Abstract
Earlier studies showed that mice primed for a few hours with the trans-sialidase (TS) of Trypanosoma cruzi, the agent of Chagas' disease, become highly susceptible to trypanosomal infection. These studies suggest that TS affects parasite virulence independent of antigenic stimulation. Potentially, TS could enhance or reduce the virulence of heterologous microbes depending on the mechanism of TS action and on the type of immune response elicited by the particular parasite. We tested this hypothesis by expressing heterologous TS in Leishmania major, a protozoan parasite that causes cutaneous leishmaniasis and lacks TS and the TS product α2-3-linked sialic acid. Leishmania cells transfected with a T. cruzi TS expression construct made high levels of active enzyme, which was present in the promastigotes and shed into the extracellular milieu. TS expression did not affect L. major binding to and entry into cultured macrophages or its tropism for macrophage infection in vivo. However, TS-expressing L. major exhibited elevated virulence in BALB/c mice, as determined by lesion progression, parasite numbers, and macro- and microscopic examination of cutaneous lesions. Several genetic tests proved that the enhanced virulence was directly attributable to TS expression. The results are consistent with TS functioning to sabotage the mouse immune system to confer a growth advantage on T. cruzi and transgenic L. major. These data suggest that heterologous expression of T. cruzi virulence factors in Leishmania may provide a new approach for dissecting their function in vivo.
The flagellate protozoan Trypanosoma cruzi, the agent of Chagas' disease, invades cells in the form of trypomastigotes. Inside the host cell, parasites escape from the initial vacuole into the cytoplasm, where they develop into amastigotes, which, after several cycles of multiplication, differentiate back into the invasive trypomastigotes. Upon exiting infected cells, trypomastigotes may invade adjacent cells by migration through the extracellular matrix or distant cells through the circulation. Coincident with the intracellular amastigote-trypomastigote differentiation is the expression of trans-sialidase (TS), an enzyme that specifically hydrolyzes α2,3-linked sialic acid (neuraminidase or sialidase activity) (17) and transfers the sialic acid to β-galactosidase acceptors (transferase activity) (16, 26, 29; for a review, see reference 27). Both host and parasite glycoconjugates can act as substrates for TS. TS is anchored to the outer membrane of trypomastigotes by a glycophosphoinositol (GPI) moiety (18, 23) and is readily shed into the extracellular environment by the action of specific phospholipases (23).
Several lines of evidence implicate TS as a direct mediator of T. cruzi-host cell interactions (27). First, surface membrane-bound TS promotes trypomastigote adhesion to host cells by direct interaction with sialyl epitopes present on the host cell plasma membrane (14, 28). Such adhesion is thought to be a consequence of the sialic acid-binding lectin activity of TS, which occurs in an environment containing relatively low concentrations of terminal nonreducing β-galactosidase residues, as in the glycoconjugate-containing outer membrane of mammalian cells (29). Second, extracellular trypomastigotes can be classified into two populations, a minor subset of 20 to 30% with relatively high TS activity (TS+ parasites), and a major subset with relatively low or no TS expression (TS− parasites) (19). In vitro, TS+ trypomastigotes are much better than the TS− parasites in adhering to and invading cells (19). What's more, the TS+ parasites exhibit greater virulence than the counterpart TS− parasites do for BALB/c mice (19). The importance of TS in T. cruzi invasion was underscored by the ability of exogenous enzyme to switch the less invasive TS− parasites to the highly invasive TS+ phenotype (19). Lastly, soluble and membrane-bound TS is thought to facilitate the rupture of the phagolysosomal membrane, which would allow the parasite to escape into the cytoplasm for completion of the intracellular cycle (8).
Other experiments suggest that TS promotes invasion without directly affecting T. cruzi penetration of mammalian cells. Specifically, mice sensitized with TS become highly permissive hosts for T. cruzi (3). In fact, TS sensitization could turn a nonlethal dose of T. cruzi into a mortal one (3). The low dosage (nanograms per mouse) and timing (1 to 2 h before parasite inoculation) of TS sensitization suggested that the enzyme enhanced virulence by altering the dynamics of innate and/or acquired immune responses to T. cruzi, independent of antigenic stimulation of B- and T-cell receptors. Accordingly, TS did not effectively potentiate parasitemia in immune-deficient mice (3).
It stands to reason that if TS sabotages the mouse defenses to spur T. cruzi growth, then TS might enhance or reduce virulence in other parasite infections as well. To test this hypothesis, we expressed TS in Leishmania major, a protozoan parasite that causes cutaneous leishmaniasis. We chose L. major because it elicits a well-characterized immune response in mammalian hosts, particularly mice (11, 22). Moreover, these parasites can be readily modified genetically, yielding lines which express high levels of exogenous transgenes (7, 12). Thus, Leishmania offers a convenient background on which to assay the function of specific T. cruzi virulence factors independent of other T. cruzi determinants. Our results show that transgenic L. major expressing TS becomes extremely virulent to mice and that the ectopically expressed trypanosomal enzyme is the cause of the enhanced virulence.
MATERIALS AND METHODS
Parasite culture.
All studies were performed with the cloned L. major strain Friedlin V1 (MHOM/IL/80/Friedlin). Promastigotes were maintained at 26°C in medium M199 (Gibco-BRL) supplemented with 10% heat-inactivated fetal calf serum, 20 mM HEPES (pH 7.4), 1% penicillin-streptomycin (Cellgro), 0.01% adenine (Sigma), 0.0004% hemin, and 0.1% biotin (Sigma) (10). Parasites were subcultured every 3 to 4 days at a 1:100 dilution. To prepare inocula for animal studies, parasites were grown in NNN medium supplemented with 20% heat-inactivated fetal calf serum overlying a solid phase composed of 3% nutrient agar, 0.6% NaCl, 10% defibrinated sheep blood, and 0.3% glucose (30). Under these conditions, densities in excess of 5 × 107 parasites per ml could be obtained.
Leishmania expression construct for T. cruzi TS.
DNA from the T. cruzi TS-containing plasmid 19Y (4, 18) was digested with SmaI and XhoI. The expected 3.8-kb DNA fragment encoding the full-length TS protein was purified and inserted into SmaI-cut plasmid pXG1a (2, 12), yielding pXG-TS. DNA from pXG1a and pXG-TS were prepared with the Qiagen Maxiprep kit.
Transfection of Leishmania.
Plasmid DNAs (40 μg) for either pXG-TS or the pXG1a vector were transfected by electroporation into 4 × 107 logarithmic-phase L. major V1 promastigotes as described before (10) except that promastigotes were plated onto semisolid M199 medium containing G418 (16 μg/ml). After incubation at 26°C for 7 days, single colonies (L1, L2, and L3 for pXG-TS, Lv for pXG1a) were picked into 1 ml of M199 lacking drug, incubated at 26°C to a density of 106 to 107/ml, and then transferred into 10 ml of growth medium containing G418 (10 μg/ml).
TS assays.
Promastigotes were cultured for 5 days to reach the stationary phase and then centrifuged at 1,250 × g for 10 min to give a supernatant (conditioned medium in Fig. 1B) and a cell pellet, which was washed twice with 0.01 M sodium phosphate buffer (PBS, pH 7.2) and resuspended in the same buffer at 108 parasites per ml. TS activity in cell lysates and conditioned medium was determined as described before (29).
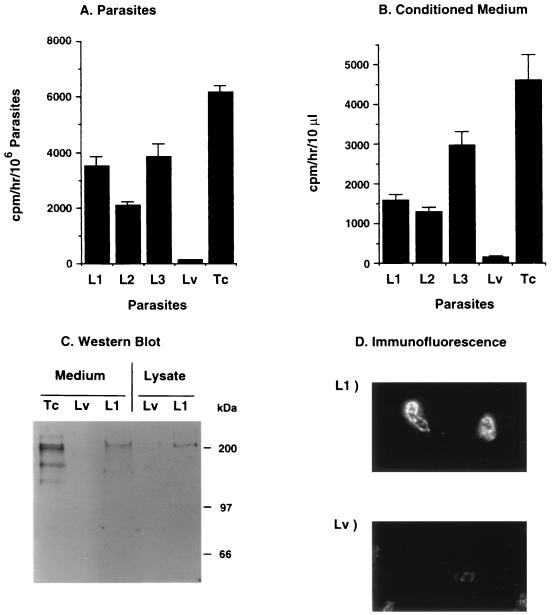
FIG. 1.
Characteristics of TS expression in L. major. (A) Specific TS activity in lysates of three promastigote clones of L. major transfected with the TS gene of T. cruzi (L1, L2, and L3) and in one clone transfected with empty vector (Lv). The specific activity of TS in T. cruzi trypomastigotes (Tc) is shown for comparison. (B) Specific activity in medium conditioned by 5 days of growth of the clones in panel A. Error bars represent standard deviations. (C) Western blot analysis of TS in conditioned medium and lysates of L1 and Lv promastigotes and conditioned medium of T. cruzi trypomastigotes (Tc). TS was detected with MAb TCN-2. (D) Immunofluorescence analysis of TS in paraformaldehyde-fixed L1 and Lv promastigotes, using MAb TCN-2 as the primary antibody and a fluorescein isothiocyanate-labeled second antibody. A control IgG1 MAb (anti-p-azophenylarsonate) did not react with L1 promastigotes (not shown).
Western blot analysis.
Stationary-phase promastigotes were washed twice with PBS (pH 7.2) and resuspended at 2 × 108/ml in PBS (pH 7.2) containing 1% Triton X-100 and protease inhibitors (10 μM pepstatin A, 0.1 mM iodoacetamide, 10 μM leupeptin, 10 μg of soybean trypsin inhibitor per ml, and 1 mU of aprotinin per ml [all from Sigma]). Conditioned medium was concentrated 10-fold using Biomax filters (Millipore) with a 50-kDa cutoff. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis, transfer to nitrocellulose, and TS detection with TCN-2 were performed as described previously (20).
Analysis of infection in vivo.
Groups of three to five female BALB/c or C57BL/6 mice (Jackson Laboratory, Bar Harbor, Maine), 4 to 6 weeks old, were injected subcutaneously in the footpad with 106 stationary-phase L. major L1 and Lv promastigotes (i.e., 14 to 17 days in culture). Most stationary-phase promastigotes were metacyclic promastigotes, as determined by peanut agglutinin binding (25). Cutaneous lesions produced by the inocula were monitored weekly by measuring the thickness of the infected footpad with a Vernier caliper and calculating the difference against uninfected-footpad measurements (30). The thickness of the uninfected contralateral footpad was measured as a control. The number of parasites in the footpad lesion was calculated by a modified limiting-dilution analysis of a single-cell suspension from individual excised lesions (12, 30). In brief, single-cell suspensions of the lesions from three mice at 4 weeks postinoculation were plated in Grace's medium in triplicate, and the mean of the negative log parasite titer was calculated 6 days after the start of the cultures.
For measurement of TS activity in tissues, BALB/c mice were killed by CO2 asphyxia, and the cutaneous lesions were cut into small pieces, which were weighed and applied directly to a tube containing TS assay reagents ([14C]N-acetyl-lactosamine and fetuin) plus 1% Triton X-100 to solubilize the tissues. After an overnight incubation (14 h) at room temperature, TS activity was determined as described previously (29).
For light microscopy, infected BALB/c mice were killed by CO2 asphyxia, and tissues from the cutaneous lesions, kidneys, spleen, and liver were fixed in 10% buffered formalin, embedded in paraffin, sectioned (5 μm thick), and stained with hematoxylin and eosin. Formalin-fixed right feet were hemisected, demineralized for 24 h in 5% nitric acid, and processed as described above. For electron microscopy, samples from the inoculation site were fixed in Trump's fixative in cacodylate buffer (pH 7.2), postfixed in 1% osmium tetroxide in 0.1 M cacodylate buffer (pH 7.4), dehydrated through graded ethanol solution, and embedded in Embed-812 epoxy resin. For orientation, 1-μm-thick sections were stained with toluidine blue. Sections 50 nm thin were cut, stained with uranyl acetate and lead citrate, and photographed with a Philips EM201 electron microscope (9).
Immunofluorescence.
Promastigotes were washed three times in PBS (pH 7.2), fixed in 4% paraformaldehyde for 1 min at room temperature, and washed with H2O. The parasites were incubated with 1% bovine serum albumin and 1% goat serum (Boehringer Mannheim) in PBS (pH 7.2) (buffer A) for 1 h at room temperature to block nonspecific binding. Primary antibody (protein G-purified TCN-2 monoclonal antibody [MAb], immunoglobulin G1 [IgG1] subtype) (20) was added to the cells for 1 h at a concentration of 1.7 μg/ml, followed by washing with PBS (pH 7.2) and addition of fluorescein 5-isothiocyanate-conjugated goat anti-mouse IgG (Boehringer Mannheim), as described previously (20).
Cloning of L1 parasites from BALB/c mouse lesions.
Five BALB/c mice were infected with L1 and Lv promastigotes and sacrificed by CO2 asphyxia when the footpads were 4 to 5 mm in circumference for the L1-infected mice (∼4 weeks postinoculation). The cutaneous lesions were dissected, homogenized in a tissue grinder, resuspended in M199 medium, and centrifuged at 500 rpm for 5 min to yield a supernatant enriched in amastigotes. The parasites were then cloned in four 96-well microtiter plates at a density of 0.25 parasites per well in growth medium without G418. When clonal growth was evident by light microscopy (minimum of 10 promastigotes per well), the parasites in each well were resuspended and subdivided into two aliquots. One was placed in another well containing growth medium without G418, and the other aliquot was placed in another well containing G418 (10 μg/ml). After 1 to 2 weeks, parasites from wells showing growth were recovered, and cultures were expanded for subsequent studies.
RESULTS
Expression of T. cruzi TS in L. major.
We constructed an expression system for the T. cruzi TS in L. major using the constitutive expression vector pXG1a (7, 10) containing the full-length coding region for TS (4, 18). pXG1a is an expression vector containing the Leishmania splice acceptor and flanking sequences required to direct gene expression in both the promastigote and amastigote stages of the Leishmania life cycle. Following transfection of the pXG-TS construct into Leishmania, we selected three independent clonal lines for further study (L1, L2, and L3). Additionally, a control line (Lv) was obtained following transfection with the empty vector pXG1a.
Evidence of TS expression in L. major was provided by direct measurement of enzymatic activity in the parasites and in the medium conditioned by Leishmania growth. Lysates of clones L1, L2, and L3 had relatively high TS activity, about 30 to 60% of the specific enzymatic activity of T. cruzi trypomastigotes (Silvio strain), while lysates of clone Lv had no detectable TS activity (Fig. 1A). The heterologously expressed TS was released into the culture medium (Fig. 1B), as observed with T. cruzi (14, 23).
Further demonstration of TS expression in L. major was provided by Western blot and immunofluorescence analysis. The MAb TCN-2, specific for the C-terminal tandem repeats of TS (20), recognized a major band of ∼200 kDa in the conditioned medium and lysate of clone L1 but not of clone Lv (Fig. 1C). A control mouse anti-p-azo-phenylarsonate IgG1 did not react with the L1 lysates (data not shown). The 200-kDa band was the expected size for the TS of the 19Y clone of T. cruzi, for the same gene gives a protein of similar size in Escherichia coli (4, 18). Under similar conditions, MAb TCN-2 reacted with a complex pattern of bands in the Silvio strain of T. cruzi trypomastigotes (Fig. 1C), in agreement with previous results, showing that TS belongs to a large protein family (20). Immunofluorescence analysis revealed that the TS-specific MAb TCN-2 did not react with Lv promastigotes while staining paraformaldehyde-fixed (Fig. 1D) and live (data not shown) L1 promastigotes.
Enhanced virulence of TS-expressing L. major promastigotes.
The TS-expressing L1, L2, and L3 promastigotes grew in liquid medium at the same rate as the control Lv promastigotes (data not shown). Likewise, there was no detectable difference in the generation of metacyclic promastigotes in stationary-phase L1 and Lv cultures, as determined by peanut agglutinin binding (data not shown). However, L1 promastigotes, upon inoculation in the footpads of genetically susceptible BALB/c mice, were much more effective than Lv promastigotes in producing local swelling (Fig. 2A and C). Most BALB/c mice died 4 to 6 weeks after inoculation with 106 L1 promastigotes, while all mice survived at least 18 weeks after inoculation with the same number of Lv promastigotes (data not shown). In addition, C57BL/6 mice, which are genetically resistant to L. major infection (15), exhibited a cutaneous swelling upon infection with TS-expressing L1 but not control Lv promastigotes (Fig. 3). However, the L1-induced swelling in the C57BL/6 mice was transient, contrary to the infection in the susceptible BALB/c mice (Fig. 2 and 3). This is similar to the lesion progression typically seen in resistant mice except that the degree of swelling was much greater. The tissues in the L1-inoculated sites of BALB/c mice had TS activity weeks after parasite inoculation, while the tissues of Lv-inoculated sites did not (Fig. 2B). This indicates that L1 parasites express TS in vivo.
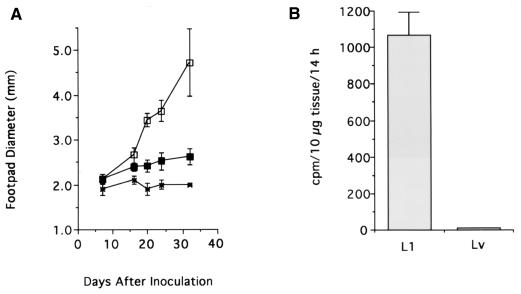
FIG. 2.
Course of infection in BALB/c mice inoculated with L1 and Lv clones of L. major. (A) Five BALB/c mice were inoculated in the right hind footpad with 106 stationary-phase (14 days in culture) L1 (□) or Lv (■) promastigotes. Lesion size was determined weekly. The size of the uninfected footpad (×) of L1-infected mice is shown for comparison. (B) TS activity in tissues harvested from L1- and Lv-infected footpads. (C) Lesions in the hind legs produced by infection with L1 (left mouse) and Lv (right mouse) 5 weeks after parasite inoculation. The right footpad of the L1-infected mouse is much more swollen than the right footpad of the Lv-infected mouse and his own left footpad.
FIG. 3.
Course of infection in BALB/c mice and C57BL/6 mice inoculated with L1 (□) and Lv (■) clones of L. major. Four C57BL/6 (B) and BALB/c (A) mice were inoculated in the right footpad with 5 × 107 stationary-phase (10 days in culture) promastigotes. Lesion size was determined as indicated in the text.
To further ascertain the ability of TS to stimulate L. major growth in vivo, we quantitated the parasites in lesions by limiting dilution 4 weeks after inoculation of 1,000,000 promastigotes in the footpads of BALB/c mice. Lesions of mice infected with L1 parasites contained a titer of log 9.4 ± 2.1 parasites, compared with a titer of log 2.8 ± 1.1 parasites from the lesions produced by Lv promastigotes.
This difference was confirmed by histologic and electron microscopic analysis of the lesions. Toluidine blue-stained paraffin sections of Lv-infected footpads at 4 weeks postinoculation of 106 promastigotes revealed only a few clusters of macrophages (Fig. 4A), some of which were vacuolated and contained a few L. major amastigotes (Fig. 4C). In contrast, the corresponding sections of L1-infected footpads revealed massive infiltration of macrophages (Fig. 4B), extending from the periosteum to just below the epidermis (data not shown). The macrophages in the TS-expressing L1-infected sites were relatively large and held many L. major amastigotes intracellularly (Fig. 4D). In agreement with the histologic examination, electron microscopic evaluation of Lv-infected tissues revealed few amastigotes in the infiltrating macrophages (Fig. 4E), whereas macrophages in the L1-infected sites were packed with parasites (Fig. 4F). Furthermore, some of the macrophages in the L1-infected site were ruptured and releasing amastigotes into the extracellular space (Fig. 4F).
FIG. 4.
Microscopic examination of lesions produced by L1 and Lv clones of L. major in BALB/c mice. (A and B) Photomicrograph of right hind legs infected with the Lv (A) and L1 (B) clones of L. major. Note mild infiltration of macrophages in the Lv-infected site (A), compared with the massive infiltration of large macrophages in the L1-infected site (B). Magnification, ×224. (C and D) Close-up view of panels A and B, respectively. Note that Lv-infected tissue contains few macrophages, some with cytoplasmic vacuoles (C), while L1-infected tissue contains many macrophages filled with Leishmania parasites and some amastigotes in the extracellular space between the macrophages (D). (E) Electron micrograph of the right footpad of an Lv-infected mouse, demonstrating a blood vessel (BV) and several macrophages, six of which contain a few parasites. Magnification, ×3,240. (F) Electron micrograph of the right footpad of an L1-infected mouse, showing several macrophages packed with parasites (asterisks), one of which is ruptured and releasing parasites (arrowhead). Magnification, ×3,240. The micrographs were taken from mice inoculated with 106 promastigotes 5 weeks prior to processing tissue specimens.
The infection of both L1- and Lv-infected BALB/c mice appeared to be largely restricted to the inoculation site, as no evidence of parasite infection was observed in histologic sections of the liver, spleen, and kidneys (data not shown). Consistent with this observation, TS activity was not detected in the liver, spleen, or kidneys of L1-infected BALB/c mice (data not shown).
Despite the robust growth of L1 at the inoculation site in BALB/c mice, these parasites were found only in macrophages and not in other cell types. This was consistent with our inability to infect cultured fibroblasts (Vero cells) and epithelial cells (mink lung epithelial cells) with L1 promastigotes. L1 promastigotes readily infected the mouse macrophage cell line RAW 267.7 in vitro, but there was no significant difference in the infection rates of L1 and Lv promastigotes (data not shown). Thus, TS expression in L. major does not appear to alter the macrophage-specific tropism of the Leishmania parasites or to enhance parasite binding to and invasion of macrophages.
Direct correlation of TS expression in L. major with enhancement of virulence.
To directly implicate TS in the enhancement of virulence, we took advantage of the instability of episomal expression constructs such as pXG1a in the absence of continued drug pressure for maintenance of the selective marker (2, 12). This allowed us to generate segregants from the highly virulent TS-expressing L1 line, which now lacked TS. First, a series of clonal lines were obtained in the absence of selective drug (G418 for the neo marker of pXG1a) from amastigotes derived from the prominent lesions of L1-infected BALB/c mice. Of the 15 lines examined, 14 had lost the pXG-TS plasmid and were now G418 sensitive, while 1 retained G418 resistance (line L1G5). Accordingly, lysates of L1G5 promastigotes expressed TS at levels similar to that of parental L1 promastigotes (Fig. 5A), while the other 14 G418-sensitive subclones, such as L1D4, did not have detectable TS activity (Fig. 5A).
FIG. 5.
Correlation of TS expression with virulence of transgenic L. major. L1G5 and L1D4 are subclones isolated by limiting dilution from a BALB/c lesion produced by infection with clone L1. (A) TS activity of parental L1 clone, subclones L1G5 and L1D4, and subclone L1D4 retransfected with TS-bearing plasmid pXG1a (L1D4-TS) and empty vector (L1D4-vector). (B) Time course of infection in BALB/c mice inoculated with subclones L1G5 and L1D4. (C) Time course of infection in BALB/c mice inoculated with subclones L1D4-TS and L1D4-vector. P.I., postinoculation.
Consistent with the results in Fig. 2 to 4, subclone L1G5, which expressed TS, exhibited the enhanced virulence phenotype in BALB/c mice, whereas its sibling L1D4, which did not express TS, showed only the normal lesion progression (Fig. 5A and B). To confirm that TS expression in Leishmania was responsible, we reintroduced the TS gene into the L1D4 promastigotes by transfection with pXG-TS, generating the L1D4-TS clonal line. As with parental L1, the retransfected promastigotes now expressed TS, while L1D4 parasites transfected with empty vector did not (Fig. 5A). Correspondingly, reexpression of TS in the otherwise low-virulence L1D4 subclone restored the enhanced virulence intrinsic to parental L1 promastigotes. This established TS as the virulence factor responsible for the enhanced virulence of the TS-expressing L. major parasites.
DISCUSSION
TS expressed by L. major is located either in the parasites, most likely on the outer membrane (Fig. 1D), or in the extracellular milieu as a soluble factor (Fig. 1B). Such location is similar to that of endogenous TS of T. cruzi, which is anchored to the trypomastigote plasma membrane through a GPI linkage (18, 23) and thus is readily shed into the extracellular environment. It is likely that ectopic TS also attaches to the L. major surface by a GPI anchor, because the TS gene introduced into the promastigotes contains a predicted GPI anchor motif. Consistent with this expectation, addition of exogenous GPI-specific lipase to L1 promastigotes increased the release of TS (M. Herrera and M. A. Pereira, unpublished observations). Thus, like the endogenous TS of T. cruzi, heterologously expressed TS is strategically located for a possible role in modifying the outcome of L. major interactions with a host.
Earlier results showed that surface-bound TS mediates attachment of T. cruzi trypomastigotes to epithelial and fibroblast host cells (14, 28). However, TS-expressing Leishmania cells were not better than control promastigotes in invading macrophages, at least the mouse macrophage cell line RAW 264.7. In addition, TS-expressing promastigotes did not bind to cultured cells that are not normally invaded by Leishmania, specifically epithelial cells (mink lung epithelial cells) and fibroblasts (Vero cells) (M. B. Carrillo and M. A. Pereira, unpublished observations). Similarly, L. major was not detected in any cell type other than macrophages of L1-infected mice. These results suggest that ectopic TS does not override the molecular mechanisms underlying direct L. major binding to and invasion of target host cells.
Remarkably, ectopic TS was highly effective in boosting the virulence of the transgenic parasites for BALB/c mice, as detected by increased lesion size and elevated parasite numbers therein (Fig. 2, 4, and 5). This was shown genetically in two ways. First, TS-expressing L1 promastigotes induced greater cutaneous lesions than control vector-transfected Lv promastigotes (Fig. 2), ruling out transfection-associated effects. Second, segregants from L1 parasites either retained or lost pXG-TS and TS expression: those that retained TS expression maintained their enhanced virulence in BALB/c mice, while those that lost the plasmid and TS expression lost the enhanced-virulence phenotype (Fig. 5A and B). As expected, reintroduction of pXG-TS into the Leishmania parasites restored TS expression (Fig. 5A) concurrently with the enhanced virulence of parental L1 promastigotes (Fig. 5C). Thus, these data show that expression of TS, and only expression of TS, is the cause for the enhanced-virulence phenotype. In this regard, TS expression satisfies the “molecular” Koch's postulates for establishment as a virulence factor (1, 6).
The TS-provoked enhancement of virulence of L. major supports earlier findings showing that mice sensitized with TS become highly susceptible to T. cruzi infection (3). The timing (1 to 2 h prior to T. cruzi inoculation) and low dose of TS priming and the reduced enhancement of virulence in mice with functionally deficient lymphocytes suggest that TS augmented T. cruzi virulence by subverting the host immune system. Such a hypothesis could also explain the augmented virulence of TS-expressing L. major reported here, particularly because TS does not seem to play a role in Leishmania invasion of macrophages in vitro.
How might TS sabotage the mouse immune system to allow greater expansion of L. major? One provocative possibility is for TS to trigger cytokine release in normal cells independent of B- and T-cell receptor activation, which should upset the balance of normal immune response patterns. It has been established that the susceptibility of BALB/c mice to L. major is determined principally by an upregulation of the anti-inflammatory cytokine interleukin-4 (IL-4) and a downregulation of the proinflammatory cytokine gamma interferon (IFN-γ) (13). Thus, in theory, TS could enhance L. major infection in BALB/c mice by further upregulating interleukin-4 and downregulating IFN-γ in mice infected with L1 promastigotes relative to mice infected with Lv parasites.
This hypothesis could indeed be the case, because we found that the level of IL-4 but not IFN-γ secreted by splenocytes from L1-infected mice in response to TS was about 10 times higher than the IL-4 level secreted by splenocytes from Lv-infected mice (W. Gao and M. A. Pereira, unpublished results). Thus, the greater virulence of L1 parasites relative to the Lv counterparts could be due to enhanced TS-driven Il-4 secretion. The interleukin-secretory power of TS was recently demonstrated in normal human cells, specifically intestinal microvascular endothelial cells and peripheral blood mononuclear cells, which secrete IL-6 in response to TS (24). A further link between TS and cytokine pathways is evident by the cooperation of the trypanosomal enzyme with ciliary neurotrophic factor and leukemia inhibitory factor, two IL-6 family members, to promote survival of neuronal cells under conditions in which the cells would otherwise die of apoptosis induced by growth factor starvation (5).
It is therefore possible that the power of TS to induce multiple cytokines in naive and immune cells and to synergize with bona fide cytokines should serve to undermine innate and/or acquired immune responses and allow the parasite to thrive in the mammalian host. However, it remains to be determined whether L1 organisms will alter the normal immune response to the leishmanial parasites. Because L. major elicits a well-characterized immune response (11, 22), infection of mice with TS-expressing L. major should prove useful to test the new concept that TS is able to sabotage the immune system to promote T. cruzi growth in vivo (3, 24).
ACKNOWLEDGMENTS
We thank David Russell for discussions, Julia Guzova for performing several DNA transfections, and Lynne Garrity for participating in the initial experiments.
This work was supported by NIH grants AI 18102 (to M.A.P.) and AI 29646 (to S.M.B.).
REFERENCES
- 1.Beverley S M. Hijacking the cell: parasites in the driver's seat. Cell. 1996;78:787–789. doi: 10.1016/s0092-8674(00)81984-4. [DOI] [PubMed] [Google Scholar]
- 2.Chakkalath H R, Siddiqui A A, Shankar A H, Dobson D E, Beverley S M, Titus R G. Priming of a β-galactosidase (βGAL)-specific type 1 response in BALB/c mice infected with βGAL-transfected Leishmania major. Infect Immun. 2000;68:809–814. doi: 10.1128/iai.68.2.809-814.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chuenkova M, Pereira M E A. Trypanosoma cruzi trans-sialidase: enhancement of virulence in a murine model of Chagas' disease. J Exp Med. 1995;181:1693–1703. doi: 10.1084/jem.181.5.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chuenkova M, Pereira M, Taylor G. trans-Sialidase of Trypanosoma cruzi: location of galactose-binding site(s) Biochem Biophys Res Commun. 1999;262:549–556. doi: 10.1006/bbrc.1999.1154. [DOI] [PubMed] [Google Scholar]
- 5.Chuenkova M, Pereira M A. A trypanosomal protein synergizes with the cytokines CNTF and LIF to prevent apoptosis in neuronal cells. 2000. Mol. Biol. Cell, in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Falkow S. Molecular Koch's postulates applied to microbial pathogenicity. Rev Infect Dis. 1988;10:S274–S276. doi: 10.1093/cid/10.supplement_2.s274. [DOI] [PubMed] [Google Scholar]
- 7.Ha D S, Schwarz J K, Turco S J, Beverley S M. Use of the green fluorescent protein as a marker in transfected Leishmania. Mol Biochem Parasitol. 1996;77:57–64. doi: 10.1016/0166-6851(96)02580-7. [DOI] [PubMed] [Google Scholar]
- 8.Hall B F, Webster P, Ma A K, Joiner K A, Andrews N W. Desialylation of lysosomal membrane glycoproteins by Trypanosoma cruzi: a role for the surface neuraminidase in facilitating parasite entry into the host cell cytoplasm. J Exp Med. 1992;176:313–325. doi: 10.1084/jem.176.2.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hamer D H, Ward H, Tzipori S, Pereira M E, Alroy J P, Keusch G T. Attachment of Cryptosporidium parvum sporozoites to MDCK cells in vitro. Infect Immun. 1994;62:2208–2213. doi: 10.1128/iai.62.6.2208-2213.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kapler G M, Coburn C C, Beverley S M. Stable transfection of the human parasite Leishmania delineates a 30-kb region sufficient for extrachromosomal replication and expression. Mol Cell Biol. 1990;10:1084–1094. doi: 10.1128/mcb.10.3.1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Launois P, Tacchini-Cottier F, Parra-Lopez C, Louis J A. Cytokines in parasitic diseases: the example of cutaneous leishmaniasis. Int Rev Immunol. 1988;17:157–180. doi: 10.3109/08830189809084491. [DOI] [PubMed] [Google Scholar]
- 12.LeBowitz J H, Coburn C M, McMahon-Pratt D, Beverley S M. Development of a stable Leishmania expression vector and application to the study of parasite surface antigen genes. Proc Natl Acad Sci USA. 1990;87:9736–9740. doi: 10.1073/pnas.87.24.9736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Locksley R M, Pingel S, Lacy D, Wakil A E, Bix M, Fowell D J. Susceptibility to infectious diseases: Leishmania as a paradigm. J Infect Dis. 1999;179:S305–S308. doi: 10.1086/513843. [DOI] [PubMed] [Google Scholar]
- 14.Ming M, Chuenkova M, Ortega-Barria E, Pereira M E A. Mediation of Trypanosoma cruzi invasion by sialic acid on the host cell and trans-sialidase on the trypanosomes. Mol Biochem Parasitol. 1993;59:243–252. doi: 10.1016/0166-6851(93)90222-j. [DOI] [PubMed] [Google Scholar]
- 15.Mitchell G F. Murine cutaneous leishmaniasis: resistance in reconstituted nude mice and several F1 hybrids infected with Leishmania tropica major. J Immunogenet. 1983;10:395–412. doi: 10.1111/j.1744-313x.1983.tb00351.x. [DOI] [PubMed] [Google Scholar]
- 16.Parodi A J, Pollevick G D, Mautner M, Buschiazzo A, Sanchez D O, Frasch A C C. Identification of the gene(s) coding for the trans-sialidase of Trypanosoma cruzi. EMBO J. 1992;11:1705–1710. doi: 10.1002/j.1460-2075.1992.tb05221.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pereira M E A. A developmentally regulated neuraminidase activity in Trypanosoma cruzi. Science. 1983;219:1444–1447. doi: 10.1126/science.6338592. [DOI] [PubMed] [Google Scholar]
- 18.Pereira M E A, Mejia J S, Ortega-Barria E, Matzilevich D, Prioli R P. The Trypanosoma cruzi neuraminidase contains sequences similar to bacterial neuraminidases, YWTD repeats of the low-density lipoprotein receptor, and type III modules of fibronectin. J Exp Med. 1991;174:179–191. doi: 10.1084/jem.174.1.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pereira M E A, Zhang K, Gong Y, Herrera E M, Ming M. Invasive phenotype of Trypanosoma cruzi restricted to a population expressing trans-sialidase. Infect Immun. 1996;64:3884–3892. doi: 10.1128/iai.64.9.3884-3892.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Prioli R P, Mejia J S, Pereira M E A. Monoclonal antibodies against Trypanosoma cruzi neuraminidase reveal enzyme polymorphism, recognize a subset of trypomastigotes, and enhance infection in vitro. J Immunol. 1990;144:4384–4391. [PubMed] [Google Scholar]
- 21.Prioli R P, Ortega-Barria E, Mejia J S, Pereira M E A. Mapping of a B-cell epitope present in the neuraminidase of Trypanosoma cruzi. Mol Biochem Parasitol. 1992;52:85–96. doi: 10.1016/0166-6851(92)90038-l. [DOI] [PubMed] [Google Scholar]
- 22.Reiner S L, Locksley R M. The regulation of immunity to Leishmania major. Annu Rev Immunol. 1995;13:151–177. doi: 10.1146/annurev.iy.13.040195.001055. [DOI] [PubMed] [Google Scholar]
- 23.Rosenberg I, Prioli R P, Ortega-Barria E, Pereira M E A. Stage-specific phospholipases C mediated release of Trypanosoma cruzi neuraminidase. Mol Biochem Parasitol. 1991;46:303–306. doi: 10.1016/0166-6851(91)90054-a. [DOI] [PubMed] [Google Scholar]
- 24.Saavedra E, Herrera M, Gao W, Uemura H, Pereira M A. The Trypanosoma cruzi trans-sialidase, through its COOH-terminal tandem repeat, up-regulates interleukin-6 secretion in normal human intestinal microvascular endothelial cells and peripheral blood mononuclear cells. J Exp Med. 1999;190:1825–1836. doi: 10.1084/jem.190.12.1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sacks D, Perkins P V. Identification of an infective stage of Leishmania promastigotes. Science. 1984;223:1417–1419. doi: 10.1126/science.6701528. [DOI] [PubMed] [Google Scholar]
- 26.Schenkman S L, de Carvalho P, Nussenzweig V. Trypanosoma cruzi trans-sialidase and neuraminidase activities can be mediated by the same enzyme. J Exp Med. 1992;175:567–575. doi: 10.1084/jem.175.2.567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schenkman S, Eichinger D, Pereira M E A, Nussenzweig V. Structural and functional properties of trypanosoma trans-sialidase. Annu Rev Microbiol. 1994;48:499–532. doi: 10.1146/annurev.mi.48.100194.002435. [DOI] [PubMed] [Google Scholar]
- 28.Schenkman R P, Vandekerckhove F, Schenkman S. Mammalian cell sialic acid enhances Trypanosoma cruzi invasion. Infect Immun. 1993;61:898–902. doi: 10.1128/iai.61.3.898-902.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Scudder P, Doom J P, Chuenkova M, Manger I D, Pereira M E A. Enzymatic characterization of the β-d-galactoside α2,3-trans-sialidase from Trypanosoma cruzi. J Biol Chem. 1993;268:9886–9891. [PubMed] [Google Scholar]
- 30.Titus R G, Gueiros-Filho F J, de Freitas L A R, Beverley S M. Development of safe live Leishmania vaccine lines by gene replacement. Proc Natl Acad Sci USA. 1995;92:10267–10271. doi: 10.1073/pnas.92.22.10267. [DOI] [PMC free article] [PubMed] [Google Scholar]