Highlights
-
•
Sustainable emerging food processing technologies (sonication and DBD plasma) are used for wheat sprout juice processing.
-
•
Both techniques significantly increased the bioactive availability; while inhibiting enzymes and microorganisms.
-
•
Significantly reduced the particle size, apparent viscosity, consistency index, and increase the flow behavior.
-
•
SERS with silver-coated gold nano-substrates were used to analyze the impact of processing on the quality of juice.
Keywords: Wheat sprouts, Juice, Sonication, DBD-plasma, Flow behavior, Non-destructive measurement
Abstract
Sonication and dielectric barrier discharge (DBD) plasma are sustainable emerging food processing technologies. The study investigates the impact of sonication, DBD-plasma, and thermal treatment (TT) on wheat sprout juice. The obtained results indicated a significant (p < 0.05) increase in chlorophyll, total phenolics, flavonoids, DPPH assay, and ORAC assay after DBD-plasma (40 V) and sonication (30 mins) treatment as compared to TT and untreated samples. Both emerging technologies significantly (p < 0.05) reduce the polyphenol oxidase and peroxidase activities, but the TT sample had the highest reduction. Moreover, the synergistic application of both technologies significantly reduced the E. coli/Coliform, aerobics, yeast and mold up to the 2 log reduction, but the TT sample had a complete reduction. DBD-plasma and sonication processing significantly decreased (p < 0.05) the particle size, reducing apparent viscosity (η) and consistency index (K); while increasing the flow behavior (n), leading to higher stability of wheat sprout juice. To assess the impact of emerging techniques on nutrient concentration, we used surface-enhance Raman spectroscopy (SERS) as an emerging method. Silver-coated gold nano-substrates were used to compare the nutritional concentration of wheat sprout juice treated with sonication, DBD-plasma, and TT-treated samples. Results showed sharp peaks for samples treated with DBD-plasma followed by sonication, untreated, and TT. The obtained results, improved quality of wheat sprout juice, and lower microbial and enzymatic loads were confirmed, showing the suitability of these sustainable processing techniques for food processing and further research.
1. Introduction
The transformation of the food system proposes an emphatic possibility to collectively address sustainability, including social, economic, health, and environmental effects of food processing and consumption [1]. Processing is essential for food safety and security; currently, highly processed food is a growing issue for food safety with negative health impacts [2]. Considering this, the food processing industry is investing in research and development to support the concept of green techniques as a tool of preservation with non-significant nutrient loss. Non-thermal food processing has gained importance among the scientific community owing to its less destructive nature toward food. These non-thermal processing techniques include cold plasma (CP) and sonication as an alternative to thermal processing [3]. These green technologies effectively decrease processing time, reduce energy requirements, are eco-friendly, and improve food safety [4]. Sonication is an economical technique due to its reduced energy inputs and processing time [5]. Literature has shown the significant effect of sonication on the end product quality of various vegetable and fruit juices, such as spinach juice [6], currant juices [7], and strawberry juice [8]. Likewise, CP technology is a versatile, safe, environment-friendly, and effective technology employed for enzymatic inactivation, microbial decontamination, food packing modifications, removal of toxic substances, and elimination of contaminants from wastewater [9]. Innovative spectroscopic techniques are the most prominently used methods for various foods. Surface-enhanced Raman spectroscopy (SERS) is a rapid, novel, and non-destructive tool that has gained importance in the scientific community owing to its sensitivity toward varied bioactive constituents [10], [11].
Wheat sprouts are young shoots of wheat (Triticum aestivum L.) used as dietary supplements due to their high nutritional value [12]. Generally, wheat sprouts are consumed in powder, fresh juice, and/or tablet forms and possess abundant amounts of vitamins (A, B-complex, C, & E), minerals (P, Ca, Zn, Fe, K, & Mg), dietary fiber, essential amino acids, bioflavonoids, and coloring compounds (chlorophyll) [13]. Wheat sprouts possess various bioactivities such as antioxidant, antiaging, anti-inflammatory, anticancer, laxative, cough, constipation, rheumatic arthritis, and chronic skin-related diseases [14]. Wheat sprout juice is green low-acid juice with elevated chlorophyll, antioxidants, vitamins, natural enzymes, and other compounds [15]. Numerous actions have been made to prolong the shelf-life of juices by adding chemical preservatives, conventional pasteurization, and hurdle technologies [16], [17]. Nevertheless, a shift in consumer preferences toward fresh and minimally processed juices has recently increased; therefore, it is necessary to develop novel and innovative processing technologies having a less detrimental effect on nutrients [6], [18].
Despite all applied biotechnological methods in the agro-industry, relatively less scientific evidence is available to analyze the impact of DBD-plasma and sonication on the quality of wheat sprout juice. Therefore, the current study is designed to analyze both techniques' effects on the biologically active compounds, enzyme activities, microbial loads, particle size, and rheological properties of wheat sprout juice. Moreover, SERS was also used as an emerging non-invasive approach to check the effect of DBD-plasma, sonication, and thermal treatment on wheat sprout juice.
2. Materials and methods
2.1. Seeds selection and cultivation
Triticum Aestivum L. (wheat) var. Inqilab-91 was procured from CCRI, Pakistan. The wheat sprouts were grown in the South China University of Technology lab. Seeds were soaked in distilled water for 24 h. Then water was drained, and the seeds were covered with a muslin cloth and kept for 12 h for germination. Later, the seeds were spread in a soil tray, sprinkled with water daily for 14 days, and cut 2 cm above the soil (Fig. 1).
Fig. 1.
Seeds selection, seeds germination, seeds spreading in trays, grown-up sprouts, lush green sprouts, and extraction of wheat sprout juice. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
2.2. Juice extraction
The collected wheat sprouts were thoroughly washed to remove unwanted dust particles, followed by air drying. The collected sprouts were subjected to a specifically designed manual juice extractor for wheat sprout juice extraction by masticating.
2.3. Thermal treatment (TT)
For the TT, wheat sprout juice was subjected to a water bath with an adjustable temperature controller. The processing conditions in this experiment were 90 °C for 5 min to achieve thermal pasteurization. The processing conditions depend on achieving juice's microbial safety with minimal loss of nutrients [19].
2.4. Sonication treatment
Wheat sprout juice (100 mL) was poured into a glass bottle, and sonication treatment was applied following the procedures described by Zafra-Rojas et al. [20]. Purposely, a sonication apparatus (JP-100S, Skymen, China) operating at power 420 W and a frequency of 40 kHz was used in this experimentation to process wheat sprout juice at varied time intervals (10 to 40 min) (Fig. 2). A water circulation system was employed to maintain the temperature (30 ± 2 °C) throughout the processing. To avoid light interference, all the samples were treated in the dark.
Fig. 2.
(Left) DBD plasma, and (Right) Sonication bath cleaner apparatus used for the study.
2.5. Dielectric barrier discharge (DBD) plasma treatment
Wheat sprout juice samples were filled in a Petri plate (60 mm) and placed in a DBD-plasma reactor. This device comprises two electrodes (outer diameter: 50 mm) and plexiglass (thickness: 1.2 mm) used as a dielectric barrier (Fig. 2). For the application of voltage, a device setup transformer having an input power of 0–220 Volt and 10 kHz was used. Wheat sprout juice samples were treated at different electromotive forces (30–45 V) at a temperature (of 30 ± 2 °C) for 2 min. All the wheat sprout juice samples subjected to different treatments (TT, DBD-plasma, and sonication) were filtered via muslin cloth (4-layered) and later cooled using an ice-water bath before storage at 4 °C till further analysis.
2.6. Total flavonoid contents (TFC) and total phenolic contents (TPC)
Total flavonoids in wheat sprout juice were determined by following the protocols of Kim, Jeong and Lee [21]. Moreover, for estimating TPC in wheat sprout juice, Slinkard and Singleton's [22] documented procedure was adopted in this study. The sample absorbance was noted at 510 and 735 nm through a UV–visible spectrophotometer (UV–vis-1810) for TFC and TPC, respectively.
2.7. ORAC assay and DPPH-scavenging activity
The DPPH-scavenging activity and ORAC assay of wheat sprout juice were estimated by Manzoor et al. [23] and Prior et al. [24], respectively. The DPPH scavenging activity and ORAC assay were described as Trolox equivalent mmol L−1.
2.8. Chlorophyll contents
Chlorophyll contents in wheat sprout juice were measured by the protocols suggested by Zhao et al. [25]. A 3 mL 80 % v/v acetone was mixed with 3 mL juice samples. The obtained solution was filtered through a 125 mm Whatman filter paper. Later, the filtrate absorbance was noted at 663 and 645 nm. The total chlorophyll contents estimation was done by the sum of chlorophyll a and b, which were estimated by the following equations;
| (1) |
| (2) |
2.9. POD (Peroxidase) and PPO (polyphenol oxidase) activity
PPO and POD activity of wheat sprout juice were estimated by slight modifications in the proposed methods [13]. Each juice sample was centrifuged for 5 min at 5000 × g, and the resultant supernatants were diluted (100 times) through distilled water. Afterward, diluted supernatants (1 mL) were transferred to a tube containing 0.32 mL K3PO4 buffer (0.1 M with pH 6) and 0.16 mL C6H3(OH)3 (5 % w/v). Later, added 0.5 % w/v H2O2 was used to start the reaction, and absorbance was noticed at 485 nm. To determine the PPO activity, 0.05 M Na3PO4 buffer with pH 6.5 containing 1 mL C6H4(OH)2 (0.07 M) was mixed in diluted supernatants, then recorded the increased absorbance at 420 nm at 25 °C.
2.10. Microbiological analysis
In this experiment, the pour plate method was adopted for microbiological analysis [26]. Three different agar media were prepared; violet red bile, nutrient, and yeast & mold agar for coliform/E.coli, aerobics, and yeast & mold, respectively. For microbial growth, the incubation needs were 37 °C for 24–36 hr, 37 °C for 48 hr, and 25 °C for 3–5 days for aerobics, coliform/E.coli, and yeast & mold. All the findings were expressed in log CFU/mL.
2.11. Rheological analysis
A rotatory rheometer (DHR, American TA, USA) was used to determine the flow property of wheat sprout juice at 25 °C [27]. The data of shear stress (σ) versus shear rate (γ) were subjected to the Herschel-Bulkley model equation as follows:
| (3) |
Where σ0: yield stress (Pa), σ: shear stress (Pa), n: index of the flow behavior, γ: shear rate (s−1) and k: consistency index (Pa•sn).
2.12. Particle size distribution (PSD)
A distribution analyzer (LA960WET, HORIBA, UK) and a polydisperse analysis model were used to determine the particle size distribution of experimented juice samples. Software version 2.19 was used to analyze the results [28]. Particle size analysis was carried out by determining the D3,2, D v,0.5, and D4,3 [29]. The equations are given below:
| (4) |
| (5) |
For the determination of Span values following equation was used:
| (6) |
Dv,0.1: 10th percentile particle diameters, Dv,0.9: 90th percentile particle diameters and Dv,0.5: 50th percentile volume median diameter.
2.13. Juice quality analysis by SERS
2.13.1. Preparation of AuNPs
The AuNPs as the seed was prepared following the protocols of Hussain, Pu, Hu and Sun [30]. In detail, HAuCl4·4H2O (1 mL of 5 g/L) was mixed with ultrapure water (60 mL) with a digital shaker (MS 3, IKA Inc., Breisgau, Germany) operated at 900 rpm. Afterward, the prepared solution was heated for 1 min at 120 °C before adding trisodium citrate (700 μL, 1 %), followed by boiling again for 5 min. The color change confirmed the AuNPs formation from colorless to purplish red.
2.13.2. Preparation of Au@AgNPS
The seed-mediated growth technique prepared silver-coated gold nanomaterials [31]. Briefly, AuNPs colloid (3 mL) was poured into an Eppendorf tube (10 mL), followed by adding 270 μL (10 mM) ascorbic acid and mixed with a digital shaker for 2 min. Then AgNO3 solution (10 mM of 120 μL) was injected drop-wise @ of 1 drop/12 s and was shaken continuously with a digital shaker for 25 min at 950 rpm. Finally, the solution color was changed from purplish red to an orange-yellow appearance. All the treated (sonication, DBD-plasma, and TT) samples (200 μL) were added with 100 μL with Au@AgNPs for 2 min, and finally, SERS detection was performed.
2.13.3. Characterization of prepared nanomaterials
The morphology of synthesized substrates was examined through TEM (transition electron microscopy). An Au@AgNPs (3 mL) was centrifuged at 4500 rpm for 10 min, and the addition and removal of ultrapure water (3 mL) followed. After repeating the dilution 2 times, the resultant solid was diluted and sonicated for 20 min. Finally, the solution was poured on ultra-thin carbon film and dried at room temperature. A high magnification TEM (transmission electron microscopy) (JEOL JEM- 2100F, Japan) and 120 kV accelerating voltage were employed. Energy-dispersive X-ray spectroscopy (EDS) and high-angle annular dark field scanning transmission electron microscope (HAADF-STEM) were also used to get the superimposed images and elemental mapping of Au@AgNPs. Moreover, the hydrodynamic diameters and aggregation (if any) in the nano substrate were also tested with DLS using a Zetasizer system (Malvern Zetasizer Nano ZS, UK).
2.13.4. Acquisition of SERS spectra
To evaluate the impact of DBD-plasma, sonication, and thermal treatments on wheat sprout juice, a confocal Raman instrument fitted with a 633 nm laser having a laser power of 50 mW was utilized in this Raman experimentation. Raman system used in this experiment had high stable confocal microscope (BX41, Olympus Co., USA), 600 grooves mm per grating, a focal length achromatic flat field monochromator (800 mm), a CCD (cooled charged-coupled device), and a confocal coupling optics between the microscope. All the Raman spectra were obtained from 400 to 1700 cm−1 with a 10 X visible objective. In this experiment, the number of accumulation, exposure time, and laser power were set as 2, 10 s, and 100 %, respectively. The raw spectra was despiked, denoised and baseline was corrected as stated by [32].
2.14. Statistical analysis
All the analyses were performed in triplicates, and data were presented as mean ± SD. SPSS 24.0 (IBM, NY, USA) was used to analyze the data statistically. The significance level of obtained values was considered as P < 0.05.
3. Results and discussion
3.1. Impact on TPC and TFC
TPC and TFC have appeared as vital food components to human health by subduing the threat of different illnesses and significantly contributing to flavor and color development in food products [33]. Table 1 shows the TFC and TPC after sonication, DBD-plasma, and TT treatments. TFC and TPC were significantly (p < 0.05) increased in sonication and DBD-plasma treated samples when the treatment time and applied voltage increased, respectively. At the same time, TFC and TPC significantly reduced (p < 0.05) after thermal treatment. The maximum level of TFC and TPC was recorded in sonication treated sample at 30 min (192.56 ± 0.13 µg CE/g and 330.56 ± 0.18 µg GAE/g, respectively) and in the DBD-plasma-treated sample at 40 V (192.34 ± 0.12 µg CE/g, and 333.36 ± 0.18 µg GAE/g, respectively).
Table 1.
Impact of DBD plasma, sonication, and TT treatment on bioactive compounds and antioxidant activities of wheat sprout juice.
| Parameters | UT | CP-30 | CP-35 | CP-40 | CP-45 | S-10 | S-20 | S-30 | S-40 | TT |
|---|---|---|---|---|---|---|---|---|---|---|
| TPC (µg GAE/g) | 315.23 ± 0.09 g | 323.62 ± 0.04e | 324.54 ± 0.11e | 333.36 ± 0.18a | 328.45 ± 0.14c | 321.34 ± 0.12f | 323.72 ± 0.11e | 330.56 ± 0.18b | 326.22 ± 0.13d | 304.62 ± 0.10 h |
| TFC (µg CE/g) | 178.34 ± 0.12f | 184.34 ± 0.16 cd | 183.17 ± 0.13e | 192.34 ± 0.12a | 187.34 ± 0.08c | 185.12 ± 0.07 cd | 186.67 ± 0.17c | 192.56 ± 0.13a | 191.34 ± 0.12ab | 168.67 ± 0.09 g |
| DPPH (TE mmol L−1) | 1.63 ± 0.02f | 1.68 ± 0.04e | 1.70 ± 0.03d | 1.76 ± 0.02a | 1.72 ± 0.05c | 1.66 ± 0.06d | 1.69 ± 0.03 cd | 1.74 ± 0.07b | 1.71 ± 0.03 cd | 1.54 ± 0.04 g |
| ORAC (TE mmol L−1) | 5.12 ± 0.03e | 5.16 ± 0.04 cd | 5.19 ± 0.05b | 5.24 ± 0.02a | 5.22 ± 0.07b | 5.14 ± 0.03c | 5.18 ± 0.04c | 5.25 ± 0.02a | 5.21 ± 0.03b | 5.02 ± 0.05f |
| Chlorophyll (mg/100 mL−1) | 1.74 ± 0.03e | 1.77 ± 0.02d | 1.80 ± 0.05 cd | 1.88 ± 0.07a | 1.85 ± 0.04b | 1.76 ± 0.03d | 1.81 ± 0.02c | 1.87 ± 0.03a | 1.86 ± 0.05b | 1.68 ± 0.02f |
| Yeast & Mold (log CFU/mL) | 0.90 ± 0.04a | 0.75 ± 0.03b | 0.60 ± 0.02c | 0.48 ± 0.01d | 0.29 ± 0.04e | 0.78 ± 0.03b | 0.61 ± 0.02c | 0.44 ± 0.05de | 0.25 ± 0.03e | ND |
| Aerobics (log CFU/mL) | 5.33 ± 0.04a | 3.65 ± 0.02c | 2.96 ± 0.04d | 2.23 ± 0.03e | 1.83 ± 0.01 fg | 3.95 ± 0.02b | 3.02 ± 0.05e | 2.18 ± 0.02e | 1.68 ± 0.03f | ND |
| E. Coli/coliform (log CFU/mL) | 1.90 ± 0.02a | 1.63 ± 0.04b | 1.41 ± 0.03c | 1.07 ± 0.02e | 0.57 ± 0.01 fg | 1.60 ± 0.03b | 1.22 ± 0.02d | 0.71 ± 0.04f | ND | ND |
Different letters within the same row are significant (p < 0.05). All values are presented as mean ± SD. UT: untreated, TT: thermal-treated, S-10, S-20, S-30, and S-40: Sonication for 10, 20, 30, and 40 min, respectively. CP-30, CP-35, CP-40, and CP-45: Dielectric barrier discharge plasma treated at 30, 35, 40, and 45 kV.
The bioactive compounds in agricultural products are either soluble in vacuoles or tied to the cell wall [34]. During sonication treatment, cavitations resulting from quick changes in pressures of the liquid by shear forces induced an increase in TPC that might be associated with enhanced plant cell wall disruption, possibly promoting the availability of bound TPC in juice. Also, generating OH radicals through bubble implosion to an aromatic ring of phenolic compounds could cause an improvement in bioactive compounds [35]. Ashokkumar, Sunartio, Kentish, Mawson, Simons, Vilkhu and Versteeg [36] already stated that the increase in phenolic compounds could be associated with adding a second OH radicals in these compounds' meta-, para-, and ortho-positions. Another potential justification for the increase in TFC and TPC; is the propagation of mechanical waves through compression and rarefactions, causing surface peeling, particle breakage, and erosion, which might help release more bioactive compounds and enhance the TFC [37]. Similarly, Ordóñez-Santos, Martínez-Girón and Arias-Jaramillo [35] reported that increasing the sonication time increased the TPC and TFC in black carrot juice could be attributed to cavitation intensity.
DBD plasma comprises the interaction of reactive species with the substrate molecules. Thirumdas, Sarangapani and Annapure [38] reported that the reactive species during plasma processing stimulates the plant cell wall disruption and the release of the phenolics present in vacuoles or bound to lignin, cellulose, and pectin traces, ultimately increasing the TFC and TPC in juice. Also, Sarangapani, O'Toole, Cullen and Bourke [39] stated that an increase in TFC and TPC in foodstuffs after plasma treatment due to depolymerization and dissolution of cell wall polysaccharides, which stimulates the release of the conjugated phenolic compounds. Liu, Zhao, Zhang, Gao and Meng [40] explained that CP discharge could generate ozone, which effectively breaks the aromatic rings of polyphenols. The reduction in TPC and TFC at higher voltage (45 V) is possibly associated with ozone sensitivity and reactive oxygen species produced by DBD plasma. Manzoor et al. [26] and Rodríguez et al. [41] reported a notable increase in TPC and TFC after increasing the applied voltage of DBD-plasma processing in sugarcane and cashew apple juice, respectively. We also observed that the TPC and TFC obeyed an inverse relationship with enzyme (PPO) activity for sonication and DBD plasma treatment. The lower activity of PPO yielded higher TFC and TPC in wheat sprout juice. The TT in juices induced intricate physical and chemical reactions impacting the composition and release of TPC and TFC from bonded structures. It also provokes polyphenols' transformation, degradation, and breakdown [42].
3.2. Impact on antioxidant activity
DPPH activity and ORAC assays were supposed to be reliable approaches to measuring the antioxidant capacity of wheat sprout juice. The DPPH activity and ORAC assays displayed the high antioxidant capacity of wheat sprout juice (Table 1). It was noted that TT significantly (p < 0.05) reduced the antioxidant capacities (DPPH and ORAC). In contrast, during DBD-plasma and sonication treatments significantly increased (p < 0.05) than untreated samples. During DBD-plasma treatments, ORAC and DPPH values increased from 5.16 ± 0.04 to 5.24 ± 0.02 TE mmol L−1 and 1.68 ± 0.04 to 1.76 ± 0.02 TE mmol L−1, respectively, while during sonication ORAC and DPPH values increased from 5.14 ± 0.03 to 5.25 ± 0.02 TE mmol L−1 and 1.66 ± 0.06 to 1.74 ± 0.07 TE mmol L−1, respectively.
During sonication processing, ORAC and DPPH activities boosted up to 30 min treatment and declined afterward. The initial increment in ORAC and DPPH activities is associated with TPC and other bioactive compounds, which increase during the cavitation and drive the flavonols' hydroxylation, generating progressive antioxidant capacities [43]. Manzoor, Xu, Khan, Shukat, Ahmad, Imran, Rehman, Karrar, Aadil and Korma [44] noted that fruits and vegetables antioxidant activities depend on vitamins, flavonoids, and phenols. The reduction after 30 min treatment is perhaps linked to the cavitation bubbles that may grow too big to collapse or collapse weakly, which could reduce the cavitation impact. Moreover, different bubbles may delay the distribution of the sonication waves [45]. Wang, Wu, Chen, Yue, Liang and Wu [46] reported the association between cavitation bubbles and sonication processing time. The cavitation bubbles raised with time, reducing the antioxidant activities (ORAC and DPPH).
During DBD plasma processing, increased DPPH and ORAC activities were recorded by increasing the treatment voltage, which agrees with the increase in TPC. Fernandes, Santos and Rodrigues [47] reported an association between antioxidant activities and TPC in acerola juice treated with glow discharge plasma. The highest antioxidant values were recorded in wheat sprout samples treated at 40 V. Sarangapani et al. [39] stated a significant increment in the antioxidant capacities of blueberries juice, Rodríguez et al. [41] in cashew apple juice, and Manzoor et al. [26] in sugarcane juice after DBD plasma treatment. The impact of DBD plasma treatment on TPC and antioxidant activities is directly associated with the chemical reactions generated by the plasma species at different voltages [48]. The inactivation of PPO, associated with enzymatic browning, might improve the juice's antioxidant properties. The reduction after 45 V treatment in antioxidant capacities of wheat sprout juice could be attributed to the decrease in TPC, as described earlier. The current results align with Ali, Cheng and Sun [49], who described the reductions in TPC, TFC, and antioxidant activities during DBD plasma treatment of tomato juice. TT significantly reduced the antioxidant activities associated with the thermal degradation of polyphenols and flavonoids [50].
3.3. Impact on chlorophyll
Table 1 presents the chlorophyll concentration in wheat sprout juice after sonication, DBD plasma, and TT treatments. Results show a significant increase (p < 0.05) after DBD-plasma and sonication treatments while a significant (p < 0.05) reduction after TT treatment than the untreated sample. Like TPC and TFC, the highest concentration of chlorophyll contents was recorded in S-30 and CP-40 samples. The sonication-treated sample exhibited the retention of chlorophyll contents; similar results were reported by Ahmed et al. [13]. The sonication treatment possibly stimulates swelling and hydration, causing an enlargement in cell wall pores; it results in greater chlorophyll content extraction during treatment [51]. During DBD plasma treatment, it was observed that chlorophyll content firstly increased by increasing treatment voltage up to 40 V and then decreased. A potential justification for the increased chlorophyll content is the breakdown of the cell membrane and covalent bonds by charged particles and chemically reactive species, promoting chlorophyll release [40]. At higher voltage treatment, chlorophyll degradation is due to pigment oxidation by denaturation enzymes, and reactive plasma species are liable for chlorophyll catabolism during treatment [52]. Earlier, Ramazzina, Berardinelli, Rizzi, Tappi, Ragni, Sacchetti and Rocculi [52] reported a similar impact of cold plasma treatment on the chlorophyll content of kiwifruit. During TT, chlorophyll reduction is associated with thermal susceptibility and the formation of pheophytins by the displacement of the Mg2+ center [53].
3.4. Impact on POD and PPO activities
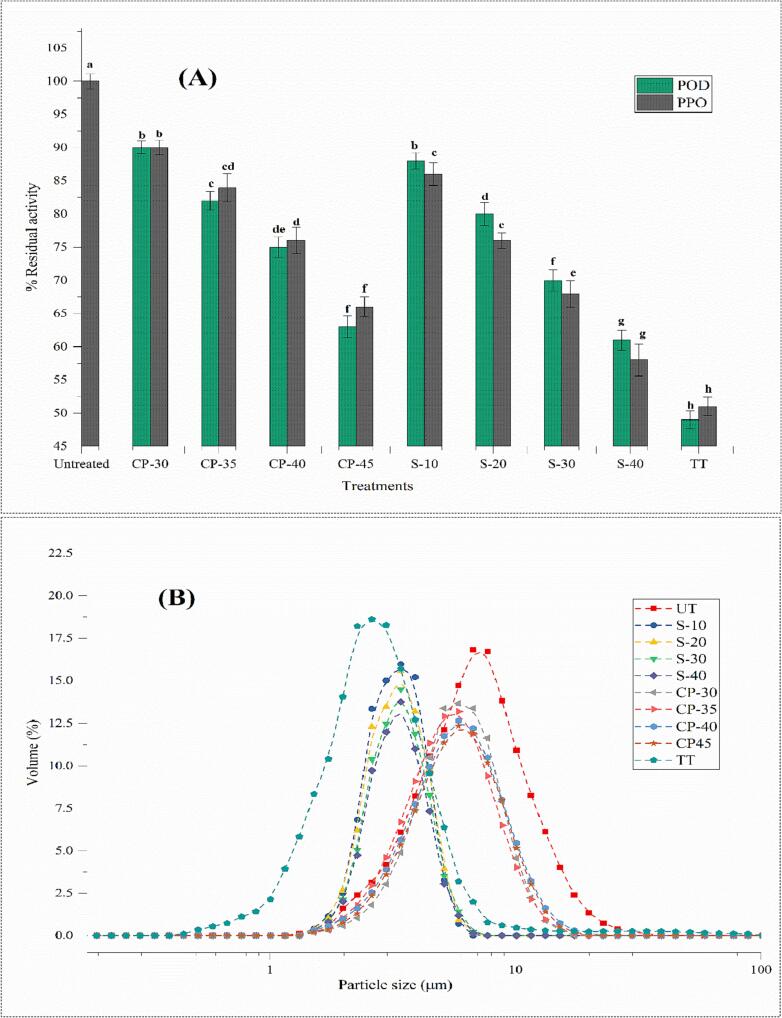
The food industry has explored endogenous enzyme inactivation techniques that can inactivate without modifying foods' nutritional and sensory properties [54]. POD and PPO generally reduce oxidative stress in plant tissue. These are spoilage-causing enzymes accountable for color changes such as browning. POD and PPO can vary in instability depending on the sources [55]. Obtained results stated that the PPO and POD enzymes activity was significantly (p < 0.05) inactivated by sonication, DBD-plasma, and TT treatments, as shown in Fig. 3A. The different treatments presented lower PPO and POD activity. The maximum reduction in PPO and POD activity was 51 %, and 49 % was observed in the TT sample, respectively, followed by sonication treatment at 40 min and DBD plasma at 45 V. TT inactivated the POD and PPO by aggregations at hydrophobic sites disclosed to denaturation. The influence of sonication treatment on enzyme activity is based on different aspects, such as food matrix, temperature, pH, processing time, and sonication intensity.
Fig. 3.
Impact of DBD plasma, sonication, and TT treatment on the (A) POD and PPO activities and (B) particle size of wheat sprouts juice. UT: untreated, TT: thermal-treated, S-10, S-20, S-30, and S-40: Sonication for 10, 20, 30, and 40 min, respectively. CP-30, CP-35, CP-40, and CP-45: Dielectric barrier discharge plasma treated at 30, 35, 40, and 45 kV.
During sonication, magnetostrictive effects are caused, inducing conformational and cavitation differences in enzymes [56]. Sonication processing can stimulate enzymes at low power and less time, whereas at a long time, higher power processing inactivates enzymes due to denaturation [56]. Sonication processing induced bubbles and cavitations that employed mechanical force, causing collapse and being responsible for enzyme inactivation. Kubo, Curet, Boillereaux and Augusto [57] reported a significant reduction in POD and PPO activities after sonication treatment than untreated juice samples. Costa, Fonteles, de Jesus, Almeida, de Miranda, Fernandes and Rodrigues [58] reported that prolonged exposure to sonication waves caused a significant reduction in PPO activity. Enzyme inactivation occurs when the rate of enzyme denaturation overcomes the rate of enzyme release. During DBD plasma treatment, free radicals are generated, mainly influencing the protein bonds N—H, C—N, and C—H [54]. Pankaj, Misra and Cullen [59] and Tinello and Lante [60] stated that the inactivation of enzymes during plasma processing due to modification in secondary structure and protein chains, induced by superoxide anion radicals, hydroxyl radicals, nitric oxide, and hydroperoxy radicals. In POD, secondary structure transformations reduce the distance between the heme group and tryptophan, increasing intramolecular energy transfer from the tryptophan heme. Therefore, tryptophan fluorescence tempering increased [61]. For PPO, structural changes cause a rise in the β-sheet region and a reduction in the α-helix content [60]. A significant reduction in PPO and POD residual activity was observed after cold plasma treatment at a different voltage in tomatoes [59].
3.5. Impact on microbial analysis
Table 1 presents a significant decrease (p < 0.05) in the microbial load after the sonication, DBD plasma, and TT treatments. The yeast & mold, aerobics, and E.coli/coliform count in freshly manufactured wheat sprout juice were 0.90, 5.33, and 1.90 logs CFU/mL, respectively. The thermal treatment completely inactivates the yeast & mold, aerobics, and E.coli/coliform. The TT generates cytolytic effects by disrupting or destroying the nucleic acids and integrity of the cellular membrane. The acquired consequences are aligned with thermally treated sugarcane juice, where a 100 % reduction in yeast & mold and aerobics mesophilic were reported by Manzoor et al. [6]. Other studies revealed that increasing the sonication time could disrupt the microbial cell wall [6]. Cell disruption during sonication is associated with different characteristics, including chemical and physical reactions that emerge during cavitation and form free radicals and hydrogen peroxide. The generation of OH− and H+ via sonolysis of water cause the inactivation of the microbial cells [44]. Earlier Ramírez et al. [62] reported that the sonication helps decrease blackberry microbial loads. In our study, at 10, 20, 30, and 40 min treatments of sonication, no complete inactivation of the microbial count was observed. Some studies reported that combining sonication and mild heat processing can enhance the rate of food sterilization. During DBD-plasma processing, a reduction in microbial loads could be associated with generating reactive nitrogen and oxygen species. A biocide influence of these reactive species possibly hinders microbial evolution and enhances the shelf life of treated products [63]. During DBD-plasma processing, it was noticed that the microbial growth was significantly reduced (p < 0.05) by raising the treatment voltage. Xu, Sanders, Tao, Garner and Keener [64] reported that high-voltage treatment generates a potent electric field that induces free electrons to interact with gas molecules to create a quasi-stable charged plasma or gas species. Consequentially reactive gas species, including NO2, NO, O, OH radical, and ozone (O3), have sporicidal, fungicidal, and bactericidal characteristics. Earlier, the higher inactivation of microbial growth was reported by Han, Patil, Boehm, Milosavljević, Cullen and Bourke [65]. Kim, Hong, Baik, Kwon, Choi, Cho, Uhm, Kim and Choi [66] noted that the most reactive short-lived species have the highest chance of contacting the surface during plasma processing. Furthermore, bombarding charged reactive gas species (RGS) in direct processing may stimulate RGS diffusion in liquid to induce microbiocidal compounds.
3.6. Impact on PSD
The impact of DBD-plasma, sonication, and TT treatments on PSD is shown in Table 2 and Fig. 3B. Increasing DBD-plasma treatment voltage and sonication treatment time reduce the particle size, leading to higher stability of wheat sprout juice. The TT-treated juice sample had significantly (p < 0.05) larger D(4,3), D(2,3), D(v,0.90), D(v,0.50), and D(v,0.10) values, while DBD-plasma and sonication treated samples had significantly lower (p < 0.05) PSD values than the untreated sample. TT increases the PSD due to more elevated temperature yielding aggregate from small to larger particles. During sonication and DBD-plasma processing, the PSD was significantly decreased by elevating the sonication time and treatment voltage, respectively. Sonication processing reduces the particle size due to shear force induced by cavitation, and sonication processing has been employed to produce emulsions [67]. Our results indicate that sonication dramatically decreases suspended particle size due to high shear; cavitation restricts the polysaccharide's aggregation and shatters them into smaller sizes [68]. Manzoor, Xu, Khan, Shukat, Ahmad, Imran, Rehman, Karrar, Aadil and Korma [44] reported a significant reduction in the PSD of spinach juice after the ultrasound processing for 30 min at 30 kHz. DBD-plasma processing significantly reduced the size of the suspended particles at higher treatment voltage, while lower treatment voltage induced an increment in the volume of PSD. Wang, Han, Niu, Zhang, Xiang and Zong [69] reported that the particle size of jujube juice was significantly reduced by increasing the treatment voltages. Illera, Chaple, Sanz, Ng, Lu, Jones, Carey and Bourke [70] reported that homogenization would occur when the particle size decreases, increasing fruit juice's stability. The sonication, DBD-plasma, and TT-treated wheat grass juice showed a monomodal distribution (Fig. 3B). The DBD-plasma and sonication treatments significantly reduced the PSD span value, while TT increased it. Fig. 3B revealed that sonication and plasma treatments reduced the % volume in a particle size ranging from 1 and 20 μm. In contrast, the % volume of TT-treated samples rose, ranging from 1 to 10 μm. The span value of wheat sprout juice treated with different techniques defines the particle distribution width, so the decline in span value indicates a juice homogenization.
Table 2.
Impact of DBD plasma, sonication, and TT treatment on the particle size of wheat sprouts juice.
| Parameters | UT | CP-30 | CP-35 | CP-40 | CP-45 | S-10 | S-20 | S-30 | S-40 | TT |
|---|---|---|---|---|---|---|---|---|---|---|
| D (v,0.10) µm | 2.23 ± 0.05a | 2.30 ± 0.03a | 1.10 ± 0.04d | 0.86 ± 0.03e | 1.08 ± 0.02d | 1.68 ± 0.02b | 1.86 ± 0.03b | 1.41 ± 0.01c | 0.95 ± 0.01e | 1.50 ± 0.02c |
| D (v,0.50) µm | 2.49 ± 0.06c | 2.61 ± 0.03c | 2.16 ± 0.04de | 2.14 ± 0.05de | 1.78 ± 0.02e | 2.39 ± 0.03d | 2.13 ± 0.01de | 2.90 ± 0.04b | 1.81 ± 0.02e | 3.92 ± 0.04a |
| D (v,0.90) µm | 65.04 ± 3b | 66.28 ± 5b | 64.34 ± 6b | 60.47 ± 3c | 52.32 ± 2e | 64.92 ± 4b | 62.95 ± 3bc | 59.09 ± 2d | 54.33 ± 4e | 87.94 ± 5a |
| D (3,2) µm | 1.73 ± 0.02b | 1.80 ± 0.05b | 1.44 ± 0.01d | 1.09 ± 0.04ef | 0.72 ± 0.03 g | 1.67 ± 0.02c | 1.54 ± 0.01c | 1.27 ± 0.06e | 0.80 ± 0.04 g | 1.98 ± 0.03a |
| D (4,3) µm | 12.96 ± 0.23c | 15.28 ± 0.14a | 11.63 ± 0.32d | 12.90 ± 0.22c | 11.34 ± 0.14d | 13.57 ± 0.36b | 11.67 ± 0.25d | 10.98 ± 0.15e | 11.28 ± 0.27d | 15.92 ± 0.19a |
| Span | 13.45 ± 0.18 cd | 20.25 ± 0.08b | 9.30 ± 0.12d | 6.34 ± 0.11f | 3.02 ± 0.06 g | 14.24 ± 0.19c | 7.03 ± 0.07e | 5.63 ± 0.11f | 5.54 ± 0.18f | 21.32 ± 0.24a |
Different letters within the same row are significant (p < 0.05). All values are presented as mean ± SD. UT: untreated, TT: thermal-treated, S-10, S-20, S-30, and S-40: Sonication for 10, 20, 30, and 40 min, respectively. CP-30, CP-35, CP-40, and CP-45: Dielectric barrier discharge plasma treated at 30, 35, 40, and 45 kV.
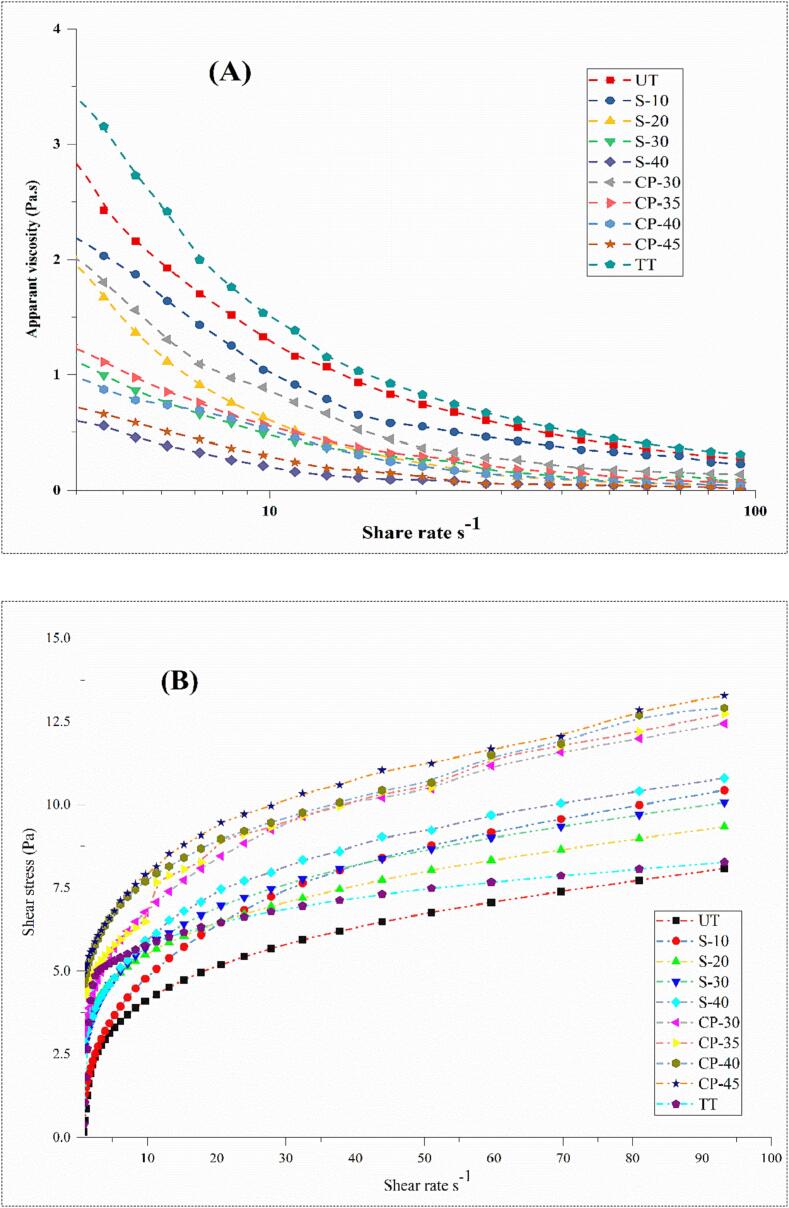
3.7. Impact of rheological behavior
Different factors influence the choice of the rheological model, which describes specific fluid behavior. Various models have been used to describe the fluid's flow behavior, whether Newtonian (n∼1) or non-Newtonian (n < 1). Different rheological parameters, including K (consistency index), R2 (nonlinear correlation coefficient), n (flow behavior), σy (yield stress), and η (apparent viscosity) at the shear rate 120 s−1 of the Herschel-Bulkley model, are shown in Table 3. The sonication (S-10), DBD-plasma (CP-30 and CP-35), TT-treated, and untreated samples displayed non-Newtonian pseudoplastic behavior, while sonication (S-20, S-30 and S-40) and DBD-plasma (CP-40 and CP-45) exhibited Newtonian behavior. Obtained results also indicate that DBD-plasma and sonication processing significantly (p < 0.05) reduce the consistency index (Table 3) and apparent viscosity (Fig. 4A) than TT-treated and untreated samples. Moreover, it also changed the macromolecules' configuration accountable for the samples' flow behavior. The decline in apparent viscosity with the rise in the shear rate indicates a quick breakdown of initial shearing. In contrast, at a high shear rate, deliberate alteration arises in the structure. These outcomes are supported by the PSD (Table 2 and Fig. 3B) because sonication and DBD-plasma ruptured the aggregates and formed many small structures, which developed liquids with lower k, and Newtonian fluid behavior. The smaller particle size increases dispersibility and occupies the spaces between the larger particles, which produces the lubricating impact with a resulting decrease in viscosity. Earlier, a similar impact of sonication and plasma treatments on the rheological properties of peach juice and guava-flavored whey-beverage was reported by Rojas et al. [71] and Silveira et al. [72]. The shear stress of all samples was plotted against the shear rate to describe wheat sprout juice's flow behavior (Fig. 4B). A notable increase in shear stress was recorded by elevating DBD-plasma voltage and sonication time than TT-treated and untreated samples. Furthermore, slower alteration occurs in the structure at high shear rates.
Table 3.
Impact of DBD plasma, sonication, and TT treatment on the rheological properties of wheat sprouts juice.
| Parameters | UT | CP-30 | CP-35 | CP-40 | CP-45 | S-10 | S-20 | S-30 | S-40 | TT |
|---|---|---|---|---|---|---|---|---|---|---|
| K Pa.sn | 5.067 ± 0.003 g | 4.165 ± 0.08a | 2.175 ± 0.06c | 3.239 ± 0.04b | 0.969 ± 0.006e | 3.156 ± 0.05b | 2.123 ± 0.03c | 1.835 ± 0.02d | 0.896 ± 0.007e | 6.235 ± 0.004f |
| σy × 10−3 (Pa) | 1.01 ± 0.03f | 3.22 ± 0.04c | 3.06 ± 0.06c | 2.02 ± 0.06de | 2.63 ± 0.05d | 4.01 ± 0.04b | 3.13 ± 0.05c | 2.85 ± 0.04d | 1.99 ± 0.03e | 5.13 ± 0.09a |
| n | 0.7212 ± 0.04c | 0.8237 ± 0.007 h | 0.8763 ± 0.005 g | 0.9056 ± 0.002e | 0.9322 ± 0.03d | 0.8511 ± 0.01b | 0.9031 ± 0.05b | 0.9109 ± 0.04a | 0.9565 ± 0.03a | 0.6134 ± 0.009ef |
| η 120 s−1 (cP) | 6.12 ± 0.05b | 4.32 ± 0.02 cd | 3.97 ± 0.03e | 2.91 ± 0.05f | 1.98 ± 0.04gh | 4.93 ± 0.01c | 4.01 ± 0.03d | 3.32 ± 0.02ef | 2.03 ± 0.04 g | 6.97 ± 0.04a |
| R2 | 0.9836 | 0.9931 | 0.9902 | 0.9841 | 0.9617 | 0.9991 | 0.9976 | 0.9516 | 0.9817 | 0.9016 |
Different letters within the same row are significant (p < 0.05). All values are presented as mean ± SD. UT: untreated, TT: thermal-treated, S-10, S-20, S-30, and S-40: Sonication for 10, 20, 30, and 40 min, respectively. CP-30, CP-35, CP-40, and CP-45: Dielectric barrier discharge plasma treated at 30, 35, 40, and 45 kV.
Abbreviations: K, consistency index; (σy), yield stress; n, flow behavior.
Fig. 4.
Impact of sonication, DBD-plasma, and TT treatments on the (A) apparent viscosity and (B) flow behavior of wheat sprout juice. UT: untreated, TT: thermal-treated, S-10, S-20, S-30, and S-40: Sonication for 10, 20, 30, and 40 min, respectively. CP-30, CP-35, CP-40, and CP-45: Dielectric barrier discharge plasma treated at 30, 35, 40, and 45 kV.
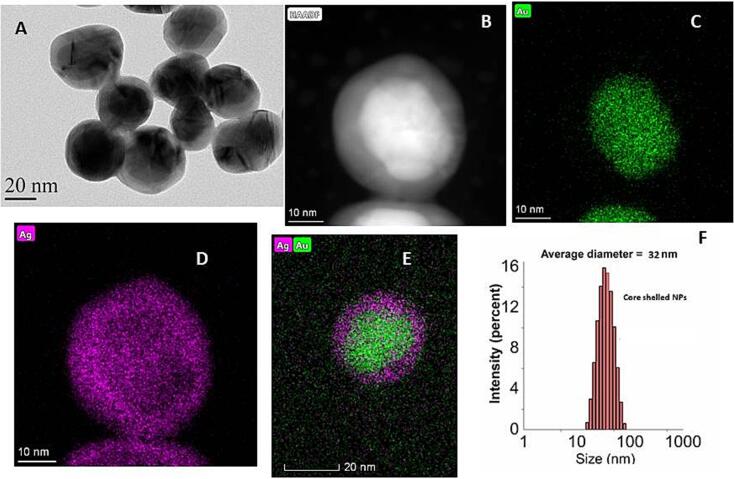
3.8. Morphology and size of prepared nanoparticles
The successful synthesis of nanoparticles was confirmed with high-resolution TEM, HAADF STEM, and EDS element mapping methods. The dark region in the center shows Au, whereas the bright region as a layer shows silver nanoparticles which can be observed with TEM (Fig. 5A) and HAADF STEM analysis (Fig. 5B). Two different layers in the figures are due to the difference in atomic numbers of both elements. Moreover, the green color shows gold, which corresponds with Ag nanoparticles, while the gold atoms coated with silver can be observed with the EDS element mapping method, as shown in Fig. 5C&D. In addition, DLS methods exhibited no aggregation in the prepared materials with an average size of 32 nm as shown in Fig. 5E&F.
Fig. 5.
High-resolution TEM (A), HDDF STEM (B), EDS elemental mapping of Au (C), Ag (D) and Ag@AuNPs (E) and DLS (F) of prepared nanomaterials.
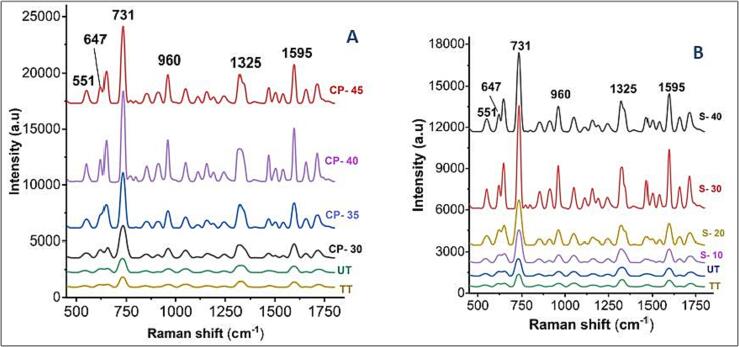
3.9. Impact of DBD plasma and sonication detection through SERS
Among the emerging spectroscopic methods, SERS proved promising in food and non-food applications and enhanced weak Raman scattering using different nanomaterials [73]. In detail, long-range electromagnetic and short-range chemical effects make SERS an ultra-sensitive tool. The electromagnetic effect enhances the Raman peaks significantly up to1010–12 folds due to the optical properties of nanoparticles. At the same time, the chemical effect enhances Raman intensity to 102 from charge transfer between the nano substrate and the sample under study [30], [31]. Gold, silver, and copper nanoparticles have been used as single substrates or modified to increase sample adsorption. Gold nanoparticles efficiently synthesize with weak sensitivity, while silver nanoparticles contribute better sensitivity; thus, combining these nanomaterials can easily achieve the targeted results. In the current study, we used silver-coated gold nano-substrates to compare the nutritional concentration of wheat sprout juice treated with sonication, DBD-plasma, and TT-treated samples. As SERS is a highly sensitive method, it can verify the presence of constituents in a sample. Our experiments observed higher intensity of Raman peaks for DBD-plasma-treated samples than sonication and TT samples. For example, characteristic peak at 731 cm−1 had higher intensity (8031 a.u) with the former compared to the latter techniques (7052 a.u and 898 a.u). The sharper peaks depict the more nutritional concentration of ingredients in the juice.
Moreover, the juices mainly comprise sugar, and the peaks correspond with carbohydrates (due to C—C and C—O stretching vibrations [13], [74]. The peak intensity was increased with the increasing DBD-plasma treatment voltage, and the highest was observed at 40 V. After this, the Raman peak intensity decreased (45 V), reducing nutritional concentration. Likewise, Raman intensity was observed with increasing sonication time up to 30 min, whereas the peak intensity declines after 30 min, as shown in Fig. 6A&B. The peak assignments for the samples are presented in Table 4. Moreover, untreated juice had a higher nutritional concentration than the TT sample due to the destructive nature of thermal methods.
Fig. 6.
(A) Raman spectra of wheat spout juice treated with DBD-plasma at different voltages (A), and sonication at different treatment times UT: untreated, TT: thermal-treated, S-10, S-20, S-30, and S-40: Sonication for 10, 20, 30, and 40 min, respectively. CP-30, CP-35, CP-40, and CP-45: Dielectric barrier discharge plasma treated at 30, 35, 40, and 45 kV.
Table 4.
Raman peaks and their assignments.
| Peak (cm−1) | Assignment | References |
|---|---|---|
| 552 | Results in aromatic C—H out-of-plane deformation and C—H deformation in phenyl rings of flavonoids | [75] |
| 647 | aromatic C—H out-of-plane deformation and C—H deformation in phenyl rings of flavonoids | [13], [76] |
| 731 | It appears due to C—C and C—O stretching vibrations of carbohydrates and bending of CH2 rocking of cutin waxes | [13], [74] |
| 960 | C—O bonds Stretching of cutin and polysaccharides | [77] |
| 1325 | Aliphatic compounds (consequents of CH2 bending) | [13], [75] |
| 1595 | Due to stretching vibrations of phenolic compounds (C C). | [78] |
4. Conclusion
The study concluded that sustainable emerging technologies (sonication and DBD-plasma) improved wheat sprout juice's various attributes. The samples were evaluated for different sonication times (10, 20, 30, and 40 min) and DBD-plasma voltages (30, 35, 40, and 45 V). The best results were obtained for bioactive compounds, antioxidants, enzymatic activities, and microbial loads with DBD-plasma (at 40 V) and sonication (at 30 min). Moreover, DBD-plasma and sonication treatment significantly reduced the particle size, reducing the apparent viscosity (η), consistency index (K) and increasing the flow behavior (n), leading to higher stability of wheat sprout juice. The impact of DBD-plasma and sonication was also determined with SERS methods based on Au@AgNPs. The substrate morphology was obtained using TEM, EDS elemental mapping, HDDF, and DLS methods. The rapid SERS method also proved to have a higher nutritional concentration of samples treated with DBD-plasma and sonication. We hope the proposed research will be extended the direction to other agricultural commodities.
CRediT authorship contribution statement
Muhammad Faisal Manzoor: Conceptualization, Methodology, Formal analysis, Writing – original draft, Writing – review & editing. Abid Hussain: Formal analysis, Writing – original draft. Gulden Goksen: Software, Data curation. Murtaza Ali: Software, Data curation. Anees Ahmed Khalil: Writing – original draft. Xin-An Zeng: Writing – review & editing, Supervision, Funding acquisition. Anet Režek Jambrak: Writing – review & editing. Jose Manuel Lorenzo: Writing – review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgment
The authors want to acknowledge the support of Guangdong Provincial Key Laboratory of Intelligent Food Manufacturing, Foshan University, Foshan 528225, China (Project ID:2022B1212010015).
Funding for open access charges Universidade de Vigo/CISUG.
Contributor Information
Xin-An Zeng, Email: xazeng@scut.edu.cn.
Jose Manuel Lorenzo, Email: jmlorenzo@ceteca.net.
Data availability
Data will be made available on request.
References
- 1.H. Nguyen, Sustainable food systems: concept and framework, Food and Agriculture Organization of the United Nations: Rome, Italy, (2018).
- 2.Elizabeth L., Machado P., Zinöcker M., Baker P., Lawrence M. Ultra-processed foods and health outcomes: a narrative review. Nutrients. 2020;12:1955. doi: 10.3390/nu12071955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chiozzi V., Agriopoulou S., Varzakas T. Advances, applications, and comparison of thermal (pasteurization, sterilization, and aseptic packaging) against non-thermal (ultrasounds, UV radiation, ozonation, high hydrostatic pressure) technologies in food processing. Appl. Sci. 2022;12:2202. [Google Scholar]
- 4.Dong X., Wang J., Raghavan V. Critical reviews and recent advances of novel non-thermal processing techniques on the modification of food allergens. Crit. Rev. Food Sci. Nutr. 2021;61:196–210. doi: 10.1080/10408398.2020.1722942. [DOI] [PubMed] [Google Scholar]
- 5.Manzoor M.F., Siddique R., Hussain A., Ahmad N., Rehman A., Siddeeg A., Alfarga A., Alshammari G.M., Yahya M.A. Thermosonication effect on bioactive compounds, enzymes activity, particle size, microbial load, and sensory properties of almond (Prunus dulcis) milk. Ultrason. Sonochem. 2021;78 doi: 10.1016/j.ultsonch.2021.105705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Manzoor M.F., Ahmed Z., Ahmad N., Aadil R.M., Rahaman A., Roobab U., Rehman A., Siddique R., Zeng X.A., Siddeeg A. Novel processing techniques and spinach juice: Quality and safety improvements. J. Food Sci. 2020;85:1018–1026. doi: 10.1111/1750-3841.15107. [DOI] [PubMed] [Google Scholar]
- 7.Kidoń M., Narasimhan G. Effect of ultrasound and enzymatic mash treatment on bioactive compounds and antioxidant capacity of black. Red White Curr. Juices Mol. 2022;27:318. doi: 10.3390/molecules27010318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xu B., Chen J., Azam S., Feng M., Wei B., Yan W., Zhou C., Ma H., Bhandari B., Ren G. Flat dual-frequency sweeping ultrasound enhances the inactivation of polyphenol oxidase in strawberry juice. J. Food Meas. Charact. 2022;16:762–771. [Google Scholar]
- 9.Sruthi N., Josna K., Pandiselvam R., Kothakota A., Gavahian M., Khaneghah A.M. Impacts of cold plasma treatment on physicochemical, functional, bioactive, textural, and sensory attributes of food: A comprehensive review. Food Chem. 2022;368 doi: 10.1016/j.foodchem.2021.130809. [DOI] [PubMed] [Google Scholar]
- 10.Manzoor M.F., Hussain A., Sameen A., Sahar A., Khan S., Siddique R., Aadil R.M., Xu B. Novel extraction, rapid assessment and bioavailability improvement of quercetin: A review. Ultrason. Sonochem. 2021;78 doi: 10.1016/j.ultsonch.2021.105686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.M.F. Manzoor, A. Hussain, N. Naumovski, M.M.A.N. Ranjha, N. Ahmad, E. Karrar, B. Xu, S.A. Ibrahim, A Narrative Review of Recent Advances in Rapid Assessment of Anthocyanins in Agricultural and Food Products, Frontiers in Nutrition, (2022) 1352. [DOI] [PMC free article] [PubMed]
- 12.Ahmed Z., Manzoor M.F., Begum N., Khan A., Shah I., Farooq U., Siddique R., Zeng X.-A., Rahaman A., Siddeeg A. Thermo-ultrasound-based sterilization approach for the quality improvement of wheat plantlets Juice. Processes. 2019;7:518. [Google Scholar]
- 13.Ahmed Z., Manzoor M.F., Hussain A., Hanif M., Zeng X.-A. Study the impact of ultra-sonication and pulsed electric field on the quality of wheat plantlet juice through FTIR and SERS. Ultrason. Sonochem. 2021;105648 doi: 10.1016/j.ultsonch.2021.105648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Parit S.B., Dawkar V.V., Tanpure R.S., Pai S.R., Chougale A.D. Nutritional quality and antioxidant activity of wheatgrass (Triticum aestivum) unwrap by proteome profiling and DPPH and FRAP assays. J. Food Sci. 2018;83:2127–2139. doi: 10.1111/1750-3841.14224. [DOI] [PubMed] [Google Scholar]
- 15.Grubišić S., Kristić M., Lisjak M., Mišković Špoljarić K., Petrović S., Vila S., Rebekić A. Effect of wheatgrass juice on nutritional quality of apple, carrot, beet, orange and lemon juice. Foods. 2022;11:445. doi: 10.3390/foods11030445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chew S., Noor M.N., Maizura M., Tan T., Rusul G. Effect of pasteurization treatment and calamansi (Fortunella japonica) juice on the physicochemical, microbiological, and sensory characteristics of black stem sugarcane juice. Int. Food Res. J. 2018;25:1007–1015. [Google Scholar]
- 17.Ahmed Z., Manzoor M.F., Ahmad N., Zeng X.A., Din Z.U., Roobab U.…Rahaman A. Impact of pulsed electric field treatments on the growth parameters of wheat seeds and nutritional properties of their wheat plantlets juice. Food Sci. Nutr. 2020;8(5):2490–2500. doi: 10.1002/fsn3.1540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ali A., Riaz S., Sameen A., Naumovski N., Iqbal M.W., Rehman A.…Manzoor M.F. The disposition of bioactive compounds from fruit waste, their extraction, and analysis using novel technologies: A review. Processes. 2022;10(10):2014. [Google Scholar]
- 19.Richa K., Ghosh A., Hiranmoy G. Study on the nutritional and microbiological changes of sugarcane juice and determination of optimum conditions during pasteurization. Asian J. Food Agro-Indust. 2010;3:453–461. [Google Scholar]
- 20.Zafra-Rojas Q.Y., Cruz-Cansino N., Ramírez-Moreno E., Delgado-Olivares L., Villanueva-Sánchez J., Alanís-García E. Effects of ultrasound treatment in purple cactus pear (Opuntia ficus-indica) juice. Ultrason. Sonochem. 2013;20:1283–1288. doi: 10.1016/j.ultsonch.2013.01.021. [DOI] [PubMed] [Google Scholar]
- 21.Kim D.-O., Jeong S.W., Lee C.Y. Antioxidant capacity of phenolic phytochemicals from various cultivars of plums. Food Chem. 2003;81:321–326. [Google Scholar]
- 22.Slinkard K., Singleton V.L. Total phenol analysis: automation and comparison with manual methods. Am. J. Enol. Viticult. 1977;28:49–55. [Google Scholar]
- 23.Manzoor M.F., Zeng X.-A., Rahaman A., Siddeeg A., Aadil R.M., Ahmed Z., Li J., Niu D. Combined impact of pulsed electric field and ultrasound on bioactive compounds and FT-IR analysis of almond extract. J. Food Sci. Technol. 2019;56:2355–2364. doi: 10.1007/s13197-019-03627-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Prior R.L., Hoang H., Gu L., Wu X., Bacchiocca M., Howard L., Hampsch-Woodill M., Huang D., Ou B., Jacob R. Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORACFL)) of plasma and other biological and food samples. J. Agric. Food Chem. 2003;51:3273–3279. doi: 10.1021/jf0262256. [DOI] [PubMed] [Google Scholar]
- 25.Zhao L., Wang S., Liu F., Dong P., Huang W., Xiong L., Liao X. Comparing the effects of high hydrostatic pressure and thermal pasteurization combined with nisin on the quality of cucumber juice drinks. Innov. Food Sci. Emerg. Technol. 2013;17:27–36. [Google Scholar]
- 26.Manzoor M.F., Ahmad N., Ahmed Z., Siddique R., Mehmood A., Usman M., Zeng X.A. Effect of dielectric barrier discharge plasma, ultra-sonication, and thermal processing on the rheological and functional properties of sugarcane juice. J. Food Sci. 2020;85:3823–3832. doi: 10.1111/1750-3841.15498. [DOI] [PubMed] [Google Scholar]
- 27.Steffe J.F. Freeman Press; 1996. Rheological methods in food process engineering. [Google Scholar]
- 28.Bernat N., Cháfer M., Rodríguez-García J., Chiralt A., González-Martínez C. Effect of high pressure homogenisation and heat treatment on physical properties and stability of almond and hazelnut milks. LWT Food Sci. Technol. 2015;62:488–496. [Google Scholar]
- 29.Syvitski J.P. Cambridge University Press; 2007. Principles, methods and application of particle size analysis. [Google Scholar]
- 30.Hussain A., Pu H., Hu B., Sun D.-W. Au@ Ag-TGANPs based SERS for facile screening of thiabendazole and ferbam in liquid milk. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021;245 doi: 10.1016/j.saa.2020.118908. [DOI] [PubMed] [Google Scholar]
- 31.Hussain A., Pu H., Sun D.-W. Cysteamine modified core-shell nanoparticles for rapid assessment of oxamyl and thiacloprid pesticides in milk using SERS. J. Food Meas. Charact. 2020;14:2021–2029. [Google Scholar]
- 32.Hussain A., Sun D.-W., Pu H. SERS detection of urea and ammonium sulfate adulterants in milk with coffee ring effect. Food Addit. Contam. Part A. 2019;36:851–862. doi: 10.1080/19440049.2019.1591643. [DOI] [PubMed] [Google Scholar]
- 33.Manzoor M.F., Ahmad N., Ahmed Z., Siddique R., Zeng X.A., Rahaman A., Muhammad Aadil R., Wahab A. Novel extraction techniques and pharmaceutical activities of luteolin and its derivatives. J. Food Biochem. 2019:e12974. doi: 10.1111/jfbc.12974. [DOI] [PubMed] [Google Scholar]
- 34.Rocchetti G., Gregorio R.P., Lorenzo J.M., Barba F.J., Oliveira P.G., Prieto M.A., Simal-Gandara J., Mosele J.I., Motilva M.J., Tomas M. Functional implications of bound phenolic compounds and phenolics–food interaction: A review. Compr. Rev. Food Sci. Saf. 2022;21:811–842. doi: 10.1111/1541-4337.12921. [DOI] [PubMed] [Google Scholar]
- 35.Ordóñez-Santos L.E., Martínez-Girón J., Arias-Jaramillo M.E. Effect of ultrasound treatment on visual color, vitamin C, total phenols, and carotenoids content in Cape gooseberry juice. Food Chem. 2017;233:96–100. doi: 10.1016/j.foodchem.2017.04.114. [DOI] [PubMed] [Google Scholar]
- 36.Ashokkumar M., Sunartio D., Kentish S., Mawson R., Simons L., Vilkhu K., Versteeg C.K. Modification of food ingredients by ultrasound to improve functionality: A preliminary study on a model system. Innov. Food Sci. Emerg. Technol. 2008;9:155–160. [Google Scholar]
- 37.Dehghannya J., Kadkhodaei S., Heshmati M.K., Ghanbarzadeh B. Ultrasound-assisted intensification of a hybrid intermittent microwave-hot air drying process of potato: Quality aspects and energy consumption. Ultrasonics. 2019;96:104–122. doi: 10.1016/j.ultras.2019.02.005. [DOI] [PubMed] [Google Scholar]
- 38.Thirumdas R., Sarangapani C., Annapure U.S. Cold plasma: a novel non-thermal technology for food processing. Food Biophys. 2015;10:1–11. [Google Scholar]
- 39.Sarangapani C., O'Toole G., Cullen P., Bourke P. Atmospheric cold plasma dissipation efficiency of agrochemicals on blueberries. Innov. Food Sci. Emerg. Technol. 2017;44:235–241. [Google Scholar]
- 40.Liu Z., Zhao W., Zhang Q., Gao G., Meng Y. Effect of cold plasma treatment on sterilizing rate and quality of kiwi turbid juice. J. Food Process Eng. 2021;44:e13711. [Google Scholar]
- 41.Rodríguez Ó., Gomes W.F., Rodrigues S., Fernandes F.A. Effect of indirect cold plasma treatment on cashew apple juice (Anacardium occidentale L.) LWT Food Sci. Technol. 2017;84:457–463. [Google Scholar]
- 42.Chen Y., Yu L.J., Rupasinghe H.V. Effect of thermal and non-thermal pasteurisation on the microbial inactivation and phenolic degradation in fruit juice: A mini-review. J. Sci. Food Agric. 2013;93:981–986. doi: 10.1002/jsfa.5989. [DOI] [PubMed] [Google Scholar]
- 43.Gouda M., Bekhit A.-E.-D., Tang Y., Huang Y., Huang L., He Y., Li X. Recent innovations of ultrasound green technology in herbal phytochemistry: A review. Ultrason. Sonochem. 2021;73 doi: 10.1016/j.ultsonch.2021.105538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Manzoor M.F., Xu B., Khan S., Shukat R., Ahmad N., Imran M., Rehman A., Karrar E., Aadil R.M., Korma S.A. Impact of high-intensity thermosonication treatment on spinach juice: Bioactive compounds, rheological, microbial, and enzymatic activities. Ultrason. Sonochem. 2021;78 doi: 10.1016/j.ultsonch.2021.105740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sun Y., Liu D., Chen J., Ye X., Yu D. Effects of different factors of ultrasound treatment on the extraction yield of the all-trans-β-carotene from citrus peels. Ultrason. Sonochem. 2011;18:243–249. doi: 10.1016/j.ultsonch.2010.05.014. [DOI] [PubMed] [Google Scholar]
- 46.Wang X., Wu Y., Chen G., Yue W., Liang Q., Wu Q. Optimisation of ultrasound assisted extraction of phenolic compounds from Sparganii rhizoma with response surface methodology. Ultrason. Sonochem. 2013;20:846–854. doi: 10.1016/j.ultsonch.2012.11.007. [DOI] [PubMed] [Google Scholar]
- 47.Fernandes F.A., Santos V.O., Rodrigues S. Effects of glow plasma technology on some bioactive compounds of acerola juice. Food Res. Int. 2019;115:16–22. doi: 10.1016/j.foodres.2018.07.042. [DOI] [PubMed] [Google Scholar]
- 48.de Castro D.R.G., Mar J.M., da Silva L.S., da Silva K.A., Sanches E.A., de Araújo Bezerra J., Rodrigues S., Fernandes F.A., Campelo P.H. Dielectric barrier atmospheric cold plasma applied on camu-camu juice processing: Effect of the excitation frequency. Food Res. Int. 2020;131 doi: 10.1016/j.foodres.2020.109044. [DOI] [PubMed] [Google Scholar]
- 49.Ali M., Cheng J.H., Sun D.W. Effects of dielectric barrier discharge cold plasma treatments on degradation of anilazine fungicide and quality of tomato (Lycopersicon esculentum Mill) juice. Int. J. Food Sci. Technol. 2021;56:69–75. [Google Scholar]
- 50.Zarei M., Fazlara A., Tulabifard N. Effect of thermal treatment on physicochemical and antioxidant properties of honey. Heliyon. 2019;5:e01894. doi: 10.1016/j.heliyon.2019.e01894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Faisal Manzoor M., Ahmed Z., Ahmad N., Karrar E., Rehman A., Muhammad Aadil R., Al-Farga A., Waheed Iqbal M., Rahaman A., Zeng X.A. Probing the combined impact of pulsed electric field and ultra-sonication on the quality of spinach juice. J. Food Process. Preserv. 2021;45:e15475. [Google Scholar]
- 52.Ramazzina I., Berardinelli A., Rizzi F., Tappi S., Ragni L., Sacchetti G., Rocculi P. Effect of cold plasma treatment on physico-chemical parameters and antioxidant activity of minimally processed kiwifruit. Postharvest Biol. Technol. 2015;107:55–65. [Google Scholar]
- 53.Popescu E.C., Buruleanu C.L. Heat degradation kinetics of the chlorophyll from spinach and its correlation with the reflection spectra. Matrix. 2017;9:12. [Google Scholar]
- 54.Tappi S., Berardinelli A., Ragni L., Dalla Rosa M., Guarnieri A., Rocculi P. Atmospheric gas plasma treatment of fresh-cut apples. Innov. Food Sci. Emerg. Technol. 2014;21:114–122. [Google Scholar]
- 55.Jiménez-Sánchez C., Lozano-Sánchez J., Segura-Carretero A., Fernandez-Gutierrez A. Alternatives to conventional thermal treatments in fruit-juice processing. Part 1: Techniques and applications. Crit. Rev. Food Sci. Nutr. 2017;57:501–523. doi: 10.1080/10408398.2013.867828. [DOI] [PubMed] [Google Scholar]
- 56.Huang G., Chen S., Dai C., Sun L., Sun W., Tang Y., Xiong F., He R., Ma H. Effects of ultrasound on microbial growth and enzyme activity. Ultrason. Sonochem. 2017;37:144–149. doi: 10.1016/j.ultsonch.2016.12.018. [DOI] [PubMed] [Google Scholar]
- 57.M. Kubo, S. Curet, L. Boillereaux, P. Augusto, Effect of ultrasound processing on the activity of polyphenoloxidase and peroxidase in a fruit juice model solution, in: SLACA–Latin American Symposium of Food Science, Campinas, 2017.
- 58.Costa M.G.M., Fonteles T.V., de Jesus A.L.T., Almeida F.D.L., de Miranda M.R.A., Fernandes F.A.N., Rodrigues S. High-intensity ultrasound processing of pineapple juice. Food Bioprocess Technol. 2013;6:997–1006. [Google Scholar]
- 59.Pankaj S., Misra N., Cullen P. Kinetics of tomato peroxidase inactivation by atmospheric pressure cold plasma based on dielectric barrier discharge. Innov. Food Sci. Emerg. Technol. 2013;19:153–157. [Google Scholar]
- 60.Tinello F., Lante A. Recent advances in controlling polyphenol oxidase activity of fruit and vegetable products. Innov. Food Sci. Emerg. Technol. 2018;50:73–83. [Google Scholar]
- 61.Surowsky B., Fischer A., Schlueter O., Knorr D. Cold plasma effects on enzyme activity in a model food system. Innov. Food Sci. Emerg. Technol. 2013;19:146–152. [Google Scholar]
- 62.Ramírez-Moreno E., Zafra-Rojas Q.Y., Arias-Rico J., Ariza-Ortega J.A., Alanís-García E., Cruz-Cansino N. Effect of ultrasound on microbiological load and antioxidant properties of blackberry juice. J. Food Process. Preserv. 2018;42:e13489. [Google Scholar]
- 63.Umair M., Jabbar S., Senan A.M., Sultana T., Nasiru M.M., Shah A.A., Zhuang H., Jianhao Z. Influence of combined effect of ultra-sonication and high-voltage cold plasma treatment on quality parameters of carrot juice. Foods. 2019;8:593. doi: 10.3390/foods8110593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.L. Xu, S.M. Sanders, B. Tao, A.L. Garner, K.M. Keener, Assessment of efficacy and reactive gas species generation for orange juice decontamination using high voltage atmospheric cold plasma, in: 2016 IEEE International Conference on Plasma Science (ICOPS), IEEE, 2016, pp. 1-1.
- 65.Han L., Patil S., Boehm D., Milosavljević V., Cullen P., Bourke P. Mechanisms of inactivation by high-voltage atmospheric cold plasma differ for Escherichia coli and Staphylococcus aureus. Appl. Environ. Microbiol. 2016;82:450–458. doi: 10.1128/AEM.02660-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kim Y.H., Hong Y.J., Baik K.Y., Kwon G.C., Choi J.J., Cho G.S., Uhm H.S., Kim D.Y., Choi E.H. Measurement of reactive hydroxyl radical species inside the biosolutions during non-thermal atmospheric pressure plasma jet bombardment onto the solution. Plasma Chem. Plasma Process. 2014;34:457–472. [Google Scholar]
- 67.Juliano P., Kutter A., Cheng L.J., Swiergon P., Mawson R., Augustin M. Enhanced creaming of milk fat globules in milk emulsions by the application of ultrasound and detection by means of optical methods. Ultrason. Sonochem. 2011;18:963–973. doi: 10.1016/j.ultsonch.2011.03.003. [DOI] [PubMed] [Google Scholar]
- 68.Bhat S., Sharma H.K. Combined effect of blanching and sonication on quality parameters of bottle gourd (Lagenaria siceraria) juice. Ultrason. Sonochem. 2016;33:182–189. doi: 10.1016/j.ultsonch.2016.04.014. [DOI] [PubMed] [Google Scholar]
- 69.Wang X., Han Y., Niu H., Zhang L., Xiang Q., Zong W. Alternaria mycotoxin degradation and quality evaluation of jujube juice by cold plasma treatment. Food Control. 2022;137 [Google Scholar]
- 70.Illera A., Chaple S., Sanz M., Ng S., Lu P., Jones J., Carey E., Bourke P. Effect of cold plasma on polyphenol oxidase inactivation in cloudy apple juice and on the quality parameters of the juice during storage. Food Chem. X. 2019;3 doi: 10.1016/j.fochx.2019.100049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Rojas M.L., Leite T.S., Cristianini M., Alvim I.D., Augusto P.E. Peach juice processed by the ultrasound technology: Changes in its microstructure improve its physical properties and stability. Food Res. Int. 2016;82:22–33. [Google Scholar]
- 72.Silveira M.R., Coutinho N.M., Rocha R.S., Moraes J., Esmerino E.A., Pimentel T.C., Freitas M.Q., Silva M.C., Raices R.S., Ranadheera C.S. Guava flavored whey-beverage processed by cold plasma: Physical characteristics, thermal behavior and microstructure. Food Res. Int. 2019;119:564–570. doi: 10.1016/j.foodres.2018.10.033. [DOI] [PubMed] [Google Scholar]
- 73.Hussain A., Pu H., Sun D.-W. SERS detection of sodium thiocyanate and benzoic acid preservatives in liquid milk using cysteamine functionalized core-shelled nanoparticles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020;229 doi: 10.1016/j.saa.2019.117994. [DOI] [PubMed] [Google Scholar]
- 74.Boyaci I.H., Temiz H.T., Geniş H.E., Soykut E.A., Yazgan N.N., Güven B., Uysal R.S., Bozkurt A.G., İlaslan K., Torun O. Dispersive and FT-Raman spectroscopic methods in food analysis. Rsc Adv. 2015;5:56606–56624. [Google Scholar]
- 75.Sharma S., Baran C., Tripathi A., Awasthi A., Jaiswal A., Uttam R., Bharti A.S., Singh R., Uttam K. Phytochemical screening of the different cultivars of ixora flowers by non-destructive, label-free, and rapid spectroscopic techniques. Anal. Lett. 2021;54:2276–2292. [Google Scholar]
- 76.Horiue H., Sasaki M., Yoshikawa Y., Toyofuku M., Shigeto S. Raman spectroscopic signatures of carotenoids and polyenes enable label-free visualization of microbial distributions within pink biofilms. Sci. Rep. 2020;10:1–10. doi: 10.1038/s41598-020-64737-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Heredia-Guerrero J.A., Benítez J.J., Domínguez E., Bayer I.S., Cingolani R., Athanassiou A., Heredia A. Infrared and Raman spectroscopic features of plant cuticles: a review. Front. Plant Sci. 2014;5:305. doi: 10.3389/fpls.2014.00305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Oleszkiewicz T., Pacia M.Z., Grzebelus E., Baranski R. Plant and Food Carotenoids. Springer; 2020. Light microscopy and Raman imaging of carotenoids in plant cells in situ and in released carotene crystals; pp. 245–260. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be made available on request.