Highlights
-
•
Mammography is an effective approach for early detection of breast cancer.
-
•
Established longitudinal patterns of adherence to breast cancer screening recommendation.
-
•
Having a family doctor can encourage compliance to breast cancer screening recommendation.
-
•
Current smokers were more likely to be non-regular screeners.
Keywords: Breast cancer, Screening, Mammography, Cohort, Alberta’s Tomorrow Project
Abstract
Breast cancer screening is an important prevention component as it can reduce cancer mortality and improve survival. Understanding patterns of adherence to screening recommendations is essential to guide health promotion strategies and policy implementation efforts. The 1999 Alberta screening guidelines were used to determine screening status for eligible female participants in Alberta’s Tomorrow Project (n = 4,972), a longitudinal province-based cohort. Screening patterns were derived based on screening status assessed at enrollment (2001–2008) and follow-up (2008–2011). Information on reason for screening was also collected at each time point. Multinomial logistic regression was used to assess potential predictors of adherence to screening recommendations. The majority of participants were up-to-date with screening at enrollment (79.3 %), and follow-up (75.2 %). Among all participants, 66.3 % were up-to-date at both time points (considered ‘regular screeners’), 8.9 % were not up-to-date or never at enrollment but up-to-date at follow-up (considered ‘new screeners’), 21.6 % were not up-to-date at follow-up (considered ‘episodic screeners’) and 3.2 % had never participated in screening (considered ‘non-screeners’). Having a family doctor was the strongest factor associated with being a regular screener (OR (95 % CI): 0.37 (0.24 0.57) when compared with new screeners. Current smokers were more likely to be non-regular screeners. The primary reason for screening was routine screening or age. In conclusions, non-regular screening patterns were more prevalent among women without a family doctor. This finding suggests having a family doctor is an important mechanism to encourage screening. Further work is required to raise awareness of current recommendations and to understand and address reasons for non-adherence.
1. Introduction
Breast cancer is the most common cancer and the second leading cause of cancer-related death in women in Canada, with 1 in 33 Canadian women expected to die from breast cancer (Gaudette et al., 2019). As breast cancer poses a significant risk to women’s health, there is a need to understand how to identify individuals at potential risk and provide appropriate prevention and early detection strategies while balancing efficient use of the health care system and risks of screening. Screening for cancer is an important component of secondary prevention as it can lead to early diagnosis and increased likelihood that cancer treatment is effective. Screening for breast cancer with mammography is widely accepted as an effective approach for early detection of breast cancer (Tonelli et al., 2011). A 2016 systematic review showed that compared to no screening, mammography screening is associated with a 38 % lower incidence of advanced stage breast cancer in women over 50 and a 14–33 % (depending on age category) lower breast cancer mortality (Nelson et al., 2016). In Canada, breast cancer screening programs using mammography are associated with a 40 % reduction in breast cancer mortality (Coldman et al., 2014).
Starting in 1988, Canadian provinces established breast cancer screening programs, and the Canadian Task Force on Preventative Health Care released updated guidelines in 2001 (Ringash and Canadian Task Force on Preventive Health Care, 2001). National guidelines in Canada recommended biennial mammography for asymptomatic women aged 50–74 (Tonelli et al., 2011). In line with this, Alberta released similar provincial breast cancer screening recommendations in 1999 (Alberta Medical Association, 1999) for women aged 50–69. In 2013, Alberta released updated guidelines (Towards Optimized Practice (TOP), 2013) to increase the eligibility to women aged 50–74. In addition, it also added specific recommendations for women deemed high-risk (women with a first-degree relative with breast cancer, who have had breast biopsies, or with a history of chest wall radiation). For these women, annual mammography is recommended and screening should start 5 to 10 years younger than the youngest case in their family, but no younger than age 25 and no older than age 40 (Towards Optimized Practice (TOP), 2013).
The Canadian Partnership Against Cancer (CPAC) set a goal to have at least 70 % of average risk individuals participate in breast cancer screening (Canadian Partnership Against Cancer, 2015). In 2012, the Canadian Community Health Survey (CCHS) reported that 61 % of women aged 50–74 across Canada met current guidelines, with at least one mammogram in the past two years. The most recent CCHS report (2017) showed an increase in screening rates, with 78.5 % of Canadian women aged 50–74 reporting having had a mammogram in the past three years (Statistics Canada, 2018). Information on screening status at any time point is important, but regular screening over time is required to reduce cancer mortality. Investigation into not only participation, but also retention in breast cancer screening is needed to provide a clear picture of screening behaviours among women who are eligible for breast cancer screening.
Alberta’s Tomorrow Project (ATP) is a large longitudinal cohort study that was established in 2000 and has collected health and lifestyle information from participants over time, including cancer screening behaviours (Ye et al., 2017). ATP’s aim is to understand factors associated with the development of cancer and chronic diseases to inform prevention strategies. Our previous study has suggested some potential promoters and barriers for colorectal cancer screening uptake (Solbak et al., 2018). The present study aims to examine cross-sectional adherence to screening recommendations and longitudinal screening patterns among women in ATP. Further, we also investigate potential predictors for women’s adherence to breast cancer screening recommendations.
2. Methods
2.1. Study population
Details on the design, enrollment, questionnaires used in ATP are described elsewhere (Robson et al., 2016). Briefly, between 2000 and 2008, 31,072 adults aged 35–69 with no previous history of cancer (except non-melanoma skin cancer) and residing in Alberta, Canada were recruited from the general population by telephone-based random digit dialling method (48.8 % enrollment rate). At enrollment, participants completed a Health and Lifestyle Questionnaire, which collected information on personal and family health history (including breast cancer in first-degree relatives, including parents, siblings and children, and age at diagnosis), smoking habits, anthropometric dimensions, sociodemographic characteristics, and cancer screening behaviours. A follow-up survey was administered between 2008 and 2011, which collected updated information on personal and family health history and cancer screening tests. Informed consent was obtained from participants upon enrollment into the study. The Health Research Board of Alberta Cancer Committee granted ethics approval for this project (HREBA.CC-17-0526) (#25985).
The initial study sample consisted of 12,919 female participants. ATP enrollment criteria included no previous history of cancer (confirmed via linkage with the Alberta Cancer Registry) other than non-melanoma skin cancer. Survey data from final sample was collected between 2001 and 2011. Minor revisions and updates to provincial breast cancer screening guidelines during this time period did not impact the 1999 recommendations, hence, the 1999 Alberta screening recommendations were used which recommended that women age 50–69 undergo routine mammography screening every 2 years. Women younger than 50 at the time of enrollment and above age 70 at follow-up were excluded from the study sample (n = 7,140; Fig. 1). Women who experienced cancer during the study period (n = 16) or reported ‘breast cancer treatment’ as their reason (described below) for mammography (n = 45) were excluded in addition to women who were pregnant within the study years (n = 6) as they could not undergo mammography. Additionally, participants who completed the mammogram at follow-up < 2 years after the date of last mammogram (assuming with upper limit of the time duration) from enrollment survey (n = 667) were excluded to reduce the chance of double reporting that same mammography screening at both surveys. Participants without screening information (i.e. missing data) at either enrollment or follow-up (n = 73) were also excluded. After all exclusions, the final sample consisted of 4,972 participants (Fig. 1).
Fig. 1.
Flow chart of Alberta’s Tomorrow Project participant selection.
2.2. Determining screening status and patterns
On each survey at enrollment and follow-up, participants were asked “have you ever had a mammogram”. If yes, participants were asked when they had their last mammogram - on the enrollment survey they chose from a list of answer options (less than 6 months ago, 6 months to less than 1 year ago, 1 year to less than 2 years ago, 2 years to less than 3 years ago, 3 years to less than 5 years ago, and 5 or more years ago) and on the follow-up survey they were asked to provide month and year. Participants were also asked why they had their last mammogram at both baseline and follow-up (described below in Reasons for Screening).
Participant’s screening status at enrollment and follow-up was categorized as:
-
•
“up-to-date” if they reported their last mammography within the past 2 years,
-
•
“not-up-to date” if their last reported mammography was >2 years ago,
-
•
“never” if they reported they had never had a mammogram.
Using the screening status at enrollment and follow-up, we derived four patterns of screening behaviours for mammography:
-
•
Regular screener: “Up-to-date” at enrollment & “Up-to-date” at follow-up
-
•
New screener: “Not up-to-date” or “Never” at enrollment & “Up-to-date” at follow-up
-
•
Episodic screener: “Up-to-date” or “Not up-to-date” or “Never” at enrollment & “Not up-to-date” at follow-up
-
•
Non-screener: “Never” at enrollment & “Never” at follow-up
2.3. Reasons for screening
Participants indicated the reason(s) for their mammography from the following options: family history of breast cancer, part of regular checkup/routine screening, age, previously detected lump, on hormone replacement therapy, breast problem, follow-up of breast cancer treatment or other (open text). Responses were allocated into three categories according to a priority hierarchy associated with the screening guidelines: 1) problem (previously detected lump or on hormone replacement therapy or breast problem or other), 2) family (family history of breast cancer), or 3) screening (part of regular checkup/routine screening or age). For this analysis, participants were assigned to only one category based on the highest priority reason given.
2.4. Assessment of high-risk participants
The 1999 screening recommendations did not include risk stratification or risk-specific guidelines, however the updated 2013 guidelines defined women considered at average and high risk, along with providing risk-stratified breast cancer screening recommendations. A recent study showed that annual screening improved early detection for women with high-risk compared with biennial screening (Chiarelli et al., 2020). We used the risk stratification criteria to determine the proportion of women at average and high-risk in order to compare screening uptake and patterns according to the 1999 guidelines. Women were considered at high-risk if they reported one or more first-degree relatives with breast cancer on the enrollment survey, in line with the 2013 guidelines. Additional information on high-risk criteria according to the 2013 TOP guidelines, such as breast biopsies or history of chest wall radiation, were not collected at enrollment. Finally, the 2013 guidelines were updated to recommend annual screening for women at high-risk. Therefore, even though the 2013 guidelines were not in use during our data capture timeframe (2001–2011), we conducted a secondary analysis to determine how many women at high-risk of breast cancer were already undergoing annual instead of biennial screening.
2.5. Statistical analysis
Sociodemographic characteristics of participants at enrollment are presented as means and standard deviations for continuous variables and as counts and percentages for categorical variables. Multinomial logistic regression models were used to evaluate the relationship, expressed as odds ratios, between breast cancer screening patterns and potential predictors, with regular screeners assigned as the reference screening pattern. Estimations were adjusted for participant characteristics at enrollment, including age, residential area (rural; urban; defined by Canadian Postcode data), marital status (divorced/ separated/ widowed/ single, never married; married/ living with someone), education level (high school or lower; college; university or post graduate education), employment status (not employed, student, home maker or other; retired; employed part-time; employed full-time), annual household income (<$70,000; ≥$70,000; cut-off was derived from the median Canadian family income in 2008 (Statista, 2021), body mass index (BMI), smoking status (current smoker; current non-smoker), self-reported health status (fair/poor; excellent/very good/good), and family history of any cancer (no; yes). Estimations were also adjusted for having a regular family doctor (no; yes), which was not available on the enrollment survey but was measured on the follow-up survey; it was assumed that health system access behaviours did not vary significantly over the study period. All analyses were performed using SAS statistical software version 9.4.
3. Results
3.1. Sociodemographic characteristics at enrollment
Table 1 summarizes the sociodemographic characteristics of the participants at enrollment. The mean age of participants was 56.7 years, most participants were married or living with partner, had post-secondary education, had an annual household income of less than $70,000, were current non-smokers, and had a family history of cancer.
Table 1.
Characteristics of ATP participants at enrollment.
| Variable | Category | All participants |
|---|---|---|
| (n = 4972) | ||
| Age (years) at enrollment |
(mean ± SD) | 56.7 ± 4.6 |
| ≥ 50 and <60 | 3542 (71.2 %) | |
| ≥ 60 and <70 | 1430 (28.8 %) | |
| Residential area | Urban | 3619 (72.8 %) |
| Rural | 1353 (27.2 %) | |
| Marital status | Married/ living with someone | 3712 (74.7 %) |
| Divorced/ separated/ widowed/ single | 1259 (25.3 %) | |
| Education level | High school or lower | 1596 (32.1 %) |
| College | 1815 (36.5 %) | |
| University | 1560 (31.4 %) | |
| Employment status | Employed full-time | 1985 (40.0 %) |
| Employed part-time | 1106 (22.2 %) | |
| Not employed, student, home maker or other | 829 (16.7 %) | |
| Retired | 1049 (21.1 %) | |
| Household income | < $70 000 | 2867 (59.7 %) |
| ≥ $70 000 | 1935 (40.3 %) | |
| BMI (kg/m2) | (mean ± SD) | 27.8 ± 5.9 |
| <25 | 1716 (34.6 %) | |
| ≥25 and < 30 | 1827 (36.8 %) | |
| ≥30 | 1418 (28.6 %) | |
| Smoking status | Current smoker | 728 (14.7 %) |
| Current non-smoker | 4239 (85.3 %) | |
| Self-reported health | Excellent/Very good | 4614 (93.6 %) |
| Others/Fair/Poor | 317 (6.4 %) | |
| Family history of any cancer | Yes | 3064 (61.6 %) |
| No | 1908 (38.4 %) | |
| Family history of breast cancer | Yes | 881 (17.7 %) |
| No | 4091 (82.3 %) | |
| Family doctor | Yes | 4680 (94.2 %) |
| No | 287 (5.8 %) | |
Except for age and BMI (mean ± SD), all values are presented as a number and percentage.
All characteristics were measured at enrollment except for currently having a family doctor, which was measured at follow-up.
Residential area was derived from reported postal code.
3.2. Screening status and patterns
At enrollment, 79.3 % of participants were up-to-date on screening and only 5.5 % reported never having been screened (Table 2). At follow-up, fewer participants were up-to-date (75.2 %), more participants were not up-to-date (21.6 %) and fewer participants reported never being screened (3.2 %). The majority of participants were regular screeners (66.3 %), being up-to-date on screening at both time points (Table 2). Over 21 % of participants were episodic screeners, and 8.9 % were new screeners, with 3.2 % reporting never having participated in breast cancer screening at both enrollment and follow-up (Table 2).
Table 2.
Breast cancer screening status and patterns of ATP participants.
| All participants1 (n = 4972) |
High-risk2 participants (n = 881) |
||
|---|---|---|---|
| Screening Status3 | |||
| Enrollment |
Up-to-date | 3943 (79.3 %) | 500 (56.8 %) |
| Not up-to-date | 756 (15.2 %) | 325 (36.9 %) | |
| Never | 273 (5.5 %) | 56 (6.3 %) | |
| Follow-up | Up-to-date | 3737 (75.2 %) | 502 (57.0 %) |
| Not up-to-date | 1075 (21.6 %) | 354 (40.2 %) | |
| Never | 160 (3.2 %) | 25 (2.8 %) | |
| Screening Pattern4 | |||
| Regular screener | 3295 (66.3 %) | 339 (38.5 %) | |
| Episodic screener | 1075 (21.6 %) | 354 (40.2 %) | |
| New screener | 442 (8.9 %) | 163 (18.5 %) | |
| Non-screener | 160 (3.2 %) | 25 (2.8 %) | |
All participants were selected based on 1999 Alberta Breast Cancer Screening guideline.
High-risk participants were a sub-group of all participants who had one or more first degree relative diagnosed with breast cancer based on the 2013 Alberta Breast Cancer Screening guideline.
Screening status was defined at enrollment and follow up as “up-to-date” if they reported their last mammography within the past 2 years (1 year for high-risk), “not-up-to date” if their last reported mammography was >2 years ago (>1 year ago for high-risk), and, “never” if they reported they had never had a mammogram.
Screening patterns were defined as follows, Regular screener: “Up-to-date” at enrollment & “Up-to-date” at follow-up; New screener: “Not up-to-date” or “Never” at enrollment & Up-to-date” at follow-up; Episodic screener: “Up-to-date” or “Not up-to-date” or “Never” at enrollment & “Not up-to-date” at follow-up; Non-screener: “Never” at enrollment & “Never” at follow-up.
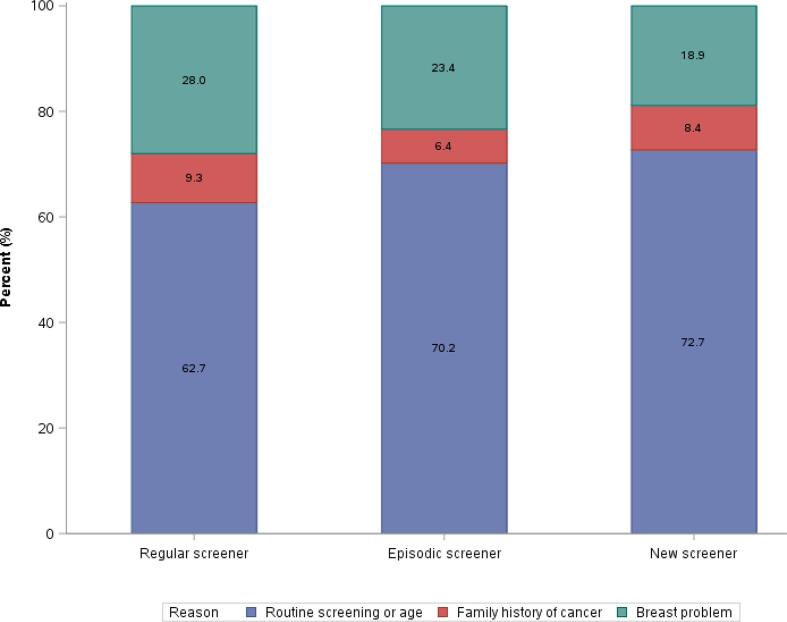
3.3. Reasons for screening
Reasons for getting a mammogram reported by participants is presented at enrollment and stratified by screening patterns (Fig. 2). The majority of participants in all three screening patterns reported routine screening or age as the reason for mammography. A breast problem was reported as the reason for obtaining a mammogram by 28 % of regular screeners, 23.4 % of episodic screeners, and 18.9 % of new screeners (Fig. 2). Trends of reasons for screening were observed to be similar at follow-up, with more participants in all screening patterns reporting routine screening or age (69.5 %, 76.4 %, and 79.4 %, for regular, episodic, and new screeners, respectively), and fewer reporting a breast problem as their reason for screening (18.3 %, 16.1 %, 13.4 %, for regular, episodic, and new screeners, respectively).
Fig. 2.
Reasons given for breast cancer screening tests at enrollment across screening patterns. Reasons for screening were reported at enrollment and the responses were allocated into three categories according to a priority hierarchy: (1) problem (previously detected lump or on hormone replace therapy or other breast problem); (2) family history of breast cancer; (3) routine screening or age (part of regular checkup or routine screening or age). Participants could select more than one category, but were assigned only one category based on the highest priority reason given.
3.4. Predictors of screening patterns
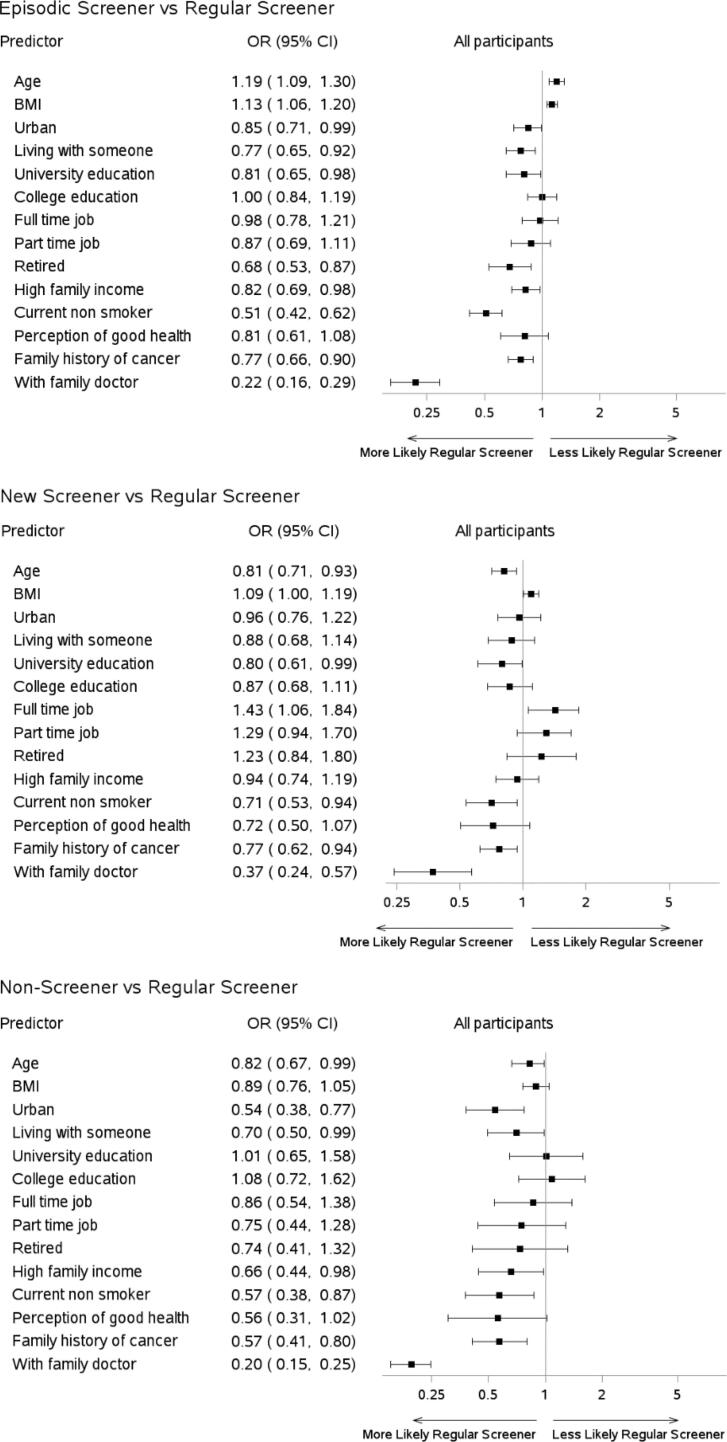
Fig. 3 illustrates the odds ratios for participant characteristics associated with mammogram screening patterns, using regular screeners as the reference group. Overall, across all screening patterns, having a family doctor was significantly associated with greater likelihood of being a regular screener (78 %, 63 %, and 80 % for comparisons to episodic, new, and non-screeners, respectively). Similarly, across all screening patterns, participants with a family history of any cancer were 23–43 % more likely to be a regular screener (Fig. 3). Retired participants were more likely to be regular screeners than episodic screeners, and participants employed full-time were more likely to be new screeners than regular screeners (Fig. 3). Participants with higher household income were more likely to be regular screeners than episodic or non-screeners. Participants living in rural areas were more likely to be non-screeners or episodic screeners than regular screeners. Even though age was one of the important confounding variables in most epidemiological models, age was not a consistent predictor across screening groups in this study. Finally, current smokers were more likely to be non-regular screeners.
Fig. 3.
Forest plot of odds ratios (ORs) for predictors of breast cancer screening patterns. Regular screeners were used as the reference groups. ORs were mutually adjusted for participant’s enrollment (except having a family doctor which was measured at follow-up) characteristics which including age, residential area (rural; urban), marital status (divorced/ separated/ widowed/ single, never married; married/ living with partner), education level (high school or lower; college; university or post graduate education), employment status (not employed, student, home maker or other; retired; employed part-time; employed full-time), annual household income (<$70 000; ≥$70 000; cut-off was derived from the median Canadian family income in 2008), body mass index (BMI), smoking status (current smoker; current non-smoker), self-reported health status (fair/ poor; excellent/ very good/ good), family history of any cancer (no; yes), and currently having a regular family doctor (no; yes).
3.5. Assessment of high risk participants
Women considered at high-risk for cancer were investigated during secondary analysis to determine what proportion were already being screened annually (as per the 2013 updated guidelines) instead of biennially (as per the 1999 guidelines). At enrollment, 56.8 % of women at high risk were up-to-date for annual screening, while 36.9 % were not up-to-date (Table 2). The proportion of women at high-risk who were up-to-date for annual screening at follow-up remained similar, with more women not up-to-date on screening, and fewer reporting never having been screened. Finally, 38.5 % were considered regular screeners for annual screening, 40.2 % were episodic screeners, 18.5 % were new screeners, and 2.8 % were non-screeners (Table 2).
4. Discussion
Participation in regular breast cancer screening is the recommended method to detect possible cancerous growths at the earliest stage to reduce cancer progression and mortality. In the present study, we examined screening behaviours over time among screening-eligible female participants of Alberta’s Tomorrow Project. The majority of participants (66 %) were reported to be up-to-date with biennial screening at both enrollment and follow-up, and among women identified as being at high-risk of breast cancer, 39 % were already participating regularly in annual mammography screening. Reasons for screening were also examined with the most common reason cited as routine screening or age. Having a family doctor was the strongest predictor of being a regular screener, while having a family history of cancer, and not current smoking strongly predicted regular participation in breast cancer screening.
The present findings are in line with the CPAC goal of at least 70 % adherence to mammogram screening and appear to be higher than the general population. A 2012 CCHS survey of over 14,000 Canadian women reported that 61 % of women age 50–74 were up-to-date for breast cancer screening (Canadian Partnership Against Cancer, 2017), compared to 79 % and 75 % of women in ATP at enrollment and follow-up, respectively over the follow-up period (average of 5.2 years). By comparison, in a population-based study of women in Ontario, 65 % of women age 50–74 reported having at least one mammogram within a 30-month period in 2011. This same study reported that among women who had a mammogram, 83 % had a subsequent mammogram within 30 months, equating to approximately 54 % of the sample being regular screeners (Cancer Care Ontario, 2016). This is lower than the present study, in which 66 % of women were determined to be regular screeners.
Information on predictors of non-regular breast cancer screening patterns is limited. A study of women screened by the Ontario Breast Screening Program found high socioeconomic status and living in an urban area were related to returning for a subsequent screening appointment (within 3 years of initial breast cancer screening) (Tatla et al., 2003), which our study supports given that women with higher household income and living in an urban area were more likely to be regular screeners than episodic or non-screeners. Interestingly, the difference in high vs low socioeconomic status did not exist for women living in rural areas in the Ontario Breast Screening Program study. In contrast, an Australian study of longitudinal patterns of breast cancer screening found women in rural areas had similar screening rates to their urban counterparts, despite having poorer access to mammograms (Leung et al., 2014a). However, a 2014 systematic review of 28 global studies concluded that rural populations are less likely to have ever had a mammogram or to have an up-to-date mammogram (Leung et al., 2014b).
A large study of 36,575 American women over the age of 40 found associations between age and mammography are non-linear, as mammography screening uptake increased until approximately age 61 but then declined (Yuan et al., 2021). Our results suggest a similar pattern, as the relationship between age and screening pattern was mixed for regular vs new and episodic screeners. Our findings echo that of Yuan et al, as it appears once women are screened, they seem to be screened consistently, and at an older age screening may plateau and become inconsistent (i.e. episodic). As the risk of breast cancer nearly doubles between the ages of 50–59 and 60–69 (Siegel et al., 2014), further research is needed to clarify why older women participate, but often only episodically.
Of particular interest is the association between modifiable risk factors and screening patterns. Women who reported having a family doctor were significantly more likely to be a regular. An analysis of 2006 CCHS data found active interaction with a family doctor doubled the odds of the participant having had a mammogram in the past 2 years (Poole et al., 2010). Other literature has thoroughly documented the role of physicians in increasing cancer screening compliance (Fox et al., 1991, Metsch et al., 1998, Wallace et al., 2006). Thus, ensuring primary care access is key and may improve cancer screening adherence. There is room for improvement, as across Canada, 7.9 % and 5.6 % of women 50–64 and over 65, respectively, report not having a primary health care provider (Statistics Canada, 2019).
We also found that new screeners were more likely to have a full-time job than regular screeners. An American study found that among female employees, having paid sick leave (to utilize preventative medical care) significantly increased the likelihood of preventative cancer screening (Wilson et al., 2014). In the Albertan context, ensuring a provincial paid leave for women without sick leave could further increase screening uptake among working women. Currently in Alberta, workers are entitled to 5 days of unpaid leave after 90 days of employment (Alberta Federation of Labour, 2020).
Despite the evidences that overweight and obesity increased risk of developing breast cancer, inconsistent findings have been reported of the impact of BMI on breast cancer screening behaviors (Samman et al., 2022, Wee et al., 2000). Our results showed that increased BMI discourage the adherence to breast cancer screening recommendations in episodic screener but not in never screener. Similarly, perception of good health did not predict mammography pattern in our study, which added the inconsistency among previous publications (Katapodi et al., 2004, Walker et al., 2014). It is possible that variations in study design, recruitment age, risk-classification criteria, and objective measurement contributed to part of these discrepancies; however, the mixed results from both cross-sectional and prospective studies warrant further research to reveal the underlying factors account for these predictive uncertainties.
Following the 1999 recommendations, the 2013 TOP guidelines were introduced to provide risk specific recommendations, suggesting women at high-risk be screened annually instead of every 2 years. Even though the guidelines were introduced in 2013, and our study data was collected between 2001 and 2011, we wanted to determine what proportion of women at high-risk were already being screened annually. When we applied the annual screening recommendations, 57 % of women at high-risk of breast cancer were up-to-date at enrollment, and 39 % were considered regular screeners for annual screening over time. Therefore, even though the 2013 guidelines for annual screening were not in place during the study period (2001–2011), some women at high-risk of breast cancer were already undergoing more frequent screening. Further research is required to verify if differences in screening frequency in the same period translated to different breast cancer detection among high-risk women.
Beyond a two-tiered recommendation hierarchy, there is growing consensus in the medical community that screening regimens should be tailored to patient risk determined by consideration of a more comprehensive suite of factors including breast density and genetics (Lee et al., 2017). The present analysis highlights the disparity in recommendation adherence between risk groups and supports the notion of risk probability-based screening recommendations. Given ATP’s longitudinal cohort platform and database of health and lifestyle factors along with biological samples, future directions for this work could include the linkage of ATP data to administrative health information, biomarkers, and genetic data. This integrated data platform could be used to examine the risk-probability of breast or other cancers related to other factors in the study population and potentially inform the creation of tailored recommendations.
Findings from the study should be interpreted considering several limitations. Mammography was self-reported by participants, and therefore may be subject to information bias. Potential misclassification for high-risk assessment may result from lacking information on breast biopsies or history of chest wall radiation. However, a study of self-reported mammography and screening status ascertained from administrative health records found women with varying levels of familial risk of breast cancer accurately reported their mammogram uptake (Walker et al., 2013). As 99 % of ATP participants have consented to link with administrative data, future studies could expand on validation of self-reported cancer screening behaviours with Alberta Breast Cancer Screening Program (ABCSP) data as ABCSP has accurate screening, and follow up data after 2012–13. Additional information from potential health administrative data linkage can also reduce the probability of misclassification of high-risk individuals. The categorization of potential confounders may have influenced the findings, however, cut-off values for binary variables such as income were derived from the median Canadian income from the same time period or were based on the statistic from Statistics Canada and limited by the availability of ATP data. Despite adjusting for a broad set of confounders, the possibility of residual confounding related to unmeasured potential confounders, such as physician referral patterns and breast self-examinations, exists. For example, ethnic minority group has been reported to have low mammography uptake compared with other populations (Raynault et al., 2020, Schwartz et al., 2021). However, the ethnicity variable was removed due to the small ethnic minority population in ATP dataset, which cannot support a model-based analysis. The use of data from a large, longitudinal cohort provides many strengths. As participants were surveyed repeatedly, there is a low recall bias. Further, the longitudinal nature of ATP allows for assessment of individual’s screening adherence over time and for determination of screening patterns. In the present study, two time points were analyzed, however as ATP is an ongoing cohort study, future research could investigate screening patterns using additional time points.
5. Conclusion
Overall, almost 80 % of women in the ATP cohort reported mammography screening at least once over the follow-up period. Women with a family doctor or family history of cancer were more adherent to mammography screening recommendations. Non-regular screening patterns were more prevalent among women who smoked, had a lower household family income, or worked full-time and therefore represent target groups for screening promotion. Our study highlights the potential interventional opportunities to increase the screening rate to reduce cancer risk by improving early detection. For example, tailored interventions adapted for high-risk group may improve the mammography uptake within specific groups. Further research is required to better understand and address reasons for non-adherence in order to identify barriers to screening to maximize cancer prevention efforts.
6. Authorship
JEV, GST, and JYX: conceptualization, supervision; NS and JYX: data curation, formal analysis; OL and JYX: writing - original draft, visualization; all authors: review & editing.
Ethical Standard Disclosure
This study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving human subjects/participants were approved by the former Alberta Cancer Board’s Research Ethics Committee and the University of Calgary Conjoint Health Research Ethics Board and the Alberta Cancer Research Ethics Committee (current analysis). Written informed consent was obtained from all subjects/participants.
CRediT authorship contribution statement
Olivia K. Loewen: Writing – original draft, Visualization, Writing – review & editing. Navjot Sandila: Data curation, Formal analysis, Writing – review & editing. Grace Shen-Tu: Conceptualization, Supervision, Writing – review & editing. Jennifer E. Vena: Conceptualization, Supervision, Writing – review & editing. Huiming Yang: Writing – review & editing. Kara Patterson: Writing – review & editing. Jian-Yi Xu: Conceptualization, Supervision, Data curation, Formal analysis, Writing – original draft, Visualization, Writing – review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
Alberta’s Tomorrow Project is only possible because of the commitment of its research participants, its staff and its funders: Alberta Health, Alberta Cancer Foundation, Canadian Partnership Against Cancer and Health Canada, and substantial in kind funding from Alberta Health Services. Cancer registry data was obtained through linkage with Surveillance & Reporting, Cancer Research & Analytics, Cancer Care Alberta. The views expressed herein represent the views of the author(s) and not Alberta’s Tomorrow Project or any of its funders. The authors would like to thank Bonnie Chiang and Michelle Sharma for their contributions and feedback on the manuscript.
Data availability
The authors do not have permission to share data.
References
- Alberta Federation of Labour, 2020. Alberta Needs Paid Sick Leave for all Workers [WWW Document]. URL https://www.afl.org/alberta_needs_paid_sick_leave_for_all_workers.
- Alberta Medical Association, 1999. The Early Detection of Breast Cancer What You and Your Doctor Can Do [WWW Document]. URL https://www.bowmont.ca/pdf/altaClinicalPract/what_you_and_doc_can_do.pdf.
- Cancer Care Ontario, 2016. Ontario cancer screening performance report 2016 [WWW Document]. URL https://www.cancercareontario.ca/en/screening-performance-report-2016.
- Canadian Partnership Against Cancer, 2015. Cancer Screening in Canada: An Overview of Screening Participation for Breast, Cervical and Colorectal Cancer [WWW Document]. URL https://www.partnershipagainstcancer.ca/topics/cancer-screening-participation-overview.
- Canadian Partnership Against Cancer, 2017. Breast Cancer Screening in Canada: An Interim Report [WWW Document]. URL https://www.partnershipagainstcancer.ca/topics/breast-cancer-screening-quality-indicators-2017.
- Chiarelli A.M., Blackmore K.M., Mirea L., Done S.J., Majpruz V., Weerasinghe A., Rabeneck L., Muradali D. Annual vs biennial screening: diagnostic accuracy among concurrent cohorts within the Ontario Breast Screening Program. J. Natl. Cancer Inst. 2020;112:400–409. doi: 10.1093/jnci/djz131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coldman A., Phillips N., Wilson C., Decker K., Chiarelli A.M., Brisson J., Zhang B., Payne J., Doyle G., Ahmad R. Pan-Canadian study of mammography screening and mortality from breast cancer. J. Natl. Cancer Inst. 2014:106. doi: 10.1093/jnci/dju261. [DOI] [PubMed] [Google Scholar]
- Fox S.A., Murata P.J., Stein J.A. The impact of physician compliance on screening mammography for older women. Arch. Intern. Med. 1991;151:50–56. [PubMed] [Google Scholar]
- Gaudette, L.A., Illing, E.M., Hill, G.B., 2019. Canadian Cancer Statistics 2019, Health reports / Statistics Canada, Canadian Centre for Health Information = Rapports sur la santé / Statistique Canada, Centre canadien d’information sur la santé.
- Katapodi M.C., Lee K.A., Facione N.C., Dodd M.J. Predictors of perceived breast cancer risk and the relation between perceived risk and breast cancer screening: a meta-analytic review. Prev. Med. (Baltim.) 2004;38:388–402. doi: 10.1016/j.ypmed.2003.11.012. [DOI] [PubMed] [Google Scholar]
- Lee C.I., Chen L.E., Elmore J.G. Risk-based breast cancer screening: implications of breast density. Med. Clin. North Am. 2017;101:725–741. doi: 10.1016/j.mcna.2017.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung J., McKenzie S., Martin J., Dobson A., McLaughlin D. Longitudinal patterns of breast cancer screening: mammography, clinical, and breast self-examinations in a rural and urban setting. Womens Health Issues. 2014;24:e139–e146. doi: 10.1016/j.whi.2013.11.005. [DOI] [PubMed] [Google Scholar]
- Leung J., McKenzie S., Martin J., McLaughlin D. Effect of rurality on screening for breast cancer: a systematic review and meta-analysis comparing mammography. Rural Remote Health. 2014;14:2730. [PubMed] [Google Scholar]
- Metsch L.R., Mccoy C.B., Pereyra M., Trapido E., Miles C. The role of the physician as an information source on mammography. Cancer Practice. 1998;6:229–236. doi: 10.1046/j.1523-5394.1998.006004229.x. [DOI] [PubMed] [Google Scholar]
- Nelson H.D., Fu R., Cantor A., Pappas M., Daeges M., Humphrey L. Effectiveness of breast cancer screening: systematic review and meta-analysis to update the 2009 U.S. Preventive Services Task Force Recommendation. Ann. Intern. Med. 2016;164:244–255. doi: 10.7326/M15-0969. [DOI] [PubMed] [Google Scholar]
- Poole B., Black C., Gelmon K., Kan L. Is Canadian women’s breast cancer screening behaviour associated with having a family doctor? Can. Fam. Physician. 2010;56:e150–e157. [PMC free article] [PubMed] [Google Scholar]
- Raynault M.-F., Féthière C., Côté D. Social inequalities in breast cancer screening: evaluating written communications with immigrant Haitian women in Montreal. Int. J. Equity Health. 2020;19:209. doi: 10.1186/s12939-020-01322-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ringash J., Canadian Task Force on Preventive Health Care Preventive health care, 2001 update: screening mammography among women aged 40–49 years at average risk of breast cancer. CMAJ. 2001;164:469–476. [PMC free article] [PubMed] [Google Scholar]
- Robson P.J., Solbak N.M., Haig T.R., Whelan H.K., Vena J.E., Akawung A.K., Rosner W.K., Brenner D.R., Cook L.S., Csizmadi I., Kopciuk K.A., McGregor S.E., Friedenreich C.M. Design, methods and demographics from phase I of Alberta’s Tomorrow Project cohort: a prospective cohort profile. CMAJ Open. 2016;4:E515–E527. doi: 10.9778/cmajo.20160005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samman, E., Mkuu, R., Zhang, X., Scummings, S., Burdine, J., 2022. Body mass index and breast and cervical cancer screening. Womens Health Rep. (New Rochelle, N.Y.) 3, 508–514. [DOI] [PMC free article] [PubMed]
- Schwartz C., Chukwudozie I.B., Tejeda S., Vijayasiri G., Abraham I., Remo M., Shah H.A., Rojas M., Carillo A., Moreno L., Warnecke R.B., Hoskins K.F. Association of population screening for breast cancer risk with use of mammography among women in medically underserved racial and ethnic minority groups. JAMA Netw. Open. 2021;4:e2123751. doi: 10.1001/jamanetworkopen.2021.23751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegel R., Ma J., Zou Z., Jemal A. Cancer statistics, 2014. CA Cancer J. Clin. 2014;64:9–29. doi: 10.3322/caac.21208. [DOI] [PubMed] [Google Scholar]
- Solbak N.M., Xu J.-Y., Vena J.E., Al Rajabi A., Vaseghi S., Whelan H.K., McGregor S.E. Patterns and predictors of adherence to colorectal cancer screening recommendations in Alberta’s Tomorrow Project participants stratified by risk. BMC Public Health. 2018;18:177. doi: 10.1186/s12889-018-5095-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Statista, 2021. Median annual family income in Canada 2000-2018 [WWW Document]. URL https://www.statista.com/statistics/465739/median-annual-family-income-in-canada-since-1990.
- Statistics Canada, 2018. Health Fact Sheets: Cancer Screening, 2017 [WWW Document]. URL https://www150.statcan.gc.ca/n1/pub/82-625-x/2018001/article/54977-eng.htm.
- Statistics Canada, 2019. Primary health care providers, 2017 [WWW Document]. URL https://www150.statcan.gc.ca/n1/pub/82-625-x/2019001/article/00001-eng.htm.
- Tatla R.K., Paszat L.F., Bondy S.J., Chen Z., Chiarelli A.M., Mai V. Socioeconomic status & returning for a second screen in the Ontario breast screening program. Breast. 2003;12:237–246. doi: 10.1016/s0960-9776(03)00100-0. [DOI] [PubMed] [Google Scholar]
- Tonelli M., Connor Gorber S., Joffres M., Dickinson J., Singh H., Lewin G., Birtwhistle R., Fitzpatrick-Lewis D., Hodgson N., Ciliska D., Gauld M., Liu Y.Y., Canadian Task Force on Preventive Health Care Recommendations on screening for breast cancer in average-risk women aged 40–74 years. CMAJ. 2011;183:1991–2001. doi: 10.1503/cmaj.110334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towards Optimized Practice (TOP) Breast cancer clinical practice guideline. Clin. Pract. Guidelines. 2013:1–14. [Google Scholar]
- Walker M.J., Chiarelli A.M., Mirea L., Glendon G., Ritvo P., Andrulis I.L., Knight J.A. Accuracy of self-reported screening mammography use: examining recall among female relatives from the Ontario Site of the Breast Cancer Family Registry. ISRN Oncol. 2013;2013:810573. doi: 10.1155/2013/810573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker M.J., Mirea L., Glendon G., Ritvo P., Andrulis I.L., Knight J.A., Chiarelli A.M. Does perceived risk predict breast cancer screening use? Findings from a prospective cohort study of female relatives from the Ontario site of the breast cancer family registry. Breast. 2014;23:482–488. doi: 10.1016/j.breast.2014.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace A.E., MacKenzie T.A., Weeks W.B. Women’s primary care providers and breast cancer screening: who’s following the guidelines? Am. J. Obstet. Gynecol. 2006;194:744–748. doi: 10.1016/j.ajog.2005.10.194. [DOI] [PubMed] [Google Scholar]
- Wee C.C., McCarthy E.P., Davis R.B., Phillips R.S. Screening for cervical and breast cancer: is obesity an unrecognized barrier to preventive care? Ann. Intern. Med. 2000;132:697–704. doi: 10.7326/0003-4819-132-9-200005020-00003. [DOI] [PubMed] [Google Scholar]
- Wilson F.A., Wang Y., Stimpson J.P. The role of sick leave in increasing breast cancer screening among female employees in the U.S. J. Cancer Policy. 2014;2:89–92. [Google Scholar]
- Ye M., Robson P.J., Eurich D.T., Vena J.E., Xu J.-Y., Johnson J.A. Cohort profile: Alberta’s Tomorrow Project. Int. J. Epidemiol. 2017;46:1097–1098l. doi: 10.1093/ije/dyw256. [DOI] [PubMed] [Google Scholar]
- Yuan C., Kulkarni K., Dashevsky B.Z. Preventive care: how mammography utilization changes as women age. J. Am. Coll. Radiol. 2021;17:238–247. doi: 10.1016/j.jacr.2019.09.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The authors do not have permission to share data.