Abstract
A key component of the insect immune response is melanin production, including within nodules, or aggregations of immune cells surrounding microbes. Melanization produces oxidative and toxic intermediates that limit microbial infections. However, a direct fungicidal role of melanin during infection has not been demonstrated. We previously reported that the fungus Cryptococcus neoformans is encapsulated with melanin within nodules of Galleria mellonella hosts. Here we developed techniques to study melanin’s role during C. neoformans infection in G. mellonella. We provided evidence that in vivo melanin-encapsulation was fungicidal. To further study immune melanization, we applied tissue-clearing techniques to visualize melanized nodules in situ throughout the larvae. Further, we developed a time-lapse microscopy protocol to visualize the melanization kinetics in extracted hemolymph following fungal exposure. Using this technique, we found that cryptococcal melanin and laccase enhance immune melanization. We extended this approach to study the fungal pathogens Candida albicans and Candida auris. We find that the yeast morphologies of these fungi elicited robust melanization responses, while hyphal and pseudohyphal morphologies were melanin-evasive. Approximately 23% of melanin-encapsulated C. albicans yeast can survive and breakthrough the encapsulation. Overall, our results provide direct evidence that immune melanization functions as a direct antifungal mechanism in G. mellonella.
Subject terms: Fungal host response, Fungal pathogenesis, Fungal immune evasion
Real-time imaging of fungal infection and tissue clearing in the wax moth, Galleria mellonella, help evaluate the role of melanin in the insect host immune response to fungal pathogens.
Introduction
Insects occupy essential niches in global ecosystems, including many that directly affect human health and survival1. In addition, insects serve as powerful model systems for infectious disease research, and help to reduce reliance on vertebrates recommended by “3R”—Replace, Reduce, and Refine—programs2. Insects are also targeted by environmental pathogens and have evolved complex immune mechanisms that partially overlap with mammalian innate immunity3. Understanding the dynamics of insect-pathogen interactions and the factors involved is vital to both ensure ecosystem stability and establish invertebrate immunological models in research.
Fungi are an important class of pathogens for insects, and emerging fungal pathogens are predicted to become bigger threats to human health and agriculture in the coming years4,5. Consequently, studying host-fungal interactions using insect models is important and timely. Although insects do not produce antibodies or other mammalian-like adaptive immune responses, the antifungal immune defenses of insects involve cell-mediated and humoral innate immune processes3. Hemocytes, the immune cells of invertebrates which circulate in the hemolymph, have roles comparable to macrophages and neutrophils in mammals. Hemocytes are responsible for clearance of fungi via phagocytosis, release of extracellular damaging reactive oxygen species (ROS) and inflammatory molecules, and the creation of granuloma-like structures through a process called nodulation6. During nodulation, hemocytes surround the microbe and form an aggregate of insect cells, within which, clotting factors, immune enzymes, and immune complexes are released and activated6–8. These structures immobilize the fungus and lead to death. In Lepidopteran species, the production of prostaglandins by the plasmatocyte subset of hemocytes cause the lysis of other hemocytes called oenocytoids, resulting in the release of antimicrobial peptides, signaling molecules, and enzymes important to immune function9–11. One class of host enzymes that are often released and activated during oenocytoid lysis and nodulation are phenoloxidases (PO)9,11. POs are enzymes responsible for converting catecholamines in the hemolymph into DOPA melanin12. DOPA melanins, and melanins more broadly, are black-brown pigments contribute to insect immune defense and wound repair13. Melanization produces oxidative species and cytotoxic intermediates that are hypothesized to result in the death of the microbe12,14. Additionally, melanin may act as a physical barrier, restricting gas exchange and nutrient uptake, and thus prevent fungal replication and dissemination to other tissues15. At this time, in vitro evidence strongly links PO activity and resulting melanin intermediates with killing of fungi, bacteria, and viruses16–18, but comparable direct evidence for the microbicidal effect of POs and their toxic intermediates in vivo during insect infections is challenging to measure directly. Consequently, obtaining direct evidence that the process of melanization is fungicidal in vivo is important for establishing insect melanin as an important antifungal mechanism.
Larvae of the wax moth Galleria mellonella are commonly used as a model organism for studying fungal pathogenesis3. G. mellonella larvae are readily available in large numbers at low cost. Their larger size (2–3 cm) relative to other model insects such as Drosophila melanogaster makes them amenable to experimental approaches requiring larger volumes of hemolymph, insect hemocytes, and soluble immune factors. The study of G. mellonella hemolymph can prove valuable for understanding insect immune responses to infection. G. mellonella are also commonly used as a model for studying mammalian pathogens, including human pathogenic fungi Cryptococcus neoformans and Candida albicans19–21. While G. mellonella is a model for mammalian fungal infections because of similarities between the G. mellonella immune responses and the mammalian innate immune responses3, a more thorough understanding of the insect immune response is needed to fully benefit from studying host-microbe interactions in G. mellonella.
The differences between mammalian versus insect hosts also provide important insights into host-microbe interactions and mechanisms of fungal virulence factors3,19,20,22,23. For example, laccase, a fungal enzyme that oxidizes mammalian and insect catecholamines, is an important virulence factor in both hosts but, seemingly by distinct and diverse mechanisms19,24,25. In insects, fungal laccase appears to oxidize and deplete host catecholamines required for encapsulating the fungus in melanin, thus weakening the host immune response. Fungal laccases also help detoxify reactive oxygen species that form during insect immune processes24. In contrast, during mammalian infection, fungal laccase enhances production of fungal melanin to evade key mammalian immune defenses26. Thus, fungal melanins increase virulence in mammals, but decrease virulence in G. mellonella27–29. These seemingly different and opposite roles in which fungal melanins interact with mammalian and insect hosts is unexplored and as of now unexplained in literature.
In this study, we demonstrate the first direct evidence, to the best of our understanding, that the melanin-based immune response in vivo is fungicidal against C. neoformans. Fungal death was visualized using an endogenously expressed GFP-based fungal viability assay. A series of in situ, ex vivo, and in vitro methods to observe and study the melanin-based immune response of G. mellonella larvae. For in situ experiments, a previously published tissue-clearing protocol was modified to visualize tissue-specificity of melanized nodules within an intact larva. We have also developed time-lapse microscopy method for observing the melanin-based immune system in vitro. We applied this method to quantify the melanization kinetics during in vitro fungal infection, which improved our understanding of how fungal components, such as laccase and melanin, interact with insect melanization. We gained insight into how Candida albicans and Candida auris activate and evade the melanin-based immune response through morphological switching. Overall, our findings strongly suggest that melanization has direct antimicrobial activity in vivo in the insect immune system.
Results
Galleria mellonella kill C. neoformans through melanin encapsulation in nodules
Our group previously identified that C. neoformans infection activates the insect-melanin-based immune response and encapsulation response in G. mellonella30, where hemocytes aggregate and produce melanin around the fungus (Fig. 1a). To evaluate whether insect-melanin encapsulation kills C. neoformans, we assessed fungal viability during infection of G. mellonella using a GFP-expressing strain of C. neoformans, which expresses GFP under an actin promotor and shows loss of fluorescence signal upon cell death. A similar assay using the GFP signal as a proxy for parasite viability within mosquitoes was previously described31. The GFP-expressing strain was validated as a reporter for fungal viability C. neoformans using the standard dead cell stain propidium iodide (PI) (Supplementary Fig. 1a, b). In these experiments, propidium iodide staining was nearly mutually exclusive with GFP fluorescence in untreated fungal cells, and GFP fluorescence was lost following fungal death when treated with high temperatures (Fig. 1b, Supplementary Fig. 1a, b).
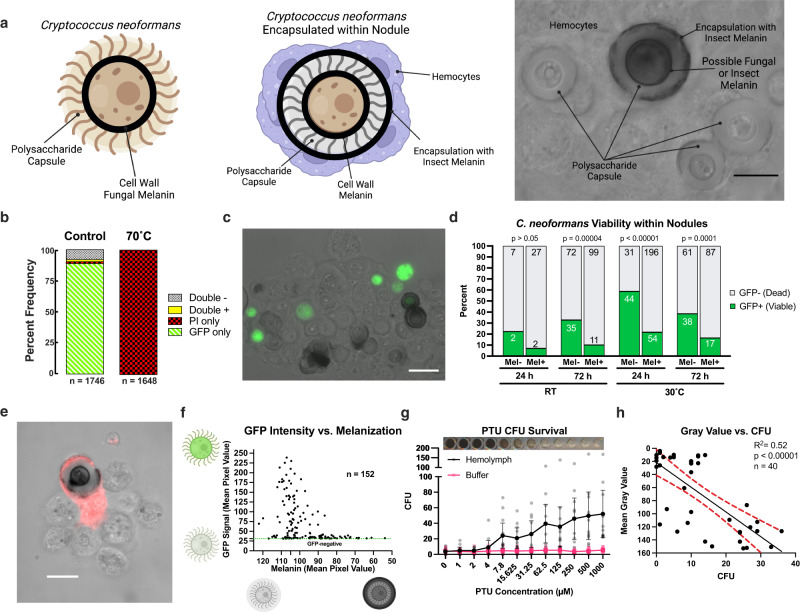
Fig. 1. The melanin-based immune response is implicated in killing C. neoformans during infection.
a During infection of G. mellonella, melanized nodules are formed within the hemolymph, which encapsulate C. neoformans. Melanin pigment produced by the fungus, is located typically within the cell wall, while the melanin produced by the insect during encapsulation within nodules is found around and within the fungal polysaccharide capsule. b GFP fluorescence is lost in heat-killed C. neoformans cells expressing GFP under an actin promoter. Data represents counts of 1640–1750 single cells from two separate biological replicates. c Using a GFP-expressing strain of C. neoformans, we can visualize cell viability within the nodule, with living fungi fluorescing green and dead cells having no signal. d Melanin-encapsulated fungi are statistically less likely to have a GFP-positive signal at 24 h after infection at 30 °C, and at 72 h post-infection at room temperature and 30 °C (Chi-squared test, [n = 217 cells] p < 0.00001, [n = 325 cells] p = 0.00004, and [n = 203 cells] p = 0.0001, respectively) indicating that melanin-encapsulation is associated with fungal death. Numbers within bars represent the number of fungi enumerated in that condition. e In GFP-positive cells, the brighter fluorescence signal correlates with little to no melanization, with no cells that have strong GFP signals and large amounts of melanin encapsulation (n = 152 independent cells). Each point represents data from one GFP-positive fungus within nodules from three biological replicates. f Propidium Iodide viability staining does not appear to penetrate the inner space of the nodule where the fungus is located, but the staining does show that the hemocytes surrounding the fungus are not viable. g Colony forming units (CFUs) correlate directly to melanin inhibition using PTU (inset, representative image of the 96-well plate) in vitro, which indicates that melanin has a role in controlling growth of C. neoformans. Line represents the mean and error bars indicate 95% Confidence Interval of four independent biological replicates with 10–12 total technical replicates. h CFUs also inversely correlated to the Mean Gray Value of these wells, meaning that the darker wells with more melanization had higher CFUs recovered compared to the melanin-inhibited wells (Simple Linear Regression, n = 40 biologically independent wells, p < 0.00001, F = 40.59). Each data point indicates measurements from individual wells from across three biological replicates, red dotted lines represent the 95% confidence interval of the linear regression. a–c, f Data shown is representative of three biological replicates. All experiments performed in biological triplicate, with g representing 5 biological replicates. Scale bars represent 10 µm.
Following infection of G. mellonella larvae, we found fewer GFP-positive melanin-encapsulated fungal cells within nodules of infected larvae compared to GFP-positive non-melanin-encapsulated fungal cells at both room temperature and at 30 °C at 24 and 72 h post-infection (Fig. 1c, d). Using the counts of GFP positivity of fungi within nodules, he non-melanin-encapsulated fungi had a survival rate of approximately 22, 33, 59, and 38% for the room temperature 24 and 72 h and the 30 °C at 24 and 72 h conditions, respectively (Fig. 1d). The melanin-encapsulated fungi had a survival rate of approximately 6, 10, 21, and 16% for the room temperature 24 and 72 h and the 30 °C at 24 and 72 h conditions, respectively (Fig. 1d). These data show that melanin-encapsulation was thus associated with approximately 21, 34, 92, and 35% greater amount of fungal death at the temperatures and times tested. This indicates that melanization contributes to a significant amount of fungal death during infection but is not the only nor predominant mechanism by which fungal cells are killed. Additional causes of fungal death in this model may include antimicrobial peptides and respiratory bursts, as previously described3. Melanin produced by C. neoformans in culture did not quench or obscure the GFP fluorescence, as determined by imaging melanized versus non-melanized C. neoformans H99-GFP (Supplementary Fig. 1c, d). Therefore, these results suggests that the immune melanization reaction resulted in fewer GFP-positive cells, which indicated that melanization kills C. neoformans in vivo during infections of G. mellonella.
Within nodules of encapsulated GFP-positive C. neoformans extracted from the hemolymph of infected G. mellonella, we measured the degree of immune melanin intensity and GFP fluorescence intensity. We found that the yeast with weaker GFP signal were associated with greater melanin encapsulation, in contrast to the population of brightly fluorescent cells that were not encapsulated with as much, or any, melanin (Fig. 1e), thus identifying an inverse correlation between degree of melanin encapsulation and fluorescence within GFP-positive cells. The occurrence of faint signal in some cells may suggest that cells are undergoing cell death or have recently been killed.
We attempted to use the standard dye propidium iodide (PI) to validate that GFP-negative cells within nodules are dead. Surprisingly, PI staining did not stain nodule-encapsulated yeast cells, although there was staining in some of the hemocytes that surrounded the yeast cells, and the external periphery of the nodules (Fig. 1f). The absence of PI staining of the fungi is likely due to inability of the dye to penetrate the nodule fully.
To further evaluate whether insect melanin promoted death of C. neoformans, fungal cells were incubated with whole extracted G. mellonella hemolymph in the presence of increasing concentrations of a phenoloxidase-specific competitive inhibitor32, phenylthiourea (PTU). This generated a range of insect-melanin inhibiting conditions. After 24 h, a small aliquot of the hemolymph-fungal mixture was plated on nutrient rich agar to assess fungal growth. Incubation of fungus melanization-inhibited hemolymph in vitro drastically enhanced fungal growth based on CFUs, while fungal growth was nearly abolished in melanin-competent hemolymph (Fig. 1g). The number of C. neoformans CFUs following 24 h incubation in the presence of hemocytes and PTU melanin inhibitor was directly proportional to the concentration of PTU (Fig. 1g), and thus inversely proportional to the degree of melanization (Fig. 1g, inset). PTU-mediated suppression of melanization was verified by quantifying melanin intensity by mean gray value of the wells of the 96-well plate (Fig. 1g, inset). The mean gray values of the wells showed that greater suppression of melanin production by PTU treatment correlated with increasing fungal growth based on CFUs (Fig. 1h). This result strongly suggests that immune melanization inhibits the growth of C. neoformans in vitro. Control conditions with buffer and PTU alone showed that incubation with PTU did not have a negative inhibitory effect on fungal growth, nor did it account for increased fungal growth in the high concentrations.
Development of a time-lapse microscopy method for studying hemocyte-fungal interactions and the melanization response
Since melanin encapsulation plays an important role in insect immunity and killing C. neoformans during infection, we sought to develop techniques to better study the melanization reaction and melanin-based host-fungal interactions. To record the kinetics of the hemolymph melanization response, we developed a protocol to extract insect hemocytes and monitor fungal interaction in vitro. (Fig. 2a). This method allowed for visualizing and quantifying the rate and magnitude of the anti-cryptococcal immune melanization response and was extended to study insect infection using different species of fungi, gene-deletion mutants, or application of isolated virulence factors (Supplementary Movie 1, 2). Using particle analysis in recorded frames, we quantified the area covered by melanization in minute intervals to determine the rate of hemolymph melanization (Fig. 2b–d). We observed variation in the extent of melanization between different experiments, likely due to variability in the fungi in the field of view, and biological variability from the non-isogenic G. mellonella larvae. To overcome the risk of interpreting variability-derived artifacts as results, each experiment was performed with a corresponding control (i.e., parental strain and mutant experiments were performed at same time, using the same stock of hemolymph and the same pool of extracted hemocytes). Despite the variation, the method was helpful in showing how morphological differences or fungal components interact with the melanin immune response, as well as comparing magnitude of melanization and speed of melanization activation between two groups.
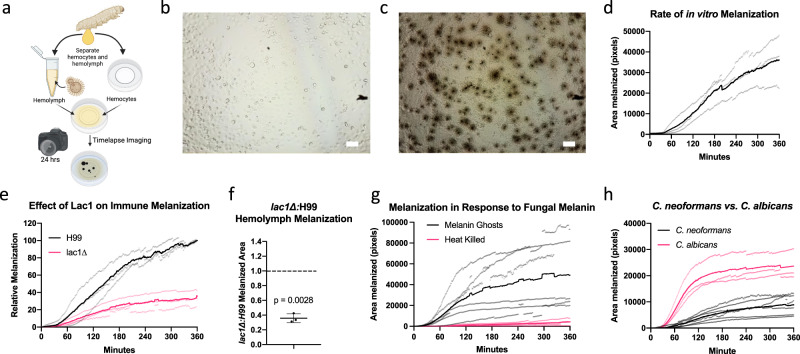
Fig. 2. Using in vitro time-lapse microscopy to visualize the melanin-based immune response.
a Using a developed time-lapse microscopy protocol, we were able to record the dynamics of hemolymph melanization in response to C. neoformans from 0 (b) up to 24 h (c). We can then use particle measurement software to visualize and quantify the melanization reactions over time (d), representative of three biological replicates. This method can be used to gain insight into how different virulence factors, such as laccase, influence the melanization response, where we see that the laccase knockout mutant causes less of a melanization response without much change in time until onset of melanization (e), represented by three biological replicates, and normalized to the wild-type control melanization area at 360 min. f Overall, the lac1∆ triggers ~40% as much of a melanization immune response compared to the WT (one sample t-test, n = 3 biologically independent movies, p = 0.0028, t = 18.78, df = 2). g Isolated fungal melanins are associated with activation of the insect-melanin-based immune response, whereas the heat-killed acapsular cryptococcal cells are not, as represented by 3–6 biological replicates. h Additionally, we can compare the activation of the melanin-based immune response from different fungal species such as C. neoformans and C. albicans, which strongly activates the melanization immune response, represented by 3–6 biological replicates. b–d, g are representative images and graphs from three biological replicates. Error bars indicate Standard Deviation. d, e, g, h Solid lines indicate mean melanization, and light colored points indicate individual replicate data points. Scale bars represent 50 µm.
By analyzing the melanization kinetics between different mutant and wild-type strains, we can determine how the mutant gene of interest affects fungal interactions with the melanization immune response. In C. neoformans, the laccase enzyme Lac1 is a major virulence factor, as it is responsible for the oxidation of catecholamines, and thus responsible for melanization33. It has also been suggested that laccase plays roles in virulence beyond melanization within mammalian hosts34. In a previous study the lac1∆ mutant showed attenuated virulence in G. mellonella.19 We found that there was a reduced rate and magnitude of hemolymph melanization following infection with lac1∆ mutant compared to the wild-type parental strain (Fig. 2e). While the overall magnitude of melanization varied between replicates, the ratio of insect melanization that occurred in the lac1∆ versus H99 (WT) was consistently lower (Fig. 2f).
Many fungi, including most human pathogens, produce either DOPA or DHN melanin in their cell walls, which may enable them to persist within mammalian hosts and avoid destruction from oxidative stress and antimicrobial agents26. However, several studies have shown that melanized fungi are less virulent in G. mellonella compared to their non-melanized or albino mutant counterparts27,28,35. We hypothesized that the fungal melanin could act as a pathogen or damage-associated molecular pattern, resulting in enhanced activation of the melanization immune reaction. We found that isolated DOPA melanin produced by C. neoformans (termed melanin ghost, which are isolated following boiling 6 N hydrochloric acid36) activated immune melanization, both with and without hemocytes present compared to heat-killed C. neoformans (Fig. 2g, Supplementary Movie 3–5). Melanin ghosts contain trace amounts of fungal cell wall components that may activate the melanization immune response37,38, therefore non-melanized heat-killed cells were used as a control for the cell wall components that would be present in small amounts. The heat-killed cells did not activate a melanization response like the fungal melanins. Hence, it appears that isolated fungal melanin is specifically recognized and activates the phenoloxidase cascade. Further, the isolated melanin ghosts are aggregated by the hemocytes throughout the course of the time-lapse microscopy, even when immune melanization is not activated (Supplementary Movie 6).
We used these techniques to compare the immune melanization between C. neoformans and C. albicans, the latter of which is known to trigger robust melanization of the hemolymph39. Following infection, C. albicans activated the melanization response faster (beginning as early as 15 min) and to a significantly greater extent than did C. neoformans (Fig. 2h). These data showing large amounts of melanization induced by C. albicans corresponds to the levels of melanization previously reported that occurs during G. mellonella infection with C. albicans versus C. neoformans and further validates the developed methodology39.
Evaluating the melanin-based immune response of G. mellonella using tissue clearing
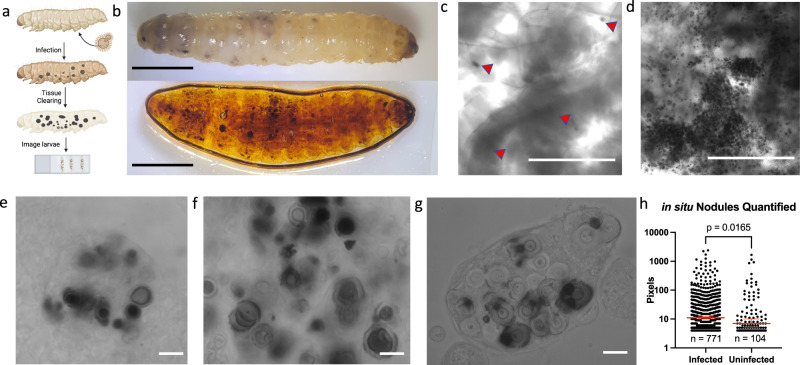
Tissue clearing is a technique that allows for visualization of structures deep within an organism or tissue sample, without significant disruption of the native tissue anatomy. A previously reported protocol40 was adapted to visualize the anatomical localization of the anti-cryptococcal melanization response in G. mellonella (Fig. 3a, b). Using this technique, we visualized melanized nodules in situ that formed only during infection with C. neoformans and not in uninfected controls (Fig. 3c, d). These in situ melanized nodules (Fig. 3d–f) appeared very similar in size and shape to those that are collected from extracted hemolymph, which represented an ex vivo method of visualizing the nodules unique to fungal infection (Figs. 1a, 3g). The visual similarities between Fig. 3e, f and Fig. 3g clarified that the nodules observed in extracted hemolymph are generally representative of the entirety of nodules in the organism. Both the ex vivo and in situ techniques could be quantified to determine the average melanized nodule area and degree of melanization (Fig. 3h). However, the cleared tissue had some opacities or normally darker tissues (i.e. digestive tract contents, legs, prolegs, spiracles, cuticle pigmentation, etc.), which can result in the detection of dark particles even in the uninfected controls, albeit at a much lower frequency (Fig. 3h). Further, while there are some large C. neoformans nodules in situ that appeared aggregated (Fig. 3i, arrows), there was no clear anatomical tropism for nodule formation and the nodules are found throughout the larvae, implying that the infection was disseminated throughout the body of larvae, possibly through the insect’s open circulatory system. The large, aggregated nodules were imaged along the Z-axis, which allowed for 3D reconstruction of the nodule for a better understanding of the native nodule structure compared to the ex vivo preparations compressed under a slide (Supplementary Movie 7). However, compared to the ex vivo experiments, the resolution of the melanin-encapsulated C. neoformans in situ is limited, and variations in opacity and tissue thickness could interfere with measurements.
Fig. 3. Using tissue clearing to visualize the melanin-based immune response against C. neoformans in situ.
Using tissue-clearing techniques (a), we allowed for better visualization of melanized nodules within the intact G. mellonella, as seen in a before (top) and after (bottom) view of the same infected larvae (b). Under microscopy, the uninfected larvae had little to no melanized nodules (red arrows indicate dark, irregular, or opaque areas in the tissue) (c), whereas the C. neoformans larvae had very clear and distinct nodules (d). These nodules were viewed under high magnification, where the nodule structure and encapsulated fungus were apparent (e, f). These structures were very similar in appearance to the nodules extracted from hemolymph (g). h The size of the melanized nodules were quantified using particle measuring software. Infected particles represent n = 771 independent particles, uninfected particles represent n = 104 independent particles, (Mann–Whitney test, p = 0.016). Error bars signify median with 95% Confidence Interval. i The melanized nodules of C. neoformans appeared to have no tissue tropism, but were found in distinct clumps or areas throughout the larvae (red arrows). b–g, i are representative images from three biological replicates. Scale bars in e–g, i represent 10 µm, c, d represent 500 µm, and b represent 50 mm.
Candida spp. interactions with the melanin-based immune response
We employed the in vitro, ex vivo, and in situ techniques described above to gain insight into the host-microbe interactions of C. albicans with the G. mellonella melanin-based immune response. Using the ex vivo technique to analyze melanized nodules in infected hemolymph following extraction, we observed melanin-encapsulated C. albicans cells within nodule structures (Fig. 4a). These melanized nodules are like those that were observed during C. neoformans infection; however, the borders of the melanin itself appeared less distinct, blurry, and smudged. An additional difference from cryptococcal infection was the presence of filamentous C. albicans structures within the nodules. These hyphae or pseudohyphae were melanin-encapsulated, but seemingly to a lesser extent than the yeast morphology.
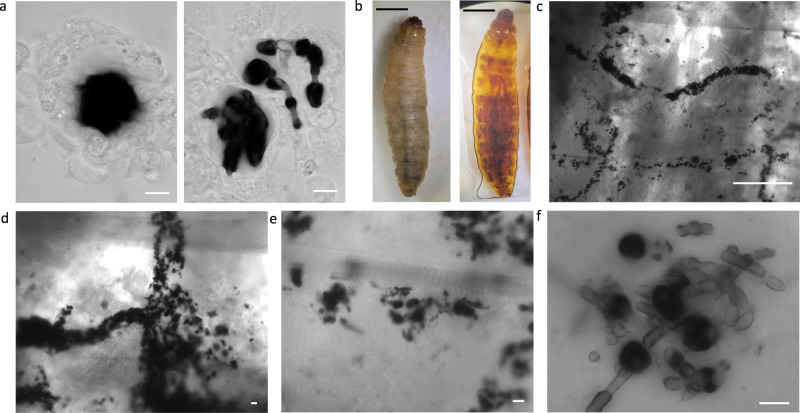
Fig. 4. Visualizing the melanization response to infection with Candida albicans.
a Similar to the hemolymph from larvae infected with C. neoformans, we are able to see melanized nodules within the hemolymph. The melanized spot within these nodules appears more diffuse/less defined, and in some, the presence of less-melanized hyphae is distinct (red arrows). b We can also use tissue clearing to visualize the melanized nodules during C. albicans infection. c, d The melanized C. albicans seem to cluster in specific areas, in long strips within the larvae (yellow arrows). e, f Under higher magnification, we can see hyphal structures of C. albicans, which corresponds to what is previously known about C. albicans morphology in G. mellonella. Interestingly, the hyphae appear less melanized (red arrows) compared to the spherical yeast (white arrows) (f). All panels show representative images from 3 biological replicates. Scale bars in a, d–f represent 10 µm, in c represent 500 µm, and in b represent 50 mm.
Analysis of the tissue of larvae infected with C. albicans for 24 h using the in situ tissue clarification technique (Fig. 4b) revealed groupings of the melanized nodules, often in long string-like patterns (Fig. 4b), that did not appear particularly associated with any organs or tissues (Fig. 4c, d, Supplementary Fig. 2a–f). Under higher magnification, we observed lightly pigmented hyphae and dark spherical melanized particles within the cleared larvae after 24 h of infection (Fig. 4c–f). These images validate that filamentation occurred within G. mellonella, which was previously observed with histology. The melanin-encapsulated fungi form large aggregates (Fig. 4c, d), which we initially thought could be indicative of a tropism for a specific tissue such as the chitinous trachea. However, upon dissection of uncleared infected larvae, there did not appear to be an association of these clusters with any specific tissues (Supplementary Fig. 2a–f). Differences in pigmentation between the two C. albicans morphologies, particularly as seen in the Z-projection in (Fig. 4f), indicated that the hyphae were encapsulated with less melanin during infection compared with the yeast form of the fungus. One potential bias in interpreting this data is that since we are only looking at melanin pigmentation, we are likely missing any non-melanin-encapsulated fungi which would be obscured within the insect tissue.
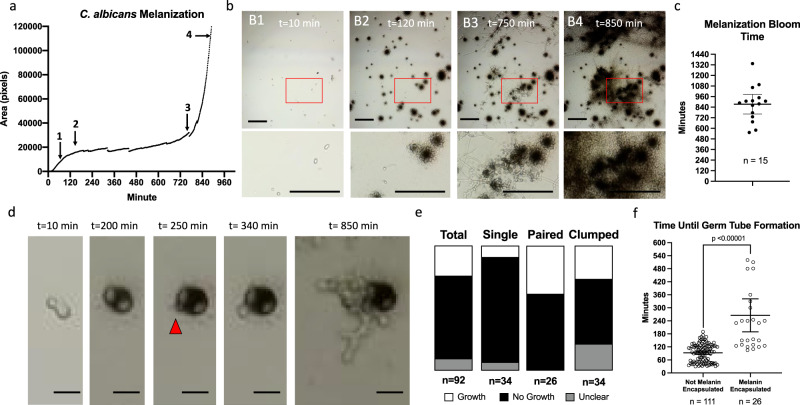
We used the in vitro time-lapse microscopy to observe the melanization dynamics of C. albicans in hemolymph. As seen earlier (Fig. 2h), C. albicans yeast triggered a more robust melanization response than C. neoformans (Fig. 5a, 5b1). In time-lapse microscopy performed without the addition of insect hemocytes, we observed that the C. albicans began to grow in filamentous forms (Fig. 5b2), consistent with the importance of filamentation in the pathogenesis of C. albicans within G. mellonella and the in situ data (Fig. 3)23. In mammalian hosts, C. albicans filamentation is previously shown to be triggered by serum, neutral pH, and temperature41,42. However, in the G. mellonella system, filamentation in vitro does not occur when the C. albicans is only incubated with hemocytes without hemolymph, indicating that a component of the hemolymph is necessary for the morphological switch. Interestingly, as the time-lapse movie progressed, we observed that the hyphae did not get encapsulated by melanin in comparison to the yeast form of C. albicans (Fig. 5b2). After about 12 h of filamentous growth, we observed the formation of blastoconidium (yeast) along the hyphae (Fig. 5b3). The formation of these yeast cells then corresponded with a subsequent bloom of melanization (Fig. 5A, 5B4, Supplementary Movie 8). A similar temporal progression of C. albicans morphology and melanin-encapsulation is seen in C. albicans infected larvae dissected at various timepoints post-infection (Supplementary Fig. 2g). The average time of this melanin bloom was about 840 min, with a 95% confidence interval between ~720 and 960 min (Fig. 5c).
Fig. 5. Using time-lapse microscopy to gain insight into C. albicans yeast and hyphae interactions with the melanization immune response.
a Kinetics of the melanization reaction within hemolymph incubated with C. albicans yeast, with a distinct tri-phastic response in which the yeast starts off non-encapsulated with melanin (1), followed by an initial peak of the melanization reaction (2), an extended plateau (3), and a greater second peak of the melanization reaction (4). b Using microscopy, we saw that the yeast start off non-melanized (B1), and most reached their peak melanin-encapsulation by 120 min (B2). After which, the yeast began to filament, and hyphae grew between 120 and 750 min with minimal melanin encapsulation occurring. At 750 min (B3), blastoconidium (yeast) began to form on the hyphae, which then corresponded to a rapid increase in melanization (B4). Scale bars in b indicate 100 µm. c The time until this melanin bloom was ~14 h (n = 15 biologically independent movies, mean = 874 min, 95% Confidence Interval 762–985 min). d Through these movies, we also saw that some melanin-encapsulated yeast were able to survive the immune response and grew from underneath the layer of pigment. The melanin-encapsulated C. albicans in panel d began to grow through the melanin by 250 min. Scale bars in d indicate 10 µm. e Overall, ~23% of melanin-encapsulated yeast were able to grow following melanin-encapsulation. Pairs of melanin-encapsulated yeast had the highest percentage of growth following melanin encapsulation, with a 33% frequency. f In the yeast that grew following melanin-encapsulation, the time until germ tube formation was delayed 265 min on average (n = 26 independent melanin-encapsulated fungi), while in non-encapsulated yeast, the average time was 92 min (n = 111 independent non-encapsulated fungi) (Two-tailed unpaired t-test, p < 0.00001, t = 8.878, df = 135) and error bars indicate mean with 95% Confidence Interval. a, b, d show representative data from three biological replicates. Each data point in c represents an independent biological replicate. e, f show data from 4 biological replicates.
We observed that some of the melanin-encapsulated C. albicans yeast survived the immune reaction and then underwent hyphal and/or pseudohyphal growth (Fig. 5d, Supplementary Movie 9). This occurred in about 23% of melanin-encapsulated yeast, with 8% of single yeast and 38% of budded/pairs of yeast being able to escape (Fig. 5e) The time until hyphal or pseudohyphal growth was significantly delayed in the melanin-encapsulated cells; the average time for a non-melanin-encapsulated C. albicans cells to begin filamentation is 92 min, while the melanin-encapsulated counterparts take 265 min, with some taking as long as 520 min (Fig. 5f). This delay could be reflective of physical barriers as the fungus breaks through the melanin layer and/or delays in initiating cellular growth because of cell damage caused by the immune response.
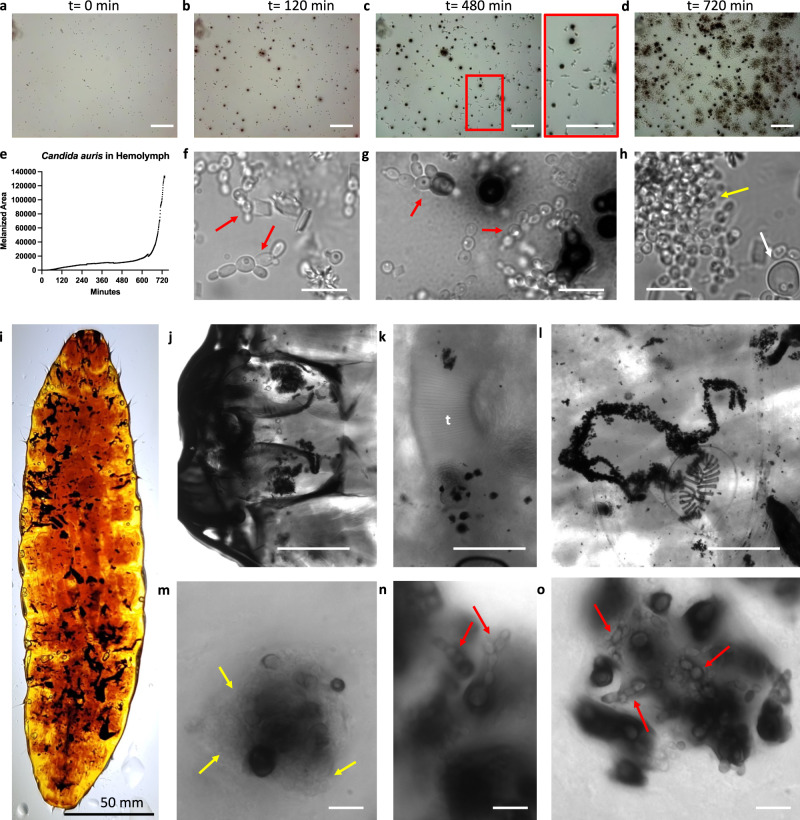
Similar to our findings with C. albicans, we observed that Candida auris yeast activated melanization of hemolymph (Fig. 6a, b). This was followed by formation of melanin-evasive pseudohyphae (Fig. 6c) and a bloom of melanization (Fig. 6d–e, Supplementary Movie 10). Formation of pseudohyphae were confirmed visually by conventional light microscopy (Fig. 6f–g). Enlarged giant cells—cells that are larger than the typical C. auris yeast—measuring roughly 8 µm were observed sporadically (Fig. 6h). Clearing of C. auris infected G. mellonella revealed large aggregates of melanin-encapsulated fungi (Fig. 6i–l), including localization within the head capsule (Fig. 6j), adjacent to trachea (Fig. 6k), and not associated with noticeable tissue structures (Fig. 6l). Similar to what was seen in vitro, in situ we saw large aggregates of non-melanized yeast (Fig. 6m, yellow arrows) and pseudohyphal fungal morphology (Fig. 6n, o).
Fig. 6. Candida auris produces pseudohypae in response to hemolymph.
Following incubation of C. auris with hemolymph, the yeast get encapsulated with melanin by 120 min (a, b). Pseudohyphae form clearly by 480 min, while not eliciting a melanization response (c, inset); however, after ~720 min, there is a sharp increase in melanization within the hemolymph (d). A representative graph of the melanization in response to C. auris over time is shown in e. The cell morphologies were confirmed as pseudohyphae through light microscopy at ×100 magnification (e.g., red arrows), while the presence of enlarged giant cells was occasionally noted (h, white arrows) and dense clusters of yeast (h, yellow arrows). i Melanized aggregates of C. auris can be seen in G. mellonella larvae following tissue clearing. Melanized clusters of fungi can be seen in the head capsule (j), associated with trachea (k, t label) and freely within the tissue (l). In these melanized areas, we can see aggregates of yeast (m, yellow arrows) and pseudohyphae (n, o, red arrows). All panels are representative images and data from three independent replicates. Scale bars in panel i represent 50 mm, j–l represent 500 µm, a–d represent 100 µm, while f–h, m–o represent 10 µm.
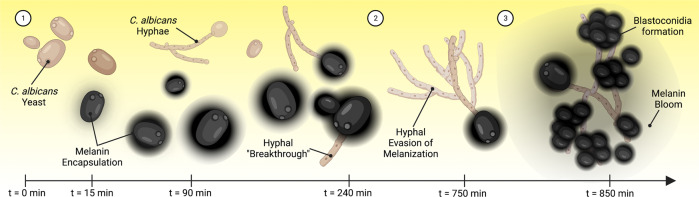
Altogether, these data demonstrate three phases of the C. albicans-immune melanization interactions: (1) yeast become encapsulated with melanin, with nearly 25% surviving and breaking through the pigment, (2) cells undergo a yeast-to-hyphal transition, with the hyphal and pseudohyphal cells evading the melanization immune response; and (3) filamentous C. albicans begins to produce more yeast cells (referred to as blastoconidia or blastospores), which then causes a second bloom of melanization to occur (Summarized in Fig. 7). While host melanization and fungal filamentation have been well-reported during the course of C. albicans infection23,43–46, we report for the first time, to the best of our knowledge, that the hyphae and pseudohyphae are melanin-evasive. Although the presence of blastoconidia has been reported in G. mellonella infected with C. albicans43, these data indicate that lateral blastoconidia growing from hyphae induce a strong second wave melanization response.
Fig. 7. Overview of the growth of C. albicans within G. mellonella and the fungus’ interaction with the melanization immune response.
(1) Within 15 min of exposure to hemolymph, immune melanin forms around the C. albicans yeast. Following melanin encapsulation, ~23% of yeast form a germ tube that grows and escapes the melanin encapsulation. Non-melanin-encapsulated yeast form germ tubes faster and more frequently. (2) Hyphal growth continues with minimal melanin encapsulation occurring on hyphae. (3) Yeast (blastoconidia) begin to form on hyphae after 12–16 h, which triggers a large-scale bloom in the melanization response.
Discussion
Melanin has been appreciated as a key part of the insect immune defense against microbes and parasites for the greater part of the past century13,47. Immune melanization has been implicated as a major process in neutralizing entomopathogenic fungi upon infection48. The insect phenoloxidase and the melanization cascade produce toxic intermediates such as dihydroxyindole and high levels of oxidative stress that can overwhelm and kill the fungus or microbe in vitro13,17. However, there have been no in vivo studies showing that melanin encapsulation within nodules directly kills fungi during these immune reactions within the insect. Here, we fill that gap by showing that melanization within nodules is associated with the death of C. neoformans using a GFP viability reporter assay and provide additional in vitro data for a fungicidal role in immune defense.
During cryptococcal infection of G. mellonella, melanin encapsulation of the fungus within nodules was associated with diminished or lost fluorescence signal in these GFP-expressing C. neoformans strains. Additionally, the melanin-encapsulated fungi that remained GFP-positive had weaker signals and the intensity of the GFP signal was more intense for the non-melanin-encapsulated fungi within the nodules. The expression of GFP in these cells is under the control of an actin promotor, and while actin is generally presumed to be constitutively expressed in cells, growth conditions have been shown to lead to some alterations in cryptococcal actin expression49,50. If the environmental conditions within the nodule abolished actin expression in some cells without killing the fungus, we would expect that condition to equally affect the melanin and non-melanin-encapsulated fungi, and as a result, see similar GFP-negative: GFP-positive ratios between the melanin-encapsulated and not melanin-encapsulated cells. The association between melanin encapsulation and disappearance in GFP fluorescence provides strong evidence for the notion that the melanization reaction kills fungal cells during infection. This is the first evidence, to the best of our understanding, that G. mellonella immune melanization directly and effectively neutralizes C. neoformans during infection and demonstrated that melanin encapsulation results in fungal death within the insect. Previously, the death of microbes, specifically bacteria, was attributed to the enzymatic activity of the melanin-producing phenoloxidase (PO) in an in vitro reaction16. In addition to our association of melanin encapsulation and fungal death in vivo, we sought to reproduce these results in vitro using extracted hemolymph in buffer. We used the PO-specific inhibitor, phenylthiourea (PTU), we found that PO-inhibited wells of hemolymph had higher recoverable CFUs of C. neoformans compared to the uninhibited wells. The inverse correlation of melanization with CFUs further supports the claim that melanin plays a role in neutralizing C. neoformans. Since we only assayed CFUs from these in vitro experiments, we cannot determine whether the melanization in the in vitro experiments directly killed the fungus or just inhibited fungal growth.
When attempting to replicate our results with a known cell viability dye, PI, we were unable to obtain staining of fungal cells within nodules. The absence of PI staining for fungal cells in nodules suggests that PI was unable to reach the center of the nodules where the fungi are found and shows that some of the hemocytes involved in surrounding the fungus may undergo cell death in the process. The permeability or access issues that may arise when using added dyes to measure microbial viability within the nodule show the usefulness of using a live-dead indicator that is endogenously present within the fungus, such as the constitutively expressed GFP.
In addition to studying the extracted hemolymph, we used a developed in vitro time-lapse microscopy assay, which allows observation of the melanization response in real time, while also using the same population of hemocytes and stock of hemolymph to reduce inter-larval variation in the melanization response. Since we only used hemocytes that adhere to the coverslip, we are making an assumption that the hemocyte population is representative, while there might be a predilection for the adhesion of some hemocyte subtypes compared to others. We investigated the impact of fungal melanins on the insect immune response. We found that isolated fungal melanins, termed melanin ghosts, activated the melanin-based immune response whereas the heat-killed C. neoformans did not. This suggests that fungal melanins can activate the immune system which could help promote fungal clearance. This is interesting in the context of naturally occurring fungal pathogens of insects, which tend to have a white color and/or do not naturally produce melanin pigment, such as Beauveria bassiana and Metarhizium anisophilae24,25. While these fungi have undergone many adaptations to effectively infect insect hosts51, their lack of melanin may be due to an additional evolutionary pressure that selects for fungi that produce less fungal melanin, and thus elicit a less robust melanin-based immune response. Since melanin is a component of the insect wound response, it is possible that these exogenous melanins are recognized by the insect as a damage-associated molecular pattern and launch an inappropriate wound repair response.
We found that the lac1∆ mutant, which is unable to produce the melanin-producing enzyme laccase, causes less melanization in the hemolymph. This implies that some of the melanization that occurred was due to fungal laccase-catalyzed melanization using host-derived catecholamines in the hemolymph. Another possible mechanism is that laccase produces fungal melanins, which in turn activates the melanization response of insects as we had seen with the isolated fungal melanins. In the lac1∆, this fungal melanin trigger would be absent and thus induce a less robust melanization response. These findings are also consistent with the observation that during B. bassiana infection of G. mellonella, laccases play a role in virulence by oxidizing the hemolymph catecholamines and preventing them from producing antifungal melanization and reducing the oxidative burden on the fungus24. It is also worth noting that the lac1∆ mutant is less virulent in G. mellonella infections compared to the parental strain19. Together, these observations paint a nuanced picture of the role that laccase and fungal melanin play during fungal pathogenesis in G. mellonella—both fungal melanin and fungal laccases activate the melanin-based immune response, while fungal melanins are associated with decreased virulence, fungal laccases enhance virulence. We note that laccase is secreted by C. neoformans and is found in extracellular vesicles, which could transport laccase away from the fungal cell and reduce the antifungal damage from its effects on triggering insect immune melanization. We compared the amount of melanization that C. neoformans triggers with the amount triggered by other fungal species such as C. albicans. The differences in hemolymph-induced melanization during exposure to C. albicans and C. neoformans were previously described39, and our results confirm those findings. The smaller magnitude of the melanization reaction in response to C. neoformans may be due to cell wall pathogen-associated molecular patterns being hidden by the fungus’ polysaccharide capsule, which is known to help the fungus evade host immunity in insects and mammals3,52.
The second method used to evaluate the melanization response to C. neoformans was tissue clearing, which enabled us to visualize melanized nodules in situ deep within the larvae. We modified a recently developed protocol40 to view the nodules that formed during infections, and saw the native structures of the nodules and their anatomical location in the larvae. This offers an advantage over dissection of uncleared larvae, because during the dissection process: (1) the tissue organization is disrupted, (2) some organs such as the nerve cord and cardiac system might be disrupted, and (3) the geography of infection patterns may not be apparent. Additionally, melanized nodules may not be visible within or behind opaque tissues and organs. Tissue dissection of opaque larvae was helpful when evaluating tissue tropism since tissue boundaries may not be fully visible in clarified larvae. A bias involved in studying fungal infections using both tissue clearing and dissection is that the non-melanized nodules or fungi may be missed, as unpigmented fungi will likely blend in with surrounding tissue. However, in the clearing method, we viewed the nodules throughout the entire depth of the larvae at a low to moderate (×4–40) magnification using light microscopy. However, the objective and microscope limitations only permitted imaging the superficial melanized nodules at ×100 magnification, which provided a lower resolution of the nodules compared to the imaging of the extracted hemolymph. While in the case of C. neoformans, the nodules within the hemolymph appeared congruent to those viewed in situ, that might not always be the case. Nodules in extracted hemolymph during other fungal infection may not be entirely representative of those found throughout the entire larvae, so only viewing the hemolymph nodules may give a biased understanding of the fungal infection.
We also examined the melanization response to Candida albicans infection. C. albicans is known to trigger large-scale systemic melanization in G. mellonella larvae39,53. Like C. neoformans, we found melanized nodules in the hemolymph from larvae infected with C. albicans. Interestingly, the center of these nodules had melanized and smoothened areas that seemed more amorphous than those seen with C. neoformans, and additionally, we saw hyphal structures appeared less melanized than the spherical yeast-like structures. Using the tissue clarification method, we noted that the melanin-encapsulated C. albicans formed large rope-like aggregates without tissue tropism, with yeast being preferentially melanized over hyphal cells. Using in vitro time-lapse microscopy, we found that rapid melanization occurred, even in the absence of hemocytes. Additionally, after the melanization plateaus, the surviving fungus can break free from the melanin encapsulation and undergo melanin-evasive filamentation. This is followed by production of laterally budding blastoconidium and a bloom in melanization around these newly formed yeast cells. Similar fungal morphologies and timelines were observed in dissected infected larvae, although the temporal kinetics were less resolved and identification of blastoconidium was less clear. Together, these data paint an interesting picture and allow insight into the pathogenesis of C. albicans within G. mellonella host. Hence, it appears that the melanin encapsulation can clear most of the yeast upon infection; however, cells that survive can then filament and evade subsequent melanin-mediated killing. The hyphae are known to penetrate and infect organs within the insect23. The hyphae then produce yeast, which again triggers a burst of melanization that would likely cause damage to the surrounding tissue and eventually death of the organism.
Notably, we observed the formation of melanin-evasive pseudohyphae in C. auris when exposed to the G. mellonella hemolymph. These findings corroborate previous studies showing that C. auris forms pseudohyphae during G. mellonella infection54, and indicate that the stress and cues from the hemolymph can induce morphological changes in multiple fungal species. Pseudohyphae and enlarged giant cells in C. auris have been previously linked to exposure to genotoxic stress, indicating that the melanization reaction can induce DNA damage55. Following tissue clearing of C. auris infected larvae, we observed pseudohyphal structures in situ. We also observed melanin-encapsulated fungi within aggregates surrounding trachea, as previously reported54,56, although the frequency of this association was difficult to ascertain, with aggregates of melanin-encapsulated C. auris was also found unassociated with trachea, including in the insect’s head capsule.
In summary, we found evidence that the G. mellonella wax moth directly kills C. neoformans by encapsulating it with melanin in vivo using a GFP-expressing strains where fluorescence indicates viability. This association between melanin encapsulation and reduced viability provides direct evidence for fungal killing via melanin encapsulation in vivo. We also describe three different methodological approaches for studying the melanization response to fungi in G. mellonella and employ these techniques to study C. neoformans, C. albicans, and C. auris’ interactions with the melanin-based immune response. With C. neoformans, we show that both fungal melanins and fungal laccases can activate the insect’s melanization immune response, furthering our understanding of how these fungal components interact with insect immunity and alter the fungus’ pathogenesis. In C. albicans, we were able to observe how some melanin-encapsulated yeasts were able to breakthrough the melanization, and form melanin-evasive hyphae and pseudohyphae during infection. The direct association of insect melanization with antifungal defense further heighten concern that pesticides that inhibit the melanin reaction30 could have untoward and unpredictable effects on insect populations.
Methods
Biological materials
G. mellonella last-instar larvae were obtained through Vanderhorst Wholesale, St. Marys, Ohio, USA. For the tissue-clearing experiments with Candida auris, G. mellonella larvae were obtained from BestBait.com, Marblehead, OH, USA. C. neoformans strain H99 (serotype A), C. neoformans acapsular mutant cap67∆, C. neoformans strain H99-GFP57, C. neoformans lac1∆ mutant (from the 2007 mutant library created by Dr. Jennifer K Lodge, and was obtained from the Fungal Genomics Stock Center), Candida albicans strain 90028, and Candida auris CDC 388 (B11098) were kept frozen in 20% glycerol stocks and subcultured into yeast peptone dextrose (YPD) broth for 48 h at 30 °C prior to each experiment. For H99-GFP infections, frozen stock was streaked out first onto YPD agar, and green colonies were inoculated into YPD broth for 48 h at 30 °C prior to each experiment. The yeast cells were washed twice with PBS, counted using a hemocytometer (Corning, New York, USA), and adjusted to 107 cells/ml for an injection inoculum of 1 × 105 cells/larva. C. albicans infections were performed at 5 × 105 cells/larva with the inoculum suspension being diluted to 5 × 107/ml.
Extraction of hemolymph from fungal-infected Galleria mellonella larvae
Infection of G. mellonella larvae was performed as previously described30. In summary, washed C. neoformans were resuspended to 107 cells/ml, and 10 µl were injected in the right rear proleg of larvae ranging from 175 to 225 mg, for an infectious inoculum of ~105 cells/larva. C. albicans was washed and resuspended in 5 × 107 cells/ml, and larvae were injected with 10 µl of culture for an infectious inoculum of ~5 × 105 cells/larva. Infected larvae were then incubated at 30 °C. 24 h following infection, or 72 h in the case of noted time course experiments with C. neoformans, larvae were removed from incubator, and hemolymph was extracted by puncturing the right rear proleg and/or side of the larvae with an 18 G needle. Removed hemolymph from three larvae was collected directly into 1 ml anticoagulation buffer at room temperature (98 mM NaOH, 186 mM NaCl, 1.7 mM EDTA, and 41 mM citric acid, pH 4.5)58. Hemolymph was centrifuged for 5 min at 4000 × g and resuspended in 200 µl insect physiological saline (IPS) (150 mM sodium chloride, 5 mM potassium chloride, 7.21 mM calcium chloride, 1 mM sodium bicarbonate, pH 6.90—adapted from refs. 59–61). Samples were placed on slides and nodules were imaged using Olympus AX70 microscope with a ×100 oil immersion objective.
C. neoformans GFP viability assay
H99-GFP strain was streaked from frozen stock on YPD agar and incubated at 30 °C. 2 ml YPD was inoculated with H99-GFP and incubated for ~18 h at 30˚C with rotation. Culture was diluted to OD 0.5 and 100 µL was incubated at 70 °C for 1 h using a thermocycler. One hundred microliters of untreated and heated samples were stained with 10 µg/ml propidium iodide (Invitrogen). Ten microliters of stained samples were loaded onto a hemocytometer and imaged using a ×10 objective and Zeiss AxioImager M2 (×60 Olympus objective) equipped with a Hamamatsu Orca R2 camera and Volocity Software (Perkin Elmer). Images were analyzed using ImageJ/FIJI software. Fluorescence channel images were processed by adjusting the minimum pixel value to 10 and maximum to 90. Number of fluorescent cells for each channel were counted using Measure Particles. Number of double fluorescence positive and double fluorescence negative cells were enumerated manually.
C. neoformans GFP fungal survival assay in vivo
Washed H99-GFP strains of C. neoformans were resuspended to 107 cells/ml, and 10 µl was injected in the right rear proleg of larvae ranging from 175 to 225 mg, for an infectious inoculum of ~105 cells/larva. Infected larvae were then incubated at room temperature 30 °C for 24–72 h as noted. Hemolymph was extracted by puncturing the larvae with an 18 G needle in their right rear proleg, and hemolymph was collected into anticoagulation buffer, centrifuged for 5 min at 4000 × g, and resuspended in 200 µl insect physiological saline. Melanized nodules were visualized using an Olympus AX70 microscope with 488 excitation/520 nm emission fluorescence microscopy to visualize the GFP signal. Images were taken at ×100 magnification at the same exposure, and manually marked as positive or negative for GFP fluorescence, and melanin-encapsulated or unencapsulated. For fluorescence and melanin intensity measurements, images were analyzed using the Measure tool in FIJI62, and the 8-bit mean gray value of each cell was measured in both channels. The region selected for the melanin measurements extended the edge of the fungal capsule, and the GFP intensity measurements were from selections limited to the fluorescent cell’s body. This protocol is summarized in Supplementary Fig. 3a.
Phenoloxidase inhibition and fungal survival assay in vitro
Serial dilutions of phenylthiourea (PTU) were performed in 100 µl IPS buffer, to which 5 µl of 106 cells of C. neoformans was added. G. mellonella hemolymph was extracted by puncturing the larvae with an 18 G needle in their right rear proleg, and letting the hemolymph drip into insect physiological saline. The mixture was mixed by pipetting up and down and 100 µl was added to each well. The mixture was incubated at room temperature for 24 h protected from light. Following the incubation, the contents of each well were resuspended and diluted 1:16 in PBS. From the dilution, 5 µl was spotted on a Sabouroud agar plate. The plate was incubated at 30 °C for 24 h and colonies were enumerated under a dissection microscope.
Tissue clearing of Galleria mellonella following fungal infection
C. neoformans was washed in PBS, counted, and resuspended to 107 cells/ml, and 10 µl was injected in the right rear proleg of G. mellonella larvae ranging from 175 to 225 mg, for an infectious inoculum of ~105 cells/larva. C. albicans was washed and resuspended in 5 × 107 cells/ml and larvae were injected with 10 µl of culture for an infectious inoculum of 5 × 105 cells/larva, while C. auris was resuspended in 108 cells/ml and larvae were injected with 10 µl of culture for an infectious inoculum of 106 cells/larva. Following infection (5 days for C. neoformans, 24 h for C. albicans, and 48 h for C. auris), groups of three larvae were removed from incubator and injected with 10 µl of 1 M ascorbic acid to inhibit new melanization and oxidation of endogenous catecholamines during the tissue-clearing process. Ten minutes following the ascorbic acid injection, larvae were placed at −20 °C for 15 min to euthanize them, then injected with an additional 10 µl of 1 M ascorbic acid. Larvae were immediately placed in 40 mL of 4% paraformaldehyde to fix overnight at 4 C. Fixed larvae were washed in PBS, and serially dehydrated in 50, 75, 100, and 100% methanol for 1 h each at 4 °C. Larvae were then incubated for 1 h in 50% methanol and 50% BABB solution (2:1 Benzyl benzoate: Benzyl alcohol), and cleared in 100% BABB solution as previously described40. Following 5–7 days of tissue clearing, larvae were removed from the BABB solution and pressed between two glass microscope slides. Once flattened, a coverslip was placed on top of the larvae and parafilmed into place. Larvae were imaged using Olympus AX70 microscope with ×4, ×20, and ×100 objectives.
Imaging Galleria mellonella hemocytes in vitro
To collect and isolate hemocytes G. mellonella larvae were surface sterilized in two sequential baths of 70% ethanol, followed by 10% bleach, then dried on sterile paper towels. Five to ten drops of hemolymph were extracted as described above into room temperature anticoagulation buffer and inverted three times. Hemolymph was centrifuged at 400 × g for 4 min, the supernatant was removed, and the hemocytes were resuspended in 1 ml anticoagulation buffer and centrifuged again at 400 × g for 4 min. The supernatant was completely removed and hemocytes were resuspended in 200 µl of insect physiological saline (IPS). The 200 µl suspension of hemocytes were added to the coverslip of a 14 mm diameter microwell MatTek dish (Cataloge #P35G-1.5-14-C, MatTek) and allowed to settle for 10 min Following the 10 min, the buffer and unsettled hemocytes were removed, and the coverslip was washed four times with 1 ml of IPS. The hemocytes were seeded into the coverslip at a cell density of 1.5 × 106 cells/ml and the resulting hemocyte density after washing was ~2–3 × 103 cells/mm2.
While the hemocytes were being isolated, cell-free hemolymph was being prepared. Approximately 10 drops of hemolymph were removed from G. mellonella larvae and collected directly into 1 ml IPS. To remove hemocytes, the mixture was filtered using a 0.22 µm syringe-driven PVDF filter. Cell-free hemolymph was stored up to a week at -80C. Penicillin-Streptomycin (Gibco, Thermo Fisher) antibiotic was added at 1× concentration to the cell-free hemolymph. For experiments looking at the interaction of hemocytes with fungi or a virulence factor, the cells or component were added at this stage.
Following the hemocyte washes, 1 ml of cell-free hemolymph was added to the entirety of the MatTek dish, followed by an addition 1 ml of IPS. The MatTek dish was covered and imaged using the OpenFlexure microscope and software and time-lapse microscopy was performed every minute for 16–24 h63. Control experiments without added fungus or fungal components, or with anticoagulation buffer to inhibit melanization and coagulation, were performed (Supplementary Movies 11 and 12). This protocol is summarized in Supplementary Fig. 3b.
All time-lapse data were analyzed using FIJI62 and particle measurements were made by converting the image sequence to 8-bit, setting a threshold of 0–50 gray value, and analyzing any particle over the size of four pixels2. Measuring the time until germ tube formation was done manually by recording the frame in which the first of the germ tube was visible.
Melanin ghost isolation
C. neoformans cultures were grown in minimal media with 1 mM L-DOPA for 7 days at 30 °C. Cells were collected and mixed 1:1 with 12 N hydrochloric acid (HCl), for a final concentration of 6 N HCl. Cells were heated for 1 h at 85 C under constant shaking at 350 RPM. Control cells from the acapsular mutant grown for 5–7 days in minimal media at at 30 °C without L-DOPA were heat-killed for 1 h at 85 °C in PBS. Cells were washed twice in PBS and subsequently used in the time-lapse microscopy.
Statistics and reproducibility
Statistical tests were performed using GraphPad Prism Version 8. All error bars represent the mean with 95% Confidence Interval with the exceptions of the ratio comparison of melanization induced by H99 versus lac1∆ where the error bars represent the ±Standard Deviation from the mean, and the size comparison of in situ nodules where the error bars represent the 95% Confidence Interval of the median. To determine the effect of melanin-encapsulation on fungal viability within nodules, we performed a Chi-Squared analysis for each timepoint and temperature condition tested. The Chi-square table was set up with melanin-encapsulated and non-encapsulated on one side of the contingency table and GFP-positive and GFP-negative on the other. The ratio comparison of melanization induced by H99 versus lac1∆ was analyzed using a one sample t-test with comparisons to the theoretical value of 1, which would indicate no difference between the H99-induced melanization and lac1∆-induced melanization. The size comparison of in situ nodules was performed using a two-tailed non-parametric Mann–Whitney Test comparing the particles from the uninfected versus infected groups. The comparison of the time it takes for the melanin-encapsulated versus non-encapsulated to escape melanization was done using an unpaired two-tailed t-test. The linear regression showing the relationship between CFUs and pigmentation of the hemolymph in vitro was performed using a simple linear regression, with the dotted lines representative of the 95% Confidence Intervals. Non-quantified data, such as microscopy images, were performed in replicates and representative images are shown, as noted in the figure legend.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Supplementary information
Description of Additional Supplementary Files
Acknowledgements
We would like to thank the entire Casadevall Lab for their contributions during lab meetings and other discussions of this project. We would like to thank Maryann Smith, Thomas Hitzelberger, and Kathy Spinnato for placing the years of weekly G. mellonella orders. Illustrations were created using Biorender.com. D.F.Q.S., Q.D., and A.C. are funded by National Institute of Allergy and Infection Disease R01 AI052733, R01 AI152078, and the National Heart, Lung, and Blood Institute R01 HL059842. D.F.Q.S. is funded by National Institutes of Health 5T32GM008752-18 and 1T32AI138953-01A1. J.M.H. and M.K. are funded by National Institutes of Health R56 AI168539 and R21 AI144373. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author contributions
Conceptualization—D.F.Q.S., Q.D., M.K., J.M.H., and A.C.; Methodology—D.F.Q.S., A.C., Q.D., and M.K.; Software—Q.D.; Validation—D.F.Q.S.; Formal analysis—D.F.Q.S. and M.K.; Investigation—D.F.Q.S. and M.K.; Resources—A.C. and J.M.H.; Data curation—D.F.Q.S.; Writing (original draft)—D.F.Q.S. and A.C.; Writing (review and editing)—D.F.Q.S., Q.D., M.K., J.M.H., and A.C.; Visualization—D.F.Q.S. and M.K.; Supervision—D.F.Q.S., J.M.H., and A.C.; Project administration—D.F.Q.S. and A.C.; Funding acquisition—J.M.H. and A.C.
Peer review
Peer review information
Communications Biology thanks Chengshu Wang, Helen Fuchs and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Meritxell Riquelme and George Inglis. Peer reviewer reports are available.
Data availability
The datasets generated during and/or analyzed during the current study are available in the Figshare repository using the following link: https://figshare.com/projects/Galleria_mellonella_immune_melanization_is_fungicidal_during_infection/13812364.
Code availability
The code used in this study is previously published open source code63 and can be accessed using the following link: https://openflexure.org/software/raspbian-openflexure/.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s42003-022-04340-6.
References
- 1.World Health Organization. Vector-borne diseases. https://www.who.int/news-room/fact-sheets/detail/vector-borne-diseases (2020).
- 2.Clark JM. The 3Rs in research: a contemporary approach to replacement, reduction and refinement. Br. J. Nutr. 2018;120:S1–S7. doi: 10.1017/S0007114517002227. [DOI] [PubMed] [Google Scholar]
- 3.Smith DFQ, Casadevall A. Fungal immunity and pathogenesis in mammals versus the invertebrate model organism Galleria mellonella. Pathogens Dis. 2021;79:1–25. doi: 10.1093/femspd/ftab013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Geddes-McAlister J, Shapiro RS. New pathogens, new tricks: emerging, drug-resistant fungal pathogens and future prospects for antifungal therapeutics. Ann. N. Y. Acad. Sci. 2019;1435:57–78. doi: 10.1111/nyas.13739. [DOI] [PubMed] [Google Scholar]
- 5.Casadevall, A., Kontoyiannis, D. P. & Robert, V. On the emergence of Candida auris: climate change, azoles, swamps, and birds. mBio10, 1–7 (2019). [DOI] [PMC free article] [PubMed]
- 6.Dubovskiy IM, Kryukova NA, Glupov VV, Ratcliffe NA. Encapsulation and nodulation in insects. Invertebr. Survival J. 2016;13:229–246. [Google Scholar]
- 7.Clark, K. D. in Vertebrate and Invertebrate Respiratory Proteins, Lipoproteins and other Body Fluid Proteins (eds. Hoeger, U. & Harris, J. R.) 123–161 (Springer International Publishing, 2020).
- 8.Phillips DR, Clark KD. Bombyx mori and Aedes aegypti form multi-functional immune complexes that integrate pattern recognition, melanization, coagulants, and hemocyte recruitment. PLoS ONE. 2017;12:e0171447. doi: 10.1371/journal.pone.0171447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim Y, Ahmed S, Stanley D, An C. Eicosanoid-mediated immunity in insects. Dev. Comp. Immunol. 2018;83:130–143. doi: 10.1016/j.dci.2017.12.005. [DOI] [PubMed] [Google Scholar]
- 10.Mandato CA, Diehl-Jones WL, Moore SJ, Downer RGH. The effects of eicosanoid biosynthesis inhibitors on prophenoloxidase activation, phagocytosis and cell spreading in Galleria mellonella. J. Insect Physiol. 1997;43:1–8. doi: 10.1016/S0022-1910(96)00100-X. [DOI] [PubMed] [Google Scholar]
- 11.Shrestha S, Kim Y. Oenocytoid cell lysis to release prophenoloxidase is induced by eicosanoid via protein kinase C. J. Asia Pac. Entomol. 2009;12:301–305. doi: 10.1016/j.aspen.2009.08.001. [DOI] [Google Scholar]
- 12.González‐Santoyo I, Córdoba‐Aguilar A. Phenoloxidase: a key component of the insect immune system. Entomol. Exp. Appl. 2012;142:1–16. doi: 10.1111/j.1570-7458.2011.01187.x. [DOI] [Google Scholar]
- 13.Nappi AJ, Christensen BM. Melanogenesis and associated cytotoxic reactions: applications to insect innate immunity. Insect Biochem. Mol. Biol. 2005;35:443–459. doi: 10.1016/j.ibmb.2005.01.014. [DOI] [PubMed] [Google Scholar]
- 14.Barillas-Mury C. CLIP proteases and Plasmodium melanization in Anopheles gambiae. Trends Parasitol. 2007;23:297–299. doi: 10.1016/j.pt.2007.05.001. [DOI] [PubMed] [Google Scholar]
- 15.Chen CC, Chen CS. Brugia bahangi: effects of melanization on the uptake of nutrients by microfilariae in vitro. Exp. Parasitol. 1995;81:72–78. doi: 10.1006/expr.1995.1094. [DOI] [PubMed] [Google Scholar]
- 16.Zhao P, Li J, Wang Y, Jiang H. Broad-spectrum antimicrobial activity of the reactive compounds generated in vitro by Manduca sexta phenoloxidase. Insect Biochem. Mol. Biol. 2007;37:952–959. doi: 10.1016/j.ibmb.2007.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhao P, Lu Z, Strand MR, Jiang H. Antiviral, anti-parasitic, and cytotoxic effects of 5,6-dihydroxyindole (DHI), a reactive compound generated by phenoloxidase during insect immune response. Insect Biochem. Mol. Biol. 2011;41:645–652. doi: 10.1016/j.ibmb.2011.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang J, et al. Prophenoloxidase-mediated ex vivo immunity to delay fungal infection after insect ecdysis. Front. Immunol. 2017;8:1445. doi: 10.3389/fimmu.2017.01445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mylonakis E, et al. Galleria mellonella as a model system to study Cryptococcus neoformans pathogenesis. Infect. Immun. 2005;73:3842–3850. doi: 10.1128/IAI.73.7.3842-3850.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kavanagh K, Fallon JP. Galleria mellonella larvae as models for studying fungal virulence. Fungal Biol. Rev. 2010;24:79–83. doi: 10.1016/j.fbr.2010.04.001. [DOI] [Google Scholar]
- 21.Ignasiak K, Maxwell A. Galleria mellonella (greater wax moth) larvae as a model for antibiotic susceptibility testing and acute toxicity trials. BMC Res. Notes. 2017;10:428. doi: 10.1186/s13104-017-2757-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Firacative C, Duan S, Meyer W. Galleria mellonella model identifies highly virulent strains among all major molecular types of Cryptococcus gattii. PLoS ONE. 2014;9:e105076. doi: 10.1371/journal.pone.0105076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fuchs BB, et al. Role of filamentation in Galleria mellonella killing by Candida albicans. Microbes Infect. 2010;12:488–496. doi: 10.1016/j.micinf.2010.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lu Z, et al. Multifunctional role of a fungal pathogen-secreted laccase 2 in evasion of insect immune defense. Environ. Microbiol. 2021;23:1256–1274. doi: 10.1111/1462-2920.15378. [DOI] [PubMed] [Google Scholar]
- 25.Fang W, Fernandes ÉKK, Roberts DW, Bidochka MJ, St. Leger RJ. A laccase exclusively expressed by Metarhizium anisopliae during isotropic growth is involved in pigmentation, tolerance to abiotic stresses and virulence. Fungal Genet. Biol. 2010;47:602–607. doi: 10.1016/j.fgb.2010.03.011. [DOI] [PubMed] [Google Scholar]
- 26.Smith, D. F. Q. & Casadevall, A. in Fungal Physiology and Immunopathogenesis (ed. Rodrigues, M. L.) 1–30 (Springer International Publishing, 2019).
- 27.Eisenman HC, Duong R, Chan H, Tsue R, McClelland EE. Reduced virulence of melanized Cryptococcus neoformans in Galleria mellonella. Virulence. 2014;5:611–618. doi: 10.4161/viru.29234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jackson JC, Higgins LA, Lin X. Conidiation color mutants of aspergillus fumigatus are highly pathogenic to the heterologous insect host Galleria mellonella. PLoS ONE. 2009;4:e4224. doi: 10.1371/journal.pone.0004224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rajendran R, et al. Acetylcholine protects against candida albicans infection by inhibiting biofilm formation and promoting hemocyte function in a Galleria mellonella infection model. Eukaryot. Cell. 2015;14:834–844. doi: 10.1128/EC.00067-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Smith DFQ, et al. Glyphosate inhibits melanization and increases susceptibility to infection in insects. PLoS Biol. 2021;19:e3001182. doi: 10.1371/journal.pbio.3001182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Blandin S, et al. Complement-Like protein TEP1 is a determinant of vectorial capacity in the malaria vector Anopheles gambiae. Cell. 2004;116:661–670. doi: 10.1016/S0092-8674(04)00173-4. [DOI] [PubMed] [Google Scholar]
- 32.Ryazanova AD, Alekseev AA, Slepneva IA. The phenylthiourea is a competitive inhibitor of the enzymatic oxidation of DOPA by phenoloxidase. J. Enzym. Inhib. Med. Chem. 2012;27:78–83. doi: 10.3109/14756366.2011.576010. [DOI] [PubMed] [Google Scholar]
- 33.Salas SD, Bennett JE, Kwon-Chung KJ, Perfect JR, Williamson PR. Effect of the laccase gene CNLAC1, on virulence of Cryptococcus neoformans. J. Exp. Med. 1996;184:377–386. doi: 10.1084/jem.184.2.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sabiiti W, et al. Efficient phagocytosis and laccase activity affect the outcome of HIV-associated cryptococcosis. J. Clin. Invest. 2014;124:2000–2008. doi: 10.1172/JCI72950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liu Y, Huang X, Liu H, Xi L, Cooper CR. Increased virulence of albino mutant of Fonsecaea monophora in Galleria mellonella. Med. Mycol. 2019;57:1018–1023. doi: 10.1093/mmy/myz007. [DOI] [PubMed] [Google Scholar]
- 36.Wang Y, Aisen P, Casadevall A. Melanin, melanin ‘ghosts,’ and melanin composition in Cryptococcus neoformans. Infect. Immun. 1996;64:2420–2424. doi: 10.1128/iai.64.7.2420-2424.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Camacho E, et al. The structural unit of melanin in the cell wall of the fungal pathogen Cryptococcus neoformans. J. Biol. Chem. 2019;294:10471–10489. doi: 10.1074/jbc.RA119.008684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chrissian C, et al. Solid-state NMR spectroscopy identifies three classes of lipids in Cryptococcus neoformans melanized cell walls and whole fungal cells. J. Biol. Chem. 2020;295:15083–15096. doi: 10.1074/jbc.RA120.015201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Trevijano-Contador N, et al. Cryptococcus neoformans induces antimicrobial responses and behaves as a facultative intracellular pathogen in the non mammalian model Galleria mellonella. Virulence. 2015;6:66–74. doi: 10.4161/21505594.2014.986412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Moya-Andérico L, Admella J, Torrents E. A clearing protocol for Galleria mellonella larvae: Visualization of internalized fluorescent nanoparticles. N. Biotechnol. 2021;60:20–26. doi: 10.1016/j.nbt.2020.08.002. [DOI] [PubMed] [Google Scholar]
- 41.Mitchell AP. Dimorphism and virulence in Candida albicans. Curr. Opin. Microbiol. 1998;1:687–692. doi: 10.1016/S1369-5274(98)80116-1. [DOI] [PubMed] [Google Scholar]
- 42.Martin R, et al. A core filamentation response network in Candida albicans is restricted to eight genes. PLoS ONE. 2013;8:e58613. doi: 10.1371/journal.pone.0058613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Borghi E, et al. Correlation between Candida albicans biofilm formation and invasion of the invertebrate host Galleria mellonella. Future Microbiol. 2014;9:163–173. doi: 10.2217/fmb.13.159. [DOI] [PubMed] [Google Scholar]
- 44.Marcos-Zambrano LJ, et al. Candida isolates causing candidemia show different degrees of virulence in Galleria mellonella. Med. Mycol. 2020;58:83–92. doi: 10.1093/mmy/myz027. [DOI] [PubMed] [Google Scholar]
- 45.Sheehan G, Kavanagh K. Proteomic analysis of the responses of Candida albicans during infection of Galleria mellonella larvae. J. Fungi. 2019;5:7. doi: 10.3390/jof5010007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Perdoni, F. et al. A histological procedure to study fungal infection in the wax moth Galleria mellonella. Eur. J. Histochem.58, 258–262 (2014). [DOI] [PMC free article] [PubMed]
- 47.Nappi AJ. The role of melanization in the immune reaction of larvae of Drosophila algonquin against Pseudeucoila bochei. Parasitology. 1973;66:23–32. doi: 10.1017/S0031182000044413. [DOI] [Google Scholar]
- 48.Ezzati-Tabizi R, Talaei-Hassanloui R, Farrokhi N, Hossininaveh V, Alavi M. Haemolymph phenoloxidase activity of larval Plodia interpunctella and Galleria mellonella in response to Beauveria bassiana and Pseudomonas fluorescens. Int. J. Agric. Innov. Res. 2013;2:217–220. [Google Scholar]
- 49.Toffaletti DL, Perfect JR. Study of Cryptococcus neoformans actin gene regulation with a beta-galactosidase-actin fusion. J. Med. Vet. Mycol. 1997;35:313–320. doi: 10.1080/02681219780001351. [DOI] [PubMed] [Google Scholar]
- 50.del Poeta, M. et al. Cryptococcus neoformans differential gene expression detected in vitro and in vivo with green fluorescent protein. Infect. Immunity10.1128/IAI.67.4.1812-1820.1999 (1999). [DOI] [PMC free article] [PubMed]
- 51.Wang C, Wang S. Insect pathogenic fungi: genomics, molecular interactions, and genetic improvements. Annu. Rev. Entomol. 2017;62:73–90. doi: 10.1146/annurev-ento-031616-035509. [DOI] [PubMed] [Google Scholar]
- 52.Casadevall A, et al. The capsule of Cryptococcus neoformans. Virulence. 2019;10:822–831. doi: 10.1080/21505594.2018.1431087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Vertyporokh L, Wojda I. Immune response of Galleria mellonella after injection with non-lethal and lethal dosages of Candida albicans. J. Invertebr. Pathol. 2020;170:107327. doi: 10.1016/j.jip.2020.107327. [DOI] [PubMed] [Google Scholar]
- 54.Garcia-Bustos V, et al. Host–pathogen interactions upon Candida auris infection: fungal behaviour and immune response in Galleria mellonella. Emerg. Microbes Infect. 2022;11:136–146. doi: 10.1080/22221751.2021.2017756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bravo Ruiz G, Ross ZK, Gow NAR, Lorenz A. Pseudohyphal growth of the emerging pathogen Candida auris is triggered by genotoxic stress through the S phase checkpoint. mSphere. 2020;5:e00151–20. doi: 10.1128/mSphere.00151-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Garcia-Bustos V, et al. Characterization of the differential pathogenicity of Candida auris in a Galleria mellonella infection model. Microbiol. Spectr. 2021;9:e00013–e00021. doi: 10.1128/Spectrum.00013-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Voelz K, Johnston SA, Rutherford JC, May RC. Automated analysis of cryptococcal macrophage parasitism using GFP-tagged cryptococci. PLoS ONE. 2010;5:e15968. doi: 10.1371/journal.pone.0015968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Stoepler, T. M., Castillo, J. C., Lill, J. T. & Eleftherianos, I. A simple protocol for extracting hemocytes from wild caterpillars. J. Vis. Exp.10.3791/4173 (2012). [DOI] [PMC free article] [PubMed]
- 59.Mandato CA, Diehl-Jones WL, Downer RGH. Insect hemocyte adhesion in vitro: inhibition by apoliphorin I and an artificial substrate. J. Insect Physiol. 1996;42:143–148. doi: 10.1016/0022-1910(95)00091-7. [DOI] [Google Scholar]
- 60.Wago, H. The role of phenoloxidase in the cellular defence reactions to foreign materials by the granular cells of the silkworm, Bombyx mori. Invertebrate Fish Tissue Culture 185–188 (1988).
- 61.Tojo S, Naganuma F, Arakawa K, Yokoo S. Involvement of both granular cells and plasmatocytes in phagocytic reactions in the greater wax moth, Galleria mellonella. J. Insect Physiol. 2000;46:1129–1135. doi: 10.1016/S0022-1910(99)00223-1. [DOI] [PubMed] [Google Scholar]
- 62.Schindelin J, et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods. 2012;9:676–682. doi: 10.1038/nmeth.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Collins, J. T. et al. Robotic microscopy for everyone: the OpenFlexure Microscope. Biomed. Opt. Express11, 2447–2460 (2020). [DOI] [PMC free article] [PubMed]
- 64.Smith, D. Galleria mellonella immune melanization is fungicidal during infection. https://figshare.com/projects/Galleria_mellonella_immune_melanization_is_fungicidal_during_infection/138123. (2022). [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Description of Additional Supplementary Files
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available in the Figshare repository using the following link: https://figshare.com/projects/Galleria_mellonella_immune_melanization_is_fungicidal_during_infection/13812364.
The code used in this study is previously published open source code63 and can be accessed using the following link: https://openflexure.org/software/raspbian-openflexure/.