Abstract
Inflammatory disease is often associated with an increased incidence of venous thromboembolism in affected patients, although in most instances, the mechanistic basis for this increased thrombogenicity remains poorly understood. Acute infection, as exemplified by sepsis, malaria and most recently, COVID-19, drives ‘immunothrombosis’, where the immune defence response to capture and neutralise invading pathogens causes concurrent activation of deleterious prothrombotic cellular and biological responses. Moreover, dysregulated innate and adaptive immune responses in patients with chronic inflammatory conditions, such as inflammatory bowel disease, allergies, and neurodegenerative disorders, are now recognised to occur in parallel with activation of coagulation. In this review, we describe the detailed cellular and biochemical mechanisms that cause inflammation-driven haemostatic dysregulation, including aberrant contact pathway activation, increased tissue factor activity and release, innate immune cell activation and programmed cell death, and T cell-mediated changes in thrombus resolution. In addition, we consider how lifestyle changes increasingly associated with modern life, such as circadian rhythm disruption, chronic stress and old age, are increasingly implicated in unbalancing haemostasis. Finally, we describe the emergence of potential therapies with broad-ranging immunothrombotic functions, and how drug development in this area is challenged by our nascent understanding of the key molecular and cellular parameters that control the shared nodes of proinflammatory and procoagulant pathways. Despite the increasing recognition and understanding of the prothrombotic nature of inflammatory disease, significant challenges remain in effectively managing affected patients, and new therapeutic approaches to curtail the key pathogenic steps in immune response-driven thrombosis are urgently required.
Keywords: haemostasis, immunothrombosis, inflammation
Haemostatic response to vessel injury
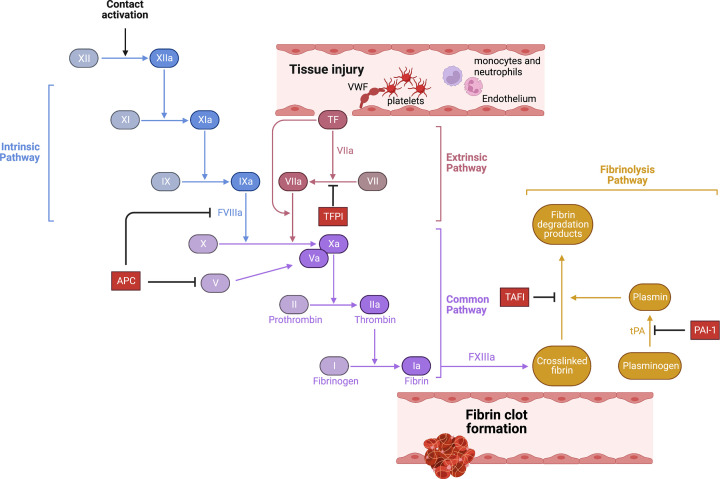
Blood coagulation is activated in response to blood vessel injury (Figure 1). Traumatic disruption of the endothelial cell layer causes the recruitment of platelets to the site of vessel injury, either via platelet glycoprotein 1b receptor interaction with plasma von Willebrand factor (VWF) bound to sub-endothelial tissue, or via direct platelet collagen receptor binding to exposed collagen. Adherent platelets become activated, prompting extensive morphological changes and secretion of granular contents to encourage further platelet recruitment to the growing clot and provide a cellular surface to amplify procoagulant enzymatic reactions. Activated platelets also secrete VWF and express P-selectin, an important cell adhesion receptor, which binds to P-selectin glycoprotein ligand 1 (PSGL-1) expressed by monocytes and neutrophils. In parallel, vessel injury causes the exposure of tissue factor (TF), expressed extensively within the sub-endothelial tissue, to blood (for detailed review, see [1]). TF bound to activated FVII (FVIIa) triggers the formation of the extrinsic tenase complex (TF, FVIIa, and factor X, FX). Activated FX (FXa), bound to its cofactor activated factor V (FVa), promotes the conversion of zymogen prothrombin to thrombin. Thrombin, in turn, activates several coagulation proteases and cofactors, including FV, FVIII, and FXI, to amplify the procoagulant response and overwhelm counteractive anticoagulant mechanisms such as tissue factor pathway inhibitor (TFPI). FXIa activation of FIX prompts intrinsic tenase complex formation (FIXa, FVIIIa, and FX) that promotes sufficient FXa generation to drive prothrombinase activity, allowing generated thrombin to convert soluble fibrinogen in the blood into an insoluble fibrin mesh clot. Thrombin also further activates platelets and endothelial cells via protease-activated receptor (PAR1) signalling [2]. Endogenous anticoagulant mechanisms, such as antithrombin and the protein C (PC) pathway, regulate clot size and spread. In addition, fibrinolytic factors, such as tissue-type plasminogen activator (tPA) and plasmin, coordinate clot resolution by enzymatic fibrin degradation.
Figure 1. Mechanisms of blood clot formation.
Blood coagulation is triggered by vessel injury and TF exposure. This triggers extrinsic pathway activation, leading to thrombin generation via the shared pathway and fibrin generation. Alternatively, contact pathway activation via FXII activation by polyP also leads to intrinsic pathway activation, thrombin generation and fibrin deposition in the absence of vessel damage, leading to pathological thrombus formation. Clot growth and size is regulated at multiple stages by endogenous anticoagulants, including tissue pathway inhibitor, the PC pathway and antithrombin. Once the vessel repair is underway, proteolytic clot lysis is orchestrated by plasmin, which breaks down cross-linked fibrin into fibrin degradation products.
Pathological venous clot formation, typically in the absence of traumatic injury, requires factor FXII (XII) activation rather than solely TF-dependent fibrin generation (for detailed review, see [3]). FXII can be activated by negatively charged synthetic materials such as glass, silica or nanoparticles [4]. Recently, however, potential endogenous FXII activators have been identified, such as misfolded protein aggregates, free collagen, glycosaminoglycans and circulating nucleic acids [5]. Of particular importance is inorganic polyphosphate (polyP) [6], which is released from activated platelets to rapidly activate FXII and drive thrombus development [7].
What is ‘immunothrombosis’?
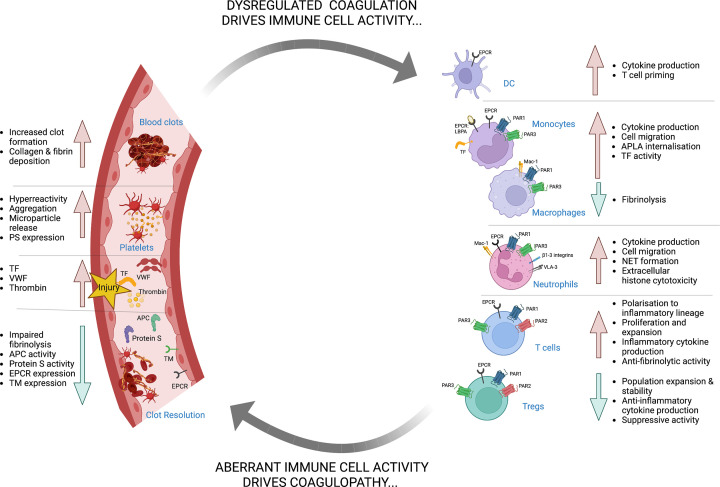
Clot development likely evolved in part as an ancient anti-bacterial response to enable localisation and neutralisation of various bloodborne pathogens. Aberrant clot formation, however, that arises from infection or excessive inflammatory response during disease is now commonly termed ‘immunothrombosis’ (Figure 2). Whereas this primitive response represents an effective strategy to slow pathogen spread during infections, this may come at the cost of immune-driven pathological thrombus formation. Currently, therapeutic approaches to control immunothrombotic disease are limited by our nascent understanding of how proinflammatory and procoagulant pathways intersect. In this review, we describe the convergent mechanisms that determine how inflammatory events precipitate an increased risk of venous thromboembolism (VTE).
Figure 2. Molecular and cellular mechanisms of immunothrombosis.
Both acute and chronic inflammatory diseases, including autoimmune diseases, allergies, and infections, promote dysregulation of haemostasis, leading to immunothrombosis. Inflammatory mediators such as enhanced pathogenic immune cell populations, increased proinflammatory cytokines and chemokines levels, and diminished tolerogenic Treg cell responses coincide with and promote aberrant coagulation. This enhanced procoagulant state feeds back to amplify inflammation, inducing further expansion of proinflammatory immune cells, enhanced TF expression in myeloid cells, NET production and inhibition of tolerogenic Treg responses that impair thrombolysis and clot resolution.
Activation of the immune response
Innate immune cells, such as monocytes, macrophages, granulocytes and dendritic cells (DCs), classically perform sentinel roles in immunosurveillance and antigen presentation to cells of the adaptive immune system, thereby inducing a robust immune response and generating immune memory [8]. These innate cells recognise intra- and extra-cellular pathogens via pathogen recognition receptors (PRRs). PRRs are sub-classified as Toll-like receptors (TLRs), nucleotide-binding oligomerization domain (Nod) like receptors (NLRs), C-type lectin receptors (CLRs), and RIG I-like receptors (RLRs). Macrophages and DCs recognise extracellular pathogens by sensing components of their cell wall such as LPS, bacterial lipopeptides, lipoteichoic acids, glycolipids, flagellin, and β-glucans via TLR1, TLR2, TLR4, TLR5, TLR6, and CLRs, such as dectin-1 and dectin-2, which are expressed on their plasma membrane [8]. TLR3, TLR7, TLR8, and TLR9, are expressed in the cell endosomes and sense intrinsic pathogens, such as viral nucleic acids [8]. RLRs, NLRs, and cytosolic DNA sensors detect viral RNA and DNA, and invading bacteria in the cytosol [8–12]. In the bloodstream, monocytes and neutrophils recognise invading bacterial, fungal and protozoan pathogens via TLRs, NLRs, and CLRs and facilitate phagocytosis, degranulation, and killing of these microorganisms [8,13]. Following pathogen recognition, innate immune cells also secrete inflammatory cytokines and chemokines into the vicinity, recruiting both other innate and adaptive cells to the site to amplify the inflammatory response. Furthermore, antigen-loaded DCs migrate to the draining lymph nodes to prime an adaptive T-cell response. The type of T-cell response induced depends on several factors including, the location of the site of injury or infection, the class of antigen being presented and the presence of other instructive cytokines and chemokines in the milieu [8]. Typically, bacteria, protozoa and fungi elicit a type 1 immune response via TLRs, RLRs, and CLRs, involving CD4+ Th1 and Th17 cells, and CD8+ cytotoxic lymphocytes (CTLs) [14–19], whereas helminths, allergens, and venoms elicit a type 2 immune response, involving CD4+ Th2 cells [20–22]. CTL induction of type 1 immunity is also required to clear viral infection via RLRs, TLR3, and CLRs [23–25]. Recent studies have highlighted the role of both adaptive and innate immune cell responses in dysregulated haemostasis.
Immune cell contribution to pathological thrombus formation
Platelets represent the classical bloodborne haemostatic cell, and their prothrombotic activity is critical for normal haemostasis. In addition to platelets, recent data have highlighted the roles played by various innate immune cells in thrombus development via distinct and often cell-specific mechanisms. Post-mortem studies and preclinical deep vein thrombosis (DVT) mouse models suggest an essential contribution of myeloid cells to VTE. Neutrophils have a crucial role in response to infection and subsequent resolution of inflammation but have recently also been identified to be critical for DVT formation. Neutrophils release neutrophil extracellular traps (NETs), which represent a last-ditch ‘damage-limitation’ approach to kill microbes during infection [26] but can also induce toxic collateral damage within the vasculature. As such, NET formation is implicated in the pathogenesis of numerous autoimmune and inflammatory diseases [26–29]. Furthermore, NETs have been demonstrated to contribute to aberrant thrombus development via multiple mechanisms [30,31]. DNA strands within NETs have been reported to capture and contribute to FXII activation [32]. Notably, NET formation can be regulated by different plasma DNAses [33]. Moreover, neutrophil enzymes, including elastase and cathepsin G, can cleave and inactivate plasma TFPI, removing a vital anticoagulant regulator from the proximity of the developing thrombus [32]. Extracellular histones within NETs promote activation of platelets [34] and interact with NET-bound VWF [35]. In addition, extracellular histones inhibit the PC anticoagulant pathway by attenuating activated protein C (APC) generation by the thrombin–thrombomodulin complex on endothelial cells [36]. Therapeutic strategies to inhibit or degrade NETs have also been reported. For example, NET inhibitors that target protein arginine deiminases (PAD4) critical for NET formation exert beneficial activity in preclinical models of arthritis, shifting the T-cell balance from a proinflammatory Th1/Th17 response to a pro-resolution Th2 response [37].
Monocytes and tissue-resident macrophages also contribute to VTE development and monocyte count have recently been reported to correlate with thrombus size and growth in murine thrombosis models [38]. Myeloid cells typically express TF on their surface, and TF expression and activity are up-regulated by exposure to either pathogen-associated molecular patterns or proinflammatory cytokines [39]. In a mouse model of stasis-induced DVT, monocyte TF was critical for initiating DVT formation [40], highlighting the complex and multi-faceted role played by innate immune cells in pathological clot formation.
Coagulopathy during acute infection
Sepsis develops due to a systemic dysregulated immune response to acute infection and accounts for ∼20% of global deaths [41]. The case fatality rate of sepsis has steadily decreased in the past 20 years; however, its incidence is the same or increasing [42–44]. Sepsis is characterised by excessive cytokine production in response to infection, leading to tissue and organ dysfunction [45]. A prominent feature of sepsis is haemostatic dysregulation, thrombocytopenia, and disseminated intravascular coagulation (DIC) [46]. DIC is associated with a worse outcome and higher mortality rate compared with sepsis patients without DIC [47–49].
Aberrant up-regulation of TF expression on circulating innate immune cells and the endothelium upon exposure to proinflammatory stimuli has long been considered an important trigger for DIC [50–52]. Moreover, recent studies have provided a mechanistic basis for cell surface TF release into the blood during bacterial infection, highlighting the crucial role of non-canonical inflammasome formation in modulating TF-rich microparticle release from activated myeloid cells. This mechanism may also be partially conserved in other cell types, such as tumour cells, which also actively release TF-containing microparticles and are associated with increased thrombgenicity [53]. Activation of the canonical or non-canonical inflammasome in macrophages following exposure to Escherichia coli EprJ type-III-secretion system rod proteins or LPS, respectively, results in the release of TF-rich microparticles [54]. In this process, pathogen exposure triggers gasdermin D (GSDMD) cleavage by activated caspase-1 and -11 to drive pyroptosis and interleukin-1β (IL-1β) release [54]. It has also been reported that a GSDMD-dependent increase in TF activity following non-canonical inflammasome activation occurs independently of cell death via increased phosphatidylserine (PS) exposure, aiding in the assembly of the extrinsic tenase complex [55]. By labelling PS on mouse splenic and peripheral blood leukocytes with lactadherin, the study showed increased PS on mouse leukocytes following transfection of LPS and a reduction in exposed PS in Gsdmd−/− leukocytes [55]. Furthermore, myeloid cell inflammasome formation has been shown to contribute to VTE development in a mouse inferior vena cava (IVC) stenosis model [56]. Mice with caspase-1 or GSDMD deficiency exhibited smaller and fewer thrombi formation following IVC stenosis compared with wild-type mice, underscoring the potential importance of canonical and non-canonical inflammasomes in immunothrombosis.
Similarly, pathogen and cytokine activation of the endothelium also contributes to a prothrombotic milieu during sepsis. Various cytokines promote the release of long, multimeric chains of ultra-large von Willebrand factor (UL-VWF) on to the endothelial cell surface. UL-VWF binds circulating platelets via platelet glycoprotein Ibα [57] and is typically broken down by the plasma metalloproteinase ADAMTS13 into smaller multimers, reducing VWF thrombogenicity [58]. UL-VWF can act as an adhesive surface to monocytes and polymorphonuclear leukocytes by binding PSGL-1 and β2-integrin [59]. VWF strings decorated with platelets and other immune cells have been suggested to contribute to microthrombi development in DIC. Notably, sepsis patients have lower levels of circulating ADAMTS13 and ADAMTS13 activity [60,61], contributing to sustained VWF strings in the intravascular space. In support of an essential role for ADAMTS13 in regulating VWF thrombogenicity during infection, low ADAMTS13 activity in patients with Staphylococcus aureus infections is associated with increased disease severity [62]. NETs formed during sepsis contribute to widespread organ damage [63] and DIC development [64,65] and their presence is reported to be especially deleterious in infant sepsis [66].
Many endogenous plasma anticoagulant proteins and cell receptors are depleted during sepsis, leading to unregulated thrombin activity and fibrin generation. For example, low levels of circulating PC, protein S and antithrombin have been associated with worse sepsis outcomes [67]. To address this, various anticoagulant therapies have been tested to mitigate sepsis-associated coagulopathy, with limited success. Most recently, recombinant APC (Xigris, Eli Lilly) was shown to reduce mortality in severe sepsis patients and was licensed on this basis [68]. Subsequent trials, however, raised questions about the efficacy and safety of APC in this setting and Xigris was eventually voluntarily withdrawn for patient use [69]. Important anticoagulant receptors, such as thrombomodulin (TM) and the endothelial PC receptor (EPCR), are cleaved from the endothelial cell surface under inflammatory conditions. Soluble forms of both receptors are elevated in the plasma of individuals with sepsis [70]. Depleting endothelial cell surface EPCR and TM restricts PC activation, skewing the haemostatic balance towards thrombin generation. Interestingly, individuals with the rs2239562 single-nucleotide polymorphism in the promoter region of the thrombomodulin (THBD) gene have been suggested to be more susceptible to sepsis and experience worse outcomes [71].
Recent studies indicate that immune cross-talk has a role in the immunothrombotic state observed in malaria infection. Coagulation dysregulation is commonly observed among malaria patients and represents an often fatal complication of the disease [72]. A recent clinical trial revealed that healthy volunteers injected with low levels of Plasmodium falciparum sporozoites exhibited a hypercoagulable state. At detection of parasitaemia, the thrombin generation potential of plasma isolated from infected subjects was 17% higher compared with their baseline measurements [73]. In addition, malaria patient studies and preclinical malaria animal studies support the role of increased endothelial cell activation and UL-VWF secretion, in sequestering infected red blood cells in the microvasculature to promote cerebral vascular occlusion [74]. Taken together, these findings indicate that coagulopathy driven by acute infection arises from aberrant activation of multiple procoagulant mechanisms that are further amplified by inflammation-induced endothelial damage and diminished systemic anticoagulant activity.
The Coronavirus Disease in 2019 (COVID-19) pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection represents an exemplar model of immunothrombotic disease driven by viral infection. VTE rates among COVID-19 patients are increased compared with healthy individuals [75,76], and COVID-19 causes a unique coagulopathy that can promote microvascular thromboses in the lung vasculature. Retrospective studies indicate that the severity of COVID-19 symptoms and the level of proinflammatory biomarkers in patients with COVID-19 positively correlated with the patient’s risk of experiencing VTE [77]. For instance, in a dataset containing 6153 COVID-19 patients, only 0.09% of non-hospitalised COVID-19 patients developed VTE [77], whereas 3.1% of hospitalised patients experienced a VTE, rising to 7.2% of COVID-19 patients who required ventilation [77]. Similarly, in a cohort of 1114 COVID-19 patients, only a small number of thrombotic events were reported in COVID-19 outpatients, whilst the highest rates of thrombotic complications appeared in those COVID-19 patients admitted to ICU [78]. Numerous studies have highlighted potential mechanisms of prothrombotic activity associated with COVID-19 pathobiology. In severely ill patients, dysregulated inflammatory cytokine release promotes endotheliopathy and innate immune cell dysfunction, driving formation of a prothrombotic milieu [79]. Neutrophils play a critical role in COVID-19-associated coagulopathy. Plasma biomarkers of NET formation correlate with COVID-19 disease severity [80], and neutrophil-platelet aggregates may also contribute to enhanced platelet reactivity in COVID-19 patients [81,82]. Interestingly, patients with severe COVID-19 often exhibit thrombocytopenia, despite having increased generation of megakaryocytes and proplatelet formation. This disproportionate platelet count is likely due to thrombus deposition in multiple vessel beds [29,81].
A persistent immunothrombotic state in COVID-19 patients may also contribute to post-viral syndrome or ‘long-COVID’, a common feature in COVID-19 patients characterised by fatigue and chronic disease symptoms that can last for weeks and months post-infection [83]. The underlying cause of this fatigue is unclear but may arise in part due to damage to the endothelium arising from chronic hypoxia and inflammation [84]. In support of this, recent data indicate that individuals with ‘long-COVID’ exhibit a persistent endotheliopathy characterised by dysregulated VWF proteolysis, endothelial dysfunction and the persistence of activated monocyte and T-cell populations [85]. Anticoagulant and anti-inflammatory strategies targeting the inflamed endothelium may therefore provide a novel approach to mitigate symptoms associated with long COVID.
Does the adaptive immune system contribute to immunothrombosis?
Although adaptive immune system cells do not appear to contribute to normal haemostasis, recent work has highlighted a new role for T effector memory (TEM) cells and regulatory T cells (Tregs) in modulating venous clot lysis. Unexpectedly, CD44highCD62LlowCD4+ and CD8+ TEM cells infiltrate the thrombus and vessel wall during DVT in a murine thrombosis model [86]. Infiltrating TEM cells secrete IFNγ, independent of antigen stimulation, and recruit monocytes and neutrophils to the injury site, delaying thrombus resolution. Deficiency in TEM cells enhanced thrombolysis and promoted increased matrix metalloproteinase-9 (MMP-9) expression by monocytes [86]. Previous studies have shown that suppression of MMP-9 in macrophages by T cell, NK cell and innate lymphoid cell-derived IFNγ delays clot resolution [87]. Tregs also play an unanticipated role in mediating efficient clot degradation, and their depletion results in delayed thrombolysis [88]. Furthermore, Treg population dynamics were found to influence monocyte recruitment and differentiation. Following TGFβ stimulation, a Treg subpopulation was found to produce secreted protein acidic and rich in cysteine (SPARC), a matricellular protein involved in cell turnover, tissue remodelling, and ECM repair. SPARC+ Tregs were reported to recruit more CD11c+ monocytes to the thrombus with enhanced MMP and fibrinolytic activity [88]. Interestingly, cancer patients undergoing T cell targeted immunotherapies, such as immune checkpoint inhibitors (ICIs), exhibit an increased VTE risk (1.5- to 6.5-fold) [89] and cumulative incidence of VTE (5–10% at 12-month follow-up) [90], which may be due to the increased activity of effector T cells. These data demonstrate an unexpected but potentially important role for T cell sub-populations in modulating clot lysis and resolution.
Why do autoimmune disorders cause an increased VTE risk?
Current estimates report a global prevalence of autoimmune disease in 3–5% of the general population [91], with a recent report highlighting an increasing incidence of 3–9% per year [92]. Notably, increased VTE risk is characteristic of many autoimmune diseases, such as inflammatory bowel disease (IBD), systemic lupus erythematosus (SLE), and graft versus host disease (GvHD), and often represents a fatal disease complication.
IBD
IBD represents an expanding global healthcare burden affecting many adults and children. Approximately 0.3% of the worldwide population are diagnosed with IBD, with 25% of these cases arising in childhood and adolescents [93–95]. Notably, thromboembolic disease represents the most significant cause of mortality among IBD patients [96], with adult and paediatric IBD patients exhibiting a 3- and 6-fold increased risk of VTE development compared with the general population [97,98]. Yet, the mechanism(s) underlying this prothrombotic state remains unknown. Individuals with IBD are characterised by increased levels of proinflammatory immune cells, such as T cells, innate lymphoid cells, monocytes and macrophages, and cytokines, such as TNFα, IFNγ, IL-1β, IL-17a, IL-36α, IL-18, IL-6 and IL-8 [99–102]. Several plasma procoagulant and anti-fibrinolytic proteins, including PAI-1, fibrinogen, and prothrombin, are elevated during an IBD ‘flare’, in which symptoms are acutely increased [103–106].
In murine IBD models, TF inhibition is associated with reduced immunothrombotic activity [107]. In addition, increased numbers of procoagulant TF+ MPs are shed by platelets and leukocytes in IBD patients [108,109]. Interestingly, these MPs also can stimulate neutrophil NETosis [110,111]. As NET density correlates with disease severity [27], targeting NET formation and MP interactions may represent a potential strategy to mitigate against immunothrombosis in IBD patients. Platelet dysregulation and hyperactivity also contribute to the prothrombotic state observed in IBD. Increased platelet activation in IBD patients has been observed, with platelet activation status correlating with disease severity [112,113].
Whereas chronic inflammation disrupts haemostatic balance to promote thrombosis, the ability of many coagulation proteases to modulate cell function, particularly the inflammatory response directly via cell signalling, suggests an additional mechanism by which aberrant coagulation protease activation, or loss of protective anticoagulant signalling responses, can modulate the mucosal immune response. The PC pathway has been implicated as an important regulator of mucosal immunity, regulating the inflammatory responses in the endothelium, epithelium, and innate and adaptive immune cells [114,115]. Transgenic mice with low plasma PC (<5% normal levels) display a constitutively enhanced proinflammatory and prothrombotic phenotype [116], and in the context of IBD, have been shown to develop spontaneous colitis due to increased TNF-mediated intestinal barrier permeability [114]. Colitogenic mice display a reduced capacity for PC activation [115] likely due to decreased EPCR and TM expression in the inflamed microvasculature [114,115]. Accordingly, EPCR-deficient mice exhibit exacerbated disease in DSS-induced colitis compared to wild-type mice [117]. Chronic inflammation may contribute to the increased VTE risk experienced by IBD patients. Interestingly, IBD patients receiving anti-TNF biologics exhibit a reduced risk of VTE compared with those on traditional corticosteroids [118], implicating TNFD-mediated inflammation as a critical feature of increased VTE risk in IBD patients. In contrast, however, other targeted immunobiologics, including ustekinumab, a monoclonal antibody targeting the p40 subunit of IL-12/23, and vedolizumab, a monoclonal antibody targeting the α3β7 integrin, confer a potential risk of severe cardiovascular events to patients, including stroke [119], thrombocytopenia [120] and reduced clot formation time [121].
Graft versus host disease
Incidence of VTE among haematopoietic stem cell transplant (HSCT) patients ranges from 0.5 to 13%, and the occurrence of VTE is typically associated with the development of Graft versus Host Disease (GvHD) [122–129]. In those who develop VTE post-transplant, there is a 65% incidence in GvHD [122]. Furthermore, there is a higher risk of other thrombotic events associated with GvHD severity and donor blood group, with non-O-donor to recipient ABO match exhibiting a 2.7-fold higher hazard ratio of thrombotic complication compared to O-donor to O-recipient match [130]. Transplant-associated thrombotic microangiopathy (TA-TMA) is a common complication of HSCT and is associated with complement activation following an endothelial injury during GvHD [131]. The association between severity of inflammation exposure and risk of thrombosis in GvHD patients is exemplified by the lowered threshold for neutrophil NETosis in GvHD patients and the consequent role in increased plasma hypercoagulability. Paediatric recipients post-HSCT have elevated levels of plasma double-stranded DNA (dsDNA), a surrogate marker for NETs, alongside an increased risk of TA-TMA, onset of gastrointestinal GvHD and mortality [132].
SLE
VTE is a common co-morbidity in patients with systemic lupus erythematosus (SLE), and the risk of its development is commonly associated with the presence of antiphospholipid antibodies (APLA). To date, the most critical APLAs identified to confer thrombotic risk are anticardiolipin antibodies (ACA), lupus anticoagulant (LAC), and anti-β2-glycoprotein I (anti-β2-GPI) [133,134]. Despite this, 40% of SLE patients diagnosed with thrombotic complications are negative for APLAs [133], indicating a multifactorial mechanism for VTE in these patients. SLE patients display endothelial dysfunction, characterised by increased expression of fibrinogen and cell adhesion molecules, ICAM, VCAM, VEGF, and VWF [135].
Furthermore, SLE patients exhibit a reduced capacity to clear neutrophil NETs [136] and an enhanced adaptive proinflammatory cytokine and immune cell profile [137]. Interestingly, β2GPI-reactive CD4+ T cells are hypothesised to drive auto-antibody production in SLE [138], thus amplifying the immunothrombotic environment. Recent work has identified an EPCR-lysophosphatidic acid (LBPA) complex expressed on monocyte cell surfaces as a receptor for APLAs that facilitates their internalisation and subsequent proinflammatory and procoagulant activities [139]. APLAs binding to EPCR-LBPA activate TF-FVIIa-FXa-PAR2 signalling via acid sphingomyelinase TF decryption to promote coagulation. APLA-mediated TF activation also induces IFN production from DCs, promoting the expansion of APLA-producing B1a cells, creating an immunothrombotic loop. Studies using EPCR-deficient transgenic mice and EPCR-blocking antibodies revealed that EPCR-LBPA deficiency protects against APLA-induced thrombosis and foetal loss, murine SLE and APS [139]. Therefore, therapeutic targeting of this pathway holds promise in regulating APLA autoimmunity and thrombosis.
Allergies
Dysregulation of haemostatic and fibrinolytic activity is evident in many allergic diseases, including chronic spontaneous urticaria (CSU), asthma and dermatitis [140–143]. Still, the inflammatory mechanisms underlying this haemostatic dysregulation, such as elevated soluble TF levels, PAI-1 and thrombin activatable fibrinolysis inhibitor (TAFI) [143] found in asthma patient sputum, are poorly understood. Interestingly, there is a significantly increased risk of pulmonary embolism in asthma patients compared to the general population, linked to the frequency of asthma exacerbation and hospitalisation [144]. In the airways, TF expression is induced by IL-13, one of the major cytokines involved in asthma pathogenesis [145]. In parallel, FX is also implicated in airway dysfunction, and FXa signalling increases airway fibroblast proliferation via the production of autocrine platelet-derived growth factor (PDGF) and procollagen synthesis through PAR1 activation, resulting in airway fibrosis and reduced lung compliance. Accordingly, FXa inhibition with fondaparinux has been shown to alleviate airway pathology in a mouse asthma model [146,147]. Enhanced thrombin activation and reduced levels of APC are also reported in asthma patients [148–150]. Platelet hyperactivity and fibrin deposition all exacerbate asthma, contributing to bronchial hyperresponsiveness, constriction, airway remodelling and inflammation [151]. Similarly, fibrin degradation products (FDPs) contribute to airway dysregulation, promoting leukocyte infiltration, proinflammatory cytokine and PAI-1 release [151–154].
Allergies in the skin, such as atopic dermatitis and contact dermatitis, are chronic inflammatory disorders of the epidermis, affecting up to 10% of adults and 20% of children globally [155]. These conditions are triggered by skin exposure to potential allergens [156], which can induce an exaggerated T cell-mediated immune response [157]. The infiltrating T cells exert cytotoxic effector mechanisms on nearby keratinocytes, thus disrupting the skin barrier and allowing the further invasion of allergens [157]. Like airway inflammation, increased myeloid cell TF expression and activity are associated with skin allergy pathogenesis, and monocytes isolated from CSU patients express significantly more TF activity than healthy controls [158]. Coagulation proteases can also induce degranulation of human skin mast cells and basophils via complement C5a and C5aR, causing edema [159]. Similarly, thrombin and fibrinogen have been shown to play a pathological role in contact dermatitis. Children with contact dermatitis show a correlation between disease severity, transepidermal water loss (TEWL), and total thrombin generation. Inhibition of either thrombin or fibrinogen in preclinical dermatitis models alleviates disease [160], as does treatment with recombinant APC [142]. APC signalling through PAR2 helps clinical signs of disease by inhibiting CD4+ T cell generation and proinflammatory responses [142]. Collectively, these studies highlight an emerging role for dysregulated haemostatic protease signalling function in the development and persistence of allergic skin disease.
Neurodegenerative disease and ageing
Aberrant coagulation pathway activity and signalling have been implicated in the pathogenesis of several neurological disorders. Multiple sclerosis (MS) is an immune-mediated demyelinating and degenerative disease of the central nervous system (CNS) [161]. MS patients have a 2-fold higher VTE risk compared with the general population [162]. Histopathological studies by Putnam in the 1930s reported an increased incidence of thrombi in plaques surrounding the CNS vasculature of MS patients that did not form in response to vessel wall injury but instead due to ‘abnormal lability in blood plasma’ [163,164]. Since then, MS patients have exhibited increased fibrin deposition around active MS lesions, impaired fibrinolytic activity, high soluble TF levels in the CNS, circulating soluble TM and elevated VWF activity reflective of endotheliopathy [165–169]. These studies indicate that proinflammatory and coagulation pathways are co-activated during MS pathogenesis, and dual targeting of both systems may be beneficial in this setting. In support of this, recent work selectively targeting a cryptic fibrin epitope (γ377-395) with a monoclonal antibody (5B8) was beneficial in preventing amyloid-driven neurodegeneration and CNS autoimmunity in preclinical models of MS and Alzheimer’s disease (AD) [170]. 5B8 inhibited fibrin binding to CR3, thus suppressing fibrin-induced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase activation, subsequent reactive oxygen species (ROS) release and innate immune-driven neurotoxicity. 5B8 down-regulated expression of genes involved in immune cell recruitment and proinflammatory responses in fibrin-treated bone marrow-derived macrophages and reduced axonal damage, ROS generation, demyelination and CD11b+ cell innate-driven inflammation in EAE [170]. Furthermore, inhibiting fibrin/CD11b interactions with 5B8 reduced loss of cholinergic neurons and microglia activation around plaques in 5XFAD mice and down-regulated critical pathways involved in neurodegeneration and oxidative stress, complement pathway, lysozyme, antigen presentation, cytokine response, and ROS pathway signalling. Significantly, this antibody did not interfere with normal haemostasis, with no differences reported in fibrin polymerisation in vitro, clotting time, or partial thromboplastin time (aPTT) in human plasma treated with 5B8 reported compared to IgG2b-treated plasma controls [170].
Elevated levels of FXII have also been observed in the plasma of MS patients and may contribute to the thrombo-inflammatory state observed in the CNS of affected individuals [171,172]. EAE mice display significantly elevated FXII levels in the plasma, cerebrospinal fluid (CSF), and lymph nodes compared with control mice. FXII deficiency ameliorates disease via inhibition of DC-mediated priming of Th17 cell pathogenic responses [171]. Importantly, mechanistic studies have revealed that FXIIa inhibition inhibits thrombus formation but does not affect haemostasis [4,173,174]. Therefore, inhibiting contact activation may prove a safe method to break the immunothrombotic loop observed in neuro-inflammatory and neurogenerative diseases, without enhancing the risk of bleeding.
The risk of thrombosis increases with age. This is probably influenced by ‘inflammaging’, which describes the chronic low-grade and systemic inflammation that accelerates during biological ageing and occurs through various mechanisms, including immunosenescent cell generation, increased intestinal barrier permeability, bacterial dysbiosis, and leakage [175,176]. Collectively, these processes combine to induce increased levels of proinflammatory cytokines, chemokines, and immune cells in the vasculature, creating a lower threshold for pro-thrombotic events.
For example, older mice are reported to exhibit higher levels of TNFα in their plasma compared with young mice [177,178] and inflammaging contributes to age-related platelet hyperactivity via exposure to enhanced constitutive levels of proinflammatory cytokines such as TNF in the blood. Following stimulation, platelets from older mice also express increased levels of αIIbβ3 and PS and display an enhanced capacity to form thrombi ex vivo. Furthermore, TNF levels are heightened in the bone marrow compartment of older mice [179], suggesting local megakaryocytes will be more susceptible to inflammaging. Interestingly, monocytes have been proposed as the cellular source of TNF in this environment [180]. Monocyte-derived TNF is implicated in the increased platelet count and reactivity observed in older patients with myeloproliferative disorders [181,182]. Enhanced platelet–monocyte interactions result in proinflammatory cytokine production [183], which may be exacerbated by platelet derived-MPs that are also increased in older individuals [184].
Elevated fibrin levels have been observed in AD patient brains post-mortem, and signalling by this protein is linked to Aβ and tau deposition, vascular dysfunction, inflammation, and neurodegeneration [185]. In an open-label study of patients with mild to moderate AD, treatment with the thrombin inhibitor hirudin with donepezil significantly reduced the rate of cognitive decline among patients [186], suggesting that coagulation inhibition may be a novel strategy for mitigating neurodegenerative and neuroinflammatory disease. Furthermore, there is emerging evidence for the role of neutrophils and NETosis in AD pathology. Neutrophils, NETs and NET-associated MPO have been found in plaques surrounding the microvasculature in post-mortem AD patient brains and murine AD models [187,188]. The contact pathway is also implicated in AD pathogenesis. FXII has been detected in Aβ plaques [188], increased plasma kallikrein activity has been observed in the AD brain parenchyma [189], and enhanced levels of HK cleavage in the cerebrospinal fluid in AD patients compared with non-disease controls [190]. In vitro studies have found that Aβ can trigger FXIIa-dependent kallikrein activity and kallikrein-mediated cleavage of HK [191–193]. In vivo studies have supported these findings, suggesting Aβ plaques can mediate the upregulation of contact activation [194]. Notably, FXII inhibition was beneficial in AD mice, which displayed improved cognitive functioning compared with untreated controls. Furthermore, FXII depletion in AD mice resulted in diminished plasma HK cleavage and reduced fibrinogen deposition, neurodegeneration and neuroinflammation compared with untreated controls [195]. Collectively, these studies indicate that activation of the contact pathway contributes to the vascular and inflammatory dysfunction observed in AD.
Stress and circadian rhythm disruption
Stress is a risk factor for neurodegenerative disease [196,197], metabolic disease [198], cancer [199] and, with particular relevance to this review, thromboembolic disease [200]. Both acute and chronic stress causes a change in the distribution of immune cells between the circulation and tissue [201,202]. Assessment of the effect of stress on hypercoagulability is challenging, and several studies have generated disparate results. Following induction of acute stress by a public speaking task and a mock job interview, individuals exhibited increased plasma levels of D-dimer and VWF [203,204]. A comprehensive acute stress test study analysed multiple plasma coagulation parameters before and after stress. They found significant increases in FVII and FVIII activity and increased VWF and D-dimer levels in acutely stressed individuals compared to unstressed individuals. No significant changes were seen in fibrinogen and soluble TF levels [205]. These studies indicate that acute stress may promote transient changes in plasma hypercoagulability. However, more extensive studies are required to assess the mechanistic basis for these biological changes and their potential long-term impact on other haemostatic parameters [206].
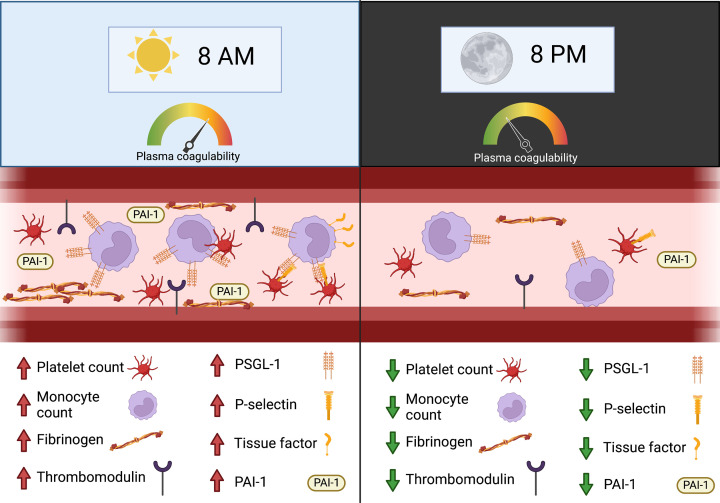
According to the 2021 edition of ‘Living and working in Europe’ [207] 19% of the EU population worked at least once during the night every month, and 21% were shift workers. Other sources of circadian disruption are social and travel-related jet lag, as well as light pollution. Circadian disruption is now well-established to disrupt normal immune cell function, and emerging data suggest a shared role in the dysregulation of haemostasis (Figure 3). The rhythmic expression of genes associated with coagulation may also contribute to the increased incidence of acute cardiovascular disease in the morning hours of the day [208,209].
Figure 3. The role of circadian variation in modulating haemostatic activity.
Levels of various procoagulant proteins, including fibrinogen and thrombomodulin, as well as fibrinolysis inhibitor PAI-1, vary with time of day, skewing the plasma and vessel surface environment in the morning towards procoagulant activity. Similarly, platelet and monocyte count increase in the morning, as is the expression of pro-adhesive cell surface receptors, such as PSGL-1. This can facilitate monocyte binding to platelet P-selectin to form aggregates, triggering increased TF activity on the monocyte surface. Collectively, circadian variation in plasma coagulation protein levels, innate immune cell number and activity, and vessel surface receptor composition may contribute to observed time-of-day effects on acute cardiovascular disease manifestation.
Circadian rhythms are controlled by a transcriptional-translational feedback loop present in almost all cell types (for detailed review, see [210]). The positive arm of the loop is controlled by transcription factors BMAL1 and CLOCK, which heterodimerise and drive the transcription of Per and Cry genes. Once phosphorylated, PER and CRY can re-enter the nucleus and act as transcriptional repressors of the BMAL1/CLOCK heterodimer, making up the negative arm of the loop. This prevents transcription of Per and Cry, reducing production of the repressors and allowing BMAL1/CLOCK to function again. Another part of the loop is formed by RORα and REV-ERBα. Their transcription is under the control of BMAL1/CLOCK and they drive or repress Bmal1 transcription, respectively. BMAL1 and CLOCK are active during daytime, while PER, CRY, RORα and REV-ERBα are active during night-time. The complete feedback loop takes 24 h to complete, giving rhythmicity to the cell and any genes under the control of these clock transcription factors, termed clock-controlled genes (CCGs). BMAL1/CLOCK bind to E-box sequences while RORα and REV-ERBα bind to RORE sequences.
Bmal1−/− mice age prematurely and lack the characteristic rhythmic expression of clock target genes [211]. Notably, several coagulation and haemostasis-related genes are CCGs, including Serpine1 [212], F7 [213,214], F12 [214] and Thbd [215]. Bmal1−/− mice exhibit a prothrombotic phenotype and clot more rapidly in a FeCl3 treatment model [216]. A shifted light schedule in mice also decreased prothrombin time, plasma fibrinogen concentration, and increased bleeding time, illustrating how coagulation changes can be driven by circadian rhythm disruption [214]. It is important to consider that mice and humans exhibit opposing circadian schedules, where mice are nocturnal and humans are diurnal, making human studies important for fully elucidating the role of circadian rhythms in coagulation. In humans, PAI-1, tPA and fibrinogen concentrations in the blood vary over 24 h [217]. PAI-1 is subject to diurnal fluctuation, independent of any day- or night-time associated behaviours [218], suggesting the capacity for thrombus formation and clearance will vary with time of day. Mouse and human neutrophils have been shown different propensities to form NETs based on time of day [219]. Mouse neutrophils isolated at night-time form more NETs than day-time neutrophils, while the opposite is seen in human neutrophils (in keeping with their opposite circadian rhythms [219]. Inflammatory (Ly6Chi) monocytes also contribute to venous thrombus formation [220] and their number is also subject to diurnal oscillations [221]. Platelet aggregation and interactions between neutrophils and monocytes is an integral arm of immunothrombosis, and their activity and distribution are also circadian. Platelet expression of activation surface markers, including GPIIb-IIIa, GpIb and P-selectin, is highest in the morning (8–9 AM). In contrast, the threshold for platelet aggregation is lowest in the evening (8 PM) [222]. Interestingly, the efficacy of the platelet inhibitor Ticagrelor (Brilinta, AstraZeneca) also varies modestly with time of day, with the lowest effectiveness at 1 PM [223]. Circadian nuclear receptor Rev-Erbα has recently been shown to have a role in platelet activation and aggregation [224]. Platelet-specific deletion of Rev-Erbα in mice and pharmacological inhibition of Rev-Erbα in human platelets showed decreased activation and aggregation by light aggregometry assays compared with untreated platelets [224]. The proposed mechanism involves GTPase activating protein Oligophrenin-1 (OPHN1), which is protected from degradation following binding to Rev-Erbα in platelets [224] and the brain [225]. Activated OPHN1 binds and activates GTPase RhoA, which is required for actin remodelling upon platelet activation [226]. Furthermore, mice whose circadian rhythms were disrupted were found to develop larger atherosclerotic lesions using a murine atherosclerotic model in which the mice were exposed to a reversed light/dark schedule. Subsequently, it was found that Bmal1 expression is altered in the foam cells in those lesions, and foam cells experience increased ER stress compared with mice in normal light conditions [227]. Collectively, these studies highlight the variance in clotting proteins and cellular components at different times of day. These studies also highlight a potential role for time of day in the effectiveness of anticoagulant and antithrombotic drugs.
New anti-immunothrombotic therapies? APC beyond sepsis
APC demonstrates potent anti-inflammatory and cytoprotective signalling properties on multiple cell types [115,228–235]. Nevertheless, APC signalling primarily occurs via activation of protease-activated receptor 1 (PAR1) when APC when bound to the EPCR [236]. Despite the failure of recombinant APC (Xigris, Eli Lily) in the treatment of severe sepsis, multiple studies have demonstrated that APC cytoprotective and anti-coagulant mechanisms can be targeted independently [237], and a variety of APC mutants altered to possess no anticoagulant activity but with cytoprotective signalling-specific activity could prove therapeutically beneficial in several inflammatory disease states. APC has been shown in many preclinical models to modulate thrombo-inflammation via regulation of both innate and adaptive immune cell signalling. For example, administration of exogenous APC ameliorates disease and induces mucosal healing by promoting barrier integrity, inhibiting leukocyte adhesion, a proinflammatory cytokine, and chemokine release in murine colitis models [114,115]. APC administration to a preclinical mouse model of asthma significantly attenuated airway inflammation via decreased neutrophil, DC and eosinophil influx, reduced type 2 cytokine levels, and IgE concentration in BAL fluid. Interestingly, these effects occurred independently of APC anticoagulant activity, indicative of a critical role for its signalling function [232,238,239]. Attempts to treat airway inflammation via FXa inhibition or by limiting TF activity resulted in severe bleeding episodes or haemorrhagic events [240–242], highlighting the challenge of traditional anticoagulant strategies to restrain the proinflammatory signalling activity of procoagulant proteases. In contrast, non-anticoagulant, signalling-selective recombinant APC variants may represent a useful strategy to address allergic airway inflammation. Administration of recombinant APC to DCs in vitro alters their population dynamics, shifting the cells from a proinflammatory conventional DC phenotype towards an anti-inflammatory plasmacytoid DC phenotype while simultaneously reducing IL-6 and IL-10 secretion [239]. As such, APC-mediated DC reprogramming renders these cells less effective in priming Th1 and Th2 cells in vitro and diminished hyperresponsiveness in an allergic airway challenge in vivo. Mice adoptively transferred with APC-treated ovalbumin-pulsed DCs exhibited significantly decreased lung concentrations of Th1 and Th2 cytokines, IFNγ and IL-5, respectively, and lower levels of immunoglobulin E (IgE) in the plasma [239].
APC can also regulate the differentiation and pathogenicity of Th17 cells. Administration of recombinant APC or PROCR deficiency inhibits the generation of Th17 cells in vitro and ameliorates Th17 mediated EAE in vivo [165,243]. Aside from inhibiting proinflammatory T-cell responses, APC also enhances anti-inflammatory Treg generation and suppressive functioning. Interestingly, administration of recombinant APC ameliorates disease in MS-like experimental autoimmune encephalomyelitis (EAE) [165,168] and APC mutants with either cytoprotective or anticoagulant mechanisms both alleviated EAE disease in mice [165]. Similarly, 3K3A-APC, a recombinant non-anticoagulant APC mutant, exerts therapeutic benefits in clinical models of AD, with reduced Aβ deposition, improved cerebrovascular integrity, and diminished neuroinflammatory responses reported [244]. 3K3A-APC also promotes BBB integrity and prevents neuronal damage during ischemic stroke in pericyte-deficient mice [228]. These studies indicate that APC treatment may benefit neurological conditions associated with pericyte loss, such as AD, dementia, and ageing.
In murine GvHD, pre-incubation of pan T-cell populations with APC before transplantation prevented the induction of disease via PAR-2/PAR-3 signalling, resulting in significantly lower levels of IFNγ, TNFα, and IL-17a plasma cytokines and reduced accumulation of IFNγ, TNFα, and IL-17+CD4+ splenic T cells when compared to non-APC treated T cell-recipient mice. Furthermore, APC-treated mice exhibited an enhanced regulatory phenotype with significantly increased numbers of FOXP3+CD4+ Treg cells in the periphery, alongside elevated levels of TGFβ and IL-10 in the plasma, compared with control mice. Mechanistic studies in vitro revealed a similar trend in human T cells, with APC incubation enhancing Treg generation and expansion, inhibiting Th1 and Th17 differentiation [233]. These studies indicate the potential use of APC in T cell immunotherapy, inhibiting T cell-mediated pathology and associated immunothrombotic effects.
Conclusions
Proinflammatory pathways overlap with and trigger many prothrombotic molecular and cellular activities, including myeloid cell activation, neutrophil NETosis, enhanced platelet reactivity and the release of procoagulant microparticles. Although well-established in sepsis, increased thrombogenicity is increasingly recognised as a common feature in disparate acute and chronic inflammatory disease settings. In addition, dysregulation of immunity caused by standard features of modern lifestyles, such as chronic stress and circadian disruption also unbalances haemostasis and understanding the biological processes underlying these changes represents a significant new challenge. Similarly, whereas an increased risk of VTE in the elderly is well-established, the growing appreciation that this may arise as a by-product of ‘inflammaging’ epitomises the emerging paradigm of immunothrombosis. Despite the increasing recognition of the prothrombotic nature of inflammatory disease, significant challenges remain in managing affected patients and new therapeutic approaches to curtail the key pathogenic steps described are urgently required.
Abbreviations
- AD
Alzheimer’s disease
- APC
activated protein C
- CLR
C-type lectin receptor
- CSF
cerebrospinal fluid
- DC
dendritic cell
- DVT
deep vein thrombosis
- EAE
experimental autoimmune encephalomyelitis
- IgE
immunoglobulin E
- NLR
Nod-like receptor
- Nod
nucleotide-binding oligomerization domain
- PAR1
protease-activated receptor 1
- PRR
pathogen recognition receptor
- PSGL-1
P-selectin glycoprotein ligand 1
- RLR
RIG I-like receptor
- ROS
reactive oxygen species
- TAFI
thrombin activatable fibrinolysis inhibitor
- TF
tissue factor
- TFPI
tissue factor pathway inhibitor
- tPA
tissue-type plasminogen activator
- VTE
venous thromboembolism
- VWF
von Willebrand factor
Data Availability
NA
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
Funding
Grant support (to R.J.S.P.) is provided by Science Foundation Ireland [grant numbers 20/COV/8511 and 21/FFP- A/8859] and the National Children’s Research Centre [grant numbers C/18/3 and PRPG/H/18/315].
CRediT Author Contribution
Paula A. Klavina: Conceptualization, Writing—original draft, Writing—review & editing. Gemma Leon: Conceptualization, Writing—original draft, Writing—review & editing. Annie M. Curtis: Conceptualization, Writing—original draft, Writing—review & editing. Roger J.S. Preston: Conceptualization, Writing—original draft, Writing—review & editing.
References
- 1.O'Donnell J.S., O'Sullivan J.M. and Preston R.J.S. (2019) Advances in understanding the molecular mechanisms that maintain normal haemostasis. Br. J. Haematol. 186, 24–36 10.1111/bjh.15872 [DOI] [PubMed] [Google Scholar]
- 2.Vu T.-K.H., Hung D.T., Wheaton V.I. and Coughlin S.R. (1991) Molecular cloning of a functional thrombin receptor reveals a novel proteolytic mechanism of receptor activation. Cell 64, 1057–1068 10.1016/0092-8674(91)90261-V [DOI] [PubMed] [Google Scholar]
- 3.Preston R.J.S., O'Sullivan J.M. and O'Donnell J.S. (2019) Advances in understanding the molecular mechanisms of venous thrombosis. Br. J. Haematol. 186, 13–23 10.1111/bjh.15869 [DOI] [PubMed] [Google Scholar]
- 4.Heestermans M., Naudin C., Mailer R.K., Konrath S., Klaetschke K., Jämsä A.et al. (2021) Identification of the factor XII contact activation site enables sensitive coagulation diagnostics. Nat. Commun. 12, 5596 10.1038/s41467-021-25888-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Grover S.P. and Mackman N. (2019) Intrinsic pathway of coagulation and thrombosis. Arterioscler. Thromb. Vasc. Biol. 39, 331–338 10.1161/ATVBAHA.118.312130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Morrissey J.H., Choi S.H. and Smith S.A. (2012) Polyphosphate: an ancient molecule that links platelets, coagulation, and inflammation. Blood 119, 5972–5979 10.1182/blood-2012-03-306605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Müller F., Mutch N.J., Schenk W.A., Smith S.A., Esterl L., Spronk H.M.et al. (2009) Platelet polyphosphates are proinflammatory and procoagulant mediators in vivo. Cell 139, 1143–1156 10.1016/j.cell.2009.11.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Iwasaki A. and Medzhitov R. (2015) Control of adaptive immunity by the innate immune system. Nat. Immunol. 16, 343–353 10.1038/ni.3123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.von Moltke J., Ayres J.S., Kofoed E.M., Chavarría-Smith J. and Vance R.E. (2013) Recognition of bacteria by inflammasomes. Annu. Rev. Immunol. 31, 73–106 10.1146/annurev-immunol-032712-095944 [DOI] [PubMed] [Google Scholar]
- 10.Gürtler C. and Bowie A.G. (2013) Innate immune detection of microbial nucleic acids. Trends Microbiol. 21, 413–420 10.1016/j.tim.2013.04.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yoneyama M., Onomoto K., Jogi M., Akaboshi T. and Fujita T. (2015) Viral RNA detection by RIG-I-like receptors. Curr. Opin. Immunol. 32, 48–53 10.1016/j.coi.2014.12.012 [DOI] [PubMed] [Google Scholar]
- 12.Rathinam V.A.K. and Fitzgerald K.A. (2011) Cytosolic surveillance and antiviral immunity. Curr. Opin. Virol. 1, 455–462 10.1016/j.coviro.2011.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Thomas C.J. and Schroder K. (2013) Pattern recognition receptor function in neutrophils. Trends Immunol. 34, 317–328 10.1016/j.it.2013.02.008 [DOI] [PubMed] [Google Scholar]
- 14.Igyártó B.Z., Haley K., Ortner D., Bobr A., Gerami-Nejad M., Edelson B.T.et al. (2011) Skin-resident murine dendritic cell subsets promote distinct and opposing antigen-specific T helper cell responses. Immunity 35, 260–272 10.1016/j.immuni.2011.06.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mashayekhi M., Sandau M.M., Dunay I.R., Frickel E.M., Khan A., Goldszmid R.S.et al. (2011) CD8α(+) dendritic cells are the critical source of interleukin-12 that controls acute infection by toxoplasma gondii tachyzoites. Immunity 35, 249–259 10.1016/j.immuni.2011.08.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Edelson B.T., Bradstreet T.R., Hildner K., Carrero J.A., Frederick K.E., Kc W.et al. (2011) CD8α(+) dendritic cells are an obligate cellular entry point for productive infection by listeria monocytogenes. Immunity 35, 236–248 10.1016/j.immuni.2011.06.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Edelson B.T., Bradstreet T.R., Kc W., Hildner K., Herzog J.W., Sim J.et al. (2011) Batf3-dependent CD11b(Low/-) peripheral dendritic cells are GM-CSF-independent and are not required for Th cell priming after subcutaneous immunization. PLoS ONE 6, e25660 10.1371/journal.pone.0025660 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hernández-Santos N. and Gaffen S.L. (2012) Th17 cells in immunity to Candida albicans. Cell Host Microbe 11, 425–435 10.1016/j.chom.2012.04.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Romani L. (2008) Cell mediated immunity to fungi: a reassessment. Med. Mycol. 46, 515–529 10.1080/13693780801971450 [DOI] [PubMed] [Google Scholar]
- 20.Murakami R., Denda-Nagai K., Hashimoto S., Nagai S., Hattori M. and Irimura T. (2013) A unique dermal dendritic cell subset that skews the immune response toward Th2. PLoS ONE 8, e73270 10.1371/journal.pone.0073270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kumamoto Y., Linehan M., Weinstein J.S., Laidlaw B.J., Craft J.E. and Iwasaki A. (2013) CD301b+ dermal dendritic cells drive T helper 2 cell-mediated immunity. Immunity 39, 733–743 10.1016/j.immuni.2013.08.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gao Y., Nish S.A., Jiang R., Hou L., Licona-Limón P., Weinstein J.S.et al. (2013) Control of T helper 2 responses by transcription factor IRF4-dependent dendritic cells. Immunity 39, 722–732 10.1016/j.immuni.2013.08.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.del Rio M.-L., Rodriguez-Barbosa J.-I., Kremmer E. and Förster R. (2007) CD103- and CD103+ bronchial lymph node dendritic cells are specialized in presenting and cross-presenting innocuous antigen to CD4+ and CD8+ T Cells. J. Immunol. Baltim. Md 1950 178, 6861–6866 [DOI] [PubMed] [Google Scholar]
- 24.Hildner K., Edelson B.T., Purtha W.E., Diamond M., Matsushita H., Kohyama M.et al. (2008) Batf3 deficiency reveals a critical role for CD8alpha+ dendritic cells in cytotoxic T cell immunity. Science 322, 1097–1100 10.1126/science.1164206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Edelson B.T., Kc W., Juang R., Kohyama M., Benoit L.A., Klekotka P.A.et al. (2010) Peripheral CD103+ dendritic cells form a unified subset developmentally related to CD8alpha+ conventional dendritic cells. J. Exp. Med. 207, 823–836 10.1084/jem.20091627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Castanheira F.V.S. and Kubes P. (2019) Neutrophils and NETs in modulating acute and chronic inflammation. Blood 133, 2178–2185 10.1182/blood-2018-11-844530 [DOI] [PubMed] [Google Scholar]
- 27.Schroder A.L., Chami B., Liu Y., Doyle C.M., El Kazzi M., Ahlenstiel G.et al. (2022) Neutrophil extracellular trap density increases with increasing histopathological severity of Crohn’s disease. Inflamm. Bowel Dis. 28, 586–598 10.1093/ibd/izab239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wolach O., Sellar R.S., Martinod K., Cherpokova D., McConkey M., Chappell R.J.et al. (2018) Increased neutrophil extracellular trap formation promotes thrombosis in myeloproliferative neoplasms. Sci. Transl. Med. 10, eaan8292 10.1126/scitranslmed.aan8292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Boeckh-Behrens T., Golkowski D., Ikenberg B., Schlegel J., Protzer U., Schulz C.et al. (2021) COVID-19-associated large vessel stroke in a 28-year-old patient : NETs and platelets possible key players in acute thrombus formation. Clin. Neuroradiol. 31, 511–514 10.1007/s00062-020-00992-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Martinod K. and Wagner D.D. (2014) Thrombosis: tangled up in NETs. Blood 123, 2768–2776 10.1182/blood-2013-10-463646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Moschonas I.C. and Tselepis A.D. (2019) The pathway of neutrophil extracellular traps towards atherosclerosis and thrombosis. Atherosclerosis 288, 9–16 10.1016/j.atherosclerosis.2019.06.919 [DOI] [PubMed] [Google Scholar]
- 32.Massberg S., Grahl L., von Bruehl M.-L., Manukyan D., Pfeiler S., Goosmann C.et al. (2010) Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat. Med. 16, 887–896 10.1038/nm.2184 [DOI] [PubMed] [Google Scholar]
- 33.Jiménez-Alcázar M., Rangaswamy C., Panda R., Bitterling J., Simsek Y.J., Long A.T.et al. (2017) Host DNases prevent vascular occlusion by neutrophil extracellular traps. Science 358, 1202–1206 10.1126/science.aam8897 [DOI] [PubMed] [Google Scholar]
- 34.Semeraro F., Ammollo C.T., Morrissey J.H., Dale G.L., Friese P., Esmon N.L.et al. (2011) Extracellular histones promote thrombin generation through platelet-dependent mechanisms: involvement of platelet TLR2 and TLR4. Blood 118, 1952–1961 10.1182/blood-2011-03-343061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang J., Wu Z., Long Q., Huang J., Hong T., Liu W.et al. (2020) Insights into immunothrombosis: the interplay among neutrophil extracellular trap, von Willebrand Factor, and ADAMTS13. Front. Immunol. 11, 10.3389/fimmu.2020.610696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ammollo C.T., Semeraro F., Xu J., Esmon N.L. and Esmon C.T. (2011) Extracellular histones increase plasma thrombin generation by impairing thrombomodulin-dependent protein C activation: histones impair TM-dependent protein C activation. J. Thromb. Haemost. 9, 1795–1803 10.1111/j.1538-7836.2011.04422.x [DOI] [PubMed] [Google Scholar]
- 37.Kawalkowska J., Quirke A.-M., Ghari F., Davis S., Subramanian V., Thompson P.R.et al. (2016) Abrogation of collagen-induced arthritis by a peptidyl arginine deiminase inhibitor is associated with modulation of T cell-mediated immune responses. Sci. Rep. 6, 26430 10.1038/srep26430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shahneh F., Christian Probst H., Wiesmann S.C., A-Gonzalez N., Ruf W., Steinbrink K.et al. (2022) Inflammatory monocyte counts determine venous blood clot formation and resolution. Arterioscler. Thromb. Vasc. Biol. 42, 145–155 10.1161/ATVBAHA.121.317176 [DOI] [PubMed] [Google Scholar]
- 39.Egorina E.M., Sovershaev M.A., Bjørkøy G., Gruber F.X.E., Olsen J.O., Parhami-Seren B.et al. (2005) Intracellular and surface distribution of monocyte tissue factor. Arterioscler. Thromb. Vasc. Biol. 25, 1493–1498 10.1161/01.ATV.0000168413.29874.d7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.von Brühl M.-L., Stark K., Steinhart A., Chandraratne S., Konrad I., Lorenz M.et al. (2012) Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J. Exp. Med. 209, 819–835 10.1084/jem.20112322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rudd K.E., Johnson S.C., Agesa K.M., Shackelford K.A., Tsoi D., Kievlan D.R.et al. (2020) Global, regional, and national sepsis incidence and mortality, 1990-2017: Analysis for the Global Burden of Disease Study. Lancet North Am. Ed. 395, 200–211 10.1016/S0140-6736(19)32989-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Álvaro-Meca A., Jiménez-Sousa M.A., Micheloud D., Sánchez-Lopez A., Heredia-Rodríguez M., Tamayo E.et al. (2018) Epidemiological Trends of Sepsis in the Twenty-First Century (2000-2013): An Analysis of Incidence, Mortality, and Associated Costs in Spain. Popul. Health Metr. 16, 4 10.1186/s12963-018-0160-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kumar G., Kumar N., Taneja A., Kaleekal T., Tarima S., McGinley E.et al. (2011) Nationwide trends of severe sepsis in the 21st century (2000-2007). Chest 140, 1223–1231 10.1378/chest.11-0352 [DOI] [PubMed] [Google Scholar]
- 44.Healy M. (2020) National sepsis report 2019. https://www.hse.ie/eng/about/who/cspd/ncps/sepsis/news/national-sepsis-report-2019.pdf [Google Scholar]
- 45.Gyawali B., Ramakrishna K. and Dhamoon A.S. (2019) Sepsis: the evolution in definition, pathophysiology, and management. SAGE Open Med. 7, 2050312119835043 10.1177/2050312119835043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Opal S.M. and van der Poll T. (2015) Endothelial barrier dysfunction in septic shock. J. Intern. Med. 277, 277–293 10.1111/joim.12331 [DOI] [PubMed] [Google Scholar]
- 47.Saito S., Uchino S., Hayakawa M., Yamakawa K., Kudo D., Iizuka Y.et al. (2019) Epidemiology of disseminated intravascular coagulation in sepsis and validation of scoring systems. J. Crit. Care 50, 23–30 10.1016/j.jcrc.2018.11.009 [DOI] [PubMed] [Google Scholar]
- 48.Gando S., Shiraishi A., Yamakawa K., Ogura H., Saitoh D., Fujishima S.et al. (2019) Role of disseminated intravascular coagulation in severe sepsis. Thromb. Res. 178, 182–188 10.1016/j.thromres.2019.04.025 [DOI] [PubMed] [Google Scholar]
- 49.Ko B.S., Cho H.Y., Ryoo S.M., Kim M.C., Jung W., Park S.H.et al. (2016) The prevalence and significance of overt disseminated intravascular coagulation in patients with septic shock in the emergency department according to the third international consensus definition. Korean J. Crit. Care Med. 31, 334–341 10.4266/kjccm.2016.00339 [DOI] [Google Scholar]
- 50.Taylor F.B., Chang A., Ruf W., Morrissey J.H., Hinshaw L., Catlett R.et al. (1991) Lethal E. coli septic shock is prevented by blocking tissue factor with monoclonal antibody. Circ. Shock 33, 127–134 [PubMed] [Google Scholar]
- 51.Creasey A.A., Chang A.C., Feigen L., Wün T.C., Taylor F.B. and Hinshaw L.B. (1993) Tissue factor pathway inhibitor reduces mortality from Escherichia coli septic shock. J. Clin. Invest. 91, 2850–2860 10.1172/JCI116529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Levi M., ten Cate H., Bauer K.A., van der Poll T., Edgington T.S., Büller H.R.et al. (1994) Inhibition of endotoxin-induced activation of coagulation and fibrinolysis by pentoxifylline or by a monoclonal anti-tissue factor antibody in chimpanzees. J. Clin. Invest. 93, 114–120 10.1172/JCI116934 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zwicker J.I., Trenor C.C., Furie B.C. and Furie B. (2011) Tissue factor-bearing microparticles and thrombus formation. Arterioscler. Thromb. Vasc. Biol. 31, 728–733 10.1161/ATVBAHA.109.200964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wu C., Lu W., Zhang Y., Zhang G., Shi X., Hisada Y.et al. (2019) Inflammasome activation triggers blood clotting and host death through pyroptosis. Immunity 50, 1401.e4–1411.e4 10.1016/j.immuni.2019.04.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yang X., Cheng X., Tang Y., Qiu X., Wang Y., Kang H.et al. (2019) Bacterial endotoxin activates the coagulation cascade through gasdermin D-dependent phosphatidylserine exposure. Immunity 51, 983.e6–996.e6 10.1016/j.immuni.2019.11.005 [DOI] [PubMed] [Google Scholar]
- 56.Zhang Y., Cui J., Zhang G., Wu C., Abdel-Latif A., Smyth S.S.et al. (2021) Inflammasome activation promotes venous thrombosis through pyroptosis. Blood Adv. 5, 2619–2623 10.1182/bloodadvances.2020003041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Denorme F., Vanhoorelbeke K. and De Meyer S.F. (2019) Von Willebrand factor and platelet glycoprotein Ib: a thromboinflammatory axis in stroke. Front. Immunol. 10, 2884 10.3389/fimmu.2019.02884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Levy G.G., Nichols W.C., Lian E.C., Foroud T., McClintick J.N., McGee B.M.et al. (2001) Mutations in a member of the ADAMTS gene family cause thrombotic thrombocytopenic purpura. Nature 413, 488–494 10.1038/35097008 [DOI] [PubMed] [Google Scholar]
- 59.Pendu R., Terraube V., Christophe O.D., Gahmberg C.G., de Groot P.G., Lenting P.J.et al. (2006) P-selectin glycoprotein ligand 1 and Β2-integrins cooperate in the adhesion of leukocytes to von Willebrand factor. Blood 108, 3746–3752 10.1182/blood-2006-03-010322 [DOI] [PubMed] [Google Scholar]
- 60.Rautiainen L., Cirko A., Pavare J., Balmaks R., Grope I., Katirlo I.et al. (2019) Assessment of ADAMTS-13 level in hospitalized children with serious bacterial infections as a possible prognostic marker. Medicina (Mex) 55, 503 10.3390/medicina55080503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Smeets N.J.L., Fijnheer R., Sebastian S. and De Mast Q. (2018) Secondary thrombotic microangiopathy with severely reduced ADAMTS13 activity in a patient with capnocytophaga canimorsus sepsis: a case report. Transfusion (Paris) 58, 2426–2429 10.1111/trf.14829 [DOI] [PubMed] [Google Scholar]
- 62.Peetermans M., Meyers S., Liesenborghs L., Vanhoorelbeke K., De Meyer S.F., Vandenbriele C.et al. (2020) Von Willebrand factor and ADAMTS13 impact on the outcome of Staphylococcus aureus sepsis. J. Thromb. Haemost. 18, 722–731 10.1111/jth.14686 [DOI] [PubMed] [Google Scholar]
- 63.Czaikoski P.G., Mota J.M.S.C., Nascimento D.C., Sônego F., e Silva Castanheira F.V., Melo P.H.et al. (2016) Neutrophil extracellular traps induce organ damage during experimental and clinical sepsis. PLoS ONE 11, e0148142 10.1371/journal.pone.0148142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Stiel L., Mayeur-Rousse C., Helms J., Meziani F. and Mauvieux L. (2019) First visualization of circulating neutrophil extracellular traps using cell fluorescence during human septic shock-induced disseminated intravascular coagulation. Thromb. Res. 183, 153–158 10.1016/j.thromres.2019.09.036 [DOI] [PubMed] [Google Scholar]
- 65.Mao J.-Y., Zhang J.-H., Cheng W., Chen J.-W. and Cui N. (2021) Effects of neutrophil extracellular traps in patients with septic coagulopathy and their interaction with autophagy. Front. Immunol. 12, 10.3389/fimmu.2021.757041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Colón D.F., Wanderley C.W., Franchin M., Silva C.M., Hiroki C.H., Castanheira F.V.S.et al. (2019) Neutrophil extracellular traps (NETs) exacerbate severity of infant sepsis. Crit. Care 23, 113 10.1186/s13054-019-2407-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Shorr A.F., Bernard G.R., Dhainaut J.-F., Russell J.R., Macias W.L., Nelson D.R.et al. (2006) Protein C concentrations in severe sepsis: an early directional change in plasma levels predicts outcome. Crit. Care 10, R92 10.1186/cc4946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bernard G.R., Vincent J.-L., Laterre P.-F., LaRosa S.P., Dhainaut J.-F., Lopez-Rodriguez A.et al. (2001) Efficacy and safety of recombinant human activated protein C for severe sepsis. N. Engl. J. Med. 344, 699–709 10.1056/NEJM200103083441001 [DOI] [PubMed] [Google Scholar]
- 69.Martí‐Carvajal A.J., Solà I., Gluud C., Lathyris D. and Anand V. (2012) Human recombinant protein C for severe sepsis and septic shock in adult and paediatric patients. Cochrane Database Syst. Rev. 12, 10.1002/14651858.CD004388.pub5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kurosawa S., Stearns-Kurosawa D.J., Carson C.W., Angelo A.D., Della Valle P. and Esmon C.T. (1998) Plasma levels of endothelial cell protein C receptor are elevated in patients with sepsis and systemic lupus erythematosus: lack of correlation with thrombomodulin suggests involvement of different pathological processes. Blood 91, 725–727 10.1182/blood.V91.2.725 [DOI] [PubMed] [Google Scholar]
- 71.Watanabe E., Takasu O., Teratake Y., Sakamoto T., Ikeda T., Kotani J.et al. (2022) A thrombomodulin promoter gene polymorphism, Rs2239562, Influences both susceptibility to and outcome of sepsis. Front. Med. 8, 10.3389/fmed.2021.762198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Moxon C.A., Alhamdi Y., Storm J., Toh J.M.H., McGuinness D., Ko J.Y.et al. (2020) Parasite histones are toxic to brain endothelium and link blood barrier breakdown and thrombosis in cerebral malaria. Blood Adv. 4, 2851–2864 10.1182/bloodadvances.2019001258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Riedl J., Mordmüller B., Koder S., Pabinger I., Kremsner P.G., Hoffman S.L.et al. (2016) Alterations of blood coagulation in controlled human malaria infection. Malar. J. 15, 15 10.1186/s12936-015-1079-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Bridges D.J., Bunn J., van Mourik J.A., Grau G., Preston R.J.S., Molyneux M.et al. (2010) Rapid activation of endothelial cells enables plasmodium falciparum adhesion to platelet-decorated von willebrand factor strings. Blood 115, 1472–1474 10.1182/blood-2009-07-235150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Helms J., Tacquard C., Severac F., Leonard-Lorant I., Ohana M., Delabranche X.et al. (2020) High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med. 46, 1089–1098 10.1007/s00134-020-06062-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Middeldorp S., Coppens M., van Haaps T.F., Foppen M., Vlaar A.P., Müller M.C.A.et al. (2020) Incidence of venous thromboembolism in hospitalized patients with COVID-19. J. Thromb. Haemost. 18, 1995–2002 10.1111/jth.14888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Hill J.B., Garcia D., Crowther M., Savage B., Peress S., Chang K.et al. (2020) Frequency of venous thromboembolism in 6513 patients with COVID-19: a retrospective study. Blood Adv. 4, 5373–5377 10.1182/bloodadvances.2020003083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Piazza G., Campia U., Hurwitz S., Snyder J.E., Rizzo S.M., Pfeferman M.B.et al. (2020) Registry of arterial and venous thromboembolic complications in patients with COVID-19. J. Am. Coll. Cardiol. 76, 2060–2072 10.1016/j.jacc.2020.08.070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Liu H., Hu T., Zhang C., Chen X., Zhang S., Li M.et al. (2021) Mechanisms of COVID-19 thrombosis in an inflammatory environment and new anticoagulant targets. Am. J. Transl. Res. 13, 3925–3941 [PMC free article] [PubMed] [Google Scholar]
- 80.Zuo Y., Yalavarthi S., Shi H., Gockman K., Zuo M., Madison J.A.et al. (2020) Neutrophil extracellular traps in COVID-19. JCI Insight 5, 10.1172/jci.insight.138999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rohlfing A.K., Rath D., Geisler T. and Gawaz M. (2021) Platelets and COVID-19. Hamostaseologie 41, 379–385 10.1055/a-1581-4355 [DOI] [PubMed] [Google Scholar]
- 82.Langnau C., Rohlfing A.K., Gekeler S., Günter M., Pöschel S., Petersen-Uribe Á.et al. (2021) Platelet activation and plasma levels of furin are associated with prognosis of patients with coronary artery disease and COVID-19. Arter. Thromb. Vasc. Biol. 41, 2080–2096 10.1161/ATVBAHA.120.315698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Borges do Nascimento I.J., von Groote T.C., O'Mathúna D.P., Abdulazeem H.M., Henderson C., Jayarajah U.et al. (2020) Clinical, laboratory and radiological characteristics and outcomes of novel coronavirus (SARS-CoV-2) infection in humans: a systematic review and series of meta-analyses. PLoS ONE 15, e0239235 10.1371/journal.pone.0239235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wang C., Yu C., Jing H., Wu X., Novakovic V.A., Xie R.et al. (2022) long covid: The Nature of Thrombotic Sequelae Determines the Necessity of Early Anticoagulation. Front. Cell Infect. Microbiol. 12, 861703 10.3389/fcimb.2022.861703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Fogarty H., Ward S.E., Townsend L., Karampini E., Elliott S., Conlon N.et al. (2022) Sustained VWF‐ADAMTS‐13 Axis Imbalance and Endotheliopathy in Long covid Syndrome Is Related to Immune Dysfunction. J. Thromb. Haemost. jth.15830 10.1111/jth.15830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Luther N., Shahneh F., Brähler M., Krebs F., Jäckel S., Subramaniam S.et al. (2016) Innate effector-memory T-cell activation regulates post-thrombotic vein wall inflammation and thrombus resolution. Circ. Res. 119, 1286–1295 10.1161/CIRCRESAHA.116.309301 [DOI] [PubMed] [Google Scholar]
- 87.Nosaka M., Ishida Y., Kimura A., Kuninaka Y., Inui M., Mukaida N.et al. (2011) Absence of IFN-γ accelerates thrombus resolution through enhanced MMP-9 and VEGF Expression in mice. J. Clin. Invest. 121, 2911–2920 10.1172/JCI40782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Shahneh F., Grill A., Klein M., Frauhammer F., Bopp T., Schäfer K.et al. (2021) Specialized regulatory T cells control venous blood clot resolution through SPARC. Blood 137, 1517–1526 10.1182/blood.2020005407 [DOI] [PubMed] [Google Scholar]
- 89.Risk of Venous and Arterial Thromboembolic Events in Patients Receiving Targeted Anti-cancer Therapy - A Nationwide Cohort Study ISTH Congress Abstracts. https://abstracts.isth.org/abstract/risk-of-venous-and-arterial-thromboembolic-events-in-patients-receiving-targeted-anti-cancer-therapy-a-nationwide-cohort-study/ accessed 2022-11-02) [Google Scholar]
- 90.Wang T.-F., Khorana A.A. and Carrier M. (2021) Thrombotic complications associated with immune checkpoint inhibitors. Cancers 13, 4606 10.3390/cancers13184606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Wang L., Wang F.S. and Gershwin M.E. (2015) Human Autoimmune diseases: a comprehensive update. J. Intern. Med. 278, 369–395 10.1111/joim.12395 [DOI] [PubMed] [Google Scholar]
- 92.Garcia P. (2018) Are you autoimmune aware? www.JDRF.org.uk Accessed: 30/08/2022 [Google Scholar]
- 93.Ng S.C., Shi H.Y., Hamidi N., Underwood F.E., Tang W., Benchimol E.I.et al. (2017) Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet Lond. Engl. 390, 2769–2778 10.1016/S0140-6736(17)32448-0 [DOI] [PubMed] [Google Scholar]
- 94.Rosen M.J., Dhawan A. and Saeed S.A. (2015) Inflammatory bowel disease in children and adolescents. JAMA Pediatr. 169, 1053–1060 10.1001/jamapediatrics.2015.1982 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Benchimol E.I., Bernstein C.N., Bitton A., Carroll M.W., Singh H., Otley A.R.et al. (2017) Trends in epidemiology of pediatric inflammatory bowel disease in Canada: distributed network analysis of multiple population-based provincial health administrative databases. Am. J. Gastroenterol. 112, 1120–1134 10.1038/ajg.2017.97 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Naito T., Botwin G.J., Haritunians T., Li D., Yang S., Khrom M.et al. (2021) Prevalence and effect of genetic risk of thromboembolic disease in inflammatory bowel disease. Gastroenterology 160, 771.e4–780.e4 10.1053/j.gastro.2020.10.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kappelman M.D., Horvath-Puho E., Sandler R.S., Rubin D.T., Ullman T.A., Pedersen L.et al. (2011) Thromboembolic risk among Danish children and adults with inflammatory bowel diseases: a population-based nationwide study. Gut 60, 937–943 10.1136/gut.2010.228585 [DOI] [PubMed] [Google Scholar]
- 98.Grainge M.J., West J. and Card T.R. (2010) Venous thromboembolism during active disease and remission in inflammatory bowel disease: a cohort study. Lancet 375, 657–663 10.1016/S0140-6736(09)61963-2 [DOI] [PubMed] [Google Scholar]
- 99.Lee S.H., Kwon J.E. and Cho M.L. (2018) Immunological pathogenesis of inflammatory bowel disease. Intest. Res. 16, 26–42 10.5217/ir.2018.16.1.26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Medina-Contreras O., Harusato A., Nishio H., Flannigan K.L., Ngo V., Leoni G.et al. (2016) Cutting edge: IL-36 receptor promotes resolution of intestinal damage. J. Immunol. 196, 34–38 10.4049/jimmunol.1501312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Leon G., Hernandez Santana Y.E., Irwin N., Giannoudaki E., O'Neill S., Csizmadia I.et al. (2022) IL-36 cytokines imprint a colitogenic phenotype on CD4+ T helper cells. Mucosal. Immunol. 15, 491–503 10.1038/s41385-022-00488-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Jones G.R., Bain C.C., Fenton T.M., Kelly A., Brown S.L., Ivens A.C.et al. (2018) Dynamics of colon monocyte and macrophage activation during colitis. Front. Immunol. 9, 2764 10.3389/fimmu.2018.02764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Alkim H., Ayaz S., Alkim C., Ulker A. and Sahin B. (2011) Continuous active state of coagulation system in patients with nonthrombotic inflammatory bowel disease. Clin. Appl. Thromb. Hemost. 17, 600–604 10.1177/1076029611405034 [DOI] [PubMed] [Google Scholar]
- 104.Kume K., Yamasaki M., Tashiro M., Yoshikawa I. and Otsuki M. (2007) Activations of coagulation and fibrinolysis secondary to bowel inflammation in patients with ulcerative colitis. Intern. Med. 46, 1323–1329 10.2169/internalmedicine.46.0237 [DOI] [PubMed] [Google Scholar]
- 105.Cakal B., Gokmen A., Yalinkilic M., Cakal E., Ayaz S., Nadir I.et al. (2010) Natural anticoagulant protein levels in Turkish patients with inflammatory bowel disease. Blood Coagul. Fibrinolysis 21, 118–121 10.1097/MBC.0b013e328335d025 [DOI] [PubMed] [Google Scholar]
- 106.Dolapcioglu C., Soylu A., Kendir T., Ince A.T., Dolapcioglu H., Purisa S.et al. (2014) Coagulation parameters in inflammatory bowel disease. Int. J. Clin. Exp. Med. 7, 1442–1448 [PMC free article] [PubMed] [Google Scholar]
- 107.Anthoni C., Russell J., Wood K.C., Stokes K.Y., Vowinkel T., Kirchhofer D.et al. (2007) Tissue factor: a mediator of inflammatory cell recruitment, tissue injury, and thrombus formation in experimental colitis. J. Exp. Med. 204, 1595–1601 10.1084/jem.20062354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Palkovits J., Novacek G., Kollars M., Hron G., Osterode W., Quehenberger P.et al. (2013) Tissue factor exposing microparticles in inflammatory bowel disease. J. Crohns. Colitis 7, 222–229 10.1016/j.crohns.2012.05.016 [DOI] [PubMed] [Google Scholar]
- 109.Deutschmann A., Schlagenhauf A., Leschnik B., Hoffmann K.M., Hauer A. and Muntean W. (2013) Increased procoagulant function of microparticles in pediatric inflammatory bowel disease: role in increased thrombin generation. J. Pediatr. Gastroenterol. Nutr. 56, 401–407 10.1097/MPG.0b013e31827daf72 [DOI] [PubMed] [Google Scholar]
- 110.Moschonas I. and Tselepis A. (2018) Platelet-derived microparticles induce the formation of neutrophil extracellular traps. Atherosclerosis 275, e106 10.1016/j.atherosclerosis.2018.06.291 [DOI] [Google Scholar]
- 111.Wang Y., Luo L., Braun O., Westman J., Madhi R., Herwald H.et al. (2018) Neutrophil extracellular trap-microparticle complexes enhance thrombin generation via the intrinsic pathway of coagulation in mice. Sci. Rep. 8, 4020 10.1038/s41598-018-22156-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Collins C.E., Cahill M.R., Newland A.C. and Rampton D.S. (1994) Platelets circulate in an activated state in inflammatory bowel disease. Gastroenterology 106, 840–845 10.1016/0016-5085(94)90741-2 [DOI] [PubMed] [Google Scholar]
- 113.Harries A.D., Fitzsimons E., Fifield R., Dew M.J. and Rhoades J. (1983) Platelet count: a simple measure of activity in Crohn’s disease. Br. Med. J. (Clin. Res. Ed) 286, 1476 10.1136/bmj.286.6376.1476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Vetrano S., Ploplis V.A., Sala E., Sandoval-Cooper M., Donahue D.L., Correale C.et al. (2011) Unexpected role of anticoagulant protein C in controlling epithelial barrier integrity and intestinal inflammation. Proc. Natl. Acad. Sci. U.S.A. 108, 19830–19835 10.1073/pnas.1107140108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Scaldaferri F., Sans M., Vetrano S., Graziani C., De Cristofaro R., Gerlitz B.et al. (2007) Crucial role of the protein C pathway in governing microvascular inflammation in inflammatory bowel disease. J. Clin. Invest. 117, 1951–1960 10.1172/JCI31027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lay A.J., Liang Z., Rosen E.D. and Castellino F.J. (2005) Mice with a severe deficiency in protein C display prothrombotic and proinflammatory phenotypes and compromised maternal reproductive capabilities. J. Clin. Invest. 115, 1552–1561 10.1172/JCI24030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Kondreddy V., Keshava S., Esmon C.T., Pendurthi U.R. and Rao L.V.M. (2020) A critical role of endothelial cell protein C receptor in the intestinal homeostasis in experimental colitis. Sci. Rep. 10, 20569 10.1038/s41598-020-77502-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Sarlos P., Szemes K., Hegyi P., Garami A., Szabo I., Illes A.et al. (2018) Steroid but not biological therapy elevates the risk of venous thromboembolic events in inflammatory bowel disease: a meta-analysis. J. Crohns. Colitis 12, 489–498 10.1093/ecco-jcc/jjx162 [DOI] [PubMed] [Google Scholar]
- 119.Poizeau F., Nowak E., Kerbrat S., Le Nautout B., Droitcourt C., Drici M.-D.et al. (2020) Association between early severe cardiovascular events and the initiation of treatment with the anti-interleukin 12/23p40 antibody ustekinumab. JAMA Dermatol. 156, 1208–1215 10.1001/jamadermatol.2020.2977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Levine I., Rolston V., Papademetriou M. and Malter L. (2018) Ustekinumab-associated thrombocytopenia in a patient with refractory Crohn’s disease: 2044. Off. J. Am. Coll. Gastroenterol. ACG 113, S1166 10.14309/00000434-201810001-02044 [DOI] [Google Scholar]
- 121.Detrez I., Ballet V., Peeters M., Van Assche G., Vermeire S., Ferrante M.et al. (2018) P484 infliximab and vedolizumab show a different effect on clot formation in inflammatory bowel disease patients. J. Crohns Colitis 12, S349–S350 10.1093/ecco-jcc/jjx180.611 [DOI] [Google Scholar]
- 122.Kekre N., Kim H.T., Ho V.T., Cutler C., Armand P., Nikiforow S.et al. (2017) Venous thromboembolism is associated with graft-. Haematologica 102, 1185–1191 10.3324/haematol.2017.164012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.de Lima M., Anagnostopoulos A., Munsell M., Shahjahan M., Ueno N., Ippoliti C.et al. (2004) Nonablative versus reduced-intensity conditioning regimens in the treatment of acute myeloid leukemia and high-risk myelodysplastic syndrome: dose is relevant for long-term disease control after allogeneic hematopoietic stem cell transplantation. Blood 104, 865–872 10.1182/blood-2003-11-3750 [DOI] [PubMed] [Google Scholar]
- 124.Russell J.A., Duan Q., Chaudhry M.A., Savoie M.L., Balogh A., Turner A.R.et al. (2008) Transplantation from matched siblings using once-daily intravenous busulfan/fludarabine with thymoglobulin: a myeloablative regimen with low nonrelapse mortality in all but older patients with high-risk disease. Biol. Blood Marrow Transpl. 14, 888–895 10.1016/j.bbmt.2008.05.010 [DOI] [PubMed] [Google Scholar]
- 125.Maris M.B., Sandmaier B.M., Storer B.E., Maloney D.G., Shizuru J.A., Agura E.et al. (2006) Unrelated donor granulocyte colony-stimulating factor-mobilized peripheral blood mononuclear cell transplantation after nonmyeloablative conditioning: the effect of postgrafting mycophenolate mofetil dosing. Biol. Blood Marrow Transpl. 12, 454–465 10.1016/j.bbmt.2005.12.030 [DOI] [PubMed] [Google Scholar]
- 126.Gonsalves A., Carrier M., Wells P.S., McDiarmid S.A., Huebsch L.B. and Allan D.S. (2008) Incidence of symptomatic venous thromboembolism following hematopoietic stem cell transplantation. J. Thromb. Haemost. 6, 1468–1473 10.1111/j.1538-7836.2008.03077.x [DOI] [PubMed] [Google Scholar]
- 127.Azık F., Gökçebay D.G., Tavil B., Işık P., Tunç B. and Uçkan D. (2015) Venous thromboembolism after allogeneic pediatric hematopoietic stem cell transplantation: a single-center study. Turk J. Haematol. 32, 228–233 10.4274/tjh.2013.0066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Labrador J., González-Rivero J., Monroy R., Lozano F.S., López-Corral L., Caballero M.D.et al. (2016) Management patterns and outcomes in symptomatic venous thromboembolism following allogeneic hematopoietic stem cell transplantation. A 15-year experience at a single center. Thromb. Res. 142, 52–56 10.1016/j.thromres.2016.02.016 [DOI] [PubMed] [Google Scholar]
- 129.Gerber D.E., Segal J.B., Levy M.Y., Kane J., Jones R.J. and Streiff M.B. (2008) The incidence of and risk factors for venous thromboembolism (VTE) and bleeding among 1514 patients undergoing hematopoietic stem cell transplantation: implications for VTE prevention. Blood 112, 504–510 10.1182/blood-2007-10-117051 [DOI] [PubMed] [Google Scholar]
- 130.El Jurdi N., Elhusseini H., Beckman J., DeFor T.E., Okoev G., Rogosheske J.et al. (2021) High incidence of thromboembolism in patients with chronic GVHD: association with severity of GVHD and donor-recipient ABO blood group. Blood Cancer J. 11, 96 10.1038/s41408-021-00488-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Laskin B.L., Goebel J., Davies S.M. and Jodele S. (2011) Small vessels, big trouble in the kidneys and beyond: hematopoietic stem cell transplantation-associated thrombotic microangiopathy. Blood 118, 1452–1462 10.1182/blood-2011-02-321315 [DOI] [PubMed] [Google Scholar]
- 132.Gloude N.J., Khandelwal P., Luebbering N., Lounder D.T., Jodele S., Alder M.N.et al. (2017) Circulating DsDNA, endothelial injury, and complement activation in thrombotic microangiopathy and GVHD. Blood 130, 1259–1266 10.1182/blood-2017-05-782870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Amoroso A., Mitterhofer A.P., Del Porto F., Garzia P., Ferri G.M., Galluzzo S.et al. (2003) Antibodies to anionic phospholipids and anti-beta2-GPI: association with thrombosis and thrombocytopenia in systemic lupus erythematosus. Hum. Immunol. 64, 265–273 10.1016/S0198-8859(02)00789-9 [DOI] [PubMed] [Google Scholar]
- 134.Love P.E. and Santoro S.A. (1990) Antiphospholipid antibodies: anticardiolipin and the lupus anticoagulant in systemic lupus erythematosus (SLE) and in non-SLE disorders. prevalence and clinical significance. Ann. Intern. Med. 112, 682–698 10.7326/0003-4819-112-9-682 [DOI] [PubMed] [Google Scholar]
- 135.Gustafsson J., Gunnarsson I., Börjesson O., Pettersson S., Möller S., Fei G.Z.et al. (2009) Predictors of the first cardiovascular event in patients with systemic lupus erythematosus - a prospective cohort study. Arthritis Res. Ther. 11, R186 10.1186/ar2878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Noordermeer T., Molhoek J.E., Schutgens R.E.G., Sebastian S.A.E., Drost-Verhoef S., van Wesel A.C.W.et al. (2021) Anti-Β2-glycoprotein I and anti-prothrombin antibodies cause lupus anticoagulant through different mechanisms of action. J. Thromb. Haemost. 19, 1018–1028 10.1111/jth.15241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ohl K. and Tenbrock K. (2011) Inflammatory cytokines in systemic lupus erythematosus. J. Biomed. Biotechnol. 2011, 432595 10.1155/2011/432595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Rauch J., Salem D., Subang R., Kuwana M. and Levine J.S. (2018) Β2-glycoprotein I-reactive T cells in autoimmune disease. Front. Immunol. 9, 2836 10.3389/fimmu.2018.02836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Müller-Calleja N., Hollerbach A., Royce J., Ritter S., Pedrosa D., Madhusudhan T.et al. (2021) Lipid presentation by the protein c receptor links coagulation with autoimmunity. Science 371, 10.1126/science.abc0956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Takahagi S., Mihara S., Iwamoto K., Morioke S., Okabe T., Kameyoshi Y.et al. (2010) Coagulation/fibrinolysis and inflammation markers are associated with disease activity in patients with chronic urticaria. Allergy 65, 649–656 10.1111/j.1398-9995.2009.02222.x [DOI] [PubMed] [Google Scholar]
- 141.Yanase Y., Takahagi S. and Hide M. (2018) Chronic spontaneous urticaria and the extrinsic coagulation system. Allergol. Int. 67, 191–194 10.1016/j.alit.2017.09.003 [DOI] [PubMed] [Google Scholar]
- 142.Xue M., Lin H., Zhao R., Fryer C., March L. and Jackson C.J. (2022) Activated protein C protects against murine contact dermatitis by suppressing protease-activated receptor 2. Int. J. Mol. Sci. 23, 10.3390/ijms23010516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Brims F.J., Chauhan A.J., Higgins B. and Shute J.K. (2009) Coagulation factors in the airways in moderate and severe asthma and the effect of inhaled steroids. Thorax 64, 1037–1043 10.1136/thx.2009.114439 [DOI] [PubMed] [Google Scholar]
- 144.Chung W.S., Lin C.L., Ho F.M., Li R.Y., Sung F.C., Kao C.H.et al. (2014) Asthma increases pulmonary thromboembolism risk: a nationwide population cohort study. Eur. Respir. J. 43, 801–807 10.1183/09031936.00043313 [DOI] [PubMed] [Google Scholar]
- 145.Mitchel J.A., Antoniak S., Lee J.H., Kim S.H., McGill M., Kasahara D.I.et al. (2016) IL-13 augments compressive stress-induced tissue factor expression in human airway epithelial cells. Am. J. Respir. Cell Mol. Biol. 54, 524–531 10.1165/rcmb.2015-0252OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Shinagawa K., Martin J.A., Ploplis V.A. and Castellino F.J. (2007) Coagulation factor Xa modulates airway remodeling in a murine model of asthma. Am. J. Respir. Crit. Care Med. 175, 136–143 10.1164/rccm.200608-1097OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Blanc-Brude O.P., Archer F., Leoni P., Derian C., Bolsover S., Laurent G.J.et al. (2005) Factor Xa Stimulates fibroblast procollagen production, proliferation, and calcium signaling via PAR1 activation. Exp. Cell. Res. 304, 16–27 10.1016/j.yexcr.2004.10.021 [DOI] [PubMed] [Google Scholar]
- 148.Reed C.E. and Kita H. (2004) The role of protease activation of inflammation in allergic respiratory diseases. J. Allergy Clin. Immunol. 114, 997–1008, quiz 1009 10.1016/j.jaci.2004.07.060 [DOI] [PubMed] [Google Scholar]
- 149.Schouten M., van de Pol M.A., Levi M., van der Poll T. and van der Zee J.S. (2009) Early activation of coagulation after allergen challenge in patients with allergic asthma. J. Thromb. Haemost. 7, 1592–1594 10.1111/j.1538-7836.2009.03523.x [DOI] [PubMed] [Google Scholar]
- 150.Hataji O., Taguchi O., Gabazza E.C., Yuda H., Fujimoto H., Suzuki K.et al. (2002) Activation of protein C pathway in the airways. Lung 180, 47–59 10.1007/s004080000080 [DOI] [PubMed] [Google Scholar]
- 151.Kornerup K.N. and Page C.P. (2007) The role of platelets in the pathophysiology of asthma. Platelets 18, 319–328 10.1080/09537100701230436 [DOI] [PubMed] [Google Scholar]
- 152.Jennewein C., Tran N., Paulus P., Ellinghaus P., Eble J.A. and Zacharowski K. (2011) Novel aspects of fibrin(ogen) fragments during inflammation. Mol. Med. 17, 568–573 10.2119/molmed.2010.00146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Levi M., Schultz M.J., Rijneveld A.W. and van der Poll T. (2003) Bronchoalveolar coagulation and fibrinolysis in endotoxemia and pneumonia. Crit. Care Med. 31, S238–S242 10.1097/01.CCM.0000057849.53689.65 [DOI] [PubMed] [Google Scholar]
- 154.Robson S.C., Shephard E.G. and Kirsch R.E. (1994) Fibrin degradation product D-dimer induces the synthesis and release of biologically active IL-1 beta, IL-6 and plasminogen activator inhibitors from monocytes in vitro. Br. J. Haematol. 86, 322–326 10.1111/j.1365-2141.1994.tb04733.x [DOI] [PubMed] [Google Scholar]
- 155.Lloyd-Lavery A., Solman L., Grindlay D.J.C., Rogers N.K., Thomas K.S. and Harman K.E. (2019) What’s new in atopic eczema? An analysis of systematic reviews published in 2016. Part 2: epidemiology, aetiology and risk factors Clin. Exp. Dermatol. 44, 370–375 10.1111/ced.13853 [DOI] [PubMed] [Google Scholar]
- 156.Nassau S. and Fonacier L. (2020) Allergic contact dermatitis. Med. Clin. North Am. 104, 61–76 10.1016/j.mcna.2019.08.012 [DOI] [PubMed] [Google Scholar]
- 157.Gittler J.K., Shemer A., Suárez-Fariñas M., Fuentes-Duculan J., Gulewicz K.J., Wang C.Q.et al. (2012) Progressive activation of T(H)2/T(H)22 cytokines and selective epidermal proteins characterizes acute and chronic atopic dermatitis. J. Allergy Clin. Immunol. 130, 1344–1354 10.1016/j.jaci.2012.07.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Saito R., Yanase Y., Kamegashira A., Takahagi S., Tanaka A., Uchida K.et al. (2020) Increase of tissue factor expression on the surface of peripheral monocytes of patients with chronic spontaneous urticaria. Allergy 75, 971–974 10.1111/all.14110 [DOI] [PubMed] [Google Scholar]
- 159.Yanase Y., Matsuo Y., Takahagi S., Kawaguchi T., Uchida K., Ishii K.et al. (2021) Coagulation factors induce human skin mast cell and basophil degranulation via activation of complement 5 and the C5a receptor. J. Allergy Clin. Immunol. 147, 1101.e7–1104.e7 10.1016/j.jaci.2020.08.018 [DOI] [PubMed] [Google Scholar]
- 160.Sherenian M., Filuta A., Amezcua P., Ruff B., Kroner J., Grashel B.et al. (2022) Thrombin and fibrinogen play a critical role in atopic dermatitis pathogenesis. J. Allergy Clin. Immunol. 149, AB6 10.1016/j.jaci.2021.12.060 [DOI] [Google Scholar]
- 161.Yadav S.K., Mindur J.E., Ito K. and Dhib-Jalbut S. (2015) Advances in the immunopathogenesis of multiple sclerosis. Curr. Opin. Neurol. 28, 206–219 10.1097/WCO.0000000000000205 [DOI] [PubMed] [Google Scholar]
- 162.Ahmed O., Geraldes R., DeLuca G.C. and Palace J. (2019) Multiple sclerosis and the risk of systemic venous thrombosis: a systematic review. Mult. Scler Relat. Disord. 27, 424–430 10.1016/j.msard.2018.10.008 [DOI] [PubMed] [Google Scholar]
- 163.Putnam T.J. (1937) Lesions of “encephalomyelitis” and multiple sclerosis: venous thrombosis as the primary alteration. J. Am. Med. Assoc. 108, 1477–1480 10.1001/jama.1937.02780180001001 [DOI] [Google Scholar]
- 164.Putnam T. (1936) Studies in multiple sclerosis. Ann. Intern. Med. 9, 854–863 10.7326/0003-4819-9-7-854 [DOI] [Google Scholar]
- 165.Han M.H., Hwang S.I., Roy D.B., Lundgren D.H., Price J.V., Ousman S.S.et al. (2008) Proteomic analysis of active multiple sclerosis lesions reveals therapeutic targets. Nature 451, 1076–1081 10.1038/nature06559 [DOI] [PubMed] [Google Scholar]
- 166.Cuzner M.L., Gveric D., Strand C., Loughlin A.J., Paemen L., Opdenakker G.et al. (1996) The expression of tissue-type plasminogen activator, matrix metalloproteases and endogenous inhibitors in the central nervous system in multiple sclerosis: comparison of stages in lesion evolution. J. Neuropathol. Exp. Neurol. 55, 1194–1204 10.1097/00005072-199612000-00002 [DOI] [PubMed] [Google Scholar]
- 167.Gveric D., Herrera B., Petzold A., Lawrence D.A. and Cuzner M.L. (2003) Impaired fibrinolysis in multiple sclerosis: a role for tissue plasminogen activator inhibitors. Brain 126, 1590–1598 10.1093/brain/awg167 [DOI] [PubMed] [Google Scholar]
- 168.Kohriyama T., Maruyama H., Kurokawa K., Harada T. and Nakamura S. (1997) Endothelial cell activation and/or injury in multiple sclerosis: analysis with von Willebrand factor and thrombomodulin. Rinsho Shinkeigaku 37, 287–291 [PubMed] [Google Scholar]
- 169.Tsukada N., Matsuda M., Miyagi K. and Yanagisawa N. (1995) Thrombomodulin in the sera of patients with multiple sclerosis and human lymphotropic virus type-1-associated myelopathy. J. Neuroimmunol. 56, 113–116 10.1016/0165-5728(94)00156-I [DOI] [PubMed] [Google Scholar]
- 170.Ryu J.K., Rafalski V.A., Meyer-Franke A., Adams R.A., Poda S.B., Rios Coronado P.E.et al. (2018) Fibrin-targeting immunotherapy protects against neuroinflammation and neurodegeneration. Nat. Immunol. 19, 1212–1223 10.1038/s41590-018-0232-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Göbel K., Pankratz S., Asaridou C.M., Herrmann A.M., Bittner S., Merker M.et al. (2016) Blood coagulation factor XII drives adaptive immunity during neuroinflammation via CD87-Mediated Modulation of Dendritic Cells. Nat Commun 7, 11626 10.1038/ncomms11626 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Ziliotto N., Baroni M., Straudi S., Manfredini F., Mari R., Menegatti E.et al. (2018) Coagulation factor XII levels and intrinsic thrombin generation in multiple sclerosis. Front. Neurol. 9, 245 10.3389/fneur.2018.00245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Chan N.C. and Weitz J.I. (2019) Antithrombotic agents. Circ. Res. 124, 426–436 10.1161/CIRCRESAHA.118.313155 [DOI] [PubMed] [Google Scholar]
- 174.Bickmann J.K., Baglin T., Meijers J.C.M. and Renné T. (2017) Novel targets for anticoagulants lacking bleeding risk. Curr. Opin. Hematol. 24, 419–426 10.1097/MOH.0000000000000367 [DOI] [PubMed] [Google Scholar]
- 175.Franceschi C., Garagnani P., Parini P., Giuliani C. and Santoro A. (2018) Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14, 576–590 10.1038/s41574-018-0059-4 [DOI] [PubMed] [Google Scholar]
- 176.Price J., Lord J.M. and Harrison P. (2020) Inflammaging and platelet hyperreactivity: a new therapeutic target? J. Thromb. Haemost. 18, 3–5 10.1111/jth.14670 [DOI] [PubMed] [Google Scholar]
- 177.Culmer D.L., Diaz J.A., Hawley A.E., Jackson T.O., Shuster K.A., Sigler R.E.et al. (2013) Circulating and vein wall P-selectin promote venous thrombogenesis during aging in a rodent model. Thromb. Res. 131, 42–48 10.1016/j.thromres.2012.10.013 [DOI] [PubMed] [Google Scholar]
- 178.Dayal S., Wilson K.M., Motto D.G., Miller F.J., Chauhan A.K. and Lentz S.R. (2013) Hydrogen peroxide promotes aging-related platelet hyperactivation and thrombosis. Circulation 127, 1308–1316 10.1161/CIRCULATIONAHA.112.000966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Puchta A., Naidoo A., Verschoor C.P., Loukov D., Thevaranjan N., Mandur T.S.et al. (2016) TNF drives monocyte dysfunction with age and results in impaired anti-pneumococcal immunity. PLoS Pathog. 12, e1005368 10.1371/journal.ppat.1005368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Parameswaran N. and Patial S. (2010) Tumor necrosis factor-α signaling in macrophages. Crit. Rev. Eukaryot. Gene Expr. 20, 87–103 10.1615/CritRevEukarGeneExpr.v20.i2.10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Taylor D.D., Senhauser D.A. and Cavazos F. (1968) Thrombocytopathy associated with nonleukemic megakaryocytic myelosis. functional and fine structure observations of the abnormal platelets. Am. J. Clin. Pathol. 49, 662–670 10.1093/ajcp/49.5.662 [DOI] [PubMed] [Google Scholar]
- 182.Hattori A., Koike K., Ito S. and Matsuoka M. (1975) Static and functional morphology of the pathological platelets in primary myelofibrosis and myeloproliferative syndrome. Ser. Haematol. 8, 126–150 [PubMed] [Google Scholar]
- 183.Stephen J., Emerson B., Fox K.A. and Dransfield I. (2013) The uncoupling of monocyte-platelet interactions from the induction of proinflammatory signaling in monocytes. J. Immunol. 191, 5677–5683 10.4049/jimmunol.1301250 [DOI] [PubMed] [Google Scholar]
- 184.van der Zee P.M., Biró E., Ko Y., de Winter R.J., Hack C.E., Sturk A.et al. (2006) P-selectin- and CD63-exposing platelet microparticles reflect platelet activation in peripheral arterial disease and myocardial infarction. Clin. Chem. 52, 657–664 10.1373/clinchem.2005.057414 [DOI] [PubMed] [Google Scholar]
- 185.Iannucci J., Renehan W. and Grammas P. (2020) Thrombin, a mediator of coagulation, inflammation, and neurotoxicity at the neurovascular interface: implications for Alzheimer’s disease. Front. Neurosci. 14, 762 10.3389/fnins.2020.00762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Li D.Q., Zhou Y.P. and Yang H. (2012) Donepezil combined with natural hirudin improves the clinical symptoms of patients with mild-to-moderate Alzheimer’s disease: a 20-week open-label pilot study. Int. J. Med. Sci. 9, 248–255 10.7150/ijms.4363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Smyth L.C.D., Murray H.C., Hill M., van Leeuwen E., Highet B., Magon N.J.et al. (2022) Neutrophil-vascular interactions drive myeloperoxidase accumulation in the brain in Alzheimer’s disease. Acta. Neuropathol. Commun. 10, 38 10.1186/s40478-022-01347-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Yasuhara O., Walker D.G. and McGeer P.L. (1994) Hageman factor and its binding sites are present in senile plaques of Alzheimer’s disease. Brain Res. 654, 234–240 10.1016/0006-8993(94)90484-7 [DOI] [PubMed] [Google Scholar]
- 189.Ashby E.L., Love S. and Kehoe P.G. (2012) Assessment of activation of the plasma kallikrein-kinin system in frontal and temporal cortex in Alzheimer’s disease and vascular dementia. Neurobiol. Aging 33, 1345–1355 10.1016/j.neurobiolaging.2010.09.024 [DOI] [PubMed] [Google Scholar]
- 190.Bergamaschini L., Parnetti L., Pareyson D., Canziani S., Cugno M. and Agostoni A. (1998) Activation of the contact system in cerebrospinal fluid of patients with Alzheimer’s disease. Alzheimer Assoc. Disord. 12, 102–108 10.1097/00002093-199806000-00008 [DOI] [PubMed] [Google Scholar]
- 191.Shibayama Y., Joseph K., Nakazawa Y., Ghebreihiwet B., Peerschke E.I. and Kaplan A.P. (1999) Zinc-dependent activation of the plasma kinin-forming cascade by aggregated beta amyloid protein. Clin. Immunol. 90, 89–99 10.1006/clim.1998.4621 [DOI] [PubMed] [Google Scholar]
- 192.Maas C., Govers-Riemslag J.W., Bouma B., Schiks B., Hazenberg B.P., Lokhorst H.M.et al. (2008) Misfolded proteins activate factor XII in humans, leading to kallikrein formation without initiating coagulation. J. Clin. Invest. 118, 3208–3218 10.1172/JCI35424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Bergamaschini L., Donarini C., Foddi C., Gobbo G., Parnetti L. and Agostoni A. (2001) The region 1-11 of alzheimer amyloid-beta is critical for activation of contact-kinin system. Neurobiol. Aging 22, 63–69 10.1016/S0197-4580(00)00174-3 [DOI] [PubMed] [Google Scholar]
- 194.Zamolodchikov D., Chen Z.L., Conti B.A., Renné T. and Strickland S. (2015) Activation of the factor XII-driven contact system in Alzheimer’s disease patient and mouse model plasma. Proc. Natl. Acad. Sci. U.S.A. 112, 4068–4073 10.1073/pnas.1423764112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Chen Z.L., Revenko A.S., Singh P., MacLeod A.R., Norris E.H. and Strickland S. (2017) Depletion of coagulation factor XII ameliorates brain pathology and cognitive impairment in Alzheimer’s disease mice. Blood 129, 2547–2556 10.1182/blood-2016-11-753202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Bisht K., Sharma K. and Tremblay M.-È. (2018) Chronic stress as a risk factor for Alzheimer’s disease: roles of microglia-mediated synaptic remodeling, inflammation, and oxidative stress. Neurobiol. Stress 9, 9–21 10.1016/j.ynstr.2018.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Song H., Sieurin J., Wirdefeldt K., Pedersen N.L., Almqvist C., Larsson H.et al. (2020) Association of stress-related disorders with subsequent neurodegenerative diseases. JAMA Neurol. 77, 700–709 10.1001/jamaneurol.2020.0117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Chandola T., Brunner E. and Marmot M. (2006) Chronic stress at work and the metabolic syndrome: prospective study. BMJ 332, 521–525 10.1136/bmj.38693.435301.80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Dai S., Mo Y., Wang Y., Xiang B., Liao Q., Zhou M.et al. (2020) Chronic stress promotes cancer development. Front. Oncol. 10, 1492 10.3389/fonc.2020.01492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Satyjeet F., Naz S., Kumar V., Aung N.H., Bansari K., Irfan S.et al. (2022) Psychological stress as a risk factor for cardiovascular disease: a case-control study. Cureus 12, e10757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Dhabhar F.S. and Mcewen B.S. (1997) Acute stress enhances while chronic stress suppresses cell-mediated immunityin vivo: a potential role for leukocyte trafficking. Brain Behav. Immun. 11, 286–306 10.1006/brbi.1997.0508 [DOI] [PubMed] [Google Scholar]
- 202.Dhabhar F.S., Malarkey W.B., Neri E. and McEwen B.S. (2012) Stress-induced redistribution of immune cells—from barracks to boulevards to battlefields: a tale of three hormones - curt richter award winner. Psychoneuroendocrinology 37, 1345–1368 10.1016/j.psyneuen.2012.05.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.von Känel R., Merz F., Pfister H., Brückl T., Zimmermann P., Uhr M.et al. (2020) Acute stress-induced coagulation activation in patients with remitted major depression versus healthy controls and the role of stress-specific coping. Ann. Behav. Med. 54, 611–618 10.1093/abm/kaaa001 [DOI] [PubMed] [Google Scholar]
- 204.von Känel R., Bellingrath S. and Kudielka B.M. (2009) Association of vital exhaustion and depressive symptoms with changes in fibrin D-dimer to acute psychosocial stress. J. Psychosom. Res. 67, 93–101 10.1016/j.jpsychores.2008.12.009 [DOI] [PubMed] [Google Scholar]
- 205.Zgraggen L., Fischer J.E., Mischler K., Preckel D., Kudielka B.M. and von Känel R. (2005) Relationship between hemoconcentration and blood coagulation responses to acute mental stress. Thromb. Res. 115, 175–183 10.1016/j.thromres.2004.08.022 [DOI] [PubMed] [Google Scholar]
- 206.von Känel R., Mills P.J., Fainman C. and Dimsdale J.E. (2001) Effects of psychological stress and psychiatric disorders on blood coagulation and fibrinolysis: a biobehavioral pathway to coronary artery disease? Psychosom. Med. 63, 531–544 10.1097/00006842-200107000-00003 [DOI] [PubMed] [Google Scholar]
- 207.Eurofound (2022) Living and Working in Europe 2021. Publ. Off. Eur. Union Luxemb. [Google Scholar]
- 208.Brown D.L., Feskanich D., Sánchez B.N., Rexrode K.M., Schernhammer E.S. and Lisabeth L.D. (2009) Rotating night shift work and the risk of ischemic stroke. Am. J. Epidemiol. 169, 1370–1377 10.1093/aje/kwp056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Casetta I., Granieri E., Fallica E., la Cecilia O., Paolino E. and Manfredini R. (2002) Patient demographic and clinical features and circadian variation in onset of ischemic stroke. Arch. Neurol. 59, 48–53 10.1001/archneur.59.1.48 [DOI] [PubMed] [Google Scholar]
- 210.Early J.O. and Curtis A.M. (2016) Immunometabolism: is it under the eye of the clock? Semin. Immunol. 28, 478–490 10.1016/j.smim.2016.10.006 [DOI] [PubMed] [Google Scholar]
- 211.Kondratov R.V., Kondratova A.A., Gorbacheva V.Y., Vykhovanets O.V. and Antoch M.P. (2006) Early aging and age-related pathologies in mice deficient in BMAL1, the core componentof the circadian clock. Genes Dev. 20, 1868–1873 10.1101/gad.1432206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Wang J., Yin L. and Lazar M.A. (2006) The Orphan nuclear receptor rev-erbα regulates circadian expression of plasminogen activator inhibitor type 1*. J. Biol. Chem. 281, 33842–33848 10.1074/jbc.M607873200 [DOI] [PubMed] [Google Scholar]
- 213.Hemmeryckx B., Frederix L. and Lijnen H.R. (2019) Deficiency of Bmal1 disrupts the diurnal rhythm of haemostasis. Exp. Gerontol. 118, 1–8 10.1016/j.exger.2018.12.017 [DOI] [PubMed] [Google Scholar]
- 214.Chen L., Li S., Nie J., Zhao J., Yu S., Li Y.et al. (2020) Bmal1 regulates coagulation factor biosynthesis in mouse liver in Streptococcus oralis infection. Front. Cell. Infect. Microbiol. 10, 530190 10.3389/fcimb.2020.530190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Takeda N., Maemura K., Horie S., Oishi K., Imai Y., Harada T.et al. (2007) Thrombomodulin is a clock-controlled gene in vascular endothelial cells*. J. Biol. Chem. 282, 32561–32567 10.1074/jbc.M705692200 [DOI] [PubMed] [Google Scholar]
- 216.Somanath P.R., Podrez E.A., Chen J., Ma Y., Marchant K., Antoch M.et al. (2011) Deficiency in core circadian protein Bmal1 is associated with a prothrombotic and vascular phenotype. J. Cell. Physiol. 226, 132–140 10.1002/jcp.22314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Budkowska M., Lebiecka A., Marcinowska Z., Woźniak J., Jastrzębska M. and Dołęgowska B. (2019) The circadian rhythm of selected parameters of the hemostasis system in healthy people. Thromb. Res. 182, 79–88 10.1016/j.thromres.2019.08.015 [DOI] [PubMed] [Google Scholar]
- 218.Scheer F.A.J.L. and Shea S.A. (2014) Human Circadian system causes a morning peak in prothrombotic plasminogen activator inhibitor-1 (PAI-1) independent of the sleep/wake cycle. Blood 123, 590–593 10.1182/blood-2013-07-517060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Adrover J.M., Aroca-Crevillén A., Crainiciuc G., Ostos F., Rojas-Vega Y., Rubio-Ponce A.et al. (2020) Programmed ‘disarming’ of the neutrophil proteome reduces the magnitude of inflammation. Nat. Immunol. 21, 135–144 10.1038/s41590-019-0571-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Shahneh F., Christian Probst H., Wiesmann S.C., A-Gonzalez N., Ruf W., Steinbrink K.et al. (2022) Inflammatory monocyte counts determine venous blood clot formation and resolution. Arterioscler. Thromb. Vasc. Biol. 42, 145–155 10.1161/ATVBAHA.121.317176 [DOI] [PubMed] [Google Scholar]
- 221.Nguyen K.D., Fentress S.J., Qiu Y., Yun K., Cox J.S. and Chawla A. (2013) Circadian gene Bmal1 regulates diurnal oscillations of Ly6Chi inflammatory monocytes. Science 341, 1483–1488 10.1126/science.1240636 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Scheer F.A.J.L., Michelson A.D., Iii A.L.F., Evoniuk H., Kelly E.E., McCarthy M.et al. (2011) The human endogenous circadian system causes greatest platelet activation during the biological morning independent of behaviors. PLoS ONE 6, e24549 10.1371/journal.pone.0024549 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Fournier S., Guenat F., Fournier A., Alberio L., Bonny O., Bertaggia Calderara D.et al. (2018) Circadian variation of ticagrelor-induced platelet inhibition in healthy adulty. Eur. Heart J. - Cardiovasc. Pharmacother. 4, 166–171 10.1093/ehjcvp/pvy003 [DOI] [PubMed] [Google Scholar]
- 224.Shi J., Tong R., Zhou M., Gao Y., Zhao Y., Chen Y.et al. (2022) Circadian nuclear receptor rev-Erbα Is expressed by platelets and potentiates platelet activation and thrombus formation. Eur. Heart J. ehac109 10.1093/eurheartj/ehac109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Valnegri P., Khelfaoui M., Dorseuil O., Bassani S., Lagneaux C., Gianfelice A.et al. (2011) A circadian clock in hippocampus is regulated by interaction between oligophrenin-1 and Rev-Erbα. Nat. Neurosci. 14, 1293–1301 10.1038/nn.2911 [DOI] [PubMed] [Google Scholar]
- 226.Khelfaoui M., Pavlowsky A., Powell A.D., Valnegri P., Cheong K.W., Blandin Y.et al. (2009) Inhibition of RhoA pathway rescues the endocytosis defects in oligophrenin1 mouse model of mental retardation. Hum. Mol. Genet. 18, 2575–2583 10.1093/hmg/ddp189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Figueiro M.G., Goo Y., Hogan R., Plitnick B., Lee J., Jahangir K.et al. (2021) Light‐dark patterns mirroring shift work accelerate atherosclerosis and promote vulnerable lesion phenotypes. J. Am. Heart Assoc. 10, e018151 10.1161/JAHA.120.018151 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Wang Y., Kisler K., Nikolakopoulou A.M., Fernandez J.A., Griffin J.H. and Zlokovic B.V. (2022) 3K3A-activated protein C protects the blood-brain barrier and neurons from accelerated ischemic injury caused by pericyte deficiency in mice. Front. Neurosci. 16, 841916 10.3389/fnins.2022.841916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Yuda H., Adachi Y., Taguchi O., Gabazza E.C., Hataji O., Fujimoto H.et al. (2004) Activated protein C inhibits bronchial hyperresponsiveness and Th2 cytokine expression in mice. Blood 103, 2196–2204 10.1182/blood-2003-06-1980 [DOI] [PubMed] [Google Scholar]
- 230.Sinha R.K., Flynn R., Zaiken M., Paz K., Gavin A.L., Nemazee D.et al. (2019) Activated protein C ameliorates chronic graft-versus-host disease by PAR1-dependent biased cell signaling on T cells. Blood 134, 776–781 10.1182/blood.2019001259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Healy L.D., Puy C., Fernández J.A., Mitrugno A., Keshari R.S., Taku N.A.et al. (2017) Activated protein C inhibits neutrophil extracellular trap formation. J. Biol. Chem. 292, 8616–8629 10.1074/jbc.M116.768309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.de Boer J.D., Berger M., Majoor C.J., Kager L.M., Meijers J.C., Terpstra S.et al. (2015) Activated protein C inhibits neutrophil migration in allergic asthma: a randomised trial. Eur. Respir. J. 46, 1636–1644 10.1183/13993003.00459-2015 [DOI] [PubMed] [Google Scholar]
- 233.Ranjan S., Goihl A., Kohli S., Gadi I., Pierau M., Shahzad K.et al. (2017) Activated protein C protects from GvHD via PAR2/PAR3 signalling in regulatory T-cells. Nat. Commun. 8, 311 10.1038/s41467-017-00169-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Xue M., Dervish S., McKelvey K.J., March L., Wang F., Little C.B.et al. (2019) Activated protein C targets immune cells and rheumatoid synovial fibroblasts to prevent inflammatory arthritis in mice. Rheumatology 58, 1850–1860 10.1093/rheumatology/key429 [DOI] [PubMed] [Google Scholar]
- 235.Riewald M., Petrovan R.J., Donner A. and Ruf W. (2003) Activated protein C signals through the thrombin receptor PAR1 in endothelial cells. J. Endotoxin Res. 9, 317–321 10.1177/09680519030090050801 [DOI] [PubMed] [Google Scholar]
- 236.Riewald M., Petrovan R.J., Donner A., Mueller B.M. and Ruf W. (2002) Activation of endothelial cell protease activated receptor 1 by the protein C pathway. Science 296, 1880–1882 10.1126/science.1071699 [DOI] [PubMed] [Google Scholar]
- 237.Lyden P., Levy H., Weymer S., Pryor K., Kramer W., Griffin J.et al. (2014) Phase 1 safety, tolerability and pharmacokinetics of 3K3A-APC in healthy adult volunteers. Curr. Pharm. Des. 19, 7479–7485 10.2174/1381612819666131230131454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Yuda H., Adachi Y., Taguchi O., Gabazza E.C., Hataji O., Fujimoto H.et al. (2004) Activated protein C inhibits bronchial hyperresponsiveness and Th2 cytokine expression in mice. Blood 103, 2196–2204 10.1182/blood-2003-06-1980 [DOI] [PubMed] [Google Scholar]
- 239.Matsumoto T., Matsushima Y., Toda M., Roeen Z., D'Alessandro-Gabazza C.N., Hinneh J.A.et al. (2015) Activated protein C modulates the proinflammatory activity of dendritic cells. J. Asthma Allergy 8, 29–37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Mackman N. (2004) Role of tissue factor in hemostasis, thrombosis, and vascular development. Arterioscler. Thromb. Vasc. Biol. 24, 1015–1022 10.1161/01.ATV.0000130465.23430.74 [DOI] [PubMed] [Google Scholar]
- 241.Peyvandi F., Palla R., Menegatti M., Siboni S.M., Halimeh S., Faeser B.et al. (2012) Coagulation factor activity and clinical bleeding severity in rare bleeding disorders: results from the european network of rare bleeding disorders. J. Thromb. Haemost. 10, 615–621 10.1111/j.1538-7836.2012.04653.x [DOI] [PubMed] [Google Scholar]
- 242.Carmeliet P., Mackman N., Moons L., Luther T., Gressens P., Van Vlaenderen I.et al. (1996) Role of tissue factor in embryonic blood vessel development. Nature 383, 73–75 10.1038/383073a0 [DOI] [PubMed] [Google Scholar]
- 243.Kishi Y., Kondo T., Xiao S., Yosef N., Gaublomme J., Wu C.et al. (2016) Protein C receptor (PROCR) is a negative regulator of Th17 pathogenicity. J. Exp. Med. 213, 2489–2501 10.1084/jem.20151118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Lazic D., Sagare A.P., Nikolakopoulou A.M., Griffin J.H., Vassar R. and Zlokovic B.V. (2019) 3K3A-activated protein C blocks amyloidogenic BACE1 pathway and improves functional outcome in mice. J. Exp. Med. 216, 279–293 10.1084/jem.20181035 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
NA