Abstract
Emphysematous gastritis and gastric emphysema are different diseases. Sometimes, we treat the diseases without distinguishing them clearly because both are rare, and the mortality rate of emphysematous gastritis cases is high (55%). Gastric emphysema is more well known than is emphysematous gastritis after percutaneous endoscopic gastrostomy (PEG) placement (80%). Particularly, it is a self-healing disease, and treatment with antibiotics is not required. CT is commonly used to diagnose emphysematous gastritis and gastric emphysema. The amount of radiation exposure is a concern for performing multiple CTs following air disappearance in the gastric wall. Here, we report the case of a 92-year-old man with gastric emphysema after PEG. It was useful to follow-up the patient by performing radiographic examination, and the disease was managed conservatively without antibiotic administration. We report that distinguishing gastric emphysema from emphysematous gastritis was necessary. Moreover, performance excessive tests and treatments should be avoided.
Keywords: Air leaks, Gas/Free Gas, Endoscopy, Stomach and duodenum
Background
It is important to distinguish emphysematous gastritis from gastric emphysema because they have different treatments.1 Emphysematous gastritis is infectious and could cause ischaemic gastric mucosa or sepsis.1 2 However, gastric emphysema is not infectious as it is an air invasion into the gastric wall. It is a trend to avoid unnecessary treatment, such as antibiotic administration use and surgery, because gastric emphysema is a self-healing benign disease.1 Gastric emphysema is often caused by iatrogenic accidents (ie, endoscopy, a gastric tube and percutaneous endoscopic gastrostomy (PEG) placement).1 3 Some cases are overtreated with antibiotics and pressure-reducing tubes without clarifying whether it is emphysematous gastritis or gastric emphysema because only a few physicians are experienced in the treatment of the latter disease, and the mortality rate of emphysematous gastritis is high (55%).2
CT is the effective diagnosis modality.1 It may be inappropriate to use multiple CTs to follow the air disappearance in the gastric wall, considering the amount of radiograph exposure. Plain radiography is also useful for detecting air in the gastric wall and is often used in paediatrics.4
Based on these findings, we concluded that minimum tests and treatments are required for cases of gastric emphysema after PEG. We report a case of gastric emphysema after PEG where radiography was used to follow-up the patient and self-healing with conservative management was provided.
Case presentation
Our case was of an elderly man in his 90s residing in a nursing home owing to high brain dysfunction following an ischaemic cerebrovascular stroke. The patient underwent PEG tube placement for nutrition. After 6 days, he underwent re-fixation surgery secondary to bleeding at the PEG site. The tube was cleared for use immediately post re-fixation with no further bleeding. Over the next 3 days, the patient had vomiting episodes, his temperature rose to 38.3°C and oxygen saturation declined. He presented to our hospital with aspiration pneumonia.
Investigations
On admission, his temperature, oxygen saturation, white cell count and lactic acid level were 37.7°C, 93% (room air), 12 500 cells/µL and 2.1 mmol/L, respectively. On physical examination, coarse crackles were heard from the bilateral lower lobe of the lung. Chest radiographic and CT examination findings showed a shadow infiltrating the bilateral lower lobe of the lung and significant gastric emphysema (figures 1 and 2).
Figure 1.
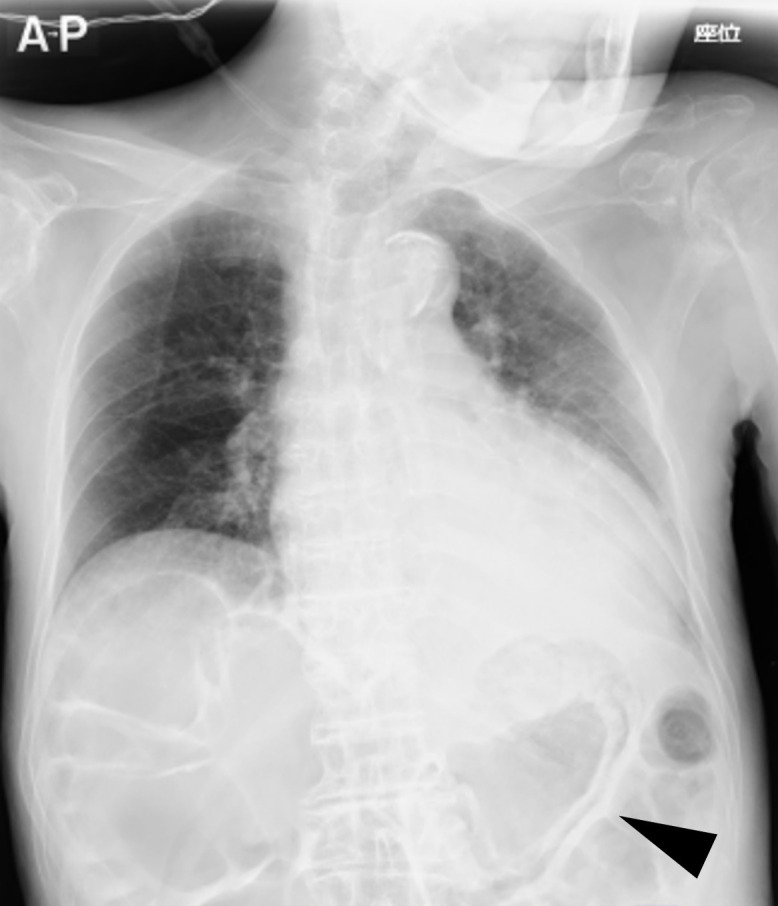
Anteroposterior chest radiograph showing the presence of air in the gastric wall (arrow), and distended intestine.
Figure 2.
Abdominal CT scan showing intramural air in the gastric wall (arrow) and a PEG tube. CT: cut (axial) and viewing window (abdominal). PEG, percutaneous endoscopic gastrostomy.
Differential diagnosis
His vital signs were stable and work-up did not show evidence of sepsis or ischaemia favouring gastric emphysema over emphysematous gastritis. Aspiration pneumonitis was likely based on his history as well as the clinical and radiological findings.
Treatment
We discontinued the use of the PEG tube; no other special treatment was provided, such as antibiotic administration and drainage tubes.
Outcome and follow-up
Gastric emphysema disappeared at 3 days posthospitalisation (figure 3). The PEG tube was re-used, and the patient was discharged thereafter.
Figure 3.
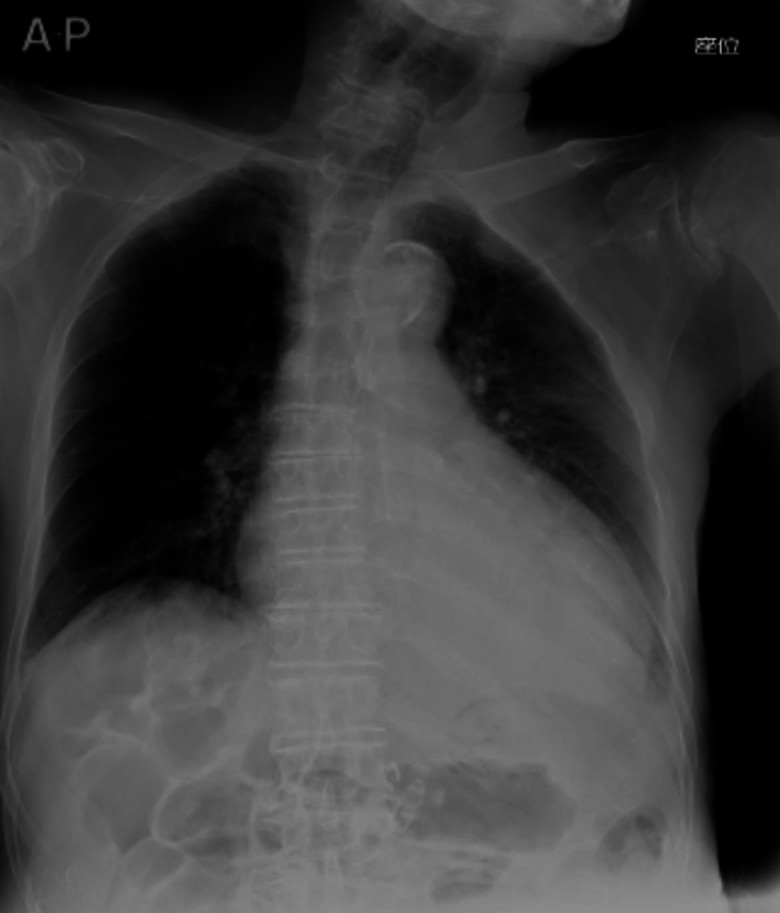
Anteroposterior chest radiograph showing that gastric emphysema disappeared at 3 days posthospitalisation.
Discussion
We suggest that radiography is useful to follow-up on air disappearance in patients with gastric emphysema after PEG.
Emphysematous gastritis and gastric emphysema are different diseases. The distinction of these diseases may be confusing,1 but it is mandatory because their treatments are different.2 Emphysematous gastritis is caused by the production of gas in the gastric wall by infection, whereas gastric emphysema is an invasion of air into the gastric wall by physical stimulation, not infection.1 Gastric emphysema is a self-healing disease with conservative management, but its prevalence is unknown owing to its rarity1 and insufficient data. Therefore, careful treatment and follow-up are necessary.2
Symptomatic air in the gastric wall in patients with PEG whose vitals were stable and had not progressed to septic shock was 80% gastric emphysema.5 It has been reported that the incidence of air in the gastric wall is observed in approximately 20%–50% of patients with PEG,6 7 but almost all the incidences are asymptomatic and self-healing.6 PEG placement is generally a safe procedure when performed by experienced physicians.8
In this case, we considered that the re-fixation surgery caused gastric wall air invasion, similar to buried bumper syndrome,9 resulting in gastric emphysema and vomiting. Fever and hypoxia after vomit are transient and, therefore, we did not have to treat them with antibiotics (aspiration pneumonitis). If his vomit was caused by aspiration pneumonia, we would need to treat him with antibiotics.10
CT is often used to diagnose emphysematous gastritis and gastric emphysema.2 We could detect small amounts of gas in the stomach wall.11 12 Nevertheless, we suggest that it is more appropriate to use radiography than it is to use CT in follow-up because of the amount of radiation exposure. The diagnostic sensitivity of radiograph is 72.2%, which is not very high,2 but we could perform follow-up examination by radiography if we had detected air in the gastric wall during diagnosis. The disappearance of air in the gastric wall takes 2–14 days, and gastric emphysema is a self-healing disease.6 13 However, the mortality rate is high (29%).2 Therefore, we must treat it safely and cautiously. It is safe to re-start using a PEG tube after confirmation of the disappearance of air in the gastric wall because endoscopy could identify an inflamed, erosive mucosa with necrosis of the stomach wall (emphysematous gastritis: 48.7%; gastric emphysema: 44.4%).2 Thus, it is important to follow the reduction or disappearance of air by radiography.
We hope that our case will help treat gastric emphysema cases after PEG by performing minimum and low-invasive treatment procedures.
Patient’s perspective
As a patient, I hope that the case will be useful for future medical care and helpful to someone in the world (Japanese to English, translated by Corresponding Author).
Learning points
Emphysematous gastritis and gastric emphysema are different diseases.
Symptomatic air in the gastric wall after percutaneous endoscopic gastrostomy indicates gastric emphysema, not emphysematous gastritis.
Radiography can be helpful in the follow-up of gastric emphysema cases.
Footnotes
Contributors: SM and HM cared for the patient and wrote the manuscript. TW revised the manuscript and supervised every process.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Case reports provide a valuable learning resource for the scientific community and can indicate areas of interest for future research. They should not be used in isolation to guide treatment choices or public health policy.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Consent obtained from parent(s)/guardian(s).
References
- 1.Shami A, Singhania R. Emphysematous gastritis: a case series and review of the current trend favoring conservative management. Clin J Gastroenterol 2021;14:1042–5. 10.1007/s12328-021-01425-3 [DOI] [PubMed] [Google Scholar]
- 2.Matsushima K, Won EJ, Tangel MR, et al. Emphysematous gastritis and gastric emphysema: similar radiographic findings, distinct clinical entities. World J Surg 2015;39:1008–17. 10.1007/s00268-014-2882-7 [DOI] [PubMed] [Google Scholar]
- 3.Iwamuro M, Abe M, Kono Y, et al. [Clinical characteristics of five patients with gastric emphysema]. Nihon Shokakibyo Gakkai Zasshi 2021;118:851–8. 10.11405/nisshoshi.118.851 [DOI] [PubMed] [Google Scholar]
- 4.Wettasinghe MC, Rosairo S, Kiriwattuduwa S, et al. Gastroduodenal emphysema with portal venous air due to congenital duodenal web in a child: a case report and review of literature. Case Rep Pediatr 2020;2020:1–5. 10.1155/2020/9897208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Iwamuro M, Okamoto Y, Kanzaki H, et al. [Conservative management of gastric emphysema and hepatic portal venous gas: a case report]. Nihon Shokakibyo Gakkai Zasshi 2018;115:655–61. 10.11405/nisshoshi.115.655 [DOI] [PubMed] [Google Scholar]
- 6.Wiesen AJ, Sideridis K, Fernandes A, et al. True incidence and clinical significance of pneumoperitoneum after PEG placement: a prospective study. Gastrointest Endosc 2006;64:886–9. 10.1016/j.gie.2006.06.088 [DOI] [PubMed] [Google Scholar]
- 7.Schrag SP, Sharma R, Jaik NP, et al. Complications related to percutaneous endoscopic gastrostomy (PEG) tubes. A comprehensive clinical review. J Gastrointestin Liver Dis 2007;16:407–18. [PubMed] [Google Scholar]
- 8.Ferraro F, Gravina AG, d'Elia A, et al. Percutaneous endoscopic gastrostomy for critically ill patients in a general intensive care unit. Acta Gastroenterol Belg 2013;76:306–10. [PubMed] [Google Scholar]
- 9.Gravina AG, Tessitore A, Ormando VM, et al. May chronic cough in chronic obstructive pulmonary disease be a contraindication of percutaneous endoscopic gastrostomy placement: a case report. BMC Gastroenterol 2021;21:31. 10.1186/s12876-021-01603-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Marik PE. Aspiration pneumonitis and aspiration pneumonia. N Engl J Med 2001;344:665–71. 10.1056/NEJM200103013440908 [DOI] [PubMed] [Google Scholar]
- 11.Loi T-H, See J-Y, Diddapur RK. Emphysematous gastritis: a case report and a review of literature. Ann Acad Med Singap 2007;36:72–3. [PubMed] [Google Scholar]
- 12.Shipman PJ, Drury P. Emphysematous gastritis: case report and literature review. Australas Radiol 2001;45:64–6. 10.1046/j.1440-1673.2001.00878.x [DOI] [PubMed] [Google Scholar]
- 13.Sawano T, Nemoto T, Tsubokura M, et al. Asymptomatic hepatic portal venous gas with gastric emphysema as a chronic complication of gastrostomy tube placement: a case report. J Med Case Rep 2016;10:234. 10.1186/s13256-016-1037-x [DOI] [PMC free article] [PubMed] [Google Scholar]



