Abstract
Composite injuries to the lower extremity from etiologies including trauma and infection present a complex dilemma for the reconstructive surgeon, and require multidisciplinary collaboration amongst plastic, vascular, and orthopaedic surgical specialties. Here we present our algorithm for lower-extremity reconstructive management, refined over the last decades to provide an optimized outcome for our patients. Reconstruction is predicated on the establishment of a clean and living wound, where quality of the wound-bed is prioritized over timing to soft-tissue coverage. Once established, soft-tissues and fractures are provisionally stabilized; our preference for definitive coverage is for microvascular free-tissue, due to the paucity of healthy soft-tissue available at the injury, and ability to avoid the zone of injury for microvascular anastomosis. Finally, definitive bony reconstruction is dictated by the length and location of long-bone defect, with a preference to utilize bone transport for defects longer than 5 cm.
Keywords: negative pressure wound therapy, fractures, lower extremity
Lower extremity trauma remains a substantial burden to the individual patient and the health system at large, accounting for more than 250,000 hospital admissions per year in the United States. 1 Associated pathology, including fracture nonunion, malunion, or osteomyelitis provides a similar paradigm of clinical challenges. Limb salvage following open fracture with soft-tissue loss remains a resource-intensive endeavor. 2 3 Indeed, data published by the landmark lower extremity assessment project group did not demonstrate a clear benefit to limb preservation when compared against amputation regarding return to work and quality of life. 4 Despite optimized treatment including provision of soft-tissue, adequate bony stabilization, and prompt wound care, rates of tibial nonunion can be as high as 50%, and delayed amputation as high as 12%. 5 6 7 The equivocal clinical results of limb salvage, while sobering, are belied and often negatively impacted by socio-economic factors. 8 9 However, continuing advances in microsurgical reconstruction and wound care, in concert with prompt osteosynthesis provides patients with an improved functional and aesthetic result following a devastating lower extremity injury. 3
Composite injuries of the lower extremity present unique dilemmas for the reconstructive surgeon. The requirements of stable bony fixation are compounded by the need for an adequate, supple, and clean soft-tissue envelope. Meeting these criteria, however, is hindered by the paucity of soft-tissue in the lower extremity, the consequence of dependent edema, and the extensive zone of injury surrounding the trauma. For this reason, the advent and routine adoption of free-tissue transfer, and the ability to augment healthy tissue, has expanded the number of potential candidates eligible for lower extremity salvage. 10 11 Factors previously thought to preclude salvage, including loss of plantar sensation, are no longer absolute contraindications. 12 Patient and injury-specific factors, including pre-existing co-morbidities can significantly impact the safety and feasibility of lower extremity reconstruction. As such, decision to proceed with reconstruction demands individualized consideration of the patient and the specific injury. 13
To be sure, successful limb salvage requires multidisciplinary collaboration, coordination, and communication. Orthopaedic, vascular, and plastic surgeons all play integral roles if reconstruction is to be successful. Here, we provide our guiding principles and practical framework for lower extremity reconstruction ( Fig. 1 .). All interventions are predicated on an adequately debrided wound-bed to create a “ Living Wound.” Temporization of the wound can be provided by negative pressure wound therapy (NPWT), but more often we utilize placement of a custom, either open or closed antibiotic bead pouch. This is especially true if there is any question of bacterial colonization. 14 15 16 At our institution, we advocate for provisional fixation that ensures maintenance of the patient's preinjury limb length. Once control of the wound and appropriate limb length are secured, soft-tissue provision and skeletal stabilization are often performed at a single stage. 17 18 19 Noted injuries, including tendon laceration or peripheral nerve repair are occasionally performed at this stage as well; if there is any suggestion of infection, reconstruction should be delayed until sterility is ensured. If a bone defect is present then one must address this in regard to its location in the bone, and, more importantly, the size of the defect. We shall present our algorithm for addressing defect size and what we have found to work most effectively.
Fig. 1.

The Buncke clinic, lower extremity reconstruction pathway.
Historical Context and Timing of Reconstruction
Multiple, validated scales have been used to classify the severity of mangling injuries to the lower-extremity. These classification systems, including the Mangled Extremity Severity Score, Limb Salvage Index, Predictive salvage index, and Nerve Injury, Ischemia, Soft Tissue Injury, Skeletal Injury, Shock, and Age of the Patient scores all provide useful schema to classify the severity of compound lower extremity injuries, and prognosticate the likelihood of lower extremity salvage. However, their clinical utility and accuracy in predicting successful salvage remain unproven. 20 Perhaps the most ubiquitous classification system used clinically is the Gustilo-Andersen grading scale of open fractures, divided into three categories with higher grades associated with increasing severity of associated soft-tissue and vascular inolvement. 21 22
Facilitated by microsurgical tissue transplantation, surgeons have long advocated for prompt soft-tissue coverage of acute open fractures. Guided by Godina's landmark paper in 1986, this window was further defined as 3 days post-injury. 23 A multitude of subsequent studies, and systematic reviews corroborated this benchmark, although, it bears noting, the majority of these studies analyze a cohort overwhelmingly dependent on Godina's original patient set. 24 The advances of improved dead-space management, including that of open antibiotic bead pouch, and the use of NPWT, have allowed further temporization of composite lower-extremity wounds. Accordingly, recent studies demonstrate a trend toward liberalization of the critical window of soft-tissue coverage without compromise of flap survival rates, or functional patient outcomes. 14 25 26 27 Indeed, over the decade of 2002 to 2011, the mean timing from injury to definitive soft-tissue coverage extended from 6 to 12.5 days. 28 The primacy of an adequately debrided wound bed prior to definitive reconstruction underlies this progression. In fact, we have learned that the most imperative factor in achieving a good result, which includes mitigation of late infection, provision of a healed and healthy soft tissue envelope, and healed bone reconstruction necessitate establishing, in the words of late George Cierny, a “ Living Wound.”
Recognition of reconstructive failure effectively re-starts the clock. Predictably, higher degrees of injury confer greater risk of reconstructive failure due to infection. 29 The etiology of wound breakdown must be determined; often underlying osteomyelitis or infected hardware will manifest as a small cutaneous wound. Refractory wound breakdown will persist, and reconstructive efforts will prove futile, unless source control is obtained, and all infected hardware, bone, and soft tissue are removed. Initial efforts should be directed toward controlling the site of non-union, or infection, and the surrounding soft-tissue and bone should be debrided as necessary. With creation of a new healthy wound bed, reconstruction can be temporized similarly to that of an acute injury. Due to the negative impact of contraction, fibrosis, and edema of the surrounding wound bed, provisional skeletal fixation and soft-tissue reconstruction should be performed in a timely fashion once a “Living Wound” is created.
Epidemiology, Etiology, and Initial Assessment
Open fractures are relatively rare, and comprise 2.6% of all long bone fractures; of these approximately 13% are of the lower leg (tibia and fibular diaphysis). 30 Despite the relative low frequency, this injury pattern is devastating and confers relatively longer hospital admissions and morbidity. 31 The most common etiology of lower extremity open fractures in the civilian population is motor vehicle accident, followed by falls from standing accounting for approximately 40 and 22% of these injuries, respectively. 31 The risk of subsequent infection and nonunion of initial reconstructive efforts remain close to 20%, and correlate with the severity of injury. 32
Lower extremity trauma with resulting open fracture occurs in a bimodal distribution, and amongst younger individuals is often the result of a high-energy injury. As such, all patients require a full Advanced Trauma Life Support primary survey to rule out potential life-threatening injuries. Efforts should be directed to mitigate hemorrhage from the injured extremity, which may require application of tourniquet, if necessary. Given the degree of tissue involvement, the presence of distracting injury, and multidisciplinary involvement, managing practitioners should maintain a high suspicion for compartment syndrome as this injury is often missed on initial presentation, and should be addressed emergently. 33 A thorough examination should take into account motor and sensory function of the limb and should grossly assess perfusion distal to the level of injury. As stated previously, damage to the tibial nerve is not, in of itself, an absolute contraindication to salvage. Antibiotic prophylaxis should be administered as soon as possible, ideally within the first hour of injury to mitigate subsequent infection risk. 34
Initial assessment should keep in mind the ultimate goals of reconstruction, specifically to ensure a controlled and clean wound, provide provisional skeletal stabilization, establish sufficient length of the injured extremity, to be followed by provision of a supple soft-tissue envelope, adequate bone stock, and definitive fixation. Once control of the wound is secured, vascular imaging of the injured extremity is performed to assess recipient vasculature for free tissue transfer. Both formal angiography and computed tomographic angiography are commonly employed for these purposes. 35 36 37 Despite the risks of complication with formal angiography, including the risk of iatrogenic injury cited in 1 to 5% of patients, our clinic prefers this modality due to the ability to assess flow directionality. 38 39 We find plain radiographs of fracture segments sufficient to perform provisional bony fixation, and routinely utilize intraoperative fluoroscopy to confirm adequate reduction. Necessary radiographs include full-length AP and lateral views of the tibia and fibula, as well as AP, lateral, and mortise views of the foot and ankle. 40 Radiographs of the contralateral extremity can also be helpful. For the majority of the high energy injuries with accompanying large open wounds and soft tissue defects, we have found that at least provisional external fixation remains the preferred choice of initial fixation, as no bridges are burned, and this can be modified as necessary.
Injury Management
Every traumatized lower extremity results in a unique injury pattern, however, a systematic approach to management keeps reconstructive goals in focus and organized. In general, our approach follows four steps: first debridement, then stabilization of soft tissue and skeleton, followed by soft tissue reconstruction, and ultimately reconstruction of the bone ( Fig. 1 ). We have found that this reconstruction ladder remains an integral part of successful limb salvage. It can be used as a framework/checklist in virtually all cases and provides a guide to follow, so that no steps are missed. It is imperative that this progression be followed in order, and bone reconstruction remains the last step in the process if one wishes to obtain eventual optimal results allowing the patient to return to more normal function and independent gait with a healed limb with proper alignment and symmetric leg length.
Debridement
Early and thorough debridement is critical to successful reconstruction. Nonviable tissue impedes physiologic healing and provides substrate for increased bacterial burden, which in turn may promote infection throughout reconstructive efforts. To definitively debride all nonviable tissue, it is often necessary to return to the operating room on multiple occasions over a series of days; provided adequate sterile conditions, this period can often last outside the 3 day period espoused in Godina's landmark study, and has been shown to mitigate subsequent risk of infection. 41 42 Through decades of experience, we have learned NOT to focus on the time frame from injury to definitive soft tissue closure/coverage, but rather to prioritize complete debridement of the injured limb with a resultant “Living Wound.” This concept is especially true in high energy traumas, wherein a large zone of injury may not be appreciated upon first look. In these scenarios, the zone of injury will declare itself over time, in turn making nonviable tissue evident. It is important to address all components of nonviable tissue including skin, subcutaneous tissue, muscle, bone, and even nerve. Staged once the wound is debrided to healthy well vascularized margins, the scale of the injury is appreciated, and the reconstructive plan can be established.
In the event of initial reconstructive failure, due to infectious, technical, or mechanical etiologies, care should be taken to debride unhealthy tissue without hesitation. All exposed hardware should be considered colonized, and duly removed. Placement of plates and screws for fixation may risk stripping of the periosteal blood supply rendering underlying bony fragments ischemic and nonviable. Ultimate determination of bone viability should be made clinically; healthy tissue should bleed when interrogated. In the setting of osteomyelitic bone, sequestrum may obstruct the medullary canal, which must be adequately obliterated to restore patency, ensure adequate endosteal circulation, and prevent persistence of biofilm. If infection is suspected, open biopsy at the time of debridement facilitates diagnosis of the pathologic organism, and can facilitate appropriate antibiotic management. 43 While IV antibiotics are routinely administered, we have found local tissue concentrations of antibiotics are sufficiently attained using the antibiotic bead pouch exclusively.
Over the years we have relied much less on the use of NPWT. This is especially true in more high energy wounds. We have found the application of continuous negative pressure at 125 mm Hg can be deleterious to cortical bone, tendon, and nerves, even in the presence of “white foam,” designed to mitigate underlying tissue desiccation. We have seen time and again cortical bone and tendon that had previously been viable, subsequently desiccated using NPWT regardless of the type of sponge material placed over it. Underlying structures can often appear white and bleached, and will exhibit no evidence of bleeding when interrogated. It is therefore our preference to use an open custom antibiotic bead pouch routinely, in such circumstances. It is far more physiologically conducive to the host tissue and far better at providing a healthy wound bed with marked reduction in bacterial colonization.
If osteomyelitis is present or suspected, then a host of other issues must be considered in the treatment of the limb and its eventual salvage. There are seven key points that must be reviewed by the Orthopaedic Surgeon and Plastic Surgeon in tandem to decide on the complete limb reconstruction plan. These steps are described and summarized in Table 1 .
Table 1. Considerations and issues to be addressed in the care of long bone osteomyelitis with accompanying impaired soft tissue injury.
| 1. | Localize the site and extent of the osteomyelitic lesion. |
| a. Is it isolated or multifocal. | |
| 2. | Define the Cierny-Mader stage (1 through 4). |
| a. If the lesion is stage 4, is a bony reconstruction possible following complete debridement? | |
| 3. | Define the Cierny-Mader type host being treated. |
| a. Type A, B local , B systemic , C | |
| b. If a B or C host define precisely the reason(s) and obstacles they may pose during the course of treatment. | |
| c. If patient is a B systemic or C host that is being considered for treatment rather than amputation, will a prolonged course of systemic antibiotics be necessary? | |
| d. If a B systemic host, can the patient undergo the type of soft tissue and/or bone reconstruction that might be necessary or should patient be reclassified as a C host? | |
| 4. | Define the condition of the soft tissue envelope. |
| a. Is the soft tissue envelope intact? | |
| b. Is there a sinus tract and does it require excision? | |
| c. If an incision is made can it then be closed? | |
| d. Will a local soft tissue rearrangement or transfer be necessary? | |
| e. Will a free tissue transfer be necessary to attain a healed soft tissue envelope? | |
| f. Would a free muscle transfer help with the local wound environment by providing improved local blood flow? | |
| 5. | Is the lesion confined enough such that it can be treated in a single surgical intervention or does treatment require a staged approach? |
| a. If treatment requires staged procedures will plastic surgical reconstruction be required? | |
| b. If plastic surgical assistance is deemed necessary then include this plan for treatment into the bone salvage and reconstruction formula for the limb. | |
| c. If staged, how will the bone and soft tissue defect be managed in the intervening periods? | |
| d. How will the dead space be managed? | |
| 6. | Will skeletal stabilization of the bone segment be required? |
| a. If so, will this best be managed by external or internal fixation? | |
| b. If external fixation is preferred, then will this in any way interfere with the long-term definitive treatment of the bone defect? | |
| c. Does the ensuing bone defect require reconstruction/grafting, and if so is it significant to the point that bone transport might be required? | |
| 7. | When and how will bone reconstruction be performed, and how will this be combined with any existing degree of bone defect? |
Source: Reproduced with permission of David W. Lowenberg, MD.
Early Skeletal Stabilization
The goal of early skeletal stabilization is to restore length and alignment of the bony skeleton. This stabilization is critical in defining apparent bone defects and dead space, and it further allows for stabilization of the soft tissue envelope about the zone of injury.
Any fixation technique that restores the length and alignment of injured bone will achieve the necessary goals of early skeletal stabilization, but external fixator placement is the technique best suited for early skeletal stabilization in devastating lower extremity injuries by spanning the zone of injury and avoiding prosthetic implant placement within a dirty or threatened wound. It also is the technique least damaging to the remaining endosteal and periosteal blood supply. The primary benefit of external fixation is that it does not utilize any permanent hardware, which is vitally important when the soft tissue coverage is lacking. External fixation also allows for further debridement and adjustment of boney reduction before definitive reconstruction is undertaken. In situations where bone defects are present, external fixation is well suited for maintaining reduction for injury stabilization. Finally, following initial placement, the external fixator can be modified to initiate and sustain bone transport via Ilizarov distraction osteogenesis if this is later determined to be necessary. At the time of frame application, care should be taken to avoid injury to soft-tissue structures, notably the common and superficial peroneal nerves, with lateral pin placement. 40
Early Soft Tissue Stabilization
After debridement of devitalized tissue and stabilization of the skeletal injuries, the soft tissue envelope must be stabilized to define the extent of defect and plan further reconstruction. The first assessment in the evaluation of soft tissue envelope is for dead space, which must be mitigated to achieve a harmonious reconstruction. Dead space can be managed through the promotion of granulation tissue or ablated during reconstruction.
Granulation tissue results from healing of well vascularized tissue and can be promoted through a variety of techniques. Ultimately, if the wound is cleanly debrided, any form of dressing will result in granulation tissue deposition. NPWT has emerged as the modality of choice at many institutions, as it robustly promotes granulation, and offers simple dressing changes. However, this may come at the cost of dessication of underlying tissues, and further obfuscation of native tissue planes. If free tissue transfer is chosen as the preferred method for definitive reconstruction then the promotion of granulation tissue is necessary. At the Buncke Clinic, where free-tissue transfer is more often than not the preferred modality of reconstruction, NPWT is avoided in favor of the custom open antibiotic bead pouch. 44
Soft Tissue Reconstruction
Definitive soft tissue reconstruction is undertaken only after orthopaedic and soft tissue injuries are adequately debrided and stable. Assessment of the soft tissue defect will account for coverage needs, dead space, and exposed or injured tendons and nerves that require repair. Despite the tenets of the “reconstructive ladder,” the unique challenges of lower extremity reconstruction often require prioritization of more complex operative solutions consistent with the framework of the “reconstructive elevator,” popularized by Gottlieb et al. 10 While workhorse local flaps exist in the lower extremity, their utility in reconstructing large defects with segmental bony defect is limited. Tenuous perfusion and proximity to the zone of injury resulting in substantial edema and fibrosis often limit the dependability of these flaps; their relatively small size and small arc of rotation similarly limits their practicality. Occasionally, smaller defects can be managed with reversed sural artery, soleus, or peroneus brevis flaps. 45 Given these limitations, free flap reconstruction is often the definitive reconstructive measure for traumatized lower limbs. Multiple permutations of free-tissue transfer including the use of chimeric, composite, and flow-through free-flaps provide elegant solutions to meet a wide spectrum of reconstructive challenges. 46
As mentioned briefly, the timing of definitive flap reconstruction has evolved over time, and now our general practice is to perform early, but not immediate, flap coverage. This approach allows for adequate debridement and control of the wound without exposing undue risk for infection. The combination of external fixation, antibiotic beads, and occasionally NPWT allows for safe and effective temporization before definitive reconstruction, generally within the first 5 to 10 days following trauma.
Flap selection should be guided by the unique clinical considerations presented by the respective defect. Size of the area to be covered, remaining perfusion to the distal foot, zone of injury including potentially traumatized recipient vessels, availability of autologous tissue for grafting (skin, vessel, nerve, etc.), and positioning restrictions should all factor into the preferred method of reconstruction and flap selection. Similarly, physician preference and comfort with certain flaps over others may reasonably inflect clinical decision making. The selection of recipient vessels should also be guided by injury and patient-specific considerations. Convention dictates vessels proximal to the zone of injury should be prioritized to restore flap inflow, however, the use of distal vasculature, and reliance on reverse arterial flow has been demonstrated to be a viable option when required.
In general, both muscle flaps and fasciocutaneous flaps demonstrate equal capacity to cover defects, resist infection, and mitigate wound breakdown. 47 Muscle flaps atrophy over time and remodel intimately to the wound, which is useful in ablating dead space. While effective in managing dead space, this intimate association between remodeled muscle flap and the underlying wound also make secondary reconstruction more difficult. In cases where flap elevation is needed for secondary reconstruction of either bone or tendon, a fasciocutaneous flap offers a tangible benefit of easier elevation and re-inset. 48 Conversely, fasciocutaneous flaps may result in relatively inferior contour and cosmetic outcome, especially in the setting of adipose hypertrophy with systemic weight gain. Regardless of the specific flap chosen to perform reconstruction, the operative surgeon should be aware of the increased rate of complications, specifically flap failure, with free-tissue transfer of the lower extremity. 49 Successful reconstruction requires technical excellence and anticipation of a difficult dissection.
At the Buncke Clinic, there is a general preference for the use of free muscle flaps in the coverage of post-traumatic wounds and infection. This is due to the increased inherent perfusion of muscle over that of subcutaneous tissue and also due the compelling basic science animal studies performed by the late Stephen Mathes on infection combatting benefits of muscle transfer versus other composites. 50 51 In the lower extremity, we have found muscle to definitely contour better over time and give a better long-term aesthetic result.
In the case of injured and exposed tendons, the scale of injury dictates the reconstructive plan. If primary tendon repair is possible, this is often performed in concert with soft tissue reconstruction. If secondary reconstruction is necessary, it is often delayed to a later point in the reconstruction. A similar concept is applied to nerve reconstruction, wherein primary repair is performed if possible. However, reconstruction with either autograft or allograft is best if performed during primary reconstruction rather than delayed to a secondary procedure. In the setting of gross infection, native tissue grafting is preferred. The use of external fixation spanning the ankle should also be leveraged to mitigate the risk of soft-tissue contracture, and can help to resist development of debilitating equinus deformity 52 ( Fig. 2 ). It is our experience that quite often if equinus is not addressed from the initiation of treatment, then down the line this becomes the predominate problem for the patient after the limb is healed. It has been found by the authors that attention to equinus at the onset of treatment gives a far superior result.
Fig. 2.

A 57-year-old male with osteomyelitis of trimalleolar right ankle fracture, requiring hardware removal, debridement of soft tissue, and fixation with external fixator spanning the ankle. Pt underwent subsequent free-tissue transfer with latissimus dorsi and split thickness skin grafting. (Panel ( A ) demonstrates initial fixator placement and debridement. Panel ( B ) demonstrates soft-tissue reconstruction). (Source: Reproduced with permission of David W. Lowenberg, MD).
Bone Reconstruction
As our treatment ladder algorithm dictates, only when dead space is obliterated, and the soft tissue envelope is adequate can bony reconstruction be performed. As discussed above, single-stage bony fixation at the time of flap coverage with a multiplanar circular fixator can expedite the patient's convalescence. 17 The ultimate technique for reconstruction is largely dictated by the size of the segmental defect, degree of comminution, and part of the bone in the lower extremity where the defect is located. One must also factor in whether the adjoining joint (knee or ankle) is preserved to allow adequate function.
For many years it has been our practice to use the segmental defect size to guide the selection of treatment method. In general, it is our preference for defects less than or equal to 2 cm to be treated with iliac crest bone grafting. We prefer the use of posterior iliac crest due to its greater volume of potential cancellous bone harvest as well as it generally being less painful to the patient. We use strictly the cancellous bone and marrow in such cases, as this incorporates must rapidly, unlike cortical bone. Our success rate with primary iliac crest bone grafting to these defects is over 90%. Actual bone grafting is usually delayed for at least 2 to 3 months following soft tissue coverage to allow for good healing of the soft tissue envelope.
Intermediate size defects, those measuring between 2 and 5 cm, represent a transition zone where the most appropriate treatment must be tailored to the individual patient. Therefore, the treatments for both small defects (<2 cm) and large defects (>5 cm) are considered. These include bone grafting, vascularized ipsilateral fibular transfer, free vascularized osseous transfer, bone transport, or the Masquelet technique. 53 54 In regards to actual cases, the vast majority of these size defects are treated by bone grafting or bone transport in our hands as these two techniques provide us with a 90% success rate for nearly all compliant patients.
The bone transport method of bone regeneration was developed by Gavril Ilizarov in Kurgan, Siberia in the 1950s and 1960s but did not make it to the Western World until the 1980s. 55 It involves creation of a low-energy, controlled fracture at a healthy portion of a long bone with preservation of the periosteal sleeve followed by a latency period then slow and controlled distraction such that the intercalary segment is moved into the bone defect with bone grown behind it at the controlled fracture site ( corticotomy ) to resultantly fill the defect ( Fig. 3 ). The length of time in a fixator for patients undergoing bone transport can be quite long and is dependent on the host conditions and length of bone defect. In some cases of large defect this can be 1 year and occasionally more.
Fig. 3.

A progression of bone transport demonstrating defect, initial corticotomy, process of distraction, and final consolidation ( A ). Example of Hoffman external fixation bone transport frame ( B ). (Source: Reproduced with permission of David W. Lowenberg, MD).
In the treatment of large defects, our pendulum for treatment swings predominately to bone transport whenever this is possible. Having actively done bone transports for nearly a third of a century, we have found that in experienced hands this has greater than a 95% success rate in properly selected patients with extremely reproducible results. This results in a “ non-implant dependent union ” in these patients with host native bone only being in the limb. This has led to long-term success in the many cases we have followed ( Figs. 4 5 6 7 ).
Fig. 4.

A 51-year-old male involved in an All-Terrain Vehicle rollover accident with a closed right subtrochanteric femur fracture and a Type 3A open right tibial pylon fracture with shaft extension. He was treated with immediate rodding of his femur fracture and debridement with spanning external fixation of his tibia fracture. Ten days later his distal tibial was treated at this outside facility with open reduction and internal fixation with seven plates and screws and closure of the wounds. Over the next 7 days there was progressive wound breakdown and dehiscence with eventual exposed hardware and bone. He was then transferred to us for a higher level of care. The patient was taken to the OR with multiple serial debridement and removal of all hardware except the anterior periarticular rim plate which was not infected or exposed. All necrotic bones were removed and the dead space was managed with custom antibiotic beads ( Fig. 2A, B demonstrating initial fixation. 2C, D wound breakdown with exposed hardware). (Source: Reproduced with permission of David W. Lowenberg, MD).
Fig. 5.
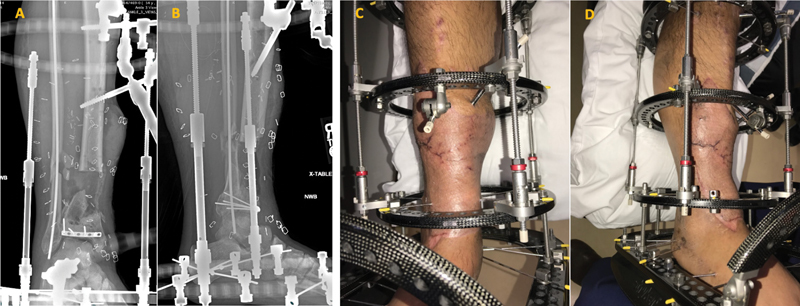
With a healthy and “Living Wound” now present, it was then covered with a free rectus abdominus flap, the pre-constructed circular fixator modified to a bone transport frame, and bone transport initiated to fill a 5.5-cm segmental bone defect ( Fig. 3A, B demonstrating circular fixator placement. 3C, D fixator in place, with healed soft-tissue envelope). (Source: Reproduced with permission of David W. Lowenberg, MD).
Fig. 6.

At the conclusion of bone transport the docking site distally is freshened up and augmented with a flap elevation and limited posterior iliac crest bone grafting to hasten bone healing. Regenerate bone maturation is allowed to continue with the patient continuing to ambulate weight bearing as tolerated during this entire time frame. At this point there is now adequate bone healing ( Fig. 4A, B Transport of tibial segment. 4C,D Distal segment docked, with supplemented iliac crest bone grafting.) (Source: Reproduced with permission of David W. Lowenberg, MD).
Fig. 7.

Radiographs 18 months, and 4 years following frame removal. The patient has no pain, is working full time as a ranch manager, and walks 3.5 miles on average per day. Patient is 56-years old and continues to work full time as a ranch manager with no limitations (Fig. 5A, B at 18 months after frame removal. 5C, D at 4 years). (Source: Reproduced with permission of David W. Lowenberg, MD ).
In a certain subset of patients, we have found continued value to the contralateral vascularized fibular transfer. These patients specifically are those that have at least 40% s of their host bone circumference remaining with a large defect. In these cases, the free fibula augments these large defects and with both host bone and the free fibula healing this leads to excellent long-term functional results. Often, fibula grafts can be transplanted concurrently with soft-tissue coverage when a “flow-through” flap design is employed. Isolated free fibula transfer in the lower extremity with a true complete segmental defect has not done as well in our hands. Fracture of the fibula transfer has been observed in many cases due to its inability to bear full forces of weightbearing and return to activities.
In essence, a spectrum exists as to the treatment methods employed for bone defects and is both dependent on the host and the size of the defect ( Fig. 8 ). This is part of the art of treating these difficult injuries in that one must be versed in numerous methods to treat the bony defect and address it individually in each patient.
Fig. 8.
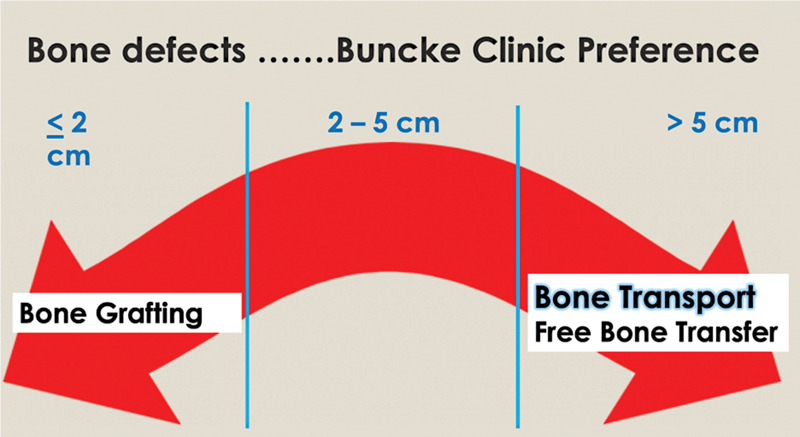
Spectrum of most common treatments we generally perform depending on size of defect. Other treatments as listed in the text can also be utilized. (Source: Reproduced with permission of David W. Lowenberg, MD).
Another concept worth mentioning is the idea of acute shortening of the limb. While this is technically feasible and can achieve bony union, it is unproven in achieving superior long-term outcomes, and negatively impacts the biomechanics of ambulation, ultimately the goal of lower extremity salvage. This is partially because of the fact that the lengthening occurs at a different site from the shortening, and this alters the blix curve of the muscle-tendon motor unit. Outside of the rare case where other reconstructive measures may be obviated, acute shortening offers little tangible benefit in our practice
Lower limb reconstruction following trauma and/or infection represents a particularly difficult form of reconstruction due to the demands of higher function and stress as well as proper mobilization in the lower extremities. Attention to detail and a fine understanding of the problems that can arise during reconstruction are necessary to achieve an optimum result. With decades of experience, we have come up with certain algorithms and principles which we have outlined, which help us achieve more acceptable outcomes.
Footnotes
Conflict of Interest None declared.
References
- 1.Nance M L. 2012 National Trauma Data Bank Annual Report. American College of Surgeons. 2012:146. [Google Scholar]
- 2.Georgiadis G M, Behrens F F, Joyce M J, Earle A S, Simmons A L. Open tibial fractures with severe soft-tissue loss. Limb salvage compared with below-the-knee amputation. J Bone Joint Surg Am. 1993;75(10):1431–1441. doi: 10.2106/00004623-199310000-00003. [DOI] [PubMed] [Google Scholar]
- 3.Lowenberg D W, Buntic R F, Buncke G M, Parrett B M. Long-term results and costs of muscle flap coverage with Ilizarov bone transport in lower limb salvage. J Orthop Trauma. 2013;27(10):576–581. doi: 10.1097/BOT.0b013e31828afde4. [DOI] [PubMed] [Google Scholar]
- 4.Bosse M J, MacKenzie E J, Kellam J F. An analysis of outcomes of reconstruction or amputation after leg-threatening injuries. N Engl J Med. 2002;347(24):1924–1931. doi: 10.1056/NEJMoa012604. [DOI] [PubMed] [Google Scholar]
- 5.Laughlin R T, Smith K L, Russell R C, Hayes J M. Late functional outcome in patients with tibia fractures covered with free muscle flaps. J Orthop Trauma. 1993;7(02):123–129. doi: 10.1097/00005131-199304000-00004. [DOI] [PubMed] [Google Scholar]
- 6.Choudry U, Moran S, Karacor Z. Soft-tissue coverage and outcome of gustilo grade IIIB midshaft tibia fractures: a 15-year experience. Plast Reconstr Surg. 2008;122(02):479–485. doi: 10.1097/PRS.0b013e31817d60e0. [DOI] [PubMed] [Google Scholar]
- 7.Lower Extremity Assessment Project (LEAP) Study Group . Harris A M, Althausen P L, Kellam J, Bosse M J, Castillo R. Complications following limb-threatening lower extremity trauma. J Orthop Trauma. 2009;23(01):1–6. doi: 10.1097/BOT.0b013e31818e43dd. [DOI] [PubMed] [Google Scholar]
- 8.MacKenzie E J, Bosse M J.Factors influencing outcome following limb-threatening lower limb trauma: lessons learned from the Lower Extremity Assessment Project (LEAP) J Am Acad Orthop Surg 200614(10 Spec No.):S205–S210. [DOI] [PubMed] [Google Scholar]
- 9.Higgins T F, Klatt J B, Beals T C. Lower Extremity Assessment Project (LEAP)—the best available evidence on limb-threatening lower extremity trauma. Orthop Clin North Am. 2010;41(02):233–239. doi: 10.1016/j.ocl.2009.12.006. [DOI] [PubMed] [Google Scholar]
- 10.Gottlieb L J, Krieger L M. From the reconstructive ladder to the reconstructive elevator. Plast Reconstr Surg. 1994;93(07):1503–1504. doi: 10.1097/00006534-199406000-00027. [DOI] [PubMed] [Google Scholar]
- 11.Xiong L, Gazyakan E, Kremer T. Free flaps for reconstruction of soft tissue defects in lower extremity: a meta-analysis on microsurgical outcome and safety. Microsurgery. 2016;36(06):511–524. doi: 10.1002/micr.30020. [DOI] [PubMed] [Google Scholar]
- 12.Momoh A O, Kumaran S, Lyons D. An argument for salvage in severe lower extremity trauma with posterior tibial nerve injury: the Ganga Hospital Experience. Plast Reconstr Surg. 2015;136(06):1337–1352. doi: 10.1097/PRS.0000000000001814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.LEAP Study Group . MacKenzie E J, Bosse M J, Kellam J F. Factors influencing the decision to amputate or reconstruct after high-energy lower extremity trauma. J Trauma. 2002;52(04):641–649. doi: 10.1097/00005373-200204000-00005. [DOI] [PubMed] [Google Scholar]
- 14.Liu D SH, Sofiadellis F, Ashton M, MacGill K, Webb A. Early soft tissue coverage and negative pressure wound therapy optimises patient outcomes in lower limb trauma. Injury. 2012;43(06):772–778. doi: 10.1016/j.injury.2011.09.003. [DOI] [PubMed] [Google Scholar]
- 15.Hill J B, Vogel J E, Sexton K W, Guillamondegui O D, Corral G AD, Shack R B. Re-evaluating the paradigm of early free flap coverage in lower extremity trauma. Microsurgery. 2013;33(01):9–13. doi: 10.1002/micr.21994. [DOI] [PubMed] [Google Scholar]
- 16.Lowenberg D W. American Academy of Orthopaedic Surgeons; 2014. Nonunions, malunions, and osteomyelitis; pp. 275–284. [Google Scholar]
- 17.Lowenberg D W, Feibel R J, Louie K W, Eshima I. Combined muscle flap and Ilizarov reconstruction for bone and soft tissue defects. Clin Orthop Relat Res. 1996;(332):37–51. doi: 10.1097/00003086-199611000-00007. [DOI] [PubMed] [Google Scholar]
- 18.Fiebel R J, Oliva A, Jackson R L, Louie K, Buncke H J. Simultaneous free-tissue transfer and Ilizarov distraction osteosynthesis in lower extremity salvage: case report and review of the literature. J Trauma. 1994;37(02):322–327. [PubMed] [Google Scholar]
- 19.McKee M D, Yoo D J, Zdero R. Combined single-stage osseous and soft tissue reconstruction of the tibia with the Ilizarov method and tissue transfer. J Orthop Trauma. 2008;22(03):183–189. doi: 10.1097/BOT.0b013e3181678a64. [DOI] [PubMed] [Google Scholar]
- 20.Bosse M J, MacKenzie E J, Kellam J F. A prospective evaluation of the clinical utility of the lower-extremity injury-severity scores. J Bone Joint Surg Am. 2001;83(01):3–14. doi: 10.2106/00004623-200101000-00002. [DOI] [PubMed] [Google Scholar]
- 21.Gustilo R B, Anderson J T. Prevention of infection in the treatment of one thousand and twenty-five open fractures of long bones: retrospective and prospective analyses. J Bone Joint Surg Am. 1976;58(04):453–458. [PubMed] [Google Scholar]
- 22.Gustilo R B, Mendoza R M, Williams D N. Problems in the management of type III (severe) open fractures: a new classification of type III open fractures. J Trauma. 1984;24(08):742–746. doi: 10.1097/00005373-198408000-00009. [DOI] [PubMed] [Google Scholar]
- 23.Godina M. Early microsurgical reconstruction of complex trauma of the extremities. Plast Reconstr Surg. 1986;78(03):285–292. doi: 10.1097/00006534-198609000-00001. [DOI] [PubMed] [Google Scholar]
- 24.Lee Z H, Stranix J T, Rifkin W J. Timing of microsurgical reconstruction in lower extremity trauma: an update of the Godina paradigm. Plast Reconstr Surg. 2019;144(03):759–767. doi: 10.1097/PRS.0000000000005955. [DOI] [PubMed] [Google Scholar]
- 25.Francel T J, Vander Kolk C A, Hoopes J E, Manson P N, Yaremchuk M J.Microvascular soft-tissue transplantation for reconstruction of acute open tibial fractures: timing of coverage and long-term functional results Plast Reconstr Surg 19928903478–487., discussion 488–489 [PubMed] [Google Scholar]
- 26.Karanas Y L, Nigriny J, Chang J. The timing of microsurgical reconstruction in lower extremity trauma. Microsurgery. 2008;28(08):632–634. doi: 10.1002/micr.20551. [DOI] [PubMed] [Google Scholar]
- 27.Steiert A E, Gohritz A, Schreiber T C, Krettek C, Vogt P M. Delayed flap coverage of open extremity fractures after previous vacuum-assisted closure (VAC) therapy - worse or worth? J Plast Reconstr Aesthet Surg. 2009;62(05):675–683. doi: 10.1016/j.bjps.2007.09.041. [DOI] [PubMed] [Google Scholar]
- 28.Sheckter C C, Pridgen B, Li A, Curtin C, Momeni A. Regional variation and trends in the timing of lower extremity reconstruction: a 10-year review of the nationwide inpatient sample. Plast Reconstr Surg. 2018;142(05):1337–1347. doi: 10.1097/PRS.0000000000004885. [DOI] [PubMed] [Google Scholar]
- 29.Patzakis M J, Wilkins J. Factors influencing infection rate in open fracture wounds. Clin Orthop Relat Res. 1989;(243):36–40. [PubMed] [Google Scholar]
- 30.Rockwood and Green's fractures in adults. - NLM Catalog - NCBIAccessed July 10, 2022, at:https://www-ncbi-nlm-nih-gov.laneproxy.stanford.edu/nlmcatalog/101258565
- 31.Court-Brown C M, Bugler K E, Clement N D, Duckworth A D, McQueen M M. The epidemiology of open fractures in adults. A 15-year review. Injury. 2012;43(06):891–897. doi: 10.1016/j.injury.2011.12.007. [DOI] [PubMed] [Google Scholar]
- 32.Hu Q, Zhao Y, Sun B, Qi W, Shi P. Surgical site infection following operative treatment of open fracture: incidence and prognostic risk factors. Int Wound J. 2020;17(03):708–715. doi: 10.1111/iwj.13330. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 33.Halawi M J, Morwood M P. Acute management of open fractures: an evidence-based review. Orthopedics. 2015;38(11):e1025–e1033. doi: 10.3928/01477447-20151020-12. [DOI] [PubMed] [Google Scholar]
- 34.Lack W D, Karunakar M A, Angerame M R. Type III open tibia fractures: immediate antibiotic prophylaxis minimizes infection. J Orthop Trauma. 2015;29(01):1–6. doi: 10.1097/BOT.0000000000000262. [DOI] [PubMed] [Google Scholar]
- 35.Johansen K, Lynch K, Paun M, Copass M.Non-invasive vascular tests reliably exclude occult arterial trauma in injured extremities J Trauma 19913104515–519., discussion 519–522 [PubMed] [Google Scholar]
- 36.Rubin G D, Schmidt A J, Logan L J, Sofilos M C. Multi-detector row CT angiography of lower extremity arterial inflow and runoff: initial experience. Radiology. 2001;221(01):146–158. doi: 10.1148/radiol.2211001325. [DOI] [PubMed] [Google Scholar]
- 37.Seamon M J, Smoger D, Torres D M.A prospective validation of a current practice: the detection of extremity vascular injury with CT angiography J Trauma 20096702238–243., discussion 243–244 [DOI] [PubMed] [Google Scholar]
- 38.Hessel S J, Adams D F, Abrams H L. Complications of angiography. Radiology. 1981;138(02):273–281. doi: 10.1148/radiology.138.2.7455105. [DOI] [PubMed] [Google Scholar]
- 39.Waugh J R, Sacharias N. Arteriographic complications in the DSA era. Radiology. 1992;182(01):243–246. doi: 10.1148/radiology.182.1.1727290. [DOI] [PubMed] [Google Scholar]
- 40.Shafiq B, Hacquebord J, Wright D J, Gupta R. Modern principles in the acute surgical management of open distal tibial fractures. J Am Acad Orthop Surg. 2021;29(11):e536–e547. doi: 10.5435/JAAOS-D-20-00502. [DOI] [PubMed] [Google Scholar]
- 41.Sirkin M, Sanders R, DiPasquale T, Herscovici D., Jr A staged protocol for soft tissue management in the treatment of complex pilon fractures. J Orthop Trauma. 1999;13(02):78–84. doi: 10.1097/00005131-199902000-00002. [DOI] [PubMed] [Google Scholar]
- 42.Patterson M J, Cole J D. Two-staged delayed open reduction and internal fixation of severe pilon fractures. J Orthop Trauma. 1999;13(02):85–91. doi: 10.1097/00005131-199902000-00003. [DOI] [PubMed] [Google Scholar]
- 43.Bishop J A, Palanca A A, Bellino M J, Lowenberg D W. Assessment of compromised fracture healing. J Am Acad Orthop Surg. 2012;20(05):273–282. doi: 10.5435/JAAOS-20-05-273. [DOI] [PubMed] [Google Scholar]
- 44.Henry S L, Ostermann P AW, Seligson D. The antibiotic bead pouch technique. The management of severe compound fractures. Clin Orthop Relat Res. 1993;(295):54–62. [PubMed] [Google Scholar]
- 45.Abd-Al-Moktader M A. Distally based peroneus brevis muscle flap for large leg, ankle, and foot defects: anatomical finding and clinical application. J Reconstr Microsurg. 2018;34(08):616–623. doi: 10.1055/s-0038-1661366. [DOI] [PubMed] [Google Scholar]
- 46.Miyamoto S, Kayano S, Umezawa H, Fujiki M, Sakuraba M. Flow-through fibula flap using soleus branch as distal runoff: a case report. Microsurgery. 2013;33(01):60–62. doi: 10.1002/micr.22043. [DOI] [PubMed] [Google Scholar]
- 47.Fischer J P, Wink J D, Nelson J A. A retrospective review of outcomes and flap selection in free tissue transfers for complex lower extremity reconstruction. J Reconstr Microsurg. 2013;29(06):407–416. doi: 10.1055/s-0033-1343952. [DOI] [PubMed] [Google Scholar]
- 48.Cho E H, Shammas R L, Carney M J. Muscle versus fasciocutaneous free flaps in lower extremity traumatic reconstruction: a multicenter outcomes analysis. Plast Reconstr Surg. 2018;141(01):191–199. doi: 10.1097/PRS.0000000000003927. [DOI] [PubMed] [Google Scholar]
- 49.Cho E H, Garcia R M, Pien I. Vascular considerations in foot and ankle free tissue transfer: analysis of 231 free flaps. Microsurgery. 2016;36(04):276–283. doi: 10.1002/micr.22406. [DOI] [PubMed] [Google Scholar]
- 50.Gosain A, Chang N, Mathes S, Hunt T K, Vasconez L.A study of the relationship between blood flow and bacterial inoculation in musculocutaneous and fasciocutaneous flaps Plast Reconstr Surg 199086061152–1162., discussion 1163 [PubMed] [Google Scholar]
- 51.Mathes S J, Alpert B S, Chang N. Use of the muscle flap in chronic osteomyelitis: experimental and clinical correlation. Plast Reconstr Surg. 1982;69(05):815–829. doi: 10.1097/00006534-198205000-00018. [DOI] [PubMed] [Google Scholar]
- 52.Lowenberg D W, Sadeghi C, Brooks D, Buncke G M, Buntic R F. Use of circular external fixation to maintain foot position during free tissue transfer to the foot and ankle. Microsurgery. 2008;28(08):623–627. doi: 10.1002/micr.20577. [DOI] [PubMed] [Google Scholar]
- 53.Masquelet A C, Fitoussi F, Begue T, Muller G P. [Reconstruction of the long bones by the induced membrane and spongy autograft] Ann Chir Plast Esthet. 2000;45(03):346–353. [PubMed] [Google Scholar]
- 54.French Society of Orthopaedic Surgery and Traumatology (SoFCOT) . Karger C, Kishi T, Schneider L, Fitoussi F, Masquelet A C. Treatment of posttraumatic bone defects by the induced membrane technique. Orthop Traumatol Surg Res. 2012;98(01):97–102. doi: 10.1016/j.otsr.2011.11.001. [DOI] [PubMed] [Google Scholar]
- 55.Lowenberg D W, Van Der Reis W. Acute shortening for tibia defects: when and where. Tech Orthop. 1996;11(02):210–215. [Google Scholar]


