Abstract
Pityrosporum folliculitis (PF) is a fungal acneiform disease of the hair follicles that often presents with pruritic papules and pustules on the upper body and face. This condition is commonly mistaken for acne vulgaris and can be distinguished from bacterial acne by the presence of fungal spores in the follicular lumen. Although studies have been performed to describe PF in cohorts, little work has been done to aggregate these data. Thus, the goal of this review is to describe the clinical characteristics and treatment outcomes of PF in immunocompetent patients. PubMed, Web of Science, and Embase were searched using the terms “Pityrosporum folliculitis” or “Malassezia folliculitis.” All cohorts reporting PF characteristics in patients classified as immunocompetent were reviewed. A total of 15 studies were included. Majority of patients were male (64%) with the average age of presentation of 24.26 years. The most common locations of lesions were the chest (70%) and back/shoulders (69.2%). Pruritus was reported by the majority of patients (71.7%). Additionally, 40.5% of patients reported a history of unsuccessful treatment regimens. Treatment was most successful with an oral antifungal (92%), followed by a topical antifungal (81.6%). In conclusion, majority of patients with PF were younger males. Many patients were primarily treated incorrectly, suggesting the importance of proper diagnosis. PF may be distinguishable from acne vulgaris by the presence of pruritus or suggested when a new acneiform eruption develops following antibiotic therapy or immunosuppression. When properly diagnosed, majority of cases of PF achieve complete response with oral or topical antifungals.
Keywords: Pityrosporum folliculitis, Malassezia folliculitis, Acne vulgaris, Antifungal, Acneiform eruption
Introduction
Pityrosporum folliculitis (PF) is fungal acneiform disease of the hair follicles that presents with papules and pustules often associated with pruritus [1]. PF lesions most commonly present on the chest, shoulders, back or face most often appear during the second and third decades of life [1]. Although other types of fungal folliculitis exist, over 90% have been associated with Malassezia spp. [2]. The pathogenesis of PF likely involves a primary occlusion event in the hair follicle, followed by an overgrowth of Malassezia spp. leading to an inflammatory cascade causing PF [3].
PF and acne vulgaris have very similar presentations, and PF is often misdiagnosed as acne vulgaris (AV) [1]. Malassezia spp. fungi require a lipid source for survival and reproduction which leads these species to overgrow in sebum-rich areas of the body, where AV often occurs [5]. Both PF and AV can present with papules and pustules and occur in similar locations across the body: face, chest, and back. New acneiform eruptions caused by PF have been associated with immunocompromised states such as post-transplant, HIV/AIDS, and malignancies [6]. This is presumed to be because these immunocompromised states allow the overgrowth of Malassezia spp. Most recently, PF has also been associated with COVID-19 in patients presenting to the hospital [7]. This may be an additional sign that may alert a practitioner to consider a diagnosis of PF rather than AV. In cases where clinicians struggle to distinguish PF and AV, a potassium hydroxide (KOH) smear can be used on lesional scrapings to reveal spores. This test has been shown to be both sensitive and specific with values as high as 84.6% and 100%, respectively [8]. Patients with PF are often incorrectly managed with traditional AV treatments, such as antibiotics, with little to no improvement [5]. Thus, in cases of AV resistant to traditional management strategies, the diagnosis of PF should be considered [4].
Given that PF is unresponsive to AV treatment, it is imperative for clinicians to properly distinguish PF and AV in order to correctly manage these two separate conditions. Although small cohort studies have been performed on patients with PF, little work has been done to aggregate these data to describe how PF in immunocompetent individuals differs in clinical presentation and treatment outcomes when compared to AV. Thus, the goal of this review is to summarize clinical characteristics and treatment response in immunocompetent individuals with PF.
Methods
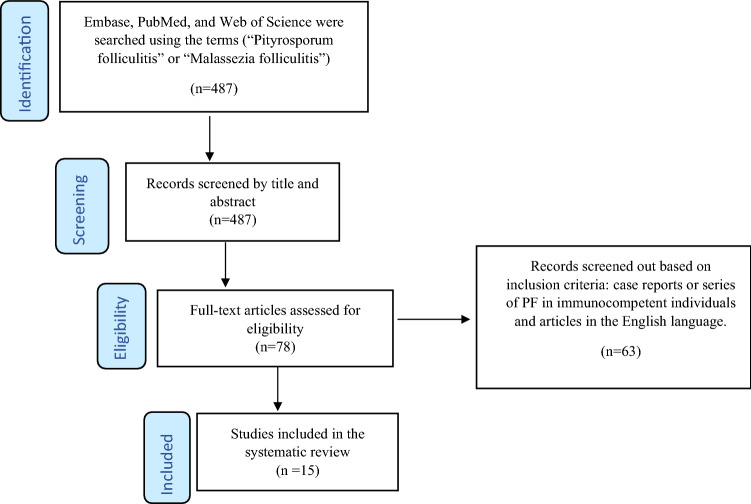
The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) were used to guide methodology and reporting (Fig. 1) [9]. A comprehensive literature search was performed using the databases PubMed, Web of Science and Embase in March of 2022 using the terms “Pityrosporum folliculitis” or “Malassezia folliculitis.” Studies were included if they described clinical characteristics or treatment outcomes in immunocompetent human patients with PF. Patients recently started on oral steroids were considered immunocompetent for the purpose of this review. Studies describing patients with an immunocompromised state were excluded from this review. No geographic or language restrictions were used.
Fig. 1.
Flow diagram of the literature search using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Adapted from http://prisma-statement.org
An initial title and abstract screening was performed on articles collected by the initial search strategy by two researchers (M.G. and A.F.). Then, those articles which passed the initial screening underwent full text review by two researchers (M.G. and A.F.). The data were collected by one researcher (M.G.) and confirmed by two additional researchers (N.K., A.F.). Any discrepancies in process for study selection were settled by a fourth independent researcher (H.M.).
Results
Ultimately, a total of 15 studies met inclusion criteria. Aggregate clinical presentation data and responses to treatment are described in Table 1. Overall, the average age of patients upon presentation was 24.3 years with the majority of patients being male (64.0%). PF lesions occurred most commonly on the chest (70.0%) and shoulders (69.2%) and less commonly on the face (30.9%). These lesions demonstrated pruritus in the majority of cases described across studies (71.7%). Additionally, a large portion of patients across studies had a history of unsuccessful treatment regimen seemingly directed toward another dermatologic diagnosis (40.5%). Finally, treatments using oral antifungals (92.0%), topical antifungals (81.6%), and combination oral and topical antifungals (77.3%) were highly successful.
Table 1.
Presentation and Treatment Response of Pityrosporum Folliculitis in 1238 Patients
| Variable | Result | Sample size | Number of studies |
|---|---|---|---|
| Age | Mean = 24.26yrs | N = 403 | 8 |
| Sex | 64.0%, male | N = 970 | 12 |
| Location | |||
| Chest | 70.0% | N = 720 | 5 |
| Back/shoulders | 69.2% | N = 409 | 5 |
| Forehead | 30.9% | N = 459 | 4 |
| Presence of pruritus | 71.7% | N = 540 | 7 |
| History of unsuccessful treatment regimen (Antibiotics, steroids, etc.) | 40.5% | N = 802 | 10 |
| Treatment | |||
| Oral antifungal | 92.0% improvement | N = 547 | 8 |
| Topical antifungal | 81.6% improvement | N = 310 | 7 |
| Combination oral + topical | 77.3% improvement | N = 78 | 4 |
Prindaville et al. (2018) found that 60/60 (100.0%) of their patients improved with topical antifungals and that 45/60 (75.0%) had been treated with an oral antibiotic previously [10]. Levy et al. (2007) also found that almost 65% of the 26 patients in their study had previously been diagnosed and treated with AV [11]. Purnak et al. (2018) used a prospective cohort design of 217 patients that PF was significantly more common in summer versus winter months [12].
The relationship of steroid acne to PF was reviewed Yu et al. (1998) which found that 26/34 (76.5%) patients with suspected steroid acne showed Malessezia fungi present on biopsy [13]. Ran et al. (1988) found that AV commonly presented alongside PF in 4/7 (57.1%) patients and often had to be managed simultaneously [14]. Ayers, Sweeney and Wiss (2005) also looked at concurrent AV and PF in a cohort of six females with all patients (100.0%) exhibiting pruritus and the majority (83.3%) receiving past treatment with oral antibiotics [15].
Tsai et al. (2018) found atypical presentations of PF in a cohort of 22/94 patients in a retrospective cohort; atypical presentation consisted of less papules, atypical macules/patches in 6/22 (27.3%), or plaques in 4/22 patients (18.2%) [16]. Tsai et al. (2019) found differences in pediatric versus adult PF, with pediatric patients showing significantly more PF on the face (n = 17/79, 21.5% pediatric; n = 28/242, 11.6% adults, p = 0.027); both age groups showed PF more commonly in males and occurring during summer months [17].
Danby (2016) found that treatment with pulsed ketoconazole treatment over 8 weeks was effective in reducing PF lesions in the vast majority (92.1%) of patients in a 151 patient cohort [18]. Suzuki et al. (2016) also researched the effectiveness of treatment and found that 37/37 (100.0%) patients treated with 2% topical ketoconazole for 27 days and 7/7 (100.0%) patients treated with oral itraconazole for 14 days all showed improvement in PF lesions [19]. Parsad, Saini, and Negi (1998) also researched the effectiveness of oral itraconazole versus placebo in a double-blind study and found the majority of those receiving itraconazole showed complete resolution (n = 9/13, 69.2%) with only one of thirteen (7.7%) showing no change in lesions. The majority of patients receiving placebo (n = 8/12, 66.7%) showed no change or worsening of their PF [20].
Lim, Giam and Tan (1987) found in a retrospective cohort of 70 patients that the majority (n = 43/48, 89.6%) were treated with antibiotic therapy for their PF with no improvement in symptoms; additionally, 34/48 (70.8%) patients also cited heat and/or sweating as an aggravating factor for their PF [21]. Yong, Tan and Tan (2021) conducted an additional cohort review on 214 patients in Singapore and found those treated with oral antifungals showed a slightly higher treatment success rate than those receiving topicals (n = 118/128, 92.2% versus n = 66/82, 80.5%, respectively) [22].
Back, Faergemann, and Hornqvist (1985) found that 20/51(39.2%) patients in a cohort previously received an incorrect diagnosis and were therefore unsuccessful on previous therapies [23]. Finally, Abdel-Razek et al. (1995) found in a clinical trial model that oral ketoconazole 200 mg once daily with application of topical 2% ketoconazole (group 1, n = 20) showed clearance of PF lesions in 100.0% of patients, while oral ketoconazole 200 mg once daily (group 2, n = 20) showed clearance in 75.0% of patients [24].
Descriptions of study design and key findings for each of the 15 studies included are summarized in Table 2.
Table 2.
Detailed description of PF studies (n = 15)
| Study characteristics | Patient characteristics | PF information | Treatment and outcomes | Other variables | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Author, year | Study type | Sample size | Age, mean | Sex (majority) | PF diagnosis method | Location | Symptoms | Concomitant AV | Prior failed treatments | Treatment | Treatment outcome | Other variables noted |
| Prindaville et al. (2018) | Retrospective cohort | 110 | 15 | F: n = 76/110, 69% | KOH test (n = 110/110) | Forehead (n = 81/110, 74%), upper back (n = 80/110, 73%) | Pruritus (n = 72/110, 65%) | NR | Previously treated for AV with antibiotics (approximately 75%) | Oral antifungals (n = 26/110); topical antifungals (n = 60/110) |
Oral antifungals: improvement or complete response (n = 25/26, 96%); topical antifungals: improvement or complete response (n = 60/60, 100%) |
NR |
| Levy et al. (2007) | Retrospective cohort | 26 | 46 | M: n = 22/26, 85% |
Microscopy or histology (n = 26/26) |
NR | NR | NR | Previously treated for AV (approximately 65%) | Oral ketoconazole; combination oral and topical ketoconazole (n = NR) | Complete response in approximately 75% of cases | NR |
| Purnak et al. (2018) | Prospective cohort | 55 | 23 | F: n = 42/55, 76% | Tzanck smear (n = 55/55) | Most commonly forehead, cheeks, and trunk | Pruritus (n = 39/55, 71%) | NR | Previously treated for AV (n = 28/38, 74%) | Oral itraconazole and ketoconazole cream (n = 38/55) | 50% or more reduction in lesions (n = 26/38, 68%) | Season: PF was significantly more common in the summer compared to the winter (n = 34/55 vs. n = 21/55, p = 0.001) |
| Yu et al. (1998) | Retrospective cohort | 75 | NR | NR | Twenty-six of 34 (76.5%) patients diagnosed with steroid acne tested positive for Pityrosporum ovale spores in the hair follicles; 19/21 (90.5%) patients diagnosed with PF tested positive for spores, and 3/20 (15%) patients diagnosed with AV tested positive for spores | NR | NR | NR | NR | Oral itraconazole (n = 29/75); oral minocycline (n = 8/75), and isoconazole nitrate or resorcinol/salicylic acid solution (n = 11/75) | Improvement in n = 27/29 (93.1%) with oral itraconazole, n = 4/8 (50%) with oral minocycline, n = 5/11 (46%) with topical isoconazole nitrate or resorcinol/salicylic acid solution | NR |
| Ran et al. (1988) | NR | 7 | 27 | M: n = 6/7, 86% | Histopathology (n = 7/7) | Back (n = 7/7, 100%), chest (n = 6/7, 85.7%) | Pruritus (n = 7/7, 100%) | AV at the time of presentation (n = 4/7, 57%) | NR | NR | NR | No history of steroid or antibiotic use (n = 7/7) |
| Ayers, Sweeney and Wiss (2005) | NR | 6 | NR | F: n = 6/6, 100% | KOH test (n = 6/6) | Most commonly face, forehead and cheeks (n = 5/6, 83.3%) or shoulders and back (n = 4/6, 66.7%) | Pruritus (n = 6/6, 100%) | Concurrent AV (n = 6/6, 100%) | Previously treated for AV with oral antibiotics (n = 5/6, 83.3%) | Oral and topical antifungal combination regimens | Improvement (n = 6/6, 100%) | NR |
| Tsai et al. (2018) | Retrospective cohort | 94 | NR | NR | Histological and periodic acid-Schiff stain (n = 94/94) | Most commonly face and scalp (n = 11/22, 50%), legs (n = 4/22, 18.2%) | NR | NR | NR | NR | NR | Morphology: typical (n = 72/94, 77%) with characteristic papules and pustules, or atypical (n = 22/94, 23%) with significantly less papules, and having atypical macules/patches, and plaques |
| Tsai et al. (2019) | Retrospective cohort | 321 |
Pediatric n = 79/321, 25% Adult: n = 242/321, 75% |
M: n = 57/79, 72.2% | Microscopy or biopsy (n = 321/321) | Most commonly chest (n = 59/79, 74.7% pediatrics; n = 177/242, 73% adults), face (n = 17/79, 22% pediatric; n = 28/242, 12% adults, p = 0.027) | Pruritus (n = 24/79, 30.4% pediatrics; n = 93/242, 38% adults) | NR | History of antibiotic use (n = 17/79, 21.5% pediatrics; n = 60/242, 24.8% adults) |
Pediatric: oral antifungals (n = 60/79, 76%); topical antifungals (n = 19/79, 24%) Adult: oral antifungals (n = 157/242, 65%); topical antifungals (n = 85/242, 35%) |
Pediatric: improvement in n = 41/47 (87%) with oral antifungals; n = 9/9, 100%) with topical antifungals Improvement in n = 119/126 (94%) with oral antifungals and in n = 34/42 (81%) with topical antifungals |
Season: most commonly during summer months (n = 34/79, 43% pediatrics, n = 98/242, 40.5% adults) Effectiveness of direct microscopy with KOH versus histology for PF diagnosis: direct microscopy increased the annual diagnosis of PF |
| Danby (2016) | NR | 151 | NR | NR | Clinically (n = 151/151) | NR | NR | NR | Oral antibiotics or steroids prior to PF presentation (51%) | Oral ketoconazole | Only 12 patients showed no change in the number of lesions after the eight-week treatment, with greater than half showing complete resolution or significant improvement | NR |
| Suzuki et al. (2016) | NR | 44 | 36 | M: n = 35/44, 80% | Methylene blue stain (n = 36/36) | Chest in approximately 60% of patients | Pruritus in approximately 80% of patients | NR | No antibiotic use was reported in any patients, but 8/44 (18.2%) patients reported recent topical steroid use | 2% ketoconazole cream (n = 37/44, 84%), oral itraconazole (n = 7/44, 16%) | Improvement in n = 37/37 (100%) with 2% ketoconazole cream, and in n = 7/7 (100%) with oral itraconazole | NR |
| Parsad, Saini, and Negi (1998) | Double-blind study | 26 | NR | M: n = 16/26, 62% | KOH test (n = 26/26) | NR | NR | NR | NR |
Itraconazole (n = 13/26), placebo 9n = 12/26) |
Complete response in n = 9/13 (69%) and no response in n = 1/13 (8%) with itracanazole. No response or worsening in n = 8/12 (67%) or marked response in n = 1/12 (8%) with placebo | NR |
| Lim, Giam and Tan (1987) | Retrospective cohort | 48 | 22 | M: n = 44/48, 92% | Histology or gram-stain (n = 48/48) | NR | Pruritus (n = 37/48, 77%) | Concurrent AV (n = 9/48, 19%) |
Antibiotic therapy (n = 43/48, 89.6%) Additionally, some patients PF was preceded by antibiotic or corticosteroid use (n = 8/48, 16/7%, n = 1/48, 2.1%) |
NR | NR |
Aggravating factors for PF: n = 34/48 (71%) cited heat and sweating as an aggravating factor Common concomitant diagnoses: seborrhea (n = 48/48, 100%), seborrheic dermatitis (n = 18/48, 38%) |
| Yong, Tan and Tan (2021) | Cohort | 214 | NR | M: n = 162/214, 76% | Gram-stain (n = 214/214) | Most commonly back (n = 137/214, 64%), chest and trunk (n = 123/214, 57.5%) | NR | NR | NR | Oral antifungals (n = 128/214, 60%); or topical antifungals (n = 82/214, 38%) | Complete response in n = 118/128 (92%) with oral antifungals and (n = 66/82, 81%) with topical antifungals | NR |
| Back, Faergemann, and Hornqvist (1985) | NR | 51 | 30 (median) | F: n = 39/51, 77% | Cellophane stripping stained with methylene blue or KOH (n = 51/51) | NR | NR | Concurrent AV (n = 3/51, 6%) | Misdiagnosed and unsuccessfully treated (n = 20/ 51, 39%) | Selenium sulfide shampoo (n = 22/25, 88%), propylene glycol 50% in water (n = 12/12, 100%), or econazole cream (n = 8/10, 80%) | NR | Common concomitant diagnoses: seborrheic dermatitis (n = 9/51, 18%), pityriasis versicolor (n = 8/51, 16%) |
| Abdel-Razek et al. (1995) | Clinical trial | 62 | 22 | F: n = 40/62, 64% | NR | Most commonly trunk (n = 59/62, 95.2%) | Pruritus (n = 59/62, 95%) | Concurrent AV (n = 8/62, 13%), | NR | Oral ketoconazole and topical 2% ketoconazole (n = 20/62), oral ketoconazole (n = 20/62), econazole nitrate 1% solution (n = 12/62), and miconazole nitrate 2% cream (n = 10/62) | Complete response in 100% (n = 20/20) with oral ketoconazole and topical 2% ketoconazole; 75% (n = 15/20) with oral ketoconazole, 0% with econazole nitrate 1% solution and miconazole nitrate 2% cream | Common concomitant diagnoses: tinea versicolor (n = 6/62, 10%) and seborrhea (n = 5/62, 8%) |
Discussion
This study summarizes clinical characteristics and treatment outcomes of 1238 immunocompetent patients with PF leading to several important clinical conclusions.
First, across studies, PF most commonly presented in male patients (64.0%) with an average age of 24.3 years. PF lesions were most commonly located on the chest (70.0%) and back (69.2%), both of which are common presentation sites for AV. These demographics and lesion sites are likely explained by the common increase in sebum production in this group of patients (young adult males) and at these anatomical sites. Sebum production is typically increased in males compared to females and tends to increase during adolescence and young adulthood, leveling off with age [25]. Additionally, pruritus was commonly described in the clinical presentation for PF in this review (71.7%). This is of relevance given that pruritus is an important distinguishing factor when present from AV [1]. The exact pathophysiology of the pruritus in PF is unknown, but it likely stems from the overgrowth of Malassezia spp. in the hair follicle leading to keratinocyte induction of an inflammatory cascade [1].
Another important finding across studies was that patients presenting with PF had commonly failed a previous treatment regimen, most commonly an antibiotic, for a previously misdiagnosed dermatologic condition (40.5%). It is thought that the use of antibiotics may predispose individuals to PF by disturbing the skin microbiome, thus allowing the overgrowth of Malassezia spp. First line antibiotic therapy for AV typically consists of either a macrolide, clindamycin or a tetracycline targeting P. acnes; in doing so, the commensal balance between bacterial and fungal species can be disrupted to cause overgrowth, thus leading to conditions such as PF [26]. However, 59.5% of patients in this study had no described history of antibiotic use or previously failed treatment regimen. With Malassezia spp. being the most common component in the normal skin flora of healthy individuals, much work remains to be done in understanding how PF develops in fully immunocompetent groups.
Additionally, the studies by Purnak et al. (2018) and Tsai et al. (2019) found that PF outbreaks were most common in summer months. This is likely due to increased sweating and subsequent sebum production providing lipids for Malassezia spp. Growth [27]. In addition, studies by Lim, Giam and Tan (1987) and Abdel-Razek et al. (1995) found the most common co-diagnoses with PF to be AV, seborrheic dermatitis and pityriasis versicolor. Malassezia spp. have been associated with AV development, especially the non-inflammatory subtype, and are the fungi responsible for pityriasis versicolor [28]. Although the exact pathogenesis of seborrheic dermatitis is still under investigation, it is known that Malassezia yeast and inflammatory responses play important roles in the disease progression [29]. Thus, patients with these previous dermatologic diagnoses may be of higher suspicion for PF.
There are currently no national guidelines in place for treating Malassezia folliculitis; however, majority of individuals across studies showed improvement in response to either oral antifungals (92.0%), topical antifungals (81.6%), or combination regimens of topical and oral antifungals (77.3%). Oral antifungals have been shown to be slightly more effective than topicals against Malassezia spp. skin conditions [30]. Given the successful response to topical antifungals in PF patients, though, topical antifungals may be recommended as first-line therapy to minimize side effects, such as hepatotoxicity, from oral antifungal therapy [30]. Given these antifungals must be prescribed off label, patients may struggle receiving reimbursement from insurance plans. This is an important health care accessibility barrier that must be considered in dermatology, as the use of off-label medications is becoming commonplace in the standard of care for a wide-range of dermatologic conditions, including PF [31].
There are a number of limitations to this review. First and most importantly, PF is likely highly underreported in the literature due to its common misdiagnosis as AV and ability to self-resolve. Additionally, the severity of disease could not be objectively compared across studies, and the definition of improvement with treatment was subjectively defined in each study. Additionally, in majority of reviewed studies, there was no control group to properly gauge treatment efficacy. Finally, in many studies, approximate percentages were used, and therefore, the exact number of patients with a given clinical history or treatment outcome had to be estimated.
Conclusion
PF is a condition commonly misdiagnosed as AV in the clinical setting. Both conditions consist of papules and pustules commonly occurring on the face, back, and chest most frequently in the second and third decades of life. The correct diagnosis of PF is essential in effectively treating patients, as many are incorrectly managed with oral antibiotics for suspected AV. With correct clinical diagnosis, PF can successfully be managed with topical and/or oral antifungals. The diagnosis of PF should be suspected over AV in patients presenting with pruritus, a history of lesions unresponsive to traditional acne vulgaris treatment, or a new onset acneiform eruption following an immunocompromised state. When in doubt, a KOH mount is highly sensitive and specific for identifying fungal Malassezia spp. in biopsied lesions.
Acknowledgements
None.
Author contributions
MG and Dr. HM worked on study concept and design. MG, AF, and NK worked on data collection, data analysis, and drafting of the manuscript. MG and Dr. HM made final edits to the manuscript.
Funding sources
None.
Data availability
Not applicable.
Declarations
Conflicts of interest
No authors have any conflict of interests to report in regards to this manuscript. Additionally, there were no sources of funding for this review.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Abdel-Razek M, Fadaly G, Abdel-Raheim M, Al-Morsy F. Pityrosporum (Malassezia) folliculitis in Saudi Arabia–diagnosis and therapeutic trials. Clin Exp Dermatol. 1995;20(5):406–409. doi: 10.1111/j.1365-2230.1995.tb01358.x. [DOI] [PubMed] [Google Scholar]
- 2.Ayers K, Sweeney SM, Wiss K. Pityrosporum folliculitis: diagnosis and management in 6 female adolescents with acne vulgaris. Arch Pediatr Adolesc Med. 2005;159(1):64–67. doi: 10.1001/archpedi.159.1.64. [DOI] [PubMed] [Google Scholar]
- 3.Bäck O, Faergemann J, Hörnqvist R. Pityrosporum folliculitis: a common disease of the young and middle-aged. J Am Acad Dermatol. 1985;12(1 Pt 1):56–61. doi: 10.1016/S0190-9622(85)70009-6. [DOI] [PubMed] [Google Scholar]
- 4.Corzo-León DE, MacCallum DM, Munro CA. Host responses in an. Front Cell Infect Microbiol. 2020;10:561382. doi: 10.3389/fcimb.2020.561382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Danby FW (2016) Malassezia infections-Management with pulsed oral ketoconazole. J Am Acad Dermatol 74(5): AB159
- 6.Del Rosso JQ, Silverberg N, Zeichner JA. When acne is not acne. Dermatol Clin. 2016;34(2):225–228. doi: 10.1016/j.det.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 7.Durdu M, Güran M, Kandemir H, Ilkit M, Seyedmousavi S. Clinical and laboratory features of six cases of candida and dermatophyte folliculitis and a review of published studies. Mycopathologia. 2016;181(1–2):97–105. doi: 10.1007/s11046-015-9939-5. [DOI] [PubMed] [Google Scholar]
- 8.Gupta AK, Lyons DC. The rise and fall of oral ketoconazole. J Cutan Med Surg. 2015;19(4):352–357. doi: 10.1177/1203475415574970. [DOI] [PubMed] [Google Scholar]
- 9.Hald M, Arendrup MC, Svejgaard EL, Lindskov R, Foged EK. Danish society of dermatology. Evidence-based danish guidelines for the treatment of malassezia-related skin diseases. Acta Derm Venereol. 2015;95(1):12–19. doi: 10.2340/00015555-1825. [DOI] [PubMed] [Google Scholar]
- 10.Hill MK, Goodfield MJ, Rodgers FG, Crowley JL, Saihan EM. Skin surface electron microscopy in Pityrosporum folliculitis. The role of follicular occlusion in disease and the response to oral ketoconazole. Arch Dermatol. 1990;126(8):1071–1074. doi: 10.1001/archderm.1990.01670320095018. [DOI] [PubMed] [Google Scholar]
- 11.Kim S, Park JW, Yeon Y, Han JY, Kim E. Influence of exposure to summer environments on skin properties. J Eur Acad Dermatol Venereol. 2019;33(11):2192–2196. doi: 10.1111/jdv.15745. [DOI] [PubMed] [Google Scholar]
- 12.Lim KB, Giam YC, Tan T. The epidemiology of Malassezia (Pityrosporon) folliculitis in Singapore. Int J Dermatol. 1987;26(7):438–441. doi: 10.1111/j.1365-4362.1987.tb00586.x. [DOI] [PubMed] [Google Scholar]
- 13.Lévy A, Feuilhade de Chauvin M, Dubertret L, Morel P, Flageul B. Folliculites à Malassezia: Caractéristiques et réponses thérapeutiques chez 26 malades [Malassezia folliculitis: characteristics and therapeutic response in 26 patients] Ann Dermatol Venereol. 2007;134(11):823–828. doi: 10.1016/S0151-9638(07)92824-0. [DOI] [PubMed] [Google Scholar]
- 14.Luebberding S, Krueger N, Kerscher M. Skin physiology in men and women: in vivo evaluation of 300 people including TEWL, SC hydration, sebum content and skin surface pH. Int J Cosmet Sci. 2013;35(5):477–483. doi: 10.1111/ics.12068. [DOI] [PubMed] [Google Scholar]
- 15.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Rev Esp Cardiol (Engl Ed) 2021;74(9):790–799. doi: 10.1016/j.recesp.2021.06.016. [DOI] [PubMed] [Google Scholar]
- 16.Parsad D, Saini R, Negi KS. Short-term treatment of pityrosporum folliculitis: a double blind placebo-controlled study. J Eur Acad Dermatol Venereol. 1998;11(2):188–190. doi: 10.1111/j.1468-3083.1998.tb00781.x. [DOI] [PubMed] [Google Scholar]
- 17.Peres FLX, Bonamigo RR, Bottega GB, Staub FL, Cartell AS, Bakos RM. Pityrosporum folliculitis in critically ill COVID-19 patients. J Eur Acad Dermatol Venereol. 2022;36(3):e186–e188. doi: 10.1111/jdv.17842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Piquero-Casals J, Hexsel D, Mir-Bonafé JF, Rozas-Muñoz E. Topical non-pharmacological treatment for facial seborrheic dermatitis. Dermatol Ther (Heidelb) 2019;9(3):469–477. doi: 10.1007/s13555-019-00319-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Prindaville B, Belazarian L, Levin NA, Wiss K. Pityrosporum folliculitis: a retrospective review of 110 cases. J Am Acad Dermatol. 2018;78(3):511–514. doi: 10.1016/j.jaad.2017.11.022. [DOI] [PubMed] [Google Scholar]
- 20.Pürnak S, Durdu M, Tekindal MA, Güleç AT, Seçkin D. The prevalence of. Skinmed. 2018;16(2):99–104. [PubMed] [Google Scholar]
- 21.Ran YP, Zhou GP. Pityrosporum folliculitis. Clinical and pathologic report of seven cases. Chin Med J (Engl). 1988;101(10):748–749. [PubMed] [Google Scholar]
- 22.Rubenstein RM, Malerich SA. Malassezia (pityrosporum) folliculitis. J Clin Aesthet Dermatol. 2014;7(3):37–41. [PMC free article] [PubMed] [Google Scholar]
- 23.Suzuki C, Hase M, Shimoyama H, Sei Y. Treatment outcomes for malassezia folliculitis in the dermatology department of a University Hospital in Japan. Med Mycol J. 2016;57(3):E63–66. doi: 10.3314/mmj.16-00003. [DOI] [PubMed] [Google Scholar]
- 24.Tsai YC, Wang JY, Wu YH, Wang YJ (2018) Atypical clinical presentations of Malassezia folliculitis: a retrospective analysis of 94 biopsy-proven cases. Int J Dermatol. 57(3):e19–e20 [DOI] [PubMed]
- 25.Tsai YC, Wang JY, Wu YH, Wang YJ. Clinical differences in pediatric and adult Malassezia folliculitis: Retrospective analysis of 321 cases over 9 years. J Am Acad Dermatol. 2019;81(1):278–280. doi: 10.1016/j.jaad.2019.03.014. [DOI] [PubMed] [Google Scholar]
- 26.Tu WT, Chin SY, Chou CL, et al. Utility of Gram staining for diagnosis of Malassezia folliculitis. J Dermatol. 2018;45(2):228–231. doi: 10.1111/1346-8138.14120. [DOI] [PubMed] [Google Scholar]
- 27.Vest BE, Krauland K (2022) Malassezia Furfur. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing
- 28.Xu H, Li H. Acne, the skin microbiome, and antibiotic treatment. Am J Clin Dermatol. 2019;20(3):335–344. doi: 10.1007/s40257-018-00417-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Xu X, Ran X, Tang J, et al. Skin microbiota in non-inflammatory and inflammatory lesions of acne vulgaris: the underlying changes within the Pilosebaceous unit. Mycopathologia. 2021;186(6):863–869. doi: 10.1007/s11046-021-00586-6. [DOI] [PubMed] [Google Scholar]
- 30.Yong AM, Tan SY, Tan CL. An update on pityrosporum folliculitis in Singapore from a single tertiary care dermatological centre. Singapore Med J. 2021;62(10):526–528. doi: 10.11622/smedj.2020068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yu HJ, Lee SK, Son SJ, Kim YS, Yang HY, Kim JH. Steroid acne vs. Pityrosporum folliculitis: the incidence of Pityrosporum ovale and the effect of antifungal drugs in steroid acne. Int J Dermatol. 1998;37(10):772–777. doi: 10.1046/j.1365-4362.1998.00229.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.