ABSTRACT
Purpose
We aimed to perform a systematic review to assess perioperative outcomes, complications, and survival in studies comparing ureteral stent and percutaneous nephrostomy in malignant ureteral obstruction.
Materials and Methods
This review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses framework. Meta-analyses were performed on procedural data; outcomes; complications (device-related, accidental dislodgement, febrile episodes, unplanned device replacement), dislodgment, and overall survival. Continuous variables were pooled using the inverse variance of the mean difference (MD) with a fixed effect, and 95% confidence interval (CI). The incidences of complications were pooled using the Cochran-Mantel-Haenszel method with the random effect model and reported as Odds Ratio (OR), and 95% CI. Statistical significance was set two-tail p-value <0.05
Results
Ten studies were included. Procedure time (MD −10.26 minutes 95%CI −12.40-8.02, p<0.00001), hospital stay (MD −1.30 days 95%CI −1.69 − −0.92, p<0.0001), number of accidental tube dislodgments (OR 0.25 95% CI 0.13 – 0.48, p<0.0001) were significantly lower in the stent group. No difference was found in mean fluoroscopy time, decrease in creatinine level post procedure, overall number of complications, interval time between the change of tubes, number of febrile episodes after diversion, unplanned device substitution, and overall survival.
Conclusion
Our meta-analysis favors stents as the preferred choice as these are easier to maintain and ureteral stent placement should be recommended whenever feasible. If the malignant obstruction precludes a stent placement, then PCN is a safe alternative.
Keywords: Ureteral Obstruction; Nephrostomy, Percutaneous; Urinary Diversion
INTRODUCTION
Malignant ureteral obstruction is the consequence of secondary, extrinsic compression/infiltration of the ureter causing obstruction in different cancers. Mechanisms include intraluminal ureteral tumour invasion, ureteral entrapment or compression by retroperitoneal/pelvic lymphadenopathy or metastasis, and as a consequence of retroperitoneal fibrosis induced by surgery, chemotherapy, and radiotherapy (1).
The primary reason for referral to urologists is to evaluate the most appropriate type of urinary diversion and its feasibility accounting for both disease and patient characteristics.
Urologists aim to relieve urinary obstruction, reduce symptoms, and improve renal function while preserving patient quality of life and potentially prolonging overall survival as many of these patients have ongoing treatments (2). Urinary diversion can be achieved externally via a percutaneous nephrostomy tube or internally using a double J ureteral stent (3).
The endoscopic approach may be technically difficult and at times even impossible in advanced pelvic or retroperitoneal disease with high failure rates particularly in pelvic malignancies. Indeed, the success of retrograde ureteral stenting in patients with pelvic malignancy is usually significantly lower in patients with extrinsic ureteral obstruction compared with those with internal ureteral obstruction due to non-progression of the hydrophilic guide and non-identification of the ureteral meatus (4). On the other hand, the percutaneous approach may negatively affect patient quality of life being more invasive and often associated with a greater incidence of infection, bleeding, discomfort, and accidental tube displacement (5). Frail patients may be even more reluctant in accepting long indwelling nephrostomy tubes that need regular change with further worsening of quality of life (2). The choice must be balanced according to operator experience and patient's choice whilst evaluating the patient clinical condition and life expectancy. Unlike temporary urinary diversion in acute urinary obstruction, there are currently no guidelines or consensus for the optimal approach in malignant ureteral obstruction.
The present study aimed to systematically review the literature to assess perioperative outcomes, complications, and survival in studies comparing double J ureteral stent and percutaneous nephrostomy in malignant ureteral obstruction to help clinicians in taking an informed decision on urinary diversion choices by understanding the nuances of both interventions.
EVIDENCE ACQUISITION
Literature search
We aimed to perform a meta-analysis comparing outcomes in patients with ureteral obstruction secondary to malignancies. This systematic review was performed according to the 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) framework. A comprehensive literature search was performed on 6th December 2021, using MEDLINE, EMBASE, and Cochrane Central Controlled Register of Trials (CENTRAL). The following term and Boolean operators were used: (ureteral stent OR urinary diversion OR double-J) AND (nephrostomy tube OR external urinary drainage) AND (extrinsic ureteral obstruction OR ureteral obstruction OR cancer ureteral obstruction). No date limits were imposed. The search was restricted to English papers, searching comparative studies between the two urinary diversions. Animal and paediatric studies were excluded. Additional articles were sought from the reference lists of the included articles. The review protocol was registered in PROSPERO (CRD42022297668).
Selection criteria
The PICOS (Patient Intervention Comparison Outcome Study type) model was used to frame and answer the clinical question. P: patients with external ureteral obstruction due to malignancy; Intervention: ureteral stent; Comparison: nephrostomy tube; Outcome: procedural time, fluoroscopy time, post-procedural complications, hospital stay, decrease in creatinine, episodes of accidental dislodgment, and overall survival; Study type: prospective randomized studies, retrospective, or prospective non-randomized studies.
Study Screening and Selection
Two independent authors screened all retrieved records through Rayyan Intelligent Systematic Review (https://www.rayyan.ai/). Discrepancies were solved by a third author. Studies were included based on PICOS eligibility criteria. Meeting abstracts, case reports, reviews, letters to editor, and editorials were excluded. The full text of the screened papers was selected if found relevant to the present review. The screening was further expanded by performing a manual search based on the references of the full-text relevant papers.
Data Synthesis and Statistical Analysis
Outcomes were split into three main domains: i) procedural data (operative time, fluoroscopy time, and the number of device replacements); ii) outcomes (postoperative creatinine, hospital stay, and overall survival); iii) complications (device-related, accidental dislodgement, febrile episodes, and unplanned device replacement).
Continuous variables (procedural time, fluoroscopy time, length of stay, creatinine, overall survival) were pooled using the inverse variance of the mean difference (MD) with a random effect, 95% confidence interval (CI), and p-values. The incidences of complications and accidental tube dislodgment were pooled using the Cochran-Mantel-Haenszel method with a random effect model and reported as Odds Ratio (OR), 95% CI, and p-values. Statistical significance was set two-tail p-value <0.05. Study heterogeneity was assessed utilizing the I2 value. Substantial heterogeneity was defined as an I2 value between 75% and 100%. Significance was set at p-value <0.05 (two tails) and 95%CI. Meta-analysis was performed using Review Manager (RevMan) 5.4 software by Cochrane Collaboration. The quality assessment of the included studies was performed using the ROBINS-I for non-randomized studies (6).
EVIDENCE SYNTHESIS
Literature screening
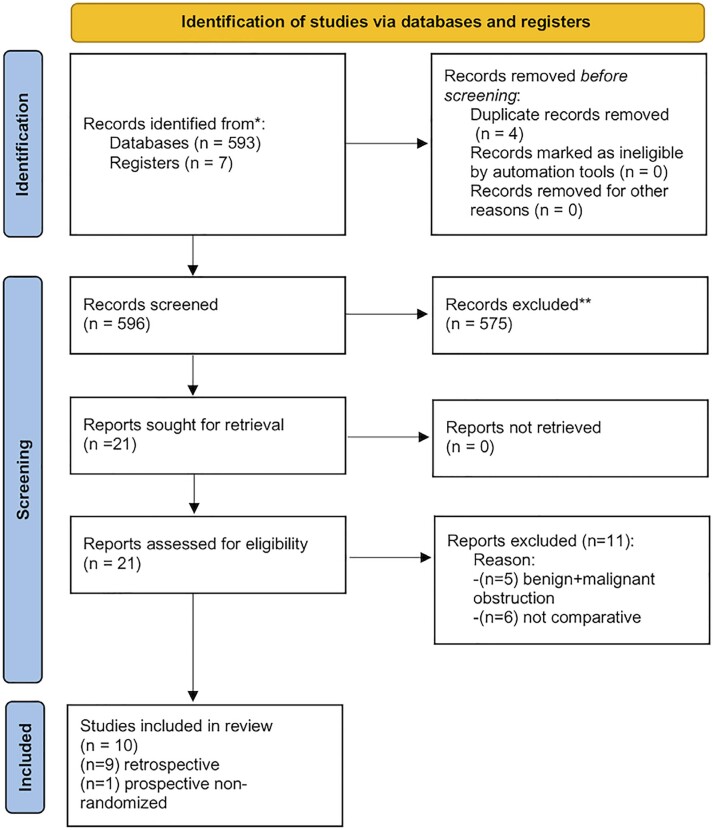
Literature search retrieved 596 papers. After title and abstract screening, 575 records were excluded because they were not related to the study purpose. The full texts of the remaining 21 studies were assessed for eligibility. Eleven studies were further excluded due to missing data. Finally, ten studies were accepted and included (7–16). Among these, only one was prospective (7) while the others were retrospective studies (8–16). No randomized study was found. Figure-1 shows the 2020 PRISMA flow diagram. Study characteristics are summarized in Table-1.
Figure 1. PRISMA flow diagram of the study.
Table 1. Characteristics of the included studies.
| Authors | Type of study: 1 RCT; 2 retrospective; 3 prospective | Ureteral stent patients (n) | Nephrostomy patients (n) | Total (n) | Type of cancer (absolute number) in ureteral stent | Type of cancer (absolute number) in nephrostomy tube |
|---|---|---|---|---|---|---|
| De Lorenzis et al. 2020 (8) | Retrospective | 27 | 24 | 51 | Upper Gastrointestinal tract (n=5); Lower Gastrointestinal tract (n=22) | Upper Gastrointestinal tract (n=4); Lower Gastrointestinal tract (n=20) |
| Gasparini et al. 1991 (9) | Retrospective | 7 | 15 | 22 | Ovarian cancer (n=2); cervical cancer (n=2); gastric cancer (n=1); colon cancer (n=1); prostate cancer (n=1) | Prostate cancer (n=1); Cervical cancer (n=3); Gastrointestinal (n=3); Ovarian cancer (n=1); Lymphoma (n=2); Unknown tumor (n=1); Bladder cancer (n=4) |
| Kanou et al. 2007 (10) | Retrospective | 51 | 24 | 75 | Cervix cancer (n=7); Rectal cancer (n=4); Prostate cancer (n=7); Bladder cancer (n=3); Ovarian cancer (n=2); Retroperitoneum tumor (n=1) | Lymphoma (n=2) |
| Ku et al. 2004 (11) | Retrospective | 68 | 80 | 148 | Not available | Not available |
| McCullough et al. 2008 (12) | Retrospective | 31 | 26 | 57 | Prostate cancer (n=5); bladder (n=5); colon (n=4); gynecological (n=7); breast (n=3); lymphoma (n=2); lung (n=1); others (n=4) | Prostate cancer (n=15); Bladder (n=7); Colon (n=3); gynecological (n=1) |
| Monsky et al. 2013 (7) | Prospective non randomized | 15 | 15 | 30 | Bladder (n=4); cervical (n=6); prostate (n=1); ovarian (n=2); endometrial (n=1); fallopian tube (n=1) | Bladder (n=5); cervical (n=3); uterine (n=2); prostate (n=2); colon (n=1); lymphoma (n=1); sarcoma (n=1) |
| Song et al. 2012 (13) | Retrospective | 50 | 25 | 75 | Cervical cancer (n=26); Endometrial cancer (n=22); Ovarian cancer (n=20); Uterine leiomyosarcoma (n=4); Vaginal carcinoma (n=1); Choriocarcinoma (n=2) | |
| Tan et al. 2019 (14) | Retrospective | 69 | 20 | 89 | Cervical cancer | Cervical cancer |
| Tibana et al. 2019 (15) | Retrospective | 26 | 15 | 41 | Bladder cancer (n=7); Uterine cancer (n=6); Metastatic colorectal cancer (n=4); Adenocarcinoma of the prostate (n=3); Sarcoma of the prostate (n=3); Colorectal adenocarcinoma (n=1); Retroperitoneal neuroendocrine tumor (n=1) | Uterine cancer (n=5); Bladder cancer (n=5); Prostate adenocarcinoma (n=3); Metastatic colorectal cancer (n=1); Ovarian cancer (n=1) |
| Zadra et al. 1987 (16) | Retrospective | 27 | 53 | 80 | Not available | Not available |
Study quality assessment
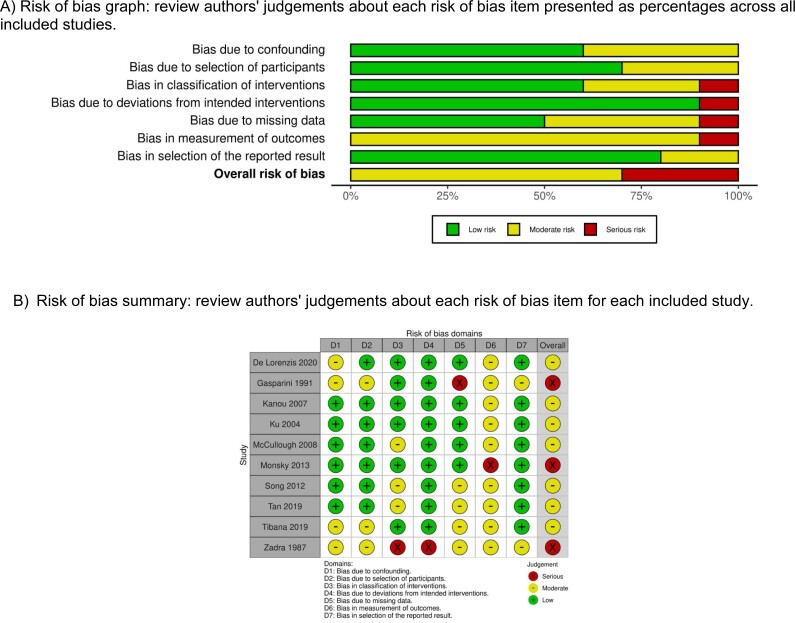
Supplementary Figure-1 demonstrates the details of the quality assessment. Seven studies exhibited a moderate risk of bias for all quality criteria, while three showed a serious risk of bias. The most common risk factor for quality assessment was the risk of bias in the classification of interventions, bias due to deviations from intended interventions, bias due to missing data, and bias in the measurement of outcomes as the studies were retrospective in design.
Supplementary Figure 1. Risk of bias in non-randomized controlled trials (ROBINS-I).
Procedural data
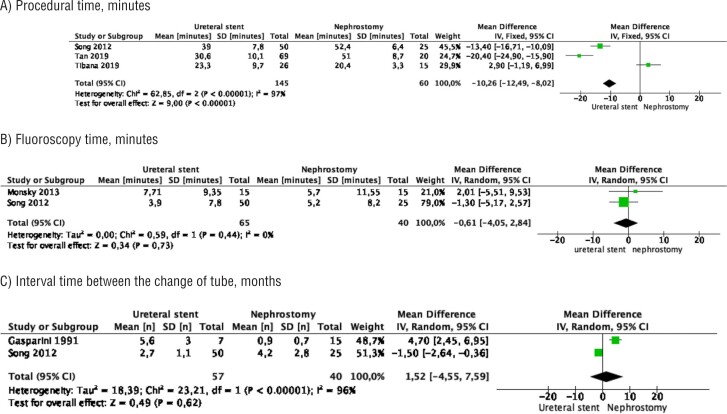
Meta-analysis from 3 studies (145 cases in stent and 60 cases in nephrostomy) showed that the mean procedure time was significantly shorter in the stent group (MD −10.26 minutes, 95% CI −12.40 −8.02, p<0.00001). Study heterogeneity was substantial (I2 97%) (Figure-2A).
Figure 2. Meta-analysis of procedural data. A) procedure time; B) fluoroscopy time; C) interval time between the change of drainage tubes over time.
Meta-analysis from 2 studies (65 cases in stent and 40 cases in nephrostomy) showed no difference between the two groups in mean fluoroscopy time (MD −0.61 minutes, 95% CI −4.05 – 2.84, p=0.73). Study heterogeneity was not significant (I2 0%) (Figure-2B).
Meta-analysis from 2 studies (65 cases in stent and 40 cases in nephrostomy) showed no difference between the two groups in the mean interval time between the change of drainage tubes over time (MD 1.52 months, 95% CI −4.55 – 7.59, p=0.62). Study heterogeneity was substantial (I2 96%) (Figure-2C).
Outcomes
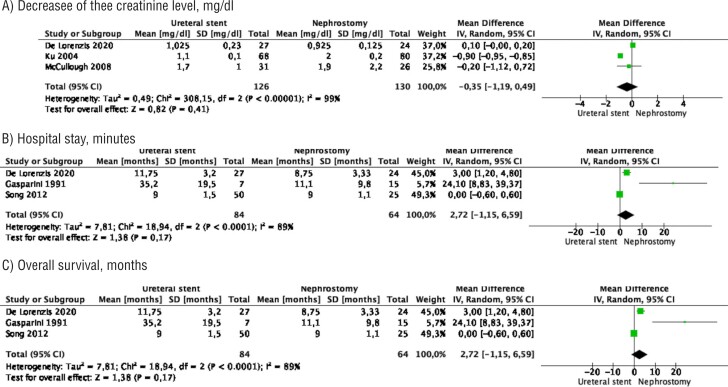
Meta-analysis from 3 studies (126 cases in stent and 130 cases in nephrostomy) showed no difference between the two groups in the decrease of the creatinine level after the procedure (MD −0.35 mg/dL, 95% CI −1.19 − 0.49, p=0.41). Study heterogeneity was substantial (I2 99%) (Figure-3A).
Figure 3. Meta-analysis of outcomes. A) decrease of the creatinine level after the procedure; B) hospital stay; C) overall survival.
Meta-analysis from 3 studies (146 cases in stent and 69 cases in nephrostomy) showed a significantly shorter hospital stay in the stent group compared to the nephrostomy tube group (MD −1.30 day, 95% CI −1.69 − −0. 92, p<0.0001). Study heterogeneity was substantial (I2 70%) (Figure-3B).
Meta-analysis from 3 studies (84 cases in stent and 64 cases in nephrostomy) showed no difference in the mean overall survival between the two groups (MD 2.72 months 95% CI −1.15 − 6.59, p=0.17). Study heterogeneity was substantial (I2 89%) (Figure-3C).
Complications
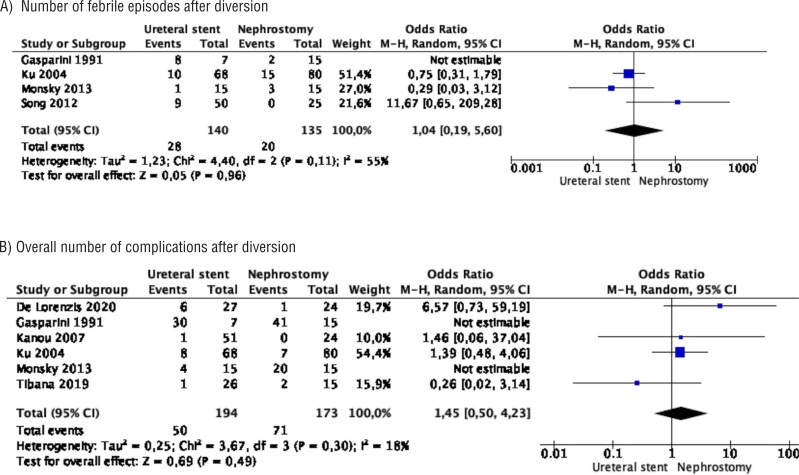
Meta-analysis from 4 studies (140 cases in stent and 135 cases in nephrostomy) showed no difference in the number of febrile episodes after diversion between the two groups (OR 1.04 95% CI 0.19 − 5.60, p=0.96). Study heterogeneity was moderate (I2 55%) (Figure-4A).
Figure 4. Meta-analysis of complications. A) number of febrile episodes after diversion; B) overall number of complications after diversion.
Meta-analysis from 4 studies (140 cases in stent and 135 cases in nephrostomy) showed no difference in the overall number of complications after diversion between the two groups (OR 1.46 95% CI 0.72 − 2.95, p=0.30). There was no study heterogeneity (I2 0%) (Figure-4B).
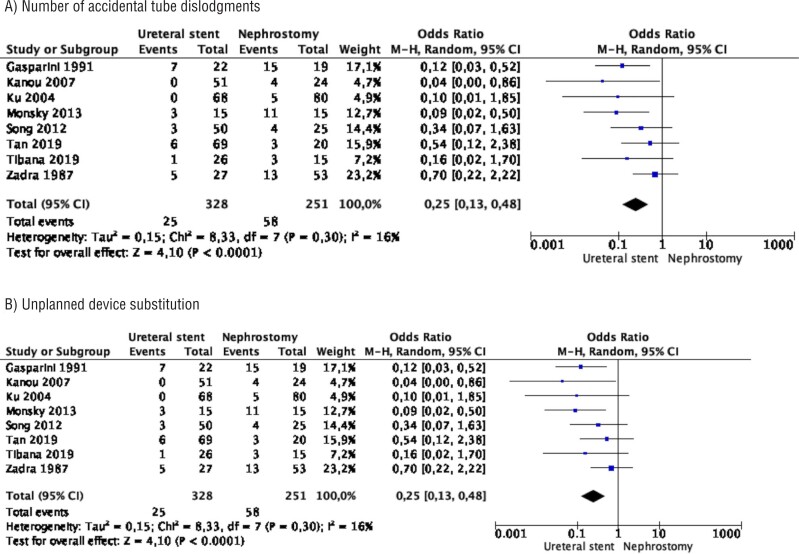
Meta-analysis from 8 studies (328 cases in stent and 251 cases in nephrostomy) showed that the number of accidental tube dislodgments was significantly lower in the stent group compared to the nephrostomy tube group (OR 0.25 95% CI 0.13 – 0.48, p<0.0001). Study heterogeneity was not important (I2 16%) (Figure-5A).
Figure 5. Meta-analysis of complications. A) number of accidental tube dislodgments; B) unplanned device substitution.
Meta-analysis from 3 studies (134 cases in stent and 119 cases in nephrostomy) showed no difference in unplanned device substitution between the two groups (OR 0.41 95% CI 0.06 – 2.98, p=0.38). Study heterogeneity was moderate (I2 43%) (Figure-5B).
DISCUSSION
Malignant ureteral obstruction commonly affects patients with advanced cancers. Development of ureteral obstruction is slow and insidious, typically causing dull pain, associated with fatigue and lethargy. Malignant ureteral obstruction is often an ominous sign frequently associated with poor survival (17).
Patients are usually referred when clinical or radiological evaluation resulting from urinary stasis often with worsening kidney function is observed as a consequence of ureteral obstruction. Occasionally, acute symptoms may occur such as fever due to urinary infection or renal colic with nausea and vomiting due to a sudden increase in pressure or stretch of the ureteral lumen with the hypercontractility of the ureteral smooth muscle and subsequent activation of nociceptors of renal afferent nerves fibers by prostaglandins (18). Sometimes, de novo acute obstructive uropathy can be the first presenting sign of advanced pelvic cancers (19).
The management of patients with ureteral obstruction needs a multi-disciplinary approach involving urologists, oncologists, palliative care physicians, interventional radiologists, along with patients and caregivers. In most cases, obstruction is primarily asymmetrical. In the case of bilateral involvement, it is common practice to drain only the symptomatic kidney or the kidney with better function in asymptomatic patients. Although there are recommendations within cancer-specific guidelines, there is a lack of consensus as well as a strong piece of evidence to support the decision process on which modality of decompression has a better outcome (20). Often, at this late stage of malignancy, the quality of life is poor and therefore the ethics of palliative decompression have often been questioned (19). Patients with advanced malignancy are poor surgical candidates, and the option of no intervention should also be discussed since the procedures themselves are not without potential morbidity (3). Indeed, the mean survival has been reported to be 120-140 days even with decompression (19, 21). Considerable variability in survival time has been reported in the literature and it is therefore important to identify objective criteria that can be used to estimate a patient's prognosis. Lapitan et al. followed up a cohort of patients who had a malignant ureteral obstruction and assessed the outcomes of those who were decompressed and those who were not (22). The authors found that the 6-month survival of patients who underwent diversion was 38% compared with 28% of those who did not. By 12 months, both groups had the same survival of 16%. In our analysis, we found no difference in mean survival between the two groups, pointing out that the type of urinary diversion does not impact the overall survival.
Since considerable variability in survival time has been reported in the literature, it is therefore important to identify objective criteria that can be used to what type of diversion will probably minimize the impact on patients’ quality of life.
Our study showed that stent placement had a shorter operative time and hospital stay but mean change interval trend over time did not differ. The overall complication rate was also not different between the two approaches, but the accidental displacement was significantly higher in the nephrostomy group. We also found no differences in creatinine level decrease after decompression or complication rates between the two procedures. Therefore, the ureteral stent placement had better procedural results, similar efficacy, and fewer handling issues than percutaneous nephrostomy tube placement. Indeed, the endoscopic approach, which represents a less invasive procedure, ensured a faster discharge of patients and a lower risk of tube displacement. This last point is very relevant because ureteral stent placement avoided repeated and unnecessary treatments, which can be very troublesome in frail patients. For all these reasons, physicians should be inclined to treat patients with malignant ureteral obstruction first with a ureteral stent, whenever possible. However, the decision to choose either should rely more on identifying risk factors associated with disease progression and resource availability at the place of practice.
The prognostic stratification model by Lienert et al. and Ishioka et al. have identified some risk factors to help decision-making for percutaneous nephrostomy placement with a 3-month to one-year benefit seen only in low or favourable risk and intermediate-risk group (19, 21). In addition, survival has been demonstrated to differ among cancers causing ureteral obstruction. For instance, tumours that originate outside the true pelvis (such as breast, pancreatic and gastric cancers) have a worse prognosis whereas patients with prostate and gynaecological cancers have longer survival times (2). For patients predicted to have relatively long survival, conversion to an internal ureteral stent may be recommended for a better quality of life [6]. In our meta-analysis, ureteral stenting had a significant advantage over the percutaneous nephrostomy cohort in terms of fewer device dislodgements (OR 0.25 95% CI 0.13 – 0.48, p<0.0001), an important consideration in patients needing a longer duration of diversion, especially in the modern era where metallic stents are available and superior to conventional polymeric stents and can stay up to 1 year before the next change (23).
As per our meta-analysis, the stent group showed better procedural results, similar efficacy, and fewer handling issues than the percutaneous nephrostomy group, however, it is not uncommon to face ureteral stent insertion failure, particularly in those cases with cystoscopy evidence of bladder or ureteral invasion (2, 4). Bladder cancer and prostate cancer-causing ureteral obstruction have shown a far higher failure rate than that caused by colon or breast cancer, probably due to the former directly invading the trigone, causing both intrinsic and extrinsic obstruction, making stent insertion, not possible (24). We also found that there was no difference in unplanned intervention for a device substitution either due to a mechanical device malfunction or for clinical reasons such as increasing febrile episodes secondary to the implants. These have a significant bearing on quality of life and hence it may be easiest for a patient to manage a ureteral stent than a percutaneous nephrostomy tube. Moreover, our meta-analysis significantly favoured ureteral stent insertion as this minimized the hospital stay (MD −1.30 day 95% CI −1.69 − −0. 92, p<0.0001) with a similar procedure-related fluoroscopy time for both procedures. These factors can influence decision-making for clinicians when faced with malignant ureteral obstruction especially as these patients are a critically vulnerable cohort. In addition, the procedural cost can also be taken into account. Only one study reported data on cost analysis and showed that the average cost of stenting was significantly lower than percutaneous nephrostomy (US$164.10 vs. US$552.20, respectively) (15).
Specific to malignant ureteral obstruction related to genitourinary malignancies, Shekarriz et al. analysed 103 patients with advanced malignancies treated with palliative urinary diversion (stent or percutaneous nephrostomy) and found that prostate cancer patients had the longest median survival, although the difference did not reach significance (25). Instead, gynaecological cancer patients survived approximately 4-fold longer than those with bladder cancer. Among patients with bladder cancer, those presenting de novo with ureteral obstruction survived significantly longer than those in whom obstruction developed after diagnosis and treatment already administered (26). These are important considerations as often in these patients if there is any difficulty in cystoscopic access for stent placement a percutaneous nephrostomy should be the best consideration for immediate diversion in advanced malignancies (11). However, a concern for percutaneous nephrostomy is that tube may need to be changed more frequently due to blockage with a reported incidence of 0.4-37% in various studies and can lead to more febrile episodes due to infection with a reported incidence of 2-8% in various studies (27). This can make clinicians and patients reluctant for this intervention, but our meta-analysis showed no significant difference between the two groups in the mean interval time between change of drainage tubes or the number of febrile episodes.
Our study pointed out two important take-home messages. First, ureteral stenting represents a less invasive procedure and has more appeal for patients, ensuring a faster discharge of patients, a longer exchange interval, and a lower risk of tube displacement. This last point is very relevant because ureteral stent placement avoided repeated and unnecessary interventions, which can save costs and precious time for other palliative procedures. Second, if stent placement is technically not possible, since no differences were noted in complication rates and unplanned need for device substitution, patients can be safely advised that percutaneous nephrostomy is not an inferior choice as a primary drainage procedure, especially in the presence of a pelvic malignancy or in patients with shorter survival that requires only palliative relief of ureteral obstruction.
The present review has some limitations. The study is based only on retrospective studies and one prospective study with no randomized data and the number of patients in each study is relatively small. We argue that this reflects two reasons. First of all, a randomized study may not be feasible, because the choice of kidney decompression is mostly patient-tailored. Second, the few comparative studies could also reflect the low interest of the scientific community in this field that, conversely, deserves attention as a pivotal role in the palliative management of end-life cancer patients. We were not able to assess the quality of life after urinary diversion due to different tools to evaluate it in the studies included in this meta-analysis and this can be considered another study limitation. We also could not assess the materials of stent as this was not reported in the studies included in our review.
CONCLUSIONS
While both forms of urinary diversion can be utilized in malignant obstruction, our meta-analysis favours stents as the preferred choice as these are easier to maintain, and ureteral stent placement should be recommended whenever feasible. If the malignant obstruction precludes a stent placement, then percutaneous nephrostomy tube is a safe alternative. The findings of our review can help clinicians in using a personalized approach to choose either option in malignant ureteral obstruction.
REFERENCES
- 1.Holden S, McPhee M, Grabstald H. The rationale of urinary diversion in cancer patients. J Urol. 1979;121:19–21. doi: 10.1016/s0022-5347(17)56644-6. [DOI] [PubMed] [Google Scholar]
- 2.Liberman D, McCormack M. Renal and urologic problems: management of ureteric obstruction. Curr Opin Support Palliat Care. 2012;6:316–321. doi: 10.1097/SPC.0b013e328354a1d8. [DOI] [PubMed] [Google Scholar]
- 3.Kouba E, Wallen EM, Pruthi RS. Management of ureteral obstruction due to advanced malignancy: optimizing therapeutic and palliative outcomes. J Urol. 2008;180:444–450. doi: 10.1016/j.juro.2008.04.008. [DOI] [PubMed] [Google Scholar]
- 4.Danilovic A, Antonopoulos IM, Mesquita JL, Lucon AM. Likelihood of retrograde double-J stenting according to ureteral obstructing pathology. Int Braz J Urol. 2005;31:431–436. doi: 10.1590/s1677-55382005000500003. discussion 436. [DOI] [PubMed] [Google Scholar]
- 5.Misra S, Coker C, Richenberg J. Percutaneous nephrostomy for ureteric obstruction due to advanced pelvic malignancy: have we got the balance right? Int Urol Nephrol. 2013;45:627–632. doi: 10.1007/s11255-013-0458-3. [DOI] [PubMed] [Google Scholar]
- 6.Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919–i4919. doi: 10.1136/bmj.i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Monsky WL, Molloy C, Jin B, Nolan T, Fernando D, Loh S, et al. Quality-of-life assessment after palliative interventions to manage malignant ureteral obstruction. Cardiovasc Intervent Radiol. 2013;36:1355–1363. doi: 10.1007/s00270-013-0571-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.De Lorenzis E, Lievore E, Turetti M, Gallioli A, Galassi B, Boeri L, et al. Ureteral Stent and Percutaneous Nephrostomy in Managing Malignant Ureteric Obstruction of Gastrointestinal Origin: A 10 Years’ Experience. Gastrointest Disord. 2020;2:456–468. [Internet]. Available at. < https://mdpi-res.com/d_attachment/gastrointestdisord/gastrointestdisord-02-00041/article_deploy/gastrointestdisord-02-00041-v2.pdf?version=1607499679>. [Google Scholar]
- 9.Gasparini M, Carroll P, Stoller M. Palliative percutaneous and endoscopic urinary diversion for malignant ureteral obstruction. Urology. 1991;38:408–412. doi: 10.1016/0090-4295(91)80227-x. [DOI] [PubMed] [Google Scholar]
- 10.Kanou T, Fujiyama C, Nishimura K, Tokuda Y, Uozumi J, Masaki Z. Management of extrinsic malignant ureteral obstruction with urinary diversion. Int J Urol. 2007;14:689–692. doi: 10.1111/j.1442-2042.2007.01747.x. [DOI] [PubMed] [Google Scholar]
- 11.Ku JH, Lee SW, Jeon HG, Kim HH, Oh SJ. Percutaneous nephrostomy versus indwelling ureteral stents in the management of extrinsic ureteral obstruction in advanced malignancies: are there differences? Urology. 2004;64:895–899. doi: 10.1016/j.urology.2004.06.029. [DOI] [PubMed] [Google Scholar]
- 12.McCullough TC, May NR, Metro MJ, Ginsberg PC, Jaffe JS, Harkaway RC. Serum creatinine predicts success in retrograde ureteral stent placement in patients with pelvic malignancies. Urology. 2008;72:370–373. doi: 10.1016/j.urology.2007.12.068. [DOI] [PubMed] [Google Scholar]
- 13.Song Y, Fei X, Song Y. Percutaneous nephrostomy versus indwelling ureteral stent in the management of gynecological malignancies. Int J Gynecol Cancer. 2012;22:697–702. doi: 10.1097/IGC.0b013e318243b475. [DOI] [PubMed] [Google Scholar]
- 14.Tan S, Tao Z, Bian X, Zhao Y, Wang N, Chen X, et al. Ureteral stent placement and percutaneous nephrostomy in the management of hydronephrosis secondary to cervical cancer. Eur J Obstet Gynecol Reprod Biol. 2019;241:99–103. doi: 10.1016/j.ejogrb.2019.08.020. [DOI] [PubMed] [Google Scholar]
- 15.Tibana TK, Grubert RM, Santos RFT, Fornazari VAV, Domingos AA, Reis WT, et al. Percutaneous nephrostomy versus antegrade double-J stent placement in the treatment of malignant obstructive uropathy: a cost-effectiveness analysis from the perspective of the Brazilian public health care system. Radiol Bras. 2019;52:305–311. doi: 10.1590/0100-3984.2018.0127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zadra JA, Jewett MA, Keresteci AG, Rankin JT, St Louis E, Grey RR, et al. Nonoperative urinary diversion for malignant ureteral obstruction. Cancer. 1987;60:1353–1357. doi: 10.1002/1097-0142(19870915)60:6<1353::aid-cncr2820600632>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 17.Wong LM, Cleeve LK, Milner AD, Pitman AG. Malignant ureteral obstruction: outcomes after intervention. Have things changed? J Urol. 2007;178:178–183. doi: 10.1016/j.juro.2007.03.026. discussion 183. [DOI] [PubMed] [Google Scholar]
- 18.Roza C, Laird JM. Pressor responses to distension of the ureter in anaesthetised rats: characterisation of a model of acute visceral pain. Neurosci Lett. 1995;198:9–12. doi: 10.1016/0304-3940(95)11947-u. [DOI] [PubMed] [Google Scholar]
- 19.Lienert A, Ing A, Mark S. Prognostic factors in malignant ureteric obstruction. BJU Int. 2009;104:938–941. doi: 10.1111/j.1464-410X.2009.08492.x. [DOI] [PubMed] [Google Scholar]
- 20.Borda AP, Charney-Sonnek F, Fonteyne V, Papaioannou EG. EAU Guidel. 2014. European Association of Urology guidelines on pain management & palliative care. [Internet]. Available at. < https://d56bochluxqnz.cloudfront.net/media/2014-Pain-Management_LR-1.pdf>. [Google Scholar]
- 21.Ishioka J, Kageyama Y, Inoue M, Higashi Y, Kihara K. Prognostic model for predicting survival after palliative urinary diversion for ureteral obstruction: analysis of 140 cases. J Urol. 2008;180:618–621. doi: 10.1016/j.juro.2008.04.011. discussion 621. [DOI] [PubMed] [Google Scholar]
- 22.Lapitan MC, Buckley BS. Impact of palliative urinary diversion by percutaneous nephrostomy drainage and ureteral stenting among patients with advanced cervical cancer and obstructive uropathy: a prospective cohort. J Obstet Gynaecol Res. 2011;37:1061–1070. doi: 10.1111/j.1447-0756.2010.01486.x. [DOI] [PubMed] [Google Scholar]
- 23.Chen Y, Liu CY, Zhang ZH, Xu PC, Chen DG, Fan XH, et al. Malignant ureteral obstruction: experience and comparative analysis of metallic versus ordinary polymer ureteral stents. World J Surg Oncol. 2019;17:74–74. doi: 10.1186/s12957-019-1608-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Allen DJ, Longhorn SE, Philp T, Smith RD, Choong S. Percutaneous urinary drainage and ureteric stenting in malignant disease. Clin Oncol (R Coll Radiol) 2010;22:733–739. doi: 10.1016/j.clon.2010.07.004. [DOI] [PubMed] [Google Scholar]
- 25.Shekarriz B, Shekarriz H, Upadhyay J, Banerjee M, Becker H, Pontes JE, et al. Outcome of palliative urinary diversion in the treatment of advanced malignancies. Cancer. 1999;85:998–1003. doi: 10.1002/(sici)1097-0142(19990215)85:4<998::aid-cncr30>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- 26.Wilson JR, Urwin GH, Stower MJ. The role of percutaneous nephrostomy in malignant ureteric obstruction. Ann R Coll Surg Engl. 2005;87:21–24. doi: 10.1308/1478708051432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ahmad I, Saeed Pansota M, Tariq M, Shahzad Saleem M, Ali Tabassum S, Hussain A. Comparison between Double J (DJ) Ureteral Stenting and Percutaneous Nephrostomy (PCN) in Obstructive Uropathy. Pak J Med Sci. 2013;29:725–729. doi: 10.12669/pjms.293.3563. [DOI] [PMC free article] [PubMed] [Google Scholar]