies have borne out this potential. Monoclonal antibodies (MAbs) directed against rhoptry-associated proteins have been shown to provide substantial inhibition of parasite invasion in vitro (7, 17, 19, 22, 29, 37). In addition, protein preparations containing both RAP1 and RAP2 have been used to immunize Saimiri monkeys. In these studies, the immunized monkeys developed a lower peak parasitemia than nonimmunized controls and were protected from subsequent lethal blood-stage challenge with P. falciparum (9, 30, 32, 34) in a way similar to the protection acquired by immunization with whole merozoite (27, 39, 46).
Two rhoptry-associated proteins, RAP1 and RAP2, have been sequenced (20, 36). Unlike many other P. falciparum antigens, RAP1 and RAP2 exhibit a high degree of sequence similarity among P. falciparum isolates. Isolates from Honduras, Sierra Leone, Tanzania, India, Thailand, The Netherlands, Uganda, and Vietnam have been sequenced for RAP1, and isolates from Honduras, Papua New Guinea, The Netherlands, Uganda, and Vietnam have been sequenced for RAP2 (9, 20, 21, 33, 36). Thus, it is thought likely that RAP1 and RAP2 proteins may provide a relatively isolate-independent protective immunity in humans compared to other P. falciparum antigens.
Immunity to the erythrocytic stages of P. falciparum is acquired following repeated clinical and subclinical infections. Antibodies play an important role in protection. This was first demonstrated by the passive transfer of antibodies from immune adults to children with acute P. falciparum infection, dramatically reducing their parasitemias (8). More recently, high levels of antibodies to certain defined asexual stage antigens (RAP1, MSP-1, RESA, MSA2, a glutamate-rich protein [GLURP], and the P. falciparum ring erythrocyte surface antigen [RESA]) have been reported to correlate with decreased parasite densities in some but not all studies (1–3, 12, 13, 18, 25, 35, 44, 45).
Production of antibody requires help from T cells which are activated by interaction with an HLA class II-peptide complex (14). Many different alleles of HLA genes exist within the population, and these allelic products differ in their abilities to bind and present different antigenic determinants of proteins. Even a single amino acid difference between HLA allelic products is sufficient to generate differences in their abilities to bind and present peptides (11, 23). Thus, definition of HLA alleles at the nucleotide level is required for adequate dissection of the interaction between HLA and an immune response. Most, but not all, recent studies, which have used DNA-based HLA typing, have reported associations between HLA class II alleles and antibody production or levels to various malarial antigens (4, 6, 16, 26, 40, 45).
In this study, we examined the effects of age and HLA-DRB1 and -DQB1 allelic products on the level of antibodies to recombinant forms of RAP1 and RAP2 in individuals between the ages of 5 and 70 years. The recombinant proteins have been found to be immunogenic in animal models and share linear epitopes with native antigens as detected by the human immune system (41, 43, 44).
MATERIALS AND METHODS
Population and sample collection.
The study was conducted in Etoa, a village of 485 individuals in central Cameroon where P. falciparum malaria is holoendemic. Etoa is located in the forest zone and has an equatorial climate. All ethical concerns were first cleared by the Cameroonian Ministry of Health and the local district health and administrative officials. The epidemiology of the study site is described fully elsewhere (I. A. Qyayki et al., submitted for publication). There are two rainy seasons (March to June and September to November). Malaria transmission is perennial, with peak infectivity during the rainy seasons. A malaria prevalence survey, performed at the beginning of the spring rainy season in 1995, indicated that P. falciparum prevalence was approximately 70% in children under 10 years of age and approximately 30% in individuals over 10 years of age. Immediately after the prevalence study was completed, peripheral whole blood was obtained from volunteers, tested for T-cell proliferation, and typed for HLA-DRB1 and HLA-DQB1 alleles, and plasma was assayed for antibody to various asexual stage antigens. Due to the amount of blood required, children under 5 years of age were not included in the study. Plasma antibodies to recombinant RAP1 and RAP2 were measured in 131 individuals from 53 different families. Family members with identical HLA-DR and -DQ alleles were removed from the data set.
rRAP1 and rRAP2.
Purified rRAP1 and rRAP2, expressed in Escherichia coli, is identical to material used in animal immunogenicity studies (41, 43). The products have been extensively purified as reported previously (41, 42). Briefly, inclusion bodies were extracted, and RAP proteins were separated on a Ni-nitriloacetic acid resin (Qiagen, Inc., Valencia, Calif.). Further purification to clinical grade was performed by a modified version of isotachophoresis (42). Recombinant RAP1 (rRAP1) contains a shortened form (amino acids 23 to 608) of the mature sequence specified by the K1 allele of RAP1, while rRAP2 contains the entire mature polypeptide sequence specified by the FCQ27-PNG allele of RAP2.
Determination of HLA alleles.
DNA was isolated from whole blood drawn in EDTA by using the Puregene DNA isolation kit (Gentra Systems, Inc., Minneapolis, Minn.). Allele level typing was performed for HLA-DRB1 and -DQB1 (except for DRB1*04 and DQB1*02) by using probes and primers obtained commercially (Lifecodes Corporation, Stamford, Conn.) and augmented, whenever necessary, for allele resolution. Because of the high occurrence of DR52 group alleles in the Cameroonian population, primer sets employing two reverse primers specific for the Val-Gly polymorphism at amino acid 86 of DRB1 were used (38). The following alleles could be resolved by the sets of primers and probes used: DRB1*0101-0104, *1501-1506, *1601-1608, *03011-0309, *04, *0701, *0801-0816, *0901, *1001, *1101-1126, *1201-1205, *1301-1315 and *1317-1327, and *1401-1425, and DQB1*02, *0301-0305, *0501-0504, *0601-0608, and *0401-0402.
ELISA.
Enzyme-linked immunosorbent assay (ELISA) procedures have been previously described (31, 41). Briefly, 96-well flat-bottom microtiter ELISA plates (Immunoplate [Maxisorp]; Nunc) were coated with 25 ng of rRAP1 or 100 ng of RAP2 per well in phosphate-buffered saline (PBS). Sera were diluted 1:200 in diluent (0.05% Tween 20–1% bovine serum albumin in PBS), added to blocked antigen plates in triplicate, and incubated for 90 min at 37°C. Positive and negative controls were set up for every plate. Wells were washed again in washing buffer (0.05% Tween in PBS), and horseradish peroxidase-conjugated goat anti-human immunoglobulin G (gamma-chain specific) (Bio-Rad Laboratories) was added. After incubation for 2 h at room temperature, the wells were washed, and substrate (o-phenylenediamine dihydrochloride) (Pharmacia/Amersham) was added. The reaction was read at an absorbance of 450 nm by using a Titretek Multiscan apparatus (Titertek; Flow Laboratories). For all of the ELISAs, the mean baseline level of positivity was an absorbance of >2 standard deviations above the mean optical density (OD) of sera from 30 nonexposed Europeans with no history of malaria (negative control sera). The mean OD plus two standard deviations was 0.2. Prior standardization of the rRAP1 and rRAP2 ELISA by titration of sera from malaria “immune” adults established that sera with an OD ranging from 0.4 to 0.79 gave an endpoint titer of <1:2,000. Sera with an OD of >0.8 corresponded to an endpoint titer of >1:10,000. Sera from malaria immune adults with high-titer or low-titer malaria antibody (positive control sera) and two MAbs (MAb 7H8/50 for RAP1 and MAb 3A9/48 for RAP2) were used as controls.
Data analysis.
The statistical analysis was performed by using SAS software (SAS Institute, Inc., Cary, N.C.). Regression analysis was performed by using the OD as the dependent continuous variable and HLA alleles and age as the independent variables. Also, age was dichotomized into two groups. However, different dichotomies were used for RAP1 and for RAP2 based on the age related changes observed in the OD to these proteins. For the RAP1 study, the two age groups were children between the ages of 5 and 15 years and individuals over the age of 15 years. For the RAP2 study, the two age groups were individuals between the ages of 5 and 30 years compared to individuals over the age of 30 years. Any variable which was significantly associated with RAP1 or RAP2 antibody levels at the simple regression was entered into a multiple regression. Variables which were nonsignificant in the multiple regression analysis at the 0.1 level (i.e., an observed P value of >0.1) were eliminated. An interaction term was included (age*allele) to test whether the association between the OD and DRB1 or DQB1 is significantly different for the two age groups. If this term was significant upon including the DRB1 or DQB1 term and the age term, then we could conclude that the age association with RAP1 (or RAP2) depended on whether or not one had a specific DRB1 or DQB1 allele.
RESULTS
Antibody levels to RAP1 and RAP2 are age related.
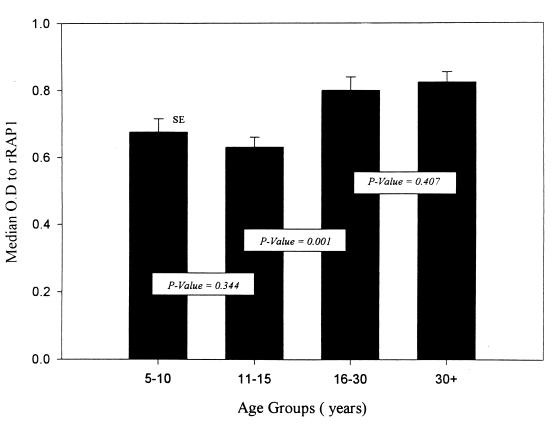
ELISAs were performed using rRAP1 and rRAP2 with the sera from 131 individuals from Etoa, Cameroon. The median OD values were compared for adjacent age groups (5 to 10 years [n = 28], 10 to 15 years [n = 33], 15 to 30 years [n = 28], and >30 years [n = 42]) using the Wilcoxon rank sum pairwise test. Thus, the median OD of one age group (e.g., 5 to 10 years) was compared to that of the adjacent age group (e.g., 10 to 15 years). Figure 1 shows the results for RAP1. A significant change in the median OD was observed only between the 10- to 15-year and the 15- to 30-year age groups. When the data were pooled for the 5- to 15- and for the >15-year age groups, the difference between the two groups was highly significant (P = 0.0002; 5 to 15 year, median OD = 0.66; >15 year, median OD = 0.81).
FIG. 1.
Median OD values of antibodies to rRAP1 within an age group. Probability values from Wilcoxon rank sum pairwise tests between the median OD values of adjacent age groups (e.g., 5 to 15 years versus 15 to 30 years) are shown.
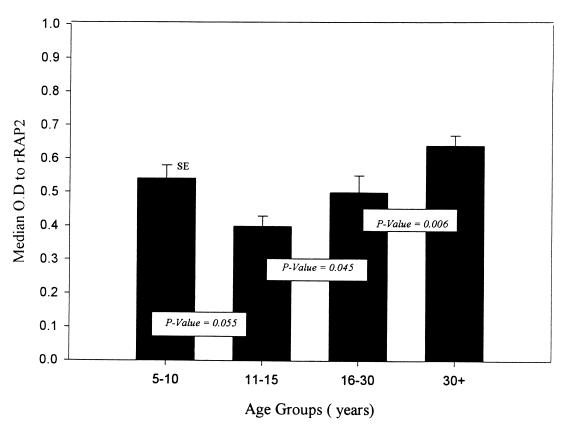
Figure 2 shows the results of Wilcoxon rank sum pairwise tests comparing the median antibody responses for rRAP2 between adjacent age groups. For RAP2, the median OD significantly decreases in the 10- to 15-year age group compared to the 5- to 10-year age group. However, the mean OD of the 15- to 30-year age group is almost equal to that of the 5- to 10-year age group. There is a significant increase in OD between the 15- to 30-year and the >30-year age groups. When the data were pooled for the 5- to 30-year and the >30-year age groups, the difference between the two groups was very significant (P = 0.006; 5 to 30 year, median OD = 0.50; >30 year, median OD = 0.61). Note that the median OD of antibody to RAP2 (i.e., 0.62) is lower than the median OD to RAP1 (i.e., 0.81) in adults over the age of 30 years. Based on prior standardization of immune sera by titration, these OD values correspond to titers of <1:2,000 for RAP2 and of >1:10,000 for RAP1.
FIG. 2.
Median OD values of antibodies to rRAP2 within an age group. Probability values from Wilcoxon rank sum pairwise tests between the median OD values of adjacent age groups (e.g., 5 to 15 years versus 15 to 30 years) are shown.
Effect of HLA and age on antibody levels to RAP1.
The effects of different HLA-DRB1 and -DQB1 alleles and age on the level of antibodies to RAP1 were analyzed. DRB1 and DQB1 alleles which are observed frequently in the population were included in the analysis: DRB1*0102, *03011, *0701, *1101, *1301, and *1503 and DQB1*02, *0301, *03032, *0501, and *0602. Other DRB1 alleles (DRB1*0101, *03021, *1303, *0901, and *1001) and DQB1 alleles (DQB1*0601, *0603, *0604, *0608, *0302, *0502, and *0402) were not evaluated because there were fewer than five individuals with the given allele.
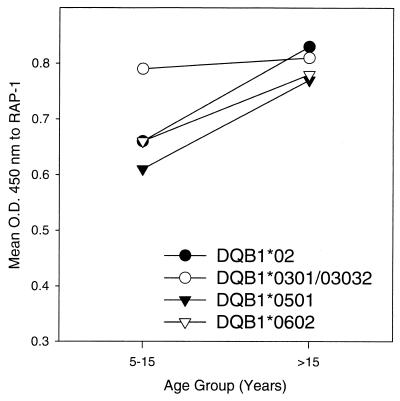
Figure 3 shows the effect of age and HLA-DQB1 alleles on the mean OD of antibodies to RAP1. The age groups, individuals between the ages of 5 and 15 years and those over the age of 15 years, were selected because of the observed age related changes in median OD shown in Fig. 1. When stratified by HLA-DQB1, the mean OD varied widely in the 5- to 15-year age group but not in the >15-year age group. Children between the ages of 5 and 15 years, who were positive for DQB1*03 (DQB1*0301 and DQB1*03032), have the highest mean OD values. Perhaps more importantly, this mean is not different from the mean OD observed in adults. Individuals positive for DQB1*0301 or DQB1*03032 had mean OD values close to that of DQB1*03 (data not shown).
FIG. 3.
Mean OD values to rRAP1 stratified by HLA-DQB1 in individuals under the age of 15 years and those over the age of 15 years. The association between HLA-DQB1*03 (-*0301 and -*03032) and the levels of antibody to rRAP1 is significant in individuals of 5 to 15 years (P = 0.0007) but not in individuals over the age of 15 years (P = 0.73) by multiple regression analysis, using age and all DQB1 alleles as independent variables.
Multiple regression was performed by using age and all DRB1 or all DQB1 alleles as independent variables. The two HLA-DQ alleles (DQB1*0301 and DQB1*03032) were analyzed together as the “DQB1*03 phenotype,” as well as separately in a different regression. A significant association was observed between DQB1*03 and antibody level, but only in the 5- to 15-year age group (P = 0.0007) and not in the >15-year age group (P = 0.73). When DQB1*0301 and DQB1*03032 alleles were regressed as two separate variables, the P value remained significant for DQB1*0301 (P = 0.003) and borderline for DQB1*03032 (P = 0.014) in the 5- to 15-year group. These data suggest that the association is predominantly with DQB1*0301. The association between the level of antibodies to RAP1 and DQB1*0301 but not DQB1*03032 was shown to be dependent on age by using an interaction term (P = 0.029). No significant association, positive or negative, was observed with any HLA-DRB1 allele and antibody levels to RAP1. No significant negative association was observed for any DQB1 allele and RAP1 antibody levels.
Effect of HLA and age on antibody levels to RAP2.
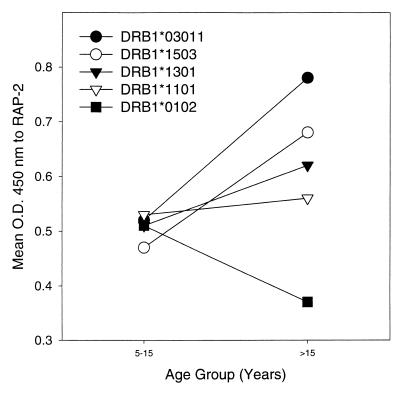
The effect of HLA class II alleles on antibody levels to RAP2 was analyzed also. The same HLA-DR and HLA-DQ alleles were investigated as for RAP1. Figure 4 shows the effect of HLA-DRB1 alleles on the mean OD of antibodies to rRAP2. When stratified by HLA-DRB1, the mean OD varied in the >30-year age group but not in the 5- to 30-year age group. For RAP2, the two age groups were selected based on the observed age related changes in median OD shown in Fig. 2. Individuals over the age of 30 years who are positive for DRB1*03011 have the highest mean OD.
FIG. 4.
Mean OD values to rRAP2 stratified by HLA-DRB1 in individuals under the age of 30 years and those over the age of 30 years. The association between HLA-DRB1*03011 and the levels of antibody to rRAP2 is significant in individuals over the age of 30 years (P = 0.0059) but not in individuals under the age of 30 years (P = 0.63) by multiple regression analysis, using age and all DRB1 alleles as independent variables.
Multiple regression was performed using age and all DRB1 or all DQB1 alleles as independent variables. A significant association between DRB1*03011 and antibody levels to RAP2 (P = 0.0075) is observed even not taking age into account. If age (5 to 30 years versus >30 years) is included as an independent variable, then DRB1*03011 remains significant in the >30-year age group (P = 0.0059) but not in the <30-year age group (P = 0.63). Therefore, the interaction of HLA and age on RAP2 antibody levels affects the final level of antibody acquired. There was no association, positive or negative, observed between HLA-DQB1 and antibody levels to RAP2. Also, no significant negative association was observed for any HLA-DRB1 allele and RAP2 antibody levels.
Antibody correlations with the hemoglobin S gene.
The effect of hemoglobin S on antibody production was evaluated, since the sickle cell trait is known to provide a strong protective factor against malaria and has been associated with higher levels of antibodies to certain asexual stage antigens. There is no significant difference between the antibody levels to rRAP1 of individuals who are AS heterozygotes compared to those who are AA homozygotes (AS mean OD = 0.677; AA mean OD = 0.644; P = 0.6). Similarly, there is no difference between the antibody levels to rRAP2 between AS heterozygotes and AA homozygotes (mean OD of AS = 0.555; mean OD of AA = 0.479; P = 0.29).
DISCUSSION
The design of our study, which looks at the acquisition of immunity to malaria from childhood to old age, has allowed us to document the interaction of HLA and age on levels of antibody to specific malarial antigens. The interactions between HLA and age on levels of antibody to rRAP1 and rRAP2 reported here show two different effects. First, the data suggest that HLA-DQB1 influences the rate of acquisition of antibody to rRAP1. The RAP1 data indicate that young subjects (5 to 15 years), who are positive for DQB1*0301 and *03032, have high levels of antibody to RAP1 earlier but that this difference in antibody level becomes insignificant in later years. The association between HLA-DQB1*0301 and DQB1*03032 and antibody levels to RAP1 in the 5- to 15-year age group is highly significant even after multiple regression in which all HLA alleles and age are included as independent variables. Such an influence could provide early protection for young children positive for HLA-DQB1*0301 and DQB1*03032 since Jakobsen and colleagues reported that young children (under 5 years) with high IgG reactivity to RAP1 have lower P. falciparum parasite densities compared to young children with low IgG (25). Unfortunately, the village of Etoa did not allow blood to be drawn from children under the age of 5 years, the age when immunity to clinical symptoms and blood-stage parasites is presumably acquired, nor did our study design allow correlation with protection in the individuals tested.
Second, HLA may influence the overall antibody levels obtained, i.e., “high producers” rather than “early producers.” The RAP2 data indicate that subjects over the age of 30 years, who are positive for DRB1*03011, have significantly higher levels of antibodies. Once again, if the protection provided by antibodies to RAP2 were sufficiently strong, individuals positive for DRB1*03011 might have a higher level of protection compared to individuals with other HLA alleles. Obviously, by the time an individual reaches 30 years of age, they have antibodies to many different malarial antigens and the isolated benefit contributed by a given HLA allele to a given malarial antigen may be masked.
Previous studies have reported that HLA class II antigens are associated with antibody response, or the lack of a response, to certain malarial antigens (4, 6, 28, 40). Interestingly, DQB1*0301 is associated with both an early response to RAP1 in Cameroon and with high antibody levels to SPf66 in a naturally exposed population in Papua New Guinea (4). The positive association of the antibody levels to SPf66, observed with DRB1*11 and DQB1*0301, becomes insignificant (P > 0.025) after multiple regression. In our study there was no significant association with DRB1*11, although both DRB1*1101 and DRB1*1102 were included in the group of individuals with high antibody levels to RAP1. Due to the combinations of HLA-DR and -DQ observed in Cameroon (N. Pimtanothai et al., submitted for publication), our data suggest that the HLA association is either with HLA-DQ or with another gene (or genes) located in the vicinity of DQB1.
The levels of antibodies to rRAP1, as suggested by the median OD, increase significantly above the age of 15 years and do not increase significantly after that time. On the other hand, the levels of antibodies to rRAP2 do not reach a maximum until after the age of 30 years. Studies in natives of Papua New Guinea have shown that maximum levels of antibody to both RAP1 and RAP2 do not appear until an individual is more than 30 years old (44). Our studies indicate that maximum antibody levels to RAP1, but not to RAP2, may be reached by the age of 15 years in Cameroon. The peak median OD is lower for rRAP2 (median OD = 0.62) than that observed for rRAP1 (median OD = 0.81). Based on titrations of “immune” sera for standardization of the rRAP1 and rRAP2 ELISA, these OD values correspond to titers of <1:2,000 for RAP2 and of >1:10,000 for RAP1. These data are consistent with other studies which suggest that RAP2 is a weaker immunogen (24, 44). Although cross-reactive antibodies to RAP1 are known to arise after immunization with RAP2 (44), the later age at which the peak median OD values are reached for RAP2, as well as the different associations between HLA and antibody level for RAP1 and RAP2, suggests that the associations observed are specific for the given molecule.
No “low-responder” alleles were identified. This is in contrast to the Papua New Guinea study in which a significant negative association was detected between the humoral responses against SPf66 and DR15 and DQB1*0601 (4). The DR15 allele associated with DQB1*0601 which is observed in Papua New Guinea is DRB1*1502 (15). The DRB1*1502 DQB1*0601 haplotype is not observed in Cameroon. The only DR15 allele observed in Cameroon is DRB1*1503, which is found exclusively in individuals of African ancestry and differs from DRB1*1502 at codon 86, the major peptide anchor binding pocket for DRB1 molecules. DQB1*0601 is observed at <1% in Cameroon (Pimtanothai et al., submitted). In the Colombian Spf66 vaccine DRB1*04-positive individuals were reported to be low responders (28). The negative association between DRB1*04 and antibody levels could not be evaluated in our study since there are no DRB1*04-positive individuals in Etoa, Cameroon. Since DRB1*04 alleles are present in such a low frequency in many African countries and in Papua New Guinea, it is interesting to hypothesize that DRB1*04 alleles might have been selected against in areas of intense malaria transmission.
The findings reported here are the first to indicate that both an HLA allele and an individual's age interact to affect the levels of antibodies to malarial antigens. Our study suggests that the effect of HLA is expressed at different ages for different malarial antigens and may influence the rate of acquisition of adult antibody levels, as well as the final level of antibodies acquired by adults. These data provide a model for investigating the influence of HLA and age on acquisition of immunity to malaria.
ACKNOWLEDGMENTS
This work was supported in part by Public Health Service grant UO1-AI-35839 from the NIAID, NIH; a grant from USAID; the Queensland Institute of Medical Research and University of Queensland, Brisbane, Australia; and a Travel Fellowship Award from the University of Queensland to I.A.Q.
We thank the chiefs and villagers of Etoa for their participation. Without the approval of the Ministry of Health of the United Republic of Cameroon, the Ethical Committee of the Faculty of Medicine and Biomedical Sciences (FMBS), and Maurice Sosso, Dean of FMBS, University of Yaounde I, this work would not have been possible. We also thank Saramane PTY, Ltd., for supplying bacterial cell pellets containing rRAP1 and rRAP2 and Anna Walduck for purifying the RAP proteins.
REFERENCES
- 1.Al-Yaman F, Genton B, Anders R, Teraika J, Ginny M, Mellor S, Alpers M P. Assessment of the role of the humoral response to Plasmodium falciparum MSA2 compared to RESA and SPf66 in protecting Papua New Guinean children from clinical malaria. Parasite Immunol. 1995;17:493–501. doi: 10.1111/j.1365-3024.1995.tb00920.x. [DOI] [PubMed] [Google Scholar]
- 2.Al-Yaman F, Genton B, Falk M, Anders R, Lewis D, Hii J, Beck H-P, Alpers M P. Humoral response to Plasmodium falciparum ring-infected erythrocyte surface antigen in a highly endemic area of Papua New Guinea. Am J Trop Med Hyg. 1995;52:66–71. doi: 10.4269/ajtmh.1995.52.66. [DOI] [PubMed] [Google Scholar]
- 3.Al-Yaman F, Genton B, Krammer K J, Chang S P, Hui G S, Baisor M, Alpers M P. Assessment of the role of naturally acquired antibody levels to Plasmodium falciparum merozoite surface protein-1 in protecting Papua New Guinean children from malaria morbidity. Am J Trop Med Hyg. 1996;54:443–448. doi: 10.4269/ajtmh.1996.54.443. [DOI] [PubMed] [Google Scholar]
- 4.Beck H P, Felger I, Barker M, Bugawan T, Genton B, Alexander N, Jazwinska E, Erlich H, Alpers M. Evidence of HLA class II association with antibody response against the malaria vaccine SPf66 in a naturally exposed population. Am J Trop Med Hyg. 1995;53:284–288. [PubMed] [Google Scholar]
- 5.Bushell G R, Ingram L T, Fardoulys C A, Cooper J A. An antigenic complex in the rhopteries of Plasmodium falciparum. Mol Biochem Parasitol. 1988;28:105–112. doi: 10.1016/0166-6851(88)90057-6. [DOI] [PubMed] [Google Scholar]
- 6.Calvo-Calle J M, Hammer J, Sinigaglia F, Clavijo P, Moya-Castro Z R, Nardin E H. Binding of malaria T cell epitopes to DR and DQ molecules in vitro correlates with immunogenicity in vivo: identification of a universal T cell epitope in the Plasmodium falciparum circumsporozoite protein. J Immunol. 1997;159:1362–1373. [PubMed] [Google Scholar]
- 7.Clark J T, Anand R, Akoglu T, McBride J S. Identification and characterization of proteins associated with the rhoptry organelles of Plasmodium falciparum merozoites. Parasitol Res. 1987;73:425–434. doi: 10.1007/BF00538200. [DOI] [PubMed] [Google Scholar]
- 8.Cohen S, Mcgregor I A, Carrington S P. Gammaglobulin and acquired immunity to human malaria. Nature. 1961;192:733–737. doi: 10.1038/192733a0. [DOI] [PubMed] [Google Scholar]
- 9.Collins, W. E., A. Salduck, J. S. Sullivan, K. Andrews, A. Stowers, C. Morris, V. Jennings, C. Yang, J. Kendall, Q. Lin, L. B. Martin, C. Diggs, and A. Saul. Efficacy of vaccines containing rhoptry-associated proteins RAP1 and RAP2 of Plasmodium falciparum in Saimiri boliviensis monkeys. Am. J. Trop. Med. Hyg., in press. [DOI] [PubMed]
- 10.Copper J A, Ingram L T, Bushell G R, Fardoulys A, Stenzel C A, Schofield L, Saul A. The 140/130/105 kilodalton protein complex in the rhoptries of Plasmodium falciparum consists of discrete polypeptides. Mol Biochem Parasitol. 1988;29:251–260. doi: 10.1016/0166-6851(88)90080-1. [DOI] [PubMed] [Google Scholar]
- 11.Demotz S, Barbey C, Corradin G, Amoroso A, Lanzavecchia A. The set of naturally processed peptides displayed by DR molecules is tuned by polymorphism of residue 86. Eur J Immunol. 1993;23:425–432. doi: 10.1002/eji.1830230219. [DOI] [PubMed] [Google Scholar]
- 12.Dziegiel M, Rowe P, Bennett S, Allen S J, Olerup O, Gorrschau A, Borre M, Riley E M. Immunoglobulin M and G antibody responses to Plasmodium falciparum glutamate-rich protein: correlation with clinical immunity in Gambian children. Infect Immun. 1993;61:103–108. doi: 10.1128/iai.61.1.103-108.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fruh K, Doumbo O, Muller H M, Koita O, McBride J, Crisanti A, Toure Y, Bujard H. Human antibody response to the major merozoite surface antigen of Plasmodium falciparum is strain specific and short-lived. Infect Immun. 1991;59:1319–1324. doi: 10.1128/iai.59.4.1319-1324.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Germain R N. Antigen processing and presentation. In: Paul W E, editor. Fundamental immunology. Philadelphia, Pa: Lippincott-Raven; 1999. pp. 287–340. [Google Scholar]
- 15.Goa X J, Serjeantson S W. Heterogeneity in HLA-DR2-related DR, DQ haplotypes in eight populations of Asia-Oceania. Immunogenetics. 1991;34:401–408. doi: 10.1007/BF01787491. [DOI] [PubMed] [Google Scholar]
- 16.Graves P M, Bhatia K, Burkot T R, Prasad M, Wirtz R A, Beckers P. Association between HLA type and antibody response to malaria sporozoite and gametocyte epitopes is not evident in immune Papua New Guineans. Clin Exp Immunol. 1989;78:418–423. [PMC free article] [PubMed] [Google Scholar]
- 17.Harnyuttanakorn P, McBride J S, Donachie S, Heidrich H G, Ridley R G. Inhibitory monoclonal antibodies recognize epitopes adjacent to a proteolytic cleavage site on the RAP1 protein of Plasmodium falciparum. Mol Biochem Parasitol. 1992;55:177–186. doi: 10.1016/0166-6851(92)90138-a. [DOI] [PubMed] [Google Scholar]
- 18.Hogh B, Petersen E, Dziegiel M, David K, Hanson A, Borre M, Holm A, Vuust J, Jepsen S. Antibodies to a recombinant glutamate-rich Plasmodium falciparum protein: evidence for protection of individuals living in a holoendemic area of Liberia. Am J Trop Med Hyg. 1992;46:307–313. doi: 10.4269/ajtmh.1992.46.307. [DOI] [PubMed] [Google Scholar]
- 19.Howard R F, Stanley H A, Campbell G H, Reese R T. Proteins responsible for a punctate fluorescence pattern in Plasmodium falciparum merozoites. Am J Trop Med Hyg. 1984;33:1055–1059. doi: 10.4269/ajtmh.1984.33.1055. [DOI] [PubMed] [Google Scholar]
- 20.Howard R F. The sequence of the p82 rhoptry protein is highly conserved between two Plasmodium falciparum isolates. Mol Biochem Parasitol. 1992;51:327–330. doi: 10.1016/0166-6851(92)90083-v. [DOI] [PubMed] [Google Scholar]
- 21.Howard R F, Peterson C. Limited RAP1 sequence diversity in field isolates of Plasmodium falciparum. Mol Biochem Parasitol. 1996;77:95–98. doi: 10.1016/0166-6851(96)02576-5. [DOI] [PubMed] [Google Scholar]
- 22.Howard R F, Jacobson K C, Rickel E, Thurman J. Analysis of inhibitory epitopes in the Plasmodium falciparum rhoptry protein RAP-1 including identification of a second inhibitory epitope. Infect Immun. 1998;66:380–386. doi: 10.1128/iai.66.1.380-386.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hurley, C. K., and N. Steiner. Differences in peptide binding of DR11 and DR13 microvariants demonstrate the power of minor variation in generating DR functional diversity. Hum. Immunol. 43:101–112. [DOI] [PubMed]
- 24.Jacobson K C, Thurman J, Schmidt C M, Rickel E, Olivera de Ferreira J, De Fatima Ferreira-da-Cruz M, Daniel-Ribeiro C T, Howard R F. A study of antibody and T cell recognition of rhoptry-associated protein-1 (RAP-1) and RAP-2 recombinant proteins and peptides of Plasmodium falciparum in migrants and residents of the state of Rondonia, Brazil. Am J Trop Med Hyg. 1998;59:208–216. doi: 10.4269/ajtmh.1998.59.208. [DOI] [PubMed] [Google Scholar]
- 25.Jakobsen P H, Lemnge M M, Abu-Zeid Y A, Msangeni H A, Salum F M, Mhina J I K, Akida J A, Ruta A S, Roon A M, Heegaard P M H, Ridley R G, Bygbjerg I C. Immunoglobulin G reactivities to rhoptry-associated protein-1 associated with decreased levels of Plasmodium falciparum parasitemia in Tanzanian children. Am J Trop Med Hyg. 1996;55:642–646. doi: 10.4269/ajtmh.1996.55.642. [DOI] [PubMed] [Google Scholar]
- 26.Migot F, Chougnet C, Perichon B, Danze P-M, Lepers J-P, Krishnamoorthy R, Deloron P. Lack of correlation between HLA class II alleles and immune responses to Pf155/ring-infected erythrocyte surface antigen (RESA) from Plasmodium falciparum in Madagascar. Am J Trop Med Hyg. 1995;52:252–257. doi: 10.4269/ajtmh.1995.52.252. [DOI] [PubMed] [Google Scholar]
- 27.Mitchell G H, Richards W H G, Butcher G A, Cohen S. Merozoite vaccination of Douroucouli monkeys against Falciparum malaria. Lancet. 1977;i:1335–1338. doi: 10.1016/S0140-6736(77)92551-X. [DOI] [PubMed] [Google Scholar]
- 28.Patarroyo M E, Vinasco J, Amador R, Espejo F, Silva Y, Moreno A, Rojas M, Salcedo M, Valero V, Goldberg A K, Kalil J. Genetic control of the immune response to a synthetic vaccine against Plasmodium falciparum. Parasite Immunol. 1991;13:509–516. doi: 10.1111/j.1365-3024.1991.tb00547.x. [DOI] [PubMed] [Google Scholar]
- 29.Perrin L H, Ramirez E, Lambert P H, Miescher P A. Inhibition of P. falciparum growth in human erythrocytes by monoclonal antibodies. Nature. 1981;289:301–303. doi: 10.1038/289301a0. [DOI] [PubMed] [Google Scholar]
- 30.Perrin L H, Merkli B, Gabra M S, Stocker J W, Chizzolini C, Richle R. Immunization with a Plasmodium falciparum merozoite surface antigen induces a partial immunity in monkeys. J Clin Investig. 1985;75:1718–1721. doi: 10.1172/JCI111881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Quakyi I A. The development and validation of an enzyme linked immunosorbent assay for malaria. Tropenmed Parasitol. 1980;31:325–333. [PubMed] [Google Scholar]
- 32.Ridley R G, Takacs B, Etlinger H, Scaife J G. A rhoptry antigen of Plasmodium falciparum is protective in Saimiri monkeys. Parasitology. 1990;101:187–192. doi: 10.1017/s0031182000063228. [DOI] [PubMed] [Google Scholar]
- 33.Ridley R G, Takacs B, Lahm H, Delves C J, Goman M, Certa U, Matile H, Woollett G R, Scaife J G. Characterization and sequence of a protective rhoptry antigen from Plasmodium falciparum. Mol Biochem Parasitol. 1990;41:125–134. doi: 10.1016/0166-6851(90)90103-s. [DOI] [PubMed] [Google Scholar]
- 34.Ridley R G, Lahm H W, Takacs B, Scaife J G. Genetic and structural relationships between components of a protective rhoptry antigen complex from Plasmodium falciparum. Mol Biochem Parasitol. 1991;47:245–246. doi: 10.1016/0166-6851(91)90184-8. [DOI] [PubMed] [Google Scholar]
- 35.Rooth I, Perlmann H, Bjorkman A. Plasmodium falciparum reinfection in children from a holoendemic area in relation to seroreactivities against oligopeptides from different malarial antigens. Am J Trop Med Hyg. 1991;45:309–318. doi: 10.4269/ajtmh.1991.45.309. [DOI] [PubMed] [Google Scholar]
- 36.Saul A, Cooper J, Hauquitz D, Irving D, Cheng Q, Stowers A, Limpaiboon T. The 42-kilodalton rhoptry-associated protein of Plasmodium falciparum. Mol Biochem Parasitol. 1992;50:139–150. doi: 10.1016/0166-6851(92)90251-e. [DOI] [PubMed] [Google Scholar]
- 37.Schofield L, Bushell G R, Cooper J A, Saul A J, Upcroft J A, Kidson C. A rhoptry antigen of Plasmodium falciparum contains conserved and variable epitopes recognized by inhibitory monoclonal antibodies. Mol Biochem Parasitol. 1986;18:183–195. doi: 10.1016/0166-6851(86)90037-x. [DOI] [PubMed] [Google Scholar]
- 38.Shaffer A L, Falk-Wade J A, Tortorelli V, Cigan A, Carter C, Hassan K, Hurley C K. HLA-DRw52-associated DRB1 alleles: identification using polymerase chain reaction amplified DNA, sequence specific oligonucleotide probes, and a chemiluminescent detection system. Tissue Antigens. 1992;39:84–91. doi: 10.1111/j.1399-0039.1992.tb01912.x. [DOI] [PubMed] [Google Scholar]
- 39.Siddiqui W A. An effective immunization of experimental monkeys against malaria parasite, Plasmodium falciparum. Science. 1977;197:388–390. doi: 10.1126/science.406671. [DOI] [PubMed] [Google Scholar]
- 40.Stephens H A F, Brown A E, Chandanayingyong D, Webster H K, Sirikong M, Longta P, Vangseratthana R, Gordon D M, Lekmak S, Rungruang E. The presence of the HLA class II allele DPB1*0501 in ethnic Thais correlates with an enhanced vaccine-induced antibody response to a malaria sporozoite antigen. Eur J Immunol. 1995;25:3142–3147. doi: 10.1002/eji.1830251123. [DOI] [PubMed] [Google Scholar]
- 41.Stowers A, Prescott N, Cooper J, Takacs B, Stueber D, Kennedy P, Saul A. Immunogenicity of recombinant Plasmodium falciparum rhoptry associated proteins 1 and 2. Parasite Immunol. 1995;17:631–642. doi: 10.1111/j.1365-3024.1995.tb01009.x. [DOI] [PubMed] [Google Scholar]
- 42.Stowers A, Spring K J, Saul A. Preparative scale purification of recombinant proteins to clinical grade by isotachophoresis. Bio/Technology. 1995;13:1498–1503. doi: 10.1038/nbt1295-1498. [DOI] [PubMed] [Google Scholar]
- 43.Stowers A W, Cooper J A, Erbhardt T, Saul A. A peptide derived from a B cell epitope of Plasmodium falciparum rhoptry-associated protein 2 specifically raises antibodies to rhoptry-associated protein 1. Mol Biochem Parasitol. 1996;82:167–180. doi: 10.1016/0166-6851(96)02730-2. [DOI] [PubMed] [Google Scholar]
- 44.Stowers A, Taylor D, Prescott N, Cheng Q, Cooper J, Saul A. Assessment of the humoral immune response against Plasmodium falciparum rhoptry-associated proteins 1 and 2. Infect Immun. 1997;65:2329–2338. doi: 10.1128/iai.65.6.2329-2338.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tolle R, Fruh K, Doumbo O, Koita O, M'Diaye M, Fischer A, Dietz K, Bujard H. A prospective study of the association between the human humoral immune response to Plasmodium falciparum blood-stage antigen gp190 and control of malarial infections. Infect Immun. 1993;61:40–47. doi: 10.1128/iai.61.1.40-47.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Trager W, Lanners H N, Stanley H A, Langreth S G. Immunization of owl monkey to Plasmodium falciparum with merozoites from cultures of a knobless clone. Parasite Immunol. 1983;5:225–231. doi: 10.1111/j.1365-3024.1983.tb00739.x. [DOI] [PubMed] [Google Scholar]
- 47.Troye-Blomberg M, Olerup O, Larsson A, Sjoberg K, Perlmann H, Riley E, Lepers J-P, Perlmann P. Failure to detect MHC class II associations of the human immune response induced by repeated malaria infections to the Plasmodium falciparum antigen Pf155/RESA. Int Immunol. 1991;3:1043–1051. doi: 10.1093/intimm/3.10.1043. [DOI] [PubMed] [Google Scholar]