Abstract
Background aims
We have previously demonstrated the safety and feasibility of adoptive cell therapy with CD45RA− memory T cells containing severe acute respiratory syndrome coronavirus 2–specific T cells for patients with coronavirus disease 2019 from an unvaccinated donor who was chosen based on human leukocyte antigen compatibility and cellular response. In this study, we examined the durability of cellular and humoral immunity within CD45RA− memory T cells and the effect of dexamethasone, the current standard of care treatment, and interleukin-15, a cytokine critically involved in T-cell maintenance and survival.
Methods
We performed a longitudinal analysis from previously severe acute respiratory syndrome coronavirus 2–infected and infection-naïve individuals covering 21 months from infection and 10 months after full vaccination with the BNT162b2 Pfizer/BioNTech vaccine.
Results
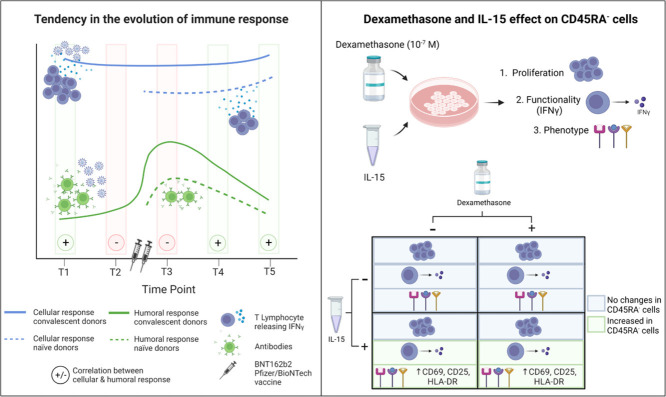
We observed that cellular responses are maintained over time. Humoral responses increased after vaccination but were gradually lost. In addition, dexamethasone did not alter cell functionality or proliferation of CD45RA- T cells, and interleukin-15 increased the memory T-cell activation state, regulatory T cell expression, and interferon gamma release.
Conclusions
Our results suggest that the best donors for adoptive cell therapy would be recovered individuals and 2 months after vaccination, although further studies with larger cohorts would be needed to confirm this finding. Dexamethasone did not affect the characteristics of the memory T cells at a concentration used in the clinical practice and IL-15 showed a positive effect on SARS-CoV-2-specific CD45RA- T cells.
Key Words: CD45RA− memory T cells, cellular immunity, COVID-19 treatment, humoral immunity
Graphical Abstract
Introduction
A novel coronavirus named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was identified in early 2020, causing a global coronavirus disease pandemic (coronavirus disease 2019 [COVID-19]). Infected patients experience a wide range of symptoms from asymptomatic to severe. Although vaccines have dramatically decreased infection rates and the number of deaths and hospitalizations, they are not 100% effective, and immunity is gradually lost [1]. Vaccines are also not completely effective at preventing coronavirus variants of concern (VOCs) [2,3].
Immune dysregulation is related to disease severity. The immune response includes humoral and cellular responses, with an adaptive immune response providing long-term protection. Plasma cells are an essential part of humoral immunity, secreting neutralizing antibodies and blocking viruses from entering cells. Cellular immunity (such as the T-cell adaptive immune response) is needed to control and eliminate SARS-CoV-2 infection [4], [5], [6]. Antibodies can protect against infection and reduce disease severity during the first months after vaccination or infection; however, memory T cells confer long-term protection even in cases in which the humoral response is poor [7,8]. Infected cells are eliminated by T cells, specifically cytotoxic T cells and T-helper cells, which coordinate the long-term immune reaction, collaborating in creating long-living plasma cells [9], [10], [11]. In addition, T-cell responses to SARS-CoV-2 can confer protection to SARS-CoV-2 variants due to the T cells’ ability to recognize parts of the virus less susceptible to mutational pressure [4,[12], [13], [14], [15], [16], [17]]. The entire immune system needs to respond to SARS-CoV-2 to efficiently eliminate the infection and avoid negative clinical outcomes.
Lymphopenia is a biomarker of disease severity, and profound lymphocytopenia correlates with severe clinical COVID-19 outcomes, whereas resolution of lymphopenia correlates with recovery [18], [19], [20], [21], [22], [23]. The mechanisms underlying lymphocytopenia are unclear, but impaired lymphocyte proliferation, lymphocyte apoptosis induction, bone marrow impairment and tissue redistribution have been suggested [4,20,24].
We and other authors have hypothesized that passive adoptive cellular immunotherapy with various T-cell subsets and/or natural killer (NK) cells might provide an effective mechanism to control COVID-19 infection due to their antiviral properties [25], [26], [27]. We have shown previously how to detect, isolate and produce, at a large clinical scale, CD45RA− memory T cells containing SARS-CoV-2–specific T cells from COVID-19–convalescent donors [25]. In the first-in-human clinical trial with adoptive cell therapy using allogeneic, off-the-shelf CD45RA− memory T cells from a convalescent donor, we demonstrated the safety and feasibility of treating hospitalized patients with moderate/severe COVID-19 [26]. The infusion of memory T cells to hospitalized patients with lymphopenia could help normalize T-cell counts and clear SARS-CoV-2 infection more efficiently than through the patient's immune system. We are currently assessing the efficacy of this treatment in a phase 2 clinical trial.
Several factors can alter the functionality of the infused CD45RA− memory T cells. The efficacy of adoptive T-cell therapy can be impaired by dexamethasone, a synthetic glucocorticoid widely used for treating human inflammatory diseases [28]. A short course of dexamethasone is the current standard of care treatment for COVID-19–hospitalized patients [29], and high doses of dexamethasone have been shown to have a negative impact on proliferation and cytokine production by T cells [30], [31], [32].
In addition, we have shown previously that interleukin-15 (IL-15), which is critically involved in the maintenance and survival of memory and naïve CD8+ T cells and NK cells [33,34], is producing an activating phenotype in the CD45RA− memory T cells of convalescent donors [25] We hypothesize that the stimulation of CD45RA− T cells with IL-15 overnight (O/N) will maintain T-cell functionality even in the presence of dexamethasone, making this approach a potential advanced cell therapy for patients with COVID-19.
This study seeks to determine several aspects of CD45RA− memory T cells for its use as an adoptive therapy for COVID-19 patients. On one hand, we have studied the durability and level of cellular immunity within CD45RA− memory T cells and the changes with vaccination, virus exposure and time. We therefore performed a longitudinal exploratory analysis of the SARS-CoV-2–specific humoral and cellular immunity within memory CD45RA− T cells in SARS-CoV-2–naive and previously infected individuals at different time points before and after two doses of the BNT162b2 BioNTech/Pfizer vaccine. On the other hand, we explored the effect of the current standard of care treatment on the adoptive cell therapy based on CD45RA− memory T cells and how this therapy could be improved to increase efficiency. For that, we evaluated the effect of dexamethasone and IL-15 on the proliferation, phenotype and functionality of SARS-CoV-2–specific CD45RA− memory T cells.
Methods
Study participants
The study included six COVID-19–recovered donors and four healthy controls (see supplementary Table 1), all of whom were health care workers. All of the recovered donors were positive for COVID-19 between March and April 2020 and had mild disease [25]. All donors tested negative for SARS-CoV-2, and none of the recovered donors experienced reinfections. The healthy donors had not been exposed to patients with COVID-19 and tested negative for anti-SARS-CoV-2 antibodies in June 2020. All participants granted their written consent, and the study was approved by the hospital institutional review board (IRB number: 254/20, La Paz University Hospital, Madrid, Spain). All participants were immunized with two doses of mRNA Pfizer vaccine between the end of January 2021 and the beginning of February 2021.
Cell processing
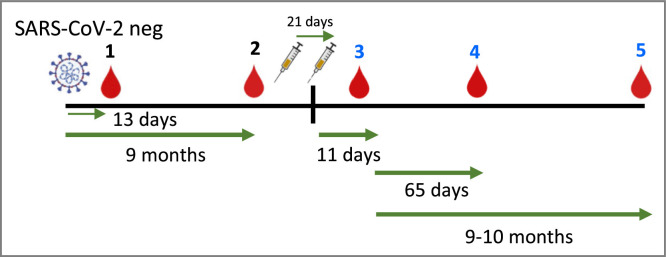
Paired plasma and peripheral blood mononuclear cells (PBMC) samples were collected from all individuals at various time points, as well as buffy coats when required (Figure 1 ). To summarize, PBMCs were isolated from peripheral blood by density gradient centrifugation using Ficoll–Paque (GE Healthcare, Chicago, IL, USA). Cells were preserved in 90% fetal bovine serum (Sigma-Aldrich, St. Louis, MO, USA) and 10% dimethyl sulfoxide and stored in liquid nitrogen. Plasma was isolated from the blood collection after centrifugation and stored at −80°C.
Figure 1.
Time point scheme for blood collection in recovered and control individuals. T1: A mean of 13 days after clearing COVID-19 infection, T2: 9 months after clearing COVID-19 infection, T3: 11 days after full BNT162b2 Pfizer/BioNTech vaccination T4: 65 days after full BNT162b2 Pfizer/BioNTech vaccination, T5: 9-10 months after full BNT162b2 Pfizer/BioNTech vaccination.
Proliferation assay of SARS-CoV-2–specific memory T cells in the presence of dexamethasone and IL-15
PBMCs from buffy coats were obtained from the Centro de Transfusion of Community of Madrid. The SARS-CoV-2 infection or vaccination status was not assessed. PBMCs from buffy coats were thawed and then incubated with or without IL-15 (50 ng/mL; Miltenyi Biotec, Bergisch Gladbach, Germany) overnight at 37°C in TexMACS Medium (Miltenyi Biotec) supplemented with 10% Human Serum Type AB (Sigma Aldrich) and 100 U/mL penicillin–100 μg/mL streptomycin sulphate (Sigma Aldrich). The cells were then washed and approximately 2 million cells per condition were resuspended in 1 mL of supplemented medium and 100 µL of a carboxyfluorescein succinimidyl ester (CFSE) stock dilution (final concentration 5 µmol/L) (CFSE, CellTrace CFSE Cell Proliferation Kit; Invitrogen, Waltham, MA, USA). The cells were then incubated for 10 min in the dark and washed three times. Lastly, the cells were cultured in a P24 plate in 1 mL of supplemented medium with 1.5% v/v Phytohemagglutinin M form (Thermo Fisher Scientific, Waltham, MA, USA) plus 0.05 µg/mL mouse anti-human CD3 (Clone OKT3; BD Biosciences, Franklin Lakes, NJ) and 5 µg/mL CD28/CD49d Purified (Clone L293 L25 RUO GMP; BD Biosciences). In addition, various concentrations of dexamethasone (Kern Pharma 4 mg/mL solution for injection EFG) (0, 10−7 mol/L, 10−6 mol/L and 10−5 mol/L) were added to the supplemented medium. PBMCs without IL-15 and without dexamethasone were employed as the negative control (see supplementary Figure 1). The dexamethasone doses were calculated considering that the patients were administered 6 mg/day of the glucocorticoid, and the volume of the drug distribution was calculated according to DrugBank's online dexamethasone information page (https://go.drugbank.com/drugs/DB01234). According to these data and previous publications [35,36], 6 mg of dexamethasone administered to an adult of approximately 60–70 kg corresponds to 1–2 × 10−7 mol/L.
The cells were incubated at 37°C for 72 h, and a proliferation assay was conducted to determine whether high doses of the drug decrease their division or proliferation index. In addition to CFSE, cells were stained with the following cell surface antibodies to define different subpopulations: CD27 APC (BD Pharmingen), CD3 Viogreen (Miltenyi Biotec), CD4 PE-Cy7 (BD Pharmingen), CD8 APC-Cy7 (BD Pharmingen), L/D 7AAD (BD Pharmingen), CD45RA Alexa Fluor 700 (BD Pharmingen), CD127 PE-CF594 (BD Horizon) and CD25 BV421 (BD Horizon). Cell acquisition was then performed using a Navios cytometer (Beckman Coulter), acquiring a mean of 200,000 cells. The analysis was performed using FlowJo 10.7.1 software (FlowJo LLC).
Proliferation cells’ capacity under different conditions was compared using two parameters: the proliferation index, which is the total number of divisions divided by the number of cells that went into division; and the relative geometric mean fluorescent intensity (GeoMFI), expressed as ratio GeoMFI /(GeoMFI of the negative control) (cells without dexamethasone and without IL-15).
Detection of SARS-CoV-2–specific memory T cells by interferon-gamma assay
The assay was performed as previously described [25]. In summary, the peptide pools were short 15-mer peptide pools with 11 amino acid overlaps that can bind MHC class I and II complexes and are therefore able to stimulate CD4+ and CD8+ T cells. The peptide pools cover the immunodominant sequence domains of the surface glycoprotein S, the complete sequence of the nucleocapsid phosphoprotein N and the membrane glycoprotein M (GenBank MN908947.3, Protein QHD43416.1, Protein QHD43423.2, Protein QHD43419.1; Miltenyi Biotec). The cells were rested O/N at 37°C and after 5 h of stimulation with individual SARS-CoV-2 peptide pools or combination of peptide pools (M, N, S), the cells were labeled with interferon-gamma (IFN-γ) catch reagent (Human IFN-γ Secretion Assay-Detection Kit; Miltenyi Biotec), and the cell surface–bound IFN-γ was targeted using the IFN-γ phycoerythrin antibody. Background subtraction was performed from parallel unstimulated cultures. A positive sample included the following acceptance criteria: 0.1% of IFN-γ+ cells out of the total cell population with a minimum of 150,000 events analyzed, at least twice the number of IFN-γ+ cells in the sample than in the negative control and a positive control based on plate-bound cells stimulated with mouse anti-human CD3 and co-stimulated with purified CD28/CD49d. Basal IFN-γ production by PBMCs was included as a background control in the absence of stimulation and co-stimulation.
The experiments with and without IL-15 incubation O/N and with different concentrations of dexamethasone were performed as described previously. After incubation, cells where stained using the following fluorochrome-conjugated anti-human surface antibodies: CD45RA FITC, CD27 APC, CD3 VioGreen, CD4 PECy7, CD8 APC Cy7, L/D 7AAD and IFN-γ phycoerythrin. Cell acquisition was then performed using a Navios cytometer (Beckman Coulter, Indianapolis, IN, USA), acquiring a mean of 200,000 cells. The analysis was performed using FlowJo 10.7.1 software (FlowJo LLC, Ashland, OR, USA).
Phenotype of memory T cells containing SARS-CoV-2–specific T cells determined by flow cytometry assay
The phenotype assay was performed as previously described [25]. In summary, we stained the cell surface for 20 min at 4°C using the following fluorochrome-conjugated antihuman antibodies: CD45RA FITC, CD27 APC, CD3 VioGreen, CD4 PECy7, CD8 APC Cy7 and L/D 7AAD. We employed other antibodies for specific cell populations: CD25 BV421 (BD Horizon, Franklin Lakes, NJ, USA) and CD127 PE-CF594 (BD Horizon) for regulatory T cells (Treg); HLA-DR BV421 (BD Pharmingen, San Diego, CA, USA), CD69 PE (Miltenyi Biotec) and CD25 BV421 (BD Horizon) for activation makers; CD279 (PD1) AF700 (BioLegend, San Diego, CA, USA) and NKG2A BV421 (BD OptiBuild) for exhaustion markers; and, CD103 BV421 (BD Horizon) and CCR7 PE-CF594 (BD Horizon) for chemokine receptor and integrin markers.
The experiments with and without IL-15 incubation O/N and with different concentrations of dexamethasone were performed as described previously. After the staining, cell acquisition was performed using a Navios cytometer (Beckman Coulter), acquiring a mean of 200,000 cells. The analysis was performed using FlowJo 10.7.1 software (FlowJo LLC).
Analysis of humoral responses
For the detection of the receptor-binding domain (RBD), spike 1 (S1), spike 2 (S2) and nucleocapsid (N) antibodies, we used the BioPlex 2200 SARS-CoV-2 IgG panel. To summarize, K3 ethylenediaminetetraacetic acid plasma samples from patients were mixed with fluoromagnetic dyed beads, each coated independently with the four different antigens, and processed in the BioPlex 2200 system, as per the manufacturer's instructions (Bio-Rad Laboratories, Hercules, CA, USA). Samples were first tested undiluted and, for results greater than 100 U/mL, serially diluted to determine the exact antibody level. Final results are shown in units (World Health Organization [WHO] BAU/mL) by transforming U/mL with the first WHO international standard for binding activity of anti-SARS-CoV-2 immunoglobulins to RBD, S1 and nucleocapsid proteins.
We also employed a test designed to mimic plasma neutralization for the interaction of RBD SARS-CoV-2 and human angiotensin-converting enzyme 2 (ACE2) by competitive chemiluminescence immunoassay (Snibe, Shenzhen, China). To summarize, donor samples, buffer, magnetic microbeads coated with hACE2 and SARS-CoV-2-RBD ABEI-labeled antigen were mixed. In the incubation, immunoglobulins of the sample compete with RBD-ACE2 binding, and the RBD label is recognized by chemiluminescence reagents after applying a magnetic device and washing process. All steps were performed in a Maglumi 2000 system. Samples were first tested undiluted and, for results greater than 30 µg/mL, serially diluted to determine the exact neutralizing capacity. The final results are shown in units (WHO IU/mL) by transforming µg/mL with the WHO standard (20/136).
Statistical analysis
Given the nature of the correlated data, lymphocyte averages were estimated using least squares means with a linear mixed-effects model employing the restricted maximum likelihood method. Time, controls, recovered donors and the interaction were included as the fixed effect, and the intercepts were included as the random effect. The least squares means were estimated and compared, and the Bonferroni adjustment was considered for multiple comparisons. The global effect of each variable in the model was assessed with the type 3 test. All statistical tests were considered bilateral, with statistically significant values having a P < 0.05. The data were analyzed with the statistical program SAS Enterprise Guide 8.2 (SAS Institute Inc., Cary, NC, USA).
To analyze the differences in proliferation, functionality (IFN-γ production) and phenotype of PBMCs due to exposure to increasing concentrations of dexamethasone in different cell subpopulations, we performed a one-way or two-way analysis of variance test. To analyze the differences due to the absence or presence of IL-15, we performed a Wilcoxon matches-pairs signed rank test for CFSE experiments and a Friedman test with paired data with Dunn's multiple comparisons test in all other cases. We employed GraphPad Prism (GraphPad v.8.0 Software, San Diego, CA, USA) for the analysis. P-values <0.05 were considered statistically significant. The quantitative variables are expressed as mean ± standard error, whereas the qualitative variables are expressed as percentages (%). For the exploratory analysis, Spearman's correlation coefficient was calculated to evaluate the linear association between quantitative variables; ±0.8–1 correlation values were considered a strong association.
Results
Participants and sample collection
Ten participants were administered two doses of the BNT162b2 Pfizer/BioNTech vaccine between January 10 and February 9, 2021, at La Paz University Hospital, Madrid, Spain. Six individuals were previously infected [25], and four were defined as SARS-CoV-2 naïve (see supplementary Table 1). The mean age was 37 years (range 23–41 years) for the recovered donors and 29.7 years (range 26–34 years) for the infection-naïve controls. Seven donors were women, and three were men. We obtained PBMCs at five time points, a mean of 13 days after the individuals who had recovered from COVID-19 tested negative (by polymerase chain reaction) for SARS-CoV-2 (T1); between 9 and 10 months after T1, which was just before the first dose of the vaccine (T2); 11 days after the second dose of the mRNA vaccine (T3), 65 days after the second dose of the mRNA vaccine (T4), and between 9 and 10 months after the second dose (T5) (Figure 1). The data at T1 for all participants have been already published by Ferreras et al. [25]. For one of the control donors, we collected data only at time points 1 and 2 because the donor was infected after the first dose of the vaccine. For one of the recovered donors, we did not collect data at the last time point (T5) because the donor was taking anakinra to treat myocarditis.
Quantification of memory T cells specific for SARS-CoV-2 immunity
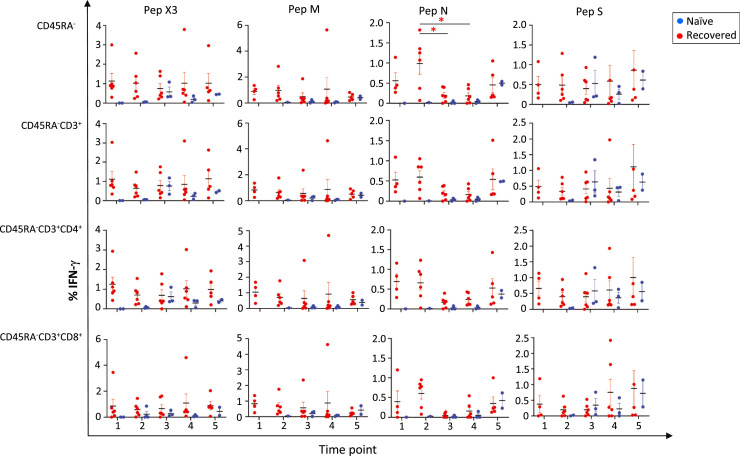
We first analyzed the changes in the percentage of T-cell subsets at the various time points and the presence of SARS-CoV-2–specific T cells within the CD45RA− memory T-cell subpopulation by the release of IFN-γ upon in vitro stimulation of PBMCs to the individual or combination of peptide pools (see supplementary Figure 2). As expected, there were no changes in the various T-cell subpopulations (see supplementary Table 2). In general, there were no statistically significant differences in cellular response (Figure 2 , supplementary Table 2 and supplementary Figure 3).
Figure 2.
Detection of SARS-CoV-2–specific T cells within the CD45RA– memory T cells and subsets. IFN-γ was measured by flow cytometry after exposure to the single SARS-CoV-2 peptides (M, N and S) and the peptide pools (PepX3). 1, 2, 3, 4 and 5 correspond to the different time points described in Figure 1. Independent data for each naïve control are shown by the blue dots, and independent data for each recovered donor are shown by the red dots. Mean and standard error of the mean are shown. *P < 0.05.
For the N single SARS-CoV-2 peptide in the recovered donors, the greatest cellular immune response occurred after COVID-19 infection. In the CD45RA− T-cell population, the mean IFN-γ expression decreased from T2 to T3 (P = 0.0382) and from the T2 to T4 time points (P = 0.0311). There were no statistically significant differences in the other CD45RA− subsets: CD4+ and CD8+, central memory T cells (TCM) (CD4+CD27+ and CD8+CD27+) and effector memory T cells (TEM) (CD4+CD27− and CD8+CD27−) subpopulations (Figure 2 and supplementary Figure 3).
For the M and S SARS-CoV-2 individual peptide pools or the combination of peptide pools, SARS-CoV-2–specific memory T-cell responses were maintained over time. Interestingly, there was no increase in cellular responses after full mRNA immunization in the recovered individuals.
We detected no cellular immune response in the control participants before the vaccination (Figure 2, supplementary Table 2 and supplementary Figure 3). However, we observed induction of SARS-CoV-2 T-cell responses to the S peptide pool at an early time point after mRNA immunization (T3), and the response was maintained at time points T4 and T5 within all CD45RA− subpopulations (Figure 2 and supplementary Figure 3). Although the sample size was small, this was expected because the S protein is the target for the mRNA vaccine. We also observed that mRNA vaccination of COVID-19–naïve participants induced an immune cellular response to the S peptide pools similar to the memory T-cell responses from recovered donors at the same time point. Surprisingly, infection-naïve individuals showed cellular responses to the M and N SARS-CoV-2 peptide pools 10 months after the second dose of the mRNA vaccine. When examining both groups, we observed a tendency toward greater cellular immune responses in the recovered individuals than in the control group (Figure 2 and supplementary Figure 3).
In summary, memory T-cell responses tended to be greater in the recovered individuals at all time points due to a pre-existing memory T-cell population formed after SARS-CoV-2 infection, this cellular response that was maintained even 10 months after mRNA immunization in both groups (recovered and infection-naïve donors). In addition, COVID-19 infection produced memory T-cells responses to the combination of SARS-CoV-2 peptide pools, whereas in our control group, the vaccine was able to only produce Spike-specific SARS-CoV-2 memory T-cell responses.
Humoral response
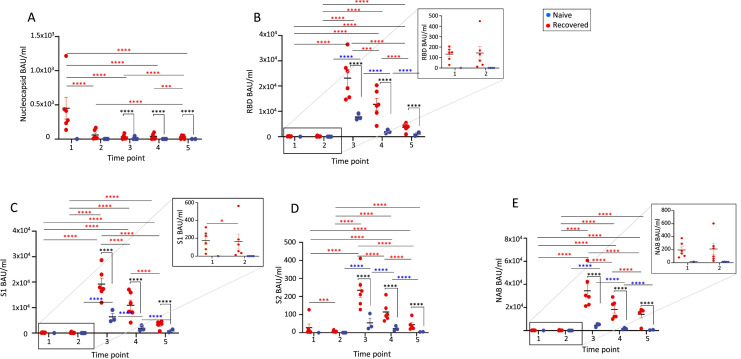
We analyzed the antibody titers to nucleocapsid (N) representing prior SARS-CoV-2 infection, RBD, S1 and S2 domains of the Spike, representing a response to either previous infection or vaccination by mRNA vaccine, and levels of the neutralizing antibody (NAB). We observed antibody production in both groups (recovered and control donors) (Figure 3 ). As expected, antibody production was observed in previously SARS-CoV-2–infected individuals at T1 but was not detected in COVID-19–naïve participants.
Figure 3.
Antibody titers for (A) nucleocapsid, (B) RBD, (C) S1, (D) S2 and (E) NAB at different time points in recovered and control individuals. 1, 2, 3, 4 and 5 correspond to the different time points described in Figure 1. Independent data for each naïve control are shown by the blue dots, and independent data for each recovered donor are shown by the red dots. P values different from P < 0.0001 are shown in the figure. *P value in recovered donors, *P value in control donors, *P value between both groups. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < <0.0001. Mean and standard error of the mean are shown.
In the recovered individuals, vaccination dramatically increased antibody titers for RBD, S1, S2 and NAB. For the N antibody, the greatest value was obtained after COVID-19 infection (T1), decreasing at T2 (Figure 3A). Vaccination did not boost the humoral immune response, with values decreasing at T3, T4, and T5 with respect to T1. There was also a significant decrease from T4 to T5, with values below T1.
For the other antibodies tested (RBD, S1, S2 and NAB), we observed vaccine-induced antibody responses with a markedly increased antibody production at T3 that gradually decreased at T4 and T5. For S2, the values at T5 were similar to the pre-vaccination values (T1) (Figure 3B–E). In the COVID-19–naïve participants, the vaccine did not elicit IgG N antibody responses as expected at any tested time point (T3, T4 and T5). For RBD, S1, S2 and NAB, early antibody production was observed at T3, with the response decreasing over time.
Values after vaccination (T3–T5) were significantly greater in the recovered individuals than in the infection-naïve controls at all time points, with a decrease in antibody titers over time. Interestingly, the antibody titers for the control individuals after full immunization (T3 and T4) seemed to be much greater than in the recovered donors after COVID-19 infection (T1 and T2; RBD, P < 0.0001; S1, P < 0.0001; S2, P = 0.0175; NAB, P < 0.0001) (Figure 3B–E).
These results showed that the BNT162b2 mRNA vaccine induced significantly greater antibody titers in the recovered donors compared with the infection-naïve individuals at all time points. However, those values decreased gradually over time in both groups, with values at 10 months after immunization resembling pre-vaccination values.
Correlation between humoral and cellular response in the recovered donors
To assess the correlation between cellular and humoral immunity over time, we performed an exploratory analysis of non-parametric Spearman's rho correlation of humoral and cellular responses in the recovered donors. We compared the humoral parameters in plasma with the cellular response to the combination of SARS-CoV-2 peptide pools (Figure 4 ) at the different time points.
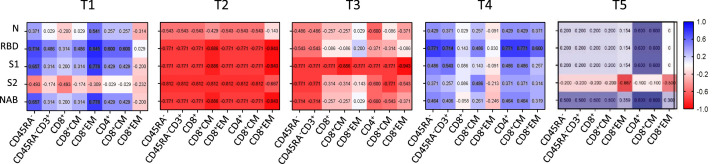
Figure 4.
Cellular and humoral immunity correlation responses in recovered donors. The nonparametric Spearman test was used for correlation analysis. A matrix heatmap shows the correlation between cellular and immune humoral responses. Cellular responses were obtained upon SARS-CoV-2 peptide pools stimulation. The Spearman's correlation matrices show the relationship between N, RBD, S1, S2 and NAB antibodies and the different CD45RA- cell subsets. T1, T2, T3, T4 and T5 correspond to the time points shown in Figure 1. Values inside the squares indicate the correlation coefficient. All data points are shown. CM, central memory; EM, effector memory. Blue color means a positive correlation, red color means a negative correlation. The intensity of the color indicates how strong this correlation is.
At T1, after the COVID-19 infection was cleared, our analysis showed a pattern of positive cellular and humoral correlation in most of the CD45RA− subsets for N, RBD, S1 and NAB antibodies. In contrast, the data 9 months after the infection and early after vaccination (T2 and T3) displayed a negative cellular and humoral correlation. However, at T4 a tendency to a positive correlation was again observed for all the studied antibodies that although less pronounced was maintained at T5. Interestingly, we observed a similar pattern at T5 to the one after COVID-19 infection (Figure 4). These data suggest that after SARS-CoV-2 infection antigen-specific T-cell responses induced upon peptide pool stimulation correlated with humoral responses. This positive correlation is lost over time and is only recovered and maintained long term after a complete vaccination regimen with BNT162b2 vaccination.
Effects of dexamethasone and IL-15 on SARS-CoV-2–specific CD45RA− memory T cells
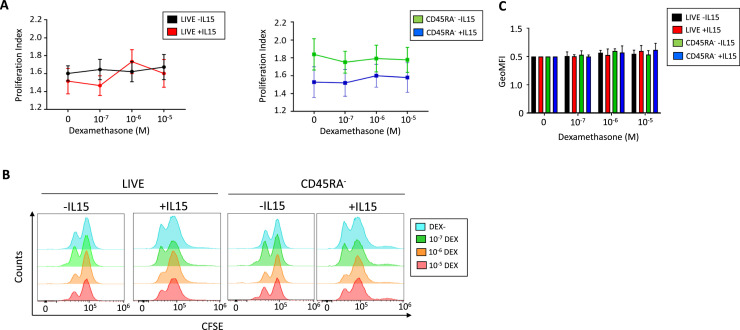
We analyzed the effect of dexamethasone on the CD45RA− memory T-cell subsets and the proliferation, functionality and phenotype of SARS-CoV-2-specific CD45RA− memory T cells. No significant differences were observed in proliferation at the studied dexamethasone concentrations in the absence or presence of IL-15 in the live cell's gate or the CD45RA− memory T cells (Figure 5 A–B). Also, the geometric mean fluorescent intensity did not show any differences (Figure 5C).
Figure 5.
Effect of dexamethasone and IL-15 on total PBMCs and CD45RA− memory T-cell proliferation. PBMCs were cultured in presence or absence of IL-15 (50 ng/mL) O/N. Then, cells were washed and labeled with CFSE and incubated for 72 h with different concentrations of dexamethasone (0, 10−7 mol/L DEX, 10−6 mol/L DEX and 10−5 mol/L) for 72 h. (A) Proliferation Index of total live cells and CD45RA− memory T cells. The PI is calculated as total number of divisions divided by the number of cells that went into division. One-way analysis of variance was performed to analyze differences due to different concentrations of dexamethasone previously cultured with IL-15 O/N. Mean ± SEM, N = 5–9 independent donors. (B) Representative determination of live cells and CD45RA− memory T cells proliferation by flow cytometry with or without IL-15 and in presence of increasing concentrations of dexamethasone. (C) Geometric mean fluorescence intensity (GeoMFI), expressed as ratio GeoMFI /(GeoMFI of the negative control) (cells without dexamethasone and without IL-15). Mean ± SEM of the ratios are represented. SEM, standard error of the mean.
We observed no changes in the percentage of each T-cell subset in the presence of dexamethasone after an O/N or 72-h incubation regardless an IL-15 pre-incubation (Table 1 ). In the absence of a previous IL-15 incubation, an O/N incubation with dexamethasone did not change the frequency of Treg, and only the greatest concentration showed a tendency to decrease Treg expression (11.3 versus 9.8, 8.2 and 6.9) (Table 1). This effect, although not statistically significant, was more pronounced after 72 h of incubation with dexamethasone (19.3 versus 11, 6.4 and 3.3) (Table 1). On the contrary, in all conditions studied, IL-15 increased the expression of Treg, and dexamethasone did not have any effect with values similar to the ones obtained with no dexamethasone (Table 1). These changes in Treg expression are the result of an activation rather than an increase in cell proliferation, since no changes were observed in proliferation index (data not shown). In general, at 72 h the cells previously exposed to IL-15 showed a lower Treg induction regardless the incubation with dexamethasone comparing with the same dexamethasone concentration after an O/N incubation. Together, these findings indicate that IL-15 alone could increase the activation state of Tregs, while dexamethasone had no effect on this activation.
Table 1.
Effect of dexamethasone on the percentage of CD45RA− memory T cell subpopulations after an O/N and 72 h-incubation with PBMCs previously treated O/N with and without IL-15.
| WO DEX O/N |
10−7 mol/L O/N |
10−6 mol/L O/N |
10−5 mol/L O/N |
|||||
|---|---|---|---|---|---|---|---|---|
| −IL15 | +IL15 | −IL15 | +IL15 | −IL15 | +IL15 | −IL15 | +IL15 | |
| O/N | ||||||||
| CD45RA− | ||||||||
| % CD45RA− | 25.2 | 18.6 | 30.1 | 21.6 | 30.8 | 21.4 | 33.0 | 28.1 |
| % CD3+ | 88.8 | 90.3 | 89.8 | 88.0 | 89.2 | 86.8 | 90.5 | 86.4 |
| % CD4+ | 76.8 | 79.2 | 75.8 | 81.3 | 77.5 | 82.6 | 78.6 | 84.1 |
| % CD8+ | 17.1 | 19.5 | 18.5 | 17.4 | 16.9 | 16.1 | 15.3 | 14.6 |
| CD45RA−CD4+ | ||||||||
| CD27+ (CM) | 80.4 | 80.5 | 80.9 | 77.7 | 80.4 | 78.4 | 78.0 | 74.7 |
| CD27− (EM) | 18.7 | 19.5 | 18.5 | 22.3 | 21.8 | 21.5 | 20.2 | 25.3 |
| CD127lowCD25+ (Treg) | 11.3 | 40.7*** | 9.8 | 42.0** | 8.2 | 40.1*** | 6.9 | 36.1*** |
| CD45RA−CD8+ | ||||||||
| CD27+ (CM) | 74.1 | 74.2 | 75.3 | 72.8 | 75.6 | 73.7 | 69.4 | 72.3 |
| CD27− (EM) | 24.8 | 25.8 | 23.8 | 27.2 | 23.8 | 26.3 | 24.2 | 27.7 |
| WO DEX 72 h |
10−7 mol/L 72 h |
10−6 mol/L 72 h |
10−5 mol/L 72 h |
|||||
| –IL15 | +IL15 | –IL15 | +IL15 | –IL15 | +IL15 | –IL15 | +IL15 | |
| 72-h incubation | ||||||||
| CD45RA− | ||||||||
| % CD45RA− | 19.1 | 20.1 | 21.8 | 22.8 | 23.5 | 23.4 | 23.5 | 25.3 |
| % CD3+ | 95.7 | 93.5 | 85.1 | 92.1 | 91.9 | 90.9 | 91.8 | 91.8 |
| % CD4+ | 88.7 | 77.5 | 84.0 | 79.7 | 85.2 | 78.5 | 84.5 | 80.2 |
| % CD8+ | 10.4 | 20.9 | 12.9 | 19.0 | 12.9 | 20.3 | 13.2 | 18.6 |
| CD45RA−CD4+ | ||||||||
| CD27+ (CM) | 71.0 | 75.7 | 73.0 | 76.9 | 76.9 | 82.0 | 76.9 | 81.2 |
| CD27− (EM) | 29.0 | 24.3 | 27.0 | 23.1 | 23.1 | 17.9 | 23.1 | 18.8 |
| CD127lowCD25+ (Treg) | 19.3 | 26.4 | 11.0 | 25.9*** | 6.4 | 27.9*** | 3.3 | 27.7*** |
| CD45RA−CD8+ | ||||||||
| CD27+ (CM) | 82.5 | 72.9 | 79.7 | 74.6 | 79.7 | 76.6 | 79.2 | 75.7 |
| CD27− (EM) | 17.5 | 26.9 | 20.3 | 25.4 | 20.3 | 23.4 | 20.8 | 24.3 |
The experiment has been designed following the scheme in supplemental Figure 1. Mean and standard error of the mean are shown. N = 3–4.
CM, central memory; EM, effector memory; IL-15, interleukin-15; O/N, overnight; PBMCs, peripheral blood mononuclear cells; Treg, regulatory T-cells.
**P ˂ 0.005.
***P ˂ 0.0001.
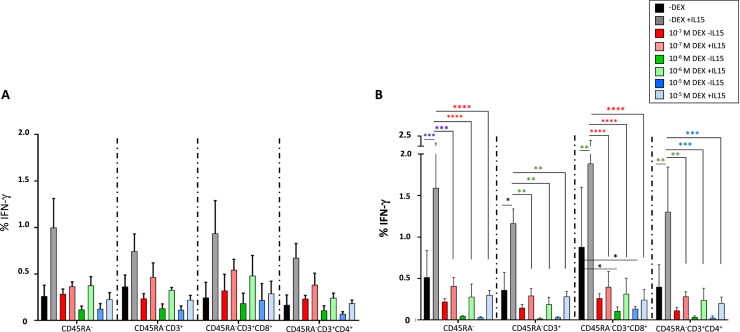
We then analyzed IFN-γ production after exposure to the combination of SARS-CoV-2 peptide pools in three separate situations: first, IFN-γ production after an O/N incubation of IL-15; second, IFN-γ production after an O/N incubation with dexamethasone and third, the effect of dexamethasone on IFN-γ production after with a previous incubation with IL-15 (Figure 6 ). Figure 6A shows that when the cells were previously incubated with IL-15 in the absence of dexamethasone there was a tendency to increase the percentage of IFN-γ in all CD45RA− memory T-cell subsets studied (Figure 6A black and gray bars). After an O/N incubation with dexamethasone, we observed no changes in IFN-γ release in the different T-cell subpopulations at the dexamethasone concentrations resembling those in clinical practice (Figure 6A red bars). Although greater concentrations tended to decrease IFN-γ values, there were no statistically significant differences (Figure 6A dark green and dark blue bars). When the cells were cultured with dexamethasone but previously exposed to IL-15, we observed IFN-γ values similar to the values with no dexamethasone and no previous IL-15 pre-incubation (Figure 6A light bars: red, green and blue).
Figure 6.
Effect of IL-15 and dexamethasone on the IFN-γ production after co-culture with the combination of the SARS-CoV-2 peptide pools. IFN-γ production of SARS-CoV-2 specific CD45RA− memory T cells after co-culture with the combination of peptide pools in the presence or absence of IL-15 O/N and (A) dexamethasone O/N or (B) for 72 h. Several subsets are represented: CD45RA−CD3+, CD45RA−CD3+CD8+, CD45RA−CD3+CD4+. Black bars represent cells cultured in the absence of dexamethasone, dark color bars represent cells incubated with the different concentrations of dexamethasone and no IL-15 previous incubation, light color bars represent cells cultures with the different concentrations of dexamethasone previously exposed to IL-15. A Two-way ANOVA test was performed *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 N = 4. All donors had a previous COVID-19 infection. Mean and ± standard error of the mean.
We then analyzed the same effect after a 72-h incubation. Figure 6B shows a statistically significant increase in IFN-γ values when the cells were previously incubated with IL-15 in the absence of dexamethasone (Figure 6B black and gray bars). A 72-h incubation with dexamethasone alone tended to decrease IFN-γ release at all the concentrations used and in all the CD45RA− memory T-cell subsets, and this effect was more pronounced at greater concentrations (Figure 6B dark bars: red, green and blue), specifically in the CD45RA−CD3+CD8+ subset at 10−6 and 10−5 mol/L (P = 0.01). When the cells were cultured with dexamethasone for 72 h and previously exposed to IL-15, we observed IFN-γ values comparable with the values with no dexamethasone and no previous IL-15 pre-incubation, similar to data for an O/N incubation (Figure 6B light red, green and blue).
These results suggest that incubation of memory T cells with dexamethasone concentrations used in clinical practice for patients hospitalized with COVID-19 did not affect the functionality of SARS-CoV-2–specific CD45RA− memory T cells based on IFN-γ release. Moreover, a previous incubation of CD45RA− memory T cells with IL-15 counteracted the effect of greater dexamethasone concentrations when co-cultured with the cells for 72 h.
We then performed a Friedman test to analyze the global effect of IL-15 regardless of the dexamethasone concentration at both time points, showing that the increased IFN-γ release was due to IL-15 incubation. We observed statistically significant differences in the following subsets: CD45RA− (P = 0.0068 O/N, P = 0.013 at 72 h), CD45RA−CD3+ (P = 0.0152 O/N, P = 0.024 at 72 h) and CD45RA−CD3+CD4+ (P = 0.033 O/N, P = 0.019 at 72 h), with no differences in the CD45RA−CD3+CD8+ subsets.
Lastly, we analyzed the changes in phenotype induced by dexamethasone and IL-15. We studied a panel of activation, exhaustion, and homing cell surface markers (supplementary Table 3 and supplementary Figures 4, 5 and 6).
The cells cultured with IL-15 showed a 2.2-fold and 5.0-fold increase in the activation markers CD69 and CD25high, respectively, in the CD45RA− memory T-cell subpopulation. A dexamethasone O/N incubation did not have any effect on this activation (supplementary Figure 4A). However, the CD25high expression showed a tendency to decrease after 72 h with dexamethasone dose-dependently in the absence of a previous incubation with IL-15 (supplementary Figure 4B). Also, no changes were observed in the presence of dexamethasone after O/N or 72-h incubation in the activation marker HLA-DR, exhaustion (NKG2A, PD1) or homing markers (CD103, CCR7) (supplementary Figures 4, 5 and 6, and supplementary Table 3). To analyze the global effect of IL-15, we performed a Friedman test and observed statistically significant differences for the activation markers HLA-DR, CD25high and CD69 in the CD45RA−, CD45RA−CD3+, CD45RA−CD3+CD4+, CD45RA−CD3+CD8+ cell subsets after an O/N incubation with dexamethasone and in CD45RA−CD3+, CD45RA−CD3+CD4+, CD45RA−CD3+CD8+, after a 72-h incubation with dexamethasone (supplementary Table 4). There were also no significant changes in the percentage of the different various T-cell subsets (data not shown). In general, IL-15 increased the activation state of cells, whereas the exhaustion or homing markers remain unchanged. Dexamethasone did not alter the expression of these markers on the CD45RA− cell surface subsets.
Discussion
Vaccination has dramatically decreased the number of deaths and hospitalizations resulting from SARS-CoV-2 infection; however, specific and effective therapeutic anti-viral therapies for COVID-19 are lacking. New COVID-19 pandemic waves and the emergence of SARS-CoV-2 VOCs with greater transmissibility risk, mortality rates and ability to evade previously acquired immunity [37] have shown that the pandemic has not yet ended and will be around for a long time.
There have been two very well-differentiated time points in the SARS-CoV-2 pandemic: before and after vaccination, with widely studied cellular and humoral responses [4,15]. We and other authors have shown the antiviral properties of CD45RA− memory T cells in hematopoietic stem cell transplantation and COVID-19 settings [25,[38], [39], [40], [41]]. We have demonstrated the safety and feasibility of adoptive cell therapy using CD45RA− memory T cells containing SARS-CoV-2–specific T cells in patients hospitalized with COVID-19. In this approach, we pursued to increase the pool of lymphocytes in patients with lymphopenia, exerting an antiviral effect on the coronavirus and protecting the patient from other viruses that the donor encountered [25]. In this trial, the patients were administered memory T cells from a convalescent and unvaccinated donor [26], who was chosen based on human leukocyte antigen compatibility and cellular response to SARS-CoV-2 peptide pools. On the basis of the results presented here, the donor's CD45RA− memory T cells had a cellular response, and a positive cellular and humoral correlation; however, the antibody titers were low because the donor had not been administered any vaccine (T1).
The present study sought to determine, on the one hand, who would be the best donor candidate for adoptive cell therapy for patients with COVID-19 in the current scenario of infections and vaccinations. To this end, we analyzed the SARS-CoV-2–specific T-cell responses within the CD45RA− memory T-cell subpopulation and subsets at different time points, covering 21 months after SARS-CoV-2 infection in the recovered donors and between 9 and 10 months after a full BNT162b2 BioNTech/Pfizer vaccination in both groups. We also analyzed the changes in humoral response by measuring antibody titers for five specific antibodies. In addition, we studied the effect of dexamethasone and the T-cell activator IL-15 on these cells to analyze the effect of these compounds on the living drug.
Our data show a tendency toward a greater cellular response in recovered individuals at all time points that is maintained over time. Humoral responses are boosted after vaccination, with greater titers in the recovered individuals than in the controls; however, these responses are gradually lost.
T-cell responses against SARS-CoV-2 have been studied extensively, showing responses against almost all proteins of the viral proteome [17,42], with spike-specific T-cell responses dominated by CD4+ T cells [4]. Our product (CD45RA−) is mainly composed of CD4+ T cells [25]. Several authors have shown the effects of the first and second immunization on cellular and humoral responses and that CD4+ T cells are primed by the vaccine [5]. CD4+ T cells are required to promote B-cell antibody production, enhance and maintain the responses of CD8+ T cells; orchestrate immune responses against a wide variety of pathogenic microorganisms and contribute to viral elimination by a direct cytotoxic effect on virus-infected cells [43], [44], [45], [46], [47]. In convalescent donors, SARS-CoV-2–specific CD4+ T cells produce a Th1 response, include cells with lymphoid and tissue-homing potential, are long-lived and are capable of proliferation [48]. CD45RA− memory T cells are composed of TCM or TEM cell subpopulations. The TCM cells can home in secondary lymphoid tissues, proliferate and create a new round of effector T cells [49], whereas TEM subsets are the first responders to infection, with a quick and strong response to pathogens and can home in on peripheral lymphoid tissues [50]. Unlike a number of authors, we observed no difference in the CD4+ TEM and TCM responses after vaccination, although this could be the result of our small sample size.
Current vaccines are targeting the S protein because it has shown high antigenicity and the ability to induce robust immune responses [51,52]. Our data are in agreement with those, indicating that the mRNA vaccine generates spike-specific memory CD4+ and CD8+ T-cell responses [5,[53], [54], [55]]. In COVID-19–naïve individuals, we observe spike-specific CD45RA− memory T-cell responses as early as 10 days after the second dose, as has been reported in greater cohorts [56,57]. Recovered donors retained SARS-CoV-2–specific T cells within the CD45RA− memory T cells that recognize different parts of the coronavirus, the M, N and S peptide pools. In line with previous publications, our data suggest that vaccine immunization does not increase the cellular responses in recovered individuals and that pre-existing immunity due to infection is maintained over time [58].
Antibody weaning has been observed 10 months after infection in non-immunized individuals [59,60] and after mRNA immunization. As others have published, we have observed that antibody levels peak after immunization in previously SARS-CoV-2–infected individuals [58,61,62]; after that, antibody production decrease dramatically over time [60,[63], [64], [65], [66]], being more profound in infection-naïve donors than in recovered individuals. Consistent with published reports, we observe no N antibody titers for SARS-CoV-2–naïve participants, the level for recovered donors peak after infection and the values are not increased by immunization with mRNA [67], [68], [69].
A number of COVID-19–naïve individuals show cellular immunity for the N and S peptides at T5. Although this might be consistent with a breakthrough infection, we would expect an increase in antibody titers after infection, which is not the case here. One possible explanation is that these individuals have been infected with the Omicron variant but did not experience any coronavirus-related symptoms [68,70]. Recent studies have reported that the Omicron variant can evade SARS-CoV-2–specific and neutralizing antibodies [71], [72], [73].
T-cell responses elicit a similar response to SARS-CoV-2 VOCs either by infection or vaccination [13,74], and that most T-cell epitopes are not affected by mutations [17], making SARS-CoV-2 evade cellular immunity unlikely.
Nevertheless, the antibody response is lower for certain variants, including Omicron; considering that antibody titers decrease over time, they might not be protective for infections [13,68,74]. In this scenario, protection might be conferred by cellular immunity due to a pre-existing memory of VOCs conferred after SARS-CoV-2 infection [75] but further experiments would confirm this hypothesis.
Coordination between humoral and cellular immune responses is necessary to eliminate SARS-CoV-2 infection and it is related to milder disease [[5], [76], [77], [78]]. Our study suggests that a positive correlation of immune responses is achieved after COVID-19 infection and at least 2 months after two doses of mRNA vaccine, with greater levels of antibodies correlating with greater levels of memory T cells responding to the infection. We can hypothesize that recurrent immunizations will maintain this pattern, although further studies are needed.
We also have addressed how current treatment can affect adoptive cell therapy with CD45RA− memory T cells. We have found that dexamethasone does not affect the proliferation, phenotype and functionality of theCD45RA− memory T cells. The previous incubation with IL-15 positively affects the release of IFN-γ when the cells are co-cultured with the combination of peptide pools without increasing cell exhaustion.
In addition, we observed an increase in the activation markers after IL-15 cell incubation that is maintained in the presence of dexamethasone. It is already known that IL-15 is a Treg inducer [79]. We have detected an increase in Treg induction with IL-15 but more importantly that induction is maintained in the presence of dexamethasone. This increase is likely not due to an increase in the percentage of Tregs but rather to their activation state, given that we observe no increase in the proliferation index. Further studies on FoxP3 [80] and CTLA-4 expression, along with certain functionality assays, would help confirm our results. In patients with COVID-19, a decrease in the number of Tregs has been associated with a poorer prognosis; therefore, keeping T-cell numbers and better functionality for the existing Tregs would result in a better prognosis [81,82].
In summary, our results suggest that the best donors for adoptive cell therapy for patients with COVID-19 would be immunized individuals who recovered from COVID-19 with mild disease and ideally 2 months after immunization. We show that the cellular response is maintained over a year post-SARS-CoV-2 infection and 2 months after full immunization, and that it seems there is a positive cellular and humoral correlation, with high antibody titers (T4). Also, dexamethasone does not affect the proliferation, phenotype and functionality of CD45RA− memory T cells at the concentration resembling the one employed in clinical practice for these patients, with IL-15 showing a positive effect on SARS-CoV2–specific CD45RA− T cells phenotype and IFN-γ release. These data are supported with the results of in our phase 1 clinical trial and the preliminary phase 2 trial (unpublished data), where donor microchimerism was detected for at least 3 weeks [26]. Our data indicate that the development of a biobank of living drugs with adoptive cell therapy is feasible as a treatment strategy for patients with COVID-19 and future viral pandemics.
Limitations
Our findings are limited by the small sample size. Larger studies are needed to confirm our findings, especially for the cellular response. Moreover, our study is biased toward young individuals, and our control cohort consisted exclusively of young women. A number of reports have observed that age and sex affect the cellular response after mRNA immunization [83]. However, other reports have found no differences in these variables [[56], [57]].
Another limitation for humoral responses is related to the study time points. We are lacking time points, which would have captured the decay in antibody titers more accurately [84]. On the basis of our results and those of other authors, cellular responses do not change and humoral responses gradually decrease [56], widening the range for our donor selection to 2 months and 6 months after full immunization.
In our study, all recovered donors had mild disease. We do not know how cellular responses will behave in individuals who have experienced moderate/severe disease [85]. Wang et al. [46] showed that asymptomatic and symptomatic patients with COVID-19 have similar levels of SARS-CoV-2–specific T-cells, supporting the idea that any recovered immunized individual would be a good donor for our product, an important consideration, given that most COVID-19 infections are asymptomatic [86], even more so after vaccination [87], [88], [89].
We used IFN-γ as a marker to detect functional activity for cellular immunity. IFN-γ is a marker of Th1, and the Th1 subset coordinates the cell-mediated response, which is essential in macrophages, cytotoxic T cells and, NK activation via IL-2 and IFN-γ. However, we cannot rule out the possibility that other CD4+ T-cell subsets are activated upon antigen-specific encounters [5,90]. Other authors have used different sets of markers to detect cellular immune response depending on the study's cell subset [5,56,91].
We studied T cells from peripheral blood, when memory T cells in peripheral blood are only a small subset of the body's memory T cells. As for other memory T cells from other viruses, the formation of memory T cells can occur at distinct sites preferentially maintained at the sites of initial effect T-cell recruitment [49]. Future studies are needed to better define the optimal donors for adoptive cell therapy in COVID-19 patients.
Declaration of Competing Interest
AP-M and CF filed patent EP20382850 on Memory T cells as adoptive cell therapy for viral diseases. All other authors declare no competing interest.
Acknowledgments
Funding
This study has been supported by CRIS Cancer Foundation and IMMUNOVACTER-CM from the European Regional Development Fund – Resources REACT-EU. KA-AS is funded by project DECODE-19 (Combinatorial Cell Therapy for high mortality risk Covid-19 patients, ICI21/00016).
Author Contributions
Conception and design of the study: KA-AS, CF, BP-M and APM. Acquisition of data: KA-AS, CF and APM. Analysis and interpretation of data: KA-AS, CF and MD-A. Drafting or revising the manuscript: all authors. All authors have approved the final article.
Acknowledgments
We thank all of the donors who participated in this study.
Footnotes
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.jcyt.2022.12.001.
Appendix. Supplementary materials
References
- 1.Israel A., et al. Elapsed time since BNT162b2 vaccine and risk of SARS-CoV-2 infection in a large cohort. medRxiv Prepr. Serv. Heal. Sci. 2021:1–7. doi: 10.1101/2021.08.03.21261496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wilder-Smith A. What is the vaccine effect on reducing transmission in the context of the SARS-CoV-2 delta variant? Lancet Infect. Dis. 2021;380:4–5. doi: 10.1016/S1473-3099(21)00690-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Singanayagam A., et al. Community transmission and viral load kinetics of the SARS-CoV-2 delta (B.1.617.2) variant in vaccinated and unvaccinated individuals in the UK: a prospective, longitudinal, cohort study. Lancet Infect. Dis. 2021;3099 doi: 10.1016/S1473-3099(21)00648-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Moss P. Vol. 23. 2022. (Review Article | SERIES The T cell immune response against SARS-CoV-2). [DOI] [PubMed] [Google Scholar]
- 5.Painter M.M., et al. Rapid induction of antigen-specific CD4+ T cells is associated with coordinated humoral and cellular immunity to SARS-CoV-2 mRNA vaccination. Immunity. 2021;54:2133–2142. doi: 10.1016/j.immuni.2021.08.001. e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zheng M., et al. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell. Mol. Immunol. 2020;17:533–535. doi: 10.1038/s41423-020-0402-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zabalza A., et al. Humoral and Cellular Responses to SARS-CoV-2 in Convalescent COVID-19 Patients With Multiple Sclerosis. Neurol Neuroimmunol Neuroinflamm. 2022;0:1–13. doi: 10.1212/NXI.0000000000001143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Riise J., et al. treated patients with lymphoma develop strong CD8 cell responses following COVID-19 vaccination. Br J Haematol. 2022:697–708. doi: 10.1111/bjh.18149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Radbruch A., Chang H.D. A long-term perspective on immunity to COVID. Nature. 2021;595:359–360. doi: 10.1038/d41586-021-01557-z. [DOI] [PubMed] [Google Scholar]
- 10.Feng C., et al. Protective humoral and cellular immune responses to SARS-CoV-2 persist up to 1 year after recovery. Nat. Commun. 2021;12:1–7. doi: 10.1038/s41467-021-25312-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cox R.J., Brokstad K.A. Not just antibodies: B cells and T cells mediate immunity to COVID-19. Nat. Rev. Immunol. 2021;20:581–582. doi: 10.1038/s41577-020-00436-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lu Z., et al. Durability of SARS-CoV-2–Specific T-Cell Responses at 12 Months Postinfection. J Infect Dis. 2021;224 doi: 10.1093/infdis/jiab543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jordan S.C., et al. T cell immune responses to SARS-CoV-2 and variants of concern (Alpha and Delta) in infected and vaccinated individuals. Cell. Mol. Immunol. 2021;18:2554–2556. doi: 10.1038/s41423-021-00767-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Marco L., De, et al. Assessment of T-cell Reactivity to the SARS-CoV-2 Omicron Variant by Immunized Individuals. JAMA Netw Open. 2022;5:1–8. doi: 10.1001/jamanetworkopen.2022.10871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kedzierska K., Thomas P.G. Count on us : T cells in SARS-CoV-2 infection and vaccination. Cell Reports Med. 2022;3 doi: 10.1016/j.xcrm.2022.100562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vardhana S., et al. Understanding T-cell responses to COVID-19 is essential for informing public health strategies. Sci Immunol. 2022;1303:1–9. doi: 10.1126/sciimmunol.abo1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tarke A., et al. Comprehensive analysis of T cell immunodominance and immunoprevalence of SARS-CoV-2 epitopes in COVID-19 cases. Cell Reports Med. 2021;2 doi: 10.1016/j.xcrm.2021.100204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Qin C., et al. Dysregulation of Immune Response in Patients with COVID-19 in Wuhan, China. SSRN Electron. J. 2020 doi: 10.2139/ssrn.3541136. [DOI] [Google Scholar]
- 19.Zhao Q., et al. Lymphopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A systemic review and meta-analysis. Int. J. Infect. Dis. 2020;96:131–135. doi: 10.1016/j.ijid.2020.04.086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jafarzadeh A., Jafarzadeh S., Nozari P., Mokhtari P., Nemati M. Lymphopenia an important immunological abnormality in patients with COVID-19: Possible mechanisms. Scand. J. Immunol. 2021;93:1–16. doi: 10.1111/sji.12967. [DOI] [PubMed] [Google Scholar]
- 21.Odak I., et al. Reappearance of effector T cells is associated with recovery from COVID-19. EBioMedicine. 2020;57 doi: 10.1016/j.ebiom.2020.102885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee J., Park S.S., Kim T.Y., Lee D.G., Kim D.W. Lymphopenia as a biological predictor of outcomes in COVID-19 patients: A nationwide cohort study. Cancers (Basel) 2021;13:1–15. doi: 10.3390/cancers13030471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Garbo R., Valent F., Gigli G.L., Valente M. Pre-Existing Lymphopenia Increases the Risk of Hospitalization and Death after SARS-CoV-2 Infection. Infect. Dis. Rep. 2022;14:20–25. doi: 10.3390/idr14010003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Xiang Q., et al. SARS-CoV-2 Induces Lymphocytopenia by Promoting Inflammation and Decimates Secondary Lymphoid Organs. Front. Immunol. 2021;12:1–13. doi: 10.3389/fimmu.2021.661052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ferreras C., et al. SARS-CoV-2-Specific Memory T Lymphocytes From COVID-19 Convalescent Donors: Identification, Biobanking, and Large-Scale Production for Adoptive Cell Therapy. Front. Cell Dev. Biol. 2021;9:1–12. doi: 10.3389/fcell.2021.620730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pérez-Martínez A., et al. Phase I dose-escalation single centre clinical trial to evaluate the safety of infusion of memory T cells as adoptive therapy in COVID-19 (RELEASE) EClinicalMedicine. 2021;39 doi: 10.1016/j.eclinm.2021.101086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Herrera L., et al. Identifying SARS-CoV-2 ‘memory’ NK cells from COVID-19 convalescent donors for adoptive cell therapy. Immunology. 2022;165:234–249. doi: 10.1111/imm.13432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Torres K.C.L., et al. Norepinephrine, dopamine and dexamethasone modulate discrete leukocyte subpopulations and cytokine profiles from human PBMC. J. Neuroimmunol. 2005;166:144–157. doi: 10.1016/j.jneuroim.2005.06.006. [DOI] [PubMed] [Google Scholar]
- 29.RECOVERY Collaborative Group. Horby P, et al. Dexamethasone in Hospitalized Patients with Covid-19. N Engl J Med. 2021 Feb 25;384(8):693–704. doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gutsol A.A., Sokhonevich N.A., Seledtsov V.I., Litvinova L.S. Dexamethasone effects on activation and proliferation of immune memory T cells. Bull. Exp. Biol. Med. 2013;155:474–476. doi: 10.1007/s10517-013-2182-5. [DOI] [PubMed] [Google Scholar]
- 31.De Antonio, S.R., et al. Effects of dexamethasone on lymphocyte proliferation and cytokine production in rheumatoid arthritis. J. Rheumatol. 2002;29:46–51. [PubMed] [Google Scholar]
- 32.Giles A.J., et al. Dexamethasone-induced immunosuppression: Mechanisms and implications for immunotherapy. J. Immunother. Cancer. 2018;6:1–13. doi: 10.1186/s40425-018-0371-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schluns K.S., Lefrançois L. Cytokine control of memory T-cell development and survival. Nat. Rev. Immunol. 2003;3:269–279. doi: 10.1038/nri1052. [DOI] [PubMed] [Google Scholar]
- 34.Arneja A., Johnson H., Gabrovsek L., Lauffenburger D.A., White F.M. Qualitatively Different T Cell Phenotypic Responses to IL-2 versus IL-15 Are Unified by Identical Dependences on Receptor Signal Strength and Duration. J. Immunol. 2014;192:123–135. doi: 10.4049/jimmunol.1302291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Knijff E.M., et al. A relative resistance of T cells to dexamethasone in bipolar disorder. Bipolar Disord. 2006;8:740–750. doi: 10.1111/j.1399-5618.2006.00359.x. [DOI] [PubMed] [Google Scholar]
- 36.Lowy M.T., Reder A.T., Gormley G.J., Meltzer H.Y. Comparison of in vivo and in vitro glucocorticoid sensitivity in depression: Relationship to the dexamethasone suppression test. Biol. Psychiatry. 1988;24:619–630. doi: 10.1016/0006-3223(88)90136-9. [DOI] [PubMed] [Google Scholar]
- 37.Fontanet A., et al. SARS-CoV-2 variants and ending the COVID-19 pandemic. Lancet. 2021;397:952–954. doi: 10.1016/S0140-6736(21)00370-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Maschan M., et al. Low-dose donor memory T-cell infusion after TCR alpha/beta depleted unrelated and haploidentical transplantation: Results of a pilot trial. Bone Marrow Transplant. 2018;53:264–273. doi: 10.1038/s41409-017-0035-y. [DOI] [PubMed] [Google Scholar]
- 39.Blagov S., et al. T-cell tracking, safety, and effect of low-dose donor memory T-cell infusions after αβ T cell-depleted hematopoietic stem cell transplantation. Bone Marrow Transplant. 2021;56:900–908. doi: 10.1038/s41409-020-01128-2. [DOI] [PubMed] [Google Scholar]
- 40.Gasior Kabat M, et al. Selective T-cell depletion targeting CD45RA as a novel approach for HLA-mismatched hematopoietic stem cell transplantation in pediatric nonmalignant hematological diseases. Int J Hematol. 2021 Jul;114(1):116–123. doi: 10.1007/s12185-021-03138-2. [DOI] [PubMed] [Google Scholar]
- 41.Gasior M., et al. The role of early natural killer cell adoptive infusion before engraftment in protecting against human herpesvirus-6B encephalitis after naïve T-cell-depleted allogeneic stem cell transplantation. Transfusion. 2021;61:1505–1517. doi: 10.1111/trf.16354. [DOI] [PubMed] [Google Scholar]
- 42.Grifoni A., et al. Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals. Cell. 2020;181:1489–1501. doi: 10.1016/j.cell.2020.05.015. e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Swain S.L., McKinstry K.K., Strutt T.M. Expanding roles for CD4 + T cells in immunity to viruses. Nat. Rev. Immunol. 2012;12:136–148. doi: 10.1038/nri3152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Luckheeram R.V., Zhou R., Verma A.D., Xia B. CD4 +T cells: Differentiation and functions. Clin. Dev. Immunol. 2012 doi: 10.1155/2012/925135. 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dong C., Flavell R.A. Cell fate decision: T-helper 1 and subsets in immune responses. Arthritis Res. 2000;2:179–188. doi: 10.1186/ar85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang Z., et al. Exposure to SARS-CoV-2 generates T-cell memory in the absence of a detectable viral infection. Nat. Commun. 2021;12:6–13. doi: 10.1038/s41467-021-22036-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ahrends T., et al. CD4+ T cell help creates memory CD8+ T cells with innate and help-independent recall capacities. Nat. Commun. 2019;10:1–13. doi: 10.1038/s41467-019-13438-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Peng Y., et al. Broad and strong memory CD4+ and CD8+ T cells induced by SARS-CoV-2 in UK convalescent individuals following COVID-19. Nat. Immunol. 2020;21:1336–1345. doi: 10.1038/s41590-020-0782-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Farber DL, Yudanin NA, Restifo NP. Human memory T cells: generation, compartmentalization and homeostasis. Nat Rev Immunol. 2014 Jan;14(1):24–35. doi: 10.1038/nri3567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mahnke Y.D., Brodie T.M., Sallusto F., Roederer M., Lugli E. The who's who of T-cell differentiation: Human memory T-cell subsets. Eur. J. Immunol. 2013;43:2797–2809. doi: 10.1002/eji.201343751. [DOI] [PubMed] [Google Scholar]
- 51.Martínez-Flores D., et al. SARS-CoV-2 Vaccines Based on the Spike Glycoprotein and Implications of New Viral Variants. Front. Immunol. 2021;12 doi: 10.3389/fimmu.2021.701501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Glycoprotein C.-S., et al. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell. 2020;181:281–292. doi: 10.1016/j.cell.2020.02.058. e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Oberhardt V., et al. Rapid and stable mobilization of CD8+ T cells by SARS-CoV-2 mRNA vaccine. Nature. 2021;597:268–273. doi: 10.1038/s41586-021-03841-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tarke A., et al. Impact of SARS-CoV-2 variants on the total CD4+ and CD8+ T cell reactivity in infected or vaccinated individuals. Cell Reports Med. 2021;2 doi: 10.1016/j.xcrm.2021.100355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mateus J., et al. Low-dose mRNA-1273 COVID-19 vaccine generates durable memory enhanced by cross-reactive T cells. Science. 2021;374(80-) doi: 10.1126/science.abj9853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Guerrera G., et al. BNT162b2 vaccination induces durable SARS-CoV-2 specific T cells with a stem cell memory phenotype. Sci. Immunol. 2021;6:eabl5344. doi: 10.1126/sciimmunol.abl5344. [DOI] [PubMed] [Google Scholar]
- 57.Goel R.R., et al. Distinct antibody and memory B cell responses in SARSCoV-2 naïve and recovered individuals following mRNA vaccination. Sci. Immunol. 2021;6:1–19. doi: 10.1126/sciimmunol.abi6950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Goel R.R., et al. mRNA vaccines induce durable immune memory to SARS-CoV-2 and variants of concern. Science. 2021;0829(80-):1–28. doi: 10.1126/science.abm0829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Long Q.X., et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020;26:1200–1204. doi: 10.1038/s41591-020-0965-6. [DOI] [PubMed] [Google Scholar]
- 60.Schiffner J., et al. Long-Term Course of Humoral and Cellular Immune Responses in Outpatients After SARS-CoV-2 Infection. Front. Public Heal. 2021;9:1–11. doi: 10.3389/fpubh.2021.732787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Prendecki M., et al. Humoral and T-cell responses to SARS-CoV-2 vaccination in patients receiving immunosuppression. Ann. Rheum. Dis. 2021;80:1322–1329. doi: 10.1136/annrheumdis-2021-220626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Krammer F., et al. Antibody Responses in Seropositive Persons after a Single Dose of SARS-CoV-2 mRNA Vaccine. N. Engl. J. Med. 2021;384:1372–1374. doi: 10.1056/NEJMc2101667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Dan J.M., et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science. 2021;371(80-) doi: 10.1126/science.abf4063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Turner J.S., et al. SARS-CoV-2 mRNA vaccines induce persistent human germinal centre responses. Nature. 2021;596:109–113. doi: 10.1038/s41586-021-03738-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Esparcia-Pinedo L., et al. CD4+ T Cell Immune Specificity Changes After Vaccination in Healthy And COVID-19 Convalescent Subjects. Front. Immunol. 2022;12:1–9. doi: 10.3389/fimmu.2021.755891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.De Giorgi V., et al. Naturally Acquired SARS-CoV-2 Immunity Persists for up to 11 Months following Infection. J. Infect. Dis. 2021;224:1294–1304. doi: 10.1093/infdis/jiab295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mazzoni A., et al. First-dose mRNA vaccination is sufficient to reactivate immunological memory to SARS-CoV-2 in subjects who have recovered from COVID-19 First-dose mRNA vaccination is sufficient to reactivate immunological memory to SARS-CoV-2 in subjects who have recover. J. Clin. Invest. 2021;131 doi: 10.1172/JCI149150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Evans J.P., et al. Neutralizing antibody responses elicited by SARS-CoV-2 mRNA vaccination wane over time and are boosted by breakthrough infection. Sci. Transl. Med. 2022;14:1–13. doi: 10.1126/scitranslmed.abn8057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ebinger J.E., et al. Antibody responses to the BNT162b2 mRNA vaccine in individuals previously infected with SARS-CoV-2. Nat. Med. 2021;27:981–984. doi: 10.1038/s41591-021-01325-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Naranbhai V., et al. T cell reactivity to the SARS-CoV-2 Omicron variant is preserved in most but not all individuals. Cell. 2022;185:1041–1051. doi: 10.1016/j.cell.2022.01.029. e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Dejnirattisai W., et al. SARS-CoV-2 Omicron-B.1.1.529 leads to widespread escape from neutralizing antibody responses. Cell. 2022;185:467–484. doi: 10.1016/j.cell.2021.12.046. e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yu X., et al. Reduced sensitivity of SARS-CoV-2 Omicron variant to antibody neutralization elicited by booster vaccination. Cell Discov. 2022;8:8–11. doi: 10.1038/s41421-022-00375-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Delamater P.L., et al. Elimination of nonmedical immunization exemptions in California and school-entry vaccine status. Pediatrics. 2019;143:2021–2023. doi: 10.1542/peds.2018-3301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Geers D., et al. SARS-CoV-2 variants of concern partially escape humoral but not T-cell responses in COVID-19 convalescent donors and vaccinees. Sci. Immunol. 2021;6:1–15. doi: 10.1126/sciimmunol.abj1750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Cele S., et al. Omicron extensively but incompletely escapes Pfizer BNT162b2 neutralization. Nature. 2022;602:654–656. doi: 10.1038/s41586-021-04387-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Agrati C., et al. Coordinate induction of humoral and spike specific t-cell response in a cohort of Italian health care workers receiving bnt162b2 mRNA vaccine. Microorganisms. 2021;9 doi: 10.3390/microorganisms9061315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Rydyznski Moderbacher C., et al. Antigen-Specific Adaptive Immunity to SARS-CoV-2 in Acute COVID-19 and Associations with Age and Disease Severity. Cell. 2020;183:996–1012. doi: 10.1016/j.cell.2020.09.038. e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ni L., et al. Detection of SARS-CoV-2-Specific Humoral and Cellular Immunity in COVID-19 Convalescent Individuals. Immunity. 2020;52:971–977. doi: 10.1016/j.immuni.2020.04.023. e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Imamichi H, Sereti I, Lane HC. IL-15 acts as a potent inducer of CD4(+)CD25(hi) cells expressing FOXP3. Eur J Immunol. 2008 Jun;38(6):1621–1630. doi: 10.1002/eji.200737607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wang, Y. et al. The role of CD4 + FoxP3 + regulatory T cells in the immunopathogenesis of COVID-19: implications for treatment. 17, (2021). [DOI] [PMC free article] [PubMed]
- 81.Wang H., et al. Regulatory T cells in COVID-19. Aging Dis. 2021;12:1545–1553. doi: 10.14336/AD.2021.0709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Caldrer S., et al. Regulatory T Cells as Predictors of Clinical Course in Hospitalised COVID-19 Patients. Front. Immunol. 2021;12:1–11. doi: 10.3389/fimmu.2021.789735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Flanagan K.L., Fink A.L., Plebanski M., Klein S.L. Sex and gender differences in the outcomes of vaccination over the life course. Annu. Rev. Cell Dev. Biol. 2017;33:577–599. doi: 10.1146/annurev-cellbio-100616-060718. [DOI] [PubMed] [Google Scholar]
- 84.Levin E.G., et al. Waning Immune Humoral Response to BNT162b2 Covid-19 Vaccine over 6 Months. N. Engl. J. Med. 2021:1–11. doi: 10.1056/nejmoa2114583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Papayanni P.G., et al. Vaccinated and Convalescent Donor-Derived Severe Acute Respiratory Syndrome Coronavirus 2-Specific T Cells as Adoptive Immunotherapy for High-Risk Coronavirus Disease 2019 Patients. Clin. Infect. Dis. 2021;73:2073–2082. doi: 10.1093/cid/ciab371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kimball A., et al. Asymptomatic and Presymptomatic SARS-CoV-2 Infections in Residents of a Long-Term Care Skilled Nursing Facility—King County, Washington, March 2020. MMWR Morb. Mortal. Wkly. Rep. 2020;69:377–381. doi: 10.15585/mmwr.mm6913e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Tang L, Hijano DR, Gaur AH, et al. Asymptomatic and Symptomatic SARS-CoV-2 Infections After BNT162b2 Vaccination in a Routinely Screened Workforce. JAMA. 2021;325(24):2500–2502. doi: 10.1001/jama.2021.656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Gohil S.K., et al. Asymptomatic and Symptomatic COVID-19 Infections among Health Care Personnel before and after Vaccination. JAMA Netw. Open. 2021;4:8–11. doi: 10.1001/jamanetworkopen.2021.15980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Shah ASV, et al. Effect of Vaccination on Transmission of SARS-CoV-2. N Engl J Med. 2021 Oct 28;385(18):1718–1720. doi: 10.1056/NEJMc2106757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Gil-Etayo F.J., et al. T-Helper Cell Subset Response Is a Determining Factor in COVID-19 Progression. Front. Cell. Infect. Microbiol. 2021;11:1–11. doi: 10.3389/fcimb.2021.624483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Payne R.P., et al. Immunogenicity of standard and extended dosing intervals of BNT162b2 mRNA vaccine. Cell. 2021;184:5699–5714. doi: 10.1016/j.cell.2021.10.011. e11. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.