Abstract
Background and purpose
Currently, human papillomavirus (HPV) positivity represents a strong prognostic factor for both reduced risk of relapse and improved survival in patients with oropharyngeal squamous cell carcinoma (OPSCC). However, a subset of HPV-positive OPSCC patients still experience poor outcomes. Furthermore, HPV-negative OPSCC patients, who have an even higher risk of relapse, are still lacking suitable prognostic biomarkers for clinical outcome. Here, we evaluated the prognostic value of LINE-1 methylation level in OPSCC patients and further addressed the relationship between LINE-1 methylation status and p53 protein expression as well as genome-wide/gene-specific DNA methylation.
Results
In this study, DNA was extracted from 163 formalin-fixed paraffin-embedded tissue samples retrospectively collected from stage III-IVB OPSCC patients managed with curative intent with up-front treatment. Quantitative methylation-specific PCR revealed that LINE-1 hypomethylation was directly associated with poor prognosis (5-year overall survival—OS: 28.1% for LINE-1 methylation < 35% vs. 69.1% for ≥ 55%; p < 0.0001). When LINE-1 methylation was dichotomized as < 55% versus ≥ 55%, interaction with HPV16 emerged: compared with hypermethylated HPV16-positive patients, subjects with hypomethylated HPV16-negative OPSCC reported an adjusted higher risk of death (HR 4.83, 95% CI 2.24–10.38) and progression (HR 4.54, 95% CI 2.18–9.48). Tumor protein p53 (TP53) gene is often mutated and overexpressed in HPV-negative OPSCC. Since p53 has been reported to repress LINE-1 promoter, we then analyzed the association between p53 protein expression and LINE-1 methylation levels. Following p53 immunohistochemistry, results indicated that among HPV16-negative patients with p53 ≥ 50%, LINE-1 methylation levels declined and remained stable at approximately 43%; any HPV16-positive patient reported p53 ≥ 50%. Finally, DNA methylation analysis demonstrated that genome-wide average methylation level at cytosine–phosphate–guanine sites was significantly lower in HPV16-negative OPSCC patients who relapsed within two years. The subsequent integrative analysis of gene expression and DNA methylation identified 20 up-regulated/hypomethylated genes in relapsed patients, and most of them contained LINE-1 elements in their promoter sequences.
Conclusions
Evaluation of the methylation level of LINE-1 may help in identifying the subset of OPSCC patients with bad prognosis regardless of their HPV status. Aberrant LINE-1 hypomethylation might occur along with TP53 mutations and lead to altered gene expression in OPSCC.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13148-022-01386-5.
Keywords: Oropharyngeal squamous cell carcinoma, HPV, LINE-1, DNA methylation, p53
Introduction
Changes in sexual habits have led to a steady increase in the incidence of human papillomavirus (HPV)-driven oropharyngeal squamous cell carcinoma (OPSCC) that in most Western countries now exceeds the proportion of tobacco- and alcohol-related counterparts [1]. Of note, HPV16 represents the most common genotype (83%) among HPV-positive OPSCC patients [2]. Hence, HPV16 is by far the most carcinogenic HPV type in OPSCC, as most non-HPV16 oncogenic infections do not progress to cancer [3]. Although HPV confers a substantial survival benefit to these malignancies [4], roughly 20% of all patients with HPV-positive OPSCC develop recurrent disease within 5 years after diagnosis [5–8]. Given the considerable interest in identifying treatment de-escalation strategies in this subset of OPSCC patients with a more favorable prognosis [9], it is of paramount importance to identify biological predictors of atypical behavior to avoid the administration of sub-optimal treatment.
Genes encoding for epigenetic regulators have been frequently found mutated in several tumors, including OPSCC [10]; hence, it is expected that epigenetic changes may play an important role in OPSCC pathogenesis and response to therapy. Consistently, HPV oncoproteins have also been demonstrated to impact on the epigenetic patterns by interacting with different epigenetic regulators [11], thus affecting the chromatin landscape of HPV-positive OPSCC cells. At present, DNA methylation represents one of the most investigated epigenetic mechanisms along with histone modifications and non-coding RNAs. DNA methylation consists of the addition of a methyl group to the fifth carbon of cytosines residues within cytosine–phosphate–guanine (CpG) sites. It has been estimated that over 90% of these CpG sites are located within DNA repetitive elements, particularly Alu and LINE-1 [12]. LINE-1 is the most abundant repetitive element in the genome, which in normal human tissues is generally found heavily methylated. Since it represents approximately 17% of the human genome, LINE-1 has been widely accepted as a surrogate marker of global DNA methylation [13]. In the last years, LINE-1 hypomethylation has been showed to be related to carcinogenesis and to the development of many tumor types [14–17]. On these grounds, we previously observed a significantly lower level of LINE-1 methylation in OPSCC patients who relapsed within 2 years, thus indicating that the overall level of genomic DNA methylation may have an impact on the risk of early relapse in OPSCC [18]. Despite these findings, however, the prognostic impact of LINE-1 methylation levels on OPSCC survival has not been established.
Hypomethylation in the LINE-1 promoter region is crucial for the transcriptional activation of LINE-1 elements, which results in retroelement transposition and genomic instability, thus providing a setting for cancer progression [19]. In the last years, it has become increasingly clear that several transcription factors and chromatin remodelers are involved in LINE-1 activation [20, 21]. Among these, a number of studies have suggested that p53 protein might silence LINE-1 through regulating the deposition of epigenetic marks within its promoter [22–25], thus affecting its retrotransposon activity in tumor cells. Notably, somatic mutations of the tumor suppressor gene TP53 are one of the most frequent alterations in head and neck squamous cell carcinoma (HNSCC) [26]. Besides mutations, p53 functions can also be disrupted by the HPV E6 protein in HPV-positive patients [27]. At present, it is largely unknown whether TP53 mutational status and/or p53 expression pattern correlates with LINE-1 methylation in OPSCC.
About 30% of the transcription start sites in the human genome are associated with repetitive elements, particularly with LINE-1 subfamilies [28]; furthermore, following loss of methylation, LINE-1 was shown to act as an alternative promoter for surrounding genes [29, 30]. Therefore, another aspect yet to be investigated is how the OPSCC epigenome evolves in relapsed patients respect to non-relapsed ones and whether the differentially methylated regions between the two subgroups map within LINE-1 elements.
Based on these premises, this study aimed to assess the impact of LINE-1 methylation level on overall survival (OS) and progression-free survival (PFS) in both HPV-positive and HPV-negative OPSCC patients. In addition, to better determine whether p53 expression might affect LINE-1 methylation status in OPSCC, we evaluated the correlation between p53 expression pattern and LINE-1 methylation levels. Finally, to shed initial light on the mechanisms through which LINE1 methylation impacts OPSCC outcome, differences in genome-wide/gene-specific DNA methylation were investigated in a subset of relapsed and not relapsed OPSCC patients and further investigated for their potential regulation by LINE-1 elements.
Methods
Patients
The study enrolled 163 stage III-IVB OPSCC patients managed with curative intent with up-front (chemo-)radiotherapy or up-front surgery followed by adjuvant (chemo-)radiotherapy, as previously described [18]. Patients have been treated between 2001 and 2019 at the National Cancer Institute in Aviano, the “Santa Maria degli Angeli” General Hospital in Pordenone, the “Ca’ Foncello” General Hospital in Treviso, and the University Hospital in Modena. All tumors were centrally reclassified according to the American Joint Committee on Cancer 7th Edition. The study was approved by the local Independent Ethic Committees (CRO-2019-13, 733/AULSS2, 5/2020/OSS/AOUMO). Participants provided written informed consent for inclusion in the study; 104 subjects overlapped with a previous study [18].
Immunohistochemical analysis of p53
For each patient, we retrieved a formalin-fixed paraffin-embedded (FFPE) tissues representative of the OPSCC, collected at the time of biopsy or surgical resection and before starting any treatment, for a total of 163 neoplastic samples. Serial 5-µm-thick FFPE tumor sections were then used for hematoxylin—eosin staining and immunohistochemistry (IHC) analysis. All stained sections were microscopically evaluated by a pathologist unaware of any clinical information (including follow-up or outcome data), and only neoplastic lesions that contained at least 70% of neoplastic cells were included in the study.
p53 expression was evaluated by IHC (Agilent Technologies DAKO; Clone DO-7) in 89 patients, for whom sufficient neoplastic sample was available. The extent of staining was estimated to the nearest 10% level of positive tumor cells. The intensity of staining was recorded as weak or strong. Strong expression in more than 50% of cells or complete absence of stain was considered a p53 mutated pattern [31–35].
Quantitation of HPV16 E6 DNA using real-time quantitative PCR analysis
Genomic DNA was extracted from OPSCC FFPE tissues using the FFPE RNA/DNA Purification Plus Kit (Norgen), following the manufacturer’s protocol. SYBR green quantitative HPV16-PCR was carried out as previously reported [18].
Quantitative methylation-specific PCR analysis for the methylation levels of LINE-1
Genomic DNA was obtained from OPSCC FFPE tissues in quantities sufficient for bisulfite treatment. Bisulfite conversion was carried out on 500 ng genomic DNA using EZ DNA Methylation-Gold™ Kit (Zymo Research), according to the manufacturer’s protocol. SYBR Green quantitative methylation-specific PCR (qMSP) was performed as previously reported [18].
Genome-wide DNA methylation analysis
DNA methylation analysis was performed by Genomix4Life S.R.L. (Baronissi, Salerno, Italy). To assess the quality of DNA isolated by FFPE samples, Illumina FFPE QC kit (Illumina, San Diego, CA, USA) was used. Only 10 FFPE DNA samples were considered eligible for restoration using the Infinium HD FFPE Restore Kit (Illumina, San Diego, CA, USA). Restored DNA was bisulfite converted using EZ DNA methylation kit (Zymo Research). For each sample, 250 ng of bisulfite converted DNA was used for analysis of whole-genome methylation using MethylationEPIC BeadChip (Illumina, San Diego, CA, USA), which contains 850,000 probes. In brief, bisulfite-converted DNA was whole-genome amplified for 20 h followed by end-point fragmentation. Fragmented DNA was precipitated, denaturated, and hybridized to the BeadChips for 20 h at 48 °C. The BeadChips were washed, and the hybridized primers were extended and labeled before scanning the BeadChips using the Illumina iScan system. GenomeStudio software was used for the extraction of DNA methylation signals from scanned arrays.
RNA extraction and transcriptome profiling
RNA isolations were performed from 5 FFPE samples using the FFPE RNA/DNA Purification Plus Kit (Norgen). Nucleic acids were quantified with Qubit 2.0 fluorimeter using Qubit RNA HS assay kit (Termo Fisher Scientifc, USA), and the assessment of nucleic acids integrity (RNA Integrity Number) was performed with Agilent 4150 TapeStation System (Agilent Technologies, USA). Only 5 samples passed the qualitative and quantitative checks required by the Illumina library protocol. Libraries preparation for transcriptome analysis was performed employing the TruSeq RNA Exome kit (Cat.20020189, Illumina) for FFPE samples starting from 200 ng of RNA as input materials, respectively, according to manufacturers’protocols; 5 libraries were sequenced on NextSeq 500 (Illumina) using 2 × 75pb paired end.
Bioinformatics analysis
EPIC methylation array was performed using ChAMP [33]. Only CpG with a detection p < 0.01 was considered for further analysis. The analysis was performed by comparing patients who relapsed within 2 years from the end of treatment with those who did not, and only the CpG associated with a p < 0.05 and DeltaBeta (DB) cutoff set to the first quartile value (|DB|≥ 0.15) of DB distribution were considered differentially methylated. Genomic annotation of CpGs was performed using the information available in Infinium MethylationEPIC v1.0 B5 Manifest file. In detail, transcriptional start site (TSS)200 refers to CpGs between 0 and 200 bases upstream of the TSS; TSS1500 refers to CpGs between 200 and 1500 bases upstream of the TSS; 5’UTR refers to those GpGs within the 5’ untranslated region, between the TSS and the ATG start site; gene body refers to CpGs between the ATG and stop codon, regardless of the presence of introns and exons. Promoter region includes TSS1500, TSS200, 5’UTR, and 1st exon regions. Annotation of LINE on selected genes was performed using the Genome Browser track “Repeating Elements by RepeatMasker” [34]. Functional analysis on potentially up- and down-regulated genes was performed using IPA (Ingenuity Pathway Analysis, Qiagen). Only molecular functions with a p ≤ 0.05 were considered.
RNA-Seq data analysis was performed as previously described [35]. In detail, quality control of sequenced reads has been performed using FASTQC (https://www.bioinformatics.babraham.ac.uk/projects/fastqc/), while adapter sequences were removed using Trimmomatic [36]. Alignment was performed on human genome (assembly hg38) considering GenCode Release 41 (GRCh38.p13) with STAR v2.7.10a [37], setting the default parameters. Quantification of expressed transcripts was performed using FeatureCounts [38] and differentially expressed transcripts were identified using DESeq2 [39]. Differential expression was performed on relapsed vs relapse-free OPSCC patients. Differentially expressed transcripts were reported as |Fold-Change|(FC) ≥ 1.5 along with associated adjusted p ≤ 0.05, computed according to Benjamini-Hochberg.
Statistical methods
Distribution of patients according to sociodemographic and clinical characteristics was reported as absolute number and corresponding percentage. LINE-1 methylation was reported as median value with interquartile range (IQR). Differences between strata were evaluated through Kruskal–Wallis test. Further, to evaluate associations between LINE-1 methylation, HPV status, and p53 expression, the analysis of variance was conducted, with post hoc Tukey test.
LINE-1 methylation was then categorized in three levels (< 35%, 35–54%, and ≥ 55%) using a recursive procedure which identifies the cutoffs which maximize the difference in OS. The optimal cutoffs were in agreement with previous findings [18]. For each patient, the time at risk was calculated from date of elective treatment completion to the event of interest or last follow-up, whichever came first. The event of interest was death (any cause) for OS and death or locoregional/distant recurrence for PFS. The Kaplan–Meier method was used to generate crude survival probabilities, and the log-rank test was used to assess the difference in time to event according to LINE-1 methylation level and HPV16 status [36]. To account for potential confounders, hazard ratios (HRs) and the corresponding 95% confidence intervals (CIs) were calculated using Cox proportional hazards models [36], adjusting for gender and age, plus covariates significantly associated with OS in the univariate analysis (i.e., T stage, N stage, and HPV16 status).
Results
Prognostic impact of LINE-1 methylation
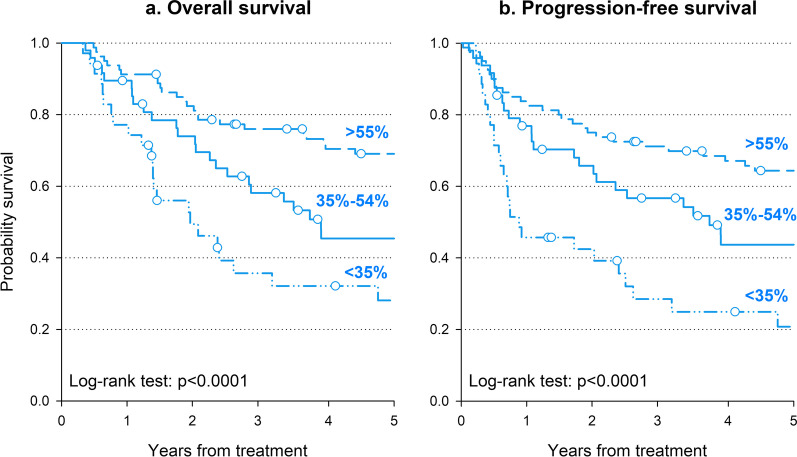
Table 1 shows the median LINE-1 expression according to sociodemographic and clinical characteristics in 163 OPSCC patients. Majority of the patients were males (71.8%), with TNM stage IVA-B (76.7%) and HPV16-negative (67.5%); 90 patients (55.2%) underwent surgery. LINE-1 expression was lower in patients aged ≥ 70 years than younger ones (p = 0.0249) and in HPV16-negative than in HPV16-positive patients (p < 0.0001). LINE-1 methylation level was directly associated with prognosis, with survival rates decreasing with LINE-1 hypomethylation (Fig. 1). In detail, patients with LINE-1 methylation ≥ 55% reported a 5-year OS of 69.1% compared to 45.5% for LINE-1 methylation between 35 and 54%, and to 28.1% for LINE-1 methylation < 35% (Fig. 1a, p < 0.0001). Similarly, 5-year PFS probabilities were 64.4%, 43.7%, and 20.8%, for LINE-1 methylation ≥ 55%, between 35 and 54%, and < 35%, respectively (Fig. 1b, p < 0.0001). Multivariate analyses confirmed that patients with LINE-1 < 35% had a worse prognosis than those with LINE-1 ≥ 55% (Table 2), with a HR of 2.76 (95% CI 1.48–5.12) for death and of 2.39 (95% CI 1.35–4.24) for progression/death. Interestingly, excess risk in patients with LINE-1 < 35% remained significant after adjustment for HPV16 status. Patients with LINE-1 35–54% were at increased risk of both death and progression/death, but the HRs were no longer statistically significant.
Table 1.
LINE-1 methylation in 163 patients with stage III-IV oropharyngeal squamous cell carcinoma according to sociodemographic and clinical characteristics
| Patients | LINE-1 (%) | |||
|---|---|---|---|---|
| N | (%) | Median (Q1–Q3) | ||
| Sex | ||||
| Man | 117 | (71.8) | 53.7 (38.5–72.7) | p = 0.8192 |
| Woman | 46 | (28.2) | 55.8 (38.1–70.6) | |
| Age (years) | ||||
| < 60 | 40 | (24.5) | 59.6 (38.6–75.3) | p = 0.0249 |
| 60–69 | 57 | (35.0) | 61.3 (42.2–73.0) | |
| ≥ 70 | 66 | (40.5) | 47.8 (24.1–59.3) | |
| T stagea | ||||
| T1 | 24 | (14.7) | 70.0 (44.8–77.8) | p = 0.1137 |
| T2 | 51 | (31.3) | 61.8 (41.0–75.5) | |
| T3 | 57 | (35.0) | 51.0 (30.2–65.0) | |
| T4 | 31 | (19.0) | 53.4 (38.6–75.3) | |
| N stagea | ||||
| N0 | 17 | (10.4) | 51.0 (20.0–66.2) | p = 0.2205 |
| N1 | 28 | (17.2) | 50.0 (34.2–74.4) | |
| N2 | 105 | (64.4) | 59.7 (41.9–73.0) | |
| N3 | 13 | (8.0) | 52.2 (30.3–69.5) | |
| Stagea | ||||
| III | 38 | (23.3) | 48.9 (27.1–70.1) | p = 0.1475 |
| IV | 125 | (76.7) | 58.2 (41.0–72.7) | |
| HPV-status | ||||
| Negative | 110 | (67.5) | 50.1 (30.3–66.2) | p < 0.0001b |
| Positive | 46 | (28.2) | 71.2 (58.6–78.2) | |
| Unknown | 7 | (4.3) | 46.1 (29.5–51.1) | |
| Surgery | ||||
| No | 73 | (44.8) | 58.6 (42.1–77.2) | p = 0.1144 |
| Yes | 90 | (55.2) | 52.5 (37.9–70.1) | |
aTNM staging according to the American Joint Committee on Cancer 7th Edition.
bExcluding missing values
Fig. 1.
Oncological outcomes according to level of LINE-1 methylation. Overall survival (a) and progression-free survival (b) were calculated through the Kaplan–Meier method. LINE-1 methylation was categorized using a recursive procedure which maximizes the difference in OS
Table 2.
Hazard ratio (HR) and corresponding 95% confidence interval (CI) for progression-free survival and overall survival in 163 patients with stage III–IV oropharyngeal squamous cell carcinoma
| Patients | Overall survival | Progression-free survival | |||||
|---|---|---|---|---|---|---|---|
| Events | HR (95% CI)a | HR (95% CI)b | Events | HR (95% CI)a | HR (95% CI)b | ||
| Sex | |||||||
| Man | 117 | 64 | Reference | Reference | 69 | Reference | Reference |
| Woman | 46 | 26 | 1.03 (0.64–1.63) | 1.05 (0.65–1.70) | 29 | 1.20 (0.77–1.87) | 1.28 (0.81–2.02) |
| Age (years) | |||||||
| < 60 | 40 | 20 | Reference | Reference | 22 | Reference | Reference |
| 60–69 | 57 | 26 | 1.00 (0.54–1.83) | 1.06 (0.57–1.98) | 31 | 1.11 (0.63–1.97) | 1.18 (0.66–2.15) |
| ≥ 70 | 66 | 44 | 1.91 (1.10–3.33) | 1.47 (0.83–2.64) | 45 | 1.80 (1.05–3.07) | 1.54 (0.88–2.58) |
| T stagec | |||||||
| T1-T2 | 75 | 35 | Reference | Reference | 37 | Reference | Reference |
| T3-T4 | 88 | 55 | 1.74 (1.13–2.69) | 1.42 (0.89–2.25) | 61 | 1.76 (1.16–2.68) | 1.32 (0.84–2.08) |
| N stagec | |||||||
| N0-N1 | 45 | 29 | Reference | Reference | 30 | Reference | Reference |
| N2 | 105 | 51 | 0.79 (0.50–1.26) | 1.20 (0.71–2.02) | 58 | 0.86 (0.55–1.34) | 1.13 (0.69–1.85) |
| N3 | 13 | 10 | 2.29 (1.10–4.78) | 2.87 (1.34–6.14) | 10 | 2.29 (1.10–4.73) | 2.65 (1.26–5.59) |
| HPV-status | |||||||
| Negative | 110 | 76 | Reference | Reference | 82 | Reference | Reference |
| Positive | 46 | 13 | 0.30 (0.16–0.54) | 0.43 (0.23–0.81) | 14 | 0.29 (0.16–0.51) | 0.39 (0.21–0.72) |
| Surgery | |||||||
| No | 73 | 40 | Reference | 43 | Reference | ||
| Yes | 90 | 50 | 1.17 (0.75–1.82) | 55 | 1.08 (0.70–1.65) | ||
| LINE-1 methylation | |||||||
| ≥ 55% | 80 | 34 | Reference | Reference | 40 | Reference | Reference |
| 35–54% | 48 | 28 | 1.60 (0.96–2.67) | 1.46 (0.85–2.49) | 29 | 1.41 (0.86–2.31) | 1.25 (0.75–2.07) |
| < 35% | 35 | 28 | 3.21 (1.89–5.45) | 2.76 (1.48–5.12) | 29 | 3.19 (1.92–5.30) | 2.39 (1.35–4.24) |
aEstimated from Cox proportional hazards model, adjusted for study center, sex, and age
bFurther adjusted for T stage, N stage, HPV status, and LINE-1 methylation. cTNM staging according to the American Joint Committee on Cancer 7th Edition
Association of LINE-1 methylation levels with HPV16 status
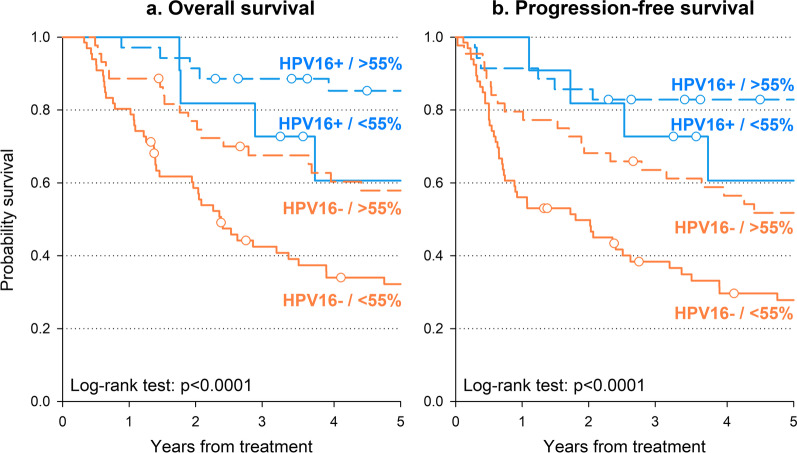
Potential interaction between HPV16 status and LINE-1 methylation levels was further investigated, dichotomizing LINE-1 as < 55% versus ≥ 55%. HPV16-positive patients with LINE-1 ≥ 55% showed the best 5-year OS (85.3%—Fig. 2a) and PFS (82.9%—Fig. 2b) in contrast with HPV16-negative patients with LINE-1 < 55% who reported the worst prognosis (32.2% and 27.8%, respectively). Interestingly, HPV16-positive patients with LINE-1 < 55% reported similar overall survival as HPV16-negative patients with LINE-1 ≥ 55%. These findings were confirmed by multivariable analyses (Additional file 1: Table S1), which showed a significantly increased risk of death or progression in HPV16-negative patients with LINE-1 < 55% compared to HPV16-positive patients with LINE-1 ≥ 55% (HR for death: 4.83, 95% CI 2.24–10.38; HR for death/progression: 4.54, 95% CI 2.18–9.48).
Fig. 2.
Oncological outcomes according to HPV16 status and level of LINE-1 methylation. Overall survival (a) and progression-free overall survival (b) were calculated through the Kaplan–Meier method, stratifying patients according to HPV16 status and LINE-1 methylation level
Association of LINE-1 methylation levels with p53 expression
Since p53 might control LINE-1 methylation, we then analyzed the association between p53 expression status and LINE-1 methylation levels in a sub-group of 89 patients (Additional file 2: Table S2). To this end, p53 expression pattern was categorized into three groups according to the overall intensity of nuclear staining of tumor cells and the extent of stained cells (i.e., 0%, 1–49%, ≥ 50%). Figure 3 shows mean LINE-1 methylation according to p53 expression and HPV16 status. For p53 expression < 50%, LINE-1 methylation increased with increasing p53 regardless of HPV16 status (p = 0.0003). Among HPV16-negative patients with p53 ≥ 50%, LINE-1 methylation levels declined and remained stable at approximately 43%. No HPV16-positive patients reported p53 ≥ 50%.
Fig. 3.
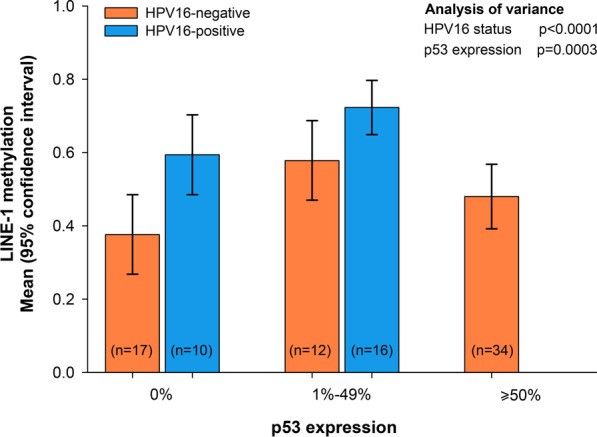
LINE-1 methylation according to HVP16 status and p53 expression pattern. Mean LINE-1 methylation, and corresponding 95% confidence intervals, was reported for HPV16 status and level of p53 expression. The independent association of HPV16 status and p53 expression with LINE-1 methylation was evaluated through the analysis of variance
Identification of differentially methylated CpG sites in relapsing patients
We recently reported a significant decrease in LINE-1 methylation in OPSCC patients who relapsed within 2 years from the end of treatment, especially in HPV16-negative ones [18]. Therefore, the methylation levels (beta-values) of CpG sites were analyzed in 5 HPV16-negative OPSCC patients who relapsed within 2 years and in 5 who did not, in order to investigate whether the differentially methylated regions between the two subgroups mapped within LINE-1 elements. Unfortunately, the sample size was limited due to the amount of genomic DNA required for the analysis (Additional file 2: Table S2).
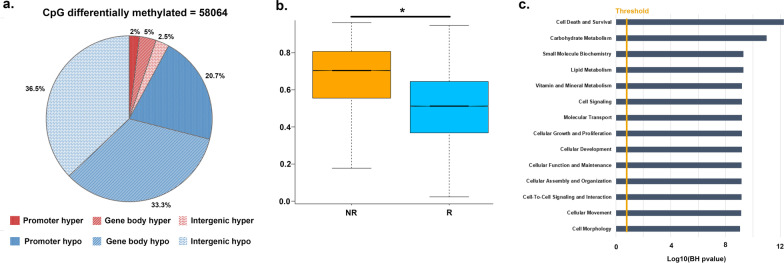
Results indicated that there were 58,064 CpG (|DB|≥ 0.15 and p < 0.05) with a difference in the methylation level between OPSCC patients who relapsed compared to those who were relapse-free for at least 24 months after the treatment; in particular, 4500 CpG sites were hypermethylated, whereas 53,564 were hypomethylated (Fig. 4a). Therefore, a significantly lower content of CpG methylation could be found in relapsed (median value = 0.51) respect to relapse-free OPSCC patients (median value = 0.70) (Fig. 4b, Additional file 5: Figure S1). Among the 58,064 differentially methylated CpG, 38.3% overlapped with gene bodies, 39.0% were intergenic, whereas 22.7% overlapped with gene promoters (Fig. 4a, Additional file 3: Table S3). To identify DNA methylation alterations within the promoter regions, we focused on CpG sites located within the TSS1500, TSS200, 5’ UTR, and first exon. A global hypomethylation pattern was still observed since 12,036 of 17,819 CpGs were significantly hypomethylated in OPSCC patients who relapsed within 2 years. To further explore the biological roles of these CpGs, we performed gene ontology (GO) enrichment analysis. Results indicated that the most significantly enriched GO terms were molecular functions of potential importance for cancer development and progression, including cellular growth and proliferation, cell-to-cell signaling and interaction, cellular movement, and cell morphology (Fig. 4c). Notably, 3743 CpGs differentially methylated overlapped with LINE-1 elements (3502 hypomethylated and 241 hypermethylated).
Fig. 4.
DNA methylation analysis in HPV16-negative OPSCC patients. a Pie chart showing the percentage of differentially methylated CpG in promoter, gene body, and intergenic regions. b Box plot showing the average methylation level (beta-value) of all CpG differentially methylated in not relapsed (NR) vs relapsed (R) HPV16-negative OPSCC patients. c Histogram showing the molecular function where the genes showing differentially methylated promoter are involved
Correlation between CpG methylation and gene expression
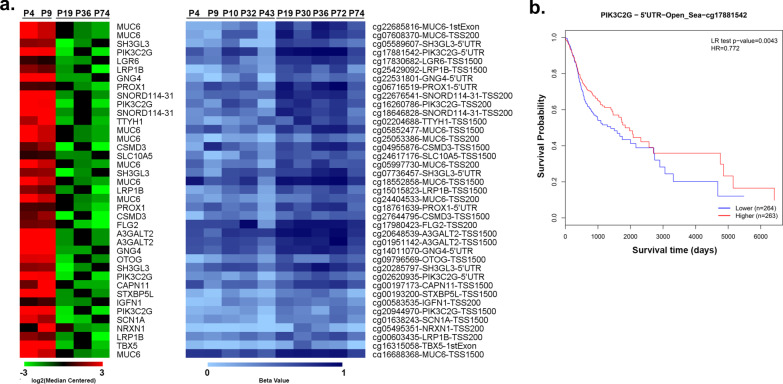
Aberrant DNA methylation patterns might contribute to differential survival through altered expression of the respective genes. Hence, gene expression profile was evaluated by RNA sequencing on 2 relapsed and 3 relapse-free HPV16-negative OPSCC patients, leading to the identification of 367 differentially expressed genes, of which 286 were down-regulated and 81 up-regulated (|FC|≥ 1.5 and adj-pval ≤ 0.05). Since it is generally accepted that promoter methylation is associated with decreased transcription of downstream genes and vice versa, gene expression and DNA methylation profiles were integrated to determine whether there were any connections. Setting |FC|≥ 1.5 and |Db|≥ 0.15 as cutoff, we identified 29 differentially expressed genes and 59 differentially methylated CpGs. We focused in particular on hypomethylated and up-regulated genes, thereby identifying 20 genes (Fig. 5a, Additional file 4: Table S4), most of which (16/20) overlapped with LINE-1 elements (Additional file 6: Figure S2–Additional file 9: Figure S5), with the exception of FLG2, MUC6, SLC10A5 and SNORD114-31 (Additional file 10: Fig. S6). These results suggest that LINE-1 hypomethylation might affect gene expression in OPSCC. Notably, a high number of LINE-1 elements were found within PIK3C2G, which represented one of the most hypomethylated and up-regulated genes in patients who early relapsed. Since PIK3C2G hypomethylation has been recently found to predict tumor relapse and shorter OS in ovarian cancer [37], the prognostic roles of PIK3C2G CpG in HNSCC were explored through the public database MethSurv (https://biit.cs.ut.ee/methsurv/) [38]. Only PIK3C2G cg17881542 was present in this dataset and, although the data were of borderline significance (log-rank test, p = 0.004; HR = 0.77, 95% CI 0.59–1.01), we could observe that high methylation levels of cg17881542 were associated with favorable prognosis in HNSCC (Fig. 5b).
Fig. 5.
Integrative analysis of gene expression and DNA methylation in HPV16-negative OPSCC patients. a Leftside: heatmap summarizing expression data for the up-regulated genes in HPV16-negative OPSCC patients (P) who relapsed within 2 years from the end of treatment (P4, P9) with those who did not (P19, P36, P74). Data are shown as normalized expression values in log2 and centered on the median value. Rightside: heatmap showing the methylation values (beta value) in relapsed (P4, P9, P10, P32, P43) vs relapsed-free (P19, P30, P36, P72, P74) HPV16-negative OPSCC patients. b Kaplan–Meier estimates overall survival according to high- and low-methylation levels of the PIK3C2G cg17881542 in MethSurv
Discussion
In the last years, accumulating evidence indicated that loss of LINE-1 methylation is crucially involved in carcinogenesis; in fact, LINE-1 demethylation was found to promote genomic and chromosomal instabilities [39] and to activate the transcription of cancer-related genes as well [29, 30]. In addition, the epigenetic status of LINE-1 has been widely associated with patient outcomes in several malignancies (for review see [14]). In this context, we have recently demonstrated that LINE-1 methylation levels were lower in OPSCC patients who relapsed within 24 months [18], thus indicating that the overall level of genomic DNA methylation might have an impact on early OPSCC relapse risk. Consistently, this study demonstrated that hypomethylation of LINE-1 correlated with significantly poorer PFS and OS in an expanded retrospective cohort of 163 OPSCC patients. Although stratified survival analyses highlighted the prognostic significance of LINE-1 hypomethylation in OPSCC patients irrespective of HPV16 status, the lowest level of LINE-1 element methylation was observed in HPV16-negative tumors. Collectively, these results corroborate the finding that LINE-1 hypomethylation may be an effective biomarker to predict OPSCC survival and further suggest that epigenetic changes could overall contribute to OPSCC biology and could be partially responsible for the biological and clinical differences between HPV16-positive and HPV16-negative OPSCC patients. Measuring LINE-1 methylation levels at diagnosis may aid the clinician to schedule the frequency of follow-up examination and/or to choose the aggressiveness of treatment, especially in HPV-negative OPSCC patients. Importantly, global hypomethylation could allow rapidly proliferating and highly mutated tumors to escape immune reaction and to become resistant to immunotherapy [40]. Therefore, LINE-1 hypomethylation may also represent an independent indicator of poor immunotherapy responses in HPV-negative OPSCC tumors.
A recent study of de Carvalho et al. has shown that HPV-negative OPSCC tumors usually have a high mutation burden respect to HPV-positive ones [41]. In particular, TP53 mutations are frequently found in OPSCC driven by alcohol and tobacco, whereas their presence has been reported in only a small subset of HPV-related OPSCC so far [42–46]. Unfortunately, in our study, OPSCC patients were not investigated for TP53 mutation by sequencing analysis. However, since the complete absence of immunolabeling or IHC overexpression for p53 (≥ 50% positive cells) has been found to closely correlate with the presence of TP53 mutations in several tumor types [31–35], p53 protein expression, as determined by IHC, was used as surrogate for TP53 mutation status. In fact, according to several studies, TP53 missense mutations resulted in nuclear accumulation and p53 overexpression, whereas absence of p53 staining was associated with nucleotide deletions or non-sense mutations that resulted in protein truncation. On the other hand, tumors with wild-type TP53 displayed intermediate immunolabeling patterns [31–35]. When we evaluated the association between LINE-1 methylation status and p53 expression, we observed that p53 absence or a strong and diffuse pattern of p53 expression correlated with lower LINE-1 methylation levels in HPV16-negative OPSCC patients, whereas no p53 overexpression was found in patients with HPV16 infection, which is consistent with the mechanism of p53 degradation by HPV16 E6 [47]. Chromatin immunoprecipitation studies indicated extensive p53 enrichment within LINE-1 promoter region of the retrotransposon element LINE-1, thus suggesting that p53 might directly bind and recruit a variety of epigenetic regulators (i.e., DNA methyltransferases) in order to silence retroelements [25]. Hence, it seems plausible that aberrant LINE-1 hypomethylation may occur along with TP53 mutations. Consistently, an increased expression of the LINE-1 retrotransposable element ORF1 protein has often been correlated with TP53 mutations and aberrant p53 expression [23, 48, 49].
DNA methylation analysis demonstrated that genome-wide average methylation level at CpGs was significantly lower in OPSCC patients who relapsed within two years, thus confirming the important role played by DNA hypomethylation in OPSCC progression. Although the sample size included was limited, our data suggested that the methylation status of PIK3C2G gene might have particular relevance in OPSCC since it appeared to be strictly associated with LINE-1 elements (Additional file 8: Figure S4). Notably, the protein encoded by PIK3C2G represents a key extracellular signaling molecule participating in the PI3K/Akt signaling pathway. Activation of this pathway has shown to contribute to the development of resistance to chemotherapy and radiotherapy in several cancers, including HNSCC [50]. Consistently, HNSCC have shown mutations in more than one PI3K pathway molecule, including PIK3C2G [51]. Of interest, by using GeneMANIA (https://genemania.org/) [52], we found that PIK3C2G is co-expressed with GNG4 and NRXN1 (Additional file 11: Figure S7) which were among the 20 hypomethylated/up-regulated genes, and contained several LINE-1 elements within their promoters as well. GNG4 is a member of the G-protein family and, similar to PIK3C2G, has been closely associated with PI3K/Akt signaling pathway in HNSCC [53]. Although GNG4 has been reported to be hypermethylated and down-regulated in bladder cancer [54], breast cancer [55], and glioblastoma [56], other studies have shown that GNG4 expression was significantly up-regulated in lung carcinoma [57], and colorectal cancer [58]. More interestingly, in a paper by You et al., GNG4 has been listed as one of the up-regulated genes potentially involved in radioresistance in HNSCC [53]. NRXN1 represents a single-pass transmembrane protein and has been recently described as a potential novel target for antibody–drug conjugate therapy in small cell lung cancer [59]. Of note, NRXN1 was indicated as hypomethylated and overexpressed in HPV-positive HNSCC [60], thus suggesting that the role of NRXN1 should be better elucidated in the two OPSCC subtypes.
Despite these findings, our study has some limitations. First, this study was carried out on a retrospective cohort. Second, the detection of HPV in our samples was restricted to HPV16, the most common high-risk subtype associated with OPSCC, whereas less common subtypes (i.e., HPV18, HPV31, HPV33 and HPV52) were not evaluated. However, although the clinical behavior and pathogenesis of non-HPV16-OPSCC are less well known, recent studies indicated that the survival benefit of HPV-positivity might be mainly attributed to HPV16 genotype in OPSCC [61], whereas OS among non-HPV16 was even poorer than for HPV-negative HNSCC patients [62]. Third, FFPE material was not sufficient to identify genetic alterations of the TP53 gene and to perform genome-wide DNA methylation and RNA-seq analyses in all OPSCC patients. Fourth, the formaldehyde-induced DNA inter-strand crosslinks might interfere with bisulfite conversion [63], which is a critical step for the quantitative analysis of LINE-1 methylation. In fact, unconverted cytosines, if present, would lead to possible bias in qMSP analysis. Finally, since qMSP assay covers a limited number of CpG sites within the promoter region, the clinical value of the LINE-1 methylation status might be representative only of the genomic location analyzed [64].
Conclusion
In conclusion, our results clearly indicated that LINE-1 hypomethylation was associated with poorer OS and PFS in OPSCC patients regardless of their HPV16 status. Intriguingly, genome-wide methylation analysis suggested that hypomethylation of LINE-1 elements might promote the transcription of genes that are potentially involved in OPSCC. At present, a prospective study is ongoing to validate the prognostic significance of LINE-1 methylation in a larger sample cohort of OPSCC patients. Future research is also needed to elucidate whether p53 may affect retrotransposon activity in OPSCC cells and to better understand whether LINE-1 activity plays a direct role in OPSCC progression.
Supplementary Information
Additional file 1: Table S1. Hazard ratio (HR) and corresponding 95% confidence interval (CI) for progression-free survival and overall survival in 163 patients with stage III–IV oropharyngeal squamous cell carcinoma according to combination of HPV status and LINE-1 methylation
Additional file 2: Table S2. Sociodemographic and clinical characteristics in sub-group analyses
Additional file 3: Table S3. List of differentially methylated CpG identified comparing relapsed vs not relapsed patients
Additional file 4: Table S4. List of differentially expressed genes correlated with differentially methylated CpGs in promoter region identified comparing relapsed versus not relapsed patients
Additional file 5: Figure S1. DNA methylation analysis in 5 relapse-free and in 5 relapsed HPV16-negative OPSCC patients. Box plot comparing the methylation level (beta-value) of the non-relapsed (NR) and the relapsed (R) OPSCC patients.
Additional file 6: Figure S2. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 7: Figure S3. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 8: Figure S4. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 9: Figure S5. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 10: Figure S6. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 11: Figure S7. Co-expression network of PIK3C2G, GNG4 and NRXN1 based on GeneMANIA. Co-expression: two genes are linked if their expression levels are similar across conditions in a gene expression study.
Acknowledgements
The authors are grateful to Mrs Luigina Mei, Unit of Cancer Epidemiology at Centro di Riferimento Oncologico (CRO) IRCCS in Aviano, for editorial assistance.
Abbreviations
- CI
Confidence interval
- CpG
Cytosine–phosphate–guanine
- DB
DeltaBeta
- FFPE
Formalin-fixed paraffin-embedded
- GO
Gene ontology
- qMSP
Quantitative methylation-specific PCR
- HNSCC
Head and neck squamous cell carcinoma
- HPV
Human papillomavirus
- HR
Hazard ratio
- IHC
Immunohistochemistry
- IQR
Interquartile range
- LINE-1
Long interspersed nuclear elements 1
- OPSCC
Oropharyngeal squamous cell carcinoma
- OS
Overall survival
- PFS
Progression-free survival
- TP53
Tumor protein p53
- TSS
Transcriptional start site
Author contributions
MC and VL participated in the design of the study, acquisition of the clinical data, data interpretation and edited the manuscript. GG performed the bioinformatics analyses, contributed in data interpretation, and drafted the manuscript. RG, AA, LB, BM, and EF contributed in molecular assays and data acquisition. SS, EDA, CF, AM, IT, SR, VB, VG, GB, MG, GF, and EV participated in the acquisition of the clinical data and data interpretation. RD, AW, AS, and LS edited the manuscript. PBR participated in the acquisition of the clinical data, data interpretation, and manuscript drafting. JP performed the statistical analyses, contributed in data interpretation, and drafted the manuscript. GF and EF designed and coordinated the study, contributed to data analysis, and drafted the manuscript. The work reported in the paper has been performed by the authors, unless clearly specified in the text. All authors read and approved the final manuscript.
Funding
This work was partially supported by Ministero della Salute Ricerca Corrente and 5 × 1000 Intramural Grant from CRO, AIRC Foundation for Cancer Research (grant n.er IG-23068) and Regione Campania (POR Campania FESR 2014/2020—Azione 1.5, grant GENOMAeSALUTE, CUP B41C17000080007 and RIS3 ‘La Campania lotta contro il cancro’, grant Rare-Plat-Net, CUP B63D18000380007).
Data availability
The Methylation EPIC raw data and RNA-Seq are publicly available in ArrayExpress repository under accession number E-MTAB-11152 and E-MTAB-12032, respectively. Other data that support the findings of this study are available from the corresponding authors upon request. Reviewers login: Username: Reviewer_E-MTAB-11152, Password: qrzridwu.
Declarations
Ethics approval and consent to participate
The study was approved by the local Independent Ethic Committees (CRO-2019-13, 733/AULSS2, 5/2020/OSS/AOUMO). Participants provided written informed consent for inclusion in the study.
Competing interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Mariateresa Casarotto and Valentina Lupato contributed equally to this work as co-first author
Giuseppe Fanetti and Elisabetta Fratta contributed equally to this work as co-last author
References
- 1.Carlander AF, Jakobsen KK, Bendtsen SK, Garset-Zamani M, Lynggaard CD, Jensen JS, Grønhøj C, Buchwald CV. A contemporary systematic review on repartition of HPV-positivity in oropharyngeal cancer worldwide. Viruses. 2021;13(7):1326. doi: 10.3390/v13071326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Castellsagué X, Alemany L, Quer M, Halec G, Quirós B, Tous S, Clavero O, Alòs L, Biegner T, Szafarowski T, et al. HPV involvement in head and neck cancers: comprehensive assessment of biomarkers in 3680 patients. J Natl Cancer Inst. 2016;108(6):djv403. doi: 10.1093/jnci/djv403. [DOI] [PubMed] [Google Scholar]
- 3.Fakhry C, Fung N, Tewari SR, D'Souza G. Unique role of HPV16 in predicting oropharyngeal cancer risk more than other oncogenic oral HPV infections. Oral Oncol. 2020;111:104981–104981. doi: 10.1016/j.oraloncology.2020.104981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tan PF, Westra WH, Chung CH, Jordan RC, Lu C, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010;363(1):24–35. doi: 10.1056/NEJMoa0912217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Faraji F, Eisele DW, Fakhry C. Emerging insights into recurrent and metastatic human papillomavirus-related oropharyngeal squamous cell carcinoma. Laryngoscope Investig Otolaryngol. 2017;2(1):10–18. doi: 10.1002/lio2.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grønhøj C, Jakobsen KK, Jensen DH, Rasmussen J, Andersen E, Friborg J, von Buchwald C. Pattern of and survival following loco-regional and distant recurrence in patients with HPV+ and HPV- oropharyngeal squamous cell carcinoma: a population-based study. Oral Oncol. 2018;83:127–133. doi: 10.1016/j.oraloncology.2018.06.012. [DOI] [PubMed] [Google Scholar]
- 7.Gillison ML, Trotti AM, Harris J, Eisbruch A, Harari PM, Adelstein DJ, Jordan RCK, Zhao W, Sturgis EM, Burtness B, et al. Radiotherapy plus cetuximab or cisplatin in human papillomavirus-positive oropharyngeal cancer (NRG Oncology RTOG 1016): a randomised, multicentre, non-inferiority trial. Lancet. 2019;393(10166):40–50. doi: 10.1016/S0140-6736(18)32779-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ryan WR, Xu MJ, Ochoa E, Plonowska-Hirschfeld KA, Zebolsky AL, Ha PK, Bewley AF, Mallen-StClair J, Joshi AS, Coffey CS, et al. Oncologic outcomes of human papillomavirus-associated oropharynx carcinoma treated with surgery alone: a 12-institution study of 344 patients. Cancer. 2021;127:3092–3106. doi: 10.1002/cncr.33611. [DOI] [PubMed] [Google Scholar]
- 9.Zakeri K, Dunn L, Lee N. HPV-associated oropharyngeal cancer de-escalation strategies and trials: past failures and future promise. J Surg Oncol. 2021;124(6):962–966. doi: 10.1002/jso.26696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Boscolo-Rizzo P, Furlan C, Lupato V, Polesel J, Fratta E. Novel insights into epigenetic drivers of oropharyngeal squamous cell carcinoma: role of HPV and lifestyle factors. Clin Epigenetics. 2017;9:124. doi: 10.1186/s13148-017-0424-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Durzynska J, Lesniewicz K, Poreba E. Human papillomaviruses in epigenetic regulations. Mutat Res. 2017;772:36–50. doi: 10.1016/j.mrrev.2016.09.006. [DOI] [PubMed] [Google Scholar]
- 12.Beisel C, Paro R. Silencing chromatin: comparing modes and mechanisms. Nat Rev Genet. 2011;12(2):123–135. doi: 10.1038/nrg2932. [DOI] [PubMed] [Google Scholar]
- 13.Ohka F, Natsume A, Motomura K, Kishida Y, Kondo Y, Abe T, Nakasu Y, Namba H, Wakai K, Fukui T, et al. The global DNA methylation surrogate LINE-1 methylation is correlated with MGMT promoter methylation and is a better prognostic factor for glioma. PLoS ONE. 2011;6(8):e23332–e23332. doi: 10.1371/journal.pone.0023332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ponomaryova AA, Rykova EY, Gervas PA, Cherdyntseva NV, Mamedov IZ, Azhikina TL. Aberrant methylation of LINE-1 transposable elements: a search for cancer biomarkers. Cells. 2020;9(9):2017. doi: 10.3390/cells9092017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lavasanifar A, Sharp CN, Korte EA, Yin T, Hosseinnejad K, Jortani SA. Long interspersed nuclear element-1 mobilization as a target in cancer diagnostics, prognostics and therapeutics. Clin Chim Acta. 2019;493:52–62. doi: 10.1016/j.cca.2019.02.015. [DOI] [PubMed] [Google Scholar]
- 16.Grundy EE, Diab N, Chiappinelli KB. Transposable element regulation and expression in cancer. FEBS J. 2022;289(5):1160–1179. doi: 10.1111/febs.15722. [DOI] [PubMed] [Google Scholar]
- 17.Chénais B. Transposable elements and human diseases: mechanisms and implication in the response to environmental pollutants. Int J Mol Sci. 2022;23(5):2551. doi: 10.3390/ijms23052551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Furlan C, Polesel J, Barzan L, Franchin G, Sulfaro S, Romeo S, Colizzi F, Rizzo A, Baggio V, Giacomarra V, et al. Prognostic significance of LINE-1 hypomethylation in oropharyngeal squamous cell carcinoma. Clin Epigenetics. 2017;9:58. doi: 10.1186/s13148-017-0357-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Saito K, Kawakami K, Matsumoto I, Oda M, Watanabe G, Minamoto T. Long interspersed nuclear element 1 hypomethylation is a marker of poor prognosis in stage IA non-small cell lung cancer. Clin Cancer Res. 2010;16(8):2418–2426. doi: 10.1158/1078-0432.CCR-09-2819. [DOI] [PubMed] [Google Scholar]
- 20.Burns KH. Transposable elements in cancer. Nat Rev Cancer. 2017;17(7):415–424. doi: 10.1038/nrc.2017.35. [DOI] [PubMed] [Google Scholar]
- 21.Sun X, Wang X, Tang Z, Grivainis M, Kahler D, Yun C, Mita P, Fenyö D, Boeke JD. Transcription factor profiling reveals molecular choreography and key regulators of human retrotransposon expression. Proc Natl Acad Sci. 2018;115(24):E5526–E5535. doi: 10.1073/pnas.1722565115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Leonova KI, Brodsky L, Lipchick B, Pal M, Novototskaya L, Chenchik AA, Sen GC, Komarova EA, Gudkov AV. p53 cooperates with DNA methylation and a suicidal interferon response to maintain epigenetic silencing of repeats and noncoding RNAs. Proc Natl Acad Sci USA. 2013;110(1):E89–98. doi: 10.1073/pnas.1216922110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wylie A, Jones AE, D'Brot A, Lu WJ, Kurtz P, Moran JV, Rakheja D, Chen KS, Hammer RE, Comerford SA, et al. p53 genes function to restrain mobile elements. Genes Dev. 2016;30(1):64–77. doi: 10.1101/gad.266098.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shin Y-J, Kim Y, Wen X, Cho N-Y, Lee S, Kim WH, Kang GH. Prognostic implications and interaction of L1 methylation and p53 expression statuses in advanced gastric cancer. Clin Epigenetics. 2019;11(1):77. doi: 10.1186/s13148-019-0661-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tiwari B, Jones AE, Caillet CJ, Das S, Royer SK, Abrams JM. p53 directly represses human LINE1 transposons. Genes Dev. 2020;34(21–22):1439–1451. doi: 10.1101/gad.343186.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Poeta ML, Manola J, Goldwasser MA, Forastiere A, Benoit N, Califano JA, Ridge JA, Goodwin J, Kenady D, Saunders J, et al. TP53 mutations and survival in squamous-cell carcinoma of the head and neck. N Engl J Med. 2007;357(25):2552–2561. doi: 10.1056/NEJMoa073770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhou G, Liu Z, Myers JN. TP53 mutations in head and neck squamous cell carcinoma and their impact on disease progression and treatment response. J Cell Biochem. 2016;117(12):2682–2692. doi: 10.1002/jcb.25592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Faulkner GJ, Kimura Y, Daub CO, Wani S, Plessy C, Irvine KM, Schroder K, Cloonan N, Steptoe AL, Lassmann T, et al. The regulated retrotransposon transcriptome of mammalian cells. Nat Genet. 2009;41(5):563–571. doi: 10.1038/ng.368. [DOI] [PubMed] [Google Scholar]
- 29.Hur K, Cejas P, Feliu J, Moreno-Rubio J, Burgos E, Boland CR, Goel A. Hypomethylation of long interspersed nuclear element-1 (LINE-1) leads to activation of proto-oncogenes in human colorectal cancer metastasis. Gut. 2014;63(4):635–646. doi: 10.1136/gutjnl-2012-304219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wolff EM, Byun H-M, Han HF, Sharma S, Nichols PW, Siegmund KD, Yang AS, Jones PA, Liang G. Hypomethylation of a LINE-1 promoter activates an alternate transcript of the MET oncogene in bladders with cancer. PLoS Genet. 2010;6(4):e1000917. doi: 10.1371/journal.pgen.1000917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Alsner J, Jensen V, Kyndi M, Vrou Offersen B, Vu P, Børresen-Dale A-L, Overgaard J. A comparison between p53 accumulation determined by immunohistochemistry and TP53 mutations as prognostic variables in tumours from breast cancer patients. Acta Oncol. 2008;47(4):600–607. doi: 10.1080/02841860802047411. [DOI] [PubMed] [Google Scholar]
- 32.Yemelyanova A, Vang R, Kshirsagar M, Lu D, Marks MA, Shih IM, Kurman RJ. Immunohistochemical staining patterns of p53 can serve as a surrogate marker for TP53 mutations in ovarian carcinoma: an immunohistochemical and nucleotide sequencing analysis. Mod Pathol. 2011;24(9):1248–1253. doi: 10.1038/modpathol.2011.85. [DOI] [PubMed] [Google Scholar]
- 33.Johnny N, McGraw KL, Komrokji RS, Sallman D, Al Ali NH, Padron E, Lancet JE, Moscinski LC, List AF, Zhang L. P53 protein overexpression by immunohistochemical staining is correlated with TP53 mutation burden and adverse clinical outcome in myelodysplastic syndromes. Blood. 2015;126(23):4121–4121. doi: 10.1182/blood.V126.23.4121.4121. [DOI] [Google Scholar]
- 34.Murnyák B, Hortobágyi T. Immunohistochemical correlates of TP53 somatic mutations in cancer. Oncotarget. 2016;7(40):64910–64920. doi: 10.18632/oncotarget.11912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fernandez-Pol S, Ma L, Ohgami RS, Arber DA. Immunohistochemistry for p53 is a useful tool to identify cases of acute myeloid leukemia with myelodysplasia-related changes that are TP53 mutated, have complex karyotype, and have poor prognosis. Mod Pathol. 2017;30(3):382–392. doi: 10.1038/modpathol.2016.206. [DOI] [PubMed] [Google Scholar]
- 36.Kalbfleisch JD, Prentice RL. The statistical analysis of failure time data. 2nd edn. 2002.
- 37.Chan DW, Lam W-Y, Chen F, Yung MMH, Chan Y-S, Chan W-S, He F, Liu SS, Chan KKL, Li B, et al. Genome-wide DNA methylome analysis identifies methylation signatures associated with survival and drug resistance of ovarian cancers. Clin Epigenetics. 2021;13(1):142. doi: 10.1186/s13148-021-01130-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Modhukur V, Iljasenko T, Metsalu T, Lokk K, Laisk-Podar T, Vilo J. MethSurv: a web tool to perform multivariable survival analysis using DNA methylation data. Epigenomics. 2018;10(3):277–288. doi: 10.2217/epi-2017-0118. [DOI] [PubMed] [Google Scholar]
- 39.Zhang X, Zhang R, Yu J. New understanding of the relevant role of LINE-1 retrotransposition in human disease and immune modulation. Front Cell Dev Biol. 2020;8:657. doi: 10.3389/fcell.2020.00657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jung H, Kim HS, Kim JY, Sun J-M, Ahn JS, Ahn M-J, Park K, Esteller M, Lee S-H, Choi JK. DNA methylation loss promotes immune evasion of tumours with high mutation and copy number load. Nat Commun. 2019;10(1):4278. doi: 10.1038/s41467-019-12159-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.de Carvalho AC, Perdomo S, Dos Santos W, Fernandes GC, de Jesus LM, Carvalho RS, Scapulatempo-Neto C, de Almeida GC, Sorroche BP, Arantes LMRB, et al. Impact of genetic variants in clinical outcome of a cohort of patients with oropharyngeal squamous cell carcinoma. Sci Rep. 2020;10(1):9970–9970. doi: 10.1038/s41598-020-66741-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mirghani H, Amen F, Blanchard P, Moreau F, Guigay J, Hartl DM. Lacau St Guily J: Treatment de-escalation in HPV-positive oropharyngeal carcinoma: ongoing trials, critical issues and perspectives. Int J Cancer. 2015;136(7):1494–1503. doi: 10.1002/ijc.28847. [DOI] [PubMed] [Google Scholar]
- 43.Cancer Genome Atlas Network Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature. 2015;517(7536):576–582. doi: 10.1038/nature14129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hong A, Zhang X, Jones D, Veillard AS, Zhang M, Martin A, Lyons JG, Lee CS, Rose B. Relationships between p53 mutation, HPV status and outcome in oropharyngeal squamous cell carcinoma. Radiother Oncol. 2016;118(2):342–349. doi: 10.1016/j.radonc.2016.02.009. [DOI] [PubMed] [Google Scholar]
- 45.de Carvalho AC, Melendez ME, da Silva SC, Palmero EI, Arantes L, Neto CS, Carvalho AL. Clinical and molecular characterization of surgically treated oropharynx squamous cell carcinoma samples. Pathol Oncol Res. 2019;25(3):1047–1058. doi: 10.1007/s12253-018-0462-0. [DOI] [PubMed] [Google Scholar]
- 46.Boscolo-Rizzo P, Schroeder L, Sacchetto V, Holzinger D, Da Mosto MC, Tirelli G, Dal Cin E, Mantovani M, Menegaldo A, Del Mistro A, et al. Absence of disruptive TP53 mutations in high-risk human papillomavirus-driven neck squamous cell carcinoma of unknown primary. Head Neck. 2019;41(11):3833–3841. doi: 10.1002/hed.25915. [DOI] [PubMed] [Google Scholar]
- 47.Crook T, Tidy JA, Vousden KH. Degradation of p53 can be targeted by HPV E6 sequences distinct from those required for p53 binding and trans-activation. Cell. 1991;67(3):547–556. doi: 10.1016/0092-8674(91)90529-8. [DOI] [PubMed] [Google Scholar]
- 48.Rodić N, Sharma R, Zampella J, Dai L, Taylor MS, Hruban RH, Iacobuzio-Donahue CA, Maitra A, Torbenson MS, Goggins M, et al. Long interspersed element-1 protein expression is a hallmark of many human cancers. Am J Pathol. 2014;184(5):1280–1286. doi: 10.1016/j.ajpath.2014.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Budania S, Sur D, Nangal J, Pilli S, Mukherjee K, Biswas M, Prasad R, Saxena S, Mandal PK. LINE-1 retrotransposon encoded ORF1p expression and promoter methylation in oral squamous cell carcinoma: a pilot study. Cancer Genet. 2020;244:21–29. doi: 10.1016/j.cancergen.2020.01.050. [DOI] [PubMed] [Google Scholar]
- 50.Glorieux M, Dok R, Nuyts S. The influence of PI3K inhibition on the radiotherapy response of head and neck cancer cells. Sci Rep. 2020;10(1):16208. doi: 10.1038/s41598-020-73249-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lui VW, Hedberg ML, Li H, Vangara BS, Pendleton K, Zeng Y, Lu Y, Zhang Q, Du Y, Gilbert BR, et al. Frequent mutation of the PI3K pathway in head and neck cancer defines predictive biomarkers. Cancer Discov. 2013;3(7):761–769. doi: 10.1158/2159-8290.CD-13-0103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Montojo J, Zuberi K, Rodriguez H, Bader GD, Morris Q. GeneMANIA: fast gene network construction and function prediction for Cytoscape. F1000Res. 2014;3:153. doi: 10.12688/f1000research.4572.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.You GR, Cheng AJ, Lee LY, Huang YC, Liu H, Chen YJ, Chang JT. Prognostic signature associated with radioresistance in head and neck cancer via transcriptomic and bioinformatic analyses. BMC Cancer. 2019;19(1):64. doi: 10.1186/s12885-018-5243-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhang Y, Fang L, Zang Y, Xu Z. Identification of core genes and key pathways via integrated analysis of gene expression and DNA methylation profiles in bladder cancer. Med Sci Monit. 2018;24:3024–3033. doi: 10.12659/MSM.909514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mao XH, Ye Q, Zhang GB, Jiang JY, Zhao HY, Shao YF, Ye ZQ, Xuan ZX, Huang P. Identification of differentially methylated genes as diagnostic and prognostic biomarkers of breast cancer. World J Surg Oncol. 2021;19(1):29. doi: 10.1186/s12957-021-02124-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Pal J, Patil V, Mondal B, Shukla S, Hegde AS, Arivazhagan A, Santosh V, Somasundaram K. Epigenetically silenced GNG4 inhibits SDF1α/CXCR4 signaling in mesenchymal glioblastoma. Genes Cancer. 2016;7(3–4):136–147. doi: 10.18632/genesandcancer.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhou B, Zhu W, Yuan S, Wang Y, Zhang Q, Zheng H, Zhu L, Xu J. High GNG4 expression is associated with poor prognosis in patients with lung adenocarcinoma. Thorac Cancer. 2022;13(3):369–379. doi: 10.1111/1759-7714.14265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Liang L, Huang J, Yao M, Li L, Jin XJ, Cai XY. GNG4 promotes tumor progression in colorectal cancer. J Oncol. 2021;2021:9931984. doi: 10.1155/2021/9931984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yotsumoto T, Maemura K, Watanabe K, Amano Y, Matsumoto Y, Zokumasu K, Ando T, Kawakami M, Kage H, Nakajima J, et al. NRXN1 as a novel potential target of antibody-drug conjugates for small cell lung cancer. Oncotarget. 2020;11(39):3590–3600. doi: 10.18632/oncotarget.27718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Degli Esposti D, Sklias A, Lima SC, Beghelli-de la Forest Divonne S, Cahais V, Fernandez-Jimenez N, Cros M-P, Ecsedi S, Cuenin C, Bouaoun L, et al. Unique DNA methylation signature in HPV-positive head and neck squamous cell carcinomas. Genome Med. 2017;9(1):33. doi: 10.1186/s13073-017-0419-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Skovvang A, Jensen JS, Garset-Zamani M, Carlander A, Grønhøj C, von Buchwald C. The impact of HPV genotypes on survival in HPV-positive oropharyngeal squamous cell carcinomas: a systematic review. Acta Otolaryngol. 2021;141(7):724–728. doi: 10.1080/00016489.2021.1927173. [DOI] [PubMed] [Google Scholar]
- 62.Chatfield-Reed K, Gui S, O'Neill WQ, Teknos TN, Pan Q. HPV33+ HNSCC is associated with poor prognosis and has unique genomic and immunologic landscapes. Oral Oncol. 2020;100:104488–104488. doi: 10.1016/j.oraloncology.2019.104488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wen X, Jeong S, Kim Y, Bae JM, Cho NY, Kim JH, Kang GH. Improved results of LINE-1 methylation analysis in formalin-fixed, paraffin-embedded tissues with the application of a heating step during the DNA extraction process. Clin Epigenetics. 2017;9(1):1. doi: 10.1186/s13148-016-0308-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.van Vlodrop IJH, Niessen HEC, Derks S, Baldewijns MMLL, van Criekinge W, Herman JG, van Engeland M. Analysis of promoter CpG island hypermethylation in cancer: location, location, location! Clin Cancer Res. 2011;17(13):4225–4231. doi: 10.1158/1078-0432.CCR-10-3394. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Hazard ratio (HR) and corresponding 95% confidence interval (CI) for progression-free survival and overall survival in 163 patients with stage III–IV oropharyngeal squamous cell carcinoma according to combination of HPV status and LINE-1 methylation
Additional file 2: Table S2. Sociodemographic and clinical characteristics in sub-group analyses
Additional file 3: Table S3. List of differentially methylated CpG identified comparing relapsed vs not relapsed patients
Additional file 4: Table S4. List of differentially expressed genes correlated with differentially methylated CpGs in promoter region identified comparing relapsed versus not relapsed patients
Additional file 5: Figure S1. DNA methylation analysis in 5 relapse-free and in 5 relapsed HPV16-negative OPSCC patients. Box plot comparing the methylation level (beta-value) of the non-relapsed (NR) and the relapsed (R) OPSCC patients.
Additional file 6: Figure S2. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 7: Figure S3. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 8: Figure S4. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 9: Figure S5. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 10: Figure S6. Mapping of the LINE-1 elements. Screenshots from Genome Browser representing the 20 up-regulated genes with hypomethylated promoter region along with “Repeating Elements by RepeatMasker” track.
Additional file 11: Figure S7. Co-expression network of PIK3C2G, GNG4 and NRXN1 based on GeneMANIA. Co-expression: two genes are linked if their expression levels are similar across conditions in a gene expression study.
Data Availability Statement
The Methylation EPIC raw data and RNA-Seq are publicly available in ArrayExpress repository under accession number E-MTAB-11152 and E-MTAB-12032, respectively. Other data that support the findings of this study are available from the corresponding authors upon request. Reviewers login: Username: Reviewer_E-MTAB-11152, Password: qrzridwu.